Association between Interleukin-6 Promoter Polymorphism (-174 G/C), Serum Interleukin-6 Levels and Mortality in Severe Septic Patients
Abstract
:1. Introduction
2. Results
3. Discussion
4. Materials and Methods
4.1. Design and Subjects
4.2. Variables Recorded
4.3. Blood Samples and Determinations
4.4. Statistical Methods
5. Conclusions
Author Contributions
Conflicts of Interest
Abbreviations
| SOFA | Sepsis-related organ failure assessment |
| COPD | Chronic obstructive pulmonary disease |
| aPTT | Activated partial thromboplastin time |
| IL | Interleukin |
| INR | International normalized ratio |
| PaO2 | Pressure of arterial oxygen |
| FIO2 | Fraction inspired oxygen |
References
- Vincent, J.L.; Sakr, Y.; Sprung, C.L.; Ranieri, V.M.; Reinhart, K.; Gerlach, H.; Moreno, R.; Carlet, J.; Le Gall, J.R.; Payen, D.; Sepsis occurrence in acutely III patients investigators. Sepsis in European intensive care units: Results of the SOAP study. Crit. Care Med. 2006, 34, 344–353. [Google Scholar] [CrossRef] [PubMed]
- Angus, D.C.; Linde-Zwirble, W.T.; Lidicker, J.; Clermont, G.; Carcillo, J.; Pinsky, M.R. Epidemiology of severe sepsis in the United States: Analysis of incidence, outcome, and associated costs of care. Crit. Care Med. 2001, 29, 1303–1310. [Google Scholar] [CrossRef] [PubMed]
- Samraj, R.S.; Zingarelli, B.; Wong, H.R. Role of biomarkers in sepsis care. Shock 2013, 40, 358–365. [Google Scholar] [CrossRef] [PubMed]
- Hack, C.E.; de Groot, E.R.; Felt-Bersma, R.J.; Nuijens, J.H.; Strack Van Schijndel, R.J.; Eerenberg-Belmer, A.J.; Thijs, L.G.; Aarden, L.A. Increased plasma levels of interleukin-6 in sepsis. Blood 1989, 74, 1704–1710. [Google Scholar] [PubMed]
- Patel, R.T.; Deen, K.I.; Youngs, D.; Warwick, J.; Keighley, M.R. Interleukin 6 is a prognostic indicator of outcome in severe intra-abdominal sepsis. Br. J. Surg. 1994, 81, 1306–1308. [Google Scholar] [CrossRef] [PubMed]
- Damas, P.; Ledoux, D.; Nys, M.; Vrindts, Y.; de Groote, D.; Franchimont, P.; Lamy, M. Cytokine serum level during severe sepsis in human IL-6 as a marker of severity. Ann. Surg. 1992, 215, 356–362. [Google Scholar] [CrossRef] [PubMed]
