Jacareubin Derivatives Increase Their Anti-Allergic Activity
Abstract
1. Introduction
2. Results
2.1. Chemical Modification of Jacareubin and Xanthone V
2.2. Effect of Xanthone Methylation and Acetylation on FcεRI-Induced BMMC Degranulation
2.3. Methylation of Compound 2 Does Not Affect Its Anti-Allergic Activity In Vivo
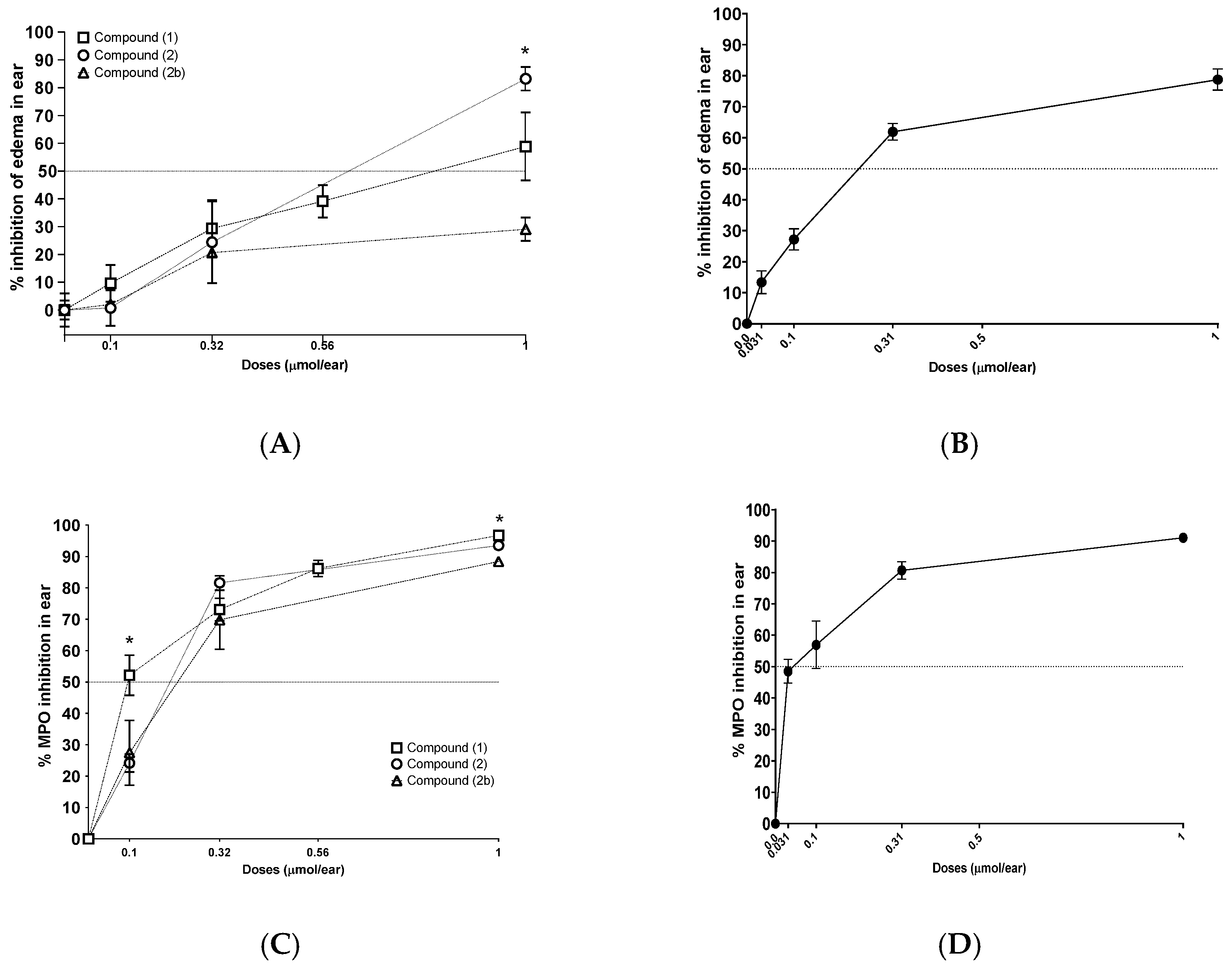
2.4. Inhibition of TPA-Induced Inflammation with Xanthones
2.5. Molecular Docking Analysis Confirms In Vitro and In Vivo Results
3. Discussion
4. Materials and Methods
4.1. Reagents
4.2. Equipment for Analytical Determinations
4.3. Collection of Plant Material and Isolation of Compounds 1 and 2
4.4. General Procedure for the Synthesis of Acetylated Derivatives of Compounds 1 and 2
4.5. General Procedure for the Synthesis of Methylated Derivatives of Compounds 1 and 2
4.6. Determination of β-Hexosaminidase Activity
4.7. Xanthine Oxidase Activity
4.8. Passive Cutaneous Anaphylaxis Model (PCA)
4.9. TPA-Induced Edema Model in the Mouse Ear
4.10. Mieloperoxidase Activity
4.11. Molecular Docking Analysis
4.12. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AHR | Airway hyper-responsiveness |
| BMMCs | Bone marrow-derived mast cells |
| CHCl3 | Chloroform |
| DHE | Dihydroethidium |
| DMF | N,N-dimethylformamide |
| DNP-HSA | Dinitrophenyl-human seric albumin |
| DPI | Diphenyleneiodonium chloride |
| ED50 | Median effective dose |
| EDTA | Ethylenediaminetetraacetic acid |
| EGTA | Ethylene glycol-bis(β-aminoethyl ether)-N,N,N′,N′-tetraacetic acid |
| ERK | Extracellular signal-regulated kinase |
| FcεRI | High affinity IgE receptor |
| H2SO4 | Sulfuric acid |
| HCl | Hydrochloric acid |
| HO-1 | Heme oxygenase-1 |
| HPLC | High performance liquid chromatography |
| HTAB | Hexadecyltrimethyl-ammonium bromide |
| IC50 | Half maximal inhibitory concentration |
| IgE | Immunoglobulin type E |
| JNK | c-Jun N-terminal kinase |
| LPS | Lipopolysaccharide |
| MAPKs | Mitogen-activated protein kinases |
| MeOH | methanol |
| MPO | Myeloperoxidase |
| Na2CO3 | Sodium carbonate |
| NADPH | Nicotinamide adenine dinucleotide phosphate |
| NBT | Nitrotetrazolium blue |
| NF-κB | Nuclear factor kappa-light-chain-enhancer of activated B cells |
| Nrf2 | Nuclear factor erythroid 2-related factor 2 |
| OVA | Ovalbumin |
| PBS | Phosphate-buffered saline |
| PCA | Passive cutaneous anaphylaxis |
