“Salicylic Acid Mutant Collection” as a Tool to Explore the Role of Salicylic Acid in Regulation of Plant Growth under a Changing Environment
Abstract
1. Introduction
2. Results
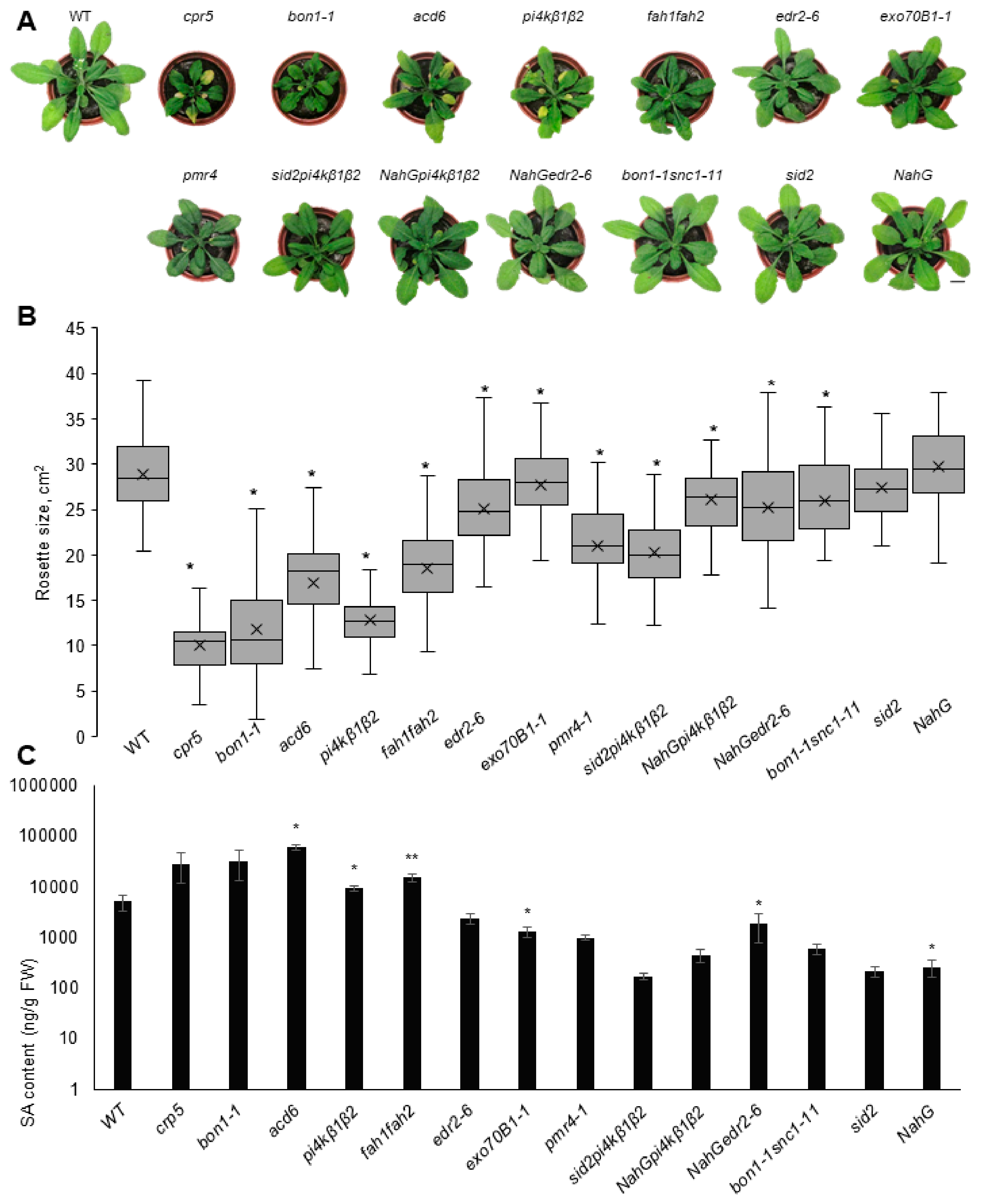
2.1. Cultivation Conditions Influence the Phenotype of the SA Collection Mutants
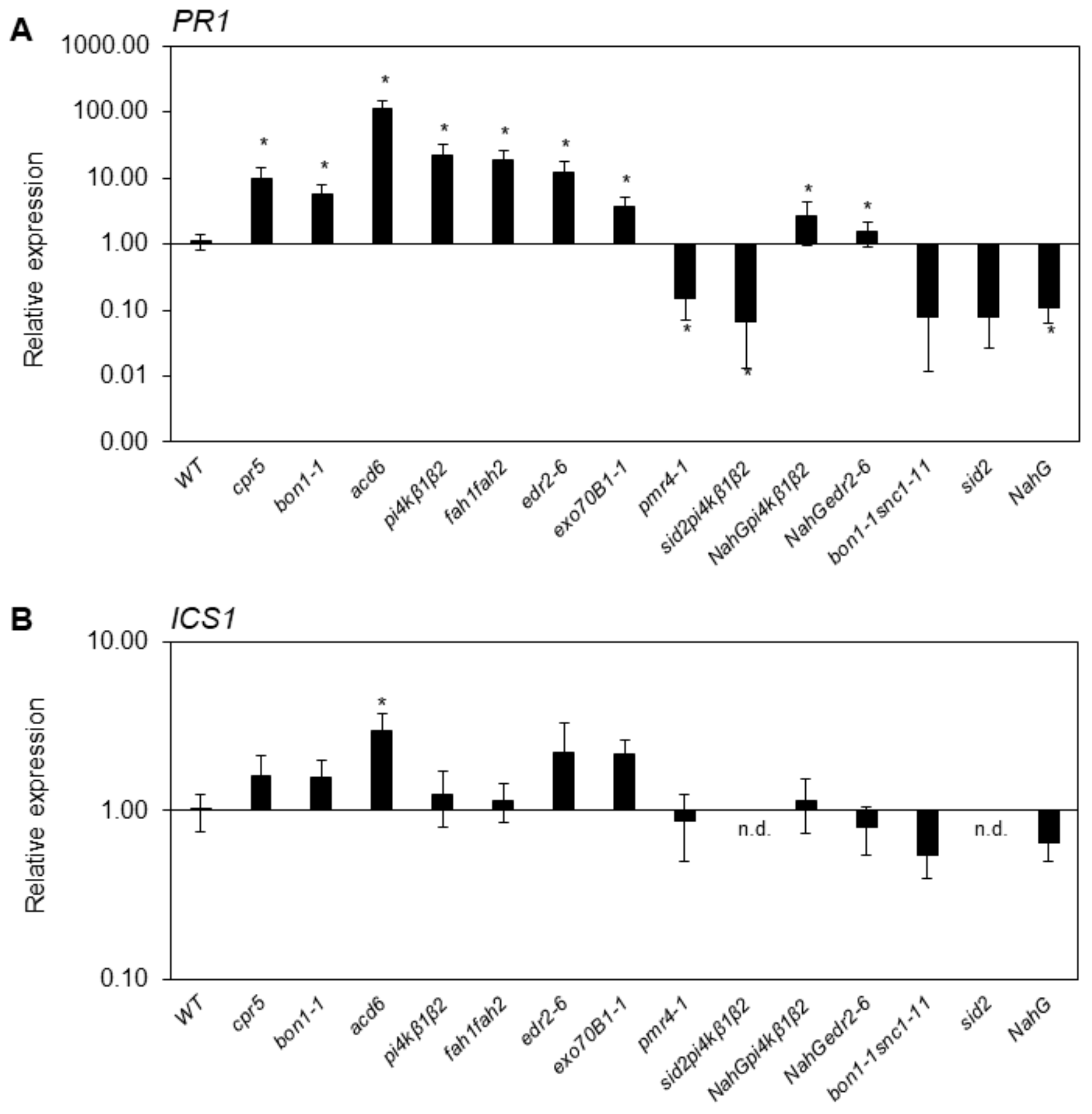
2.2. SA-Related Gene Transcription Varies in Different Growth Conditions
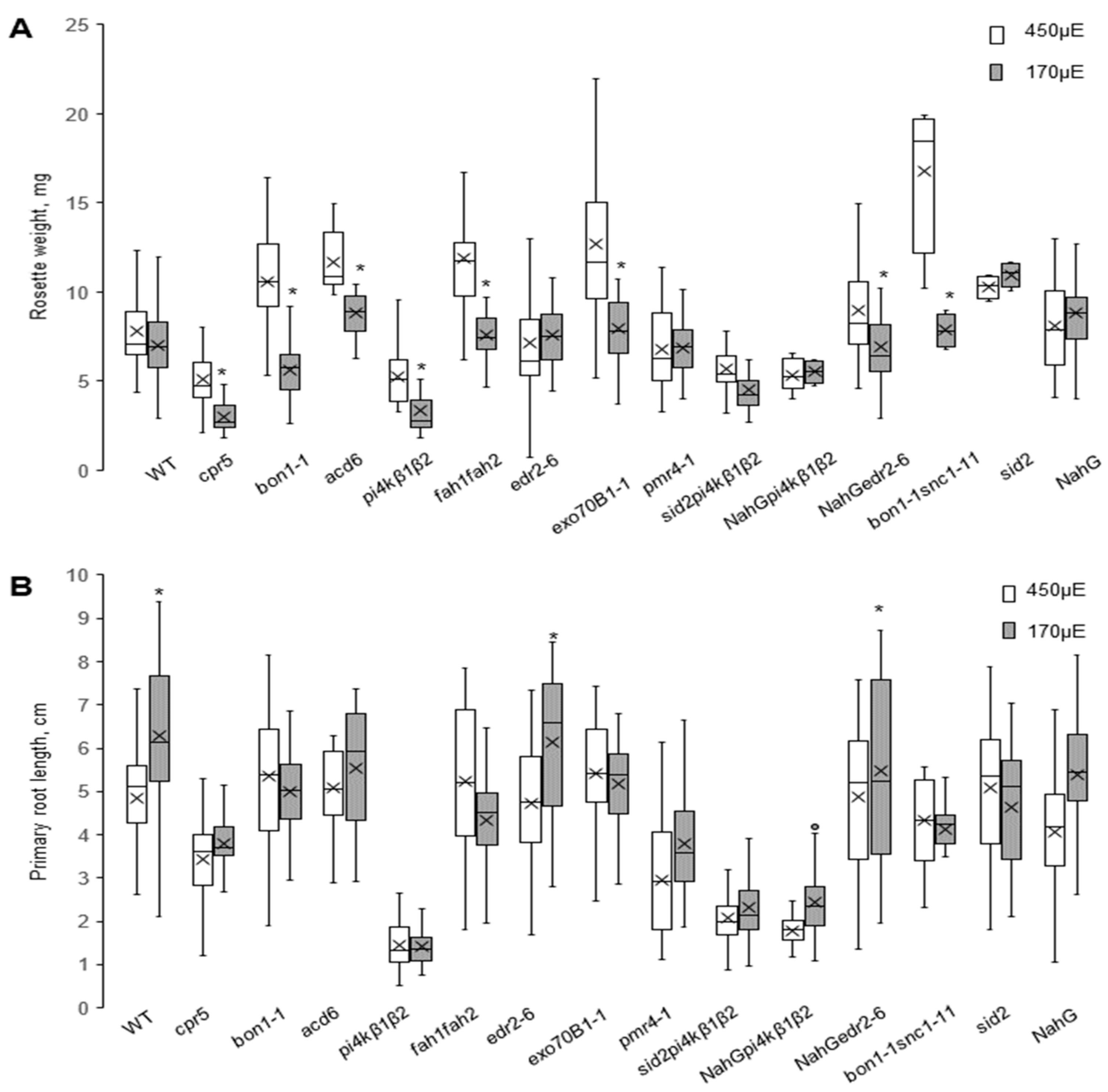
2.3. Overaccumulation of SA Increases High Light Sensitivity in In-Vitro-Grown Seedlings
2.4. Salicylic Acid’s Effect on the Root Growth and Shoot Growth is Distinct
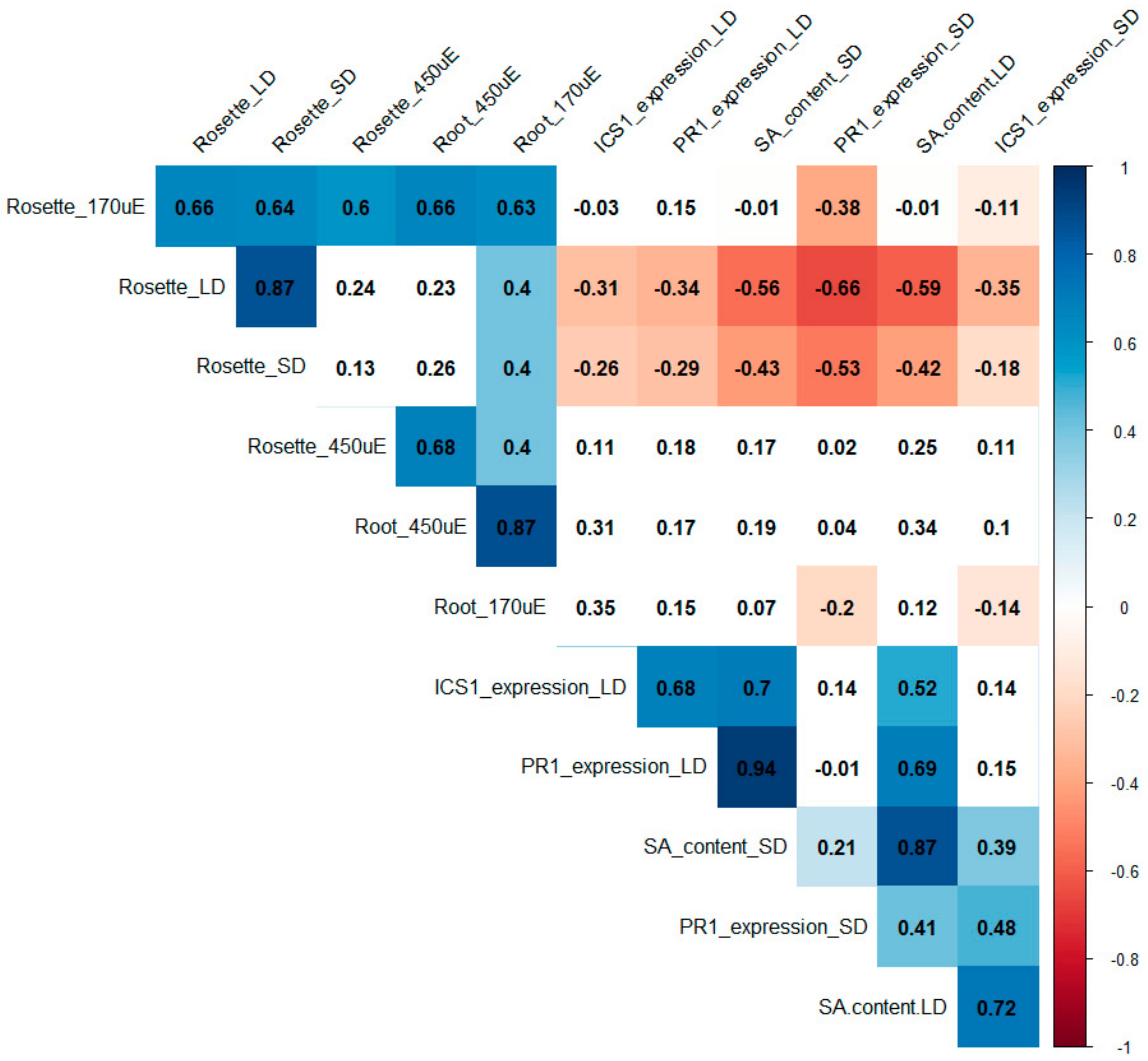
3. Discussion
4. Materials and Methods
4.1. Plant Material and Growth Conditions
4.2. Plant Phenotyping
4.3. SA Concentration Measurements
4.4. Gene Transcription Analysis
4.5. Photosynthetic Parameter Analysis
4.6. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| SA | Salicylic acid |
| SA-OA | Salicylic acid overaccumulating mutants |
| ICS1 | Isochorismate synthase 1 |
| PR1 | Pathogenesis related protein 1 |
| SD | Short day conditions |
| LD | Long day conditions |
References
- Rajjou, L.; Belghazi, M.; Huguet, R.; Robin, C.; Moreau, A.; Job, C.; Job, D. Proteomic Investigation of the Effect of Salicylic Acid on Arabidopsis Seed Germination and Establishment of Early Defense Mechanisms. Plant Physiol. 2006, 141, 910–923. [Google Scholar] [CrossRef] [PubMed]
- Vanacker, H.; Lu, H.; Rate, D.N.; Greenberg, J.T. A role for salicylic acid and NPR1 in regulating cell growth in Arabidopsis. Plant J. Cell Mol. Biol. 2001, 28, 209–216. [Google Scholar] [CrossRef] [PubMed]
- Martínez, C.; Pons, E.; Prats, G.; León, J. Salicylic acid regulates flowering time and links defence responses and reproductive development. Plant J. Cell Mol. Biol. 2004, 37, 209–217. [Google Scholar] [CrossRef]
- Jayakannan, M.; Bose, J.; Babourina, O.; Rengel, Z.; Shabala, S. Salicylic acid improves salinity tolerance in Arabidopsis by restoring membrane potential and preventing salt-induced K+ loss via a GORK channel. J. Exp. Bot. 2013, 64, 2255–2268. [Google Scholar] [CrossRef] [PubMed]
