Abstract
Reactive oxygen species (ROS) are produced in various cell compartments by an array of enzymes and processes. An excess of ROS production can be hazardous for normal cell functioning, whereas at normal levels, ROS act as vital regulators of many signal transduction pathways and transcription factors. ROS production is affected by a wide range of viruses. However, to date, the impact of viral infections has been studied only in respect to selected ROS-generating enzymes. The role of several ROS-generating and -scavenging enzymes or cellular systems in viral infections has never been addressed. In this review, we focus on the roles of biogenic polyamines and oxidative protein folding in the endoplasmic reticulum (ER) and their interplay with viruses. Polyamines act as ROS scavengers, however, their catabolism is accompanied by H2O2 production. Hydrogen peroxide is also produced during oxidative protein folding, with ER oxidoreductin 1 (Ero1) being a major source of oxidative equivalents. In addition, Ero1 controls Ca2+ efflux from the ER in response to e.g., ER stress. Here, we briefly summarize the current knowledge on the physiological roles of biogenic polyamines and the role of Ero1 at the ER, and present available data on their interplay with viral infections.
1. Introduction
Viral infections contribute significantly to human disease. Some viruses cause an acute infection that resolves spontaneously. This is the case for influenza, respiratory syncytial, rhino-, and other respiratory viruses, as well as for hepatitis A and E, measles, and dengue viruses [1]; they all trigger a wide range of symptoms including inflammation of the infected organ or cell type. Other viruses establish chronic, often life-long disease. The most prominent amongst this group of pathogens are human immunodeficiency virus (HIV), hepatitis B (HBV), C (HCV) and delta viruses (HDV), Epstein-Barr virus (EBV), human papilloma virus (HPV), and type 8 human herpes virus. Chronic viral infections are associated with various pathologies. For instance, chronic virally-induced liver hepatitis causes long-lasting liver inflammation, fibrosis, and metabolic disorders [2]. HIV causes immunodeficiency as well as neurological disorders [3]. In addition, a number of viruses are oncogenic: HBV and HCV induce hepatocellular carcinoma, EBV causes Burkett or Hodgkin lymphomas, and type 8 herpes virus causes Kaposi sarcoma [4]. As a result, both acute and chronic infections may cause permanent or persistent disability and contribute to premature mortality. Elaboration of approaches for treatment and/or prevention of virus-associated pathologies is hampered because the life cycles of these viruses and their impact on the host are not fully understood.
To date, it is clear that pathogenesis of a variety of viruses is at least partially due to altered metabolism of reactive oxygen species (ROS). ROS represent a group of relatively short-lived oxygen intermediates such as hydroxyl-radical (HO●), superoxide anion (O2●−), singlet oxygen (1O2), and hydrogen peroxide (H2O2). Indeed, many viruses including HBV, HCV, HIV, influenza, respiratory syncytial, rhino, dengue and tick-borne encephalitis viruses enhance ROS production in infected cells [5,6,7,8,9,10,11]. They also affect various components of the host’s antioxidant defense system. Most of the studies published on this topic so far were based on the concept of “oxidative stress” as a mere imbalance between ROS production and neutralization [12] and therefore focused on particular ROS generating and ROS-scavenging systems. In terms of ROS production, studies focused on the oxidative phosphorylation system in mitochondria, extramitochondrial NADPH-oxidases (Nox), and xanthine oxidase. However, other mitochondrial and non-mitochondrial ROS-generating enzymes and processes have been largely ignored. Amongst them are the non-respiratory mitochondrial enzymes monoaminooxidase (MAO) and α-ketoglutarate dehydrogenase (reviewed in [13]). However, here we will summarize data on two important extra-mitochondrial processes that can be important sources of ROS and affect cellular redox pathways and associated signaling. The first are biogenic polyamines, which act as ROS scavengers, even though their catabolism is associated with H2O2 production. Polyamines are involved metabolic homeostasis and developmental processes. The second source of ROS we will address is the major protein that ensures oxidative protein folding in the endoplasmic reticulum (ER): oxidoreductin 1 (Ero1). The interplay between these ROS-producing systems and viral infections will be detailed.
2. Biogenic Polyamines at the Crossroads of Redox Status
Biogenic polyamines are low molecular weight compounds composed of an aliphatic carbon chain with several amino groups. They include spermine, spermidine, and their precursor diamine putrescine and are present in all types of mammalian cells (Figure 1). Other species such as plants, bacteria and archaea may lack some of these polyamines but contain other members of this class of compounds [14], such as agmatine, cadaverine, thermospermine, and norspermidine (Figure 1), as well as a wide array of branched polyamines. The first polyamine identified by crystallization by Antonie van Leeuwenhoek in 1678 was spermine [15]. However, its structure was determined only in 1926 [16]. Purification of spermine from biological samples with the methods available at that time was only possible due to very high levels of polyamines in mammalian tissues and cells that can reach millimolar concentrations. In prokaryotes, polyamine concentration is estimated as 2 mM [17,18].

Figure 1.
Structure of biogenic polyamines.
2.1. Polyamine Biosynthesis and Catabolism
Putrescine, a precursor of spermidine and spermine, is synthesized by decarboxylation of the amino acid l-ornithine by a pyridoxal phosphate-dependent enzyme—ornithine decarboxylase (ODC) (Figure 2) [19]. Plants and bacteria have an alternative pathway in which l-arginine is decarboxylated into agmatine by arginine decarboxylase (ADC) [20]. The latter can then be converted into putrescine by agmatinase (in Escherichia coli) [21], or by a combined action of agmatine deiminase and N-carbamoylputrescine amidase [22]. Sixteen years ago human agmatinase has been cloned suggesting that in mammalian cells putrescine can be produced from agmatine as well [23]. Indeed, agmatine is present in mammalian tissues [24,25,26,27], and has even been claimed to exhibit neurotransmitter activity [28]. Since then, several papers reported the existence of ADC in mammals, and this enzymatic activity was assigned to antizyme inhibitor 2 (AZIn2) [29,30], a regulatory protein whose place in polyamine metabolism is discussed below. However, other groups did not find evidence for a conversion of arginine into agmatine in the presence of the recombinant AZIn2 or extracts of mammalian tissues (i.e., liver) (for example, [31]). So the current view of the field is that polyamine precursor putrescine in mammals is synthesized either from arginine via ornithine, or via a reaction catalyzed by agmatinase from dietary agmatine or the one produced by gut microbes.
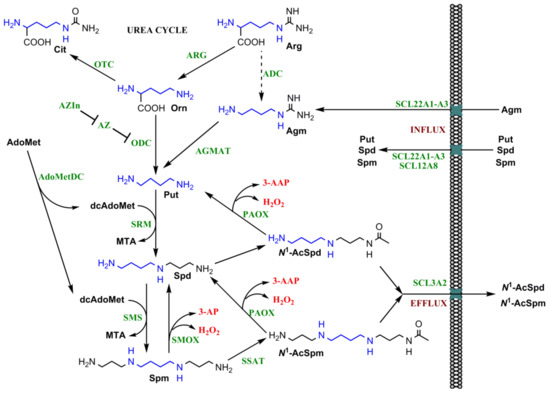
Figure 2.
Scheme of metabolism of biogenic polyamines. Polyamines are synthesized from two metabolites of the urea cycle, namely arginine (Arg) and ornithine (Orn). Within this cycle, Arg is converted by arginase (ARG) into Orn, and the latter is transformed into citrulline (Cit) by an ornithine transcarbamylase (OTC). Polyamine precursor putrescine (Put) is synthesized from Orn by ornithine decarboxylase (ODC), or from Agm by agmatinase (AGMAT). The latter is a product of bacterial arginine decarboxylase (ADC) (a dashed arrow indicates absence of the enzyme in mammalian cells), whereas in mammals it is absorbed from the gut. Putrescine is converted into spermidine (Spd) and then to spermine (Spm) by spermidine and spermine synthases (SRM and SMS). The aminopropyl fragment is transferred by them from a decarboxylated S-adenosyl methionine (dcAdoMet) that is synthesized by S-adenosyl methionine decarboxylase (AdoMetDC). Polyamine catabolism is mediated by spermidine/spermine-N1-acetyltransferase (SSAT) with subsequent degradation of the acetylated spermine (N1-AcSpm) and spermidine (N1-AcSpd) by acetylpolyamine oxidase (PAOX). Spermine can also be directly converted into spermidine by spermine oxidase (SMOX). Both catabolic pathways produce toxic hydrogen peroxide and either N-acetyl-3-aminopropanal (3-AAP) or 3-aminopropanal (3-AP) (given in red) that are readily converted into acrolein. The bended arrows indicate utilization of a second substrate by spermine and spermidine synthases and formation of by-products during both polyamine biosynthesis and catabolism. A negative regulation of polyamine biosynthesis is achieved by antizyme (AZ) that targets ODC to proteasome, whereas functions of AZ are also inhibited by antizyme inhibitor (AZIn) (T-bar). The green abbreviations indicate enzymes and regulatory proteins. An 1,4-diaminobutyl group of polyamines deriving from arginine is marked blue.
Spermine and spermidine are synthesized from putrescine by addition of aminopropyl groups (Figure 2). This group is taken from a decarboxylated S-adenosylmethionine (dcAdoMet) that is produced from S-adenosylmethionine by S-adenosylmethionine decarboxylase (AdoMetDC) [32]. The enzymes that transfer aminopropyl groups to putrescine and spermidine are known as spermidine and spermine synthases, respectively [33]. Noteworthy both ODC and AdoMetDC have cysteine residues in their active sites that readily react with nitric oxide leading to inactivation of these enzymes [34]. Nitric oxide is, therefore, a potent inhibitor of polyamine synthesis, thus forming a first link between polyamines and redox biology.
Degradation of polyamines is achieved by a combination of acetylation, oxidation, and excretion to vacuoles or extracellular fluids. In the extracellular space, polyamines are utilized by diamine oxidase and other copper-dependent extracellular enzymes [35]. In the cell spermine and spermidine undergo acetylation by spermidine/spermine-N1-acetyltransferase (SSAT) with subsequent conversion by a flavin adenine dinucleotide (FAD)—dependent acetylpolyamine oxidase (PAOX) into spermidine and putrescine, respectively [36]. Alternatively, spermine can be directly converted into spermidine by another FAD-dependent enzyme—spermine oxidase (SMOX) [37]. These extracellular and intracellular oxidases generate H2O2 and N-acetyl-3-aminopropanal (in case of PAOX) and 3-aminopropanal (in case of SMOX), and the latter two are readily and spontaneously metabolized into a highly toxic acrolein [38]. Noteworthy, production of these two by-products is generally associated with activity of other metabolic pathways such as lipid degradation. However, the input of polyamine-catabolizing enzymes towards ROS production is generally neglected.
SMOX and PAOX are localized in different cellular compartments: PAOX is a peroxisomal enzyme [39], whereas several different isoforms of SMOX are present in the cytoplasm and nucleus [40]. Among them, only two SMOX isoforms exhibit enzymatic activity, namely SMOX1 and SMOX5. Moreover, recently, existence of a novel SMOX-like protein in mitochondria was reported [41]. These observations may account for the greater input of SMOX compared to PAOX into production of H2O2, at least during enhanced polyamine catabolism [42]. In line with this, overexpression of SMOX leads to substantial changes in cellular redox status [43], whereas SSAT induction has no notable effect on ROS production, as revealed by commonly used ROS-sensitive dyes [42,44].
2.2. Regulation of Polyamine Metabolism
Intracellular levels of spermine and spermidine are maintained and tightly controlled by enzymes that catalyze rate-limiting steps of their biosynthesis (ODC) and catabolism (SMOX and SSAT) [45], as well as by their uptake [46]. Intracellular ODC and SSAT activities depend on levels of their gene transcription, translation, mRNA stability and protein degradation [47]. Expression of ODC (ODC1) is controlled by the transcription factors cMYC [48,49] and nuclear factor-kappa B (NF-κB) [50], whereas SSAT transcription is driven by NF-κB [51,52,53] and nuclear factor erythroid 2 (NFE2)-related factor 2 (Nrf2) transcription factors [54,55,56]. Thus, their expression is enhanced in response to inflammation, activation of phosphatidylinositol-4,5-bisphosphate 3-kinase (PI3K)/AKT [57], mitogen-activated protein kinase (MAPK) [58], and Wnt/β-catenin signaling [59], altered levels of polyamines [54,60] and enhanced ROS production [56]. Of note, the SAT1 gene encoding SSAT contains a polyamine response element (PRE), which acts as a binding site for the classical ROS-sensitive Nrf2 factor [54,55]. Our group also demonstrated that ODC is induced in response to H2O2 via Nrf2 [56]. We did not map the binding site for this factor within the promoter, but the latter contains three TGACnnnGC sequences at −1.5, −2.1 and −4.9 kb before the transcriptional start site [61], that represent classical antioxidant response elements (ARE) to which Nrf2 is known to bind [62]. Finally, cMYC was also shown to drive transcription of genes encoding spermine synthase (SMS) and AdoMetDC [63,64].
In addition, ODC and SSAT have a very short half-life. Mechanisms of control of ODC degradation have been extensively studied and are controlled by two proteins—ODC antizyme (AZ) and antizyme inhibitor (AZIn). AZ is an inhibitor of ODC since it binds to ODC monomer and prevents assembly of the active homodimer [65]. In addition, AZ targets ODC for degradation by the 26S proteasome. These mechanisms are highly responsive to the levels of polyamines, since the active AZ is produced by a +1 frameshift of its mRNA. This frameshift is enhanced by polyamines, presumably by stabilization of a stem-loop structure in the proximity of the frameshift site. The half-life of ODC in the cell is likely also affected by ROS, since ODC can also bind to a classical Nrf2-inducible protein—NAD(P)H:quinone oxidoreductase 1 (Nqo1) [66]. Nqo1 targets ODC to the 20S proteasomal degradation pathway, which is characterized by a lower efficiency than 26S proteasomal pathway, thus prolonging the half-life of the enzyme. A second component of the system regulating ODC protein stability is AZIn. This protein, which has a structure similar to that ODC, binds to AZ more tightly than ODC. It can, therefore, displace ODC from ODC-AZ complexes or prevent their formation [65]. It should be noted that mammalian genomes contain one functional ODC1 gene and at least four AZ and two AZIn genes that encode proteins with different expression profiles in various tissues and different properties [31,67,68].
SMOX activity is regulated only at the transcriptional level [69]. It is highly inducible by polyamine analogs and other stimuli such as ischemia-reperfusion and treatment with tumor necrosis factor alpha [37,40,51,70,71]. Increased SMOX expression was also shown to occur during differentiation of mouse myoblast C2C12 cells [72]. The other oxidase, PAOX, is generally expressed constitutively, and in most cells, this enzyme catalyzes a non-rate-limiting step [73].
Intracellular levels of polyamines are also regulated by their influx. Spermine and spermidine are imported into the cell by an active transport mechanism; however the exact transporters remain unknown. So far, several transporters have been implicated in polyamine influx and efflux. These include solute carrier (SLC) 22A1–A3 (Oct1–3), SLC12A8, SLC3A2 etc. (reviewed by Abdulhussein and Wallace in [46]). Polyamine transport is suppressed by AZ, presenting another mechanism by which antizyme reduces polyamine levels [65]. Finally, polyamines were also shown to penetrate into the cells by endocytosis [74].
2.3. Polyamines Can Act as Antioxidants
Although enhanced turnover of spermine and spermidine contribute to overproduction of H2O2, polyamines also contribute to the protection of the cells against ROS. Initially, it was observed that spermine and spermidine, as well as other amines, can quench 1O2● [17]. Later a more detailed study from Casero’s group confirmed, that spermine indeed acts as a direct ROS scavenger [75]. Similar data were also obtained for spermidine [76], agmatine [77] as well as synthetic polyamine analogs [78]. Putrescine and cadaverine exhibit low efficacy in ROS neutralization [78,79]. Polyamines can neutralize a wide spectrum of ROS including H2O2 [76], O2●− [78], HO● [75,79,80], 1O2 [17,79], as well as synthetic radicals including 1,1-diphenyl-2-picrylhydrazyl (DPPH) radical [76] and peroxyl radicals, the latter formed from 2,2′-azo-bis-(2-amidinopropane) [78]. These studies led to the assumption that polyamines can act as bona fide ROS scavengers. However, the rate constants of the ROS scavenging reactions and therefore the input of polyamines into ROS neutralization has not been assessed in a precise manner.
Antioxidant properties of polyamines are observed at relatively high concentrations, as revealed from an example of agmatine exhibiting prooxidant activity at low (10–100 µM) and antioxidant at high (1–2 mM) concentrations [77]. At the same time, spermine exhibits protective effect towards isolated rat liver mitochondria at relatively high concentrations (25–100 mM) [81]. In particular, spermine prevents mitochondrial swelling and a decrease of mitochondrial potential triggered by the combined action of Ca2+ and phosphate. Moreover, polyamines bound to DNA protect the latter from ROS-induced damage [82]. This is observed at physiological (~1 mM) concentrations [82,83].
Spermidine plays a significant role in protection of cells against H2O2-induced oxidative stress [84]. Importantly, the mechanism of action of polyamines in protection of the cells against H2O2 is different from that observed for classical antioxidants like glutathione [84]. Polyamine-deficient bacteria (Escherichia coli) [85] or yeast cells (Saccharomyces cerevisiae) [86,87] exhibit more pronounced signs of oxidative stress compared to their wild-type strains. Polyamines also serve as messengers for induction of expression of antioxidant defense enzymes such as heme oxygenase 1 (HO-1) in response to e.g., β2-adrenoceptor agonists [88]. Spermine was also shown to decrease H2O2 levels and to prevent dysfunction of rat liver mitochondria [80]. The antioxidant effect of spermine at physiological concentrations in cultured myocytes is comparable to that of vitamin E [89]. In addition, at physiological concentrations spermine suppresses accumulation of free iron in cultured myocytes, with the effect being comparable to that of deferoxamine [89]. The latter may suppress generation of hydroxyl radicals via the Fenton reaction. Yeast with a defect in spe1 gene, leading to inability to synthesize polyamines, exhibit signs of elevated ROS production, whereas supplementation of medium with spermidine reduces ROS back to the level observed in wild type cells [87]. In addition, food supplementation with spermidine decreased oxidative stress markers in aged mice [87]. Finally, such supplementation of food of Drosophila melanogaster confirms resistance to H2O2 and paraquat, an inducer of superoxide anion production [90]. In a zebrafish model, spermidine supplementation significantly suppressed production of ROS and NO and consequently alleviated lipopolysaccharide-triggered inflammation [91]. Altogether, these data support that polyamines contain antioxidant activity in physiological settings.