- Miguel-Bayarri, V.; Casanoves-Laparra, E.B.; Pallás-Beneyto, L.; Sancho-Chinesta, S.; Martín-Osorio, L.F.; Tormo-Calandín, C.; Bautista-Rentero, D. Prognostic value of the biomarkers procalcitonin, interleukin-6 and C-reactive protein in severe sepsis. Med. Intensiv. 2012, 36, 556–562. [Google Scholar] [CrossRef] [PubMed]
- Tschaikowsky, K.; Hedwig-Geissing, M.; Braun, G.G.; Radespiel-Troeger, M. Predictive value of procalcitonin, interleukin-6, and C-reactive protein for survival in postoperative patients with severe sepsis. J. Crit. Care 2011, 26, 54–64. [Google Scholar] [CrossRef] [PubMed]
- DeMichele, A.; Martin, A.M.; Mick, R.; Gor, P.; Wray, L.; Klein-Cabral, M.; Athanasiadis, G.; Colligan, T.; Stadtmauer, E.; Weber, B. Interleukin-6-174G>C polymorphism is associated with improved outcome in high-risk breast cancer. Cancer Res. 2003, 63, 8051–8056. [Google Scholar] [PubMed]
- Hefler, L.A.; Grimm, C.; Ackermann, S.; Malur, S.; Radjabi-Rahat, A.R.; Leodolter, S.; Beckmann, M.W.; Zeillinger, R.; Koelbl, H.; Tempfer, C.B. An interleukin-6 gene promoter polymorphism influences the biological phenotype of ovarian cancer. Cancer Res. 2003, 63, 3066–3068. [Google Scholar] [PubMed]
- Burzotta, F.; Iacoviello, L.; di Castelnuovo, A.; Glieca, F.; Luciani, N.; Zamparelli, R.; Schiavello, R.; Donati, M.B.; Maseri, A.; Possati, G.; et al. Relation of the -174 G/C polymorphism of interleukin-6 to interleukin-6 plasma levels and to length of hospitalization after surgical coronary revascularization. Am. J. Cardiol. 2001, 88, 1125–1128. [Google Scholar] [CrossRef]
- Gomes, M.; Coelho, A.; Araújo, A.; Azevedo, A.; Teixeira, A.L.; Catarino, R.; Medeiros, R. IL-6 polymorphism in non-small cell lung cancer: A prognostic value? Tumour Biol. 2015, 36, 3679–3684. [Google Scholar] [CrossRef] [PubMed]
- Balakrishnan, V.S.; Guo, D.; Rao, M.; Jaber, B.L.; Tighiouart, H.; Freeman, R.L.; Huang, C.; King, A.J.; Pereira, B.J.; HEMO Study Group. Cytokine gene polymorphisms in hemodialysis patients: Association with comorbidity, functionality, and serum albumin. Kidney Int. 2004, 65, 1449–1460. [Google Scholar] [CrossRef] [PubMed]
