Pericytes in Microvessels: From “Mural” Function to Brain and Retina Regeneration
Abstract
1. Introduction
2. Pericytes: Canonical and New Functions
2.1. Angiogenesis Modulation and Vessel Stabilization
2.2. Blood Flow Regulation
2.3. Anatomical Stabilization of the BBB and BRB
2.4. Mesenchymal Stem Cell Features
3. Pericytes in Brain Disease, Repair and Preservation
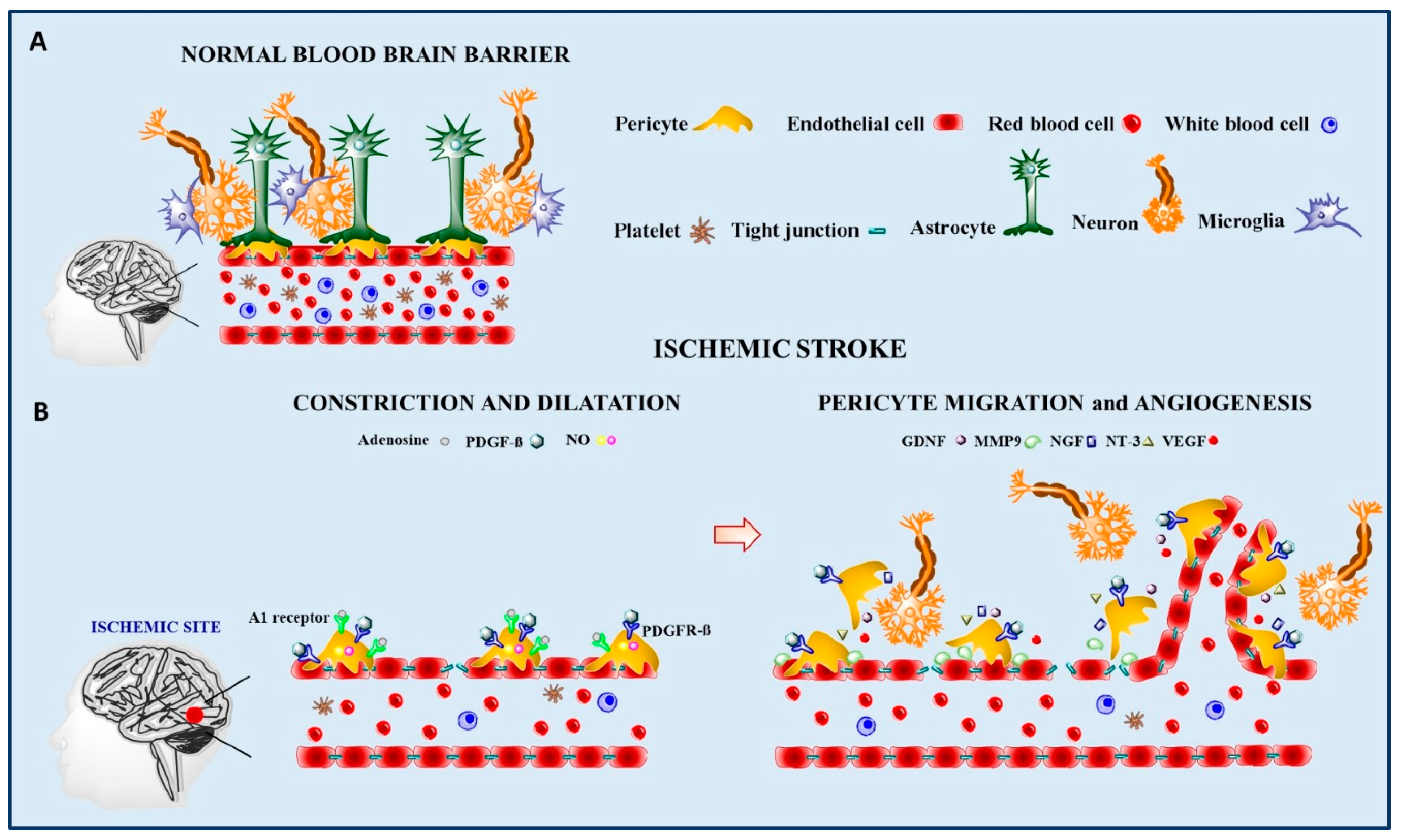
3.1. Ischemia-Reperfusion
3.2. Pericyte Regenerative Potential in Ischemia-Reperfusion
3.3. Tumor
3.4. Pericyte-Targeted Anti-Tumor Therapy
4. Pericytes in Retinal Diseases and Repair
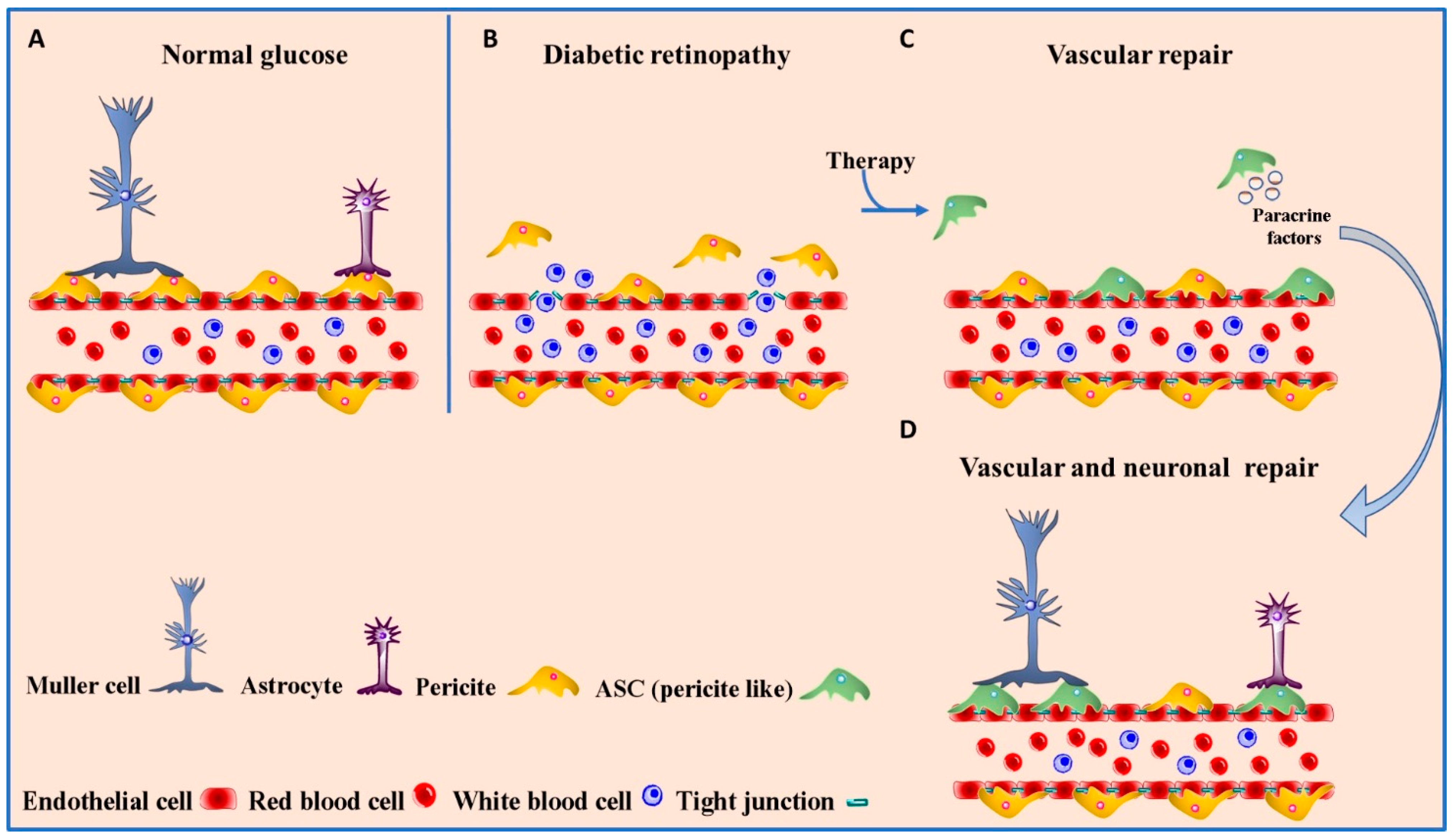
4.1. Diabetic Retinopathy
4.2. Uveal Melanoma
5. Conclusions and Future Perspectives
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Diaz-Flores, L.; Gutierrez, R.; Madrid, J.F.; Varela, H.; Valladares, F.; Acosta, E.; Martin-Vasallo, P.; Diaz-Flores, L. Pericytes. Morphofunction, interactions and pathology in a quiescent and activated mesenchymal cell niche. Histol Histopathol. 2009, 24, 909–969. [Google Scholar] [PubMed]
- Zimmermann, K.W. Der feinere Bau der Blutcapillaren. Z. Anat. Entwickl. 1923, 68, 29–109. [Google Scholar] [CrossRef]
- Mills, S.J.; Cowin, A.J.; Kaur, P. Pericytes, mesenchymal stem cells and the wound healing process. Cells 2013, 2, 621–634. [Google Scholar] [CrossRef] [PubMed]
- Armulik, A.; Abramsson, A.; Betsholtz, C. Endothelial/pericyte interactions. Circ Res. 2005, 97, 512–523. [Google Scholar] [CrossRef]
- Bergers, G.; Song, S. The role of pericytes in blood-vessel formation and maintenance. Neuro-Oncol. 2005, 7, 452–464. [Google Scholar] [CrossRef]
- Gerhardt, H.; Betsholtz, C. Endothelial-pericyte interactions in angiogenesis. Cell Tissue Res. 2003, 314, 15–23. [Google Scholar] [CrossRef]
- Shepro, D.; Morel, N.M. Pericyte physiology. FASEB J. 1993, 7, 1031–1038. [Google Scholar] [CrossRef]
- Rucker, H.K.; Wynder, H.J.; Thomas, W.E. Cellular mechanisms of CNS pericytes. Brain Res. Bull. 2000, 51, 363–369. [Google Scholar] [CrossRef]
- Von Tell, D.; Armulik, A.; Betsholtz, C. Pericytes and vascular stability. Exp. Cell Res. 2006, 312, 623–629. [Google Scholar] [CrossRef]
- Hamilton, N.B.; Attwell, D.; Hall, C.N. Pericyte-mediated regulation of capillary diameter: A component of neurovascular coupling in health and disease. Front. Neuroenergetics 2010, 2, 5. [Google Scholar] [CrossRef]
- Lupo, G.; Caporarello, N.; Olivieri, M.; Cristaldi, M.; Motta, C.; Bramanti, V.; Avola, R.; Salmeri, M.; Nicoletti, F.; Anfuso, C.D. Anti-angiogenic Therapy in Cancer: Downsides and New Pivots for Precision Medicine. Front. Pharmacol. 2016, 7, 519. [Google Scholar] [CrossRef] [PubMed]
- Crisan, M.; Chen, C.W.; Corselli, M.; Andriolo, G.; Lazzari, L.; Peault, B. Perivascular multipotent progenitor cells in human organs. Ann. N. Y. Acad. Sci. 2009, 1176, 118–123. [Google Scholar] [CrossRef] [PubMed]
- Betsholtz, C. Insight into the physiological functions of PDGF through genetic studies in mice. Cytokine Growth Factor Rev. 2004, 15, 215–228. [Google Scholar] [CrossRef] [PubMed]
- Lindahl, P.; Johansson, B.R.; Leveen, P.; Betsholtz, C. Pericyte loss and microaneurysm formation in PDGF-B-deficient mice. Science 1997, 277, 242–245. [Google Scholar] [CrossRef] [PubMed]
- Leveen, P.; Pekny, M.; Gebre-Medhin, S.; Swolin, B.; Larsson, E.; Betsholtz, C. Mice deficient for PDGF B show renal, cardiovascular, and hematological abnormalities. Genes Dev. 1994, 8, 1875–1887. [Google Scholar] [CrossRef] [PubMed]
- Soriano, P. Abnormal kidney development and hematological disorders in PDGF beta-receptor mutant mice. Genes Dev. 1994, 8, 1888–1896. [Google Scholar] [CrossRef] [PubMed]
- Enge, M.; Bjarnegard, M.; Gerhardt, H.; Gustafsson, E.; Kalen, M.; Asker, N.; Hammes, H.P.; Shani, M.; Fassler, R.; Betsholtz, C. Endothelium-specific platelet-derived growth factor-B ablation mimics diabetic retinopathy. EMBO J. 2002, 21, 4307–4316. [Google Scholar] [CrossRef]
- Hellstrom, M.; Kalen, M.; Lindahl, P.; Abramsson, A.; Betsholtz, C. Role of PDGF-B and PDGFR-beta in recruitment of vascular smooth muscle cells and pericytes during embryonic blood vessel formation in the mouse. Development 1999, 126, 3047–3055. [Google Scholar]
- Gaengel, K.; Genove, G.; Armulik, A.; Betsholtz, C. Endothelial-mural cell signaling in vascular development and angiogenesis. Arterioscler. Thromb. Vasc. Biol. 2009, 29, 630–638. [Google Scholar] [CrossRef]
- Jeansson, M.; Gawlik, A.; Anderson, G.; Li, C.J.; Kerjaschki, D.; Henkelman, M.; Quaggin, S.E. Angiopoietin-1 is essential in mouse vasculature during development and in response to injury. J. Clin. Invest. 2011, 121, 2278–2289. [Google Scholar] [CrossRef]
