Chemistry of Substituted Quinolinones. Part VI. Synthesis and Nucleophilic Reactions of 4-Chloro-8-methylquinolin-2(1H)-one and its Thione Analogue
Abstract
:Introduction
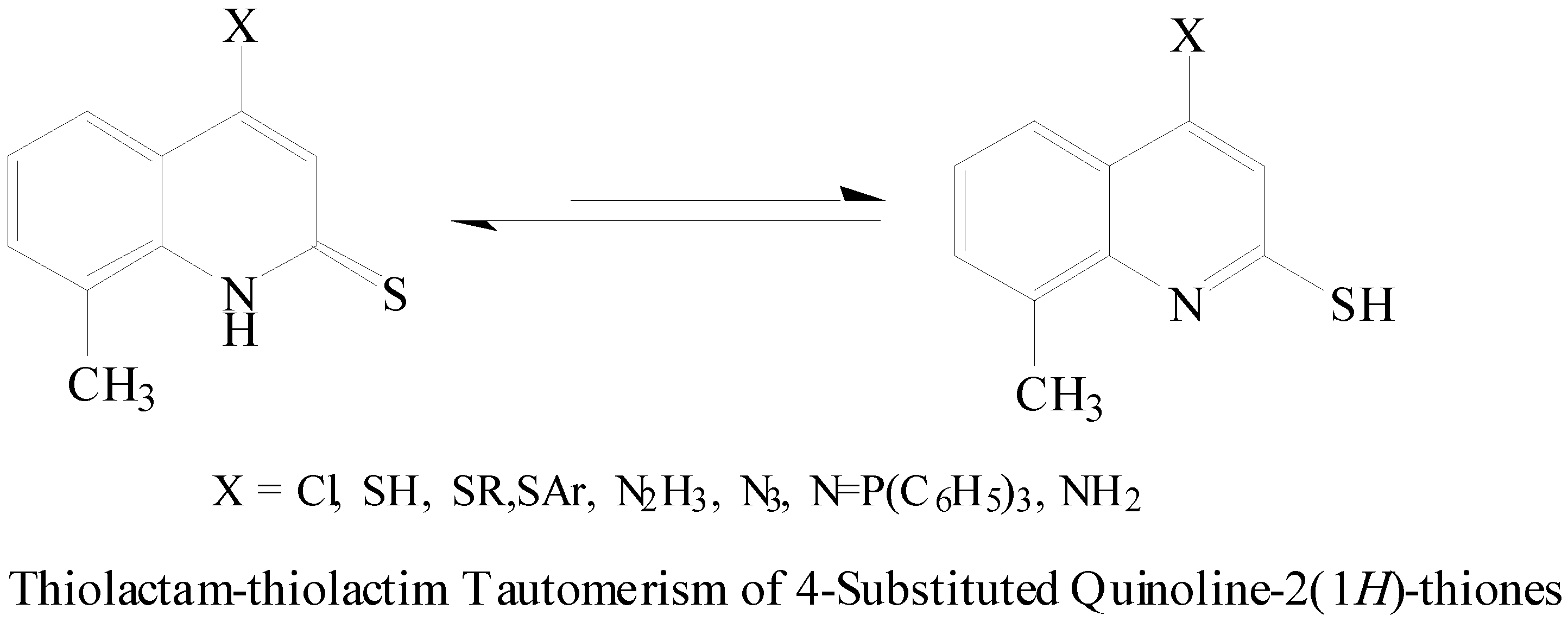
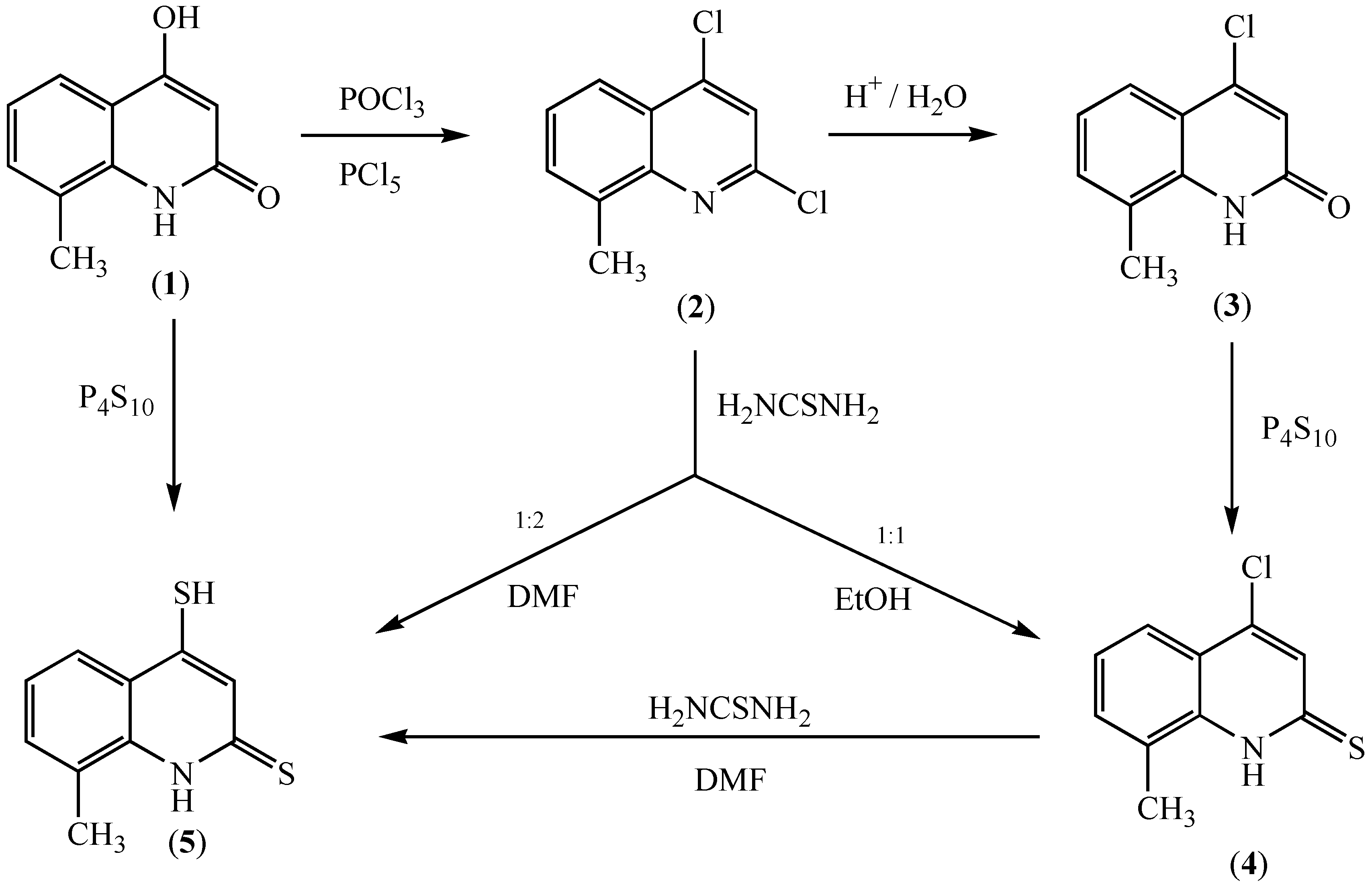
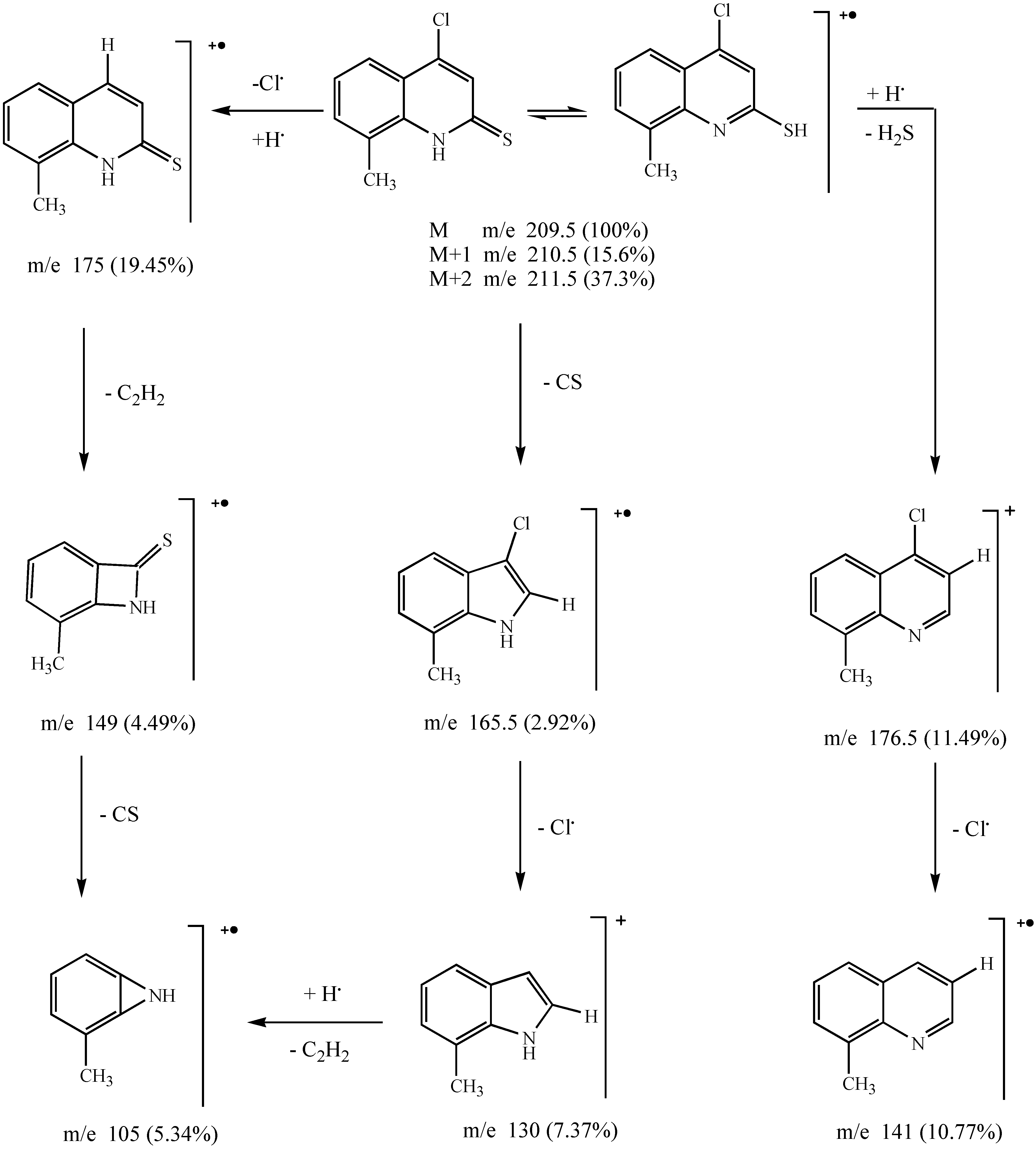
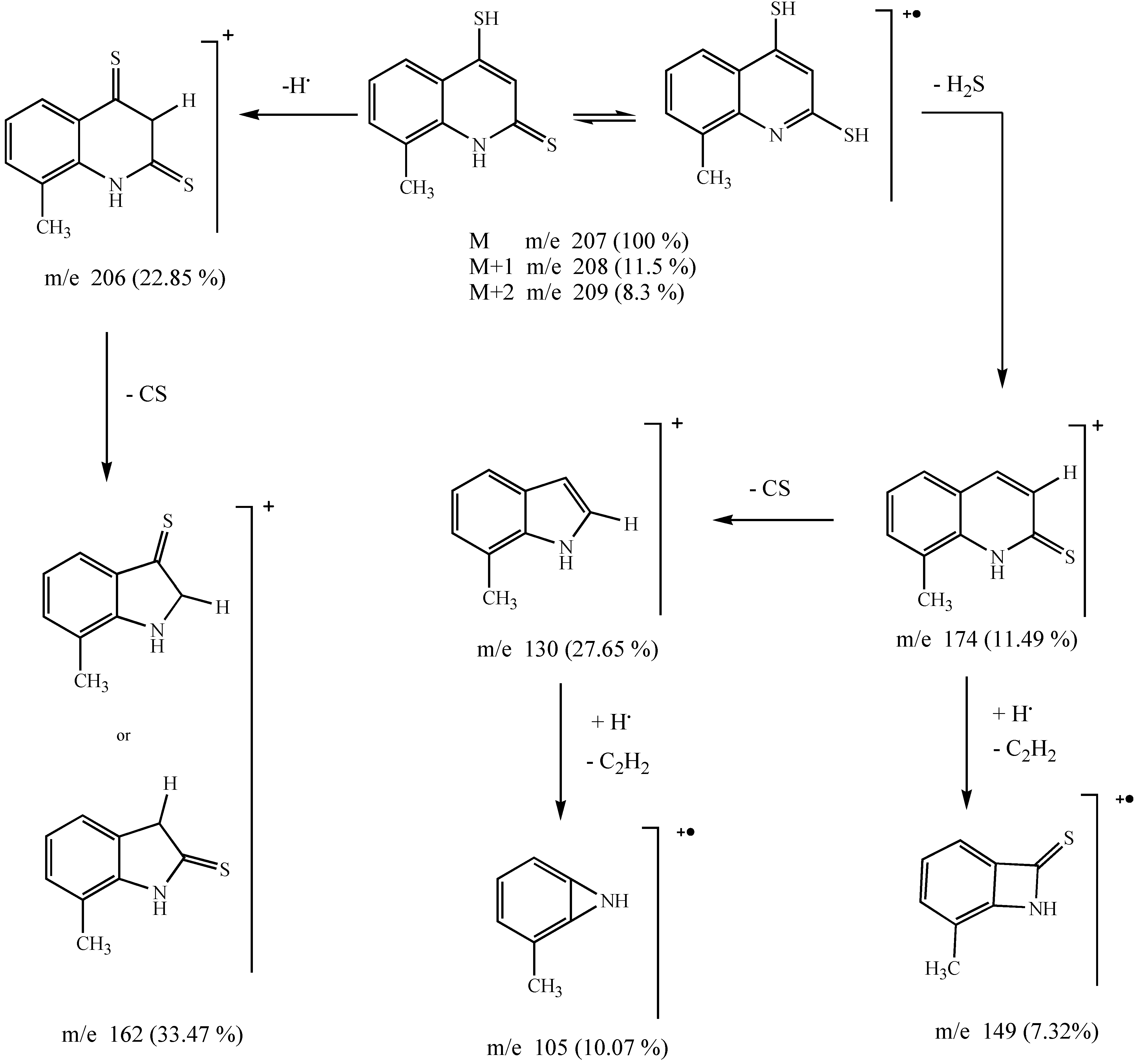
Results and Discussion

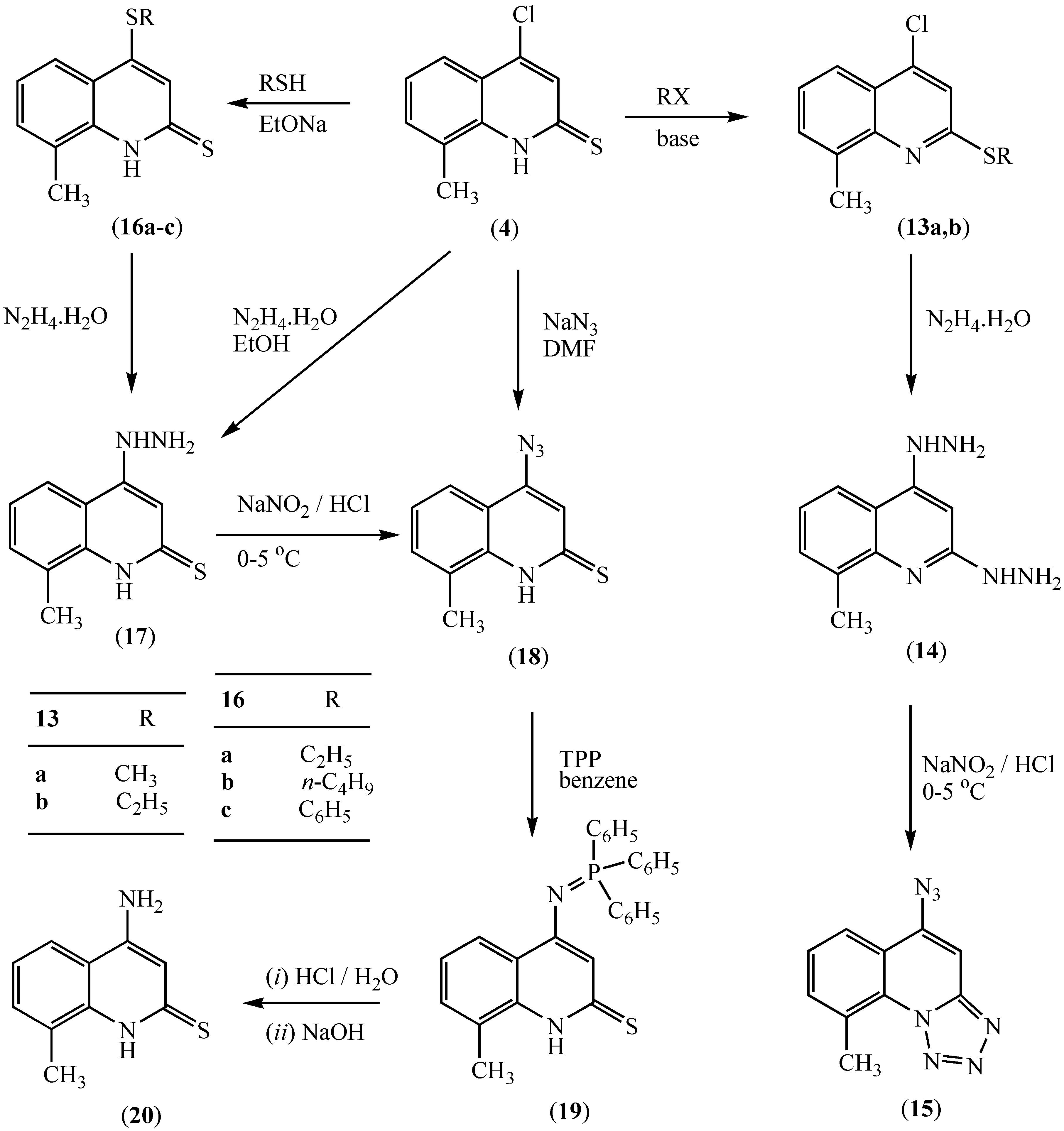

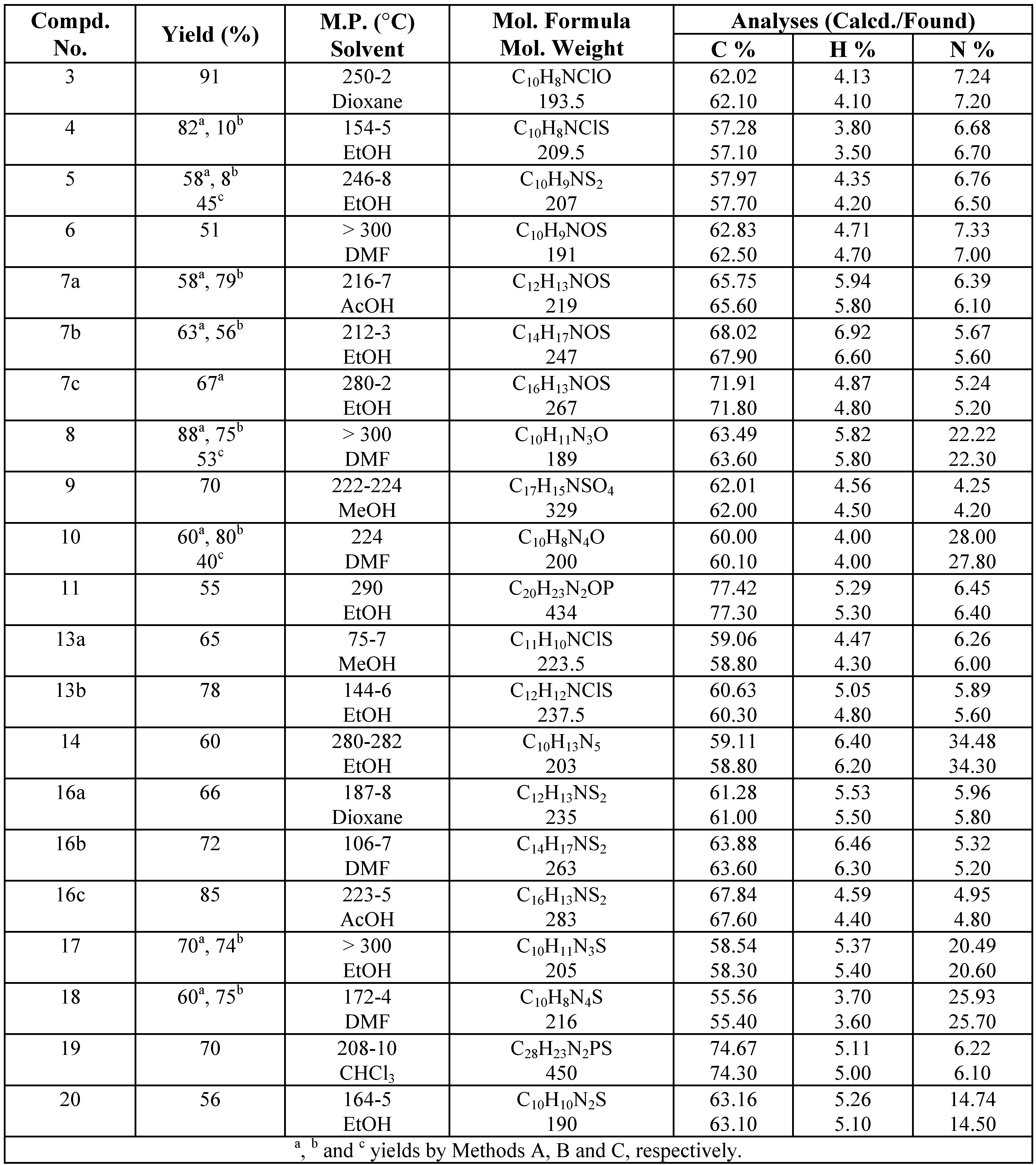
