Advances in Therapeutic Implications of Inorganic Drug Delivery Nano-Platforms for Cancer
Abstract
1. Introduction
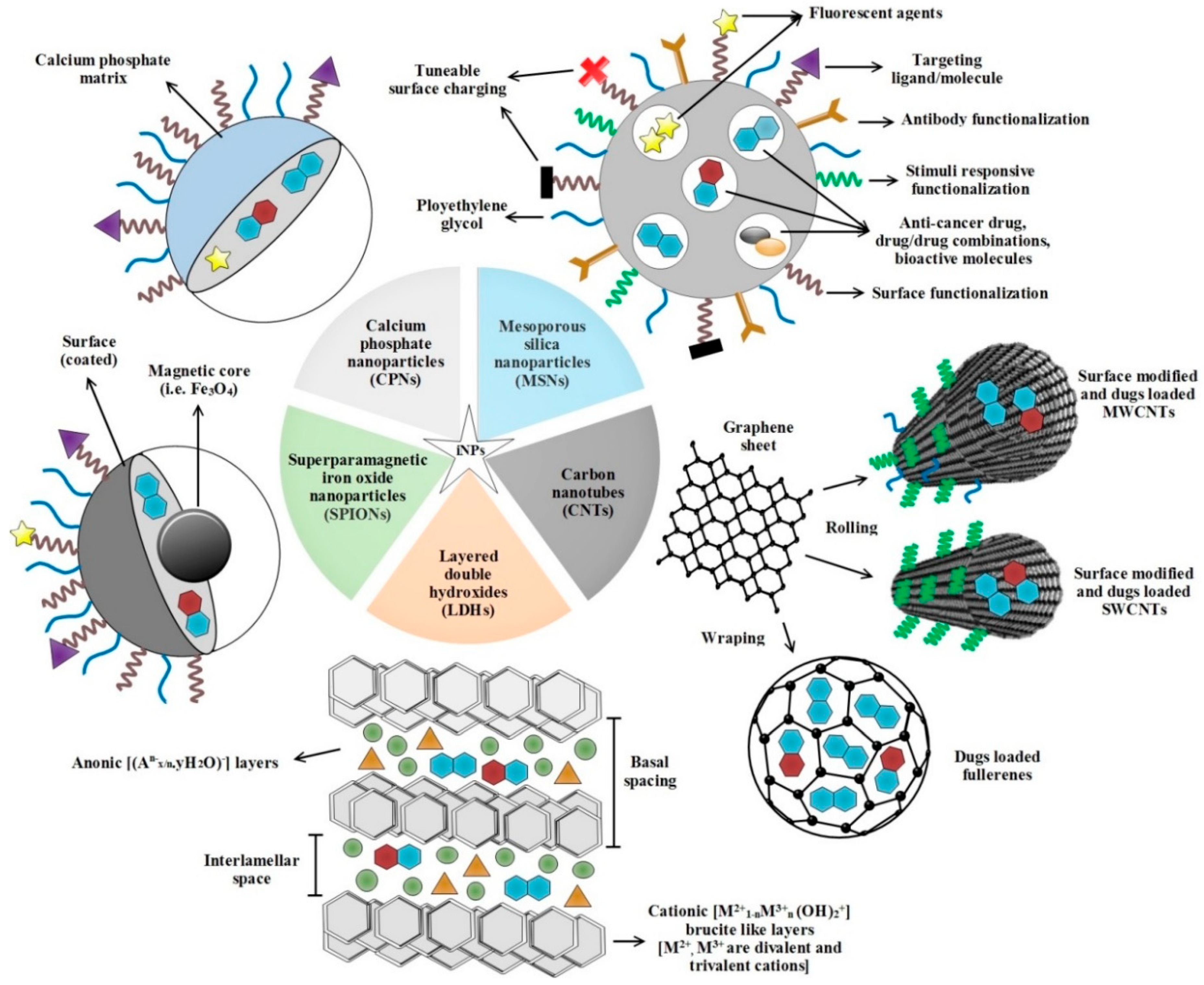
2. Inorganic NDDPs Implicated in Anti-Cancer Therapy
2.1. MSNs based NDDPs Implicated in Anti-Cancer Therapy
2.2. CNTs based NDDPs Implicated in Anti-Cancer Therapy
2.3. LDHs Based NDDPs Implicated in Anti-Cancer Therapy
2.4. SPIONs Based NDDPs Implicated in Anti-Cancer Therapy
2.5. CPNs based NDDPs Implicated in Anti-Cancer Therapy
3. Conclusions and Future Outlook
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2019. CA A Cancer J. Clin. 2019, 69, 7–34. [Google Scholar] [CrossRef] [PubMed]
- Bray, F.; Jemal, A.; Grey, N.; Ferlay, J.; Forman, D. Global cancer transitions according to the Human Development Index (2008–2030): A population-based study. Lancet Oncol. 2012, 13, 790–801. [Google Scholar] [CrossRef]
- Wang, S.H.; Yu, J. Structure-based design for binding peptides in anti-cancer therapy. Biomaterials 2018, 156, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Luo, C.; Sun, J.; Sun, B.; He, Z. Prodrug-based nanoparticulate drug delivery strategies for cancer therapy. Trends Pharmacol. Sci. 2014, 35, 556–566. [Google Scholar] [CrossRef]
- Dissanayake, S.; Denny, W.A.; Gamage, S.; Sarojini, V. Recent developments in anticancer drug delivery using cell penetrating and tumor targeting peptides. J. Control. Release 2017, 250, 62–76. [Google Scholar] [CrossRef] [PubMed]
- Thapa, R.K.; Byeon, J.H.; Ku, S.K.; Yong, C.S.; Kim, J.O. Easy on-demand self-assembly of lateral nanodimensional hybrid graphene oxide flakes for near-infrared-induced chemothermal therapy. NPG Asia Mater. 2017, 9, e416. [Google Scholar] [CrossRef]
- Yin, T.; Liu, J.; Zhao, Z.; Zhao, Y.; Dong, L.; Yang, M.; Zhou, J.; Huo, M. Redox Sensitive Hyaluronic Acid-Decorated Graphene Oxide for Photothermally Controlled Tumor-Cytoplasm-Selective Rapid Drug Delivery. Adv. Funct. Mater. 2017, 27, 1604620. [Google Scholar] [CrossRef]
- Liu, X.; Cheng, X.; Wang, F.; Feng, L.; Wang, Y.; Zheng, Y.; Guo, R. Targeted delivery of SNX-2112 by polysaccharide-modified graphene oxide nanocomposites for treatment of lung cancer. Carbohydr. Polym. 2018, 185, 85–95. [Google Scholar] [CrossRef]
- Diaz-Diestra, D.; Thapa, B.; Badillo-Diaz, D.; Beltran-Huarac, J.; Morell, G.; Weiner, B. Graphene Oxide/ZnS:Mn Nanocomposite Functionalized with Folic Acid as a Nontoxic and Effective Theranostic Platform for Breast Cancer Treatment. Nanomaterials 2018, 8, 484. [Google Scholar] [CrossRef]
- Yang, G.; Xu, L.; Chao, Y.; Xu, J.; Sun, X.; Wu, Y.; Peng, R.; Liu, Z. Hollow MnO2 as a tumor-microenvironment-responsive biodegradable nano-platform for combination therapy favoring antitumor immune responses. Nat. Commun. 2017, 8, 902. [Google Scholar] [CrossRef]
- Ramalingam, V.; Varunkumar, K.; Ravikumar, V.; Rajaram, R. Target delivery of doxorubicin tethered with PVP stabilized gold nanoparticles for effective treatment of lung cancer. Sci. Rep. 2018, 8, 3815. [Google Scholar] [CrossRef] [PubMed]
- Farooq, M.U.; Novosad, V.; Rozhkova, E.A.; Wali, H.; Ali, A.; Fateh, A.A.; Neogi, P.B.; Neogi, A.; Wang, Z. Gold Nanoparticles-enabled Efficient Dual Delivery of Anticancer Therapeutics to HeLa Cells. Sci. Rep. 2018, 8, 2907. [Google Scholar] [CrossRef] [PubMed]
- Ramasamy, T.; Ruttala, H.B.; Sundaramoorthy, P.; Poudel, B.K.; Youn, Y.S.; Ku, S.K.; Choi, H.-G.; Yong, C.S.; Kim, J.O. Multimodal selenium nanoshell-capped Au@mSiO2 nanoplatform for NIR-responsive chemo-photothermal therapy against metastatic breast cancer. NPG Asia Mater. 2018, 10, 197–216. [Google Scholar] [CrossRef]
- Habiba, K.; Encarnacion-Rosado, J.; Garcia-Pabon, K.; Villalobos-Santos, J.C.; Makarov, V.I.; Avalos, J.A.; Weiner, B.R.; Morell, G. Improving cytotoxicity against cancer cells by chemo-photodynamic combined modalities using silver-graphene quantum dots nanocomposites. Int. J. Nanomed. 2016, 11, 107–119. [Google Scholar] [CrossRef] [PubMed]
- Krishnan, P.; Rajan, M.; Kumari, S.; Sakinah, S.; Priya, S.P.; Amira, F.; Danjuma, L.; Pooi Ling, M.; Fakurazi, S.; Arulselvan, P.; et al. Efficiency of newly formulated camptothecin with β-cyclodextrin-EDTA-Fe3O4 nanoparticle-conjugated nanocarriers as an anti-colon cancer (HT29) drug. Sci. Rep. 2017, 7, 10962. [Google Scholar] [CrossRef] [PubMed]
