Fish, Fish Oils and Cardioprotection: Promise or Fish Tale?
Abstract
1. Introduction
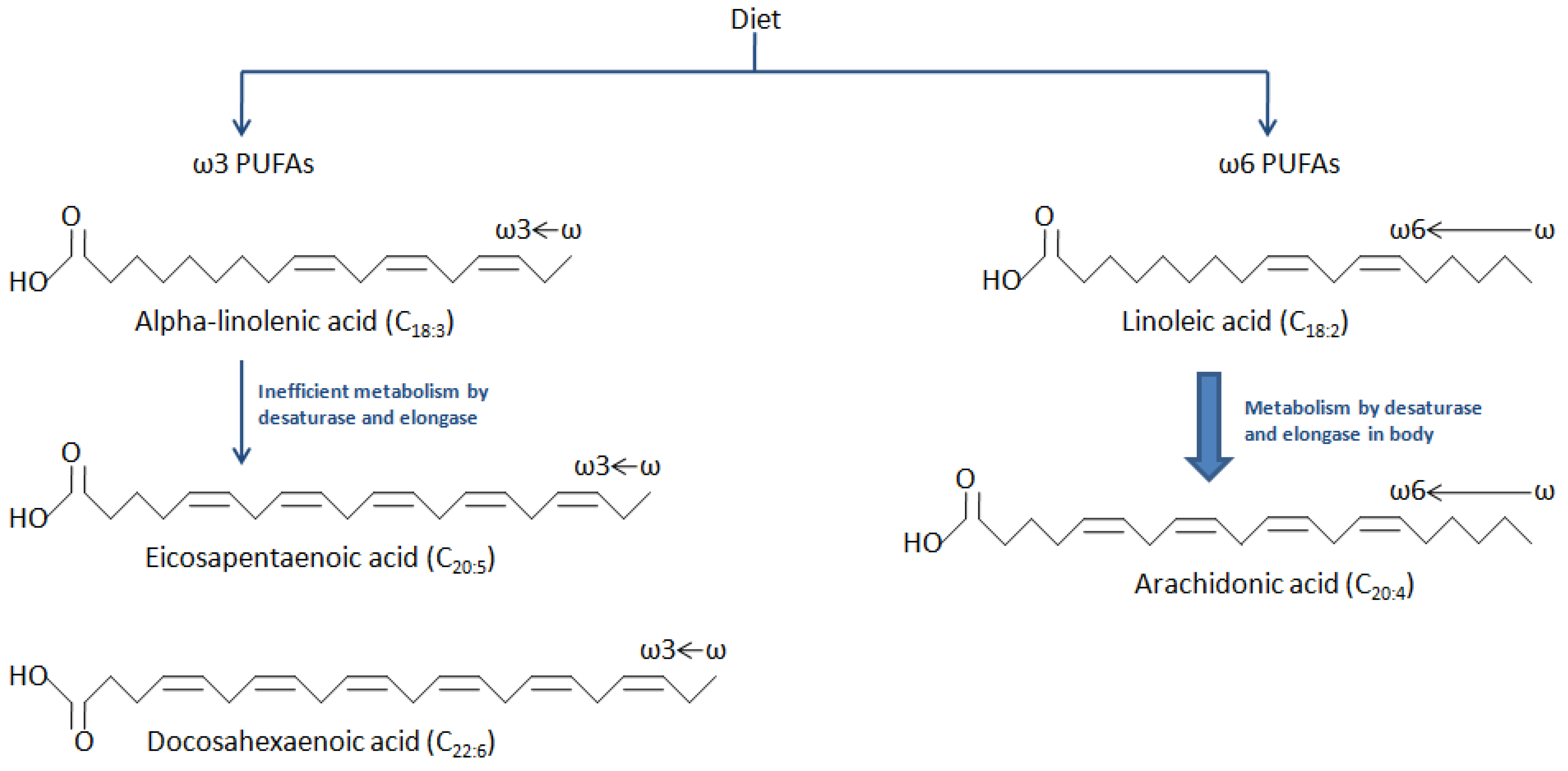
2. Structure and Metabolism of PUFAs
3. Dietary Sources of Major PUFAs
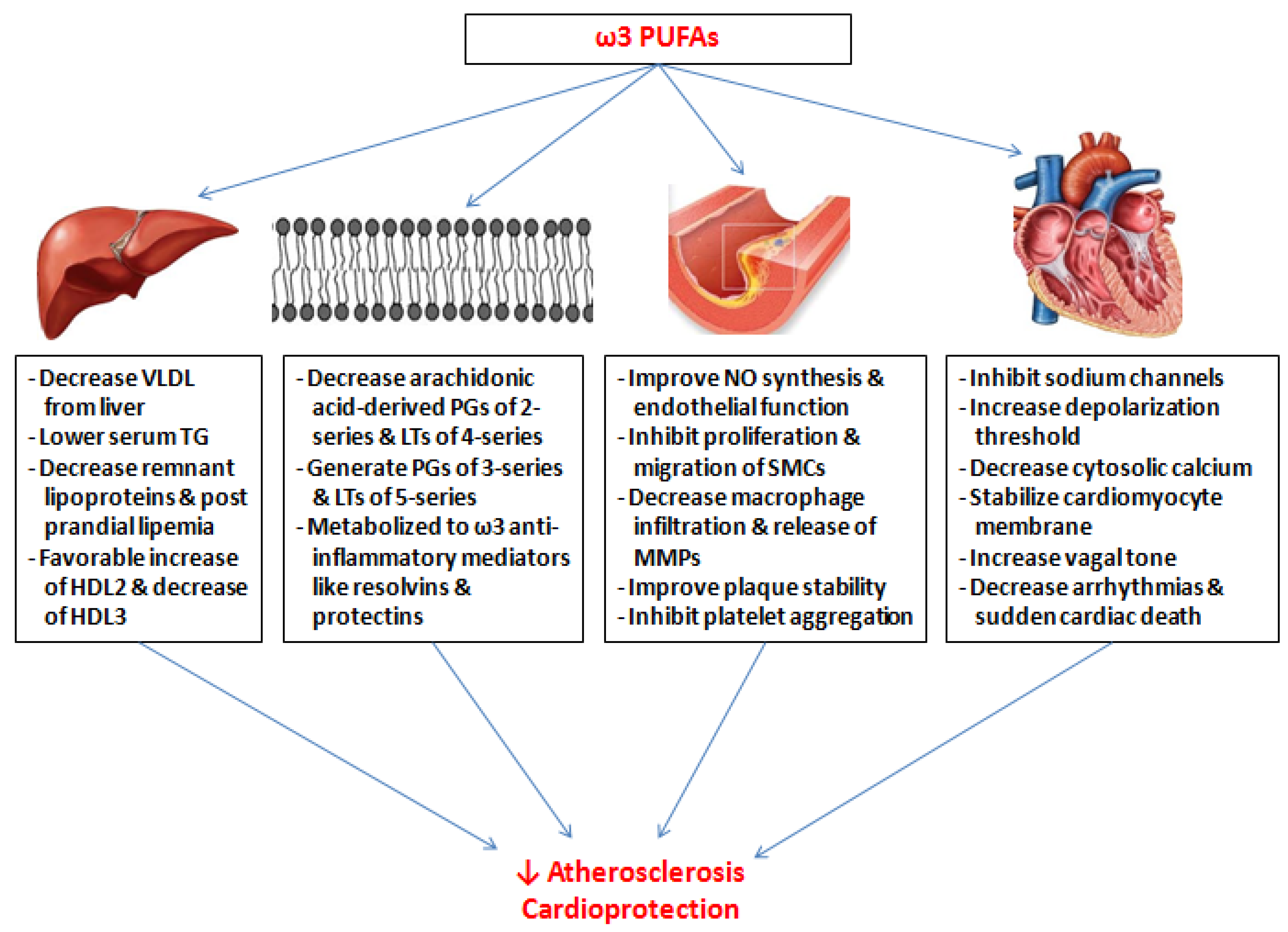
4. Proposed Cardioprotective Benefits of ω-3 PUFAs and Their Molecular Mechanisms of Action
4.1. Anti-Inflammatory Effect
4.2. Improved Endothelial Function
4.3. Atherosclerotic Plaque Stabilization
4.4. Effect on Lipid Metabolism
4.5. Anti-Thrombotic Effect
4.6. Anti-Arrhythmic Effect
4.7. Cardiac Remodeling
4.8. Improved Exercise Tolerance
4.9. Improved Cognitive Function
5. Evidence from Trials and Meta-Analyses
6. Available ω-3 PUFA Formulations
7. Side-Effects and Safety Concerns
8. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Bauer, U.E.; Briss, P.A.; Goodman, R.A.; Bowman, B.A. Prevention of chronic disease in the 21st century: Elimination of the leading preventable causes of premature death and disability in the USA. Lancet 2014, 384, 45–52. [Google Scholar] [CrossRef]
- Bang, H.O.; Dyerberg, J.; Nielsen, A. Plasma lipid and lipoprotein pattern in Greenlandic West-coast Eskimos. Lancet 1971, 297, 1143–1146. [Google Scholar] [CrossRef]
