New Insights in Prostate Cancer Development and Tumor Therapy: Modulation of Nuclear Receptors and the Specific Role of Liver X Receptors
Abstract
1. Introduction
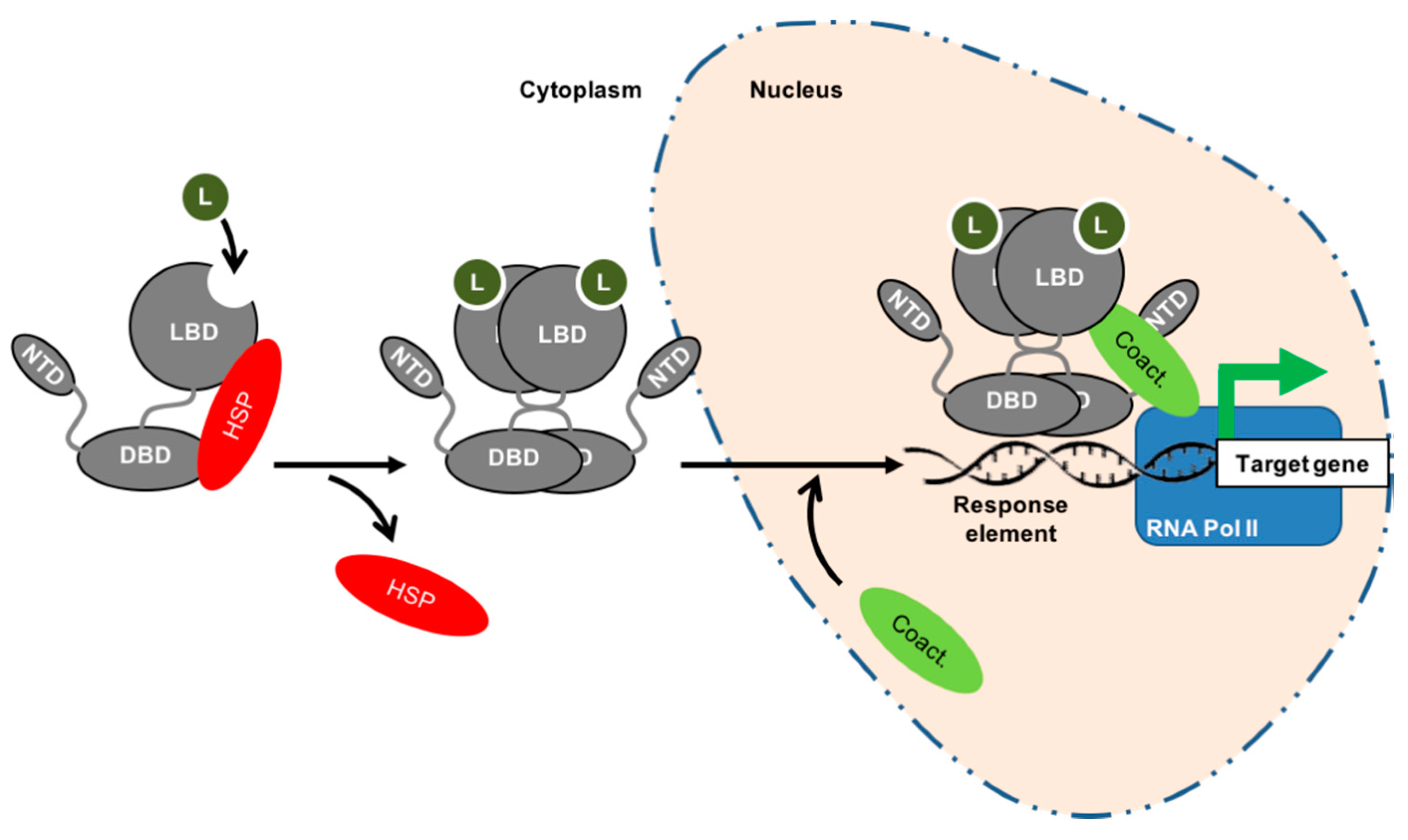
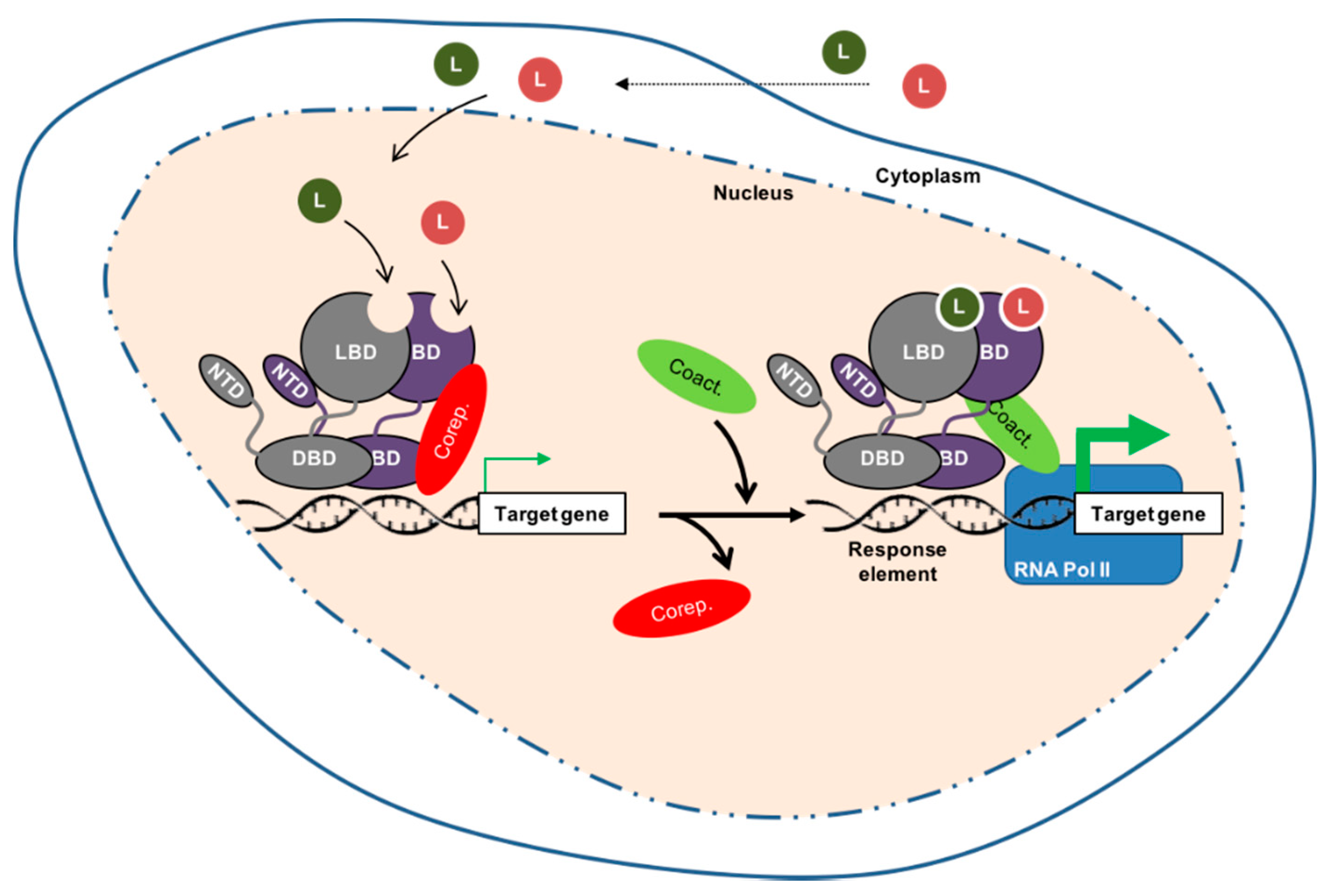
2. Nuclear Receptors Are Fundamental for Prostate Physiology
2.1. Role of AR in Prostate Physiology and in PCa
2.1.1. Androgens Are Involved in Prostate Differentiation, Growth, and Functioning
2.1.2. Androgens Drive Proliferation of Luminal Epithelial Cells in PCa
2.2. Role of ER in Prostate Physiology and in PCa
2.3. LXRs as Emerging Factors Regulating Prostate Physiology
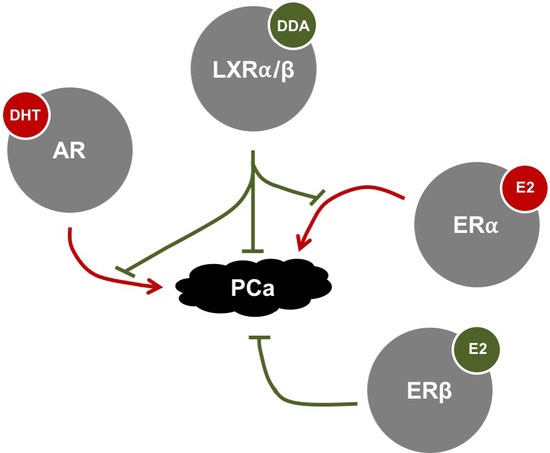
2.4. Androgens, Estrogens, and Oxysterols Interact in the Prostate
2.4.1. LXRs Regulate the Availability of Active Steroids
2.4.2. AR, ER, and LXR Interaction within the Prostate
2.5. NR Activity Can Be Altered in Prostate Physiology by Environmental Disrupting Chemicals
3. Lipid Metabolism, Angiogenesis and Immunity Are Altered in PCa
3.1. Lipid and Cholesterol Metabolism in Cancer Cells
3.2. Modification of Angiogenesis and Immunity
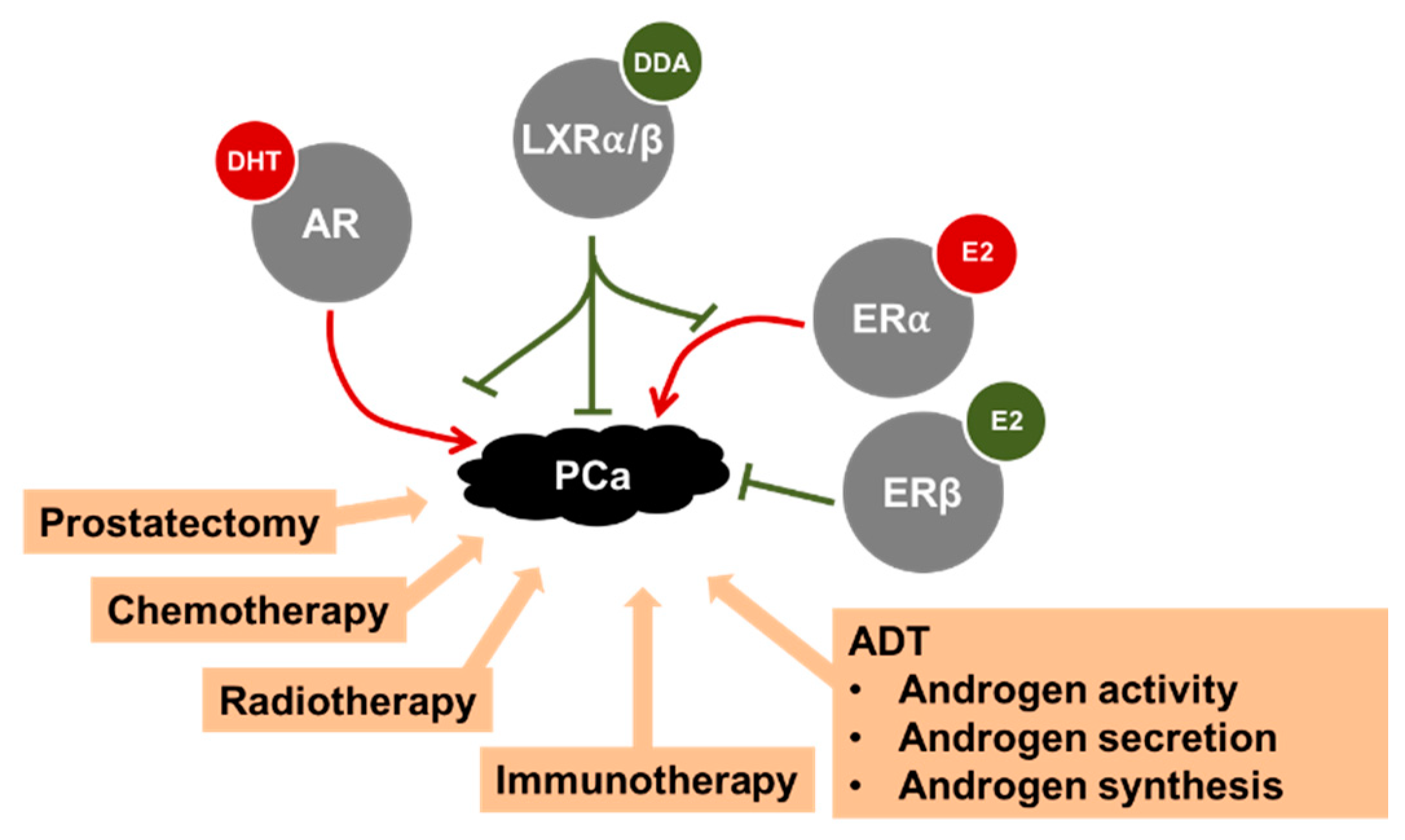
4. Management of Prostate Cancer and Treatments
4.1. Local PCa
4.2. Advanced and Metastatic PCa
4.3. Castration Resistant Prostate Cancer
4.4. New Therapeutic Strategies for PCa Beside Steroids and NRs
5. Conclusions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| ABCA1/G1 | ATP-binding cassette A1/G1 |
| ADT | androgen deprivation therapy |
| AF1/2 | activating function 1/2 |
| AR | androgen receptor |
| COX2 | cyclooxygenase 2 |
| DHT | dihydrotestosterone |
| CRPC | castration-resistant prostate cancer |
| mCRPC | metastatic CRPC |
| ER | estrogen receptor |
| EST | estrogen sulfotransferase |
| EZH2 | enhancer of Zeste homolog 2 |
| Her-2/neu | receptor tyrosine-protein kinase erbB-2 |
| HMGCR | hydroxy-3-methylglutaryl-coenzyme A reductase |
| IDOL | inducible degrader of the LDL receptor |
| iNOS | inducible nitric oxide synthase |
| JAK/STAT | Janus kinase/signal transducers and activators of transcription |
| LDLR | LDL receptor |
| LXR | liver X receptor |
| MAPK | mitogen-activated protein kinases |
| MSMB | beta-microseminoprotein |
| NKX3.1 | homeobox protein Nkx-3.1 |
| NR | nuclear receptor |
| PCa | prostate cancer |
| PI3K | phosphoinositide 3-kinase |
| PSA | prostate specific antigen |
| RXR | retinoid X receptor |
| SOCS3 | suppressor of cytokine signaling 3 |
| SR5A2 | 5α-reductase type 2 |
| SREBP | sterol response element binding protein |
| SLiMs | selective liver X receptor modulators |
| SULT2A1 | sulfotransferase family 2A member 1 |
| VEGF | vascular endothelial growth factor |
| Wnt | Wingless/Integrated |
References
- Bostwick, D.G.; Burke, H.B.; Djakiew, D.; Euling, S.; Ho, S.; Landolph, J.; Morrison, H.; Sonawane, B.; Shifflett, T.; Waters, D.J.; et al. Human prostate cancer risk factors. Cancer 2004, 101, 2371–2490. [Google Scholar] [CrossRef] [PubMed]
- Hsing, A.W.; Chu, L.W.; Stanczyk, F.Z. Androgen and prostate cancer: Is the hypothesis dead? Cancer Epidemiol. Biomark. 2008, 17, 2525–2530. [Google Scholar] [CrossRef] [PubMed]
- Härkönen, P.L.; Mäkelä, S.I. Role of estrogens in development of prostate cancer. J. Steroid Biochem. Mol. Biol. 2004, 92, 297–305. [Google Scholar] [CrossRef] [PubMed]
- Prins, G.S.; Korach, K.S. The role of estrogens and estrogen receptors in normal prostate growth and disease. Steroids 2008, 73, 233–244. [Google Scholar] [CrossRef] [PubMed]
- Evans, R.M.; Mangelsdorf, D.J. Nuclear Receptors, RXR, and the Big Bang. Cell 2014, 157, 255–266. [Google Scholar] [CrossRef] [PubMed]
- McKenna, N.J. Research Resources for Nuclear Receptor Signaling Pathways. Mol. Pharmacol. 2016, 90, 153–159. [Google Scholar] [CrossRef] [PubMed]
- Helsen, C.; Claessens, F. Looking at nuclear receptors from a new angle. Mol. Cell. Endocrinol. 2014, 382, 97–106. [Google Scholar] [CrossRef] [PubMed]
- Huggins, C.; Hodges, C.V. Studies on Prostatic Cancer. I. The Effect of Castration, of Estrogen and of Androgen Injection on Serum Phosphatases in Metastatic Carcinoma of the Prostate. Cancer Res. 1941, 1, 293–297. [Google Scholar] [CrossRef]
- Bonkhoff, H. Estrogen receptor signaling in prostate cancer: Implications for carcinogenesis and tumor progression. Prostate 2018, 78, 2–10. [Google Scholar] [CrossRef] [PubMed]
- Maqdasy, S.; Trousson, A.; Tauveron, I.; Volle, D.H.; Baron, S.; Lobaccaro, J.-M.A. Once and for all, LXRα and LXRβ are gatekeepers of the endocrine system. Mol. Aspects Med. 2016, 49, 31–46. [Google Scholar] [CrossRef] [PubMed]
- De Boussac, H.; Pommier, A.J.; Dufour, J.; Trousson, A.; Caira, F.; Volle, D.H.; Baron, S.; Lobaccaro, J.-M.A. LXR, prostate cancer and cholesterol: The Good, the Bad and the Ugly. Am. J. Cancer Res. 2013, 3, 58–69. [Google Scholar] [PubMed]
- Dufour, J.; Pommier, A.; Alves, G.; de Boussac, H.; Lours-Calet, C.; Volle, D.H.; Lobaccaro, J.-M.A.; Baron, S. Lack of Liver X Receptors Leads to Cell Proliferation in a Model of Mouse Dorsal Prostate Epithelial Cell. PLoS ONE 2013, 8, e58876. [Google Scholar] [CrossRef] [PubMed]