- Bravo, R.E.; Chen, G.; Grosser, K.; Dam, N.M.V.; Leiss, K.A.; Klinkhamer, P.G.L. Ultraviolet radiation enhances salicylic acid-mediated defense signaling and resistance to Pseudomonas syringae DC3000 in a jasmonic acid-deficient tomato mutant. Plant Signal. Behav. 2019, 14, e1581560. [Google Scholar] [CrossRef] [PubMed]
- Klessig, D.F.; Choi, H.W.; Dempsey, D.A. Systemic Acquired Resistance and Salicylic Acid: Past, Present, and Future. Mol. Plant Microbe Interact. MPMI 2018, 31, 871–888. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Li, X. Salicylic acid: Biosynthesis, perception, and contributions to plant immunity. Curr. Opin. Plant Biol. 2019, 50, 29–36. [Google Scholar] [CrossRef]
- Wildermuth, M.C.; Dewdney, J.; Wu, G.; Ausubel, F.M. Isochorismate synthase is required to synthesize salicylic acid for plant defence. Nature 2001, 414, 562–565. [Google Scholar] [CrossRef]
- Delaney, T.P.; Uknes, S.; Vernooij, B.; Friedrich, L.B.; Weymann, K.; Negrotto, D.V.; Gaffney, T.; Gut-Rella, M.; Kessmann, H.; Ward, E.; et al. A central role of salicylic Acid in plant disease resistance. Science 1994, 266, 1247–1250. [Google Scholar] [CrossRef]
- Fragnière, C.; Serrano, M.; Abou-Mansour, E.; Métraux, J.P.; L’Haridon, F. Salicylic acid and its location in response to biotic and abiotic stress. FEBS Lett. 2011, 585, 1847–1852. [Google Scholar] [CrossRef]
- Glazebrook, J. Contrasting mechanisms of defense against biotrophic and necrotrophic pathogens. Annu. Rev. Phytopathol. 2005, 43, 205–227. [Google Scholar] [CrossRef] [PubMed]
- Canet, J.V.; Dobón, A.; Roig, A.; Tornero, P. Structure-function analysis of npr1 alleles in Arabidopsis reveals a role for its paralogs in the perception of salicylic acid. Plant Cell Environ. 2010, 33, 1911–1922. [Google Scholar] [CrossRef] [PubMed]