2.4. Polyamines in Health and Disease
Spermine and spermidine are compounds that are positively charged at physiological pH and thus bind to acidic groups of cellular macromolecules (proteins, nucleic acids or phospholipids) [60,92]. They are involved in regulation of a wide spectrum of biological processes in the cell including DNA replication and transcription, RNA processing and translation and regulation of ion channels [93,94,95]. Interaction with nucleic acids results in enhanced stability of DNA and RNA duplexes. In case of interaction with RNA, this can lead to stabilization of regulatory structural elements. Exhaustion of polyamine levels may lead to decreased expression of particular genes, as can be exemplified by decreased Heat shock protein 70 (Hsp70) [96] and cMYC [97], and cyclin-dependent kinase 4 (CDK4) [98] mRNA levels in cells treated with ODC inhibitor or in cells with decreased expression of HO-1, c-Jun and c-Fos transcription factors in response to AdoMetDC inhibition [96]. Other genes such as p21 [97] and JunD [98] are up-regulated in cells with depleted polyamine content. Since CDK4 and p21 are involved in regulation of the cell cycle, changes in their expression suggest that altered polyamine levels affect cell proliferation.
Spermine and spermidine can also affect cell fate. Monti et al. showed that HL-60 cell apoptosis triggered by 2-deoxyglucose (an inhibitor of glycolysis) can be prevented by exhaustion of the polyamine pool using ODC inhibitors, whereas addition of exogenous polyamines blocks this effect [99]. In the presence of 2-deoxyribose, polyamines do not affect cell survival rate but merely regulate the death scenario between apoptosis (high polyamine levels) and necrosis (low polyamine levels) [100]. Contrary data reported that ageing in yeast and human peripheral blood mononuclear cells (PBMCs) is accompanied by a decrease of spermidine content, elevation of oxidative stress markers and, as a result, necrotic cell death [87].
Polyamines are important for cell differentiation. For example, exogenous polyamines promote differentiation of human marrow-derived mesenchymal stem cells by regulating osteogenic and adipogenic gene expression [101]. They also affect differentiation of other types of cells including myoblasts [72], germ cells [102], and keratinocytes [103].
Spermidine is a precursor for a unique posttranslational modification of a lysine residue known as hypusination [104]. Hypusination has been described in the case of only one protein-eukaryotic initiation factor 5A (eIF5A) [105] (Figure 3). A mature form of eIF5A is essential for protein biosynthesis. According to the current concept, hypusinated eIF5A acts as an elongation factor [106] that allows translation bypass polyproline-rich sequences [107].

Figure 3.
Spermidine serves as a substrate for a hypusination of eukaryotic translation initiation factor 5A (eIF5A). Lysine 50 residue of the latter is conjugated with the aminobutyl fragment from spermine by deoxyhypusine synthase (DHPS), and the resulting deoxyhypusinated factor undergoes oxidation by a deoxyhypusine hydroxylase (DOHH).
The importance of polyamines for mammalian development is supported by data from knockout mice. Animals lacking functional ODC or AdoMetDC-encoding genes are incapable of polyamine biosynthesis and die during very early embryonic stages [108]. Addition of spermidine to the culture medium during yeast/PBMCs cultivation or as a food supplement for Drosophila melanogaster and mice restored polyamine levels, that had decreased during ageing, counteracted ROS production and significantly prolonged lifespan [87,109,110]. Life-long food supplementation with spermidine diminished rates of occurrence of liver fibrosis and hepatocellular carcinoma in mice [110]. Investigation of the underlying mechanisms revealed that depletion of polyamines during ageing leads to activation of histone acetyl transferase (HAT) complexes and increased histone H3 acetylation [87]. Spermidine-induced longevity is promoted via histone hypoacetylation, increased expression of autophagy-related genes and, as a result, increased formation of autophagosomes [87,90,109,110]. Interestingly, these changes correlate with ROS production. Previously it was shown by several groups that oxidative stress triggered by exogenous H2O2 or ethanol in hepatocytes or lung alveolar A549 cells, induced histone acetylation through modulation of HAT complexes or their co-activators [111,112,113,114]. Similarly, autophagy is induced by ROS of various origins including mitochondria [115], ER-residing Nox4 activated by endoplasmic reticulum (ER) stress, expression of HIV transactivator (Tat)-protein [116], or treatment of cells with H2O2 [117,118]. So, spermidine might be one of the central regulators of these events.
Substantial elevation in polyamine content is also associated with pathologies including cancer and other types of cell hyperproliferation [119,120]. It is widely acknowledged that a majority of cancers exhibit increased levels of spermine and spermidine. These include liver, colon, gastric, esophagus, lung, prostate, and many others cancers (reviewed in [120]). Increased expression of polyamine-synthesizing enzymes contributes to carcinogenesis. As an example, increase in polyamine levels in skin of mice due to overexpression of ODC was accompanied by spontaneous occurrence of skin tumors both in the absence [119] and presence [121] of carcinogens. In combination with other pro-cancerous events such as expression of a mutated Ras oncogene ODC promotes invasiveness of skin cancer cells [122]. Overexpression of this enzyme in normal prostate cells is sufficient for their malignant transformation [123]. In this study, a strong up-regulation of the main polyamine enzymes (ODC, spermine oxidase, spermine synthase) and a concomitant reduction of spermine was observed in high-grade prostate intraepithelial neoplasia (HGPIN) tissues as compared to normal prostate glands with the expression levels for SMOX expression being the highest. Elevated polyamine levels also promote angiogenesis, which is one of the tumors hallmarks [124]. So, enhanced polyamine biosynthesis can be an early event in tumorigenesis, and its combination with other oncogenic events may result in formation of tumor cells.
Alteration of polyamine metabolism also contributes to bacteria-induced tumorigenesis in the gastrointestinal tract. Wilson’s group clearly demonstrated that development of gastric cancer during Helicobacter pylori infection is due to induction of spermine oxidase [125,126]. The procarcinogenic effect of this enzyme is at least partly associated with production of H2O2, since its expression and concomitant development of tumors were strongly associated with levels of H2O2 and oxidative DNA damage. Similar findings were also reported for the enteric bacterial strain Bacteroides fragilis [127,128].
2.5. Polyamines and Viral Infections
Data on the interplay between polyamines and viral infections are controversial. The majority of them were published in the 1970s–1980s, when several key players of their metabolism were just discovered, but the technical approaches for their investigation not yet elaborated. These old data refer mostly to incorporation of polyamines in virions, and to sensitivity of virus replication to inhibition of polyamine biosynthesis.
Since spermine and spermidine can bind to DNA and RNA, it is not surprising that they were found in viral particles of various viruses. Virions from densonucleosis viruses 1 and 2 contain putrescine, spermidine, and spermine comprising 1.41% (w/w) of the virus particle and neutralizing 26% of the single-stranded viral DNA [129]. Vaccinia virus of the Poxviridae family [130] and bacteriophage R17 [131] also incorporate polyamines into virions. Encephalomyocarditis virus contains approximately 200 molecules of putrescine, 100 molecules of spermidine, and 40 molecules of spermine that neutralize 11% of the negative charge of the viral genome [132]. Herpes simplex virus (HSV) also contains both spermine and spermidine, which are compartmentalized in capsid and envelope, respectively [133]. Presence of polyamines was also shown for influenza virus (in which they neutralize approximately 38% of RNA phosphates) [134] and rhinovirus [135]. Other viruses such as Semliki Forest virus do not contain polyamines in their capsids [136].
Both old and recent papers suggest that polyamines are crucial for replication of many viruses. Inhibition of polyamine biosynthesis inhibits replication of a variety of RNA viruses belonging to the families Picornaviridae, Flaviviridae, Coronaviridae, Filoviridae, and Buniaviridae [137,138,139] as well as of DNA-containing HSV [140], human cytomegalovirus (HCMV) [141,142,143], and vaccinia viruses (Tyms et al. 1983). However, the mechanisms by which polyamine depletion suppresses their replication is likely to be different. In the case of HCMV, polyamines are required for virion assembly, either at the level of DNA packaging or capsid envelopment [141]. For HSV, arboviruses, and poxviruses, polyamines are needed for viral genome replication [130,138,140]. Another mechanism by which polyamines contribute to viral replication is maintaining hypusination of eIF5A factor, as shown for Ebola and Marburg viruses [139] and human immunodeficiency virus (HIV) [144]. In HIV-infected cells, hypusination inhibitors suppress viral gene expression at the level of transcription initiation [144]. Finally, induction of SSAT and a concomitant decrease in polyamines were shown to inhibit replication of Zika and chikungunya viruses [138]. The same effect was observed for both pathogens when biosynthesis of polyamines was blocked [138]. Depletion of polyamines affected both, viral genome replication and protein biosynthesis. Interestingly, passaging chikungunya virus in the presence of increasing d,l-difluoromethylornithine (DFMO) concentrations led to establishing clones resistant to polyamine depletions [145]. The latter was associated with mutations in the non-structural protein 4 (nsP4) gene that enhanced methylation of viral genomes and their binding to cell membranes.
The least studied is the effect of viral infection on polyamine metabolism itself. Cells infected with HSV-1 exhibit decreased levels of spermine and spermidine, presumably due to suppressed polyamine biosynthesis [146,147]. In addition, HSV-2 was shown to suppress subsequent steps of polyamine biosynthesis, i.e., conversion of putrescine into spermidine and then to spermidine [148] At the same time, HCMV, that also belongs to the Herpesviridae family, triggers pronounced induction of ODC [149] and a concomitant increase in spermine and spermidine levels [142,150]. However, other authors noted, that elevation of putrescine and spermidine levels was accompanied by reduced levels of spermine [151]. The latter implies induction of SSAT and/or SMOX, however, expression of the former was not studied, whereas the latter was not yet discovered at the time. Transient increase in ODC activity was also shown for cells infected with vaccinia virus [152]. Some data also suggest inhibited polyamine biosynthesis by human adenovirus type 5 [152]. In the lymphocytes from HIV-1-infected patients an increase in polyamine levels and SSAT activity was reported [153], though such changes do not occur in the infected lymphocytes in vitro [154]. Several years ago, HIV Tat protein was shown to induce SMOX, which plays role in virally-induced neurotoxicity [155]. Finally, our group recently reported that transient expression of HCV core and nonstructural protein (NS) 5A (NS5A) proteins leads to activation of polyamine metabolism and elevation of intracellular polyamine levels [156]. However, long-lasting expression of viral proteins in the context of the full length replicon resulted in a decrease in spermine and spermidine, accompanied by an induction of spermine oxidase. Spermine oxidase may be required for efficient replication of the viral genome, since polyamines were previously shown to inhibit helicase activity of HCV NS3 protein [157]. Finally, Chlorella virus that infects some algae species encodes enzymes of polyamine biosynthesis itself [158,159]. However, similar examples are not known for mammalian viruses.
3. ER Oxidoreductins as Key Players in Oxidative Protein Folding, Calcium Efflux from the ER, and Evasion of Cancer Cells from Immune Responses
3.1. Ero1 Is the Key Player in Oxidative Protein Folding
The endoplasmic reticulum is an organelle that ensures production and folding of secreted, plasmatic, Golgi, and lysosomal proteins, that serves as intracellular storage of calcium ions and as site for lipid synthesis. Protein folding involves formation of disulfide bonds between cysteine residues of the same protein or between two different proteins. Disulfide bonds are introduced into the nascent polypeptide via dithiol-disulfide exchange reaction with assistance of oxidoreductases including protein disulfide isomerase (PDI), ERp44, ERp46, ERp72 and other members of the PDI family [160] (Figure 4). PDI is also capable of reducing incorrect S–S bonds formed spontaneously, thus allowing subsequent formation of the correct disulfide bonds [161]. Specifically, PDI family members form mixed disulfides with the client proteins, with subsequent release of oxidized substrates and reduced PDIs [162,163]. Regeneration of PDI into its oxidized form is tightly linked to H2O2 production or scavenging.
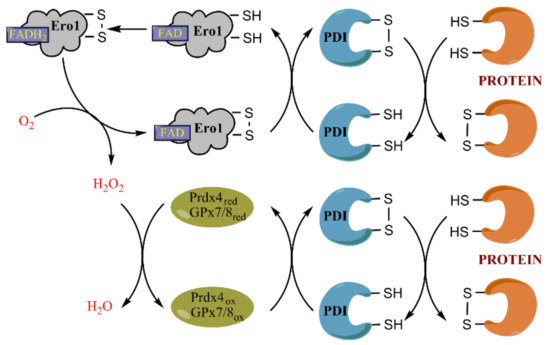
Figure 4.
Role of ER oxidoreductin 1 (Ero1), peroxiredoxin 4 (Prdx4) and glutathione peroxidases (GPx) 7 and 8 in oxidative protein folding. The primary oxidative equivalents are generated by a FAD-dependent enzyme Ero1 that reduces molecular oxygen (O2) into hydrogen peroxide (H2O2). It leads to formation of a disulfide bond between two cysteines of Ero1 followed by a disulfide-dithiol exchange with a protein disulfide isomerases (PDI) and later with the target protein. Noteworthy, the figure does not show exchanges between several pairs of cysteines within Ero1. The generated H2O2 is utilized by Prdx4 or GPx7/8, that also oxidize PDI. Thus, a reduction of one oxygen molecule generates equivalents for oxidation of two pairs of cysteines of a target protein.
The primary oxidation equivalents for protein folding are generated by an enzyme referred to as ER oxidoreductase (Ero1). It was originally identified in yeast in 1998 as the ER-residing glycosylated enzyme Ero1p, and shown to be involved in oxidative protein folding [164,165]. In 2000, two human orthologs were found and termed Ero1-like (Ero1L) α [166] and β [167]. Ero1α is highly expressed in esophagus, liver, heart, stomach, duodenum, and placenta, whereas Ero1β is expressed mostly in the pancreas and at lower levels in the stomach and testis [167,168]. Ero1β exhibits a two-fold higher enzymatic activity compared to Ero1α [169].
Both yeast and mammalian ER oxidoreductins transfer electrons from PDI to molecular oxygen, leading to its reduction into H2O2 [170,171]. Ero1 is a flavoprotein, so the reaction is dependent on levels of FAD [170,171,172]. Reduction of O2 is accompanied by formation of an intramolecular disulfide bond between Cys352 and Cys355 of the enzyme (the numbers correspond to positions of amino acids within yeast Ero1p) [173]. This disulfide bond is later passed to another pair of cysteines (Cys100 and Cys105) within Ero1 and the resulting disulfide bond is cleaved during formation of an intermolecular disulfide with PDI and subsequent regeneration of the latter [174]. Other intramolecular disulfide bonds also exist that are implicated in regulation of Ero1p activity [175]. Similar intra- and intermolecular disulfide-dithiol exchange has been demonstrated for human Ero1α and Ero1β isoforms [176,177,178]. Kinetic analysis revealed that formation of the correct regulatory S–S bond within Ero1α in the presence of O2 or H2O2 is slow, but is significantly enhanced in the presence of PDI family members [179]. Again, enzymatic activity of the mammalian oxidoreductase is regulated by several additional pairs of cysteines. For example, a disulfide bond between Cys208 and Cys241 residues of Ero1α forms a seal for FAD in the site of molecular oxygen reduction, and its reduction by PDI leads to release of FAD and, therefore, to inactivation of the enzyme [176]. Substitution of some of these cysteine residues results in a constantly active Ero1 enzyme, i.e., in a constituent H2O2 production [180]. It is also worth noting, that PDI is the major but not the sole substrate of Ero1α and Ero1β [169,174,181], they can also oxidize other members of PDI family such as ERp46, ERp57, and ERp72 [179,181,182,183]. Several PDI family members are also implicated in retention of Ero1α/β in the ER [184,185].
Before identification of Ero1, protein oxidation folding was thought to be mediated by oxidized glutathione (GSSG). However, later it was found that GSSG is not required [164]. In contrast, reduced glutathione (GSH) can interact with oxidized PDI, leading to reduction of the latter and formation of GSSG [186,187]. Since the reduced PDI activates Ero1, increase in GSH content in the ER enhances H2O2 production [188]. Recently, a negative feedback loop was described: GSH-induced Ero1α activation results in massive production of H2O2, leading to suppressed GSH import into ER [189]. On the other hand, absence of GSH would result in hyperoxidation of proteins [186]. So, a balance between GSH and GSSG in the ER lumen forms a sort of a “buffer” for oxidative protein folding, that allows efficient isomerization of S–S bonds and conversion of non-native disulfides into native ones [182,187,190,191].
3.2. Regulation of Ero1 Expression
Expression of yeast Ero1p and its mammalian orthologs Ero1α and Ero1β is not constitutive. ER stress and concomitant unfolded protein response (UPR) cause significant increase in Ero1p/Ero1β and moderate increase in Ero1α levels [164,165,167,192,193,194]. Specifically, Ero1α is induced by the protein kinase R (PKR)-like ER kinase (PERK)/CCAAT/enhancer-binding protein homologous protein (CHOP) pathway of the UPR [193,194]. Another factor that plays an even greater impact on Ero1α expression is the level of O2, as hypoxia leads to a profound overexpression of this oxidoreductase via induction of hypoxia-inducible factor 1α (HIF-1α) [192,195,196]. In addition, Ero1α is upregulated in response to depletion of iron content, triggered, for example, by the chelating agent deferoxamine [192]. Finally, induction of Ero1α and other UPR-responsive genes has been described during nitrosative stress, and the underlying mechanism involved S-glutathionylation of PDI cysteine(s) [197].
