Can Nrf2 Modulate the Development of Intestinal Fibrosis and Cancer in Inflammatory Bowel Disease?
Abstract
1. Introduction
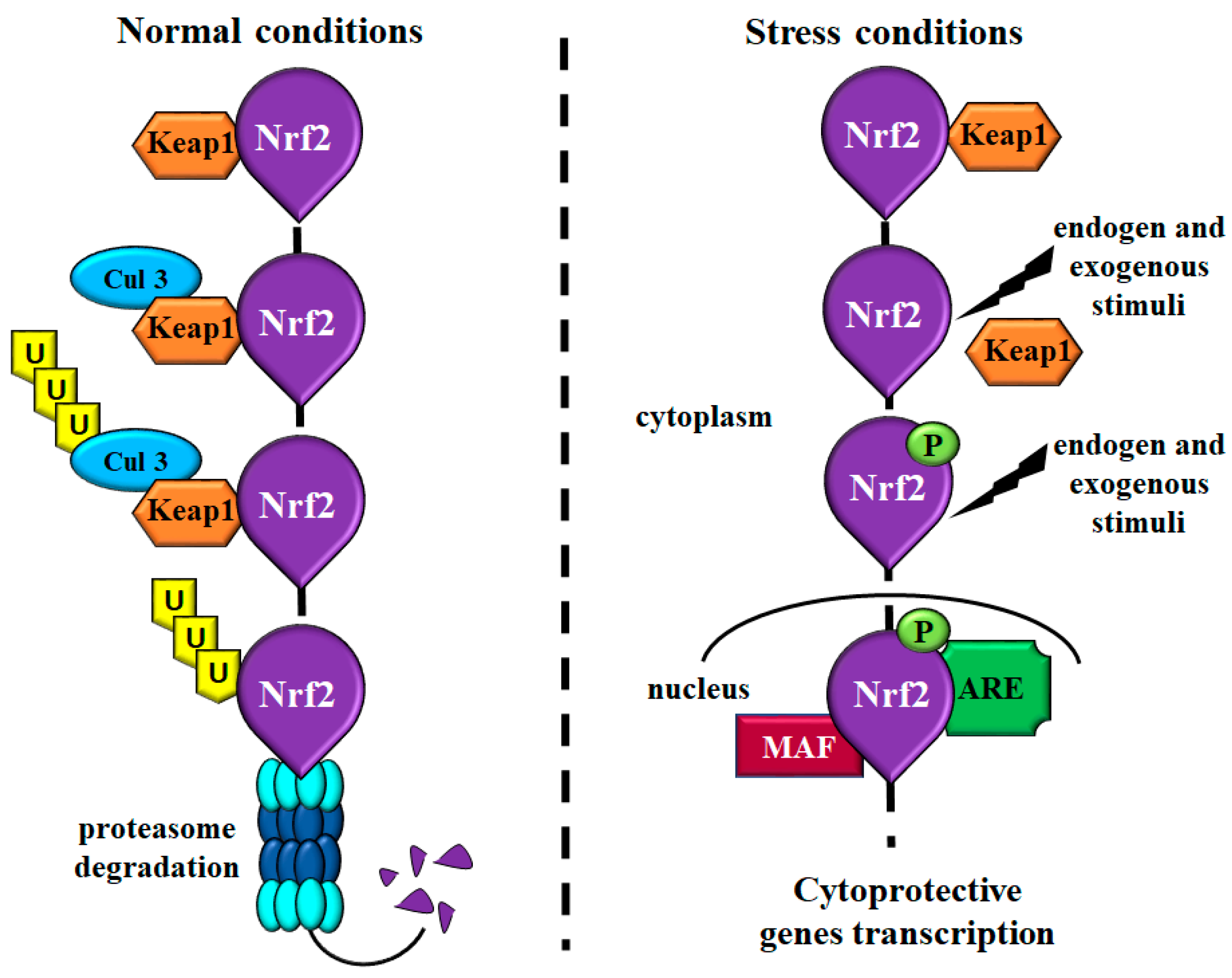
2. What Is Nrf2?
3. Role of Nrf2 in Inflammation and Fibrosis in Various Organs
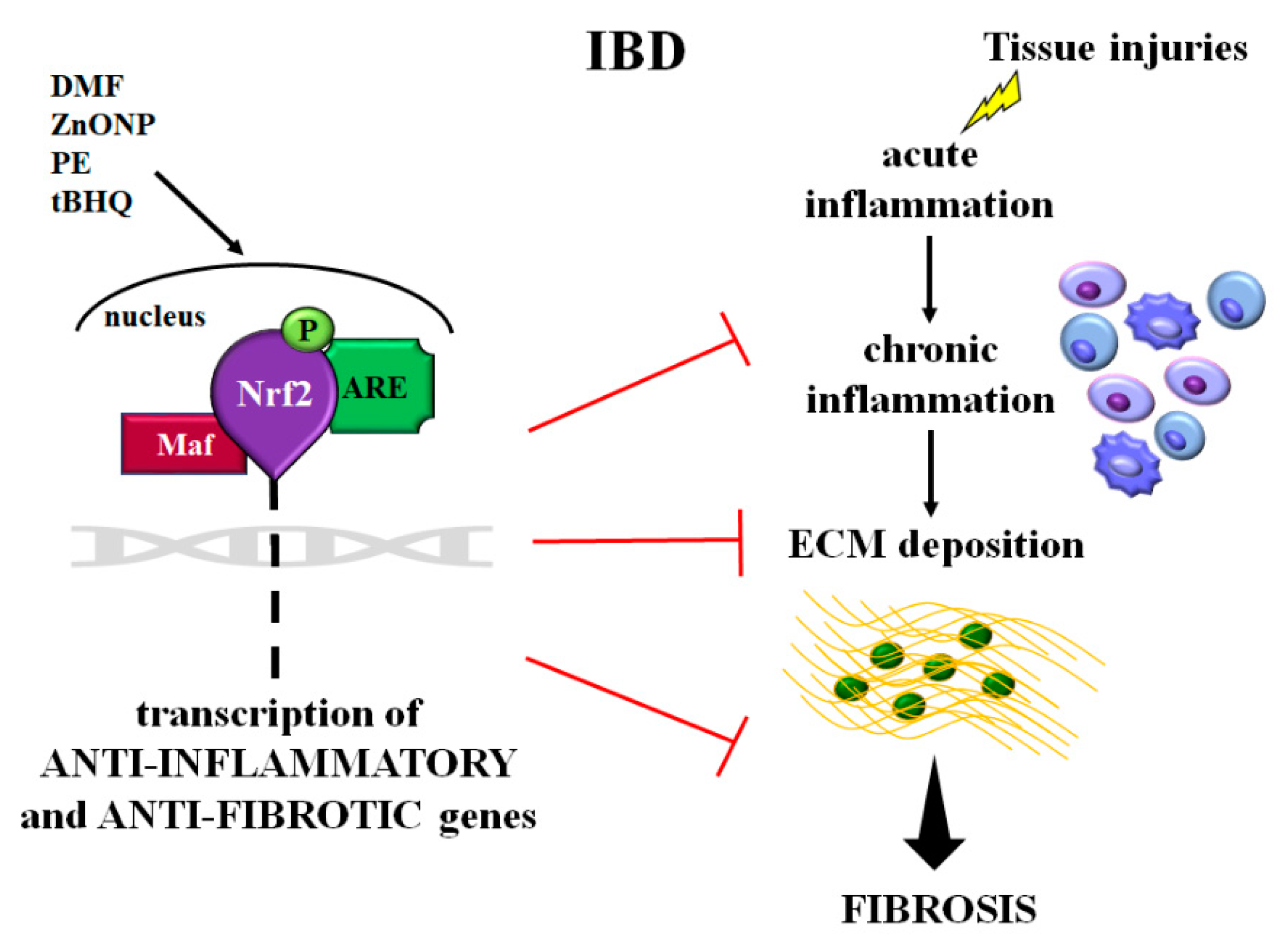
4. Role of Nrf2 Signaling in Intestinal Inflammation and Fibrosis
4.1. Nrf2 in Experimental Models of Colitis Induced in Transgenic Mice
4.2. Nrf2 Inhibition in Experimental Models of Colitis
4.3. Nrf2 Activation in Experimental Models of Colitis
5. Role of Nrf2 in the Cancer of Various Organs
6. Role of Nrf2 in IBD-Associated Colorectal Cancer
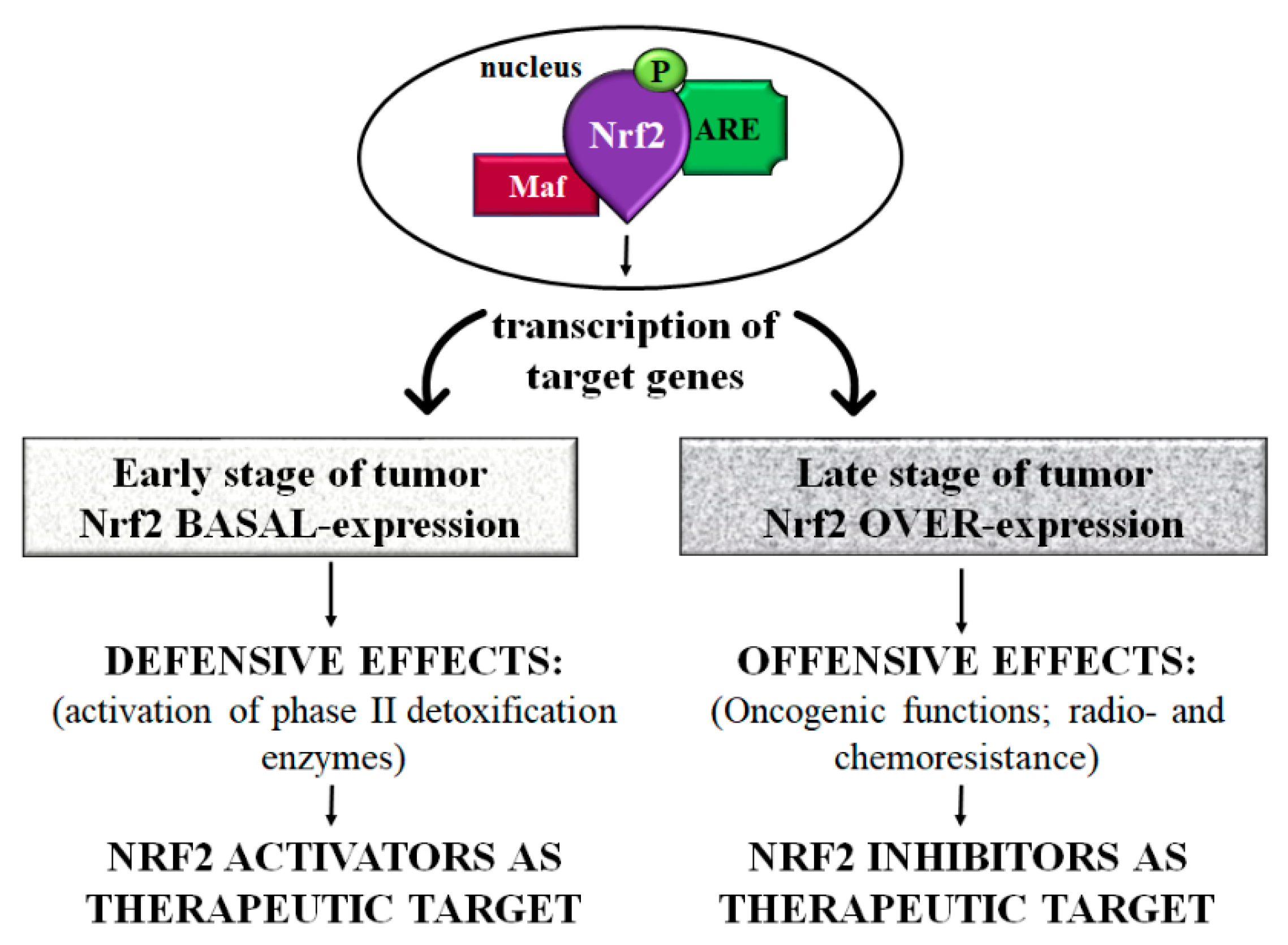
6.1. Nrf2 Plays a “Protective Effect” in the Early Stage of Colorectal Cancer
6.2. Nrf2 Plays an “Offensive Effect” in Late-Stage Colorectal Cancer
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Wynn, T.A. Cellular and molecular mechanisms of fibrosis. J. Pathol. 2008, 214, 199–210. [Google Scholar] [CrossRef] [PubMed]
- Speca, S.; Giusti, I.; Rieder, F.; Latella, G. Cellular and molecular mechanisms of intestinal fibrosis. World J. Gastroenterol. 2012, 18, 3635–3661. [Google Scholar] [CrossRef] [PubMed]
- Vetuschi, A.; Latella, G.; Pompili, S.; Gaudio, E.; Sferra, R. Features of intestinal lesions in the clinical course of inflammatory bowel diseases. Ital. J. Anat. Embryol. 2014, 119, 286–303. [Google Scholar] [PubMed]
- Latella, G.; Di Gregorio, J.; Flati, V.; Rieder, F.; Lawrance, I.C. Mechanisms of initiation and progression of intestinal fibrosis in IBD. Scand. J. Gastroenterol. 2015, 50, 53–65. [Google Scholar] [CrossRef] [PubMed]
- Sferra, R.; Pompili, S.; Festuccia, C.; Marampon, F.; Gravina, G.L.; Ventura, L.; Di Cesare, E.; Cicchinelli, S.; Gaudio, E.; Vetuschi, A. The possible prognostic role of histone deacetylase and transforming growth factor β/Smad signaling in high grade gliomas treated by radio-chemotherapy: A preliminary immunohistochemical study. Eur. J. Histochem. 2017, 61, 2732. [Google Scholar] [CrossRef] [PubMed]
- Vetuschi, A.; Pompili, S.; Gaudio, E.; Latella, G.; Sferra, R. PPAR-γ with its anti-inflammatory and anti-fibrotic action could be an effective therapeutic target in IBD. Eur. Rev. Med. Pharmacol. Sci. 2018, 22, 8839–8848. [Google Scholar]
- Samarakoon, R.; Overstreet, J.M.; Higgins, P.J. TGF-βsignaling in tissue fibrosis: Redox controls, target genes and therapeutic opportunities. Cell Signal. 2013, 25, 264–268. [Google Scholar] [CrossRef]
- Liu, R.M.; Desai, L.P. Reciprocal regulation of TGF-βand reactive oxygen species: A perverse cycle for fibrosis. Redox Biol. 2015, 6, 565–577. [Google Scholar] [CrossRef]
- Richter, K.; Kietzmann, T. Reactive oxygen species and fibrosis: Further evidence of a significant liaison. Cell Tissue Res. 2016, 365, 591–605. [Google Scholar] [CrossRef]
- Swamy, S.M.; Rajasekaran, N.S.; Thannickal, V.J. Nuclear factor erythroid-2 related factor 2 in aging and lung fibrosis. Am. J. Pathol. 2016, 186, 1712–1723. [Google Scholar] [CrossRef]
- Ahamed, J.; Laurence, J. Role of platelet-derived transforming growth factor-β1 and reactive oxygen species in radiation-induced organ fibrosis. Antioxid. Redox Signal. 2017, 27, 977–988. [Google Scholar] [CrossRef] [PubMed]
- Khor, T.O.; Huang, M.T.; Prawan, A.; Liu, Y.; Hao, X.; Yu, S.; Cheung, W.K.; Chan, J.Y.; Reddy, B.S.; Yang, C.S.; et al. Increased susceptibility of Nrf2 knockout mice to colitis-associated colorectal cancer. Cancer Prev. Res. 2008, 1, 187–191. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.; Misra, V.; Thimmulappa, R.K.; Lee, H.; Ames, S.; Hoque, M.O.; Herman, J.G.; Baylin, S.B.; Sidransky, D.; Gabrielson, E.; et al. Dysfunctional KEAP1-NRF2 interaction in non-small-cell lung cancer. PLoS Med. 2006, 3, e420. [Google Scholar] [CrossRef]
- Shibata, T.; Kokubu, A.; Gotoh, M.; Ojima, H.; Ohta, T.; Yamamoto, M.; Hirohashi, S. Genetic alteration of Keap1 confers constitutive Nrf2 activation and resistance to chemotherapy in gallbladder cancer. Gastroenterology 2008, 135, 1358–1368. [Google Scholar] [CrossRef]
- Wang, R.; An, J.; Ji, F.; Jiao, H.; Sun, H.; Zhou, D. Hypermethylation of the Keap1 gene in human lung cancer cell lines and lung cancer tissues. Biochem. Biophys. Res. Commun. 2008, 373, 151–154. [Google Scholar] [CrossRef] [PubMed]
- Solis, L.M.; Behrens, C.; Dong, W.; Suraokar, M.; Ozburn, N.C.; Moran, C.A.; Corvalan, A.H.; Biswal, S.; Swisher, S.G.; Bekele, B.N.; et al. Nrf2 and Keap1 abnormalities in non-small cell lung carcinoma and association with clinicopathologic features. Clin. Cancer Res. 2010, 16, 3743–3753. [Google Scholar] [CrossRef]
- Zhang, P.; Singh, A.; Yegnasubramanian, S.; Esopi, D.; Kombairaju, P.; Bodas, M.; Wu, H.; Bova, S.G.; Biswal, S. Loss of Kelch-like ECH-associated protein 1 function in prostate cancer cells causes chemoresistance and radioresistance and promotes tumor growth. Mol. Cancer Ther. 2010, 9, 336–346. [Google Scholar] [CrossRef] [PubMed]
- Jung, B.J.; Yoo, H.S.; Shin, S.; Park, Y.J.; Jeon, S.M. Dysregulation of NRF2 in Cancer: From Molecular Mechanisms to Therapeutic Opportunities. Biomol. Ther. 2018, 26, 57–68. [Google Scholar] [CrossRef] [PubMed]
- Latella, G. Redox Imbalance in Intestinal Fibrosis: Beware of the TGF-β1, ROS, and Nrf2 Connection. Dig. Dis. Sci. 2018, 63, 312–320. [Google Scholar] [CrossRef] [PubMed]
- Latella, G. Concise Commentary: Is Nrf2 a Master Regulator of Intestinal Fibrosis? Dig. Dis. Sci. 2018, 63, 381–382. [Google Scholar] [CrossRef] [PubMed]
- Guan, Y.; Tan, Y.; Liu, W.; Wang, D.; Pan, D.; Sun, Y.; Zheng, C. NF-E2-Related Factor 2 Suppresses Intestinal Fibrosis by Inhibiting Reactive Oxygen Species-Dependent TGF-β1/SMADs Pathway. Dig. Dis. Sci. 2018, 63, 366–380. [Google Scholar] [CrossRef] [PubMed]
- Osburn, W.O.; Kensler, T.W. Nrf2 signaling: An adaptive response pathway for protection against environmental toxic insults. Rev. Mutat. Res. 2008, 659, 31–39. [Google Scholar] [CrossRef]
- Cuadrado, A.; Manda, G.; Hassan, A.; Alcaraz, M.J.; Barbas, C.; Daiber, A.; Ghezzi, P.; Leόn, R.; Lόpez, M.G.; Oliva, B.; et al. Transcription Factor NRF2 as a Therapeutic Target for Chronic Diseases: A Systems Medicine Approach. Pharmacol. Rev. 2018, 70, 348–383. [Google Scholar] [CrossRef] [PubMed]
- Tonelli, C.; Chio, C.I.I.; Tuveson, D.A. Transcriptional Regulation by Nrf2. Antioxid. Redox Signal. 2018, 29, 1727–1745. [Google Scholar] [CrossRef]
- Cullinan, S.; Gordan, J.; Jin, J.; Harper, J.; Diehl, J. The keap1-BTB protein is an adaptor that bridges Nrf2 to a Cul3-based E3 ligase: Oxidative stress sensing by Cul3-Keap1 ligase. Mol. Cell. Biol. 2004, 24, 8477–8486. [Google Scholar] [CrossRef]
- Ahmed, S.M.; Luo, L.; Namani, A.; Wang, X.J.; Tang, X. Nrf2 signaling pathway: Pivotal roles in inflammation. Biochim. Biophys. Acta Mol. Bas. Dis. 2017, 1863, 585–597. [Google Scholar] [CrossRef] [PubMed]
