2.1. Analysis of the Reduction Reaction Energetics and Thermal Effects
In all three experimental variants, high-temperature reduction was successfully initiated, leading to the synthesis of a Cr–Mn-containing metallic phase. The process was characterized by distinct gravitational separation between the metallic and oxide phases, confirming that the system achieved a stable metal–slag equilibrium under the applied conditions.
To evaluate the energetics of the reduction process and identify characteristic exothermic responses, the temperature–current profiles (
Figure 3,
Figure 4 and
Figure 5) were subjected to detailed thermal analysis. Unlike standard heating curves, these dependencies reflect the non-stationary nature of the chemical interactions occurring within the melt as a function of the inductor current (A). The analysis of these profiles allows for the identification of specific “ignition” points where internal chemical energy begins to dominate the external induction heating, providing a mechanistic understanding of the thermal evolution of the system.
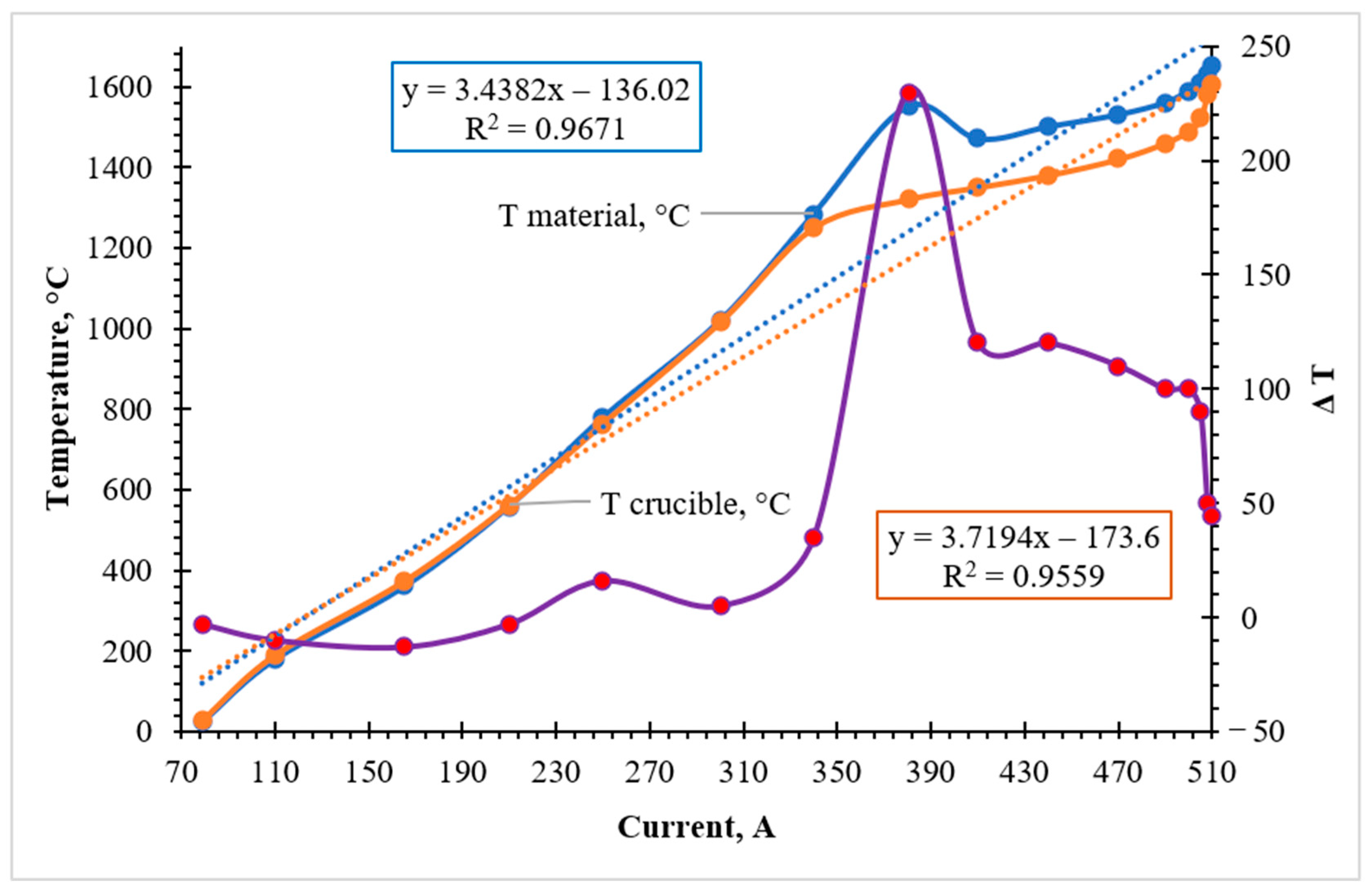
The temperature–current profiles obtained for the first experimental variant (Cr-ore + AlSiMn reductant + lime) provide critical insight into the energetic evolution of the system (
Figure 3). At the initial heating stage (up to ~1250 °C), the thermal state was governed strictly by the external power input, with the temperatures of the melt (T
material) and the crucible (T
crucible) increasing linearly as the inductor current was raised from 80 to 300 A. This coordinated heating indicates the absence of significant internal chemical heat generation during the pre-reduction phase.
