Olive Oil, Palm Oil, and Hybrid Palm Oil Distinctly Modulate Liver Transcriptome and Induce NAFLD in Mice Fed a High-Fat Diet
Abstract
1. Introduction
2. Results
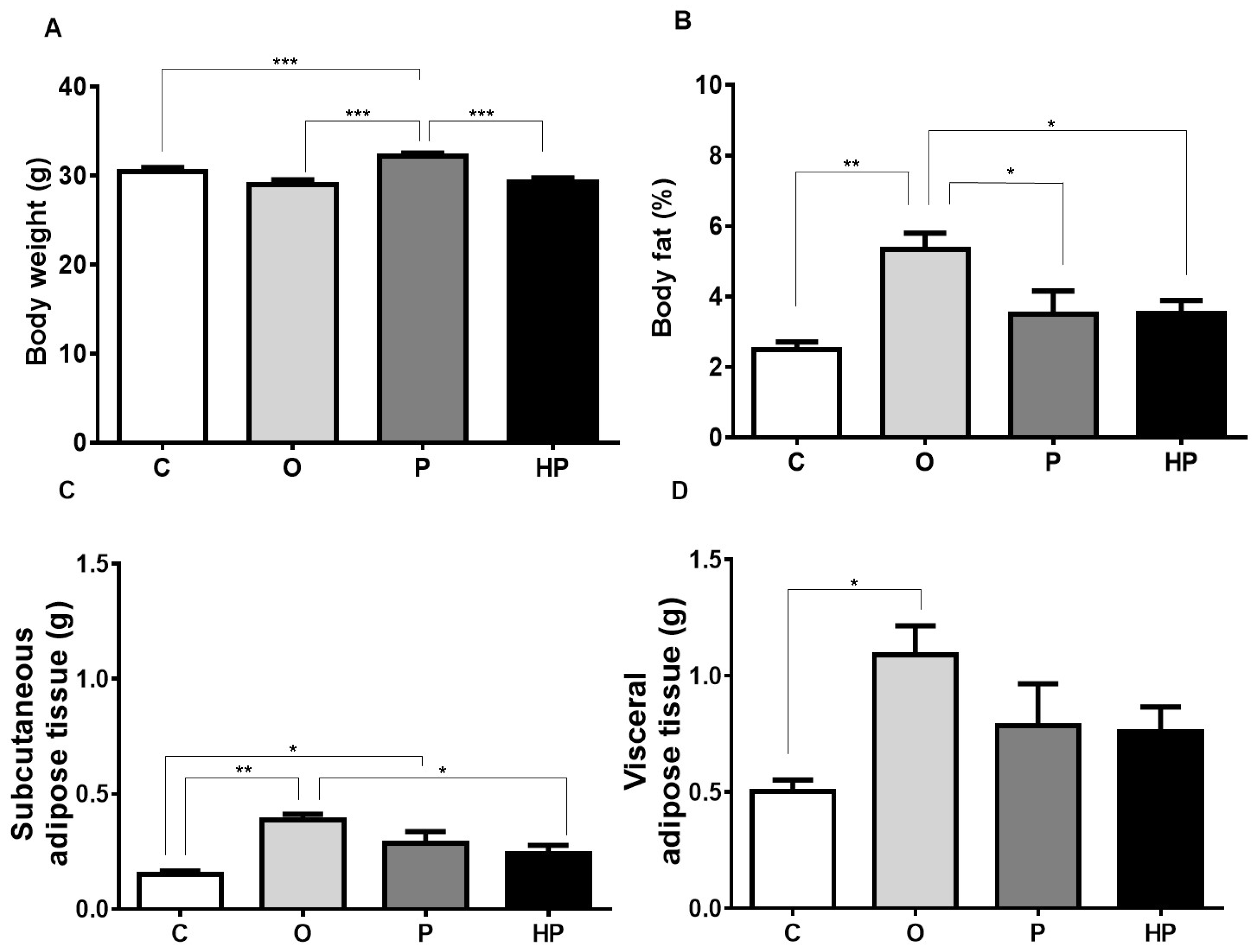
2.1. Body Weight and Adipose Tissue
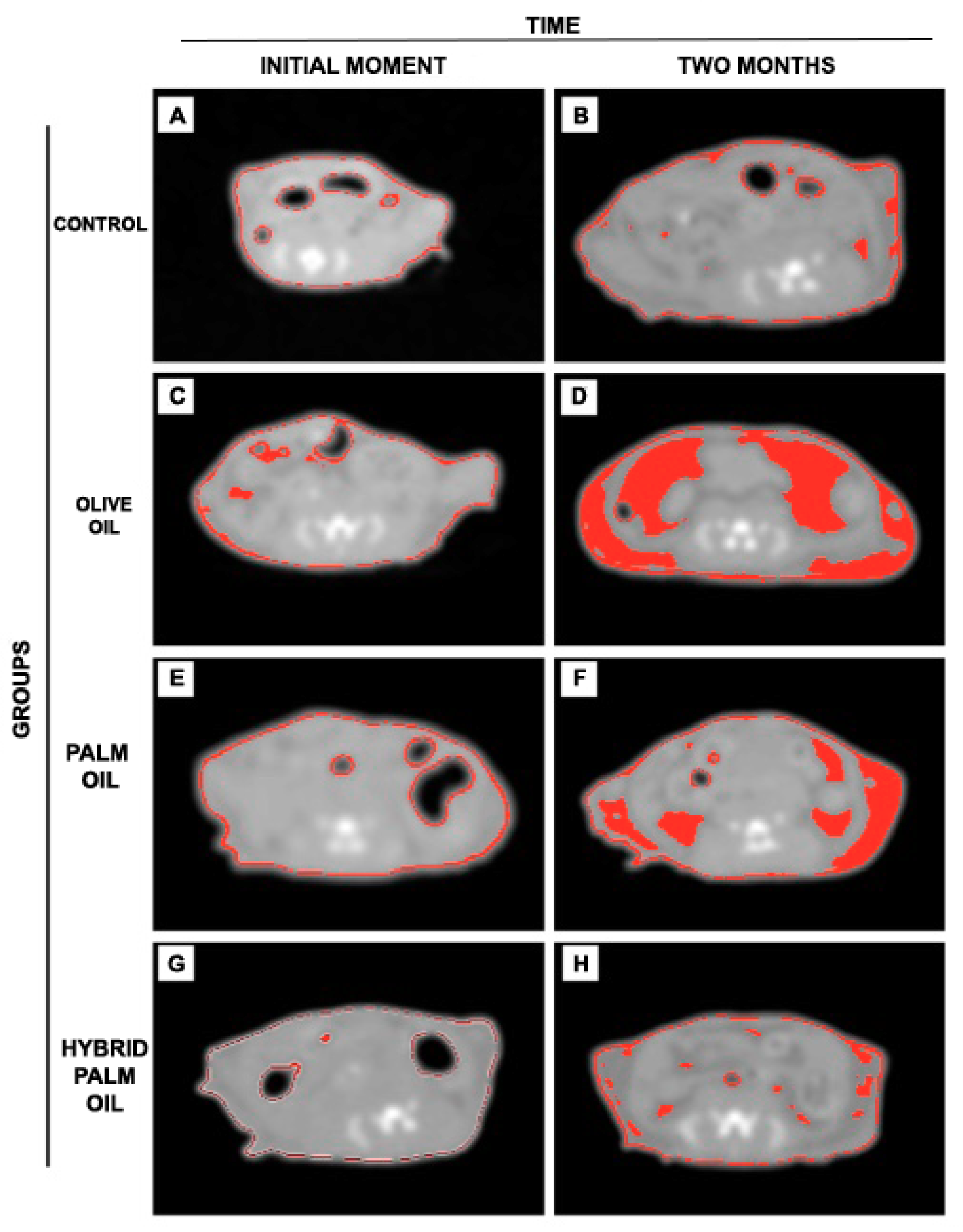
2.2. Adiposity Measurement by Computed Tomography (CT) in Mice
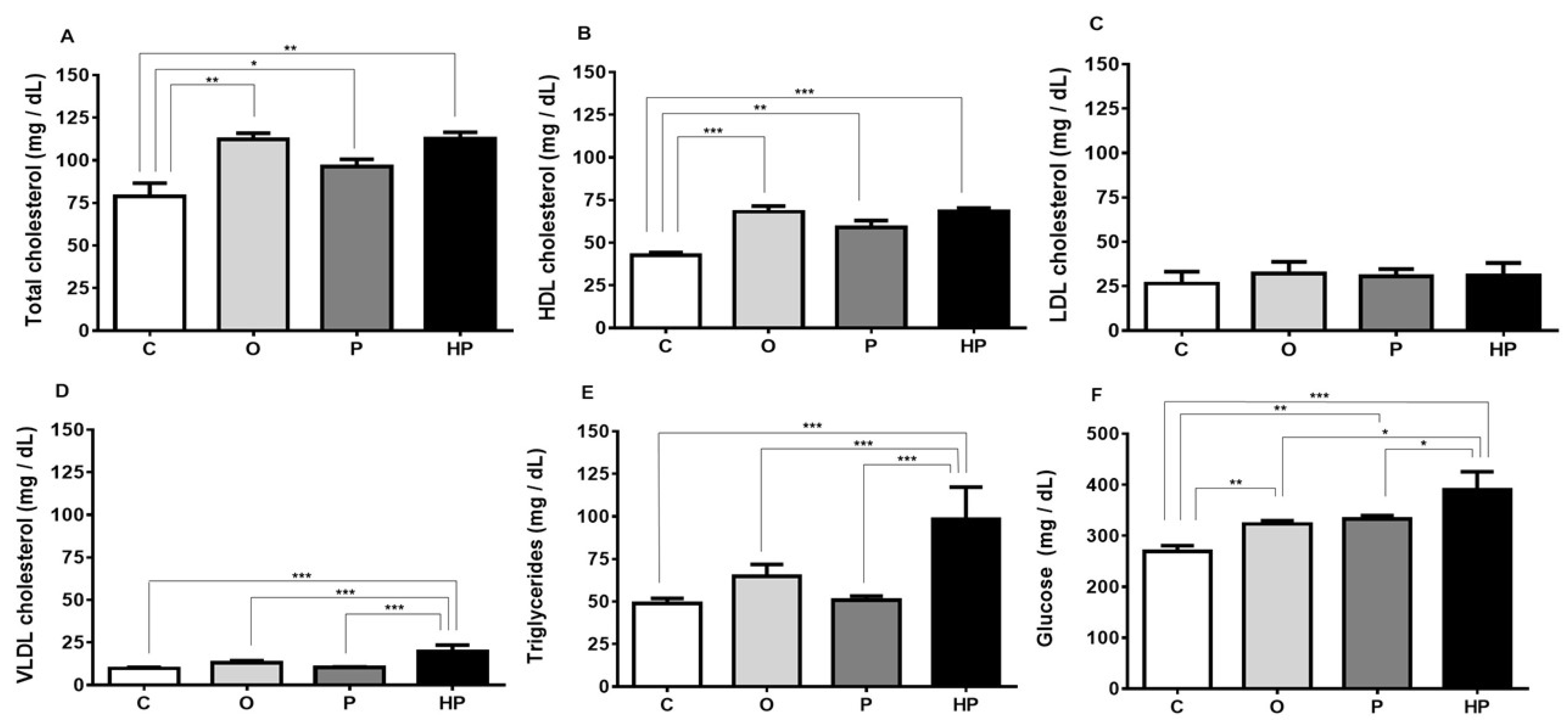
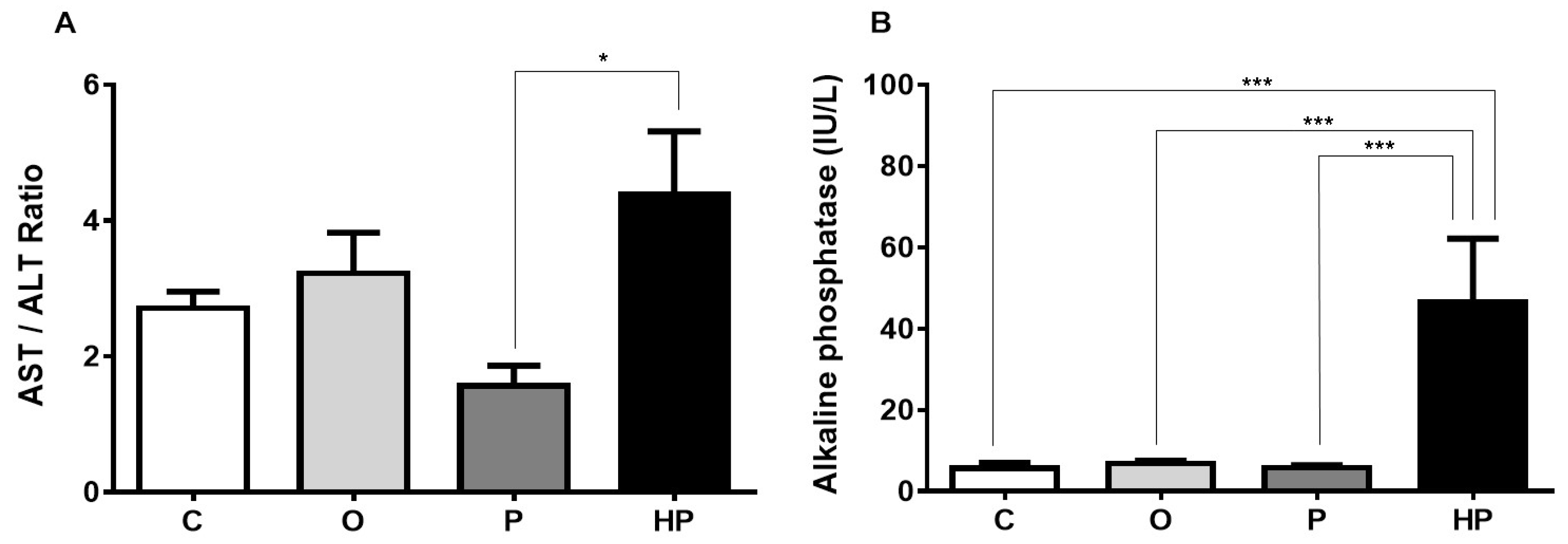
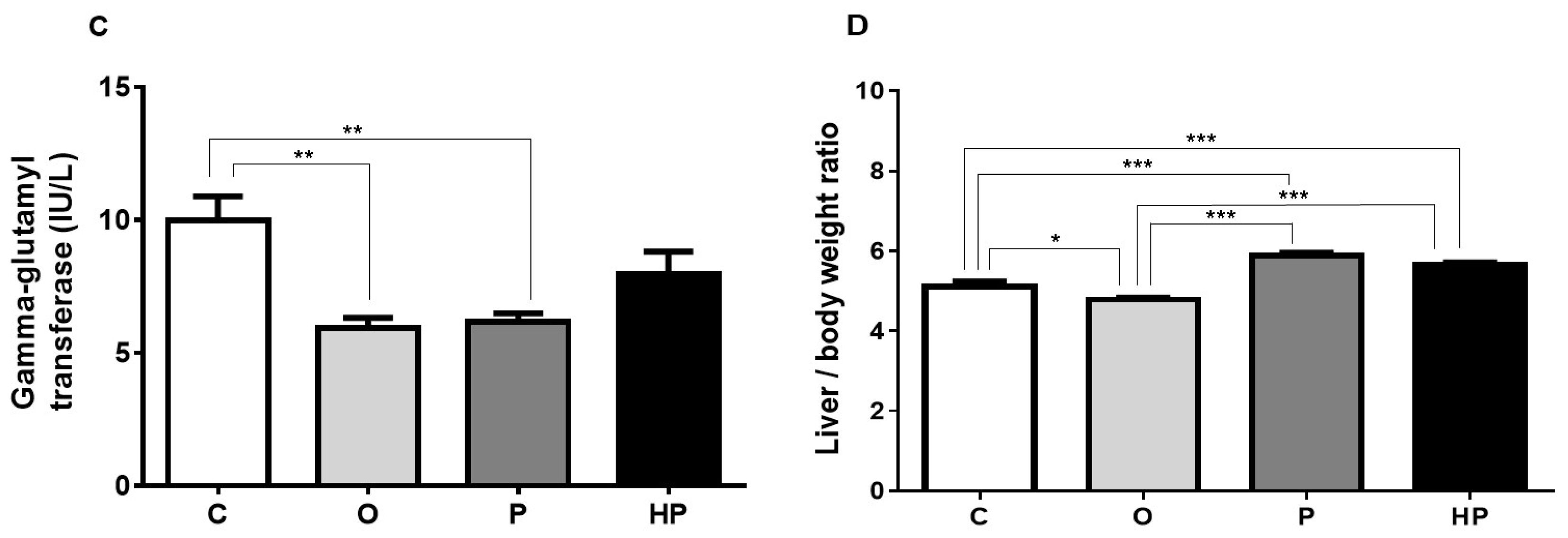
2.3. Biochemical and Liver Analyses
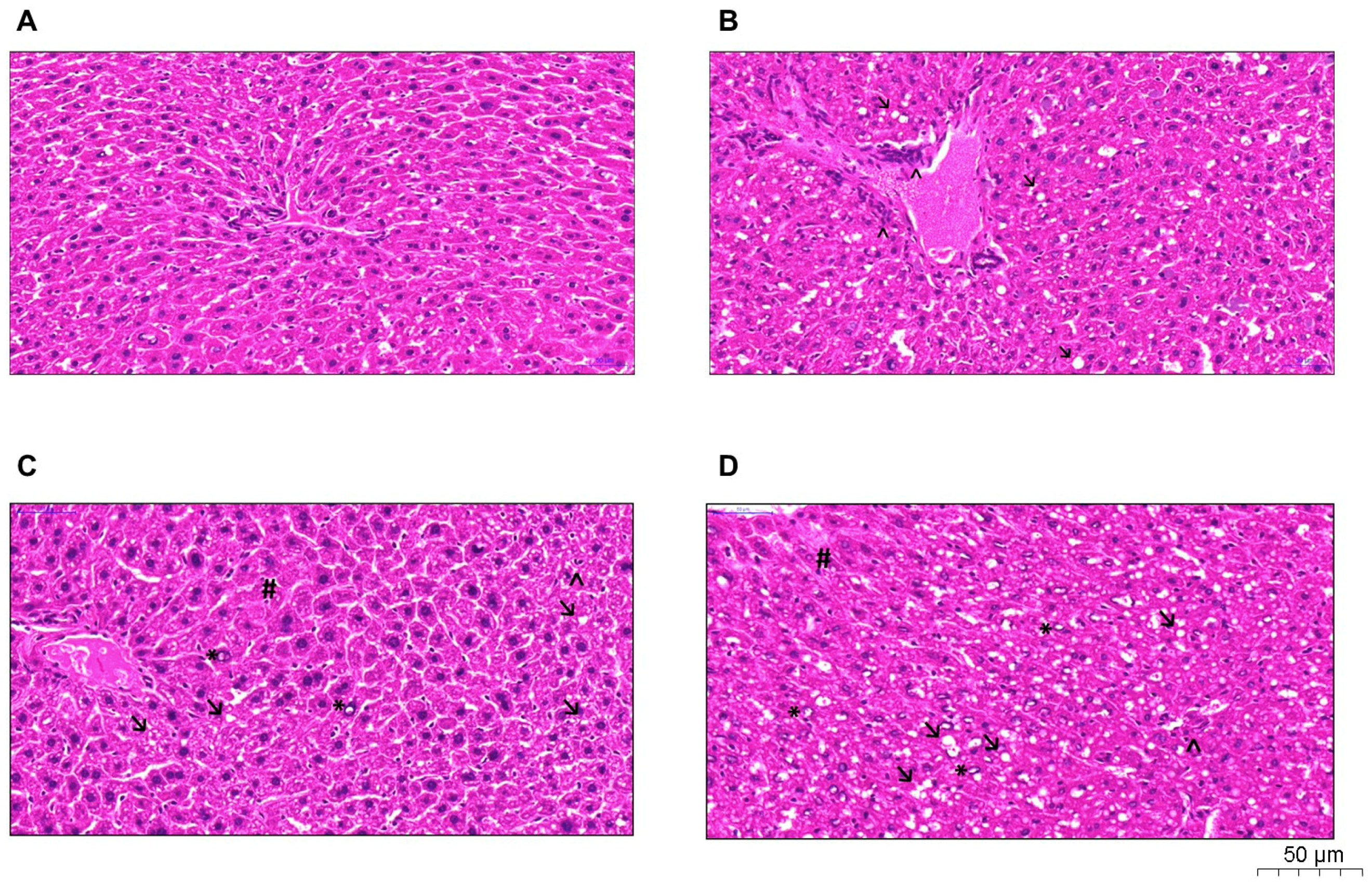
2.4. Histological Analysis
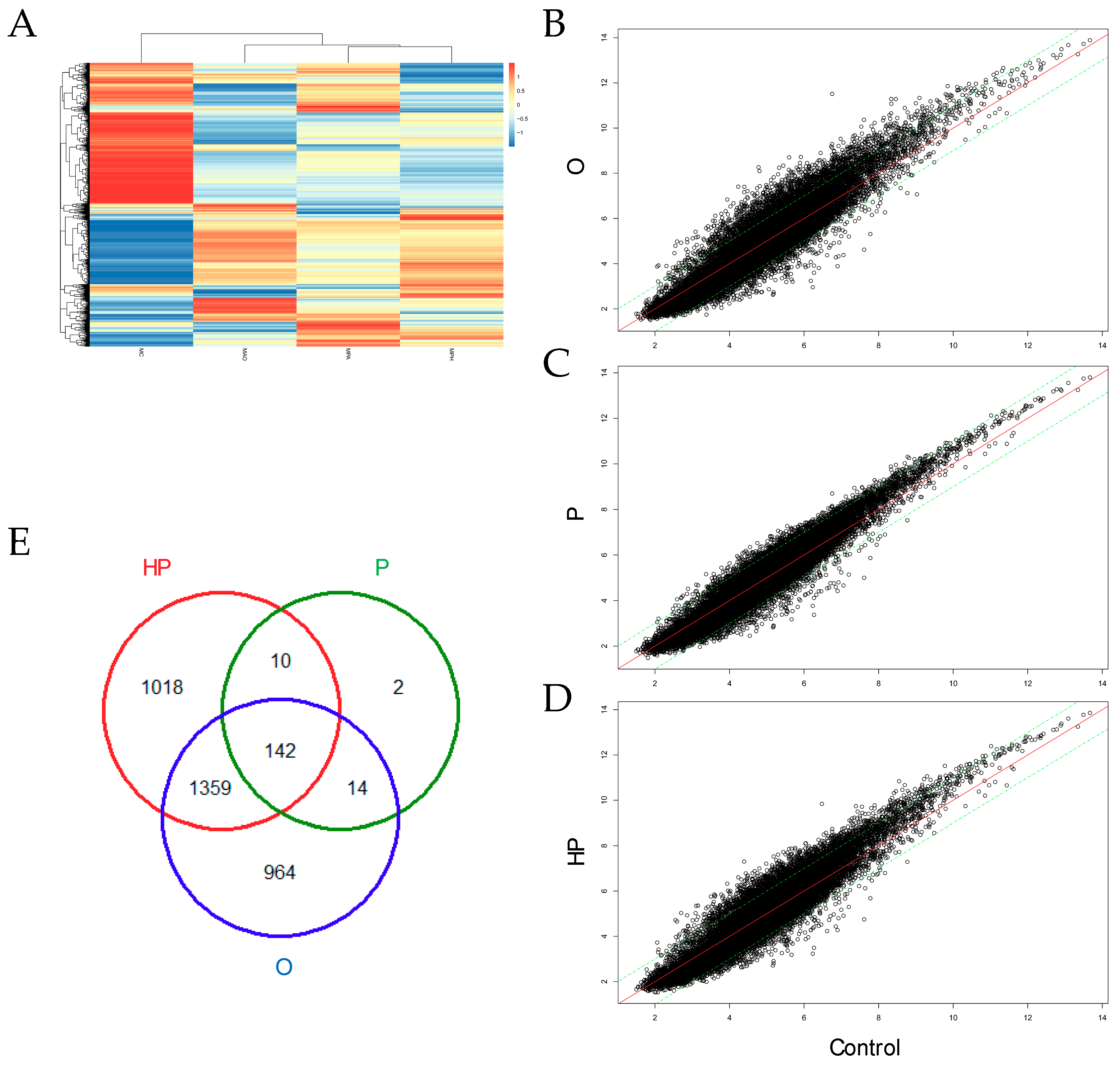
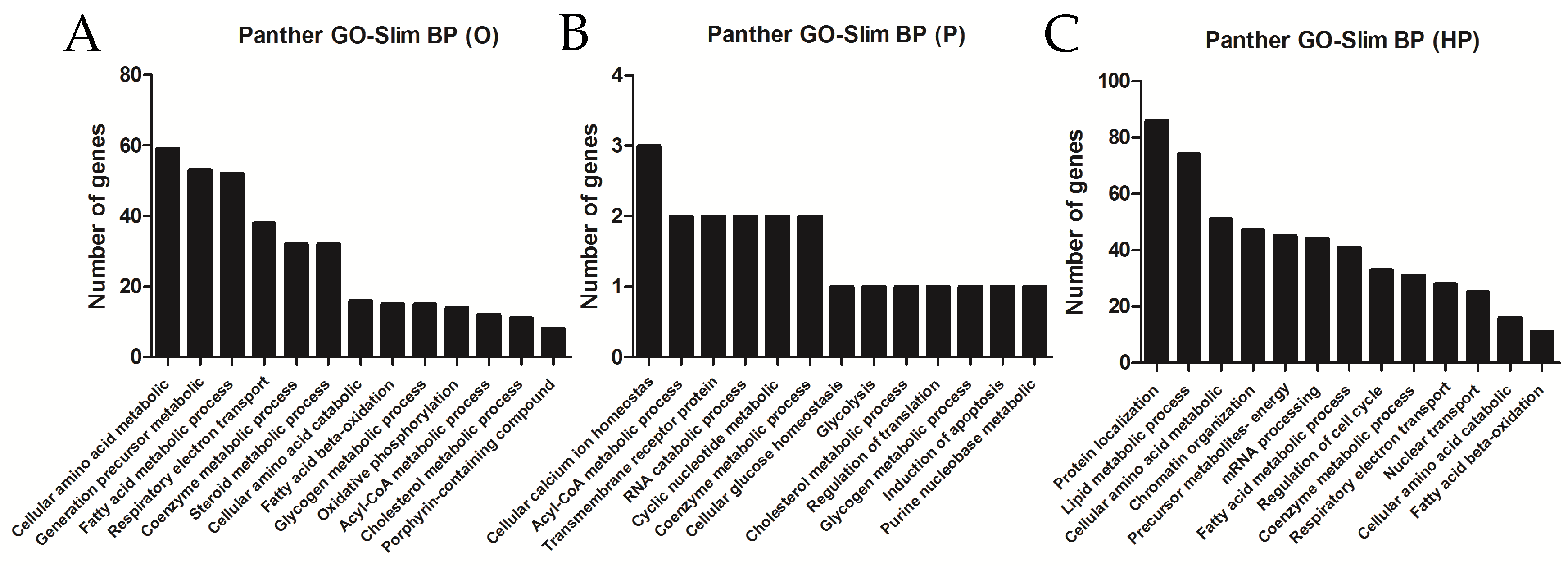
2.5. Transcriptomic Analyses
3. Discussion
4. Materials and Methods
4.1. Animals
4.2. Experimental Dietetic Protocol
4.3. Body Weight and Computed Tomography
4.4. Biological Sample Collection and Liver Histological Analyzes
4.5. Liver Fat Assessment
4.6. Liver Transcriptome
4.7. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| NAFLD | Nonalcoholic fatty liver disease |
| NAFL | Nonalcoholic fatty liver |
| NASH | Nonalcoholic steatohepatitis |
| FA | Fatty acids |
| MUFA | Monounsaturated fatty acids |
| PPAR | Peroxisome proliferator–activated receptors |
| SREBP-1 | Sterol-regulatory element binding protein 1 |
| TLR | Toll-like receptors |
| P | Palm |
| HP | Hybrid palm |
| O | Olive |
| CT | Computed tomography |
| NAS | Nonalcoholic fatty liver disease activity score |
| AST/ALT | Aspartate aminotransferase/alanine aminotransferase |
| DEG | Differentially expressed genes |
| SFA | Saturated fatty acids |
| NLRP3 | NOD-like receptor family pyrin containing 3 |
| T0 | Initial day |
| T1 | Final time |
| HU | Hounsfield unit |
| HDL | High-density lipoprotein cholesterol |
| LDL | Low-density lipoprotein cholesterol |
| VLDL | Very low-density lipoprotein cholesterol |
| GO | Gene ontology |
| BP | Biological process |
References
- Chalasani, N.; Younossi, Z.; Lavine, J.E.; Charlton, M.; Cusi, K.; Rinella, M.; Harrison, S.A.; Brunt, E.M.; Sanyal, A.J. The Diagnosis and Management of Nonalcoholic Fatty Liver Disease: Practice Guidance From the American Association for the Study of Liver Diseases. Hepatology 2018, 67, 328–357. [Google Scholar] [CrossRef] [PubMed]