3.3. Role of Prdx4 and GPx7/8 in Scavenging H2O2 in the ER and in Protein Folding
Since oxidative protein folding is accompanied by constant production of H2O2 in the ER, intensive search identified several lumenal enzymes, that efficiently scavenge H2O2. These include the ER-residing peroxiredoxin 4 (Prdx4) [198] and glutathione peroxidases 7 (GPx7) [199] and 8 (GPx8) [200]. However, the exact role of each of these factors is still not fully understood. Initially it was proposed that Prdx4 is the bona fide scavenger of H2O2 generated by Ero1α, as the levels of peroxide production correlated to hyperoxidation of this peroxiredoxin, which also plays a cytoprotective role [198]. However, Appenzeller-Herzog’s group suggested that Prdx4 is implicated in neutralization of H2O2 only in the settings of constantly active Ero1α, whereas in cells expressing the wild type enzyme the major antioxidant role is attributed to GPx8 [200]. Noteworthy, the latter efficiently prevents leakage of H2O2 from the ER to the cytoplasm [200]. At the same time Ero1α is controlling luminal levels of hydrogen peroxide, that correlate with the expression levels of this enzyme [201].
Recent data suggest, that this scavenging of H2O2 is not a mere antioxidant reaction, but an element of oxidative protein folding. All these three enzymes contribute to the formation of disulfide bonds. It was proposed that both GPx7 and GPx8 are not the real glutathione peroxidases but protein disulfide isomerase peroxidases, that enhance peroxidase activity of PDI family members [202]. Indeed, they interact with Ero1α, thereby enhancing its enzymatic activity and concomitant oxygen consumption [202], and with PDI [199], thus forming an Ero1α/PDI/GPx7/8 triad that drives protein folding. Prdx4 also interacts with and oxidizes PDI [203,204,205] and other members of its family such as ERp56 and P5 [161]. Interestingly, Prdx4 drives peroxide-dependent oxidative protein folding even in cells lacking Ero1α, thus suggesting existence of alternative, yet undiscovered, sources of H2O2 in the ER lumen [204]. To sum up, these antioxidant enzymes allow formation of two disulfide bonds at a cost of consumption of one oxygen molecule, without affecting the redox status in the ER lumen.
3.4. Ero1 Controls Efflux of Calcium Ions from the ER
Redox biology is tightly linked to homeostasis of calcium ions in the cell. In most compartments the concentration of Ca2+ is low (~10−7 M), whereas the ER acts as a storage for calcium ions [206]. An estimated concentration of Ca2+ in the ER lumen is ~10−4 M [206]. Thus, it is not surprising that alteration of normal ER functioning may result in efflux of Ca2+ from the ER, yielding a significant increase in calcium levels in the cytoplasm and mitochondria and causing a wide spectrum of events. More and more data describe Ero1 as one of the key regulators of calcium homeostasis.
Elevated Ca2+ levels in the ER, compared to other organelles, are maintained by sarco/endoplasmic reticulum Ca2+-ATPase (SERCA). SERCA mediates uptake of Ca2+ and controls activity of inositol 1,4,5-trisphosphate receptor (IP3R) and ryanodine receptor (RyR) that mediate Ca2+ efflux [206]. There are three isoforms of SERCA: SERCA1 is expressed in the skeletal muscle, SERCA3—Primarily in cells of hematopoetic origin and some epithelial cells, whereas SERCA2 is expressed ubiquitously [207]. Since one of the SERCA2 cysteines undergo S-glutathionylation [208], it is tempting to speculate, that the status and level of Ero1 may affect the functioning of this pump and, therefore, calcium homeostasis. Indeed, the mutant of the target cysteine residue leads to Ca2+ depletion of the ER stores as well as in altered Ero1α induction in response to hypoxia [209]. Since decrease in Ca2+ levels in the ER is known to down-regulate SERCA2 expression [210], this would amplify the signal. SERCA2 is also known to be sensitive to inactivation due to hyperoxidation of its cysteine residue(s) [211]. However, the pump is protected from hyperoxidation by selenoprotein N (SEPN1), which interacts with SERCA2. Moreover, increased expression of Ero1α is accompanied by induction of SEPN1, which protects the pump from the hazardous effects of the oxidoreductase [211]. However, it could be assumed, that any other factor that targets this selenoprotein may lead to altered calcium homeostasis.
Ero1α also affects calcium storage by interaction with the IP3R. The latter is localized at mitochondria-associated membranes (MAMs), i.e., at the site of physical interaction between ER and mitochondria [212]. IP3R interacts with voltage-dependent anion channel (VDAC) through Grp75, whereas VDAC acts as a channel for Ca2+ and ROS in the outer membrane of mitochondria [213]. Activation of IP3R leads to efflux of calcium ions from the ER and their accumulation in mitochondria [166]. Ca2+ influx through the inner mitochondrial membrane is mediated by mitochondrial calcium uniporter channel (MCU). Interestingly, MCU regulatory protein termed mitochondrial calcium uptake 1 (MICU1) is activated when calcium concentrations reach ~2–10 μM [214]. While efflux of Ca2+ from the ER results in <0.8 μM general concentration in the cytoplasm, the local concentration at MAMs reaches the target values, thus allowing a direct flux from the ER to mitochondria [214,215]. Ero1α is also localized at MAMs under normal oxidizing ER conditions, whereas reducing agents and hypoxia lead to its relocalization [216]. In 2012, Sitia’s group revealed that Ero1α controls efflux of Ca2+ from the ER to mitochondria, and its induction leads to decreased levels of the ions in the ER, their accumulation in the mitochondrial matrix and concomitant mitochondrial dysfunction [217].
Both SERCA2 and IP3R are also regulated by PDI family members. ERp44 was shown to inhibit Ca2+ efflux by IP3R through direct interaction with either of them. This interaction was disrupted during saturation of ER Ca2+stores [218]. ERp57, in contrast, suppressed activity of SERCA2b, which resulted in increased periods and length of Ca2+ oscillations [219]. Currently, no direct data exist on the effect of Ero1α on these interactions. However, it can be assumed that overexpression of this oxidoreductase, which leads to changes in redox status of members of the PDI family, may constitute an additional mechanism of Ca2+ efflux from the ER. This is consistent with evidence that shows that ER stress leads to calcium release from the ER through Ero1α-mediated IP3R activation [194].
3.5. Role of Ero1 in Pathology
Ero1α is associated with carcinogenesis and other pathologies. It has been clearly established that breast [220], gastric [221], esophagus [222], and colon [222,223] tumors exhibit significantly higher levels of Ero1α expression compared to the respective normal tissues. Moreover, levels of its expression correlate with rates of recurrence of the disease and with reduced survival rates [220,221,224]. Silencing Ero1α inhibits growth and metastatic potential of breast cancer cells [220]. To our knowledge, there are no data about interference of this enzyme with the cell cycle machinery, proapoptotic factors or tumor suppressors. At the same time, several lines of evidence suggest, that Ero1α facilitates evasion of tumor cells from host immune responses and tumor vascularization. Tanaka et al. showed that Ero1α plays important roles in production of vascular endothelial growth factor (VEGF), with a significant correlation between expression of the oxidoreductase and angiogenesis in breast tumors [225]. The same group also reported that overexpression of Ero1α in human breast cancer up-regulates programmed cell-death 1 ligand 1 (PD-L1) [226], a well-known regulator of an immune checkpoint [227]. Mechanistically, this was due to enhanced oxidative protein folding and by induction of PD-L1 transcription. Production of PD-L1 by tumor cells promotes their escape from immune responses by triggering death of T-cells [228]. Another mechanism by which Ero1α contributes to evasion of tumor cells from immune responses is by promoting infiltration of myeloid-derived suppressor cells (MDSCs) into the tumor [229]. The latter results from enhanced production of granulocyte colony-stimulating factor (G-CSF), chemokine (C-X-C motif) ligand (CXCL) 1 and 2 cytokines/chemokines. MDSCs exhibit immunosuppressive activity by preventing activation of T-cells [230]. Therefore it is not surprising, that mice with Ero1α knock-out are characterized by slower tumor growth [229]. However, other groups revealed that ER oxidoreductin 1α can also enhance T-cell responses. It was exemplified by demonstrating that Ero1α is involved in oxidative folding of major histocompatibility complex 1 (MHC-1) [223], that is crucial for interaction with cytotoxic CD8+ T-cells [231]. Indeed, colon carcinoma cells exhibit elevated expression of Ero1α compared to normal cells, and in these cells, oxidative MHC-1 folding is enhanced [223]. Moreover, expression of Ero1α and concomitant MHC-1 folding are enhanced in hypoxic conditions, as shown for both colon cancer and thymoma cells [223,232]. As a result, Ero1α-overexpressing cells trigger an enhanced cytotoxic T-cell response, whereas Ero1α down-regulation inhibits the response, and hypoxia partially restores it. The discrepancy between pro- and antitumor activity of Ero1α is unknown.
3.6. Ero1α in Viral Infections
Data on the status of Ero1α in human viral infections are scarce and limited to just two human and one plant virus. Most of the data concern HCV. HCV is known to trigger massive ROS production in the infected hepatocytes, both in vitro and in the context of the liver (reviewed in [233]). Amongst the ten viral proteins, two are considered as main inducers of ROS: core and NS5A [234,235,236]. For a long time, it has been considered, that ROS production in HCV core-expressing cells was enhanced primarily in mitochondria and resulted from induced activity of mitochondrial Ca2+ uniporter [237] or interaction of the viral protein with mitochondrial chaperone Hsp60 [238]. In addition, HCV core was shown to cause UPR stress, leading to depletion of ER calcium stores [239]. We demonstrated, that HCV core up-regulated expression of Ero1α, and that prevention of its induction not only reduced H2O2 levels in the cytoplasm, but also blocked mitochondrial Ca2+ uniporter-mediated ROS production in mitochondria [240]. In contrast, Ero1α expression was not altered in cells overexpressing NS5A [241], another HCV protein that was considered to up-regulate ROS production via alteration of calcium homeostasis [234]. Nevertheless, Ero1α is indeed one of the key players in HCV-induced redox alterations, whereas its role in the viral life cycle still remains to be uncovered.
Chikungunya virus is another pathogen that is thought to cause oxidative stress in infected cells [242]. Nothing is known about the underlying molecular mechanisms and the potential involvement of Ero1α. However, recent data suggest, that Ero1α is important for the chikungunya viral life cycle, since its inhibitor EN460, as well as inhibitors of PDI, suppress viral replication, especially during its early stages [243].
Finally, a recently published data suggest, that Ero1 is a crucial component of replication of a plant brome mosaic virus (BMV) [244]. This oxidoreductase facilitates formation of a disulfide-linked complex of viral replication protein 1a protein, thus driving capping of BMV RNA. In addition, Ero1p triggers leakage of oxidizing equivalents from ER lumen. The latter is likely to contribute to enhancement of pathogen replication, as both plant and human viruses are known to exploit ROS for efficient capping and synthesis of their genomes [245,246].
4. Interplay between Polyamine Metabolism and ER Stress/Unfolded Protein Response
In the current literature, polyamine metabolism and oxidative protein folding in the ER are still being considered as separate events, even though tight interlinks have been reported between them. It has been known for a long time that polyamine depletion, resulting from their inhibited biosynthesis, causes swelling of ER [247]. Prunotto et al. provided evidence for increased expression of the classical UPR markers Grp78 and Grp94 in MDCK cells treated with profibrotic transforming growth factor β1 (TGFβ1) and DMFO, an inhibitor of polyamine biosynthesis, compared to TGFβ1-treated cells [248]. This effect was due to depletion of polyamines, as exogenous putrescine, spermidine, and spermine prevented expression of Grp78 and Grp94. Unfortunately, this study presented no data on expression of UPR marker genes in TGFβ1-untreated cells and did not study oxidative protein folding in the ER. This gap was later filled in by Kahana’s group, who studied changes in gene expression in NIH 3T3 cells treated with DFMO using Affimetrix microarrays [249]. They revealed, that polyamine depletion leads to specific activation of the PERK pathway, but no changes in expression of genes regulated by the other two branches of the UPR. At the same time, the microarray dataset demonstrated pronounced induction of protein disulfide isomerase A6 (PDIA6) and peroxiredoxin 4. Since the latter factor is a key component of the oxidative protein folding machinery, and Ero1α expression is regulated by the PERK-activating transcription factor 4 (ATF4)-CHOP pathway, that is activated in these cells (see above), this finding suggests existence of an interplay between polyamines and protein folding. Moreover, a depletion of ER calcium stores observed in DFMO-treated cells further supports this observation, taking into account the known role of Ero1α in Ca2+ homeostasis.
Similar changes occur in response to activation of polyamine catabolism. In rats with acute pancreatitis, due to conditional overexpression of SSAT, pancreatic acinus cells exhibited pronounced signs of endoplasmic reticulum dilatation soon after activation of polyamine catabolism [250]. At the same time, it was shown that SSAT overexpression and concomitant polyamine depletion trigger mitochondrial dysfunction leading to cell death [251]. Several papers report, that enhanced polyamine catabolism interferes with exogenous and endogenous gene translation [252,253] and triggers ER stress and UPR. The latter was demonstrated in various systems based on cell lines overexpressing SSAT [254] and animals with induced SSAT in response to cisplatin [254]. Translation inhibition was caused by depletion of polyamines, since restoration of polyamine content in SSAT-overexpressing cells with spermine and spermidine as well as their mimetics restored translation [253]. The observed ER stress was probably triggered by toxic by-products of spermine and spermidine degradation, as both ER stress as well as concomitant cell death could be prevented by compounds, that scavenge aldehydes alone or in combination with pegylated catalase [254]. The status of oxidative protein folding was not accessed in any of these studies. One could speculate that induction of SSAT does not lead to induction of Ero1α. Indeed, SSAT1 targets HIF-1α, which controls Ero1α expression for degradation [195]. However, Ero1α could still be induced by the UPR PERK pathway [194], which is induced by SSAT overexpression.
ER stress can also result from dysregulated eIF5α hypusination. In a yeast model, Rossi et al. found that eIF5A is involved in cotranslational translocation of the forming polypeptide into the ER, and that defects in hypusination of this factor lead to induction of several stress-induced chaperones [255]. Park’s group revealed that depletion of eIF5α from HeLa cells does not affect overall translation, but specifically upregulates many genes of the UPR [256]. Interestingly, eIF5α depletion did not affect translation of genes encoding proteins containing polyproline sequences. Indeed, the majority of dysregulated proteins did not possess any polyproline stretches. Again, this study did not investigate expression of Ero1α. However, the provided data set clearly showed increase in expression of several key ER chaperones and proline-disulfide isomerases as well as of mitochondrial chaperones [256]. Changes were also found in expression of VDAC1 and VDAC3, which are involved in maintaining calcium homeostasis. Therefore, it is tempting to speculate, that affecting this part of polyamine metabolism may affect cellular redox status by altering oxidative protein folding in the ER. However, this assumption needs to be validated experimentally.
Polyamines not only modulate proper ER functioning, but also affect processes downstream of calcium efflux from the ER. Indeed, overexpression of ODC has been reported to protect cells from death triggered by thapsigargin (Tg) [257], a widely known non-competitive inhibitor or SERCA. This agent depletes ER Ca2+ stores, thus inducing ER stress, subsequent accumulation of calcium ions in mitochondria and, as a result, apoptosis. ODC overexpression in Tg-treated cells prevented induction of UPR-responsive genes and decrease in SERCA levels, suppressed increase in intramitochondrial Ca2+ concentration, mitochondrial membrane depolarization, release of cytochrome c and other preapoptotic events [257]. Induction of UPR-inducible proapoptotic genes (i.e., CHOP) at the translational level and subsequent apoptosis could also be prevented by inhibition of deoxyhypusine synthase with its inhibitor GC7, as was shown in a pancreatic β-cell line treated with Tg [258]. This provides additional evidence that polyamine metabolism may affect ER homeostasis, and changes in spermine and spermidine levels can affect protein folding in the ER and cause mitochondrial dysregulation. Thus, mitochondrial dysfunction and production of ROS in this organelle, observed during many viral infections, may not necessarily be directly induced by a virus but can also occur as a secondary or even tertiary event.
Most of the data discussed in this chapter concern uninfected cells or animal studies, while few data based on infectious models are discussed. This does not mean that the same interlinks do not exist in the context of viral infections. Indeed, our group revealed that hepatitis C virus alters polyamine metabolism specifically by inducing the two catabolic enzymes SSAT and SMOX in a transient and persistent fashion, respectively [156]. This effect was mediated by two viral proteins: core and NS5A [156]. These two proteins are known to trigger ROS production in infected cells [234,235,259]. In addition, HCV core, the viral protein that displays the highest prooxidant activity [235], upregulates expression of Ero1α, and this event was found to be upstream to core-triggered superoxide production in mitochondria, as well as to accumulation of hydrogen peroxide in cytoplasm [240]. This clearly shows that polyamine-metabolizing enzymes and oxidative protein folding in ER can (i) significantly contribute to virus-induced oxidative stress, (ii) generate ROS outside mitochondria, and (iii) regulate proper functioning of this organelle. Other examples come from studies of parasitic infections and from plant viruses. It is acknowledged that Arabidopsis thaliana, like many other species, defend against pathogens by inducing an event referred to as hypersensitive response (HR). Its induction during infection with mosaic cucumber virus is accompanied by enhanced polyamine biosynthesis [260]. Detailed analysis of changes in gene expression between control plants and those treated with spermine revealed induction of UPR [261] and in particular of AtbZIP60 [260]—one of its key regulators [262]. Given that AtbZIP60 controls expression of several protein disulfide isomerases (i.e., AtPDI6, AtPDI9, AtPDI10, and AtPDI11) [263], these facts provide another line of support of a tight interplay between polyamine metabolism and oxidative protein folding in the ER. Finally, Yarlett’s group performed analysis of changes in HTC-8 cells infected with the Cryptosporidium parvum parasite [264]. They revealed (i) perturbation of polyamine metabolism (i.e., induction of SSAT and ODC leading to changes in the ratio between individual polyamines and accumulation of acetylated spermine and spermidine), (ii) signs of UPR and (iii) enhanced H2O2 production and concomitant activation of the antioxidant Nrf2/ARE pathway.
