Potential Biomarkers for Predicting Congenital Cytomegalovirus Infection
Abstract
1. Introduction
2. Cytomegalovirus-Specific Antibody Tests
3. Cytomegalovirus Immunoglobulin G Avidity Measurements
4. Epitope-Specific Antibody Detection
5. Cytomegalovirus DNA Polymerase Chain Reaction Assay
6. Assays for Measuring Cytomegalovirus-Specific, T Cell-Mediated Immunity
7. Imaging Examinations
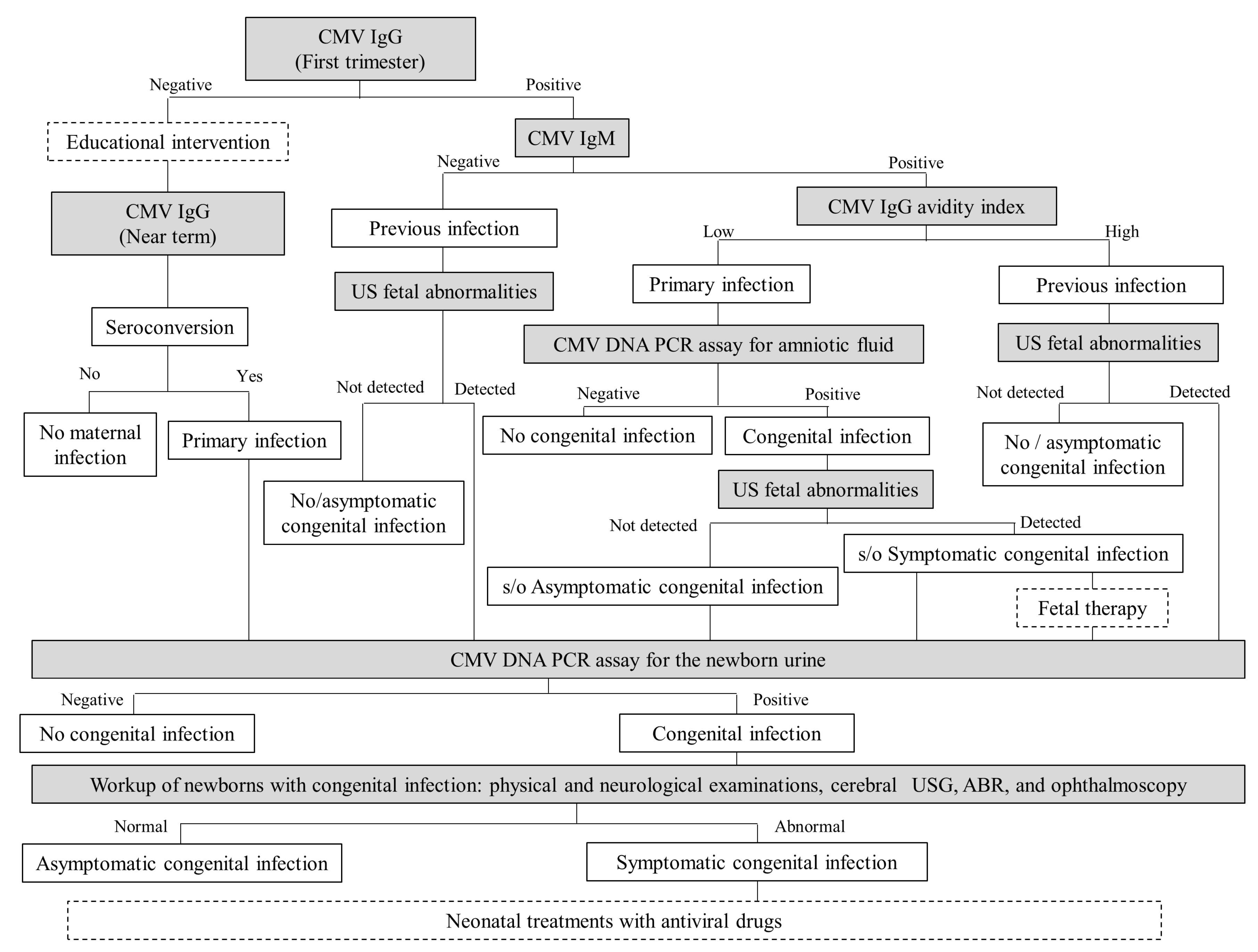
8. Clinical Factors Associated with the Occurrence of Congenital Cytomegalovirus Infection
9. Neonatal Screening and Definitive Diagnosis of Congenital Cytomegalovirus Infection
10. Potential Strategies to Improve the Outcomes of Children with Congenital CMV Infection
11. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| CMV | Cytomegalovirus |
| PCR | Polymerase chain reaction |
| FGR | Fetal growth restriction |
| IgG | Immunoglobulin G |
| IgM | Immunoglobulin M |
| ROC | Receiver operating characteristic |
| GW | Weeks of gestation |
| PPV | Positive predictive value |
| NPV | Negative predictive value |
| gp | glycoproteins |
| gB | glycoprotein B |
| gH | glycoprotein H |
| ELISA | Enzyme-linked immunosorbent assay |
| OR | Odds ratio |
| CI | Confidence interval |
| ELISPOT | Enzyme-linked immunosorbent spot |
| QFT | QuantiFERON |
| MRI | Magnetic resonance imaging |
References
- Revello, M.G.; Gerna, G. Diagnosis and management of human cytomegalovirus infection in the mother, fetus, and newborn infant. Clin. Microbiol. Rev. 2002, 15, 680–715. [Google Scholar] [CrossRef] [PubMed]
- Stagno, S.; Whitley, R.J. Herpesvirus infections of pregnancy. Part I: Cytomegalovirus and Epstein-Barr virus infections. N. Engl. J. Med. 1985, 313, 1270–1274. [Google Scholar] [CrossRef] [PubMed]
- Marsico, C.; Kimberlin, D.W. Congenital cytomegalovirus infection: Advances and challenges in diagnosis, prevention and treatment. Ital. J. Pediatr. 2017, 43, 38. [Google Scholar] [CrossRef] [PubMed]
- Kimberlin, D.W.; Lin, C.Y.; Sanchez, P.J.; Demmler, G.J.; Dankner, W.; Shelton, M.; Jacobs, R.F.; Vaudry, W.; Pass, R.F.; Kiell, J.M.; et al. Effect of ganciclovir therapy on hearing in symptomatic congenital cytomegalovirus disease involving the central nervous system: A randomized, controlled trial. J. Pediatr. 2003, 143, 16–25. [Google Scholar] [CrossRef]
- Nishida, K.; Morioka, I.; Nakamachi, Y.; Kobayashi, Y.; Imanishi, T.; Kawano, S.; Iwatani, S.; Koda, T.; Deguchi, M.; Tanimura, K.; et al. Neurological outcomes in symptomatic congenital cytomegalovirus-infected infants after introduction of newborn urine screening and antiviral treatment. Brain Dev. 2016, 38, 209–216. [Google Scholar] [CrossRef] [PubMed]
- Soper, D.E. Congenital cytomegalovirus infection: An obstetrician’s point of view. Clin. Infect. Dis. 2013, 57 (Suppl. S4), S171–S173. [Google Scholar] [CrossRef] [PubMed]