- Bertot, L.C.; Adams, L. The Natural Course of Non-Alcoholic Fatty Liver Disease. Int. J. Mol. Sci. 2016, 17, 774. [Google Scholar] [CrossRef] [PubMed]
- Lonardo, A.; Byrne, C.D.; Caldwell, S.H.; Cortez-Pinto, H.; Targher, G. Global Epidemiology of Nonalcoholic Fatty Liver Disease: Meta-Analytic Assessment of Prevalence, Incidence, and Outcomes. Hepatology 2016, 64, 1388–1389. [Google Scholar] [CrossRef] [PubMed]
- Araújo, A.R.; Rosso, N.; Bedogni, G.; Tiribelli, C.; Bellentani, S. Global Epidemiology of Non-Alcoholic Fatty Liver Disease/Non-Alcoholic Steatohepatitis: What We Need in the Future. Liver Int. 2018, 38, 47–51. [Google Scholar] [CrossRef] [PubMed]
- Caligiuri, A.; Gentilini, A.; Marra, F. Molecular Pathogenesis of NASH. Int. J. Mol. Sci. 2016, 17, 1575. [Google Scholar] [CrossRef]
- Fan, J.G.; Cao, H.X. Role of Diet and Nutritional Management in Non-Alcoholic Fatty Liver Disease. J. Gastroenterol. Hepatol. 2013, 28, 81–87. [Google Scholar] [CrossRef]
- Nakamura, A.; Terauchi, Y. Lessons From Mouse Models of High-Fat Diet-Induced NAFLD. Int. J. Mol. Sci. 2013, 14, 21240–21257. [Google Scholar] [CrossRef]
- Yasutake, K.; Kohjima, M.; Kotoh, K.; Nakashima, M.; Nakamuta, M.; Enjoji, M. Dietary Habits and Behaviors Associated with Nonalcoholic Fatty Liver Disease. World J. Gastroenterol. 2014, 20, 1756. [Google Scholar] [CrossRef]
- Westerbacka, J.; Lammi, K.; HäKkinen, A.M.; Rissanen, A.; Salminen, I.; Aro, A.; Yki-JaRvinen, H. Dietary Fat Content Modifies Liver Fat in Overweight Nondiabetic Subjects. J. Clin. Endocrinol. Metab. 2005, 90, 2804–2809. [Google Scholar] [CrossRef]
- Van Herck, M.; Vonghia, L.; Francque, S. Animal models of nonalcoholic fatty liver disease—A starter’s guide. Nutrients 2017, 9, 1072. [Google Scholar] [CrossRef]
- Kakimoto, P.A.; Kowaltowski, A.J. Effects of high fat diets on rodent liver bioenergetics and oxidative imbalance. Redox Biol. 2016, 8, 216–225. [Google Scholar] [CrossRef] [PubMed]
- Juárez-Hernández, E.; Chávez-Tapia, N.C.; Uribe, M.; Barbero-Becerra, V.J. Role of Bioactive Fatty Acids in Nonalcoholic Fatty Liver Disease. Nutr. J. 2015, 15, 72. [Google Scholar] [CrossRef] [PubMed]
- Cansanção, K.; Monteiro, L.S.; Leite, N.C.; Dávalos, A.; Carmo, M.G.T.; Peres, W.A.F. Advanced Liver Fibrosis is Independently Associated with Palmitic Acid and Insulin Levels in Patients with Non-Alcoholic Fatty Liver Disease. Nutrients 2018, 10, 1586. [Google Scholar] [CrossRef] [PubMed]
