Nanoparticle Exposure and Hormetic Dose–Responses: An Update
Abstract
1. Introduction
2. Results
2.1. In Vitro Studies
2.1.1. Lung Cells
2.1.2. Gastrointestinal and Hepatic Cells
2.1.3. Dermal Cells
2.2. In Vivo Studies
2.2.1. Bacteria Strains
2.2.2. Environmental Microbial Communities
2.2.3. Algae
2.2.4. Plants
2.2.5. Nematodes and Larvae
2.2.6. Superior Aquatic Organisms
2.2.7. Rats
3. Discussion
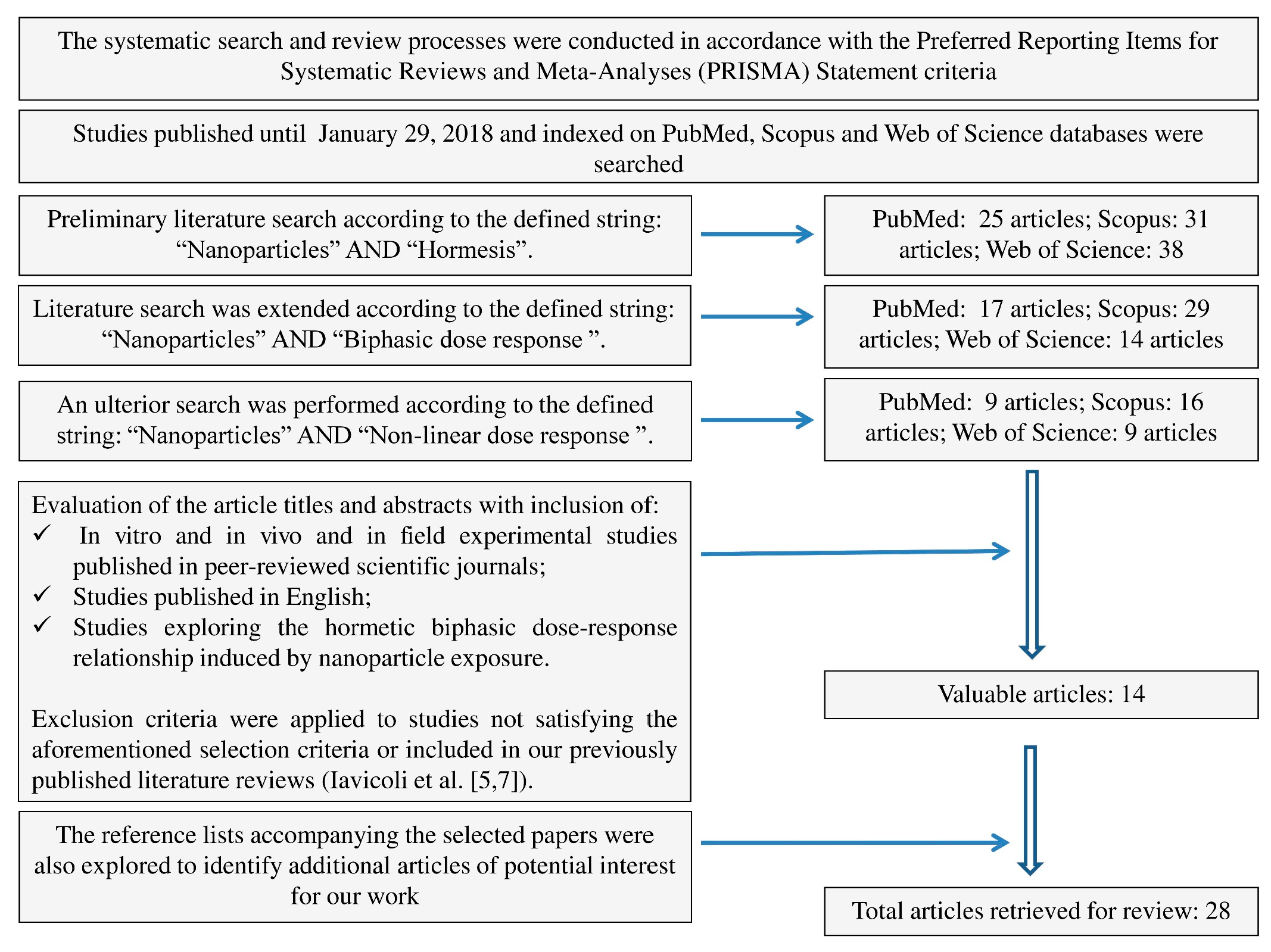
4. Materials and Methods
- -
- In vitro and in vivo and in field experimental studies published in peer-reviewed scientific journals;
- -
- Published in English; and
- -
- Exploring the hormetic biphasic dose–response relationship induced by nanoparticle exposure.
5. Conclusions
Acknowledgments
Conflicts of Interest
Abbreviations
| A549 | Human lung adenocarcinoma cells |
| Ag-NPs | Silver Nanoparticles |
| AgNO3 | Silver nitrate |
| Caco2 | Human epithelial colorectal adenocarcinoma cells |
| CeO2-NPs | Cerium oxide nanoparticles |
| Cu-NPs | Copper Nanoparticles |
| ENM | Engineered Nanomaterial |
| Fe | Iron |
| FSF1 | Normal diploid human facial skin fibroblasts |
| HepG2 | Human hepatoma derived cell line |
| GM-CSF | Granulocyte macrophage- colony stimulating factor |
| K | Potassium |
| IL | Interleukin |
| INF-γ | Interferon-γ |
| MIC | Minimum Inhibitory Concentrations |
| Mn | Manganese |
| MW-CNTs | Multi-Walled Carbon Nanotubes |
| N | Nitrogen |
| N2O | Nitrous oxide |
| NDs | Nano Diamonds |
| NPs | Nanoparticles |
| n.s. | Not significant |
| P | Phosphorous |
| Pd-NPs | Palladium Nanoparticles |
| PEG | Polyethylene-Glycol |
| PVP | Polyvinylpyrrolidone |
| QD | Quantum Dot |
| RR | Respiration Rate |
| SiO2-NPs | Silica Nanoparticles |
| SVL | Snout Vent Length |
| TNF-α | Tumor necrosis factor-α |
| ZnO-NPs | Zinc Oxide Nanoparticles |
References
- Leso, V.; Fontana, L.; Mauriello, M.C.; Iavicoli, I. Occupational Risk Assessment of Engineered Nanomaterials: Limits, Challenges and Opportunities. Curr. Nanosci. 2017, 13, 55–78. [Google Scholar] [CrossRef]
- Gottschalk, F.; Nowack, B. The release of engineered nanomaterials to the environment. J. Environ. Monit. 2011, 13, 1145–1155. [Google Scholar] [CrossRef] [PubMed]
- Gottschalk, F.; Sun, T.; Nowack, B. Environmental concentrations of engineered nanomaterials: Review of modeling and analytical studies. Environ. Pollut. 2013, 181, 287–300. [Google Scholar] [CrossRef] [PubMed]
- Calabrese, E.J. Hormesis: Why it is important to toxicology and toxicologists. Environ. Toxicol. Chem. 2008, 27, 1451–1474. [Google Scholar] [CrossRef] [PubMed]
- Iavicoli, I.; Calabrese, E.J.; Nascarella, M.A. Exposure to nanoparticles and hormesis. Dose Response 2010, 8, 501–517. [Google Scholar] [CrossRef] [PubMed]
- Iavicoli, I.; Fontana, L.; Leso, V.; Bergamaschi, A. The effects of nanomaterials as endocrine disruptors. Int. J. Mol. Sci. 2013, 14, 16732–16801. [Google Scholar] [CrossRef] [PubMed]
- Iavicoli, I.; Fontana, L.; Leso, V.; Calabrese, E.J. Hormetic dose-responses in nanotechnology studies. Sci. Total Environ. 2014, 487, 361–374. [Google Scholar] [CrossRef] [PubMed]
- Nascarella, M.A.; Calabrese, E.J. A method to evaluate hormesis in nanoparticle dose–responses. Dose Response 2012, 10, 344–354. [Google Scholar] [CrossRef] [PubMed]
- Quadros, M.E.; Marr, L.C. Silver nanoparticles and total aerosols emitted by nanotechnology-related consumer spray products. Environ. Sci. Technol. 2011, 45, 10713–10719. [Google Scholar] [CrossRef]
- Fabrega, J.; Zhang, R.; Renshaw, J.C.; Liu, W.-T.; Lead, J.R. Impact of silver nanoparticles on natural marine biofilm bacteria. Chemosphere 2011, 85, 961–966. [Google Scholar] [CrossRef] [PubMed]
- Faunce, T.; Watal, A. Nanosilver and global public health: International regulatory issues. Nanomedicine 2010, 5, 617–632. [Google Scholar] [CrossRef] [PubMed]
