Environmental Factors Associated with Altered Gut Microbiota in Children with Eczema: A Systematic Review
Abstract
:1. Introduction
2. Methodology of Review
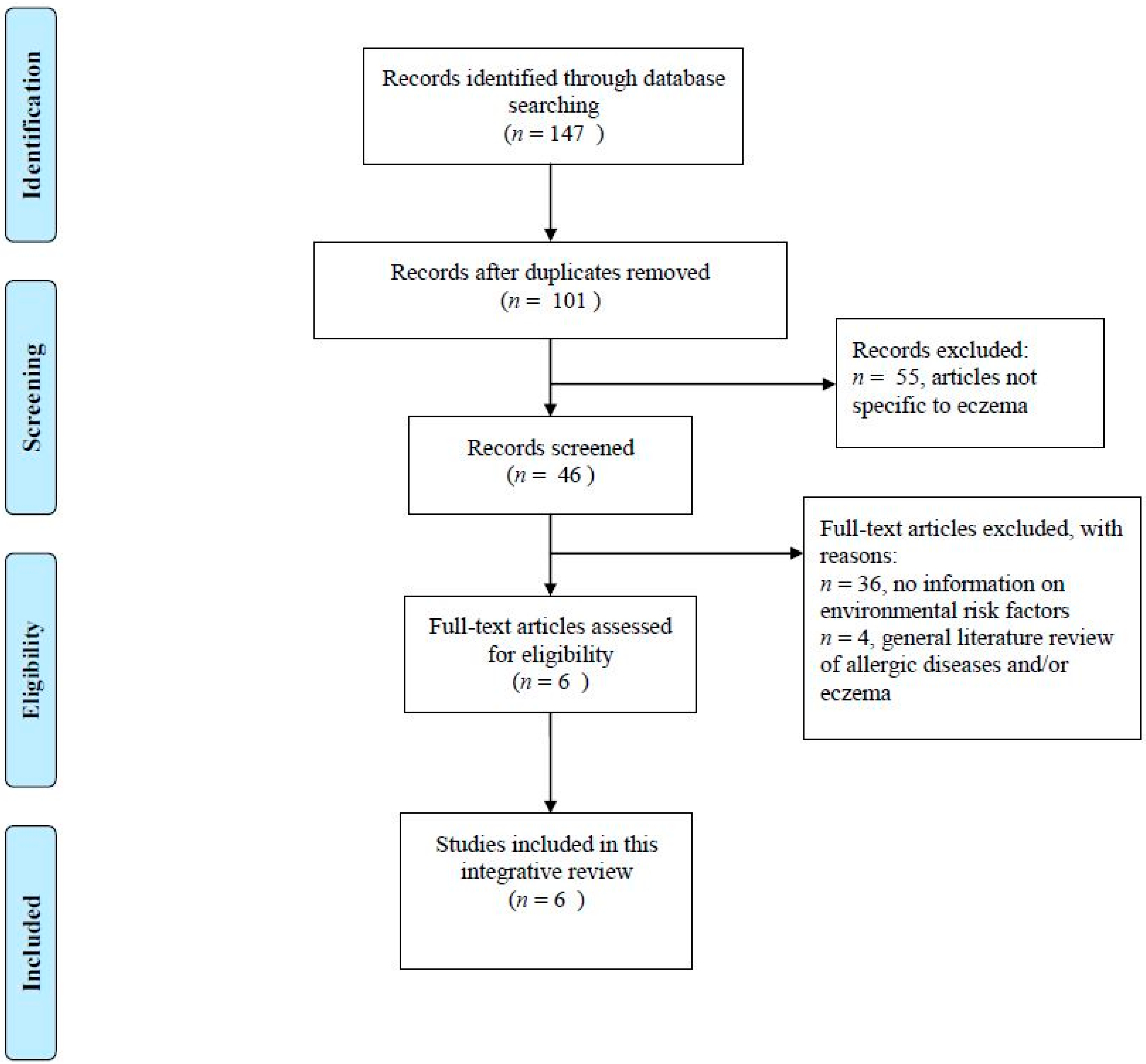
2.1. Identification and Selection of Articles
2.2. Study Quality Assessment
3. Results
3.1. Study Appraisals
3.2. Environmental Factors Associated with Low Microbial Diversity Specific to Eczema
3.2.1. Length of Gestation
3.2.2. Mode of Delivery
3.2.3. Type of Feeding
3.2.4. Treatment
3.2.5. Number of Older Siblings
3.2.6. Other Factors
4. Discussion
4.1. Strength and Limitations
4.2. Implication for Future Research and Practice
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Barnetson, R.S.C.; Rogers, M. Childhood atopic eczema. Br. Med. J. 2002, 324, 1376–1379. [Google Scholar] [CrossRef]
- Lewis-Jones, S. Quality of life and childhood atopic dermatitis: The misery of living with childhood eczema. Int. J. Clin. Pract. 2006, 60, 984–992. [Google Scholar] [CrossRef] [PubMed]
- Van der Hulst, A.E.; Klip, H.; Brand, P.L. Risk of developing asthma in young children with atopic eczema: A systematic review. J. Allergy Clin. Immunol. 2007, 120, 565–569. [Google Scholar] [CrossRef] [PubMed]
- Brown, S.; Reynolds, N.J. Atopic and non-atopic eczema. Br. Med. J. 2006, 7541, 584. [Google Scholar] [CrossRef] [PubMed]
- Arshad, S.H.; Karmaus, W.; Raza, A.; Kurukulaaratchy, R.J.; Matthews, S.M.; Holloway, J.W.; Sadeghnejad, A.; Zhang, H.; Roberts, G.; Ewart, S.L. The effect of parental allergy on childhood allergic diseases depends on the sex of the child. J. Allergy Clin. Immunol. 2012, 130, 427–434. [Google Scholar] [CrossRef] [PubMed]
- Klüken, H.; Wienker, T.; Bieber, T. Atopic eczema/dermatitis syndrome—A genetically complex disease. New advances in discovering the genetic contribution. Allergy 2003, 58, 5–12. [Google Scholar] [CrossRef] [PubMed]
- Von Mutius, E.; Fritzsch, C.; Weiland, S.K.; Röll, G.; Magnussen, H. Prevalence of asthma and allergic disorders among children in united Germany: A descriptive comparison. BMJ 1992, 305, 1395–1399. [Google Scholar] [CrossRef] [PubMed]
- Okada, H.; Kuhn, C.; Feillet, H.; Bach, J.F. The “hygiene hypothesis” for autoimmune and allergic diseases: An update. Clin. Exp. Immunol. 2010, 160, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Williams, H.C.; Strachan, D.P.; Hay, R.J. Childhood eczema: Disease of the advantaged? BMJ 1994, 308, 1132–1135. [Google Scholar] [CrossRef] [PubMed]
- Round, J.L.; Mazmanian, S.K. The gut microbiota shapes intestinal immune responses during health and disease. Nat. Rev. Immunol. 2009, 9, 313–323. [Google Scholar] [CrossRef] [PubMed]
- Penders, J.; Stobberingh, E.E.; van den Brandt, P.A.; Thijs, C. The role of the intestinal microbiota in the development of atopic disorders. Allergy 2007, 62, 1223–1236. [Google Scholar] [CrossRef] [PubMed]
- Marques, T.M.; Wall, R.; Ross, R.P.; Fitzgerald, G.F.; Ryan, C.A.; Stanton, C. Programming infant gut microbiota: Influence of dietary and environmental factors. Curr. Opin. Biotechnol. 2010, 21, 149–156. [Google Scholar] [CrossRef] [PubMed]