- Sen, A.; Paine, S.K.; Chowdhury, I.H.; Mukherjee, A.; Choudhuri, S.; Saha, A.; Mandal, L.K.; Bhattacharya, B. Impact of interleukin-6 promoter polymorphism and serum interleukin-6 level on the acute inflammation and neovascularization stages of patients with Eales’disease. Mol. Vis. 2011, 17, 2552–2563. [Google Scholar] [PubMed]
- Giannitrapani, L.; Soresi, M.; Giacalone, A.; Campagna, M.E.; Marasà, M.; Cervello, M.; Marasà, S.; Montalto, G. IL-6 -174G/C polymorphism and IL-6 serum levels in patients with liver cirrhosis and hepatocellular carcinoma. OMICS 2011, 15, 183–186. [Google Scholar] [CrossRef] [PubMed]
- Talar-Wojnarowska, R.; Gasiorowska, A.; Smolarz, B.; Romanowicz-Makowska, H.; Kulig, A.; Malecka-Panas, E. Clinical significance of interleukin-6 (IL-6) gene polymorphism and IL-6 serum level in pancreatic adenocarcinoma and chronic pancreatitis. Dig. Dis. Sci. 2009, 54, 683–689. [Google Scholar] [CrossRef] [PubMed]
- Fishman, D.; Faulds, G.; Jeffery, R.; Mohamed-Ali, V.; Yudkin, J.S.; Humphries, S.; Woo, P. The effect of novel polymorphisms in the interleukin-6 (IL-6) gene on IL-6 transcription and plasma IL-6 levels, and an association with systemic-onset juvenile chronic arthritis. J. Clin. Investig. 1998, 102, 1369–1376. [Google Scholar] [CrossRef] [PubMed]
- Feng, B.; Mao, Z.R.; Pang, K.; Zhang, S.L.; Li, L. Association of tumor necrosis factor α-308G/A and interleukin-6 -174G/C gene polymorphism with pneumonia-induced sepsis. J. Crit. Care 2015, 30, 920–233. [Google Scholar] [CrossRef] [PubMed]
- Zidan, H.E.; Elbehedy, R.M.; Azab, S.F. IL6-174 G/C gene polymorphism and its relation to serum IL6 in Egyptian children with community-acquired pneumonia. Cytokine 2014, 67, 60–64. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Hady, H.; El-Naggar, M.; El-Nady, G.; Badr, R.; El-Daker, M. Genetic polymorphisms of IL-6-174 and IL-10–1082 in full term neonates with late onset blood stream infections. J. Pediatr. Infect. Dis. 2009, 4, 357–365. [Google Scholar]
- Schluter, B.; Raufhake, C.; Erren, M.; Schotte, H.; Kipp, F.; Rust, S.; Van Aken, H.; Assmann, G.; Berendes, E. Effect of interleukin-6 promoter polymorphism (-174 G/C) on the incidence and outcome of sepsis. Crit. Care Med. 2002, 30, 32–37. [Google Scholar] [CrossRef] [PubMed]