- Ding, Y.; Shaholli, D.; Mou, Z. A large-scale genetic screen for mutants with altered salicylic acid accumulation in Arabidopsis. Front. Plant Sci. 2015, 5, 763. [Google Scholar] [CrossRef] [PubMed]
- Rivas-San Vicente, M.; Plasencia, J. Salicylic acid beyond defence: Its role in plant growth and development. J. Exp. Bot. 2011, 62, 3321–3338. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Yang, S.; Yang, H.; Hua, J. The TIR-NB-LRR Gene SNC1 Is Regulated at the Transcript Level by Multiple Factors. Mol. Plant. Microbe Interact. 2007, 20, 1449–1456. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Lin, F.; Krishnamoorthy, P.; Schubert, V.; Hause, G.; Heilmann, M.; Heilmann, I. A dual role for cell plate-associated PI4Kβ in endocytosis and phragmoplast dynamics during plant somatic cytokinesis. EMBO J. 2019, 38, e100303. [Google Scholar] [CrossRef] [PubMed]
- Kalachova, T.; Janda, M.; Šašek, V.; Ortmannová, J.; Nováková, P.; Dobrev, I.P.; Kravets, V.; Guivarc’h, A.; Moura, D.; Burketová, L.; et al. Identification of salicylic acid-independent responses in an Arabidopsis phosphatidylinositol 4-kinase beta double mutant. Ann. Bot. 2019. [Google Scholar] [CrossRef]
- Kulich, I.; Pečenková, T.; Sekereš, J.; Smetana, O.; Fendrych, M.; Foissner, I.; Höftberger, M.; Žárský, V. Arabidopsis Exocyst Subcomplex Containing Subunit EXO70B1 Is Involved in Autophagy-Related Transport to the Vacuole. Traffic 2013, 14, 1155–1165. [Google Scholar] [CrossRef]
- König, S.; Feussner, K.; Schwarz, M.; Kaever, A.; Iven, T.; Landesfeind, M.; Ternes, P.; Karlovsky, P.; Lipka, V.; Feussner, I. Arabidopsis mutants of sphingolipid fatty acid α-hydroxylases accumulate ceramides and salicylates. New Phytol. 2012, 196, 1086–1097. [Google Scholar] [CrossRef]
- Yoshida, S.; Ito, M.; Nishida, I.; Watanabe, A. Identification of a novel gene HYS1/CPR5 that has a repressive role in the induction of leaf senescence and pathogen-defence responses in Arabidopsis thaliana. Plant J. 2002, 29, 427–437. [Google Scholar] [CrossRef]
