Novel Therapeutics for Multiple Sclerosis Designed by Parasitic Worms
Abstract
1. Introduction
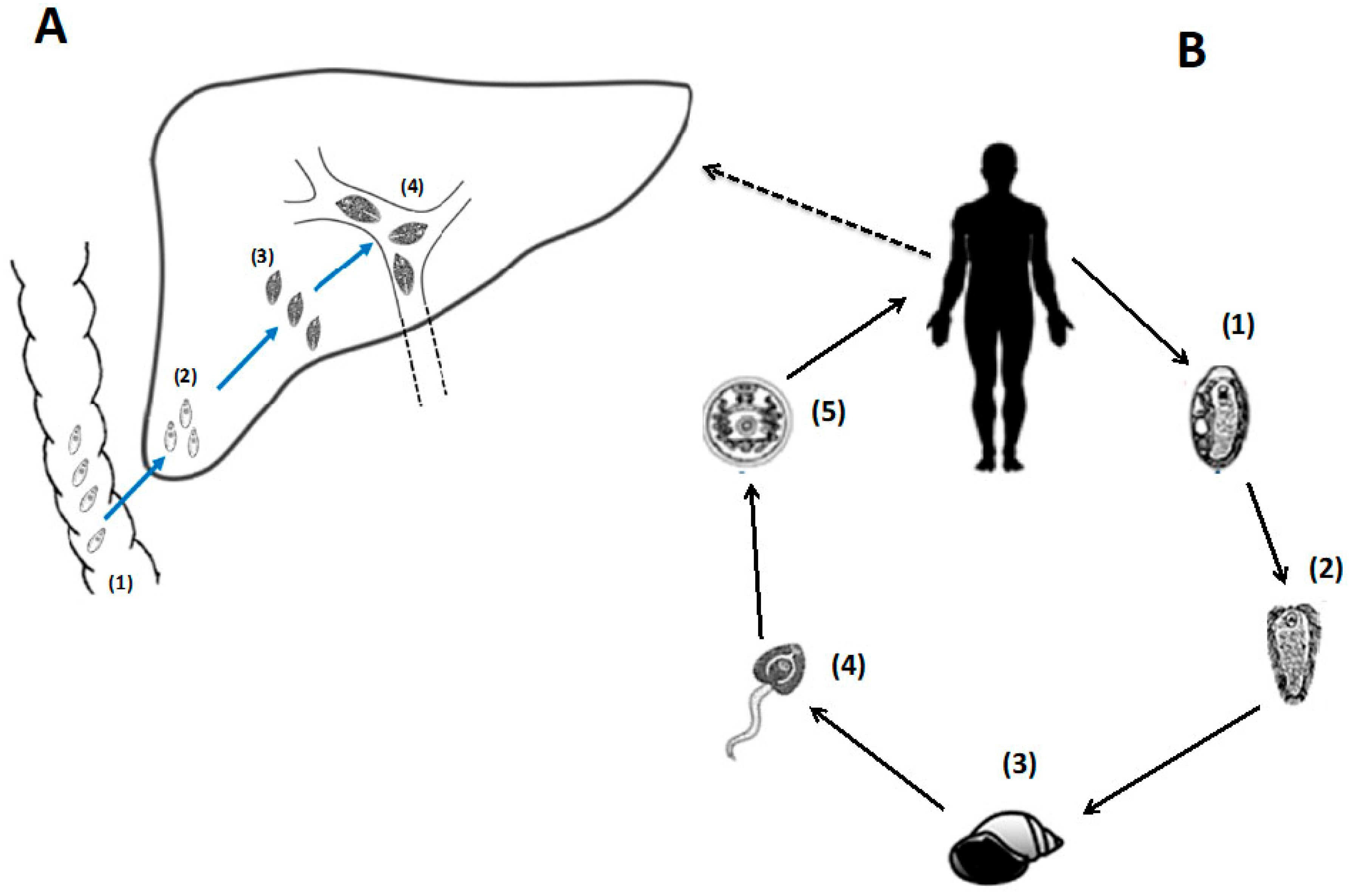
2. Should We Reunite with Our Old Friends the Helminth Parasites?
3. Worm Therapy: Proof-of-Principle in Experimental and Clinical Trials
3.1. Testing the Effectiveness of Helminth Infection in Animal Models of MS
3.2. Testing the Effectiveness of Helminth Infection in Human Trials
4. Translating to the Clinic: Worms or Molecules?
5. The Peptide FhHDM-1, Secreted by Fasciola hepatica, Shows Therapeutic Efficacy in EAE
6. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| MS | Multiple sclerosis |
| GWAS | Genome wide association studies |
| ILC2 | Type 2 Innate lymphoid cells |
| IL | Interleukin |
| Th | T helper |
| DC | Dendritic cell |
| Treg | T-regulatory cell |
| TGF-β | Transforming growth factor-beta |
| EAE | Experimental autoimmune encephalomyelitis |
| CNS | Central nervous system |
| MOG | Myelin oligodendrocyte glycoprotein |
| PLP | Proteolipid protein |
| MBP | Myelin basic protein |
| po | Oral administration |
| ip | Intraperitoneal injection |
| Sc | Subcutaneous injection |
| IFN | Interferon |
| DA | Dark Agouti |
| TSO | Trichuris suis ova |
| RRMS | Relapsing remitting multiple sclerosis |
| SPMS | Secondary progressive multiple sclerosis |
| MRI | Magnetic resonance imaging |
| NCT | National Clinical Trial |
| HINT | Helminth-induced immunomodulation therapy |
| TRIMS-A | Trichuris suis ova therapy for multiple sclerosis—a safety study |
| TRIOMS | Trichuris suis ova in recurrent remittent multiple sclerosis and clinically isolated syndrome |
| WIRMS | Worms for immune regulation of multiple sclerosis |
| GMP | Good manufacturing practices |
| SEA | Schistosoma egg antigen |
| ESP | Excretory secretory products |
| TNF | Tumour necrosis factor |
| NEJ | Newly excysted juveniles |
| FhCL | Fasciola hepatica cathepsin L |
| FhPrx | Fasciola hepatica peroxiredoxin |
| FhHDM-1 | Fasciola hepatica helminth defense molecule |
References
- Sawcer, S. The major cause of multiple sclerosis is environmental: Genetics has a minor role–no. Mult. Scler. 2011, 17, 1174–1175. [Google Scholar] [CrossRef] [PubMed]
- Taylor, B.V. The major cause of multiple sclerosis is environmental: Genetics has a minor role–yes. Mult. Scler. 2011, 17, 1171–1173. [Google Scholar] [CrossRef] [PubMed]
- Gourraud, P.A.; Harbo, H.F.; Hauser, S.L.; Baranzini, S.E. The genetics of multiple sclerosis: An up-to-date review. Immunol. Rev. 2012, 248, 87–103. [Google Scholar] [CrossRef] [PubMed]
- Correale, J.; Farez, M.F.; Gaitán, M.I. Environmental factors influencing multiple sclerosis in Latin America. Mult. Scler. J. Exp. Transl. Clin. 2017, 3, 2055217317715049. [Google Scholar] [CrossRef] [PubMed]
- Ebers, G.C.; Bulman, D.E.; Sadovnik, A.D.; Paty, D.W.; Warren, S.; Hader, W.; Murray, T.J.; Seland, T.P.; Duquette, P.; Grey, T. A population-based study of multiple sclerosis in twins. N. Engl. J. Med. 1986, 315, 1638–1642. [Google Scholar] [CrossRef] [PubMed]
- Bach, J.F. The effect of infections on susceptibility to autoimmune and allergic diseases. N. Engl. J. Med. 2002, 347, 911–920. [Google Scholar] [CrossRef] [PubMed]
- Bilbo, S.D.; Wray, G.A.; Perkins, S.E.; Parker, W. Reconstitution of the human biome as the most reasonable solution for epidemics of allergic and autoimmune diseases. Med. Hypotheses 2011, 77, 494–504. [Google Scholar] [CrossRef] [PubMed]
- Cooke, A. Infection and autoimmunity. Blood Cells Mol. Dis. 2009, 42, 105–107. [Google Scholar] [CrossRef] [PubMed]