- Pommier, A.J.C.; Alves, G.; Viennois, E.; Bernard, S.; Communal, Y.; Sion, B.; Marceau, G.; Damon, C.; Mouzat, K.; Caira, F.; et al. Liver X Receptor activation downregulates AKT survival signaling in lipid rafts and induces apoptosis of prostate cancer cells. Oncogene 2010, 29, 2712–2723. [Google Scholar] [CrossRef] [PubMed]
- Abdelgadir, S.E.; Connolly, P.B.; Resko, J.A. Androgen binding in peripheral tissues of fetal rhesus macaques: Effects of androgen metabolism in liver. J. Steroid Biochem. Mol. Biol. 1990, 37, 545–551. [Google Scholar] [CrossRef]
- Heinlein, C.A.; Chang, C. The Roles of Androgen Receptors and Androgen-Binding Proteins in Nongenomic Androgen Actions. Mol. Endocrinol. 2002, 16, 2181–2187. [Google Scholar] [CrossRef] [PubMed]
- Yeh, S.; Tsai, M.-Y.; Xu, Q.; Mu, X.-M.; Lardy, H.; Huang, K.-E.; Lin, H.; Yeh, S.-D.; Altuwaijri, S.; Zhou, X.; et al. Generation and characterization of androgen receptor knockout (ARKO) mice: An in vivo model for the study of androgen functions in selective tissues. Proc. Natl. Acad. Sci. USA 2002, 99, 13498–13503. [Google Scholar] [CrossRef] [PubMed]
- Cunha, G.R. The role of androgens in the epithelio-mesenchymal interactions involved in prostatic morphogenesis in embryonic mice. Anat. Rec. 1973, 175, 87–96. [Google Scholar] [CrossRef] [PubMed]
- Lu, W.; Luo, Y.; Kan, M.; McKeehan, W.L. Fibroblast Growth Factor-10 A second candidate stromal to epithelial cell andromedin in prostate. J. Biol. Chem. 1999, 274, 12827–12834. [Google Scholar] [CrossRef] [PubMed]
- Planz, B.; Aretz, H.T.; Wang, Q.; Tabatabaei, S.; Kirley, S.D.; Lin, C.W.; McDougal, W.S. Immunolocalization of the keratinocyte growth factor in benign and neoplastic human prostate and its relation to androgen receptor. Prostate 1999, 41, 233–242. [Google Scholar] [CrossRef]
- Wikström, P.; Westin, P.; Stattin, P.; Damber, J.E.; Bergh, A. Early castration-induced upregulation of transforming growth factor beta1 and its receptors is associated with tumor cell apoptosis and a major decline in serum prostate-specific antigen in prostate cancer patients. Prostate 1999, 38, 268–277. [Google Scholar] [CrossRef]
- Bonkhoff, H.; Remberger, K. Widespread distribution of nuclear androgen receptors in the basal cell layer of the normal and hyperplastic human prostate. Virchows Arch. Pathol. Anat. Histopathol. 1993, 422, 35–38. [Google Scholar] [CrossRef]
- Mirosevich, J.; Bentel, J.M.; Zeps, N.; Redmond, S.L.; D’Antuono, M.F.; Dawkins, H.J. Androgen receptor expression of proliferating basal and luminal cells in adult murine ventral prostate. J. Endocrinol. 1999, 162, 341–350. [Google Scholar] [CrossRef] [PubMed]
- Berry, S.J.; Isaacs, J.T. Comparative Aspects of Prostatic Growth and Androgen Metabolism with Aging in the Dog Versus the Rat. Endocrinology 1984, 114, 511–520. [Google Scholar] [CrossRef] [PubMed]
- English, H.F.; Kyprianou, N.; Isaacs, J.T. Relationship between DNA fragmentation and apoptosis in the programmed cell death in the rat prostate following castration. Prostate 1989, 15, 233–250. [Google Scholar] [CrossRef] [PubMed]
- Gao, J.; Isaacs, J.T. Development of an androgen receptor-null model for identifying the initiation site for androgen stimulation of proliferation and suppression of programmed (apoptotic) death of PC-82 human prostate cancer cells. Cancer Res. 1998, 58, 3299–3306. [Google Scholar] [PubMed]
- Gao, J.; Arnold, J.T.; Isaacs, J.T. Conversion from a paracrine to an autocrine mechanism of androgen-stimulated growth during malignant transformation of prostatic epithelial cells. Cancer Res. 2001, 61, 5038–5044. [Google Scholar] [PubMed]
- Memarzadeh, S.; Cai, H.; Janzen, D.M.; Xin, L.; Lukacs, R.; Riedinger, M.; Zong, Y.; DeGendt, K.; Verhoeven, G.; Huang, J.; et al. Role of autonomous androgen receptor signaling in prostate cancer initiation is dichotomous and depends on the oncogenic signal. Proc. Natl. Acad. Sci. USA 2011, 108, 7962–7967. [Google Scholar] [CrossRef] [PubMed]
- Massie, C.E.; Lynch, A.; Ramos-Montoya, A.; Boren, J.; Stark, R.; Fazli, L.; Warren, A.; Scott, H.; Madhu, B.; Sharma, N.; et al. The androgen receptor fuels prostate cancer by regulating central metabolism and biosynthesis. EMBO J. 2011, 30, 2719–2733. [Google Scholar] [CrossRef] [PubMed]
- Jin, H.-J.; Kim, J.; Yu, J. Androgen receptor genomic regulation. Transl. Androl. Urol. 2013, 2, 157–177. [Google Scholar] [CrossRef] [PubMed]
- Sharma, N.L.; Massie, C.E.; Ramos-Montoya, A.; Zecchini, V.; Scott, H.E.; Lamb, A.D.; MacArthur, S.; Stark, R.; Warren, A.Y.; Mills, I.G.; et al. The androgen receptor induces a distinct transcriptional program in castration-resistant prostate cancer in man. Cancer Cell 2013, 23, 35–47. [Google Scholar] [CrossRef] [PubMed]
- Yaşar, P.; Ayaz, G.; User, S.D.; Güpür, G.; Muyan, M. Molecular mechanism of estrogen-estrogen receptor signaling. Reprod. Med. Biol. 2017, 16, 4–20. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Hsu, I.; Wolfe, A.; Radovick, S.; Huang, K.; Yu, S.; Chang, C.; Messing, E.M.; Yeh, S. Defects of prostate development and reproductive system in the estrogen receptor-alpha null male mice. Endocrinology 2009, 150, 251–259. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Yeh, C.-R.; Chang, H.-C.; Vitkus, S.; Wen, X.-Q.; Bhowmick, N.A.; Wolfe, A.; Yeh, S. Loss of epithelial oestrogen receptor α inhibits oestrogen-stimulated prostate proliferation and squamous metaplasia via in vivo tissue selective knockout models. J. Pathol. 2012, 226, 17–27. [Google Scholar] [CrossRef] [PubMed]
