Plasmodesmata-Related Structural and Functional Proteins: The Long Sought-After Secrets of a Cytoplasmic Channel in Plant Cell Walls
Abstract
1. Introduction
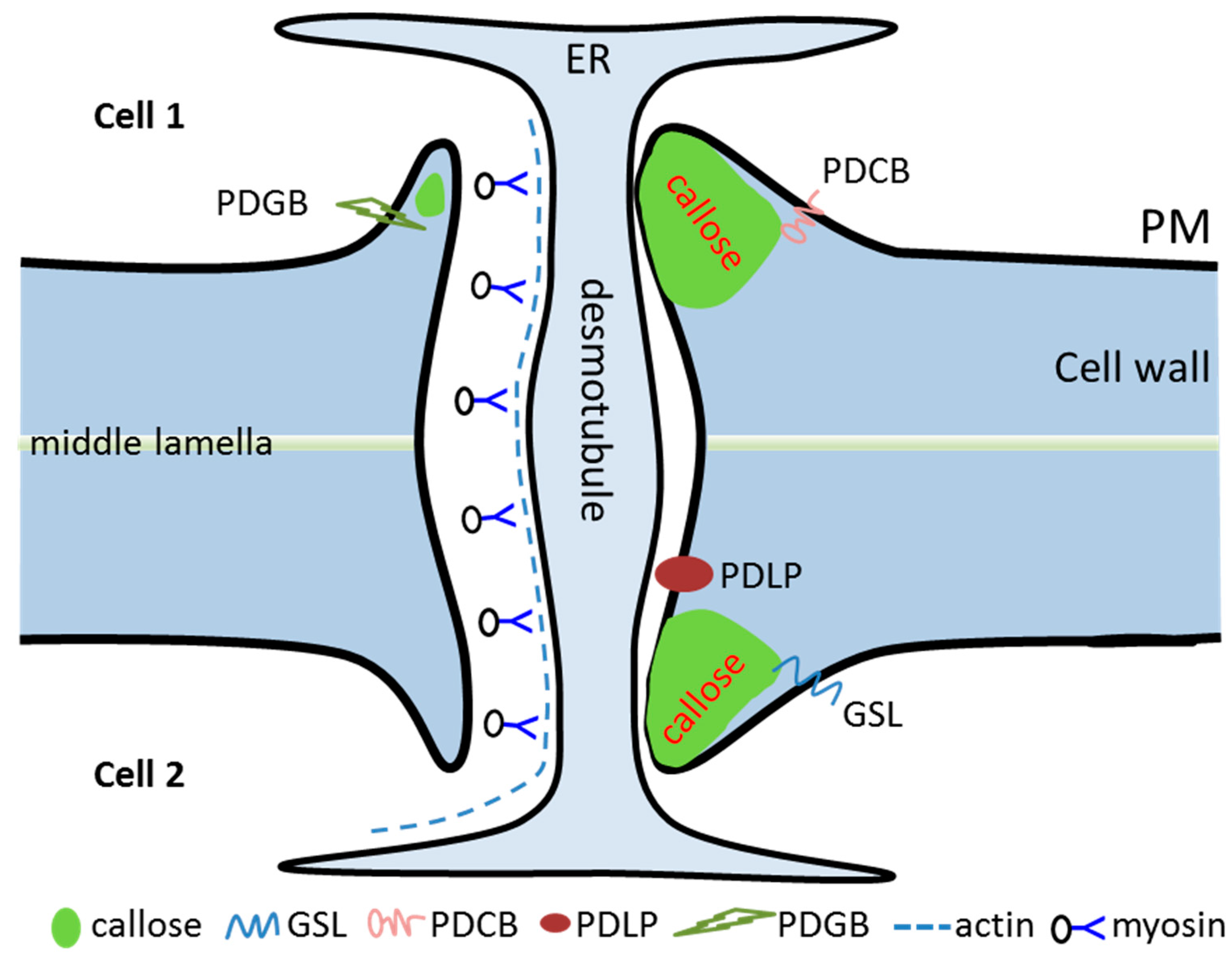
2. The Formation and Structure of PD
3. PD-Associated Structural Proteins
3.1. Actin
3.2. Myosin
3.3. Tubulin
4. PD-Associated Regulatory Proteins
4.1. Callose Synthases Modify Cell Wall PD to Regulate Plant Development
4.2. Callose Hydrolases Degrade Callose to Enhance Intercellular Communication
4.3. PD-Associated Callose Binding Proteins (PDCBs) Regulate Callose Stability
4.4. PD-Located Receptor-Like Proteins (PDLPs) Regulate Plant Defense Responses
4.5. Other PD-Related Cell Wall Proteins and Even More to Be Identified
5. Further Perspectives
Acknowledgments
Conflicts of Interest
References
- Lucas, W.J.; Ham, B.-K.; Kim, J.-Y. Plasmodesmata—Bridging the gap between neighboring plant cells. Trends Cell Biol. 2009, 19, 495–503. [Google Scholar] [CrossRef] [PubMed]
- Epel, B.L. Plasmodesmata: Composition, structure and trafficking. Plant Mol. Biol. 1994, 26, 1343–1356. [Google Scholar] [CrossRef] [PubMed]
- Lucas, W.J. Plasmodesmata: Intercellular channels for macromolecular transport in plants. Curr. Opin. Cell Biol. 1995, 7, 673–680. [Google Scholar] [CrossRef]
- McLean, B.G.; Hempel, F.D.; Zambryski, P.C. Plant intercellular communication via plasmodesmata. Plant Cell 1997, 9, 1043–1054. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.-Y. Regulation of short-distance transport of RNA and protein. Curr. Opin. Plant Biol. 2005, 8, 45–52. [Google Scholar] [CrossRef] [PubMed]
- Ehlers, K.; Kollmann, R. Primary and secondary plasmodesmata: Structure, origin, and functioning. Protoplasma 2001, 216, 1–30. [Google Scholar] [CrossRef]
- Maule, A.J. Plasmodesmata: Structure, function and biogenesis. Curr. Opin. Plant Biol. 2008, 11, 680–686. [Google Scholar] [CrossRef]
- Reagan, B.C.; Ganusova, E.E.; Fernandez, J.C.; McCray, T.N.; Burch-Smith, T.M. RNA on the move: The plasmodesmata perspective. Plant Sci. 2018, 275, 1–10. [Google Scholar] [CrossRef]
- Han, X.; Kumar, D.; Chen, H.; Wu, S.; Kim, J.-Y. Transcription factor-mediated cell-to-cell signalling in plants. J. Exp. Bot. 2014, 65, 1737–1749. [Google Scholar] [CrossRef]
- Lee, J.-Y. New and old roles of plasmodesmata in immunity and parallels to tunneling nanotubes. Plant Sci. 2014, 221, 13–20. [Google Scholar] [CrossRef]
- Zambryski, P.; Crawford, K. Plasmodesmata: Gatekeepers for cell-to-cell transport of developmental signals in plants. Annu. Rev. Cell Dev. Biol. 2000, 16, 393–421. [Google Scholar] [CrossRef] [PubMed]
- Haywood, V.; Kragler, F.; Lucas, W.J. Plasmodesmata: Pathways for protein and ribonucleoprotein signaling. Plant Cell 2002, 14 (Suppl.), S303–S325. [Google Scholar] [CrossRef]
- Otero, S.; Helariutta, Y.; Benitez-Alfonso, Y. Symplastic communication in organ formation and tissue patterning. Curr. Opin. Plant Biol. 2016, 29, 21–28. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.-Y.; Frank, M. Plasmodesmata in phloem: Different gateways for different cargoes. Curr. Opin. Plant Biol. 2018, 43, 119–124. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.-Y. Symplasmic intercellular communication through plasmodesmata. Plants 2018, 7, 23. [Google Scholar] [CrossRef]
