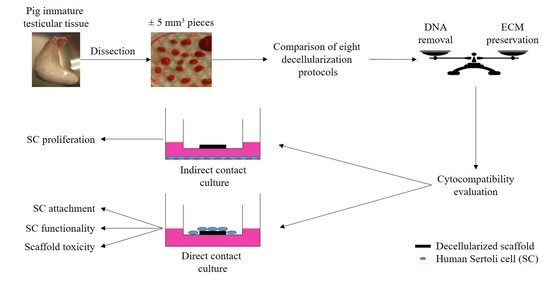
Development of a Cytocompatible Scaffold from Pig Immature Testicular Tissue Allowing Human Sertoli Cell Attachment, Proliferation and Functionality
Abstract
:1. Introduction
2. Results
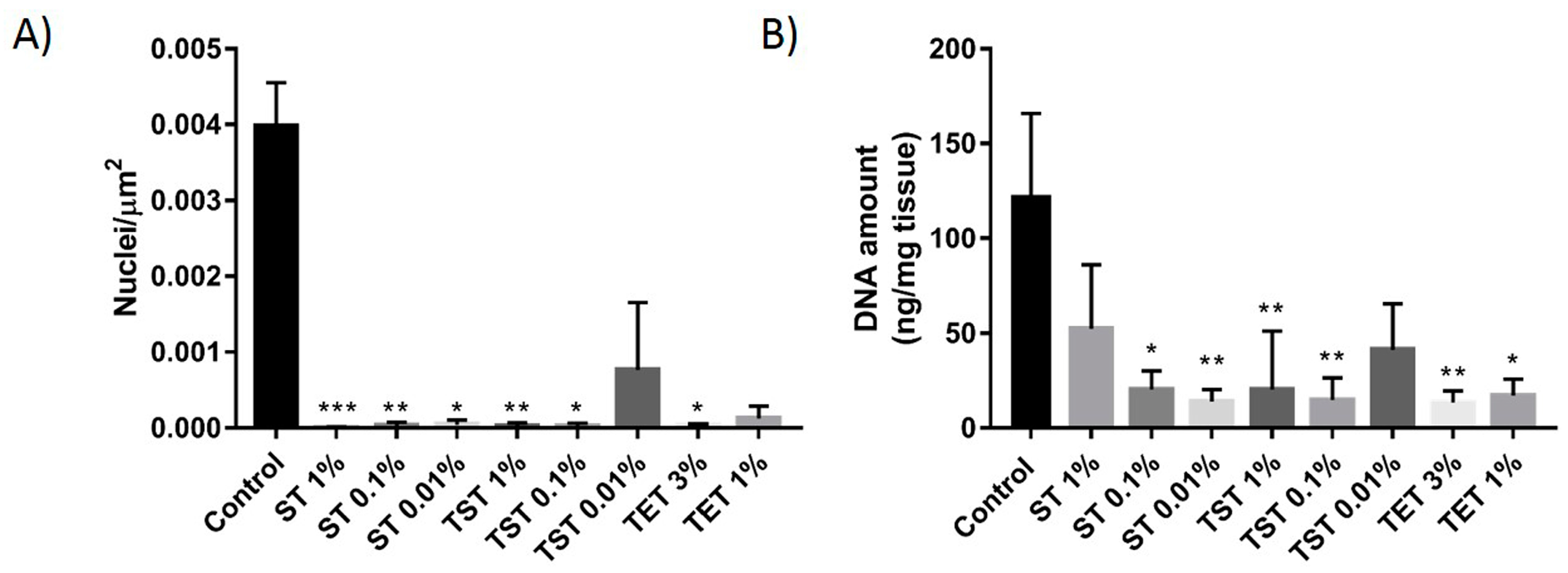
2.1. DNA Content Evaluation
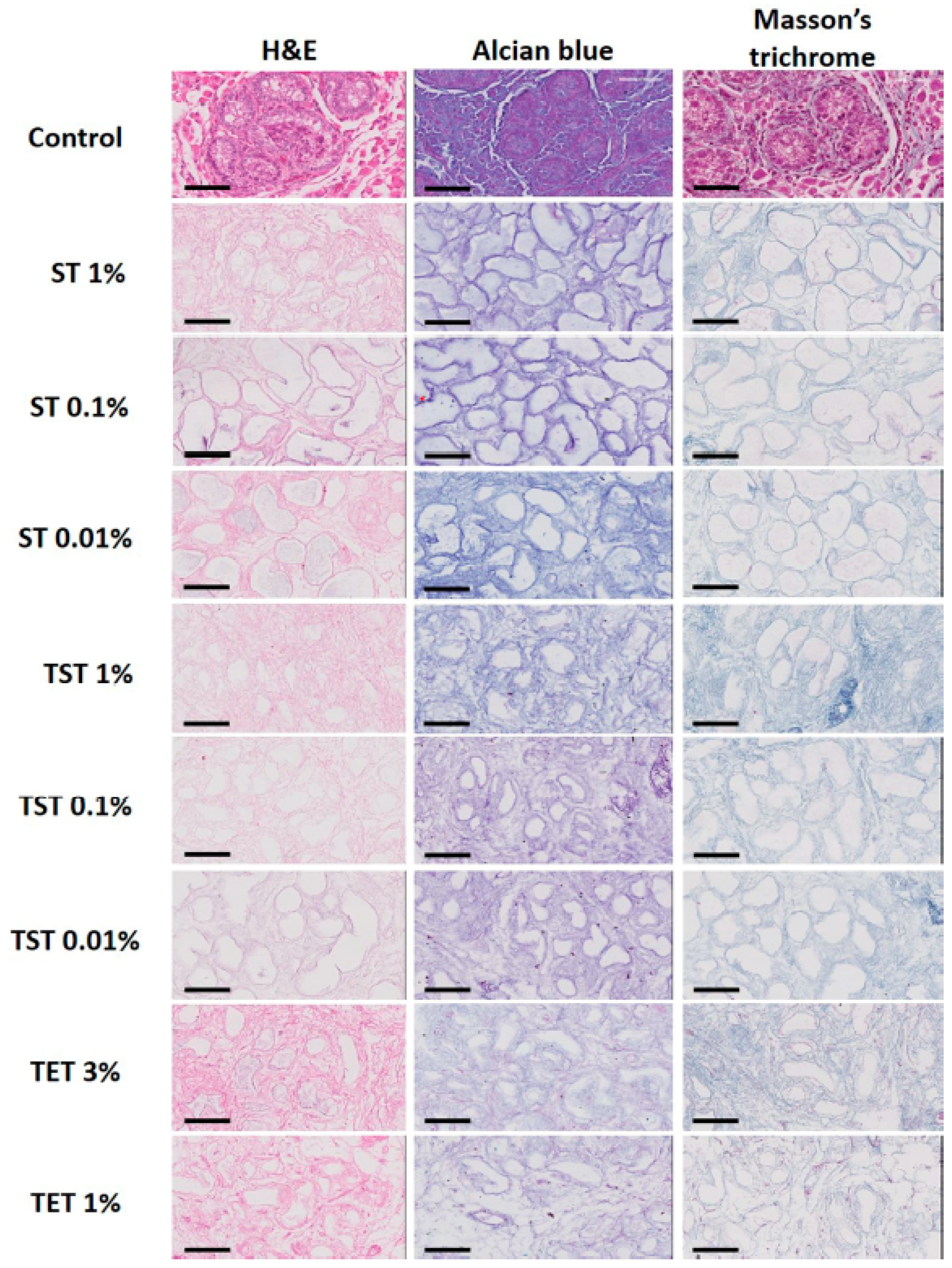
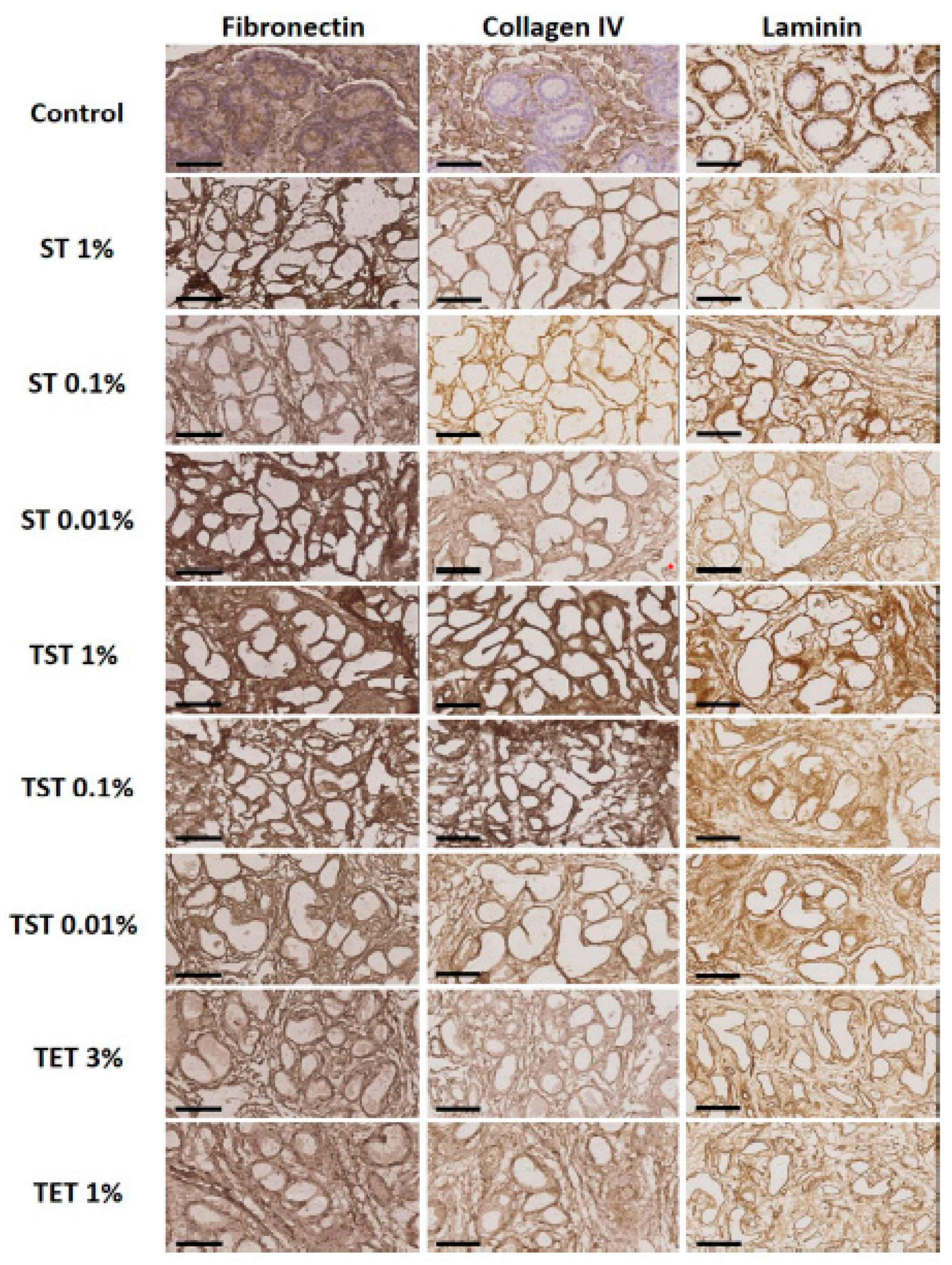
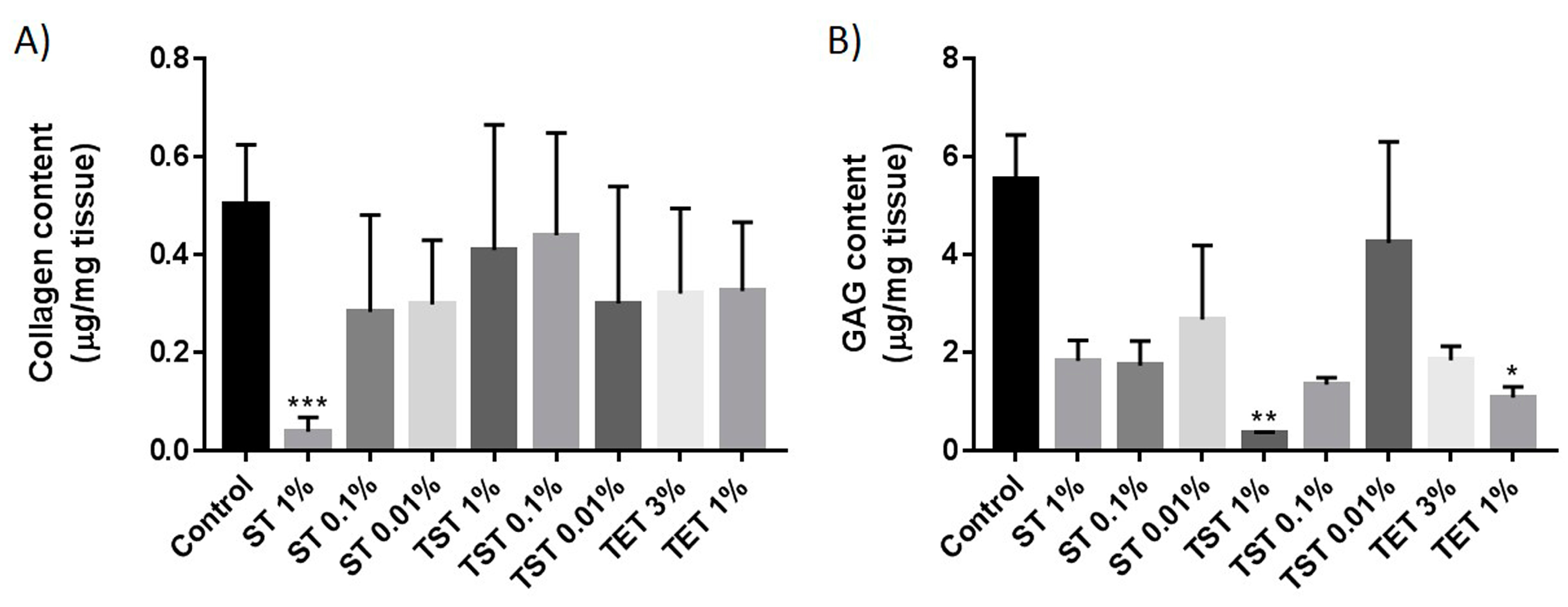
2.2. Composition of the Testicular ECM
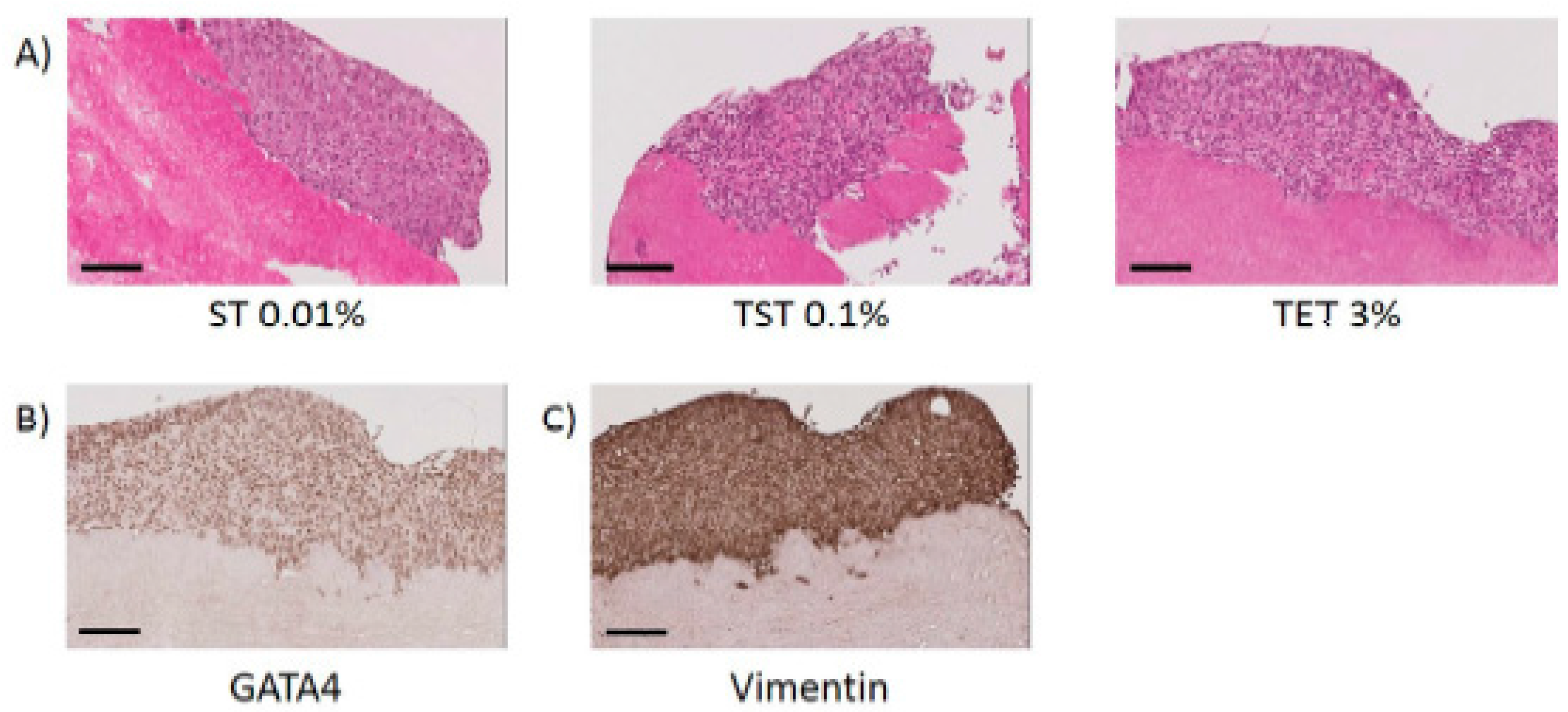
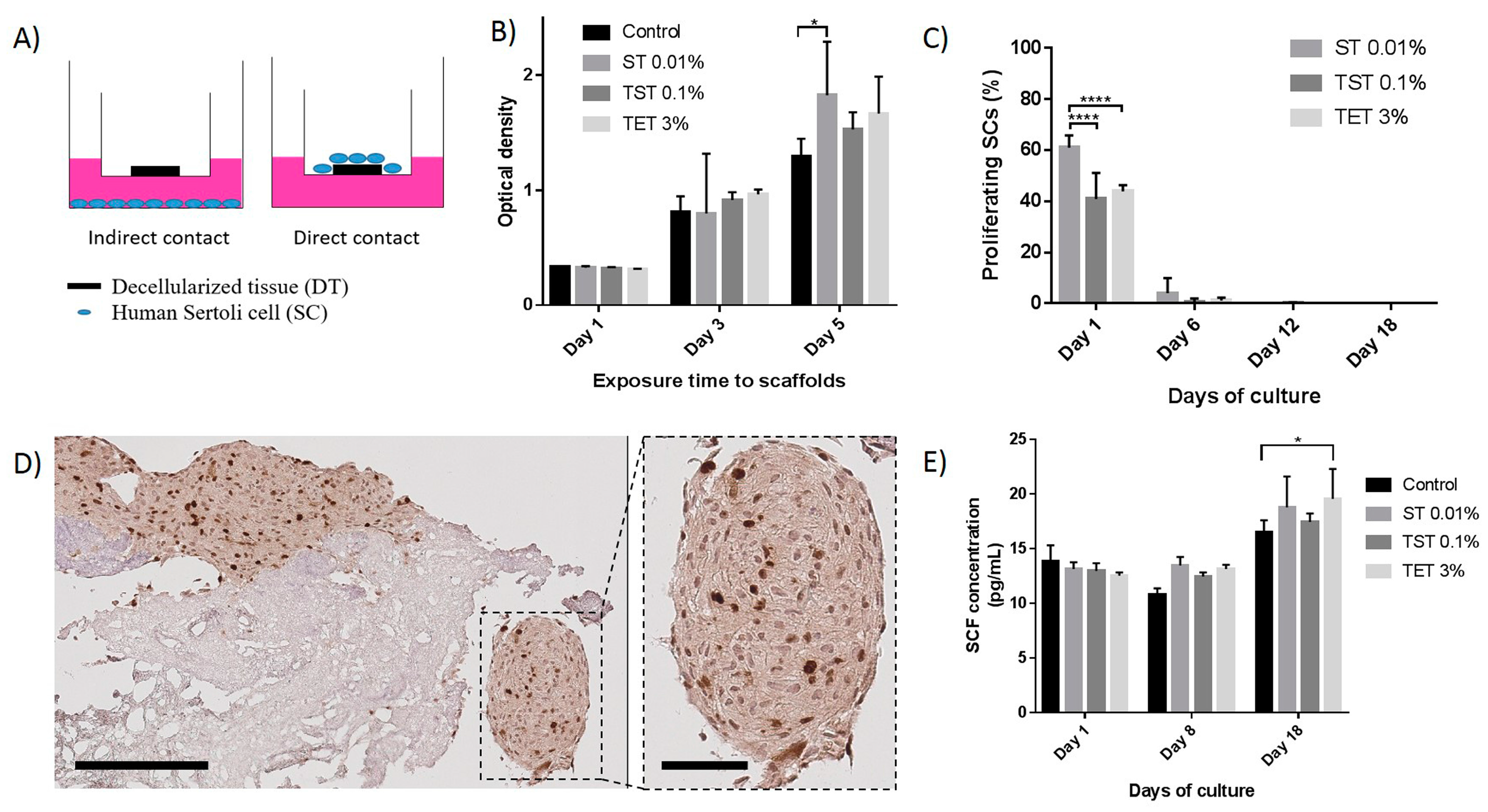
2.3. Scaffold Cytocompatibility
3. Discussion
4. Materials and Methods
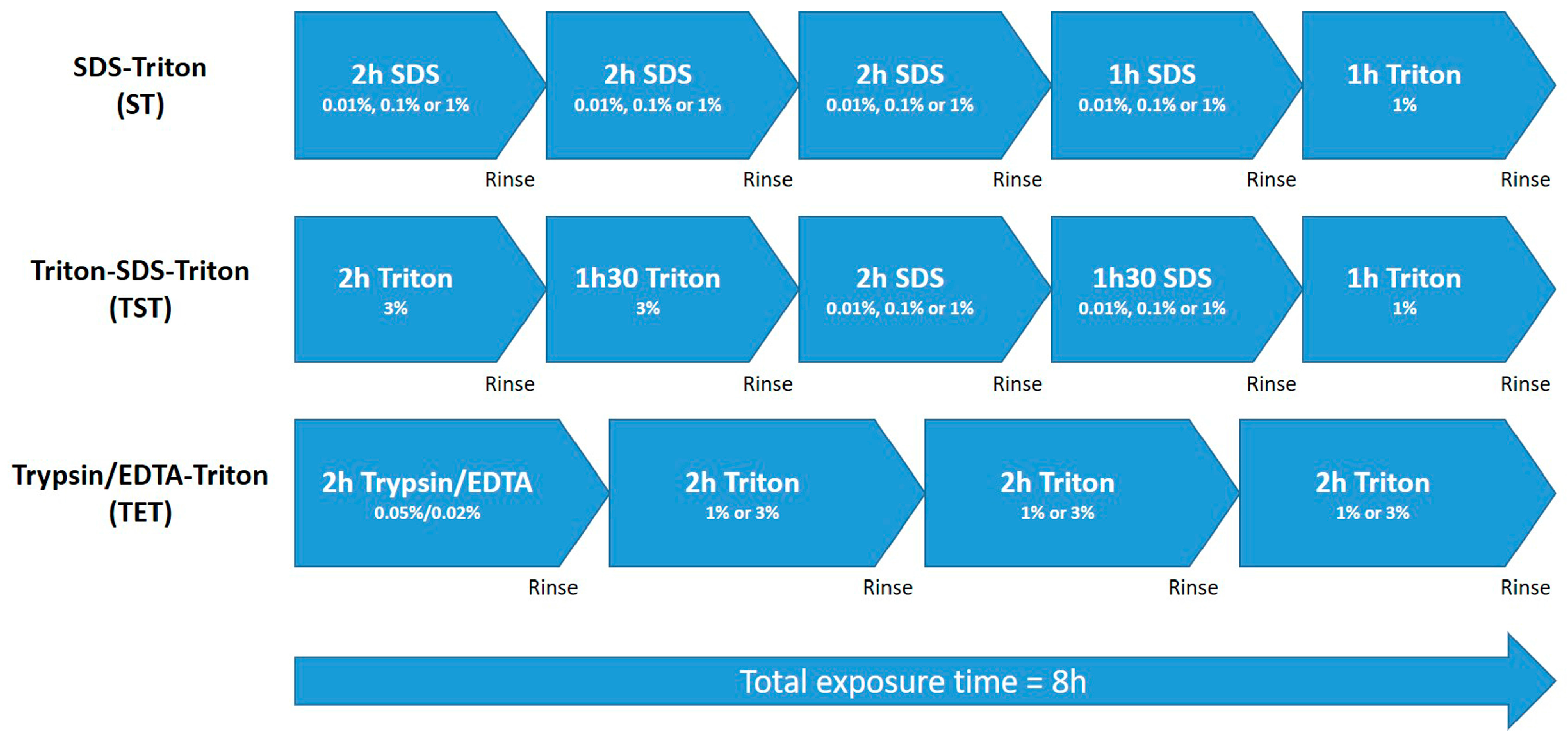
4.1. Decellularization of Pig Testicular Tissue
4.2. Histology
4.3. Assessment of Nuclear Material Content
4.4. Immunohistochemistry
4.5. Glycosaminoglycan Quantification
4.6. Collagen Quantification
4.7. Cell Culture
4.8. SC Proliferation in Indirect Contact with DTs
4.9. Scaffold Preparation and Culture
4.10. SC Attachment, Proliferation, Functionality and Viability on Scaffolds
4.11. Statistical Analyses
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| α-gal KO | alpha 1,3 galactosyltransferase knockout |
| BSA | bovine serum albumin |
| DNA | deoxyribonucleic acid |
| DT | decellularized tissue |
| ECM | extracellular matrix |
| EDTA | ethylenediaminetetraacetic acid |
| ELISA | enzyme-linked immunosorbent assay |
| GAG | glycosaminoglycan |
| H and E | hematoxylin and eosin |
| HEPES | 4-(2-hydroxyethyl) piperazine-1-ethanesulfonic acid |
| HRP | horseradish peroxidase |
| IHC | immunohistochemistry |
| ITT | immature testicular tissue |
| LDH | lactate dehydrogenase |
| NGS | normal goat serum |
| PBS | phosphate buffer saline |
| SC | sertoli cell |
| SCF | stem cell factor |
| SDS | sodium dodecyl sulfate |
| SSC | spermatogonial stem cell |
| ST | SDS-Triton |
| TET | Trypsin/EDTA-Triton |