- Suto, H.; Matsuda, H.; Mitsuishi, K.; Hira, K.; Uchida, T.; Unno, T.; Ogawa, H.; Ra, C. NC/Nga mice: A mouse model for atopic dermatitis. Int. Arch. Allergy Immunol. 1999, 120 (Suppl. S1), 70–75. [Google Scholar] [CrossRef] [PubMed]
- Marsella, R.; Olivry, T.; Nicklin, C.; Lopez, J. Pilot investigation of a model for canine atopic dermatitis: Environmental house dust mite challenge of high-IgE–producing beagles, mite hypersensitive dogs with atopic dermatitis and normal dogs. Vet. Dermatol. 2006, 17, 24–35. [Google Scholar] [CrossRef] [PubMed]
- Martin, R.; Nauta, A.; Ben Amor, K.; Knippels, L.; Knol, J.; Garssen, J. Early life: Gut microbiota and immune development in infancy. Benef. Microbes 2010, 1, 367–382. [Google Scholar] [CrossRef] [PubMed]
- West, C.E. Gut microbiota and allergic disease: New findings. Curr Opin. Clin. Nutr. Metab. Care 2014, 17, 261–266. [Google Scholar] [CrossRef] [PubMed]
- Marsland, B.J.; Salami, O. Microbiome influences on allergy in mice and humans. Curr. Opin. Immunol. 2015, 36, 94–100. [Google Scholar] [CrossRef] [PubMed]
- Flohr, C.; Mann, J. New insights into the epidemiology of childhood atopic dermatitis. Allergy 2014, 69, 3–16. [Google Scholar] [CrossRef] [PubMed]
- Von Elm, E.; Altman, D.G.; Egger, M.; Pocock, S.J.; Gøtzsche, P.C.; Vandenbroucke, J.P. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: Guidelines for reporting observational studies. Prev. Med. 2007, 45, 247–251. [Google Scholar] [CrossRef] [PubMed]
- Storrø, O.; Øien, T.; Langsrud, Ø.; Rudi, K.; Dotterud, C.; Johnsen, R. Temporal variations in early gut microbial colonization are associated with allergen-specific immunoglobulin E but not atopic eczema at 2 years of age. Clin. Exp. Allergy 2011, 41, 1545–1554. [Google Scholar] [CrossRef] [PubMed]
- Adlerberth, I.; Strachan, D.P.; Matricardi, P.M.; Ahrné, S.; Orfei, L.; Åberg, N.; Perkin, M.R.; Tripodi, S.; Hesselmar, B.; Saalman, R.; et al. Gut microbiota and development of atopic eczema in 3 European birth cohorts. J. Allergy Clin. Immunol. 2007, 120, 343–350. [Google Scholar] [CrossRef] [PubMed]
- Penders, J.; Gerhold, K.; Stobberingh, E.E.; Thijs, C.; Zimmermann, K.; Lau, S.; Hamelmann, E. Establishment of the intestinal microbiota and its role for atopic dermatitis in early childhood. J. Allergy Clin. Immunol. 2013, 132, 601–607. [Google Scholar] [PubMed]
- Penders, J.; Thijs, C.; van den Brandt, P.A.; Kummeling, I.; Snijders, B.; Stelma, F.; Adams, H.; van Ree, R.; Stobberingh, E.E. Gut microbiota composition and development of atopic manifestations in infancy: The KOALA Birth Cohort Study. Gut 2007, 56, 661–667. [Google Scholar] [CrossRef] [PubMed]
- Ismail, I.H.; Oppedisano, F.; Joseph, S.J.; Boyle, R.J.; Licciardi, P.V.; Robins-Browne, R.M.; Tang, M.L. Reduced gut microbial diversity in early life is associated with later development of eczema but not atopy in high-risk infants. Pediatr. Allergy Immunol. 2012, 23, 674–681. [Google Scholar] [CrossRef] [PubMed]
- West, C.E.; Rydén, P.; Lundin, D.; Engstrand, L.; Tulic, M.K.; Prescott, S.L. Gut microbiome and innate immune response patterns in IgE-associated eczema. Clin. Exp. Allergy 2015, 45, 1419–1429. [Google Scholar] [CrossRef] [PubMed]