- Vitkus, S.; Yeh, C.-R.; Lin, H.-H.; Hsu, I.; Yu, J.; Chen, M.; Yeh, S. Distinct function of estrogen receptor α in smooth muscle and fibroblast cells in prostate development. Mol. Endocrinol. 2013, 27, 38–49. [Google Scholar] [CrossRef] [PubMed]
- Leav, I.; Lau, K.M.; Adams, J.Y.; McNeal, J.E.; Taplin, M.E.; Wang, J.; Singh, H.; Ho, S.M. Comparative studies of the estrogen receptors beta and alpha and the androgen receptor in normal human prostate glands, dysplasia, and in primary and metastatic carcinoma. Am. J. Pathol. 2001, 159, 79–92. [Google Scholar] [CrossRef]
- Lau, K.M.; LaSpina, M.; Long, J.; Ho, S.M. Expression of estrogen receptor (ER)-α and ER-β in normal and malignant prostatic epithelial cells: Regulation by methylation and involvement in growth regulation. Cancer Res. 2000, 60, 3175–3182. [Google Scholar] [PubMed]
- Royuela, M.; de Miguel, M.P.; Bethencourt, F.R.; Sánchez-Chapado, M.; Fraile, B.; Arenas, M.I.; Paniagua, R. Estrogen receptors α and β in the normal, hyperplastic and carcinomatous human prostate. J. Endocrinol. 2001, 168, 447–454. [Google Scholar] [CrossRef] [PubMed]
- Imamov, O.; Morani, A.; Shim, G.-J.; Omoto, Y.; Thulin-Andersson, C.; Warner, M.; Gustafsson, J.-A. Estrogen receptor β regulates epithelial cellular differentiation in the mouse ventral prostate. Proc. Natl. Acad. Sci. USA 2004, 101, 9375–9380. [Google Scholar] [CrossRef] [PubMed]
- Latil, A.; Bièche, I.; Vidaud, D.; Lidereau, R.; Berthon, P.; Cussenot, O.; Vidaud, M. Evaluation of androgen, estrogen (ER α and ER β), and progesterone receptor expression in human prostate cancer by real-time quantitative reverse transcription-polymerase chain reaction assays. Cancer Res. 2001, 61, 1919–1926. [Google Scholar] [PubMed]
- Bonkhoff, H.; Fixemer, T.; Hunsicker, I.; Remberger, K. Estrogen Receptor Expression in Prostate Cancer and Premalignant Prostatic Lesions. Am. J. Pathol. 1999, 155, 641–647. [Google Scholar] [CrossRef]
- Slusarz, A.; Jackson, G.A.; Day, J.K.; Shenouda, N.S.; Bogener, J.L.; Browning, J.D.; Fritsche, K.L.; MacDonald, R.S.; Besch-Williford, C.L.; Lubahn, D.B. Aggressive prostate cancer is prevented in ERαKO mice and stimulated in ERβKO TRAMP mice. Endocrinology 2012, 153, 4160–4170. [Google Scholar] [CrossRef] [PubMed]
- Mohler, J.L.; Gregory, C.W.; Ford, O.H.; Kim, D.; Weaver, C.M.; Petrusz, P.; Wilson, E.M.; French, F.S. The androgen axis in recurrent prostate cancer. Clin. Cancer Res. 2004, 10, 440–448. [Google Scholar] [CrossRef] [PubMed]
- Ellem, S.J.; Schmitt, J.F.; Pedersen, J.S.; Frydenberg, M.; Risbridger, G.P. Local aromatase expression in human prostate is altered in malignancy. J. Clin. Endocrinol. Metab. 2004, 89, 2434–2441. [Google Scholar] [CrossRef] [PubMed]
- Montgomery, R.B.; Mostaghel, E.A.; Vessella, R.; Hess, D.L.; Kalhorn, T.F.; Higano, C.S.; True, L.D.; Nelson, P.S. Maintenance of intratumoral androgens in metastatic prostate cancer: A mechanism for castration-resistant tumor growth. Cancer Res. 2008, 68, 4447–4454. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Tontonoz, P. Liver X receptors in lipid signalling and membrane homeostasis. Nat. Rev. Endocrinol. 2018, 14, 452–463. [Google Scholar] [CrossRef] [PubMed]
- Janowski, B.A.; Willy, P.J.; Devi, T.R.; Falck, J.R.; Mangelsdorf, D.J. An oxysterol signalling pathway mediated by the nuclear receptor LXRα. Nature 1996, 383, 728–731. [Google Scholar] [CrossRef] [PubMed]
- Janowski, B.A.; Grogan, M.J.; Jones, S.A.; Wisely, G.B.; Kliewer, S.A.; Corey, E.J.; Mangelsdorf, D.J. Structural requirements of ligands for the oxysterol liver X receptors LXRα and LXRβ. Proc. Natl. Acad. Sci. USA 1999, 96, 266–271. [Google Scholar] [CrossRef] [PubMed]
- Peet, D.J.; Turley, S.D.; Ma, W.; Janowski, B.A.; Lobaccaro, J.M.; Hammer, R.E.; Mangelsdorf, D.J. Cholesterol and bile acid metabolism are impaired in mice lacking the nuclear oxysterol receptor LXRα. Cell 1998, 93, 693–704. [Google Scholar] [CrossRef]
- Viennois, E.; Esposito, T.; Dufour, J.; Pommier, A.; Fabre, S.; Kemeny, J.-L.; Guy, L.; Morel, L.; Lobaccaro, J.-M.; Baron, S. LXRα Regulates the Androgen Response in Prostate Epithelium. Endocrinology 2012, 153, 3211–3223. [Google Scholar] [CrossRef] [PubMed]
- Fukuchi, J.; Hiipakka, R.A.; Kokontis, J.M.; Hsu, S.; Ko, A.L.; Fitzgerald, M.L.; Liao, S. Androgenic suppression of ATP-binding cassette transporter A1 expression in LNCaP human prostate cancer cells. Cancer Res. 2004, 64, 7682–7685. [Google Scholar] [CrossRef] [PubMed]
- Fukuchi, J.; Kokontis, J.M.; Hiipakka, R.A.; Chuu, C.; Liao, S. Antiproliferative Effect of Liver X Receptor Agonists on LNCaP Human Prostate Cancer Cells. Cancer Res. 2004, 64, 7686–7689. [Google Scholar] [CrossRef] [PubMed]
- Chuu, C.; Hiipakka, R.A.; Kokontis, J.M.; Fukuchi, J.; Chen, R.-Y.; Liao, S. Inhibition of tumor growth and progression of LNCaP prostate cancer cells in athymic mice by androgen and liver X receptor agonist. Cancer Res. 2006, 66, 6482–6486. [Google Scholar] [CrossRef] [PubMed]
- Taylor, B.S.; Schultz, N.; Hieronymus, H.; Gopalan, A.; Xiao, Y.; Carver, B.S.; Arora, V.K.; Kaushik, P.; Cerami, E.; Reva, B.; et al. Integrative Genomic Profiling of Human Prostate Cancer. Cancer Cell 2010, 18, 11–22. [Google Scholar] [CrossRef] [PubMed]