- Revello, M.G.; Fabbri, E.; Furione, M.; Zavattoni, M.; Lilleri, D.; Tassis, B.; Quarenghi, A.; Cena, C.; Arossa, A.; Montanari, L.; et al. Role of prenatal diagnosis and counseling in the management of 735 pregnancies complicated by primary human cytomegalovirus infection: A 20-year experience. J. Clin. Virol. 2011, 50, 303–307. [Google Scholar] [CrossRef] [PubMed]
- Griffiths, P.D.; Stagno, S.; Pass, R.F.; Smith, R.J.; Alford, C.A., Jr. Infection with cytomegalovirus during pregnancy: Specific IgM antibodies as a marker of recent primary infection. J. Infect. Dis. 1982, 145, 647–653. [Google Scholar] [CrossRef] [PubMed]
- Lazzarotto, T.; Guerra, B.; Gabrielli, L.; Lanari, M.; Landini, M.P. Update on the prevention, diagnosis and management of cytomegalovirus infection during pregnancy. Clin. Microbiol. Infect. 2011, 17, 1285–1293. [Google Scholar] [CrossRef] [PubMed]
- Lagrou, K.; Bodeus, M.; Van Ranst, M.; Goubau, P. Evaluation of the new architect cytomegalovirus immunoglobulin M (IgM), IgG, and IgG avidity assays. J. Clin. Microbiol. 2009, 47, 1695–1699. [Google Scholar] [CrossRef] [PubMed]
- Sonoyama, A.; Ebina, Y.; Morioka, I.; Tanimura, K.; Morizane, M.; Tairaku, S.; Minematsu, T.; Inoue, N.; Yamada, H. Low IgG avidity and ultrasound fetal abnormality predict congenital cytomegalovirus infection. J. Med. Virol. 2012, 84, 1928–1933. [Google Scholar] [CrossRef] [PubMed]
- Ebina, Y.; Minematsu, T.; Sonoyama, A.; Morioka, I.; Inoue, N.; Tairaku, S.; Nagamata, S.; Tanimura, K.; Morizane, M.; Deguchi, M.; et al. The IgG avidity value for the prediction of congenital cytomegalovirus infection in a prospective cohort study. J. Perinat. Med. 2014, 42, 755–759. [Google Scholar] [CrossRef] [PubMed]
- Ebina, Y.; Minematsu, T.; Morioka, I.; Deguchi, M.; Tairaku, S.; Tanimura, K.; Sonoyama, A.; Nagamata, S.; Morizane, M.; Yamada, H. Rapid increase in the serum cytomegalovirus IgG avidity index in women with a congenitally infected fetus. J. Clin. Virol. 2015, 66, 44–47. [Google Scholar] [CrossRef] [PubMed]
- Tanimura, K.; Tairaku, S.; Morioka, I.; Ozaki, K.; Nagamata, S.; Morizane, M.; Deguchi, M.; Ebina, Y.; Minematsu, T.; Yamada, H. Universal screening with use of immunoglobulin G avidity for congenital cytomegalovirus infection. Clin. Infect. Dis. 2017, 65, 1652–1658. [Google Scholar] [CrossRef] [PubMed]
- Britt, W.J. Maternal immunity and the natural history of congenital human cytomegalovirus infection. Viruses 2018, 10, 405. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Shenk, T. Human cytomegalovirus virion protein complex required for epithelial and endothelial cell tropism. Proc. Natl. Acad. Sci. USA 2005, 102, 18153–18158. [Google Scholar] [CrossRef] [PubMed]