- Moi, P.; Chan, K.; Asunis, I.; Cao, A.; Kan, Y.W. Isolation of NF-E2-related factor 2 (Nrf2), a NF-E2-like basic leucine zipper transcriptional activator that binds to the tandem NF-E2/AP1 repeat of the beta-globin locus control region. Proc. Natl. Acad. Sci. USA 1994, 91, 9926–9930. [Google Scholar] [CrossRef]
- Itho, K.; Chiba, T.; Takahashi, S.; Ishii, T.; Igarashi, K.; Katoh, Y.; Oyake, T.; Hayashi, N.; Satoh, K.; Hatayama, I.; et al. An Nrf2/small Maf heterodimer mediates the induction of phase II detoxifying enzyme genes through antioxidant response elements. Biochem. Biophys. Res. Commun. 1997, 236, 313–322. [Google Scholar] [CrossRef]
- Telakowski-Hopkins, C.A.; King, R.G.; Pickett, C.B. Glutathione S-transferase Ya subunit gene: Identification of regulatory elements required for basal level and inducible expression. Proc. Natl. Acad. Sci. USA 1988, 85, 1000–1004. [Google Scholar] [CrossRef] [PubMed]
- Rushmore, T.H.; Morton, M.R.; Pickett, C.B. The antioxidant responsive element. Activation by oxidative stress and identification of the DNA consensus sequence required for functional activity. J. Biol. Chem. 1991, 266, 11632–11639. [Google Scholar]
- Friling, R.S.; Bergelson, S.; Daniel, V. Two adjacent AP1-like binding sites from the electrophile-responsive element of the murine glutathione S-transferase Ya subunit gene. Proc. Natl. Acad. Sci. USA 1992, 89, 668–672. [Google Scholar] [CrossRef] [PubMed]
- Hayes, J.D.; Dinkova-Kostova, A.T. The Nrf2 regulatory network provides an interface between redox and intermediary metabolism. Trends Biochem. Sci. 2014, 39, 199–218. [Google Scholar] [CrossRef]
- Hirayama, A.; Yoh, K.; Nagase, S.; Ueda, A.; Itoh, K.; Morito, N.; Hirayama, K.; Takahashi, S.; Yamamoto, M.; Koyama, A. EPR imaging of reducing activity in Nrf2 transcriptional factor-deficient mice. Free Radic. Biol. Med. 2003, 34, 1236–1242. [Google Scholar] [CrossRef]
- Morito, N.; Yoh, K.; Itoh, K.; Hirayama, A.; Koyama, A.; Yamamoto, M.; Takahashi, S. Nrf2 regulates the sensitivity of death receptor signals by affecting intracellular glutathione levels. Oncogene 2003, 22, 9275–9281. [Google Scholar] [CrossRef] [PubMed]
- Cho, H.Y.; Jedlicka, A.E.; Reddy, S.P.; Kensler, T.W.; Yamamoto, M.; Zhang, L.Y.; Kleeberger, S.R. Role of NRF2 in protection against hyperoxic lung injury in mice. Am. J. Respir. Cell Mol. Biol. 2002, 26, 175–182. [Google Scholar] [CrossRef]
- Ma, Q. Role of nrf2 in oxidative stress and toxicity. Annu. Rev. Pharmacol. Toxicol. 2013, 53, 401–426. [Google Scholar] [CrossRef] [PubMed]
- Hybertson, B.M.; Gao, B.; Bose, S.K.; McCord, J.M. Oxidative stress in health and disease: The therapeutic potential of Nrf2 activation. Mol. Asp. Med. 2011, 32, 234–246. [Google Scholar] [CrossRef] [PubMed]
- Gorrini, C.; Harris, I.S.; Mark, T.W. Modulation of oxidative stress as an anticancer strategy. Nat. Rev. Drug Discov. 2013, 12, 931–947. [Google Scholar] [CrossRef]
- Tomashefski, J.F. Pulmonary pathology of acute respiratory distress syndrome. Clin. Chest Med. 2000, 21, 435–466. [Google Scholar] [CrossRef]
- Ware, L.B.; Matthay, M.A. The acute respiratory distress syndrome. N. Engl. J. Med. 2000, 342, 1334–1349. [Google Scholar] [CrossRef] [PubMed]
- Kataoka, K.; Handa, H.; Nishizawa, M. Induction of cellular anti-oxidative stress genes through heterodimeric transcription factor Nrf2/ small Maf by antirheumatic gold(I) compounds. J. Biol. Chem. 2001, 276, 34074–34081. [Google Scholar] [CrossRef] [PubMed]
- Abraham, E. Neutrophils and acute lung injury. Crit. Care Med. 2003, 31, S195–S199. [Google Scholar] [CrossRef] [PubMed]
- Arisawa, T.; Tahara, T.; Shibata, T.; Nagasaka, M.; Nakamura, M.; Kamiya, Y.; Fujita, H.; Hasegawa, S.; Takagi, T.; Wang, F.Y.; et al. The relationship between Helicobacter pylori infection and promoter polymorphism of the Nrf2 gene in chronic gastritis. Int. J. Mol. Med. 2007, 19, 143–148. [Google Scholar] [CrossRef] [PubMed]
- Levonen, A.L.; Inkala, M.; Heikura, S.; Jauhiainen, S.; Jykkanen, H.K.; Kansanen, E.; Maatta, K.; Romppanen, E.; Turenen, P.; Rutanen, J.; et al. Nrf2 gene transfer induces antioxidant enzymes and suppresses smooth muscle cell growth in vitro and reduces oxidative stress in rabbit aorta in vivo. Arterioscler. Thromb. Vasc. Biol. 2007, 27, 741–747. [Google Scholar] [CrossRef] [PubMed]
- Nathan, C. Points of control in inflammation. Nature 2002, 420, 846–852. [Google Scholar] [CrossRef] [PubMed]
- Itoh, K.; Mochizuki, M.; Ishii, Y.; Ishii, T.; Shibata, T.; Kawamoto, Y.; Kelly, W.; Sekizawa, K.; Uchida, K.; Yamamoto, M. Transcription Factor Nrf2 Regulates Inflammation by Mediating the Effect of 15-Deoxy-∆12,14-Prostaglandin J2. Mol. Cell. Biol. 2004, 24, 36–45. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.M.; Chan, K.; Kan, Y.W.; Johnson, J.A. Targeted disruption of Nrf2 causes regenerative immune-mediated hemolytic anemia. Proc. Natl. Acad. Sci. USA 2004, 101, 9751–9756. [Google Scholar] [CrossRef] [PubMed]
- Banning, A.; Brigelius-Flohe, R. NF-kappaB, Nrf2 and HO-1 interplay in redox regulated VCAM -1 expression. Antioxid. Redox Signal. 2005, 7, 889–899. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.H.; Sohn, D.H.; Jin, X.Y.; Kim, S.W.; Choi, S.C.; Seo, G.S. 2′,4′,6′Tris(methoxymethoxy) chalcone protects against trinitrobenzene sulfonic acid-induced colitis and blocks tumor necrosis factor-alpha-induced intestinal epithelial inflammation via heme oxygenase 1-dependent and independent pathways. Biochem. Pharmacol. 2007, 74, 870–880. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Young-Nam, C.; Young-Joon, S. A protective role of nuclear factor-erythroid 2-related factor-2 (Nrf2) in inflammatory disorders. Mutat. Res. Fundam. Mol. Mech. Mutagen. 2009, 690, 12–23. [Google Scholar] [CrossRef] [PubMed]
- Xu, W.; Hellerbrand, C.; Köhler, A.U.; Bugnon, P.; Kan, Y.W.; Werner, S.; Beyer, T.A. The Nrf2 transcription factor protects from toxin-induced liver injury and fibrosis. Lab. Investig. 2008, 88, 1068–1078. [Google Scholar] [CrossRef] [PubMed]
- Sriram, N.; Kalayarasan, S.; Sudhandiran, G. Enhancement of antioxidant defense system by epigallocatechin-3-gallateduring bleomycin-induced experimental pulmonary fibrosis. Biol. Pharm. Bull. 2008, 31, 1306–1311. [Google Scholar] [CrossRef] [PubMed]
- Kikuchi, N.; Ishii, Y.; Morishima, Y.; Yageta, Y.; Haraguchi, N.; Itoh, K.; Yamamoto, M.; Hizawa, N. Nrf2 protects against pulmonary fibrosis by regulating the lung oxidant level and Th1/Th2 balance. Respir. Res. 2010, 11, 31. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Zou, L.; Li, L.; Wu, T. The Protective Effect of Glycyrrhetinic Acid on Carbon Tetrachloride-Induced Chronic Liver Fibrosis in Mice via Upregulation of Nrf2. PLoS ONE 2013, 8, e53662. [Google Scholar] [CrossRef] [PubMed]
