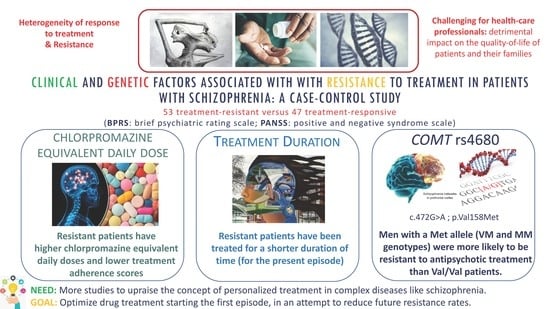
Clinical and Genetic Factors Associated with Resistance to Treatment in Patients with Schizophrenia: A Case-Control Study
Abstract
1. Introduction
2. Results
2.1. Demographic and Clinical Data of Patients
2.2. Genotype and Allele Distribution
2.3. Variable Associated with Resistance to Treatment
2.4. Gender Differences
2.5. Multivariable Analyses
2.6. Gene-Gene Interaction
3. Methods
3.1. Study Design and Patients
3.2. Compliance with Ethical Standards
3.3. Clinical and Demographic Information
3.4. Response and Resistance to Treatment
3.5. Assessment of Adherence to Treatment
3.6. DNA Sampling and Genotyping
3.7. Data and Statistical Analysis
4. Discussion
Limitations and Strengths
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Leucht, S.; Davis, J.M.; Engel, R.R.; Kissling, W.; Kane, J.M. Definitions of response and remission in schizophrenia: Recommendations for their use and their presentation. Acta Psychiatr. Scand. Suppl. 2009, 7–14. [Google Scholar] [CrossRef] [PubMed]
- Leucht, S.; Kane, J.M.; Etschel, E.; Kissling, W.; Hamann, J.; Engel, R.R. Linking the panss, bprs, and cgi: Clinical implications. Neuropsychopharmacology 2006, 31, 2318–2325. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, J.L.; Altar, C.A.; Taylor, D.L.; Degtiar, I.; Hornberger, J.C. The social and economic burden of treatment-resistant schizophrenia: A systematic literature review. Int. Clin. Psychopharmacol. 2014, 29, 63–76. [Google Scholar] [CrossRef] [PubMed]
- Nucifora, F.C., Jr.; Woznica, E.; Lee, B.J.; Cascella, N.; Sawa, A. Treatment resistant schizophrenia: Clinical, biological, and therapeutic perspectives. Neurobiol. Dis. 2018. [Google Scholar] [CrossRef] [PubMed]
- Terzic, T.; Kastelic, M.; Dolzan, V.; Plesnicar, B.K. Influence of 5-ht1a and 5-httlpr genetic variants on the schizophrenia symptoms and occurrence of treatment-resistant schizophrenia. Neuropsychiatr. Dis. Treat. 2015, 11, 453–459. [Google Scholar]
- Terzic, T.; Kastelic, M.; Dolzan, V.; Plesnicar, B.K. Genetic polymorphisms in dopaminergic system and treatment-resistant schizophrenia. Psychiatr. Danub. 2016, 28, 127–131. [Google Scholar] [PubMed]
- Lally, J.; Gaughran, F.; Timms, P.; Curran, S.R. Treatment-resistant schizophrenia: Current insights on the pharmacogenomics of antipsychotics. Pharmgenom. Pers. Med. 2016, 9, 117–129. [Google Scholar] [CrossRef]
- Iasevoli, F.; Giordano, S.; Balletta, R.; Latte, G.; Formato, M.V.; Prinzivalli, E.; De Berardis, D.; Tomasetti, C.; de Bartolomeis, A. Treatment resistant schizophrenia is associated with the worst community functioning among severely-ill highly-disabling psychiatric conditions and is the most relevant predictor of poorer achievements in functional milestones. Prog. Neuropsychopharmacol. Biol. Psychiatry 2016, 65, 34–48. [Google Scholar] [CrossRef]
- Galderisi, S.; Rucci, P.; Kirkpatrick, B.; Mucci, A.; Gibertoni, D.; Rocca, P.; Rossi, A.; Bertolino, A.; Strauss, G.P.; Aguglia, E.; et al. Interplay among psychopathologic variables, personal resources, context-related factors, and real-life functioning in individuals with schizophrenia: A network analysis. JAMA Psychiatry 2018, 75, 396–404. [Google Scholar] [CrossRef]
- Howes, O.D.; McCutcheon, R.; Agid, O.; de Bartolomeis, A.; van Beveren, N.J.; Birnbaum, M.L.; Bloomfield, M.A.; Bressan, R.A.; Buchanan, R.W.; Carpenter, W.T.; et al. Treatment-resistant schizophrenia: Treatment response and resistance in psychosis (trrip) working group consensus guidelines on diagnosis and terminology. Am. J. Psychiatry 2017, 174, 216–229. [Google Scholar] [CrossRef]
- Perkins, D.O.; Gu, H.; Weiden, P.J.; McEvoy, J.P.; Hamer, R.M.; Lieberman, J.A.; Comparison of Atypicals in First Episode study, g. Predictors of treatment discontinuation and medication nonadherence in patients recovering from a first episode of schizophrenia, schizophreniform disorder, or schizoaffective disorder: A randomized, double-blind, flexible-dose, multicenter study. J. Clin. Psychiatry 2008, 69, 106–113. [Google Scholar] [CrossRef] [PubMed]
- Wimberley, T.; Stovring, H.; Sorensen, H.J.; Horsdal, H.T.; MacCabe, J.H.; Gasse, C. Predictors of treatment resistance in patients with schizophrenia: A population-based cohort study. Lancet Psychiatry 2016, 3, 358–366. [Google Scholar] [CrossRef]