- Ryckman, B.J.; Rainish, B.L.; Chase, M.C.; Borton, J.A.; Nelson, J.A.; Jarvis, M.A.; Johnson, D.C. Characterization of the human cytomegalovirus gH/gL/UL128-131 complex that mediates entry into epithelial and endothelial cells. J. Virol. 2008, 82, 60–70. [Google Scholar] [CrossRef] [PubMed]
- Wen, Y.; Monroe, J.; Linton, C.; Archer, J.; Beard, C.W.; Barnett, S.W.; Palladino, G.; Mason, P.W.; Carfi, A.; Lilja, A.E. Human cytomegalovirus gH/gL/UL128/UL130/UL131A complex elicits potently neutralizing antibodies in mice. Vaccine 2014, 32, 3796–3804. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, R.; Abe, M.; Oguri, K.; Torikai, M.; Nishimura, T.; Mori, H.; Koshizuka, T.; Inoue, N. Analysis of relationships between polymorphisms in the genes encoding the pentameric complex and neutralization of clinical cytomegalovirus isolates. Vaccine 2018, 36, 5983–5989. [Google Scholar] [CrossRef] [PubMed]
- Schleiss, M.R.; Permar, S.R.; Plotkin, S.A. Progress toward development of a vaccine against congenital cytomegalovirus infection. Clin. Vaccine Immunol. 2017, 24. [Google Scholar] [CrossRef] [PubMed]
- Lilleri, D.; Kabanova, A.; Revello, M.G.; Percivalle, E.; Sarasini, A.; Genini, E.; Sallusto, F.; Lanzavecchia, A.; Corti, D.; Gerna, G. Fetal human cytomegalovirus transmission correlates with delayed maternal antibodies to gH/gL/pUL128-130-131 complex during primary infection. PLoS ONE 2013, 8, e59863. [Google Scholar] [CrossRef] [PubMed]
- Kaneko, M.; Ohhashi, M.; Minematsu, T.; Muraoka, J.; Kusumoto, K.; Sameshima, H. Immunoglobulin G avidity as a diagnostic tool to identify pregnant women at risk of congenital cytomegalovirus infection. J. Infect. Chemother. 2017, 23, 173–176. [Google Scholar] [CrossRef] [PubMed]
- Eggers, M.; Radsak, K.; Enders, G.; Reschke, M. Use of recombinant glycoprotein antigens gB and gH for diagnosis of primary human cytomegalovirus infection during pregnancy. J. Med. Virol. 2001, 63, 135–142. [Google Scholar] [CrossRef]
- Boppana, S.B.; Rivera, L.B.; Fowler, K.B.; Mach, M.; Britt, W.J. Intrauterine transmission of cytomegalovirus to infants of women with preconceptional immunity. N. Engl. J. Med. 2001, 344, 1366–1371. [Google Scholar] [CrossRef] [PubMed]
- Novak, Z.; Ross, S.A.; Patro, R.K.; Pati, S.K.; Reddy, M.K.; Purser, M.; Britt, W.J.; Boppana, S.B. Enzyme-linked immunosorbent assay method for detection of cytomegalovirus strain-specific antibody responses. Clin. Vaccine Immunol. 2009, 16, 288–290. [Google Scholar] [CrossRef] [PubMed]
- Liesnard, C.; Donner, C.; Brancart, F.; Gosselin, F.; Delforge, M.L.; Rodesch, F. Prenatal diagnosis of congenital cytomegalovirus infection: Prospective study of 237 pregnancies at risk. Obstet. Gynecol. 2000, 95, 881–888. [Google Scholar] [CrossRef] [PubMed]
- Guerra, B.; Lazzarotto, T.; Quarta, S.; Lanari, M.; Bovicelli, L.; Nicolosi, A.; Landini, M.P. Prenatal diagnosis of symptomatic congenital cytomegalovirus infection. Am. J. Obstet. Gynecol. 2000, 183, 476–482. [Google Scholar] [CrossRef] [PubMed]