5. Conclusions
Despite a tremendous significance of polyamines and Ero1α for normal cell growth, their interplay with viral infections remains poorly studied. However, there are several key facts that merit further investigations. First, various groups demonstrated that inhibitors of polyamine biosynthesis exhibit antiviral activity against a wide spectrum of DNA and RNA viruses. However, there is only limited data unveiling changes in polyamine levels and levels of expression of polyamine-metabolizing enzymes in infected cells. Should these scarce data hold true, these changes could contribute to various virus-associated pathologies such as tissue damage and neoplastic transformation in chronic infections, or barrier dysfunction and concomitant susceptibility to bacterial pathogens in respiratory viral infections. Secondly, it has been clearly shown that polyamine catabolism is associated with production of H2O2 as a by-product. At the same time, with the exception of HIV, it is not known if augmented ROS production in infected cells is due to SMOX or PAOX. Again, SMOX-derived peroxide and acrolein could be partially responsible for the pathogenicity of these infections. Third, the alternative polyamine biosynthesis pathway requires further studies, both in normal and infected cells. Fourth, the effect(s) of viruses on the ER oxidative protein folding machinery also requires future studies. Since Ero1α is induced in response to UPR and hypoxia, it may turn out that many infections that trigger ER stress and induce HIF-1α up-regulate its expression. If this is the case, this might be the upstream cause for many well-characterized events in infected cells, such as disrupted calcium homeostasis, mitochondrial dysfunction and changes in redox status of the cells in total and in the ER in particular. Moreover, for HCV, which induces GPx8 proteolysis, induction of Ero1α could lead to leakage of H2O2 from the ER. Thus, the interplay between viruses and the ER oxidative folding machinery can contribute to infection-associated pathologies. In conclusion, a significant amount of data demonstrate ER stress, altered protein folding and dysregulated polyamine metabolism in infected cells, but the molecular links between these events and their respective roles in the cellular stress and pathophysiology associated with infection will require further studies.
Acknowledgments
This work was supported by Russian ministry of education and science (Agreement 14.616.21.0043, identification number RFMEFI61615X0043), French National Agency for AIDS and Viral Hepatitis Research (14370), Comite de Saone-et-Loire de la Ligue contre le cancer, Agence Nationale de Recherche, the Region Rhone-Alpes, DevWeCan French Laboratories of Excellence Network (Labex, Grant ANR-10-LABX-61) and the OpeRa International Hellenic University (IHU) program (GRANT ANR-10-IBHU-004), and le partenariat Hubert Curien (PHC) Kolmogorov.
Conflicts of Interest
The authors declare no conflict of interest.
Abbreviations
| ROS | Reactive oxygen species |
| ER | Endoplasmic reticulum |
| Ero1 | ER oxidoreductin 1 |
| Ero1L | Ero1-like |
| HIV | Human immunodeficiency virus |
| HBV | Hepatitis B virus |
| HCV | Hepatitis C virus |
| HDV | Hepatitis D virus |
| EBV | Epstein-Barr virus |
| HPV | Human papilloma virus |
| Nox | NADPH-oxidase |
| MAO | Monoaminooxidase |
| ODC | Ornithine decarboxylase |
| ADC | Arginine decarboxylase |
| AZInI | Antizyme inhibitor |
| ARG | Arginase |
| OTC | Ornithine transcarbamylase |
| AGMAT | Agmatinase |
| SRM | Spermidine synthase |
| SMS | Spermine synthase |
| 3-AAP | N-acetyl-3-aminopropanal |
| 3-AP | 3-Aminopropanal |
| dcAdoMet | Decarboxylated S-adenosylmethionine |
| AdoMetDC | S-Adenosylmethionine decarboxylase |
| SSAT | Spermidine/spermine-N1-acetyltransferase |
| FAD | Flavin adenine dinucleotide |
| PAOX | Acetylpolyamine oxidase |
| SMOX | Spermine oxidase |
| DHPS | Deoxyhypusine synthase |
| DOHH | Deoxyhypusine hydroxylase |
| NF-κB | Nuclear factor-kappa B |
| Nrf2 | nuclear factor erythroid 2 (NFE2)-related factor 2 |
| PI3K | Phosphatidylinositol-4,5-bisphosphate 3-kinase |
| MAPK | Mitogen-activated protein kinase |
| SLC | Solute carrier |
| PRE | Polyamine response element |
| ARE | Antioxidant response element |
| AZ | Antizyme |
| Nqo1 | NADPH:quinone oxidoreductase 1 |
| PERK | Protein kinase R (PKR)-like ER kinase |
| CHOP | CCAAT/Enhancer-Binding Protein Homologous Protein |
| ATF4 | Activating transcription factor 4 |
| HIF-1α | Hypoxia inducible factor 1α |
| G-CSF | Granulocyte colony-stimulating factor |
| CXCL | Chemokine (C-X-C motif) ligand |
| FDMO | d,l-difluoromethylornithine |
| DPPH | 1,1-Diphenyl-2-picrylhydrazyl |
| PBMC | Peripheral blood mononuclear cells |
| eIF5A | Eukaryotic translation initiation factor 5A |
| HAT | Histone acetyl transferase |
| HSV | Human herpes virus |
| HCMV | Human cytomegalovirus |
| PDI | Protein disulfide isomerase |
| GSSG | Oxidized glutathione |
| GSH | Reduced glutathione |
| UPR | Unfolded protein response |
| Prdx4 | Peroxiredoxin 4 |
| GPx | Glutathione peroxidase |
| SERCA | Sarco/endoplasmic reticulum Ca2+-ATPase |
| IP3R | Inositol 1,4,5-trisphosphate receptor |
| RyR | Ryanodine receptor |
| SEPN1 | Selenoprotein N |
| VDAC | Voltage-dependent anion channel |
| MCU | Mitochondrial calcium uniporter channel |
| MICU1 | Mitochondrial calcium uptake 1 |
| MAMs | Mitochondria-associated membranes |
| PD-L1 | Programmed cell-death 1 ligand 1 |
| MDSCs | Myeloid-derived suppressor cells |
| MHC-1 | Major histocompatibility complex 1 |
References
- Fields, B.N.; Knipe, D.M.; Howley, P.M. Fields Virology, 5th ed.; Wolters Kluwer Health/Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2007. [Google Scholar]
- Lemon, S.M.; Walker, C.M.; Alter, M.J.; Yi, M.-K. Hepatitis C virus. In Fields Virology, 5th ed.; Knipe, D.M., Howley, P.M., Eds.; Lippincott, Williams & Wilkins: Philadelphia, PA, USA, 2007; Volume 1, pp. 1253–1304. [Google Scholar]
- Kuritzkes, D.R.; Walker, B.D. HIV-1 pathigenesis, clinical manifestations and treatment. In Fields Virology, 5th ed.; Knipe, D.M., Howley, P.M., Eds.; Lippincott, Williams & Wilkins: Philadelphia, PA, USA, 2007; Volume 1, pp. 2187–2214. [Google Scholar]
- Oh, J.K.; Weiderpass, E. Infection and cancer: Global distribution and burden of diseases. Ann. Glob. Health 2014, 80, 384–392. [Google Scholar] [CrossRef] [PubMed]
- Biagioli, M.C.; Kaul, P.; Singh, I.; Turner, R.B. The role of oxidative stress in rhinovirus induced elaboration of IL-8 by respiratory epithelial cells. Free Radic. Biol. Med. 1999, 26, 454–462. [Google Scholar] [CrossRef]
- Ivanov, A.V.; Valuev-Ellison, V.T.; Ivanova, O.N.; Kochetkov, S.N.; Starodubova, E.S.; Bartosch, B.; Isaguliants, M.G. Oxidative stress during HIV infection: Mechanisms and consequences. Oxid. Med. Cell. Longev. 2016, 2016, 8910396. [Google Scholar] [CrossRef] [PubMed]
- Ivanov, A.V.; Valuev-Elliston, V.T.; Tyurina, D.A.; Ivanova, O.N.; Kochetkov, S.N.; Bartosch, B.; Isaguliants, M.G. Oxidative stress, a trigger of hepatitis C and B virus-induced liver carcinogenesis. Oncotarget 2017, 8, 3895–3932. [Google Scholar] [CrossRef] [PubMed]
- Kuzmenko, Y.V.; Smirnova, O.A.; Ivanov, A.V.; Starodubova, E.S.; Karpov, V.L. Nonstructural Protein 1 of Tick-Borne Encephalitis Virus Induces Oxidative Stress and Activates Antioxidant Defense by the Nrf2/ARE Pathway. Intervirology 2016, 59, 111–117. [Google Scholar] [CrossRef] [PubMed]
- Ng, M.P.; Lee, J.C.; Loke, W.M.; Yeo, L.L.; Quek, A.M.; Lim, E.C.; Halliwell, B.; Seet, R.C. Does influenza A infection increase oxidative damage? Antioxid. Redox Signal. 2014, 21, 1025–1031. [Google Scholar] [CrossRef] [PubMed]
- Olagnier, D.; Peri, S.; Steel, C.; van Montfoort, N.; Chiang, C.; Beljanski, V.; Slifker, M.; He, Z.; Nichols, C.N.; Lin, R.; et al. Cellular oxidative stress response controls the antiviral and apoptotic programs in dengue virus-infected dendritic cells. PLoS Pathog. 2014, 10, e1004566. [Google Scholar] [CrossRef] [PubMed]
- Jamaluddin, M.; Tian, B.; Boldogh, I.; Garofalo, R.P.; Brasier, A.R. Respiratory syncytial virus infection induces a reactive oxygen species-MSK1-phospho-Ser-276 RelA pathway required for cytokine expression. J. Virol. 2009, 83, 10605–10615. [Google Scholar] [CrossRef] [PubMed]
- Sies, H. Oxidative stress: A concept in redox biology and medicine. Redox Biol. 2015, 4, 180–183. [Google Scholar] [CrossRef] [PubMed]
- Zorov, D.B.; Juhaszova, M.; Sollott, S.J. Mitochondrial reactive oxygen species (ROS) and ROS-induced ROS release. Physiol. Rev. 2014, 94, 909–950. [Google Scholar] [CrossRef] [PubMed]
- Michael, A.J. Biosynthesis of polyamines and polyamine-containing molecules. Biochem. J. 2016, 473, 2315–2329. [Google Scholar] [CrossRef] [PubMed]
- Van Leeuwenhoek, A. Observationes, D. Anthonii Leeuwenhoek, de natis e semine genitali animalculis. Philos. Trans. R. Soc. Lond. 1678, 12, 1040–1046. [Google Scholar] [CrossRef]
- Dudley, H.W.; Rosenheim, O.; Starling, W.W. The Chemical Constitution of Spermine: Structure and Synthesis. Biochem. J. 1926, 20, 1082–1094. [Google Scholar] [CrossRef] [PubMed]
- Khan, A.U.; Mei, Y.H.; Wilson, T. A proposed function for spermine and spermidine: Protection of replicating DNA against damage by singlet oxygen. Proc. Natl. Acad. Sci. USA 1992, 89, 11426–11427. [Google Scholar] [CrossRef] [PubMed]
- Tabor, H.; Tabor, C.W.; Irreverre, F. Quantitative determination of aliphatic diamines and polyamines by an automated liquid chromatography procedure. Anal. Biochem. 1973, 55, 457–467. [Google Scholar] [CrossRef]
- Pegg, A.E.; Wechter, R.S.; Clark, R.S.; Wiest, L.; Erwin, B.G. Acetylation of decarboxylated S-adenosylmethionine by mammalian cells. Biochemistry 1986, 25, 379–384. [Google Scholar] [CrossRef] [PubMed]
- Burtin, D.; Martin-Tanguy, J.; Paynot, M.; Rossin, N. Effects of the suicide inhibitors of arginine and ornithine decarboxylase activities on organogenesis, growth, free polyamine and hydroxycinnamoyl putrescine levels in leaf explants of Nicotiana Xanthi n.c. Cultivated in vitro in a medium producing callus formation. Plant Physiol. 1989, 89, 104–110. [Google Scholar] [PubMed]
- Satishchandran, C.; Boyle, S.M. Purification and properties of agmatine ureohydrolyase, a putrescine biosynthetic enzyme in Escherichia coli. J. Bacteriol. 1986, 165, 843–848. [Google Scholar] [CrossRef] [PubMed]
- Nakada, Y.; Itoh, Y. Identification of the putrescine biosynthetic genes in Pseudomonas aeruginosa and characterization of agmatine deiminase and N-carbamoylputrescine amidohydrolase of the arginine decarboxylase pathway. Microbiology 2003, 149, 707–714. [Google Scholar] [CrossRef] [PubMed]
- Mistry, S.K.; Burwell, T.J.; Chambers, R.M.; Rudolph-Owen, L.; Spaltmann, F.; Cook, W.J.; Morris, S.M., Jr. Cloning of human agmatinase. An alternate path for polyamine synthesis induced in liver by hepatitis B virus. Am. J. Physiol. Gastrointest. Liver Physiol. 2002, 282, G375–G381. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Regunathan, S.; Barrow, C.J.; Eshraghi, J.; Cooper, R.; Reis, D.J. Agmatine: An endogenous clonidine-displacing substance in the brain. Science 1994, 263, 966–969. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Regunathan, S.; Youngson, C.; Bramwell, S.; Reis, D.J. An antibody to agmatine localizes the amine in bovine adrenal chromaffin cells. Neurosci. Lett. 1995, 183, 17–21. [Google Scholar] [CrossRef]
- Regunathan, S.; Youngson, C.; Raasch, W.; Wang, H.; Reis, D.J. Imidazoline receptors and agmatine in blood vessels: A novel system inhibiting vascular smooth muscle proliferation. J. Pharmacol. Exp. Ther. 1996, 276, 1272–1282. [Google Scholar] [PubMed]
- Raasch, W.; Regunathan, S.; Li, G.; Reis, D.J. Agmatine, the bacterial amine, is widely distributed in mammalian tissues. Life Sci. 1995, 56, 2319–2330. [Google Scholar] [CrossRef]
- Bence, A.K.; Worthen, D.R.; Stables, J.P.; Crooks, P.A. An in vivo evaluation of the antiseizure activity and acute neurotoxicity of agmatine. Pharmacol. Biochem. Behav. 2003, 74, 771–775. [Google Scholar] [CrossRef]
- Regunathan, S.; Reis, D.J. Characterization of arginine decarboxylase in rat brain and liver: Distinction from ornithine decarboxylase. J. Neurochem. 2000, 74, 2201–2208. [Google Scholar] [CrossRef] [PubMed]
- Ramos-Molina, B.; Lopez-Contreras, A.J.; Lambertos, A.; Dardonville, C.; Cremades, A.; Penafiel, R. Influence of ornithine decarboxylase antizymes and antizyme inhibitors on agmatine uptake by mammalian cells. Amino Acids 2015, 47, 1025–1034. [Google Scholar] [CrossRef] [PubMed]
- Coleman, C.S.; Hu, G.; Pegg, A.E. Putrescine biosynthesis in mammalian tissues. Biochem. J. 2004, 379, 849–855. [Google Scholar] [CrossRef] [PubMed]
- Balasundaram, D.; Xie, Q.W.; Tabor, C.W.; Tabor, H. The presence of an active S-adenosylmethionine decarboxylase gene increases the growth defect observed in Saccharomyces cerevisiae mutants unable to synthesize putrescine, spermidine, and spermine. J. Bacteriol. 1994, 176, 6407–6409. [Google Scholar] [CrossRef] [PubMed]
- Pegg, A.E.; Xiong, H.; Feith, D.J.; Shantz, L.M. S-adenosylmethionine decarboxylase: Structure, function and regulation by polyamines. Biochem. Soc. Trans. 1998, 26, 580–586. [Google Scholar] [CrossRef] [PubMed]
- Hillary, R.A.; Pegg, A.E. Decarboxylases involved in polyamine biosynthesis and their inactivation by nitric oxide. Biochim. Biophys. Acta 2003, 1647, 161–166. [Google Scholar] [CrossRef]
- Finney, J.; Moon, H.J.; Ronnebaum, T.; Lantz, M.; Mure, M. Human copper-dependent amine oxidases. Arch. Biochem. Biophys. 2014, 546, 19–32. [Google Scholar] [CrossRef] [PubMed]
- Pegg, A.E.; Casero, R.A., Jr. Current status of the polyamine research field. Methods Mol. Biol. 2011, 720, 3–35. [Google Scholar] [PubMed]
- Wang, Y.; Devereux, W.; Woster, P.M.; Stewart, T.M.; Hacker, A.; Casero, R.A., Jr. Cloning and characterization of a human polyamine oxidase that is inducible by polyamine analogue exposure. Cancer Res. 2001, 61, 5370–5373. [Google Scholar] [PubMed]
- Yoshida, M.; Tomitori, H.; Machi, Y.; Hagihara, M.; Higashi, K.; Goda, H.; Ohya, T.; Niitsu, M.; Kashiwagi, K.; Igarashi, K. Acrolein toxicity: Comparison with reactive oxygen species. Biochem. Biophys. Res. Commun. 2009, 378, 313–318. [Google Scholar] [CrossRef] [PubMed]