- Rate, D.N.; Cuenca, J.V.; Bowman, G.R.; Guttman, D.S.; Greenberg, J.T. The Gain-of-Function Arabidopsis acd6 Mutant Reveals Novel Regulation and Function of the Salicylic Acid Signaling Pathway in Controlling Cell Death, Defenses, and Cell Growth. Plant Cell 1999, 11, 1695–1708. [Google Scholar] [CrossRef] [PubMed]
- Preuss, M.L.; Schmitz, A.J.; Thole, J.M.; Bonner, H.K.S.; Otegui, M.S.; Nielsen, E. A role for the RabA4b effector protein PI-4Kbeta1 in polarized expansion of root hair cells in Arabidopsis thaliana. J. Cell Biol. 2006, 172, 991–998. [Google Scholar] [CrossRef] [PubMed]
- Vorwerk, S.; Schiff, C.; Santamaria, M.; Koh, S.; Nishimura, M.; Vogel, J.; Somerville, C.; Somerville, S. EDR2 negatively regulates salicylic acid-based defenses and cell death during powdery mildew infections of Arabidopsis thaliana. BMC Plant Biol. 2007, 7, 35. [Google Scholar] [CrossRef] [PubMed]
- Nishimura, M.T.; Stein, M.; Hou, B.H.; Vogel, J.P.; Edwards, H.; Somerville, S.C. Loss of a callose synthase results in salicylic acid-dependent disease resistance. Science 2003, 301, 969–972. [Google Scholar] [CrossRef] [PubMed]
- Šašek, V.; Janda, M.; Delage, E.; Puyaubert, J.; Guivarc’h, A.; Maseda, E.L.; Dobrev, P.I.; Caius, J.; Bóka, K.; Valentová, O.; et al. Constitutive salicylic acid accumulation in pi4kIIIβ1β2 Arabidopsis plants stunts rosette but not root growth. New Phytol. 2014, 203, 805–816. [Google Scholar] [CrossRef] [PubMed]
- Nawrath, C.; Métraux, J.P. Salicylic Acid Induction—Deficient Mutants of Arabidopsis Express PR-2 and PR-5 and Accumulate High Levels of Camalexin after Pathogen Inoculation. Plant Cell 1999, 11, 1393–1404. [Google Scholar]
- Maxwell, K.; Johnson, G.N. Chlorophyll fluorescence—A practical guide. J. Exp. Bot. 2000, 51, 659–668. [Google Scholar] [CrossRef]
- Hendrickson, L.; Furbank, R.T.; Chow, W.S. A Simple Alternative Approach to Assessing the Fate of Absorbed Light Energy Using Chlorophyll Fluorescence. Photosynth. Res. 2004, 82, 73. [Google Scholar] [CrossRef]
