Quantitative Structure – Antioxidant Activity Relationships of Flavonoid Compounds
Abstract
:Introduction
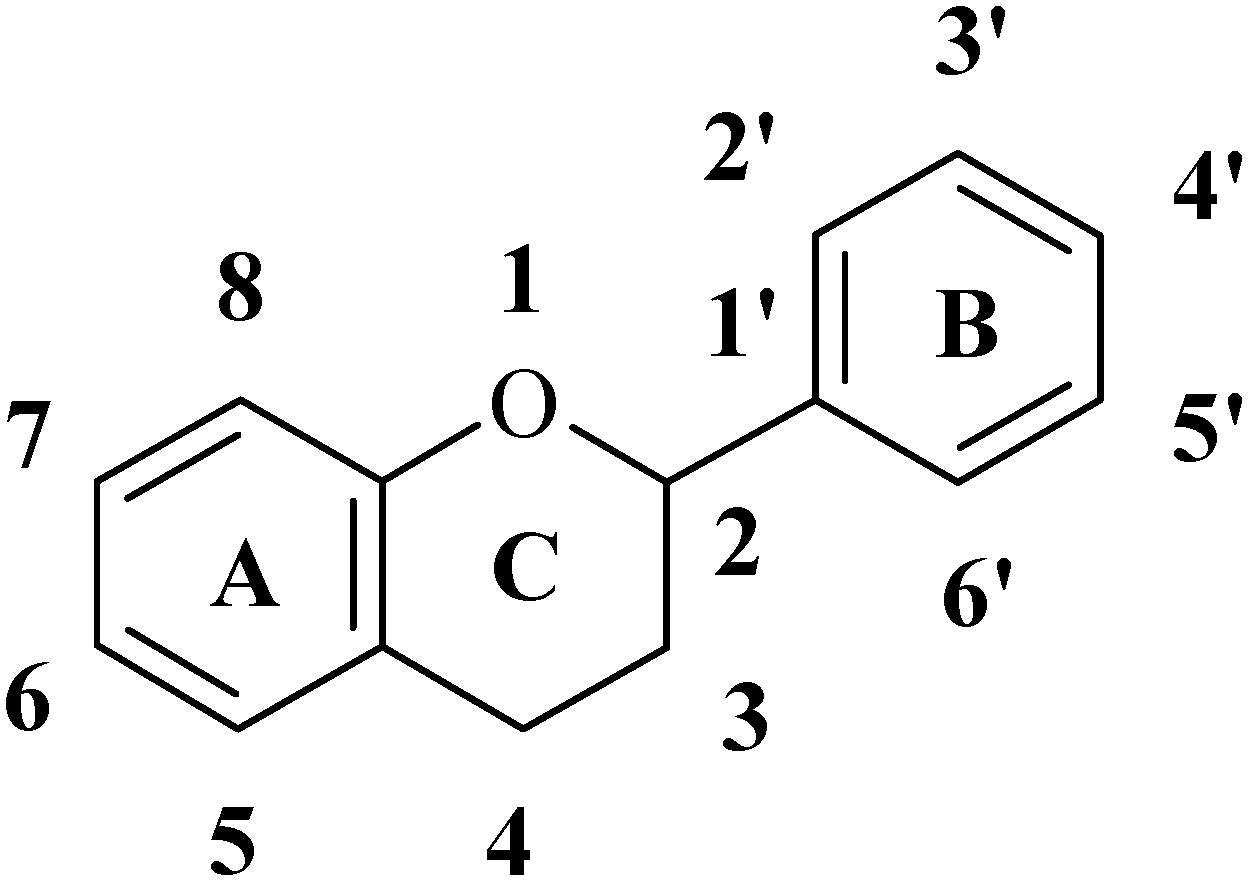
Experimental
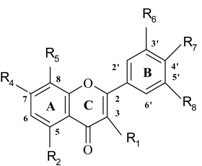
| No | Name | Code | Set** | R1 | R2 | R3 | R4 | R5 | R6 | R7 | R8 | Antioxidant activity (%) |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | kaempferol | kl | 1 | OH | OH | OH | H | H | H | OH | H | 65.3 |
| 2 | galangin | gl | 2 | OH | OH | OH | H | H | H | H | H | 64.9 |
| 3 | quercetin | qu | 1 | OH | OH | OH | H | H | OH | OH | H | 63.6 |
| 4 | robinetin | ro | 1 | OH | H | OH | H | H | OH | OH | OH | 61.7 |
| 5 | fisetin | fi | 1 | OH | H | OH | H | H | OH | OH | H | 61.6 |
| 6 | kaempferide | kd | 1 | OH | OH | OH | H | H | H | OMe | H | 60.0 |
| 7 | 3-hydroxy-flavone | h3 | 1 | OH | H | H | H | H | H | H | H | 59.4 |
| 8 | laricytrin | la | 2 | OH | OH | OH | H | H | OH | OH | OMe | 28.5 |
| 9 | laricytrin 3’- O-glucoside | l3 | 1 | OH | OH | OH | H | H | O-glu* | OH | OMe | 26.2 |
| 10 | myricetin | my | 1 | OH | OH | OH | H | H | OH | OH | OH | 18.4 |
| 11 | 3,5,7,3’,4’,5’-hexamethoxy-flavone | hm | 1 | OMe | OMe | OMe | H | H | OMe | OMe | OMe | 2.6 |
| 12 | 3,5,7,3’,4’-pentamethoxy-flavone | pm | 2 | OMe | OMe | OMe | H | H | O-glu | OMe | OH | 1.1 |
| 13 | larycitrin 3,3’- O-diglucoside | ld | 2 | O-glu | OH | OH | H | H | OH | OH | OMe | 1.1 |
| 14 | quercetin 3- O-glucoside-7-O-rhamnoside | qg | 1 | O-glu | OH | O-rha* | H | H | O-glu | OH | H | -6.2 |
| 15 | laricyrin 3,7,3’- O-triglucoside | lt | 1 | O-glu | OH | O-glu | H | H | OH | OH | OMe | -6.2 |
| 16 | rutin | ru | 1 | O-rut* | OH | OH | H | H | H | OH | H | -10.2 |