| TST | Triton-SDS-Triton |
References
- Gatta, G.; Botta, L.; Rossi, S.; Aareleid, T.; Bielska-Lasota, M.; Clavel, J.; Dimitrova, N.; Jakab, Z.; Kaatsch, P.; Lacour, B.; et al. Childhood cancer survival in Europe 1999–2007: Results of eurocare-5—A population-based study. Lancet Oncol. 2014, 15, 35–47. [Google Scholar] [CrossRef]
- Wyns, C.; Curaba, M.; Petit, S.; Vanabelle, B.; Laurent, P.; Wese, J.F.; Donnez, J. Management of fertility preservation in prepubertal patients: 5 years’ experience at the catholic university of louvain. Hum. Reprod. 2011, 26, 737–747. [Google Scholar] [CrossRef] [PubMed]
- Wyns, C.; Collienne, C.; Shenfield, F.; Robert, A.; Laurent, P.; Roegiers, L.; Brichard, B. Fertility preservation in the male pediatric population: Factors influencing the decision of parents and children. Hum. Reprod. 2015, 30, 2022–2030. [Google Scholar] [CrossRef] [PubMed]
- Sato, T.; Katagiri, K.; Gohbara, A.; Inoue, K.; Ogonuki, N.; Ogura, A.; Kubota, Y.; Ogawa, T. In vitro production of functional sperm in cultured neonatal mouse testes. Nature 2011, 471, 504–507. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Nie, Y.H.; Zhang, C.C.; Cai, Y.J.; Wang, Y.; Lu, H.P.; Li, Y.Z.; Cheng, C.; Qiu, Z.L.; Sun, Q. Generation of macaques with sperm derived from juvenile monkey testicular xenografts. Cell Res. 2016, 26, 139–142. [Google Scholar] [CrossRef] [PubMed]
- Brinster, R.L.; Avarbock, M.R. Germline transmission of donor haplotype following spermatogonial transplantation. Proc. Natl. Acad. Sci. USA 1994, 91, 11303–11307. [Google Scholar] [CrossRef] [PubMed]
- Hamra, F.K.; Gatlin, J.; Chapman, K.M.; Grellhesl, D.M.; Garcia, J.V.; Hammer, R.E.; Garbers, D.L. Production of transgenic rats by lentiviral transduction of male germ-line stem cells. Proc. Natl. Acad. Sci. USA 2002, 99, 14931–14936. [Google Scholar] [CrossRef] [PubMed]