- Sthijns, M.M.; Thongkam, W.; Albrecht, C.; Hellack, B.; Bast, A.; Haenen, G.R.; Schins, R.P. Silver nanoparticles induce hormesis in A549 human epithelial cells. Toxicol. In Vitro 2017, 40, 223–233. [Google Scholar] [CrossRef] [PubMed]
- Jiao, Z.H.; Li, M.; Feng, Y.X.; Shi, J.C.; Zhang, J.; Shao, B. Hormesis effects of silver nanoparticles at non-cytotoxic doses to human hepatoma cells. PLoS ONE 2014, 9, e102564. [Google Scholar] [CrossRef] [PubMed]
- Kang, T.; Guan, R.; Song, Y.; Lyu, F.; Ye, X.; Jiang, H. Cytotoxicity of zinc oxide nanoparticles and silver nanoparticles in human epithelial colorectal adenocarcinoma cells. LWT Food Sci. Technol. 2015, 60, 1143–1148. [Google Scholar] [CrossRef]
- Mytych, J.; Wnuk, M.; Rattan, S.I. Low doses of nanodiamonds and silica nanoparticles have beneficial hormetic effects in normal human skin fibroblasts in culture. Chemosphere 2016, 148, 307–315. [Google Scholar] [CrossRef] [PubMed]
- Xiu, Z.M.; Zhang, Q.B.; Puppala, H.L.; Colvin, V.L.; Alvarez, P.J. Negligible particle-specific antibacterial activity of silver nanoparticles. Nano Lett. 2012, 12, 4271–4275. [Google Scholar] [CrossRef] [PubMed]
- Guo, Z.; Chen, G.; Zeng, G.; Huang, Z.; Chen, A.; Hu, L.; Wangab, J.; Jiangab, L. Cysteine-induced hormesis effect of silver nanoparticles. Toxicol. Res. 2016, 5, 1268–1272. [Google Scholar] [CrossRef]
- Echavarri-Bravo, V.; Paterson, L.; Aspray, T.J.; Porter, J.S.; Winson, M.K.; Hartl, M.G.J. Natural marine bacteria as model organisms for the hazard-assessment of consumer products containing silver nanoparticles. Mar. Environ. Res. 2017, 130, 293–302. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Wang, J.; Xiu, Z.; Alvarez, P.J. Impacts of silver nanoparticles on cellular and transcriptional activity of nitrogen-cycling bacteria. Environ. Toxicol. Chem. 2013, 32, 1488–1494. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Alvarez, P.J.J. Sublethal Concentrations of Silver Nanoparticles Stimulate Biofilm Development. Environ. Sci. Technol. Lett. 2015, 2, 221–226. [Google Scholar] [CrossRef]
- Zheng, Y.; Hou, L.; Liu, M.; Newell, S.E.; Yin, G.; Yu, C.; Zhang, H.; Li, X.; Gao, D.; Gao, J.; et al. Effects of silver nanoparticles on nitrification and associated nitrous oxide production in aquatic environments. Sci. Adv. 2017, 3, e1603229. [Google Scholar] [CrossRef] [PubMed]
- Sheng, Z.; Van Nostrand, J.D.; Zhou, J.; Liu, Y. Contradictory effects of silver nanoparticles on activated sludge wastewater treatment. J. Hazard. Mater. 2018, 341, 448–456. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Xu, J.; Lu, T.; Zhang, M.; Ke, M.; Fu, Z.; Pan, X.; Qian, H. A comparison of the effects of copper nanoparticles and copper sulfate on Phaeodactylum tricornutum physiology and transcription. Environ. Toxicol. Pharmacol. 2017, 56, 43–49. [Google Scholar] [CrossRef] [PubMed]
- Mykhaylenko, N.F.; Zolotareva, E.K. The Effect of Copper and Selenium Nanocarboxylates on Biomass Accumulation and Photosynthetic Energy Transduction Efficiency of the Green Algae Chlorella Vulgaris. Nanoscale Res. Lett. 2017, 12, 147. [Google Scholar] [CrossRef] [PubMed]
- Tiwari, D.K.; Dasgupta-Schubert, N.; Villaseñor Cendejas, L.M.; Villegas, J.; Carreto Montoya, L.; Borjas Garcıa, S.E. Interfacing carbon nanotubes (CNT) with plants: Enhancement of growth, water and ionic nutrient uptake in maize (Zea mays) and implications for nanoagriculture. Appl. Nanosci. 2014, 4, 577–591. [Google Scholar] [CrossRef]
- Wang, J.; Koo, Y.; Alexander, A.; Yang, Y.; Westerhof, S.; Zhang, Q.; Schnoor, J.L.; Colvin, V.L.; Braam, J.; Alvarez, P.J. Phytostimulation of poplars and Arabidopsis exposed to silver nanoparticles and Ag+ at sublethal concentrations. Environ. Sci. Technol. 2013, 47, 5442–5449. [Google Scholar] [CrossRef] [PubMed]
- Salama, H. Effects of silver nanoparticles in some crop plants, Common bean (Phaseolus vulgaris L.) and corn (Zea mays L.). Int. Res. J. Biotechnol. 2012, 3, 190–197. [Google Scholar]