- Nagao, K.; Yanagita, T. Bioactive Lipids in Metabolic Syndrome. Prog. Lipid Res. 2008, 47, 127–146. [Google Scholar] [CrossRef] [PubMed]
- Georgiadi, A.; Kersten, S. Mechanisms of Gene Regulation by Fatty Acids. Adv. Nutr. 2012, 3, 127–134. [Google Scholar] [CrossRef] [PubMed]
- Kucera, O.; Cervinkova, Z. Experimental Models of Non-Alcoholic Fatty Liver Disease in Rats. World J. Gastroenterol. 2014, 20, 8364. [Google Scholar] [CrossRef]
- Nam, Y.; Jmn, M.; Long, K. Composition and Thermal Analysis of Lard Stearin and Lard Olein. J. Oleo Sci. 2011, 60, 333–338. [Google Scholar] [CrossRef]
- Mba, O.I.; Dumont, M.J.; Ngadi, M. Palm Oil: Processing, Characterization and Utilization in the Food Industry—A Review. Food Biosci. 2015, 10, 26–41. [Google Scholar] [CrossRef]
- Orsavova, J.; Misurcova, L.; Ambrozova, J.V.; Vicha, R.; Mlcek, J. Fatty Acids Composition of Vegetable Oils and its Contribution to Dietary Energy Intake and Dependence of Cardiovascular Mortality on Dietary Intake of Fatty Acids. Int. J. Mol. Sci. 2015, 16, 12871–12890. [Google Scholar] [CrossRef]
- Mancini, A.; Imperlini, E.; Nigro, E.; Montagnese, C.; Daniele, A.; Orrù, S.; Buono, P. Biological and Nutritional Properties of Palm Oil and Palmitic Acid: Effects on Health. Molecules 2015, 20, 17339–17361. [Google Scholar] [CrossRef]
- EMBRAPA. BRS Manicoré: Híbrido Interespecífico Entre o Caiaué e o Dendezeiro Africano Recomendado Para Áreas de Incidência de Amarelecimento-Fatal. Available online: https://www.embrapa.br/solos/busca-de-publicacoes/-/publicacao/867099/brs-manicore-hibrido-interespecifico-entre-o-caiaue-e-o-dendezeiro-africano-recomendado-para-areas-de-incidencia-de-amarelecimento-fatal (accessed on 13 November 2018).
- Ojeda, M.; Borrero, M.; Sequeda, G.; Diez, O.; Castro, V.; García, Á.; Ruiz, Á.; Pacetti, D.; Frega, N.; Gagliardi, R.; et al. Hybrid Palm Oil (Elaeis Oleifera × Elaeis Guineensis) Supplementation Improves Plasma Antioxidant Capacity in Humans. Eur. J. Lipid Sci. Technol. 2017, 119, 1600070. [Google Scholar] [CrossRef]