- Honaramooz, A.; Behboodi, E.; Megee, S.O.; Overton, S.A.; Galantino-Homer, H.; Echelard, Y.; Dobrinski, I. Fertility and germline transmission of donor haplotype following germ cell transplantation in immunocompetent goats. Biol. Reprod. 2003, 69, 1260–1264. [Google Scholar] [CrossRef] [PubMed]
- Trefil, P.; Micakova, A.; Mucksova, J.; Hejnar, J.; Poplstein, M.; Bakst, M.R.; Kalina, J.; Brillard, J.P. Restoration of spermatogenesis and male fertility by transplantation of dispersed testicular cells in the chicken. Biol. Reprod. 2006, 75, 575–581. [Google Scholar] [CrossRef] [PubMed]
- Herrid, M.; Olejnik, J.; Jackson, M.; Suchowerska, N.; Stockwell, S.; Davey, R.; Hutton, K.; Hope, S.; Hill, J.R. Irradiation enhances the efficiency of testicular germ cell transplantation in sheep. Biol. Reprod. 2009, 81, 898–905. [Google Scholar] [CrossRef] [PubMed]
- Kawasaki, T.; Saito, K.; Sakai, C.; Shinya, M.; Sakai, N. Production of zebrafish offspring from cultured spermatogonial stem cells. Genes Cells 2012, 17, 316–325. [Google Scholar] [CrossRef] [PubMed]
- Hermann, B.P.; Sukhwani, M.; Winkler, F.; Pascarella, J.N.; Peters, K.A.; Sheng, Y.; Valli, H.; Rodriguez, M.; Ezzelarab, M.; Dargo, G.; et al. Spermatogonial stem cell transplantation into rhesus testes regenerates spermatogenesis producing functional sperm. Cell Stem Cell 2012, 11, 715–726. [Google Scholar] [CrossRef] [PubMed]
- Picton, H.M.; Wyns, C.; Anderson, R.A.; Goossens, E.; Jahnukainen, K.; Kliesch, S.; Mitchell, R.T.; Pennings, G.; Rives, N.; Tournaye, H.; et al. A european perspective on testicular tissue cryopreservation for fertility preservation in prepubertal and adolescent boys. Hum. Reprod. 2015, 30, 2463–2475. [Google Scholar] [CrossRef] [PubMed]
- Ginsberg, J.P.; Carlson, C.A.; Lin, K.; Hobbie, W.L.; Wigo, E.; Wu, X.; Brinster, R.L.; Kolon, T.F. An experimental protocol for fertility preservation in prepubertal boys recently diagnosed with cancer: A report of acceptability and safety. Hum. Reprod. 2010, 25, 37–41. [Google Scholar] [CrossRef] [PubMed]