- Spinoso-Castillo, J.L.; Chavez-Santoscoy, R.A.; Bogdanchikova, N.; Pérez-Sato, J.A.; Morales-Ramos, V.; Bello-Bello, J.J. Antimicrobial and hormetic effects of silver nanoparticles on in vitro regeneration of vanilla (Vanilla planifolia Jacks. Ex Andrews) using a temporary immersion system. Plant Cell Tissue Organ Cult. 2017, 129, 195. [Google Scholar] [CrossRef]
- Bello-Bello, J.J.; Chavez-Santoscoy, R.A.; Lecona-Guzmán, C.A.; Bogdanchikova, N.; Salinas-Ruíz, J.; Gómez-Merino, F.C.; Pestryakov, A. Hormetic Response by Silver Nanoparticles on In Vitro Multiplication of Sugarcane (Saccharum spp. Cv. Mex 69-290) Using a Temporary Immersion System. Dose Response 2017, 15, 1559325817744945. [Google Scholar] [CrossRef] [PubMed]
- Jhanzab, H.M.; Razzaq, A.; Jilani, G.; Rehman, A.; Hafeez, A.; Yasmeen, F. Silver nano-particles enhance the growth, yield and nutrient use efficiency of wheat. Int. J. Agron. Agric. Res. 2015, 7, 15–22. [Google Scholar]
- Razzaq, A.R.; Ammara, H.M.; Jhanzab, T.; Mahmood, A.; Hafeez, S.; Hussain, S. A Novel Nanomaterial to Enhance Growth and Yield of Wheat. J. Nanosci. Technol. 2016, 2, 55–58. [Google Scholar]
- Taran, N.; Batsmanova, L.; Kovalenko, M.; Okanenko, A. Impact of Metal Nanoform Colloidal Solution on the Adaptive Potential of Plants. Nanoscale Res. Lett. 2016, 11, 89. [Google Scholar] [CrossRef] [PubMed]
- Tyne, W.; Little, S.; Spurgeon, D.J.; Svendsen, C. Hormesis depends upon the life-stage and duration of exposure: Examples for a pesticide and a nanomaterial. Ecotoxicol. Environ. Saf. 2015, 120, 117–123. [Google Scholar] [CrossRef] [PubMed]
- Bour, F.; Mouchet, S.; Cadarsi, J.; Silvestre, E.; Chauvet, J.; Bonzom, C.; Pagnout, H.; Clivot, L.; Gauthier, L.; Pinelli, E. Impact of CeO2 nanoparticles on the functions of freshwater ecosystems: A microcosm study. Environ. Sci. Nano 2016. [Google Scholar] [CrossRef]
- Nations, S.; Long, M.; Wages, M.; Canas, J.; Maul, J.D.; Theodorakis, C.; Cobb, G.P. Effects of ZnO nanomaterials on Xenopus laevis growth and development. Ecotoxicol. Environ. Saf. 2011, 74, 203–210. [Google Scholar] [CrossRef] [PubMed]
- Nations, S.; Wages, M.; Cañas, J.E.; Maul, J.; Theodorakis, C.; Cobb, G.P. Acute effects of Fe2O3, TiO2, ZnO and CuO nanomaterials on Xenopus laevis. Chemosphere 2011, 83, 1053–1061. [Google Scholar] [CrossRef] [PubMed]
- Nations, S.; Long, M.; Wages, M.; Maul, J.D.; Theodorakis, C.W.; Cobb, G.P. Subchronic and chronic developmental effects of copper oxide (CuO) nanoparticles on Xenopus laevis. Chemosphere 2015, 135, 166–174. [Google Scholar] [CrossRef] [PubMed]
- Saggese, I.; Sarà, G.; Dondero, F. Silver Nanoparticles Affect Functional Bioenergetic Traits in the Invasive Red Sea Mussel Brachidontes pharaonis. BioMed Res. Int. 2016, 2016, 1872351. [Google Scholar] [CrossRef] [PubMed]
- Iavicoli, I.; Fontana, L.; Corbi, M.; Leso, V.; Marinaccio, A.; Leopold, K.; Schindl, R.; Sgambato, A. Exposure to Palladium Nanoparticles Affects Serum Levels of Cytokines in Female Wistar Rats. PLoS ONE 2015, 10, e0143801. [Google Scholar] [CrossRef] [PubMed]
- Sheng, Z.; Liu, Y. Potential impacts of silver nanoparticles on bacteria in the aquatic environment. J. Environ. Manag. 2017, 191, 290–296. [Google Scholar] [CrossRef] [PubMed]
- Gallon, C.; Flegal, A.R. Sources, fluxes, and biogeochemical cycling of silver in the oceans. Rev. Environ. Contam. Toxicol. 2015, 235, 27–48. [Google Scholar] [CrossRef] [PubMed]
- Mueller, N.C.; Nowack, B. Exposure modeling of engineered nanoparticles in the environment. Environ. Sci. Technol. 2008, 42, 4447–4453. [Google Scholar] [CrossRef] [PubMed]
- Lu, D.; Liu, Q.; Zhang, T.; Cai, Y.; Yin, Y.; Jiang, G. Stable silver isotope fractionation in the natural transformation process of silver nanoparticles. Nat. Nanotechnol. 2016, 11, 682–686. [Google Scholar] [CrossRef] [PubMed]
- Siripong, S.; Rittmann, B.E. Diversity study of nitrifying bacteria in full-scale municipal wastewater treatment plants. Water Res. 2007, 41, 1110–1120. [Google Scholar] [CrossRef] [PubMed]