- Bang, H.O.; Dyerberg, J.; Sinclair, H.M. The composition of the Eskimo food in north western Greenland. Am. J. Clin. Nutr. 1980, 33, 2657–2661. [Google Scholar] [CrossRef] [PubMed]
- Burr, M.L.; Gilbert, J.F.; Holliday, R.A.; Elwood, P.C.; Fehily, A.M.; Rogers, S.; Sweetnam, P.M.; Deadman, N.M. Effects of changes in fat, fish, and fibre intakes on death and myocardial reinfarction: Diet and reinfarction trial (DART). Lancet 1989, 334, 757–761. [Google Scholar] [CrossRef]
- Yang, B.; Saldeen, T.G.; Nichols, W.W.; Mehta, J.L. Dietary fish oil supplementation attenuates myocardial dysfunction and injury caused by global ischemia and reperfusion in isolated rat hearts. J. Nutr. 1993, 123, 2067–2074. [Google Scholar] [PubMed]
- GISSI-Prevenzione Investigators. Dietary supplementation with n-3 polyunsaturated fatty acids and vitamin E after myocardial infarction: Results of the GISSI-Prevenzione trial. Lancet 1999, 354, 447–455. [Google Scholar] [CrossRef]
- Whelton, S.P.; He, J.; Whelton, P.K.; Muntner, P. Meta-analysis of observational studies on fish intake and coronary heart disease. Am. J. Cardiol. 2004, 93, 1119–1123. [Google Scholar] [CrossRef] [PubMed]
- He, K.; Song, Y.; Daviglus, M.L.; Liu, K.; Van Horn, L.; Dyer, A.R.; Greenland, P. Accumulated evidence on fish consumption and coronary heart disease mortality: A Meta-analysis of cohort studies. Circulation 2004, 109, 2705–2711. [Google Scholar] [CrossRef] [PubMed]