- Ginsberg, J.P.; Li, Y.; Carlson, C.A.; Gracia, C.R.; Hobbie, W.L.; Miller, V.A.; Mulhall, J.; Shnorhavorian, M.; Brinster, R.L.; Kolon, T.F. Testicular tissue cryopreservation in prepubertal male children: An analysis of parental decision-making. Pediatr. Blood Cancer 2014, 61, 1673–1678. [Google Scholar] [CrossRef] [PubMed]
- Davidoff, M.S.; Breucker, H.; Holstein, A.F.; Seidl, K. Cellular architecture of the lamina propria of human seminiferous tubules. Cell Tissue Res. 1990, 262, 253–261. [Google Scholar] [CrossRef] [PubMed]
- Kerr, J.; Loveland, K.; O’Bryan, M.; de Kretser, D. Cytology of the testis and intrinsic control mechanisms. In Knobil and Neill’s Physiology of Reproduction; Elsevier: Amsterdam, The Netherlands, 2006; Volume 1, pp. 827–947. [Google Scholar]
- Skinner, M.K.; Tung, P.S.; Fritz, I.B. Cooperativity between sertoli cells and testicular peritubular cells in the production and deposition of extracellular matrix components. J. Cell Biol. 1985, 100, 1941–1947. [Google Scholar] [CrossRef] [PubMed]
- Tung, P.S.; Skinner, M.K.; Fritz, I.B. Cooperativity between sertoli cells and peritubular myoid cells in the formation of the basal lamina in the seminiferous tubule. Ann. N. Y. Acad. Sci. 1984, 438, 435–446. [Google Scholar] [CrossRef] [PubMed]
- Tung, P.S.; Fritz, I.B. Morphogenetic restructuring and formation of basement membranes by sertoli cells and testis peritubular cells in co-culture: Inhibition of the morphogenetic cascade by cyclic amp derivatives and by blocking direct cell contact. Dev. Biol. 1987, 120, 139–153. [Google Scholar] [CrossRef]
- Hadley, M.A.; Byers, S.W.; Suarez-Quian, C.A.; Kleinman, H.K.; Dym, M. Extracellular matrix regulates sertoli cell differentiation, testicular cord formation, and germ cell development in vitro. J. Cell Biol. 1985, 101, 1511–1522. [Google Scholar] [CrossRef] [PubMed]