- Vance, M.E.; Kuiken, T.; Vejerano, E.P.; McGinnis, S.P.; Hochella, M.F., Jr.; Rejeski, D.; Hull, M.S. Nanotechnology in the real world: Redeveloping the nanomaterial consumer products inventory. Beilstein J. Nanotechnol. 2015, 6, 1769–1780. [Google Scholar] [CrossRef] [PubMed]
- Morelli, E.; Salvadori, E.; Bizzarri, R.; Cioni, P.; Gabellieri, E. Interaction of CdSe/ZnS quantum dots with the marine diatom Phaeodactylum tricornutum and the green alga Dunaliella tertiolecta: A biophysical approach. Biophys. Chem. 2013, 182, 4–10. [Google Scholar] [CrossRef] [PubMed]
- Lee, W.M.; Kwak, J.I.; An, Y.J. Effect of silver nanoparticles in crop plants Phaseolus radiatus and Sorghum bicolor: Media effect on phytotoxicity. Chemosphere 2012, 86, 491–499. [Google Scholar] [CrossRef] [PubMed]
- Poschenrieder, C.; Cabot, C.; Martos, S.; Gallego, B.; Barceló, J. Do toxic ions induce hormesis in plants? Plant Sci. 2013, 212, 15–25. [Google Scholar] [CrossRef] [PubMed]
- Iavicoli, I.; Leso, V.; Beezhold, D.H.; Shvedova, A.A. Nanotechnology in agriculture: Opportunities, toxicological implications, and occupational risks. Toxicol. Appl. Pharmacol. 2017, 329, 96–111. [Google Scholar] [CrossRef] [PubMed]
- Calabrese, E.J.; Mattson, M.P. How does hormesis impact biology, toxicology, and medicine? NPJ Aging Mech. Dis. 2017, 3, 13. [Google Scholar] [CrossRef] [PubMed]
- Calabrese, E.J. Preconditioning is hormesis. Part I: Documentation, dose-response features and mechanistic foundations. Pharm. Res. 2016, 110, 242–264. [Google Scholar] [CrossRef] [PubMed]
- Calabrese, E.J. Preconditioning is hormesis. Part II: How the conditioning dose mediates protection. Dose optimization within temporal and mechanistic frameworks. Pharm. Res. 2016, 110, 265–275. [Google Scholar] [CrossRef] [PubMed]
- Lawler, J.M.; Rodriguez, D.A.; Hord, J.M. Mitochondria in the middle: Exercise preconditioning protection of striated muscle. J. Physiol. 2016, 594, 5161–5183. [Google Scholar] [CrossRef] [PubMed]
- Moher, D.; Shamseer, L.; Clarke, M.; Ghersi, D.; Liberati, A.; Petticrew, M.; Shekelle, P.; Stewart, L.A.; PRISMA-P Group. Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015 statement. Syst. Rev. 2015, 4, 1. [Google Scholar] [CrossRef] [PubMed]
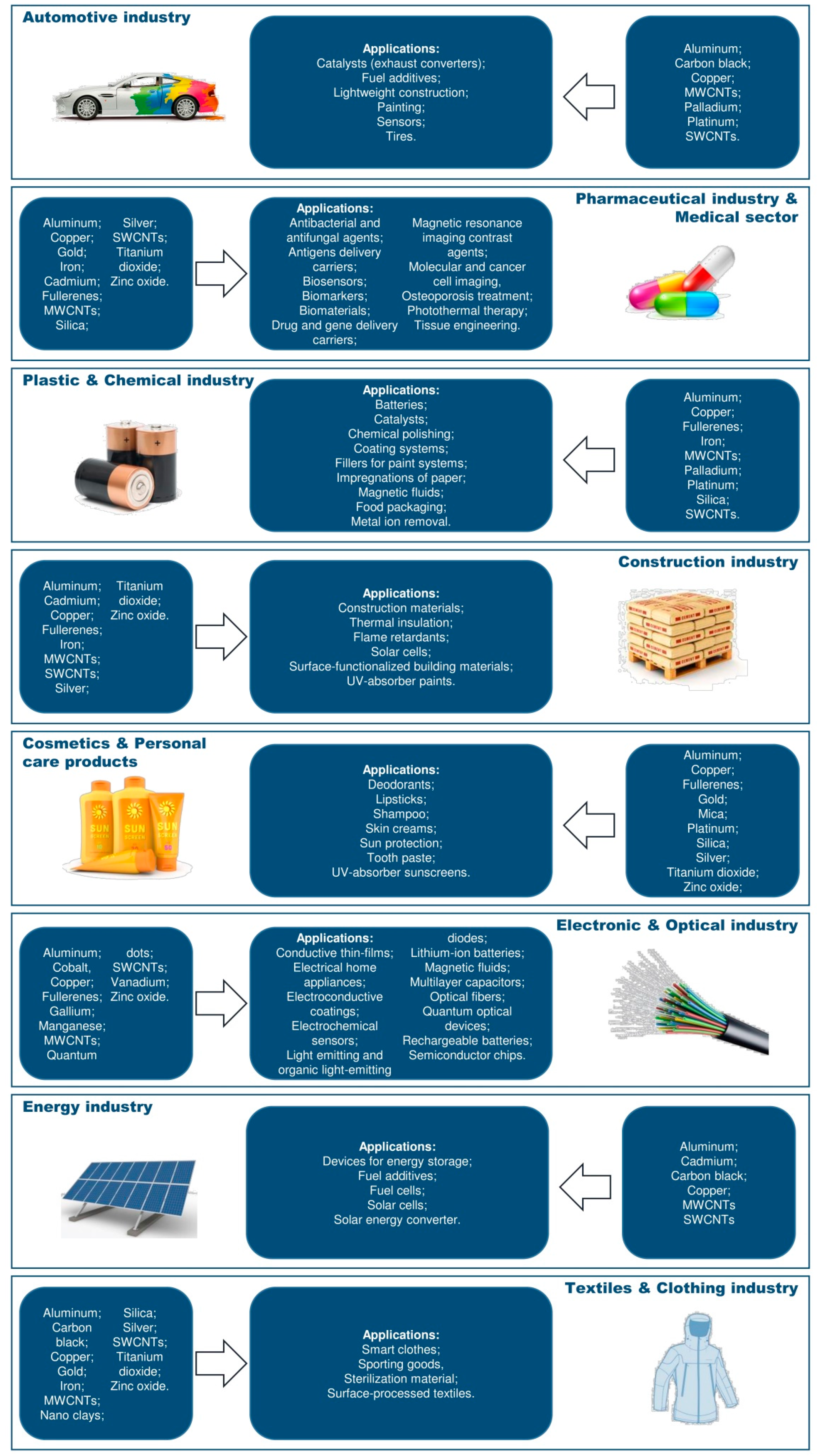
| Type of Nanoparticles | NP-Physicochemical Characterization | Cell Line Investigated | Experimental Design | Assessed Endpoint | Results | References |
|---|---|---|---|---|---|---|
| Ag-NPs | Shape: spherical; Size: 10 and 100 nm; Hydrodynamic diameter: 19.6 ± 0.5, 99.5 ± 0.3 nm. | HepG2 | Cells were exposed to 1 and 2 μg/mL NPs for up to 72 h. | Cell viability | Cell viability increased to ~125% and ~150% relative to controls with 10 and 100 nm Ag-NPs after 48 and 72 h treatments with both exposure concentrations. | Jiao et al. [13] |
| Ag-NPs ZnO-NPs | Size: 30 nm. | Caco2 | Cells were exposed to 0–100 μg/mL NPs for up to 36 h. | Cell viability | Cell viability was slightly (n.s.) increased by 10, 25, 50 μg/mL Ag-NPs for 36 h, and was reduced by higher doses at all 3 time points. ZnO-NPs induced a dose-dependent decrease in cell viability at all 3 time points. | Kang et al. [14] |
| Colloidal (c-) and powder (p-) Ag-NPs | Shape: near-spherical to elongated (p-Ag-NPs); spherical c-Ag-NPs; Size: 37.0 ± 13.0 nm (p-Ag-NPs); 16.6 ± 4.4 nm (c-Ag-NPs); Hydrodynamic diameter: 690.6 ± 74.5 (p-Ag-NPs); 242.5 ± 93.5 nm (c-Ag-NPs). | A549 | Cells were exposed to c-Ag-NPs (5 μg/cm2) or p-Ag-NPs (2.5 μg/cm2) for 24 h followed by 80 or 60 μg/cm2 NPs, respectively, or acrolein as an environmental pollutant (100 μM). | Cell viability | Cell viability increased in cells pre-treated with low concentrations of NPs (2.5 and 5 μg/cm2), compared to cells directly exposed to the toxic challenge. | Sthijns et al. [12] |
| NDs and SiO2-NPs | Size: <10 nm (NDs); 12 nm SiO2-NPs. | FSF1 | Cells were exposed to 0–100 μg/mL NPs for 48 h. | Cell viability | Cell viability was significantly increased by 0.5–2.5 μg/mL SiO2-NPs and 0.5 μg/mL NDs. Above 5 μg/mL there was a progressive decrease. | Mytych et al. [15] |
