Induced Pluripotent Stem Cell Therapies for Cervical Spinal Cord Injury
Abstract
:1. Introduction
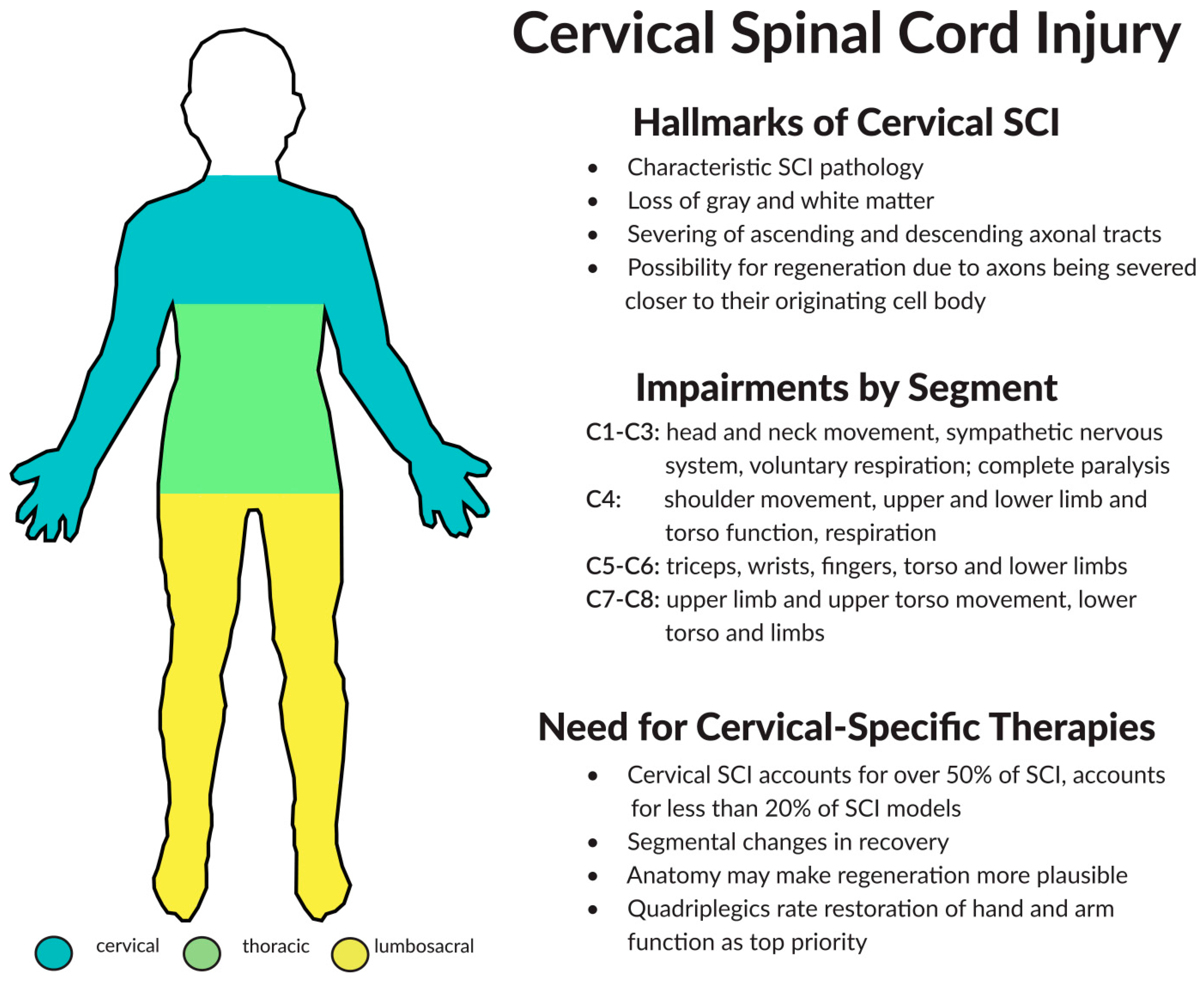
2. Components of Cervical Spinal Cord Injury (SCI)
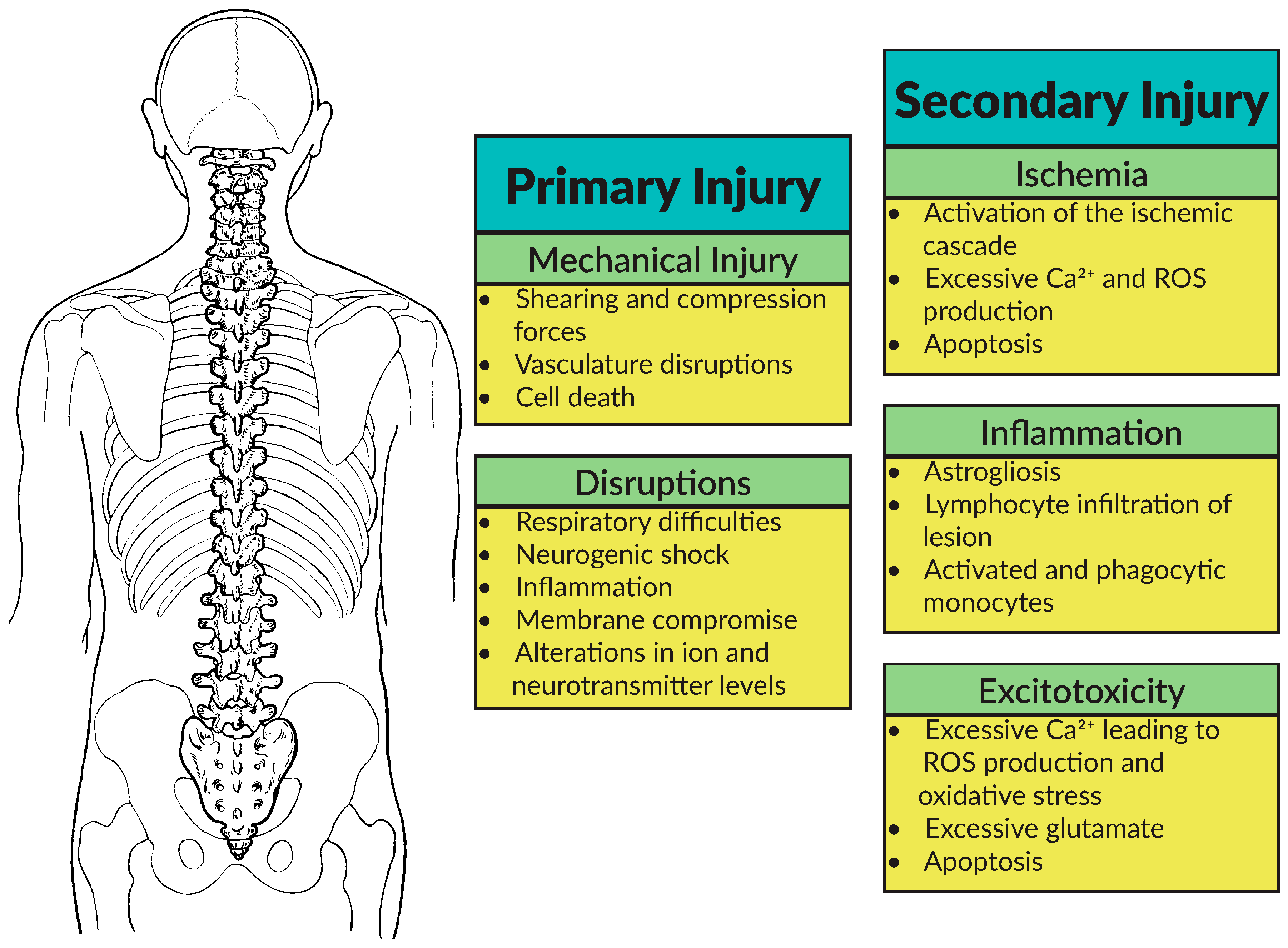
2.1. Pathophysiology
2.2. Regeneration and Plasticity
2.3. Considerations for Treating Cervical SCI
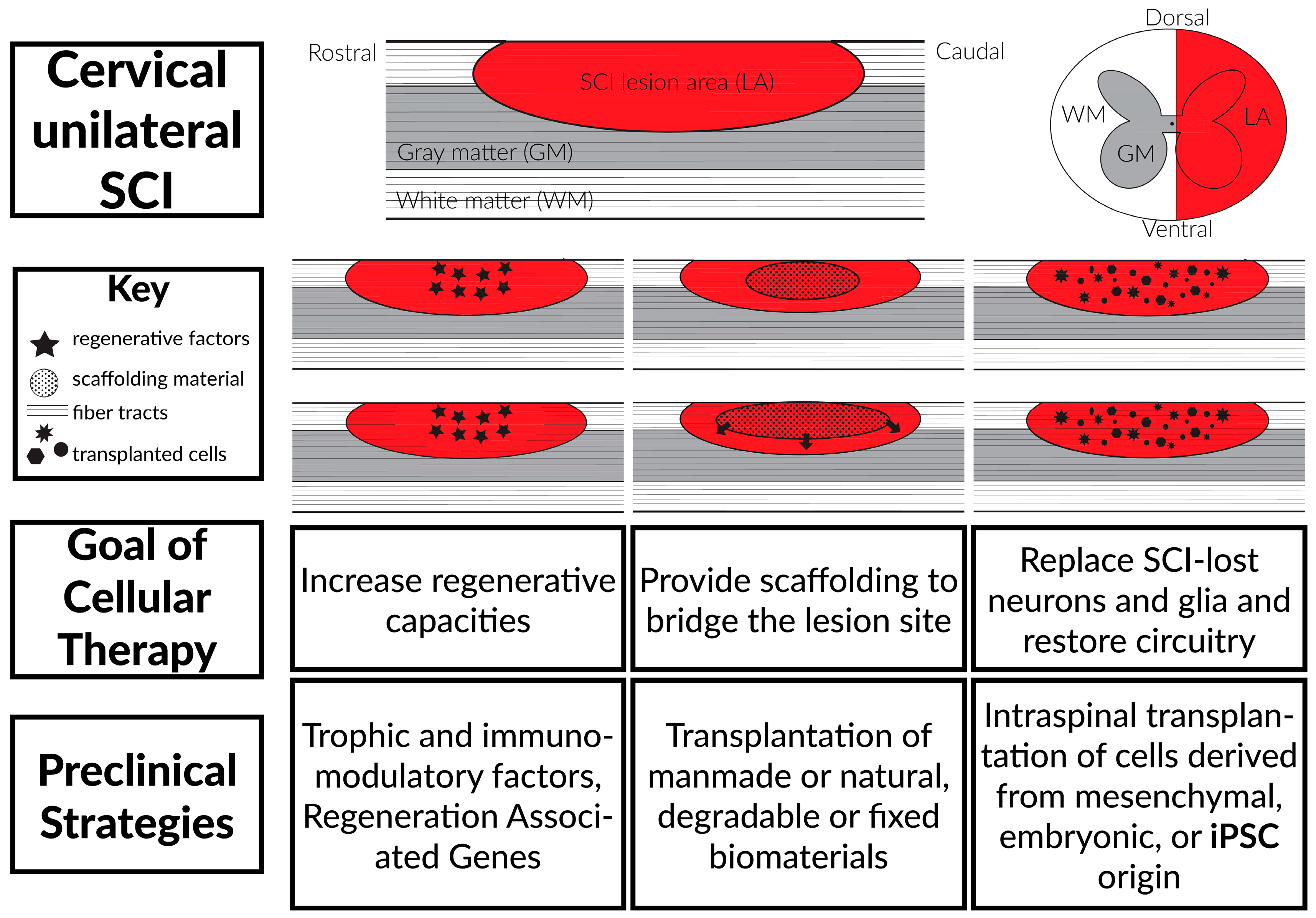
3. Stem Cell Transplantation Therapies
3.1. Background
3.2. Mesenchymal Stem Cells (MSCs)
3.3. Embryonic Stem Cells (ESCs)
3.4. Potential Drawbacks of Adult and Embryonic Stem Cell Therapies
4. Induced Pluripotent Stem Cell-Derived Therapies
4.1. Induced Pluripotent Stem Cells
4.2. Differentiation of Induced Pluripotent Stem Cells (iPSCs)
4.3. Treating Cervical SCI with iPSC Technologies
5. Conclusions and Future Considerations
Acknowledgments
Author Contributions
Conflicts of Interest
References
- National Spinal Cord Injury Statistical Center. Facts and Figures at a Glance; University of Alabama: Birmingham, AL, USA, 2015. [Google Scholar]
- Berkowitz, M. Spinal cord injury: An analysis of medical and social costs. Am. J. Phys. Med. Rehabil. 1999, 78, 568–569. [Google Scholar]
- Schoenfeld, A.J.; Sielski, B.; Rivera, K.P.; Bader, J.O.; Harris, M.B. Epidemiology of cervical spine fractures in the US military. Spine J. 2012, 12, 777–783. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Yoganandan, N.; Stemper, B.D.; Pintar, F.A.; Maiman, D.J.; McEntire, B.J.; Chancey, V.C. Cervical spine injury biomechanics: Applications for under body blast loadings in military environments. Clin. Biomech. 2013, 28, 602–609. [Google Scholar] [CrossRef] [PubMed]
- Inoue, T.; Lin, A.; Ma, X.; McKenna, S.L.; Creasey, G.H.; Manley, G.T.; Ferguson, A.R.; Bresnahan, J.C.; Beattie, M.S. Combined SCI and TBI: Recovery of forelimb function after unilateral cervical spinal cord injury (SCI) is retarded by contralateral traumatic brain injury (TBI), and ipsilateral TBI balances the effects of SCI on paw placement. Exp. Neurol. 2013, 248, 136–147. [Google Scholar] [CrossRef] [PubMed]
- Breeze, J.; Fryer, R.; Hare, J.; Delaney, R.; Hunt, N.C.; Lewis, E.A.; Clasper, J.C. Clinical and post mortem analysis of combat neck injury used to inform a novel coverage of armour tool. Injury 2015, 46, 629–633. [Google Scholar] [CrossRef] [PubMed]
- Wagner, L.S.C.; Lehman, L.R.A. Cervical spine and neck injuries. In Musculoskeletal Injuries in the Military; Springer: New York, NY, USA, 2016; pp. 229–245. [Google Scholar]
- Satyanand, V.; Gopalakrishnaiah, T.; Panneerselvam, E.; Mahaboobvali, S.; Basha, S.A.; Sarala, V. Effects of yogasanas on cervical spondylosis. Int. Arch. Integr. Med. 2015, 2, 6–10. [Google Scholar]
- Smith, L.; Louw, Q.; Crous, L.; Grimmer-Somers, K. Prevalence of neck pain and headaches: Impact of computer use and other associative factors. Cephalalgia 2009, 29, 250–257. [Google Scholar] [CrossRef] [PubMed]
- Ropper, A.E.; Matthew, T.N.; Nicholas, T. Acute management of traumatic cervical spinal cord injury. Pract. Neurol. 2015, 15, 266–272. [Google Scholar] [CrossRef] [PubMed]
- Tetreault, L.A.; Karpova, A.; Fehlings, M.G. Predictors of outcome in patients with degenerative cervical spondylotic myelopathy undergoing surgical treatment: Results of a systematic review. Eur. Spine J. 2015, 24, 236–251. [Google Scholar] [CrossRef] [PubMed]
- Laing, A.C.; Brenneman, E.C.; Yung, A.; Liu, J.; Kozlowski, P.; Oxland, T. The effects of age on the morphometry of the cervical spinal cord and spinal column in adult rats: An MRI-based study. Anat. Rec. 2014, 297, 1885–1895. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Tian, F.; Zhou, Y.; He, W.; Cai, Z. The incidence of cervical spondylosis decreases with aging in the elderly, and increases with aging in the young and adult population: A hospital-based clinical analysis. Clin. Interv. Aging 2016, 11, 47–53. [Google Scholar] [PubMed]
- Sabapathy, V.; George, T.; Sanjay, K. Cell therapy augments functional recovery subsequent to spinal cord injury under experimental conditions. Stem Cells Int. 2014, 2015, 640–645. [Google Scholar] [CrossRef] [PubMed]
- Peitzman, A.B.; Fabian, T.C.; Rhodes, M.; Yealy, D.M.; Schwab, C.W. (Eds.) The Trauma Manual: Trauma and Acute Care Surgery; Lippincott Williams & Wilkins: Pasadena, CA, USA, 2012.
- Newman, M.F.; Lee, A.F.; Mitchell, P.F. Perioperative Medicine: Managing for Outcome; Elsevier Health Sciences: New York, NY, USA, 2008. [Google Scholar]
- Tator, C.H. Biology of neurological recovery and functional restoration after spinal cord injury. Neurosurgery 1998, 42, 696–707. [Google Scholar] [CrossRef] [PubMed]
- Ramer, L.M.; Ramer, M.S.; Steeves, J.D. Setting the stage for functional repair of spinal cord injuries: A cast of thousands. Spinal Cord 2005, 43, 134–161. [Google Scholar] [CrossRef] [PubMed]
- Park, E.; Alexander, A.V.; Michael, G.F. The role of excitotoxicity in secondary mechanisms of spinal cord injury: A review with an emphasis on the implications for white matter degeneration. J. Neurotrauma 2004, 21, 754–774. [Google Scholar] [CrossRef] [PubMed]
- Dumont, R.J.; Okonkwo, D.O.; Verma, S.; Hurlbert, R.J.; Boulos, P.T.; Ellegala, D.B.; Dumont, A.S. Acute spinal cord injury, part I: Pathophysiologic mechanisms. Clin. Neuropharmacol. 2001, 24, 254–264. [Google Scholar] [CrossRef] [PubMed]
- Evelyne, E.; Aldana, P.; Bunge, M.B.; Puckett, W.; Srinivasan, A.; Keane, R.W.; Bethea, J.; Levi, A.D.O. Apoptosis after traumatic human spinal cord injury. J. Neurosurg. 1998, 89, 911–920. [Google Scholar]
- Hausmann, O.N. Post-traumatic inflammation following spinal cord injury. Spinal Cord 2003, 41, 369–378. [Google Scholar] [CrossRef] [PubMed]
- Beattie, M.S.; Akhlaq, A.F.; Jacqueline, C.B. Review of current evidence for apoptosis after spinal cord injury. J. Neurotrauma 2000, 17, 915–925. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.; Ken, W.S.A.; Phil, W. Advances in secondary spinal cord injury: Role of apoptosis. Spine 2000, 25, 1859–1866. [Google Scholar] [CrossRef] [PubMed]
- Mautes, A.E.; Weinzierl, M.R.; Donovan, F.; Noble, L.J. Vascular events after spinal cord injury: Contribution to secondary pathogenesis. Phys. Ther. 2000, 80, 673–687. [Google Scholar] [PubMed]
- Horner, P.J.; Fred, H.G. Regenerating the damaged central nervous system. Nature 2000, 407, 963–970. [Google Scholar] [PubMed]
- Ramer, M.S.; John, V.P.; Stephen, B.M. Functional regeneration of sensory axons into the adult spinal cord. Nature 2000, 403, 312–316. [Google Scholar] [CrossRef] [PubMed]
- Bernstein, J.J.; Mary, E.B. Axonal regeneration and formation of synapses proximal to the site of lesion following hemisection of the rat spinal cord. Exp. Neurol. 1971, 30, 336–351. [Google Scholar] [CrossRef]
- Ye, J.-H.; Houle, J.D. Treatment of the chronically injured spinal cord with neurotrophic factors can promote axonal regeneration from supraspinal neurons. Exp. Neurol. 1997, 143, 70–81. [Google Scholar] [CrossRef] [PubMed]
- Namiki, J.; Kojima, A.; Tator, C.H. Effect of brain-derived neurotrophic factor, nerve growth factor, and neurotrophin-3 on functional recovery and regeneration after spinal cord injury in adult rats. J. Neurotrauma 2000, 17, 1219–1231. [Google Scholar] [CrossRef] [PubMed]
- McTigue, D.M.; Horner, P.J.; Stokes, B.T.; Gage, F.H. Neurotrophin-3 and brain-derived neurotrophic factor induce oligodendrocyte proliferation and myelination of regenerating axons in the contused adult rat spinal cord. J. Neurosci. 1998, 18, 5354–5365. [Google Scholar] [PubMed]
- Grill, R.; Murai, K.; Blesch, A.; Gage, F.H.; Tuszynski, M.H. Cellular delivery of neurotrophin-3 promotes corticospinal axonal growth and partial functional recovery after spinal cord injury. J. Neurosci. 1997, 17, 5560–5572. [Google Scholar] [PubMed]
- Simonen, M.; Pedersen, V.; Weinmann, O.; Schnell, L.; Buss, A.; Ledermann, B.; Christ, F.; Sansig, G.; van der Putten, H.; Schwab, M.E. Systemic deletion of the myelin-associated outgrowth inhibitor Nogo-A improves regenerative and plastic responses after spinal cord injury. Neuron 2003, 38, 201–211. [Google Scholar] [CrossRef]
