Defining Aldol Chemoselectivity in the Presence of Henry Nucleophiles (Nitroalkanes)
Abstract
1. Introduction
2. Discussion and Results
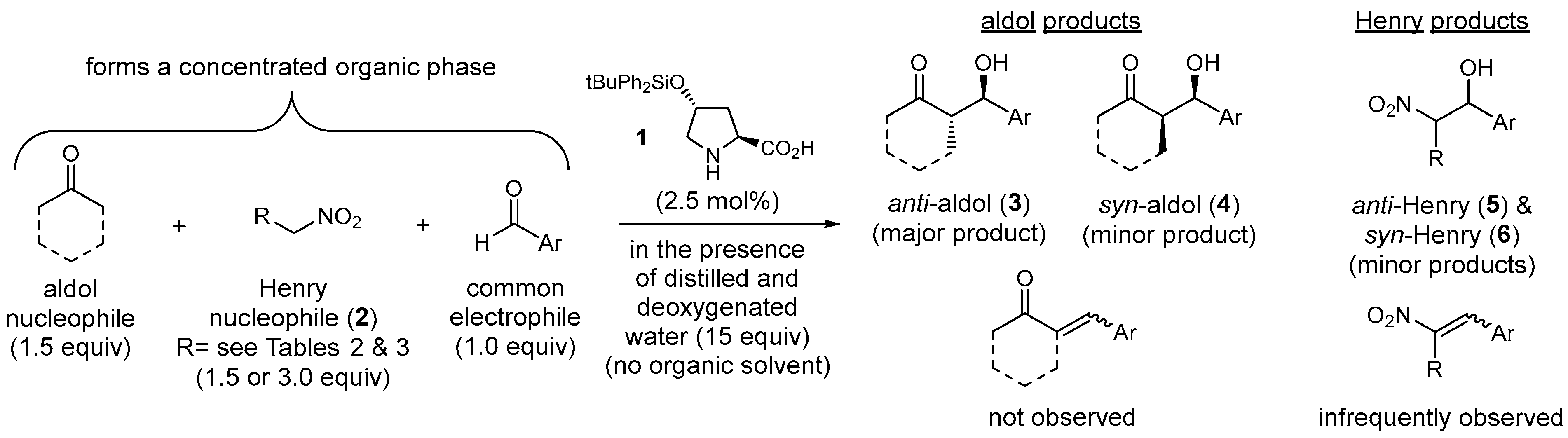
2.1. Aldol Versus Henry Competition Reactions
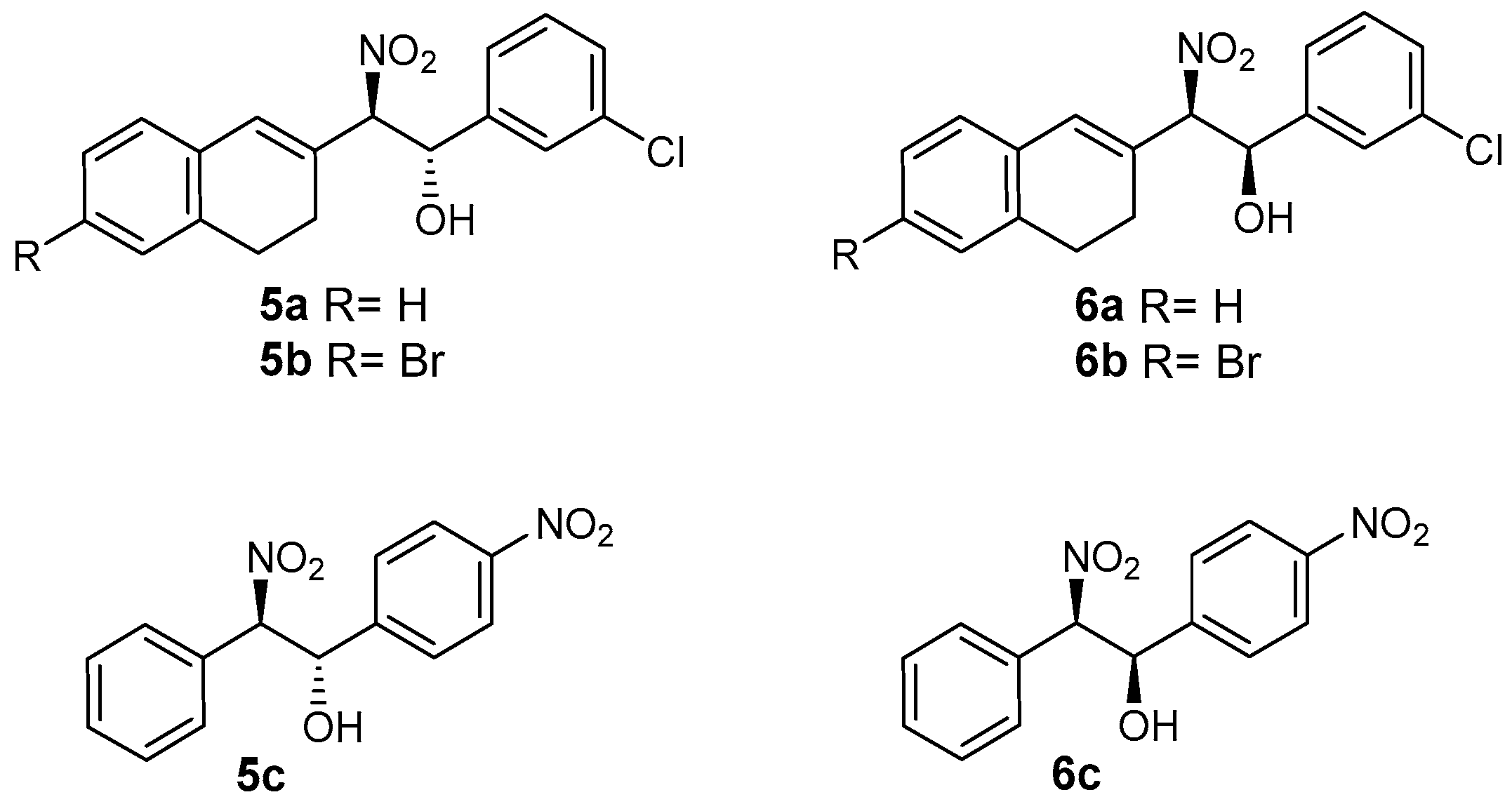
2.2. Henry Product Reference Standards
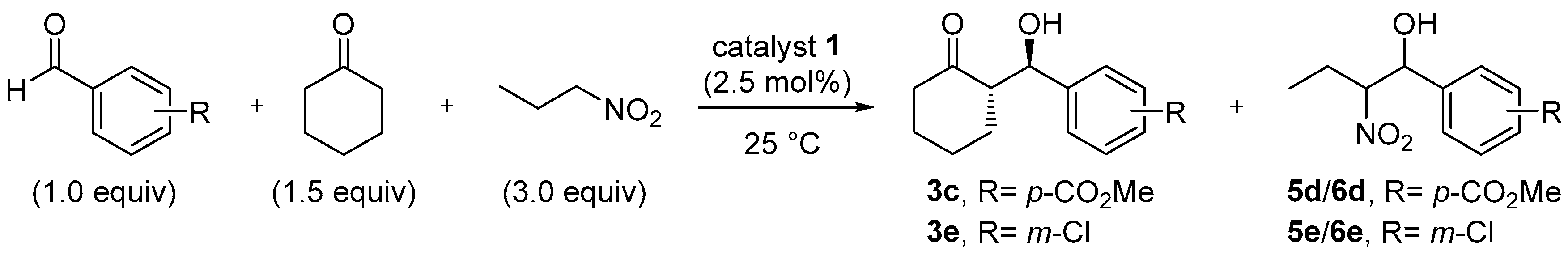
2.3. Solvent Studies: Uniqueness of Water-Based Solvent Systems
3. Materials and Methods
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- List, B.; Lerner, R.A.; Barbas, C.F., III. Proline-catalyzed Direct Asymmetric Aldol Reactions. J. Am. Chem. Soc. 2000, 122, 2395–2396. [Google Scholar] [CrossRef]
- Ahrendt, K.A.; Borths, C.J.; MacMillan, D.W.C. New Strategies for Organic Catalysis: The First Highly Enantioselective Organocatalytic Diels-Alder Reaction. J. Am. Chem. Soc. 2000, 122, 4243–4244. [Google Scholar] [CrossRef]
- Marigo, M.; Melchiorre, P. Chemoselectivity in Asymmetric Aminocatalysis. ChemCatChem 2010, 2, 621–623. [Google Scholar] [CrossRef]
- Ma, J.; Gao, B.; Song, G.; Zhang, R.; Wang, Q.; Ye, Z.; Chen, W.-W.; Zhao, B. Asymmetric α-Allylation of Glycinate with Switched Chemoselectivity Enabled by Customized Bifunctional Pyridoxal Catalysts. Angew. Chem. Int. Ed. 2022, 61, e202200850. [Google Scholar] [CrossRef]
- Nugent, T.C.; Najafian, F.T.; Hussein, H.A.E.D.; Hussain, I. Beyond Chemoselectivity: Catalytic Site-Selective Aldolization of Diketones and Exploitation for Enantioselective Alzheimer Drug Candidate Synthesis. Chem. Eur. J. 2016, 22, 14342–14348. [Google Scholar] [CrossRef]
- Jhuo, D.-H.; Hong, B.-C.; Chang, C.-W.; Lee, G.-H. One-Pot Organocatalytic Enantioselective Michael−Michael−Aldol−Henry Reaction Cascade. A Facile Entry to the Steroid System with Six Contiguous Stereogenic Centers. Org. Lett. 2014, 16, 2724–2727. [Google Scholar] [CrossRef]