- Mah, K.W.; Björkstén, B.; Lee, B.W.; Van Bever, H.P.; Shek, L.P.; Tan, T.N.; Lee, Y.K.; Chua, K.Y. Distinct pattern of commensal gut microbiota in toddlers with eczema. Int. Arch. Allergy Immunol. 2006, 140, 157–163. [Google Scholar] [CrossRef] [PubMed]
- Smidesang, I.; Saunes, M.; Storrø, O.; Øien, T.; Holmen, T.L.; Johnsen, R.; Henriksen, A.H. Atopic dermatitis among 2-year olds; high prevalence, but predominantly mild disease—The PACT study, Norway. Pediatr. Dermatol. 2008, 25, 13–18. [Google Scholar] [CrossRef] [PubMed]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. The PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA Statement. PLoS Med 2009, 6, e1000097. [Google Scholar] [CrossRef] [PubMed]
| Study | Study Type | Study Place | Participants | Data Collection Time | Outcome Measures | Techniques Used to Determine the Microbial Diversity | Results | |
|---|---|---|---|---|---|---|---|---|
| Evaluation of Eczema | Collection of Samples | |||||||
| Penders et al. (2007) [23] | Prospective cohort study | The Netherlands | 957 infants | Postpartum questionnaires on child eczema status in the seven, 12 and 24 months | Infant feces collected at age one month; Infant blood samples collected at age two years | Bacteria counts, total and specific IgE, atopic manifestations and sensitization | Quantitative real-time PCR | (1) More Escherichia coli was associated with higher eczema risk after adjusting for subcohort, parental history of atopy, sibling history of atopy, age at collection of fecal sample, infant’s gender and total bacterial count; (2) infants colonized with Clostridium difficile were at higher risk of developing eczema, recurrent wheeze and allergic sensitization; and (3) the presence of C difficile was associated with higher eczema risk |
| Adlerberth et al. (2007) [21] | Prospective cohort study | Multiple | 116 Goteborg children, 108 London children, 100 Rome children | Atopic eczema and total and food-specific IgE levels assessed at age 18 months | Rectal swab collected at age three days; Fecal samples collected at seven, 14, and 28 days and age two, six, and 12 months | Atopic eczema diagnosis according to Williams’ criteria, SCORAD, Serum total and specific IgE levels against common food antigens | Subculture, Gram staining and biochemical/genetic tests | (1) No association between atopic eczema or food-specific IgE by age 18 months and any particular bacterial group acquisition time after adjusting for mode of delivery, parity, and breastfeeding at six months; (2) Cesarean section delayed colonization by Escherichia coli and Bacteroides and Bifidobacterium species, giving way to Clostridium; and (3) Lack of older siblings was associated with earlier colonization by Clostridium species and lower strict anaerobic/facultative anaerobic ratio at 12 months after adjusting for mode of delivery, parity, breastfeeding at six months, and dietary correlates. |