- Cheng, C.Y.; Wong, E.W.; Yan, H.H.; Mruk, D.D. Regulation of spermatogenesis in the microenvironment of the seminiferous epithelium: New insights and advances. Mol. Cell. Endocrinol. 2010, 315, 49–56. [Google Scholar] [CrossRef] [PubMed]
- Pollanen, P.P.; Kallajoki, M.; Risteli, L.; Risteli, J.; Suominen, J.J. Laminin and type iv collagen in the human testis. Int. J. Androl. 1985, 8, 337–347. [Google Scholar] [CrossRef] [PubMed]
- Santamaria, L.; Martinez-Onsurbe, P.; Paniagua, R.; Nistal, M. Laminin, type iv collagen, and fibronectin in normal and cryptorchid human testes. An immunohistochemical study. Int. J. Androl. 1990, 13, 135–146. [Google Scholar] [CrossRef] [PubMed]
- Hussein, K.H.; Park, K.M.; Kang, K.S.; Woo, H.M. Biocompatibility evaluation of tissue-engineered decellularized scaffolds for biomedical application. Mater. Sci. Eng. C Mater. Biol. Appl. 2016, 67, 766–778. [Google Scholar] [CrossRef] [PubMed]
- Crapo, P.M.; Gilbert, T.W.; Badylak, S.F. An overview of tissue and whole organ decellularization processes. Biomaterials 2011, 32, 3233–3243. [Google Scholar] [CrossRef] [PubMed]
- Brown, B.N.; Badylak, S.F. Extracellular matrix as an inductive scaffold for functional tissue reconstruction. Transl. Res. 2014, 163, 268–285. [Google Scholar] [CrossRef] [PubMed]
- Baert, Y.; Stukenborg, J.B.; Landreh, M.; De Kock, J.; Jornvall, H.; Soder, O.; Goossens, E. Derivation and characterization of a cytocompatible scaffold from human testis. Hum. Reprod. 2015, 30, 256–267. [Google Scholar] [CrossRef] [PubMed]
- Baert, Y.; De Kock, J.; Alves-Lopes, J.P.; Soder, O.; Stukenborg, J.B.; Goossens, E. Primary human testicular cells self-organize into organoids with testicular properties. Stem Cell Rep. 2016, 8, 30–38. [Google Scholar] [CrossRef] [PubMed]