A pronounced shift in the thermal regime was observed in the current range of 340–380 A. Within a 12–14 min interval, while the inductor current was held in a quasi-stationary regime, a rapid temperature surge of the melt from 1285 to 1550 °C was recorded. This sharp increase of 250–270 °C cannot be attributed to the induction heating alone, providing direct experimental evidence of an internal exothermic heat source.
From a mechanistic perspective, this thermal pulse is driven by the high negative enthalpy of the metallothermic reduction of chromium oxides by the active components of the aluminosilicate reductant (Al and Si). Since manganese is introduced into the system in its metallic form, the exothermic effect is primarily localized within the chromium reduction reactions:
The sharp increase in the temperature difference (ΔT) between the melt and the crucible at ~380 A confirms that the heat release occurred directly within the reaction zone. Following the peak, the temperature stabilized at ~1470 °C as the chemical energy was dissipated and absorbed by the surrounding slag phase. At the final stage, the heating was again controlled by the induction power as the current reached 510 A, with the final melt temperature reaching approximately 1650 °C.
Thus, the exothermic effect in Variant 1 manifests as a short-lived transient thermal pulse. This localized energy release is functionally significant: it provides the necessary heat to facilitate the rapid depolymerization of the oxide network, thereby lowering the viscosity of the multicomponent melt and ensuring efficient gravitational separation of the synthesized Cr–Mn metallic phase from the slag.
The temperature–current profiles recorded for the second experimental variant (chromium ore + iron–manganese ore + FeSiAl + lime) provide a comprehensive view of the thermal behavior of the system during complex reduction (
Figure 4). During the initial heating phase (up to 900 °C), the system followed a stable induction heating regime where the melt (T
material) and crucible (T
crucible) temperatures increased linearly with the inductor current.
A transition in the kinetic regime was observed starting from ~950 °C. Initially, a slight plateau in the heating rate occurred, which is attributed to the endothermic energy requirements for initial slag formation and the rearrangement of the oxide lattice. However, as the current reached the 300–350 A range, a powerful exothermic pulse was triggered. The melt temperature exhibited a sharp jump from ~1240 °C to ~1440 °C (a ΔT of 200 °C) while the external power input remained quasi-stationary. This provides definitive experimental evidence of internal chemical heat generation.
Unlike Variant 1, where the reduction processes were primarily limited to chromium and iron oxides, the introduction of iron–manganese ore in this variant significantly alters the energetic profile. The presence of manganese oxides expands the range of exothermic interactions. From a mechanistic standpoint, this thermal surge is the cumulative result of the following reactions:
Aluminothermic stage (Primary heat pulse):
Silicothermic stage (Thermal sustainment and slag formation):
The participation of silicon (Si) in the reduction of manganese and chromium oxides ensures sustained heat release following the initial aluminum-driven pulse. The reaction products, specifically SiO2, are instrumental in forming a fluid silicate-based slag. This combined energy release promotes the rapid depolymerization of the oxide melt, significantly reducing its viscosity. This reduction in viscosity is the key factor that allows the freshly reduced metallic droplets to coalesce and settle through the slag, ensuring high recovery rates of the Cr–Mn–Fe alloy.
After the reaction front passed, the temperature stabilized at ~1290 °C before the induction heating brought the final melt to ~1640 °C at 508 A. Thus, the thermal behavior in Variant 2 is characterized by a transient, high-intensity thermal pulse that optimizes the conditions for phase separation and alloy formation.
The temperature–current profiles for the third experimental variant (ferromanganese ore + FeSiCr dust + lime) reveal a distinct thermal regime under a constant frequency of 50 Hz (
Figure 5). Up to a current of approximately 290 A, the heating followed a stable linear trend, governed predominantly by the external induction energy input.