- Puhr, M.; Santer, F.R.; Neuwirt, H.; Marcias, G.; Hobisch, A.; Culig, Z. SOCS-3 antagonises the proliferative and migratory effects of fibroblast growth factor-2 in prostate cancer by inhibition of p44/p42 MAPK signalling. Endocr. Relat. Cancer 2010, 17, 525–538. [Google Scholar] [CrossRef] [PubMed]
- Fu, W.; Yao, J.; Huang, Y.; Li, Q.; Li, W.; Chen, Z.; He, F.; Zhou, Z.; Yan, J. LXR Agonist Regulates the Carcinogenesis of PCa via the SOCS3 Pathway. Cell. Physiol. Biochem. 2014, 33, 195–204. [Google Scholar] [CrossRef] [PubMed]
- Xiong, H.; Zhang, Y.; Chen, S.; Ni, Z.; He, J.; Li, X.; Li, B.; Zhao, K.; Yang, F.; Zeng, Y.; et al. Induction of SOCS3 by liver X receptor suppresses the proliferation of hepatocellular carcinoma cells. Oncotarget 2017, 8, 64083–64094. [Google Scholar] [CrossRef] [PubMed]
- Youlin, K.; Li, Z.; Weiyang, H.; Jian, K.; Siming, L.; Xin, G. Liver X receptor activation inhibits PC-3 prostate cancer cells via the beta-catenin pathway. Pathol. Res. Pract. 2017, 213, 267–270. [Google Scholar] [CrossRef] [PubMed]
- Pommier, A.J.C.; Dufour, J.; Alves, G.; Viennois, E.; de Boussac, H.; Trousson, A.; Volle, D.H.; Caira, F.; Val, P.; Arnaud, P.; et al. Liver X receptors protect from development of prostatic intra-epithelial neoplasia in mice. PLoS Genet. 2013, 9, e1003483. [Google Scholar] [CrossRef] [PubMed]
- Varambally, S.; Dhanasekaran, S.M.; Zhou, M.; Barrette, T.R.; Kumar-Sinha, C.; Sanda, M.G.; Ghosh, D.; Pienta, K.J.; Sewalt, R.G.A.B.; Otte, A.P.; et al. The polycomb group protein EZH2 is involved in progression of prostate cancer. Nature 2002, 419, 624–629. [Google Scholar] [CrossRef] [PubMed]
- Tamae, D.; Mostaghel, E.; Montgomery, B.; Nelson, P.S.; Balk, S.P.; Kantoff, P.W.; Taplin, M.-E.; Penning, T.M. The DHEA-sulfate depot following P450c17 inhibition supports the case for AKR1C3 inhibition in high risk localized and advanced castration resistant prostate cancer. Chem. Biol. Interact. 2015, 234, 332–338. [Google Scholar] [CrossRef] [PubMed]
- Pasqualini, J.R. Estrogen sulfotransferases in breast and endometrial cancers. Ann. N. Y. Acad. Sci. 2009, 1155, 88–98. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H.; Gong, H.; Khadem, S.; Lu, Y.; Gao, X.; Li, S.; Zhang, J.; Xie, W. Androgen deprivation by activating the liver X receptor. Endocrinology 2008, 149, 3778–3788. [Google Scholar] [CrossRef] [PubMed]
- Gong, H.; Guo, P.; Zhai, Y.; Zhou, J.; Uppal, H.; Jarzynka, M.J.; Song, W.-C.; Cheng, S.-Y.; Xie, W. Estrogen deprivation and inhibition of breast cancer growth in vivo through activation of the orphan nuclear receptor liver X receptor. Mol. Endocrinol. 2007, 21, 1781–1790. [Google Scholar] [CrossRef] [PubMed]
- Song, W.C. Biochemistry and reproductive endocrinology of estrogen sulfotransferase. Ann. N. Y. Acad. Sci. 2001, 948, 43–50. [Google Scholar] [CrossRef] [PubMed]
- Dozmorov, M.G.; Hurst, R.E.; Culkin, D.J.; Kropp, B.P.; Frank, M.B.; Osban, J.; Penning, T.M.; Lin, H.-K. Unique patterns of molecular profiling between human prostate cancer LNCaP and PC-3 cells. Prostate 2009, 69, 1077–1090. [Google Scholar] [CrossRef] [PubMed]
- Sekine, Y.; Demosky, S.J.; Stonik, J.A.; Furuya, Y.; Koike, H.; Suzuki, K.; Remaley, A.T. High-density lipoprotein induces proliferation and migration of human prostate androgen-independent cancer cells by an ABCA1-dependent mechanism. Mol. Cancer Res. 2010, 8, 1284–1294. [Google Scholar] [CrossRef] [PubMed]
- Füllhase, C.; Schneider, M.P. 5-α-Reductase Inhibitors and Combination Therapy. Urol. Clin. 2016, 43, 325–336. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.-J.; Andersson, L.C.; Bouton, D.; Warner, M.; Gustafsson, J.-A. Stromal growth and epithelial cell proliferation in ventral prostates of liver X receptor knockout mice. Proc. Natl. Acad. Sci. USA 2009, 106, 558–563. [Google Scholar] [CrossRef] [PubMed]
- Tsui, K.-H.; Chung, L.-C.; Feng, T.-H.; Lee, T.-Y.; Chang, P.-L.; Chen, W.-T.; Juang, H.-H. Divergent effect of liver X receptor agonists on prostate-specific antigen expression is dependent on androgen receptor in prostate carcinoma cells. Prostate 2015, 75, 603–615. [Google Scholar] [CrossRef] [PubMed]
- Lundholm, L.; Moverare, S.; Steffensen, K.R.; Nilsson, M.; Otsuki, M.; Ohlsson, C.; Gustafsson, J.A.; Dahlman-Wright, K. Gene expression profiling identifies liver X receptor α as an estrogen-regulated gene in mouse adipose tissue. J. Mol. Endocrinol. 2004, 32, 879–892. [Google Scholar] [CrossRef] [PubMed]
- Kramer, P.R.; Wray, S. 17-β-estradiol regulates expression of genes that function in macrophage activation and cholesterol homeostasis. J. Steroid Biochem. Mol. Biol. 2002, 81, 203–216. [Google Scholar] [CrossRef]
- Delfosse, V.; le Maire, A.; Balaguer, P.; Bourguet, W. A structural perspective on nuclear receptors as targets of environmental compounds. Acta Pharmacol. Sin. 2015, 36, 88–101. [Google Scholar] [CrossRef] [PubMed]
- Schulze, A.; Harris, A.L. How cancer metabolism is tuned for proliferation and vulnerable to disruption. Nature 2012, 491, 364–373. [Google Scholar] [CrossRef] [PubMed]
- Warburg, O. On respiratory impairment in cancer cells. Science 1956, 124, 269–270. [Google Scholar] [PubMed]