- Sager, R.E.; Lee, J.-Y. Plasmodesmata at a glance. J. Cell. Sci. 2018, 131, jcs209346. [Google Scholar] [CrossRef]
- Bell, K.; Oparka, K. Imaging plasmodesmata. Protoplasma 2011, 248, 9–25. [Google Scholar] [CrossRef]
- Lee, J.-Y.; Taoka, K.; Yoo, B.-C.; Ben-Nissan, G.; Kim, D.-J.; Lucas, W.J. Plasmodesmal-associated protein kinase in tobacco and Arabidopsis recognizes a subset of non-cell-autonomous proteins. Plant Cell 2005, 17, 2817–2831. [Google Scholar] [CrossRef]
- Baluska, F.; Samaj, J.; Napier, R.; Volkmann, D. Maize calreticulin localizes preferentially to plasmodesmata in root apex. Plant J. 1999, 19, 481–488. [Google Scholar] [CrossRef]
- Faulkner, C.; Maule, A. Opportunities and successes in the search for plasmodesmal proteins. Protoplasma 2011, 248, 27–38. [Google Scholar] [CrossRef]
- Sun, Y.; Huang, D.; Chen, X. Dynamic regulation of plasmodesmatal permeability and its application to horticultural research. Hortic. Res. 2019, 6, 47. [Google Scholar] [CrossRef]
- Wu, S.-W.; Kumar, R.; Iswanto, A.B.B.; Kim, J.-Y. Callose balancing at plasmodesmata. J. Exp. Bot. 2018, 69, 5325–5339. [Google Scholar] [CrossRef] [PubMed]
- Amsbury, S.; Kirk, P.; Benitez-Alfonso, Y. Emerging models on the regulation of intercellular transport by plasmodesmata-associated callose. J. Exp. Bot. 2017, 69, 105–115. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.-Y.; Kim, J.-Y. Callose synthesis in higher plants. Plant Signal. Behav. 2009, 4, 489–492. [Google Scholar] [CrossRef] [PubMed]
- De Storme, N.; Geelen, D. Callose homeostasis at plasmodesmata: Molecular regulators and developmental relevance. Front. Plant Sci. 2014, 5, 138. [Google Scholar] [CrossRef] [PubMed]
- Heinlein, M.; Epel, B.L. Macromolecular transport and signaling through plasmodesmata. Int. Rev. Cytol. 2004, 235, 93–164. [Google Scholar] [PubMed]
- Guenoune-Gelbart, D.; Elbaum, M.; Sagi, G.; Levy, A.; Epel, B.L. Tobacco mosaic virus (TMV) replicase and movement protein function synergistically in facilitating TMV spread by lateral diffusion in the plasmodesmal desmotubule of Nicotiana benthamiana. Mol. Plant Microbe Interact. 2008, 21, 335–345. [Google Scholar] [CrossRef]
- Levy, A.; Erlanger, M.; Rosenthal, M.; Epel, B.L. A plasmodesmata-associated beta-1,3-glucanase in Arabidopsis. Plant J. 2007, 49, 669–682. [Google Scholar] [CrossRef]
- Simpson, C.; Thomas, C.; Findlay, K.; Bayer, E.; Maule, A.J. An Arabidopsis GPI-anchor plasmodesmal neck protein with callose binding activity and potential to regulate cell-to-cell trafficking. Plant Cell 2009, 21, 581–594. [Google Scholar] [CrossRef]
- Levy, A.; Guenoune-Gelbart, D.; Epel, B.L. Beta-1,3-glucanases: Plasmodesmal gate keepers for intercellular communication. Plant Signal. Behav. 2007, 2, 404–407. [Google Scholar] [CrossRef]
- Knox, K.; Wang, P.; Kriechbaumer, V.; Tilsner, J.; Frigerio, L.; Sparkes, I.; Hawes, C.; Oparka, K. Putting the squeeze on plasmodesmata: A role for reticulons in primary plasmodesmata formation. Plant Physiol. 2015, 168, 1563–1572. [Google Scholar] [CrossRef] [PubMed]
- Fitzgibbon, J.; Bell, K.; King, E.; Oparka, K. Super-resolution imaging of plasmodesmata using three-dimensional structured illumination microscopy. Plant Physiol. 2010, 153, 1453–1463. [Google Scholar] [CrossRef] [PubMed]