- Prestigiacomo, V.; Suter-Dick, L. Nrf2 protects stellate cells from Smad-dependent cell activation. PLoS ONE 2018, 13, e0201044. [Google Scholar] [CrossRef] [PubMed]
- Zhang, G.; Kang, Y.; Zhou, C.; Cui, R.; Jia, M.; Hu, S.; Ji, X.; Yuan, J.; Cui, H.; Shi, G. Amelioratory Effects of Testosterone Propionate on Age-related Renal Fibrosis via Suppression of TGF-β1/Smad Signaling and Activation of Nrf2ARE Signaling. Sci. Rep. 2018, 8, 10726. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.; Rong, Y.; Sun, D.; Li, W.; Chen, H.; Cao, B.; Wang, T. Costunolide inhibits pulmonary fibrosis via regulating NF-kB and TGF-β1/Smad2/Nrf2-NOX4 signaling pathways. Biochem. Biophys. Res. Commun. 2019, 510, 329–333. [Google Scholar] [CrossRef] [PubMed]
- Chen, R.R.; Fan, X.H.; Chen, G.; Zeng, G.W.; Xue, Y.G.; Liu, X.T.; Wang, C.Y. Irisin attenuates angiotensin II-induced cardiac fibrosis via Nrf2 mediated inhibition of ROS/ TGFβ1/Smad2/3 signaling axis. Chem. Biol. Interact. 2019, 302, 11–21. [Google Scholar] [CrossRef] [PubMed]
- Lee, B.S.; Kalluri, R. Mechanistic connection between inflammation and fibrosis. Kidney Int. 2010, 119, S22–S26. [Google Scholar] [CrossRef] [PubMed]
- Latella, G.; Vetuschi, A.; Sferra, R.; Speca, S.; Gaudio, E. Localization of ανβ6 integrin-TGF-β1/Smad3, mTOR and PPARγ in experimental colorectal fibrosis. Eur. J. Histochem. 2013, 57, e40. [Google Scholar] [CrossRef] [PubMed]
- Sferra, R.; Pompili, S.; Ventura, L.; Dubuquoy, C.; Speca, S.; Gaudio, E.; Latella, G.; Vetuschi, A. Interaction between sphingosine kinase/sphingosine 1 phosphate and transforming growth factor-β/Smads pathways in experimental intestinal fibrosis. An in vivo immunohistochemical study. Eur. J. Histochem. 2018, 31, 62. [Google Scholar] [CrossRef] [PubMed]
- Khor, T.O.O.; Huang, M.T.; Kwon, H.K.; Chan, J.Y.; Reddy, B.S.; Kong, A.N. Nrf2-Deficient Mice Have an Increased Susceptibility to Dextran Sulfate Sodium-Induced Colitis. Cancer Res. 2006, 66, 11580–11584. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.S.; An, J.M.; Kang, E.A.; Han, Y.M.; Kim, Y.S.; Lee, H.J.; Kim, K.J.; Surh, Y.J.; Hahm, K.B. Host nuclear factor erythroid 2-related factor-2 defense system determines the outcome of dextran sulfate sodium-induced colitis in mice. J. Physiol Pharmacol. 2018, 69, 755–767. [Google Scholar] [CrossRef]
- Li, J.; Wang, H.; Zheng, Z.; Luo, L.; Wang, P.; Liu, K.; Namani, A.; Jiang, Z.; Wang, X.J.; Tang, X. Mkp-1 cross-talks with Nrf2/Ho-1 pathway protecting against intestinal inflammation. Free Radic. Biol. Med. 2018, 124, 541–549. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wang, H.; Qian, C.; Tang, J.; Zhou, W.; Liu, X.; You, Q.; Hu, R. 3-(2-Oxo-2-phenylethylidene)-2,3,6,7-tetrahydro-1H-pyrazino [2,1-a] isoquinolin-4 (11bH)-one (compound 1), a novel potent Nrf2/ARE inducer, protects against DSS-induced colitis via inhibiting NLRP3 inflammasome. Biochem. Pharmacol. 2016, 101, 71–86. [Google Scholar] [CrossRef] [PubMed]
- Poss, K.D.; Tonegawa, S. Reduced stress defense in heme oxygenase 1-deficient cells. Proc. Natl. Acad. Sci. USA 1996, 20, 10925–10930. [Google Scholar] [CrossRef] [PubMed]
- Willis, D.; Moore, A.R.; Frederick, R.; Willoughby, D.A. Heme oxygenase: A novel target for the modulation of the inflammatory response. Nat. Med. 1996, 87, 87–90. [Google Scholar] [CrossRef]
- Laniado-Schwartzman, M.; Abraham, N.G.; Conners, M.; Dunn, M.W.; Levere, R.D. Heme oxygenase induction with attenuation of experimentally induced corneal inflammation. Biochem. Pharmacol. 1997, 53, 1069–1075. [Google Scholar] [CrossRef]
- Otterbein, L.E.; Bach, F.H.; Alam, J.; Soares, M.; Lu, H.T.; Wysk, M.; Davis, R.J.; Flavell, R.A.; Choi, A.M.K. Carbon monoxide has anti-inflammatory effects involving the mitogen-activated protein kinase pathway. Nat. Med. 2000, 6, 422–428. [Google Scholar] [CrossRef]
- Wang, W.P.; Guo, X.; Koo, M.V.L.; Wong, B.C.Y.; Lam, S.K.; Ye, Y.N.; Cho, C.H. Protective role of heme oxygenase-1 on trinitrobenzene sulfonic acid-induced colitis in rats. Am. J. Physiol. Gastrointest. Liver Physiol. 2001, 281, G586–G594. [Google Scholar] [CrossRef]
- Lu, M.C.; Ji, J.A.; Jiang, Y.L.; Chen, Y.Z.; Yuan, Z.W.; You, Q.D.; Jiang, Z.Y. An inhibitor of the Keap1-Nrf2 protein-protein interaction protects NCM460 colonic cells and alleviates experimental colitis. Sci. Rep. 2016, 6, 26585. [Google Scholar] [CrossRef] [PubMed]
- Yalniz, M.; Demirel, U.; Orhan, C.; Bahcecioglu, I.H.; Ozercan, I.H.; Aygun, C.; Tuzcu, M.; Sahin, K. Nadroparin Sodium Activates Nrf2/HO-1 Pathway in Acetic Acid-Induced Colitis in Rats. Inflammation 2012, 35, 1213–1221. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Zhou, W.; Zhang, X.; Lu, P.; Du, Q.; Tao, L.; Ding, Y.; Wang, Y.; Hu, R. Dimethyl fumarate ameliorates dextran sulphate sodium-induced murine experimental colitis by activating Nrf2 and suppressing NLRP3 inflammasome activation. Biochem. Pharmacol. 2016, 112, 37–49. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Shen, L.; Li, Y.; Li, Y.; Yu, S.; Wang, S. Hyperoside attenuates dextran sulfate sodium-induced colitis in mice possibly via activation of the Nrf2 signalling pathway. J. Inflamm. 2017, 14, 25. [Google Scholar] [CrossRef] [PubMed]
- Park, D.D.; Yum, H.W.; Zhong, Z.; Kim, S.H.; Kim, S.H.; Kim, D.H.; Kim, S.J.; Na, H.K.; Sato, A.; Miura, T.; et al. Perilla frutescens Extracts Protects against Dextran Sulphate Sodium-Induced Murine Colitis: NF-kB, STAT3, and Nrf2 as Putative Targets. Front. Pharmacol. 2017, 8, 1–14. [Google Scholar]
- Jing, W.; Safarpour, Y.; Zhang, T.; Guo, P.; Chen, G.; Wu, X.; Fu, Q.; Wang, Y. Berberine Upregulates P-Glycoprotein in Human Caco-2 Cells and in an Experimental Model of Colitis in the Rat via Activation of Nrf2-Dependent Mechanisms. J. Pharmacol. Exp. Ther. 2018, 366, 332–340. [Google Scholar] [CrossRef]
- Li, Y.; Shen, L.; Luo, H. Luteolin ameliorates dextran sulfate sodium-induced colitis in mice possibly through activation of the Nrf2 signaling pathway. Int. Immunopharmacol. 2016, 40, 24–31. [Google Scholar] [CrossRef]
- Kim, K.J.; Park, J.M.; Lee, J.S.; Kim, Y.S.; Kangwan, N.; Han, Y.M.; Kang, E.A.; An, J.M.; Park, Y.K.; Hahm, K.B. Oligonol prevented the relapse of dextran sulfate sodium-ulcerative colitis through enhancing NRF2-mediated antioxidative defense mechanism. J. Physiol. Pharmacol. 2018, 69, 359–371. [Google Scholar] [CrossRef]
- Zhou, Y.; Liu, H.; Song, J.; Cao, L.; Tang, L.; Qi, C. Sinomenine alleviates dextran sulfate sodium-induced colitis via the Nrf2/NQO-1 signaling pathway. Mol. Med. Rep. 2018, 18, 3691–3698. [Google Scholar] [CrossRef]
