Urinary Metabolic Phenotyping Reveals Differences in the Metabolic Status of Healthy and Inflammatory Bowel Disease (IBD) Children in Relation to Growth and Disease Activity
Abstract
:1. Introduction
2. Results
2.1. Clinical Parameters of IBD and Healthy Subjects
2.2. Urine Metabonomics Describes Differences between IBD and Healthy Subjects
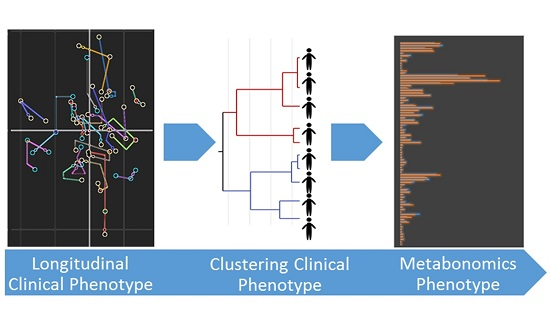
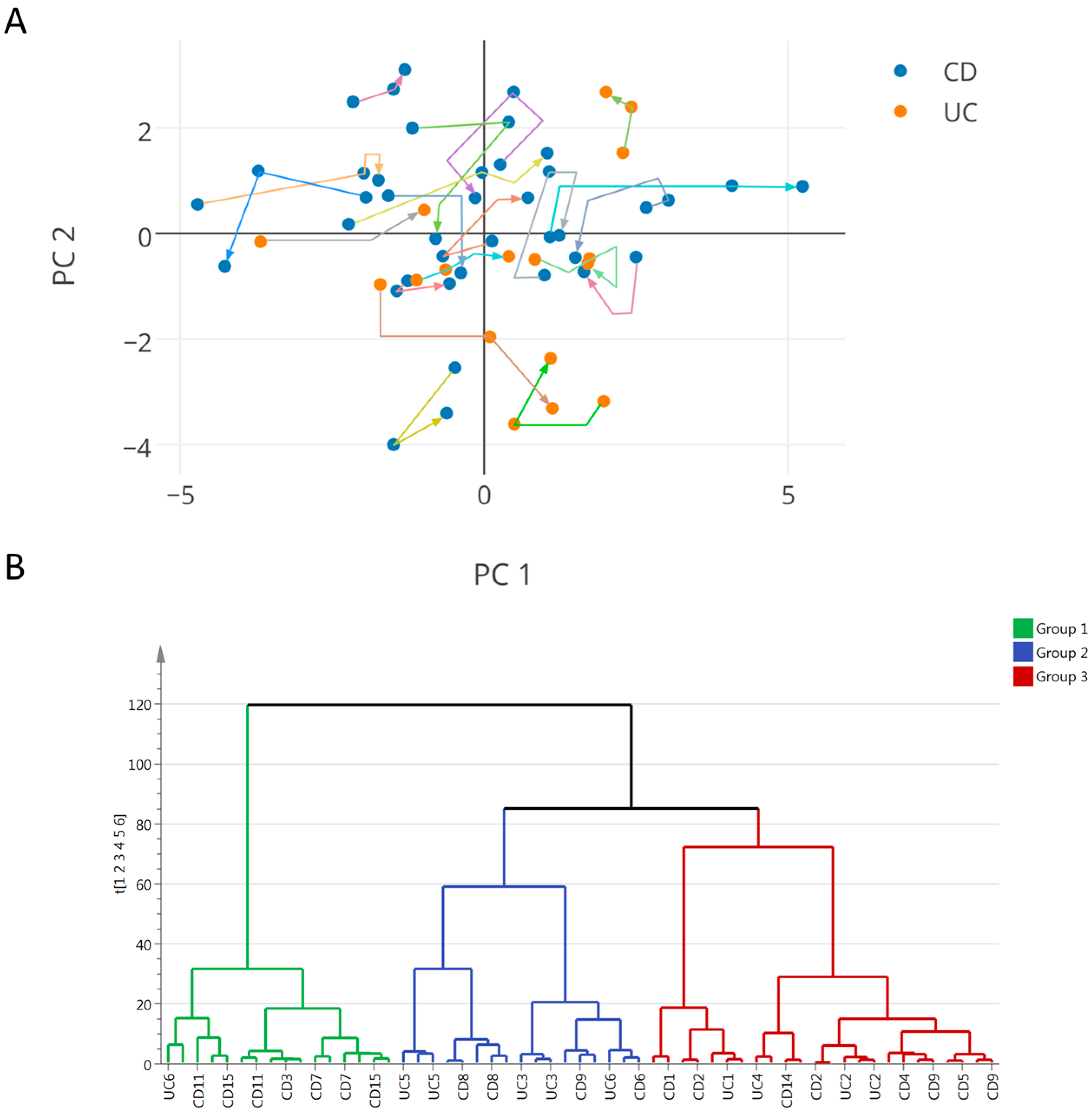
2.3. Integration of Clinical and Urine Metabonomics Data in IBD Patients
3. Discussion
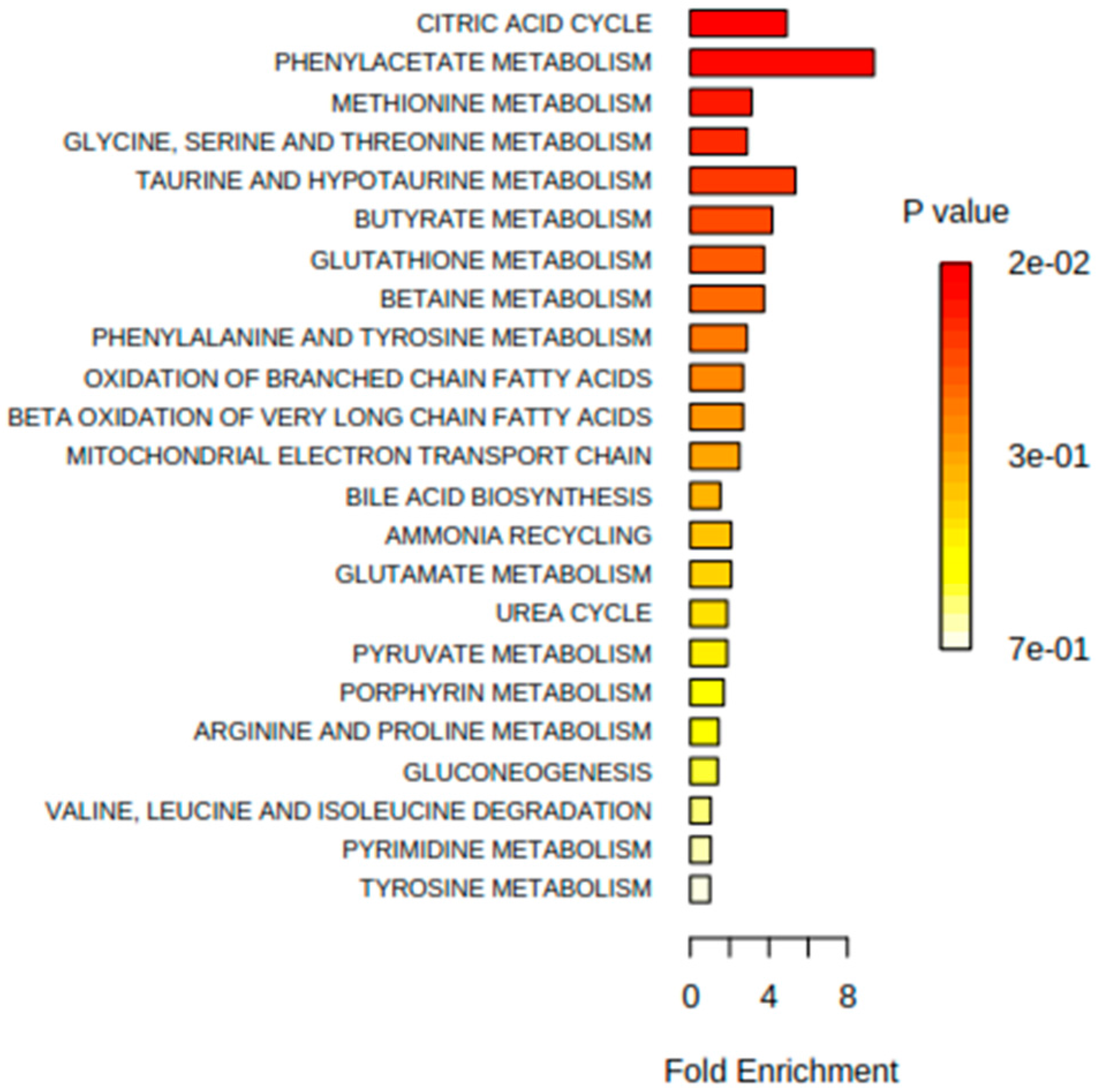
3.1. Urine Metabonomics Reflects Different Metabolic Requirements in Pediatric IBD Patients Compared to Healthy Subjects
3.2. IBD Clinical Sub-Phenotypes Link to Different Metabolic Status
3.3. Host-Gut Microbial Urinary Co-Metabolites Describe Relationships between Dietary Sources of Nitrogen, Carbamyl Phosphate Synthetase, and Host Metabolism
4. Materials and Methods
4.1. Subjects
4.2. Anthropometric and Clinical Measures
4.2.1. Anthropometric Assessment
4.2.2. Body Composition
4.2.3. Disease Activity in Patients with IBD
4.2.4. Blood and Stool Markers
4.2.5. Dietary Intake
4.2.6. Resting Energy Expenditure
4.3. Metabonomics Analysis
4.4. Statistical Analysis
4.5. Ethics
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Moco, S.; Candela, M.; Chuang, E.; Draper, C.; Cominetti, O.; Montoliu, I.; Barron, D.; Kussmann, M.; Brigidi, P.; Gionchetti, P.; et al. Systems biology approaches for inflammatory bowel disease: Emphasis on gut microbial metabolism. Inflamm. Bowel Dis. 2014, 20, 2104–2114. [Google Scholar] [CrossRef] [PubMed]
- Ezri, J.; Marques-Vidal, P.; Nydegger, A. Impact of disease and treatments on growth and puberty of pediatric patients with inflammatory bowel disease. Digestion 2012, 85, 308–319. [Google Scholar] [CrossRef] [PubMed]
- Sauer, C.G.; Kugathasan, S. Pediatric inflammatory bowel disease: Highlighting pediatric differences in IBD. Gastroenterol. Clin. N. Am. 2009, 38, 611–628. [Google Scholar] [CrossRef] [PubMed]
- Markowitz, J.; Grancher, K.; Rosa, J.; Aiges, H.; Daum, F. Growth failure in pediatric inflammatory bowel disease. J. Pediatr. Gastroenterol. Nutr. 1993, 16, 373–380. [Google Scholar] [CrossRef] [PubMed]
- Diamanti, A.; Basso, M.S.; Gambarara, M.; Papadatou, B.; Bracci, F.; Noto, C.; Castro, M. Positive impact of blocking tumor necrosis factor alpha on the nutritional status in pediatric crohn’s disease patients. Int. J. Colorect. Dis. 2009, 24, 19–25. [Google Scholar] [CrossRef] [PubMed]
- Pfefferkorn, M.; Burke, G.; Griffiths, A.; Markowitz, J.; Rosh, J.; Mack, D.; Otley, A.; Kugathasan, S.; Evans, J.; Bousvaros, A.; et al. Growth abnormalities persist in newly diagnosed children with crohn disease despite current treatment paradigms. J. Pediatr. Gastroenterol. Nutr. 2009, 48, 168–174. [Google Scholar] [CrossRef] [PubMed]