- Mycielska, M.E.; Patel, A.; Rizaner, N.; Mazurek, M.P.; Keun, H.; Patel, A.; Ganapathy, V.; Djamgoz, M.B.A. Citrate transport and metabolism in mammalian cells. BioEssays 2009, 31, 10–20. [Google Scholar] [CrossRef] [PubMed]
- Swinnen, J.V.; Roskams, T.; Joniau, S.; van Poppel, H.; Oyen, R.; Baert, L.; Heyns, W.; Verhoeven, G. Overexpression of fatty acid synthase is an early and common event in the development of prostate cancer. Int. J. Cancer 2002, 98, 19–22. [Google Scholar] [CrossRef] [PubMed]
- Swyer, G.I.M. The Cholesterol Content of Normal and Enlarged Prostates. Cancer Res. 1942, 2, 372–375. [Google Scholar]
- White, C.P. On the occurrence of crystals in tumours. J. Pathol. Bacteriol. 1909, 13, 3–10. [Google Scholar] [CrossRef]
- Yue, S.; Li, J.; Lee, S.-Y.; Lee, H.J.; Shao, T.; Song, B.; Cheng, L.; Masterson, T.A.; Liu, X.; Ratliff, T.L.; et al. Cholesteryl Ester Accumulation Induced by PTEN Loss and PI3K/AKT Activation Underlies Human Prostate Cancer Aggressiveness. Cell Metab. 2014, 19, 393–406. [Google Scholar] [CrossRef] [PubMed]
- Wettstein, M.S.; Saba, K.; Umbehr, M.H.; Murtola, T.J.; Fankhauser, C.D.; Adank, J.-P.; Hofmann, M.; Sulser, T.; Hermanns, T.; Moch, H.; et al. Prognostic Role of Preoperative Serum Lipid Levels in Patients Undergoing Radical Prostatectomy for Clinically Localized Prostate Cancer. Prostate 2017, 77, 549–556. [Google Scholar] [CrossRef] [PubMed]
- Alfaqih, M.A.; Allott, E.H.; Hamilton, R.J.; Freeman, M.R.; Freedland, S.J. The current evidence on statin use and prostate cancer prevention: Are we there yet? Nat. Rev. Urol. 2017, 14, 107–119. [Google Scholar] [CrossRef] [PubMed]
- Krycer, J.R.; Brown, A.J. Cross-talk between the androgen receptor and the liver X receptor: Implications for cholesterol homeostasis. J. Biol. Chem. 2011, 286, 20637–20647. [Google Scholar] [CrossRef] [PubMed]
- Cariello, M.; Ducheix, S.; Maqdasy, S.; Baron, S.; Moschetta, A.; Lobaccaro, J. LXRs, SHP and FXR in prostate cancer: Enemies or Ménage-à-quatre with AR? Nucl. Recpt. Signal. 2018, in press. [Google Scholar]
- Morris, M.D.; Chaikoff, I.L. The origin of cholesterol in liver, small intestine, adrenal gland, and testis of the rat: Dietary versus endogenous contributions. J. Biol. Chem. 1959, 234, 1095–1097. [Google Scholar] [PubMed]
- Schaffner, C.P. Prostatic cholesterol metabolism: Regulation and alteration. Prog. Clin. Biol. Res. 1981, 75A, 279–324. [Google Scholar] [PubMed]
- Michaud, D.S.; Augustsson, K.; Rimm, E.B.; Stampfer, M.J.; Willet, W.C.; Giovannucci, E. A prospective study on intake of animal products and risk of prostate cancer. Cancer Causes Control 2001, 12, 557–567. [Google Scholar] [CrossRef] [PubMed]
- De Marzo, A.M.; Platz, E.A.; Sutcliffe, S.; Xu, J.; Grönberg, H.; Drake, C.G.; Nakai, Y.; Isaacs, W.B.; Nelson, W.G. Inflammation in prostate carcinogenesis. Nat. Rev. Cancer 2007, 7, 256–269. [Google Scholar] [CrossRef] [PubMed]
- Aaltoma, S.H.; Lipponen, P.K.; Kosma, V.M. Inducible nitric oxide synthase (iNOS) expression and its prognostic value in prostate cancer. Anticancer Res. 2001, 21, 3101–3106. [Google Scholar] [PubMed]
- Tsai, C.-S.; Chen, F.-H.; Wang, C.-C.; Huang, H.-L.; Jung, S.-M.; Wu, C.-J.; Lee, C.-C.; McBride, W.H.; Chiang, C.-S.; Hong, J.-H. Macrophages From Irradiated Tumors Express Higher Levels of iNOS, Arginase-I and COX-2, and Promote Tumor Growth. Int. J. Radiat. Oncol. 2007, 68, 499–507. [Google Scholar] [CrossRef] [PubMed]
- Twillie, D.A.; Eisenberger, M.A.; Carducci, M.A.; Hseih, W.-S.; Kim, W.Y.; Simons, J.W. Interleukin-6: A candidate mediator of human prostate cancer morbidity. Urology 1995, 45, 542–549. [Google Scholar] [CrossRef]
- Siegsmund, M.J.; Yamazaki, H.; Pastan, I. Interleukin 6 receptor mRNA in prostate carcinomas and benign prostate hyperplasia. J. Urol. 1994, 151, 1396–1399. [Google Scholar] [CrossRef]
- Castrillo, A.; Joseph, S.B.; Vaidya, S.A.; Haberland, M.; Fogelman, A.M.; Cheng, G.; Tontonoz, P. Crosstalk between LXR and toll-like receptor signaling mediates bacterial and viral antagonism of cholesterol metabolism. Mol. Cell 2003, 12, 805–816. [Google Scholar] [CrossRef]
- Joseph, S.B.; Castrillo, A.; Laffitte, B.A.; Mangelsdorf, D.J.; Tontonoz, P. Reciprocal regulation of inflammation and lipid metabolism by liver X receptors. Nat. Med. 2003, 9, 213–219. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Ma, X.; Chen, Y.; Zhang, L.; Jiang, M.; Li, X.; Xiang, R.; Miao, R.; Hajjar, D.P.; Duan, Y.; et al. Identification of interferon-γ as a new molecular target of liver X receptor. Biochem. J. 2014, 459, 345–354. [Google Scholar] [CrossRef] [PubMed]
- Labrecque, L.; Royal, I.; Surprenant, D.S.; Patterson, C.; Gingras, D.; Béliveau, R. Regulation of Vascular Endothelial Growth Factor Receptor-2 Activity by Caveolin-1 and Plasma Membrane Cholesterol. Mol. Biol. Cell 2003, 14, 334–347. [Google Scholar] [CrossRef] [PubMed]
- Noghero, A.; Perino, A.; Seano, G.; Saglio, E.; Lo Sasso, G.; Veglio, F.; Primo, L.; Hirsch, E.; Bussolino, F.; Morello, F. Liver X receptor activation reduces angiogenesis by impairing lipid raft localization and signaling of vascular endothelial growth factor receptor-2. Arterioscler. Thromb. Vasc. Biol. Thromb. Vasc. Biol. 2012, 32, 2280–2288. [Google Scholar] [CrossRef] [PubMed]