- Kim, E.; Howes, O.D.; Veronese, M.; Beck, K.; Seo, S.; Park, J.W.; Lee, J.S.; Lee, Y.S.; Kwon, J.S. Presynaptic dopamine capacity in patients with treatment-resistant schizophrenia taking clozapine: An [(18)f]dopa pet study. Neuropsychopharmacology 2017, 42, 941–950. [Google Scholar] [CrossRef] [PubMed]
- Bora, E. Peripheral inflammatory and neurotrophic biomarkers of cognitive impairment in schizophrenia: A meta-analysis. Psychol. Med. 2019, 49, 1971–1979. [Google Scholar] [CrossRef]
- Fraguas, D.; Diaz-Caneja, C.M.; Ayora, M.; Hernandez-Alvarez, F.; Rodriguez-Quiroga, A.; Recio, S.; Leza, J.C.; Arango, C. Oxidative stress and inflammation in first-episode psychosis: A systematic review and meta-analysis. Schizophr. Bull. 2019, 45, 742–751. [Google Scholar] [CrossRef]
- Garcia-Bueno, B.; Bioque, M.; Mac-Dowell, K.S.; Barcones, M.F.; Martinez-Cengotitabengoa, M.; Pina-Camacho, L.; Rodriguez-Jimenez, R.; Saiz, P.A.; Castro, C.; Lafuente, A.; et al. Pro-/anti-inflammatory dysregulation in patients with first episode of psychosis: Toward an integrative inflammatory hypothesis of schizophrenia. Schizophr. Bull. 2014, 40, 376–387. [Google Scholar] [CrossRef]
- Marini, S.; De Berardis, D.; Vellante, F.; Santacroce, R.; Orsolini, L.; Valchera, A.; Girinelli, G.; Carano, A.; Fornaro, M.; Gambi, F.; et al. Celecoxib adjunctive treatment to antipsychotics in schizophrenia: A review of randomized clinical add-on trials. Mediat. Inflamm. 2016, 2016, 3476240. [Google Scholar] [CrossRef]
- Muller, N. Inflammation in schizophrenia: Pathogenetic aspects and therapeutic considerations. Schizophr. Bull. 2018, 44, 973–982. [Google Scholar] [CrossRef]
- Orsolini, L.; Sarchione, F.; Vellante, F.; Fornaro, M.; Matarazzo, I.; Martinotti, G.; Valchera, A.; Di Nicola, M.; Carano, A.; Di Giannantonio, M.; et al. Protein-c reactive as biomarker predictor of schizophrenia phases of illness? A systematic review. Curr. Neuropharmacol. 2018, 16, 583–606. [Google Scholar] [CrossRef]
- Rakvag, T.T.; Klepstad, P.; Baar, C.; Kvam, T.M.; Dale, O.; Kaasa, S.; Krokan, H.E.; Skorpen, F. The val158met polymorphism of the human catechol-o-methyltransferase (comt) gene may influence morphine requirements in cancer pain patients. Pain 2005, 116, 73–78. [Google Scholar] [CrossRef]
- Lachman, H.M.; Papolos, D.F.; Saito, T.; Yu, Y.M.; Szumlanski, C.L.; Weinshilboum, R.M. Human catechol-o-methyltransferase pharmacogenetics: Description of a functional polymorphism and its potential application to neuropsychiatric disorders. Pharmacogenetics 1996, 6, 243–250. [Google Scholar] [CrossRef] [PubMed]
- Lotta, T.; Vidgren, J.; Tilgmann, C.; Ulmanen, I.; Melen, K.; Julkunen, I.; Taskinen, J. Kinetics of human soluble and membrane-bound catechol o-methyltransferase: A revised mechanism and description of the thermolabile variant of the enzyme. Biochemistry 1995, 34, 4202–4210. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Lipska, B.K.; Halim, N.; Ma, Q.D.; Matsumoto, M.; Melhem, S.; Kolachana, B.S.; Hyde, T.M.; Herman, M.M.; Apud, J.; et al. Functional analysis of genetic variation in catechol-o-methyltransferase (comt): Effects on mrna, protein, and enzyme activity in postmortem human brain. Am. J. Hum. Genet. 2004, 75, 807–821. [Google Scholar] [CrossRef] [PubMed]
- Dempster, E.L.; Mill, J.; Craig, I.W.; Collier, D.A. The quantification of comt mrna in post mortem cerebellum tissue: Diagnosis, genotype, methylation and expression. BMC Med. Genet. 2006, 7, 10. [Google Scholar] [CrossRef] [PubMed]
- Moretti, P.N.; Ota, V.K.; Gouvea, E.S.; Pedrini, M.; Santoro, M.L.; Talarico, F.; Spindola, L.M.; Carvalho, C.M.; Noto, C.; Xavier, G.; et al. Accessing gene expression in treatment-resistant schizophrenia. Mol. Neurobiol. 2018, 55, 7000–7008. [Google Scholar] [CrossRef] [PubMed]
- Inada, T.; Nakamura, A.; Iijima, Y. Relationship between catechol-o-methyltransferase polymorphism and treatment-resistant schizophrenia. Am. J. Med. Genet. B Neuropsychiatr. Genet. 2003, 120B, 35–39. [Google Scholar] [CrossRef]
- Sagud, M.; Tudor, L.; Uzun, S.; Perkovic, M.N.; Zivkovic, M.; Konjevod, M.; Kozumplik, O.; Vuksan Cusa, B.; Svob Strac, D.; Rados, I.; et al. Haplotypic and genotypic association of catechol-o-methyltransferase rs4680 and rs4818 polymorphisms and treatment resistance in schizophrenia. Front. Pharm. 2018, 9, 705. [Google Scholar] [CrossRef]