- Okada, H.; Kuhn, C.; Feillet, H.; Bach, J.F. The ‘hygiene hypothesis’ for autoimmune and allergic diseases: An update. Clin. Exp. Immunol. 2010, 160, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Orton, S.M.; Ramagopalan, S.V.; Brocklebank, D.; Herrera, B.M.; Dyment, D.A.; Yee, I.M.; Sadovnick, A.D.; Ebers, G.C. Effect of immigration on multiple sclerosis sex ratio in Canada: The Canadian Collaborative Study. J. Neurol. Neurosurg. Psychiatry 2010, 81, 31–36. [Google Scholar] [CrossRef] [PubMed]
- McLeod, J.G.; Hammond, S.R.; Kurtzke, J.F. Migration and multiple sclerosis in United Kingdom and Ireland immigrants to Australia: A reassessment. II. Characteristics of early (pre-1947) compared to later migrants. J. Neurol. 2012, 259, 684–693. [Google Scholar] [CrossRef] [PubMed]
- Ascherio, A.; Munger, K.L. Environmental risk factors for multiple sclerosis. Part I: The role of infection. Ann. Neurol. 2007, 61, 288–299. [Google Scholar] [CrossRef] [PubMed]
- Ascherio, A.; Munger, K.L. Environmental risk factors for multiple sclerosis. Part II: Noninfectious factors. Ann. Neurol. 2007, 61, 504–513. [Google Scholar] [CrossRef] [PubMed]
- Taylor, B.V.; Lucas, R.M.; Dear, K.; Kilpatrick, T.J.; Pender, M.P.; van der Mei, I.A.; Chapman, C.; Coulthard, A.; Dwyer, T.; McMichael, A.J.; et al. Latitudinal variation in incidence and type of first central nervous system demyelinating events. Mult. Scler. 2010, 16, 398–405. [Google Scholar] [CrossRef] [PubMed]
- Fleming, J.O. Helminth therapy and multiple sclerosis. Int. J. Parasitol. 2013, 43, 259–274. [Google Scholar] [CrossRef] [PubMed]
- Correale, J.; Gaitán, M.I. Multiple sclerosis and environmental factors: The role of vitamin D, parasites, and Epstein-Barr virus infection. Acta Neurol. Scand. 2015, 132, 46–55. [Google Scholar] [CrossRef] [PubMed]
- Olsson, T.; Barcellos, L.F.; Alfredsson, L. Interactions between genetic, lifestyle and environmental risk factors for multiple sclerosis. Nat. Rev. Neurol. 2017, 13, 25–36. [Google Scholar] [CrossRef] [PubMed]
- Zaccone, P.; Fehervari, Z.; Phillips, J.M.; Dunne, D.W.; Cooke, A. Parasitic worms and inflammatory diseases. Parasite Immunol. 2006, 28, 515–523. [Google Scholar] [CrossRef] [PubMed]
- Cabre, P.; Signate, A.; Olindo, S.; Merle, H.; Caparros-Lefebvre, D.; Béra, O.; Smadja, D. Role of return migration in the emergence of multiple sclerosis in the French West Indies. Brain 2005, 128, 2899–2910. [Google Scholar] [CrossRef] [PubMed]
- Fleming, J.O.; Cook, T.D. Multiple sclerosis and the hygiene hypothesis. Neurology 2006, 67, 2085–2086. [Google Scholar] [CrossRef] [PubMed]
- Correale, J.; Farez, M. The impact of parasite infections on the course of multiple sclerosis. J. Neuroimmunol. 2011, 233, 6–11. [Google Scholar] [CrossRef] [PubMed]
- Hotez, P.J.; Brindley, P.J.; Bethony, J.M.; King, C.H.; Pearce, E.J.; Jacobson, J. Helminth infections: The great neglected tropical diseases. J. Clin. Investig. 2008, 118, 1311–1321. [Google Scholar] [CrossRef] [PubMed]
- Cox, F.E. History of human parasitology. Clin. Microbiol. Rev. 2002, 15, 595–612. [Google Scholar] [CrossRef] [PubMed]
- Jackson, J.A.; Friberg, I.M.; Little, S.; Bradley, J.E. Review series on helminths, immune modulation and the hygiene hypothesis: Immunity against helminths and immunological phenomena in modern human populations: Coevolutionary legacies? Immunology 2009, 126, 18–27. [Google Scholar] [CrossRef] [PubMed]
- Finkelman, F.D.; Shea-Donohue, T.; Morris, S.C.; Gildea, L.; Strait, R.; Madden, K.B.; Schopf, L.; Urban, J.F. Interleukin-4- and interleukin-13- mediated host protection against intestinal nematode parasites. Immunol. Rev. 2004, 201, 139–155. [Google Scholar] [CrossRef] [PubMed]
- Jenkins, S.J.; Allen, J.E. Similarity and diversity in macrophage activation by nematodes, trematodes, and cestodes. J. Biomed. Biotechnol. 2010, 2010, 262609. [Google Scholar] [CrossRef] [PubMed]