- Lee, A.; Michrowska, A.; Sulzer-Mosse, S.; List, B. The Catalytic Asymmetric Knoevenagel Condensation. Angew. Chem. Int. Ed. 2011, 50, 1707–1710. [Google Scholar] [CrossRef] [PubMed]
- Ibrahem, I.; Zou, W.; Casas, J.; Sundén, H.; Córdova, A. Direct Organocatalytic Enantioselective α-Aminomethylation of Ketones. Tetrahedron 2006, 62, 357–364. [Google Scholar] [CrossRef]
- Enders, D.; Hüttl, M.R.M.; Grondal, C.; Raabe, G. Control of Four Stereocentres in a Triple Cascade Organocatalytic Reaction. Nature 2006, 441, 861–863. [Google Scholar] [CrossRef] [PubMed]
- Al Beiruty, H.; Zhylinska, S.-S.; Kutateladze, N.; Cheong, H.K.T.; Ñíguez, J.A.; Burlingham, S.J.; Marset, X.; Guillena, G.; Chinchilla, R.; Alonso, D.A.; et al. Enantioselective Catalytic Aldol Reactions in the Presence of Knoevenagel Nucleophiles: A Chemoselective Switch Optimized in Deep Eutectic Solvents Using Mechanochemistry. Molecules 2024, 29, 4. [Google Scholar] [CrossRef]
- Nugent, T.C.; Goswami, F.; Debnath, S.; Hussain, I.; Hussein, H.A.E.D.; Karn, A.; Nakka, S. Harnessing Additional Capability from in Water Reaction Conditions: Aldol versus Knoevenagel Chemoselectivity. Adv. Synth. Catal. 2021, 363, 3539–3545. [Google Scholar] [CrossRef]
- Smith, M.B.; March, J. March’s Advanced Organic Chemistry: Reactions, Mechanisms, and Structure, 6th ed.; Wiley-Interscience: Hoboken, NJ, USA, 2007. [Google Scholar]
- Mukherjee, S.; Yang, J.W.; Hoffmann, S.; List, B. Asymmetric Enamine Catalysis. Chem. Rev. 2007, 107, 5471–5569. [Google Scholar] [CrossRef] [PubMed]
- Trost, B.M.; Brindle, C.S. The Direct Catalytic Asymmetric Aldol Reaction. Chem. Soc. Rev. 2010, 39, 1600–1632. [Google Scholar] [CrossRef]
- Mlynarski, J.; Baś, S. Catalytic Asymmetric Aldol Reactions in Aqueous Media—A 5 Year Update. Chem. Soc. Rev. 2014, 43, 577–587. [Google Scholar] [CrossRef]
