Cultivar and Metal-Specific Effects of Endophytic Bacteria in Helianthus tuberosus Exposed to Cd and Zn
Abstract
1. Introduction
2. Results and Discussion
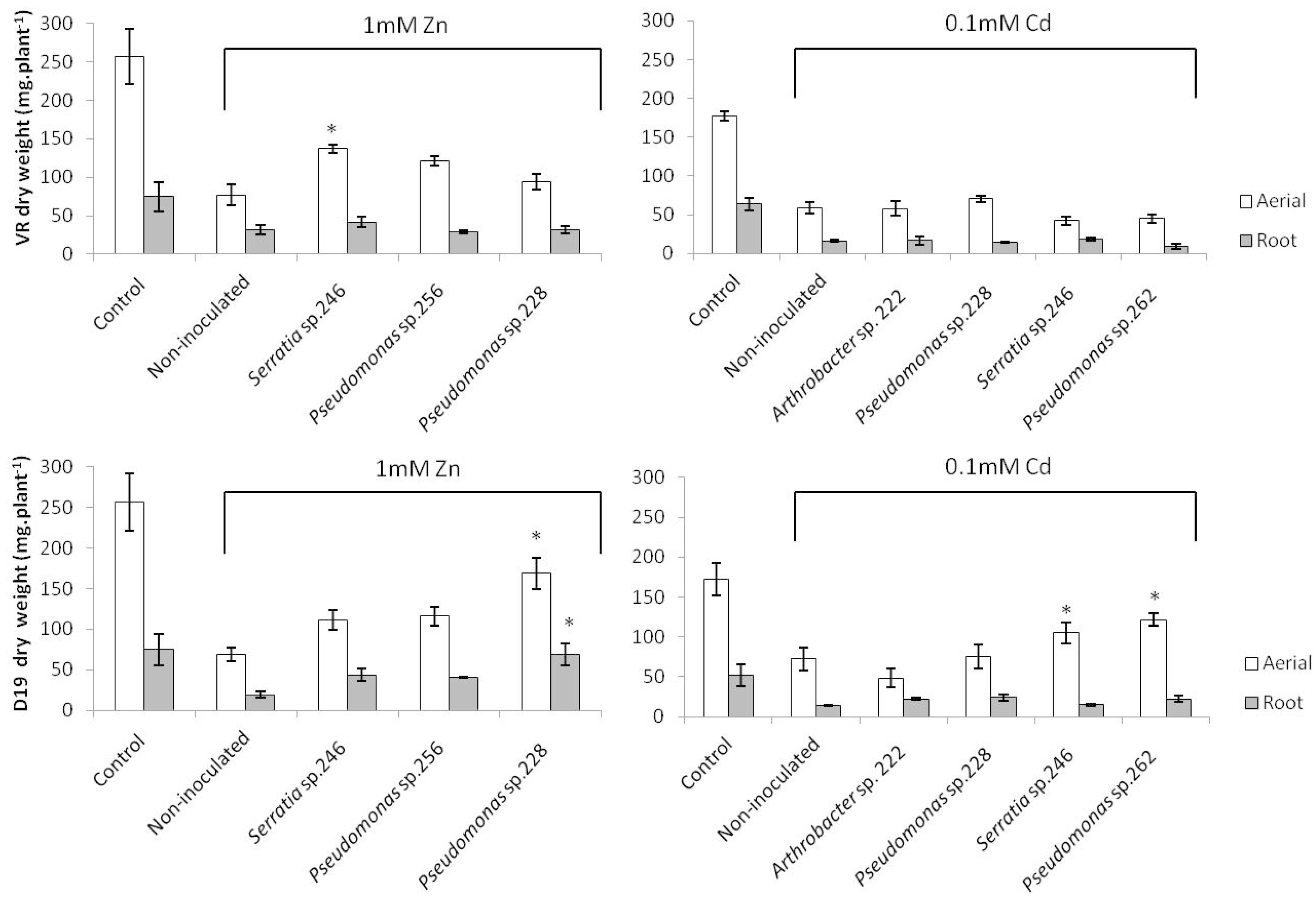
2.1. Plant Growth and Metal Uptake
2.2. Nutrient Status
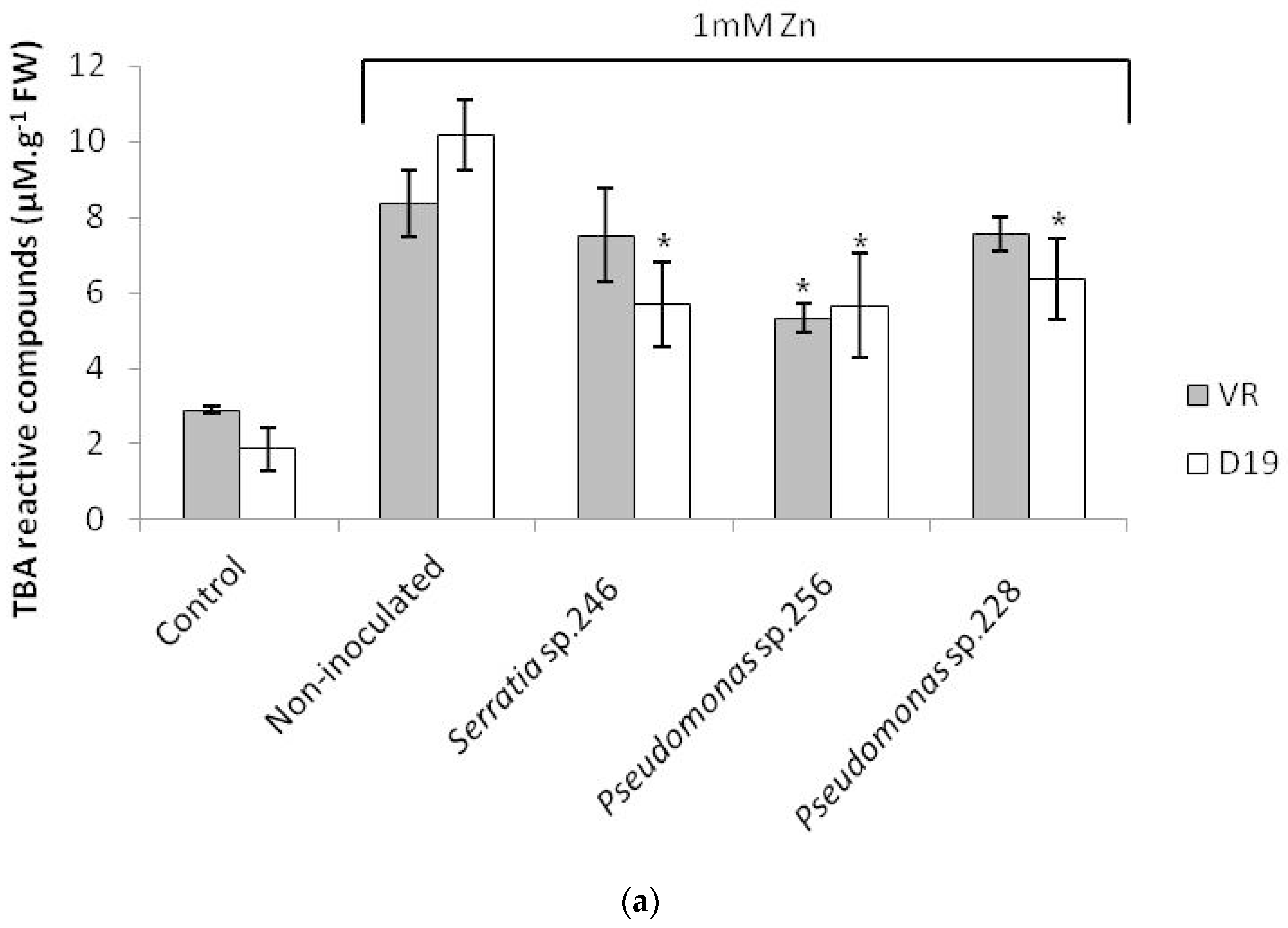
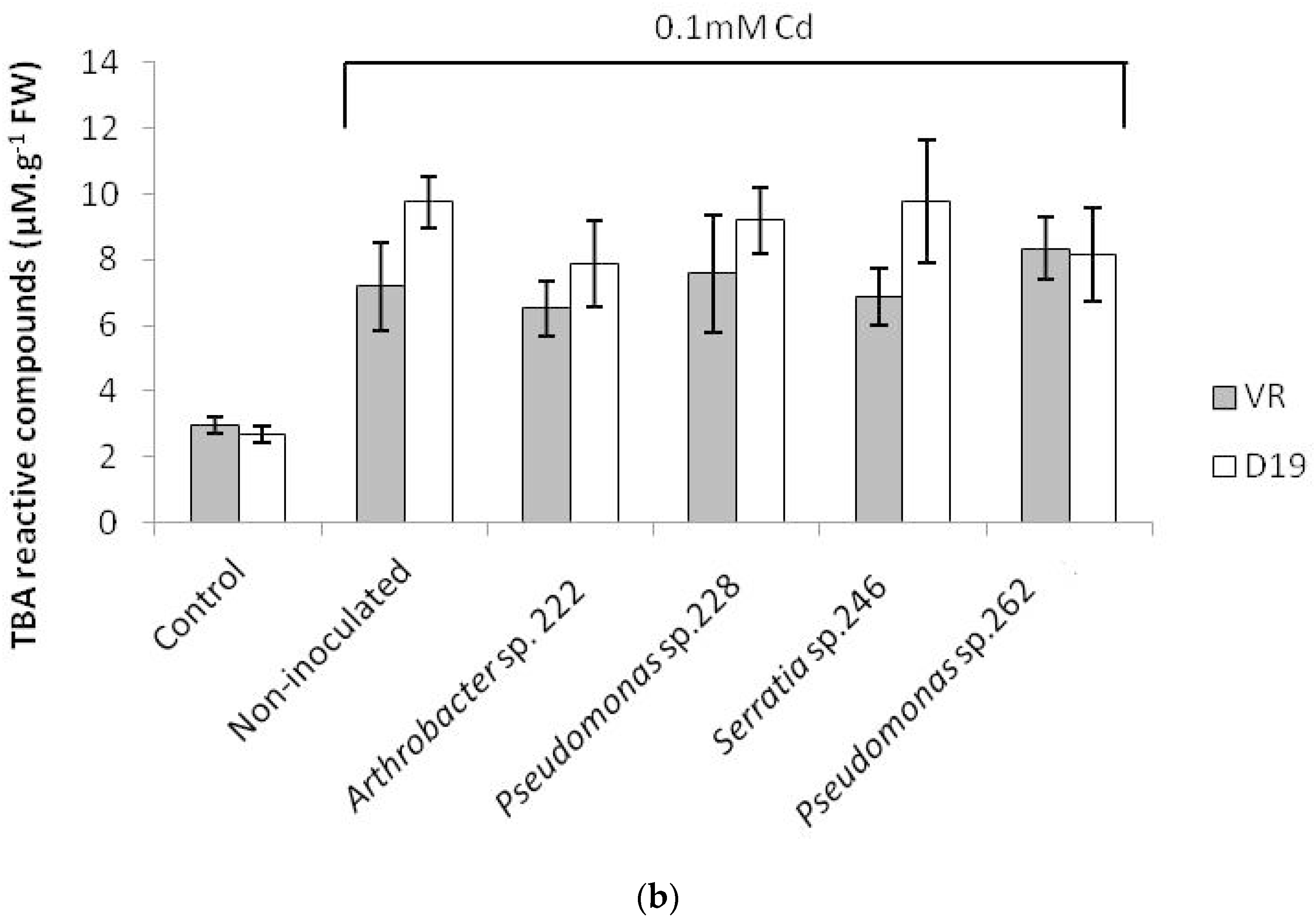
2.3. Lipid Peroxidation
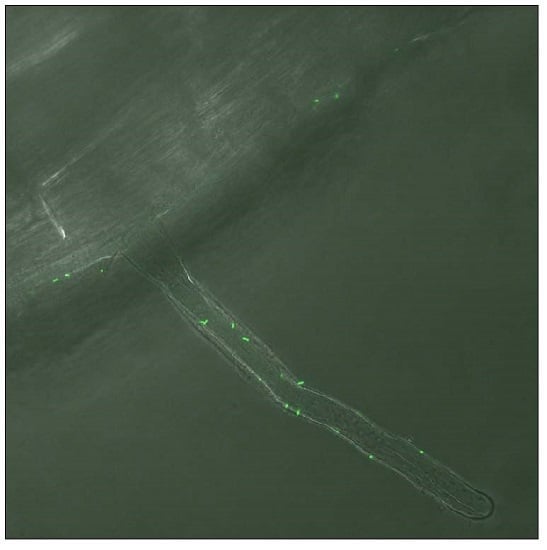
2.4. Colonization of Enhanced Green Fluorescent Protein (EGFP): Tetracycline® Pseudomonas sp. 262 in the Roots of H. tuberosus
3. Materials and Methods
3.1. Plant Material
3.2. PGPB Strains
3.3. Inoculation of PGPB Strains in H. tuberosus
3.4. Plant Analysis
3.5. Evaluation of the Colonization Process: Localization of Inoculated EGFP Labeled Pseudomonas sp. 262
3.5.1. Bacterial Strains and Growth Conditions
3.5.2. Introduction of the EGFP: Tetracycline into Pseudomonas sp. 262
3.5.3. Inoculation of EGFP Pseudomonas sp. 262 on Roots of H. tuberosus
3.5.4. Confocal Laser Scanning Microscopy
3.6. Statistical Analysis
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| PGPB | Plant Growth Promoting Bacteria |
| TBA | Thiobarbituric Acid |
| EGFP | Enhanced Green Fluorescent Protein |
| EDTA | Ethylenediaminetetraacetic Acid |
| MDA | Malondialdehyde |
References
- Mulligan, C.N.; Yong, R.N.; Gibbs, B.F. Heavy metal removal from sediments by biosurfactants. J. Hazard Mater. 2001, 85, 111–125. [Google Scholar] [CrossRef]
- Weyens, N.; van der Lelie, D.; Taghavi, S.; Newman, L.; Vangronsveld, J. Exploiting plant-microbe partnerships to improve biomass production and remediation. Trends Biotechnol. 2009, 27, 591–598. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Rajkumar, M.; Zhang, C.H.; Freitas, H. Beneficial role of bacterial endophytes in heavy metal phytoremediation. J. Environ. Manag. 2016, 174, 14–25. [Google Scholar] [CrossRef] [PubMed]
- Coninx, L.; Martinova, V.; Rineau, F. Mycorrhiza-Assisted Phytoremediation. In Phytoremediation. Advances in Botanical Research; Cuypers, A., Vangronsveld, J., Eds.; Elsevier: Amsterdam, The Netherlands, 2017; Volume 83, pp. 127–188. [Google Scholar]
- Kidd, P.S.; Álvarez-López, V.; Becerra-Castro, C.; Cabello-Conejo, M.; Prieto-Fernández, Á. Potential role of plant-associated bacteria in plant metal uptake and implications in phytotechnologies. In Phytoremediation. Advances in Botanical Research; Cuypers, A., Vangronsveld, J., Eds.; Elsevier: Amsterdam, The Netherlands, 2017; Volume 83, pp. 87–126. [Google Scholar]
- Germida, J.J.; Siciliano, S.D.; Renato de Freitas, J.; Seib, A.M. Diversity of root-associated bacteria associated with field-grown canola (Brassica napus L.) and wheat (Triticuma estivum L.). FEMS Microbiol. Ecol. 1998, 26, 43–50. [Google Scholar] [CrossRef]