In the 290–350 A range, a moderate transient thermal pulse was recorded, causing the melt temperature to rise from ~1080 °C to a peak of ~1230 °C. Compared to the previous variants, this exothermic response is significantly less intense and exhibits a shorter duration.
Mechanistic Analysis: The lower intensity of the exothermic effect in Variant 3 is fundamentally linked to the physical and chemical state of the charge components. Unlike Variants 1 and 2, where chromium was introduced in the oxide form (requiring highly exothermic reduction), Variant 3 utilizes FeSiCr dust. In this technogenic material, chromium is already present in its metallic state. Consequently, it does not participate in exothermic redox reactions; instead, the metallic chromium acts as a thermal “sink,” consuming a portion of the generated heat for its own melting and superheating.
Therefore, the observed thermal pulse is attributed exclusively to the silicothermic reduction of manganese oxides from the Kerege-Tas ore by the silicon present in the reductant:
Despite the moderate nature of this heat release, the thermal energy is sufficient to initiate the depolymerization of the multicomponent oxide melt, thereby reducing its viscosity. This provides the necessary kinetic conditions for the coalescence of Cr–Mn metallic droplets and their subsequent gravitational separation from the slag. Following the reaction stage, the system stabilized, reaching a final equilibrium temperature of 1600 °C at 510 A.
In summary, the thermal and kinetic analysis of the three experimental variants was focused primarily on the reduction of chromium (Cr) and manganese (Mn) oxides. While secondary reactions occur within the multicomponent system, these primary transitions are the fundamental drivers of the system’s energetics and the formation of the metallic phase. The experimental data confirms that the localized thermal pulses, regardless of their intensity, are sufficient to trigger the depolymerization of the slag and ensure high recovery rates of the target elements.
To verify the efficiency of the reduction process, the final metallic and slag phases were subjected to rigorous chemical analysis. The elemental compositions were determined using the wet chemical method in accordance with GOST standards (for low-carbon FeCr and FeMn alloys). This analytical approach ensured high precision in measuring the distribution of Cr and Mn between the alloy and the oxide melt. The results of the chemical analysis for all three variants are presented in
Table 1.
Analysis of the chemical composition of the metallic phase shows that the obtained Cr–Mn-containing samples represent multicomponent alloys of the Fe–Cr–Mn system with a high total content of the main transition elements. For all investigated variants, a stable redistribution of chromium and manganese into the metallic phase is observed, which is reflected in the close and reproducible composition of the obtained products.
The obtained data indicate the formation of a metallic matrix enriched with transition metals, while local compositional variations point to non-equilibrium crystallization conditions caused by the intense progression of reduction reactions and subsequent cooling of the melt. The contents of Cr and Mn compounds in the slag phase remain limited, as confirmed by the low residual concentrations of the corresponding oxides in the smelting products.
2.2. XRD and SEM Analysis
To determine the phase composition and microstructural features of the obtained Cr–Mn-containing metallic phase, a combination of X-ray diffraction and qualitative scanning electron microscopy methods was employed. The combined use of XRD and SEM makes it possible to identify crystalline phases, assess their morphological distribution characteristics, and reveal features of non-equilibrium crystallization caused by the intensive progression of reduction reactions and specific thermal conditions. The obtained data (
Figure 6,
Figure 7,
Figure 8 and
Figure 9) provide the basis for interpreting the chemical composition of the metallic phase and for further analysis of the relationships between phase state, microstructure, and the conditions of high-temperature experiments.
