Furosine Posed Toxic Effects on Primary Sertoli Cells through Regulating Cep55/NF-κB/PI3K/Akt/FOX01/TNF-α Pathway
Abstract
1. Introduction
2. Results
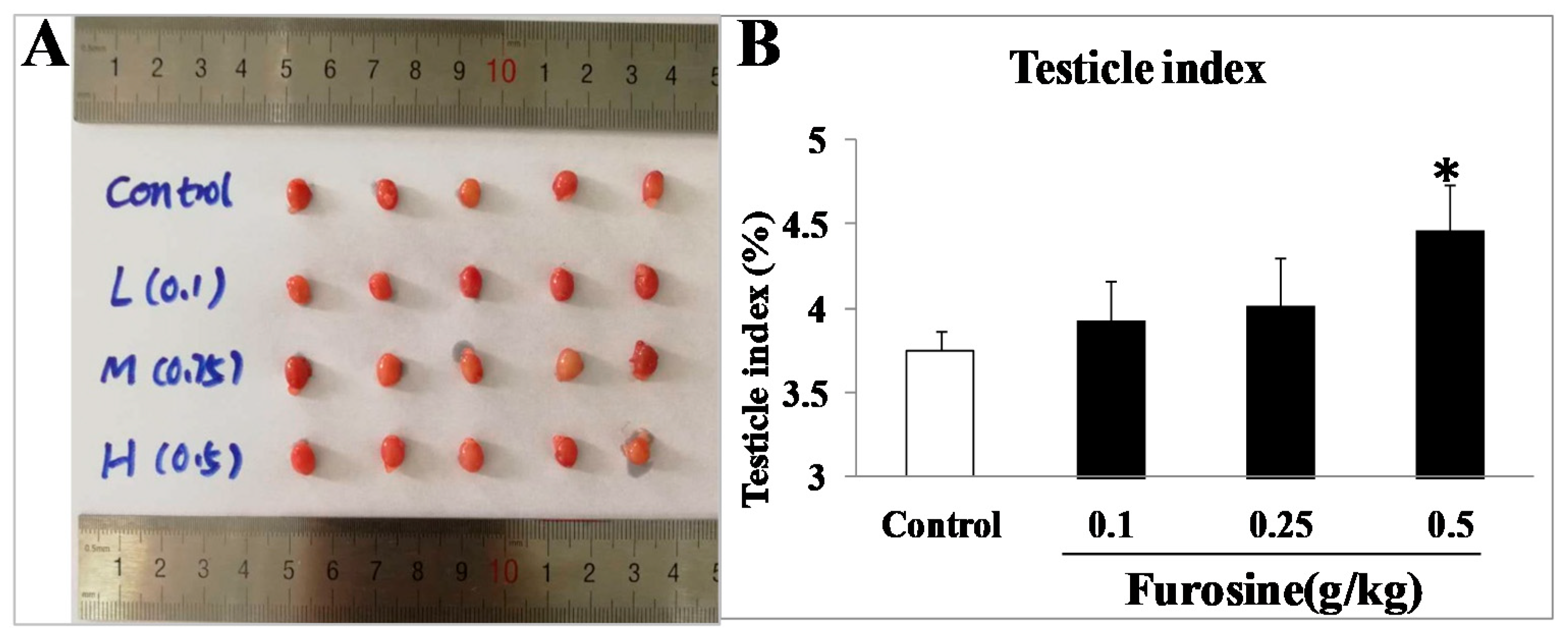
2.1. Furosine Affected Testicle Index of Male Mice.
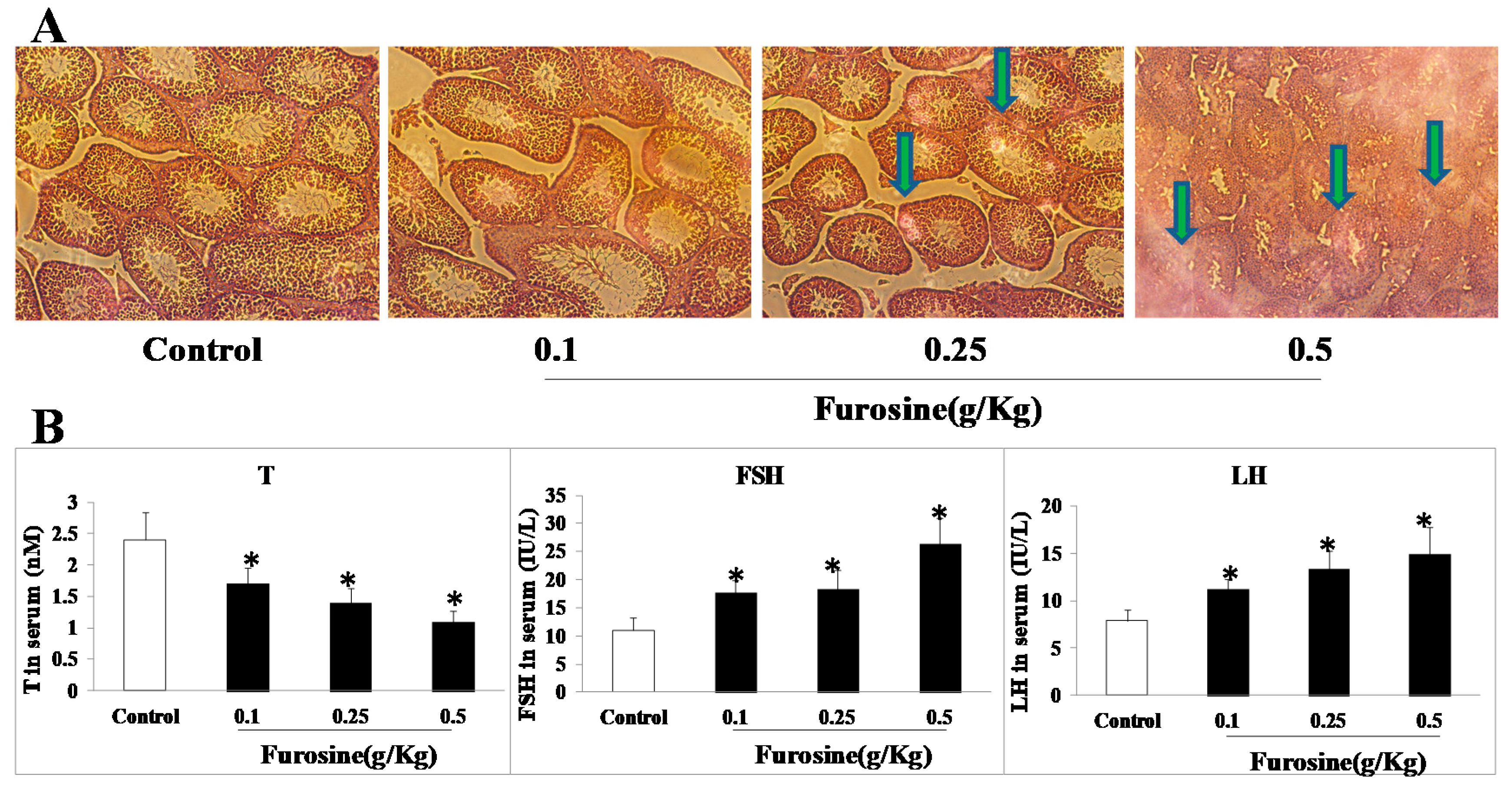
2.2. Furosine Affected Pathological Condition of Testicle Tissue and Reproductive Hormones Level in Serum
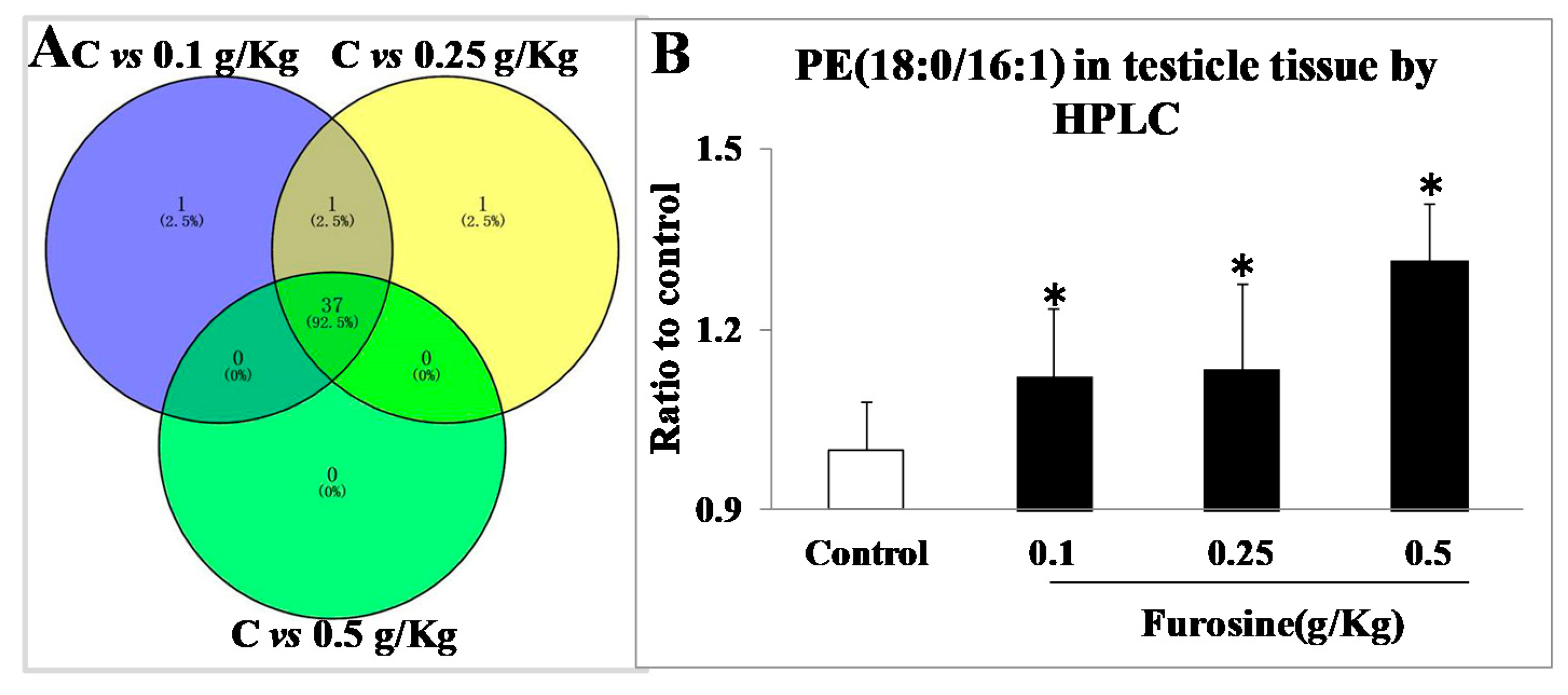
2.3. Special Metabolites Screening and Data Analysis
2.4. Seperation and Culturing of Sertoli Cells
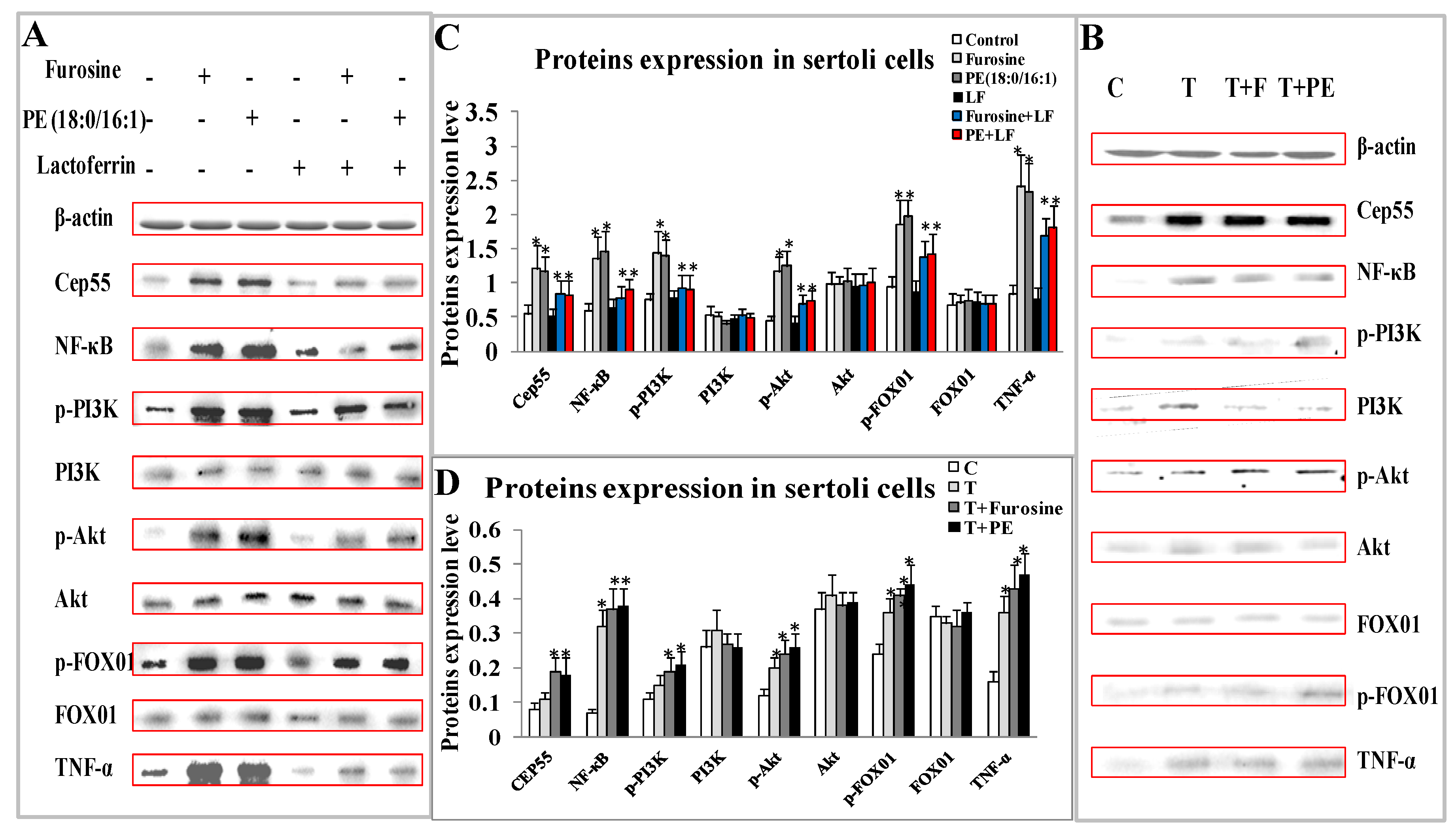
2.5. Expression of Cep55/NF-κB/PI3K/Akt/FOX01/TNF-α in Testicle Tissue
2.6. Expression of Cep55/NF-κB/PI3K/Akt/FOX01/TNF-α Affected by Furosine and Special Metabolite in Sertoli Cells
2.7. Sperm Quality Affected by 42-day Treatment of Furosine
3. Discussion
4. Materials and Methods
4.1. Chemicals
4.2. Animal Model Construction
4.3. Lipid Metabolism Detection and Data Analysis
4.4. Seperation and Culturing of Sertoli Cells
4.5. Cep55-related Pathway Detection By q-PCR and Western Blotting
4.6. Detection of Reproductive Hormone
4.7. Pathological Observation by HE Staining
4.8. Assessment of Sperm Parameters
4.9. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| PE | Phosphatidylethanolamine |
| Cep55 | centro-somal protein 55 |
| FOX01 | forkhead protein in rhabdomyosarcoma |
| NF-κB | nuclear factor-kappa B |
| T | Testosterone |
| FSH | follicle-stimulating hormone |
| LH | luteinizing hormone |
References
- Henle, T. Protein-bound advanced glycation endproducts (ages) as bioactive amino acid derivatives in foods. Amino Acids 2005, 29, 313–322. [Google Scholar] [CrossRef] [PubMed]
- Ledl, F.; Schleicher, E. New aspects of the maillard reaction in foods and in the human body. Angew. Chem. Int. Edit. 2010, 29, 565–594. [Google Scholar] [CrossRef]
- Ames, J.M. Control of the maillard reaction in food systems. Trends Food Sci. Technol. 1990, 1, 150–154. [Google Scholar] [CrossRef]
- Huang, M.; Zhang, X.; Karangwa, E. Comparation sensory characteristin, non-volatile compounds, volatile compounds and antioxidant activity of mrps by novel gradient temperature-elevating and traditional isothermal methods. J. Food Sci. Technol. 2015, 52, 858–866. [Google Scholar] [CrossRef] [PubMed]