- Genrich, I.; Burd, D.; George, D.; Glick, B.R. Plant growth promoting bacteria that decrease heavy metal toxicity in plants. Can. J. Microbiol. 2000, 46, 237–245. [Google Scholar]
- Rajkumar, M.; Sandhya, S.; Prasad, M.N.V.; Freitas, H. Perspectives of plant-associated microbes in heavy metal phytoremediation. Biotechnol. Adv. 2012, 30, 1562–1574. [Google Scholar] [CrossRef] [PubMed]
- Ullah, A.; Heng, S.; Munis, M.F.H.; Fahads, S.; Yang, X. Phytoremediation of heavy metals assisted by plant growth promoting (PGP) bacteria: A review. Environ. Exp. Bot. 2015, 117, 28–40. [Google Scholar] [CrossRef]
- Lugtenberg, B.; Kamilova, F. Plant-Growth-Promoting Rhizobacteria. Annu. Rev. Microbiol. 2009, 63, 541–556. [Google Scholar] [CrossRef] [PubMed]
- Roper, M.M.; Ladha, J.K. Biological N2-fixation by heterotrophic and phototrophic bacteria in association with straw. Plant Soil 1995, 174, 211–224. [Google Scholar] [CrossRef]
- Kim, K.Y.; Jordan, D.; McDonald, G.A. Enterobacter agglomerans, phosphate solubilizing bacteria, and microbial activity in soil: Effect of carbon sources. Soil Biol. Biochem. 1998, 30, 995–1003. [Google Scholar] [CrossRef]
- Ryan, R.P.; Germaine, K.; Franks, A.; Ryan, D.J.; Dowling, D.N. Bacterial endophytes: Recent developments and applications. FEMS Microbiol. Lett. 2008, 278, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Dobbelaere, S.; Croonenborghs, A.; Thys, A.; Vandebroek, A.; Vanderleyden, J. Phytostimulatory effect of Azospirillum brasilense wild type and mutant strains altered in IAA production on wheat. Plant Soil 1999, 212, 155–164. [Google Scholar] [CrossRef]
- Glick, B.R.; Penrose, D.M.; Li, J. A model for the lowering of plant ethylene concentrations by plant growth promoting bacteria. J. Theor. Biol. 1998, 190, 63–68. [Google Scholar] [CrossRef] [PubMed]
- Glick, B.R. Phytoremediation: Synergistic use of plants and bacteria to clean up the environment. Biotechnol. Adv. 2003, 21, 383–393. [Google Scholar] [CrossRef]
- Glick, B.R.; Bashan, Y. Genetic manipulation of plant growth-promoting bacteria to enhance biocontrol of phytopathogens. Biotechnol. Adv. 1997, 15, 353–378. [Google Scholar] [CrossRef]
- Fasim, F.; Ahmed, N.; Parsons, R.; Gadd, G.M. Solubilization of zinc salts by a bacterium isolated from the air environment of a tannery. FEMS Microbiol. Lett. 2002, 213, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Diels, L.; De Smet, M.; Hooyberghs, L.; Corbisier, P. Heavy metals bioremediation of soil. Mol. Biotechnol. 1999, 12, 149–158. [Google Scholar] [CrossRef]
- Rouch, D.A.; Lee, T.O.B.; Morby, A.P. Understanding cellular responses to toxic agents: A model for mechanisms-choice in bacterial resistance. J. Ind. Microbiol. 1995, 14, 132–141. [Google Scholar] [CrossRef] [PubMed]