| Storrø et al. (2011) [20] | Prospective cohort study | Norway | 94 infants | Eczema survey assessment at six weeks after delivery and at age one and two years | Infant venous blood for slgE quantification collected at age two years; Infant feces collected at 10 days postnatal, age 4 months and 1 and 2 years | Eczema diagnosis using UK Working Party (UKWP) criteria, slgE concentrations, microbiome composition analyzed through feces samples | Quantitative real-time PCR | (1) Less Escherichia coli at four months and one year and Bacteroides fragilis at two years plus more Bifidobacterium longum at one year among subjects with atopic sensitization; and (2) Higher likelihood of family history of atopy, antibiotic treatment and breastfeeding plus lower likelihood of parental smoking among subjects with atopic sensitization; (3) no association between colonization patterns and atopic eczema. |
| Ismail et al. (2012) [24] | Case-control study | Australia | cases: 33 (33.7%) infants with eczema; controls: 65 (66.3%) healthy infants | Infants evaluated at three, six, and 12 months for the presence of eczema | Infant fecal samples collected at day three, seven, 28, 90 and 180 of life | Skin prick testing, the Scoring Atopic Dermatitis (SCORAD) scale, bacterial DNA from infant fecal specimens | Terminal restriction fragment length polymorphism (T-RFLP) | (1) At day seven, higher microbial diversity in healthy infants than infants with eczema at age 12 months after adjusting for mode of delivery, number of siblings, antibiotics use during pregnancy, breastfeeding, household pets, and maternal allergy |
| Penders et al. (2013) [22] | Prospective cohort study | Germany | 497 infants | Infants evaluated regularly by a pediatrician for atopic dermatitis (AD) signs from the start of intervention until age three years | Fecal samples collected at age five weeks, 13 weeks and 31 weeks | Bacterial DNA from faeces, AD diagnosis | Quantitative real-time PCR | (1) Higher colonization rates of lactobacilli and bacteroides but lower rates of clostridia at age five weeks were associated with more older siblings; (2) colonization of clostridia at age five and 13 weeks was associated with higher eczema risk in the subsequent six months of life; and (3) indirect effect of Clostridium cluster I colonization on birth mode and birth order in association to eczema |
| West et al. (2015) [25] | Case-control study | Australia | cases: 10 children with IgE-associated eczema; controls: 10 non-allegic children | Infants evaluated at age six and 12 months and 2.5 years | Stool samples collected from pregnant mother at inclusion and infant at age one week, one month, and 12 months | Skin prick testing, the Scoring Atopic Dermatitis (SCORAD) scale, bacterial DNA from infant fecal specimens | 16S rRNA 454 pyrosequencing | (1) Association between reduced relative abundance of potentially immunomodulatory gut bacteria and exaggerated inflammatory cytokine responses to TLR-ligands and subsequent development of IgE-associated eczema; (2) More proteobacteria and less bacteroidetes in caesarean section born children than vaginally delivered children; and (3) More bacteroidetes in stool samples of mothers whose infants developed IgE-associated eczema and also in infants later diagnosed with atopic eczema of previous study |