- Sawczenko, A.; Ballinger, A.B.; Savage, M.O.; Sanderson, I.R. Clinical features affecting final adult height in patients with pediatric-onset crohn’s disease. Pediatrics 2006, 118, 124–129. [Google Scholar] [CrossRef] [PubMed]
- Shamir, R.; Phillip, M.; Levine, A. Growth retardation in pediatric crohn’s disease: Pathogenesis and interventions. Inflamm. Bowel Dis. 2007, 13, 620–628. [Google Scholar] [CrossRef] [PubMed]
- Day, A.S.; Ledder, O.; Leach, S.T.; Lemberg, D.A. Crohn’s and colitis in children and adolescents. World J. Gastroenterol. 2012, 18, 5862–5869. [Google Scholar] [CrossRef] [PubMed]
- Powell-Tuck, J. Nutritional consequences of gastrointestinal disease. Bailliere’s Clin. Gastroenterol. 1988, 2, 715–727. [Google Scholar] [CrossRef]
- Rigaud, D.; Angel, L.A.; Cerf, M.; Carduner, M.J.; Melchior, J.C.; Sautier, C.; Rene, E.; Apfelbaum, M.; Mignon, M. Mechanisms of decreased food intake during weight loss in adult crohn’s disease patients without obvious malabsorption. Am. J. Clin. Nutr. 1994, 60, 775–781. [Google Scholar] [PubMed]
- Ballinger, A. Fundamental mechanisms of growth failure in inflammatory bowel disease. Horm. Res. 2002, 58, 7–10. [Google Scholar] [CrossRef] [PubMed]
- Nicholson, J.K.; Wilson, I.D. High resolution proton NMR spectroscopy of biological fluids. Prog. Nucl. Magn. Reson. Spectrosc. 1989, 21, 449–501. [Google Scholar] [CrossRef]
- Nicholson, J.K.; Lindon, J.C.; Holmes, E. ‘Metabonomics’: Understanding the metabolic responses of living systems to pathophysiological stimuli via multivariate statistical analysis of biological NMR spectroscopic data. Xenobiotica 1999, 29, 1181–1189. [Google Scholar] [CrossRef] [PubMed]
- Martin, F.P.; Collino, S.; Rezzi, S.; Kochhar, S. Metabolomic applications to decipher gut microbial metabolic influence in health and disease. Front. Physiol. 2012, 3, 113. [Google Scholar] [CrossRef] [PubMed]
- Nicholson, J.K.; Holmes, E.; Kinross, J.M.; Darzi, A.W.; Takats, Z.; Lindon, J.C. Metabolic phenotyping in clinical and surgical environments. Nature 2012, 491, 384–392. [Google Scholar] [CrossRef] [PubMed]
- Montoliu, I.; Martin, F.P.; Collino, S.; Rezzi, S.; Kochhar, S. Multivariate modeling strategy for intercompartmental analysis of tissue and plasma (1) h NMR spectrotypes. J. Proteome Res. 2009, 8, 2397–2406. [Google Scholar] [CrossRef] [PubMed]
- Eriksson, L.; Johansson, E.; Kettanah-Wold, N.; Wold, S. Multi and Megavariate Data Analysis—Principles and Methods; Umetrics Academy: Malmö, Sweden, 1999. [Google Scholar]
- Baur, P.; Martin, F.P.; Gruber, L.; Bosco, N.; Brahmbhatt, V.; Collino, S.; Guy, P.; Montoliu, I.; Rozman, J.; Klingenspor, M.; et al. Metabolic phenotyping of the Crohn’s disease-like IBD etiopathology in the tnf(deltaare/wt) mouse model. J. Proteome Res. 2011, 10, 5523–5535. [Google Scholar] [CrossRef] [PubMed]