- Madhaiyan, M.; Poonguzhali, S.; Sa, T. Influence of plant species and environmental conditions on epiphytic and endophytic pink-pigmented facultative methylotrophic bacterial populations associated with field-grown rice cultivars. J. Microbiol. Biotechnol. 2007, 17, 1645–1654. [Google Scholar] [PubMed]
- Vivas, A.; Biró, B.; Ruíz-Lozano, J.M.; Barea, J.M.; Azcón, R. Two bacterial strains isolated from a Zn-polluted soil enhance plant growth and mycorrhizal efficiency under Zn-toxicity. Chemosphere 2006, 62, 1523–1533. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Prasad, M.N.V.; Rajkumar, M.; Freitas, H. Plant growth promoting rhizobacteria and endophytes accelerate phytoremediation of metalliferous soils. Biotechnol. Adv. 2011, 29, 248–258. [Google Scholar] [CrossRef] [PubMed]
- Montalbán, B.; Croes, S.; Weyens, N.; Lobo, M.C.; Pérez-Sanz, A.; Vangronsveld, J. Characterization of bacterial communities associated with Brassica napus L. growing on a Zn-contaminated soil and their effects on root growth. Int. J. Phytoremediat. 2016, 18, 985–993. [Google Scholar]
- Chen, B.; Shen, J.; Zhang, X.; Pan, F.; Yang, X.; Feng, Y. The endophytic bacterium, Sphingomonas SaMR12, improves the potential for zinc phytoremediation by its host, Sedum alfredii. PLoS ONE 2014, 9, e106826. [Google Scholar] [CrossRef] [PubMed]
- Sheng, X.; Sun, L.; Huang, Z.; He, L.; Zhang, W.; Chen, Z. Promotion of growth and Cu accumulation of bio-energy crop (Zea mays) by bacteria: Implications for energy plant biomass production and phytoremediation. J. Environ. Manag. 2012, 103, 58–64. [Google Scholar] [CrossRef] [PubMed]
- He, H.; Ye, Z.; Yang, D.; Yan, J.; Xiao, L.; Zhong, T.; Yuan, M.; Cai, X.; Fang, Z.; Jing, Y. Characterization of endophytic Rahnella sp. JN6 from Polygonum pubescens and its potential in promoting growth and Cd, Pb, Zn uptake by Brassica napus. Chemosphere 2013, 90, 1960–1965. [Google Scholar] [CrossRef] [PubMed]
- Sessitsch, A.; Kuffner, M.; Kidd, P.; Vangronsveld, J.; Wenzel, W.W.; Fallmann, K.; Puschenreiter, M. The role of plant-associated bacteria in the mobilization and phytoextraction of trace elements in contaminated soils. Soil Biol. Biochem. 2013, 60, 182–194. [Google Scholar] [CrossRef] [PubMed]
- Rajkumar, M.; Ae, N.; Freitas, H. Endophytic bacteria and their potential to enhance heavy metal phytoextraction. Chemosphere 2009, 77, 153–160. [Google Scholar] [CrossRef] [PubMed]
- Wan, Y.; Luo, S.; Chen, J.; Xiao, X.; Chen, L.; Zeng, G.; Liu, C.; He, Y. Effect of endophyte-infection on growth parameters and Cd-induced phytotoxicity of Cd-hyperaccumulator Solanum nigrum L. Chemosphere 2012, 89, 743–750. [Google Scholar] [CrossRef] [PubMed]