- Yokoyama, M.; Origasa, H.; Matsuzaki, M.; Matsuzawa, Y.; Saito, Y.; Ishikawa, Y.; Oikawa, S.; Sasaki, J.; Hishida, H.; Itakura, H.; et al. Effects of eicosapentaenoic acid on major coronary events in hypercholesterolaemic patients (JELIS): A randomised open-label, blinded endpoint analysis. Lancet 2007, 369, 1090–1098. [Google Scholar] [CrossRef]
- Tavazzi, L.; Maggioni, A.P.; Marchioli, R.; Barlera, S.; Franzosi, M.G.; Latini, R.; Lucci, D.; Nicolosi, G.L.; Porcu, M.; Tognoni, G. Effect of n-3 polyunsaturated fatty acids in patients with chronic heart failure (the GISSI-HF trial): A randomised, double-blind, placebo-controlled trial. Lancet 2008, 372, 1223–1230. [Google Scholar] [PubMed]
- Kris-Etherton, P.M.; Harris, W.S.; Appel, L.J. Fish consumption, fish oil, omega-3 fatty acids, and cardiovascular disease. Circulation 2002, 106, 2747–2757. [Google Scholar] [CrossRef] [PubMed]
- Clarke, T.C.; Black, L.I.; Stussman, B.J.; Barnes, P.M.; Nahin, R.L. Trends in the Use of Complementary Health Approaches among Adults: United States, 2002–2012; National Health Statistics Reports No. 79; National Center for Health Statistics: Hyattsville, MD, USA, 2015; pp. 1–16.