- Bosco, N.; Brahmbhatt, V.; Oliveira, M.; Martin, F.P.; Lichti, P.; Raymond, F.; Mansourian, R.; Metairon, S.; Pace-Asciak, C.; Bastic Schmid, V.; et al. Effects of increase in fish oil intake on intestinal eicosanoids and inflammation in a mouse model of colitis. Lipids Health Dis. 2013, 12, 81. [Google Scholar] [CrossRef] [PubMed]
- Martin, F.P.; Lichti, P.; Bosco, N.; Brahmbhatt, V.; Oliveira, M.; Haller, D.; Benyacoub, J. Metabolic phenotyping of an adoptive transfer mouse model of experimental colitis and impact of dietary fish oil intake. J. Proteome Res. 2015, 14, 1911–1919. [Google Scholar] [CrossRef] [PubMed]
- Claus, S.P.; Swann, J.R. Nutrimetabonomics: Applications for nutritional sciences, with specific reference to gut microbial interactions. Annu. Rev. Food Sci. Technol. 2013, 4, 381–399. [Google Scholar] [CrossRef] [PubMed]
- Holmes, E.; Li, J.V.; Marchesi, J.R.; Nicholson, J.K. Gut microbiota composition and activity in relation to host metabolic phenotype and disease risk. Cell Metab. 2012, 16, 559–564. [Google Scholar] [CrossRef] [PubMed]
- Xia, J.; Sinelnikov, I.V.; Han, B.; Wishart, D.S. Metaboanalyst 3.0-making metabolomics more meaningful. Nucleic Acids Res. 2015, 43, W251–W257. [Google Scholar] [CrossRef] [PubMed]
- Heuschkel, R.; Salvestrini, C.; Beattie, R.M.; Hildebrand, H.; Walters, T.; Griffiths, A. Guidelines for the management of growth failure in childhood inflammatory bowel disease. Inflamm. Bowel Dis. 2008, 14, 839–849. [Google Scholar] [CrossRef] [PubMed]
- Hill, R.J.; Cleghorn, G.J.; Withers, G.D.; Lewindon, P.J.; Ee, L.C.; Connor, F.; Davies, P.S. Resting energy expenditure in children with inflammatory bowel disease. J. Pediatr. Gastroenterol. Nutr. 2007, 45, 342–346. [Google Scholar] [CrossRef] [PubMed]
- Steiner, S.J.; Pfefferkorn, M.D.; Fitzgerald, J.F.; Denne, S.C. Protein and energy metabolism response to the initial dose of infliximab in children with Crohn’s disease. Inflamm. Bowel Dis. 2007, 13, 737–744. [Google Scholar] [CrossRef] [PubMed]
- Mokhtarani, M.; Diaz, G.A.; Rhead, W.; Lichter-Konecki, U.; Bartley, J.; Feigenbaum, A.; Longo, N.; Berquist, W.; Berry, S.A.; Gallagher, R.; et al. Urinary phenylacetylglutamine as dosing biomarker for patients with urea cycle disorders. Mol. Genet. Metab. 2012, 107, 308–314. [Google Scholar] [CrossRef] [PubMed]
- Brusilow, S.W. Phenylacetylglutamine may replace urea as a vehicle for waste nitrogen excretion. Pediatr. Res. 1991, 29, 147–150. [Google Scholar] [CrossRef] [PubMed]
- Zimmerman, L.; Egestad, B.; Jornvall, H.; Bergstrom, J. Identification and determination of phenylacetylglutamine, a major nitrogenous metabolite in plasma of uremic patients. Clin. Nephrol. 1989, 32, 124–128. [Google Scholar] [PubMed]
- Schicho, R.; Shaykhutdinov, R.; Ngo, J.; Nazyrova, A.; Schneider, C.; Panaccione, R.; Kaplan, G.G.; Vogel, H.J.; Storr, M. Quantitative metabolomic profiling of serum, plasma, and urine by (1)h NMR spectroscopy discriminates between patients with inflammatory bowel disease and healthy individuals. J. Proteome Res. 2012, 11, 3344–3357. [Google Scholar] [CrossRef] [PubMed]