2.2.1. XRD Analysis of the Metallic Phase
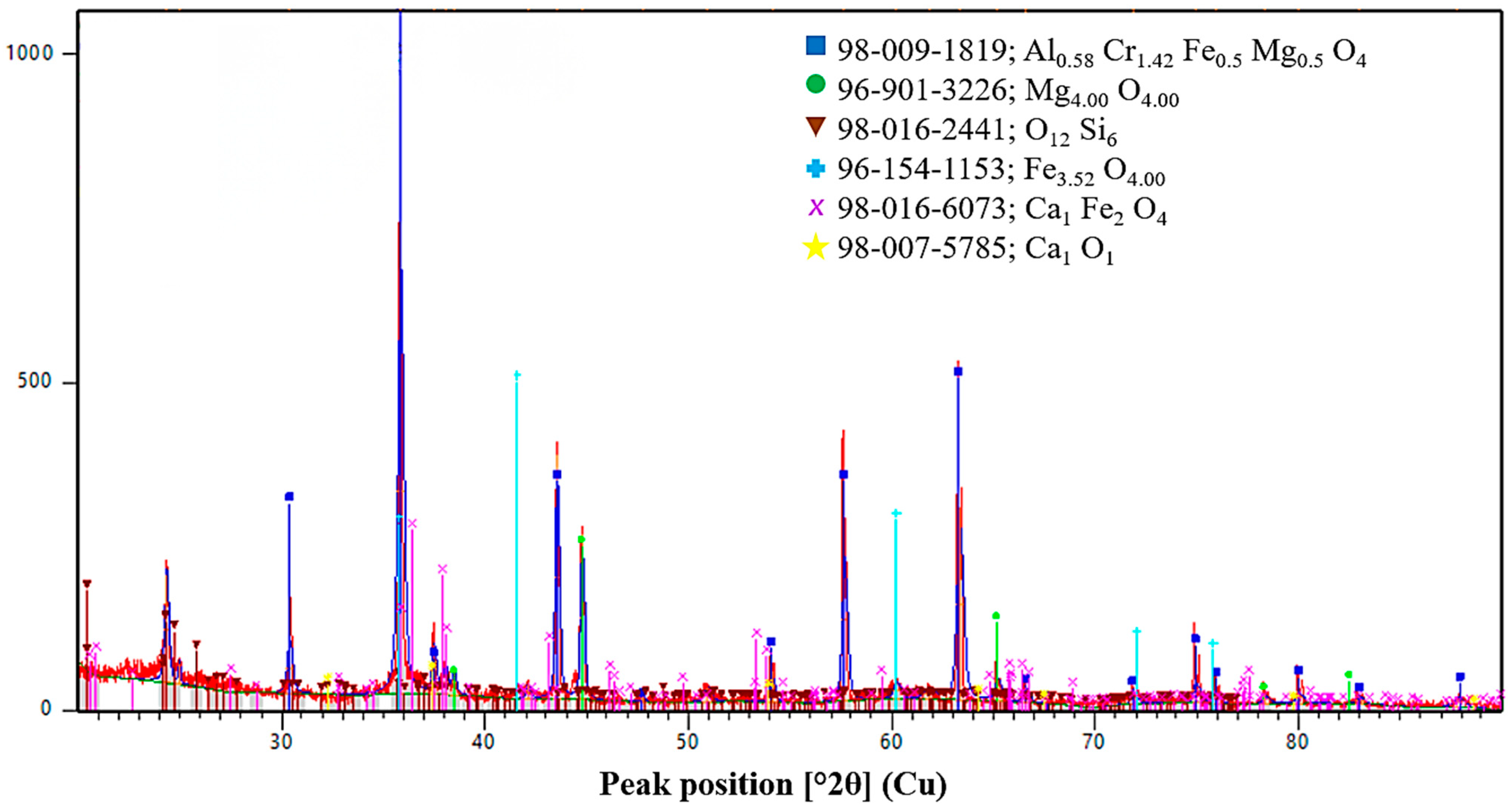
V1. The XRD pattern of the metallic product obtained in Variant V1 indicates a clearly multiphase structure. The dominant diffraction peaks correspond to α-Fe with a body-centered cubic (BCC) lattice, as evidenced by intense reflections in the 2θ range of approximately 42–45°, as well as higher-angle reflections near 65° and 78–80°. These peaks are partially broadened and split due to the formation of Fe–Mn solid solutions, indicating the incorporation of manganese into the iron lattice.
In addition to the Fe-based phases, distinct reflections of metallic chromium are observed, confirming the effective reduction of chromium oxides. The presence of ε-FeSi, Cr3Si, and Fe–Mn–Si silicide phases is also evident from characteristic peaks in the 38–41° and 45–50° ranges. The formation of these silicides demonstrates the active participation of silicon in the reduction process and reflects non-equilibrium crystallization under conditions of rapid heat release. Overall, the phase assemblage of Variant V1 corresponds to a Cr–Mn–Fe alloy with a significant fraction of secondary silicide phases.
V2. The metallic phase obtained in Variant V2 is characterized by a different phase balance. The XRD pattern is dominated by α-Fe-based solid solutions alloyed with chromium, as indicated by intense BCC reflections and their systematic shifts relative to pure iron. The strongest peak at approximately 44.7° (2θ) confirms the prevalence of the Fe–Cr metallic matrix.