- Pandey, S.; Ghosh, P.K.; Ghosh, S.; Kumar, D.T.; Maiti, T.K. Role of heavy metal resistant Ochrobactrum sp. and Bacillus spp. strains in bioremediation of a rice cultivar and their PGPR like activities. J. Microbiol. 2013, 5, 7–11. [Google Scholar] [CrossRef] [PubMed]
- Weyens, N.; van der Lelie, D.; Taghavi, S.; Vangronsveld, J. Phytoremediation: Plant-endophyte partnerships take the challenge. Curr. Opin. Biotechnol. 2009, 20, 248–254. [Google Scholar] [CrossRef] [PubMed]
- Lugtenberg, B.J.J.; Dekkers, L.; Bloemberg, G.V. Molecular determinants of rhizosphere colonization by Pseudomonas. Annu. Rev. Phytopathol. 2001, 39, 461–490. [Google Scholar] [CrossRef] [PubMed]
- Compant, S.; Clément, C.; Sessitsch, A. Plant growth-promoting bacteria in the rhizo- and endosphere of plants: Their role, colonization, mechanisms involved and prospects for utilization. Soil Biol. Biochem. 2010, 42, 669–678. [Google Scholar] [CrossRef]
- Lagendijk, E.L.; Validov, S.; Lamers, G.E.M.; de Weert, S.; Bloemberg, G.V. Genetic tools for tagging Gram-negative bacteria with mCherry for visualization in vitro and in natural habitats, biofilm and pathogenicity studies. FEMS Microbiol. Lett. 2010, 305, 81–90. [Google Scholar] [CrossRef] [PubMed]
- Bloemberg, G.V.; Lugtenberg, B.J.J. Molecular basis of plant growth promotion and biocontrol by rhizobacteria. Curr. Opin. Plant Biol. 2001, 4, 343–350. [Google Scholar] [CrossRef]
- Germaine, K.; Keogh, E.; Borremans, B.; van der Lelie, D.; Barac, T.; Oeyen, L.; Vangronsveld, J.; Moore, F.P.; Moore, E.R.B.; Campbel, C.D.; et al. Colonisation of poplar trees by gfp expressing endophytes. FEMS Microbiol. Ecol. 2004, 48, 109–118. [Google Scholar] [CrossRef] [PubMed]
- Weyens, N.; Boulet, J.; Adriaensen, D.; Timmermans, J.P.; Prinsen, E.; Van Oevelen, S.; D’Haen, J.; Smeets, K.; van der Lelie, D.; Taghavi, S.; et al. Contrasting colonization and plant growth promoting capacity between wild type and a gfp-derative of the endophyte Pseudomonas putida W619 in hybrid poplar. Plant Soil 2012, 356, 217–230. [Google Scholar] [CrossRef]
- Bloemberg, G.V.; Wijfjes, A.H.; Lamers, G.E.; Stuurman, N.; Lugtenberg, B.J. Simultaneous imaging of Pseudomonas fluorescens WCS365 populations expressing three different autofluorescent proteins in the rhizosphere: New perspectives for studying microbial communities. Mol. Plant Microbe Interact. 2000, 13, 1170–1176. [Google Scholar] [CrossRef] [PubMed]
- Newman, K.L.; Almeida, R.P.P.; Purcell, A.H.; Lindow, S.E. Use of a green fluorescent strain for analysis of Xylella fastidiosa colonization of Vitis vinifera. Appl. Environ. Microbiol. 2003, 69, 7319–7327. [Google Scholar] [CrossRef] [PubMed]
- Serieys, H.; Souyris, I.; Gil, A.; Poinso, B.; Berville, A. Diversity of Jerusalem artichoke clones (Helianthus tuberosus L.) from the INRA-Montpellier collection. Genet. Resour. Crop Evol. 2010, 57, 1207–1215. [Google Scholar] [CrossRef]