- Yokawa, K.; Kagenishi, T.; Kawano, T.; Mancuso, S.; Baluška, F. Illumination of Arabidopsis roots induces immediate burst of ROS production. Plant Signal. Behav. 2011, 6, 1460–1464. [Google Scholar] [CrossRef]
- Silva-Navas, J.; Moreno-Risueno, M.A.; Manzano, C.; Pallero-Baena, M.; Navarro-Neila, S.; Téllez-Robledo, B.; Garcia-Mina, J.M.; Baigorri, R.; Gallego, F.J.; Pozo, J.C. D-Root: A system for cultivating plants with the roots in darkness or under different light conditions. Plant J. 2015, 84, 244–255. [Google Scholar] [CrossRef]
- Janda, M.; Ruelland, E. Magical mystery tour: Salicylic acid signalling. Environ. Exp. Bot. 2015, 114, 117–128. [Google Scholar] [CrossRef]
- Nie, H.; Wu, Y.; Yao, C.; Tang, D. Suppression of edr2-mediated powdery mildew resistance, cell death and ethylene-induced senescence by mutations in ALD1 in Arabidopsis. J. Genet. Genom. Yi Chuan Xue Bao 2011, 38, 137–148. [Google Scholar] [CrossRef] [PubMed]
- Smith, L.M.; Bomblies, K.; Weigel, D. Complex evolutionary events at a tandem cluster of Arabidopsis thaliana genes resulting in a single-locus genetic incompatibility. PLoS Genet. 2011, 7, e1002164. [Google Scholar] [CrossRef] [PubMed]
- Zhu, W.; Zaidem, M.; Van de Weyer, A.L.; Gutaker, R.M.; Chae, E.; Kim, S.T.; Bemm, F.; Li, L.; Todesco, M.; Schwab, R.; et al. Modulation of ACD6 dependent hyperimmunity by natural alleles of an Arabidopsis thaliana NLR resistance gene. PLOS Genet. 2018, 14, e1007628. [Google Scholar] [CrossRef] [PubMed]
- Miura, K.; Lee, J.; Miura, T.; Hasegawa, P.M. SIZ1 controls cell growth and plant development in Arabidopsis through salicylic acid. Plant Cell Physiol. 2010, 51, 103–113. [Google Scholar] [CrossRef]
- Todesco, M.; Kim, S.T.; Chae, E.; Bomblies, K.; Zaidem, M.; Smith, L.M.; Weigel, D.; Laitinen, R.A.E. Activation of the Arabidopsis thaliana Immune System by Combinations of Common ACD6 Alleles. PLoS Genet. 2014, 10, e1004459. [Google Scholar] [CrossRef] [PubMed]
- Ellinger, D.; Naumann, M.; Falter, C.; Zwikowics, C.; Jamrow, T.; Manisseri, C.; Somerville, S.C.; Voigt, C.A. Elevated Early Callose Deposition Results in Complete Penetration Resistance to Powdery Mildew in Arabidopsis. Plant Physiol. 2013, 161, 1433–1444. [Google Scholar] [CrossRef]