- Martin-Loeches, I.; Sole-Violan, J.; Rodriguez de Castro, F.; Garcia-Laorden, M.I.; Borderias, L.; Blanquer, J.; Rajas, O.; Briones, M.L.; Aspa, J.; Herrera-Ramos, E.; et al. Variants at the promoter of the interleukin-6 gene are associated with severity and outcome of pneumococcal community-acquired pneumonia. Intensive Care Med. 2012, 38, 256–262. [Google Scholar] [CrossRef] [PubMed]
- Sole-Violan, J.; Rodriguez de Castro, F.; Garcia-Laorden, M.I.; Blanquer, J.; Aspa, J.; Borderías, L.; Briones, M.L.; Rajas, O.; Carrondo, I.M.; Marcos-Ramos, J.A.; et al. Genetic variability in the severity and outcome of community-acquired pneumonia. Respir. Med. 2010, 104, 440–447. [Google Scholar] [CrossRef] [PubMed]
- Sipahi, T.; Pocan, H.; Akar, N. Effect of various genetic polymorphisms on the incidence and outcome of severe sepsis. Clin. Appl. Thromb. Hemost. 2006, 12, 47–54. [Google Scholar] [CrossRef] [PubMed]
- Baier, R.J.; Loggins, J.; Yanamandra, K. IL-10, IL-6 and CD14 polymorphisms and sepsis outcome in ventilated very low birth weight infants. BMC Med. 2006, 4, 10–21. [Google Scholar] [CrossRef] [PubMed]
- Sabelnikovs, O.; Nikitina-Zake, L.; Vanags, I. Association of interleukin 6 promoter polymorphism (-174G/C) with IL-6 level and outcome in severe sepsis. Proc. Latv. Acad. Sci. Sect. B Nat. Exact Appl. Sci. 2008, 62, 162–164. [Google Scholar] [CrossRef]
- Shimada, T.; Oda, S.; Sadahiro, T.; Nakamura, M.; Hirayama, Y.; Watanabe, E.; Abe, R.; Nakada, T.A.; Tateishi, Y.; Otani, S.; et al. Outcome prediction in sepsis combined use of genetic polymorphisms—A study in Japanese population. Cytokine 2011, 54, 79–84. [Google Scholar] [CrossRef] [PubMed]
- Barber, R.C.; Aragaki, C.C.; Rivera-Chavez, F.A.; Purdue, G.F.; Hunt, J.L.; Horton, J.W. TLR4 and TNF-alpha polymorphisms are associated with an increased risk for severe sepsis following burn injury. J. Med. Genet. 2004, 41, 808–813. [Google Scholar] [CrossRef] [PubMed]
- Balding, J.; Healy, C.M.; Livingstone, W.J.; White, B.; Mynett-Johnson, L.; Cafferkey, M.; Smith, O.P. Genomic polymorphic profiles in an Irish population with meningococcaemia: Is it possible to predict severity and outcome of disease? Genes Immun. 2003, 4, 533–540. [Google Scholar] [CrossRef] [PubMed]