| Study | Methods | Results | Total Quality Score | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Study Design (Report Key Elements?) | Setting (Report Study Setting Details?) | Participants (Give Eligibility Criteria?) | Variables (Define All Variables?) | Data Sources/Measurement (Give Sources of Data?) | Bias (Address Potential Bias?) | Sample Size (How to Measure Study Size?) | Statistical Methods (Describe Statistical Methods Used?) | Participants (Report the Numbers of Individuals at Each Study Stage?) | Descriptive Data (Report Study Subject Characteristics?) | Outcome Data (Report Number in Each Exposure Category?) | Main Results (Report Unadjusted Estimates?) | ||
| Penders et al. (2007) [23] | * | * | * | * | * | * | – | * | * | * | * | * | 11 |
| Adlerberth et al. (2007) [21] | * | * | * | * | * | – | – | * | * | * | * | * | 10 |
| Storrø et al. (2011) [20] | * | * | * | * | * | – | – | * | * | * | * | – | 9 |
| Ismail et al. (2012) [24] | * | * | * | * | * | * | – | * | * | * | * | * | 11 |
| Penders et al. (2013) [22] | * | * | * | * | * | * | – | * | * | * | * | * | 11 |
| West et al. (2015) [25] | * | * | * | * | * | * | – | * | * | * | * | – | 10 |
| Study | Target Organisms | Primer/Probe | Sequence (5′-3′) |
|---|---|---|---|
| Penders et al. (2007) [23] | Bifidobacterium spp. | Forward primer | GCGTGCTTAACACATGCAAGTC |
| Reverse primer | CACCCGTTTCCAGGAGCTATT | ||
| Probe | TCACGCATTACTCACCCGTTCGCC | ||
| Escherichia coli | Forward primer | CATGCCGCGTGTATGAAGAA | |
| Reverse primer | CGGGTAACGTCAATGAGCAAA | ||
| Probe | TATTAACTTTACTCCCTTCCTCCCCGCTGAA | ||
| Clostridium difficile | Forward primer | TTGAGCGATTTACTTCGGTAAAGA | |
| Reverse primer | TGTACTGGCTCACCTTTGATATTCA | ||
| Probe | CCACGCGTTACTCACCCGTCCG | ||
| Bacteroides fragilis group | Forward primer | CGGAGGATCCGAGCGTTA | |
| Reverse primer | CCGCAAACTTTCACAACTGACTTA | ||
| Probe | CGCTCCCTTTAAACCCAATAAATCCGG | ||
| Lactobacillus spp. | Forward primer | AGCAGTAGGGAATCTTCCA | |
| Reverse primer | CACCGCTACACATGGAG | ||
| Storrø, et al. (2011b) [20] | Bacteria | Forward primer | TCCTACGGGAGGCAGCAGT |
| Probe | FAM-CTGATTACCGCGGCTGCTGGCAC-TAMRA | ||
| Reverse primer | GGACTACCAGGGTATCTAATCCTGTT | ||
| Bacteroides fragilis | Forward primer | GAAAGCATTAAGTATTCCACCTG | |
| Probe | FAM-TGAAACTCAAAGGAATTGACGGGG-TAMRA | ||
| Reverse primer | CGGTGATTGGTCACTGACA | ||
| Escherichia coli | Forward primer | GTGTGATATCTACCCGCTTCGC | |
| Probe | FAM-TCGGCATCCGGTCAGTGGCAGT-TAMRA | ||
| Reverse primer | AGAACGGTTTGTGGTTAATCAGGA | ||
| Bifidobacterium breve | Forward primer | GTGGTGGCTTGAGAACTGGATAG | |
| Probe | FAM-TGATTCCTCGTTCTTGCTGT-MGB | ||
| Reverse primer | CAAAACGATCGAAACAAACACTAAA | ||
| Bifidobacterium lactis | Forward primer | AGAACCACGGCGGCGTC | |
| Probe | FAM-TGCGCTCGCCGACG-MGB | ||
| Reverse primer | CGCGGTCTTCTCGAGCACT | ||
| Bifidobacterium longum | Forward primer | TGGAAGACGTCGTTGGCTTT | |
| Probe | FAM-CGCACCCACCGCA-MGB | ||
| Reverse primer | ATCGCGCCAGGCAAAA | ||
| Bifidobacterium spp. | Forward primer | GGGATGCTGGTGTGGAAGAGA | |
| Probe | FAM-TCAAACCACCACGCGCCA-MGB | ||
| Reverse primer | TGCTCGCGTCCACTATCCAGT | ||