- Lai, C.-J.; Cheng, H.-C.; Lin, C.-Y.; Huang, S.-H.; Chen, T.-H.; Chung, C.-J.; Chang, C.-H.; Wang, H.-D.; Chuu, C.-P. Activation of liver X receptor suppresses angiogenesis via induction of ApoD. FASEB J. 2017, 31. [Google Scholar] [CrossRef] [PubMed]
- Albertsen, P.C. Observational studies and the natural history of screen-detected prostate cancer. Curr. Opin. Urol. 2015, 25, 232–237. [Google Scholar] [CrossRef] [PubMed]
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2016. CA. Cancer J. Clin. 2016, 66, 7–30. [Google Scholar] [CrossRef] [PubMed]
- James, N.D.; Sydes, M.R.; Clarke, N.W.; Mason, M.D.; Dearnaley, D.P.; Spears, M.R.; Ritchie, A.W.S.; Parker, C.C.; Russell, J.M.; Attard, G.; et al. Addition of docetaxel, zoledronic acid, or both to first-line long-term hormone therapy in prostate cancer (STAMPEDE): Survival results from an adaptive, multiarm, multistage, platform randomised controlled trial. Lancet Lond. Engl. 2016, 387, 1163–1177. [Google Scholar] [CrossRef]
- Sweeney, C.J.; Chen, Y.-H.; Carducci, M.; Liu, G.; Jarrard, D.F.; Eisenberger, M.; Wong, Y.-N.; Hahn, N.; Kohli, M.; Cooney, M.M.; et al. Chemohormonal Therapy in Metastatic Hormone-Sensitive Prostate Cancer. N. Engl. J. Med. 2015, 373, 737–746. [Google Scholar] [CrossRef] [PubMed]
- Klotz, L.; Boccon-Gibod, L.; Shore, N.D.; Andreou, C.; Persson, B.-E.; Cantor, P.; Jensen, J.-K.; Olesen, T.K.; Schröder, F.H. The efficacy and safety of degarelix: A 12-month, comparative, randomized, open-label, parallel-group phase III study in patients with prostate cancer. BJU Int. 2008, 102, 1531–1538. [Google Scholar] [CrossRef] [PubMed]
- Attard, G.; Reid, A.H.M.; A’Hern, R.; Parker, C.; Oommen, N.B.; Folkerd, E.; Messiou, C.; Molife, L.R.; Maier, G.; Thompson, E.; et al. Selective Inhibition of CYP17 With Abiraterone Acetate Is Highly Active in the Treatment of Castration-Resistant Prostate Cancer. J. Clin. Oncol. 2009, 27, 3742–3748. [Google Scholar] [CrossRef] [PubMed]
- Chang, K.-H.; Li, R.; Kuri, B.; Lotan, Y.; Roehrborn, C.G.; Liu, J.; Vessella, R.; Nelson, P.S.; Kapur, P.; Guo, X.; et al. A gain-of-function mutation in DHT synthesis in castration-resistant prostate cancer. Cell 2013, 154, 1074–1084. [Google Scholar] [CrossRef] [PubMed]
- Hamid, A.R.A.H.; Pfeiffer, M.J.; Verhaegh, G.W.; Schaafsma, E.; Brandt, A.; Sweep, F.C.G.J.; Sedelaar, J.P.M.; Schalken, J.A. Aldo-keto reductase family 1 member C3 (AKR1C3) is a biomarker and therapeutic target for castration-resistant prostate cancer. Mol. Med. Camb. Mass 2013, 18, 1449–1455. [Google Scholar] [CrossRef] [PubMed]
- Tran, C.; Ouk, S.; Clegg, N.J.; Chen, Y.; Watson, P.A.; Arora, V.; Wongvipat, J.; Smith-Jones, P.M.; Yoo, D.; Kwon, A.; et al. Development of a Second-Generation Antiandrogen for Treatment of Advanced Prostate Cancer. Science 2009, 324, 787–790. [Google Scholar] [CrossRef] [PubMed]
- Sharifi, N.; Dahut, W.L.; Steinberg, S.M.; Figg, W.D.; Tarassoff, C.; Arlen, P.; Gulley, J.L. A retrospective study of the time to clinical endpoints for advanced prostate cancer. BJU Int. 2005, 96, 985–989. [Google Scholar] [CrossRef] [PubMed]
- Cornford, P.; Bellmunt, J.; Bolla, M.; Briers, E.; de Santis, M.; Gross, T.; Henry, A.M.; Joniau, S.; Lam, T.B.; Mason, M.D.; et al. EAU-ESTRO-SIOG Guidelines on Prostate Cancer. Part II: Treatment of Relapsing, Metastatic, and Castration-Resistant Prostate Cancer. Eur. Urol. 2017, 71, 630–642. [Google Scholar] [CrossRef] [PubMed]
- Smith, M.R.; Kabbinavar, F.; Saad, F.; Hussain, A.; Gittelman, M.C.; Bilhartz, D.L.; Wynne, C.; Murray, R.; Zinner, N.R.; Schulman, C.; et al. Natural history of rising serum prostate-specific antigen in men with castrate nonmetastatic prostate cancer. J. Clin. Oncol. 2005, 23, 2918–2925. [Google Scholar] [CrossRef] [PubMed]
- Robinson, D.; van Allen, E.M.; Wu, Y.-M.; Schultz, N.; Lonigro, R.J.; Mosquera, J.-M.; Montgomery, B.; Taplin, M.-E.; Pritchard, C.C.; Attard, G.; et al. Integrative Clinical Genomics of Advanced Prostate Cancer. Cell 2015, 162, 454. [Google Scholar] [CrossRef] [PubMed]
- Visakorpi, T.; Hyytinen, E.; Koivisto, P.; Tanner, M.; Keinänen, R.; Palmberg, C.; Palotie, A.; Tammela, T.; Isola, J.; Kallioniemi, O.-P. In vivo amplification of the androgen receptor gene and progression of human prostate cancer. Nat. Genet. 1995, 9, 401–406. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.D.; Welsbie, D.S.; Tran, C.; Baek, S.H.; Chen, R.; Vessella, R.; Rosenfeld, M.G.; Sawyers, C.L. Molecular determinants of resistance to antiandrogen therapy. Nat. Med. 2004, 10, 33–39. [Google Scholar] [CrossRef] [PubMed]
- Komiya, A.; Yasuda, K.; Watanabe, A.; Fujiuchi, Y.; Tsuzuki, T.; Fuse, H. The prognostic significance of loss of the androgen receptor and neuroendocrine differentiation in prostate biopsy specimens among castration-resistant prostate cancer patients. Mol. Clin. Oncol. 2013, 1, 257–262. [Google Scholar] [CrossRef] [PubMed]
- Waltering, K.K.; Urbanucci, A.; Visakorpi, T. Androgen receptor (AR) aberrations in castration-resistant prostate cancer. Mol. Cell. Endocrinol. 2012, 360, 38–43. [Google Scholar] [CrossRef] [PubMed]
- Miyamoto, H.; Yeh, S.; Wilding, G.; Chang, C. Promotion of agonist activity of antiandrogens by the androgen receptor coactivator, ARA70, in human prostate cancer DU145 cells. Proc. Natl. Acad. Sci. USA 1998, 95, 7379–7384. [Google Scholar] [CrossRef] [PubMed]