| ROS | Reactive oxygen species |
| SEM | Standard error of the mean |
| SOD | Superoxide dismutase |
| Syk | Spleen tyrosine kinase |
| TMB | 3,3′,5,5′,tetramethyl-benzidine |
| TPA | 12-O-tetradecanoylphorbol-13-acetate |
| X-V | Xanthone V |
References
- Oriola, A.O.; Kar, P. Naturally Occurring Xanthones and Their Biological Implications. Molecules 2024, 29, 4241. [Google Scholar] [CrossRef]
- Masters, K.S.; Bräse, S. Xanthones from Fungi, Lichens, and Bacteria: The Natural Products and Their Synthesis. Chem. Rev. 2012, 112, 3717–3776. [Google Scholar] [CrossRef] [PubMed]
- Bedi, P.; Gupta, R.; Pramanik, T. Synthesis and biological properties of pharmaceutically important xanthones and benzoxanthone analogs: A brief review. Asian J. Pharm. Clin. Res. 2018, 11, 12–20. [Google Scholar] [CrossRef]
- Feng, Z.; Lu, X.; Gan, L.; Zhang, Q.; Lin, L. Xanthones, A Promising Anti-Inflammatory Scaffold: Structure, Activity, and Drug Likeness Analysis. Molecules 2020, 25, 598. [Google Scholar] [CrossRef]
- Lu, Y.; Cai, S.; Nie, J.; Li, Y.; Shi, G.; Hao, J.; Fu, W.; Tan, H.; Chen, S.; Li, B.; et al. The Natural Compound Nujiangexanthone A Suppresses Mast Cell Activation and Allergic Asthma. Biochem. Pharmacol. 2016, 100, 61–72. [Google Scholar] [CrossRef]
- Castillo-Arellano, J.I.; Guzmán-Gutiérrez, S.L.; Ibarra-Sánchez, A.; Hernández-Ortega, S.; Nieto-Camacho, A.; Medina-Campos, O.N.; Pedraza-Chaverri, J.; Reyes-Chilpa, R.; González-Espinosa, C. Jacareubin Inhibits FcεRI-Induced Extracellular Calcium Entry and Production of Reactive Oxygen Species Required for Anaphylactic Degranulation of Mast Cells. Biochem. Pharmacol. 2018, 154, 344–356. [Google Scholar] [CrossRef]
- Gunter, N.V.; Teh, S.S.; Lim, Y.M.; Mah, S.H. Natural Xanthones and Skin Inflammatory Diseases: Multitargeting Mechanisms of Action and Potential Application. Front. Pharmacol. 2020, 11, 594202. [Google Scholar] [CrossRef]
- Piao, C.H.; Fan, Y.J.; Van Nguyen, T.; Song, C.H.; Chai, O.H. Mangiferin Alleviates Ovalbumin-Induced Allergic Rhinitis via Nrf2/HO-1/NF-ΚB Signaling Pathways. Int. J. Mol. Sci. 2020, 21, 3415. [Google Scholar] [CrossRef]
- Chae, H.S.; Oh, S.R.; Lee, H.K.; Joo, S.H.; Chin, Y.W. Mangosteen Xanthones, α-and γ-Mangostins, Inhibit Allergic Mediators in Bone Marrow-Derived Mast Cell. Food Chem. 2012, 134, 397–400. [Google Scholar] [CrossRef]