- Denoroy, P. The crop physiology of Helianthus tuberosus L.: A model oriented view. Biomass Bioenergy 1996, 11, 11–32. [Google Scholar] [CrossRef]
- Kays, S.J.; Nottingham, S.F. Biology and Chemistry of Jerusalem Artichoke Helianthus tuberosus L.; Taylor and Francis Group: Boca Raton, FL, USA; Abingdon, UK; New York, NY, USA, 2007; pp. 1–496. ISBN 9781420044959. [Google Scholar]
- Cui, S.; Zhou, Q.; Chao, L. Potential hyperaccumulation of Pb, Zn, Cu and Cd in endurant plants distributed in an old smeltery, northeast China. Environ. Geol. 2007, 51, 1043–1048. [Google Scholar] [CrossRef]
- Chen, L.; Long, X.; Zhang, Z.; Zheng, X.; Rengel, Z.; Liu, Z. Cadmium accumulation and translocation in two Jerusalem Artichoke (Helianthus tuberosus L.) Cultivars. Pedosphere 2011, 21, 573–580. [Google Scholar] [CrossRef]
- Long, X.; Ni, N.; Wang, L.; Wang, X.; Wang, Y.; Zhang, Z.; Zed, R.; Liu, Z.; Shao, H. Phytoremediation of cadmium-contaminated soil by two Jerusalem Artichoke (Helianthus tuberosus L.) genotypes. CLEAN Soil Air Water 2013, 41, 202–209. [Google Scholar] [CrossRef]
- Montalbán, B.; Lobo, M.C.; Alonso, J.; Pérez-Sanz, A. Metal(loid)s uptake and effects on the growth of Helianthus tuberosus cultivar-clones under multi-polluted hydroponic cultures. CLEAN Soil Air Water 2016, 44, 1368–1374. [Google Scholar] [CrossRef]
- Willscher, S.; Jablonski, L.; Fona, Z.; Rahmi, R.; Wittig, J. Phytoremediation experiments with Helianthus tuberosus under different pH and heavy metal soil concentrations. Hydrometallurgy 2016, 168, 153–158. [Google Scholar] [CrossRef]
- Zaidi, S.; Usmani, S.; Singh, B.R.; Musarrat, J. Significance of Bacillus subtilis strain SJ-101 as a bioinoculant for concurrent plant growth promotion and nickel accumulation in Brassica juncea. Chemosphere 2006, 64, 991–997. [Google Scholar] [CrossRef] [PubMed]
- Sheng, X.F.; Xia, J.J. Improvement of rape (Brassica napus) plant growth and cadmium uptake by cadmium-resistant bacteria. Chemosphere 2006, 64, 1036–1042. [Google Scholar] [CrossRef] [PubMed]
- Sheng, X.F.; Xia, J.J.; Jiang, C.Y.; He, L.Y.; Qian, M. Characterization of heavy metal-resistant endophytic bacteria from rape (Brassica napus) roots and their potential in promoting the growth and lead accumulation of rape. Environ. Pollut. 2008, 156, 1164–1170. [Google Scholar] [CrossRef] [PubMed]
- Duan, J.; Jiang, W.; Cheng, Z.; Heikkila, J.J.; Glick, B.R. The Complete Genome Sequence of the Plant Growth-Promoting Bacterium Pseudomonas sp. UW4. PLoS ONE 2013, 8, e58640. [Google Scholar] [CrossRef] [PubMed]
- Glick, B.R. Using soil bacteria to facilitate phytoremediation. Biotechnol. Adv. 2010, 28, 367–374. [Google Scholar] [CrossRef] [PubMed]