| 17 | morin | mo | 1 | OH | OH | OH | H | OH | H | OH | H | 63.5 |
| 18 | flavone | fl | 1 | H | H | H | H | H | H | H | H | -1.5 |
| 19 | 5-hydroxy-flavone | h5 | 2 | H | OH | H | H | H | H | H | H | -4.0 |
| 20 | 7-hydroxy-flavone | h7 | 1 | H | H | OH | H | H | H | H | H | 0.0 |
| 21 | chrysin | cr | 2 | H | OH | OH | H | H | H | H | H | -20.8 |
| 22 | 8-methoxy-flavone | m8 | 2 | H | H | OMe | H | H | H | H | H | -29.2 |
| 23 | apigenin 8- C-glucoside | a8 | 1 | H | OH | OH | glu | H | H | OH | H | -29.6 |
| 24 | luteolin 7- O-glucoside | lu | 2 | H | OH | O-glu | H | H | OH | OH | H | -25.3 |
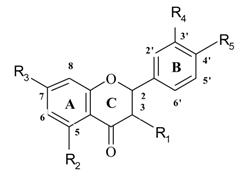
| No | Name | Code | Set** | R1 | R2 | R3 | R4 | R5 | Antioxidant activity |
|---|---|---|---|---|---|---|---|---|---|
| 25 | flavanone | fn | 1 | H | H | H | H | H | -23.0 |
| 26 | naringin | nh | 1 | H | OH | O-neohesp* | H | OH | 47.4 |
| 27 | hesperitin | he | 2 | H | OH | OH | OH | OMe | 4.7 |
| 28 | fustin | fu | 1 | OH | H | OH | OH | OH | -23.4 |
| 29 | taxifolin | ta | 1 | OH | OH | OH | OH | OH | -16.8 |
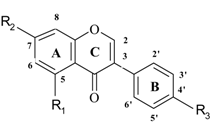
| No | Name | Code | Set** | R1 | R2 | R3 | Antioxidant activity |
|---|---|---|---|---|---|---|---|
| 30 | GB-1 | gb | /1 | OH | OH | H | -30.1 |
| 31 | GB-1a | ga | /2 | H | OH | H | -16.9 |
| No | Name | Code | Set** | R1 | R2 | R3 | Antioxidant activity |
|---|---|---|---|---|---|---|---|
| 32 | daidzein | da | 1 | H | OH | OH | 32.9 |
| 33 | formononetin | fm | 1 | H | OH | OMe | -20.4 |
| 34 | genistein | ge | 1 | OH | OH | OH | -24.6 |
| 35 | biochanin A | bi | 2 | OH | OH | OMe | -20.4 |
| No | Name | Code | Set | Antioxidant activity |
|---|---|---|---|---|
| 36 | coumestrol | cu | 1 | 38.7 |
Results and Discussion