- Rivera, D.G.; Balmaseda, I.H.; León, A.Á.; Hernández, B.C.; Montiel, L.M.; Garrido, G.G.; Hernández, R.D.; Cuzzocrea, S. Anti-Allergic Properties of Mangifera indica L. Extract (Vimang) and Contribution of Its Glucosylxanthone Mangiferin. J. Pharm. Pharmacol. 2006, 58, 385–392. [Google Scholar] [CrossRef] [PubMed]
- García, D.; Escalante, M.; Delgado, R.; Ubeira, F.M.; Leiro, J. Anthelminthic and Antiallergic Activities of Mangifera indica L. Stem Bark Components Vimang and Mangiferin. Phytother. Res. 2003, 17, 1203–1208. [Google Scholar] [CrossRef]
- Itoh, T.; Ohguchi, K.; Iinuma, M.; Nozawa, Y.; Akao, Y. Inhibitory Effect of Xanthones Isolated from the Pericarp of Garcinia mangostana L. on Rat Basophilic Leukemia RBL-2H3 Cell Degranulation. Bioorg. Med. Chem. 2008, 16, 4500–4508. [Google Scholar] [CrossRef]
- Lee, B.; Trinh, H.T.; Bae, E.A.; Jung, K.; Kim, D.H. Mangiferin Inhibits Passive Cutaneous Anaphylaxis Reaction and Pruritus in Mice. Planta Med. 2009, 75, 1415–1417. [Google Scholar] [CrossRef] [PubMed]
- Jang, H.Y.; Kwon, O.K.; Oh, S.R.; Lee, H.K.; Ahn, K.S.; Chin, Y.W. Mangosteen Xanthones Mitigate Ovalbumin-Induced Airway Inflammation in a Mouse Model of Asthma. Food Chem. Toxicol. 2012, 50, 4042–4050. [Google Scholar] [CrossRef] [PubMed]
- Ishida, M.; Sasaki, T.; Nishi, K.; Tamamoto, T.; Sugahara, T. Suppressive Effect of Ethanol Extract from Mango (Mangifera indica L.) Peel on IgE Production in Vitro and in Vivo. Biosci. Biotechnol. Biochem. 2018, 82, 732–739. [Google Scholar] [CrossRef]
- Rivera, D.G.; Hernández, I.; Merino, N.; Luque, Y.; Álvarez, A.; Martín, Y.; Amador, A.; Nuevas, L.; Delgado, R. Mangifera indica L. Extract (Vimang) and Mangiferin Reduce the Airway Inflammation and Th2 Cytokines in Murine Model of Allergic Asthma. J. Pharm. Pharmacol. 2011, 63, 1336–1345. [Google Scholar] [CrossRef]
- Guo, H.W.; Yun, C.X.; Hou, G.H.; Du, J.; Huang, X.; Lu, Y.; Keller, E.T.; Zhang, J.; Deng, J.G. Mangiferin Attenuates Th1/Th2 Cytokine Imbalance in an Ovalbumin-Induced Asthmatic Mouse Model. PLoS ONE 2014, 9, e100394. [Google Scholar] [CrossRef]
- Yun, C.; Chang, M.; Hou, G.; Lan, T.; Yuan, H.; Su, Z.; Zhu, D.; Liang, W.; Li, Q.; Zhu, H.; et al. Mangiferin Suppresses Allergic Asthma Symptoms by Decreased Th9 and Th17 Responses and Increased Treg Response. Mol. Immunol. 2019, 114, 233–242. [Google Scholar] [CrossRef]