- Lam, Y.-H.; Grayson, M.N.; Holland, M.C.; Simon, A.; Houk, K.N. Theory and Modeling of Asymmetric Catalytic Reactions. Acc. Chem. Res. 2016, 49, 750–762. [Google Scholar] [CrossRef]
- Yamashita, Y.; Yasukawa, T.; Yoo, W.-J.; Kitanosono, T.; Kobayashi, S. Catalytic Enantioselective Aldol Reactions. Chem. Soc. Rev. 2018, 47, 4388–4480. [Google Scholar] [CrossRef]
- Nugent, T.C.; de Vos, A.E.; Hussain, I.; Hussein, H.A.E.D.; Goswami, F. A 2000 to 2020 Practitioner’s Guide to Chiral Amine Based Enantioselective Aldol Reactions: Ketone Substrates, Best Methods, in Water Reaction Environments, and Defining Nuances. Eur. J. Org. Chem. 2022, 2022, e202100529. [Google Scholar] [CrossRef]
- Boruwa, J.; Gogoi, N.; Saikia, P.P.; Barua, N.C. Catalytic Asymmetric Henry Reaction. Tetrahedron Asymmetry 2006, 17, 3315–3326. [Google Scholar] [CrossRef]
- Palomo, C.; Oiarbide, M.; Laso, A. Recent Advances in the Catalytic Asymmetric Nitroaldol (Henry) Reaction. Eur. J. Org. Chem. 2007, 2007, 2561–2574. [Google Scholar] [CrossRef]
- Sasai, H. Chapter 2.13: The Henry (Nitroaldol) Reaction. In Comprehensive Organic Synthesis; Johnson, J., Ed.; Elsevier: Oxford, UK, 2014; Volume 2, pp. 543–570. [Google Scholar]
- Saranya, S.; Harry, N.A.; Ujwaldev, S.M.; Anilkumar, G. Recent Advances and Perspectives on the Zinc-Catalyzed Nitroaldol (Henry) Reaction. Asian J. Org. Chem. 2017, 6, 1349–1360. [Google Scholar] [CrossRef]
- Zhang, S.; Li, Y.; Xu, Y.; Wang, Z. Recent Progress in Copper Catalyzed Asymmetric Henry Reaction. Chin. Chem. Lett. 2018, 29, 873–883. [Google Scholar] [CrossRef]
- Alvarez-Casao, Y.; Marques-Lopez, E.; Herrera, R.P. Organocatalytic Enantioselective Henry Reactions. Symmetry 2011, 3, 220–245. [Google Scholar] [CrossRef]
- Oriyama, T.; Aoyagi, M.; Iwanami, K. Henry Reaction and 1,4-Addition of Nitroalkanes to α,β-Unsaturated Carbonyl Compounds under the Influence of MS 4Å in DMSO. Chem. Lett. 2007, 36, 612–613. [Google Scholar] [CrossRef]