| Lactobacillus reuteri | Forward primer | ATGGCTTTTGTTTGAAAGATGGC | |
| Probe | FAM-TGGCTATCACTCTGGGATG-MGB | ||
| Reverse primer | CCTTACCAACTAGCTAATGCACCG | ||
| Lactobacillus rhamnosus | Forward primer | CATAAATCCAAGAACCGCATGG | |
| Probe | FAM-CTTGGCTGAAAGATG-MGB | ||
| Reverse primer | CACGCCGACAACAGTTACTCTGC | ||
| Clostridium difficile | Forward primer | ATATCAGAGACTGATGAG | |
| Probe | FAM-TGGAGAATCTATATTTGTAGAAAC-MGB | ||
| Reverse primer | TAGCATATTCAGAGAATATTGT | ||
| Clostridium perfringens | Forward primer | TTCTATCTTGGAGAGGCTATGCACTATTTT | |
| Probe | FAM-TAGATACTCCATATCATCCTGCTAATGTTACTGCCGTTGA-TAMRA | ||
| Reverse primer | TTTCAAACTTAACATGTCCTGCGC | ||
| Lactobacillus plantarum | Forward primer | TGGACCGCATGGTCCGAG | |
| Probe | FAM-TCCCGCGGCGTATTA-MGB | ||
| Reverse primer | GTGAGCCGTTACCCCACCAT | ||
| Helicobacter pylori | Forward primer | CGTGGCAAGCATGATCCAT | |
| Probe | FAM-TCAGGAAACATCGCTTCAATACCCACTT-TAMRA | ||
| Reverse primer | GGGTATGCACGGTTACGAGTTT | ||
| Penders et al. (2013) [22] | Bifidobacterium spp. | Forward primer | GCGTGCTTAACACATGCAAGTC |
| Reverse primer | CACCCGTTTCCAGGAGCTATT | ||
| Probe | TCACGCATTACTCACCCGTTCGCC | ||
| E. coli | Forward primer | CATGCCGCGTGTATGAAGAA | |
| Reverse primer | CGGGTAACGTCAATGAGCAAA | ||
| Probe | TATTAACTTTACTCCCTTCCTCCCCGCTGAA | ||
| C difficile | Forward primer | TTGAGCGATTTACTTCGGTAAAGA | |
| Reverse primer | TGTACTGGCTCACCTTTGATATTCA | ||
| Probe | CCACGCGTTACTCACCCGTCCG | ||
| B fragilis group | Forward primer | CGGAGGATCCGAGCGTTA | |
| Reverse primer | CCGCAAACTTTCACAACTGACTTA | ||
| Probe | CGCTCCCTTTAAACCCAATAAATCCGG | ||
| Lactobacillus spp. | Forward primer | AGCAGTAGGGAATCTTCCA | |
| Reverse primer | CACCGCTACACATGGAG | ||
| Clostridium cluster I | Forward primer | TACCHRAGGAGGAAGCCAC | |
| Reverse primer | GTTCTTCCTAATCTCTACGCAT | ||
| Probe | GTGCCAGCAGCCGCGGTAATACG |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chan, C.W.H.; Wong, R.S.; Law, P.T.W.; Wong, C.L.; Tsui, S.K.W.; Tang, W.P.Y.; Sit, J.W.H. Environmental Factors Associated with Altered Gut Microbiota in Children with Eczema: A Systematic Review. Int. J. Mol. Sci. 2016, 17, 1147. https://doi.org/10.3390/ijms17071147
Chan CWH, Wong RS, Law PTW, Wong CL, Tsui SKW, Tang WPY, Sit JWH. Environmental Factors Associated with Altered Gut Microbiota in Children with Eczema: A Systematic Review. International Journal of Molecular Sciences. 2016; 17(7):1147. https://doi.org/10.3390/ijms17071147
Chicago/Turabian StyleChan, Carmen W. H., Rosa S. Wong, Patrick T. W. Law, Cho Lee Wong, Stephen K. W. Tsui, Winnie P. Y. Tang, and Janet W. H. Sit. 2016. "Environmental Factors Associated with Altered Gut Microbiota in Children with Eczema: A Systematic Review" International Journal of Molecular Sciences 17, no. 7: 1147. https://doi.org/10.3390/ijms17071147
APA StyleChan, C. W. H., Wong, R. S., Law, P. T. W., Wong, C. L., Tsui, S. K. W., Tang, W. P. Y., & Sit, J. W. H. (2016). Environmental Factors Associated with Altered Gut Microbiota in Children with Eczema: A Systematic Review. International Journal of Molecular Sciences, 17(7), 1147. https://doi.org/10.3390/ijms17071147