- Villamiel, M.; Mddel, C.; Corzo, N.; Olano, A. Presence of furosine in honeys. J. Sci. Food Agric. 2001, 81, 790–793. [Google Scholar] [CrossRef]
- Rajchl, A.; Čížková, H.; Voldřich, M.; Jirušková, M.; Ševčík, R. Evaluation of shelf life and heat treatment of tomato products. Czech J. Food Sci. 2009, 27, 130–133. [Google Scholar] [CrossRef]
- Seiquer, I.; Díaz-Alguacil, J.; Delgado-Andrade, C.; López-Frías, M.; Muñoz, H.A.; Galdó, G.; Navarro, M.P. Diets rich in maillard reaction products affect protein digestibility in adolescent males aged 11–14 y. Am. J. Clin. Nutr. 2006, 83, 1082–1088. [Google Scholar] [CrossRef] [PubMed]
- Troise, A.D.; Fiore, A.; Wiltafsky, M.; Fogliano, V. Quantification of nε-(2-furoylmethyl)-l-lysine(furosine), nε-(carboxymethyl)-l-lysine(cml), nε-(carboxyethyl)-l-lysine(cel) and total lysine through stable isotope dilution assay and tandem mass spectrometry. Food Chem. 2015, 188, 357–364. [Google Scholar] [CrossRef] [PubMed]
- Li, H.Y.; Xing, L.; Wang, J.Q.; Zheng, N. Toxicology studies of furosine in vitro/in vivo and exploration of the related mechanism. Toxicol. Lett. 2018, 291, 101–111. [Google Scholar] [CrossRef] [PubMed]
- Erbersdobler, H.F.; Dehn, M.B. Formation of early Maillard products during UHT treatment of milk. In Heat Induced Changes in Milk; Bulletin 238 of the International Dairy Federation; The Federation Bruxelles: The Netherlands, 1989; Volume 238, pp. 62–67. [Google Scholar]
- Ohta, T.; Yoshida, T.; Kanzaki, B.; Hosono, A.; Suyama, K. Quantitative determination of furosine in cow’s milk containing reconstituted skim milk. Milchwissenschaft 2002, 57, 70–73. [Google Scholar]
- Tokuşoǧlu, Ö.; Akalin, A.S.; Unal, M.K. A rapid high-performance liquid chromatographic detection of furosine (ε-n-2-furoylmethyl-l-lysine) in pasteurized and uht milks. Milchwissenschaft 2004, 59, 502–505. [Google Scholar]
- Tokuşoğlu, O.; Akalin, A.S.; Unal, K. Rapid high performance liquid chromatographic detection of furosine (ε-n-2-furoylmethyl-l-lysine) in yogurt and cheese marketed in turkey. J. Food Qual. 2006, 29, 38–46. [Google Scholar] [CrossRef]