- Lucci, P.; Borrero, M.; Ruiz, A.; Pacetti, D.; Frega, N.G.; Diez, O.; Ojeda, M.; Gagliardi, R.; Parra, L.; Angel, M. Palm Oil and Cardiovascular Disease: A Randomized Trial of the Effects of Hybrid Palm Oil Supplementation on Human Plasma Lipid Patterns. Food Funct. 2016, 7, 347–354. [Google Scholar] [CrossRef] [PubMed]
- Spreafico, F.; Sales, R.C.; Gil-Zamorano, J.; Medeiros, P.C.; Latasa, M.J.; Lima, M.R.; Souza, S.A.L.; Martin-Hernández, R.; Gómez-Coronado, D.; Iglesias-Gutierrez, E.; et al. Dietary Supplementation with Hybrid Palm Oil Alters Liver Function in the Common Marmoset. Sci. Rep. 2018, 8, 2765. [Google Scholar] [CrossRef] [PubMed]
- Monteiro, C.A.; Moubarac, J.C.; Cannon, G.; Ng, S.W.; Popkin, B. Ultra-Processed Products are Becoming Dominant in the Global Food System. Obes. Rev. 2013, 14, 21–28. [Google Scholar] [CrossRef] [PubMed]
- Hariri, N.; Thibault, L. High-Fat Diet-Induced Obesity in Animal Models. Nutr. Res. Rev. 2010, 23, 270–299. [Google Scholar] [CrossRef] [PubMed]
- Kanuri, G.; Bergheim, I. In Vitro and In Vivo Models of Non-Alcoholic Fatty Liver Disease (NAFLD). Int. J. Mol. Sci. 2013, 14, 11963–11980. [Google Scholar] [CrossRef] [PubMed]
- Lau, J.K.C.; Zhang, X.; Yu, J. Animal Models of Non-Alcoholic Fatty Liver Disease: Current Perspectives and Recent Advances. J. Pathol. 2017, 241, 36–44. [Google Scholar] [CrossRef] [PubMed]
- Schwingshackl, L.; Hoffmann, G. Monounsaturated Fatty Acids, Olive Oil and Health Status: A Systematic Review and Meta-Analysis of Cohort Studies. Lipids Health Dis. 2014, 13, 154. [Google Scholar] [CrossRef] [PubMed]
- Elias, S.; Wisam, S.; Luai, A.; Massad, B.; Nimer, A. Lipotoxicity in Obesity: Benefit of Olive Oil. Adv. Exp. Med. Biol. 2017, 960, 607–617. [Google Scholar] [CrossRef] [PubMed]
- Garaulet, M.; Marin, C.; Pérez-Llamas, F.; Canteras, M.; Tebar, F.J.; Zamora, S. Adiposity and Dietary Intake in Cardiovascular Risk in an Obese Population from a Mediterranean Area. J. Physiol. Biochem. 2004, 60, 39–49. [Google Scholar] [CrossRef] [PubMed]
- Benner, K.G.; Sasaki, A.; Gowen, D.R.; Weaver, A.; Connor, W.E. The Differential Effect of Eicosapentaenoic Acid and Oleic Acid on Lipid Synthesis and VLDL Secretion in Rabbit Hepatocytes. Lipids 1990, 25, 534–540. [Google Scholar] [CrossRef] [PubMed]