- Khurana, J.P.; Cleland, C.F. Role of Salicylic Acid and Benzoic Acid in Flowering of a Photoperiod-Insensitive Strain, Lemna paucicostata LP6 1. Plant Physiol. 1992, 100, 1541–1546. [Google Scholar] [CrossRef]
- Martínez-Medina, A.; Appels, F.V.W.; van Wees, S.C.M. Impact of salicylic acid- and jasmonic acid-regulated defences on root colonization by Trichoderma harzianum T-78. Plant Signal. Behav. 2017, 12, e1345404. [Google Scholar] [CrossRef]
- Badri, D.V.; Loyola-Vargas, V.M.; Du, J.; Stermitz, F.R.; Broeckling, C.D.; Iglesias-Andreu, L.; Vivanco, J.M. Transcriptome analysis of Arabidopsis roots treated with signaling compounds: A focus on signal transduction, metabolic regulation and secretion. New Phytol. 2008, 179, 209–223. [Google Scholar] [CrossRef]
- Pasternak, T.; Groot, E.P.; Kazantsev, F.V.; Teale, W.; Omelyanchuk, N.; Kovrizhnykh, V.; Palme, K.; Mironova, V.V. Salicylic Acid Affects Root Meristem Patterning via Auxin Distribution in a Concentration-Dependent Manner. Plant Physiol. 2019, 180, 1725–1739. [Google Scholar] [CrossRef]
- Liu, P.; Xu, Z.S.; Pan-Pan, L.; Hu, D.; Chen, M.; Li, L.C.; Ma, Y.Z. A wheat PI4K gene whose product possesses threonine autophophorylation activity confers tolerance to drought and salt in Arabidopsis. J. Exp. Bot. 2013, 64, 2915–2927. [Google Scholar] [CrossRef] [PubMed]
- Borghi, M.; Rus, A.; Salt, D.E. Loss-of-Function of Constitutive Expresser of Pathogenesis Related Genes5 Affects Potassium Homeostasis in Arabidopsis thaliana. PLoS ONE 2011, 6, e26360. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Gao, G.; Zhang, S.; Wang, C.; Yang, X.; Wang, Y.; Su, X.; Du, J.; Yang, C. Arabidopsis CPR5 independently regulates seed germination and postgermination arrest of development through LOX pathway and ABA signaling. PLoS ONE 2011, 6, e19406. [Google Scholar] [CrossRef] [PubMed]