- Choi, H.W.; Park, Y.S.; Lee, S.H.; Lim, C.K.; Seo, J.T.; Yang, K.M. Effects of maternal age on embryo quality and pregnancy outcomes using testicular sperm with intracytoplasmic sperm injection. Clin. Exp. Reprod. Med. 2016, 43, 221–227. [Google Scholar] [CrossRef] [PubMed]
- Niederberger, C. Re: Aberrant sperm dna methylation predicts male fertility status and embryo quality. J. Urol. 2016, 196, 1231. [Google Scholar] [CrossRef] [PubMed]
- Fabbro, M.; Zhou, B.B.; Takahashi, M.; Sarcevic, B.; Lal, P.; Graham, M.E.; Gabrielli, B.G.; Robinson, P.J.; Nigg, E.A.; Ono, Y.; et al. Cdk1/erk2- and plk1-dependent phosphorylation of a centrosome protein, Cep55, is required for its recruitment to midbody and cytokinesis. Dev. Cell 2005, 9, 477–488. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; Burke, L.J.; Rasko, J.E.; Lobanenkov, V.; Renkawitz, R. Dynamic association of the mammalian insulator protein ctcf with centrosomes and the midbody. Exp. Cell Res. 2004, 294, 86–93. [Google Scholar] [CrossRef] [PubMed]
- Van, D.H.A.; Simmons, J.; Khanna, K.K. Cep55 stabilization is required for normal execution of cytokinesis. Cell Cycle. 2009, 8, 3742–3749. [Google Scholar]
- Sinha, D.; Kalimutho, M.; Bowles, J.; Chan, A.L.; Merriner, D.J.; Bain, A.L.; Simmons, J.L.; Freire, R.; Lopez, J.A.; Hobbs, R.M.; et al. Cep55 overexpression causes male-specific sterility in mice by suppressing foxo1 nuclear retention through sustained activation of pi3k/akt signaling. FASEB J. 2018, 32, 4984–4999. [Google Scholar] [CrossRef] [PubMed]
- Van, D.H.A.; Burgering, B.M. Stressing the role of foxo proteins in lifespan and disease. Nat. Rev. Mol. Cell Biol. 2007, 8, 440–450. [Google Scholar]
- Kobayashi, M.; Kikuchi, O.; Sasaki, T.; Kim, H.J.; Yokota, H.H.; Lee, Y.S.; Amano, K.; Kitazumi, T.; Susanti, V.Y.; Kitamura, Y.I.; et al. Foxo1 as a double-edged sword in the pancreas: Analysis of pancreas- and β-cell-specific foxo1 knockout mice. Am. J. Physiol. Endocrinol. Metab. 2012, 302, 603–613. [Google Scholar] [CrossRef]
- Storz, P. Forkhead homeobox type o transcription factors in the responses to oxidative stress. Antioxid Redox Sign. 2011, 14, 593–605. [Google Scholar] [CrossRef] [PubMed]