- Kim, G.-M.; Xu, J.; Xu, J.; Song, S.K.; Yan, P.; Ku, G.; Xu, X.M.; Hsu, C.Y. Tumor necrosis factor receptor deletion reduces nuclear factor-κB activation, cellular inhibitor of apoptosis protein 2 expression, and functional recovery after traumatic spinal cord injury. J. Neurosci. 2001, 21, 6617–6625. [Google Scholar] [PubMed]
- Nishio, Y.; Koda, M.; Hashimoto, M.; Kamada, T.; Koshizuka, S.; Yoshinaga, K.; Onodera, S.; Nishihira, J.; Okawa, A.; Yamazaki, M. Deletion of macrophage migration inhibitory factor attenuates neuronal death and promotes functional recovery after compression-induced spinal cord injury in mice. Acta Neuropathol. 2009, 117, 321–328. [Google Scholar] [CrossRef] [PubMed]
- Hamid, S.; Ray, H. Role of electrical stimulation for rehabilitation and regeneration after spinal cord injury: An overview. Eur. Spine J. 2008, 17, 1256–1269. [Google Scholar] [CrossRef] [PubMed]
- Smith, R.R.; Shum-Siu, A.; Baltzley, R.; Bunger, M.; Baldini, A.; Burke, D.A.; Magnuson, D.S. Effects of swimming on functional recovery after incomplete spinal cord injury in rats. J. Neurotrauma 2006, 23, 908–919. [Google Scholar] [CrossRef] [PubMed]
- Engesser-Cesar, C.; Anderson, A.J.; Basso, D.M.; Edgerton, V.R.; Cotman, C.W. Voluntary wheel running improves recovery from a moderate spinal cord injury. J. Neurotrauma 2005, 22, 157–171. [Google Scholar] [CrossRef] [PubMed]
- Donnelly, D.J.; Phillip, G.P. Inflammation and its role in neuroprotection, axonal regeneration and functional recovery after spinal cord injury. Exp. Neurol. 2008, 209, 378–388. [Google Scholar] [CrossRef] [PubMed]
- Popovich, P.G.; Guan, Z.; Wei, P.; Huitinga, I.; van Rooijen, N.; Stokes, B.T. Depletion of hematogenous macrophages promotes partial hindlimb recovery and neuroanatomical repair after experimental spinal cord injury. Exp. Neurol. 1999, 158, 351–365. [Google Scholar] [CrossRef] [PubMed]
- Ikegami, T.; Nakamura, M.; Yamane, J.; Katoh, H.; Okada, S.; Iwanami, A.; Watanabe, K.; Ishii, K.; Kato, F.; Fujita, H.; et al. Chondroitinase ABC combined with neural stem/progenitor cell transplantation enhances graft cell migration and outgrowth of growth-associated protein-43-positive fibers after rat spinal cord injury. Eur. J. Neurosci. 2005, 22, 3036–3046. [Google Scholar] [CrossRef] [PubMed]
- Bregman, B.S.; McAtee, M.; Dai, H.N.; Kuhn, P.L. Neurotrophic factors increase axonal growth after spinal cord injury and transplantation in the adult rat. Exp. Neurol. 1997, 148, 475–494. [Google Scholar] [CrossRef] [PubMed]
- Chopp, M.; Zhang, X.H.; Li, Y.; Wang, L.; Chen, J.; Lu, D.; Lu, M.; Rosenblum, M. Spinal cord injury in rat: Treatment with bone marrow stromal cell transplantation. Neuroreport 2000, 11, 3001–3005. [Google Scholar] [CrossRef] [PubMed]
- Jones, L.L.; Oudega, M.; Bunge, M.B.; Tuszynski, M.H. Neurotrophic factors, cellular bridges and gene therapy for spinal cord injury. J. Physiol. 2001, 533, 83–89. [Google Scholar] [CrossRef] [PubMed]
- Raisman, G. Olfactory ensheathing cells—Another miracle cure for spinal cord injury? Nat. Rev. Neurosci. 2001, 2, 369–375. [Google Scholar] [CrossRef] [PubMed]
- Thuret, S.; Lawrence, D.F.M.; Fred, H.G. Therapeutic interventions after spinal cord injury. Nat. Rev. Neurosci. 2006, 7, 628–643. [Google Scholar] [CrossRef] [PubMed]
- Plant, G.W.; Margaret, L.B.; Mary, B.B. Inhibitory proteoglycan immunoreactivity is higher at the caudal than the rostral Schwann cell graft-transected spinal cord interface. Mol. Cell. Neurosci. 2001, 17, 471–487. [Google Scholar] [CrossRef] [PubMed]
- Ruitenberg, M.J.; Plant, G.W.; Christensen, C.L.; Blits, B.; Niclou, S.P.; Harvey, A.R.; Boer, G.J.; Verhaagen, J. Viral vector-mediated gene expression in olfactory ensheathing glia implants in the lesioned rat spinal cord. Gene Ther. 2002, 9, 135–146. [Google Scholar] [CrossRef] [PubMed]
- Ramer, L.M.; Au, E.; Richter, M.W.; Liu, J.; Tetzlaff, W.; Roskams, A.J. Peripheral olfactory ensheathing cells reduce scar and cavity formation and promote regeneration after spinal cord injury. J. Comp. Neurol. 2004, 473, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Kwon, B.K.; Tetzlaff, W.; Grauer, J.N.; Beiner, J.; Vaccaro, A.R. Pathophysiology and pharmacologic treatment of acute spinal cord injury. Spine J. 2004, 4, 451–464. [Google Scholar] [CrossRef] [PubMed]
- Snyder, E.Y.; Yang, D.T. Stem cells and spinal cord repair. N. Engl. J. Med. 2012, 366, 1940–1942. [Google Scholar] [CrossRef] [PubMed]
- Barry, F.P.; Murphy, J.M. Mesenchymal stem cells: Clinical applications and biological characterization. Int. J. Biochem. Cell Biol. 2004, 36, 568–584. [Google Scholar] [CrossRef] [PubMed]
- Parr, A.M.; Kulbatski, I.; Zahir, T.; Wang, X.; Yue, C.; Keating, A.; Tator, C.H. Transplanted adult spinal cord–Derived neural stem/progenitor cells promote early functional recovery after rat spinal cord injury. Neuroscience 2008, 155, 760–770. [Google Scholar] [CrossRef] [PubMed]
- Cummings, B.J.; Uchida, N.; Tamaki, S.J.; Salazar, D.L.; Hooshmand, M.; Summers, R.; Gage, F.H.; Anderson, A.J. Human neural stem cells differentiate and promote locomotor recovery in spinal cord-injured mice. Proc. Natl. Acad. Sci. USA 2005, 102, 14069–14074. [Google Scholar] [CrossRef] [PubMed]
- Reier, P.J.; Bregman, B.S.; Wujek, J.R. Intraspinal transplantation of embyronic spinal cord tissue in neonatal and adult rats. J. Comp. Neurol. 1986, 247, 275–296. [Google Scholar] [CrossRef] [PubMed]
- Reubinoff, B.E.; Itsykson, P.; Turetsky, T.; Pera, M.F.; Reinhartz, E.; Itzik, A.; Ben-Hur, T. Neural progenitors from human embryonic stem cells. Nat. Biotechnol. 2001, 19, 1134–1140. [Google Scholar] [CrossRef] [PubMed]
- Carpenter, M.K.; Rosler, E.; Rao, M.S. Characterization and differentiation of human embryonic stem cells. Cloning Stem Cells 2003, 5, 79–88. [Google Scholar] [CrossRef] [PubMed]
- Tetzlaff, W.; Okon, E.B.; Karimi-Abdolrezaee, S.; Hill, C.E.; Sparling, J.S.; Plemel, J.R.; Plunet, W.T.; Tsai, E.C.; Baptiste, D.; Smithson, L.J.; et al. A systematic review of cellular transplantation therapies for spinal cord injury. J. Neurotrauma 2011, 28, 1611–1682. [Google Scholar] [CrossRef] [PubMed]
- Hedel, H.J.A.V.; Armin, C. Fighting for each segment: Estimating the clinical value of cervical and thoracic segments in SCI. J. Neurotrauma 2006, 23, 1621–1631. [Google Scholar] [CrossRef] [PubMed]
- Hill, C.E.; Beattie, M.S.; Bresnahan, J.C. Degeneration and sprouting of identified descending supraspinal axons after contusive spinal cord injury in the rat. Exp. Neurol. 2001, 171, 153–169. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, K.J.; Fan, D.P.; Tsui, B.J.; Cassar, S.L.; Tetzlaff, W. Influence of the axotomy to cell body distance in rat rubrospinal and spinal motoneurons: Differential regulation of GAP-43, tubulins, and neurofilament-M. J. Comp. Neurol. 1999, 414, 495–510. [Google Scholar] [CrossRef]
- Richardson, P.M.; Lu, X. Inflammation and axonal regeneration. J. Neurol. 1994, 242, S57–S60. [Google Scholar] [CrossRef] [PubMed]
- Dimitrijevic, M.R.; Gerasimenko, Y.; Pinter, M.M. Evidence for a spinal central pattern generator in humansa. Ann. N. Y. Acad. Sci. 1998, 860, 360–376. [Google Scholar] [CrossRef] [PubMed]
- Bareyre, F.M.; Kerschensteiner, M.O.; Mettenleiter, T.C.; Weinmann, O.; Schwab, M.E. The injured spinal cord spontaneously forms a new intraspinal circuit in adult rats. Nat. Neurosci. 2004, 7, 269–277. [Google Scholar] [CrossRef] [PubMed]
- Grillner, S.; Zangger, P. On the central generation of locomotion in the low spinal cat. Exp. Brain Res. 1979, 34, 241–261. [Google Scholar] [CrossRef] [PubMed]
- Grillner, S.; Peter, Z. The effect of dorsal root transection on the efferent motor pattern in the cat’s hindlimb during locomotion. Acta Physiol. Scand. 1984, 120, 393–405. [Google Scholar] [CrossRef] [PubMed]
- Whelan, P.J. Control of locomotion in the decerebrate cat. Prog. Neurobiol. 1996, 49, 481–515. [Google Scholar] [CrossRef]