- Stephens, N.S.; Siffledeen, J.; Su, X.; Murdoch, T.B.; Fedorak, R.N.; Slupsky, C.M. Urinary nmr metabolomic profiles discriminate inflammatory bowel disease from healthy. J. Crohns Colitis 2013, 7, e42–e48. [Google Scholar] [CrossRef] [PubMed]
- Williams, H.R.; Cox, I.J.; Walker, D.G.; North, B.V.; Patel, V.M.; Marshall, S.E.; Jewell, D.P.; Ghosh, S.; Thomas, H.J.; Teare, J.P.; et al. Characterization of inflammatory bowel disease with urinary metabolic profiling. Am. J. Gastroenterol. 2009, 104, 1435–1444. [Google Scholar] [CrossRef] [PubMed]
- Le Chatelier, E.; Nielsen, T.; Qin, J.; Prifti, E.; Hildebrand, F.; Falony, G.; Almeida, M.; Arumugam, M.; Batto, J.M.; Kennedy, S.; et al. Richness of human gut microbiome correlates with metabolic markers. Nature 2013, 500, 541–546. [Google Scholar] [CrossRef] [PubMed]
- Forbes, G.B.; Bruining, G.J. Urinary creatinine excretion and lean body mass. Am. J. Clin. Nutr. 1976, 29, 1359–1366. [Google Scholar] [PubMed]
- Richmond, A.L.; Kabi, A.; Homer, C.R.; Marina-Garcia, N.; Nickerson, K.P.; Nesvizhskii, A.I.; Sreekumar, A.; Chinnaiyan, A.M.; Nunez, G.; McDonald, C. The nucleotide synthesis enzyme CAD inhibits nod2 antibacterial function in human intestinal epithelial cells. Gastroenterology 2012, 142, 1483–1492. [Google Scholar] [CrossRef] [PubMed]
- Lees, H.J.; Swann, J.R.; Wilson, I.D.; Nicholson, J.K.; Holmes, E. Hippurate: The natural history of a mammalian-microbial cometabolite. J. Proteome Res. 2013, 12, 1527–1546. [Google Scholar] [CrossRef] [PubMed]
- Moco, S.; Martin, F.P.; Rezzi, S. A metabolomics view on gut microbiome modulation by polyphenol-rich foods. J. Proteome Res. 2012, 11, 4781–4790. [Google Scholar] [CrossRef] [PubMed]
- Wong, J.M.; de, S.R.; Kendall, C.W.; Emam, A.; Jenkins, D.J. Colonic health: Fermentation and short chain fatty acids. J. Clin. Gastroenterol. 2006, 40, 235–243. [Google Scholar] [CrossRef] [PubMed]
- Levine, A.; Koletzko, S.; Turner, D.; Escher, J.C.; Cucchiara, S.; de Ridder, L.; Kolho, K.L.; Veres, G.; Russell, R.K.; Paerregaard, A.; et al. Espghan revised porto criteria for the diagnosis of inflammatory bowel disease in children and adolescents. J. Pediatr. Gastroenterol. Nutr. 2014, 58, 795–806. [Google Scholar] [CrossRef] [PubMed]
- De Onis, M.; Onyango, A.; Borghi, E.; Siyam, A.; Nishida, C.; Siekmann, J. Development of a who growth reference for scholl-aged children and adolescents. Bull. World Health Organ. 2007, 85, 660–667. [Google Scholar] [CrossRef] [PubMed]
- Lohman, T.; Roche, A.; Martorell, R. Anthropometric StandarDisation Reference Manual; Human Kinetics Books: Champaign, IL, USA, 1988. [Google Scholar]
- Tanner, J.M.; Whitehouse, R.H. Clinical longitudinal standards for height, weight, height velocity, weight velocity, and stages of puberty. Arch. Dis. Child. 1976, 51, 170–179. [Google Scholar] [CrossRef] [PubMed]