- Liu, L.; Luo, Y.; Zhang, G.; Jin, C.; Zhou, Z.; Cheng, Z.; Yuan, G. The mrna expression of drd2, pi3kcb, and akt1 in the blood of acute schizophrenia patients. Psychiatry Res. 2016, 243, 397–402. [Google Scholar] [CrossRef]
- Liu, L.; Luo, Y.; Zhang, G.; Jin, C.; Zhou, Z.; Cheng, Z.; Yuan, G. Correlation of drd2 mrna expression levels with deficit syndrome severity in chronic schizophrenia patients receiving clozapine treatment. Oncotarget 2017, 8, 86515–86526. [Google Scholar] [CrossRef]
- Han, J.; Li, Y.; Wang, X. Potential link between genetic polymorphisms of catechol-o-methyltransferase and dopamine receptors and treatment efficacy of risperidone on schizophrenia. Neuropsychiatr. Dis. Treat. 2017, 13, 2935–2943. [Google Scholar] [CrossRef]
- Xu, Q.; Wu, X.; Li, M.; Huang, H.; Minica, C.; Yi, Z.; Wang, G.; Shen, L.; Xing, Q.; Shi, Y.; et al. Association studies of genomic variants with treatment response to risperidone, clozapine, quetiapine and chlorpromazine in the chinese han population. Pharm. J. 2016, 16, 357–365. [Google Scholar] [CrossRef] [PubMed]
- Leclerc, D.; Christensen, K.E.; Cauvi, O.; Yang, E.; Fournelle, F.; Bahous, R.H.; Malysheva, O.V.; Deng, L.; Wu, Q.; Zhou, Z.; et al. Mild methylenetetrahydrofolate reductase deficiency alters inflammatory and lipid pathways in liver. Mol. Nutr. Food Res. 2019, 63, e1801001. [Google Scholar] [CrossRef] [PubMed]
- Moustafa, A.A.; Hewedi, D.H.; Eissa, A.M.; Frydecka, D.; Misiak, B. Homocysteine levels in schizophrenia and affective disorders-focus on cognition. Front. Behav. Neurosci. 2014, 8, 343. [Google Scholar] [CrossRef]
- Roffman, J.L.; Weiss, A.P.; Deckersbach, T.; Freudenreich, O.; Henderson, D.C.; Wong, D.H.; Halsted, C.H.; Goff, D.C. Interactive effects of comt val108/158met and mthfr c677t on executive function in schizophrenia. Am. J. Med. Genet. B Neuropsychiatr. Genet. 2008, 147B, 990–995. [Google Scholar] [CrossRef] [PubMed]
- Allen, N.C.; Bagade, S.; McQueen, M.B.; Ioannidis, J.P.; Kavvoura, F.K.; Khoury, M.J.; Tanzi, R.E.; Bertram, L. Systematic meta-analyses and field synopsis of genetic association studies in schizophrenia: The szgene database. Nat. Genet. 2008, 40, 827–834. [Google Scholar] [CrossRef]
- Gilbody, S.; Lewis, S.; Lightfoot, T. Methylenetetrahydrofolate reductase (mthfr) genetic polymorphisms and psychiatric disorders: A huge review. Am. J. Epidemiol. 2007, 165, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Roffman, J.L.; Weiss, A.P.; Purcell, S.; Caffalette, C.A.; Freudenreich, O.; Henderson, D.C.; Bottiglieri, T.; Wong, D.H.; Halsted, C.H.; Goff, D.C. Contribution of methylenetetrahydrofolate reductase (mthfr) polymorphisms to negative symptoms in schizophrenia. Biol. Psychiatry 2008, 63, 42–48. [Google Scholar] [CrossRef]
- Roffman, J.L.; Weiss, A.P.; Deckersbach, T.; Freudenreich, O.; Henderson, D.C.; Purcell, S.; Wong, D.H.; Halsted, C.H.; Goff, D.C. Effects of the methylenetetrahydrofolate reductase (mthfr) c677t polymorphism on executive function in schizophrenia. Schizophr. Res. 2007, 92, 181–188. [Google Scholar] [CrossRef] [PubMed]
- Roffeei, S.N.; Mohamed, Z.; Reynolds, G.P.; Said, M.A.; Hatim, A.; Mohamed, E.H.; Aida, S.A.; Zainal, N.Z. Association of fto, lepr and mthfr gene polymorphisms with metabolic syndrome in schizophrenia patients receiving antipsychotics. Pharmacogenomics 2014, 15, 477–485. [Google Scholar] [CrossRef]
- Ikeda, H.; Kamei, J.; Koshikawa, N.; Cools, A.R. Nucleus accumbens and dopamine-mediated turning behavior of the rat: Role of accumbal non-dopaminergic receptors. J. Pharm. Sci. 2012, 120, 152–164. [Google Scholar] [CrossRef]
- Suzuki, T.; Remington, G.; Mulsant, B.H.; Uchida, H.; Rajji, T.K.; Graff-Guerrero, A.; Mimura, M.; Mamo, D.C. Defining treatment-resistant schizophrenia and response to antipsychotics: A review and recommendation. Psychiatry Res. 2012, 197, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Molins, C.; Roldan, A.; Corripio, I.; Isohanni, M.; Miettunen, J.; Seppala, J.; Seppala, A.; Koponen, H.; Moilanen, J.; Jaaskelainen, E.; et al. Response to antipsychotic drugs in treatment-resistant schizophrenia: Conclusions based on systematic review. Schizophr. Res. 2016, 178, 64–67. [Google Scholar] [CrossRef] [PubMed]
- National Center for Biotechnology Information. Available online: https://www.ncbi.nlm.nih.gov/snp/rs6275 (accessed on 12 December 2017).