- Anderson, K.D.; Marim, A.; Oswald, S. Quantitative assessment of deficits and recovery of forelimb motor function after cervical spinal cord injury in mice. Exp. Neurol. 2004, 190, 184–191. [Google Scholar] [CrossRef] [PubMed]
- Webb, A.A.; Gillian, D.M. Sensorimotor behaviour following incomplete cervical spinal cord injury in the rat. Behav. Brain Res. 2005, 165, 147–159. [Google Scholar] [CrossRef] [PubMed]
- Anderson, K.D.; Ardi, G.; Oswald, S. Spinal pathways involved in the control of forelimb motor function in rats. Exp. Neurol. 2007, 206, 318–331. [Google Scholar] [CrossRef] [PubMed]
- Zörner, B.; Filli, L.; Starkey, M.L.; Gonzenbach, R.; Kasper, H.; Röthlisberger, M.; Bolliger, M.; Schwab, M.E. Profiling locomotor recovery: Comprehensive quantification of impairments after CNS damage in rodents. Nat. Methods 2010, 7, 701–708. [Google Scholar] [CrossRef] [PubMed]
- Friedli, L.; Rosenzweig, E.S.; Barraud, Q.; Schubert, M.; Dominici, N.; Awai, L.; Nielson, J.L.; Musienko, P.; Nout-Lomas, Y.; Zhong, H.; et al. Pronounced species divergence in corticospinal tract reorganization and functional recovery after lateralized spinal cord injury favors primates. Sci. Transl. Med. 2015, 7. [Google Scholar] [CrossRef] [PubMed]
- Anderson, K.D. Targeting recovery: Priorities of the spinal cord-injured population. J. Neurotrauma 2004, 21, 1371–1383. [Google Scholar] [CrossRef] [PubMed]
- Antonic, A.; Wenger, N.; Gorod, A. Stem cell transplantation in traumatic spinal cord injury: A systematic review and meta-analysis of animal studies. PLoS Biol. 2013, 11, e1001738. [Google Scholar] [CrossRef] [PubMed]
- Vawda, R.; Michael, G.F. Mesenchymal cells in the treatment of spinal cord injury: Current & future perspectives. Curr. Stem Cell Res. Ther. 2013, 8, 25–38. [Google Scholar] [PubMed]
- Caplan, A.I. Adult mesenchymal stem cells for tissue engineering versus regenerative medicine. J. Cell. Physiol. 2007, 213, 341–347. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Shao, J.Z.; Xiang, L.X.; Dong, X.J.; Zhang, G.R. Mesenchymal stem cells: A promising candidate in regenerative medicine. Int. J. Biochem. Cell Biol. 2008, 40, 815–820. [Google Scholar] [CrossRef] [PubMed]
- Deans, R.J.; Moseley, A.B. Mesenchymal stem cells: biology and potential clinical uses. Exp. Hematol. 2000, 28, 875–884. [Google Scholar] [CrossRef]
- Richardson, S.M.; Hoyland, J.A.; Mobasheri, R.; Csaki, C.; Shakibaei, M.; Mobasheri, A. Mesenchymal stem cells in regenerative medicine: Opportunities and challenges for articular cartilage and intervertebral disc tissue engineering. J. Cell. Physiol. 2010, 222, 23–32. [Google Scholar] [CrossRef] [PubMed]
- Tabar, V.; Lorenz, S. Pluripotent stem cells in regenerative medicine: Challenges and recent progress. Nat. Rev. Genet. 2014, 15, 82–92. [Google Scholar] [CrossRef] [PubMed]
- Martello, G.; Austin, S. The nature of embryonic stem cells. Annu. Rev. Cell Dev. Biol. 2014, 30, 647–675. [Google Scholar] [CrossRef] [PubMed]
- Mothe, A.J.; Charles, H.T. Review of transplantation of neural stem/progenitor cells for spinal cord injury. Int. J. Dev. Neurosci. 2013, 31, 701–713. [Google Scholar] [CrossRef] [PubMed]
- Vibhu, S.; Kessler, J.A. Stem cell therapies for spinal cord injury. Nat. Rev. Neurol. 2010, 6, 363–372. [Google Scholar]
- Paul, L.; Ahmad, R.; Tuszynski, M.H. Neural Stem Cells for Spinal Cord Injury. Transl. Neurosci. 2016, 5, 297–315. [Google Scholar]
- Youmna, K.; Scadden, D.T. Mesenchymal cell contributions to the stem cell niche. Cell Stem Cell 2015, 16, 239–253. [Google Scholar]
- Morayma, R.; Verfaillie, C.M. Characterization of multipotent adult progenitor cells, a subpopulation of mesenchymal stem cells. Ann. N. Y. Acad. Sci. 2001, 938, 231–235. [Google Scholar]
- Amit, M.; Carpenter, M.K.; Inokuma, M.S.; Chiu, C.P.; Harris, C.P.; Waknitz-Eldor, J.; Thomson, J.A. Clonally derived human embryonic stem cell lines maintain pluripotency and proliferative potential for prolonged periods of culture. Dev. Biol. 2000, 227, 271–278. [Google Scholar] [CrossRef] [PubMed]
- Biswas, A.; Robert, H. Embryonic stem cells. Stem Cells Dev. 2007, 16, 213–222. [Google Scholar] [CrossRef] [PubMed]
- Hodgetts, S.I.; Paul, J.S.; Giles, W.P. Human mesenchymal precursor cells (Stro-1+) from spinal cord injury patients improve functional recovery and tissue sparing in an acute spinal cord injury rat model. Cell Transplant. 2013, 22, 393–412. [Google Scholar] [CrossRef] [PubMed]
- Dominici, M.; Le, B.K.; Mueller, I.; Slaper-Cortenbach, I.; Marini, F.; Krause, D.; Deans, R.; Keating, A.; Prockop, D.J.; Horwitz, E. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy 2006, 8, 315–317. [Google Scholar] [CrossRef] [PubMed]
- Wuchter, P.; Bieback, K.; Schrezenmeier, H.; Bornhäuser, M.; Müller, L.P.; Bönig, H.; Wagner, W.; Meisel, R.; Pavel, P.; Tonn, T.; et al. Standardization of Good Manufacturing Practice-compliant production of bone marrow-derived human mesenchymal stromal cells for immunotherapeutic applications. Cytotherapy 2015, 17, 128–139. [Google Scholar] [CrossRef] [PubMed]
- Torres-Espín, A.; Corona-Quintanilla, D.L.; Forés, J.; Allodi, I.; González, F.; Udina, E.; Navarro, X. Neuroprotection and axonal regeneration after lumbar ventral root avulsion by re-implantation and mesenchymal stem cells transplant combined therapy. Neurotherapeutics 2013, 10, 354–368. [Google Scholar] [CrossRef] [PubMed]
- Lalu, M.M.; McIntyre, L.; Pugliese, C.; Fergusson, D.; Winston, B.W.; Marshall, J.C.; Granton, J.; Stewart, D.J. Safety of cell therapy with mesenchymal stromal cells (SafeCell): A systematic review and meta-analysis of clinical trials. PLoS ONE 2012, 7, e47559. [Google Scholar] [CrossRef] [PubMed]
- Lalu, M.M.; Lauralyn, L.M.; Duncan, J.S. Mesenchymal stromal cells: Cautious optimism for their potential role in the treatment of acute lung injury. Crit. Care Med. 2012, 40, 1373–1375. [Google Scholar] [CrossRef] [PubMed]
- Ra, J.C.; Shin, I.S.; Kim, S.H.; Kang, S.K.; Kang, B.C.; Lee, H.Y.; Kim, Y.J.; Jo, J.Y.; Yoon, E.J.; Choi, H.J.; et al. Safety of intravenous infusion of human adipose tissue-derived mesenchymal stem cells in animals and humans. Stem Cells Dev. 2011, 20, 1297–1308. [Google Scholar] [CrossRef] [PubMed]
- Uccelli, A.; Federica, B.; Alice, L.; Debora, G. Neuroprotective features of mesenchymal stem cells. Best Pract. Res. Clin. Haematol. 2011, 24, 59–64. [Google Scholar] [CrossRef] [PubMed]
- Tollervey, J.R.; Victoria, V.L. Adult stem cells: Simply a tool for regenerative medicine or an additional piece in the puzzle of human aging? Cell Cycle 2011, 10, 4173–4176. [Google Scholar] [CrossRef] [PubMed]
- Tidball, J.G.; Villalta, S.A. Regulatory interactions between muscle and the immune system during muscle regeneration. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2010, 298, R1173–R1187. [Google Scholar] [CrossRef] [PubMed]
- Bai, L.; Lennon, D.P.; Eaton, V.; Maier, K.; Caplan, A.I.; Miller, S.D.; Miller, R.H. Human bone marrow-derived mesenchymal stem cells induce Th2-polarized immune response and promote endogenous repair in animal models of multiple sclerosis. Glia 2009, 57, 1192–1203. [Google Scholar] [CrossRef] [PubMed]
- Kong, D.; Li, Y.; Wang, Z.; Banerjee, S.; Ahmad, A.; Kim, H.R.C.; Sarkar, F.H. miR-200 Regulates PDGF-D-mediated epithelial-mesenchymal transition, adhesion, and invasion of prostate cancer cells. Stem Cells 2009, 27, 1712–1721. [Google Scholar] [CrossRef] [PubMed]
- Uccelli, A.; Lorenzo, M.; Vito, P. Mesenchymal stem cells in health and disease. Nat. Rev. Immunol. 2008, 8, 726–736. [Google Scholar] [CrossRef] [PubMed]
- Aggarwal, S.; Mark, F.P. Human mesenchymal stem cells modulate allogeneic immune cell responses. Blood 2005, 105, 1815–1822. [Google Scholar] [CrossRef] [PubMed]
- Di Nicola, M.; Carlo-Stella, C.; Magni, M.; Milanesi, M.; Longoni, P.D.; Matteucci, P.; Grisanti, S.; Gianni, A.M. Human bone marrow stromal cells suppress T-lymphocyte proliferation induced by cellular or nonspecific mitogenic stimuli. Blood 2002, 99, 3838–3843. [Google Scholar] [CrossRef] [PubMed]