- Uemura, A.; Ogawa, M.; Hirashima, M.; Fujiwara, T.; Koyama, S.; Takagi, H.; Honda, Y.; Wiegand, S.J.; Yancopoulos, G.D.; Nishikawa, S. Recombinant angiopoietin-1 restores higher-order architecture of growing blood vessels in mice in the absence of mural cells. J. Clin. Invest. 2002, 110, 1619–1628. [Google Scholar] [CrossRef] [PubMed]
- Teichert-Kuliszewska, K.; Maisonpierre, P.C.; Jones, N.; Campbell, A.I.M.; Master, Z.; Bendeck, M.P.; Alitalo, K.; Dumont, D.J.; Yancopoulos, G.D.; Stewart, D.J. Biological action of angiopoietin-2 in a fibrin matrix model of angiogenesis is associated with activation of Tie2. Cardiovasc. Res. 2001, 49, 659–670. [Google Scholar] [CrossRef]
- Sweeney, M.D.; Ayyadurai, S.; Zlokovic, B.V. Pericytes of the neurovascular unit: Key functions and signaling pathways. Nat. Neurosci. 2016, 19, 771–783. [Google Scholar] [CrossRef] [PubMed]
- Lupo, G.; Motta, C.; Salmeri, M.; Spina-Purrello, V.; Alberghina, M.; Anfuso, C.D. An in vitro retinoblastoma human triple culture model of angiogenesis: A modulatory effect of TGF-beta. Cancer Lett. 2014, 354, 181–188. [Google Scholar] [CrossRef] [PubMed]
- Larsson, J.; Goumans, M.J.; Sjostrand, L.J.; van Rooijen, M.A.; Ward, D.; Leveen, P.; Xu, X.F.; ten Dijke, P.; Mummery, C.L.; Karlsson, S. Abnormal angiogenesis but intact hematopoietic potential in TGF-beta type I receptor-deficient mice. EMBO J. 2001, 20, 1663–1673. [Google Scholar] [CrossRef]
- Darland, D.C.; Massingham, L.J.; Smith, S.R.; Piek, E.; St-Geniez, M.; D’Amore, P.A. Pericyte production of cell-associated VEGF is differentiation-dependent and is associated with endothelial survival. Dev. Biol. 2003, 264, 275–288. [Google Scholar] [CrossRef]
- Chen, C.W.; Okada, M.; Proto, J.D.; Gao, X.Q.; Sekiya, N.; Beckman, S.A.; Corselli, M.; Crisan, M.; Saparov, A.; Tobita, K.; et al. Human Pericytes for Ischemic Heart Repair. Stem Cells 2013, 31, 305–316. [Google Scholar] [CrossRef]
- Eilken, H.M.; Dieguez-Hurtado, R.; Schmidt, I.; Nakayama, M.; Jeong, H.W.; Arf, H.; Adams, S.; Ferrara, N.; Adams, R.H. Pericytes regulate VEGF-induced endothelial sprouting through VEGFR-1. Nat. Commun. 2017, 8, 1574. [Google Scholar] [CrossRef]
- Koch, S.; Claesson-Welsh, L. Signal Transduction by Vascular Endothelial Growth Factor Receptors. Cold Spring Harb. Perspect. Med. 2012, 2, a006502. [Google Scholar] [CrossRef]
- Tilton, R.G.; Kilo, C.; Williamson, J.R. Pericyte-endothelial relationships in cardiac and skeletal muscle capillaries. Microvasc. Res. 1979, 18, 325–335. [Google Scholar] [CrossRef]
- Kelley, C.; D’Amore, P.; Hechtman, H.B.; Shepro, D. Microvascular pericyte contractility in vitro: Comparison with other cells of the vascular wall. J. Cell Biol. 1987, 104, 483–490. [Google Scholar] [CrossRef] [PubMed]
- Matsugi, T.; Chen, Q.; Anderson, D.R. Suppression of CO2-induced relaxation of bovine retinal pericytes by angiotensin II. Invest. Ophthalmol. Vis. Sci. 1997, 38, 652–657. [Google Scholar] [PubMed]
- Dore-Duffy, P.; Wang, S.; Mehedi, A.; Katyshev, V.; Cleary, K.; Tapper, A.; Reynolds, C.; Ding, Y.; Zhan, P.; Rafols, J.; et al. Pericyte-mediated vasoconstriction underlies TBI-induced hypoperfusion. Neurol. Res. 2011, 33, 176–186. [Google Scholar] [CrossRef] [PubMed]
- Hall, C.N.; Reynell, C.; Gesslein, B.; Hamilton, N.B.; Mishra, A.; Sutherland, B.A.; O’Farrell, F.M.; Buchan, A.M.; Lauritzen, M.; Attwell, D. Capillary pericytes regulate cerebral blood flow in health and disease. Nature 2014, 508, 55–60. [Google Scholar] [CrossRef]
- Mishra, A.; Reynolds, J.P.; Chen, Y.; Gourine, A.V.; Rusakov, D.A.; Attwell, D. Astrocytes mediate neurovascular signaling to capillary pericytes but not to arterioles. Nat. Neurosci. 2016, 19, 1619–1627. [Google Scholar] [CrossRef]
- Hauck, E.F.; Apostel, S.; Hoffmann, J.F.; Heimann, A.; Kempski, O. Capillary flow and diameter changes during reperfusion after global cerebral ischemia studied by intravital video microscopy. J. Cereb. Blood Flow Metab. 2004, 24, 383–391. [Google Scholar] [CrossRef]
- Mapelli, L.; Gagliano, G.; Soda, T.; Laforenza, U.; Moccia, F.; D’Angelo, E.U. Granular Layer Neurons Control Cerebellar Neurovascular Coupling Through an NMDA Receptor/NO-Dependent System. J. Neurosci. 2017, 37, 1340–1351. [Google Scholar] [CrossRef]
- Hill, R.A.; Tong, L.; Yuan, P.; Murikinati, S.; Gupta, S.; Grutzendler, J. Regional Blood Flow in the Normal and Ischemic Brain Is Controlled by Arteriolar Smooth Muscle Cell Contractility and Not by Capillary Pericytes. Neuron 2015, 87, 95–110. [Google Scholar] [CrossRef]