- Van den Munckhof, R.J.; Denyn, M.; Tigchelaar-Gutter, W.; Schipper, R.G.; Verhofstad, A.A.; Van Noorden, C.J.; Frederiks, W.M. In situ substrate specificity and ultrastructural localization of polyamine oxidase activity in unfixed rat tissues. J. Histochem. Cytochem. 1995, 43, 1155–1162. [Google Scholar] [CrossRef] [PubMed]
- Murray-Stewart, T.; Wang, Y.; Goodwin, A.; Hacker, A.; Meeker, A.; Casero, R.A., Jr. Nuclear localization of human spermine oxidase isoforms—Possible implications in drug response and disease etiology. FEBS J. 2008, 275, 2795–2806. [Google Scholar] [CrossRef] [PubMed]
- Bonaiuto, E.; Grancara, S.; Martinis, P.; Stringaro, A.; Colone, M.; Agostinelli, E.; Macone, A.; Stevanato, R.; Vianello, F.; Toninello, A.; et al. A novel enzyme with spermine oxidase properties in bovine liver mitochondria: Identification and kinetic characterization. Free Radic. Biol. Med. 2015, 81, 88–99. [Google Scholar] [CrossRef] [PubMed]
- Pledgie, A.; Huang, Y.; Hacker, A.; Zhang, Z.; Woster, P.M.; Davidson, N.E.; Casero, R.A., Jr. Spermine oxidase SMO(PAOh1), Not N1-acetylpolyamine oxidase PAO, is the primary source of cytotoxic H2O2 in polyamine analogue-treated human breast cancer cell lines. J. Biol. Chem. 2005, 280, 39843–39851. [Google Scholar] [CrossRef] [PubMed]
- Ceci, R.; Duranti, G.; Leonetti, A.; Pietropaoli, S.; Spinozzi, F.; Marcocci, L.; Amendola, R.; Cecconi, F.; Sabatini, S.; Mariottini, P.; et al. Adaptive responses of heart and skeletal muscle to spermine oxidase overexpression: Evaluation of a new transgenic mouse model. Free Radic. Biol. Med. 2017, 103, 216–225. [Google Scholar] [CrossRef] [PubMed]
- Ou, Y.; Wang, S.J.; Li, D.; Chu, B.; Gu, W. Activation of SAT1 engages polyamine metabolism with p53-mediated ferroptotic responses. Proc. Natl. Acad. Sci. USA 2016, 113, E6806–E6812. [Google Scholar] [CrossRef] [PubMed]
- Pegg, A.E. Mammalian polyamine metabolism and function. IUBMB Life 2009, 61, 880–894. [Google Scholar] [CrossRef] [PubMed]
- Abdulhussein, A.A.; Wallace, H.M. Polyamines and membrane transporters. Amino Acids 2014, 46, 655–660. [Google Scholar] [CrossRef] [PubMed]
- Perez-Leal, O.; Merali, S. Regulation of polyamine metabolism by translational control. Amino Acids 2012, 42, 611–617. [Google Scholar] [CrossRef] [PubMed]
- Wagner, A.J.; Meyers, C.; Laimins, L.A.; Hay, N. c-Myc induces the expression and activity of ornithine decarboxylase. Cell Growth Differ. 1993, 4, 879–883. [Google Scholar] [PubMed]
- Packham, G.; Cleveland, J.L. Ornithine decarboxylase is a mediator of c-Myc-induced apoptosis. Mol. Cell. Biol. 1994, 14, 5741–5747. [Google Scholar] [CrossRef] [PubMed]
- Tacchini, L.; De Ponti, C.; Matteucci, E.; Follis, R.; Desiderio, M.A. Hepatocyte growth factor-activated NF-κB regulates HIF-1 activity and ODC expression, implicated in survival, differently in different carcinoma cell lines. Carcinogenesis 2004, 25, 2089–2100. [Google Scholar] [CrossRef] [PubMed]
- Babbar, N.; Casero, R.A., Jr. Tumor necrosis factor-α increases reactive oxygen species by inducing spermine oxidase in human lung epithelial cells: A potential mechanism for inflammation-induced carcinogenesis. Cancer Res. 2006, 66, 11125–11130. [Google Scholar] [CrossRef] [PubMed]
- Babbar, N.; Gerner, E.W.; Casero, R.A., Jr. Induction of spermidine/spermine N1-acetyltransferase (SSAT) by aspirin in Caco-2 colon cancer cells. Biochem. J. 2006, 394, 317–324. [Google Scholar] [CrossRef] [PubMed]
- Babbar, N.; Hacker, A.; Huang, Y.; Casero, R.A., Jr. Tumor necrosis factor α induces spermidine/spermine N1-acetyltransferase through nuclear factor κB in non-small cell lung cancer cells. J. Biol. Chem. 2006, 281, 24182–24192. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Xiao, L.; Thiagalingam, A.; Nelkin, B.D.; Casero, R.A., Jr. The identification of a cis-element and a trans-acting factor involved in the response to polyamines and polyamine analogues in the regulation of the human spermidine/spermine N1-acetyltransferase gene transcription. J. Biol. Chem. 1998, 273, 34623–34630. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Devereux, W.; Stewart, T.M.; Casero, R.A., Jr. Cloning and characterization of human polyamine-modulated factor-1, a transcriptional cofactor that regulates the transcription of the spermidine/spermine N1-acetyltransferase gene. J. Biol. Chem. 1999, 274, 22095–22101. [Google Scholar] [CrossRef] [PubMed]
- Smirnova, O.A.; Isaguliants, M.G.; Hyvonen, M.T.; Keinanen, T.A.; Tunitskaya, V.L.; Vepsalainen, J.; Alhonen, L.; Kochetkov, S.N.; Ivanov, A.V. Chemically induced oxidative stress increases polyamine levels by activating the transcription of ornithine decarboxylase and spermidine/spermine-N1-acetyltransferase in human hepatoma HUH7 cells. Biochimie 2012, 94, 1876–1883. [Google Scholar] [CrossRef] [PubMed]
- Wei, L.H.; Yang, Y.; Wu, G.; Ignarro, L.J. IL-4 and IL-13 upregulate ornithine decarboxylase expression by PI3K and MAP kinase pathways in vascular smooth muscle cells. Am. J. Physiol. Cell Physiol. 2008, 294, C1198–C1205. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Chen, Y.; Alm, K.; Vujcic, S.; Kramer, D.L.; Kee, K.; Diegelman, P.; Porter, C.W. The role of mitogen-activated protein kinase activation in determining cellular outcomes in polyamine analogue-treated human melanoma cells. Cancer Res. 2003, 63, 3619–3625. [Google Scholar] [PubMed]
- Wang, C.; Ruan, P.; Zhao, Y.; Li, X.; Wang, J.; Wu, X.; Liu, T.; Wang, S.; Hou, J.; Li, W.; et al. Spermidine/spermine N1-acetyltransferase regulates cell growth and metastasis via AKT/β-catenin signaling pathways in hepatocellular and colorectal carcinoma cells. Oncotarget 2017, 8, 1092–1109. [Google Scholar] [PubMed]
- Pegg, A.E.; McCann, P.P. Polyamine metabolism and function. Am. J. Physiol. 1982, 243, C212–C221. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.J.; Pan, H.; Verma, A.K. Non-AP-1 tumor promoter 12-O-tetradecanoylphorbol-13-acetate-responsive sequences in the human ornithine decarboxylase gene. Mol. Carcinog. 1994, 10, 169–179. [Google Scholar] [CrossRef] [PubMed]
- Aleksunes, L.M.; Manautou, J.E. Emerging role of Nrf2 in protecting against hepatic and gastrointestinal disease. Toxicol. Pathol. 2007, 35, 459–473. [Google Scholar] [CrossRef] [PubMed]
- Forshell, T.P.; Rimpi, S.; Nilsson, J.A. Chemoprevention of B-cell lymphomas by inhibition of the Myc target spermidine synthase. Cancer Prev. Res. 2010, 3, 140–147. [Google Scholar] [CrossRef] [PubMed]
- Nilsson, J.A.; Keller, U.B.; Baudino, T.A.; Yang, C.; Norton, S.; Old, J.A.; Nilsson, L.M.; Neale, G.; Kramer, D.L.; Porter, C.W.; et al. Targeting ornithine decarboxylase in Myc-induced lymphomagenesis prevents tumor formation. Cancer Cell 2005, 7, 433–444. [Google Scholar] [CrossRef] [PubMed]
- Pegg, A.E. Regulation of ornithine decarboxylase. J. Biol. Chem. 2006, 281, 14529–14532. [Google Scholar] [CrossRef] [PubMed]
- Asher, G.; Bercovich, Z.; Tsvetkov, P.; Shaul, Y.; Kahana, C. 20S proteasomal degradation of ornithine decarboxylase is regulated by NQO1. Mol. Cell 2005, 17, 645–655. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Contreras, A.J.; Ramos-Molina, B.; Martinez-de-la-Torre, M.; Penafiel-Verdu, C.; Puelles, L.; Cremades, A.; Penafiel, R. Expression of antizyme inhibitor 2 in male haploid germinal cells suggests a role in spermiogenesis. Int. J. Biochem. Cell Biol. 2009, 41, 1070–1078. [Google Scholar] [CrossRef] [PubMed]
- Pitkanen, L.T.; Heiskala, M.; Andersson, L.C. Expression of a novel human ornithine decarboxylase-like protein in the central nervous system and testes. Biochem. Biophys. Res. Commun. 2001, 287, 1051–1057. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Hacker, A.; Murray-Stewart, T.; Fleischer, J.G.; Woster, P.M.; Casero, R.A., Jr. Induction of human spermine oxidase SMO(PAOh1) is regulated at the levels of new mRNA synthesis, mRNA stabilization and newly synthesized protein. Biochem. J. 2005, 386, 543–547. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Casero, R.A., Jr. Mammalian polyamine catabolism: A therapeutic target, a pathological problem, or both? J. Biochem. 2006, 139, 17–25. [Google Scholar] [CrossRef] [PubMed]
- Zahedi, K.; Wang, Z.; Barone, S.; Prada, A.E.; Kelly, C.N.; Casero, R.A.; Yokota, N.; Porter, C.W.; Rabb, H.; Soleimani, M. Expression of SSAT, a novel biomarker of tubular cell damage, increases in kidney ischemia-reperfusion injury. Am. J. Physiol. Renal Physiol. 2003, 284, F1046–F1055. [Google Scholar] [CrossRef] [PubMed]
- Cervelli, M.; Fratini, E.; Amendola, R.; Bianchi, M.; Signori, E.; Ferraro, E.; Lisi, A.; Federico, R.; Marcocci, L.; Mariottini, P. Increased spermine oxidase (SMO) activity as a novel differentiation marker of myogenic C2C12 cells. Int. J. Biochem. Cell Biol. 2009, 41, 934–944. [Google Scholar] [CrossRef] [PubMed]
- Casero, R.A.; Pegg, A.E. Polyamine catabolism and disease. Biochem. J. 2009, 421, 323–338. [Google Scholar] [CrossRef] [PubMed]
- Soulet, D.; Gagnon, B.; Rivest, S.; Audette, M.; Poulin, R. A fluorescent probe of polyamine transport accumulates into intracellular acidic vesicles via a two-step mechanism. J. Biol. Chem. 2004, 279, 49355–49366. [Google Scholar] [CrossRef] [PubMed]
- Ha, H.C.; Sirisoma, N.S.; Kuppusamy, P.; Zweier, J.L.; Woster, P.M.; Casero, R.A., Jr. The natural polyamine spermine functions directly as a free radical scavenger. Proc. Natl. Acad. Sci. USA 1998, 95, 11140–11145. [Google Scholar] [CrossRef] [PubMed]
- Park, I.-H.; Lim, M.-M. Inhibitory Effect of Spermidine with Antioxidant Activity on Oxidative Stress in Human Dermal Fibroblasts. J. Life Sci. 2011, 21, 693–699. [Google Scholar] [CrossRef]
- Battaglia, V.; Rossi, C.A.; Colombatto, S.; Grillo, M.A.; Toninello, A. Different behavior of agmatine in liver mitochondria: Inducer of oxidative stress or scavenger of reactive oxygen species? Biochim. Biophys. Acta 2007, 1768, 1147–1153. [Google Scholar] [CrossRef] [PubMed]
- Gaboriau, F.; Vaultier, M.; Moulinoux, J.P.; Delcros, J.G. Antioxidative properties of natural polyamines and dimethylsilane analogues. Redox Rep. 2005, 10, 9–18. [Google Scholar] [CrossRef] [PubMed]
- Das, K.C.; Misra, H.P. Hydroxyl radical scavenging and singlet oxygen quenching properties of polyamines. Mol. Cell. Biochem. 2004, 262, 127–133. [Google Scholar] [CrossRef] [PubMed]
- Sava, I.G.; Battaglia, V.; Rossi, C.A.; Salvi, M.; Toninello, A. Free radical scavenging action of the natural polyamine spermine in rat liver mitochondria. Free Radic. Biol. Med. 2006, 41, 1272–1281. [Google Scholar] [CrossRef] [PubMed]
- Rigobello, M.P.; Toninello, A.; Siliprandi, D.; Bindoli, A. Effect of spermine on mitochondrial glutathione release. Biochem. Biophys. Res. Commun. 1993, 194, 1276–1281. [Google Scholar] [CrossRef] [PubMed]
- Muscari, C.; Guarnieri, C.; Stefanelli, C.; Giaccari, A.; Caldarera, C.M. Protective effect of spermine on DNA exposed to oxidative stress. Mol. Cell. Biochem. 1995, 144, 125–129. [Google Scholar] [CrossRef] [PubMed]
- Ha, H.C.; Yager, J.D.; Woster, P.A.; Casero, R.A., Jr. Structural specificity of polyamines and polyamine analogues in the protection of DNA from strand breaks induced by reactive oxygen species. Biochem. Biophys. Res. Commun. 1998, 244, 298–303. [Google Scholar] [CrossRef] [PubMed]
- Rider, J.E.; Hacker, A.; Mackintosh, C.A.; Pegg, A.E.; Woster, P.M.; Casero, R.A. Spermine and spermidine mediate protection against oxidative damage caused by hydrogen peroxide. Amino Acids 2007, 33, 231–240. [Google Scholar] [CrossRef] [PubMed]
- Jung, I.L.; Oh, T.J.; Kim, I.G. Abnormal growth of polyamine-deficient Escherichia coli mutant is partially caused by oxidative stress-induced damage. Arch. Biochem. Biophys. 2003, 418, 125–132. [Google Scholar] [CrossRef] [PubMed]
- Chattopadhyay, M.K.; Tabor, C.W.; Tabor, H. Polyamine deficiency leads to accumulation of reactive oxygen species in a spe2Delta mutant of Saccharomyces cerevisiae. Yeast 2006, 23, 751–761. [Google Scholar] [CrossRef] [PubMed]
- Eisenberg, T.; Knauer, H.; Schauer, A.; Buttner, S.; Ruckenstuhl, C.; Carmona-Gutierrez, D.; Ring, J.; Schroeder, S.; Magnes, C.; Antonacci, L.; et al. Induction of autophagy by spermidine promotes longevity. Nat. Cell Biol. 2009, 11, 1305–1314. [Google Scholar] [CrossRef] [PubMed]
- Von Deutsch, A.W.; Mitchell, C.D.; Williams, C.E.; Dutt, K.; Silvestrov, N.A.; Klement, B.J.; Abukhalaf, I.K.; von Deutsch, D.A. Polyamines protect against radiation-induced oxidative stress. Gravit. Space Biol. Bull. 2005, 18, 109–110. [Google Scholar] [PubMed]
- Marzabadi, M.R.; Llvaas, E. Spermine prevent iron accumulation and depress lipofuscin accumulation in cultured myocardial cells. Free Radic. Biol. Med. 1996, 21, 375–381. [Google Scholar] [CrossRef]
- Minois, N.; Carmona-Gutierrez, D.; Bauer, M.A.; Rockenfeller, P.; Eisenberg, T.; Brandhorst, S.; Sigrist, S.J.; Kroemer, G.; Madeo, F. Spermidine promotes stress resistance in Drosophila melanogaster through autophagy-dependent and -independent pathways. Cell Death Dis. 2012, 3, e401. [Google Scholar] [CrossRef] [PubMed]
- Jeong, J.W.; Cha, H.J.; Han, M.H.; Hwang, S.J.; Lee, D.S.; Yoo, J.S.; Choi, I.W.; Kim, S.; Kim, H.S.; Kim, G.Y.; et al. Spermidine Protects against Oxidative Stress in Inflammation Models Using Macrophages and Zebrafish. Biomol. Ther. 2018, 26, 146–156. [Google Scholar] [CrossRef] [PubMed]
- Childs, A.C.; Mehta, D.J.; Gerner, E.W. Polyamine-dependent gene expression. Cell. Mol. Life Sci. 2003, 60, 1394–1406. [Google Scholar] [CrossRef] [PubMed]
- Han, X.; Tomitori, H.; Mizuno, S.; Higashi, K.; Full, C.; Fukiwake, T.; Terui, Y.; Leewanich, P.; Nishimura, K.; Toida, T.; et al. Binding of spermine and ifenprodil to a purified, soluble regulatory domain of the N-methyl-d-aspartate receptor. J. Neurochem. 2008, 107, 1566–1577. [Google Scholar] [CrossRef] [PubMed]
- Mott, D.D.; Washburn, M.S.; Zhang, S.; Dingledine, R.J. Subunit-dependent modulation of kainate receptors by extracellular protons and polyamines. J. Neurosci. 2003, 23, 1179–1188. [Google Scholar] [CrossRef] [PubMed]
- Ishihara, K.; Yan, D.H. Low-affinity spermine block mediating outward currents through Kir2.1 and Kir2.2 inward rectifier potassium channels. J. Physiol. 2007, 583, 891–908. [Google Scholar] [CrossRef] [PubMed]
- Desiderio, M.A.; Tacchini, L.; Anzon, E.; Pogliaghi, G.; Radice, L.; Bernelli-Zazzera, A. Effects of polyamine imbalance on the induction of stress genes in hepatocarcinoma cells exposed to heat shock. Hepatology 1996, 24, 150–156. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Guo, X.; Rao, J.N.; Zou, T.; Marasa, B.S.; Chen, J.; Greenspon, J.; Casero, R.A., Jr.; Wang, J.Y. Polyamine-modulated c-Myc expression in normal intestinal epithelial cells regulates p21Cip1 transcription through a proximal promoter region. Biochem. J. 2006, 398, 257–267. [Google Scholar] [CrossRef] [PubMed]