| Type of Nanoparticles | NP-Physicochemical Characterization | In Vivo Model | Experimental Design | Assessed Endpoint | Results | References |
|---|---|---|---|---|---|---|
| Bacteria strains | ||||||
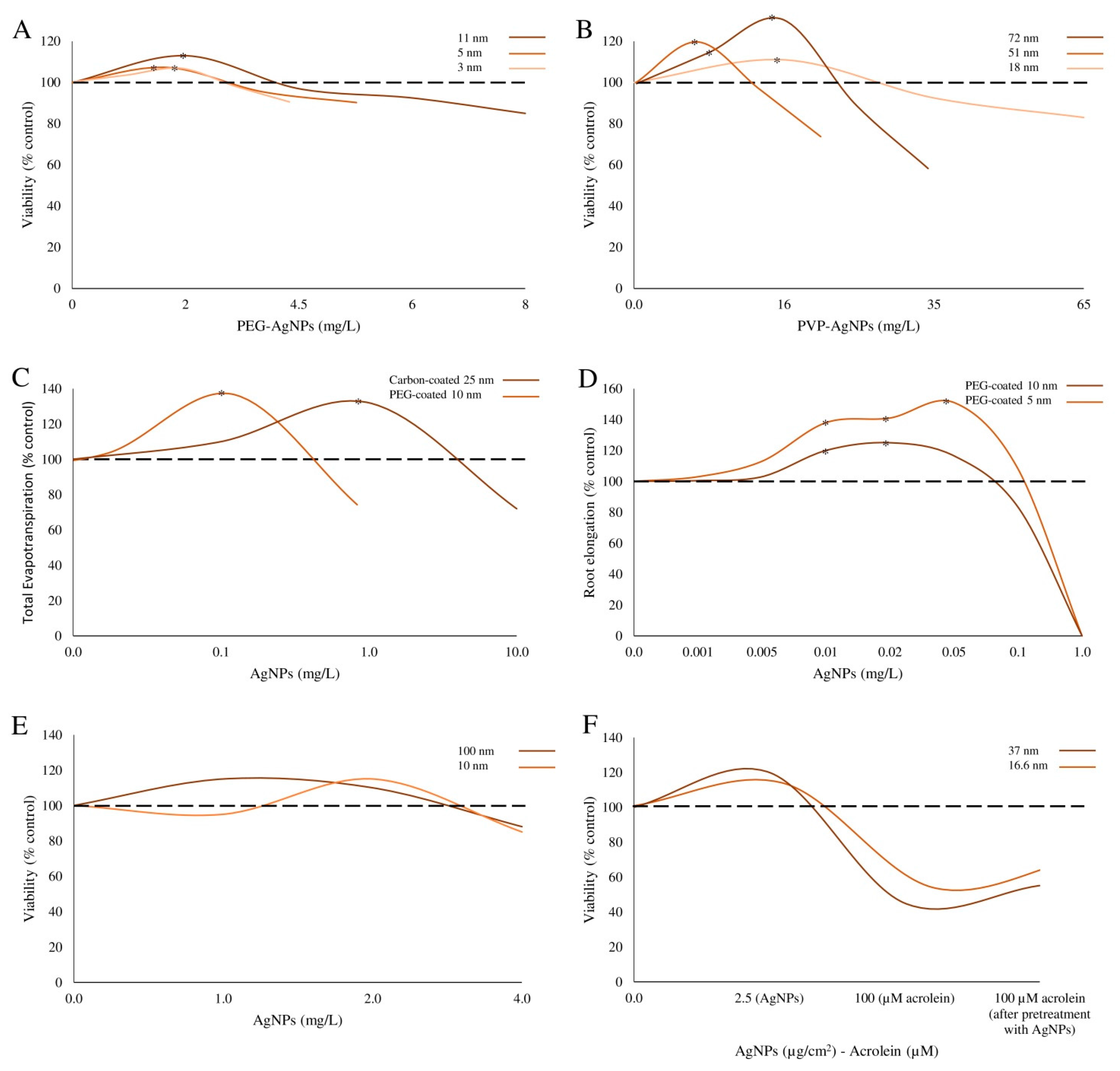
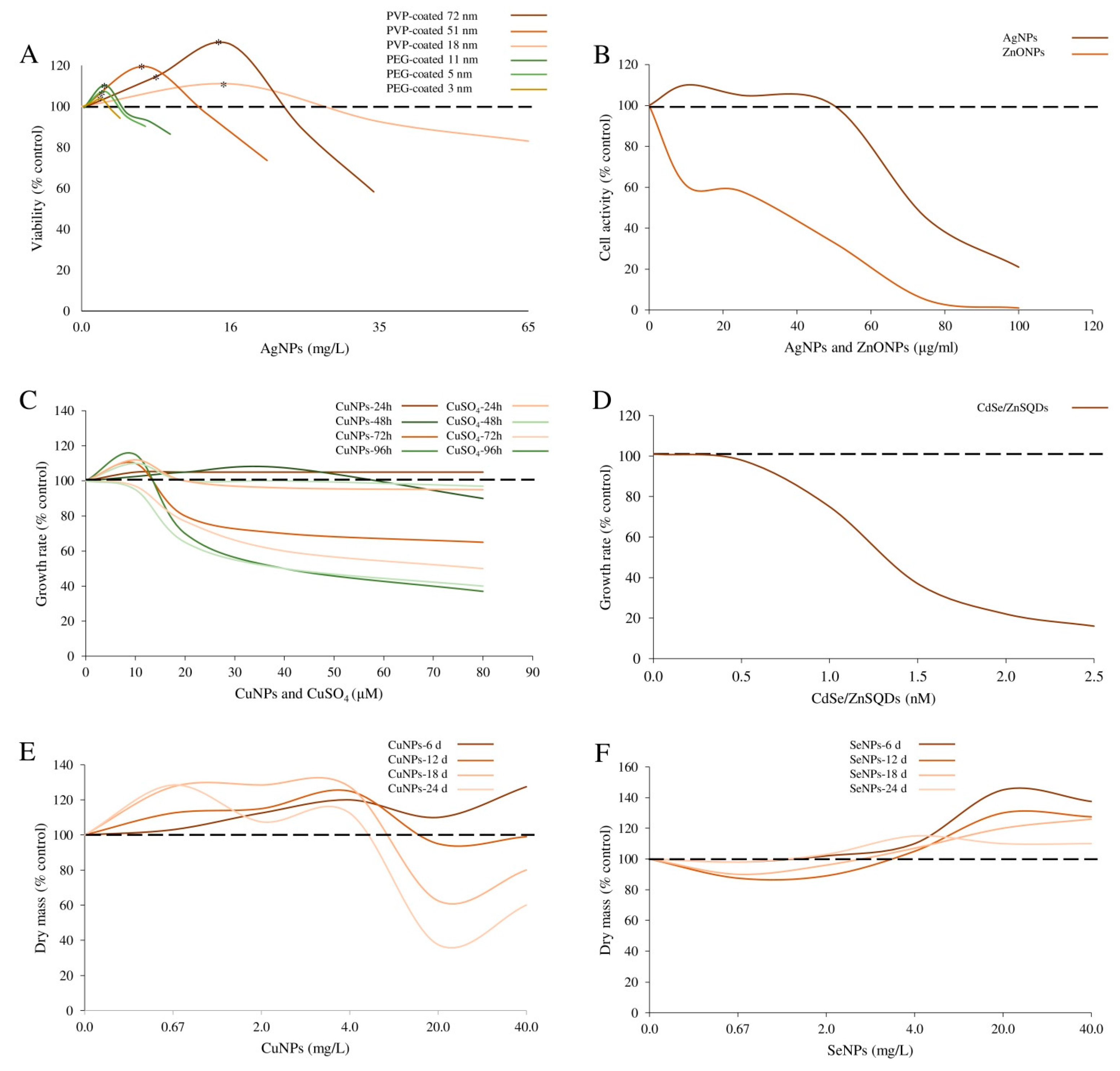
| PEG- and PVP-coated Ag-NPs | PEG-Ag-NPs Shape: spherical; Size: 2.8 ± 0.47; 4.7 ± 0.20; 10.5 ± 0.59 nm. PVP-Ag-NPs Size: 20, 40 and 80 nm. | E. coli strain K12 (ATCC 25404) | Bacteria were exposed to 0–65 μg/mL NPs for 6 h. | Bacteria viability | PEG-Ag-NPs: higher survival rates, 6%, 7% and 13% were induced by 2.8, 4.7 and 10.5 nm NPs at 2.2, 1.8 and 2.0 μg/mL, respectively. PVP-Ag-NPs: higher survival rates, 11%, 17%, and 21% were induced by 20, 40 and 80 nm NPs at 16.4, 5.7 and 6.7 μg/mL, respectively. | Xiu et al. [16] |
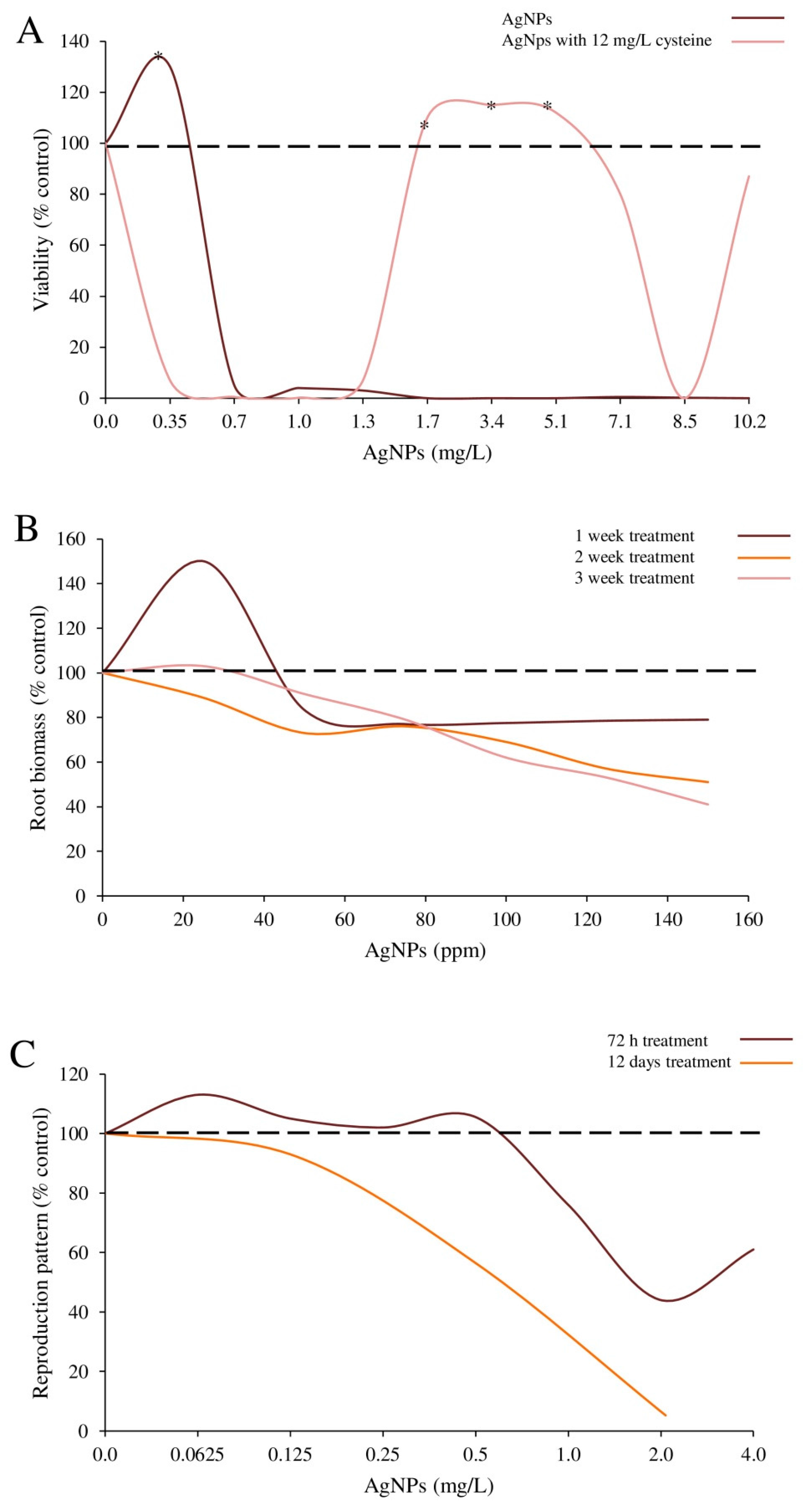
| PVP coated-Ag-NPs | Shape: spherical; Size: 7.59 ± 2.92 nm; Hydrodynamic diameter: 27.1 ± 2.2 nm. | E. coli strain ATCC 25922 | Bacteria were exposed to 0.34–5.1 μg/mL NPs with or without cysteine (12.5 μg/mL). | Bacterial viability | Viability (without cysteine): 29.9% increase in bacteria treated with 0.34 μg/mL NPs compared to controls. Viability (with cysteine): stimulated by 1.7–5.1 μg/mL NPs. | Guo et al. [17] |
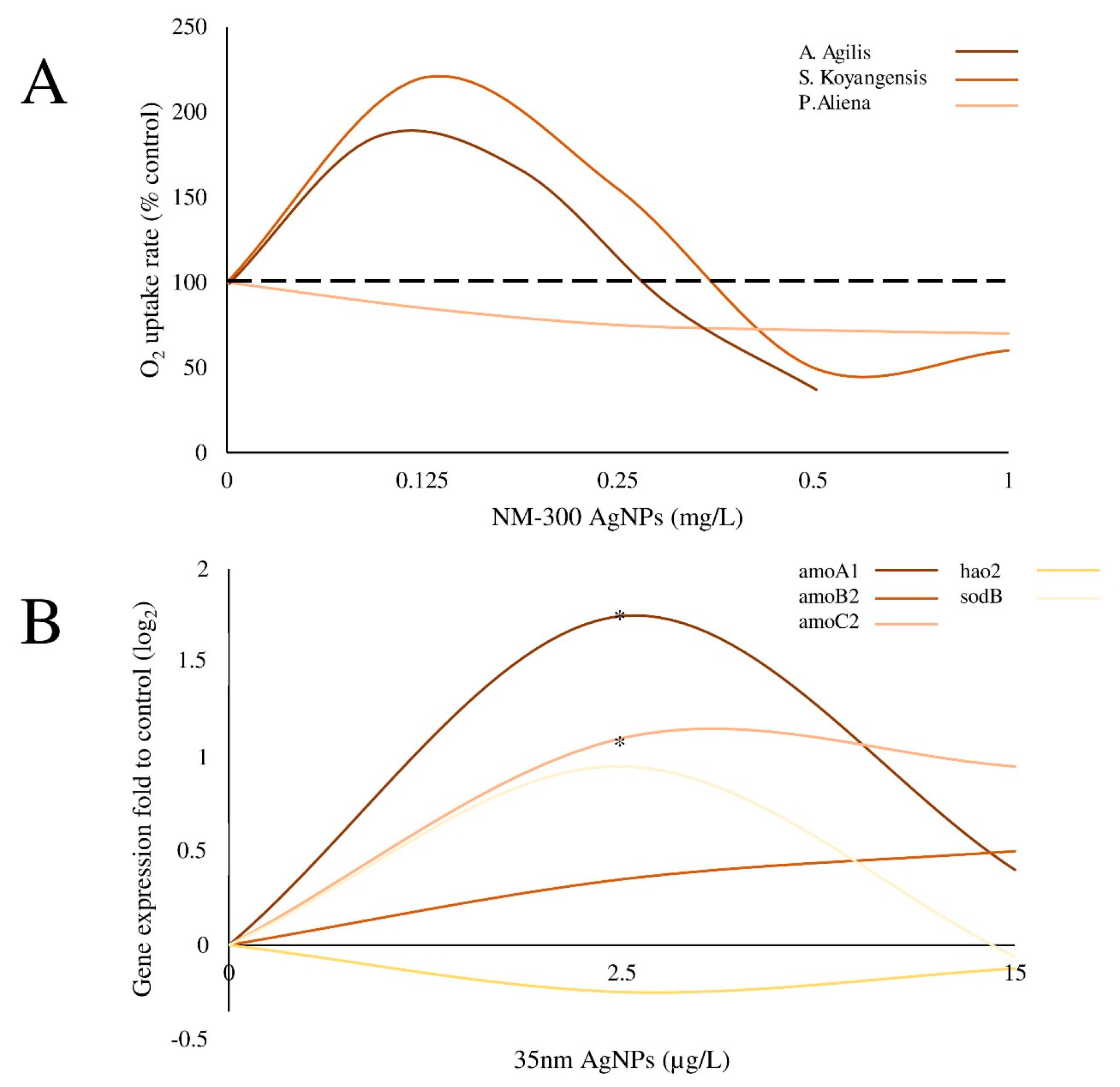
| NM-300 Ag-NPs; Mesosilver containing Ag-NPs | Size: 18.2 ± 7.3 and 14.0 ± 6.9 nm for NM-300 and Mesosilver, respectively. | Gram-positive bacteria A. agilis and S. koyangensis and the Gram-negative bacterium P. aliena | Bacteria were exposed to 0–1 μg/mL Ag-NPs (NM- 300) and Ag-NPs in a household product. Responses could be detected within 0.5–1 h. | O2 uptake | Gram-negative strain P. aliena: reduced O2 uptake rate in a concentration dependent manner Gram-positive strains (A. agilis and S. koyangesis): increased uptake rate at low concentrations (<0.25 μg/mL) compared to controls; reduced uptake at ≥0.5 μg/mL. | Echarri-Bravo et al. [18] |
| Environmental microbial communities | ||||||
| Ag-NPs | Size: 35 nm; Hydrodynamic diameter: 35.4 ± 5.1 nm Surface functionalization: amorphous carbon coating. | Pseudomonas stutzeri, Azotobacter vinelandii, and Nitrosomonas europaea | N. europaea, P. stutzeri and A. vinelandii were exposed to 2.5, 20 and 25 μg/L NPs (10% of the AgNO3-MIC), respectively. N. europaea was also exposed to 15 μg/L NPs. | Transcriptional response to Ag-NP exposure | Ag-NPs had no significant effect on P. stutzeri, and A. vinelandii expression of denitrifying genes or nitrogen-fixing genes. In N. europaea, Ag-NPs (2.5 μg/mL) up-regulated the ammonia mono-oxygenase genes, while such effect was not evident at higher concentrations (15 μg/mL, 60% of the Ag + MIC). | Yang et al. [19] |
| PVP-coated Ag-NPs | Size: 10 nm. | Mixed culture of the effluent from West University Place wastewater treatment plant | Mixed and P. aeruginosa pure cultures were exposed to 45.7; 21.6 and 21.6 μg/L of PVP, Ag+, and NPs for 24 h, respectively. | Biofilm formation | The biofilm coverage fractions were 0.9 ± 0.2% for control, 0.9 ± 0.3% for PVP, 1.1 ± 0.3% for Ag+, and 5.2 ± 2.1% for PVP-Ag-NPs in the mixed culture. Biofilm development was increased by PVP-coated Ag-NPs in the P. aeruginosa pure culture. | Yang and Alvarez [20] |
| PVP-coated Ag-NPs | Size: 10, 30 and 100 nm. | Sediment slurry of the Yangtze Estuary | Sediment slurry was exposed to NPs 0–10,000 μg/L for 30 h. | N2O production | Nitrifier N2O production to Ag-NPs exhibited low-dose stimulation (<534, 1476, and 2473 μg/L for 10-, 30-, and 100-nm Ag-NPs, respectively) and higher dose inhibition. | Zheng et al. [21] |