- Hajj, A.; Halepian, L.; Osta, N.E.; Chahine, G.; Kattan, J.; Rabbaa Khabbaz, L. Oprm1 c.118a>g polymorphism and duration of morphine treatment associated with morphine doses and quality-of-life in palliative cancer pain settings. Int. J. Mol. Sci. 2017, 18, 669. [Google Scholar] [CrossRef] [PubMed]
- Hajj, A.; Peoc’h, K.; Laplanche, J.L.; Jabbour, H.; Naccache, N.; Abou Zeid, H.; Yazbeck, P.; Khabbaz, L.R. Genotyping test with clinical factors: Better management of acute postoperative pain? Int. J. Mol. Sci. 2015, 16, 6298–6311. [Google Scholar] [CrossRef] [PubMed]
- Almawi, W.Y.; Finan, R.R.; Tamim, H.; Daccache, J.L.; Irani-Hakime, N. Differences in the frequency of the c677t mutation in the methylenetetrahydrofolate reductase (mthfr) gene among the lebanese population. Am. J. Hematol. 2004, 76, 85–87. [Google Scholar] [CrossRef] [PubMed]
- Mahfouz, R.A.; Cortas, N.K.; Charafeddine, K.M.; Abdul Khalik, R.N.; Sarieddine, D.S.; Kadi, R.H.; Daher, R.T. Correlation of methylenetetrahydrofolate reductase polymorphisms with homocysteine metabolism in healthy lebanese adults. Gene 2012, 504, 175–180. [Google Scholar] [CrossRef]
- Sabbagh, A.S.; Mahfoud, Z.; Taher, A.; Zaatari, G.; Daher, R.; Mahfouz, R.A. High prevalence of mthfr gene a1298c polymorphism in lebanon. Genet. Test. 2008, 12, 75–80. [Google Scholar] [CrossRef]
- National Center for Biotechnology Information. Available online: https://www.ncbi.nlm.nih.gov/projects/snp/snp_ref.Cgi?Rs = 4680 (accessed on 12 December 2016).
- National Center for Biotechnology Information. Available online: http://www.ncbi.nlm.nih.gov/snp/?Term = rs1799971 (accessed on 12 December 2016).
- National Center for Biotechnology Information. Available online: http://www.ncbi.nlm.nih.gov/snp/?Term = rs1045642 (accessed on 12 December 2016).