- Xiao, L.; Rao, J.N.; Zou, T.; Liu, L.; Marasa, B.S.; Chen, J.; Turner, D.J.; Passaniti, A.; Wang, J.Y. Induced JunD in intestinal epithelial cells represses CDK4 transcription through its proximal promoter region following polyamine depletion. Biochem. J. 2007, 403, 573–581. [Google Scholar] [CrossRef] [PubMed]
- Monti, M.G.; Ghiaroni, S.; Pernecco, L.; Barbieri, D.; Marverti, G.; Franceschi, C. Polyamine depletion protects HL-60 cells from 2-deoxy-d-ribose-induced apoptosis. Life Sci. 1998, 62, 799–806. [Google Scholar] [CrossRef]
- Monti, M.G.; Ghiaroni, S.; Marverti, G.; Montanari, M.; Moruzzi, M.S. Polyamine depletion switches the form of 2-deoxy-d-ribose-induced cell death from apoptosis to necrosis in HL-60 cells. Int. J. Biochem. Cell Biol. 2004, 36, 1238–1248. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.J.; Chen, Y.; Huang, Y.P.; Hsu, Y.C.; Chiang, L.H.; Chen, T.Y.; Wang, G.J. Exogenous polyamines promote osteogenic differentiation by reciprocally regulating osteogenic and adipogenic gene expression. J. Cell. Biochem. 2013, 114, 2718–2728. [Google Scholar] [CrossRef] [PubMed]
- Quemener, V.; Blanchard, Y.; Lescoat, D.; Havouis, R.; Moulinoux, J.P. Depletion in nuclear spermine during human spermatogenesis, a natural process of cell differentiation. Am. J. Physiol. 1992, 263, C343–C347. [Google Scholar] [CrossRef] [PubMed]
- Pietila, M.; Pirinen, E.; Keskitalo, S.; Juutinen, S.; Pasonen-Seppanen, S.; Keinanen, T.; Alhonen, L.; Janne, J. Disturbed keratinocyte differentiation in transgenic mice and organotypic keratinocyte cultures as a result of spermidine/spermine N-acetyltransferase overexpression. J. Investig. Dermatol. 2005, 124, 596–601. [Google Scholar] [CrossRef] [PubMed]
- Park, M.H.; Cooper, H.L.; Folk, J.E. Identification of hypusine, an unusual amino acid, in a protein from human lymphocytes and of spermidine as its biosynthetic precursor. Proc. Natl. Acad. Sci. USA 1981, 78, 2869–2873. [Google Scholar] [CrossRef] [PubMed]
- Park, M.H. The post-translational synthesis of a polyamine-derived amino acid, hypusine, in the eukaryotic translation initiation factor 5A (eIF5A). J. Biochem. 2006, 139, 161–169. [Google Scholar] [CrossRef] [PubMed]
- Saini, P.; Eyler, D.E.; Green, R.; Dever, T.E. Hypusine-containing protein eIF5A promotes translation elongation. Nature 2009, 459, 118–121. [Google Scholar] [CrossRef] [PubMed]
- Gutierrez, E.; Shin, B.S.; Woolstenhulme, C.J.; Kim, J.R.; Saini, P.; Buskirk, A.R.; Dever, T.E. eIF5A promotes translation of polyproline motifs. Mol. Cell 2013, 51, 35–45. [Google Scholar] [CrossRef] [PubMed]
- Nishimura, K.; Nakatsu, F.; Kashiwagi, K.; Ohno, H.; Saito, T.; Igarashi, K. Essential role of S-adenosylmethionine decarboxylase in mouse embryonic development. Genes Cells 2002, 7, 41–47. [Google Scholar] [CrossRef] [PubMed]
- Eisenberg, T.; Abdellatif, M.; Schroeder, S.; Primessnig, U.; Stekovic, S.; Pendl, T.; Harger, A.; Schipke, J.; Zimmermann, A.; Schmidt, A.; et al. Cardioprotection and lifespan extension by the natural polyamine spermidine. Nat. Med. 2016, 22, 1428–1438. [Google Scholar] [CrossRef] [PubMed]
- Yue, F.; Li, W.; Zou, J.; Jiang, X.; Xu, G.; Huang, H.; Liu, L. Spermidine Prolongs Lifespan and Prevents Liver Fibrosis and Hepatocellular Carcinoma by Activating MAP1S-Mediated Autophagy. Cancer Res. 2017, 77, 2938–2951. [Google Scholar] [CrossRef] [PubMed]
- Rahman, I. Oxidative stress, transcription factors and chromatin remodelling in lung inflammation. Biochem. Pharmacol. 2002, 64, 935–942. [Google Scholar] [CrossRef]
- Rahman, I.; Gilmour, P.S.; Jimenez, L.A.; MacNee, W. Oxidative stress and TNF-α induce histone acetylation and NF-κB/AP-1 activation in alveolar epithelial cells: Potential mechanism in gene transcription in lung inflammation. Mol. Cell. Biochem. 2002, 234–235, 239–248. [Google Scholar] [CrossRef] [PubMed]
- Rahman, I.; Marwick, J.; Kirkham, P. Redox modulation of chromatin remodeling: Impact on histone acetylation and deacetylation, NF-κB and pro-inflammatory gene expression. Biochem. Pharmacol. 2004, 68, 1255–1267. [Google Scholar] [CrossRef] [PubMed]
- Choudhury, M.; Park, P.H.; Jackson, D.; Shukla, S.D. Evidence for the role of oxidative stress in the acetylation of histone H3 by ethanol in rat hepatocytes. Alcohol 2010, 44, 531–540. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Gibson, S.B. Is mitochondrial generation of reactive oxygen species a trigger for autophagy? Autophagy 2008, 4, 246–248. [Google Scholar] [CrossRef] [PubMed]
- Wu, R.F.; Ma, Z.; Liu, Z.; Terada, L.S. Nox4-derived H2O2 mediates endoplasmic reticulum signaling through local Ras activation. Mol. Cell. Biol. 2010, 30, 3553–3568. [Google Scholar] [CrossRef] [PubMed]
- Zhao, S.; Luo, H.; Kan, G.; Zhao, Y.; Lin, L.; Tang, Q.; Yu, C.; Sun, W.; Cai, L.; Cui, S. The protective role of autophagy in Heterocephalus glaber hepatic stellate cells exposed to H2O2 or nutritional stress. Cell. Physiol. Biochem. 2014, 34, 463–473. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.J.; Cho, K.S.; Koh, J.Y. Oxidative injury triggers autophagy in astrocytes: The role of endogenous zinc. Glia 2009, 57, 1351–1361. [Google Scholar] [CrossRef] [PubMed]
- Megosh, L.; Gilmour, S.K.; Rosson, D.; Soler, A.P.; Blessing, M.; Sawicki, J.A.; O’Brien, T.G. Increased frequency of spontaneous skin tumors in transgenic mice which overexpress ornithine decarboxylase. Cancer Res. 1995, 55, 4205–4209. [Google Scholar] [PubMed]
- Nowotarski, S.L.; Woster, P.M.; Casero, R.A., Jr. Polyamines and cancer: Implications for chemotherapy and chemoprevention. Expert Rev. Mol. Med. 2013, 15, e3. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, T.G.; Megosh, L.C.; Gilliard, G.; Soler, A.P. Ornithine decarboxylase overexpression is a sufficient condition for tumor promotion in mouse skin. Cancer Res. 1997, 57, 2630–2637. [Google Scholar] [PubMed]
- Hayes, C.S.; DeFeo, K.; Lan, L.; Paul, B.; Sell, C.; Gilmour, S.K. Elevated levels of ornithine decarboxylase cooperate with Raf/ERK activation to convert normal keratinocytes into invasive malignant cells. Oncogene 2006, 25, 1543–1553. [Google Scholar] [CrossRef] [PubMed]
- Shukla-Dave, A.; Castillo-Martin, M.; Chen, M.; Lobo, J.; Gladoun, N.; Collazo-Lorduy, A.; Khan, F.M.; Ponomarev, V.; Yi, Z.; Zhang, W.; et al. Ornithine Decarboxylase Is Sufficient for Prostate Tumorigenesis via Androgen Receptor Signaling. Am. J. Pathol. 2016, 186, 3131–3145. [Google Scholar] [CrossRef] [PubMed]
- Lan, L.; Hayes, C.S.; Laury-Kleintop, L.; Gilmour, S.K. Suprabasal induction of ornithine decarboxylase in adult mouse skin is sufficient to activate keratinocytes. J. Investig. Dermatol. 2005, 124, 602–614. [Google Scholar] [CrossRef] [PubMed]
- Chaturvedi, R.; Asim, M.; Romero-Gallo, J.; Barry, D.P.; Hoge, S.; de Sablet, T.; Delgado, A.G.; Wroblewski, L.E.; Piazuelo, M.B.; Yan, F.; et al. Spermine oxidase mediates the gastric cancer risk associated with Helicobacter pylori CagA. Gastroenterology 2011, 141. [Google Scholar] [CrossRef] [PubMed]
- Chaturvedi, R.; de Sablet, T.; Asim, M.; Piazuelo, M.B.; Barry, D.P.; Verriere, T.G.; Sierra, J.C.; Hardbower, D.M.; Delgado, A.G.; Schneider, B.G.; et al. Increased Helicobacter pylori-associated gastric cancer risk in the Andean region of Colombia is mediated by spermine oxidase. Oncogene 2015, 34, 3429–3440. [Google Scholar] [CrossRef] [PubMed]
- Goodwin, A.C.; Destefano Shields, C.E.; Wu, S.; Huso, D.L.; Wu, X.; Murray-Stewart, T.R.; Hacker-Prietz, A.; Rabizadeh, S.; Woster, P.M.; Sears, C.L.; et al. Polyamine catabolism contributes to enterotoxigenic Bacteroides fragilis-induced colon tumorigenesis. Proc. Natl. Acad. Sci. USA 2011, 108, 15354–15359. [Google Scholar] [CrossRef] [PubMed]
- Snezhkina, A.V.; Krasnov, G.S.; Lipatova, A.V.; Sadritdinova, A.F.; Kardymon, O.L.; Fedorova, M.S.; Melnikova, N.V.; Stepanov, O.A.; Zaretsky, A.R.; Kaprin, A.D.; et al. The Dysregulation of Polyamine Metabolism in Colorectal Cancer Is Associated with Overexpression of c-Myc and C/EBPβ rather than Enterotoxigenic Bacteroides fragilis Infection. Oxid. Med. Cell. Longev. 2016, 2016, 2353560. [Google Scholar] [CrossRef] [PubMed]
- Kelly, D.C.; Barwise, A.H.; Walker, I.O. DNA contained by two densonucleosis viruses. J. Virol. 1977, 21, 396–407. [Google Scholar] [PubMed]
- Lanzer, W.; Holowczak, J.A. Polyamines in vaccinia virions and polypeptides released from viral cores by acid extraction. J. Virol. 1975, 16, 1254–1264. [Google Scholar] [PubMed]
- Fukuma, I.; Cohen, S.S. Polyamines in bacteriophage R17 and its RNA. J. Virol. 1975, 16, 222–227. [Google Scholar] [PubMed]
- Sheppard, S.L.; Burness, A.T.; Boyle, S.M. Polyamines in encephalomyocarditis virus. J. Virol. 1980, 34, 266–267. [Google Scholar] [PubMed]
- Gibson, W.; Roizman, B. Compartmentalization of spermine and spermidine in the herpes simplex virion. Proc. Natl. Acad. Sci. USA 1971, 68, 2818–2821. [Google Scholar] [CrossRef] [PubMed]
- Bachrach, U.; Don, S.; Wiener, H. Occurrence of polyamines in myxoviruses. J. Gen. Virol. 1974, 22, 451–454. [Google Scholar] [CrossRef] [PubMed]
- Fout, G.S.; Medappa, K.C.; Mapoles, J.E.; Rueckert, R.R. Radiochemical determination of polyamines in poliovirus and human rhinovirus 14. J. Biol. Chem. 1984, 259, 3639–3643. [Google Scholar] [PubMed]
- Raina, A.; Tuomi, K.; Mantyjarvi, R. Roles of polyamines in the replication of animal viruses. Med. Biol. 1981, 59, 428–432. [Google Scholar] [PubMed]
- Mounce, B.C.; Cesaro, T.; Moratorio, G.; Hooikaas, P.J.; Yakovleva, A.; Werneke, S.W.; Smith, E.C.; Poirier, E.Z.; Simon-Loriere, E.; Prot, M.; et al. Inhibition of Polyamine Biosynthesis Is a Broad-Spectrum Strategy against RNA Viruses. J. Virol. 2016, 90, 9683–9692. [Google Scholar] [CrossRef] [PubMed]
- Mounce, B.C.; Poirier, E.Z.; Passoni, G.; Simon-Loriere, E.; Cesaro, T.; Prot, M.; Stapleford, K.A.; Moratorio, G.; Sakuntabhai, A.; Levraud, J.P.; et al. Interferon-Induced Spermidine-Spermine Acetyltransferase and Polyamine Depletion Restrict Zika and Chikungunya Viruses. Cell Host Microbe 2016, 20, 167–177. [Google Scholar] [CrossRef] [PubMed]
- Olsen, M.E.; Filone, C.M.; Rozelle, D.; Mire, C.E.; Agans, K.N.; Hensley, L.; Connor, J.H. Polyamines and Hypusination Are Required for Ebolavirus Gene Expression and Replication. mBio 2016, 7. [Google Scholar] [CrossRef] [PubMed]
- Greco, A.; Calle, A.; Morfin, F.; Thouvenot, D.; Cayre, M.; Kindbeiter, K.; Martin, L.; Levillain, O.; Diaz, J.J. S-adenosyl methionine decarboxylase activity is required for the outcome of herpes simplex virus type 1 infection and represents a new potential therapeutic target. FASEB J. 2005, 19, 1128–1130. [Google Scholar] [CrossRef] [PubMed]
- Gibson, W.; van Breemen, R.; Fields, A.; LaFemina, R.; Irmiere, A. d,l-α-Difluoromethylornithine inhibits human cytomegalovirus replication. J. Virol. 1984, 50, 145–154. [Google Scholar] [PubMed]
- Tyms, A.S.; Scamans, E.; Williamson, J.D. Polyamine metabolism in MRC5 cells infected with different herpesviruses. Biochem. Biophys. Res. Commun. 1979, 86, 312–318. [Google Scholar] [CrossRef]
- Tyms, A.S.; Williamson, J.D. Inhibitors of polyamine biosynthesis block human cytomegalovirus replication. Nature 1982, 297, 690–691. [Google Scholar] [CrossRef] [PubMed]
- Hoque, M.; Hanauske-Abel, H.M.; Palumbo, P.; Saxena, D.; D’Alliessi Gandolfi, D.; Park, M.H.; Pe’ery, T.; Mathews, M.B. Inhibition of HIV-1 gene expression by Ciclopirox and Deferiprone, drugs that prevent hypusination of eukaryotic initiation factor 5A. Retrovirology 2009, 6, 90. [Google Scholar] [CrossRef] [PubMed]
- Mounce, B.C.; Cesaro, T.; Vlajnic, L.; Vidina, A.; Vallet, T.; Weger-Lucarelli, J.; Passoni, G.; Stapleford, K.A.; Levraud, J.P.; Vignuzzi, M. Chikungunya Virus Overcomes Polyamine Depletion by Mutation of nsP1 and the Opal Stop Codon to Confer Enhanced Replication and Fitness. J. Virol. 2017, 91. [Google Scholar] [CrossRef] [PubMed]
- McCormick, F. Polyamine turnover and leakage during infection of HeLa and L-cells with herpes simplex virus type 1. Virology 1978, 91, 496–503. [Google Scholar] [CrossRef]
- Gibson, W.; Roizman, B. The structural role and metabolic involvement of polyamines with herpes simplex virus. In Polyamines in Normal and Neoplastic Growth; Russell, D.H., Ed.; Raven Press, Inc.: New York, NY, USA, 1973. [Google Scholar]
- McCormick, F.P.; Newton, A.A. Polyamine metabolism in cells infected with herpes simplex virus. J. Gen. Virol. 1975, 27, 25–33. [Google Scholar] [CrossRef] [PubMed]
- Isom, H.C. Stimulation of ornithine decarboxylase by human cytomegalovirus. J. Gen. Virol. 1979, 42, 265–278. [Google Scholar] [CrossRef] [PubMed]
- Clarke, J.R.; Tyms, A.S. Polyamine biosynthesis in cells infected with different clinical isolates of human cytomegalovirus. J. Med. Virol. 1991, 34, 212–216. [Google Scholar] [CrossRef] [PubMed]
- Garnett, H.M. Altered polyamine concentrations in cytomegalovirus-infected human cells. S. Afr. Med. J. 1988, 73, 209–211. [Google Scholar] [PubMed]
- Hodgson, J.; Williamson, J.D. Ornithine decarboxylase activity in uninfected and vaccinia virus-infected HeLa cells. Biochem. Biophys. Res. Commun. 1975, 63, 308–312. [Google Scholar] [CrossRef]
- Colombatto, S.; De Agostini, M.; Corsi, D.; Sinicco, A. Polyamines in lymphocytes from patients infected by human immunodeficiency virus. Biol. Chem. Hoppe Seyler 1989, 370, 745–748. [Google Scholar] [CrossRef] [PubMed]
- White, E.L.; Rose, L.M.; Allan, P.W.; Buckheit, R.W., Jr.; Shannon, W.M.; Secrist, J.A., III. Polyamine pools in HIV-infected cells. J. Acquir. Immune Defic. Syndr. Hum. Retrovirol. 1998, 17, 101–103. [Google Scholar] [CrossRef] [PubMed]
- Capone, C.; Cervelli, M.; Angelucci, E.; Colasanti, M.; Macone, A.; Mariottini, P.; Persichini, T. A role for spermine oxidase as a mediator of reactive oxygen species production in HIV-Tat-induced neuronal toxicity. Free Radic. Biol. Med. 2013, 63, 99–107. [Google Scholar] [CrossRef] [PubMed]
- Smirnova, O.A.; Keinanen, T.A.; Ivanova, O.N.; Hyvonen, M.T.; Khomutov, A.R.; Kochetkov, S.N.; Bartosch, B.; Ivanov, A.V. Hepatitis C virus alters metabolism of biogenic polyamines by affecting expression of key enzymes of their metabolism. Biochem. Biophys. Res. Commun. 2017, 483, 904–909. [Google Scholar] [CrossRef] [PubMed]