- Parmaksiz, M.; Dogan, A.; Odabas, S.; Elcin, A.E.; Elcin, Y.M. Clinical applications of decellularized extracellular matrices for tissue engineering and regenerative medicine. Biomed. Mater. 2016, 11, 022003. [Google Scholar] [CrossRef] [PubMed]
- Liao, J.; Joyce, E.M.; Sacks, M.S. Effects of decellularization on the mechanical and structural properties of the porcine aortic valve leaflet. Biomaterials 2008, 29, 1065–1074. [Google Scholar] [CrossRef] [PubMed]
- Gilbert, T.W.; Sellaro, T.L.; Badylak, S.F. Decellularization of tissues and organs. Biomaterials 2006, 27, 3675–3683. [Google Scholar] [CrossRef] [PubMed]
- Gilpin, S.E.; Guyette, J.P.; Gonzalez, G.; Ren, X.; Asara, J.M.; Mathisen, D.J.; Vacanti, J.P.; Ott, H.C. Perfusion decellularization of human and porcine lungs: Bringing the matrix to clinical scale. J. Heart Lung Trans. 2014, 33, 298–308. [Google Scholar] [CrossRef] [PubMed]
- Hadley, M.A.; Weeks, B.S.; Kleinman, H.K.; Dym, M. Laminin promotes formation of cord-like structures by sertoli cells in vitro. Dev. Biol. 1990, 140, 318–327. [Google Scholar] [CrossRef]
- van der Wee, K.; Hofmann, M.C. An in vitro tubule assay identifies hgf as a morphogen for the formation of seminiferous tubules in the postnatal mouse testis. Exp. Cell Res. 1999, 252, 175–185. [Google Scholar] [CrossRef] [PubMed]
- Chui, K.; Trivedi, A.; Cheng, C.Y.; Cherbavaz, D.B.; Dazin, P.F.; Huynh, A.L.; Mitchell, J.B.; Rabinovich, G.A.; Noble-Haeusslein, L.J.; John, C.M. Characterization and functionality of proliferative human sertoli cells. Cell Trans. 2011, 20, 619–635. [Google Scholar] [CrossRef] [PubMed]
- Ren, H.; Shi, X.; Tao, L.; Xiao, J.; Han, B.; Zhang, Y.; Yuan, X.; Ding, Y. Evaluation of two decellularization methods in the development of a whole-organ decellularized rat liver scaffold. Liver Int. 2013, 33, 448–458. [Google Scholar] [CrossRef] [PubMed]