- Phan, T.T.V.; Bui, N.Q.; Cho, S.W.; Bharathiraja, S.; Manivasagan, P.; Moorthy, M.S.; Mondal, S.; Kim, C.-S.; Oh, J. Photoacoustic Imaging-Guided Photothermal Therapy with Tumor-Targeting HA-FeOOH@PPy Nanorods. Sci. Rep. 2018, 8, 8809. [Google Scholar] [CrossRef]
- Truffi, M.; Colombo, M.; Sorrentino, L.; Pandolfi, L.; Mazzucchelli, S.; Pappalardo, F.; Pacini, C.; Allevi, R.; Bonizzi, A.; Corsi, F.; et al. Multivalent exposure of trastuzumab on iron oxide nanoparticles improves antitumor potential and reduces resistance in HER2-positive breast cancer cells. Sci. Rep. 2018, 8, 6563. [Google Scholar] [CrossRef]
- Wang, L.; Jang, G.; Ban, D.K.; Sant, V.; Seth, J.; Kazmi, S.; Patel, N.; Yang, Q.; Lee, J.; Janetanakit, W.; et al. Multifunctional stimuli responsive polymer-gated iron and gold-embedded silica nano golf balls: Nanoshuttles for targeted on-demand theranostics. Bone Res. 2017, 5, 17051. [Google Scholar] [CrossRef]
- Liu, H.N.; Guo, N.N.; Guo, W.W.; Huang-Fu, M.Y.; Vakili, M.R.; Chen, J.J.; Xu, W.-H.; Wei, Q.C.; Han, M.; Lavasanifar, A.; et al. Delivery of mitochondriotropic doxorubicin derivatives using self-assembling hyaluronic acid nanocarriers in doxorubicin-resistant breast cancer. Acta Pharmacol. Sin. 2018, 10, 1681–1692. [Google Scholar] [CrossRef]
- Qiu, M.; Wang, D.; Liang, W.; Liu, L.; Zhang, Y.; Chen, X.; Sang, D.K.; Xing, C.; Li, Z.; Dong, B.; et al. Novel concept of the smart NIR-light–controlled drug release of black phosphorus nanostructure for cancer therapy. Proc. Natl. Acad. Sci. USA 2018, 115, 501–506. [Google Scholar] [CrossRef]
- Hurria, A.; Togawa, K.; Mohile, S.G.; Owusu, C.; Klepin, H.D.; Gross, C.P.; Lichtman, S.M.; Gajra, A.; Bhatia, S.; Katheria, V.; et al. Predicting chemotherapy toxicity in older adults with cancer: A prospective multicenter study. J. Clin. Oncol. 2011, 29, 3457–3465. [Google Scholar] [CrossRef] [PubMed]
- Rodzinski, A.; Guduru, R.; Liang, P.; Hadjikhani, A.; Stewart, T.; Stimphil, E.; Runowicz, C.; Cote, R.; Altman, N.; Datar, R.; et al. Targeted and controlled anticancer drug delivery and release with magnetoelectric nanoparticles. Sci. Rep. 2016, 6, 20867. [Google Scholar] [CrossRef] [PubMed]
- George, J.; Yan, I.K.; Patel, T. Nanovesicle-mediated delivery of anticancer agents effectively induced cell death and regressed intrahepatic tumors in athymic mice. Lab. Investig. 2018, 98, 895–910. [Google Scholar] [CrossRef] [PubMed]
- Murugan, C.; Rayappan, K.; Thangam, R.; Bhanumathi, R.; Shanthi, K.; Vivek, R.; Thirumurugan, R.; Bhattacharyya, A.; Sivasubramanian, S.; Gunasekaran, P.; et al. Combinatorial nanocarrier based drug delivery approach for amalgamation of anti-tumor agents in breast cancer cells: An improved nanomedicine strategy. Sci. Rep. 2016, 6, 34053. [Google Scholar] [CrossRef]
- Shi, J.; Kantoff, P.W.; Wooster, R.; Farokhzad, O.C. Cancer nanomedicine: Progress, challenges and opportunities. Nat. Rev. Cancer 2016, 17, 20–37. [Google Scholar] [CrossRef] [PubMed]
- Quader, S.; Kataoka, K. Nanomaterial-enabled cancer therapy. Mol. Ther. 2017, 25, 1501–1513. [Google Scholar] [CrossRef]
- Gomes, B.S.; Simões, B.; Mendes, P.M. The increasing dynamic, functional complexity of bio-interface materials. Nat. Rev. Chem. 2018, 2, 0120. [Google Scholar] [CrossRef]
- Moreira, A.F.; Dias, D.R.; Correia, I.J. Stimuli-responsive mesoporous silica nanoparticles for cancer therapy: A review. Microporous Mesoporous Mater. 2016, 236, 141–157. [Google Scholar] [CrossRef]
- Feng, Y.; Panwar, N.; Tng, D.J.H.; Tjin, S.C.; Wang, K.; Yong, K.T. The application of mesoporous silica nanoparticle family in cancer theranostics. Coord. Chem. Rev. 2016, 319, 86–109. [Google Scholar] [CrossRef]
- Zhou, Y.; Quan, G.; Wu, Q.; Zhang, X.; Niu, B.; Wu, B.; Huang, Y.; Pan, X.; Wu, C. Mesoporous silica nanoparticles for drug and gene delivery. Acta Pharm. Sin. B 2018, 8, 165–177. [Google Scholar] [CrossRef] [PubMed]
- Freitas, L.B.D.O.; Corgosinho, L.D.M.; Faria, J.A.Q.A.; dos Santos, V.M.; Resende, J.M.; Leal, A.S.; Gomes, D.A.; Sousa, E.M.B.d. Multifunctional mesoporous silica nanoparticles for cancer-targeted, controlled drug delivery and imaging. Microporous Mesoporous Mater. 2017, 242, 271–283. [Google Scholar] [CrossRef]
- Wang, F.; Zhang, L.; Bai, X.; Cao, X.; Jiao, X.; Huang, Y.; Li, Y.; Qin, Y.; Wen, Y. Stimuli-Responsive Nanocarrier for Co-delivery of MiR-31 and Doxorubicin To Suppress High MtEF4 Cancer. ACS Appl. Mater. Interfaces 2018, 10, 22767–22775. [Google Scholar] [CrossRef] [PubMed]