- Anthony, R.M.; Rutitzky, L.I.; Urban, J.F.; Stadecker, M.J.; Gause, W.C. Protective immune mechanisms in helminth infection. Nat. Rev. Immunol. 2007, 7, 975–987. [Google Scholar] [CrossRef] [PubMed]
- McSorley, H.J.; Hewitson, J.P.; Maizels, R.M. Immunomodulation by helminth parasites: Defining mechanisms and mediators. Int. Jnl. Parasitol. 2013, 43, 301–310. [Google Scholar] [CrossRef] [PubMed]
- Fallon, P.G.; Ballantyne, S.J.; Mangan, N.E.; Barlow, J.L.; Dasvarma, A.; Hewett, D.R.; McIlgorm, A.; Jolin, H.E.; McKenzie, A.N. Identification of an interleukin (IL)-25-dependent cell population that provides IL-4, IL-5, and IL-13 at the onset of helminth expulsion. J. Exp. Med. 2006, 203, 1105–1116. [Google Scholar] [CrossRef] [PubMed]
- Howitt, M.R.; Lavoie, S.; Michaud, M.; Blum, A.M.; Tran, S.V.; Weinstock, J.V.; Gallini, C.A.; Redding, K.; Margolskee, R.F.; Osborne, L.C.; et al. Tuft cells, taste-chemosensory cells, orchestrate parasite type 2 immunity in the gut. Science 2016, 351, 1329–1333. [Google Scholar] [CrossRef] [PubMed]
- Klose, C.S.; Artis, D. Innate lymphoid cells as regulators of immunity, inflammation and tissue homeostasis. Nat. Immunol. 2016, 17, 765–774. [Google Scholar] [CrossRef] [PubMed]
- Mosser, D.M.; Edwards, J.P. Exploring the full spectrum of macrophage activation. Nat. Rev. Immunol. 2008, 8, 958–969. [Google Scholar] [CrossRef] [PubMed]
- Walsh, K.P.; Brady, M.T.; Finlay, C.M.; Boon, L.; Mills, K.H. Infection with a helminth parasite attenuates autoimmunity through TGF-β-mediated suppression of Th17 and Th1 responses. J. Immunol. 2009, 183, 1577–1586. [Google Scholar] [CrossRef] [PubMed]
- Gause, W.C.; Wynn, T.A.; Allen, J.E. Type 2 immunity and wound healing: Evolutionary refinement of adaptive immunity by helminths. Nat. Rev. Immunol. 2013, 13, 607–614. [Google Scholar] [CrossRef] [PubMed]
- Allen, J.E.; Wynn, T.A. Evolution of Th2 immunity: A rapid repair response to the tissue destructive pathogens. PLoS Pathog. 2011, 7, e1002003. [Google Scholar] [CrossRef] [PubMed]
- Fumagalli, M.; Pozzoli, U.; Cagliani, R.; Comi, G.P.; Riva, S.; Clerici, M.; Bresolin, N.; Sironi, M. Parasites represent a major selective force for interleukin genes and shape the genetic predisposition to autoimmune conditions. J. Exp. Med. 2009, 206, 1395–1408. [Google Scholar] [CrossRef] [PubMed]
- Maizels, R.M. Parasite immunomodulation and polymorphisms of the immune system. J Biol. 2009, 8, 62. [Google Scholar] [CrossRef] [PubMed]
- Allen, J.E.; Maizels, R.M. Diversity and dialogue in immunity to helminths. Nat. Rev. Immunol. 2011, 11, 375–388. [Google Scholar] [CrossRef] [PubMed]
- Edwards, L.J.; Constantinescu, C.S. Parasite immunomodulation in autoimmune disease: Focus on multiple sclerosis. Expert. Rev. Clin. Immunol. 2009, 5, 487–489. [Google Scholar] [CrossRef] [PubMed]
- Hasseldam, H.; Hansen, C.S.; Johansen, F.F. Immunomodulatory effects of helminths and protozoa in multiple sclerosis and experimental autoimmune encephalomyelitis. Parasite Immunol. 2013, 35, 103–108. [Google Scholar] [CrossRef] [PubMed]
- Tanasescu, R.; Constantinescu, C.S. Helminth Therapy for MS. Curr. Top. Behav. Neurosci. 2015, 26, 195–220. [Google Scholar] [PubMed]
- Peón, A.N.; Terrazas, L.I. Immune-Regulatory Mechanisms of Classical and Experimental Multiple Sclerosis Drugs: A Special Focus on Helminth-Derived Treatments. Curr. Med. Chem. 2016, 23, 1152–1170. [Google Scholar] [CrossRef] [PubMed]
- Filyk, H.A.; Osborne, L.C. The Multibiome: The Intestinal Ecosystem’s Influence on Immune Homeostasis, Health, and Disease. EBioMedicine 2016, 13, 46–54. [Google Scholar] [CrossRef] [PubMed]
- Kuerten, S.; Lehmann, P.V. The immune pathogenesis of experimental autoimmune encephalomyelitis: Lessons learned for multiple sclerosis? J. Interferon Cytokine Res. 2011, 31, 907–916. [Google Scholar] [CrossRef] [PubMed]