- Danivas, V.; Venkatasubramanian, G. Current perspectives on chlorpromazine equivalents: Comparing apples and oranges! Indian J. Psychiatry 2013, 55, 207–208. [Google Scholar]
- Hallit, S.; Obeid, S.; Haddad, C.; Kazour, F.; Kazour, G. Validation of the arabic version of the panss scale among lebanese schizophrenic patients. J. Psychopathol. G. Di Psicopatol. 2017, 23, 60–66. [Google Scholar]
- Kay, S.R.; Opler, L.A.; Lindenmayer, J.-P. Reliability and validity of the positive and negative syndrome scale for schizophrenics. Psychiatry Res. 1988, 23, 99–110. [Google Scholar] [CrossRef]
- Preston, C.C.; Colman, A.M. Optimal number of response categories in rating scales: Reliability, validity, discriminating power, and respondent preferences. Acta. Psychol. (Amst) 2000, 104, 1–15. [Google Scholar] [CrossRef]
- Lu, M.; Safren, S.A.; Skolnik, P.R.; Rogers, W.H.; Coady, W.; Hardy, H.; Wilson, I.B. Optimal recall period and response task for self-reported hiv medication adherence. Aids Behav. 2008, 12, 86–94. [Google Scholar] [CrossRef] [PubMed]
- Garfield, S.; Eliasson, L.; Clifford, S.; Willson, A.; Barber, N. Developing the diagnostic adherence to medication scale (the dams) for use in clinical practice. BMC Health Serv. Res. 2012, 12, 350. [Google Scholar] [CrossRef] [PubMed]
- Harrison, P.J.; Tunbridge, E.M. Catechol-o-methyltransferase (comt): A gene contributing to sex differences in brain function, and to sexual dimorphism in the predisposition to psychiatric disorders. Neuropsychopharmacology 2008, 33, 3037–3045. [Google Scholar] [CrossRef] [PubMed]
- Laatikainen, L.M.; Sharp, T.; Harrison, P.J.; Tunbridge, E.M. Sexually dimorphic effects of catechol-o-methyltransferase (comt) inhibition on dopamine metabolism in multiple brain regions. PLoS ONE 2013, 8, e61839. [Google Scholar] [CrossRef]
- Bursac, Z.; Gauss, C.H.; Williams, D.K.; Hosmer, D.W. Purposeful selection of variables in logistic regression. Source Code Biol. Med. 2008, 3, 17. [Google Scholar] [CrossRef]
- Lally, J.; Ajnakina, O.; Di Forti, M.; Trotta, A.; Demjaha, A.; Kolliakou, A.; Mondelli, V.; Reis Marques, T.; Pariante, C.; Dazzan, P.; et al. Two distinct patterns of treatment resistance: Clinical predictors of treatment resistance in first-episode schizophrenia spectrum psychoses. Psychol. Med. 2016, 46, 3231–3240. [Google Scholar] [CrossRef]
- Joober, R.; Rouleau, G.A.; Lal, S.; Bloom, D.; Lalonde, P.; Labelle, A.; Benkelfat, C. Increased prevalence of schizophrenia spectrum disorders in relatives of neuroleptic-nonresponsive schizophrenic patients. Schizophr. Res. 2005, 77, 35–41. [Google Scholar] [CrossRef]
- Hassan, A.N.; De Luca, V. The effect of lifetime adversities on resistance to antipsychotic treatment in schizophrenia patients. Schizophr. Res. 2015, 161, 496–500. [Google Scholar] [CrossRef]
- Crespo-Facorro, B.; de la Foz, V.O.; Ayesa-Arriola, R.; Perez-Iglesias, R.; Mata, I.; Suarez-Pinilla, P.; Tabares-Seisdedos, R.; Vazquez-Barquero, J.L. Prediction of acute clinical response following a first episode of non affective psychosis: Results of a cohort of 375 patients from the spanish pafip study. Prog. Neuropsychopharmacol. Biol. Psychiatry 2013, 44, 162–167. [Google Scholar] [CrossRef]
- Perkins, D.; Lieberman, J.; Gu, H.; Tohen, M.; McEvoy, J.; Green, A.; Zipursky, R.; Strakowski, S.; Sharma, T.; Kahn, R.; et al. Predictors of antipsychotic treatment response in patients with first-episode schizophrenia, schizoaffective and schizophreniform disorders. Br. J. Psychiatry 2004, 185, 18–24. [Google Scholar] [CrossRef] [PubMed]
- Zalloua, P.A.; Xue, Y.; Khalife, J.; Makhoul, N.; Debiane, L.; Platt, D.E.; Royyuru, A.K.; Herrera, R.J.; Hernanz, D.F.; Blue-Smith, J.; et al. Y-chromosomal diversity in lebanon is structured by recent historical events. Am. J. Hum. Genet. 2008, 82, 873–882. [Google Scholar] [CrossRef] [PubMed]