- Burd, G.I.; Dixon, D.G.; Glick, B.R. A plant growth-promoting bacterium that decreases nickel toxicity in seedlings. Appl. Environ. Microbiol. 1998, 64, 3663–3668. [Google Scholar] [PubMed]
- Marques, A.P.G.C.; Moreira, H.; Franco, A.R.; Rangel, A.O.S.S.; Castro, P.M.L. Inoculating Helianthus annuus (sunflower) grown in zinc and cadmium contaminated soils with plant growth promoting bacteria—Effects on phytoremediation strategies. Chemosphere 2013, 92, 74–83. [Google Scholar] [CrossRef] [PubMed]
- Tripathi, M.; Munot, H.P.; Shouche, Y.; Meyer, J.M.; Goel, R. Isolation and functional characterization of siderophore-producing lead- and cadmium-resistant Pseudomonas putida KNP9. Curr. Microbiol. 2005, 50, 233–237. [Google Scholar] [CrossRef] [PubMed]
- Aafi, N.E.; Brhada, F.; Dary, M.; Maltouf, A.F.; Pajuelo, E. Rhizostabilization of metals in soils using Lupinus luteus inoculated with the metal resistant rhizobacterium Serratia sp. MSMC 541. Int. J. Phytoremediat. 2012, 14, 261–274. [Google Scholar] [CrossRef] [PubMed]
- Nies, D.H. Microbial heavy-metal resistance. Appl. Microbiol. Biotechnol. 1999, 51, 730–750. [Google Scholar] [CrossRef] [PubMed]
- Bruins, M.R.; Kapil, S.; Oehme, F.W. Microbial resistance to metals in the environment. Ecotoxicol. Environ. Saf. 2000, 45, 198–207. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Cutright, T.J. Preliminary evaluation of microbiallymediated precipitation of cadmium, chromium, and nickel by rhizosphere consortium. J. Environ. Eng. 2003, 129, 4–9. [Google Scholar] [CrossRef]
- Kidd, P.; Barceló, J.; Bernal, M.P.; Navari-Izzo, F.; Poschenrieder, C.; Shilev, S.; Clemente, R.; Monterroso, C. Trace element behaviour at the root–soil interface: Implications in Phytoremediation. Environ. Exp. Bot. 2009, 67, 243–259. [Google Scholar] [CrossRef]
- Vecchio, A.; Finoli, C.; Di Simine, D.; Andreoni, V. Heavy metal biosorption by bacterial cells. Fresen. J. Anal. Chem. 1998, 361, 338–342. [Google Scholar] [CrossRef]
- Li, Y.; Zhang, S.; Jiang, W.; Liu, D. Cadmium accumulation, activities of antioxidant enzymes, and malondialdehyde (MDA) content in Pistiastratiotes L. Environ. Sci. Pollut. Res. 2013, 20, 1117–1123. [Google Scholar] [CrossRef] [PubMed]
- Smeets, K.; Cuypers, A.; Lambrechts, A.; Semane, B.; Hoet, P.; Van Laere, A.; Vangronsveld, J. Induction of oxidative stress and antioxidative mechanisms in Phaseolus vulgaris after Cd application. Plant Physiol. Biochem. 2005, 43, 437–444. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Zhang, S.H.; Wang, P.F.; Hou, J.; Zhang, W.J.; Li, W.; Lin, Z.P. The effect of excess Zn on mineral nutrition and antioxidative response in rapeseed seedlings. Chemosphere 2009, 75, 1468–1476. [Google Scholar] [CrossRef] [PubMed]