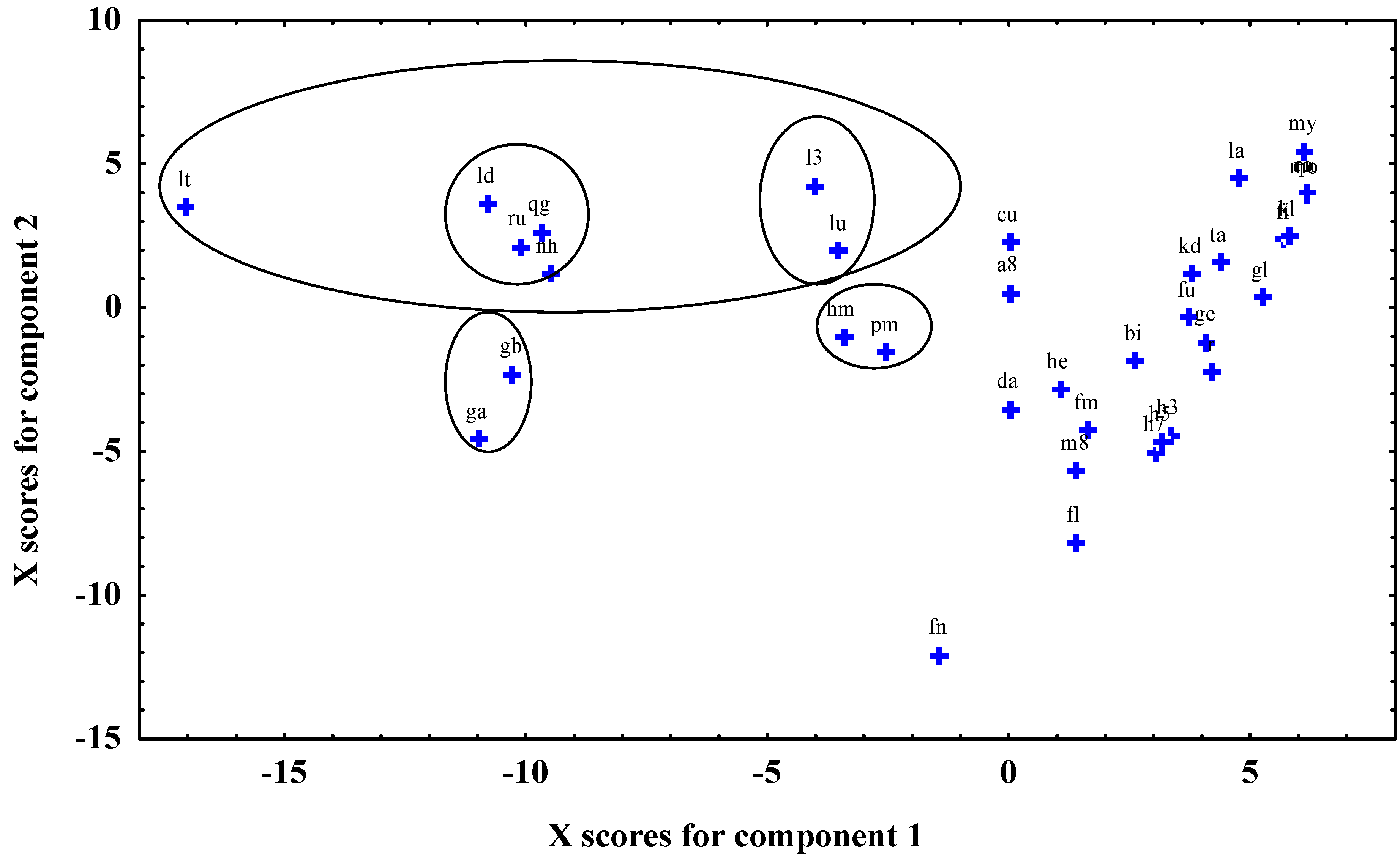
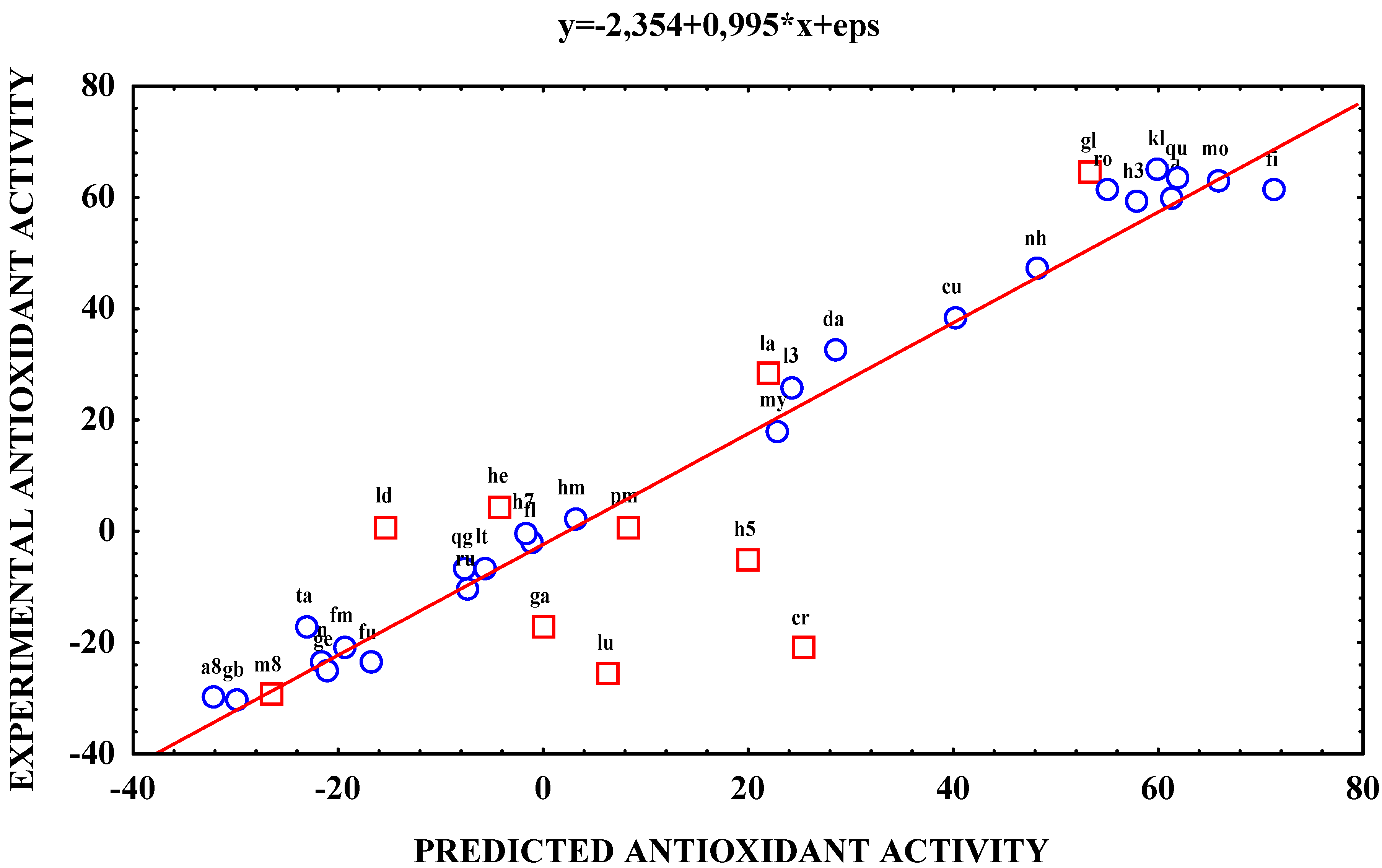
| Antioxidant activity | Antioxidant activity | ||||
|---|---|---|---|---|---|
| Code | Experimental | Predicted | Code | Experimental | Predicted |
| a8 | -29.6 | 53.3 | hm | 2.6 | 22.7 |
| bi | -20.4 | -30.1 | kd | 60.0 | 48.1 |
| cr | -20.8 | -0.2 | kl | 65.3 | 8.1 |
| cu | 38.7 | -21.3 | la | 28.5 | 61.8 |
| da | 32.9 | -4.3 | l3 | 26.2 | -7.8 |
| fi | 61.6 | 57.7 | ld | 1.1 | 54.8 |
| fl | -1.5 | 19.8 | lt | -6.2 | -7.4 |
| fn | -23.0 | -1.9 | lu | -25.3 | -23.2 |
| fm | -20.4 | 2.9 | m8 | -29.2 | -26.7 |
| fu | -23.4 | 61.1 | mo | 63.5 | 65.8 |
| gl | 64.9 | 59.8 | my | 18.4 | 22.7 |
| gb | -30.1 | 22.0 | nh | 47.4 | 48.1 |
| ga | -16.9 | 24.2 | pm | 1.1 | 8.1 |
| ge | -24.6 | -15.5 | qu | 63.6 | 61.8 |
| he | 4.7 | -5.9 | qg | -6.2 | -7.8 |
| h3 | 59.4 | 6.1 | ro | 61.7 | 54.8 |
| h5 | -4.9 | -26.7 | ru | -10.2 | -7.4 |
| h7 | 0.0 | 65.8 | ta | -16.8 | -23.2 |
Conclusions
Acknowledgements
References
- Block, G. A role for antioxidants in reducing cancer risk. Nutr. Rev. 1992, 50, 207–213. [Google Scholar] [PubMed]
- Elangovan, V.; Sekar, N.; Govindasamy, S. Chemoprotective potential of dietary bioflavonoids against 20-methylcholanthrene-induced tumorgenesis. Cancer Lett. 1994, 87, 278–284. [Google Scholar]
- Middleton, E. J.; Kandaswami, C.; Theoharides, T. C. The effects of plant flavonoids on mammalian cells: implications for inflammation, heart disease, and cancer. Pharmacol. Rev. 2000, 52, 673–651. [Google Scholar] [PubMed]
- Selway, J. W. Antiviral activity of flavones and flavans. Prog. Clin. Biol. Res. 1986, 213, 521–536. [Google Scholar] [PubMed]