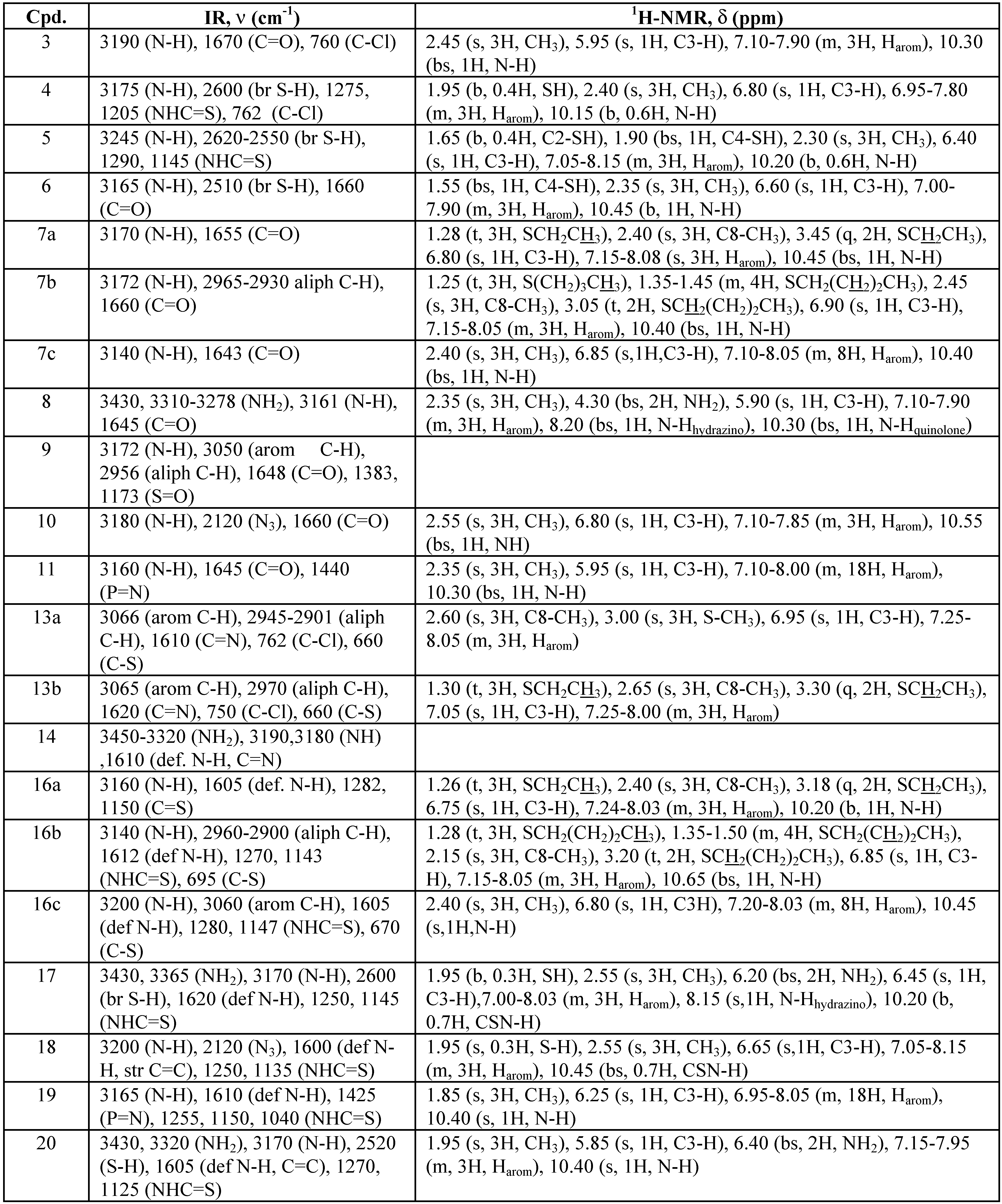
Experimental
General
4-Chloro-8-methylquinolin-2(1H)-one (3)
4-Chloro-8-methylquinoline-2(1H)-thione (4)
Method A
Method B
8-Methyl-4-sulfanylquinoline-2(1H)-thione (5)
Method A
Method B
Method C
8-Methyl-4-sulfanylquinolin-2(1H)-one (6)
4-Alkyl (or aryl)thio-8-methylquinolin-2(1H)-ones 7a-7c
Method A
Method B
4-Hydrazino-8-methylquinolin-2(1H)-one (8)
Method A
Method B
Method C