- Attwell, D.; Mishra, A.; Hall, C.N.; O’Farrell, F.M.; Dalkara, T. What is a pericyte? J. Cereb. Blood Flow Met. 2016, 36, 451–455. [Google Scholar] [CrossRef]
- Lange, S.; Trost, A.; Tempfer, H.; Bauer, H.C.; Bauer, H.; Rohde, E.; Reitsamer, H.A.; Franklin, R.J.M.; Aigner, L.; Rivera, F.J. Brain pericyte plasticity as a potential drug target in CNS repair. Drug Discov. Today 2013, 18, 456–463. [Google Scholar] [CrossRef]
- Winkler, E.A.; Sagare, A.P.; Zlokovic, B.V. The Pericyte: A Forgotten Cell Type with Important Implications for Alzheimer’s Disease? Brain Pathol. 2014, 24, 371–386. [Google Scholar] [CrossRef] [PubMed]
- Beltramo, E.; Porta, M. Pericyte loss in diabetic retinopathy: Mechanisms and consequences. Curr. Med. Chem. 2013, 20, 3218–3225. [Google Scholar] [CrossRef] [PubMed]
- Assero, G.; Lupo, G.; Anfuso, C.D.; Ragusa, N.; Alberghina, M. High glucose and advanced glycation end products induce phospholipid hydrolysis and phospholipid enzyme inhibition in bovine retinal pericytes. Biochim. Biophys. Acta 2001, 1533, 128–140. [Google Scholar] [CrossRef]
- Braun, A.; Xu, H.M.; Hu, F.R.; Kocherlakota, P.; Siegel, D.; Chander, P.; Ungvari, Z.; Csiszar, A.; Nedergaard, M.; Ballabh, P. Paucity of Pericytes in germinal matrix vasculature of premature infants. J. Neurosci. 2007, 27, 12012–12024. [Google Scholar] [CrossRef] [PubMed]
- Winkler, E.A.; Sengillo, J.D.; Sullivan, J.S.; Henkel, J.S.; Appel, S.H.; Zlokovic, B.V. Blood-spinal cord barrier breakdown and pericyte reductions in amyotrophic lateral sclerosis. Acta Neuropathol. 2013, 125, 111–120. [Google Scholar] [CrossRef] [PubMed]
- Bell, R.D.; Winkler, E.A.; Sagare, A.P.; Singh, I.; LaRue, B.; Deane, R.; Zlokovic, B.V. Pericytes control key neurovascular functions and neuronal phenotype in the adult brain and during brain aging. Neuron 2010, 68, 409–427. [Google Scholar] [CrossRef] [PubMed]
- Salmeri, M.; Motta, C.; Anfuso, C.D.; Amodeo, A.; Scalia, M.; Toscano, M.A.; Alberghina, M.; Lupo, G. VEGF receptor-1 involvement in pericyte loss induced by Escherichia coli in an in vitro model of blood brain barrier. Cell Microbiol. 2013, 15, 1367–1384. [Google Scholar] [CrossRef]
- Anfuso, C.D.; Motta, C.; Giurdanella, G.; Arena, V.; Alberghina, M.; Lupo, G. Endothelial PKC alpha-MAPK/ERK-phospholipase A(2) pathway activation as a response of glioma in a triple culture model. A new role for pericytes? Biochimie 2014, 99, 77–87. [Google Scholar] [CrossRef]
- Benjamin, L.E.; Golijanin, D.; Itin, A.; Pode, D.; Keshet, E. Selective ablation of immature blood vessels in established human tumors follows vascular endothelial growth factor withdrawal. J. Clin. Invest. 1999, 103, 159–165. [Google Scholar] [CrossRef]
- Daneman, R.; Zhou, L.; Kebede, A.A.; Barres, B.A. Pericytes are required for blood-brain barrier integrity during embryogenesis. Nature 2010, 468, 562–566. [Google Scholar] [CrossRef]
- Lindblom, P.; Gerhardt, H.; Liebner, S.; Abramsson, A.; Enge, M.; Hellstrom, M.; Backstrom, G.; Fredriksson, S.; Landegren, U.; Nystrom, H.C.; et al. Endothelial PDGF-B retention is required for proper investment of pericytes in the microvessel wall. Gene Dev. 2003, 17, 1835–1840. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Kim, J.H.; Yu, Y.S.; Kim, D.H.; Kim, K.W. Recruitment of Pericytes and Astrocytes Is Closely Related to the Formation of Tight junction in Developing Retinal Vessels. J. Neurosci. Res. 2009, 87, 653–659. [Google Scholar] [CrossRef] [PubMed]
- Paquet-Fifield, S.; Schluter, H.; Li, A.; Aitken, T.; Gangatirkar, P.; Blashki, D.; Koelmeyer, R.; Pouliot, N.; Palatsides, M.; Ellis, S.; et al. A role for pericytes as microenvironmental regulators of human skin tissue regeneration. J. Clin. Invest. 2009, 119, 2795–2806. [Google Scholar] [CrossRef] [PubMed]
- Farrington-Rock, C.; Crofts, N.J.; Doherty, M.J.; Ashton, B.A.; Griffin-Jones, C.; Canfield, A.E. Chondrogenic and adipogenic potential of microvascular pericytes. Circulation 2004, 110, 2226–2232. [Google Scholar] [CrossRef] [PubMed]
- Richardson, R.L.; Hausman, G.J.; Campion, D.R. Response of pericytes to thermal lesion in the inguinal fat pad of 10-day-old rats. Acta Anat. (Basel) 1982, 114, 41–57. [Google Scholar] [CrossRef] [PubMed]
- Wong, S.P.; Rowley, J.E.; Redpath, A.N.; Tilman, J.D.; Fellous, T.G.; Johnson, J.R. Pericytes, mesenchymal stem cells and their contributions to tissue repair. Pharmacol. Therapeut. 2015, 151, 107–120. [Google Scholar] [CrossRef] [PubMed]