- Mandal, T.; Beck, M.; Kirsten, N.; Lindén, M.; Buske, C. Targeting murine leukemic stem cells by antibody functionalized mesoporous silica nanoparticles. Sci. Rep. 2018, 8, 989. [Google Scholar] [CrossRef] [PubMed]
- Hadipour Moghaddam, S.P.; Yazdimamaghani, M.; Ghandehari, H. Glutathione-sensitive hollow mesoporous silica nanoparticles for controlled drug delivery. J. Control. Release 2018, 282, 62–75. [Google Scholar] [CrossRef] [PubMed]
- Jin, R.; Liu, Z.; Bai, Y.; Zhou, Y.; Chen, X. Multiple-Responsive Mesoporous Silica Nanoparticles for Highly Accurate Drugs Delivery to Tumor Cells. ACS Omega 2018, 3, 4306–4315. [Google Scholar] [CrossRef] [PubMed]
- Velusamy, P.; Srinivasa, C.M.; Kumar, G.V.; Qurishi, Y.; Su, C.H.; Gopinath, S.C.B. A pH stimuli thiol modified mesoporous silica nanoparticles: Doxorubicin carrier for cancer therapy. J. Taiwan Inst. Chem. Engin. 2018, 87, 264–271. [Google Scholar] [CrossRef]
- Liang, C.; Wang, H.; Zhang, M.; Cheng, W.; Li, Z.; Nie, J.; Liu, G.; Lian, D.; Xie, Z.; Huang, L.; et al. Self-controlled release of Oxaliplatin prodrug from d-α-tocopheryl polyethylene glycol 1000 succinate (TPGS) functionalized mesoporous silica nanoparticles for cancer therapy. J. Colloid Interface Sci. 2018, 525, 1–10. [Google Scholar] [CrossRef]
- Yao, Z.; Han, L.; Chen, Y.; He, F.; Sun, B.; kamar, S.; Zhang, Y.; Yang, Y.; Wang, C.; Yang, Z. Hedgehog signalling in the tumourigenesis and metastasis of osteosarcoma, and its potential value in the clinical therapy of osteosarcoma. Cell Death Dis. 2018, 9, 1–12. [Google Scholar] [CrossRef]
- Martínez-Carmona, M.; Lozano, D.; Colilla, M.; Vallet-Regí, M. Lectin-conjugated pH-responsive mesoporous silica nanoparticles for targeted bone cancer treatment. Acta Biomater. 2018, 65, 393–404. [Google Scholar] [CrossRef]
- Rajesh, Y.; Pal, I.; Banik, P.; Chakraborty, S.; Borkar, S.A.; Dey, G.; Mukherjee, A.; Mandal, M. Insights into molecular therapy of glioma: Current challenges and next generation blueprint. Acta Pharm. Sin. 2017, 38, 591–613. [Google Scholar] [CrossRef]
- Heggannavar, G.B.; Hiremath, C.G.; Achari, D.D.; Pangarkar, V.G.; Kariduraganavar, M.Y. Development of Doxorubicin-Loaded Magnetic Silica–Pluronic F-127 Nanocarriers Conjugated with Transferrin for Treating Glioblastoma across the Blood–Brain Barrier Using an in Vitro Model. ACS Omega 2018, 3, 8017–8026. [Google Scholar] [CrossRef] [PubMed]
- AbouAitah, K.; Swiderska-Sroda, A.; Farghali, A.A.; Wojnarowicz, J.; Stefanek, A.; Gierlotka, S.; Opalinska, A.; Allayeh, A.K.; Ciach, T.; Lojkowski, W. Folic acid-conjugated mesoporous silica particles as nanocarriers of natural prodrugs for cancer targeting and antioxidant action. Oncotarget 2018, 9, 26466–26490. [Google Scholar] [CrossRef] [PubMed]
- Wani, A.; Savithra, G.H.L.; Abyad, A.; Kanvinde, S.; Li, J.; Brock, S.; Oupický, D. Surface PEGylation of Mesoporous Silica Nanorods (MSNR): Effect on loading, release, and delivery of mitoxantrone in hypoxic cancer cells. Sci. Rep. 2017, 7, 2274. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, W.; Elhissi, A.; Dhanak, V.; Subramani, K. Carbon nanotubes: Applications in cancer therapy and drug delivery research. In Emerging Nanotechnologies in Dentistry, 2nd ed.; Subramani, K., Ahmed, W., Eds.; William Andrew Publishing: Norwich, CT, USA, 2018; pp. 371–389. [Google Scholar]
- Mahajan, S.; Patharkar, A.; Kuche, K.; Maheshwari, R.; Deb, P.K.; Kalia, K.; Tekade, R.K. Functionalized carbon nanotubes as emerging delivery system for the treatment of cancer. Int. J. Pharm. 2018, 548, 540–558. [Google Scholar] [CrossRef] [PubMed]
- Raval, J.P.; Joshi, P.; Chejara, D.R. Carbon nanotube for targeted drug delivery. In Applications of Nanocomposite Materials in Drug Delivery; Inamuddin, A.M.A., Mohammad, A., Eds.; Woodhead Publishing: Duxford, UK, 2018; pp. 203–216. [Google Scholar]
- Ates, M.; Eker, A.A.; Eker, B. Carbon nanotube-based nanocomposites and their applications. J. Adhes. Sci. Technol. 2017, 31, 1977–1997. [Google Scholar] [CrossRef]
- Liu, X.; Xu, D.; Liao, C.; Fang, Y.; Guo, B. Development of a promising drug delivery for formononetin: Cyclodextrin-modified single-walled carbon nanotubes. J. Drug Deliv. Sci. Technol. 2018, 43, 461–468. [Google Scholar] [CrossRef]
- Huang, N.; Wang, H.; Zhao, J.; Lui, H.; Korbelik, M.; Zeng, H. Single-wall carbon nanotubes assisted photothermal cancer therapy: Animal study with a murine model of squamous cell carcinoma. Lasers Surg. Med. 2010, 42, 798–808. [Google Scholar] [CrossRef]