- Escamilla, R.; Camarena, B.; Saracco-Alvarez, R.; Fresan, A.; Hernandez, S.; Aguilar-Garcia, A. Association study between comt, drd2, and drd3 gene variants and antipsychotic treatment response in mexican patients with schizophrenia. Neuropsychiatr. Dis. Treat. 2018, 14, 2981–2987. [Google Scholar] [CrossRef] [PubMed]
- Sery, O.; Prikryl, R.; Castulik, L.; St’astny, F. A118g polymorphism of oprm1 gene is associated with schizophrenia. J. Mol. Neurosci. 2010, 41, 219–222. [Google Scholar] [CrossRef] [PubMed]
- Rajagopal, V.M.; Rajkumar, A.P.; Jacob, K.S.; Jacob, M. Gene-gene interaction between drd4 and comt modulates clinical response to clozapine in treatment-resistant schizophrenia. Pharm. Genom. 2018, 28, 31–35. [Google Scholar] [CrossRef] [PubMed]
| Frequency (%) * | ||
|---|---|---|
| Gender | Male | 27 (27.0%) |
| Female | 73 (73.0%) | |
| Marital status | Married | 18 (19.4%) |
| Single | 68 (73.1%) | |
| Divorced | 7 (7.5%) | |
| Level of education | Primary | 42 (51.2%) |
| Secondary | 32 (39.0%) | |
| University | 8 (9.8%) | |
| Consumption of psychoactive substances | No | 92 (95.8%) |
| Yes | 4 (4.2%) | |
| Alcohol consumption | No | 87 (89.7%) |
| Yes | 10 (10.3%) | |
| Smoking | No | 35 (36.1%) |
| Yes | 62 (63.9%) | |
| Family history of schizophrenic episodes | No | 62 (66.7%) |
| Yes | 31 (33.3%) | |
| Family history of other psychiatric disorders | No | 62 (70.5%) |
| Yes | 26 (29.5%) | |
| Personal history of schizophrenic episodes | No | 11 (11.6%) |
| Yes | 84 (88.4%) | |
| Personal history of other psychiatric disorders | No | 59 (81.9%) |
| Yes | 13 (18.1%) | |
| Mean ± SD | ||
| Age (years) | 49.35 ± 12.56 | |
| Time since the beginning of treatment (Years) | 2.48 ± 1.89 | |
| Chlorpromazine equivalent-dose (mg) | 1210.83 ± 1247.74 | |
| Number of episodes | 6.96 ± 5.43 | |
| Alternative medicine: | ||
| Psychotherapy | 15 (17%) | |
| Social assistance | 6 (6.8%) | |
| Art therapy | 12 (13.6%) | |
| Ergotherapy | 7 (8%) | |
| Physical therapy | 7 (8%) | |
| Gene dbSNP | Genotype Frequencies 1 | Allelic Frequencies | p2 | |||
|---|---|---|---|---|---|---|
| DRD2 rs6275 | CC | CT | TT | C | T | |
| Schizophrenic patients n = 92 3 (current study, schizophrenia) | 16 (17.4) | 47 (51.1) | 29 (31.5) | 0.43 | 0.57 | - |
| HapMap European n = 113 [43] | 48 (42.5) | 55 (48.7) | 10 (8.8) | 0.67 | 0.33 | <0.001 * |
| HapMap Japanese = 86 [43] | 22 (25.6) | 39 (45.4) | 25 (29) | 0.48 | 0.52 | 0.4 |
| HapMap Chinese n = 43 [43] | 10 (23.3) | 27 (62.8) | 6 (13.9) | 0.55 | 0.45 | 0.09 |
| HapMap Sub-Saharan African n = 113 [43] | 14 (12.4) | 39 (34.5) | 60 (53.1) | 0.30 | 0.70 | 0.008 * |
| OPRM1 rs1799971 | AA | AG | GG | A | G | |
| Schizophrenic patients n = 100 (current study, schizophrenia) | 77 (77.0) | 23 (23.0) | 0 (0) | 0.89 | 0.12 | - |
| Lebanese patients n = 84 [44] | 67 (79.8) | 17 (20.2) | 0 (0) | 0.90 | 0.10 | 0.9 |
| Lebansee patients n = 96 [45] | 76 (79.2) | 18 (18.8) | 2 (2.1) | 0.89 | 0.11 | 0.28 |
| COMT rs4680 | Val/Val | Val/Met | Met/Met | Val | Met | |
| Schizophrenic patients n = 100 (current study, schizophrenia) | 34 (34.0) | 41 (41.0) | 25 (25.0) | 0.55 | 0.46 | - |
| Lebanese patients n = 84 [44] | 22 (26.2) | 42 (50) | 20 (23.8) | 0.51 | 0.49 | 0.42 |
| Lebanese patients n = 96 [45] | 23 (24) | 48 (50) | 25 (26) | 0.49 | 0.51 | 0.27 |
| MTHFR rs1801133 | CC | CT | TT | C | T | |
| Schizophrenic patients n = 100 (current study, schizophrenia) | 42 (42.0) | 42 (42.0) | 16 (16.0) | 0.63 | 0.37 | - |
| Lebanese patients n = 589 [46] | 290 (49.2) | 234 (39.7) | 65 (11.0) | 0.69 | 0.31 | 0.24 |
| Lebanese patients n = 233 [47] | 105 (45.0) | 101 (43.3) | 27 (11.6) | 0.67 | 0.33 | 0.54 |
| Lebanese patients n = 205 [48] | 134 (65.3) | 63 (30.8) | 8 (3.9) | 0.81 | 0.19 | <0.0001 * |
| Resistance to Treatment (BPRS) | ||||
|---|---|---|---|---|
| No | Yes | p-Value | ||
| Frequency (%) | Frequency (%) | |||
| Gender | Male | 4 (7.5%) | 23 (48.9%) | <0.001 |