Compared to Variant V1, Variant V2 exhibits a more pronounced development of iron–chromium and iron–chromium–silicon silicide phases, including Cr0.3Fe2.7Si, ε-FeSi, and Fe5Si3 (xifengite). These phases are identified by multiple reflections in the 38–42°, 50–55°, and 70–80° regions. The occurrence of Fe–Mn–Si silicides and trace manganese-containing phases indicates partial participation of manganese in silicide formation rather than extensive incorporation into the Fe lattice. This phase composition suggests a stronger influence of silicon-driven reactions and a higher degree of chemical heterogeneity in the metallic product.
V3. In contrast to Variants V1 and V2, the XRD pattern of Variant V3 reveals a comparatively simpler phase composition. The metallic product is predominantly composed of α-Fe-based solid solutions with minor alloying by chromium and manganese, as reflected by sharp and well-defined BCC diffraction peaks. The reduced number and intensity of secondary reflections indicate a lower fraction of silicide phases.
Only weak reflections corresponding to FeSi and Fe3Si are detected, suggesting limited interaction between silicon and the metallic melt under the thermal conditions of Variant V3. The absence of pronounced Cr3Si or complex Fe–Mn–Si silicides implies a more equilibrium-like crystallization behavior and a lower degree of chemical segregation during solidification.
Comparative discussion. A comparison of the three variants demonstrates that the phase composition of the metallic products is strongly governed by the reduction conditions and the availability of active reductants. Variant V1 exhibits the most complex multiphase structure, combining Fe–Mn solid solutions, metallic chromium, and several silicide phases formed under highly non-equilibrium conditions. Variant V2 is characterized by a Fe–Cr-dominated metallic matrix with an increased contribution of iron–chromium–silicon silicides, reflecting the enhanced role of silicon in the reduction process. Variant V3 shows the simplest phase assemblage, dominated by Fe-based solid solutions with minimal silicide formation.
2.2.2. SEM Analysis of the Metallic Phase
To complement the XRD findings, SEM combined with energy-dispersive X-ray spectroscopy (EDS) was performed on the metallic product of V1 (
Figure 7). The microstructure analysis reveals a highly heterogeneous, multiphase morphology, which directly correlates with the complex diffraction pattern observed in the XRD analysis.
Microstructural features the SEM micrographs show a well-developed crystalline structure characterized by dendritic formations and intermetallic inclusions. This confirms the non-equilibrium crystallization process mentioned earlier, where rapid thermal shifts lead to the segregation of various phases. The elemental mapping displays a non-uniform distribution of Fe, Cr, Mn, and Si, indicating the formation of distinct chemical zones within the alloy.
Elemental composition (EDS Results) the quantitative EDS analysis (
Table 2) of selected local areas (Spectra 1–3) provides specific insights into the phase distribution:
Spectrum 1 (Cr-rich matrix): This region is dominated by chromium (69.63 wt.%) with significant iron (19.21 wt.%) and manganese (6.88 wt.%). This composition supports the XRD detection of metallic Cr reflections and the formation of α-(Fe,Cr) solid solutions.
Spectrum 2 (High Mn-Si zone): A sharp increase in manganese (21.32 wt.%) and silicon (6.73 wt.%) is observed here. This local chemistry corresponds to the Fe–Mn–Si and Cr3Si silicide phases identified in the 38–41° and 45–50° (2theta) ranges of the XRD pattern.
Spectrum 3 (Fe-Cr-Mn balance): This area shows a more balanced distribution of iron (29.11 wt.%) and chromium (44.39 wt.%) with high manganese (22.51 wt.%), confirming the presence of the complex Fe–Mn and BCC α-Fe based solid solutions.
The microstructural evolution and elemental distribution of V2 (
Figure 8) exhibit distinct differences from V1, directly supporting the phase shifts observed in the XRD patterns. The transition from a multi-component Cr-rich system to a more dominated iron–chromium matrix is evident in both the morphology and the localized chemical analysis.
Microstructural Observations SEM imaging of Variant V2 (
Figure 8) reveals a heterogeneous surface with clearer grain boundaries and specific clusters of silicide inclusions. The elemental maps for Fe, Cr, Mn, and Si show that while iron and chromium form the primary matrix, silicon and manganese are concentrated in specific secondary phases. This microstructural heterogeneity explains the multiple reflections of complex silicides identified in the XRD analysis.