- Bauddh, K.; Singh, R.P. Growth, tolerance, efficiency and phytoremediation potential of Ricinuscommunis (L.) and Brassica juncea (L.) in salinity and drought affected cadmium contaminated soil. Ecotoxicol. Environ. Saf. 2012, 85, 13–22. [Google Scholar] [CrossRef] [PubMed]
- Jozefczak, M.; Keunen, E.; Schat, H.; Bliek, M.; Hernández, L.E.; Carleer, R.; Remans, T.; Bohler, S.; Vangronsveld, J.; Cuypers, A. Differential response of Arabidopsis leaves and roots to cadmium: Glutathione-related chelating capacity vs antioxidant capacity. Plant Physiol. Biochem. 2014, 83, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Nouairi, I.; Ammar, W.B.; Youssef, N.B.; Miled, D.D.B.; Ghorbal, M.H.; Zarrouk, M. Antioxidant defense system in leaves of Indian mustard (Brassica juncea) and rape (Brassica napus) under cadmium stress. Acta Physiol. Plant. 2009, 31, 237–247. [Google Scholar] [CrossRef]
- Zhang, F.Q.; Wang, Y.S.; Lou, Z.P.; Dong, J.D. Effect of heavy metal stress on antioxidative enzymes and lipid peroxidation in leaves and roots of two mangrove plant seedlings (Kandelia candel and Bruguiera gymnorrhiza). Chemosphere 2007, 67, 44–50. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Rajkumar, M.; Luo, Y.M.; Freitas, H. Inoculation of endophytic bacteria on host and non-host plants—Effects on plant growth and Ni uptake. J. Hazard. Mater. 2011, 195, 230–237. [Google Scholar] [CrossRef] [PubMed]
- Khan, Z.; Doty, S.L. Characterization of bacterial endophytes of sweet potato plants. Plant Soil 2009, 322, 197–207. [Google Scholar] [CrossRef]
- Mergeay, M.; Nies, D.; Schlegel, H.G.; Gerits, J.; Charles, P.; Van Gijsegem, F. Alcaligenes eutrophus CH34 is a facultative chemolithotroph with plasmid-bound resistgance to heavy metals. J. Bacteriol. 1985, 162, 328–334. [Google Scholar] [PubMed]
- Montalbán, B.; García-Gonzalo, P.; Pradas del Real, A.E.; Alonso, J.; Lobo, M.C.; Pérez-Sanz, A. Brachypodium distachyon tolerance to metals under in vitro conditions: A comparison with two metal-tolerant energy crops. Fresen. Environ. Bull. 2014, 23, 2086–2092. [Google Scholar]
- Reilly, C.A.; Aust, S.D. Measurement of lipid peroxidation. Curr. Protoc. Toxicol. 2001, 28, 659–671. [Google Scholar]
- Catalá, M.; Gasulla, F.; Pradas del Real, A.E.; Garcia-Breijo, F.; Reig-Arminana, J.; Barreno, E. Fungal-associated NO is involved in the regulation of oxidative stress during rehydration in lichen symbiosis. BMC Microbiol. 2010, 10, 297. [Google Scholar] [CrossRef] [PubMed]
- Schlegel, H.G.; Kaltwasser, H.; Gottschalk, G. Ein Submersverfahren zur Kultur wassers toffoxy dieren der Bakterien: Wachstums physiologische Untersuchungen. Arch. Mikrobiol. 1961, 38, 209–222. [Google Scholar] [CrossRef] [PubMed]