- Okuma, E.; Nozawa, R.; Murata, Y.; Miura, K. Accumulation of endogenous salicylic acid confers drought tolerance to Arabidopsis. Plant Signal. Behav. 2014, 9. [Google Scholar] [CrossRef] [PubMed]
- Aki, T.; Konishi, M.; Kikuchi, T.; Fujimori, T.; Yoneyama, T.; Yanagisawa, S. Distinct modulations of the hexokinase1-mediated glucose response and hexokinase1-independent processes by HYS1/CPR5 in Arabidopsis. J. Exp. Bot. 2007, 58, 3239–3248. [Google Scholar] [CrossRef] [PubMed]
- Miller, R.N.G.; Costa Alves, G.S.; Van Sluys, M.A. Plant immunity: Unravelling the complexity of plant responses to biotic stresses. Ann. Bot. 2017, 119, 681–687. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Wang, Z.Y. At the intersection of plant growth and immunity. Cell Host Microbe 2014, 15, 400–402. [Google Scholar] [CrossRef]
- Karasov, T.L.; Chae, E.; Herman, J.J.; Bergelson, J. Mechanisms to Mitigate the Trade-Off between Growth and Defense. Plant Cell 2017, 29, 666–680. [Google Scholar] [CrossRef]
- Schindelin, J.; Arganda-Carreras, I.; Frise, E.; Kaynig, V.; Longair, M.; Pietzsch, T.; Preibisch, S.; Rueden, C.; Saalfeld, S.; Schmid, B.; et al. Fiji: An open-source platform for biological-image analysis. Nat. Methods 2012, 9, 676–682. [Google Scholar] [CrossRef]
- Kalachova, T.; Leontovyčová, H.; Iakovenko, O.; Pospíchalová, R.; Maršík, P.; Klouček, P.; Janda, M.; Valentová, O.; Kocourková, D.; Martinec, J.; et al. Interplay between phosphoinositides and actin cytoskeleton in the regulation of immunity related responses in Arabidopsis thaliana seedlings. Environ. Exp. Bot. 2019, 167, 103867. [Google Scholar]
- Huang, J.; Gu, M.; Lai, Z.; Fan, B.; Shi, K.; Zhou, Y.H.; Yu, J.Q.; Chen, Z. Functional Analysis of the Arabidopsis PAL Gene Family in Plant Growth, Development, and Response to Environmental Stress. Plant Physiol. 2010, 153, 1526–1538. [Google Scholar] [CrossRef] [PubMed]
- Simko, W. R Package “Corrplot”: Visualization of a Correlation Matrix (Version 0.84). Available online: https://github.com/taiyun/corrplot (accessed on 2 August 2018).