- Gerhardt, J.; Montani, M.; Wild, P.; Beer, M.; Huber, F.; Hermanns, T.; Müntener, M.; Kristiansen, G. FOXA1 Promotes Tumor Progression in Prostate Cancer and Represents a Novel Hallmark of Castration-Resistant Prostate Cancer. Am. J. Pathol. 2012, 180, 848–861. [Google Scholar] [CrossRef] [PubMed]
- Gregory, C.W.; He, B.; Johnson, R.T.; Ford, O.H.; Mohler, J.L.; French, F.S.; Wilson, E.M. A Mechanism for Androgen Receptor-mediated Prostate Cancer Recurrence after Androgen Deprivation Therapy. Cancer Res. 2001, 61, 4315–4319. [Google Scholar] [PubMed]
- Xu, K.; Wu, Z.J.; Groner, A.C.; He, H.H.; Cai, C.; Lis, R.T.; Wu, X.; Stack, E.C.; Loda, M.; Liu, T.; et al. EZH2 oncogenic activity in castration-resistant prostate cancer cells is Polycomb-independent. Science 2012, 338, 1465–1469. [Google Scholar] [CrossRef] [PubMed]
- Yepuru, M.; Wu, Z.; Kulkarni, A.; Yin, F.; Barrett, C.M.; Kim, J.; Steiner, M.S.; Miller, D.D.; Dalton, J.T.; Narayanan, R. Steroidogenic enzyme AKR1C3 is a novel androgen receptor-selective coactivator that promotes prostate cancer growth. Clin. Cancer Res. 2013, 19, 5613–5625. [Google Scholar] [CrossRef] [PubMed]
- Culig, Z.; Hobisch, A.; Cronauer, M.V.; Radmayr, C.; Trapman, J.; Hittmair, A.; Bartsch, G.; Klocker, H. Androgen receptor activation in prostatic tumor cell lines by insulin-like growth factor-I, keratinocyte growth factor, and epidermal growth factor. Cancer Res. 1994, 54, 5474–5478. [Google Scholar] [CrossRef] [PubMed]
- Signoretti, S.; Montironi, R.; Manola, J.; Altimari, A.; Tam, C.; Bubley, G.; Balk, S.; Thomas, G.; Kaplan, I.; Hlatky, L.; et al. Her-2-neu Expression and Progression Toward Androgen Independence in Human Prostate Cancer. JNCI J. Natl. Cancer Inst. 2000, 92, 1918–1925. [Google Scholar] [CrossRef] [PubMed]
- Osman, I.; Scher, H.I.; Drobnjak, M.; Verbel, D.; Morris, M.; Agus, D.; Ross, J.S.; Cordon-Cardo, C. HER-2/neu (p185neu) Protein Expression in the Natural or Treated History of Prostate Cancer. Clin. Cancer Res. 2001, 7, 2643–2647. [Google Scholar] [PubMed]
- Yeh, S.; Lin, H.-K.; Kang, H.-Y.; Thin, T.H.; Lin, M.-F.; Chang, C. From HER2/Neu signal cascade to androgen receptor and its coactivators: A novel pathway by induction of androgen target genes through MAP kinase in prostate cancer cells. Proc. Natl. Acad. Sci. USA 1999, 96, 5458–5463. [Google Scholar] [CrossRef] [PubMed]
- Mellinghoff, I.K.; Vivanco, I.; Kwon, A.; Tran, C.; Wongvipat, J.; Sawyers, C.L. HER2/neu kinase-dependent modulation of androgen receptor function through effects on DNA binding and stability. Cancer Cell 2004, 6, 517–527. [Google Scholar] [CrossRef] [PubMed]
- Labrie, F. Adrenal androgens and intracrinology. Semin. Reprod. Med. 2004, 22, 299–309. [Google Scholar] [CrossRef] [PubMed]
- Mitsiades, N.; Sung, C.C.; Schultz, N.; Danila, D.C.; He, B.; Eedunuri, V.K.; Fleisher, M.; Sander, C.; Sawyers, C.L.; Scher, H.I. Distinct patterns of dysregulated expression of enzymes involved in androgen synthesis and metabolism in metastatic prostate cancer tumors. Cancer Res. 2012, 72, 6142–6152. [Google Scholar] [CrossRef] [PubMed]
- De Bono, J.S.; Oudard, S.; Ozguroglu, M.; Hansen, S.; Machiels, J.-P.; Kocak, I.; Gravis, G.; Bodrogi, I.; Mackenzie, M.J.; Shen, L.; et al. Prednisone plus cabazitaxel or mitoxantrone for metastatic castration-resistant prostate cancer progressing after docetaxel treatment: A randomised open-label trial. Lancet 2010, 376, 1147–1154. [Google Scholar] [CrossRef]
- Bubendorf, L.; Schöpfer, A.; Wagner, U.; Sauter, G.; Moch, H.; Willi, N.; Gasser, T.C.; Mihatsch, M.J. Metastatic patterns of prostate cancer: An autopsy study of 1589 patients. Hum. Pathol. 2000, 31, 578–583. [Google Scholar] [CrossRef] [PubMed]
- Weinfurt, K.P.; Li, Y.; Castel, L.D.; Saad, F.; Timbie, J.W.; Glendenning, G.A.; Schulman, K.A. The significance of skeletal-related events for the health-related quality of life of patients with metastatic prostate cancer. Ann. Oncol. 2005, 16, 579–584. [Google Scholar] [CrossRef] [PubMed]
- Parker, C.; Nilsson, S.; Heinrich, D.; Helle, S.I.; O’Sullivan, J.M.; Fosså, S.D.; Chodacki, A.; Wiechno, P.; Logue, J.; Seke, M.; et al. Alpha emitter radium-223 and survival in metastatic prostate cancer. N. Engl. J. Med. 2013, 369, 213–223. [Google Scholar] [CrossRef] [PubMed]
- Saad, F.; Gleason, D.M.; Murray, R.; Tchekmedyian, S.; Venner, P.; Lacombe, L.; Chin, J.L.; Vinholes, J.J.; Goas, J.A.; Zheng, M. Long-term efficacy of zoledronic acid for the prevention of skeletal complications in patients with metastatic hormone-refractory prostate cancer. J. Natl. Cancer Inst. 2004, 96, 879–882. [Google Scholar] [CrossRef] [PubMed]
- Smith, M.R.; Saad, F.; Coleman, R.; Shore, N.; Fizazi, K.; Tombal, B.; Miller, K.; Sieber, P.; Karsh, L.; Damião, R.; et al. Denosumab and bone-metastasis-free survival in men with castration-resistant prostate cancer: Results of a phase 3, randomised, placebo-controlled trial. Lancet Lond. Engl. 2012, 379, 39–46. [Google Scholar] [CrossRef]
- Kantoff, P.W.; Higano, C.S.; Shore, N.D.; Berger, E.R.; Small, E.J.; Penson, D.F.; Redfern, C.H.; Ferrari, A.C.; Dreicer, R.; Sims, R.B.; et al. Sipuleucel-T immunotherapy for castration-resistant prostate cancer. N. Engl. J. Med. 2010, 363, 411–422. [Google Scholar] [CrossRef] [PubMed]