- De Velasco, P.C.; Chicaybam, G.; Ramos-Filho, D.M.; Dos Santos, R.M.; Mairink, C.; Sardinha, F.L.; El-Bacha, T.; Galina, A.; Do Carmo, M.G.T. Maternal Intake of Trans-Unsaturated or Interesterified Fatty Acids During Pregnancy and Lactation Modifies Mitochondrial Bioenergetics in the Liver of Adult Offspring in Mice. Br. J. Nutr. 2017, 118, 41–52. [Google Scholar] [CrossRef] [PubMed]
- Zhu, W.; Feng, P.P.; He, K.; Li, S.W.; Gong, J.P. Liraglutide Protects Non-Alcoholic Fatty Liver Disease Via Inhibiting NLRP3 Inflammasome Activation in a Mouse Model Induced by High-Fat Diet. Biochem. Biophys. Res. Commun. 2018, 505, 523–529. [Google Scholar] [CrossRef]
- Dowman, J.K.; Hopkins, L.J.; Reynolds, G.M.; Nikolaou, N.; Armstrong, M.J.; Shaw, J.C.; Houlihan, D.D.; Lalor, P.F.; Tomlinson, J.W.; Hübscher, S.G.; et al. Development of hepatocellular carcinoma in a murine model of nonalcoholic steatohepatitis induced by use of a high-fat/fructose diet and sedentary lifestyle. Am. J. Pathol. 2014, 184, 1550–1561. [Google Scholar] [CrossRef] [PubMed]
- Karin, M.; Dhar, D. Liver carcinogenesis: From naughty chemicals to soothing fat and the surprising role of NRF2. Carcinogenesis 2016, 37, 541–546. [Google Scholar] [CrossRef]
- Zhang, H.; Temel, R.E.; Martel, C. Cholesterol and Lipoprotein Metabolism: Early Career Committee Contribution. Arterioscler. Thromb. Vasc. Biol. 2014, 34, 1791–1794. [Google Scholar] [CrossRef] [PubMed]
- Katan, M.B.; Zock, P.L.; Mensink, R.P. Effects of Fats and Fatty Acids on Blood Lipids in Humans: An Overview. Am. J. Clin. Nutr. 1994, 60, S1017–S1022. [Google Scholar] [CrossRef] [PubMed]
- Sparks, J.D.; Sparks, C.E.; Adeli, K. Selective Hepatic Insulin Resistance, VLDL Overproduction, and Hypertriglyceridemia. Arterioscler. Thromb. Vasc. Biol. 2012, 32, 2104–2112. [Google Scholar] [CrossRef] [PubMed]
- Giannini, E.; Botta, F.; Fasoli, A.; Ceppa, P.; Risso, D.; Lantieri, P.B.; Celle, G.; Testa, R. Progressive Liver Functional Impairment is Associated With an Increase in AST/ALT Ratio. Dig. Dis. Sci. 1999, 44, 1249–1253. [Google Scholar] [CrossRef]
- Zhao, L.; Cheng, J.; Chen, Y.; Li, Q.; Han, B.; Chen, Y.; Xia, F.; Chen, C.; Lin, D.; Yu, X.; et al. Serum Alanine Aminotransferase/Aspartate Aminotransferase Ratio Is One Of The Best Markers Of Insulin Resistance In The Chinese Population. Nutr. Metab. (London) 2017, 14, 64. [Google Scholar] [CrossRef]