- Jones, M.G.; Dropkin, V.H. Scanning electron microscopy in nematode-induced giant transfer cells. Cytobios 1976, 15, 149–161. [Google Scholar] [PubMed]
- Ding, B.; Turgeon, R.; Parthasarathy, M.V. Substructure of freeze-substituted plasmodesmata. Protoplasma 1992, 169, 28–41. [Google Scholar] [CrossRef]
- Zavaliev, R.; Ueki, S.; Epel, B.L.; Citovsky, V. Biology of callose (β-1,3-glucan) turnover at plasmodesmata. Protoplasma 2011, 248, 117–130. [Google Scholar] [CrossRef] [PubMed]
- Vatén, A.; Dettmer, J.; Wu, S.; Stierhof, Y.-D.; Miyashima, S.; Yadav, S.R.; Roberts, C.J.; Campilho, A.; Bulone, V.; Lichtenberger, R.; et al. Callose biosynthesis regulates symplastic trafficking during root development. Dev. Cell 2011, 21, 1144–1155. [Google Scholar] [CrossRef] [PubMed]
- Brecknock, S.; Dibbayawan, T.P.; Vesk, M.; Vesk, P.A.; Faulkner, C.; Barton, D.A.; Overall, R.L. High resolution scanning electron microscopy of plasmodesmata. Planta 2011, 234, 749–758. [Google Scholar] [CrossRef]
- Tilsner, J.; Amari, K.; Torrance, L. Plasmodesmata viewed as specialised membrane adhesion sites. Protoplasma 2011, 248, 39–60. [Google Scholar] [CrossRef]
- White, R.G.; Barton, D.A. The cytoskeleton in plasmodesmata: A role in intercellular transport? J. Exp. Bot. 2011, 62, 5249–5266. [Google Scholar] [CrossRef][Green Version]
- Kumar, D.; Kumar, R.; Hyun, T.K.; Kim, J.-Y. Cell-to-cell movement of viruses via plasmodesmata. J. Plant Res. 2015, 128, 37–47. [Google Scholar] [CrossRef]
- Botha, C.E.J.; Hartley, B.J.; Cross, R.H.M. The ultrastructure and computer-enhanced digital image analysis of plasmodesmata at the Kranz mesophyll-bundle sheath interface of Themeda triandra var. imberbis (Retz) A. Camus in conventionally-fixed blades. Ann. Bot. 1993, 72, 255–261. [Google Scholar]
- Wright, K.M.; Oparka, K.J. The ER Within Plasmodesmata. In The Plant Endoplasmic Reticulum; Robinson, D.G., Ed.; Plant Cell Monographs; Springer: Berlin/Heidelberg, Germany, 2006; pp. 279–308. [Google Scholar]
- Gamalei, Y.V.; van Bel, A.J.E.; Pakhomova, M.V.; Sjutkina, A.V. Effects of temperature on the conformation of the endoplasmic reticulum and on starch accumulation in leaves with the symplasmic minor-vein configuration. Planta 1994, 194, 443–453. [Google Scholar] [CrossRef]
- Cook, M.E.; Graham, L.E. Evolution of Plasmodesmata. In Plasmodesmata: Structure, Function, Role in Cell Communication; Van Bel, A.J.E., Van Kesteren, W.J.P., Eds.; Springer: Berlin/Heidelberg, Germany, 1999; pp. 101–117. [Google Scholar]
- Overall, R.L.; Blackman, L.M. A model of the macromolecular structure of plasmodesmata. Trends Plant Sci. 1996, 1, 307–311. [Google Scholar] [CrossRef]
- Chaffey, N.; Barlow, P. Myosin, microtubules, and microfilaments: Co-operation between cytoskeletal components during cambial cell division and secondary vascular differentiation in trees. Planta 2002, 214, 526–536. [Google Scholar] [CrossRef] [PubMed]
- Vale, R.D. The molecular motor toolbox for intracellular transport. Cell 2003, 112, 467–480. [Google Scholar] [CrossRef]
- Ehrhardt, D.W.; Shaw, S.L. Microtubule dynamics and organization in the plant cortical array. Ann. Rev. Plant Biol. 2006, 57, 859–875. [Google Scholar] [CrossRef]