- Crisan, M.; Yap, S.; Casteilla, L.; Chen, C.W.; Corselli, M.; Park, T.S.; Andriolo, G.; Sun, B.; Zheng, B.; Zhang, L.; et al. A perivascular origin for mesenchymal stem cells in multiple human organs. Cell Stem Cell 2008, 3, 301–313. [Google Scholar] [CrossRef]
- Meirelles, L.D.; Caplan, A.I.; Nardi, N.B. In search of the in vivo identity of mesenchymal stem cells. Stem Cells 2008, 26, 2287–2299. [Google Scholar] [CrossRef]
- Feng, J.F.; Mantesso, A.; Sharpe, P.T. Perivascular cells as mesenchymal stem cells. Expert Opin. Biol. Therapy 2010, 10, 1441–1451. [Google Scholar] [CrossRef]
- Thomas, W.E. Brain macrophages: On the role of pericytes and perivascular cells. Brain Res. Rev. 1999, 31, 42–57. [Google Scholar] [CrossRef]
- Nicosia, R.F.; Villaschi, S. Rat Aortic Smooth-Muscle Cells Become Pericytes during Angiogenesis in-Vitro. Lab. Invest. 1995, 73, 658–666. [Google Scholar] [PubMed]
- Minutti, C.M.; Modak, R.V.; Macdonald, F.; Li, F.; Smyth, D.J.; Dorward, D.A.; Blair, N.; Husovsky, C.; Muir, A.; Giampazolias, E.; et al. A Macrophage-Pericyte Axis Directs Tissue Restoration via Amphiregulin-Induced Transforming Growth Factor Beta Activation. Immunity 2019, 50, 645–654. [Google Scholar] [CrossRef] [PubMed]
- Birbrair, A.; Zhang, T.; Wang, Z.M.; Messi, M.L.; Enikolopov, G.N.; Mintz, A.; Delbono, O. Skeletal muscle pericyte subtypes differ in their differentiation potential. Stem Cell Res. 2013, 10, 67–84. [Google Scholar] [CrossRef] [PubMed]
- Erdener, S.E.; Dalkara, T. Small Vessels Are a Big Problem in Neurodegeneration and Neuroprotection. Front. Neurol. 2019, 10, 889. [Google Scholar] [CrossRef]
- Li, Q.; Khatibi, N.; Zhang, J.H. Vascular neural network: The importance of vein drainage in stroke. Transl. Stroke Res. 2014, 5, 163–166. [Google Scholar] [CrossRef][Green Version]
- Linfante, I.; Cipolla, M.J. Improving Reperfusion Therapies in the Era of Mechanical Thrombectomy. Transl. Stroke Res. 2016, 7, 294–302. [Google Scholar] [CrossRef]
- O’Farrell, F.M.; Mastitskaya, S.; Hammond-Haley, M.; Freitas, F.; Wah, W.R.; Attwell, D. Capillary pericytes mediate coronary no-reflow after myocardial ischaemia. Elife 2017, 6, e29280. [Google Scholar] [CrossRef]
- Li, Q.; Puro, D.G. Adenosine activates ATP-sensitive K+ currents in pericytes of rat retinal microvessels: Role of A(1) and A(2a) receptors. Brain Res. 2001, 907, 93–99. [Google Scholar] [CrossRef]
- Arimura, K.; Ago, T.; Kamouchi, M.; Nakamura, K.; Ishitsuka, K.; Kuroda, J.; Sugimori, H.; Ooboshi, H.; Sasaki, T.; Kitazono, T. PDGF receptor beta signaling in pericytes following ischemic brain injury. Curr. Neurovasc. Res. 2012, 9, 1–9. [Google Scholar] [CrossRef]
- Jung, K.H.; Chu, K.; Lee, S.T.; Bahn, J.J.; Jeon, D.; Kim, J.H.; Kim, S.; Won, C.H.; Kim, M.; Lee, S.K.; et al. Multipotent PDGFRbeta-expressing cells in the circulation of stroke patients. Neurobiol. Dis. 2011, 41, 489–497. [Google Scholar] [CrossRef]
- Nakamura, K.; Arimura, K.; Nishimura, A.; Tachibana, M.; Yoshikawa, Y.; Makihara, N.; Wakisaka, Y.; Kuroda, J.; Kamouchi, M.; Ooboshi, H.; et al. Possible involvement of basic FGF in the upregulation of PDGFRbeta in pericytes after ischemic stroke. Brain Res. 2016, 1630, 98–108. [Google Scholar] [CrossRef] [PubMed]
- Shimizu, F.; Sano, Y.; Saito, K.; Abe, M.A.; Maeda, T.; Haruki, H.; Kanda, T. Pericyte-derived glial cell line-derived neurotrophic factor increase the expression of claudin-5 in the blood-brain barrier and the blood-nerve barrier. Neurochem. Res. 2012, 37, 401–409. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.L.; Hui, Y.N.; Guo, B.; Ma, J.X. Strengthening tight junctions of retinal microvascular endothelial cells by pericytes under normoxia and hypoxia involving angiopoietin-1 signal way. Eye 2007, 21, 1501–1510. [Google Scholar] [CrossRef] [PubMed]
- Shih, S.C.; Ju, M.H.; Liu, N.; Mo, J.R.; Ney, J.J.; Smith, L.E.H. Transforming growth factor beta 1 induction of vascular endothelial growth factor receptor 1: Mechanism of pericyte-induced vascular survival in vivo. Proc. Natl. Acad. Sci. USA 2003, 100, 15859–15864. [Google Scholar] [CrossRef]
- Zechariah, A.; ElAli, A.; Doeppner, T.R.; Jin, F.; Hasan, M.R.; Helfrich, I.; Mies, G.; Hermann, D.M. Vascular endothelial growth factor promotes pericyte coverage of brain capillaries, improves cerebral blood flow during subsequent focal cerebral ischemia, and preserves the metabolic penumbra. Stroke 2013, 44, 1690–1697. [Google Scholar] [CrossRef]
- Yemisci, M.; Gursoy-Ozdemir, Y.; Vural, A.; Can, A.; Topalkara, K.; Dalkara, T. Pericyte contraction induced by oxidative-nitrative stress impairs capillary reflow despite successful opening of an occluded cerebral artery. Nat. Med. 2009, 15, 1031–1037. [Google Scholar] [CrossRef]