- Chiuso-Minicucci, F.; Van, D.B.; Zorzella-Pezavento, S.F.; Peres, R.S.; Ishikawa, L.L.; Rosa, L.C.; França, T.G.; Turato, W.M.; Amarante, A.F.; Sartori, A. Experimental autoimmune encephalomyelitis evolution was not modified by multiple infections with Strongyloides venezuelensis. Parasite Immunol. 2011, 33, 303–308. [Google Scholar] [CrossRef] [PubMed]
- Sewell, D.; Qing, Z.; Reinke, E.; Elliot, D.; Weinstock, J.; Sandor, M.; Fabry, Z. Immunomodulation of experimental autoimmune encephalomyelitis by helminth ova immunization. Int. Immunol. 2003, 15, 59–69. [Google Scholar] [CrossRef] [PubMed]
- Correale, J.; Farez, M. Association between parasite infection and immune responses in multiple sclerosis. Ann. Neurol. 2007, 61, 97–108. [Google Scholar] [CrossRef] [PubMed]
- Fleming, J.O.; Isaak, A.; Lee, J.E.; Luzzio, C.C.; Carrithers, M.D.; Cook, T.D.; Field, A.S.; Boland, J.; Fabry, Z. Probiotic helminth administration in relapsing-remitting multiple sclerosis: A phase 1 study. Mult. Scler. 2011, 17, 743–754. [Google Scholar] [CrossRef] [PubMed]
- Benzel, F.; Erdur, H.; Kohler, S.; Frentsch, M.; Thiel, A.; Harms, L.; Wandinger, K.P.; Rosche, B. Immune monitoring of Trichuris suis egg therapy in multiple sclerosis patients. J. Helminthol. 2012, 86, 339–347. [Google Scholar] [CrossRef] [PubMed]
- Broadhurst, M.J.; Leung, J.M.; Kashyap, V.; McCune, J.M.; Mahadevan, U.; McKerrow, J.H.; Loke, P. IL-22+ CD4+ T cells are associated with therapeutic Trichuris trichiura infection in an ulcerative colitis patient. Sci. Transl. Med. 2010, 2, 60ra88. [Google Scholar] [CrossRef] [PubMed]
- Gaze, S.; McSorley, H.J.; Daveson, J.; Jones, D.; Bethony, J.M.; Oliveira, L.M.; Speare, R.; McCarthy, J.S.; Engwerda, C.R.; Croese, J.; et al. Characterising the mucosal and systemic immune responses to experimental human hookworm infection. PLoS Pathog. 2012, 8, e1002520. [Google Scholar] [CrossRef] [PubMed]
- La Flamme, A.C.; Ruddenklau, K.; Backstrom, B.T. Schistosomiasis decreases central nervous system inflammation and alters the progression of experimental autoimmune encephalomyelitis. Infect. Immun. 2003, 71, 4996–5004. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.; Nagano, I.; Asanom, K.; Takahashim, Y. Infection of non-encapsulated species of Trichinella ameliorates experimental autoimmune encephalomyelitis involving suppression of Th17 and Th1 response. Parasitol. Res. 2010, 107, 1173–1188. [Google Scholar] [CrossRef] [PubMed]
- Reyes, J.L.; Espinoza-Jiménez, A.F.; González, M.I.; Verdin, L.; Terrazas, L.I. Taenia crassiceps infection abrogates experimental autoimmune encephalomyelitis. Cell Immunol. 2011, 267, 77–87. [Google Scholar] [CrossRef] [PubMed]
- Donskow-Lysoniewska, K.; Krawczak, K.; Doligalska, M. Heligmosomoides polygyrus: EAE remission is correlated with different systemic cytokine profiles provoked by L4 and adult nematodes. Exp. Parasitol. 2012, 132, 243–248. [Google Scholar] [CrossRef] [PubMed]
- Gruden-Movsesijan, A.; Ilic, N.; Mostarica-Stojkovic, M.; Stosic-Grujicic, S.; Milic, M.; Sofronic-Milosavljevic, L. Trichinella spiralis: Modulation of experimental autoimmune encephalomyelitis in DA rats. Exp. Parasitol. 2008, 118, 641–647. [Google Scholar] [CrossRef] [PubMed]
- Gruden-Movsesijan, A.; Ilic, N.; Mostarica-Stojkovic, M.; Stosic-Grujicic, S.; Milic, M.; Sofronic-Milosavljevic, L. Mechanisms of modulation of experimental autoimmune encephalomyelitis by chronic Trichinella spiralis infection in Dark Agouti rats. Parasite Immunol. 2010, 32, 450–459. [Google Scholar] [CrossRef] [PubMed]