- Ben Sahra, I.; Le Marchand-Brustel, Y.; Tanti, J.F.; Bost, F. Metformin in Cancer Therapy: A New Perspective for an Old Antidiabetic Drug? Mol. Cancer Ther. 2010, 9, 1092–1099. [Google Scholar] [CrossRef] [PubMed]
- Yoo, S.; Hou, J.; Yi, W.; Li, Y.; Chen, W.; Meng, L.; Si, J.; Hou, X. Enhanced Response of Metformin towards the Cancer Cells due to Synergism with Multi-walled Carbon Nanotubes in Photothermal Therapy. Sci. Rep. 2017, 7, 1071. [Google Scholar] [CrossRef] [PubMed]
- Wei, C.; Dong, X.; Zhang, Y.; Liang, J.; Yang, A.; Zhu, D.; Liu, T.; Kong, D.; Lv, F. Simultaneous fluorescence imaging monitoring of the programmed release of dual drugs from a hydrogel-carbon nanotube delivery system. Sens. Actuators B Chem. 2018, 273, 264–275. [Google Scholar] [CrossRef]
- Karthika, V.; Kaleeswarran, P.; Gopinath, K.; Arumugam, A.; Govindarajan, M.; Alharbi, N.S.; Khaled, J.M.; Al-Anbr, M.N.; Benelli, G. Biocompatible properties of nano-drug carriers using TiO2-Au embedded on multiwall carbon nanotubes for targeted drug delivery. Mater. Sci. Eng. C 2018, 90, 589–601. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Tian, K.; Zhou, T.; Jia, X.; Li, J.; Liu, P. PEGylated multi-walled carbon nanotubes as versatile vector for tumor-specific intracellular triggered release with enhanced anti-cancer efficiency: Optimization of length and PEGylation degree. Colloids Surf. B Biointerfaces 2018, 168, 43–49. [Google Scholar] [CrossRef] [PubMed]
- Fadel, T.R.; Fahmy, T.M. Immunotherapy applications of carbon nanotubes: From design to safe applications. Trends Biotechnol. 2014, 32, 198–209. [Google Scholar] [CrossRef]
- Villa, C.H.; Dao, T.; Ahearn, I.; Fehrenbacher, N.; Casey, E.; Rey, D.A.; Korontsvit, T.; Zakhaleva, V.; Batt, C.A.; Philips, M.R.; et al. Single-Walled Carbon Nanotubes Deliver Peptide Antigen into Dendritic Cells and Enhance IgG Responses to Tumor-Associated Antigens. ACS Nano 2011, 5, 5300–5311. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Hu, Z.; Xu, J.; Zhao, Y. Therapeutic applications of low-toxicity spherical nanocarbon materials. NPG Asia Mater. 2014, 6, e84. [Google Scholar] [CrossRef]
- Mohajeri, M.; Behnam, B.; Sahebkar, A. Biomedical applications of carbon nanomaterials: Drug and gene delivery potentials. J. Cell Physiol. 2019, 234, 298–319. [Google Scholar] [CrossRef] [PubMed]
- Saleem, J.; Wang, L.; Chen, C. Carbon-Based Nanomaterials for Cancer Therapy via Targeting Tumor Microenvironment. Adv. Healthc. Mater. 2018, 7, 1800525. [Google Scholar] [CrossRef] [PubMed]
- Hazrati, M.K.; Javanshir, Z.; Bagheri, Z. B24N24 fullerene as a carrier for 5-fluorouracil anti-cancer drug delivery: DFT studies. J. Mol. Graph. Model. 2017, 77, 17–24. [Google Scholar] [CrossRef] [PubMed]
- Guan, M.; Ge, J.; Wu, J.; Zhang, G.; Chen, D.; Zhang, W.; Zhang, Y.; Zou, T.; Zhen, M.; Wang, C.; et al. Fullerene/photosensitizer nanovesicles as highly efficient and clearable phototheranostics with enhanced tumor accumulation for cancer therapy. Biomaterials 2016, 103, 75–85. [Google Scholar] [CrossRef]
- Lin, M.S.; Chen, R.T.; Yu, N.Y.; Sun, L.C.; Liu, Y.; Cui, C.H.; Xie, S.Y.; Huang, R.B.; Zheng, L.S. Fullerene-based amino acid ester chlorides self-assembled as spherical nano-vesicles for drug delayed release. Colloids Surf. B Biointerfaces 2017, 159, 613–619. [Google Scholar] [CrossRef]
- Thotakura, N.; Sharma, G.; Singh, B.; Kumar, V.; Raza, K. Aspartic acid derivatized hydroxylated fullerenes as drug delivery vehicles for docetaxel: An explorative study. Artif. Cells Nanomed. Biotechnol. 2018, 46, 1763–1772. [Google Scholar] [CrossRef]
- Raza, K.; Thotakura, N.; Kumar, P.; Joshi, M.; Bhushan, S.; Bhatia, A.; Kumar, V.; Malik, R.; Sharma, G.; Guru, S.K.; et al. C60-fullerenes for delivery of docetaxel to breast cancer cells: A promising approach for enhanced efficacy and better pharmacokinetic profile. Int. J. Pharm. 2015, 495, 551–559. [Google Scholar] [CrossRef]
- Mishra, G.; Dash, B.; Pandey, S. Layered double hydroxides: A brief review from fundamentals to application as evolving biomaterials. Appl. Clay Sci. 2018, 153, 172–186. [Google Scholar] [CrossRef]
- Choy, J.H.; Oh, J.M.; Choi, S.J. Layered Double Hydroxides as Controlled Release Materials. In Comprehensive Biomaterials II; Ducheyne, P., Ed.; Elsevier: Oxford, UK, 2017; pp. 705–718. [Google Scholar]