- Yadav, D.; Choi, E.; Ahn, S.V.; Baik, S.K.; Zoo Cho, Y.; Koh, S.B.; Huh, J.H.; Chang, Y.; Sung, K.C.; Kim, J.Y. Incremental Predictive Value of Serum AST-to-ALT Ratio for Incident Metabolic Syndrome: The ARIRANG Study. PLoS ONE 2016, 11, E0161304. [Google Scholar] [CrossRef] [PubMed]
- Kwo, P.Y.; Cohen, S.M.; Lim, J.K. ACG Clinical Guideline: Evaluation of Abnormal Liver Chemistries. Am. J. Gastroenterol. 2017, 112, 18. [Google Scholar] [CrossRef] [PubMed]
- Pantsari, M.W.; Harrison, S.A. Nonalcoholic Fatty Liver Disease Presenting with an Isolated Elevated Alkaline Phosphatase. J. Clin. Gastroenterol. 2006, 40, 633–635. [Google Scholar] [CrossRef] [PubMed]
- Ndrepepa, G.; Kastrati, A. Gamma-Glutamyl Transferase and Cardiovascular Disease. Ann. Transl. Med. 2016, 4. [Google Scholar] [CrossRef] [PubMed]
- Ndrepepa, G.; Colleran, R.; Kastrati, A. Gamma-Glutamyl Transferase and the Risk of Atherosclerosis and Coronary Heart Disease. Clin. Chim. Acta 2017, 476, 130–138. [Google Scholar] [CrossRef]
- Kris-Etherton, P.M.; Ho, C.Y.; Fosmire, M.A. The Effect of Dietary Fat Saturation on Plasma and Hepatic Lipoproteins in the Rat. J. Nutr. 1994, 114, 1675–1682. [Google Scholar] [CrossRef]
- Meidan, E.; Kolesnikov, Y.; Tirosh, O. High Fat Diets Composed of Palm Stearin and Olive Oil Equally Exacerbate Liver Inflammatory Damage and Metabolic Stress in Mice. Mol. Nutr. Food Res. 2018, 1700915. [Google Scholar] [CrossRef]
- Djohan, Y.F.; Badia, E.; Bonafos, B.; Fouret, G.; Lauret, C.; Dupuy, A.M.; Pinot, E.; Sutra, T.; Gaillet, S.; Lambert, K.; et al. High Dietary Intake of Palm Oils Compromises Glucose Tolerance Whereas High Dietary Intake of Olive Oil Compromises Liver Lipid Metabolism and Integrity. Eur. J. Nutr. 2018, 1–17. [Google Scholar] [CrossRef]
- Lopes, L.L.; Rocha, D.M.U.; Silva, A.D.; Peluzio, M.D.C.G.; Bressan, J.; Hermsdorff, H.H.M. Postprandial Lipid Response to High-Saturated and High-Monounsaturated Fat Meals in Normal-Weight or Overweight Women. J. Am. Coll. Nutr. 2018, 37, 308–315. [Google Scholar] [CrossRef]
- Roberts, M.D.; Mobley, C.B.; Toedebush, R.G.; Heese, A.J.; Zhu, C.; Krieger, A.E.; Cruthirds, C.L.; Lockwood, C.M.; Hofheins, J.C.; Wiedmeyer, C.E.; et al. Western Diet-Induced Hepatic Steatosis and Alterations in the Liver Transcriptome in Adult Brown-Norway Rats. BMC Gastroenterol. 2015, 15, 151. [Google Scholar] [CrossRef]