- Sekiya, I.; Larson, B.L.; Smith, J.R.; Pochampally, R.; Cui, J.G.; Prockop, D.J. Expansion of human adult stem cells from bone marrow stroma: Conditions that maximize the yields of early progenitors and evaluate their quality. Stem Cells 2002, 20, 530–541. [Google Scholar] [CrossRef] [PubMed]
- Parekkadan, B.; Jack, M.M. Mesenchymal stem cells as therapeutics. Annu. Rev. Biomed. Eng. 2010, 12, 87–117. [Google Scholar] [CrossRef] [PubMed]
- Koshizuka, S.; Okada, S.; Okawa, A.; Koda, M.; Murasawa, M.; Hashimoto, M.; Kamada, T.; Yoshinaga, K.; Murakami, M.; Moriya, H.; et al. Transplanted hematopoietic stem cells from bone marrow differentiate into neural lineage cells and promote functional recovery after spinal cord injury in mice. J. Neuropathol. Exp. Neurol. 2004, 63, 64–72. [Google Scholar] [CrossRef] [PubMed]
- Martinez, A.M.; Goulart, C.O.; Ramalho, B.S.; Oliveira, J.T.; Almeida, F.M. Neurotrauma and mesenchymal stem cells treatment: From experimental studies to clinical trials. World J. Stem Cell. 2014, 6, 179–194. [Google Scholar] [CrossRef] [PubMed]
- Hofstetter, C.P.; Schwarz, E.J.; Hess, D.; Widenfalk, J.; El Manira, A.; Prockop, D.J.; Olson, L. Marrow stromal cells form guiding strands in the injured spinal cord and promote recovery. Proc. Natl. Acad. Sci. USA 2002, 99, 2199–2204. [Google Scholar] [CrossRef] [PubMed]
- Kopen, G.C.; Darwin, J.P.; Donald, G.P. Marrow stromal cells migrate throughout forebrain and cerebellum, and they differentiate into astrocytes after injection into neonatal mouse brains. Proc. Natl. Acad. Sci. USA 1999, 96, 10711–10716. [Google Scholar] [CrossRef] [PubMed]
- Lu, P.; Hong, Y.; Jones, L.L.; Filbin, M.T.; Tuszynski, M.H. Combinatorial therapy with neurotrophins and cAMP promotes axonal regeneration beyond sites of spinal cord injury. J. Neurosci. 2004, 24, 6402–6409. [Google Scholar] [CrossRef] [PubMed]
- Lu, P.; Jones, L.L.; Tuszynski, M.H. BDNF-expressing marrow stromal cells support extensive axonal growth at sites of spinal cord injury. Exp. Neurol. 2005, 191, 344–360. [Google Scholar] [CrossRef] [PubMed]
- Lu, P.; Jones, L.L.; Tuszynski, M.H. Axon regeneration through scars and into sites of chronic spinal cord injury. Exp. Neurol. 2007, 203, 8–21. [Google Scholar] [CrossRef] [PubMed]
- Novikova, L.N.; Brohlin, M.; Kingham, P.J.; Novikov, L.N.; Wiberg, M. Neuroprotective and growth-promoting effects of bone marrow stromal cells after cervical spinal cord injury in adult rats. Cytotherapy 2011, 13, 873–887. [Google Scholar] [CrossRef] [PubMed]
- White, S.V.; Czisch, C.E.; Han, M.H.; Plant, C.D.; Harvey, A.R.; Plant, G.W. Intravenous transplantation of mesenchymal progenitors distribute solely to the lungs and improve outcomes in cervical spinal cord injury. Stem Cell 2016. [Google Scholar] [CrossRef] [PubMed]
- Sandner, B.; Rivera, F.J.; Caioni, M.; Nicholson, L.; Eckstein, V.; Bogdahn, U.; Aigner, L.; Blesch, A.; Weidner, N. Bone morphogenetic proteins prevent bone marrow stromal cell-mediated oligodendroglial differentiation of transplanted adult neural progenitor cells in the injured spinal cord. Stem Cell Res. 2013, 11, 758–771. [Google Scholar] [CrossRef] [PubMed]
- Neuhuber, B.; Himes, B.T.; Shumsky, J.S.; Gallo, G.; Fischer, I. Axon growth and recovery of function supported by human bone marrow stromal cells in the injured spinal cord exhibit donor variations. Brain Res. 2005, 1035, 73–85. [Google Scholar] [CrossRef] [PubMed]
- Goldschlager, T.; Jenkin, G.; Ghosh, P.; Zannettino, A.; Victor Rosenfeld, J. Potential applications for using stem cells in spine surgery. Curr. Stem Cell Res. Ther. 2010, 5, 345–355. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Syková, E.; Homola, A.; Mazanec, R.; Lachmann, H.; Langkramer Konrádová, Š.; Kobylka, P.; Pádr, R.; Neuwirth, J.; Komrska, V.; Vávra, V.; et al. Autologous bone marrow transplantation in patients with subacute and chronic spinal cord injury. Cell Transpl. 2006, 15, 675–687. [Google Scholar] [CrossRef]
- Saporta, S.; Kim, J.J.; Willing, A.E.; Fu, E.S.; Davis, C.D.; Sanberg, P.R. Human umbilical cord blood stem cells infusion in spinal cord injury: Engraftment and beneficial influence on behavior. J. Hematother. Stem Cell Res. 2003, 12, 271–278. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Ramos, J.R.; Song, S.; Kamath, S.G.; Zigova, T.; Willing, A.; Cardozo-Pelaez, F.; Stedeford, T.; Chopp, M.; Sanberg, P.R. Expression of neural markers in human umbilical cord blood. Exp. Neurol. 2001, 171, 109–115. [Google Scholar] [CrossRef] [PubMed]
- Nishio, Y.; Koda, M.; Kamada, T.; Someya, Y.; Yoshinaga, K.; Okada, S.; Harada, H.; Okawa, A.; Moriya, H.; Yamazaki, M. The use of hemopoietic stem cells derived from human umbilical cord blood to promote restoration of spinal cord tissue and recovery of hindlimb function in adult rats. J. Neurosurg. Spine 2006, 5, 424–433. [Google Scholar] [CrossRef] [PubMed]
- Quertainmont, R.; Cantinieaux, D.; Botman, O.; Sid, S.; Schoenen, J.; Franzen, R. Mesenchymal stem cell graft improves recovery after spinal cord injury in adult rats through neurotrophic and pro-angiogenic actions. PLoS ONE 2012, 7, e39500. [Google Scholar] [CrossRef] [PubMed]
- Cantinieaux, D.; Quertainmont, R.; Blacher, S.; Rossi, L.; Wanet, T.; Noël, A.; Brook, G.; Schoenen, J.; Franzen, R. Conditioned medium from bone marrow-derived mesenchymal stem cells improves recovery after spinal cord injury in rats: An original strategy to avoid cell transplantation. PLoS ONE 2013, 8, e69515. [Google Scholar] [CrossRef] [PubMed]
- Boido, M.; Garbossa, D.; Fontanella, M.; Ducati, A.; Vercelli, A. Mesenchymal stem cell transplantation reduces glial cyst and improves functional outcome after spinal cord compression. World Neurosurg. 2014, 81, 183–190. [Google Scholar] [CrossRef] [PubMed]
- Karaoz, E.; Kabatas, S.; Duruksu, G.; Okcu, A.; Subasi, C.; Ay, B.; Musluman, M.; Civelek, E. Reduction of lesion in injured rat spinal cord and partial functional recovery of motility after bone marrow derived mesenchymal stem cell transplantation. Turk. Neurosurg. 2012, 22, 207–217. [Google Scholar] [CrossRef] [PubMed]
- Nakajima, H.; Uchida, K.; Guerrero, A.R.; Watanabe, S.; Sugita, D.; Takeura, N.; Yoshida, A.; Long, G.; Wright, K.T.; Johnson, W.E.; et al. Transplantation of mesenchymal stem cells promotes an alternative pathway of macrophage activation and functional recovery after spinal cord injury. J. Neurotrauma 2012, 29, 1614–1625. [Google Scholar] [CrossRef] [PubMed]
- Dasari, V.R.; Spomar, D.G.; Gondi, C.S.; Sloffer, C.A.; Saving, K.L.; Gujrati, M.; Rao, J.S.; Dinh, D.H. Axonal remyelination by cord blood stem cells after spinal cord injury. J. Neurotrauma 2007, 24, 391–410. [Google Scholar] [CrossRef] [PubMed]
- Osaka, M.; Honmou, O.; Murakami, T.; Nonaka, T.; Houkin, K.; Hamada, H.; Kocsis, J.D. Intravenous administration of mesenchymal stem cells derived from bone marrow after contusive spinal cord injury improves functional outcome. Brain Res. 2010, 1343, 226–235. [Google Scholar] [CrossRef] [PubMed]
- Ide, C.; Nakai, Y.; Nakano, N.; Seo, T.B.; Yamada, Y.; Endo, K.; Noda, T.; Saito, F.; Suzuki, Y.; Fukushima, M.; et al. Bone marrow stromal cell transplantation for treatment of sub-acute spinal cord injury in the rat. Brain Res. 2010, 1332, 32–47. [Google Scholar] [CrossRef] [PubMed]
- Hodgetts, S.I.; Simmons, P.J.; Plant, G.W. A comparison of the behavioral and anatomical outcomes in sub-acute and chronic spinal cord injury models following treatment with human mesenchymal precursor cell transplantation and recombinant decorin. Exp. Neurol. 2013, 248, 343–359. [Google Scholar] [CrossRef] [PubMed]
- Kumagai, G.; Tsoulfas, P.; Toh, S.; McNiece, I.; Bramlett, H.M.; Dietrich, W.D. Genetically modified mesenchymal stem cells (MSCs) promote axonal regeneration and prevent hypersensitivity after spinal cord injury. Exp. Neurol. 2013, 248, 369–380. [Google Scholar] [CrossRef] [PubMed]