- Oberwallner, B.; Brodarac, A.; Choi, Y.H.; Saric, T.; Anic, P.; Morawietz, L.; Stamm, C. Preparation of cardiac extracellular matrix scaffolds by decellularization of human myocardium. J. Biomed. Mater. Res. A 2014, 102, 3263–3272. [Google Scholar] [CrossRef] [PubMed]
- Svingen, T.; Koopman, P. Building the mammalian testis: Origins, differentiation, and assembly of the component cell populations. Genes Dev. 2013, 27, 2409–2426. [Google Scholar] [CrossRef] [PubMed]
- Enders, G.C.; Henson, J.H.; Millette, C.F. Sertoli cell binding to isolated testicular basement membrane. J. Cell Biol. 1986, 103, 1109–1119. [Google Scholar] [CrossRef] [PubMed]
- Mohiuddin, M.M.; Singh, A.K.; Corcoran, P.C.; Hoyt, R.F.; Thomas, M.L., 3rd; Ayares, D.; Horvath, K.A. Genetically engineered pigs and target-specific immunomodulation provide significant graft survival and hope for clinical cardiac xenotransplantation. J. Thorac. Cardiovasc. Surg. 2014, 148, 1106–1113. [Google Scholar] [CrossRef] [PubMed]
- Platz, J.; Bonenfant, N.R.; Uhl, F.E.; Coffey, A.L.; McKnight, T.; Parsons, C.; Sokocevic, D.; Borg, Z.D.; Lam, Y.W.; Deng, B.; et al. Comparative decellularization and recellularization of wild-type and alpha 1,3 galactosyltransferase knockout pig lungs: A model for ex vivo xenogeneic lung bioengineering and transplantation. Tissue Eng. Part C Methods 2016, 22, 725–739. [Google Scholar] [CrossRef] [PubMed]
- Schlatt, S.; de Kretser, D.M.; Loveland, K.L. Discriminative analysis of rat sertoli and peritubular cells and their proliferation in vitro: Evidence for follicle-stimulating hormone-mediated contact inhibition of sertoli cell mitosis. Biol. Reprod. 1996, 55, 227–235. [Google Scholar] [CrossRef] [PubMed]
- Feng, L.X.; Ravindranath, N.; Dym, M. Stem cell factor/c-kit up-regulates cyclin d3 and promotes cell cycle progression via the phosphoinositide 3-kinase/p70 s6 kinase pathway in spermatogonia. J. Biol. Chem. 2000, 275, 25572–25576. [Google Scholar] [CrossRef] [PubMed]