Quantitative EDS Interpretation The point EDS analysis for Variant V2 (
Table 3) clarifies the chemical nature of the phases:
Spectrum 1 (Fe-Cr-Mn matrix): This area shows a high concentration of iron (48.36 wt.%) and chromium (23.38 wt.%), which aligns with the XRD results showing the prevalence of α-(Fe,Cr) BCC and Fe0.8Cr0.2 solid solutions.
Spectrum 2 (Mn-rich phase): A significant concentration of manganese (38.22 wt.%) and silicon (6.11 wt.%) is detected. This chemical signature correlates with the Fe4MnSi3 and manganese-containing silicide phases noted in the diffractogram.
Spectrum 3 (High Chromium area): This region is dominated by chromium (52.16 wt.%) and iron (36.37 wt.%), supporting the identification of chromium-rich silicides like Cr0.3Fe2.7Si and Cr0.2Fe0.8 observed in the higher angle XRD reflections.
The microstructure of V3 (
Figure 9) is noticeably more uniform compared to the previous samples, which is consistent with the simplified phase composition shown in the XRD patterns.
Microstructural observations the SEM images of Variant V3 exhibit a more homogeneous metallic matrix with a significant reduction in the size and quantity of secondary silicide inclusions. The elemental maps show a more even distribution of iron and chromium, indicating a closer approach to equilibrium crystallization conditions compared to Variants V1 and V2.
Quantitative EDS interpretation point EDS analysis of V3 (
Table 4) reflects a stabilized chemical composition across the metallic product:
Spectrum 1 (Main Matrix): Characterized by high manganese (39.29 wt.%) and iron (39.98 wt.%) content with moderate chromium (16.72 wt.%), confirming the formation of complex α-(Fe,Cr,Mn) solid solutions identified by XRD.
Spectrum 2 (Mn-enriched zone): Shows a substantial manganese concentration (61.32 wt.%), which aligns with the intense reflections of manganese-incorporated phases in the diffraction study.
Spectrum 3 (Balanced alloy zone): Demonstrates a balanced distribution of iron (41.77 wt.%) and manganese (27.03 wt.%) with a low silicon content (4.88 wt.%), correlating with the minor FeSi and Fe3Si reflections detected in the 48–50° (2theta) range.
Summary of Phase and Microstructural Evolution. The comparative analysis of the three variants demonstrates a clear correlation between the crystalline phases identified by XRD and the microstructural features observed via SEM/EDS. In V1, the complex multiphase diffraction pattern, including distinct Cr3Si and ε-FeSi reflections, is fully validated by the high degree of chemical segregation and the presence of Cr-rich dendritic zones in the SEM micrographs. V2 represents an intermediate state where the matrix shifts toward an iron–chromium base, with EDS confirming that manganese and silicon are primarily concentrated in specific silicide clusters like Fe4MnSi3 and Fe5Si3, as detected in the corresponding XRD patterns. Finally, Variant V3 exhibits the most homogeneous structure, where XRD and EDS maps reflect a stable α-(Fe,Cr,Mn) solid solution with minimal secondary inclusions.
Ultimately, these results confirm that the phase composition and morphology of the Cr–Mn metallic products are highly sensitive to the specific reduction conditions defined for each variant. The presence of silicon (ranging from 2.16 wt.% to 6.73 wt.% across all variants) acts as a key driver for silicide formation. While the AlSiMn reductant ensured high efficiency in chromium and manganese recovery, the variations in thermal regimes and reactant ratios between V1, V2, and V3 directly influenced the final phase distribution. The transition from the complex, non-equilibrium silicide-rich structure of V1 to the more stabilized solid solutions of V3 demonstrates that the degree of alloying and structural homogeneity can be precisely controlled. This consistency across XRD and SEM/EDS methods provides a reliable basis for interpreting the relationship between experimental parameters and the resulting alloy properties.
2.3. Viscosity of the Slag Melts
To elucidate the influence of thermal effects and phase composition on the behavior of the oxide phase at high temperatures, the viscosity properties of slags formed during reduction reactions in the Cr–Mn system were investigated. Slag viscosity is considered an integral parameter sensitive to chemical composition, oxide network structure, and temperature regime, allowing relationships between phase transformations, the energetics of reduction processes, and macroscopic melt behavior to be established.
In the present study, the viscosity behavior of the slag phase was determined using a combination of experimental high-temperature measurements and thermodynamic modeling. This integrated approach provides a comprehensive assessment of the effects of slag composition and exothermic thermal effects on viscosity and enables comparison of experimentally observed system behavior with calculated trends derived from equilibrium models.