- Senapati, S.; Thakur, R.; Verma, S.P.; Duggal, S.; Mishra, D.P.; Das, P.; Shripathi, T.; Kumar, M.; Rana, D.; Maiti, P. Layered double hydroxides as effective carrier for anticancer drugs and tailoring of release rate through interlayer anions. J. Control. Release 2016, 224, 186–198. [Google Scholar] [CrossRef]
- Zhang, L.X.; Xie, X.X.; Liu, D.Q.; Xu, Z.P.; Liu, R.T. Efficient co-delivery of neo-epitopes using dispersion-stable layered double hydroxide nanoparticles for enhanced melanoma immunotherapy. Biomaterials 2018, 174, 54–66. [Google Scholar] [CrossRef]
- Allou, N.B.; Yadav, A.; Pal, M.; Goswamee, R.L. Biocompatible nanocomposite of carboxymethyl cellulose and functionalized carbon–norfloxacin intercalated layered double hydroxides. Carbohydr. Polym. 2018, 186, 282–289. [Google Scholar] [CrossRef]
- Li, L.; Gu, Z.; Gu, W.; Liu, J.; Xu, Z.P. Efficient drug delivery using SiO2-layered double hydroxide nanocomposites. J. Colloid Interface Sci. 2016, 470, 47–55. [Google Scholar] [CrossRef]
- Ray, S.; Saha, S.; Sa, B.; Chakraborty, J. In vivo pharmacological evaluation and efficacy study of methotrexate-encapsulated polymer-coated layered double hydroxide nanoparticles for possible application in the treatment of osteosarcoma. Drug Deliv. Trans. Res. 2017, 7, 259–275. [Google Scholar] [CrossRef]
- Choi, G.; Piao, H.; Alothman, Z.A.; Vinu, A.; Yun, C.O.; Choy, J.H. Anionic clay as the drug delivery vehicle: Tumor targeting function of layered double hydroxide-methotrexate nanohybrid in C33A orthotopic cervical cancer model. Int. J. Nanomed. 2016, 11, 337–348. [Google Scholar] [CrossRef]
- Zhu, R.; Wang, Q.; Zhu, Y.; Wang, Z.; Zhang, H.; Wu, B.; Wu, X.; Wang, S. pH sensitive nano layered double hydroxides reduce the hematotoxicity and enhance the anticancer efficacy of etoposide on non-small cell lung cancer. Acta Biomater. 2016, 29, 320–332. [Google Scholar] [CrossRef]
- Li, L.; Gu, W.; Chen, J.; Chen, W.; Xu, Z.P. Co-delivery of siRNAs and anti-cancer drugs using layered double hydroxide nanoparticles. Biomaterials 2014, 35, 3331–3339. [Google Scholar] [CrossRef]
- Li, L.; Zhang, R.; Gu, W.; Xu, Z.P. Mannose-conjugated layered double hydroxide nanocomposite for targeted siRNA delivery to enhance cancer therapy. Nanomed. Nanotechnol. 2017, 14, 2355–2364. [Google Scholar] [CrossRef]
- Cianciosi, D.; Varela-Lopez, A.; Forbes-Hernandez, T.Y.; Gasparrini, M.; Afrin, S.; Reboredo-Rodriguez, P.; Zhang, J.; Quiles, J.L.; Nabav, S.F.; Battino, M.; et al. Targeting Molecular Pathways in Cancer Stem Cells by Natural Bioactive Compounds. Pharmacol. Res. 2018, 135, 150–165. [Google Scholar] [CrossRef]
- Kim, C.; Kim, B. Anti-Cancer Natural Products and Their Bioactive Compounds Inducing ER Stress-Mediated Apoptosis: A Review. Nutrients 2018, 10, 1021. [Google Scholar] [CrossRef]
- Zhang, H.; Zhu, Y.; Sun, X.; He, X.; Wang, M.; Wang, Z.; Wang, Q.; Zhu, R.; Wang, S. Curcumin-Loaded Layered Double Hydroxide Nanoparticles-Induced Autophagy for Reducing Glioma Cell Migration and Invasion. J. Biomed. Nanotechnol. 2016, 12, 2051–2062. [Google Scholar] [CrossRef]
- Arratia-Quijada, J.; Rivas-Fuentes, S.; Saavedra, K.J.; Lamas, A.M.; Carbajal Arizaga, G.G. Layered Double Hydroxide as a Vehicle to Increase Toxicity of Gallate Ions against Adenocarcinoma Cells. Molecules 2016, 21, 928. [Google Scholar] [CrossRef]
- Chee, C.F.; Leo, B.F.; Lai, C.W. Superparamagnetic iron oxide nanoparticles for drug delivery. In Applications of Nanocomposite Materials in Drug Delivery; Inamuddin, A.M.A., Mohammad, A., Eds.; Woodhead Publishing: Duxford, UK, 2018; pp. 861–903. [Google Scholar]
- Ansari, M.O.; Ahmad, M.F.; Shadab, G.G.H.A.; Siddique, H.R. Superparamagnetic iron oxide nanoparticles based cancer theranostics: A double edge sword to fight against cancer. J. Drug Deliv. Sci. Technol. 2018, 45, 177–183. [Google Scholar] [CrossRef]
- Laurent, S.; Saei, A.A.; Behzadi, S.; Panahifar, A.; Mahmoudi, M. Superparamagnetic iron oxide nanoparticles for delivery of therapeutic agents: Opportunities and challenges. Expert Opin. Drug Deliv. 2014, 11, 1449–1470. [Google Scholar] [CrossRef]
- Mahmoudi, M.; Sant, S.; Wang, B.; Laurent, S.; Sen, T. Superparamagnetic iron oxide nanoparticles (SPIONs): Development, surface modification and applications in chemotherapy. Adv. Drug Deliv. Rev. 2011, 63, 24–46. [Google Scholar] [CrossRef]