- Korovina, A.N.; Tunitskaya, V.L.; Khomutov, M.A.; Simonian, A.R.; Khomutov, A.R.; Ivanov, A.I.; Kochetkov, S.N. Biogenic Polyamines Spermine and Spermidine Activate RNA Polymerase and Inhibit RNA Helicase of Hepatitis C Virus. Biochemistry 2012, 77, 1172–1180. [Google Scholar] [CrossRef] [PubMed]
- Kaiser, A.; Vollmert, M.; Tholl, D.; Graves, M.V.; Gurnon, J.R.; Xing, W.; Lisec, A.D.; Nickerson, K.W.; Van Etten, J.L. Chlorella virus PBCV-1 encodes a functional homospermidine synthase. Virology 1999, 263, 254–262. [Google Scholar] [CrossRef] [PubMed]
- Morehead, T.A.; Gurnon, J.R.; Adams, B.; Nickerson, K.W.; Fitzgerald, L.A.; Van Etten, J.L. Ornithine decarboxylase encoded by chlorella virus PBCV-1. Virology 2002, 301, 165–175. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ellgaard, L.; Ruddock, L.W. The human protein disulphide isomerase family: Substrate interactions and functional properties. EMBO Rep. 2005, 6, 28–32. [Google Scholar] [CrossRef] [PubMed]
- Sato, Y.; Kojima, R.; Okumura, M.; Hagiwara, M.; Masui, S.; Maegawa, K.; Saiki, M.; Horibe, T.; Suzuki, M.; Inaba, K. Synergistic cooperation of PDI family members in peroxiredoxin 4-driven oxidative protein folding. Sci. Rep. 2013, 3, 2456. [Google Scholar] [CrossRef] [PubMed]
- Oka, O.B.; Bulleid, N.J. Forming disulfides in the endoplasmic reticulum. Biochim. Biophys. Acta 2013, 1833, 2425–2429. [Google Scholar] [CrossRef] [PubMed]
- Jessop, C.E.; Watkins, R.H.; Simmons, J.J.; Tasab, M.; Bulleid, N.J. Protein disulphide isomerase family members show distinct substrate specificity: P5 is targeted to BiP client proteins. J. Cell Sci. 2009, 122, 4287–4295. [Google Scholar] [CrossRef] [PubMed]
- Frand, A.R.; Kaiser, C.A. The ERO1 gene of yeast is required for oxidation of protein dithiols in the endoplasmic reticulum. Mol. Cell 1998, 1, 161–170. [Google Scholar] [CrossRef]
- Pollard, M.G.; Travers, K.J.; Weissman, J.S. Ero1p: A novel and ubiquitous protein with an essential role in oxidative protein folding in the endoplasmic reticulum. Mol. Cell 1998, 1, 171–182. [Google Scholar] [CrossRef]
- Cabibbo, A.; Pagani, M.; Fabbri, M.; Rocchi, M.; Farmery, M.R.; Bulleid, N.J.; Sitia, R. ERO1-L, a human protein that favors disulfide bond formation in the endoplasmic reticulum. J. Biol. Chem. 2000, 275, 4827–4833. [Google Scholar] [CrossRef] [PubMed]
- Pagani, M.; Fabbri, M.; Benedetti, C.; Fassio, A.; Pilati, S.; Bulleid, N.J.; Cabibbo, A.; Sitia, R. Endoplasmic reticulum oxidoreductin 1-Lβ (ERO1-Lβ), a human gene induced in the course of the unfolded protein response. J. Biol. Chem. 2000, 275, 23685–23692. [Google Scholar] [CrossRef] [PubMed]
- Dias-Gunasekara, S.; Gubbens, J.; van Lith, M.; Dunne, C.; Williams, J.A.; Kataky, R.; Scoones, D.; Lapthorn, A.; Bulleid, N.J.; Benham, A.M. Tissue-specific expression and dimerization of the endoplasmic reticulum oxidoreductase Ero1β. J. Biol. Chem. 2005, 280, 33066–33075. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Zhu, L.; Wang, C.C. The endoplasmic reticulum sulfhydryl oxidase Ero1β drives efficient oxidative protein folding with loose regulation. Biochem. J. 2011, 434, 113–121. [Google Scholar] [CrossRef] [PubMed]
- Gross, E.; Sevier, C.S.; Heldman, N.; Vitu, E.; Bentzur, M.; Kaiser, C.A.; Thorpe, C.; Fass, D. Generating disulfides enzymatically: Reaction products and electron acceptors of the endoplasmic reticulum thiol oxidase Ero1p. Proc. Natl. Acad. Sci. USA 2006, 103, 299–304. [Google Scholar] [CrossRef] [PubMed]
- Tu, B.P.; Weissman, J.S. The FAD- and O2-dependent reaction cycle of Ero1-mediated oxidative protein folding in the endoplasmic reticulum. Mol. Cell 2002, 10, 983–994. [Google Scholar] [CrossRef]
- Tu, B.P.; Ho-Schleyer, S.C.; Travers, K.J.; Weissman, J.S. Biochemical basis of oxidative protein folding in the endoplasmic reticulum. Science 2000, 290, 1571–1574. [Google Scholar] [CrossRef] [PubMed]
- Sevier, C.S.; Kaiser, C.A. Disulfide transfer between two conserved cysteine pairs imparts selectivity to protein oxidation by Ero1. Mol. Biol. Cell 2006, 17, 2256–2266. [Google Scholar] [CrossRef] [PubMed]
- Frand, A.R.; Kaiser, C.A. Ero1p oxidizes protein disulfide isomerase in a pathway for disulfide bond formation in the endoplasmic reticulum. Mol. Cell 1999, 4, 469–477. [Google Scholar] [CrossRef]
- Sevier, C.S.; Qu, H.; Heldman, N.; Gross, E.; Fass, D.; Kaiser, C.A. Modulation of cellular disulfide-bond formation and the ER redox environment by feedback regulation of Ero1. Cell 2007, 129, 333–344. [Google Scholar] [CrossRef] [PubMed]
- Ramming, T.; Okumura, M.; Kanemura, S.; Baday, S.; Birk, J.; Moes, S.; Spiess, M.; Jeno, P.; Berneche, S.; Inaba, K.; et al. A PDI-catalyzed thiol-disulfide switch regulates the production of hydrogen peroxide by human Ero1. Free Radic. Biol. Med. 2015, 83, 361–372. [Google Scholar] [CrossRef] [PubMed]
- Bertoli, G.; Simmen, T.; Anelli, T.; Molteni, S.N.; Fesce, R.; Sitia, R. Two conserved cysteine triads in human Ero1α cooperate for efficient disulfide bond formation in the endoplasmic reticulum. J. Biol. Chem. 2004, 279, 30047–30052. [Google Scholar] [CrossRef] [PubMed]
- Hansen, H.G.; Soltoft, C.L.; Schmidt, J.D.; Birk, J.; Appenzeller-Herzog, C.; Ellgaard, L. Biochemical evidence that regulation of Ero1β activity in human cells does not involve the isoform-specific cysteine 262. Biosci. Rep. 2014, 34, e00103. [Google Scholar] [CrossRef] [PubMed]
- Shepherd, C.; Oka, O.B.; Bulleid, N.J. Inactivation of mammalian Ero1α is catalysed by specific protein disulfide-isomerases. Biochem. J. 2014, 461, 107–113. [Google Scholar] [CrossRef] [PubMed]
- Araki, K.; Nagata, K. Functional in vitro analysis of the ERO1 protein and protein-disulfide isomerase pathway. J. Biol. Chem. 2011, 286, 32705–32712. [Google Scholar] [CrossRef] [PubMed]
- Mezghrani, A.; Fassio, A.; Benham, A.; Simmen, T.; Braakman, I.; Sitia, R. Manipulation of oxidative protein folding and PDI redox state in mammalian cells. EMBO J. 2001, 20, 6288–6296. [Google Scholar] [CrossRef] [PubMed]
- Appenzeller-Herzog, C.; Riemer, J.; Zito, E.; Chin, K.T.; Ron, D.; Spiess, M.; Ellgaard, L. Disulphide production by Ero1α-PDI relay is rapid and effectively regulated. EMBO J. 2010, 29, 3318–3329. [Google Scholar] [CrossRef] [PubMed]
- Araki, K.; Iemura, S.; Kamiya, Y.; Ron, D.; Kato, K.; Natsume, T.; Nagata, K. Ero1-α and PDIs constitute a hierarchical electron transfer network of endoplasmic reticulum oxidoreductases. J. Cell Biol. 2013, 202, 861–874. [Google Scholar] [CrossRef] [PubMed]
- Anelli, T.; Alessio, M.; Bachi, A.; Bergamelli, L.; Bertoli, G.; Camerini, S.; Mezghrani, A.; Ruffato, E.; Simmen, T.; Sitia, R. Thiol-mediated protein retention in the endoplasmic reticulum: The role of ERp44. EMBO J. 2003, 22, 5015–5022. [Google Scholar] [CrossRef] [PubMed]
- Kakihana, T.; Araki, K.; Vavassori, S.; Iemura, S.; Cortini, M.; Fagioli, C.; Natsume, T.; Sitia, R.; Nagata, K. Dynamic regulation of Ero1α and peroxiredoxin 4 localization in the secretory pathway. J. Biol. Chem. 2013, 288, 29586–29594. [Google Scholar] [CrossRef] [PubMed]
- Cuozzo, J.W.; Kaiser, C.A. Competition between glutathione and protein thiols for disulphide-bond formation. Nat. Cell Biol. 1999, 1, 130–135. [Google Scholar] [CrossRef] [PubMed]
- Molteni, S.N.; Fassio, A.; Ciriolo, M.R.; Filomeni, G.; Pasqualetto, E.; Fagioli, C.; Sitia, R. Glutathione limits Ero1-dependent oxidation in the endoplasmic reticulum. J. Biol. Chem. 2004, 279, 32667–32673. [Google Scholar] [CrossRef] [PubMed]
- Baker, K.M.; Chakravarthi, S.; Langton, K.P.; Sheppard, A.M.; Lu, H.; Bulleid, N.J. Low reduction potential of Ero1α regulatory disulphides ensures tight control of substrate oxidation. EMBO J. 2008, 27, 2988–2997. [Google Scholar] [CrossRef] [PubMed]
- Ponsero, A.J.; Igbaria, A.; Darch, M.A.; Miled, S.; Outten, C.E.; Winther, J.R.; Palais, G.; D’Autreaux, B.; Delaunay-Moisan, A.; Toledano, M.B. Endoplasmic Reticulum Transport of Glutathione by Sec61 Is Regulated by Ero1 and Bip. Mol. Cell 2017, 67, 67. [Google Scholar] [CrossRef] [PubMed]
- Birk, J.; Meyer, M.; Aller, I.; Hansen, H.G.; Odermatt, A.; Dick, T.P.; Meyer, A.J.; Appenzeller-Herzog, C. Endoplasmic reticulum: Reduced and oxidized glutathione revisited. J. Cell Sci. 2013, 126, 1604–1617. [Google Scholar] [CrossRef] [PubMed]
- Chakravarthi, S.; Bulleid, N.J. Glutathione is required to regulate the formation of native disulfide bonds within proteins entering the secretory pathway. J. Biol. Chem. 2004, 279, 39872–39879. [Google Scholar] [CrossRef] [PubMed]
- Gess, B.; Hofbauer, K.H.; Wenger, R.H.; Lohaus, C.; Meyer, H.E.; Kurtz, A. The cellular oxygen tension regulates expression of the endoplasmic oxidoreductase ERO1-Lα. Eur. J. Biochem. 2003, 270, 2228–2235. [Google Scholar] [CrossRef] [PubMed]
- Marciniak, S.J.; Yun, C.Y.; Oyadomari, S.; Novoa, I.; Zhang, Y.; Jungreis, R.; Nagata, K.; Harding, H.P.; Ron, D. CHOP induces death by promoting protein synthesis and oxidation in the stressed endoplasmic reticulum. Genes Dev. 2004, 18, 3066–3077. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Mongillo, M.; Chin, K.T.; Harding, H.; Ron, D.; Marks, A.R.; Tabas, I. Role of ERO1-α-mediated stimulation of inositol 1,4,5-triphosphate receptor activity in endoplasmic reticulum stress-induced apoptosis. J. Cell Biol. 2009, 186, 783–792. [Google Scholar] [CrossRef] [PubMed]
- May, D.; Itin, A.; Gal, O.; Kalinski, H.; Feinstein, E.; Keshet, E. Ero1-Lα plays a key role in a HIF-1-mediated pathway to improve disulfide bond formation and VEGF secretion under hypoxia: Implication for cancer. Oncogene 2005, 24, 1011–1020. [Google Scholar] [CrossRef] [PubMed]
- Takei, N.; Yoneda, A.; Sakai-Sawada, K.; Kosaka, M.; Minomi, K.; Tamura, Y. Hypoxia-inducible ERO1α promotes cancer progression through modulation of integrin-β1 modification and signalling in HCT116 colorectal cancer cells. Sci. Rep. 2017, 7, 9389. [Google Scholar] [CrossRef] [PubMed]
- Townsend, D.M.; Manevich, Y.; He, L.; Xiong, Y.; Bowers, R.R., Jr.; Hutchens, S.; Tew, K.D. Nitrosative stress-induced S-glutathionylation of protein disulfide isomerase leads to activation of the unfolded protein response. Cancer Res. 2009, 69, 7626–7634. [Google Scholar] [CrossRef] [PubMed]
- Tavender, T.J.; Bulleid, N.J. Peroxiredoxin IV protects cells from oxidative stress by removing H2O2 produced during disulphide formation. J. Cell Sci. 2010, 123, 2672–2679. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Zhang, L.; Niu, Y.; Sitia, R.; Wang, C.C. Glutathione peroxidase 7 utilizes hydrogen peroxide generated by Ero1α to promote oxidative protein folding. Antioxid. Redox Signal. 2014, 20, 545–556. [Google Scholar] [CrossRef] [PubMed]
- Ramming, T.; Hansen, H.G.; Nagata, K.; Ellgaard, L.; Appenzeller-Herzog, C. GPx8 peroxidase prevents leakage of H2O2 from the endoplasmic reticulum. Free Radic. Biol. Med. 2014, 70, 106–116. [Google Scholar] [CrossRef] [PubMed]
- Enyedi, B.; Varnai, P.; Geiszt, M. Redox state of the endoplasmic reticulum is controlled by Ero1L-alpha and intraluminal calcium. Antioxid. Redox Signal. 2010, 13, 721–729. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, V.D.; Saaranen, M.J.; Karala, A.R.; Lappi, A.K.; Wang, L.; Raykhel, I.B.; Alanen, H.I.; Salo, K.E.; Wang, C.C.; Ruddock, L.W. Two endoplasmic reticulum PDI peroxidases increase the efficiency of the use of peroxide during disulfide bond formation. J. Mol. Biol. 2011, 406, 503–515. [Google Scholar] [CrossRef] [PubMed]
- Tavender, T.J.; Springate, J.J.; Bulleid, N.J. Recycling of peroxiredoxin IV provides a novel pathway for disulphide formation in the endoplasmic reticulum. EMBO J. 2010, 29, 4185–4197. [Google Scholar] [CrossRef] [PubMed]
- Konno, T.; Pinho Melo, E.; Lopes, C.; Mehmeti, I.; Lenzen, S.; Ron, D.; Avezov, E. ERO1-independent production of H2O2 within the endoplasmic reticulum fuels Prdx4-mediated oxidative protein folding. J. Cell Biol. 2015, 211, 253–259. [Google Scholar] [CrossRef] [PubMed]
- Zito, E.; Melo, E.P.; Yang, Y.; Wahlander, A.; Neubert, T.A.; Ron, D. Oxidative protein folding by an endoplasmic reticulum-localized peroxiredoxin. Mol. Cell 2010, 40, 787–797. [Google Scholar] [CrossRef] [PubMed]
- Bagur, R.; Hajnoczky, G. Intracellular Ca2+ Sensing: Its Role in Calcium Homeostasis and Signaling. Mol. Cell 2017, 66, 780–788. [Google Scholar] [CrossRef] [PubMed]
- Baba-Aissa, F.; Raeymaekers, L.; Wuytack, F.; Dode, L.; Casteels, R. Distribution and isoform diversity of the organellar Ca2+ pumps in the brain. Mol. Chem. Neuropathol. 1998, 33, 199–208. [Google Scholar] [CrossRef] [PubMed]
- Adachi, T.; Weisbrod, R.M.; Pimentel, D.R.; Ying, J.; Sharov, V.S.; Schoneich, C.; Cohen, R.A. S-Glutathiolation by peroxynitrite activates SERCA during arterial relaxation by nitric oxide. Nat. Med. 2004, 10, 1200–1207. [Google Scholar] [CrossRef] [PubMed]
- Mei, Y.; Thompson, M.D.; Shiraishi, Y.; Cohen, R.A.; Tong, X. Sarcoplasmic/endoplasmic reticulum Ca2+ ATPase C674 promotes ischemia- and hypoxia-induced angiogenesis via coordinated endothelial cell and macrophage function. J. Mol. Cell. Cardiol. 2014, 76, 275–282. [Google Scholar] [CrossRef] [PubMed]
- Liang, C.P.; Han, S.; Li, G.; Tabas, I.; Tall, A.R. Impaired MEK signaling and SERCA expression promote ER stress and apoptosis in insulin-resistant macrophages and are reversed by exenatide treatment. Diabetes 2012, 61, 2609–2620. [Google Scholar] [CrossRef] [PubMed]
- Marino, M.; Stoilova, T.; Giorgi, C.; Bachi, A.; Cattaneo, A.; Auricchio, A.; Pinton, P.; Zito, E. SEPN1, an endoplasmic reticulum-localized selenoprotein linked to skeletal muscle pathology, counteracts hyperoxidation by means of redox-regulating SERCA2 pump activity. Hum. Mol. Genet. 2015, 24, 1843–1855. [Google Scholar] [CrossRef] [PubMed]
- Szabadkai, G.; Bianchi, K.; Varnai, P.; De Stefani, D.; Wieckowski, M.R.; Cavagna, D.; Nagy, A.I.; Balla, T.; Rizzuto, R. Chaperone-mediated coupling of endoplasmic reticulum and mitochondrial Ca2+ channels. J. Cell Biol. 2006, 175, 901–911. [Google Scholar] [CrossRef] [PubMed]
- Shoshan-Barmatz, V.; Ben-Hail, D.; Admoni, L.; Krelin, Y.; Tripathi, S.S. The mitochondrial voltage-dependent anion channel 1 in tumor cells. Biochim. Biophys. Acta 2015, 1848, 2547–2575. [Google Scholar] [CrossRef] [PubMed]