- Antonarakis, E.S.; Lu, C.; Wang, H.; Luber, B.; Nakazawa, M.; Roeser, J.C.; Chen, Y.; Mohammad, T.A.; Chen, Y.; Fedor, H.L.; et al. AR-V7 and Resistance to Enzalutamide and Abiraterone in Prostate Cancer. N. Engl. J. Med. 2014, 371, 1028–1038. [Google Scholar] [CrossRef] [PubMed]
- Raza, S.; Meyer, M.; Goodyear, C.; Hammer, K.D.P.; Guo, B.; Ghribi, O. The cholesterol metabolite 27-hydroxycholesterol stimulates cell proliferation via ERβ in prostate cancer cells. Cancer Cell Int. 2017, 17, 52. [Google Scholar] [CrossRef] [PubMed]
- Pencheva, N.; Buss, C.G.; Posada, J.; Merghoub, T.; Tavazoie, S.F. Broad-Spectrum Therapeutic Suppression of Metastatic Melanoma through Nuclear Hormone Receptor Activation. Cell 2014, 156, 986–1001. [Google Scholar] [CrossRef] [PubMed]
- Segala, G.; David, M.; de Medina, P.; Poirot, M.C.; Serhan, N.; Vergez, F.; Mougel, A.; Saland, E.; Carayon, K.; Leignadier, J.; et al. Dendrogenin A drives LXR to trigger lethal autophagy in cancers. Nat. Commun. 2017, 8, 1903. [Google Scholar] [CrossRef] [PubMed]
- Zhong, S.; Zhang, X.; Chen, L.; Ma, T.; Tang, J.; Zhao, J. Statin use and mortality in cancer patients: Systematic review and meta-analysis of observational studies. Cancer Treat. Rev. 2015, 41, 554–567. [Google Scholar] [CrossRef] [PubMed]
- Platz, E.A.; Leitzmann, M.F.; Visvanathan, K.; Rimm, E.B.; Stampfer, M.J.; Willett, W.C.; Giovannucci, E. Statin drugs and risk of advanced prostate cancer. J. Natl. Cancer Inst. 2006, 98, 1819–1825. [Google Scholar] [CrossRef] [PubMed]
- Loeb, S.; Kan, D.; Helfand, B.T.; Nadler, R.B.; Catalona, W.J. Is statin use associated with prostate cancer aggressiveness? BJU Int. 2010, 105, 1222–1225. [Google Scholar] [CrossRef] [PubMed]
- Harshman, L.C.; Wang, X.; Nakabayashi, M.; Xie, W.; Valenca, L.; Werner, L.; Yu, Y.; Kantoff, A.M.; Sweeney, C.J.; Mucci, L.A.; et al. Statin Use at the Time of Initiation of Androgen Deprivation Therapy and Time to Progression in Patients With Hormone-Sensitive Prostate Cancer. JAMA Oncol. 2015, 1, 495–504. [Google Scholar] [CrossRef] [PubMed]
- Harshman, L.C.; Werner, L.; Tripathi, A.; Wang, X.; Maughan, B.L.; Antonarakis, E.S.; Nakabayashi, M.; McKay, R.; Pomerantz, M.; Mucci, L.A.; et al. The impact of statin use on the efficacy of abiraterone acetate in patients with castration-resistant prostate cancer. Prostate 2017, 77, 1303–1311. [Google Scholar] [CrossRef] [PubMed]
- Viennois, E.; Pommier, A.J.C.; Mouzat, K.; Oumeddour, A.; El Hajjaji, F.-Z.; Dufour, J.; Caira, F.; Volle, D.H.; Baron, S.; Lobaccaro, J.-M.A. Targeting liver X receptors in human health: Deadlock or promising trail? Expert. Opin. Ther. Targets 2011, 15, 219–232. [Google Scholar] [CrossRef] [PubMed]
- Yasuda, T.; Grillot, D.; Billheimer, J.T.; Briand, F.; Delerive, P.; Huet, S.; Rader, D.J. Tissue-specific liver X receptor activation promotes macrophage RCT in vivo. Arterioscler. Thromb. Vasc. Biol. 2010, 30, 781–786. [Google Scholar] [CrossRef] [PubMed]
- Stachel, S.J.; Zerbinatti, C.; Rudd, M.T.; Cosden, M.; Suon, S.; Nanda, K.K.; Wessner, K.; DiMuzio, J.; Maxwell, J.; Wu, Z.; et al. Identification and in Vivo Evaluation of Liver X Receptor β-Selective Agonists for the Potential Treatment of Alzheimer’s Disease. J. Med. Chem. 2016, 59, 3489–3498. [Google Scholar] [CrossRef] [PubMed]
- Kirchgessner, T.G.; Sleph, P.; Ostrowski, J.; Lupisella, J.; Ryan, C.S.; Liu, X.; Fernando, G.; Grimm, D.; Shipkova, P.; Zhang, R.; et al. Beneficial and Adverse Effects of an LXR Agonist on Human Lipid and Lipoprotein Metabolism and Circulating Neutrophils. Cell Metab. 2016, 24, 223–233. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bousset, L.; Rambur, A.; Fouache, A.; Bunay, J.; Morel, L.; Lobaccaro, J.-M.A.; Baron, S.; Trousson, A.; De Joussineau, C. New Insights in Prostate Cancer Development and Tumor Therapy: Modulation of Nuclear Receptors and the Specific Role of Liver X Receptors. Int. J. Mol. Sci. 2018, 19, 2545. https://doi.org/10.3390/ijms19092545
Bousset L, Rambur A, Fouache A, Bunay J, Morel L, Lobaccaro J-MA, Baron S, Trousson A, De Joussineau C. New Insights in Prostate Cancer Development and Tumor Therapy: Modulation of Nuclear Receptors and the Specific Role of Liver X Receptors. International Journal of Molecular Sciences. 2018; 19(9):2545. https://doi.org/10.3390/ijms19092545
Chicago/Turabian StyleBousset, Laura, Amandine Rambur, Allan Fouache, Julio Bunay, Laurent Morel, Jean-Marc A. Lobaccaro, Silvère Baron, Amalia Trousson, and Cyrille De Joussineau. 2018. "New Insights in Prostate Cancer Development and Tumor Therapy: Modulation of Nuclear Receptors and the Specific Role of Liver X Receptors" International Journal of Molecular Sciences 19, no. 9: 2545. https://doi.org/10.3390/ijms19092545
APA StyleBousset, L., Rambur, A., Fouache, A., Bunay, J., Morel, L., Lobaccaro, J.-M. A., Baron, S., Trousson, A., & De Joussineau, C. (2018). New Insights in Prostate Cancer Development and Tumor Therapy: Modulation of Nuclear Receptors and the Specific Role of Liver X Receptors. International Journal of Molecular Sciences, 19(9), 2545. https://doi.org/10.3390/ijms19092545