- Kheirkhah, P.; Denyer, S.; Bhimani, A.D.; Arnone, G.D.; Esfahani, D.R.; Aguilar, T.; Zakrzewski, J.; Venugopal, I.; Habib, N.; Gallia, G.L.; et al. Magnetic Drug Targeting: A Novel Treatment for Intramedullary Spinal Cord Tumors. Sci. Rep. 2018, 8, 11417. [Google Scholar] [CrossRef]
- Foglia, S.; Ledda, M.; Fioretti, D.; Iucci, G.; Papi, M.; Capellini, G.; Lolli, M.G.; Grimaldi, S.; Rinaldi, M.; Lisi, A. In vitro biocompatibility study of sub-5 nm silica-coated magnetic iron oxide fluorescent nanoparticles for potential biomedical application. Sci. Rep. 2017, 7, 46513. [Google Scholar] [CrossRef] [PubMed]
- Srisa-nga, K.; Mankhetkorn, S.; Okonogi, S.; Khonkarn, R. Delivery of Superparamagnetic Polymeric Micelles Loaded with Quercetin to Hepatocellular Carcinoma Cells. J. Pharm. Sci. 2019, 108, 996–1006. [Google Scholar] [CrossRef] [PubMed]
- Nagesh, P.K.B.; Johnson, N.R.; Boya, V.K.N.; Chowdhury, P.; Othman, S.F.; Khalilzad-Sharghi, V.; Hafeez, B.B.; Ganju, A.; Khan, S.; Behrman, S.W.; et al. PSMA targeted docetaxel-loaded superparamagnetic iron oxide nanoparticles for prostate cancer. Colloids Surf. B Biointerfaces 2016, 144, 8–20. [Google Scholar] [CrossRef]
- Quinto, C.A.; Mohindra, P.; Tong, S.; Bao, G. Multifunctional Superparamagnetic Iron Oxide Nanoparticles for Combined Chemotherapy and Hyperthermia Cancer Treatment. Nanoscale 2015, 7, 12728–12736. [Google Scholar] [CrossRef]
- Fortin, J.-P.; Wilhelm, C.; Servais, J.; Ménager, C.; Bacri, J.-C.; Gazeau, F. Size-Sorted Anionic Iron Oxide Nanomagnets as Colloidal Mediators for Magnetic Hyperthermia. J. Am. Chem. Soc. 2007, 129, 2628–2635. [Google Scholar] [CrossRef]
- Zhou, Y.; Wang, R.; Teng, Z.; Wang, Z.; Hu, B.; Kolios, M.; Chen, H.; Zhang, N.; Wang, Y.; Li, P.; et al. Magnetic nanoparticle-promoted droplet vaporization for in vivo stimuli-responsive cancer theranostics. NPG Asia Mater. 2016, 8, e313. [Google Scholar] [CrossRef]
- Victor, S.P.; Paul, W.; Sharma, C.P. Calcium phosphate nanoplatforms for drug delivery and theranostic applications. In Drug Delivery Nanosystems for Biomedical Applications; Sharma, C.P., Ed.; Elsevier: Amsterdam, The Netherlands, 2018; pp. 163–179. [Google Scholar]
- Bakan, F.; Kara, G.; Cokol Cakmak, M.; Cokol, M.; Denkbas, E.B. Synthesis and characterization of amino acid-functionalized calcium phosphate nanoparticles for siRNA delivery. Colloids Surf. B Biointerfaces 2017, 158, 175–181. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.S.; Lee, J.E.; Byun, E.; Kim, N.W.; Lee, K.; Lee, H.; Sim, S.J.; Lee, D.S.; Jeong, J.H. Target-specific delivery of siRNA by stabilized calcium phosphate nanoparticles using dopa–hyaluronic acid conjugate. J. Control. Release 2014, 192, 122–130. [Google Scholar] [CrossRef]
- Zhang, N.N.; Yu, R.S.; Xu, M.; Cheng, X.Y.; Chen, C.M.; Xu, X.L.; Lu, C.Y.; Lu, K.J.; Chen, M.J.; Zhu, M.L.; et al. Visual targeted therapy of hepatic cancer using homing peptide modified calcium phosphate nanoparticles loading doxorubicin guided by T1 weighted MRI. Nanomed. Nanotechnol. 2018, 14, 2167–2178. [Google Scholar] [CrossRef]
- Liu, L.; Xiong, X.; Shen, M.; Ru, D.; Gao, P.; Zhang, X.; Huang, C.; Sun, Y.; Li, H.; Duan, Y. Co-Delivery of Triptolide and Curcumin for Ovarian Cancer Targeting Therapy via mPEG-DPPE/CaP Nanoparticle. J. Biomed. Nanotechnol. 2018, 14, 1761–1772. [Google Scholar] [CrossRef]
- Jiang, W.; von Roemeling, C.A.; Chen, Y.; Qie, Y.; Liu, X.; Chen, J.; Kim, B.Y.S. Designing nanomedicine for immuno-oncology. Nat. Biomed. Eng. 2017, 1, 0029. [Google Scholar] [CrossRef]
- Wu, J.Y.; Wang, Z.X.; Zhang, G.; Lu, X.; Qiang, G.H.; Hu, W.; Ji, A.L.; Wu, J.H.; Jiang, C.P. Targeted co-delivery of Beclin 1 siRNA and FTY720 to hepatocellular carcinoma by calcium phosphate nanoparticles for enhanced anticancer efficacy. Int. J. Nanomed. 2018, 13, 1265–1280. [Google Scholar] [CrossRef] [PubMed]
- Tseng, Y.-C.; Xu, Z.; Guley, K.; Yuan, H.; Huang, L. Lipid-Calcium Phosphate Nanoparticles for Delivery to the Lymphatic System and SPECT/CT Imaging of Lymph Node Metastases. Biomaterials 2014, 35, 4688–4698. [Google Scholar] [CrossRef] [PubMed]