- Voldsgaard, A.; Bager, P.; Garde, E.; Åkeson, P.; Leffers, A.M.; Madsen, C.G.; Kapel, C.; Roepstorff, A.; Thamsborg, S.M.; Melbye, M.; et al. Trichuris suis ova therapy in relapsing multiple sclerosis is safe but without signals of beneficial effect. Mult. Scler. 2015, 21, 1723–1729. [Google Scholar] [CrossRef] [PubMed]
- Rosche, B.; Wernecke, K.D.; Ohlraun, S.; Dörr, J.M.; Paul, F. Trichuris suis ova in relapsing-remitting multiple sclerosis and clinically isolated syndrome (TRIOMS): Study protocol for a randomized controlled trial. Trials 2013, 14, 112. [Google Scholar] [CrossRef] [PubMed]
- Fleming, J.O.; Weinstock, J.V. Clinical trials of helminth therapy in autoimmune diseases: Rationale and findings. Parasite Immunol. 2015, 37, 277–292. [Google Scholar] [CrossRef] [PubMed]
- Khan, A.R.; Fallon, P.G. Helminth therapies: Translating the unknown unknowns into known knowns. Int. J. Parasitol. 2013, 43, 293–299. [Google Scholar] [CrossRef] [PubMed]
- Ruyssers, N.E.; De Winter, B.Y.; De Man, J.G.; Loukas, A.; Herman, A.G.; Pelckmans, P.A.; Moreels, T.G. Worms and the treatment of inflammatory bowel disease: Are molecules the answer? Clin. Dev. Immunol. 2008, 567314. [Google Scholar] [CrossRef] [PubMed]
- Briggs, N.; Weatherhead, J.; Sastry, K.J.; Hotez, P.J. The Hygiene Hypothesis and Its Inconvenient Truths about Helminth Infections. PLoS Negl. Trop. Dis. 2016, 10, e0004944. [Google Scholar] [CrossRef] [PubMed]
- Jouvin, M.H.; Kinet, J.P. Trichuris suis ova: Testing a helminth-based therapy as an extension of the hygiene hypothesis. J. Allergy Clin. Immunol. 2012, 130, 3–10. [Google Scholar] [CrossRef] [PubMed]
- Summers, R.W.; Elliott, D.E.; Urban, J.F.; Thompson, R.; Weinstock, J.V. Trichuris suis therapy in Crohn’s disease Gut 2005, 54, 87–90. Gut 2005, 54, 87–90. [Google Scholar] [CrossRef] [PubMed]
- Maizels, R.M.; Yazdanbakhsh, M. T-cell regulation in helminth parasite infections: Implications for inflammatory diseases. Chem. Immunol. Allergy 2008, 94, 112–123. [Google Scholar] [PubMed]
- Farah, I.O.; Langoi, D.; Nyaundi, J.; Hau, J. Schistosome-induced pathology is exacerbated and Th2 polarization is enhanced during pregnancy. In Vivo 2007, 21, 599–602. [Google Scholar] [PubMed]
- Mejia, R.; Nutman, T.B. Screening, prevention, and treatment for hyperinfection syndrome and disseminated infections caused by Strongyloides stercoralis. Curr. Opin. Infect. Dis. 2012, 25, 458–463. [Google Scholar] [CrossRef] [PubMed]
- Wammes, L.J.; Mpairwe, H.; Elliott, A.M.; Yazdanbakhsh, M. Helminth therapy or elimination: Epidemiological, immunological, and clinical considerations. Lancet Infect. Dis. 2014, 14, 1150–1162. [Google Scholar] [CrossRef]
- Johnston, M.J.; MacDonald, J.A.; McKay, D.M. Parasitic helminths: A pharmacopeia of anti-inflammatory molecules. Parasitology 2009, 136, 125–147. [Google Scholar] [CrossRef] [PubMed]
- Hewitson, J.P.; Grainger, J.R.; Maizels, R.M. Helminth immunoregulation: The role of parasite secreted proteins in modulating host immunity. Mol. Biochem. Parasitol. 2009, 167, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Harnett, W.; Harnett, M.M. Helminth-derived immunomodulators: Can understanding the worm produce the pill? Nat. Rev. Immunol. 2010, 10, 278–284. [Google Scholar] [CrossRef] [PubMed]
- Ditgen, D.; Anandarajah, E.M.; Meissner, K.A.; Brattig, N.; Wrenger, C.; Liebau, E. Harnessing the Helminth Secretome for Therapeutic Immunomodulators. Bio. Med. Res. Int. 2014, 2014, 964350. [Google Scholar] [CrossRef] [PubMed]
- Zheng, X.; Hu, X.; Zhou, G.; Lu, Z.; Qiu, W.; Bao, J.; Dai, Y. Soluble egg antigen from Schistosoma japonicum modulates the progression of chronic progressive experimental autoimmune encephalomyelitis via Th2-shift response. J. Neuroimmunol. 2008, 194, 107–114. [Google Scholar] [CrossRef] [PubMed]