- Rauch, B.; Schiele, R.; Schneider, S.; Diller, F.; Victor, N.; Gohlke, H.; Gottwik, M.; Steinbeck, G.; Del Castillo, U.; Sack, R.; et al. OMEGA, a randomized, placebo-controlled trial to test the effect of highly purified omega-3 fatty acids on top of modern guideline-adjusted therapy after myocardial infarction. Circulation 2010, 122, 2152–2159. [Google Scholar] [CrossRef] [PubMed]
- Kromhout, D.; Giltay, E.J.; Geleijnse, J.M. n–3 Fatty acids and cardiovascular events after myocardial infarction. N. Engl. J. Med. 2010, 363, 2015–2026. [Google Scholar] [CrossRef] [PubMed]
- Galan, P.; Kesse-Guyot, E.; Czernichow, S.; Briancon, S.; Blacher, J.; Hercberg, S. Effects of B vitamins and omega 3 fatty acids on cardiovascular diseases: A randomised placebo controlled trial. BMJ 2010, 341, c6273. [Google Scholar] [CrossRef] [PubMed]
- ORIGIN Trial Investigators. n–3 Fatty acids and cardiovascular outcomes in patients with dysglycemia. N. Engl. J. Med. 2012, 367, 309–318. [Google Scholar] [CrossRef] [PubMed]
- Risk and Prevention Study Collaborative Group. N–3 fatty acids in patients with multiple cardiovascular risk factors. N. Engl. J. Med. 2013, 368, 1800–1808. [Google Scholar] [CrossRef] [PubMed]
- Bowen, K.J.; Harris, W.S.; Kris-Etherton, P.M. Omega-3 fatty acids and cardiovascular disease: Are there benefits? Curr. Treat. Opt. Cardiovasc. Med. 2016, 18, 69. [Google Scholar] [CrossRef] [PubMed]
- Dennis, E.A.; Norris, P.C. Eicosanoid storm in infection and inflammation. Nat. Rev. Immunol. 2015, 15, 511. [Google Scholar] [CrossRef] [PubMed]
- Saini, R.K.; Keum, Y.S. Omega-3 and omega-6 polyunsaturated fatty acids: Dietary sources, metabolism, and significance—A review. Life Sci. 2018, 203, 255–267. [Google Scholar] [CrossRef] [PubMed]
- Dietary Guidelines Advisory Committee. Scientific Report of the 2015 Dietary Guidelines Advisory Committee; US Department of Health and Human Services: Washington, DC, USA, 2015.
- Burke, M.F.; Burke, F.M.; Soffer, D.E. Review of cardiometabolic effects of prescription omega-3 fatty acids. Curr. Atheroscler. Rep. 2017, 19, 60. [Google Scholar] [CrossRef] [PubMed]
- Kromhout, D.; Yasuda, S.; Geleijnse, J.M.; Shimokawa, H. Fish oil and omega-3 fatty acids in cardiovascular disease: Do they really work? Eur. Heart J. 2011, 33, 436–443. [Google Scholar] [CrossRef] [PubMed]
- Endo, J.; Arita, M. Cardioprotective mechanism of omega-3 polyunsaturated fatty acids. J. Cardiol. 2016, 67, 22–27. [Google Scholar] [CrossRef] [PubMed]
- Yagi, S.; Fukuda, D.; Aihara, K.I.; Akaike, M.; Shimabukuro, M.; Sata, M. N-3 polyunsaturated fatty acids: Promising nutrients for preventing cardiovascular disease. J. Atheroscler. Thromb. 2017, 24, 999–1010. [Google Scholar] [CrossRef] [PubMed]
- Calder, P.C. The role of marine omega-3 (n-fatty acids in inflammatory processes, atherosclerosis and plaque stability. Mol. Nutr. Food Res. 2012, 56, 1073–1080. [Google Scholar] [CrossRef] [PubMed]
- De Winther, M.P.; Kanters, E.; Kraal, G.; Hofker, M.H. Nuclear factor κB signaling in atherogenesis. Arter. Thromb. Vasc. Biol. 2005, 25, 904–914. [Google Scholar] [CrossRef] [PubMed]