- Middleton, E., Jr. Effect of plant flavonoids on immune and inflammatory cell function. Adv. Exp. Med. Biol. 1998, 439, 175–182. [Google Scholar] [PubMed]
- Gabor, M. Anti-inflammatory and anti-allergic properties of flavonoids. Prog. Clin. Biol. Res. 1986, 213, 471–480. [Google Scholar] [PubMed]
- Facino, R. M.; Carini, M.; Aldini, G.; Berti, F.; Rossoni, G.; Bombardelli, E.; Morazzoni, P. Diet enriched with procyanidins enhances antioxidant activity and reduces myocardial post-ischemic damage in rats. Life Sci 1999, 64, 943–949. [Google Scholar] [CrossRef]
- Herteg, M. G. L.; Feskens, E. J. M.; Hollman, P. C. H.; Katan, M. B.; Kromhout, D. Dietary flavonoids and risk of coronary heart disease. Lancet 1993, 342, 1007–1011. [Google Scholar] [PubMed]
- Mazur, A.; Bayle, D.; Lab, C.; Rock, E.; Rayssiguier, Y. Inhibitory effect of procyanidin-rich extracts on LDL-oxidation in vitro. Atherosclerosis 1999, 149, 421–422. [Google Scholar]
- Alves, C. N.; Pinheiro, J. C.; Camargo, A. J.; Ferreira, M. M. C.; Romero, R. A. F.; da Silva, A. B. F. A multiple linear regression and partial least squares study of flavonoid compounds with anti-HIV activity. J. Mol. Struct. 2001, 541, 81–88. [Google Scholar]
- Alves, C. N.; Pinheiro, J. C.; Camargo, A. J.; de Souza, A. J.; Carvalho, R. B.; da Silva, A. B. F. A multiple linear regression and partial least squares study of flavonoid compounds with anti-HIV activity. J. Mol. Struct. 1999, 491, 123–131. [Google Scholar] [CrossRef]
- Moriani, M. Z.; Galati, G.; O’Brien, P. J. Comparative quantitative structure toxicitz relationships for flavonoids evaluated in isolated rat hepatocytes and HeLa tumor cells. Chem. Bio. Interact. 2002, 139, 251–264. [Google Scholar] [CrossRef]