- Pendergraft, S.S.; Sadri-Ardekani, H.; Atala, A.; Bishop, C.E. Three-dimensional testicular organoid: A novel tool for the study of human spermatogenesis and gonadotoxicity in vitro. Biol. Reprod. 2017, 96, 720–732. [Google Scholar] [CrossRef] [PubMed]
- Alves-Lopes, J.P.; Soder, O.; Stukenborg, J.B. Testicular organoid generation by a novel in vitro three-layer gradient system. Biomaterials 2017, 130, 76–89. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vermeulen, M.; Del Vento, F.; De Michele, F.; Poels, J.; Wyns, C. Development of a Cytocompatible Scaffold from Pig Immature Testicular Tissue Allowing Human Sertoli Cell Attachment, Proliferation and Functionality. Int. J. Mol. Sci. 2018, 19, 227. https://doi.org/10.3390/ijms19010227
Vermeulen M, Del Vento F, De Michele F, Poels J, Wyns C. Development of a Cytocompatible Scaffold from Pig Immature Testicular Tissue Allowing Human Sertoli Cell Attachment, Proliferation and Functionality. International Journal of Molecular Sciences. 2018; 19(1):227. https://doi.org/10.3390/ijms19010227
Chicago/Turabian StyleVermeulen, Maxime, Federico Del Vento, Francesca De Michele, Jonathan Poels, and Christine Wyns. 2018. "Development of a Cytocompatible Scaffold from Pig Immature Testicular Tissue Allowing Human Sertoli Cell Attachment, Proliferation and Functionality" International Journal of Molecular Sciences 19, no. 1: 227. https://doi.org/10.3390/ijms19010227
APA StyleVermeulen, M., Del Vento, F., De Michele, F., Poels, J., & Wyns, C. (2018). Development of a Cytocompatible Scaffold from Pig Immature Testicular Tissue Allowing Human Sertoli Cell Attachment, Proliferation and Functionality. International Journal of Molecular Sciences, 19(1), 227. https://doi.org/10.3390/ijms19010227