- Gao, J.W.; Zhang, A.Q.; Pan, W.; Yue, C.L.; Zeng, L.; Gu, W.; Jiang, J. Association between IL-6-174G/C polymorphism and the risk of sepsis and mortality: A systematic review and meta-analysis. PLoS ONE 2015, 10, e0118843. [Google Scholar] [CrossRef] [PubMed]
- Tischendorf, J.J.; Yagmur, E.; Scholten, D.; Vidacek, D.; Koch, A.; Winograd, R.; Gressner, A.M.; Trautwein, C.; Wasmuth, H.E.; Lammert, F. The interleukin-6 (IL6)-174 G/C promoter genotype is associated with the presence of septic shock and the ex vivo secretion of IL6. Int. J. Immunogenet. 2007, 34, 413–418. [Google Scholar] [CrossRef] [PubMed]
- Feezor, R.J.; Oberholzer, C.; Baker, H.V.; Novick, D.; Rubinstein, M.; Moldawer, L.L.; Pribble, J.; Souza, S.; Dinarello, C.A.; Ertel, W.; et al. Molecular characterization of the acute inflammatory response to infections with gram-negative versus gram-positive bacteria. Infect. Immun. 2003, 71, 5803–5813. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.L.; Chen, H.W.; Yang, P.C.; Peck, K.; Tsai, M.H.; Chen, J.J.; Lin, F.Y. Differential gene expression in gram-negative and gram-positive sepsis. Am. J. Respir. Crit. Care Med. 2004, 169, 1135–1143. [Google Scholar] [CrossRef] [PubMed]
- Accardo Palumbo, A.; Forte, G.I.; Pileri, D.; Vaccarino, L.; Conte, F.; D'Amelio, L.; Palmeri, M.; Triolo, A.; D’Arpa, N.; Scola, L.; et al. Analysis of IL-6, IL-10 and IL-17 genetic polymorphisms as risk factors for sepsis development in burned patients. Burns 2012, 38, 208–213. [Google Scholar] [CrossRef] [PubMed]
- Dellinger, R.P.; Levy, M.M.; Rhodes, A.; Annane, D.; Gerlach, H.; Opal, S.M.; Sevransky, J.E.; Sprung, C.L.; Douglas, I.S.; Jaeschke, R.; et al. Surviving Sepsis Campaign Guidelines Committee including the Pediatric Subgroup. Surviving sepsis campaign: International guidelines for management of severe sepsis and septic shock: 2012. Crit. Care Med. 2013, 41, 580–637. [Google Scholar] [CrossRef] [PubMed]
- Lorente, L.; Martín, M.M.; González-Rivero, A.F.; Ferreres, J.; Solé-Violán, J.; Labarta, L.; Díaz, C.; Jiménez, A.; Borreguero-León, J.M. Serum levels of caspase-cleaved cytokeratin-18 and mortality are associated in severe septic patients: Pilot study. PLoS ONE 2014, 9, e109618. [Google Scholar] [CrossRef] [PubMed]