- Gao, Y.; Bai, D.; Zhao, Y.; Zhu, Q.; Zhou, Y.; Li, Z.; Lu, N. LL202 ameliorates colitis against oxidative stress of macrophage by activation of the Nrf2/HO-1 pathway. J. Cell. Physiol. 2019, 234, 10625–10639. [Google Scholar] [CrossRef]
- Saber, S.; Khalil, R.M.; Abdo, W.S.; Nassif, D.; El-Ahwany, E. Olmesartan ameliorates chemically-induced ulcerative colitis in rats via modulating NFκB and Nrf-2/HO-1 signaling crosstalk. Toxicol. Appl. Pharmacol. 2019, 364, 120–132. [Google Scholar] [CrossRef] [PubMed]
- Cheon, H.J.; Nam, S.H.; Kim, J.K. Tussilagone, a major active component in Tussilago farfara, ameliorates inflammatory responses in dextran sulphate sodium-induced murine colitis. Chem. Biol. Interact. 2018, 294, 74–80. [Google Scholar] [CrossRef] [PubMed]
- Yan, M.; Vemu, B.; Veenstra, J.; Petiwala, S.M.; Johnson, J.J. Carnosol, a dietary diterpene from rosemary (Rosmarinus officinalis) activates Nrf2 leading to sestrin 2 induction in colon cells. Integr. Mol. Med. 2018, 5. [Google Scholar] [CrossRef] [PubMed]
- Yang, N.; Xia, Z.; Shao, N.; Li, B.; Xue, L.; Peng, Y.; Zhi, F.; Yang, Y. Carnosic acid prevents dextran sulfate sodium-induced acute colitis associated with the regulation of the Keap1/Nrf2 pathway. Sci. Rep. 2017, 7, 11036. [Google Scholar] [CrossRef] [PubMed]
- Sangaraju, R.; Nalban, N.; Alavala, S.; Rajendran, V.; Jerald, M.K.; Sistla, R. Protective effect of galangin against dextran sulfate sodium (DSS)-induced ulcerative colitis in Balb/c mice. Inflamm. Res. 2019, 68, 1–14. [Google Scholar] [CrossRef]
- Hwang, Y.J.; Nam, S.J.; Chun, W.; Kim, S.I.; Park, S.C.; Kang, C.D.; Lee, S.J. Anti-inflammatory effects of apocynin on dextran sulfate sodium-induced mouse colitis model. PLoS ONE 2019, 14, e0217642. [Google Scholar] [CrossRef]
- Liu, G.; Yu, L.; Fang, J.; Hu, C.A.; Yin, J.; Ni, H.; Ren, W.; Duraipandiyan, V.; Chen, S.; Al-Dhabi, N.A.; et al. Methionine restriction on oxidative stress and immune response in DSS-induced colitis mice. Oncotarget 2017, 8, 44511–44520. [Google Scholar] [CrossRef]
- Fan, F.Y.; Sang, L.X.; Jiang, M. Catechins and Their Therapeutic Benefits to Inflammatory Bowel Disease. Molecules 2017, 22, 484. [Google Scholar] [CrossRef]
- Liu, D.; Huo, X.; Gao, L.; Zhang, J.; Ni, H.; Cao, L. NF-κB and Nrf2 pathways contribute to the protective effect of Licochalcone A on dextran sulfate sodium-induced ulcerative colitis in mice. Biomed. Pharmacother. 2018, 102, 922–929. [Google Scholar] [CrossRef]
- Li, J.; Chen, H.; Wang, B.; Cai, C.; Yang, X.; Chai, Z.; Feng, W. ZnO nanoparticles act as supportive therapy in DSS-induced ulcerative colitis in mice by maintaining gut homeostasis and activating Nrf2 signaling. Sci. Rep. 2017, 7, 43126. [Google Scholar] [CrossRef]
- Ramos-Gomez, M.; Kwak, M.K.; Dolan, P.M.; Itho, K.; Yamamoto, M.; Talalay, P.; Kensler, T.W. Interactive effects of nrf2 genotype and oltipraz on benzo[a]pyrene-DNA adducts and tumor yield in mice. Carcinogenesis 2003, 24, 461–467. [Google Scholar] [CrossRef] [PubMed]
- Osburn, W.O.; Karim, B.; Dolan, P.M.; Liu, G.; Yamamoto, M.; Huso, D.L.; Kensler, T.W. Increased colonic inflammatory injury and formation of aberrant crypt foci in Nrf2-deficient mice upon dextran sulfate treatment. Int. J. Cancer 2007, 121, 1883–1891. [Google Scholar] [CrossRef] [PubMed]
- Fahey, J.W.; Haristoy, X.; Dolan, P.M.; Kensler, T.W.; Scholtus, I.; Stephenson, K.K.; Talalay, P.; Lozniewski, A. Sulforaphane inhibits extracellular, intracellular, and antibiotic-resistant strains of Helicobacter pylori and prevents benzo[a]pyrene-induced stomach tumors. Proc. Natl. Acad. Sci. USA 2002, 99, 7610–7615. [Google Scholar] [CrossRef] [PubMed]
- Xu, C.; Huang, M.T.; Shen, G.; Yuan, X.; Lin, W.; Khor, T.O.; Conney, A.H.; Kong, A.N. Inhibition of 7,12-dimethylbenz(a)anthracene-induced skin tumorigenesis in C57BL/6 mice by sulforaphane is mediated by nuclear factor E2-related factor 2. Cancer Res. 2006, 66, 8293–8296. [Google Scholar] [CrossRef] [PubMed]
- Iida, K.; Itoh, K.; Kumagai, Y.; Oyasu, R.; Hattori, K.; Kawai, K.; Shimazui, T.; Akaza, H.; Yamamoto, M. Nrf2 is essential for the chemopreventive efficacy of oltipraz against urinary bladder carcinogenesis. Cancer Res. 2004, 64, 6424–6431. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.R.; Oh, J.E.; Kim, M.S.; Kang, M.R.; Park, S.W.; Han, J.Y.; Eom, H.S.; Yoo, N.J.; Lee, S.H. Oncogenic NRF2 mutations in squamous cell carcinomas of oesophagus and skin. J. Pathol. 2010, 220, 446–451. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.; Venkannagari, S.; Oh, K.H.; Zhang, Y.Q.; Rohde, J.M.; Liu, M.J.; Wang, A.; Xu, X.; Shahane, S.A.; Xia, M.; et al. Small molecule inhibitor of NRF2 selectively intervenes therapeutic resistance in KEAP1-deficient NSCLC tumors. ACS Chem. Biol. 2016, 11, 3214–3225. [Google Scholar] [CrossRef]
- Mitsuishi, Y.; Taguchi, K.; Kawatani, Y.; Shibata, T.; Nukiwa, T.; Aburatani, H.; Yamamoto, M.; Motohashi, H. Nrf2 redirects glucose and glutamine into anabolic pathways in metabolic reprogramming. Cancer Cell 2012, 22, 66–79. [Google Scholar] [CrossRef]
- Satoh, H.; Moriguchi, T.; Takai, J.; Ebina, M.; Yamamoto, M. Nrf2 prevents initiation but accelerates progression through the Kras signaling pathway during lung carcinogenesis. Cancer Res. 2013, 73, 4158–4168. [Google Scholar] [CrossRef]
- Satoh, H.; Moriguchi, T.; Saigusa, D.; Baird, L.; Yu, L.; Rokutan, H.; Igarashi, K.; Ebina, M.; Shibata, T.; Yamamoto, M. NRF2 Intensifies Host Defense Systems to Prevent Lung Carcinogenesis, but After Tumor Initiation Accelerates Malignant Cell Growth. Cancer Res. 2016, 76, 3088–3096. [Google Scholar] [CrossRef]
- Shim, G.S.; Manandhar, S.; Shin, D.H.; Kim, T.H.; Kwak, M.K. Acquisition of doxorubicin resistance in ovarian carcinoma cells accompanies activation of the NRF2 pathway. Free Radic. Biol. Med. 2009, 47, 1619–1631. [Google Scholar] [CrossRef] [PubMed]
- Jaramillo, M.C.; Zhang, D.D. The emerging role of the Nrf2–Keap1 signaling pathway in cancer. Genes Dev. 2013, 27, 2179–2191. [Google Scholar] [CrossRef] [PubMed]
- Ballinger, A.B.; Anggiansah, C. Colorectal cancer. BMJ 2007, 335, 715–718. [Google Scholar] [CrossRef] [PubMed]
- Siegel, R.; Naishadham, D.; Jemal, A. Cancer statistics. CA Cancer J. Clin. 2013, 63, 11–30. [Google Scholar] [CrossRef] [PubMed]
- Arnold, M.; Sierra, M.S.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Brai, F. Global patterns and trends in colorectal cancer incidence and mortality. Gut 2016, 66, 683–691. [Google Scholar] [CrossRef] [PubMed]
- Ferlay, J.; Soerjomataram, I.; Dikshit, R.; Eser, S.; Mathers, C.; Rebelo, M.; Parkin, D.M.; Forman, D.; Bray, F. Cancer incidence and mortality worldwide: Sources, methods and major patterns in GLOBOCAN 2012. Int. J. Cancer 2015, 136, E359–E386. [Google Scholar] [CrossRef]