- Penha, E.M.; Meira, C.S.; Guimarães, E.T.; Mendonça, M.V.P.; Gravely, F.A.; Pinheiro, C.M.B.; Pinheiro, T.M.B.; Barrouin-Melo, S.M.; Ribeiro-dos-Santos, R.; Soares, M.B.P. Use of autologous mesenchymal stem cells derived from bone marrow for the treatment of naturally injured spinal cord in dogs. Stem Cell Int. 2014, 2014. [Google Scholar] [CrossRef] [PubMed]
- Kang, K.; Kim, S.W.; Oh, Y.H.; Yu, J.W.; Kim, K.Y.; Park, H.K.; Han, H. A 37-year-old spinal cord-injured female patient, transplanted of multipotent stem cells from human UC blood, with improved sensory perception and mobility, both functionally and morphologically: A case study. Cytotherapy 2005, 7, 368–373. [Google Scholar] [CrossRef] [PubMed]
- Adewumi, O.; Aflatoonian, B.; Ahrlund-Richter, L.; Amit, M.; Andrews, P.W.; Beighton, G.; Bello, P.A.; Benvenisty, N.; Berry, L.S.; Bevan, S.; et al. Characterization of human embryonic stem cell lines by the International Stem Cell Initiative. Nat. Biotechnol. 2007, 25, 803–816. [Google Scholar] [CrossRef] [PubMed]
- Nógrádi, A. Transplantation of Neural Tissue into the Spinal Cord; Landes Bioscience: Austin, TX, USA, 2006. [Google Scholar]
- Reier, P.J.; Anderson, D.K.; Thompson, F.J.; Stokes, B.T. Neural tissue transplantation and CNS trauma: Anatomical and functional repair of the injured spinal cord. J. Neurotrauma 1992, 9, S223–S248. [Google Scholar] [PubMed]
- Evans, M.J.; Kaufman, M.H. Establishment in culture of pluripotential cells from mouse embryos. Nature 1981, 292, 154–156. [Google Scholar] [CrossRef] [PubMed]
- Martin, G.R. Isolation of a pluripotent cell line from early mouse embryos cultured in medium conditioned by teratocarcinoma stem cells. Proc. Natl. Acad. Sci. USA 1981, 78, 7634–7638. [Google Scholar] [CrossRef] [PubMed]
- Pera, M.F.; Trounson, A.O. Human embryonic stem cells: Prospects for development. Development 2004, 131, 5515–5525. [Google Scholar] [CrossRef] [PubMed]
- Thomson, J.A.; Itskovitz-Eldor, J.; Shapiro, S.S.; Waknitz, M.A.; Swiergiel, J.J.; Marshall, V.S.; Jones, J.M. Embryonic stem cell lines derived from human blastocysts. Science 1998, 282, 1145–1147. [Google Scholar] [CrossRef] [PubMed]
- Willerth, S.M. Neural tissue engineering using embryonic and induced pluripotent stem cells. Stem Cell Res. Ther. 2011, 2. [Google Scholar] [CrossRef] [PubMed]
- Johnson, P.J.; Tatara, A.; Shiu, A.; Sakiyama-Elbert, S.E. Controlled release of neurotrophin-3 and platelet derived growth factor from fibrin scaffolds containing neural progenitor cells enhances survival and differentiation into neurons in a subacute model of SCI. Cell Transplant. 2010, 19, 89–101. [Google Scholar] [CrossRef] [PubMed]
- Lu, P.; Jonesa, L.L.; Snyderb, E.Y.; Tuszynskia, M.H. Neural stem cells constitutively secrete neurotrophic factors and promote extensive host axonal growth after spinal cord injury. Exp. Neurol. 2003, 181, 115–129. [Google Scholar] [CrossRef]
- Ogawa, Y.; Sawamoto, K.; Miyata, T.; Miyao, S.; Watanabe, M.; Nakamura, M.; Bregman, B.S.; Koike, M.; Uchiyama, Y.; Toyama, Y.; et al. Transplantation of in vitro—expanded fetal neural progenitor cells results in neurogenesis and functional recovery after spinal cord contusion injury in adult rats. J. Neurosci. Res. 2002, 69, 925–933. [Google Scholar] [CrossRef] [PubMed]
- Iwanami, A.; Kaneko, S.; Nakamura, M.; Kanemura, Y.; Mori, H.; Kobayashi, S.; Yamasaki, M.; Momoshima, S.; Ishii, H.; Ando, K.; et al. Transplantation of human neural stem cells for spinal cord injury in primates. J. Neurosci. Res. 2005, 80, 182–190. [Google Scholar] [CrossRef] [PubMed]
- Keirstead, H.S.; Nistor, G.; Bernal, G.; Totoiu, M.; Cloutier, F.; Sharp, K.; Steward, O. Human embryonic stem cell-derived oligodendrocyte progenitor cell transplants remyelinate and restore locomotion after spinal cord injury. J. Neurosci. 2005, 25, 4694–4705. [Google Scholar] [CrossRef] [PubMed]
- Cloutier, F.; Siegenthaler, M.M.; Nistor, G.; Keirstead, H.S. Transplantation of human embryonic stem cell-derived oligodendrocyte progenitors into rat spinal cord injuries does not cause harm. Rengen. Med. 2006, 1, 469–479. [Google Scholar] [CrossRef] [PubMed]
- Bain, G.; Kitchens, D.; Yao, M.; Huettner, J.E.; Gottlieb, D.I. Embryonic stem cells express neuronal properties in vitro. Dev. Biol. 1995, 168, 342–357. [Google Scholar] [CrossRef] [PubMed]
- McDonald, J.W.; Liu, X.Z.; Qu, Y.; Liu, S.; Mickey, S.K.; Turetsky, D.; Gottlieb, D.I.; Choi, D.W. Transplanted embryonic stem cells survive, differentiate and promote recovery in injured rat spinal cord. Nat. Med. 1999, 5, 1410–1412. [Google Scholar] [CrossRef] [PubMed]
- Kayama, M.; Kurokawa, M.S.; Ueda, Y.; Ueno, H.; Kumagai, Y.; Chiba, S.; Takada, E.; Ueno, S.; Tadokoro, M.; Suzuki, N. Transfection with pax6 gene of mouse embryonic stem cells and subsequent cell cloning induced retinal neuron progenitors, including retinal ganglion cell-like cells, in vitro. Ophthalmic Res. 2009, 43, 79–91. [Google Scholar] [CrossRef] [PubMed]
- Sharp, J.; Frame, J.; Siegenthaler, M.; Nistor, G.; Keirstead, H.S. Human embryonic stem cell-derived oligodendrocyte progenitor cell transplants improve recovery after cervical spinal cord injury. Stem Cells 2010, 28, 152–163. [Google Scholar] [CrossRef] [PubMed]
- Karimi-Abdolrezaee, S.; Eftekharpour, E.; Wang, J.; Morshead, C.M.; Fehlings, M.G. Delayed transplantation of adult neural precursor cells promotes remyelination and functional neurological recovery after spinal cord injury. J. Neurosci. 2006, 26, 3377–3389. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Qu, Y.; Stewart, T.J.; Howard, M.J.; Chakrabortty, S.; Holekamp, T.F.; McDonald, J.W. Embryonic stem cells differentiate into oligodendrocytes and myelinate in culture and after spinal cord transplantation. Proc. Natl. Acad. Sci. USA 2000, 97, 6126–6131. [Google Scholar] [CrossRef] [PubMed]
- Faulkner, J.; Keirstead, H.S. Human embryonic stem cell-derived oligodendrocyte progenitors for the treatment of spinal cord injury. Transp. Immunol. 2005, 15, 131–142. [Google Scholar] [CrossRef] [PubMed]
- Hasegawa, K.; Chang, Y.W.; Li, H.; Berlin, Y.; Ikeda, O.; Kane-Goldsmith, N.; Grumet, M. Embryonic radial glia bridge spinal cord lesions and promote functional recovery following spinal cord injury. Exp. Neurol. 2005, 193, 394–410. [Google Scholar] [CrossRef] [PubMed]
- Nistor, G.; Siegenthaler, M.M.; Poirier, S.N.; Rossi, S.; Poole, A.J.; Charlton, M.E.; McNeish, J.D.; Airriess, C.N.; Keirstead, H.S. Derivation of high purity neuronal progenitors from human embryonic stem cells. PLoS ONE 2011, 6, e20692. [Google Scholar] [CrossRef] [PubMed]
- Peljto, M.; Hynek, W. Programming embryonic stem cells to neuronal subtypes. Curr. Opin. Neurobiol. 2011, 21, 43–51. [Google Scholar] [CrossRef] [PubMed]
- Gabut, M.; Samavarchi-Tehrani, P.; Wang, X.; Slobodeniuc, V.; O'Hanlon, D.; Sung, H.K.; Alvarez, M.; Talukder, S.; Pan, Q.; Mazzoni, E.O.; et al. An alternative splicing switch regulates embryonic stem cell pluripotency and reprogramming. Cell 2011, 147, 132–146. [Google Scholar] [CrossRef] [PubMed]
- Son, E.Y.; Ichida, J.K.; Wainger, B.J.; Toma, J.S.; Rafuse, V.F.; Woolf, C.J.; Eggan, K. Conversion of mouse and human fibroblasts into functional spinal motor neurons. Cell Stem Cell 2011, 9, 205–218. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.A.; Huang, Y.P.; Mazzoni, E.O.; Tan, G.C.; Zavadil, J.; Wichterle, H. miR-17-3p controls spinal neural progenitor patterning by regulating Olig2/Irx3 cross-repressive loop. Neuron 2011, 69, 721–735. [Google Scholar] [CrossRef] [PubMed]
- Wyatt, T.J.; Rossi, S.L.; Siegenthaler, M.M.; Frame, J.; Robles, R.; Nistor, G.; Keirstead, H.S. Human motor neuron progenitor transplantation leads to endogenous neuronal sparing in 3 models of motor neuron loss. Stem Cells Int. 2011, 2011. [Google Scholar] [CrossRef] [PubMed]
- McCreedy, D.A.; Rieger, C.R.; Gottlieb, D.I.; Sakiyama-Elbert, S.E. Transgenic enrichment of mouse embryonic stem cell-derived progenitor motor neurons. Stem Cell Res. 2012, 8, 368–378. [Google Scholar] [CrossRef] [PubMed]