- Shojaee, N.; Patton, W.F.; Hechtman, H.B.; Shepro, D. Myosin translocation in retinal pericytes during free-radical induced apoptosis. J. Cell. Biochem. 1999, 75, 118–129. [Google Scholar] [CrossRef]
- Miller, A.A.; Drummond, G.R.; Schmidt, H.H.; Sobey, C.G. NADPH oxidase activity and function are profoundly greater in cerebral versus systemic arteries. Circ. Res. 2005, 97, 1055–1062. [Google Scholar] [CrossRef]
- Deguchi, K.; Liu, N.; Liu, W.T.; Omote, Y.; Kono, S.; Yunoki, T.; Deguchi, S.; Yamashita, T.; Ikeda, Y.; Abe, K. Pericyte Protection by Edaravone After Tissue Plasminogen Activator Treatment in Rat Cerebral Ischemia. J. Neurosci. Res. 2014, 92, 1509–1519. [Google Scholar] [CrossRef]
- Lukic-Panin, V.; Deguchi, K.; Yamashita, T.; Shang, J.; Zhang, X.; Tian, F.; Liu, N.; Kawai, H.; Matsuura, T.; Abe, K. Free radical scavenger edaravone administration protects against tissue plasminogen activator induced oxidative stress and blood brain barrier damage. Curr. Neurovasc. Res. 2010, 7, 319–329. [Google Scholar] [CrossRef]
- Ozen, I.; Deierborg, T.; Miharada, K.; Padel, T.; Englund, E.; Genove, G.; Paul, G. Brain pericytes acquire a microglial phenotype after stroke. Acta Neuropathol. 2014, 128, 381–396. [Google Scholar] [CrossRef] [PubMed]
- Underly, R.G.; Levy, M.; Hartmann, D.A.; Grant, R.I.; Watson, A.N.; Shih, A.Y. Pericytes as Inducers of Rapid, Matrix Metalloproteinase-9-Dependent Capillary Damage during Ischemia. J. Neurosci. 2017, 37, 129–140. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Agalliu, D.; Yu, C.H.; Fisher, M. The Role of Pericytes in Blood-Brain Barrier Function and Stroke. Curr. Pharm. Design 2012, 18, 3653–3662. [Google Scholar] [CrossRef] [PubMed]
- Shaikh, H.; Lechpammer, M.; Jensen, F.E.; Warfield, S.K.; Hansen, A.H.; Kosaras, B.; Shevell, M.; Wintermark, P. Increased Brain Perfusion Persists over the First Month of Life in Term Asphyxiated Newborns Treated with Hypothermia: Does it Reflect Activated Angiogenesis? Transl. Stroke Res. 2015, 6, 224–233. [Google Scholar] [CrossRef]
- Ishitsuka, K.; Ago, T.; Arimura, K.; Nakamura, K.; Tokami, H.; Makihara, N.; Kuroda, J.; Kamouchi, M.; Kitazono, T. Neurotrophin production in brain pericytes during hypoxia: A role of pericytes for neuroprotection. Microvasc. Res. 2012, 83, 352–359. [Google Scholar] [CrossRef]
- Lapchak, P.A. Critical early thrombolytic and endovascular reperfusion therapy for acute ischemic stroke victims: A call for adjunct neuroprotection. Transl. Stroke Res. 2015, 6, 345–354. [Google Scholar] [CrossRef]
- Yamagishi, S.; Yonekura, H.; Yamamoto, Y.; Fujimori, H.; Sakurai, S.; Tanaka, N.; Yamamoto, H. Vascular endothelial growth factor acts as a pericyte mitogen under hypoxic conditions. Lab. Invest. 1999, 79, 501–509. [Google Scholar]
- Takagi, H.; King, G.L.; Aiello, L.P. Identification and characterization of vascular endothelial growth factor receptor (Flt) in bovine retinal pericytes. Diabetes 1996, 45, 1016–1023. [Google Scholar] [CrossRef]
- Park, Y.S.; Kim, N.H.; Jo, I. Hypoxia and vascular endothelial growth factor acutely up-regulate angiopoietin-1 and Tie2 mRNA in bovine retinal pericytes. Microvasc. Res. 2003, 65, 125–131. [Google Scholar] [CrossRef]
- Berger, M.; Bergers, G.; Arnold, B.; Hammerling, G.J.; Ganss, R. Regulator of G-protein signaling-5 induction in pericytes coincides with active vessel remodeling during neovascularization. Blood 2005, 105, 1094–1101. [Google Scholar] [CrossRef]
- Seifert, H.A.; Pennypacker, K.R. Molecular and cellular immune responses to ischemic brain injury. Transl. Stroke Res. 2014, 5, 543–553. [Google Scholar] [CrossRef] [PubMed]
- Lyck, R.; Enzmann, G. The physiological roles of ICAM-1 and ICAM-2 in neutrophil migration into tissues. Curr. Opin. Hematol. 2015, 22, 53–59. [Google Scholar] [CrossRef] [PubMed]
- Youn, S.W.; Jung, K.H.; Chu, K.; Lee, J.Y.; Lee, S.T.; Bahn, J.J.; Park, D.K.; Yu, J.S.; Kim, S.Y.; Kim, M.; et al. Feasibility and Safety of Intra-arterial Pericyte Progenitor Cell Delivery Following Mannitol-Induced Transient Blood-Brain Barrier Opening in a Canine Model. Cell Transplant. 2015, 24, 1469–1479. [Google Scholar] [CrossRef] [PubMed]
- Sakuma, R.; Kawahara, M.; Nakano-Doi, A.; Takahashi, A.; Tanaka, Y.; Narita, A.; Kuwahara-Otani, S.; Hayakawa, T.; Yagi, H.; Matsuyama, T.; et al. Brain pericytes serve as microglia-generating multipotent vascular stem cells following ischemic stroke. J. Neuroinflammation 2016, 13, 57. [Google Scholar] [CrossRef]