The viscosity of the investigated slags was calculated using the FactSage 8.4 software package, which allows evaluation of the physicochemical properties of multicomponent oxide melts based on thermodynamic models and phase equilibria. Experimental viscosity values were obtained using an electrovibrational viscometer installed in a resistance furnace with a graphite heater, enabling assessment of the actual slag fluidity under conditions close to metallurgical practice.
At the first stage, the temperature dependence of slag viscosity was analyzed based on data obtained using the FactSage software (
Figure 10a,b). For the three slag compositions considered, characteristic trends in viscosity variation were identified in the temperature range of 1400–1800 °C. The calculated results show that the viscosity of all investigated slags decreases monotonically with increasing temperature, which is attributed to gradual thermal disruption of the oxide structural network, a reduction in the degree of polymerization, and increased mobility of liquid-phase components.
As shown in
Figure 10a, the viscosity of all investigated slags decreases monotonically with increasing temperature, which is a characteristic feature of oxide melts. This behavior is associated with the gradual thermal disruption and depolymerization of the oxide structural network, leading to increased mobility of structural units and ions in the liquid phase. Among the studied compositions, slag V2 exhibits the highest viscosity values over the entire investigated temperature range, whereas slags V1 and V3 show lower viscosities, indicating differences in their chemical composition and structural organization.
Figure 10b presents the linearization of the experimental and calculated data in ln(η)–1/T coordinates, demonstrating an approximately linear relationship with high correlation coefficients (R
2 > 0.97). This confirms that the temperature dependence of slag viscosity within the investigated temperature range is adequately described by an Arrhenius-type equation. Differences in the slopes of the fitted lines indicate variations in the apparent activation energy of viscous flow, which are governed by the degree of polymerization of the oxide network and the ratio of network-forming to network-modifying oxides.
However, the FactSage software calculates viscosity only for an idealized homogeneous liquid phase and does not account for the possible presence of solid inclusions, microcrystalline phases, or structural heterogeneity of real melts. Therefore, to correctly describe the viscous state of slags under metallurgical conditions, the effective viscosity determined experimentally was used in this study.
For this purpose, slag samples were subjected to high-temperature measurements using an electrovibrational viscometer installed in a resistance furnace with a graphite heater. Pre-crushed slag samples (30–50 g) were placed in a molybdenum crucible (inner diameter 18 mm, outer diameter 30 mm, height 65 mm) positioned in the constant-temperature zone of the furnace.
After complete melting of the sample in the temperature range of 1550–1650 °C, the melt was stirred with a molybdenum rod for approximately 5 min to ensure chemical and thermal homogeneity. Subsequently, a molybdenum probe (spindle) with a diameter of 2 mm and a length of 40 mm was introduced into the central part of the crucible using a screw lifting mechanism to a depth of 10–12 mm from the melt surface.
Viscosity measurements were carried out under homogeneous liquid conditions during controlled cooling at a rate of 3–5 °C·min
−1 until the onset of solidification. Although the measurements were continuous, specific data points were selected for
Figure 11 to facilitate a clear comparison between the experimental results for different compositions (V1–V3) and the linearized plots.
The temperature in the furnace working zone was monitored using a tungsten–rhenium thermocouple protected by a corundum sheath, with the measuring junction positioned in close proximity to the crucible bottom, ensuring high accuracy of melt temperature determination.
Comparison of experimentally determined effective viscosity with FactSage-calculated values revealed discrepancies caused by the influence of melt structural heterogeneity and the possible presence of solid phases. The obtained results confirm that the use of effective viscosity provides a more adequate description of slag behavior under conditions close to real metallurgical processes and enables reliable assessment of slag fluidity and technological suitability.
Figure 11a shows the temperature dependence of the effective viscosity for the three investigated slag variants (V1–V3), determined experimentally using the electrovibrational method. The temperature ranges for measurements (ranging from 1400 °C to 1650 °C) were individually selected for each variant based on their specific crystallization onset temperatures. For all systems, a monotonic decrease in viscosity with increasing temperature is observed, which is attributed to the thermally induced restructuring of the melt. At elevated temperatures, the breakdown of bridging bonds in the silicate–aluminate network occurs, reducing the degree of polymerization and increasing the mobility of modifying cations (Ca
2+, Mg
2+, Fe
2+, Mn
2+, etc.).