- Brown, C.R.; Butts, J.C.; McCreedy, D.A.; Sakiyama-Elbert, S.E. Generation of V2a interneurons from mouse embryonic stem cells. Stem Cells Dev. 2014, 23, 1765–1776. [Google Scholar] [CrossRef] [PubMed]
- McCreedy, D.A.; Brown, C.R.; Butts, J.C.; Xu, H.; Huettner, J.E.; Sakiyama-Elbert, S.E. A new method for generating high purity motoneurons from mouse embryonic stem cells. Biotechnol. Bioeng. 2014, 111, 2041–2055. [Google Scholar] [CrossRef] [PubMed]
- McCreedy, D.A.; Wilems, T.S.; Xu, H.; Butts, J.C.; Brown, C.R.; Smith, A.W.; Sakiyama-Elbert, S.E. Survival, differentiation, and migration of high-purity mouse embryonic stem cell-derived progenitor motor neurons in fibrin scaffolds after sub-acute spinal cord injury. Biomater. Sci. 2014, 2, 1672–1682. [Google Scholar] [CrossRef] [PubMed]
- Ideguchi, M.; Palmer, T.D.; Recht, L.D.; Weimann, J.M. Murine embryonic stem cell-derived pyramidal neurons integrate into the cerebral cortex and appropriately project axons to subcortical targets. J. Neurosci. 2010, 30, 894–904. [Google Scholar] [CrossRef] [PubMed]
- Yamamori, T.; Fukada, K.; Aebersold, R.; Korsching, S.; Fann, M.J. The cholinergic neuronal differentiation factor from heart cells is identical to leukemia inhibitory factor. Science 1989, 246, 1412–1416. [Google Scholar] [CrossRef] [PubMed]
- Diener, P.S.; Bregman, B.S. Fetal spinal cord transplants support growth of supraspinal and segmental projections after cervical spinal cord hemisection in the neonatal rat. J. Neurosci. 1998, 18, 779–793. [Google Scholar] [PubMed]
- Lepore, A.C.; Bakshi, A.; Swanger, S.A.; Rao, M.S.; Fischer, I. Neural precursor cells can be delivered into the injured cervical spinal cord by intrathecal injection at the lumbar cord. Brain Res. 2005, 1045, 206–216. [Google Scholar] [CrossRef] [PubMed]
- Mitsui, T.; Shumsky, J.S.; Lepore, A.C.; Murray, M.; Fischer, I. Transplantation of neuronal and glial restricted precursors into contused spinal cord improves bladder and motor functions, decreases thermal hypersensitivity, and modifies intraspinal circuitry. J. Neurosci. 2005, 25, 9624–9636. [Google Scholar] [CrossRef] [PubMed]
- Lepore, A.C.; Fischer, I. Lineage-restricted neural precursors survive, migrate, and differentiate following transplantation into the injured adult spinal cord. Exp. Neurol. 2005, 194, 230–242. [Google Scholar] [CrossRef] [PubMed]
- Ruff, C.A.; Wilcox, J.T.; Fehlings, M.G. Cell-based transplantation strategies to promote plasticity following spinal cord injury. Exp. Neurol. 2012, 235, 78–90. [Google Scholar] [CrossRef] [PubMed]
- Church, E.W.; Halpern, C.H.; Faught, R.W.; Balmuri, U.; Attiah, M.A.; Hayden, S.; Kerr, M.; Maloney-Wilensky, E.; Bynum, J.; Dante, S.J.; et al. Cervical laminoforaminotomy for radiculopathy: Symptomatic and functional outcomes in a large cohort with long-term follow-up. Surg. Neurol. Int. 2014, 5, S536–S543. [Google Scholar] [CrossRef] [PubMed]
- Giusto, E.; Donegà, M.; Cossetti, C.; Pluchino, S. Neuro-immune interactions of neural stem cell transplants: From animal disease models to human trials. Exp. Neurol. 2014, 260, 19–32. [Google Scholar] [CrossRef] [PubMed]
- Trounson, A.; Thakar, R.G.; Lomax, G.; Gibbons, D. Clinical trials for stem cell therapies. BMC Med. 2011, 9. [Google Scholar] [CrossRef] [PubMed]
- Guzman, R.; Uchida, N.; Bliss, T.M.; He, D.; Christopherson, K.K.; Stellwagen, D.; Capela, A.; Greve, J.; Malenka, R.C.; Moseley, M.E.; et al. Long-term monitoring of transplanted human neural stem cells in developmental and pathological contexts with MRI. Proc. Natl. Acad. Sci. USA 2007, 104, 10211–10216. [Google Scholar] [CrossRef] [PubMed]
- Mothe, A.J.; Tator, C.H. Advances in stem cell therapy for spinal cord injury. J. Clin. Investig. 2012, 122, 3824–3834. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Xu, C.C.; Li, J.; Guan, X.Y.; Gao, L.; Ma, L.X.; Li, R.X.; Peng, Y.W.; Zhu, G.P. Transplantation of oligodendrocyte precursor cells improves locomotion deficits in rats with spinal cord irradiation injury. PLoS ONE 2013, 8, e57534. [Google Scholar] [CrossRef] [PubMed]
- Rossi, S.L.; Nistor, G.; Wyatt, T.; Yin, H.Z.; Poole, A.J.; Weiss, J.H.; Gardener, M.J.; Dijkstra, S.; Fischer, D.F.; Keirstead, H.S. Histological and functional benefit following transplantation of motor neuron progenitors to the injured rat spinal cord. PLoS ONE 2010, 5, e11852. [Google Scholar] [CrossRef] [PubMed]
- Sandner, B.; Ciatipis, M.; Motsch, M.; Soljanik, I.; Weidner, N.; Blesch, A. Limited functional effects of subacute syngeneic bone marrow stromal cell transplantation after rat spinal cord contusion injury. Cell Transplant. 2016, 25, 125–139. [Google Scholar] [CrossRef] [PubMed]
- Prockop, D.J. Further proof of the plasticity of adult stem cells and their role in tissue repair. J. Cell Biol. 2003, 160, 807–809. [Google Scholar] [CrossRef] [PubMed]
- Gallo, M.P.; Ramella, R.; Alloatti, G.; Penna, C.; Pagliaro, P.; Marcantoni, A.; Bonafé, F.; Losano, G.; Levi, R. Limited plasticity of mesenchymal stem cells cocultured with adult cardiomyocytes. J. Cell. Biochem. 2007, 100, 86–99. [Google Scholar] [CrossRef] [PubMed]
- Zouani, O.F.; Yifeng, L.; Marie-Christine, D. Pericytes, stem-cell-like cells, but not mesenchymal stem cells are recruited to support microvascular tube stabilization. Small 2013, 9, 3070–3075. [Google Scholar] [CrossRef] [PubMed]
- Prockop, D.J. Repair of tissues by adult stem/progenitor cells (MSCs): Controversies, myths, and changing paradigms. Mol. Ther. 2009, 17, 939–946. [Google Scholar] [CrossRef] [PubMed]
- Prockop, D.J.; Brenner, M.; Fibbe, W.E.; Horwitz, E.; Le Blanc, K.; Phinney, D.G.; Simmons, P.J.; Sensebe, L.; Keating, A. Defining the risks of mesenchymal stromal cell therapy. Cytotherapy 2010, 12, 576–578. [Google Scholar] [CrossRef] [PubMed]
- Ankrum, J.A.; Joon, F.O.; Karp, J.M. Mesenchymal stem cells: Immune evasive, not immune privileged. Nat. Biotechnol. 2014, 32, 252–260. [Google Scholar] [CrossRef] [PubMed]
- Nisbet, M.C.; Dominique, B.; Adrianne, K. Framing science the stem cell controversy in an age of press/politics. Int. J. Press 2003, 8, 36–70. [Google Scholar] [CrossRef]
- Cao, Q.L.; Howard, R.M.; Dennison, J.B.; Whittemore, S.R. Differentiation of engrafted neuronal-restricted precursor cells is inhibited in the traumatically injured spinal cord. Exp. Neurol. 2002, 177, 349–359. [Google Scholar] [CrossRef] [PubMed]
- Cao, Q.L.; Zhang, Y.P.; Howard, R.M.; Walters, W.M.; Tsoulfas, P.; Whittemore, S.R. Pluripotent stem cells engrafted into the normal or lesioned adult rat spinal cord are restricted to a glial lineage. Exp. Neurol. 2001, 167, 48–58. [Google Scholar] [CrossRef] [PubMed]
- Dressel, R. Effects of histocompatibility and host immune responses on the tumorigenicity of pluripotent stem cells. Semin. Immunopathol. 2011, 33, 573. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, Z.; Huang, M.; Hu, S.; Wilson, K.D.; Dey, D.; Wu, J.C. Dissecting the oncogenic and tumorigenic potential of differentiated human induced pluripotent stem cells and human embryonic stem cells. Cancer Res. 2011, 71, 5030–5039. [Google Scholar] [CrossRef] [PubMed]
- Iwai, H.; Shimada, H.; Nishimura, S.; Kobayashi, Y.; Itakura, G.; Hori, K.; Hikishima, K.; Ebise, H.; Negishi, N.; Shibata, S.; et al. Allogeneic neural stem/progenitor cells derived from embryonic stem cells promote functional recovery after transplantation into injured spinal cord of nonhuman primates. Stem Cells Transl. Med. 2015. [Google Scholar] [CrossRef] [PubMed]
- Blum, B.; Nissim, B. The tumorigenicity of human embryonic stem cells. Adv. Cancer Res. 2008, 100, 133–158. [Google Scholar] [PubMed]
- Baker, D.E.; Harrison, N.J.; Maltby, E.; Smith, K.; Moore, H.D.; Shaw, P.J.; Heath, P.R.; Holden, H.; Andrews, P.W. Adaptation to culture of human embryonic stem cells and oncogenesis in vivo. Nat. Biotechnol. 2007, 25, 207–215. [Google Scholar] [CrossRef] [PubMed]
- Lefort, N.; Feyeux, M.; Bas, C.; Féraud, O.; Bennaceur-Griscelli, A.; Tachdjian, G.; Peschanski, M.; Perrier, A.L. Human embryonic stem cells reveal recurrent genomic instability at 20q11.21. Nat. Biotechnol. 2008, 26, 1364–1366. [Google Scholar] [CrossRef] [PubMed]