- Pietrobelli, A.; Andreoli, A.; Cervelli, V.; Carbonelli, M.G.; Peroni, D.G.; de Lorenzo, A. Predicting fat-free mass in children using bioimpedance analysis. Acta Diabetol. 2003, 40, S212–S215. [Google Scholar] [CrossRef] [PubMed]
- Houtkooper, L.B.; Lohman, T.G.; Going, S.B.; Hall, M.C. Validity of bioelectric impedance for body composition assessment in children. J. Appl. Physiol. 1989, 66, 814–821. [Google Scholar] [PubMed]
- Hyams, J.S.; Ferry, G.D.; Mandel, F.S.; Gryboski, J.D.; Kibort, P.M.; Kirschner, B.S.; Griffiths, A.M.; Katz, A.J.; Grand, R.J.; Boyle, J.T.; et al. Development and validation of a pediatric Crohn’s disease activity index. J. Pediatr. Gastroenterol. Nutr. 1991, 12, 439–447. [Google Scholar] [CrossRef] [PubMed]
- Turner, D.; Otley, A.R.; Mack, D.; Hyams, J.; de Bruijne, J.; Uusoue, K.; Walters, T.D.; Zachos, M.; Mamula, P.; Beaton, D.E.; et al. Development, validation, and evaluation of a pediatric ulcerative colitis activity index: A prospective multicenter study. Gastroenterology 2007, 133, 423–432. [Google Scholar] [CrossRef] [PubMed]
- Diamanti, A.; Colistro, F.; Basso, M.S.; Papadatou, B.; Francalanci, P.; Bracci, F.; Muraca, M.; Knafelz, D.; de Angelis, P.; Castro, M. Clinical role of calprotectin assay in determining histological relapses in children affected by inflammatory bowel diseases. Inflamm. Bowel Dis. 2008, 14, 1229–1235. [Google Scholar] [CrossRef] [PubMed]
- Trygg, J.; Wold, S. O2-PLS, a two-block (x–y) latent variable regression (LVR) method with an integrated osc filter. J. Chemom. 2003, 17, 53–64. [Google Scholar] [CrossRef]
| Group | Healthy Controls | Crohn’s Disease (CD) | Ulcerative Colitis (UC) | ||||
|---|---|---|---|---|---|---|---|
| Visit | T0 | T0 | T6 | T12 | T0 | T6 | T12 |
| N total (males) | 27 (16) | 15 (8) | 14 (8) | 12 (7) | 5 * (2) | 6 (2) | 6 (2) |
| Age (Years) | 12.9 ± 1.9 (10.1–16.7) | 14.9 ± 1.3 b (12.4–16.7) | 15.2 ± 1.2 (12.9–17.2) | 15.7 ± 1.3 (13.4–17.7) | 15 ± 1.2 a (12.7–16.1) | 15.2 ± 1.3 (13.1–16.7) | 15.8 ± 1.2 (13.8–17.2) |
| Tanner Score | 3 ± 1 (1–5) | 3 ± 1 (2–5) | 4 ± 1 (2–5) | 4 ± 1(2–5) | 4 ± 1 (3–5) | 4 ± 1 (2–5) | 4 ± 1 (3–5) |
| Weight z-score | 0.5 ± 1 (−2.1–1.8) | −0.7 ± 0.9 b (−2.1–1.2) | −0.7 ± 0.8 (−1.8–0.9) | −0.7 ± 0.9 (−2.2–0.6) | −0.1 ± 0.9 (−1.3–1.2) | −0.3 ± 1 (−1.5–0.9) | −0.1 ± 1 (−1.6–1) |
| Height z-score | 0.5 ± 0.9 (−1.7–2.2) | −0.8 ± 1 b (−3.2–0.5) | −0.7 ± 1 (−3.1–1.1) | −0.5 ± 1.1 (−2.5–1.7) | −0.1 ± 1.5 (−2.4–1.8) | −0.3 ± 1.2 (−2.1–1.6) | −0.2 ± 1.1 (−1.8–1.5) |
| BMI z-score | 0.4 ± 0.9 (−1.7–1.6) | −0.5 ± 1.2 a (−2.8–1.7) | −0.5 ± 1 (−2.3–1.4) | −0.7 ± 1.1 (−2.7–1.1) | 0 ± 0.9 (−1.1–1.1) | −0.2 ± 1 (−1.3–1.2) | −0.1 ± 1 (−1.3–1.6) |
| GV z-score | NA | 0.8 ± 1.7 (−1.4–3.8) | 0.7 ± 1.6 (−1.1–4.3) | 0.9 ± 1.7 (−1–4.6) | 0.1 ± 0.6 (−0.4–1.4) | −0.3 ± 1.4 (−1.8–2.1) | 0.4 ± 1.2 (−0.8–2.7) |