- Omura, M.; Kobayashi, S.; Mizukami, Y.; Mogami, K.; Todoroki-Ikeda, N.; Miyake, T.; Matsuzaki, M. Eicosapentaenoic acid (EPA) induces Ca2+-independent activation and translocation of endothelial nitric oxide synthase and endothelium-dependent vasorelaxation. FEBS Lett. 2001, 487, 361–366. [Google Scholar] [CrossRef]
- Ishida, T.; Naoe, S.; Nakakuki, M.; Kawano, H.; Imada, K. Eicosapentaenoic acid prevents saturated fatty acid-induced vascular endothelial dysfunction: Involvement of long-chain acyl-CoA synthetase. J. Atheroscler. Thromb. 2015, 22, 1172–1185. [Google Scholar] [CrossRef] [PubMed]
- Jain, A.P.; Aggarwal, K.K.; Zhang, P.Y. Omega-3 fatty acids and cardiovascular disease. Eur. Rev. Med. Pharmacol. Sci. 2015, 19, 441–445. [Google Scholar] [PubMed]
- Haglund, O.; Mehta, J.L.; Saldeen, T. Effects of fish oil on some parameters of fibrinolysis and lipoprotein(a) in healthy subjects. Am. J. Cardiol. 1994, 74, 189–192. [Google Scholar] [CrossRef]
- Niki, T.; Wakatsuki, T.; Yamaguchi, K.; Taketani, Y.; Oeduka, H.; Kusunose, K.; Ise, T.; Iwase, T.; Yamada, H.; Soeki, T.; et al. Effects of the addition of eicosapentaenoic acid to strong statin therapy on inflammatory cytokines and coronary plaque components assessed by integrated backscatter intravascular ultrasound. Circ. J. 2016, 80, 450–460. [Google Scholar] [CrossRef] [PubMed]
- Thies, F.; Garry, J.M.; Yaqoob, P.; Rerkasem, K.; Williams, J.; Shearman, C.P.; Gallagher, P.J.; Calder, P.C.; Grimble, R.F. Association of n-3 polyunsaturated fatty acids with stability of atherosclerotic plaques: A randomised controlled trial. Lancet 2003, 361, 477–485. [Google Scholar] [CrossRef]
- Swann, P.G.; Venton, D.L.; Le Breton, G.C. Eicosapentaenoic acid and docosahexaenoic acid are antagonists at the thromboxane A2/prostaglandin H2 receptor in human platelets. FEBS Lett. 1989, 243, 244–246. [Google Scholar] [CrossRef]
- Atakisi, O.; Atakisi, E.; Ozcan, A.; Karapehlivan, M.; Kart, A. Protective effect of omega-3 fatty acids on diethylnitrosamine toxicity in rats. Eur. Rev. Med. Pharmacol. Sci. 2013, 17, 467–471. [Google Scholar] [PubMed]
- Christensen, J.H.; Schmidt, E.B. Autonomic nervous system, heart rate variability and n-3 fatty acids. J. Cardiovasc. Med. 2007, 8, 19–22. [Google Scholar] [CrossRef] [PubMed]
- Yagi, S.; Soeki, T.; Aihara, K.I.; Fukuda, D.; Ise, T.; Kadota, M.; Bando, S.; Matsuura, T.; Tobiume, T.; Yamaguchi, K.; et al. Low serum levels of eicosapentaenoic acid and docosahexaenoic acid are risk factors for cardiogenic syncope in patients with Brugada syndrome. Int. Heart J. 2017, 58, 720–723. [Google Scholar] [CrossRef] [PubMed]
- Heydari, B.; Abdullah, S.; Pottala, J.V.; Shah, R.; Abbasi, S.; Mandry, D.; Francis, S.A.; Lumish, H.; Ghoshhajra, B.B.; Hoffmann, U.; et al. Effect of Omega-3 Acid Ethyl Esters on Left Ventricular Remodeling After Acute Myocardial Infarction Clinical Perspective: The OMEGA-REMODEL Randomized Clinical Trial. Circulation 2016, 134, 378–391. [Google Scholar] [CrossRef] [PubMed]
- Endo, J.; Sano, M.; Isobe, Y.; Fukuda, K.; Kang, J.X.; Arai, H.; Arita, M. 18-HEPE, an n-3 fatty acid metabolite released by macrophages, prevents pressure overload–induced maladaptive cardiac remodeling. J. Exp. Med. 2014, 211, 1673–1687. [Google Scholar] [CrossRef] [PubMed]