- Yinon, Y.; Farine, D.; Yudin, M.H. Cytomegalovirus infection in pregnancy. J. Obstet. Gynaecol. Can. 2010, 32, 348–354. [Google Scholar] [CrossRef]
- Tanimura, K.; Tairaku, S.; Ebina, Y.; Morioka, I.; Nagamata, S.; Deguchi, K.; Morizane, M.; Deguchi, M.; Minematsu, T.; Yamada, H. Prediction of congenital cytomegalovirus infection in high-risk pregnant women. Clin. Infect. Dis. 2017, 64, 159–165. [Google Scholar] [CrossRef] [PubMed]
- La Rosa, C.; Diamond, D.J. The immune response to human CMV. Future Virol. 2012, 7, 279–293. [Google Scholar] [CrossRef] [PubMed]
- Gratama, J.W.; van Esser, J.W.; Lamers, C.H.; Tournay, C.; Löwenberg, B.; Bolhuis, R.L.; Cornelissen, J.J. Tetramer-based quantification of cytomegalovirus (CMV)-specific CD8+ T lymphocytes in T-cell-depleted stem cell grafts and after transplantation may identify patients at risk for progressive CMV infection. Blood 2001, 98, 1358–1364. [Google Scholar] [CrossRef] [PubMed]
- Bunde, T.; Kirchner, A.; Hoffmeister, B.; Habedank, D.; Hetzer, R.; Cherepnev, G.; Proesch, S.; Reinke, P.; Volk, H.D.; Lehmkuhl, H.; et al. Protection from cytomegalovirus after transplantation is correlated with immediate early 1-specific CD8 T cells. J. Exp. Med. 2005, 201, 1031–1036. [Google Scholar] [CrossRef] [PubMed]
- Fornara, C.; Cassaniti, I.; Zavattoni, M.; Furione, M.; Adzasehoun, K.M.; De Silvestri, A.; Comolli, G.; Baldanti, F. Human cytomegalovirus-specific memory CD4_ T-cell response and its correlation with virus transmission to the fetus in pregnant women with primary infection. Clin. Infect. Dis. 2017, 65, 1659–1665. [Google Scholar] [CrossRef] [PubMed]
- Saldan, A.; Forner, G.; Mengoli, C.; Gussetti, N.; Palu, G.; Abate, D. Strong cell-mediated immune response to human cytomegalovirus is associated with increased risk of fetal infection in primarily infected pregnant women. Clin. Infect. Dis. 2015, 61, 1228–1234. [Google Scholar] [CrossRef] [PubMed]
- Saldan, A.; Forner, G.; Mengoli, C.; Tinto, D.; Fallico, L.; Peracchi, M.; Gussetti, N.; Palu, G.; Abate, D. Comparison of the cytomegalovirus (CMV) enzyme-linked immunosorbent spot and CMV QuantiFERON cell-mediated immune assays in CMV-seropositive and -seronegative pregnant and nonpregnant women. J. Clin. Microbiol. 2016, 54, 1352–1356. [Google Scholar] [CrossRef] [PubMed]
- Forner, G.; Saldan, A.; Mengoli, C.; Gussetti, N.; Palu, G.; Abate, D. Cytomegalovirus (CMV) enzyme-linked immunosorbent spot assay but not CMV QuantiFERON assay is a novel biomarker to determine risk of congenital CMV infection in pregnant women. J. Clin. Microbiol. 2016, 54, 2149–2154. [Google Scholar] [CrossRef] [PubMed]
- Malinger, G.; Lev, D.; Lerman-Sagie, T. Imaging of fetal cytomegalovirus infection. Fetal Diagn. Ther. 2011, 29, 117–126. [Google Scholar] [CrossRef] [PubMed]