- Rubenstein, J.H.; Enns, R.; Heidelbaugh, J.; Barkun, A. Clinical Guidelines Committee. American Gastroenterological Association Institute Guideline on the Diagnosis and Management of Lynch Syndrome. Gastroenterology 2015, 149, 777–782. [Google Scholar] [CrossRef]
- Cannon, J. Colorectal neoplasia and inflammatory Bowel disease. Surg. Clin. 2015, 95, 1261–1269. [Google Scholar] [CrossRef]
- Sasso, A.; Latella, G. Role of Heme Iron in the Association between Red Meat Consumption and Colorectal Cancer. Nutr. Cancer 2018, 70, 1173–1183. [Google Scholar] [CrossRef]
- Sasso, A.; Latella, G. Dietary components that counteract the increased risk of colorectal cancer related to red meat consumption. Int. J. Food Sci. Nutr. 2018, 69, 536–548. [Google Scholar] [CrossRef]
- Triantafillidis, J.K.; Nasioulas, G.; Kosmidis, P.A. Colorectal cancer and inflammatory bowel disease: Epidemiology, risk factors, mechanisms of carcinogenesis and prevention strategies. Anticancer Res. 2009, 29, 2727–2737. [Google Scholar] [PubMed]
- Ullman, T.A.; Itzkowitz, S.H. Intestinal inflammation and cancer. Gastroenterology 2011, 140, 1807–1816. [Google Scholar] [CrossRef] [PubMed]
- Azer, S.A. Overview of molecular pathways in inflammatory bowel disease associated with colorectal cancer development. Eur. J. Gastroenterol. Hepatol. 2013, 25, 271–281. [Google Scholar] [CrossRef] [PubMed]
- Ekbom, A.; Helmick, C.; Zack, M.; Adami, H.O. Ulcerative colitis and colorectal cancer. A population-based study. N. Engl. J. Med. 1990, 323, 1228–1233. [Google Scholar] [CrossRef] [PubMed]
- Choi, P.M.; Zelig, M.P. Similarity of colorectal cancer in Crohn’s disease and ulcerative colitis: Implications for carcinogenesis and prevention. Gut 1994, 35, 950–954. [Google Scholar] [CrossRef] [PubMed]
- Eaden, J.A.; Abrams, K.R.; Mayberry, J.F. The risk of colorectal cancer in ulcerative colitis: A meta-analysis. Gut 2001, 48, 526–535. [Google Scholar] [CrossRef] [PubMed]
- Friedman, S.; Rubin, P.H.; Bodian, C.; Harpaz, N.; Present, D.H. Screening and surveillance colonoscopy in chronic Crohn’s colitis: Results of a surveillance program spanning 25 years. Gastroenterology 2001, 120, 820–826. [Google Scholar] [CrossRef]
- Itzkowitz, S.H.; Yio, X. Inflammation and cancer IV. Colorectal cancer in inflammatory bowel disease: The role of inflammation. Am. J. Physiol. Gastrointest. Liver Physiol. 2004, 287, G7–G17. [Google Scholar] [CrossRef]
- Jess, T.; Loftus, E.V.; Velayos, F.S.; Harmsen, W.S.; Zinsmeister, A.R.; Smyrk, T.C.; Schleck, C.D.; Tremaine, W.J.; Melton, L.J.; Munkholm, P.; et al. Risk of intestinal cancer in inflammatory bowel disease: A population-based study from Olmsted County, Minnesota. Gastroenterology 2006, 130, 1039–1046. [Google Scholar] [CrossRef]
- Rachmilewitz, D.; Stamler, J.S.; Bachwich, D.; Karmeli, F.; Ackerman, Z.; Podolsky, D.K. Enhanced colonic nitric oxide generation and nitric oxide synthase activity in ulcerative colitis and Crohn’s disease. Gut 1995, 36, 718–723. [Google Scholar] [CrossRef]
- Kimura, H.; Hokari, R.; Miura, S.; Shigematsu, T.; Hirokawa, M.; Akiba, Y.; Kurose, I.; Higuchi, H.; Fujimori, H.; Tsuzuki, Y.; et al. Increased expression of an inducible isoform of nitric oxide synthase and the formation of peroxynitrite in colonic mucosa of patients with active ulcerative colitis. Gut 1998, 42, 180–187. [Google Scholar] [CrossRef] [PubMed]
- Hussain, S.P.; Amstad, P.; Raja, K.; Ambs, S.; Nagashima, M.; Bennett, W.P.; Shields, P.G.; Ham, A.J.; Swenberg, J.A.; Marrogi, A.J.; et al. Increased p53 mutation load in noncancerous colon tissue from ulcerative colitis: A cancer-prone chronic inflammatory disease. Cancer Res. 2000, 60, 3333–3337. [Google Scholar] [PubMed]
- Sadeghi, M.R.; Jeddi, F.; Soozangar, N.; Somi, M.H.; Samadi, N. The role of Nrf2-Keap1 axis in colorectal cancer, progression, and chemoresistance. Tumor Biol. 2017, 39, 1010428317705510. [Google Scholar] [CrossRef] [PubMed]
- Song, C.H.; Kim, N.; Lee, S.M.; Nam, R.H.; Choi, S.I.; Kang, S.R.; Shin, E.; Lee, D.H.; Lee, H.N.; Surh, Y.J. Effects of 17β-estradiol on colorectal cancer development after azoxymethane/dextran sulfate sodium treatment of ovariectomized mice. Biochem. Pharmacol. 2019, 164, 139–151. [Google Scholar] [CrossRef] [PubMed]
- Spitz, M.R.; Durphorne, C.M.; Detry, M.A.; Pillow, P.C.; Amos, C.I.; Lei, L.; de Andrade, M.; Gu, X.; Hong, W.K.; Wu, X. Dietary intake of isothiocyanates: Evidence of joint effect with glutathione S-transferase polymorphisms in lung cancer risk. Cancer Epidemiol. Biomark. Prev. 2000, 9, 1017–1020. [Google Scholar]
- Joseph, M.A.; Moysich, K.B.; Freudenheim, J.L.; Shields, P.G.; Bowman, E.D.; Zhang, Y.; Marshall, J.R.; Ambrosone, C.B. Cruciferous vegetables, genetic polymorphisms in glutathione S-transferases M1 and T1, and prostate cancer risk. Nutr. Cancer 2004, 50, 206–213. [Google Scholar] [CrossRef] [PubMed]
- Arisawa, T.; Tahara, T.; Shibata, T.; Nagasaka, M.; Nakamura, M.; Kamiya, Y.; Fujita, H.; Yoshioka, D.; Okubo, M.; Sakata, M.; et al. Nrf2 gene promoter polymorphism is associated with ulcerative colitis in a Japanese population. Hepatogastroenterology 2008, 55, 394–397. [Google Scholar]
- Marzec, J.M.; Christie, J.D.; Reddy, S.P.; Jedlicka, A.E.; Vuong, H.; Lanken, P.N.; Aplenc, R.; Yamamoto, T.; Yamamoto, M.; Cho, H.Y.; et al. Functional polymorphisms in the transcription factor NRF2 in humans increase the risk of acute lung injury. FASEB J. 2007, 21, 2237–2246. [Google Scholar] [CrossRef]
- Yokoo, Y.; Kijima, A.; Ishii, Y.; Takasu, S.; Tsuchiya, T.; Umemura, T. Effects of Nrf2 silencing on oxidative stress-associated intestinal carcinogenesis in mice. Cancer Medicine. 2016, 5, 1228–1238. [Google Scholar] [CrossRef]
- Jang, H.J.; Hong, E.M.; Kim, M.; Kim, J.H.; Jang, J.; Park, S.W.; Byun, H.W.; Koh, D.H.; Choi, M.H.; Kae, S.H.; et al. Simvastatin induces heme oxygenase-1 via NF-E2-related factor 2 (Nrf2) activation through ERK and PI3K/Akt pathway in colon cancer. Oncotarget 2016, 7, 46219–46229. [Google Scholar] [CrossRef]
- Kou, X.; Kirberger, M.; Yang, Y.; Chen, N. Natural products for cancer prevention associated with Nrf2-ARE pathway. Food Sci. Hum. Wellness 2013, 2, 22–28. [Google Scholar] [CrossRef]
- Trivedi, P.P.; Jena, G.B.; Tikoo, K.B.; Kumar, V. Melatonin modulated autophagy and Nrf2 signaling pathways in mice with colitis-associated colon carcinogenesis. Mol. Carcinog. 2016, 55, 255–267. [Google Scholar] [CrossRef] [PubMed]
- Zuo, Q.; Wu, R.; Xiao, X.; Yang, C.; Yang, Y.; Wang, C.; Lin, L.; Kong, A.N. The dietary flavone luteolin epigenetically activates the Nrf2 pathway and blocks cell transformation in human colorectal cancer HCT116 cells. J. Cell. Biochem. 2018, 119, 9573–9582. [Google Scholar] [CrossRef] [PubMed]