| %FFM | 39.6 ± 10.9 (23.5–60.6) | 35.0 ± 5.1 (26.7–43.7) | 37.3 ± 5.4 (27.7–48.1) | 38.9 ± 7.3 (29.7–57.7) | 37.8 ± 4.1 (33.4–43.8) | 36.7 ± 4.2 (27.8–40.2) | 39.4 ± 5.1 (29.6–44.8) |
| REE (Kcal) | 1531.2 ± 275.6 (958–2036) | 1338.1 ± 147 a (1065–1702) | 1381.3 ± 188.6 (1050–1824) | 1374 ± 213.2 (1177–1942) | 1472.6 ± 86.6 (1362–1561) | 1355.3 ± 201.8 (1118–1599) | 1467.7 ± 174.6 (1249–1761) |
| Blood Urea (mmol/L) | 438.8 ± 109.9 (116.8–647.9) | 379.7 ± 111.2 (208.5–548.4) | 395.9 ± 101.2 (224.4–541.5) | 401.3 ± 123.4 (189.8–615.6) | 394.4 ± 86 (310.7–545.7) | 382.1 ± 145.1 (156.1–586.4) | 389.5 ± 182.1 (90.7–598.6) |
| PCDAI in CD/PUCAI in UC | NA | 9.8 ± 9 (0-30) | 7.7 ± 7 (0-22.5) | 6.3 ± 8.9 (0-25) | 5 ± 4.5 (0-10) | 5.8 ± 5.3 (0-15) | 3 ± 4 (0-10) |
| ESR (mm/h) | NA | 15.1 ± 8.5 (2–32) | 16 ± 17.8 (3–70) | 18 ± 20.2 (1–70) | 26.6 ± 25.9 (9–78) | 33.2 ± 31 (10–94) | 23.8 ± 12.2 (13–47) |
| CRP ** (mg/L) | NA | 3.5 ± 1.9 (2–8) | 7.1 ± 10.1 (2–35) | 11.1 ± 17.3 (2–60) | 5 ± 3.3 (2–11) | 6.5 ± 6.5 (2–18) | 7.7 ± 11.4 (1–33) |
| Fecal calprotectin (µg/g) | NA | 660.8 ± 673.9 (10–1500) | 372.3 ± 464.4 (20–1500) | 714.8 ± 643 (20–1500) | 1046.7 ± 501.5 (367–1500) | 966.7 ± 644.2 (20–1500) | 1500 ± 0 (1500–1500) |
| IGF-1 z-score | NA | −0.6 ± 0.4 (−1.1–0.3) | −0.7 ± 0.3 (−1.1–0.0) | −0.7 ± 0.3 (−1.2–0.1) | −0.5 ± 0.5 (−1.0–0.1) | −0.4 ± 0.5 (−0.9–0.4) | −0.4 ± 0.4 (−0.9–0.4) |
| IGFBP-3 z-score | NA | −0.5 ± 0.2 (−0.9–−0.2) | −0.5 ± 0.1 (−0.8–−0.3) | −0.5 ± 0.2 (−0.7–0.1) | −0.4 ± 0.5 (−1.0–0.0) | −0.4 ± 0.4 (−0.8–0.1) | −0.4 ± 0.3 (−0.8–0.1) |
| Group | Healthy Controls | CD | UC | ||||
|---|---|---|---|---|---|---|---|
| Metabolites (a.u.)/Visit | T0 | T0 | T6 | T12 | T0 | T6 | T12 |
| Uk1 | 4.6 ± 2.5 | 4.1 ± 2.3 | 4.3 ± 2.0 | 3.2 ± 1.2 b | 3.7 ± 1.5 | 6.2 ± 3.2 | 7.9 ± 7.5 b |
| Uk2 | 13.1 ± 1.2 | 13.5 ± 1.6 | 13.4 ± 1.9 | 14.2 ± 3.5 | 15.4 ± 3.2 a | 13.8 ± 1.5 | 16.4 ± 5.1 a |
| Uk3 | 40.9 ± 4.6 | 59.7 ± 53.0 b | 50 ± 22.0 a | 96.7 ± 150.9 b | 184.4 ± 166.3 a | 90 ± 83.8 a | 263.9 ± 259.6 a |
| Methanol | 37.4 ± 8.7 | 36.0 ± 12.3 | 30.2 ± 6.6 a | 33.9 ± 9.3 | 35.0 ± 4.0 | 35.5 ± 14.3 | 39.7 ± 17.7 |
| Acyl-carnitine | 95.7 ± 15.6 | 99.3 ± 22.1 | 108.6 ± 30.6 b | 99 ± 27.4 | 93.6 ± 18.3 | 81.9 ± 16.6 b | 82.9 ± 12.9 b |
| cis-Aconitate | 36.4 ± 4.6 | 31.1 ± 7.7 a | 30.2 ± 6.9 a | 30.1 ± 5.6 a | 30.2 ± 1.7 a | 29.2 ± 3.0 a | 27.9 ± 4.7 a |
| Betaine | 179.9 ± 62.3 | 215.2 ± 228.6 | 176.8 ± 86.9 | 163.1 ± 55.6 | 202.7 ± 53.3 | 144.5 ± 68.5 | 165.5 ± 78.9 |
| Urea | 380.0 ± 124.8 | 281.3 ± 153 a | 227.4 ± 139.9 a | 258.5 ± 147.7 a | 357.9 ± 89.6 | 289.1 ± 139.1 | 273.1 ± 142.5 b |
| 4-Hydroxyphenylacetate | 3.4 ± 0.6 | 3.4 ± 0.7 | 3.1 ± 0.9 | 10.5 ± 20.1 b | 11.8 ± 14.2 a | 7.3 ± 6.6 a | 24.9 ± 28.7 a |
| 4-Hydroxyphenylpyruvate | 13.3 ± 6.0 | 12.8 ± 4.8 | 9.6 ± 3.9 b | 20.8 ± 22.5 | 31.2 ± 26.5 a | 14.1 ± 10.3 | 35.2 ± 28.6 a |
| Phenylacetylglutamine | 6.9 ± 1.1 | 9.0 ± 4.5 a | 7.5 ± 1.2 b | 9.6 ± 3.8 a | 8.9 ± 1.6 a | 13.1 ± 12.4 a | 9.6 ± 2.5 a |