- García-Niño, W.R.; Estrada-Muñiz, E.; Valverde, M.; Reyes-Chilpa, R.; Vega, L. Cytogenetic Effects of Jacareubin from Calophyllum Brasiliense on Human Peripheral Blood Mononucleated Cells in Vitro and on Mouse Polychromatic Erythrocytes in Vivo. Toxicol. Appl. Pharmacol. 2017, 335, 6–15. [Google Scholar] [CrossRef]
- Castillo-Arellano, J.; González-Espinosa, C.; Jiménez-Estrada, M.; Reyes-Chilpa, R. Natural Compounds against Allergic and Pseudoallergic Reactions: Molecular Mechanisms of Action. Stud. Nat. Prod. Chem. 2024, 80, 269–325. [Google Scholar] [CrossRef]
- Reyes-Chilpa, R.; Jimenez-Estrada, M.; Estrada-Muñiz, E. Antifungal Xanthones from Calophyllum Brasiliensis Heartwood. J. Chem. Ecol. 1997, 23, 1901–1911. [Google Scholar] [CrossRef]
- Colquhoun, H.M.; Lewis, D.F.; Williams, D.J. Synthesis of Dixanthones and Poly(Dixanthone)s by Cyclization of 2-Aryloxybenzonitriles in Trifluoromethanesulfonic Acid. Org. Lett. 2001, 3, 2337–2340. [Google Scholar] [CrossRef]
- Yen, C.T.; Nakagawa-Goto, K.; Hwang, T.L.; Morris-Natschke, S.L.; Bastow, K.F.; Wu, Y.C.; Lee, K.H. Design and Synthesis of Gambogic Acid Analogs as Potent Cytotoxic and Anti-Inflammatory Agents. Bioorg. Med. Chem. Lett. 2012, 22, 4018–4022. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, L.A.; Deka, B.; Sharmah, H.; Patowary, P.; Bharali, D.; Sahariah, B.J.; Talukdar, A. Exploring Synthetic Xanthone Derivatives as Potential Anti-Inflammatory Agents: A Comprehensive Review. Chem. Pap. 2024, 78, 7313–7333. [Google Scholar] [CrossRef]
- Hu, L.; Hu, H.; Wu, W.; Chai, X.; Luo, J.; Wu, Q. Discovery of Novel Xanthone Derivatives as Xanthine Oxidase Inhibitors. Bioorg. Med. Chem. Lett. 2011, 21, 4013–4015. [Google Scholar] [CrossRef]
- Manetz, T.S.; Gonzalez-Espinosa, C.; Arudchandran, R.; Xirasagar, S.; Tybulewicz, V.; Rivera, J. Vav1 Regulates Phospholipase Cγ Activation and Calcium Responses in Mast Cells. Mol. Cell. Biol. 2001, 21, 3763–3774. [Google Scholar] [CrossRef]
- Saitoh, S.; Arudchandran, R.; Manetz, T.S.; Zhang, W.; Sommers, C.L.; Love, P.E.; Rivera, J.; Samelson, L.E. LAT Is Essential for FcεRI-Mediated Mast Cell Activation. Immunity 2000, 12, 525–535. [Google Scholar] [CrossRef]
- Pineda-Ramírez, N.; Calzada, F.; Alquisiras-Burgos, I.; Medina-Campos, O.N.; Pedraza-Chaverri, J.; Ortiz-Plata, A.; Estrada, E.P.; Torres, I.; Aguilera, P. Antioxidant Properties and Protective Effects of Some Species of the Annonaceae, Lamiaceae, and Geraniaceae Families against Neuronal Damage Induced by Excitotoxicity and Cerebral Ischemia. Antioxidants 2020, 9, 253. [Google Scholar] [CrossRef]