- White, R.G.; Badelt, K.; Overall, R.L.; Vesk, M. Actin associated with plasmodesmata. Protoplasma 1994, 180, 169–184. [Google Scholar] [CrossRef]
- Ding, B.; Kwon, M.-O.; Warnberg, L. Evidence that actin filaments are involved in controlling the permeability of plasmodesmata in tobacco mesophyll. Plant J. 1996, 10, 157–164. [Google Scholar] [CrossRef]
- Blackman, L.M.; Overall, R.L. Immunolocalisation of the cytoskeleton to plasmodesmata of Chara corallina. Plant J. 1998, 14, 733–741. [Google Scholar] [CrossRef]
- Radford, J.E.; White, R.G. Localization of a myosin-like protein to plasmodesmata. Plant J. 1998, 14, 743–750. [Google Scholar] [CrossRef]
- Han, X.; Hyun, T.K.; Zhang, M.; Kumar, R.; Koh, E.; Kang, B.-H.; Lucas, W.J.; Kim, J.-Y. Auxin-callose-mediated plasmodesmal gating is essential for tropic auxin gradient formation and signaling. Dev. Cell 2014, 28, 132–146. [Google Scholar] [CrossRef] [PubMed]
- Benitez-Alfonso, Y.; Faulkner, C.; Pendle, A.; Miyashima, S.; Helariutta, Y.; Maule, A. Symplastic intercellular connectivity regulates lateral root patterning. Dev. Cell 2013, 26, 136–147. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Calvino, L.; Faulkner, C.; Walshaw, J.; Saalbach, G.; Bayer, E.; Benitez-Alfonso, Y.; Maule, A. Arabidopsis plasmodesmal proteome. PLoS ONE 2011, 6, e18880. [Google Scholar] [CrossRef]
- Amari, K.; Boutant, E.; Hofmann, C.; Schmitt-Keichinger, C.; Fernandez-Calvino, L.; Didier, P.; Lerich, A.; Mutterer, J.; Thomas, C.L.; Heinlein, M.; et al. A family of plasmodesmal proteins with receptor-like properties for plant viral movement proteins. PLoS Pathog. 2010, 6, e1001119. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.-Y.; Wang, X.; Cui, W.; Sager, R.; Modla, S.; Czymmek, K.; Zybaliov, B.; van Wijk, K.; Zhang, C.; Lu, H.; et al. A plasmodesmata-localized protein mediates crosstalk between cell-to-cell communication and innate immunity in Arabidopsis. Plant Cell 2011, 23, 3353–3373. [Google Scholar] [CrossRef] [PubMed]
- Thomas, C.L.; Bayer, E.M.; Ritzenthaler, C.; Fernandez-Calvino, L.; Maule, A.J. Specific targeting of a plasmodesmal protein affecting cell-to-cell communication. PLoS Biol. 2008, 6, e7. [Google Scholar] [CrossRef] [PubMed]
- Caillaud, M.-C.; Wirthmueller, L.; Sklenar, J.; Findlay, K.; Piquerez, S.J.M.; Jones, A.M.E.; Robatzek, S.; Jones, J.D.G.; Faulkner, C. The plasmodesmal protein PDLP1 localises to haustoria-associated membranes during downy mildew infection and regulates callose deposition. PLoS Pathog. 2014, 10, e1004496. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Zhang, Y.; Zhu, L.; Yuan, M. The actin cytoskeleton is involved in the regulation of the plasmodesmal size exclusion limit. Plant Signal. Behav. 2010, 5, 1663–1665. [Google Scholar] [CrossRef][Green Version]
- Badelt, K.; White, R.G.; Overall, R.L.; Vesk, M. Ultrastructural specializations of the cell wall sleeve around plasmodesmata. Am. J. Plant Bot. 1994, 81, 1422–1427. [Google Scholar] [CrossRef]
- Aaziz, R.; Dinant, S.; Epel, B.L. Plasmodesmata and plant cytoskeleton. Trends Plant Sci. 2001, 6, 326–330. [Google Scholar] [CrossRef]