| Comp. 1 | Strain | Identification | Accesion | Zn | Cd | Fe 0 µM | Fe 0.25 µM | OA | ACC | IAA | Ace | Psol | N fix |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Soil | 222 | Arthrobacter sp. | KT461847 | +++ | +++ | − | − | ++ | +++ | − | − | − | + |
| Root | 228 | Pseudomonas sp. | KT461831 | ++ | ++ | + | + | + | ++ | + | − | ++ | − |
| Root | 246 | Serratia sp. | KT461863 | +++ | +++ | + | + | ++ | +++ | ++ | − | − | − |
| Root | 256 | Pseudomonas sp. | KT461831 | +++ | + | + | + | - | + | ++ | + | +++ | ++ |
| Root | 262 | Pseudomonas sp. | KT461831 | + | + | ++ | − | + | +++ | ++ | + | − | − |
| Treatments | VR | D19 | |||
|---|---|---|---|---|---|
| Zn | |||||
| Aerial | Root | Aerial | Root | ||
| Zn | Control | 58 ± 14a | 40 ± 10a | 75 ± 23a | 41 ± 5a |
| Non-inoculated | 1533 ± 149b | 4533 ± 945c | 1097 ± 175b | 3862 ± 1063bc | |
| Serratia sp. 246 | 1155 ± 23b | 4195 ± 355bc | 1283 ± 207b | 3455 ± 1767b | |
| Pseudomonas sp. 256 | 1349 ± 183b | 4368 ± 442bc | 1554 ± 299b | 3484 ± 651b | |
| Pseudomonas sp. 228 | 975 ± 154b | 2237 ± 368b | 1317 ± 177b | 3504 ± 1167b | |
| Cd | |||||
| Cd | Control | 0.43 ± 0.09a | 1.2 ± 0.2a | 0.6 ± 0.1a | 0.5 ± 0.2a |
| Non-inoculated | 152 ± 10c | 1118 ± 177def | 106 ± 44bc | 889 ± 196cde | |
| Arthrobacter sp. 222 | 83 ± 6bc | 492 ± 85bc | 58 ± 8b | 631 ± 140bc | |
| Pseudomonas sp. 228 | 112 ± 23bc | 1250 ± 320ef | 106 ± 18bc | 1365 ± 145f | |
| Serratia sp. 246 | 24 ± 4b | 908 ± 314cde | 129 ± 45c | 798 ± 65bcd | |
| Pseudomonas sp. 262 | 145 ± 29c | 487 ± 57bc | 81 ± 5bc | 383 ± 107b | |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Montalbán, B.; Thijs, S.; Lobo, M.C.; Weyens, N.; Ameloot, M.; Vangronsveld, J.; Pérez-Sanz, A. Cultivar and Metal-Specific Effects of Endophytic Bacteria in Helianthus tuberosus Exposed to Cd and Zn. Int. J. Mol. Sci. 2017, 18, 2026. https://doi.org/10.3390/ijms18102026
Montalbán B, Thijs S, Lobo MC, Weyens N, Ameloot M, Vangronsveld J, Pérez-Sanz A. Cultivar and Metal-Specific Effects of Endophytic Bacteria in Helianthus tuberosus Exposed to Cd and Zn. International Journal of Molecular Sciences. 2017; 18(10):2026. https://doi.org/10.3390/ijms18102026
Chicago/Turabian StyleMontalbán, Blanca, Sofie Thijs, Mª Carmen Lobo, Nele Weyens, Marcel Ameloot, Jaco Vangronsveld, and Araceli Pérez-Sanz. 2017. "Cultivar and Metal-Specific Effects of Endophytic Bacteria in Helianthus tuberosus Exposed to Cd and Zn" International Journal of Molecular Sciences 18, no. 10: 2026. https://doi.org/10.3390/ijms18102026
APA StyleMontalbán, B., Thijs, S., Lobo, M. C., Weyens, N., Ameloot, M., Vangronsveld, J., & Pérez-Sanz, A. (2017). Cultivar and Metal-Specific Effects of Endophytic Bacteria in Helianthus tuberosus Exposed to Cd and Zn. International Journal of Molecular Sciences, 18(10), 2026. https://doi.org/10.3390/ijms18102026