- Mayshar, Y.; Ofra, Y.; Nissim, B. Teratogen screening using transcriptome profiling of differentiating human embryonic stem cells. J. Cell. Mol. Med. 2011, 15, 1393–1401. [Google Scholar] [CrossRef] [PubMed]
- Klimanskaya, I.; Chung, Y.; Becker, S.; Lu, S.J.; Lanza, R. Human embryonic stem cell lines derived from single blastomeres. Nature 2006, 444, 481–485. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, K.; Shinya, Y. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 2006, 126, 663–676. [Google Scholar] [CrossRef] [PubMed]
- Miura, K.; Okada, Y.; Aoi, T.; Okada, A.; Takahashi, K.; Okita, K.; Nakagawa, M.; Koyanagi, M.; Tanabe, K.; Ohnuki, M.; et al. Variation in the safety of induced pluripotent stem cell lines. Nat. Biotechnol. 2009, 27, 743–745. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, K.; Tanabe, K.; Ohnuki, M.; Narita, M.; Ichisaka, T.; Tomoda, K.; Yamanaka, S. Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell 2007, 131, 861–872. [Google Scholar] [CrossRef] [PubMed]
- Yamanaka, S. Patient-specific pluripotent stem cells become even more accessible. Cell Stem Cell 2010, 7, 1–2. [Google Scholar] [CrossRef] [PubMed]
- Fusaki, N.; Ban, H.; Nishiyama, A.; Saeki, K.; Hasegawa, M. Efficient induction of transgene-free human pluripotent stem cells using a vector based on Sendai virus, an RNA virus that does not integrate into the host genome. Proc. Jpn. Acad. Ser. B 2009, 85, 348–362. [Google Scholar] [CrossRef]
- Nori, S.; Okada, Y.; Nishimura, S.; Sasaki, T.; Itakura, G.; Kobayashi, Y.; Renault-Mihara, F.; Shimizu, A.; Koya, I.; Yoshida, R. Long-term safety issues of iPSC-based cell therapy in a spinal cord injury model: Oncogenic transformation with epithelial-mesenchymal transition. Stem Cell Rep. 2015, 4, 360–373. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.; Wu, S.; Joo, J.Y.; Zhu, S.; Han, D.W.; Lin, T.; Trauger, S.; Bien, G.; Yao, S.; Zhu, Y.; et al. Generation of induced pluripotent stem cells using recombinant proteins. Cell Stem Cell 2009, 4, 381–384. [Google Scholar] [CrossRef] [PubMed]
- Warren, L.; Manos, P.D.; Ahfeldt, T.; Loh, Y.H.; Li, H.; Lau, F.; Ebina, W.; Mandal, P.K.; Smith, Z.D.; Meissner, A.; et al. Highly efficient reprogramming to pluripotency and directed differentiation of human cells with synthetic modified mRNA. Cell Stem Cell 2010, 7, 618–630. [Google Scholar] [CrossRef] [PubMed]
- Rao, M.S.; Nasir, M. Assessing iPSC reprogramming methods for their suitability in translational medicine. J. Cell. Biochem. 2012, 113, 3061–3068. [Google Scholar] [CrossRef] [PubMed]
- Yamanaka, S. Induced pluripotent stem cells: Past, present, and future. Cell Stem Cell 2012, 10, 678–684. [Google Scholar] [CrossRef] [PubMed]
- Chin, M.H.; Mason, M.J.; Xie, W.; Volinia, S.; Singer, M.; Peterson, C.; Ambartsumyan, G.; Aimiuwu, O.; Richter, L.; Zhang, J.; et al. Induced pluripotent stem cells and embryonic stem cells are distinguished by gene expression signatures. Cell Stem Cell 2009, 5, 111–123. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.; Lee, S.; Mallard, W.; Clement, K.; Tagliazucchi, G.M.; Lim, H.; Choi, I.Y.; Ferrari, F.; Tsankov, A.M.; Pop, R.; et al. A comparison of genetically matched cell lines reveals the equivalence of human iPSCs and ESCs. Nat. Biotechnol. 2015, 33, 1173–1181. [Google Scholar] [CrossRef] [PubMed]
- Phanstiel, D.H.; Brumbaugh, J.; Wenger, C.D.; Tian, S.; Probasco, M.D.; Bailey, D.J.; Swaney, D.L.; Tervo, M.A.; Bolin, J.M.; Ruotti, V.; et al. Proteomic and phosphoproteomic comparison of human ES and iPS cells. Nat. Methods 2011, 8, 821–827. [Google Scholar] [CrossRef] [PubMed]
- Guenther, M.G.; Frampton, G.M.; Soldner, F.; Hockemeyer, D.; Mitalipova, M.; Jaenisch, R.; Young, R.A. Chromatin structure and gene expression programs of human embryonic and induced pluripotent stem cells. Cell Stem Cell 2010, 7, 249–257. [Google Scholar] [CrossRef] [PubMed]
- Munoz, J.; Low, T.Y.; Kok, Y.J.; Chin, A.; Frese, C.K.; Ding, V.; Choo, A.; Heck, A.J. The quantitative proteomes of human-induced pluripotent stem cells and embryonic stem cells. Mol. Syst. Biol. 2011, 7. [Google Scholar] [CrossRef] [PubMed]
- Prigione, A.; Fauler, B.; Lurz, R.; Lehrach, H.; Adjaye, J. The senescence-related mitochondrial/oxidative stress pathway is repressed in human induced pluripotent stem cells. Stem Cells 2010, 28, 721–733. [Google Scholar] [CrossRef] [PubMed]
- Puri, M.C.; Andras, N. Concise review: Embryonic stem cells versus induced pluripotent stem cells: The game is on. Stem Cells 2012, 30, 10–14. [Google Scholar] [CrossRef] [PubMed]
- Hirasaki, M.; Hiraki-Kamon, K.; Kamon, M.; Suzuki, A.; Katano, M.; Nishimoto, M.; Okuda, A. Striking similarity in the gene expression levels of individual Myc module members among ESCs, EpiSCs, and partial iPSCs. PLoS ONE 2013, 8, e83769. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.G.; Mallon, B.S.; McKay, R.D.; Robey, P.G. Human pluripotent stem cell culture: Considerations for maintenance, expansion, and therapeutics. Cell Stem Cell 2014, 14, 13–26. [Google Scholar] [CrossRef] [PubMed]
- Mallon, B.S.; Hamilton, R.S.; Kozhich, O.A.; Johnson, K.R.; Fann, Y.C.; Rao, M.S.; Robey, P.G. Comparison of the molecular profiles of human embryonic and induced pluripotent stem cells of isogenic origin. Stem Cell Res. 2014, 12, 376–386. [Google Scholar] [CrossRef] [PubMed]
- Taylor, C.J.; Peacock, S.; Chaudhry, A.N.; Bradley, J.A.; Bolton, E.M. Generating an iPSC bank for HLA-matched tissue transplantation based on known donor and recipient HLA types. Cell Stem Cell 2012, 11, 147–152. [Google Scholar] [CrossRef] [PubMed]
- Okano, H.; Shinya, Y. iPS cell technologies: Significance and applications to CNS regeneration and disease. Mol. Brain 2014, 7. [Google Scholar] [CrossRef] [PubMed]
- Polo, J.M.; Liu, S.; Figueroa, M.E.; Kulalert, W.; Eminli, S.; Tan, K.Y.; Apostolou, E.; Stadtfeld, M.; Li, Y.; Shioda, T.; et al. Cell type of origin influences the molecular and functional properties of mouse induced pluripotent stem cells. Nat. Biotechnol. 2010, 28, 848–855. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.; Doi, A.; Wen, B.; Ng, K.; Zhao, R.; Cahan, P.; Kim, J.; Aryee, M.J.; Ji, H.; Ehrlich, L.I.R.; et al. Epigenetic memory in induced pluripotent stem cells. Nature 2010, 467, 285–290. [Google Scholar] [CrossRef] [PubMed]
- Han, S.W.; Williams, L.A.; Eggan, K.C. Constructing and deconstructing stem cell models of neurological disease. Neuron 2011, 70, 626–644. [Google Scholar] [CrossRef] [PubMed]
- Marchetto, M.C.; Gage, F.H. Modeling brain disease in a dish: Really? Cell Stem Cell 2012, 10, 642–645. [Google Scholar] [CrossRef] [PubMed]
- Brennand, K.J.; Simone, A.; Tran, N.; Gage, F.H. Modeling psychiatric disorders at the cellular and network levels. Mol. Psychiatr. 2012, 17, 1239–1253. [Google Scholar] [CrossRef] [PubMed]
- Barker, R.A. Developing stem cell therapies for Parkinson’s disease: Waiting until the time is right. Cell Stem Cell 2014, 15, 539–542. [Google Scholar] [CrossRef] [PubMed]
- Beevers, J.E.; Caffrey, T.M.; Wade-Martins, R. Induced pluripotent stem cell (iPSC)-derived dopaminergic models of Parkinson’s disease. Biochem. Soc. Trans. 2013, 41, 1503–1508. [Google Scholar] [CrossRef] [PubMed]
- Perrier, A.; Marc, P. How can human pluripotent stem cells help decipher and cure Huntington’s disease? Cell Stem Cell 2012, 11, 153–161. [Google Scholar] [CrossRef] [PubMed]
- Israel, M.A.; Yuan, S.H.; Bardy, C.; Reyna, S.M.; Mu, Y.; Herrera, C.; Hefferan, M.P.; van Gorp, S.; Nazor, K.L.; Boscolo, F.S.; et al. Probing sporadic and familial Alzheimer’s disease using induced pluripotent stem cells. Nature 2012, 482, 216–220. [Google Scholar] [CrossRef] [PubMed]
- Liu, G.-H.; Zhichao, D.; Belmonte, J.C.I. iPSC technology to study human aging and aging-related disorders. Curr. Opin. Cell Biol. 2012, 24, 765–774. [Google Scholar] [CrossRef] [PubMed]