- Mathieson, W.; Wilson, R.A. A comparative proteomic study of the undeveloped and developed Schistosoma mansoni egg and its contents: The miracidium, hatch fluid and secretions. Int. J. Parasitol. 2010, 40, 617–628. [Google Scholar] [CrossRef] [PubMed]
- Robinson, M.W.; Menon, R.; Donnelly, S.M.; Dalton, J.P.; Ranganathan, S. An integrated transcriptomics and proteomics analysis of the secretome of the helminth pathogen Fasciola hepatica: Proteins associated with invasion and infection of the mammalian host. Mol. Cell Proteom. 2009, 8, 1891–1907. [Google Scholar] [CrossRef] [PubMed]
- Kuijk, L.M.; Klaver, E.J.; Kooij, G.; van der Pol, S.M.; Heijnen, P.; Bruijns, S.C.; Kringel, H.; Pinelli, E.; Kraal, G.; de Vries, H.E.; et al. Soluble helminth products suppress clinical signs in murine experimental autoimmune encephalomyelitis and differentially modulate human dendritic cell activation. Mol. Immunol. 2012, 51, 210–218. [Google Scholar] [CrossRef] [PubMed]
- Hansen, C.S.; Hasseldam, H.; Bacher, I.H.; Thamsborg, S.M.; Johansen, F.F.; Kringel, H. Trichuris suis secrete products that reduce disease severity in a multiple sclerosis model. Acta Parasitol. 2017, 62, 22–28. [Google Scholar] [CrossRef] [PubMed]
- Finlay, C.M.; Stefanskam, A.M.; Walsh, K.P.; Kelly, P.J.; Boon, L.; Lavelle, E.C.; Walsh, P.T.; Mills, K.H. Helminth products protect against autoimmunity via innate type 2 cytokines IL-5 and IL-33, which promote eosinophilia. J. Immunol. 2016, 196, 703–714. [Google Scholar] [CrossRef] [PubMed]
- Peón, A.N.; Ledesma-Soto, Y.; Olguín, J.E.; Bautista-Donis, M.; Sciutto, E.; Terrazas, L.I. Helminth products potently modulate experimental autoimmune encephalomyelitis by downregulating neuroinflammation and promoting a suppressive microenvironment. Mediators Inflamm. 2017, 2017. [Google Scholar] [CrossRef] [PubMed]
- Radovic, I.; Gruden-Movsesijan, A.; Ilic, N.; Cvetkovic, J.; Mojsilovic, S.; Devic, M.; Sofronic-Milosavljevic, L. Immunomodulatory effects of Trichinella spiralis-derived excretory-secretory antigens. Immunol. Res. 2015, 61, 312–325. [Google Scholar] [CrossRef] [PubMed]
- Andrews, S.J. The life-cycle of Fasciola hepatica. In Fasciolosis; Dalton, J.P., Ed.; CAB International: Oxford, UK, 1999; pp. 1–29. [Google Scholar]
- Mas-Coma, S.; Valero, M.A.; Bargues, M.D. Fasciola, lymnaeids and human fascioliasis, with a global overview on disease transmission, epidemiology, evolutionary genetics, molecular epidemiology and control. Adv. Parasitol. 2009, 69, 41–146. [Google Scholar] [PubMed]
- Garcia, H.H.; Moro, P.L.; Schantz, P.M. Zoonotic helminth infections of humans: Echinococcosis, cysticercosis and fascioliasis. Curr. Opin. Infect. Dis. 2007, 20, 489–494. [Google Scholar] [CrossRef] [PubMed]
- Mas-Coma, S.; Bargues, M.D.; Valero, M.A. Fascioliasis and other plant-borne trematode zoonoses. Int. J. Parasitol. 2005, 35, 1255–1278. [Google Scholar] [CrossRef] [PubMed]
- Cwiklinski, K.; Dalton, J.P.; Dufresne, P.J.; La Course, J.; Williams, D.J.; Hodgkinson, J.; Paterson, S. The Fasciola hepatica genome: Gene duplication and polymorphism reveals adaptation to the host environment and the capacity for rapid evolution. Genome Biol. 2015, 16, 71. [Google Scholar] [CrossRef] [PubMed]
- McGonigle, S.; Curley, G.P.; Dalton, J.P. Cloning of peroxiredoxin, a novel antioxidant enzyme, from the helminth parasite Fasciola hepatica. Parasitology 1997, 115, 101–104. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Sernández, V.; Mezo, M.; González-Warleta, M.; Perteguer, M.J.; Muiño, L.; Guitián, E.; Gárate, T.; Ubeira, F.M. The MF6p/FhHDM-1 major antigen secreted by the trematode parasite Fasciola hepatica is a heme-binding protein. J. Biol. Chem. 2014, 289, 1441–1456. [Google Scholar] [CrossRef] [PubMed]