8-Methyl-4-tosyloxyquinolin-2(1H)-one (9)
4-Azido-8-methylquinolin-2(1H)-one (10)
Method A
Method B
Method C
8-Methyl-4-(triphenylphosphoranylideneamino)quinolin-2(1H)-one (11)
4-Amino-8-methylquinolin-2(1H)-one (12)
4-Chloro-2-methylthio-8-methylquinoline (13a)
Chloro-2-ethylthio-8-methylquinoline (13b)
2,4-Dihydrazino-8-methylquinoline (14)
Azido-9-methyltetrazolo[1,5-a] quinoline (15)
4-Alkyl(or phenyl)thio-8-methylquinoline-2(1H)-thiones 16a-c
4-Hydrazino-8-methylquinoline-2(1H)-thione (17)
Method A
Method B
4-Azido-8-methylquinoline-2(1H)-thione (18)
Method A
Method B
8-Methyl-4-(triphenylphosphoranylideneamino)quinoline-2(1H)-thione (19)
4-Amino-8-methylquinolin-2(1H)-thione (20)
References and Notes
- Ismail, M. M.; Abass, M.; Hassan, M. M. Phosph. Sulfur Silicon. http://www.reprints.net/ecsoc-4/a0088/a0088.htm.
- Mohamed, E. A.; Ismail, M. M.; Gabr, Y.; Abass, M. J. Serb. Chem. Soc. 1993, 58, 737.