- Lorente, L.; Martín, M.M.; Almeida, T.; Abreu-González, P.; Ferreres, J.; Solé-Violán, J.; Labarta, L.; Díaz, C.; Jiménez, A. Association between serum total antioxidant capacity and mortality in severe septic patients. J. Crit. Care 2015, 30, 7–12. [Google Scholar] [CrossRef] [PubMed]
- Lorente, L.; Martín, M.M.; Almeida, T.; Hernández, M.; Ferreres, J.; Solé-Violán, J.; Labarta, L.; Díaz, C.; Jiménez, A. Association between serum substance P levels and mortality in patients with severe sepsis. J. Crit. Care 2015, 30, 924–928. [Google Scholar] [CrossRef] [PubMed]
- Lorente, L.; Martín, M.M.; López-Gallardo, E.; Blanquer, J.; Solé-Violán, J.; Labarta, L.; Díaz, C.; Jiménez, A.; Montoya, J.; Ruiz-Pesini, E. Decrease of oxidative phosphorylation system function in severe septic patients. J. Crit. Care 2015, 30, 935–939. [Google Scholar] [CrossRef] [PubMed]
- Vincent, J.L.; Moreno, R.; Takala, J.; Willatts, S.; de Mendonça, A.; Bruining, H.; Reinhart, C.K.; Suter, P.M.; Thijs, L.G. The Sepsis-related Organ Failure Assessment (SOFA) score to describe organ dysfunction/failure. Intensive Care Med. 1996, 22, 707–710. [Google Scholar] [CrossRef] [PubMed]
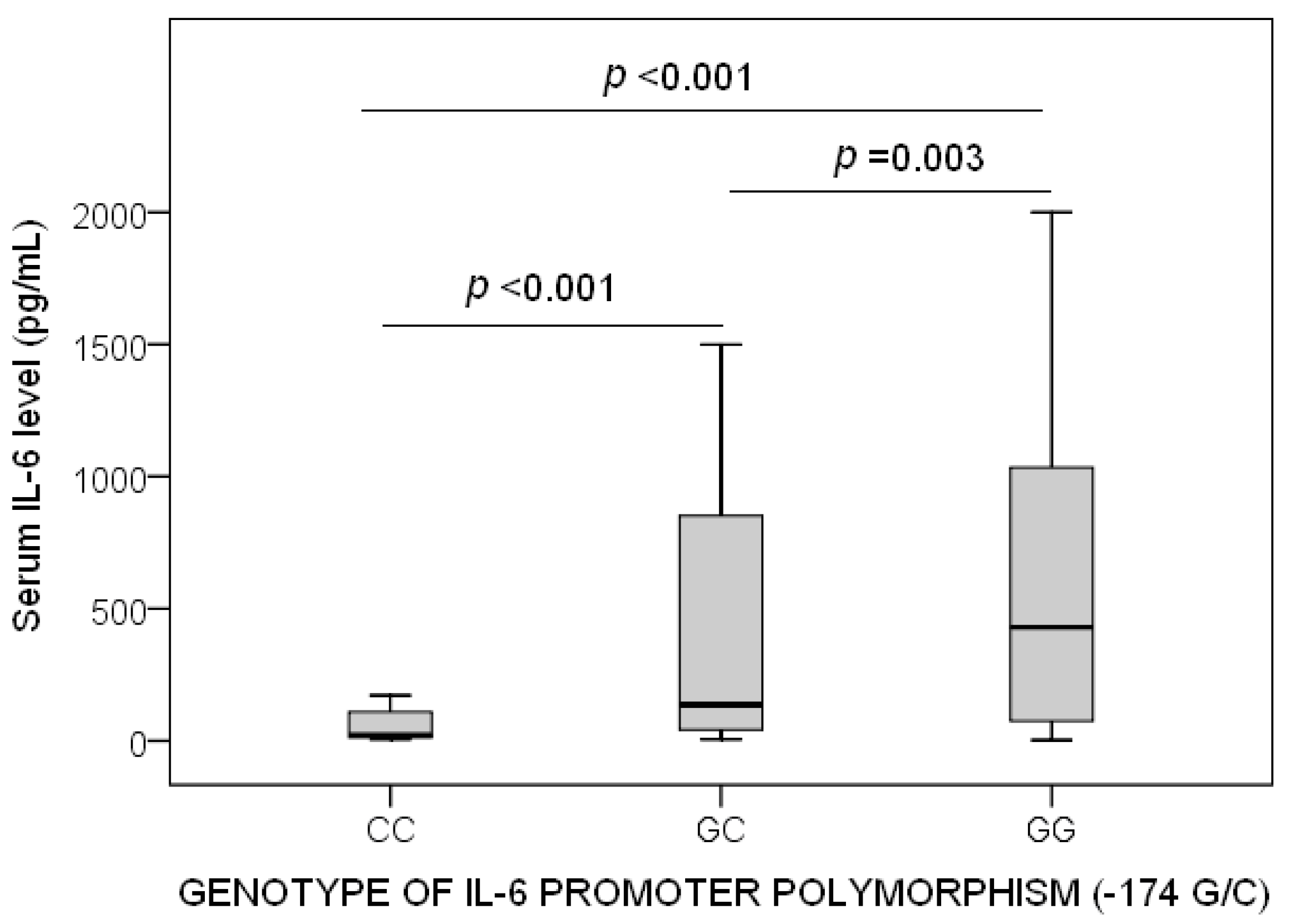
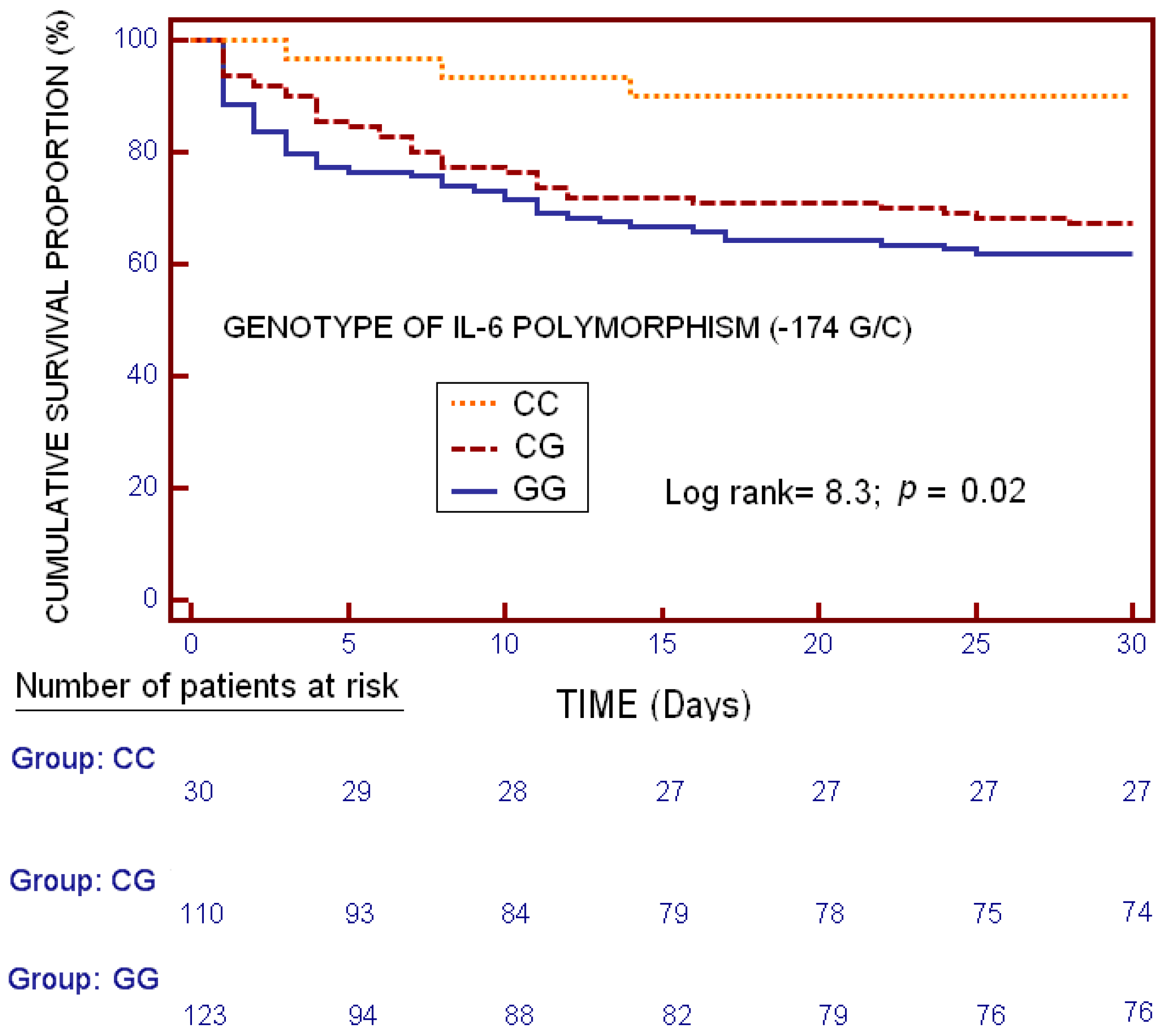
| Demographic and Clinical Characteristics | GG (n = 123) | GC (n = 110) | CC (n = 30) | p-Value |
|---|---|---|---|---|
| Bloodstream infection—n (%) | 17 (13.8) | 22 (20.0) | 3 (10.0) | 0.28 |
| COPD—n (%) | 15 (12.2) | 14 (12.7) | 6 (20.0) | 0.52 |
| Diabetes mellitus—n (%) | 39 (31.7) | 34 (30.9) | 6 (20.0) | 0.44 |
| Empiric antimicrobial treatment—n (%) | – | – | – | 0.28 |
| Unknown whether adequate due to negative cultures | 63 (51.2) | 52 (47.3) | 17 (56.7) | – |
| Unknown whether adequate due to diagnosis by antigenuria | 5 (4.1) | 1 (0.9) | 1 (3.3) | – |
| Adequate | 45 (36.6) | 52 (47.3) | 11 (36.7) | – |
| Inadequate | 11 (8.9) | 4 (3.6) | 1 (3.3) | – |
| Female gender—n (%) | 43 (35.0) | 35 (31.8) | 12 (40.0) | 0.69 |
| Ischemic heart disease—n (%) | 16 (13.0) | 9 (8.2) | 4 (13.3) | 0.46 |
| Mechanical ventilation—n (%) | 104 (84.6) | 95 (86.3) | 24 (80.0) | 0.51 |
| Microorganism responsible—n (%) | – | – | – | – |