- Sergediene, E.; Jönsson, K.; Szymusiak, H.; Tyrakowska, B.; Rietjens, I. M. C. M.; Cenas, N. Prooxidant toxicity of polyphenolic antioxidants to HL-60 cells: description of quantitative structure-activity relationships. FEBS Letters 1999, 462, 392–396. [Google Scholar] [PubMed]
- Moon, T.; Chi, M. H.; Kim, D. H.; Yoon, C. N.; Choi, Y. K. Quantitative structure-activity relationships (QSAR) study of flavonoid derivatives for inhibition of cytochrome P450 1A2. Quant. Struct. Act. Relat. 2000, 19, 257–263. [Google Scholar]
- Nikolovska-Coleska, Z.; Suturkova, L.; Dorevski, K.; Krbavcic, A.; Solmajer, T. Quantitative structure-activity relationship of flavonoid inhibitors of p56(lck) protein tyrosine kinase: A classical/quantum chemical approach. Quant. Struct. Act. Relat. 1998, 17, 7–13. [Google Scholar]
- Oblak, M.; Randic, M.; Solmajer, T. Quantitative structure-activity relationship of flavonoid analogues. 3. Inhibition of p56(lck) protein tyrosine kinase. J. Chem. Inf. Comput. Sci. 2000, 40, 994–1001. [Google Scholar] [PubMed]
- Stefanic-Petek, A.; Krbavcic, A.; Solmajer, T. QSAR of flavonoids: 4. Differential inhibition of aldose reductase and p56(lck) protein tyrosine kinase. Croat. Chem. Act. 2000, 5, 517–529. [Google Scholar]
- Rice-Evans, C. A.; Miller, N. J.; Paganga, G. Structure-antioxidant activity relationships of flavonoids and phenolic acids. Free. Rad. Biol. Med 1996, 20, 933–956. [Google Scholar] [CrossRef] [PubMed]
- Heim, K. E.; Tagliaferro, A. R.; Bobilya, D. J. Flavonoid antioxidants: chemistry, metabolism and sructure-activity relationships. J. Nutr. Biochem. 2002, 13, 572–584. [Google Scholar] [PubMed]