| Tryptophan | 6.6 ± 2.6 | 8.2 ± 7.0 | 7.4 ± 10.1 | 17.9 ± 43.9 | 49.2 ± 51.7 a | 20.3 ± 29.0 a | 68.4 ± 74.9 a |
| Hippurate | 140.9 ± 92.3 | 53.6 ± 32.8 a | 57.3 ± 45.6 a | 81.5 ± 69.6 b | 67.1 ± 32.1 b | 62.8 ± 59.6 b | 64.4 ± 50.2 b |
| Glycine | 91.7 ± 32.2 | 102 ± 39.4 | 103.5 ± 35.4 | 119.7 ± 68 b | 106.1 ± 38.9 | 128.6 ± 59 a | 122.9 ± 71.3 |
| Taurine | 117.2 ± 28.7 | 99.9 ± 37.0 | 118.1 ± 33.4 | 94.8 ± 37.1 a | 101.8 ± 35.9 | 81.8 ± 30 a | 112.7 ± 68 |
| Mannitol | 364.7 ± 67.9 | 404.5 ± 77.9 b | 379.7 ± 44.5 | 369 ± 30.4 | 379.4 ± 28.6 | 391.1 ± 95.2 | 351.7 ± 79 |
| Carnitine | 57.1 ± 31.3 | 35.4 ± 15.7 a | 51.4 ± 47.0 | 27.6 ± 7.9 a | 44.2 ± 27.6 | 41.8 ± 13.8 | 30.4 ± 12.6 b |
| Succinate | 27.2 ± 4.9 | 24.6 ± 5.9 | 24.6 ± 4.8 | 26.3 ± 6.4 | 22.1 ± 3.8 a | 28.8 ± 9.5 | 23.9 ± 3.7 |
| 3-Methyl-2-oxovalerate | 16.1 ± 1.6 | 16.9 ± 2.6 | 17.4 ± 3.4 | 15.9 ± 2.1 | 15.5 ± 1.5 | 35.6 ± 46.9 a | 20.3 ± 11 a |
| 3-Hydroxyisobutyrate | 15.0 ± 2.5 | 13.2 ± 3.2 a | 13.9 ± 3.9 | 13.4 ± 3.4 | 12.6 ± 1.8 a | 35.9 ± 55.4 a | 17.4 ± 9.4 |
| 2-Oxoisocaproate | 21.7 ± 3.3 | 22.3 ± 3.9 | 22.5 ± 2.4 | 21.0 ± 3.4 | 20.1 ± 3.5 | 31.0 ± 20.0 a | 26.5 ± 9.6 a |
| Citrate | 180.9 ± 54.3 | 168 ± 79.2 | 178.1 ± 65.4 | 180.9 ± 85.0 | 152.3 ± 31.1 | 197.4 ± 156 | 196.5 ± 58 |
| Creatinine | 862.7 ± 152.6 | 866.3 ± 152.1 | 856.9 ± 152.6 | 844.7 ± 107.4 | 832 ± 101.7 | 841.0 ± 263.3 | 846.6 ± 140.5 |
| 3-aminoisobutyrate | 17.9 ± 2.6 | 17.3 ± 3.2 | 18.9 ± 4.9 | 17.9 ± 4.3 | 18.2 ± 2.5 | 20.9 ± 9.4 | 18.4 ± 2.6 |
| Lactate | 49.9 ± 12.5 | 52.3 ± 8.9 | 156.4 ± 372.2 | 51.2 ± 12.1 | 47.9 ± 6.1 | 69.8 ± 50.5 b | 76.6 ± 67.6 b |
| Formate | 5.7 ± 1.8 | 4.6 ± 2.4 | 4.5 ± 2.7 | 4 ± 1.7 a | 4.6 ± 1.4 | 17.7 ± 30.4 a | 4.6 ± 1.7 |
| Clinical Parameters | Group 1 (6% UC) | Group 2 (53% UC) | Group 3 (30% UC) | Healthy | p-Values |
|---|---|---|---|---|---|
| Weight z-score | −1.5 ± 0.5 | 0.4 ± 0.6 | −0.7 ± 0.7 | 0.5 ± 1 | x,y,z,a,c |
| Height z-score | −0.8 ± 0.4 | -0.2 ± 1.2 | −0.7 ± 1.3 | 0.5 ± 0.9 | x,a,b,c |
| BMI z-score | −1.6 ± 0.7 | 0.5 ± 0.8 | −0.4 ± 0.7 | 0.4 ± 0.9 | x,y,z,a,c |
| GV z-score | −0.2 ± 0.9 | 0.4 ± 1.5 | 1.2 ± 1.8 | NA | z |
| %FFM (kg on the weight) | 32.3 ± 3.7 | 38.1 ± 4 | 39.7 ± 6.3 | 39.8 ± 11.3 | x,z,a |
| REE (kcal) | 1284.9 ± 128.6 | 1452.9 ± 152.9 | 1401.6 ± 216.4 | 1537.1 ± 284.8 | x,a |
| Blood urea (mmol/L) | 381.7 ± 128.9 | 321.1 ± 133.6 | 442 ± 90.4 | 437.9 ± 114.1 | y,b |
| Caloric intake (Kcal/day) | 2094.1 ± 422.5 | 1504.3 ± 256.5 | 1834 ± 581.6 | 1935 ± 461.1 | x,y,b |
| ESR (mm/h) | 21.6 ± 19.2 | 32.2 ± 26.3 | 9.8 ± 5.5 | NA | y,z |
| CRP (mg/L) | 11.6 ± 16.9 | 8.4 ± 9.3 | 2.7 ± 1.4 | NA | y,z |
| Fecal calprotectin (µg/g) | 689.2 ± 614.7 | 1086.4 ± 598.1 | 516.7 ± 635.2 | NA | y |
| IGF-1 z-score | −0.7 ± 0.2 | −0.4 ± 0.5 | −0.6 ± 0.3 | NA | x |
| IGFBP-3 z-score | −0.5 ± 0.2 | −0.4 ± 0.3 | −0.5 ± 0.2 | NA | x |