| Unknown | 63 (51.2) | 52 (47.3) | 17 (56.7) | 0.63 |
| Gram-positive | 28 (22.8) | 30 (27.3) | 7 (23.3) | 0.72 |
| Gram-negative | 32 (26.0) | 27 (24.5) | 8 (26.7) | 0.96 |
| Fungii | 6 (4.9) | 1 (0.9) | 0 | 0.11 |
| Anaerobe | 1 (0.8) | 1 (0.9) | 0 | 0.88 |
| Mortality at 30 days—n (%) | 47 (38.2) | 36 (32.7) | 3 (10.0) | 0.01 |
| Septic shock—n (%) | 104 (84.6) | 95 (87.2) | 24 (80.0) | 0.60 |
| Site of infection—n (%) | – | – | – | 0.84 |
| Respiratory | 64 (52.0) | 67 (60.9) | 14 (46.7) | – |
| Abdominal | 39 (31.7) | 28 (25.5) | 10 (33.3) | – |
| Neurological | 4 (3.3) | 1 (0.9) | 0 | – |
| Urinary | 6 (4.9) | 5 (4.5) | 2 (6.7) | – |
| Skin | 5 (4.1) | 4 (3.6) | 2 (6.7) | – |
| Endocarditis | 5 (4.1) | 4 (3.6) | 2 (6.7) | – |
| Osteomyelitis | 0 | 1 (0.9) | 0 | – |
| Age—median years (p25–75) | 56 (43–68) | 61 (50–70) | 63 (51–78) | 0.32 |
| APACHE-II score—median (p25–75) | 20 (15–25) | 21 (16–25) | 20 (17–22) | 0.62 |
| aPTT (seconds)—median (p25–75) | 35 (29–44) | 33 (28–43) | 28 (27–43) | 0.09 |
| Bilirubin (mg/dl)—median (p25–75) | 0.90 (0.50–2.49) | 0.80 (0.41–1.50) | 0.80 (0.51–1.63) | 0.20 |
| Creatinine (mg/dl)—median (p25–75) | 1.30 (0.70–2.28) | 1.40 (0.90–2.45) | 1.60 (0.90–2.55) | 0.94 |
| Hospitalization before enrollment in the study (days)—median (p25–75) | 0 (0–5) | 0 (0–5) | 0 (0–3) | 0.20 |
| GCS—median (p25–75) | 15 (13–15) | 15 (8–15) | 14 (10–15) | 0.40 |
| Interleukin-6 levels (pg/mL)—median (p25-75) | 429 (74–1034) | 136 (41–852) | 21 (13–116) | <0.001 |
| INR—median (p25–75) | 1.42 (1.20–1.71) | 1.20 (1.06–1.50) | 1.36 (1.11–1.50) | 0.0033 |
| Lactic acid (mmol/L)—median (p25–75) | 2.15 (1.13–4.38) | 2.20 (1.25–4.45) | 2.00 (1.15–3.15) | 0.11 |
| Leukocytes (cells/mm3)—median × 103 (p25–75) | 14.1 (8.9–20.1) | 15.2 (7.9–22.0) | 16.2 (13.4–22.4) | 0.57 |
| Pa02/FI02 ratio—median (p25–75) | 194 (111–265) | 160 (102–221) | 250 (172–298) | 0.001 |
| Platelets (cells/mm3)—median × 103 (p25–75) | 165 (90–243) | 179 (101–270) | 210 (93–267) | 0.17 |
| SOFA score—median (p25−75) | 10 (7–13) | 10 (7–12) | 8 (6–11) | 0.02 |
| Predictors | Odds Ratio | 95% Confidence Interval | p-Value |
|---|---|---|---|
| First Model | – | – | – |
| SOFA (points) | 1.15 | 1.023–1.284 | 0.02 |