- Brunkard, J.O.; Zambryski, P.C. Plasmodesmata enable multicellularity: New insights into their evolution, biogenesis, and functions in development and immunity. Curr. Opin. Plant Biol. 2017, 35, 76–83. [Google Scholar] [CrossRef] [PubMed]
- Ganusova, E.E.; Burch-Smith, T.M. Review: Plant-pathogen interactions through the plasmodesma prism. Plant Sci. 2019, 279, 70–80. [Google Scholar] [CrossRef] [PubMed]
- Turner, A.; Bacic, A.; Harris, P.J.; Read, S.M. Membrane fractionation and enrichment of callose synthase from pollen tubes of Nicotiana alata Link et Otto. Planta 1998, 205, 380–388. [Google Scholar] [CrossRef] [PubMed]
- Yadav, S.R.; Yan, D.; Sevilem, I.; Helariutta, Y. Plasmodesmata-mediated intercellular signaling during plant growth and development. Front. Plant Sci. 2014, 5, 44. [Google Scholar] [CrossRef] [PubMed]
- Olatunji, D.; Geelen, D.; Verstraeten, I. Control of endogenous auxin levels in plant root development. Int. J. Mol. Sci. 2017, 18, 2587. [Google Scholar] [CrossRef] [PubMed]
- Han, X.; Kim, J.-Y. Integrating hormone- and micromolecule-mediated signaling with plasmodesmal communication. Mol. Plant 2016, 9, 46–56. [Google Scholar] [CrossRef] [PubMed]
- Faulkner, C. Plasmodesmata and the symplast. Curr. Biol. 2018, 28, R1374–R1378. [Google Scholar] [CrossRef]
- Liu, Y.; Xu, M.; Liang, N.; Zheng, Y.; Yu, Q.; Wu, S. Symplastic communication spatially directs local auxin biosynthesis to maintain root stem cell niche in Arabidopsis. Proc. Natl. Acad. Sci. USA 2017, 114, 4005–4010. [Google Scholar] [CrossRef]
- Slewinski, T.L.; Baker, R.F.; Stubert, A.; Braun, D.M. Tie-dyed2 encodes a callose synthase that functions in vein development and affects symplastic trafficking within the phloem of maize leaves. Plant Physiol. 2012, 160, 1540–1550. [Google Scholar] [CrossRef]
- Baker, R.F.; Slewinski, T.L.; Braun, D.M. The tie-dyed pathway promotes symplastic trafficking in the phloem. Plant Signal. Behav. 2013, 8, e24540. [Google Scholar] [CrossRef]
- Song, L.; Wang, R.; Zhang, L.; Wang, Y.; Yao, S. CRR1 encoding callose synthase functions in ovary expansion by affecting vascular cell patterning in rice. Plant J. 2016, 88, 620–632. [Google Scholar] [CrossRef] [PubMed]
- Beffa, R.; Meins, F. Pathogenesis-related functions of plant beta-1,3-glucanases investigated by antisense transformation—A review. Gene 1996, 179, 97–103. [Google Scholar] [CrossRef]
- Iglesias, V.A.; Meins, F. Movement of plant viruses is delayed in a beta-1,3-glucanase-deficient mutant showing a reduced plasmodesmatal size exclusion limit and enhanced callose deposition. Plant J. 2000, 21, 157–166. [Google Scholar] [CrossRef] [PubMed]
- Bucher, G.L.; Tarina, C.; Heinlein, M.; Di Serio, F.; Meins, F.; Iglesias, V.A. Local expression of enzymatically active class I beta-1, 3-glucanase enhances symptoms of TMV infection in tobacco. Plant J. 2001, 28, 361–369. [Google Scholar] [CrossRef] [PubMed]
- Rinne, P.L.H.; Welling, A.; Vahala, J.; Ripel, L.; Ruonala, R.; Kangasjärvi, J.; van der Schoot, C. Chilling of dormant buds hyperinduces FLOWERING LOCUS T and recruits GA-inducible 1,3-beta-glucanases to reopen signal conduits and release dormancy in Populus. Plant Cell 2011, 23, 130–146. [Google Scholar] [CrossRef] [PubMed]