- Karow, M.; Schichor, C.; Beckervordersandforth, R.; Berninger, B. Lineage-reprogramming of Pericyte-derived Cells of the Adult Human Brain into Induced Neurons. Jove-J. Vis. Exp. 2014, 87, e51433. [Google Scholar] [CrossRef]
- Nakagomi, T.; Nakano-Doi, A.; Kawamura, M.; Matsuyama, T. Do Vascular Pericytes Contribute to Neurovasculogenesis in the Central Nervous System as Multipotent Vascular Stem Cells? Stem Cells Dev. 2015, 24, 1730–1739. [Google Scholar] [CrossRef]
- Cheng, J.P.; Korte, N.; Nortley, R.; Sethi, H.; Tang, Y.M.; Attwell, D. Targeting pericytes for therapeutic approaches to neurological disorders. Acta Neuropathol. 2018, 136, 507–523. [Google Scholar] [CrossRef]
- Stebbins, M.J.; Gastfriend, B.D.; Canfield, S.G.; Lee, M.S.; Richards, D.; Faubion, M.G.; Li, W.J.; Daneman, R.; Palecek, S.P.; Shusta, E.V. Human pluripotent stem cell-derived brain pericyte-like cells induce blood-brain barrier properties. Sci. Adv. 2019, 5, eaau7375. [Google Scholar] [CrossRef]
- Faal, T.; Phan, D.T.T.; Davtyan, H.; Scarfone, V.M.; Varady, E.; Blurton-Jones, M.; Hughes, C.C.W.; Inlay, M.A. Induction of Mesoderm and Neural Crest-Derived Pericytes from Human Pluripotent Stem Cells to Study Blood-Brain Barrier Interactions. Stem Cell Rep. 2019, 12, 451–460. [Google Scholar] [CrossRef]
- Onishi, M.; Ichikawa, T.; Kurozumi, K.; Date, I. Angiogenesis and invasion in glioma. Brain Tumor Pathol. 2011, 28, 13–24. [Google Scholar] [CrossRef]
- Yang, X.F.-E. Yuan.; Qian-Xue, Chen.; Bao-Hui, Liu., Molecular mechanisms involved in angiogenesis and potential target of antiangiogenesis in human glioblastomas. Glioma 2018, 1, 35–42. [Google Scholar]
- Barlow, K.D.; Sanders, A.M.; Soker, S.; Ergun, S.; Metheny-Barlow, L.J. Pericytes on the tumor vasculature: Jekyll or hyde? Cancer Microenviron 2013, 6, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Filatova, A.; Acker, T.; Garvalov, B.K. The cancer stem cell niche(s): The crosstalk between glioma stem cells and their microenvironment. BBA-Gen. Subj. 2013, 1830, 2496–2508. [Google Scholar] [CrossRef] [PubMed]
- Hambardzumyan, D.; Bergers, G. Glioblastoma: Defining Tumor Niches. Trends Cancer 2015, 1, 252–265. [Google Scholar] [CrossRef]
- Cheng, L.; Huang, Z.; Zhou, W.C.; Wu, Q.L.; Donnola, S.; Liu, J.K.; Fang, X.G.; Sloan, A.E.; Mao, Y.B.; Lathia, J.D.; et al. Glioblastoma Stem Cells Generate Vascular Pericytes to Support Vessel Function and Tumor Growth. Cell 2013, 153, 139–152. [Google Scholar] [CrossRef]
- Valdor, R.; Garcia-Bernal, D.; Bueno, C.; Rodenas, M.; Moraleda, J.M.; Macian, F.; Martinez, S. Glioblastoma progression is assisted by induction of immunosuppressive function of pericytes through interaction with tumor cells. Oncotarget 2017, 8, 68614–68626. [Google Scholar] [CrossRef]
- Stout, A.P.; Murray, M.R. Hemangiopericytoma: A Vascular Tumor Featuring Zimmermann’s Pericytes. Ann. Surg 1942, 116, 26–33. [Google Scholar] [CrossRef]
- Park, M.S.; Ravi, V.; Araujo, D.M. Inhibiting the VEGF-VEGFR pathway in angiosarcoma, epithelioid hemangioendothelioma, and hemangiopericytoma/solitary fibrous tumor. Curr. Opin. Oncol. 2010, 22, 351–355. [Google Scholar] [CrossRef]
- Pries, A.R.; Hopfner, M.; le Noble, F.; Dewhirst, M.W.; Secomb, T.W. The shunt problem: Control of functional shunting in normal and tumour vasculature. Nat. Rev. Cancer 2010, 10, 587–593. [Google Scholar] [CrossRef]
- Watkins, S.; Robel, S.; Kimbrough, I.F.; Robert, S.M.; Ellis-Davies, G.; Sontheimer, H. Disruption of astrocyte-vascular coupling and the blood-brain barrier by invading glioma cells. Nat. Commun. 2014, 5, 4196. [Google Scholar] [CrossRef]
- Zhou, W.; Chen, C.; Shi, Y.; Wu, Q.; Gimple, R.C.; Fang, X.; Huang, Z.; Zhai, K.; Ke, S.Q.; Ping, Y.F.; et al. Targeting Glioma Stem Cell-Derived Pericytes Disrupts the Blood-Tumor Barrier and Improves Chemotherapeutic Efficacy. Cell Stem Cell 2017, 21, 591–603. [Google Scholar] [CrossRef] [PubMed]
- Nisancioglu, M.H.; Betsholtz, C.; Genove, G. The Absence of Pericytes Does Not Increase the Sensitivity of Tumor Vasculature to Vascular Endothelial Growth Factor-A Blockade. Cancer Res. 2010, 70, 5109–5115. [Google Scholar] [CrossRef] [PubMed]
- Winkler, F.; Kozin, S.V.; Tong, R.T.; Chae, S.S.; Booth, M.F.; Garkavtsev, I.; Xu, L.; Hicklin, D.J.; Fukumura, D.; di Tomaso, E.; et al. Kinetics of vascular normalization by VEGFR2 blockade governs brain tumor response to radiation: Role of oxygenation, angiopoietin-1, and matrix metalloproteinases. Cancer Cell 2004, 6, 553–563. [Google Scholar] [PubMed]
- Hamzah, J.; Jugold, M.; Kiessling, F.; Rigby, P.; Manzur, M.; Marti, H.H.; Rabie, T.; Kaden, S.; Grone, H.J.; Hammerling, G.J.; et al. Vascular normalization in Rgs5-deficient tumours promotes immune destruction. Nature 2008, 453, 410–414. [Google Scholar] [CrossRef] [PubMed]