In the temperature range of 1400–1450 °C, the differences between the slag variants are most pronounced. Variant 2 (Curve 2) exhibits the highest effective viscosity values (up to ~7.5 Pa·s), indicating a more highly polymerized structural network with a larger fraction of complex silicate chains. Variants 1 and 3 show lower viscosity values in this range, suggesting a more open structural network. As the temperature rises above 1550 °C, the viscosity curves of all three variants converge, indicating a transition to a near-homogeneous state where the influence of structural heterogeneity and microcrystalline inclusions is minimized.
Figure 11b presents the experimental data in Arrhenius coordinates (ln
η − 1/T). While high linearity is observed in the high-temperature regions (R
2 = 0.94–0.98), the plots exhibit a noticeable deviation from strict linearity as the temperature approaches the solidification point. This “curvature” in the Arrhenius plots is explained by the transition from a purely viscous flow to a heterogeneous state, where the onset of crystallization increases the apparent activation energy of the flow. This behavior is characteristic of real metallurgical slags and reflects the systematic increase in energy barriers for ion transport during cooling.
The variation in the slopes of the Arrhenius plots confirms that the temperature sensitivity of viscosity is governed by the slag chemical composition, particularly basicity and the ratio of network-forming to network-modifying oxides (CaO, MgO). Thus, the combined analysis of
Figure 11a,b demonstrates that the viscosity behavior of the investigated melts is controlled by the degree of polymerization and the strength of ionic bonds within the oxide network under conditions closely mimicking metallurgical practice.
Table 5 summarizes the Arrhenius equation parameters, the activation energy of viscous flow (E
a), and the crystallization onset temperatures (T
cr) for the three investigated slag variants. These parameters were determined based on experimental effective viscosity data processed in lnη–1/T coordinates, as well as the analysis of viscosity anomalies identified on the temperature dependences.
The crystallization onset temperatures (Tcr) were determined from characteristic inflection points on the effective viscosity versus temperature curves. At these points, the formation of the first solid phases in the melt begins, structural homogeneity is disrupted, and a sharp increase in the rate of viscosity growth is observed. According to the obtained results, Tcr values for variants 1, 2, and 3 are 1415, 1380, and 1435 °C, respectively.
The Arrhenius equations were obtained by fitting the experimental effective viscosity data and provide a quantitative description of the temperature dependence of the viscosity properties of the investigated melts. The coefficient B = Ea/R characterizes the energy barrier associated with ion transport and structural rearrangement of the slag matrix. The calculated activation energies of viscous flow are 189.8 kJ·mol−1 for variant 1, 175.8 kJ·mol−1 for variant 2, and 161.7 kJ·mol−1 for variant 3.
The gradual decrease in activation energy from variant 1 to variant 3 reflects weakening of the melt structural network and a reduction in the degree of polymerization. Variant 1 is characterized by the most robust silicate–aluminate structure, whereas variant 3 exhibits a more mobile and thermally sensitive structural organization. Variant 2 occupies an intermediate position between these limiting cases.
The combined analysis of experimentally determined effective viscosity, Arrhenius parameters, and crystallization onset temperatures demonstrates that all three slag compositions exhibit low viscosity, high thermal stability, and a wide temperature range of homogeneous liquid-phase existence in the working temperature interval of approximately 1600 °C. Despite differences in Ea and Tcr, all investigated variants provide favorable conditions for efficient metal–slag separation and stable smelting operation, confirming their technological suitability for producing Cr–Mn ligatures.
To interpret the experimental viscosity data, the investigated slag compositions were projected onto the phase diagram (
Figure 12a–c). The analysis shows that Variant 1 (V1) is located at the boundary of the Merwinite/Periclase fields, with a liquidus temperature in the range of 1550–1600 °C. This positioning within a relatively fluid mineralogical zone explains its stable viscosity at operating temperatures.
Variant 2 (V2) is situated in a transition zone beyond the Merwinite field, with a higher liquidus temperature of 1600–1650 °C. The shift toward this more refractory region correlates with the observed increase in effective viscosity and a higher activation energy (Ea), as the melt structure becomes more prone to polymerization and early phase precipitation during cooling.
Variant 3 (V3) lies at the boundary of the Rankinite/Belite fields (liquidus ~1600 °C). Despite the presence of these phases, the composition maintains high fluidity, which is consistent with the experimental results showing the lowest crystallization onset temperature (Tcr). This phase-based evidence confirms that all three variants are optimized for smelting, with V1 and V3 providing the most favorable conditions for efficient metal–slag separation.