- Picone, O.; Vauloup-Fellous, C.; Cordier, A.G.; Guitton, S.; Senat, M.V.; Fuchs, F.; Ayoubi, J.M.; Keros, L.G.; Benachi, A. A series of 238 cytomegalovirus primary infections during pregnancy: Description and outcome. Prenat. Diagn. 2013, 33, 751–758. [Google Scholar] [CrossRef] [PubMed]
- Guerra, B.; Simonazzi, G.; Puccetti, C.; Puccetti, C.; Lanari, M.; Farina, A.; Lazzarotto, T.; Rizzo, N. Ultrasound prediction of symptomatic congenital cytomegalovirus infection. Am. J. Obstet. Gynecol. 2008, 198, 380.e1–380.e7. [Google Scholar] [CrossRef] [PubMed]
- Averill, L.W.; Kandula, V.V.; Akyol, Y.; Epelman, M. Fetal brain magnetic resonance imaging findings in congenital cytomegalovirus infection with postnatal imaging correlation. Semin. Ultrasound CT MRI 2015, 36, 476–486. [Google Scholar] [CrossRef] [PubMed]
- Giannattasio, A.; Di Costanzo, P.; De Matteis, A.; Milite, P.; De Martino, D.; Bucci, L.; Augurio, M.R.; Bravaccio, C.; Ferrara, T.; Capasso, L.; et al. Outcomes of congenital cytomegalovirus disease following maternal primary and non-primary infection. J. Clin. Virol. 2017, 96, 32–36. [Google Scholar] [CrossRef] [PubMed]
- Puhakka, L.; Renko, M.; Helminen, M.; Peltola, V.; Heiskanen-Kosma, T.; Lappalainen, M.; Surcel, H.M.; Lonnqvist, T.; Saxen, H. Primary versus non-primary maternal cytomegalovirus infection as a cause of symptomatic congenital infection—Register-based study from Finland. Infect. Dis. 2017, 49, 445–453. [Google Scholar] [CrossRef] [PubMed]
- Manicklal, S.; Emery, V.C.; Lazzarotto, T.; Boppana, S.B.; Gupta, R.K. The “silent” global burden of congenital cytomegalovirus. Clin. Microbiol. Rev. 2013, 26, 86–102. [Google Scholar] [CrossRef] [PubMed]
- Yamada, H.; Tanimura, K.; Tairaku, S.; Morioka, I.; Deguchi, M.; Morizane, M.; Nagamata, S.; Ozaki, K.; Ebina, Y.; Minematsu, T. Clinical factor associated with congenital cytomegalovirus infection in pregnant women with non-primary infection. J. Infect. Chemother. 2018, 24, 702–706. [Google Scholar] [CrossRef] [PubMed]
- Barkai, G.; Ari-Even, R.D.; Barzilai, A.; Tepperberg, O.M.; Mendlson, E.; Hildesheimer, M.; Kuint, J. Universal neonatal cytomegalovirus screening using saliva—Report of clinical experience. J. Clin. Virol. 2014, 60, 361–366. [Google Scholar] [CrossRef] [PubMed]
- Hamprecht, K.; Maschmann, J.; Vochem, M.; Dietz, K.; Speer, C.P.; Jahn, G. Epidemiology of transmission of cytomegalovirus from mother to preterm infant by breastfeeding. Lancet 2001, 357, 513–518. [Google Scholar] [CrossRef]
- Boppana, S.B.; Ross, S.A.; Novak, Z.; Shimamura, M.; Tolan, R.W.; Palmer, A.L.; Ahmed, A.; Michaels, M.G.; Sánchez, P.J.; Bernstein, D.I.; et al. Dried blood spot real-time polymerase chain reaction assays to screen newborns for congenital cytomegalovirus infection. JAMA 2010, 303, 1375–1382. [Google Scholar] [CrossRef] [PubMed]