- Xu, F.; Rong, X.; Huang, X.; Cheng, S. Recent advances of flowering locus T gene in higher plants. Int. J. Mol. Sci. 2012, 13, 3773–3781. [Google Scholar] [CrossRef] [PubMed]
- Conti, L.; Bradley, D. TERMINAL FLOWER1 is a mobile signal controlling Arabidopsis architecture. Plant Cell 2007, 19, 767–778. [Google Scholar] [CrossRef]
- Ruonala, R.; Rinne, P.L.H.; Kangasjarvi, J.; van der Schoot, C. CENL1 expression in the rib meristem affects stem elongation and the transition to dormancy in Populus. Plant Cell 2008, 20, 59–74. [Google Scholar] [CrossRef]
- Rinne, P.L.H.; Paul, L.K.; Vahala, J.; Kangasjärvi, J.; van der Schoot, C. Axillary buds are dwarfed shoots that tightly regulate GA pathway and GA-inducible 1,3-β-glucanase genes during branching in hybrid aspen. J. Exp. Bot. 2016, 67, 5975–5991. [Google Scholar] [CrossRef]
- Tylewicz, S.; Petterle, A.; Marttila, S.; Miskolczi, P.; Azeez, A.; Singh, R.K.; Immanen, J.; Mähler, N.; Hvidsten, T.R.; Eklund, D.M.; et al. Photoperiodic control of seasonal growth is mediated by ABA acting on cell-cell communication. Science 2018, 360, 212–215. [Google Scholar] [CrossRef]
- Singh, R.K.; Miskolczi, P.; Maurya, J.P.; Bhalerao, R.P. A tree ortholog of SHORT VEGETATIVE PHASE floral repressor mediates photoperiodic control of bud dormancy. Curr. Biol. 2019, 29, 128–133. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.K.; Maurya, J.P.; Azeez, A.; Miskolczi, P.; Tylewicz, S.; Stojkovič, K.; Delhomme, N.; Busov, V.; Bhalerao, R.P. A genetic network mediating the control of bud break in hybrid aspen. Nat. Commun. 2018, 9, 4173. [Google Scholar] [CrossRef] [PubMed]
- Ruan, Y.L.; Llewellyn, D.J.; Furbank, R.T. The control of single-celled cotton fiber elongation by developmentally reversible gating of plasmodesmata and coordinated expression of sucrose and K+ transporters and expansin. Plant Cell 2001, 13, 47–60. [Google Scholar] [PubMed]
- Ruan, Y.-L.; Xu, S.-M.; White, R.; Furbank, R.T. Genotypic and developmental evidence for the role of plasmodesmatal regulation in cotton fiber elongation mediated by callose turnover. Plant Physiol. 2004, 136, 4104–4113. [Google Scholar] [CrossRef] [PubMed]
- Zavaliev, R.; Dong, X.; Epel, B.L. Glycosylphosphatidylinositol (GPI) modification serves as a primary plasmodesmal sorting signal. Plant Physiol. 2016, 172, 1061–1073. [Google Scholar]
- Brunkard, J.O.; Runkel, A.M.; Zambryski, P.C. The cytosol must flow: Intercellular transport through plasmodesmata. Curr. Opin. Cell Biol. 2015, 35, 13–20. [Google Scholar] [CrossRef]
- Vaattovaara, A.; Brandt, B.; Rajaraman, S.; Safronov, O.; Veidenberg, A.; Luklová, M.; Kangasjärvi, J.; Löytynoja, A.; Hothorn, M.; Salojärvi, J.; et al. Mechanistic insights into the evolution of DUF26-containing proteins in land plants. Commun. Biol. 2019, 2, 56. [Google Scholar] [CrossRef]
- Klessig, D.F.; Choi, H.W.; Dempsey, D.A. Systemic acquired resistance and salicylic acid: Past, present, and future. Mol. Plant Microbe Interact. 2018, 31, 871–888. [Google Scholar] [CrossRef]
- Ádám, A.L.; Nagy, Z.Á.; Kátay, G.; Mergenthaler, E.; Viczián, O. Signals of systemic immunity in plants: Progress and open questions. Int. J. Mol. Sci. 2018, 19, 1146. [Google Scholar] [CrossRef]