| Fresh and aged Ag-NPs | Size: 74.2 ± 5.1 (aged NPs), 67.9 ± 1.0 nm (fresh NPs). | Sequencing batch reactors (n = 4) operating for over 3 months | Ag-NPs were added, 27 days after start-up, at a concentration of 1 mg Ag/L in influent (0.5 mg/L in the reactor) for over 2 months. | Microbial community analysis | Microbial analysis (16S gene-based sequencing): fresh Ag-NPs had the highest number of genes detected and richness. | Sheng et al. [22] |
| Algae | ||||||
| Cu-NPs | Size: 10–30 nm; Purity: 99.9%. | Marin diatom Phaeodactylum tricornutum | Algal cultures were supplied with NPs (0–80 μM) for 48–96 h. | Algal growth; photosynthetic pigment content | Algal growth: slightly stimulated by 48 h of 10 μM Cu-NPs and inhibited by the same dose for 96 h. Greater concentrations (20, 40, 80 μM) significantly inhibited algal growth in a time dependent manner. Photosynthetic pigment content: slightly increased by 10 μM and decreased by 40 μM Cu-NPs (by 41.4%, 47.2%, 44.3% and 76.6%, 83.6%, and 76.1% for chlorophyll a, c and carotenoids, after 48 and 96 h, respectively). | Zhu et al. [23] |
| Cu- and Se-carboxylated with citric acid nanoaquachelates | Size: ~100 nm. | Green algae Chlorella vulgaris | Algal cultures were supplied with NPs (0.67–40 μg/mL) for 6–24 days. | Algal growth: biomass production | Increased by 0.67–4 μg/mL of Cu-nanocarboxylates (~20% biomass increase); inhibited by 20 to 40 μg/mL after the 12th day of cultivation. Decreased by 0.07 and 0.2 μg/mL of Se-nanocarboxylates (6 and 12 days of culture); stimulated by 0.4–4 μg/mL (40–45% biomass increase). | Mykhaylenko and Zolotareva, [24] |
| Plants and seedlings | ||||||
| MW-CNTs | Size: 6–9 nm large, 5 μm length; Purity: 95%. | Zea mays seedlings | Agar culture medium containing MW-CNTs (0–60 μg/mL) was used for seed growth (7 days) | Plant indices of growth and water absorption | Water content (shoots): increased at 10 μg/mL, decreased at higher concentrations. Dry weight (seedlings, roots, shoots): increased at 20 μg/mL, decreased thereafter. | Tiwari et al. [25] |
| PEG-coated and carbon-coated Ag-NPs | Size: 5 and 10 (PEG-Ag-NPs); 25 nm (carbon coated Ag-NPs). | Poplars (Populus deltoides × nigra) and Arabidopsis thaliana | Plants were exposed to 0–100 μg/mL NPs. | Growth parameters | Evotranspiration (poplar): enhanced by 1 μg/mL carbon-Ag-NPs (42% vs. controls) and by 0.1 μg/mL PEG-Ag-NPs (43% vs. controls); decreased by 100 μg/mL carbon-Ag-NPs (87%). Root and stem biomass (poplar): increased by 63% and 46% after exposure to 1 μg/mL for 11 days carbon-Ag-NPs. Fresh weights of roots, stem and leaves (poplar): increased by 0.1 μg/mL PEG-Ag-NPs (48%, 50%, 39% vs. controls); reduced by 100 μg/mL carbon Ag-NPs (87%, 42%%, 81%). Growth (Arabidopsis): enhanced (more extensively expanded leaves) by 1 μg/mL, and inhibited by 100 μg/mL carbon-Ag-NPs. Root growth and shoot weights were increased by 0.01 and 0.05 μg/mL of 5 nm or 0.01 and 0.02 μg/mL fo 10 nm PEG-Ag-NPs and inhibited by 1 μg/mL of these NPs. | Wang et al. [26] |
| Ag-NPs | Shape: spherical; Size: 10–30 nm. | Common bean (Phaseolus vulgaris) and corn (Zea mays) plants | Ag-NPs (15 mL at 0–100 μg/mL) were daily supplied to plants for 12 days. | Plant growth parameters | Shoot and root length; fresh and dry weight; leaf area; chlorophyll and carbohydrate content: increased with concentrations up to 60 μg/mL, decreased by higher, 80 and 100 μg/mL. | Salama et al. [27] |
| Ag-NPs | Shape: spherical; Size: 35 ± 15 nm; Surface functionalization: PVP. | Vanilla shoots (2 cm long) | Shoots were exposed to 0–200 μg/mL NPs for 30 days of culture in a recipient for automated temporary immersion. | Shoot multiplication and length | Number of shoot per explant: 25 and 50 μg/mL NPs: 14.33 and 14.89 respectively; 200 μg/mL: 4.55. Shoot length: 25 and 50 μg/mL NP: 14.33 and 14.89 cm, respectively; 100 and 200 μg/mL: 1.14 ± 0.07 and 0.82 ± 0.6 cm, respectively. | Spinoso-Castillo et al. [28] |