- Trujillo, J.; Molina-Jijón, E.; Medina-Campos, O.N.; Rodríguez-Muñoz, R.; Reyes, J.L.; Barrera, D.; Pedraza-Chaverri, J. Superoxide Anion Production and Expression of Gp91phox and P47phox Are Increased in Glomeruli and Proximal Tubules of Cisplatin-Treated Rats. J. Biochem. Mol. Toxicol. 2015, 29, 149–156. [Google Scholar] [CrossRef] [PubMed]
- Weng, Z.; Patel, A.B.; Panagiotidou, S.; Theoharides, T.C. The Novel Flavone Tetramethoxyluteolin Is a Potent Inhibitor of Human Mast Cells. J. Allergy Clin. Immunol. 2015, 135, 1044–1052.e5. [Google Scholar] [CrossRef] [PubMed]
- Klemm, S.; Gutermuth, J.; Hültner, L.; Sparwasser, T.; Behrendt, H.; Peschel, C.; Mak, T.W.; Jakob, T.; Ruland, J. The Bcl10–Malt1 Complex Segregates FcεRI-Mediated Nuclear Factor ΚB Activation and Cytokine Production from Mast Cell Degranulation. J. Exp. Med. 2006, 203, 337–347. [Google Scholar] [CrossRef] [PubMed]
- Carlson, R.P.; Lynn, O.D.; Chang, J.; Lewis, A.J. Modulation of Mouse Ear Edema by Cyclooxygenase and Lipoxygenase Inhibitors and Other Pharmacologic Agents. Agents Actions 1985, 17, 197–204. [Google Scholar] [CrossRef] [PubMed]
- Bradley, P.P.; Priebat, D.A.; Christensen, R.D.; Rothstein, G. Measurement of Cutaneous Inflammation: Estimation of Neutrophil Content with an Enzyme Marker. J. Investig. Dermatol. 1982, 78, 206–209. [Google Scholar] [CrossRef]
- Suzuki, K.; Ota, H.; Sasagawa, S.; Sakatani, T.; Fujikura, T. Assay Method for Myeloperoxidase in Human Polymorphonuclear Leukocytes. Anal. Biochem. 1983, 132, 345–352. [Google Scholar] [CrossRef]
- Berman, H.M.; Westbrook, J.; Feng, Z.; Gilliland, G.; Bhat, T.N.; Weissig, H.; Shindyalov, I.N.; Bourne, P.E. The Protein Data Bank. Nucleic Acids Res. 2000, 28, 235–242. [Google Scholar] [CrossRef] [PubMed]









| Xanthine Oxidase (kJ/mol) | ||||
| No. | Title | Tot Q | docking score | glide gscore |
| 4 | Compound 2 | −1 | −10.251 | −10.913 |
| Residues of XO that interact with compound 2 | ||||
| Residue | Substituent with which it interacts | |||
| 1 | GLH802 | HO-C1 | ||
| 2 | THR1010 | HO-C5 and HO-C6 | ||
| 3 | VAL1011 | HO-C5 | ||
| 4 | PHE1009 | Ring B and C | ||
| 5 | ARG880 | HO-C6 | ||
| 6 | PHE914 | Ring C | ||
| No. | Title | Tot Q | docking score | glide gscore |