| Metabolites (a.u.) | Group 1 | Group 2 | Group 3 | Healthy Controls | p-Values |
|---|---|---|---|---|---|
| Uk3 | 44.7 ± 5.6 | 142.7 ± 186.2 | 108.8 ± 131.2 | 40.9 ± 4.6 | x,a,b,c |
| Methanol | 36.5 ± 11.9 | 39.9 ± 12.8 | 29.4 ± 5.6 | 37.4 ± 8.7 | y,z,c |
| Acyl-carnitine | 115 ± 26.7 | 90.6 ± 16 | 90.2 ± 23.5 | 95.7 ± 15.6 | x,z,a |
| cis-Aconitate | 31.5 ± 5.2 | 30.3 ± 7.5 | 28.9 ± 5.1 | 36.4 ± 4.6 | a,b,c |
| Urea | 295.2 ± 157.1 | 308.1 ± 111.7 | 228.4 ± 140.6 | 380 ± 124.8 | c |
| 4-Hydroxyphenylacetate | 4.3 ± 3.6 | 11.7 ± 19.6 | 8.5 ± 15.2 | 3.4 ± 0.6 | b |
| 4-Hydroxyphenylpyruvate | 10 ± 5.8 | 24 ± 23.9 | 18.8 ± 16.6 | 13.3 ± 6 | x,z,b |
| Phenylacetylglutamine | 7.8 ± 2.2 | 11.5 ± 8 | 8.7 ± 2.7 | 6.9 ± 1.1 | b,c |
| Tryptophane | 5.1 ± 2.2 | 32.5 ± 54.2 | 24.1 ± 39 | 6.6 ± 2.6 | b,c |
| Hippurate | 57.9 ± 37.2 | 55.2 ± 60.1 | 73.4 ± 47.2 | 140.9 ± 92.3 | a,b,c |
| Glycine | 97.7 ± 31.5 | 108 ± 43.2 | 123 ± 62.9 | 91.7 ± 32.2 | c |
| Taurine | 106.8 ± 33.7 | 95.3 ± 38.8 | 104.9 ± 44.3 | 117.2 ± 28.7 | b |
| Mannitol | 381.7 ± 53.7 | 411.5 ± 67.7 | 361.8 ± 56.5 | 364.7 ± 67.9 | y,b |
| Carnitine | 38.1 ± 21.2 | 36.2 ± 11.9 | 39.9 ± 36.5 | 57.1 ± 31.3 | a,b |
| Succinate | 28.2 ± 5.9 | 23.6 ± 7 | 24.2 ± 4.4 | 27.2 ± 4.9 | z,c |
| Creatinine | 785.6 ± 107.6 | 826.1 ± 179.3 | 914.3 ± 125.3 | 862.7 ± 152.6 | z |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martin, F.-P.; Ezri, J.; Cominetti, O.; Da Silva, L.; Kussmann, M.; Godin, J.-P.; Nydegger, A. Urinary Metabolic Phenotyping Reveals Differences in the Metabolic Status of Healthy and Inflammatory Bowel Disease (IBD) Children in Relation to Growth and Disease Activity. Int. J. Mol. Sci. 2016, 17, 1310. https://doi.org/10.3390/ijms17081310
Martin F-P, Ezri J, Cominetti O, Da Silva L, Kussmann M, Godin J-P, Nydegger A. Urinary Metabolic Phenotyping Reveals Differences in the Metabolic Status of Healthy and Inflammatory Bowel Disease (IBD) Children in Relation to Growth and Disease Activity. International Journal of Molecular Sciences. 2016; 17(8):1310. https://doi.org/10.3390/ijms17081310
Chicago/Turabian StyleMartin, Francois-Pierre, Jessica Ezri, Ornella Cominetti, Laeticia Da Silva, Martin Kussmann, Jean-Philippe Godin, and Andreas Nydegger. 2016. "Urinary Metabolic Phenotyping Reveals Differences in the Metabolic Status of Healthy and Inflammatory Bowel Disease (IBD) Children in Relation to Growth and Disease Activity" International Journal of Molecular Sciences 17, no. 8: 1310. https://doi.org/10.3390/ijms17081310
APA StyleMartin, F.-P., Ezri, J., Cominetti, O., Da Silva, L., Kussmann, M., Godin, J.-P., & Nydegger, A. (2016). Urinary Metabolic Phenotyping Reveals Differences in the Metabolic Status of Healthy and Inflammatory Bowel Disease (IBD) Children in Relation to Growth and Disease Activity. International Journal of Molecular Sciences, 17(8), 1310. https://doi.org/10.3390/ijms17081310