- Trounson, A.; Kelly, A.S.; DeWitt, N.D. Human disease modeling with induced pluripotent stem cells. Curr. Opin. Genet. Dev. 2012, 22, 509–516. [Google Scholar] [CrossRef] [PubMed]
- Guo, Z.; Zhang, L.; Wu, Z.; Chen, Y.; Wang, F.; Chen, G. In vivo direct reprogramming of reactive glial cells into functional neurons after brain injury and in an Alzheimer’s disease model. Cell Stem Cell 2014, 14, 188–202. [Google Scholar] [CrossRef] [PubMed]
- Krencik, R.; Su-Chun, Z. Directed differentiation of functional astroglial subtypes from human pluripotent stem cells. Nat. Protoc. 2011, 6, 1710–1717. [Google Scholar] [CrossRef] [PubMed]
- Yang, N.; Zuchero, J.B.; Ahlenius, H.; Marro, S.; Ng, Y.H.; Vierbuchen, T.; Hawkins, J.S.; Geissler, R.; Barres, B.A.; Wernig, M. Generation of oligodendroglial cells by direct lineage conversion. Nat. Biotechnol. 2013, 31, 434–439. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Pak, C.; Han, Y.; Ahlenius, H.; Zhang, Z.; Chanda, S.; Marro, S.; Patzke, C.; Acuna, C.; Covy, J.; et al. Rapid single-step induction of functional neurons from human pluripotent stem cells. Neuron 2013, 78, 785–798. [Google Scholar] [CrossRef] [PubMed]
- Ambasudhan, R.; Talantova, M.; Coleman, R.; Yuan, X.; Zhu, S.; Lipton, S.A.; Ding, S. Direct reprogramming of adult human fibroblasts to functional neurons under defined conditions. Cell Stem Cell 2011, 9, 113–118. [Google Scholar] [CrossRef] [PubMed]
- Hodgetts, S.I.; Michael, E.; Harvey, A.R. The state of play with iPSCs and spinal cord injury models. J. Clin. Med. 2015, 4, 193–203. [Google Scholar] [CrossRef] [PubMed]
- Marchetto, M.C.; Carromeu, C.; Acab, A.; Yu, D.; Yeo, G.W.; Mu, Y.; Chen, G.; Gage, F.H.; Muotri, A.R. A model for neural development and treatment of Rett syndrome using human induced pluripotent stem cells. Cell 2010, 143, 527–539. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Efea, J.A.; Zhua, S.; Talantovac, M.; Yuana, X.; Wang, S.; Liptonc, S.A.; Zhang, K.; Ding, S. Direct reprogramming of mouse fibroblasts to neural progenitors. Proc. Natl. Acad. Sci. USA 2011, 108, 7838–7843. [Google Scholar] [CrossRef] [PubMed]
- Swistowski, A.; Peng, J.; Liu, Q.; Mali, P.; Rao, M.S.; Cheng, L.; Zeng, X. Efficient generation of functional dopaminergic neurons from human induced pluripotent stem cells under defined conditions. Stem Cells 2010, 28, 1893–1904. [Google Scholar] [CrossRef] [PubMed]
- Takebe, T.; Sekine, K.; Enomura, M.; Koike, H.; Kimura, M.; Ogaeri, T.; Zhang, R.R.; Ueno, Y.; Zheng, Y.W.; Koike, N.; et al. Vascularized and functional human liver from an iPSC-derived organ bud transplant. Nature 2013, 499, 481–484. [Google Scholar] [CrossRef] [PubMed]
- Zeng, H.; Guo, M.; Martins-Taylor, K.; Wang, X.; Zhang, Z.; Park, J.W.; Zhan, S.; Kronenberg, M.S.; Lichtler, A.; Liu, H.X.; et al. Specification of region-specific neurons including forebrain glutamatergic neurons from human induced pluripotent stem cells. PLoS ONE 2010, 5, e11853. [Google Scholar] [CrossRef] [PubMed]
- Brennand, K.J.; Simone, A.; Jou, J.; Gelboin-Burkhart, C.; Tran, N.; Sangar, S.; Li, Y.; Mu, Y.; Chen, G.; Yu, D.; et al. Modelling schizophrenia using human induced pluripotent stem cells. Nature 2011, 473, 221–225. [Google Scholar] [CrossRef] [PubMed]
- Hester, M.E.; Murtha, M.J.; Song, S.; Rao, M.; Miranda, C.J.; Meyer, K.; Tian, J.; Boulting, G.; Schaffer, D.V.; Zhu, M.X.; et al. Rapid and efficient generation of functional motor neurons from human pluripotent stem cells using gene delivered transcription factor codes. Mol. Ther. 2011, 19, 1905–1912. [Google Scholar] [CrossRef] [PubMed]
- Boulting, G.L.; Kiskinis, E.; Croft, G.F.; Amoroso, M.W.; Oakley, D.H.; Wainger, B.J.; Williams, D.J.; Kahler, D.J.; Yamaki, M.; Davidow, L.; et al. A functionally characterized test set of human induced pluripotent stem cells. Nat. Biotechnol. 2011, 29, 279–286. [Google Scholar] [CrossRef] [PubMed]
- Faravelli, I.; Frattini, E.; Ramirez, A.; Stuppia, G.; Nizzardo, M.; Corti, S. iPSC-based models to unravel key pathogenetic processes underlying motor neuron disease development. J. Clin. Med. 2014, 3, 1124–1145. [Google Scholar] [CrossRef] [PubMed]
- Hu, B.Y.; Weick, J.P.; Yu, J.; Ma, L.X.; Zhang, X.Q.; Thomson, J.A.; Zhang, S.C. Neural differentiation of human induced pluripotent stem cells follows developmental principles but with variable potency. Proc. Natl. Acad. Sci. USA 2010, 107, 4335–4340. [Google Scholar] [CrossRef] [PubMed]
- Tucker, B.A.; Mullins, R.F.; Streb, L.M.; Anfinson, K.; Eyestone, M.E.; Kaalberg, E.; Riker, M.J.; Drack, A.V.; Braun, T.A.; Stone, E.M. Patient-specific iPSC-derived photoreceptor precursor cells as a means to investigate retinitis pigmentosa. Elife 2013, 2, e00824. [Google Scholar] [CrossRef] [PubMed]
- Buchholz, D.E.; Hikita, S.T.; Rowland, T.J.; Friedrich, A.M.; Hinman, C.R.; Johnson, L.V.; Clegg, D.O. Derivation of functional retinal pigmented epithelium from induced pluripotent stem cells. Stem Cells 2009, 27, 2427–2434. [Google Scholar] [CrossRef] [PubMed]
- Gensel, J.C.; Tovar, C.A.; Hamers, F.P.; Deibert, R.J.; Beattie, M.S.; Bresnahan, J.C. Behavioral and histological characterization of unilateral cervical spinal cord contusion injury in rats. J. Neurotrauma 2006, 23, 36–54. [Google Scholar] [CrossRef] [PubMed]
- Nutt, S.E.; Chang, E.A.; Suhr, S.T.; Schlosser, L.O.; Mondello, S.E.; Moritz, C.T.; Cibelli, J.B.; Horner, P.J. Caudalized human iPSC-derived neural progenitor cells produce neurons and glia but fail to restore function in an early chronic spinal cord injury model. Exp. Neurol. 2013, 248, 491–503. [Google Scholar] [CrossRef] [PubMed]
- Li, K.; Javed, E.; Scura, D.; Hala, T.J.; Seetharam, S.; Falnikar, A.; Richard, J.P.; Chorath, A.; Maragakis, N.J.; Wright, M.C.; et al. Human iPS cell-derived astrocyte transplants preserve respiratory function after spinal cord injury. Exp. Neurol. 2015, 271, 479–492. [Google Scholar] [CrossRef] [PubMed]
- Lu, P.; Woodruff, G.; Wang, Y.; Graham, L.; Hunt, M.; Wu, D.; Boehle, E.; Ahmad, R.; Poplawski, G.; Brock, J.; Goldstein, L.S. Long-distance axonal growth from human induced pluripotent stem cells after spinal cord injury. Neuron 2014, 83, 789–796. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, Y.; Okada, Y.; Itakura, G.; Iwai, H.; Nishimura, S.; Yasuda, A.; Nori, S.; Hikishima, K.; Konomi, T.; Fujiyoshi, K.; et al. Pre-evaluated safe human iPSC-derived neural stem cells promote functional recovery after spinal cord injury in common marmoset without tumorigenicity. PLoS ONE 2012, 7, e52787. [Google Scholar] [CrossRef] [PubMed]
- Sroga, J.M.; Jones, T.; Kigerl, K.A.; McGaughy, V.M.; Popovich, P.G. Rats and mice exhibit distinct inflammatory reactions after spinal cord injury. J. Comp. Neurol. 2003, 462, 223–240. [Google Scholar] [CrossRef] [PubMed]
- Norenberg, M.D.; Jon, S.; Alex, M. The pathology of human spinal cord injury: Defining the problems. J. Neurotrauma 2004, 21, 429–440. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Lenart, B.A.; Lee, J.K.; Chen, D.; Shi, P.; Ren, J.; Muehleman, C.; Chen, D.; An, H.S. Histological features of endplates of the mammalian spine: From mice to men. Spine 2014, 39, E312–E317. [Google Scholar] [CrossRef] [PubMed]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Doulames, V.M.; Plant, G.W. Induced Pluripotent Stem Cell Therapies for Cervical Spinal Cord Injury. Int. J. Mol. Sci. 2016, 17, 530. https://doi.org/10.3390/ijms17040530
Doulames VM, Plant GW. Induced Pluripotent Stem Cell Therapies for Cervical Spinal Cord Injury. International Journal of Molecular Sciences. 2016; 17(4):530. https://doi.org/10.3390/ijms17040530
Chicago/Turabian StyleDoulames, Vanessa M., and Giles W. Plant. 2016. "Induced Pluripotent Stem Cell Therapies for Cervical Spinal Cord Injury" International Journal of Molecular Sciences 17, no. 4: 530. https://doi.org/10.3390/ijms17040530
APA StyleDoulames, V. M., & Plant, G. W. (2016). Induced Pluripotent Stem Cell Therapies for Cervical Spinal Cord Injury. International Journal of Molecular Sciences, 17(4), 530. https://doi.org/10.3390/ijms17040530