- Meng, M.B.; Zaorsky, N.G.; Deng, L.; Wang, H.H.; Chao, J.; Zhao, L.J.; Yuan, Z.Y.; Ping, W. Pericytes: a double-edged sword in cancer therapy. Future Oncol. 2015, 11, 169–179. [Google Scholar] [CrossRef]
- Caporarello, N.; Lupo, G.; Olivieri, M.; Cristaldi, M.; Cambria, M.T.; Salmeri, M.; Anfuso, C.D. Classical VEGF, Notch and Ang signalling in cancer angiogenesis, alternative approaches and future directions (Review). Mol. Med. Rep. 2017, 16, 4393–4402. [Google Scholar] [CrossRef]
- Ruan, J.; Luo, M.; Wang, C.; Fan, L.; Yang, S.N.; Cardenas, M.; Geng, H.; Leonard, J.P.; Melnick, A.; Cerchietti, L.; et al. Imatinib disrupts lymphoma angiogenesis by targeting vascular pericytes. Blood 2013, 121, 5192–5202. [Google Scholar] [CrossRef]
- Yang, Y.; Andersson, P.; Hosaka, K.; Zhang, Y.; Cao, R.; Iwamoto, H.; Yang, X.; Nakamura, M.; Wang, J.; Zhuang, R.; et al. The PDGF-BB-SOX7 axis-modulated IL-33 in pericytes and stromal cells promotes metastasis through tumour-associated macrophages. Nat. Commun. 2016, 7, 11385. [Google Scholar] [CrossRef]
- Guan, Y.Y.; Luan, X.; Xu, J.R.; Liu, Y.R.; Lu, Q.; Wang, C.; Liu, H.J.; Gao, Y.G.; Chen, H.Z.; Fang, C. Selective eradication of tumor vascular pericytes by peptide-conjugated nanoparticles for antiangiogenic therapy of melanoma lung metastasis. Biomaterials 2014, 35, 3060–3070. [Google Scholar] [CrossRef]
- Aly, A.E.; Harmon, B.; Padegimas, L.; Sesenoglu-Laird, O.; Cooper, M.J.; Yurek, D.M.; Waszczak, B.L. Intranasal delivery of hGDNF plasmid DNA nanoparticles results in long-term and widespread transfection of perivascular cells in rat brain. Nanomedicine 2019, 16, 20–33. [Google Scholar] [CrossRef]
- Trost, A.; Lange, S.; Schroedl, F.; Bruckner, D.; Motloch, K.A.; Bogner, B.; Kaser-Eichberger, A.; Strohmaier, C.; Runge, C.; Aigner, L.; et al. Brain and Retinal Pericytes: Origin, Function and Role. Front. Cell Neurosci. 2016, 10, 20. [Google Scholar] [CrossRef] [PubMed]
- Giurdanella, G.; Anfuso, C.D.; Olivieri, M.; Lupo, G.; Caporarello, N.; Eandi, C.M.; Drago, F.; Bucolo, C.; Salomone, S. Aflibercept, bevacizumab and ranibizumab prevent glucose-induced damage in human retinal pericytes in vitro, through a PLA(2)/COX-2/VEGF-A pathway. Biochem. Pharmacol. 2015, 96, 278–287. [Google Scholar] [CrossRef] [PubMed]
- Scott, A.; Fruttiger, M. Oxygen-induced retinopathy: A model for vascular pathology in the retina. Eye 2010, 24, 416–421. [Google Scholar] [CrossRef] [PubMed]
- Ouyang, H.; Goldberg, J.L.; Chen, S.Y.; Li, W.; Xu, G.T.; Li, W.; Zhang, K.; Nussenblatt, R.B.; Liu, Y.Z.; Xie, T.; et al. Ocular Stem Cell Research from Basic Science to Clinical Application: A Report from Zhongshan Ophthalmic Center Ocular Stem Cell Symposium. Int. J. Mol. Sci. 2016, 17, 415. [Google Scholar] [CrossRef]
- Singh, R.; Cuzzani, O.; Binette, F.; Sternberg, H.; West, M.D.; Nasonkin, I.O. Pluripotent Stem Cells for Retinal Tissue Engineering: Current Status and Future Prospects. Stem Cell Rev. Rep. 2018, 14, 463–483. [Google Scholar] [CrossRef]
- Caballero, S.; Sengupta, N.; Afzal, A.; Chang, K.H.; Calzi, S.L.; Guberski, D.L.; Kern, T.S.; Grant, M.B. Ischemic vascular damage can be repaired by healthy, but not diabetic, endothelial progenitor cells. Diabetes 2007, 56, 960–967. [Google Scholar] [CrossRef]
- Canfield, A.E.; Sutton, A.B.; Hoyland, J.A.; Schor, A.M. Association of thrombospondin-1 with osteogenic differentiation of retinal pericytes in vitro. J. Cell Sci. 1996, 109, 343–353. [Google Scholar]
- Yoshida, Y.; Kabara, M.; Kano, K.; Horiuchi, K.; Hayasaka, T.; Tomita, Y.; Takehara, N.; Minoshima, A.; Aonuma, T.; Maruyama, K.; et al. Capillary-Resident EphA7(+) Pericytes Are Multipotent Cells with Anti-Ischemic Effects Through Capillary Formation. Stem Cells Transl. Med. 2019. [Google Scholar] [CrossRef]
- Vidhya, S.; Ramya, R.; Coral, K.; Sulochana, K.N.; Bharathidevi, S.R. Free amino acids hydroxyproline, lysine, and glycine promote differentiation of retinal pericytes to adipocytes: A protective role against proliferative diabetic retinopathy. Exp. Eye Res. 2018, 173, 179–187. [Google Scholar] [CrossRef]
- Teichert, M.; Milde, L.; Annegret, H.W.; Stanicek, L.; Gengenbacher, N.; Savant, S.; Ruckdeschel, T.; Hasanov, Z.; Srivastava, K.; Hu, J.H.; et al. Pericyte-expressed Tie2 controls angiogenesis and vessel maturation. Nat. Commun. 2017, 8, 16106. [Google Scholar] [CrossRef]
- Rubsam, A.; Parikh, S.; Fort, P.E. Role of Inflammation in Diabetic Retinopathy. Int J. Mol Sci 2018, 19, 942. [Google Scholar] [CrossRef] [PubMed]