- Duda, M.K.; O’shea, K.M.; Stanley, W.C. ω-3 polyunsaturated fatty acid supplementation for the treatment of heart failure: Mechanisms and clinical potential. Cardiovasc. Res. 2009, 84, 33–41. [Google Scholar] [CrossRef] [PubMed]
- Da Boit, M.; Hunter, A.M.; Gray, S.R. Fit with good fat? The role of n-3 polyunsaturated fatty acids on exercise performance. Metabolism 2017, 66, 45–54. [Google Scholar] [CrossRef] [PubMed]
- Yagi, S.; Akaike, M.; Ise, T.; Ueda, Y.; Iwase, T.; Sata, M. Renin–angiotensin–aldosterone system has a pivotal role in cognitive impairment. Hypertens. Res. 2013, 36, 753. [Google Scholar] [CrossRef] [PubMed]
- Yagi, S.; Hara, T.; Ueno, R.; Aihara, K.I.; Fukuda, D.; Takashima, A.; Hotchi, J.; Ise, T.; Yamaguchi, K.; Tobiume, T.; et al. Serum concentration of eicosapentaenoic acid is associated with cognitive function in patients with coronary artery disease. Nutr. J. 2014, 13, 112. [Google Scholar] [CrossRef] [PubMed]
- Burr, M.L.; Ashfield-Watt, P.A.; Dunstan, F.D.; Fehily, A.M.; Breay, P.; Ashton, T.; Zotos, P.C.; Haboubi, N.A.; Elwood, P.C. Lack of benefit of dietary advice to men with angina: Results of a controlled trial. Eur. J. Clin. Nutr. 2003, 57, 193. [Google Scholar] [CrossRef] [PubMed]
- Estruch, R.; Ros, E.; Salas-Salvadó, J.; Covas, M.I.; Corella, D.; Arós, F.; Gómez-Gracia, E.; Ruiz-Gutiérrez, V.; Fiol, M.; Lapetra, J.; et al. Primary prevention of cardiovascular disease with a Mediterranean diet. N. Engl. J. Med. 2013, 368, 1279–1290. [Google Scholar] [CrossRef] [PubMed]
- Bonds, D.E.; Harrington, M.; Worrall, B.B.; Bertoni, A.G.; Eaton, C.B.; Hsia, J.; Robinson, J.; Clemons, T.E.; Fine, L.J.; Chew, E.Y. Effect of long-chain ω-3 fatty acids and lutein+ zeaxanthin supplements on cardiovascular outcomes: Results of the Age-Related Eye Disease Study 2 (AREDSrandomized clinical trial. JAMA Intern. Med. 2014, 174, 763–771. [Google Scholar] [CrossRef] [PubMed]
- Rizos, E.C.; Ntzani, E.E.; Bika, E.; Kostapanos, M.S.; Elisaf, M.S. Association between omega-3 fatty acid supplementation and risk of major cardiovascular disease events: A systematic review and meta-analysis. JAMA 2012, 308, 1024–1033. [Google Scholar] [CrossRef] [PubMed]
- Macchia, A.; Grancelli, H.; Varini, S.; Nul, D.; Laffaye, N.; Mariani, J.; Ferrante, D.; Badra, R.; Figal, J.; Ramos, S.; et al. Omega-3 fatty acids for the prevention of recurrent symptomatic atrial fibrillation: Results of the FORWARD (Randomized Trial to Assess Efficacy of PUFA for the Maintenance of Sinus Rhythm in Persistent Atrial Fibrillation) trial. J. Am. Coll. Cardiol. 2013, 61, 463–468. [Google Scholar] [CrossRef] [PubMed]
- Mariani, J.; Doval, H.C.; Nul, D.; Varini, S.; Grancelli, H.; Ferrante, D.; Tognoni, G.; Macchia, A. N-3 polyunsaturated fatty acids to prevent atrial fibrillation: Updated systematic review and meta-analysis of randomized controlled trials. J. Am. Heart Assoc. 2013, 2, e005033. [Google Scholar] [CrossRef] [PubMed]
- The ASCEND Study Collaborative Group. Effects of n-3 fatty acid supplements in diabetes mellitus. N. Engl. J. Med. 2018, 379, 1540–1550. [Google Scholar] [CrossRef] [PubMed]
- A Study of AMR101 to Evaluate Its Ability to Reduce Cardiovascular Events in High Risk Patients with Hypertriglyceridemia and on Statin. 2016. Available online: https://clinicaltrials.gov/ct2/show/NCT01492361 (accessed on 13 October 2018).