- Csordas, G.; Golenar, T.; Seifert, E.L.; Kamer, K.J.; Sancak, Y.; Perocchi, F.; Moffat, C.; Weaver, D.; de la Fuente Perez, S.; Bogorad, R.; et al. MICU1 controls both the threshold and cooperative activation of the mitochondrial Ca2+ uniporter. Cell Metab. 2013, 17, 976–987. [Google Scholar] [CrossRef] [PubMed]
- Csordas, G.; Varnai, P.; Golenar, T.; Roy, S.; Purkins, G.; Schneider, T.G.; Balla, T.; Hajnoczky, G. Imaging interorganelle contacts and local calcium dynamics at the ER-mitochondrial interface. Mol. Cell 2010, 39, 121–132. [Google Scholar] [CrossRef] [PubMed]
- Gilady, S.Y.; Bui, M.; Lynes, E.M.; Benson, M.D.; Watts, R.; Vance, J.E.; Simmen, T. Ero1α requires oxidizing and normoxic conditions to localize to the mitochondria-associated membrane (MAM). Cell Stress Chaperones 2010, 15, 619–629. [Google Scholar] [CrossRef] [PubMed]
- Anelli, T.; Bergamelli, L.; Margittai, E.; Rimessi, A.; Fagioli, C.; Malgaroli, A.; Pinton, P.; Ripamonti, M.; Rizzuto, R.; Sitia, R. Ero1α regulates Ca2+ fluxes at the endoplasmic reticulum-mitochondria interface (MAM). Antioxid. Redox Signal. 2012, 16, 1077–1087. [Google Scholar] [CrossRef] [PubMed]
- Higo, T.; Hattori, M.; Nakamura, T.; Natsume, T.; Michikawa, T.; Mikoshiba, K. Subtype-specific and ER lumenal environment-dependent regulation of inositol 1,4,5-trisphosphate receptor type 1 by ERp44. Cell 2005, 120, 85–98. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Camacho, P. Ca2+-dependent redox modulation of SERCA 2b by ERp57. J. Cell Biol. 2004, 164, 35–46. [Google Scholar] [CrossRef] [PubMed]
- Kutomi, G.; Tamura, Y.; Tanaka, T.; Kajiwara, T.; Kukita, K.; Ohmura, T.; Shima, H.; Takamaru, T.; Satomi, F.; Suzuki, Y.; et al. Human endoplasmic reticulum oxidoreductin 1-α is a novel predictor for poor prognosis of breast cancer. Cancer Sci. 2013, 104, 1091–1096. [Google Scholar] [CrossRef] [PubMed]
- Zhou, B.; Wang, G.; Gao, S.; Chen, Y.; Jin, C.; Wang, Z.; Yang, Y.; Ma, Z.; Zhang, W.; Feng, X. Expression of ERO1L in gastric cancer and its association with patient prognosis. Exp. Ther. Med. 2017, 14, 2298–2302. [Google Scholar] [CrossRef] [PubMed]
- Battle, D.M.; Gunasekara, S.D.; Watson, G.R.; Ahmed, E.M.; Saysell, C.G.; Altaf, N.; Sanusi, A.L.; Munipalle, P.C.; Scoones, D.; Walker, J.; et al. Expression of the endoplasmic reticulum oxidoreductase Ero1α in gastro-intestinal cancer reveals a link between homocysteine and oxidative protein folding. Antioxid. Redox Signal. 2013, 19, 24–35. [Google Scholar] [CrossRef] [PubMed]
- Kukita, K.; Tamura, Y.; Tanaka, T.; Kajiwara, T.; Kutomi, G.; Saito, K.; Okuya, K.; Takaya, A.; Kanaseki, T.; Tsukahara, T.; et al. Cancer-Associated Oxidase ERO1-α Regulates the Expression of MHC Class I Molecule via Oxidative Folding. J. Immunol. 2015, 194, 4988–4996. [Google Scholar] [CrossRef] [PubMed]
- Seol, S.Y.; Kim, C.; Lim, J.Y.; Yoon, S.O.; Hong, S.W.; Kim, J.W.; Choi, S.H.; Cho, J.Y. Overexpression of Endoplasmic Reticulum Oxidoreductin 1-α (ERO1L) Is Associated with Poor Prognosis of Gastric Cancer. Cancer Res. Treat. 2016, 48, 1196–1209. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, T.; Kutomi, G.; Kajiwara, T.; Kukita, K.; Kochin, V.; Kanaseki, T.; Tsukahara, T.; Hirohashi, Y.; Torigoe, T.; Okamoto, Y.; et al. Cancer-associated oxidoreductase ERO1-α drives the production of VEGF via oxidative protein folding and regulating the mRNA level. Br. J. Cancer 2016, 114, 1227–1234. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, T.; Kutomi, G.; Kajiwara, T.; Kukita, K.; Kochin, V.; Kanaseki, T.; Tsukahara, T.; Hirohashi, Y.; Torigoe, T.; Okamoto, Y.; et al. Cancer-associated oxidoreductase ERO1-α promotes immune escape through up-regulation of PD-L1 in human breast cancer. Oncotarget 2017, 8, 24706–24718. [Google Scholar] [CrossRef] [PubMed]
- Smyth, M.J.; Ngiow, S.F.; Ribas, A.; Teng, M.W. Combination cancer immunotherapies tailored to the tumour microenvironment. Nat. Rev. Clin. Oncol. 2016, 13, 143–158. [Google Scholar] [CrossRef] [PubMed]
- Black, M.; Barsoum, I.B.; Truesdell, P.; Cotechini, T.; Macdonald-Goodfellow, S.K.; Petroff, M.; Siemens, D.R.; Koti, M.; Craig, A.W.; Graham, C.H. Activation of the PD-1/PD-L1 immune checkpoint confers tumor cell chemoresistance associated with increased metastasis. Oncotarget 2016, 7, 10557–10567. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, T.; Kajiwara, T.; Torigoe, T.; Okamoto, Y.; Sato, N.; Tamura, Y. Cancer-associated oxidoreductase ERO1-α drives the production of tumor-promoting myeloid-derived suppressor cells via oxidative protein folding. J. Immunol. 2015, 194, 2004–2010. [Google Scholar] [CrossRef] [PubMed]
- Talmadge, J.E.; Gabrilovich, D.I. History of myeloid-derived suppressor cells. Nat. Rev. Cancer 2013, 13, 739–752. [Google Scholar] [CrossRef] [PubMed]
- Leone, P.; Shin, E.C.; Perosa, F.; Vacca, A.; Dammacco, F.; Racanelli, V. MHC class I antigen processing and presenting machinery: Organization, function, and defects in tumor cells. J. Natl. Cancer Inst. 2013, 105, 1172–1187. [Google Scholar] [CrossRef] [PubMed]
- Kajiwara, T.; Tanaka, T.; Kukita, K.; Kutomi, G.; Saito, K.; Okuya, K.; Takaya, A.; Kochin, V.; Kanaseki, T.; Tsukahara, T.; et al. Hypoxia augments MHC class I antigen presentation via facilitation of ERO1-α-mediated oxidative folding in murine tumor cells. Eur. J. Immunol. 2016, 46, 2842–2851. [Google Scholar] [CrossRef] [PubMed]
- Ching, R.H.H.; Sze, K.M.F.; Lau, E.Y.T.; Chiu, Y.T.; Lee, J.M.F.; Ng, I.O.L.; Lee, T.K.W. C-terminal truncated hepatitis B virus X protein regulates tumorigenicity, self-renewal and drug resistance via STAT3/Nanog signaling pathway. Oncotarget 2017, 8, 23507–23516. [Google Scholar] [CrossRef] [PubMed]
- Gong, G.; Waris, G.; Tanveer, R.; Siddiqui, A. Human hepatitis C virus NS5A protein alters intracellular calcium levels, induces oxidative stress, and activates STAT-3 and NF-κB. Proc. Natl. Acad. Sci. USA 2001, 98, 9599–9604. [Google Scholar] [CrossRef] [PubMed]
- Ivanov, A.V.; Smirnova, O.A.; Ivanova, O.N.; Masalova, O.V.; Kochetkov, S.N.; Isaguliants, M.G. Hepatitis C virus proteins activate NRF2/ARE pathway by distinct ROS-dependent and independent mechanisms in HUH7 cells. PLoS ONE 2011, 6, e24957. [Google Scholar] [CrossRef] [PubMed]
- Okuda, M.; Li, K.; Beard, M.R.; Showalter, L.A.; Scholle, F.; Lemon, S.M.; Weinman, S.A. Mitochondrial injury, oxidative stress, and antioxidant gene expression are induced by hepatitis C virus core protein. Gastroenterology 2002, 122, 366–375. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Boehning, D.F.; Qian, T.; Popov, V.L.; Weinman, S.A. Hepatitis C virus core protein increases mitochondrial ROS production by stimulation of Ca2+ uniporter activity. FASEB J. 2007, 21, 2474–2485. [Google Scholar] [CrossRef] [PubMed]
- Kang, S.M.; Kim, S.J.; Kim, J.H.; Lee, W.; Kim, G.W.; Lee, K.H.; Choi, K.Y.; Oh, J.W. Interaction of hepatitis C virus core protein with Hsp60 triggers the production of reactive oxygen species and enhances TNF-α-mediated apoptosis. Cancer Lett. 2009, 279, 230–237. [Google Scholar] [CrossRef] [PubMed]
- Benali-Furet, N.L.; Chami, M.; Houel, L.; De Giorgi, F.; Vernejoul, F.; Lagorce, D.; Buscail, L.; Bartenschlager, R.; Ichas, F.; Rizzuto, R.; et al. Hepatitis C virus core triggers apoptosis in liver cells by inducing ER stress and ER calcium depletion. Oncogene 2005, 24, 4921–4933. [Google Scholar] [CrossRef] [PubMed]
- Ivanov, A.V.; Smirnova, O.A.; Petrushanko, I.Y.; Ivanova, O.N.; Karpenko, I.L.; Alekseeva, E.; Sominskaya, I.; Makarov, A.A.; Bartosch, B.; Kochetkov, S.N.; et al. HCV core protein uses multiple mechanisms to induce oxidative stress in human hepatoma Huh7 cells. Viruses 2015, 7, 2745–2770. [Google Scholar] [CrossRef] [PubMed]
- Smirnova, O.A.; Ivanova, O.N.; Bartosch, B.; Valuev-Elliston, V.T.; Mukhtarov, F.; Kochetkov, S.N.; Ivanov, A.V. Hepatitis C Virus NS5A Protein Triggers Oxidative Stress by Inducing NADPH Oxidases 1 and 4 and Cytochrome P450 2E1. Oxid. Med. Cell. Longev. 2016, 2016, 8341937. [Google Scholar] [CrossRef] [PubMed]
- Joubert, P.E.; Werneke, S.W.; de la Calle, C.; Guivel-Benhassine, F.; Giodini, A.; Peduto, L.; Levine, B.; Schwartz, O.; Lenschow, D.J.; Albert, M.L. Chikungunya virus-induced autophagy delays caspase-dependent cell death. J. Exp. Med. 2012, 209, 1029–1047. [Google Scholar] [CrossRef] [PubMed]
- Langsjoen, R.M.; Auguste, A.J.; Rossi, S.L.; Roundy, C.M.; Penate, H.N.; Kastis, M.; Schnizlein, M.K.; Le, K.C.; Haller, S.L.; Chen, R.; et al. Host oxidative folding pathways offer novel anti-chikungunya virus drug targets with broad spectrum potential. Antivir. Res. 2017, 143, 246–251. [Google Scholar] [CrossRef] [PubMed]
- Nishikiori, M.; Ahlquist, P. Organelle luminal dependence of (+)strand RNA virus replication reveals a hidden druggable target. Sci. Adv. 2018, 4, eaap8258. [Google Scholar] [CrossRef] [PubMed]
- Gullberg, R.C.; Jordan Steel, J.; Moon, S.L.; Soltani, E.; Geiss, B.J. Oxidative stress influences positive strand RNA virus genome synthesis and capping. Virology 2015, 475, 219–229. [Google Scholar] [CrossRef] [PubMed]
- Hyodo, K.; Hashimoto, K.; Kuchitsu, K.; Suzuki, N.; Okuno, T. Harnessing host ROS-generating machinery for the robust genome replication of a plant RNA virus. Proc. Natl. Acad. Sci. USA 2017, 114, E1282–E1290. [Google Scholar] [CrossRef] [PubMed]
- Parkkinen, J.J.; Lammi, M.J.; Agren, U.; Tammi, M.; Keinanen, T.A.; Hyvonen, T.; Eloranta, T.O. Polyamine-dependent alterations in the structure of microfilaments, Golgi apparatus, endoplasmic reticulum, and proteoglycan synthesis in BHK cells. J. Cell. Biochem. 1997, 66, 165–174. [Google Scholar] [CrossRef]
- Prunotto, M.; Compagnone, A.; Bruschi, M.; Candiano, G.; Colombatto, S.; Bandino, A.; Petretto, A.; Moll, S.; Bochaton-Piallat, M.L.; Gabbiani, G.; et al. Endocellular polyamine availability modulates epithelial-to-mesenchymal transition and unfolded protein response in MDCK cells. Lab. Investig. 2010, 90, 929–939. [Google Scholar] [CrossRef] [PubMed]
- Landau, G.; Ran, A.; Bercovich, Z.; Feldmesser, E.; Horn-Saban, S.; Korkotian, E.; Jacob-Hirsh, J.; Rechavi, G.; Ron, D.; Kahana, C. Expression profiling and biochemical analysis suggest stress response as a potential mechanism inhibiting proliferation of polyamine-depleted cells. J. Biol. Chem. 2012, 287, 35825–35837. [Google Scholar] [CrossRef] [PubMed]
- Hyvonen, M.T.; Merentie, M.; Uimari, A.; Keinanen, T.A.; Janne, J.; Alhonen, L. Mechanisms of polyamine catabolism-induced acute pancreatitis. Biochem. Soc. Trans. 2007, 35, 326–330. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Mandal, S.; Mandal, A.; Park, M.H. Depletion of the polyamines spermidine and spermine by overexpression of spermidine/spermine N1-acetyltransferase 1 (SAT1) leads to mitochondria-mediated apoptosis in mammalian cells. Biochem. J. 2015, 468, 435–447. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.B.; Park, J.H.; Woster, P.M.; Casero, R.A., Jr.; Park, M.H. Suppression of exogenous gene expression by spermidine/spermine N1-acetyltransferase 1 (SSAT1) cotransfection. J. Biol. Chem. 2010, 285, 15548–15556. [Google Scholar] [CrossRef] [PubMed]
- Mandal, S.; Mandal, A.; Johansson, H.E.; Orjalo, A.V.; Park, M.H. Depletion of cellular polyamines, spermidine and spermine, causes a total arrest in translation and growth in mammalian cells. Proc. Natl. Acad. Sci. USA 2013, 110, 2169–2174. [Google Scholar] [CrossRef] [PubMed]
- Zahedi, K.; Barone, S.; Destefano-Shields, C.; Brooks, M.; Murray-Stewart, T.; Dunworth, M.; Li, W.; Doherty, J.R.; Hall, M.A.; Smith, R.D.; et al. Activation of endoplasmic reticulum stress response by enhanced polyamine catabolism is important in the mediation of cisplatin-induced acute kidney injury. PLoS ONE 2017, 12, e0184570. [Google Scholar] [CrossRef] [PubMed]
- Rossi, D.; Galvao, F.C.; Bellato, H.M.; Boldrin, P.E.; Andrews, B.J.; Valentini, S.R.; Zanelli, C.F. eIF5A has a function in the cotranslational translocation of proteins into the ER. Amino Acids 2014, 46, 645–653. [Google Scholar] [CrossRef] [PubMed]
- Mandal, A.; Mandal, S.; Park, M.H. Global quantitative proteomics reveal up-regulation of endoplasmic reticulum stress response proteins upon depletion of eIF5A in HeLa cells. Sci. Rep. 2016, 6, 25795. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, W.C.; Hsu, P.C.; Liao, Y.F.; Young, S.T.; Wang, Z.W.; Lin, C.L.; Tsay, G.J.; Lee, H.; Hung, H.C.; Liu, G.Y. Overexpression of ornithine decarboxylase suppresses thapsigargin-induced apoptosis. Mol. Cells 2010, 30, 311–318. [Google Scholar] [CrossRef] [PubMed]
- Robbins, R.D.; Tersey, S.A.; Ogihara, T.; Gupta, D.; Farb, T.B.; Ficorilli, J.; Bokvist, K.; Maier, B.; Mirmira, R.G. Inhibition of deoxyhypusine synthase enhances islet β cell function and survival in the setting of endoplasmic reticulum stress and type 2 diabetes. J. Biol. Chem. 2010, 285, 39943–39952. [Google Scholar] [CrossRef] [PubMed]
- Korenaga, M.; Wang, T.; Li, Y.; Showalter, L.A.; Chan, T.; Sun, J.; Weinman, S.A. Hepatitis C virus core protein inhibits mitochondrial electron transport and increases reactive oxygen species (ROS) production. J. Biol. Chem. 2005, 280, 37481–37488. [Google Scholar] [CrossRef] [PubMed]
- Mitsuya, Y.; Takahashi, Y.; Berberich, T.; Miyazaki, A.; Matsumura, H.; Takahashi, H.; Terauchi, R.; Kusano, T. Spermine signaling plays a significant role in the defense response of Arabidopsis thaliana to cucumber mosaic virus. J. Plant Physiol. 2009, 166, 626–643. [Google Scholar] [CrossRef] [PubMed]
- Sagor, G.H.; Chawla, P.; Kim, D.W.; Berberich, T.; Kojima, S.; Niitsu, M.; Kusano, T. The polyamine spermine induces the unfolded protein response via the MAPK cascade in Arabidopsis. Front. Plant Sci. 2015, 6, 687. [Google Scholar] [CrossRef] [PubMed]
- Iwata, Y.; Koizumi, N. An Arabidopsis transcription factor, AtbZIP60, regulates the endoplasmic reticulum stress response in a manner unique to plants. Proc. Natl. Acad. Sci. USA 2005, 102, 5280–5285. [Google Scholar] [CrossRef] [PubMed]
- Lu, D.P.; Christopher, D.A. Endoplasmic reticulum stress activates the expression of a sub-group of protein disulfide isomerase genes and AtbZIP60 modulates the response in Arabidopsis thaliana. Mol. Genet. Genom. 2008, 280, 199–210. [Google Scholar] [CrossRef] [PubMed]
- Morada, M.; Pendyala, L.; Wu, G.; Merali, S.; Yarlett, N. Cryptosporidium parvum induces an endoplasmic stress response in the intestinal adenocarcinoma HCT-8 cell line. J. Biol. Chem. 2013, 288, 30356–30364. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).