- Donnelly, S.; O’Neill, S.M.; Stack, C.M.; Robinson, M.W.; Turnbull, L.; Whitchurch, C.; Dalton, J.P. Helminth cysteine proteases inhibit TRIF-dependent activation of macrophages via degradation of TLR3. J. Biol. Chem. 2010, 285, 3383–3392. [Google Scholar] [CrossRef]
- Donnelly, S.; O’Neill, S.M.; Sekiya, M.; Mulcahy, G.; Dalton, J.P. Thioredoxin peroxidase secreted by Fasciola hepatica induces the alternative activation of macrophages. Infect. Immun. 2005, 73, 166–173. [Google Scholar] [CrossRef] [PubMed]
- Donnelly, S.; Stack, C.M.; O’Neill, S.M.; Sayed, A.A.; Williams, D.L.; Dalton, J.P. Helminth 2-Cys peroxiredoxin drives Th2 responses through a mechanism involving alternatively activated macrophages. FASEB J. 2008, 22, 4022–4032. [Google Scholar] [CrossRef]
- Robinson, M.W.; Donnelly, S.; Hutchinson, A.T.; To, J.; Taylor, N.L.; Norton, R.S.; Perugini, M.A.; Dalton, J.P. A family of helminth molecules that modulate innate cell responses via molecular mimicry of host antimicrobial peptides. PLoS Pathog. 2011, 7, e1002042. [Google Scholar] [CrossRef] [PubMed]
- Robinson, M.W.; Alvarado, R.; To, J.; Hutchinson, A.T.; Dowdell, S.N.; Lund, M.; Turnbull, L.; Whitchurch, C.B.; O’Brien, B.A.; Dalton, J.P.; et al. A helminth cathelicidin-like protein suppresses antigen processing and presentation in macrophages via inhibition of lysosomal vATPase. FASEB J. 2012, 26, 4614–4627. [Google Scholar] [CrossRef] [PubMed]
- Lund, M.E.; Greer, J.; Dixit, A.; Alvarado, R.; McCauley-Winter, P.; To, J.; Tanaka, A.; Hutchinson, A.T.; Robinson, M.W.; Simpson, A.M.; et al. A parasite-derived 68-mer peptide ameliorates autoimmune disease in murine models of Type 1 diabetes and multiple sclerosis. Sci. Rep. 2016, 6, 37789. [Google Scholar] [CrossRef] [PubMed]
| Animal Model a | Helminth Treatment b | Effects When Treatment Administered at c | Mechanism | Ref. | ||
|---|---|---|---|---|---|---|
| Pre-Induction | Induction | Effector | ||||
| PLP139-151 SJL mice | Schistosoma mansoni 5000–20,000 eggs i.p. | ++ | + | - | Polarisation of immune response to Th2 | [46] |
| Schistosoma mansoni 5000–10,000 eggs i.p./s.c. | ++ | ++ | - | Increase in IL-4, reduction in Interferon (IFN)-γ | [46] | |
| MOG35-55C57BL/6 mice | Schistosoma mansoni 5000–10,000 eggs i.p./ s.c. | ++ | ++ | - | Increase in IL-4, reduction in IFN-γ | [46] |
| Schistosoma mansoni 70 cercariae cutaneous | ++ | n.d. | n.d. | Reduction of Th1 pro-inflammatory cytokines | [52] | |
| Fasciola hepatica 10 metacercariae p.o. | ++ | n.d. | n.d. | Bystander attenuation of Th17 and Th1 responses by means of Transforming growth factor (TGF)-β | [33] | |
| Trichinella pseudospiralis 200 larvae p.o. | ++ | n.d. | n.d. | Strong Th2 responses; Th1 and Th17 responses suppressed | [53] | |
| Taenia crassiceps 400 metacestodes i.p. | +++ | n.d. | n.d. | Anti-inflammatory cytokine environment leads to reduced T cell activation | [54] | |
| Heligmosomoides polygyrus 200 larvae p.o. | n.d. | n.d. | ++ | Reduction of IL-17A, IL-12 and promotion of regulatory cytokines | [55] | |
| MBP-EAE in Lewis rats | Strongyloides venezuelensis 4000 larvae s.c. | - | n.d. | n.d. | No effect on disease course; The host may be resistant to helminth infection | [45] |
| Spinal cord homogenate in Dark agouti (DA) rats | Trichinella spiralis larvae Multiple doses by gastric inoculation | 500 & 1000 ++ | n.d. | n.d. | Th2 response and regulatory response increase, also increase in IL-10 | [56] |
| Trichinella spiralia Larvae p.o. | + | ++ | n.d. | Th2 cytokine bias; anti-inflammatory responses likely due to regulatory T cells | [57] | |
| ClinicalTrialsgov a | End Date | Subjects b | Helminth Treatment c | Clinical Evaluation Parameters d | Results | Status e | Ref. |
|---|---|---|---|---|---|---|---|