- Nemoto, S.; Finkel, T. Redox regulation of forkhead proteins through a p66shc-dependent signaling pathway. Science 2002, 295, 2450–2452. [Google Scholar] [CrossRef] [PubMed]
- Sedding, D.G. Foxo transcription factors in oxidative stress response and ageing-a new fork on the way to longevity? J. Biol. Chem. 2008, 389, 279–283. [Google Scholar] [CrossRef] [PubMed]
- Takashi, T.; Mito, K.S.; Lei, Z.; Rao, C.V.; Takashi, S. The luteinizing hormone-testosterone pathway regulates mouse spermatogonial stem cell self-renewal by suppressing WNT5A expression in sertoli cells. Stem Cell Rep. 2016, 7, 279–291. [Google Scholar]
- Liegibel, U.M.; Sommer, U.; Boercsoek, I.; Hilscher, U.; Bierhaus, A.; Schweikert, H.U.; Nawroth, P.; Kasperk, C. Androgen receptor isoforms ar-a and ar-b display functional differences in cultured human bone cells and genital skin fibroblasts. Steroids 2003, 68, 1179–1187. [Google Scholar] [CrossRef] [PubMed]
- Bukovsky, A.; Ayala, M.E.; Dominguez, R.; Keenan, J.A.; Wimalasena, J.; Elder, R.F.; Caudle, M.R. Changes of ovarian interstitial cell hormone receptors and behavior of resident mesenchymal cells in developing and adult rats with steroid-induced sterility. Steroids 2002, 67, 277–289. [Google Scholar] [CrossRef]
- Kim, J.S.; Griswold, M.D. E2f and gata-1 are required for the sertoli cell-specific promoter activity of the follicle-stimulating hormone receptor gene. J. Androl. 2001, 22, 629–639. [Google Scholar]
- Dym, M.; Fawcett, D.W. The blood-testis barrier in the rat and the physiological compartmentation of the seminiferous epithelium. Biol. Reprod. 1970, 3, 308–326. [Google Scholar] [CrossRef]
- Park, H.J.; Lee, R.; Lee, W.Y.; Kim, J.H.; Do, J.T.; Park, C.; Song, H. Stage-specific expression of sal-like protein 4 in boar testicular germ cells. Theriogenology 2017, 101, 44–52. [Google Scholar] [CrossRef]
- Mecklenburg, J.M.; Hermann, B.P. Mechanisms regulating spermatogonial differentiation. Results Probl. Cell Differ. 2016, 58, 253–287. [Google Scholar]
- Meng, X.; Lindahl, M.; Hyvönen, M.E.; Parvinen, M.; De, R.D.G.; Hess, M.W.; Raatikainen-Ahokas, A.; Sainio, K.; Rauvala, H.; Lakso, M.; et al. Regulation of cell fate decision of undifferentiated spermatogonia by gdnf. Science 2000, 287, 1489–1493. [Google Scholar] [CrossRef] [PubMed]