| PVP coated Ag-NPs | Shape: spherical; Size: 35 ± 15 nm. | Sugarcane shoots | Shoots were placed in a temporary immersion bioreactor in which NP solutions were added (0–250 μg/mL) for 30 days. | Shoot multiplication rate and length | The treatment with 50 and 100 mg/L Ag-NPs induced the greatest shoot number (increased by 35% and 28%, respectively) and length (increased by 52% and 48%, respectively). The lowest Ag-NP concentration (25 μg/mL) had no effect on the evaluated variables. The highest Ag-NP concentration (250 μg/mL) caused a reduction in shoot number and length. | Bello-Bello et al. [29] |
| Ag-NPs | Size: 10–20 nm. | Seedlings of wheat variety Narc-2009 (n = 10 per pot) | Pot soil was soaked with Ag-NPs (0–150 μg/mL) solution or distilled water in control treatment. | Seedling growth; yield attributes and nutrient use efficiency | Ag-NPs at 25 and 50 μg/mL significantly improved maximum leaf area (19.7 and 18.18 cm2, respectively, vs. 15 cm2 in controls); grain yield (n = 29 grains per spike at 25 μg/mL); and nutrient use efficiency (N: 74.3%; K: 89.0%; P: 72.5% at 25 μg/mL). Greater concentrations decreased leaf area, grain yielded (n = 11.5 grains per spike at 150 μg/mL) and nutrient use efficiency (N: 36.4%; K: 67.88%; P: 41.3% at 150 μg/mL). | Jhanzab et al. [30] |
| Ag-NPs | Size: 10–20 nm. | Seedlings of wheat variety Narc-2009 (n = 10 per pot) | Seedlings were applied Ag-NPs (0–150 μg/mL) through a blended Murashige and Skoog medium. | Germination, seedling growth, yield attributes | Germination (medium): Ag-NPs had no effect (25–75 μg/mL), and reduced germination (100–150 μg/mL) compared to controls. Number of seminal roots (medium): increased by 25–75 μg/mL, decreased by 100–150 μg/mL, compared to controls. Growth parameters (medium): higher leaf area, fresh weight, dry weight and root biomass was observed with 25 μg/mL Ag-NPs as compared to control after one week of growth in nutrient medium. Growth parameters (NP added soil): more number of grains per spike was produced with 25 and 50 μg/mL, while a decrease was observed with 75–100 μg/mL compared to controls; grain yield per pot was higher with 25 μg/mL, maximum reduction was evident with 150 μg/mL compared to controls. | Razzaq et al. [31] |
| Metal nanoform colloidal solution | Metal NPs in solution (size in nm; dose in μg/mL): Ag (30–50; 150); Cu (100–150; 200); Fe (20–30; 300); Zn (30–50; 150); Mn (20–30; 150). | Soybean seeds and plants | Pre-sowing seed treatment with metal colloids at 120 or 240 μg/mL; pre-sowing (120 μg/mL) combined with vegetative treatment. | Oxidative stress; lipid peroxidation | Pre-sowing treatment with 120 μg/mL nanosolution increased (12%) lipid peroxidation, while treatment with 240 μg/mL and 120 μg/mL + vegetative treatment decreased the oxidative process (19% and 10%, respectively). | Taran et al. [32] |
| Nematodes and larvae | ||||||
| Ag-NPs | Size: 8 nm. | Caenorhabditis elegans nematodes | Nematodes (n = 6) were exposed to 0–4 μg/mL NPs for 72 h. | Reproduction outcome: number of offspring | Short-term test: mean number of offspring (89 and 117, at the 1st and 2nd test, respectively) at 0.0625 μg/mL was higher compared to controls (75) (n.s.). Mean number of offspring at higher doses (2, 4 μg/mL) was significantly reduced compared to controls. Full life-cycle brood size (mean): 182 controls compared to 168, 98 and 5 juveniles at 0.0625, 0.7 and 2.1 μg/mL. No eggs were laid by nematodes exposed to 6.5 μg/mL. | Tyne et al. [33] |
| CeO2-NPs | CeO2-NPs (1) Shape: spherical; Size: 2–5 nm; Surface functionalization: tri ammonium citrate layer; Uncoated CeO2- spherical NPs (2) Size: 2–5 nm; Uncoated CeO2-nano-plates (3) Size: 10–60 nm. | Microcosms containing microbial communities, diatoms and chironomid larvae. | Fresh NP suspensions (50 mL of 93.4 mg/L) were added 12 times over 4 weeks to obtain the final concentration of 1 mg/L. Chironomid larvae were added after 1 week of contamination. | Larval growth and teratogenicity | The teratogenicity induced by CeO2-NPs (1) (frequency and seriousness of deformities) was significantly less than that in the control conditions. CeO2-NPs (2) and (3) conditions did not show differences compared to controls, although a larger number of larvae lacking one or more teeth was observed with CeO2-NPs (2). | Bour et al. [34] |