- Li, N.; Han, X.; Feng, D.; Yuan, D.; Huang, L.-J. Signaling crosstalk between salicylic acid and ethylene/jasmonate in plant defense: Do we understand what they are whispering? Int. J. Mol. Sci. 2019, 20, 671. [Google Scholar] [CrossRef]
- Yu, K.; Soares, J.M.; Mandal, M.K.; Wang, C.; Chanda, B.; Gifford, A.N.; Fowler, J.S.; Navarre, D.; Kachroo, A.; Kachroo, P. A feedback regulatory loop between G3P and lipid transfer proteins DIR1 and AZI1 mediates azelaic-acid-induced systemic immunity. Cell Rep. 2013, 3, 1266–1278. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; El-Shetehy, M.; Shine, M.B.; Yu, K.; Navarre, D.; Wendehenne, D.; Kachroo, A.; Kachroo, P. Free radicals mediate systemic acquired resistance. Cell Rep. 2014, 7, 348–355. [Google Scholar] [CrossRef] [PubMed]
- Lim, G.-H.; Shine, M.B.; de Lorenzo, L.; Yu, K.; Cui, W.; Navarre, D.; Hunt, A.G.; Lee, J.-Y.; Kachroo, A.; Kachroo, P. Plasmodesmata localizing proteins regulate transport and signaling during systemic acquired immunity in plants. Cell Host Microbe 2016, 19, 541–549. [Google Scholar] [CrossRef] [PubMed]
- Lim, G.-H.; Kachroo, A.; Kachroo, P. Role of plasmodesmata and plasmodesmata localizing proteins in systemic immunity. Plant Signal. Behav. 2016, 11, e1219829. [Google Scholar] [CrossRef] [PubMed]
- Ye, Z.-W.; Chen, Q.-F.; Chye, M.-L. Arabidopsis thaliana acyl-CoA-binding protein ACBP6 interacts with plasmodesmata-located protein PDLP8. Plant Signal. Behav. 2017, 12, e1359365. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Cui, W.; Lee, J.-Y. Arabidopsis callose synthases CalS1/8 regulate plasmodesmal permeability during stress. Nat. Plants 2016, 2, 16034. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.; Chen, X.-Y.; Rim, Y.; Han, X.; Cho, W.K.; Kim, S.-W.; Kim, J.-Y. Arabidopsis glucan synthase-like 10 functions in male gametogenesis. J. Plant Physiol. 2009, 166, 344–352. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Zhu, P.; Qu, S.; Zhao, J.; Singh, P.K.; Wang, W. Ectodomain of plasmodesmata-localized protein 5 in Arabidopsis: Expression, purification, crystallization and crystallographic analysis. Acta Crystallogr. F-Struct. Biol. Commun. 2017, 73, 532–535. [Google Scholar] [CrossRef] [PubMed]
- Cheval, C.; Faulkner, C. Plasmodesmal regulation during plant-pathogen interactions. New Phytol. 2018, 217, 62–67. [Google Scholar] [CrossRef] [PubMed]
- Sonnino, S.; Prinetti, A. Lipids and membrane lateral organization. Front. Physiol. 2010, 1, 153. [Google Scholar] [CrossRef] [PubMed]
- Jarsch, I.K.; Konrad, S.S.A.; Stratil, T.F.; Urbanus, S.L.; Szymanski, W.; Braun, P.; Braun, K.-H.; Ott, T. Plasma membranes are subcompartmentalized into a plethora of coexisting and diverse microdomains in Arabidopsis and Nicotiana benthamiana. Plant Cell 2014, 26, 1698–1711. [Google Scholar] [CrossRef] [PubMed]
- Iswanto, A.B.B.; Kim, J.-Y. Lipid raft, regulator of plasmodesmal callose homeostasis. Plants 2017, 6, 15. [Google Scholar] [CrossRef] [PubMed]
- Grison, M.S.; Brocard, L.; Fouillen, L.; Nicolas, W.; Wewer, V.; Dörmann, P.; Nacir, H.; Benitez-Alfonso, Y.; Claverol, S.; Germain, V.; et al. Specific membrane lipid composition is important for plasmodesmata function in Arabidopsis. Plant Cell 2015, 27, 1228–1250. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Ruan, Y.-L.; Zhou, N.; Wang, F.; Guan, X.; Fang, L.; Shang, X.; Guo, W.; Zhu, S.; Zhang, T. Suppressing a putative sterol carrier gene reduces plasmodesmal permeability and activates sucrose transporter genes during cotton fiber elongation. Plant Cell 2017, 29, 2027–2046. [Google Scholar] [CrossRef] [PubMed]