- Rajashekhar, G. Mesenchymal stem cells: New players in retinopathy therapy. Front. Endocrinol. (Lausanne) 2014, 5, 59. [Google Scholar] [CrossRef] [PubMed]
- Mendel, T.A.; Clabough, E.B.; Kao, D.S.; Demidova-Rice, T.N.; Durham, J.T.; Zotter, B.C.; Seaman, S.A.; Cronk, S.M.; Rakoczy, E.P.; Katz, A.J.; et al. Pericytes derived from adipose-derived stem cells protect against retinal vasculopathy. PLoS ONE 2013, 8, e65691. [Google Scholar] [CrossRef]
- Wang, S.; Gusenoff, J.A.; Rubin, J.P.; Kokai, L. Molecular Mechanisms of Adipose Tissue Survival during Severe Hypoxia: Implications for Autologous Fat Graft Performance. Plast. Reconstr. Surg. Glob. Open 2019, 7, e2275. [Google Scholar] [CrossRef] [PubMed]
- Dong, Y.X.; Rodrigues, M.; Kwon, S.H.; Li, X.L.; Sigen, A.; Brett, E.A.; Elvassore, N.; Wang, W.X.; Gurtner, G.C. Acceleration of Diabetic Wound Regeneration using an In Situ-Formed Stem-Cell-Based Skin Substitute. Adv. Healthc. Mater. 2018, 7. [Google Scholar] [CrossRef]
- Fiori, A.; Terlizzi, V.; Kremer, H.; Gebauer, J.; Hammes, H.P.; Harmsen, M.C.; Bieback, K. Mesenchymal stromal/stem cells as potential therapy in diabetic retinopathy. Immunobiology 2018, 223, 729–743. [Google Scholar] [CrossRef]
- Rennert, R.C.; Sorkin, M.; Januszyk, M.; Duscher, D.; Kosaraju, R.; Chung, M.T.; Lennon, J.; Radiya-Dixit, A.; Raghvendra, S.; Maan, Z.N.; et al. Diabetes impairs the angiogenic potential of adipose-derived stem cells by selectively depleting cellular subpopulations. Stem Cell Res. Ther. 2014, 5, 79. [Google Scholar] [CrossRef]
- Ozerdem, U. Targeting pericytes diminishes neovascularization in orthotopic uveal melanoma in nerve/glial antigen 2 proteoglycan knockout mouse. Ophthal. Res. 2006, 38, 251–254. [Google Scholar] [CrossRef]
- Clarijs, R.; van Dijk, M.; Ruiter, D.J.; de Waal, R.M.W. Functional and morphologic analysis of the fluid-conducting meshwork in xenografted cutaneous and primary uveal melanoma. Invest. Ophth. Vis. Sci. 2005, 46, 3013–3020. [Google Scholar] [CrossRef]
- Van der Wijk, A.E.; Vogels, I.M.C.; van Veen, H.A.; van Noorden, C.J.F.; Schlingemann, R.O.; Klaassen, I. Spatial and temporal recruitment of the neurovascular unit during development of the mouse blood-retinal barrier. Tissue Cell 2018, 52, 42–50. [Google Scholar] [CrossRef]
- Correa, D.; Somoza, R.A.; Lin, P.; Schiemann, W.P.; Caplan, A.I. Mesenchymal stem cells regulate melanoma cancer cells extravasation to bone and liver at their perivascular niche. Int. J. Cancer 2016, 138, 417–427. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.F.; Lei, X.P.; Shi, C.Z.; Huang, M.H.; Li, X.B.; Wu, B.J.; Li, Z.Q.; Han, W.L.; Du, B.; Hu, J.Y.; et al. Pericyte-targeting prodrug overcomes tumor resistance to vascular disrupting agents. J. Clin. Invest. 2017, 127, 3689–3701. [Google Scholar] [CrossRef] [PubMed]
- Jo, N.; Mailhos, C.; Ju, M.; Cheung, E.; Bradley, J.; Nishijima, K.; Robinson, G.S.; Adamis, A.P.; Shima, D.T. Inhibition of platelet-derived growth factor B signaling enhances the efficacy of anti-vascular endothelial growth factor therapy in multiple models of ocular neovascularization. Am. J. Pathol. 2006, 168, 2036–2053. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Caporarello, N.; D’Angeli, F.; Cambria, M.T.; Candido, S.; Giallongo, C.; Salmeri, M.; Lombardo, C.; Longo, A.; Giurdanella, G.; Anfuso, C.D.; et al. Pericytes in Microvessels: From “Mural” Function to Brain and Retina Regeneration. Int. J. Mol. Sci. 2019, 20, 6351. https://doi.org/10.3390/ijms20246351
Caporarello N, D’Angeli F, Cambria MT, Candido S, Giallongo C, Salmeri M, Lombardo C, Longo A, Giurdanella G, Anfuso CD, et al. Pericytes in Microvessels: From “Mural” Function to Brain and Retina Regeneration. International Journal of Molecular Sciences. 2019; 20(24):6351. https://doi.org/10.3390/ijms20246351
Chicago/Turabian StyleCaporarello, Nunzia, Floriana D’Angeli, Maria Teresa Cambria, Saverio Candido, Cesarina Giallongo, Mario Salmeri, Cinzia Lombardo, Anna Longo, Giovanni Giurdanella, Carmelina Daniela Anfuso, and et al. 2019. "Pericytes in Microvessels: From “Mural” Function to Brain and Retina Regeneration" International Journal of Molecular Sciences 20, no. 24: 6351. https://doi.org/10.3390/ijms20246351
APA StyleCaporarello, N., D’Angeli, F., Cambria, M. T., Candido, S., Giallongo, C., Salmeri, M., Lombardo, C., Longo, A., Giurdanella, G., Anfuso, C. D., & Lupo, G. (2019). Pericytes in Microvessels: From “Mural” Function to Brain and Retina Regeneration. International Journal of Molecular Sciences, 20(24), 6351. https://doi.org/10.3390/ijms20246351