- Manson, J.E.; Cook, N.R.; Lee, I.M.; Christen, W.; Bassuk, S.S.; Mora, S.; Gibson, H.; Albert, C.M.; Gordon, D.; Copeland, T.; et al. Marine n− 3 fatty acids and prevention of cardiovascular disease and cancer. N. Engl. J. Med. 2018. [Google Scholar] [CrossRef] [PubMed]
- US National Institutes of Health. Outcomes Study to Assess STatin Residual Risk Reduction With EpaNova in HiGh CV Risk PatienTs With Hypertriglyceridemia (STRENGTH); US National Institutes of Health: Bethesda, MD, USA, 2015.
- Benes, L.B.; Bassi, N.S.; Kalot, M.A.; Davidson, M.H. Evolution of Omega-3 Fatty Acid Therapy and Current and Future Role in the Management of Dyslipidemia. Cardiol. Clin. 2018, 36, 277–285. [Google Scholar] [CrossRef] [PubMed]
- Davidson, M.H.; Johnson, J.; Rooney, M.W.; Kyle, M.L.; Kling, D.F. A novel omega-3 free fatty acid formulation has dramatically improved bioavailability during a low-fat diet compared with omega-3-acid ethyl esters: The ECLIPSE (Epanova® compared to Lovaza® in a pharmacokinetic single-dose evaluation) study. J. Clin. Lipidol. 2012, 6, 573–584. [Google Scholar] [CrossRef] [PubMed]
- Offman, E.; Marenco, T.; Ferber, S.; Johnson, J.; Kling, D.; Curcio, D.; Davidson, M. Steady-state bioavailability of prescription omega-3 on a low-fat diet is significantly improved with a free fatty acid formulation compared with an ethyl ester formulation: The ECLIPSE II study. Vasc. Health Risk Manag. 2013, 9, 563. [Google Scholar] [CrossRef] [PubMed]
| PUFA | Dietary Source |
|---|---|
| Linoleic acid (ω-6) | Corn, safflower, soybean, sunflower oils |
| Alpha-linolenic acid (plant-derived ω-3) | Flaxseed oil, canola (rapeseed) oil, walnuts, seeds of chia, perilla, green leafy vegetables |
| Eicosapentaenoic and Docosahexaenoic acids (marine-derived ω-3) | Fish, fish oil, other seafood, beef, lamb, ω-3 fortified foods |
| Study | Study Design | Number of Patients | Intervention | Follow-Up | Outcome |
|---|---|---|---|---|---|
| DART [4] 1989 | Secondary prevention RCT | 2033 | 200–400 g fish per week | 2 years | 29% reduction in mortality |
| GISSI Prevenzione [6] 1999 | Secondary prevention RCT | 11,324 | 882 mg EPA and DHA daily | 3.5 years | 15–20% reduction in mortality and CV events |
| DART-2 [44] 2003 | Secondary prevention RCT | 3114 | 2 fish servings per week or 3 fish oil capsules daily | 3-9 years | Higher cardiac mortality and SCD |
| JELIS [9] 2007 | Primary and secondary prevention RCT | 18,645 | 1.8 g EPA daily | 4.6 years | 19% reduction in coronary events in CAD patients, no benefit in primary prevention |
| GISSI-HF [10] 2008 | Secondary prevention RCT | 6975 | 840 mg EPA and DHA daily | 3.9 years | 9% reduction in mortality and 8% reduction in hospitalizations |