- Eaton, S.; Bartlett, K.; Quant, P.A. Carnitine Palmitoyl Transferase I and the Control of Β-Oxidation in Heart Mitochondria. Biochem. Biophys. Res. Commun. 2001, 285, 537–539. [Google Scholar] [CrossRef] [PubMed]
- Pop, M.; Raluca, P. Using Abdominal CT Data for Visceral Fat Evaluation. Acta Med. Marisiensis 2013, 59, 254–256. [Google Scholar] [CrossRef]
- Irving, B.A.; Weltman, J.Y.; Brock, D.W.; Davis, C.K.; Gaesser, G.A.; Weltman, A. NIH Imagej And Slice-O-Matic Computed Tomography Imaging Software To Quantify Soft Tissue. Obesity (Silver Spring) 2007, 15, 370–376. [Google Scholar] [CrossRef] [PubMed]
- Van Vugt, J.L.; Levolger, S.; Gharbharan, A.; Koek, M.; Niessen, W.J.; Burger, J.W.; Willemsen, S.P.; De Bruin, R.W.; Ijzermans, J.N. A Comparative Study of Software Programmes for Cross-Sectional Skeletal Muscle and Adipose Tissue Measurements on Abdominal Computed Tomography Scans of Rectal Cancer Patients. J. Cachexia Sarcopenia Muscle 2017, 8, 285–297. [Google Scholar] [CrossRef] [PubMed]
- Kleiner, D.E.; Brunt, E.M.; Van Natta, M.; Behling, C.; Contos, M.J.; Cummings, O.W.; Ferrell, L.D.; Liu, Y.C.; Torbenson, M.S.; Unalp-Arida, A.; et al. Design and Validation of a Histological Scoring System for Nonalcoholic Fatty Liver Disease. Hepatology 2005, 41, 1313–1321. [Google Scholar] [CrossRef] [PubMed]
- Bligh, E.G.; Dyer, W.J. A Rapid Method of Total Lipid Extraction and Purification. Can. J. Biochem. Physiol. 1959, 37, 911–917. [Google Scholar] [CrossRef] [PubMed]
| Group | Liver Fat (%) | Statistical Significance Versus Control | Statistical Significance Versus Olive oil | Statistical Significance Versus Palm Oil | Statistical Significance Versus Hybrid Palm Oil |
|---|---|---|---|---|---|
| Control | 14.71 ± 0.70 | - | p = 0.1189 | p < 0.0001 | p < 0.0001 |
| Olive oil | 16.46 ± 0.34 | p = 0.1189 | - | p < 0.0001 | p < 0.0001 |
| Palm oil | 33.94 ± 1.13 | p < 0.0001 | p < 0.0001 | - | p < 0.0001 |
| Hybrid Palm oil | 42.76 ± 1.58 | p < 0.0001 | p < 0.0001 | p < 0.0001 | - |
| Group | Grade of Steatosis | Lobular Inflammation | Ballooning | NAFLD Activity Score |
|---|---|---|---|---|
| Control | 1 | 0 | 0 | 1 |
| Olive oil | 1 | 2 | 0 | 3 |