| Female | 49 (92.5%) | 24 (51.1%) | ||
| Consumption of psychoactive substances | No | 53 (100.0%) | 39 (90.7%) | 0.023 |
| Yes | 0 (0.0%) | 4 (9.3%) | ||
| COMT | VV | 18 (34%) | 16 (34%) | 0.991 |
| VM | 22 (41.5%) | 19 (40.4%) | ||
| MM | 13 (24.5%) | 12 (25.5%) | ||
| COMT (male) | VV | 4 (100%) | 5 (21.7%) | 0.008 |
| VM | 0 (0%) | 12 (52.3%) | ||
| MM | 0 (0%) | 6 (26.1%) | ||
| COMT (female) | VV | 14 (28.6%) | 11 (45.8%) | 0.298 |
| VM | 22 (44.9%) | 7 (29.2%) | ||
| MM | 13 (26.5%) | 6 (25.0%) | ||
| DRD2 | CC | 9 (18%) | 7 (16.7%) | 0.529 |
| CT | 23 (46%) | 24 (57.1%) | ||
| TT | 18 (36%) | 11 (26.2%) | ||
| MTHFR | CC | 24 (45.3%) | 18 (38.3%) | 0.390 |
| CT | 19 (35.8%) | 23 (48.9%) | ||
| TT | 10 (18.9%) | 6 (12.8%) | ||
| OPRM1 | AA | 41 (77.4%) | 36 (76.6%) | 0.928 |
| AG | 12 (22.6%) | 11 (23.4%) | ||
| GG | 0 (0.0%) | 0 (0.0%) | ||
| Mean ± SD | Mean ± SD | p-value | ||
| Adherence to treatment score (over 8) | 6.22 ± 1.28 | 5.37 ± 1.59 | 0.005 | |
| Time since the beginning of treatment (years) | 2.84 ± 1.89 | 1.85 ± 1.74 | 0.029 | |
| Chlorpromazine-equivalent dose (mg) | 984.11 ± 1299.53 | 1466.48 ± 1231.12 | 0.053 | |
| Resistance to Treatment (PANSS) | ||||
|---|---|---|---|---|
| No | Yes | p-Value | ||
| Frequency (%) | Frequency (%) | |||
| Gender | Male | 11(16.9%) | 16(45.7%) | 0.002 |
| Female | 54(83.1%) | 19(54.3%) | ||
| Level of education | Primary | 29(53.7%) | 13(46.4%) | 0.036 |
| Secondary | 23(42.6%) | 9(32.1%) | ||
| University | 2(3.7%) | 6(21.4%) | ||
| Consumption of psychoactive substances | No | 65(100.0%) | 27(87.1%) | 0.003 |
| Yes | 0(0.0%) | 4(12.9%) | ||
| Family history of schizophrenic episodes | No | 47(73.4%) | 15(51.7%) | 0.040 |
| Yes | 17(26.6%) | 14(48.3%) | ||
| Psychotherapy | No | 53(88.3%) | 20(71.4%) | 0.050 |
| Yes | 7(11.7%) | 8(28.6%) | ||
| Treatment by benzodiazepines | No | 41(63.1%) | 29(82.9%) | 0.040 |
| Yes | 24(36.9%) | 6(17.1%) | ||
| Treatment by anticholinergic drugs | No | 23 (35.4%) | 3 (8.6%) | 0.004 |
| Yes | 42 (64.6%) | 32 (91.4%) | ||
| COMT | VV | 23 (35.4%) | 11 (31.4%) | 0.822 |
| VM | 27 (41.5%) | 14 (40.0%) | ||
| MM | 15 (23.1%) | 10 (28.6%) | ||
| COMT (male) | VV | 7 (63.6%) | 2 (12.5%) | 0.023 |
| VM | 3 (27.3%) | 9 (56.3%) | ||
| MM | 1 (9.1%) | 5 (31.3%) | ||
| COMT (female) | VV | 16 (29.6%) | 9 (47.4%) | 0.273 |
| VM | 24 (44.4%) | 5 (26.3%) | ||
| MM | 14 (25.9%) | 5 (26.3%) | ||
| DRD2 | CC | 10 (16.1%) | 6 (20.0%) | 0.897 |
| CT | 32 (51.6%) | 15 (50.0%) | ||
| TT | 20 (32.3%) | 9 (30.0%) | ||
| MTHFR | CC | 28 (43.1%) | 14 (40.0%) | 0.520 |
| CT | 25 (38.5%) | 17 (48.6%) | ||
| TT | 12 (18.5%) | 4 (11.4%) | ||
| OPRM1 | AA | 52 (80.0%) | 25 (71.4%) | 0.331 |
| AG | 13 (20.0%) | 10 (28.6%) | ||
| GG | 0 (0.0%) | 0 (0.0%) | ||
| Mean ± SD | Mean ± SD | |||
| Time since the beginning of treatment (years) | 2.71 ± 1.92 | 1.85 ± 1.69 | 0.080 | |
| Chlorpromazine equivalent dose (mg) | 1006.28 ± 1232.53 | 1590.71 ± 1202.11 | 0.025 | |
| Adherence to treatment score (over 8) | 6.10 ± 1.37 | 5.31 ± 1.61 | 0.010 | |
| Resistance to Treatment (BPRS or PANSS) | ||||
|---|---|---|---|---|
| No | Yes | p-Value | ||
| Frequency (%) | Frequency (%) | |||
| Gender | Male | 2 (4.0%) | 23 (47.9%) | <0.001 |
| Female | 48 (96.0%) | 25 (52.1%) | ||
| Consumption of psychoactive substances | No | 50 (100.0%) | 40 (90.9%) | 0.029 |
| Yes | 0 (0.0%) | 4 (9.1%) | ||
| Family history of schizophrenic episodes | No | 38 (76.0%) | 22 (53.7%) | 0.025 |
| Yes | 12 (24.0%) | 19 (46.3%) | ||
| COMT | VV | 16 (32.0%) | 16 (33.3%) | 0.896 |
| VM | 22 (44.0%) | 19 (39.6%) | ||
| MM | 12 (24.0%) | 13 (27.1%) | ||
| DRD2 | CC | 7 (14.9%) | 8 (18.6%) | 0.549 |
| CT | 23 (48.9%) | 24 (55.8%) | ||
| TT | 17 (36.2%) | 11 (25.6%) | ||
| MTHFR | CC | 22 (44.0%) | 18 (37.5%) | 0.609 |
| CT | 19 (38.0%) | 23 (47.9%) | ||
| TT | 9 (18.0%) | 7 (14.6%) | ||
| OPRM1 | AA | 39 (78.0%) | 37 (77.1%) | 0.913 |