- Harborne, J. B.; Williams, C. A. Advances in flavonoid research since 1992. Phytochem. 2000, 55, 481–504. [Google Scholar]
- Lien, E. J.; Ren, S. J.; Bui, H. Y. H.; Wang, R. B. Quantitative structure-activity relationship analysis of phenolic antioxidants. Free. Rad. Biol. Med. 1999, 26, 285–297. [Google Scholar]
- Amić, D.; Davidovic-Amić, D.; Bešlo, D.; Trinajstić, N. Structure-radical scavenging activity relationships of flavonoids. Croat. Chem. Act. 2003, 76, 55–61. [Google Scholar]
- Burda, S.; Oleszek, W. Antioxidant and antiradical activities of flavonoids. J. Agric. Food Chem. 2001, 49, 2774–2779. [Google Scholar] [CrossRef] [PubMed]
- HyperChem 7.0. HyperCube Inc.: Toronto, Canada, 2002.
- Todeschini, R.; Consonni, V.; Pavan, M. Dragon Software version 2.1; 2002.
- Statistica 5.5 software package. StatSoft Inc.: Tulsa, OK, USA.
© 2004 by MDPI (http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.
Share and Cite
Farkas, O.; Jakus, J.; Héberger, K. Quantitative Structure – Antioxidant Activity Relationships of Flavonoid Compounds. Molecules 2004, 9, 1079-1088. https://doi.org/10.3390/91201079
Farkas O, Jakus J, Héberger K. Quantitative Structure – Antioxidant Activity Relationships of Flavonoid Compounds. Molecules. 2004; 9(12):1079-1088. https://doi.org/10.3390/91201079
Chicago/Turabian StyleFarkas, Orsolya, Judit Jakus, and Károly Héberger. 2004. "Quantitative Structure – Antioxidant Activity Relationships of Flavonoid Compounds" Molecules 9, no. 12: 1079-1088. https://doi.org/10.3390/91201079
APA StyleFarkas, O., Jakus, J., & Héberger, K. (2004). Quantitative Structure – Antioxidant Activity Relationships of Flavonoid Compounds. Molecules, 9(12), 1079-1088. https://doi.org/10.3390/91201079