| HINT 1 NCT00645749 | 2011 | 5 RRMS | 2500 TSO orally 2 weeks × 12 weeks | Number of new gadolinium enhancing lesions on serial MRI scans (monthly) | Treatment was safe. No adverse effects. No. of lesions fell from 6.6 at baseline to 2. Serum IL-4, IL-10 increased in 4 patients | C | [48] |
| TRIMS-A 2010 NCT01006941 | 2011 | 10 RRMS | 2,500 TSO orally 2 weeks × 12 weeks | No. of new or enlarging T2 lesions, no. of Gd enhancing lesions, volume of T2 lesions | Well tolerated; minor gastrointestinal symptoms. No beneficial effect. | C | [58] |
| Charite safety study | 2011 | 4 SPMS | 2500 TSO orally 2 weeks × 4 weeks | Immunological & clinical parameters were assessed | Treatment was safe. Lower Th1 & increase in Th2 (IL-4) | C | [49] |
| TRIOMS 2012 NCT01413243 | 2016 | 50 RRMS | 2,500 TSO orally 2 weeks × 12 weeks | Number of new gadolinium enhancing lesions on serial MRI scans; Vol of new T2 hyperintensive in cerebral MRI | Terminated | T | [59] |
| WIRMS 2011 NCT01470521 | 2016 | 72 RRMS | 25 live Necator americanus dermally | Number of new gadolinium enhancing lesions on serial MRI scans (at month 9), change in expanded disability status scale | Final results not released—expecting lower number of lesions | C | [39] |
| HINT 2 NCT00645749 | 2017 | 18 RRMS | 2,500 TSO orally 2 weeks × 10 months | Number of new gadolinium enhancing lesions on serial MRI scans (monthly) | Safety confirmed. Interim MRI and immunological measures positive. | O | [15,60] |
| Animal Model | Helminth Treatment a | Time Point of Administration b | Mechanism of Protection | Ref. | |
|---|---|---|---|---|---|
| Pre-Induction | Induction | ||||
| MOG35-55 in C57BL/6 mice | Schistosoma japonicum SEA 100 μg i.p. once a week for 4 weeks | + | ++ | Th2 environment established. | [74] |
| Trichuris suis SP 100 μg i.p. once a week for 4 weeks | ++ | n.d. | Unknown. In vitro suppression of pro-inflammatory dendritic cells | [77] | |
| Trichuris spiralis SP 100 μg i.p. once a week for 4 weeks | ++ | n.d. | Unknown. In vitro suppression of pro-inflammatory dendritic cells | [77] | |
| Trichuris suis ESP 250 μg i.p. alternate days over 22 days | n.d. | ++ | Reduced number of splenic Th1 and Th17 cells | [78] | |
| F. hepatica ESP Five or Six daily doses of 50 μg/dose i.p. | + | ++ | Production of innate type 2 cytokines IL-5 and IL-33. | [79] | |
| Taenia Crassiceps ESP 250 μg i.p. alternate days × 7 times | n.d. | +++ | Induction of Th2. Suppression of Tumour Necrosis Factor (TNF) and IL-17. Redirected cell migration from the central nervous system to peritoneal cavity | [80] | |
| Spinal cord homogenate in DA rats | Trichinella spiralis larvae ESP Multiple doses of soluble products i.p. | ++ | n.d. | Strong Th2-type response and increased proportion of CD4+CD25-Foxp3+ regulatory cells | [81] |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dixit, A.; Tanaka, A.; Greer, J.M.; Donnelly, S. Novel Therapeutics for Multiple Sclerosis Designed by Parasitic Worms. Int. J. Mol. Sci. 2017, 18, 2141. https://doi.org/10.3390/ijms18102141
Dixit A, Tanaka A, Greer JM, Donnelly S. Novel Therapeutics for Multiple Sclerosis Designed by Parasitic Worms. International Journal of Molecular Sciences. 2017; 18(10):2141. https://doi.org/10.3390/ijms18102141
Chicago/Turabian StyleDixit, Aakanksha, Akane Tanaka, Judith M. Greer, and Sheila Donnelly. 2017. "Novel Therapeutics for Multiple Sclerosis Designed by Parasitic Worms" International Journal of Molecular Sciences 18, no. 10: 2141. https://doi.org/10.3390/ijms18102141
APA StyleDixit, A., Tanaka, A., Greer, J. M., & Donnelly, S. (2017). Novel Therapeutics for Multiple Sclerosis Designed by Parasitic Worms. International Journal of Molecular Sciences, 18(10), 2141. https://doi.org/10.3390/ijms18102141