- Ohta, H.; Yomogida, K.; Dohmae, K.; Nishimune, Y. Regulation of proliferation and differentiation in spermatogonial stem cells: The role of c-kit and its ligand scf. Development 2000, 127, 2125–2131. [Google Scholar] [PubMed]
- Zhou, Q.; Nie, R.; Li, Y.; Friel, P.; Mitchell, D.; Hess, R.A.; Small, C.; Griswold, M.D. Expression of stimulated by retinoic acid gene 8 (Stra8) and maturation of murine gonocytes and spermatogonia induced by retinoic acid in vitro. Biol. Reprod. 2008, 9, 35–42. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Wang, S.; Wang, X.; Liao, S.; Wu, Y.; Han, C. Endogenously produced fgf2 is essential for the survival and proliferation of cultured mouse spermatogonial stem cells. Cell Res. 2012, 22, 773–776. [Google Scholar] [CrossRef] [PubMed]
- Ebata, K.T.; Yeh, J.R.; Zhang, X.; Nagano, M.C. Soluble growth factors stimulate spermatogonial stem cell divisions that maintain a stem cell pool and produce progenitors in vitro. Exp. Cell Res. 2011, 317, 1319–1329. [Google Scholar] [CrossRef] [PubMed]
- Kadam, P.H.; Kala, S.; Agrawal, H.; Singh, K.P.; Singh, M.K.; Chauhan, M.S.; Palta, P.; Singla, S.K.; Manik, R.S. Effects of glial cell line-derived neurotrophic factor, fibroblast growth factor 2 and epidermal growth factor on proliferation and the expression of some genes in buffalo (bubalus bubalis) spermatogonial cells. Reprod. Fert. Devel. 2013, 25, 1149–1157. [Google Scholar] [CrossRef] [PubMed]
- Holdcraft, R.W.; Braun, R.E. Androgen receptor function is required in sertoli cells for the terminal differentiation of haploid spermatids. Development 2004, 131, 459–467. [Google Scholar] [CrossRef] [PubMed]
- Dym, M. The fine structure of the monkey (macaca) sertoli cell and its role in maintaining the blood-testis barrier. Anat. Rec. 1973, 175, 639–656. [Google Scholar] [CrossRef]
- Regadera, J.; Martínez-García, F.; González-Peramato, P.; Serrano, A.; Nistal, M.; Suárez-Quian, C. Androgen receptor expression in sertoli cells as a function of seminiferous tubule maturation in the human cryptorchid testis. J. Clin. Endocrinol. Metab. 2001, 86, 413–421. [Google Scholar] [CrossRef]