| 4 | Compound 1 | −1 | −9.317 | −10.019 |
| Residues of XO that interact with compound 1 | ||||
| Residue | Substituent with which it interacts | |||
| 1 | GLH802 | HO-C1 | ||
| 2 | THR1010 | HO-C5 and HO-C6 | ||
| 3 | ARG880 | HO-C6 | ||
| PHE1009 | Ring B and C | |||
| PHE914 | Ring B and C | |||
| Myeloperoxidase (kJ/mol) | ||||
| No. | Title | Tot Q | docking score | glide gscore |
| 6 | Compound 1 | −1 | −6.049 | −6.751 |
| Residues of MPO that interact with compound 1 | ||||
| Residue | Substituent with which it interacts | |||
| 1 | GLU102 | HO-C5 | ||
| 2 | PHE147 | HO-C3 | ||
| 3 | ARG239 | HO-C6 | ||
| No. | Title | Tot Q | docking score | glide gscore |
| 5 | Compound 2 | −1 | −5.151 | −5.814 |
| Residues of MPO that interact with compound 1 | ||||
| Residue | Substituent with which it interacts | |||
| 1 | GLN91 | HO-C6 | ||
| 2 | HEM601 | HO-C6 and Ring C | ||
| 3 | GLU102 | HO-C4 | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Tavera-Hernández, R.; Pérez-Rodríguez, J.; Nieto-Camacho, A.; Medina-Campos, O.N.; Pedraza-Chaverri, J.; León, F.; González-Espinosa, C.; Jiménez-Estrada, M.; Reyes-Chilpa, R.; Castillo-Arellano, J.I. Jacareubin Derivatives Increase Their Anti-Allergic Activity. Molecules 2026, 31, 1666. https://doi.org/10.3390/molecules31101666
Tavera-Hernández R, Pérez-Rodríguez J, Nieto-Camacho A, Medina-Campos ON, Pedraza-Chaverri J, León F, González-Espinosa C, Jiménez-Estrada M, Reyes-Chilpa R, Castillo-Arellano JI. Jacareubin Derivatives Increase Their Anti-Allergic Activity. Molecules. 2026; 31(10):1666. https://doi.org/10.3390/molecules31101666
Chicago/Turabian StyleTavera-Hernández, Rosario, Jesabel Pérez-Rodríguez, Antonio Nieto-Camacho, Omar Noel Medina-Campos, José Pedraza-Chaverri, Francisco León, Claudia González-Espinosa, Manuel Jiménez-Estrada, Ricardo Reyes-Chilpa, and Jorge Ivan Castillo-Arellano. 2026. "Jacareubin Derivatives Increase Their Anti-Allergic Activity" Molecules 31, no. 10: 1666. https://doi.org/10.3390/molecules31101666
APA StyleTavera-Hernández, R., Pérez-Rodríguez, J., Nieto-Camacho, A., Medina-Campos, O. N., Pedraza-Chaverri, J., León, F., González-Espinosa, C., Jiménez-Estrada, M., Reyes-Chilpa, R., & Castillo-Arellano, J. I. (2026). Jacareubin Derivatives Increase Their Anti-Allergic Activity. Molecules, 31(10), 1666. https://doi.org/10.3390/molecules31101666