- Kensler, T.W.; Chen, J.G.; Egner, P.A.; Fahey, J.W.; Jacobson, L.P.; Stephenson, K.K.; Ye, L.; Coady, J.L.; Wang, J.B.; Wu, Y.; et al. Effects of glucosinolate-rich broccoli sprouts on urinary levels of aflatoxin-DNA adducts and phenanthrene tetraols in a randomized clinical trial in He Zuo township, Qidong, People’s Republic of China. Cancer Epidemiol. Biomark. Prev. 2005, 4, 2605–2613. [Google Scholar] [CrossRef] [PubMed]
- Seow, A.; Yuan, J.M.; Sun, C.L.; Van Der Berg, D.; Lee, H.P.; Yu, M.C. Dietary isothiocyanates, glutathione S-transferase polymorphisms and colorectal cancer risk in the Singapore Chinese Health Study. Carcinogenesis 2002, 23, 2055–2061. [Google Scholar] [CrossRef] [PubMed]
- Ambrosone, C.B.; McCann, S.E.; Freudenheim, J.L.; Marshall, J.R.; Zhang, Y.; Shields, P.G. Breast cancer risk in premenopausal women is inversely associated with consumption of broccoli, a source of isothiocyanates, but is not modified by GST genotype. J. Nutr. 2004, 134, 1134–1138. [Google Scholar] [CrossRef] [PubMed]
- Rao, C.V.; Rivenson, A.; Zang, E.; Steele, V.; Kelloff, G.; Reddy, B.S. Inhibition of 2-amino-1-methyl-6-phenylimidazo [4,5]-pyridine-induced lymphoma formation by oltipraz. Cancer Res. 1996, 56, 3395–3398. [Google Scholar]
- Moon, R.; Rao, K.; Detrisac, C.; Kelloff, G.; Steele, V.; Doody, L. Chemoprevention of respiratory-tract neoplasia in the hamster by oltipraz, alone and in combination. Int. J. Oncol. 1994, 4, 661–667. [Google Scholar] [CrossRef]
- Nishikawa, A.; Tanakamura, Z.; Furukawa, F.; Lee, I.S.; Kasahara, K.; Ikezaki, S.; Takahashi, M. Chemopreventive activity of oltipraz against induction of glandular stomach carcinogenesis in rats by N-methyl-N-nitro-N-nitrosoguanidine. Carcinogenesis 1998, 19, 365–368. [Google Scholar] [CrossRef]
- Szarka, C.E.; Yao, K.S.; Pfeiffer, G.R.; Balshem, A.M.; Litwin, S.; Frucht, H.; Goosenberg, E.B.; Engstrom, P.F.; Clapper, M.L.; O’Dwyer, P.J. Chronic dosing of oltipraz in people at increased risk for colorectal cancer. Cancer Detect. Prev. 2001, 24, 352–361. [Google Scholar]
- Roebuck, B.D.; Liu, X.L.; Rogers, A.E.; Groopmen, J.D.; Kensler, T.W. Protection against aflatoxin B1-induced hepatocarcinogenesis in F344 rats by 5-(2-pyrazynil)-4-methyl-1,2-dithiole-3thione (oltipraz): Predictive role of short-term moleculary dosimetry. Cancer Res. 1991, 51, 5501–5506. [Google Scholar] [PubMed]
- Kang, K.A.; Piao, M.J.; Hyun, Y.J.; Zhen, A.X.; Cho, S.J.; Ahn, M.J.; Yi, J.M.; Hyun, J.W. Luteolin promotes apoptotic cell death via upregulation of Nrf2 expression by DNA demethylase and the interaction of Nrf2 with p53 in human colon cancer cells. Exp. Mol. Med. 2019, 51, 40. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Saud, S.M.; Zhang, X.; Li, W.; Hua, B. Protective effect of Shaoyao Decoction against colorectal cancer via the Keap1-Nrf2-ARE signaling pathway. J. Ethnopharmacol. 2019, 241, 111981. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.N.; Park, G.H.; Park, S.B.; Kim, J.D.; Eo, H.J.; Son, H.J.; Song, J.H.; Jeong, J.B. Sageretia thea Inhibits Inflammation through Suppression of NF-κ B and MAPK and Activation of Nrf2/HO-1 Signaling Pathways in RAW264.7 Cells. Am. J. Chin. Med. 2019, 47, 385–403. [Google Scholar] [CrossRef] [PubMed]
- Rao, C.V.; Rivenson, A.; Katiwalla, M.; Kelloff, G.J.; Reddy, B.S. Chemopreventive effect of oltipraz during different stages of experimental colon carcinogenesis induced by azoxymethane in male F344 rats. Cancer Res. 1993, 53, 2502–2506. [Google Scholar]
- Clapper, M.L.; Wood, M.; Leahy, K.; Lang, D.; Miknyoczki, S.; Ruggeri, B.A. Chemopreventive activity of Oltipraz against N-nitrosobis (2-oxopropyl) amine (BOP)-induced ductal pancreatic carcinoma development and effects on survival of Syrian golden hamsters. Carcinogenesis 1995, 16, 2159–2165. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.; Gao, P.; Steele, V.E. The chemopreventive efficacy of inhaled oltipraz particulates in the B[a]P-induced A/J mouse lung adenoma model. Carcinogenesis 2006, 27, 1721–1727. [Google Scholar] [CrossRef][Green Version]
- Kensler, T.W.; Egner, P.A.; Dolan, P.M.; Groopman, J.D.; Roebuck, B.D. Mechanism of protection against aflatoxin tumorigenicity in rats fed 5-(2-pyrazinyl)-4-methyl-1,2-dithiol-3-thione (oltipraz) and related 1,2-dithiol-3-thiones and 1,2-dithiol-3-ones. Cancer Res. 1987, 47, 4271–4277. [Google Scholar]
- Kensler, T.W.; Curphey, T.J.; Maxiutenko, Y.; Roebuck, B.D. Chemoprotection by organosulfur inducers of phase 2 enzymes: Dithiolethiones and dithiins. Drug Metabol. Drug Interact. 2000, 17, 3–22. [Google Scholar] [CrossRef]
- Hu, T.; Hua, T.; Yu, Y.; Yub, S.; Guoa, H.; Hana, L.; Wanga, W.; Tiana, T.; Haoc, Y.; Liud, Z.; et al. Clinicopathologic significance of CXCR4 and Nrf2 in colorectal cancer. J. Biomed. Res. 2013, 27, 283–290. [Google Scholar]
- Zhao, X.Q.; Zhang, Y.F.; Xia, Y.F.; Zhou, Z.M.; Cao, Y.Q. Promoter demethylation of nuclear factor-erythroid 2-related factor 2 gene in drug-resistant colon cancer cells. Oncol. Lett. 2015, 10, 1287–1292. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; He, L.J.; Ye, H.Z.; Liu, D.F.; Zhu, Y.B.; Miao, D.D.; Zhang, S.P.; Chen, Y.Y.; Jia, Y.W.; Shen, J.; et al. Nrf2 is a key factor in the reversal effect of curcumin on multidrug resistance in the HCT-8/5-Fu human colorectal cancer cell line. Mol. Med. Rep. 2018, 18, 5409–5416. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Zhang, Z.Y.; Du, H.; Li, S.Z.; Tu, R.; Jia, Y.F.; Zheng, Z.; Song, X.M.; Du, R.L.; Zhang, X.D. DUB3 deubiquitinates and stabilizes NRF2 in chemotherapy resistance of colorectal cancer. Cell Death Differ. 2019. [Google Scholar] [CrossRef] [PubMed]
- Lin, P.O.; Chang, J.T.; Wu, D.W.; Huang, C.C.; Lee, H. Cytoplasmatic localization of Nrf2 promotes colorectal cancer with more aggressive tumors via upregulation of PSMD4. Free Radic. Biol. Med. 2016, 95, 121–132. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Y.M.; Lin, P.L.; Wu, D.W.; Wang, L.; Huang, C.C.; Lee, H. PSMD4 is a novel therapeutic target in chemoresistant colorectal cancer activated by cytoplasmic localization of Nrf2. Oncotarget 2018, 9, 26342–26352. [Google Scholar] [CrossRef] [PubMed]
- Tajmohammadi, I.; Mohammadian, J.; Sabzichi, M.; Mahmuodi, S.; Ramezani, M.; Aghajani, M.; Ramezani, F. Identification of Nrf2/STAT3 axis in induction of apoptosis through sub-G 1 cell cycle arrest mechanism in HT-29 colon cancer cells. J. Cell. Biochem. 2019, 120, 14035–14043. [Google Scholar] [CrossRef] [PubMed]
- Cernigliaro, C.; D’Anneo, A.; Carlisi, D.; Giuliano, M.; Marino Gammazza, A.; Barone, R.; Longhitano, L.; Cappello, F.; Emanuele, S.; Distefano, A.; et al. Ethanol-Mediated Stress Promotes Autophagic Survival and Aggressiveness of Colon Cancer Cells via Activation of Nrf2/HO-1 Pathway. Cancers 2019, 11, 505. [Google Scholar] [CrossRef]