| Palm oil | 2 | 2 | 1 | 5 |
| Hybrid Palm oil | 2 | 2 | 1 | 5 |
| Ingredients | g/100 g |
|---|---|
| Ground corn | 8.80 |
| Wheat middlings | 8.50 |
| Rice bran | 8.00 |
| Sucrose | 19.50 |
| Dehulled soybean meal | 5.00 |
| Casein | 7.00 |
| Powder milk | 7.00 |
| Albumin | 11.20 |
| Chicken meal | 2.90 |
| Soybean oil | 2.00 |
| P, HP, or O oils | 14.30 |
| Fiber | 2.00 |
| Vitamins and minerals mix | 3,86 |
| Butylhydroxytoluene | 0.02 |
| Kcal/100 g | 422.08 |
| Protein | 16% |
| Carbohydrate | 43% |
| Lipid | 41% |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sales, R.C.; Medeiros, P.C.; Spreafico, F.; De Velasco, P.C.; Gonçalves, F.K.A.; Martín-Hernández, R.; Mantilla-Escalante, D.C.; Gil-Zamorano, J.; Peres, W.A.F.; De Souza, S.A.L.; et al. Olive Oil, Palm Oil, and Hybrid Palm Oil Distinctly Modulate Liver Transcriptome and Induce NAFLD in Mice Fed a High-Fat Diet. Int. J. Mol. Sci. 2019, 20, 8. https://doi.org/10.3390/ijms20010008
Sales RC, Medeiros PC, Spreafico F, De Velasco PC, Gonçalves FKA, Martín-Hernández R, Mantilla-Escalante DC, Gil-Zamorano J, Peres WAF, De Souza SAL, et al. Olive Oil, Palm Oil, and Hybrid Palm Oil Distinctly Modulate Liver Transcriptome and Induce NAFLD in Mice Fed a High-Fat Diet. International Journal of Molecular Sciences. 2019; 20(1):8. https://doi.org/10.3390/ijms20010008
Chicago/Turabian StyleSales, Rafael C., Priscylla C. Medeiros, Flavia Spreafico, Patrícia C. De Velasco, Fernanda K. A. Gonçalves, Roberto Martín-Hernández, Diana C. Mantilla-Escalante, Judit Gil-Zamorano, Wilza A. F. Peres, Sergio A. L. De Souza, and et al. 2019. "Olive Oil, Palm Oil, and Hybrid Palm Oil Distinctly Modulate Liver Transcriptome and Induce NAFLD in Mice Fed a High-Fat Diet" International Journal of Molecular Sciences 20, no. 1: 8. https://doi.org/10.3390/ijms20010008
APA StyleSales, R. C., Medeiros, P. C., Spreafico, F., De Velasco, P. C., Gonçalves, F. K. A., Martín-Hernández, R., Mantilla-Escalante, D. C., Gil-Zamorano, J., Peres, W. A. F., De Souza, S. A. L., Dávalos, A., & Tavares do Carmo, M. G. (2019). Olive Oil, Palm Oil, and Hybrid Palm Oil Distinctly Modulate Liver Transcriptome and Induce NAFLD in Mice Fed a High-Fat Diet. International Journal of Molecular Sciences, 20(1), 8. https://doi.org/10.3390/ijms20010008