| Lactic acid levels (mmol/L) | 1.23 | 1.063–1.430 | 0.006 |
| Age (years) | 1.02 | 0.990–1.042 | 0.23 |
| Genotype of interleukin-6 (CC vs. GG) | 0.21 | 0.053–0.838 | 0.03 |
| Second Model | – | – | – |
| SOFA (points) | 1.22 | 1.068–1.395 | 0.003 |
| Lactic acid levels (mmol/L) | 1.08 | 0.933–1.254 | 0.30 |
| Age (years) | 1.02 | 0.988–1.052 | 0.22 |
| Genotype of interleukin-6 (CC vs. GC) | 0.28 | 0.074–1.037 | 0.057 |
| Third Model | – | – | – |
| SOFA (points) | 1.16 | 1.062–1.263 | 0.001 |
| Lactic acid levels (mmol/L) | 1.14 | 1.029–1.033 | 0.01 |
| Age (years) | 1.01 | 0.993–1.033 | 0.22 |
| Serum interleukin-6 levels (pg/mL) | 1.001 | 1.0001–1.001 | 0.002 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lorente, L.; Martín, M.M.; Pérez-Cejas, A.; Barrios, Y.; Solé-Violán, J.; Ferreres, J.; Labarta, L.; Díaz, C.; Jiménez, A. Association between Interleukin-6 Promoter Polymorphism (-174 G/C), Serum Interleukin-6 Levels and Mortality in Severe Septic Patients. Int. J. Mol. Sci. 2016, 17, 1861. https://doi.org/10.3390/ijms17111861
Lorente L, Martín MM, Pérez-Cejas A, Barrios Y, Solé-Violán J, Ferreres J, Labarta L, Díaz C, Jiménez A. Association between Interleukin-6 Promoter Polymorphism (-174 G/C), Serum Interleukin-6 Levels and Mortality in Severe Septic Patients. International Journal of Molecular Sciences. 2016; 17(11):1861. https://doi.org/10.3390/ijms17111861
Chicago/Turabian StyleLorente, Leonardo, María M. Martín, Antonia Pérez-Cejas, Ysamar Barrios, Jordi Solé-Violán, José Ferreres, Lorenzo Labarta, César Díaz, and Alejandro Jiménez. 2016. "Association between Interleukin-6 Promoter Polymorphism (-174 G/C), Serum Interleukin-6 Levels and Mortality in Severe Septic Patients" International Journal of Molecular Sciences 17, no. 11: 1861. https://doi.org/10.3390/ijms17111861
APA StyleLorente, L., Martín, M. M., Pérez-Cejas, A., Barrios, Y., Solé-Violán, J., Ferreres, J., Labarta, L., Díaz, C., & Jiménez, A. (2016). Association between Interleukin-6 Promoter Polymorphism (-174 G/C), Serum Interleukin-6 Levels and Mortality in Severe Septic Patients. International Journal of Molecular Sciences, 17(11), 1861. https://doi.org/10.3390/ijms17111861