- Boppana, S.B.; Ross, S.A.; Shimamura, M.; Palmer, A.L.; Ahmed, A.; Michaels, M.G.; Sánchez, P.J.; Bernstein, D.I.; Tolan, R.W., Jr.; Novak, Z.; et al. Saliva polymerase-chain reaction assay for cytomegalovirus screening in newborns. N. Engl. J. Med. 2011, 364, 2111–2118. [Google Scholar] [CrossRef] [PubMed]
- Vancor, E.; Shapiro, E.D. Results of a targeted screening program for congenital cytomegalovirus infection in infants who fail newborn hearing screening. J. Pediatr. Infect. Dis. Soc. 2018. [Google Scholar] [CrossRef] [PubMed]
- Dreher, A.M.; Arora, N.; Fowler, K.B.; Novak, Z.; Britt, W.J.; Boppana, S.B.; Ross, S.A. Spectrum of disease and outcome in children with symptomatic congenital cytomegalovirus infection. J. Pediatr. 2014, 164, 855–859. [Google Scholar] [CrossRef] [PubMed]
- Nigro, G.; Adler, S.P.; La Torre, R.; Best, A.M. Congenital Cytomegalovirus Collaborating, G. Passive immunization during pregnancy for congenital cytomegalovirus infection. N. Engl. J. Med. 2005, 353, 1350–1362. [Google Scholar] [CrossRef] [PubMed]
- Japanese Congenital Cytomegalovirus Infection Immunoglobulin Fetal Therapy Study. A trial of immunoglobulin fetal therapy for symptomatic congenital cytomegalovirus infection. J. Reprod. Immunol. 2012, 915, 73–79. [Google Scholar]
- Leruez-Ville, M.; Ghout, I.; Bussieres, L.; Stirnemann, J.; Magny, J.F.; Couderc, S.; Salomon, L.J.; Guilleminot, T.; Aegerter, P.; Benoist, G.; et al. In utero treatment of congenital cytomegalovirus infection with valacyclovir in a multicenter, open-label, phase II study. Am. J. Obstet. Gynecol. 2016, 215, 462.e1–462.e10. [Google Scholar] [CrossRef] [PubMed]
- Revello, M.G.; Lazzarotto, T.; Guerra, B.; Spinillo, A.; Ferrazzi, E.; Kustermann, A.; Guaschino, S.; Vergani, P.; Todros, T.; Frusca, T.; et al. A randomized trial of hyperimmune globulin to prevent congenital cytomegalovirus. N. Engl. J. Med. 2014, 370, 1316–1326. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tanimura, K.; Yamada, H. Potential Biomarkers for Predicting Congenital Cytomegalovirus Infection. Int. J. Mol. Sci. 2018, 19, 3760. https://doi.org/10.3390/ijms19123760
Tanimura K, Yamada H. Potential Biomarkers for Predicting Congenital Cytomegalovirus Infection. International Journal of Molecular Sciences. 2018; 19(12):3760. https://doi.org/10.3390/ijms19123760
Chicago/Turabian StyleTanimura, Kenji, and Hideto Yamada. 2018. "Potential Biomarkers for Predicting Congenital Cytomegalovirus Infection" International Journal of Molecular Sciences 19, no. 12: 3760. https://doi.org/10.3390/ijms19123760
APA StyleTanimura, K., & Yamada, H. (2018). Potential Biomarkers for Predicting Congenital Cytomegalovirus Infection. International Journal of Molecular Sciences, 19(12), 3760. https://doi.org/10.3390/ijms19123760