- Tang, J.; Howard, C.B.; Mahler, S.M.; Thurecht, K.J.; Huang, L.; Xu, Z.P. Enhanced delivery of siRNA to triple negative breast cancer cells in vitro and in vivo through functionalizing lipid-coated calcium phosphate nanoparticles with dual target ligands. Nanoscale 2018, 10, 4258–4266. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.; Xu, J.; Hao, Y.; Zhao, Y.; Qiu, Y.; Jiang, J.; Yu, T.; Ji, P.; Liu, Y. Application of a lipid-coated hollow calcium phosphate nanoparticle in synergistic co-delivery of doxorubicin and paclitaxel for the treatment of human lung cancer A549 cells. Int. J. Nanomed. 2017, 12, 7979–7992. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Zhang, M.; Zhang, L.; Li, L.; Li, S.; Wang, C.; Su, Z.; Yuan, Y.; Pan, W. Designed Synthesis of Lipid-Coated Polyacrylic Acid/Calcium Phosphate Nanoparticles as Dual pH-Responsive Drug-Delivery Vehicles for Cancer Chemotherapy. Chem. A Eur. J. 2017, 23, 6586–6595. [Google Scholar] [CrossRef] [PubMed]
- Pittella, F.; Cabral, H.; Maeda, Y.; Mi, P.; Watanabe, S.; Takemoto, H.; Kim, H.J.; Nishiyama, N.; Miyata, K.; Kataoka, K. Systemic siRNA delivery to a spontaneous pancreatic tumor model in transgenic mice by PEGylated calcium phosphate hybrid micelles. J. Control. Release 2014, 178, 18–24. [Google Scholar] [CrossRef]
- Zhou, Z.; Kennell, C.; Lee, J.Y.; Leung, Y.K.; Tarapore, P. Calcium phosphate-polymer hybrid nanoparticles for enhanced triple negative breast cancer treatment via co-delivery of paclitaxel and miR-221/222 inhibitors. Nanomed. Nanotechnol. 2017, 13, 403–410. [Google Scholar] [CrossRef]
| NDDP | Inorganic Agent(s) | Ther. Agent | DLC/DRE (%) | Stimuli | Study Model | Cancer Type | Preventive Outcomes of NDDPs in Cancer |
|---|---|---|---|---|---|---|---|
| nGO-DOX-cPEG [6] | Graphene | DOX | ~70/>60 | NIR, pH | In vitro: PC3, DU145, LNCaP In vivo: BALB/c nude | Prostate | In vitro: ↑ cancer cells killing by ~80%, ↑ cellular drug uptake, ↑ apoptotic effects (higher expression of p53, p21, bax, c-caspase 3). ↓ cytotoxicity for normal cells. In vivo: ↑ site selective accumulation and prolong blood circulation (>24 h), ↑ sustained drug release and retention. ↓ tumor growth and volume. |
| HSG-DOX [7] | Graphene | DOX | 49.2/>50 | NIR, Redox | In vitro: MDA-MB-231 In vivo: BALB/c nude | Breast | In vitro: ↑ targeted delivery to HA-receptor over expressing tumors, ↑ endo/lysosomal escape, ↑ cytoplasmic release. ↓ cytotoxic even at <10 µg mL−1. In vivo: ↑ preferential accumulation (36 h) and tumor targeting, ↑ sustained drug release and retention. ↓ tumor growth and volume, ↓ side effects (evidenced by improvements in body weight). |
| GO-CHI-HA-SNX-2112 [8] | Graphene | SNX-2112 | >110/>40 | pH | In vitro: A549, NHBE In vivo: Sprague-Dawley | Lung | In vitro: ↑ CD44 receptor mediated cellular uptake and intracellular drug distribution, ↑ cell apoptosis as high as 36.59%, ↑ blood compatibility (haemolysis analysis). ↓ cytotoxic (evidenced by cell viability). In vivo: ↑ immune-tolerant, ↓ tissue swelling, inflammation and lesion formation, ↓ necrocytosis and fibrous proliferation, ↓ systemic side effects. |
| DOX-FA-rGO/ZnS:Mn [9] | Graphene, Zinc, Manganese | DOX | ~35/95 | pH | In vitro: MDA-MB-231, NIH-3T3 In vivo: NS | Breast | In vitro: ↓ cancer cells viability by 50%, FA functionalization decreased the toxic response up to 72 h, FA functionalized system further improved the drug disperseability, bioavailability and selective drug release. In vivo: NS |
| H-MnO2-PEG-C&D [10] | Manganese | Ce6, DOX | >86/>90 | pH, NIR | In vitro: 4T1 In vivo: Balb/c mice | Breast | In vitro: relieve tumor hypoxia, ↓ high photo-toxicity for cancer cells, ↑ intracellular accumulation, ↑ killing cancer. In vivo: ↑ efficient tumor accumulation, ↑ decomposition of H2O2 into O2 and accumulation tumor, ↓ reduce tumor hypoxia, ↓ tumor size, ↓ side effects (evidenced by improvements in body weight), ↑ macrophages infiltration, ↑ cytotoxic T lymphocytes, ↑ TNF-α secretion |
| DOX-PVP-AuNPs [11] | Gold | DOX | NR/91 | pH | In vitro: 549, H460, H540 In vivo: NS | Lung | In vitro: ↑ inhibition of lung cancer cells proliferation, ↑ ROS generation, ↑ cancer cell apoptosis, ↑ mitochondrial membrane depolarization potential (more cellular uptake), ↑ expression of apoptotic cytochrome oxidase C and bax, ↑ expression of tumor suppressing p21, p53, ↑ expression of caspases 3 and 9. In vivo: NS |
| BLM-DOX-PEG-AuNPs [12] | Gold | BLM, DOX | 33/2.3 | pH | In vitro: HeLa In vivo: NS | Ovarian | In vitro: ↑ cytotoxicity for cancerous cells at lower concentration, ↑ active targeting, ↑ cancer cell apoptosis, ↑EC50 values. ↓ agglomeration. In vivo: NS |