| Mutant Name | Targeted Gene | Targeted Process | Reference | |
|---|---|---|---|---|
| cpr5 | CPR5 | Constitutive Expression of Pathogenesis-related genes 5 | Constitutive expression of pathogenesis-related genes 5 | Yoshida et el. 2002 [20] |
| bon1-1 | BON1 | BONZAI 1 | Negative regulator of cell death, defence responses and several R genes | Li et al. 2007 [15] |
| acd6 | ACD6 | Accelerated Cell Death 6 | Dose-dependent activation of defence signalling, accelerated cell death observed | Rate et al. 1999 [21] |
| pi4kβ1β2 | PI4Kβ1, PI4Kβ2 | Phosphatidylinositol-4-kinase β1 and β2 | Second messenger, phosphatidyl inositol-4-phosphate production | Preuss et al. 2006 [22] |
| fah1fah2 | FAH1, FAH2 | Fatty acid5-hydroxylase 1 and 2 | Fatty acid hydroxylation | Konig et al. 2012 [19] |
| edr2-6 | EDR2 | Enhanced Disease Resistance 2 | Negative regulation of cell death | Vorwerk et al. 2008 [23] |
| exo70B1-1 | EXO70B1 | Exocyst Complex Component EXO70B1 | Endomembrane trafficking | Kulich et al. 2013 [18] |
| pmr4-1 | CALS12 | Callose Synthase 12 | Pathogen-induced callose synthesis | Nishimura et al. 2003 [24] |
| sid2 pi4kβ1β2 | ICS1, PI4Kβ1, PI4Kβ2 | Isochorismate synthase 1, phosphatidylinositol-4-kinase β1 and β2 | SA biosynthesis, second messenger inositol-1,4,5-trisphosphate production | Sasek et al. 2014 [25] |
| NahG pi4kβ1β2 | NahG, PI4Kβ1, PI4Kβ2 | SA hydroxylase, phosphatidylinositol-4-kinase β1 and β2 | SA degradation, second messenger inositol-1,4,5-trisphosphate production | Sasek et al. 2014 [25] |
| NahG edr2-6 | NahG, EDR2 | SA hydroxylase, enhanced disease resistance 2 | SA degradation, negative regulation of cell death | Vorwerk et al. 2008 [23] |
| bon1-1 snc1-11 | BON1, SNC1 | BONZAI 1, Suppresssor npr1-1, constitutive 1 | bon1-1 crossed to the snc1-11, loss-of-function point mutation of the SNC1 | Li et al. 2007 [15] |
| sid2 | ICS1 | Isochorismate synthase 1 | SA biosynthesis | Wildermuth et al. 2001 [8] |
| NahG | NahG | SA hydroxylase | SA degradation | Nawrath and Metraux 1999 [26] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pluhařová, K.; Leontovyčová, H.; Stoudková, V.; Pospíchalová, R.; Maršík, P.; Klouček, P.; Starodubtseva, A.; Iakovenko, O.; Krčková, Z.; Valentová, O.; et al. “Salicylic Acid Mutant Collection” as a Tool to Explore the Role of Salicylic Acid in Regulation of Plant Growth under a Changing Environment. Int. J. Mol. Sci. 2019, 20, 6365. https://doi.org/10.3390/ijms20246365
Pluhařová K, Leontovyčová H, Stoudková V, Pospíchalová R, Maršík P, Klouček P, Starodubtseva A, Iakovenko O, Krčková Z, Valentová O, et al. “Salicylic Acid Mutant Collection” as a Tool to Explore the Role of Salicylic Acid in Regulation of Plant Growth under a Changing Environment. International Journal of Molecular Sciences. 2019; 20(24):6365. https://doi.org/10.3390/ijms20246365
Chicago/Turabian StylePluhařová, Kamila, Hana Leontovyčová, Věra Stoudková, Romana Pospíchalová, Petr Maršík, Pavel Klouček, Anastasiia Starodubtseva, Oksana Iakovenko, Zuzana Krčková, Olga Valentová, and et al. 2019. "“Salicylic Acid Mutant Collection” as a Tool to Explore the Role of Salicylic Acid in Regulation of Plant Growth under a Changing Environment" International Journal of Molecular Sciences 20, no. 24: 6365. https://doi.org/10.3390/ijms20246365
APA StylePluhařová, K., Leontovyčová, H., Stoudková, V., Pospíchalová, R., Maršík, P., Klouček, P., Starodubtseva, A., Iakovenko, O., Krčková, Z., Valentová, O., Burketová, L., Janda, M., & Kalachova, T. (2019). “Salicylic Acid Mutant Collection” as a Tool to Explore the Role of Salicylic Acid in Regulation of Plant Growth under a Changing Environment. International Journal of Molecular Sciences, 20(24), 6365. https://doi.org/10.3390/ijms20246365