| Alpha Omega [14] 2010 | Secondary prevention RCT | 4837 | 226 mg EPA and 150 mg DHA daily | 3.4 years | No benefit |
| OMEGA [13] 2010 | Secondary prevention RCT | 3851 | 460 mg EPA and 380 mg DHA daily (vs olive oil control) | 1 year | No benefit |
| SU.FOL.OM3 [15] 2010 | Secondary prevention RCT | 2501 | 600 mg EPA and DHA daily | 4.7 years | No benefit |
| ORIGIN [16] 2012 | Secondary prevention RCT | 12,536 | 465 mg EPA and 375 mg DHA daily (vs olive oil control) | 6.2 years | No benefit |
| Rizos et al. [47] 2012 | Meta-analysis of 20 RCTs | 68,680 | 1000 mg EPA and DHA daily (median) | - | No benefit |
| Risk and Prevention [17] 2013 | Primary prevention RCT | 12,513 | 850 mg of EPA and DHA daily (vs olive oil control) | 5 years | No benefit |
| AREDS2 [46] 2014 | Primary prevention RCT | 4203 | 650 mg EPA plus 350 mg DHA daily | 4.8 years | No benefit |
| ASCEND [50] 2018 | Primary prevention RCT | 15,480 | 1 g ω-3 PUFA daily (vs olive oil control) | 7.4 years | No benefit |
| REDUCE-IT [51] 2018 | Primary prevention RCT | 8179 | 4 g ethyl EPA daily | 4.9 years | 25% reduction in major CV events |
| VITAL [52] 2018 | Primary prevention RCT | 25,871 | 1 g ω-3 PUFA daily | 5.3 years | No benefit |
| Ethyl Esters of EPA and DHA | Ethyl Esters of EPA Only | Free Fatty Acids of EPA and DHA | |
|---|---|---|---|
| Brand name | Lovaza® (GlaxoSmithKline) | Vascepa® (Amarin Pharmaceuticals) | Epanova® (AstraZeneca) |
| Approval date | 2004 | 2012 | 2014 |
| EPA/DHA (g per capsule) | EPA 0.465 g DHA0.375 g | EPA 1 g | EPA 0.550 g DHA 0.200 g |
| Dosing | 2 g (2 capsules) twice daily or 4 g (4 capsules)once daily WITH MEALS | 2 g (2 capsules) twice daily WITH MEALS | 2 g (2 capsules)or 4 g (4 capsules) once daily WITH OR WITHOUT MEALS |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Goel, A.; Pothineni, N.V.; Singhal, M.; Paydak, H.; Saldeen, T.; Mehta, J.L. Fish, Fish Oils and Cardioprotection: Promise or Fish Tale? Int. J. Mol. Sci. 2018, 19, 3703. https://doi.org/10.3390/ijms19123703
Goel A, Pothineni NV, Singhal M, Paydak H, Saldeen T, Mehta JL. Fish, Fish Oils and Cardioprotection: Promise or Fish Tale? International Journal of Molecular Sciences. 2018; 19(12):3703. https://doi.org/10.3390/ijms19123703
Chicago/Turabian StyleGoel, Akshay, Naga Venkata Pothineni, Mayank Singhal, Hakan Paydak, Tom Saldeen, and Jawahar L. Mehta. 2018. "Fish, Fish Oils and Cardioprotection: Promise or Fish Tale?" International Journal of Molecular Sciences 19, no. 12: 3703. https://doi.org/10.3390/ijms19123703
APA StyleGoel, A., Pothineni, N. V., Singhal, M., Paydak, H., Saldeen, T., & Mehta, J. L. (2018). Fish, Fish Oils and Cardioprotection: Promise or Fish Tale? International Journal of Molecular Sciences, 19(12), 3703. https://doi.org/10.3390/ijms19123703