| AG | 11 (22.0%) | 11 (22.9%) | ||
| GG | 0 (0.0%) | 0 (0.0%) | ||
| Mean ± SD | Mean ± SD | |||
| Adherence to treatment score (over 8) | 6.40 ± 1.02 | 5.37 ± 1.58 | <0.001 | |
| Time since the beginning of treatment (years) | 2.85 ± 1.91 | 1.82 ± 1.72 | 0.022 | |
| Chlorpromazine equivalent dose (mg) | 851.17 ± 821.37 | 1479.69 ± 1221.38 | 0.004 | |
| Logistic Regression 1: Taking Resistance to Treatment Based on the BPRS as the Dependent Variable | ||||
| Variable | p-Value | ORa | 95% Confidence Interval (CI) | |
| Lower Bound | Lower Bound | |||
| Time since the beginning of treatment (years) | 0.047 | 0.730 | 0.535 | 0.996 |
| Adherence to treatment score | 0.094 | 0.679 | 0.432 | 1.068 |
| Chlorpromazine equivalent daily dose (mg) | 0.062 | 1.001 | 1.000 | 1.001 |
| Variables entered in model 1: Gender, time since the beginning of treatment (years), adherence to treatment score, consumption of psychoactive substances, chlorpromazine equivalent daily dose (mg). | ||||
| Logistic Regression 2: Taking Resistance to Treatment Based on the PANSS as the Dependent Variable | ||||
| Variable | p-Value | ORa | 95% Confidence Interval (CI) | |
| Lower Bound | Lower Bound | |||
| Level of education: Secondary | 0.369 | 0.481 | 0.097 | 2.377 |
| Level of education: University | 0.042 | 11.962 | 1.095 | 130.661 |
| Family history of schizophrenic episodes | 0.043 | 4.617 | 1.048 | 20.329 |
| Time since the beginning of treatment (years) | 0.097 | 0.675 | 0.425 | 1.074 |
| Chlorpromazine equivalent daily dose (mg) | 0.020 | 1.001 | 1.000 | 1.002 |
| Variables entered in model 2: Gender, level of education, family history of schizophrenic episodes, psychotherapy, time since the beginning of treatment (years), adherence to treatment score, consumption of benzodiazepines, chlorpromazine equivalent daily dose (mg). | ||||
| Logistic Regression 3: Taking Resistance to Treatment Based on the BPRS or PANSS as the Dependent Variable | ||||
| Variable | p-Value | ORa | 95% Confidence Interval (CI) | |
| Lower Bound | Lower Bound | |||
| Time since the beginning of treatment (years) | 0.023 | 0.675 | 0.481 | 0.946 |
| Adherence to treatment score | 0.068 | 0.646 | 0.405 | 1.032 |
| Chlorpromazine equivalent daily dose (mg) | 0.026 | 1.001 | 1.000 | 1.002 |
| Family history of schizophrenic episodes | 0.088 | 2.779 | 0.858 | 9.003 |
| Variables entered in model 3: Gender, family history of schizophrenic episodes, time since the beginning of treatment (years), adherence to treatment score, consumption of psychoactive substances, chlorpromazine equivalent daily dose (mg). | ||||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hajj, A.; Obeid, S.; Sahyoun, S.; Haddad, C.; Azar, J.; Rabbaa Khabbaz, L.; Hallit, S. Clinical and Genetic Factors Associated with Resistance to Treatment in Patients with Schizophrenia: A Case-Control Study. Int. J. Mol. Sci. 2019, 20, 4753. https://doi.org/10.3390/ijms20194753
Hajj A, Obeid S, Sahyoun S, Haddad C, Azar J, Rabbaa Khabbaz L, Hallit S. Clinical and Genetic Factors Associated with Resistance to Treatment in Patients with Schizophrenia: A Case-Control Study. International Journal of Molecular Sciences. 2019; 20(19):4753. https://doi.org/10.3390/ijms20194753
Chicago/Turabian StyleHajj, Aline, Sahar Obeid, Saria Sahyoun, Chadia Haddad, Jocelyne Azar, Lydia Rabbaa Khabbaz, and Souheil Hallit. 2019. "Clinical and Genetic Factors Associated with Resistance to Treatment in Patients with Schizophrenia: A Case-Control Study" International Journal of Molecular Sciences 20, no. 19: 4753. https://doi.org/10.3390/ijms20194753
APA StyleHajj, A., Obeid, S., Sahyoun, S., Haddad, C., Azar, J., Rabbaa Khabbaz, L., & Hallit, S. (2019). Clinical and Genetic Factors Associated with Resistance to Treatment in Patients with Schizophrenia: A Case-Control Study. International Journal of Molecular Sciences, 20(19), 4753. https://doi.org/10.3390/ijms20194753