- Park, S.-H.; Li, F.; Renaud, J.; Shen, W.; Li, Y.; Guo, L.; Cui, H.; Sumarah, M.; Wang, A. NbEXPA1, an α-expansin, is plasmodesmata-specific and a novel host factor for potyviral infection. Plant J. 2017, 92, 846–861. [Google Scholar] [CrossRef] [PubMed]
- Oparka, K.J. Signalling via plasmodesmata—The neglected pathway. Semin. Cell Biol. 1993, 4, 131–138. [Google Scholar] [CrossRef]
- Tilsner, J.; Nicolas, W.; Rosado, A.; Bayer, E.M. Staying tight: Plasmodesmal membrane contact sites and the control of cell-to-cell connectivity in plants. Ann. Rev. Plant Biol. 2016, 67, 337–364. [Google Scholar] [CrossRef]
- Schubert, V. Super-resolution microscopy—Applications in plant cell research. Front. Plant Sci. 2017, 8, 531. [Google Scholar] [CrossRef]
| Gene | Protein Function | Method | Biological Function | References |
|---|---|---|---|---|
| Actin | Cell cytoskeleton | Immunolocalization | PD structure component | [49,50,51] |
| Myosin | Cell cytoskeleton | Immunolocalization | PD structure component | [46,51,52] |
| Tubulin | Cell cytoskeleton | Immunolocalization | PD structure component | [51] |
| GSL8 (CALS10) | Callose synthase | Genetics and cell biology | Callose deposition and auxin response | [53] |
| GSL12 (CALS3) | Callose synthase | Genetics and cell biology | Callose deposition and phloem development | [36] |
| PDBGs (AtBG_ppap) | β-1,3-glucanase | Proteomics and cell biology | Callose degradation and lateral root development | [28,54,55] |
| PDCBs | Callose binding protein | Proteomics and cell biology | PD localization and callose stabilization at neck region | [29] |
| PDLPs | Transmembrane receptor-like protein kinase | Proteomics and cell biology | SAR- and SA-induced callose accumulation | [55,56,57,58,59] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Han, X.; Huang, L.-J.; Feng, D.; Jiang, W.; Miu, W.; Li, N. Plasmodesmata-Related Structural and Functional Proteins: The Long Sought-After Secrets of a Cytoplasmic Channel in Plant Cell Walls. Int. J. Mol. Sci. 2019, 20, 2946. https://doi.org/10.3390/ijms20122946
Han X, Huang L-J, Feng D, Jiang W, Miu W, Li N. Plasmodesmata-Related Structural and Functional Proteins: The Long Sought-After Secrets of a Cytoplasmic Channel in Plant Cell Walls. International Journal of Molecular Sciences. 2019; 20(12):2946. https://doi.org/10.3390/ijms20122946
Chicago/Turabian StyleHan, Xiao, Li-Jun Huang, Dan Feng, Wenhan Jiang, Wenzhuo Miu, and Ning Li. 2019. "Plasmodesmata-Related Structural and Functional Proteins: The Long Sought-After Secrets of a Cytoplasmic Channel in Plant Cell Walls" International Journal of Molecular Sciences 20, no. 12: 2946. https://doi.org/10.3390/ijms20122946
APA StyleHan, X., Huang, L.-J., Feng, D., Jiang, W., Miu, W., & Li, N. (2019). Plasmodesmata-Related Structural and Functional Proteins: The Long Sought-After Secrets of a Cytoplasmic Channel in Plant Cell Walls. International Journal of Molecular Sciences, 20(12), 2946. https://doi.org/10.3390/ijms20122946