- Jeong, G.; Oh, J.; Kim, J.S. Glyceollins Modulate Tumor Development and Growth in a Mouse Xenograft Model of Human Colon Cancer in a p53-Dependent Manner. J. Med. Food 2019, 22, 521–528. [Google Scholar] [CrossRef] [PubMed]
- Taira, J.; Miyazato, H.; Ueda, K. Marine Peroxy Sesquiterpenoids Induce Apoptosis by Modulation of Nrf2-ARE Signaling in HCT116 Colon Cancer Cells. Mar. Drugs 2018, 23, 16. [Google Scholar] [CrossRef]
- Sadeghi, M.R.; Jeddi, F.; Soozanger, N.; Somi, M.H.; Shirmohamadi, M.; Khaze, V.; Samadi, N. Nrf2/P-glycoprotein axis is associated with clinicopathological characteristics in colorectal cancer. Biomed. Pharmacother. 2018, 104, 458–464. [Google Scholar] [CrossRef]
- Ballong, M.J.; Yun, H.; Sherwood, L.; Woods, A.K.; Lairson, L.L.; Schultz, P.G. A small molecule inhibits deregulated NRF2 transcriptional activity in cancer. ACS Chem. Biol. 2015, 10, 2193–2198. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Wang, H.; Chen, F.; Fu, J.; Xu, Y.; Hou, Y.; Kou, H.H.; Zhai, C.; Nelson, M.B.; Zhang, Q.; et al. An overview of chemical inhibitors of the Nrf2-ARE signaling pathway and their potential applications in cancer therapy. Free Radic. Biol. Med. 2016, 99, 544–556. [Google Scholar] [CrossRef] [PubMed]
- Yao, J.; Zhao, L.; Zhao, Q.; Sun, Y.; Zhang, Y.; Miao, H.; You, Q.; Hu, R.; Guo, Q.L. NF-Kappa B and Nrf2 signaling pathways contribute to Wogonin-mediated inhibition of inflammation-associated colorectal carcinogenesis. J. Cell Death Dis. 2014, 5, e1283. [Google Scholar] [CrossRef] [PubMed]
- Evans, J.P.; Winiarski, B.K.; Sutton, P.A.; Jones, R.P.; Ressel, L.; Duckworth, C.A.; Pritchard, D.M.; Lin, Z.X.; Vicky, F.L.; Tweedle, M.E.; et al. The Nrf2 inhibitor brusatol is a potent antitumour agent in an orthotopic mouse model of colorectal cancer. Oncotarget 2018, 9, 27104–27116. [Google Scholar] [CrossRef] [PubMed]
- Hendersen, D.; Frieson, D.; Zuber, J.; Solomon, S.S. Metformin has positive therapeutic effects in colon cancer and lung cancer. Am. J. Med. Sci. 2017, 354, 246–251. [Google Scholar] [CrossRef]
- Sena, P.; Mancini, S.; Benincasa, M.; Mariani, F.; Palumbo, C.; Roncucci, L. Metformin Induces Apoptosis and Alters Cellular Responses to Oxidative Stresses in Ht29 Colon Cancer Cells: Preliminary Findings. Int. J. Mol. Sci. 2018, 19, 1478. [Google Scholar] [CrossRef] [PubMed]
| References | Organ | Nrf2 Action |
|---|---|---|
| Xu et al., 2008 [51] | Liver | Nrf2-deficient mice showed a decreased expression of cytoprotective proteins together with reduced development and progression of liver fibrosis compared to wild-type mice. |
| Chen et al., 2013 [54] | Upregulation of Nrf2 induced by glycyrrhetinic acid prevented CCl4-induced liver fibrosis in mice. | |
| Prestigiacomo et al., 2018 [55] | Overexpression of the main markers of fibrosis (α-SMA, collagens, fibronectin) in Nrf2/Keap1 knockout mice compared to wild-type animals. The TGFβ-1/Smad pathway was able to mediate hepatic stellate cells activation and then fibrogenesis in mice with Nrf2 depletion. | |
| Kikuchi et al., 2010 [53] | Lung | An increase of oxidative stress markers associated with a higher degree of inflammation and fibrosis was found in Nrf2−/− KO mice compared to controls. |
| Liu et al., 2019 [57] | Costunolide suppressed the development of pulmonary fibrosis through its ability to inhibit NF-kB and to regulate TGF-β1/Smad2/Nrf2-NOX4 signaling. | |
| Zhang et al., 2018 [56] | Kidney | Testosterone propionate attenuated renal fibrosis age-related through TGF-β1/Smad deletion and parallel Nrf2/ARE activation in aged rats. |
| Chen et al., 2019 [58] | Hearth | Irisin was able to play an antifibrotic effect via Nrf2 through the inhibition of ROS/TGFβ1/Smad2-3 signaling. |
| References | Nrf2 protective effect |
|---|---|
| Khor et al., 2006 [62] | Nrf2−/− KO mice were more susceptible to colitis compared to WT mice demonstrating the significant role played by Nrf2 in intestinal inflammation. |
| Lee et al., 2018 [63] | COX-2−/− KO mice were protected to colitis progression, while Nrf2−/− KO mice showed severe signs of colitis demonstrating the protective effect of Nrf2 in intestinal inflammation. |
| Li et al., 2018 [64] | Mkp-1−/− mice were more sensitive to DSS insult. Mkp-1 correlated with Nrf2/HO-1 expression in intestinal colitis. |
| Wang et al., 2016 [65] | NLRP3 inhibition, concomitantly to Nrf2 activation, protected from DSS-induced colitis. |
| Wang et al., 2001 [70] | Mesoporphyrin, an HO-inhibitor, induced progression of colonic injuries in TNBS-induced colitis. |
| Lu et al., 2016 [71] | The inhibitor of Keap1/Nrf2 (CPUY192018) alleviated DSS-induced colitis in mice. |
| Yalniz et al., 2012 [72] | Nadroparin sodium exerted anti-inflammatory and antioxidative effects through Nrf2/HO-1/NF-kB signaling. |
| Liu et al., 2016 [73] | Dimethyl fumarate activated Nrf2 and inhibited NLRP3, attenuating signs of DSS-induced colitis. |
| Yang et al., 2017 [74] | Hyperoside, by increasing Nrf2, attenuated inflammation in DSS-induced colitis in mice. |
| Park et al., 2017 [75] | A natural compound, Perilla frutescens, increased Nrf2, decreased NF-kB, and improved features of DSS-induced colitis. |
| Jing et al., 2018 [76] | Berberine induced Nrf2 and P-gp both in vitro and in vivo, leading to an attenuation of colitis. |
| Li et al., 2016 [77] | Luteolin, through the activation of Nrf2, mitigates intestinal inflammation. |
| Kim et al., 2018 [78] | Oligonol activated Nrf2, exerting a protective effect in experimental colitis. |
| Zhou et al., 2018 [79] | Sinomenine induced Nrf2 and NQO-1 and ameliorated signs of colitis. |
| Gao et al., 2019 [80] | Synthetic flavonoid molecule LL202 showed a protective effect on colitis, stimulating Nrf2/HO-1 signaling. |
| Saber et al., 2019 [81] | Olmesartan blocking Angiotensin II receptor type 1 activated Nrf2 and mitigated colitis in acetic acid-induced colitis. |
| Cheon et al., 2018 [82] | Tussilagone inhibited NF-kB and induced Nrf2, reducing the main markers of colitis induced by DSS in mice. |
| Yan et al., 2018 [83] | Carnosol increased the expression of Nrf2 and its downstream genes leading to antioxidant effects. |
| Yang et al., 2017 [84] | Carnosic acid exerted a protective effect in DSS-induced colitis inducing overexpression of Nrf2. |
| Sangaraju et al., 2019 [85] | Galangin was able to increase HO-1 and to reduce pro-inflammatory cytokines expression in DSS-induced colitis. |
| Hwang et al., 2019 [86] | Apocynin activated the Nrf2/HO-1 pathway and decreased intestinal inflammation and fibrosis in DSS-induced colitis in mice. |
| Liu et al., 2017 [87] | Low methionine diet protected from the progression of colitis and fibrosis induced by DSS mice. |