- Suárez-QuianSharpe, R.M.; Mckinnell, C.; Kivlin, C.; Fisher, J.S. Proliferation and functional maturation of sertoli cells, and their relevance to disorders of testis function in adulthood. Reproduction 2003, 125, 769–784. [Google Scholar]
- Mazaudguittot, S.; Meugnier, E.; Pesenti, S.; Wu, X.; Vidal, H.; Gow, A.; Le, M.B.B. Claudin 11 deficiency in mice results in loss of the sertoli cell epithelial phenotype in the testis. Biol. Reprod. 2010, 82, 202–213. [Google Scholar] [CrossRef] [PubMed]
- Brehm, R.; Zeiler, M.; Rüttinger, C.; Herde, K.; Kibschull, M.; Winterhager, E.; Willecke, K.; Guillou, F.; Lécureuil, C.; Steger, K.; et al. A sertoli cell-specific knockout of connexin 43 prevents initiation of spermatogenesis. Am. J. Pathol. 2007, 171, 19–31. [Google Scholar] [CrossRef] [PubMed]
- Oatley, J.M.; Brinster, R.L. The germline stem cell niche unit in mammalian testes. Physiol. Rev. 2012, 92, 577–595. [Google Scholar] [CrossRef] [PubMed]
- Cory, A.T.; Boyer, A.; Pilon, N.; Lussier, J.G.; Silversides, D.W. Presumptive pre-sertoli cells express genes involved in cell proliferation and cell signalling during a critical window in early testis differentiation. Mol. Reprod. Dev. 2010, 74, 1491–1504. [Google Scholar] [CrossRef] [PubMed]
- Mccoard, S. Gata-4 marks genital ridge formation and sertoli cells in the developing porcine gonad. Angew. Chem. Int. Edit. 2001, 40, 376–378. [Google Scholar]
- Kretser, D.M.D. Sertoli cell-leydig cell interaction in the regulation of testicular function. Int. J. Androl. 2010, 5, 11–17. [Google Scholar] [CrossRef]
- Michel, G.P.; Starka, J. Origin and fate of the lysophosphatidylethanolamine in a chain-forming mutant (envc) of escherichia coli. J. Gen. Microbiol. 1984, 130, 1391–1398. [Google Scholar] [CrossRef]
- Jones, R.E. Synthesis of ether lipids and phosphatidylethanolamine by ejaculated human spermatozoa. Arch. Androl. 1997, 38, 181–189. [Google Scholar] [CrossRef]
- Gulaya, N.M.; Margitich, V.M.; Govseeva, N.M.; Klimashevsky, V.M.; Gorpynchenko, I.I.; Boyko, M.I. Phospholipid composition of human sperm and seminal plasma in relation to sperm fertility. Arch. Androl. 2001, 46, 169–175. [Google Scholar]