| Superior aquatic organisms | ||||||
| ZnO-NPs | Size: 40–100 nm; Surface area: 10–25 m2/g. | Xenopus laevis embryos (n = 15 per dose, per replicate) | Tadpoles were exposed to aqueous suspensions beginning in ovo through metamorphosis (0–2 μg/mL). | Developmental assessment | SVL: on days 10–20, 0.067 μg/mL had significantly longer SVL than controls. On days 10–46, 0.305, 0.513 and 0.799 μg/mL tadpoles were shorter than controls. Total body length: decreased from day 10 to 46, by 0.305, 0.513 and 0.799 μg/mL ZnO-NPs; increased, on days 10, 15, and 35, by 0.067 μg/mL ZnO-NPs compared to controls. Hind limb length: decreased by 0.305, 0.513 and 0.799 μg/mL from day 10 to 46, increased, from day 20 to 35, by 0.067 μg/mL ZnO-NPs compared to controls. | Nations et al. [35] |
| ZnO-NPs | Size: 40–100 nm; Surface area: 10–25 m2/g. | Xenopus laevis embryos (n = 10 per dose, per replicate) | Nominal solutions of ZnO-NPs (0.1–31.6 μg/mL) were employed for a subacute 96 h exposure protocol for daily solution exchange. | Total body length | Increased by 0.1, 0.316, and 1 μg/mL ZnO; decreased by 10 and 31 μg/mL. | Nations et al. [36] |
| CuO-NPs | Size: 23–37 nm; Surface area: 25–40 m2/g. | Xenopus laevis embryos (n = 15 per dose, per replicate) | Nominal solutions of CuO-NPs (0.01875–2.5 μg/mL) were employed for 14 and 47 days. | Total body length | All tadpoles in solutions containing less than 0.15 μg/mL CuO-NPs achieved significantly longer total body length than controls in chronic treatment. Tadpoles in the 0.3 μg/mL had lower total body length than controls in chronic treatment. | Nations et al. [37] |
| Ag-NPs | Size: 5 nm. | Lessepsian-entry bivalve sea mussel B. pharaonis | Animals were treated with 2–40 μg/L NPs for 8 days. | Respiration and heartbeat rate | Overall (8 days) respiration rate: 2 and 40 μg/L increased the rate, while 20 μg/L did not show differences compared to controls. RR dynamics during the 8-day period of exposure: a fair increase followed by acclimation to control values was induced by 40 μg/L. A delayed RR intensification was seen with 2 μg/L NPs. | Saggese et al. [38] |
| Rats | ||||||
| Pd-NPs | Shape: spherical; Size: 10 nm; Size distribution: 4–16 nm. | 20 female pathogen-free Wistar rats | Single intravenous injection (via the tail vein) of vehicle (control group) and 0.012, 0.12, 1.2 and 12 μg/kg Pd-NPs (exposed rats). | Production and release of different cytokines (IL-1α, IL-2, IL-4, IL-6, IL-10, IL-12, GM-CSF, INF-γ and TNF-α) | Exposure to Pd-NPs was able to affect immune response since the mean serum concentrations of all cytokines decreased after the administration of 0.012 μg/kg Pd-NPs, while their levels exceeded the control values at higher doses of exposure (0.12, 1.2 and 12 μg/kg). | Iavicoli et al. [39] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Iavicoli, I.; Leso, V.; Fontana, L.; Calabrese, E.J. Nanoparticle Exposure and Hormetic Dose–Responses: An Update. Int. J. Mol. Sci. 2018, 19, 805. https://doi.org/10.3390/ijms19030805
Iavicoli I, Leso V, Fontana L, Calabrese EJ. Nanoparticle Exposure and Hormetic Dose–Responses: An Update. International Journal of Molecular Sciences. 2018; 19(3):805. https://doi.org/10.3390/ijms19030805
Chicago/Turabian StyleIavicoli, Ivo, Veruscka Leso, Luca Fontana, and Edward J. Calabrese. 2018. "Nanoparticle Exposure and Hormetic Dose–Responses: An Update" International Journal of Molecular Sciences 19, no. 3: 805. https://doi.org/10.3390/ijms19030805
APA StyleIavicoli, I., Leso, V., Fontana, L., & Calabrese, E. J. (2018). Nanoparticle Exposure and Hormetic Dose–Responses: An Update. International Journal of Molecular Sciences, 19(3), 805. https://doi.org/10.3390/ijms19030805