| Se-Au-mSiO2-DOX [13] | Gold, Selenium, Silicon | DOX | 8.1/80 | Laser NIR | In vitro: MCF-7, MDA-MB-231 In vivo: BALB/c nude | Breast | In vitro: ↑ reversing multidrug resistance strategy, ↑ intracellular drug release in lysosomes, ↑ cytotoxic effect and cancer cells killing (evidenced by cell shrinkage, chromatin condensation, degeneration, nuclear fragmentation), ↑ apoptosis (~40% of cells in late apoptosis), ↑ ROS generated mediated mitochondrial dysfunction, ↑ expression of tumor suppressing p21, p53, ↑ Bid and Bad expression. ↓ GSH/GSSG ratio, ↓ PARP expression, ↓ Bcl-xl expression, ↓ Src/FAK/AKT pathways. In vivo: ↑ nanoparticles accumulation in tumor sites (24 h), ↑ cellular apoptosis by 10 fold, ↑ caspase 3 by 11 fold, ↑ PARP by 13 fold. ↓ tumor progression, ↓ tumor volume by 5 fold, ↓ adverse effects. |
| Ag-GQDs-DOX [14] | Silver | DOX | NR/NR | pH | In vitro: DU145, HeLa In vivo: NS | Prostate, Cervical | In vitro: ↑ antitumor activity, ↑ apoptosis in cancer cells, ↓ affecting the viability of normal cells, ↑ caspase-3 activity, ↑ caspase-7 activity, ↑ cellular drug uptake. In vivo: NS |
| CPT-CEF [15] | Iron | CPT | NR/65 | pH, Temp. | In vitro: HT29, A549 In vivo: NS | Colon, Lung | In vitro: ↓ cancer cells viability, ↑ membrane rupturing and nuclear fragmentation, ↑ induction of cell toxicity cancer cells, ↑ early apoptosis, ↓ cancer cells proliferation, ↑ mitochondrial membrane depolarization, ↑ caspase-3 activity. In vivo: NS |
| HA-FeOOH-PPy NRs [16] | Iron | HA | NR/NR | Laser | In vitro: MDA-MB-231 In vivo: BALB/c nude | Breast | In vitro: ↓ cancer cells viability, ↑ quick cellular uptake, ↑ selectivity toward CD44 expressing cancer cells, ↑ biocompatible, ↑ cancer cells killing efficiency, ↑ localized drug distribution around the tumor. In vivo: ↑ tumor volume growth (by 11 times), ↓ side effects (evidenced by improvements in body weight). |
| MNP-HC [17] | Iron | TZ | NR/NR | NS | In vitro: SKBR3, MDA-MB-231 In vivo: NS | Breast | In vitro: ↑ up to 97% binding to the tested cell lines, ↑ site-specific phosphorylation in catalytic domain of HER2, ↑ cellular uptake, ↑ anti-cancer proliferative effect (antibody dependent cell mediated cytotoxicity) by 41.8%, ↑ p27kip1 expression. In vivo: NS |
| DOX-MGNSs [18] | Iron, Gold, Silica | DOX | 65.8/86.2 | pH, Temp. | In vitro: ATCC CCL-2 In vivo: NS | Ovarian | In vitro: ↑ cytoxicity for cancer cells, ↑ cellular uptake, ↑ anti-cancer proliferative effect, ↑ disruption of ruffles in cells (loss of morphology). In vivo: NS |
| HA-ionic-TPP-DOX [19] | Phosphonium | DOX | 31.4/91 | pH | In vitro: MCF-7/ADR In vivo: BALB/c nude, Tg(fli1a:eGFP)+/− | Breast | In vitro: ↑ cellular uptake and cytoxicity in tumor cells, ↑ prolong blood circulation, ↑ drug distribution in tumors, ↑ enhanced permeability and retention, punctate distribution (selectively mitochondrial accumulation), ↑ ROS generation. In vivo: ↑ tumor targeting, ↓ tumor growth and tumor progression, ↓ tumor volume, ↓ adverse effects (cardiotoxicity). |
| DOX-BP-Hyd [20] | Phosphorus | DOX | NR/38.8 | NIR, Light | In vitro: MDA-MB-231, A549, HeLa, B16 In vivo: BALB/c nude | Breast, Lung, Cervical, Melanoma | In vitro: ↑ cell killing ability, ↑ tumor ablation effect, ↑ biodegradability, ↑ biosafety. In vivo: ↑ localized drug distribution around the tumor site, ↑ sustained release (<12 h), ↓ tumor growth and volume, ↓ acute side effects (evidenced by improvements in body weight), ↓ toxic to normal tissues. |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Naz, S.; Shamoon, M.; Wang, R.; Zhang, L.; Zhou, J.; Chen, J. Advances in Therapeutic Implications of Inorganic Drug Delivery Nano-Platforms for Cancer. Int. J. Mol. Sci. 2019, 20, 965. https://doi.org/10.3390/ijms20040965
Naz S, Shamoon M, Wang R, Zhang L, Zhou J, Chen J. Advances in Therapeutic Implications of Inorganic Drug Delivery Nano-Platforms for Cancer. International Journal of Molecular Sciences. 2019; 20(4):965. https://doi.org/10.3390/ijms20040965
Chicago/Turabian StyleNaz, Safia, Muhammad Shamoon, Rui Wang, Li Zhang, Juan Zhou, and Jinghua Chen. 2019. "Advances in Therapeutic Implications of Inorganic Drug Delivery Nano-Platforms for Cancer" International Journal of Molecular Sciences 20, no. 4: 965. https://doi.org/10.3390/ijms20040965
APA StyleNaz, S., Shamoon, M., Wang, R., Zhang, L., Zhou, J., & Chen, J. (2019). Advances in Therapeutic Implications of Inorganic Drug Delivery Nano-Platforms for Cancer. International Journal of Molecular Sciences, 20(4), 965. https://doi.org/10.3390/ijms20040965