- Campbell, E.J. Human leukocyte elastase, cathepsin g, and lactoferrin: Family of neutrophil granule glycoproteins that bind to an alveolar macrophage receptor. Proc. Natl. Acad. Sci. USA 1982, 79, 6941–6945. [Google Scholar] [CrossRef]
- Małaczewska, J.; Rotkiewicz, Z. Lactoferrin—A multipotential protein. Med. Weter. 2007, 63, 136–139. [Google Scholar]
- Chung, S.H.; Kang, H.B.; Kim, J.W.; Yoon, S.S.; Nam, M.S. The Biological Effects of Bovine Lactoferrin on Inflammatory Cytokine Expression in the PMA Stimulated Cells. Korean J. Food Sci. Anim. 2012, 32, 364–368. [Google Scholar] [CrossRef]
- Redwan, E.M.; El-Baky, N.A.; Al-Hejin, A.M.; Baeshen, M.N.; Almehdar, H.A.; Elsaway, A.; Gomaa, A.B.; Al-Masaudi, S.B.; Al-Fassi, F.A.; AbuZeid, I.E.; et al. Significant antibacterial activity and synergistic effects of camel lactoferrin with antibiotics against methicillin-resistant Staphylococcus aureus (MRSA). Res. Microbiol. 2016, 167, 480–491. [Google Scholar] [CrossRef] [PubMed]
- Okubo, K.; Kamiya, M.; Urano, Y.; Nishi, H.; Herter, J.M.; Mayadas, T.; Hirohama, D.; Suzuki, K.; Kawakami, H.; Tanaka, M.; et al. Lactoferrin Supresses Neutrophil Extracellular Traps Release in Inflammation. EBioMedicine 2016, 10, 204–215. [Google Scholar] [CrossRef] [PubMed]
- Artym, J.; Zimecki, M.; Paprocka, M.; Kruzel, M.L. Orally administered lactoferrin restores humoral immune response in immunocompromised mice. Immunol. Lett. 2003, 89, 9–15. [Google Scholar] [CrossRef]
- González-Chávez, S.A.; Arévalo-Gallegos, S.; Rascón-Cruz, Q. Lactoferrin: Structure, function and applications. Int. J. Antimic. Agents 2008, 33, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Ren, F.; Xiong, L.; Zhao, L.; Guo, H.Y. Bovine lactoferrin suppresses high-fat diet induced obesity and modulates gut microbiota in C57BL/6J mice. J. Funct. Foods 2006, 22, 189–200. [Google Scholar] [CrossRef]
- Dominique, L. Overview of Lactoferrin as a Natural Immune Modulator. J. Pediatr. 2016, 173, 10–15. [Google Scholar]
- Sharon, M.D. The Role of Lactoferrin in Gastrointestinal and Immune Development and Function: A Preclinical Perspective. J. Pediat. 2016, 173, 16–28. [Google Scholar]