| Fan et al., 2017 [88] | Catechins reduced ROS, increased Nrf2 levels, and modulate intestinal flora, exerting beneficial effects in colitis. |
| Liu et al., 2018 [89] | Licochalcone A, a molecule able to attenuate oxidative stress and inflammation by decreasing NF-kB and increasing Nrf2 expression. |
| Li et al., 2017 [90] | ZnONP activated Nrf2, suppressed ROS and pro-inflammatory cytokines, and preserved gut microbiota. |
| Guan et al., 2018 [21] | An agonist of Nrf2 (tBHQ) ameliorated markers of fibrosis in TNBS-induced chronic colitis. |
| Advantages of Nrf2 | ||
| References | Tumor Site | Nrf2 Action |
| Ramos-Gomez et al., 2003 [91] Fahey et al., 2002 [93] | Stomach | Benzo(a)pyrene-induced gastric neoplasia was reduced in Nrf2 knockout compared to wild-type mice. Sulforaphane blocked benzo(a)pyrene-evoked forestomach tumors in mice. This protection was abrogated in mice lacking the nrf2 gene. |
| Iida K et al., 2004 [95] | Bladder | Increased incidence of bladder tumors was found in Nrf2−/− knockout mice compared to wild-type animals following N-nitrosobutyl(4-hydroxybutyl)amine administration. |
| Xu et al., 2006 [94] | Skin | Higher incidence of skin tumors after 7,12-dimethylbenz(a)anthracene or 12-O-tetradecanoylphorbol-13-acetate administration was detected in Nrf2-deficient mice compared to wild-type mice. |
| Disadvantages of Nrf2 | ||
| References | Tumor Site | Nrf2 Action |
| Singh et al., 2006 [13] Shibata et al., 2008 [14] Zhang et al., 2010 [17] Kim et al., 2010 [96] Singh et al., 2016 [97] | Lung, gallbladder, esophagus, skin prostate, breast, head, neck, ovary, endometrium | Increased levels of Nrf2 expression leading to a progression of all these types of cancer and, consequently, to a poor prognosis. |
| Mitsuishi et al., 2012 [98] | Lung | High levels of Nrf2 accelerate cancer cell proliferation, inducing purine nucleotide and glutathione synthesis. Nrf2 directly activates metabolic genes under active PI3K-Akt signaling. |
| Satho et al., 2013 [99] Satho et al., 2016 [100] | Nrf2 prevents initiation but accelerates progression through the Kras signaling pathway during lung carcinogenesis. | |
| Shim et al., 2009 [101] | Ovary | Activation of the Nrf2 pathway can participate in the acquisition of doxorubicin resistance in ovarian carcinoma cells. |
| Protective Effects of Nrf2 | |
| References | Nrf2 Action |
| Khor et al., 2008 [12] | Nrf2−/− KO mice showed an increased susceptibility to colitis-associated colorectal cancer (CRC) compared to Nrf2+/+ WT mice. |
| Osburn et al., 2007 [92] | Nrf2-deficient mice upon DSS treatment showed an increased formation of colorectal aberrant crypt foci and cancer compared to wild-type mice. |
| Song et al., 2019 [124] | Estrogens exerted a protective effect in the development of colon cancer through estrogen receptor, and NF-kB and Nrf2 pathways. |
| Yokoo et al., 2016 [129] | Incidence of colorectal preneoplastic and neoplastic lesions was significantly higher in Nrf2-deficient mice. |
| Jang et al., 2016 [130] | Simvastatin was able to induce a protective effect inducing Nrf2 overexpression and cooperating with PI3K/Akt and Erk pathways. |
| Kou et al., 2013 [131] | EGCG induced Nrf2, exerting antioxidant, anti-inflammatory, and antineoplastic functions. |
| Trivedi et al., 2016 [132] | Melatonin activated Nrf2 and p62 and decreased autophagy. In colitis-associated colorectal carcinogenesis, melatonin reduced the progression of colorectal cancer. |
| Zuo et al., 2018 [133] | Luteolin is an epigenetic modulator of Nrf2 and suppressed carcinogenesis in colon cancer cells. |
| Kang et al., 2019 [142] | Luteolin increased Nrf2 expression, showed an interaction between Nrf2 and p53, and induced apoptosis. |
| Wang et al., 2019 [143] | SYD reduced oxidative stress and inflammation in an AOM/DSS mouse model, activating Keap1–Nrf2–ARE signaling and preventing colitis-associated CRC. |
| Kim et al., 2019 [144] | Sageretia thea induced the Nrf2/HO1 pathway and decreased viability of colorectal cancer cells. |
| Offensive Effects of Nrf2 | |
| References | Nrf2 Action |
| Hu et al., 2013 [150] | Nrf2 and CXCR4 were correlated with lymph node and distant metastasis in CRC patients. |
| Zhao et al., 2015 [151] | Nuclear translocation of Nrf2 and high expression of HO-1 and ROS were found in colorectal carcinoma cell line resistant to 5-Fu. |
| Zhang et al., 2019 [152] | Deubiquitination of Nrf2 by DUB3 stabilized Nrf2–Keap1 complex promoting chemoresistance in colorectal cancer cells. |
| Zhang et al., 2018 [153] | Curcumin corroborated 5-Fu effect in colon cancer reducing Nrf2 and Bcl2-Bax expression and inducing apoptosis. |
| Lin et al., 2016 [154] | Cytoplasmic Nrf2-positive tumors had a poor outcome, increasing tumor invasiveness by upregulation of PSMD4. |
| Cheng et al., 2018 [155] | Cytoplasmic Nrf2 conferred chemoresistance to 5-Fu and oxaliplatin, in both in vitro and in vivo studies. |
| Tajmohammadi et al., 2019 [156] | STAT3 inhibitor improved 5-Fu effect in colon cancer cells, decreasing Nrf2 and Bcl2. |
| Cernigliaro et al., 2019 [157] | Ethanol increased Nrf2 nuclear translocation and activated Nrf2/HO-1 signaling, favoring survival and aggressiveness of CRC cells. |
| Jeong et al., 2019 [158] | Glyceollins activated Nrf2 and HO-1, promoting tumor growth in a BALB/c xenograft. |
| Taira et al., 2018 [159] | Coral extract suppressed Nrf2 signaling, inducing apoptosis in human colon cancer cells. |
| Kensler et al., 2005 [134] | Nrf2 induced ABCB1 expression and consequent chemoresistance in CRC. |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pompili, S.; Sferra, R.; Gaudio, E.; Viscido, A.; Frieri, G.; Vetuschi, A.; Latella, G. Can Nrf2 Modulate the Development of Intestinal Fibrosis and Cancer in Inflammatory Bowel Disease? Int. J. Mol. Sci. 2019, 20, 4061. https://doi.org/10.3390/ijms20164061
Pompili S, Sferra R, Gaudio E, Viscido A, Frieri G, Vetuschi A, Latella G. Can Nrf2 Modulate the Development of Intestinal Fibrosis and Cancer in Inflammatory Bowel Disease? International Journal of Molecular Sciences. 2019; 20(16):4061. https://doi.org/10.3390/ijms20164061
Chicago/Turabian StylePompili, Simona, Roberta Sferra, Eugenio Gaudio, Angelo Viscido, Giuseppe Frieri, Antonella Vetuschi, and Giovanni Latella. 2019. "Can Nrf2 Modulate the Development of Intestinal Fibrosis and Cancer in Inflammatory Bowel Disease?" International Journal of Molecular Sciences 20, no. 16: 4061. https://doi.org/10.3390/ijms20164061
APA StylePompili, S., Sferra, R., Gaudio, E., Viscido, A., Frieri, G., Vetuschi, A., & Latella, G. (2019). Can Nrf2 Modulate the Development of Intestinal Fibrosis and Cancer in Inflammatory Bowel Disease? International Journal of Molecular Sciences, 20(16), 4061. https://doi.org/10.3390/ijms20164061