- Ismail, M. M.; Abass, M. Acta Chim. Slov. 2000, 47, 327.
- Sarac-Arneri, R.; Mintas, M.; Pustec, N.; Mannschreck, A. Monatsh. Chem. 1994, 125, 457.
- Urquhart, G. G.; Gates, T. W.; Connor, R. Org. Synth. 1955, 3, 363.
- Bourguignon, J.; Lemarchand, M.; Queguiner, G. J. Heterocycl. Chem. 1980, 17, 257.
- Abass, M.; Ismail, M. M. Chem. Papers 2000, 51, 186.
- Mirek, J.; Sygula, A. Z. Naturforsch 1982, 37a, 1276.
- Staudinger, H.; Meyer, J. Helv. Chim. Acta 1919, 2, 635.
- Rylander, P. N. Hydrogenation Methods; 1985. [Google Scholar]
- Samples Availability: Available from the authors.
© 2000 by MDPI (http://www.mdpi.org) All rights reserved.
Share and Cite
Ismail, M.M.; Abass, M.; Hassan, M.M. Chemistry of Substituted Quinolinones. Part VI. Synthesis and Nucleophilic Reactions of 4-Chloro-8-methylquinolin-2(1H)-one and its Thione Analogue. Molecules 2000, 5, 1224-1239. https://doi.org/10.3390/51201224
Ismail MM, Abass M, Hassan MM. Chemistry of Substituted Quinolinones. Part VI. Synthesis and Nucleophilic Reactions of 4-Chloro-8-methylquinolin-2(1H)-one and its Thione Analogue. Molecules. 2000; 5(12):1224-1239. https://doi.org/10.3390/51201224
Chicago/Turabian StyleIsmail, Mostafa M., Mohamed Abass, and Mohamed M. Hassan. 2000. "Chemistry of Substituted Quinolinones. Part VI. Synthesis and Nucleophilic Reactions of 4-Chloro-8-methylquinolin-2(1H)-one and its Thione Analogue" Molecules 5, no. 12: 1224-1239. https://doi.org/10.3390/51201224
APA StyleIsmail, M. M., Abass, M., & Hassan, M. M. (2000). Chemistry of Substituted Quinolinones. Part VI. Synthesis and Nucleophilic Reactions of 4-Chloro-8-methylquinolin-2(1H)-one and its Thione Analogue. Molecules, 5(12), 1224-1239. https://doi.org/10.3390/51201224