- Le, G.F.; Loir, M.; Le, B.P.; Ollitrault, M. Insulin-like growth factor (igf-i) mrna and igf-i receptor in trout testis and in isolated spermatogenic and sertoli cells. Mol. Reprod. Dev. 2015, 44, 23–35. [Google Scholar]


| Parameter | Control | Treatment |
|---|---|---|
| Progressive sperm (%) | 52.1 ± 11.2 | 45.6 ± 6.9 * |
| VAP (μm/s) | 43.8 ± 5.3 | 40.6 ± 9.3 * |
| VSL (μm/s) | 28.5 ± 5.5 | 22.1 ± 7.6 * |
| VCL (μm/s) | 82.7 ± 8.7 | 75.9 ± 18.1 * |
| Gene Name | Primer Sequences (5′ → 3′) | |
|---|---|---|
| Forward Primer | Reverse Primer | |
| Cep55 | CGACAAGCCGTGACTCAGT | TCCTCCGACCTCTTCCTCTC |
| FOX01 | GTTGTTGACTTCTGACTCTCCT | CCATCCTACCATAGCCATTGC |
| NF-κB | CAAGAGTGATGACGAGGAGAGT | GTGGAGGTGGATGATGGCTAA |
| PI3K | TAGGAGGAGGTTGGAAGAAGAC | CTACGGAGCAGGCATAGCA |
| Akt | ATGGACTCAAGAGGCAGGAAG | GCAGGACACGGTTCTCAGT |
| TNF-α | CGTGGAACTGGCAGAAGAG | GTAGACAGAAGAGCGTGGTG |
| β-actin | CCTGTATGCCTCTGGTCGTA | CGCTCGTTGCCAATAGTGAT |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, H.; Wang, B.; Yang, H.; Wang, Y.; Xing, L.; Chen, W.; Wang, J.; Zheng, N. Furosine Posed Toxic Effects on Primary Sertoli Cells through Regulating Cep55/NF-κB/PI3K/Akt/FOX01/TNF-α Pathway. Int. J. Mol. Sci. 2019, 20, 3716. https://doi.org/10.3390/ijms20153716
Li H, Wang B, Yang H, Wang Y, Xing L, Chen W, Wang J, Zheng N. Furosine Posed Toxic Effects on Primary Sertoli Cells through Regulating Cep55/NF-κB/PI3K/Akt/FOX01/TNF-α Pathway. International Journal of Molecular Sciences. 2019; 20(15):3716. https://doi.org/10.3390/ijms20153716
Chicago/Turabian StyleLi, Huiying, Bingyuan Wang, Huaigu Yang, Yizhen Wang, Lei Xing, Wei Chen, Jiaqi Wang, and Nan Zheng. 2019. "Furosine Posed Toxic Effects on Primary Sertoli Cells through Regulating Cep55/NF-κB/PI3K/Akt/FOX01/TNF-α Pathway" International Journal of Molecular Sciences 20, no. 15: 3716. https://doi.org/10.3390/ijms20153716
APA StyleLi, H., Wang, B., Yang, H., Wang, Y., Xing, L., Chen, W., Wang, J., & Zheng, N. (2019). Furosine Posed Toxic Effects on Primary Sertoli Cells through Regulating Cep55/NF-κB/PI3K/Akt/FOX01/TNF-α Pathway. International Journal of Molecular Sciences, 20(15), 3716. https://doi.org/10.3390/ijms20153716