- Bora, P.P.; Bez, G. Henry Reaction in Aqueous Media at Neutral pH. Eur. J. Org. Chem. 2013, 2013, 2922–2929. [Google Scholar] [CrossRef]
- Pirola, M.; Compostella, M.E.; Raimondi, L.; Puglisi, A.; Benaglia, M. A Continuous-Flow, Two-Step, Metal-Free Process for the Synthesis of Differently Substituted Chiral 1,2-Diamino Derivatives. Synthesis 2018, 50, 1430–1438. [Google Scholar]
- Phukan, M.; Borah, K.J.; Borah, R. Imidazole-Catalyzed Henry Reactions in Aqueous Medium. Synth. Commun. 2008, 38, 3068–3073. [Google Scholar] [CrossRef]
- Ai, W.; Shi, R.; Zhu, L.; Jiang, D.; Ma, X.; Yuan, J.; Wang, Z. One-Pot Protocol to Synthesize N-(b-nitro)amides by Tandem Henry/Ritter reaction. RSC Adv. 2015, 5, 24044–24048. [Google Scholar] [CrossRef]
- Vovard-LeBray, C.; Jiang, F.; Wu, X.-F.; Sortais, J.-B.; Darcel, C. Cyclen-Catalyzed Henry Reaction Under Neutral Conditions. Tetrahedron Lett. 2010, 51, 4555–4557. [Google Scholar] [CrossRef]
- Luzzio, F.A. The Henry Reaction: Recent Examples. Tetrahedron 2001, 57, 915–945. [Google Scholar] [CrossRef]
- Sahoo, S.C.; Parida, C.; Pan, S.C. Organocatalytic Asymmetric Domino [3 + 2]-Cycloaddition-Acyl Transfer Reaction Between Azomethine Ylides and α-Nitro-α,β-Unsaturated Ketones. Chem. Asian J. 2023, 18, e202300563. [Google Scholar] [CrossRef] [PubMed]
- Gharui, C.; Behera, D.; Pan, S.C. Organocatalytic Asymmetric Domino Michael/Acyl Transfer Reaction Between a-Nitroketones and in situ-Generated ortho-Quinone Methides: Route to 2-(1-Arylethyl)phenols. Adv. Synth. Catal. 2018, 360, 4502–4508. [Google Scholar] [CrossRef]
- Concellón, C.; Martín, J.; Gallegos, M.; Fanjul-Mosteirín, N.; Costales, A.; Pendás, Á.M.; del Amo, V. Mimicking Enzymes: Asymmetric Induction Inside a Carbamate−Based Steroidal Cleft. Org. Lett. 2019, 21, 3994–3997. [Google Scholar] [CrossRef]
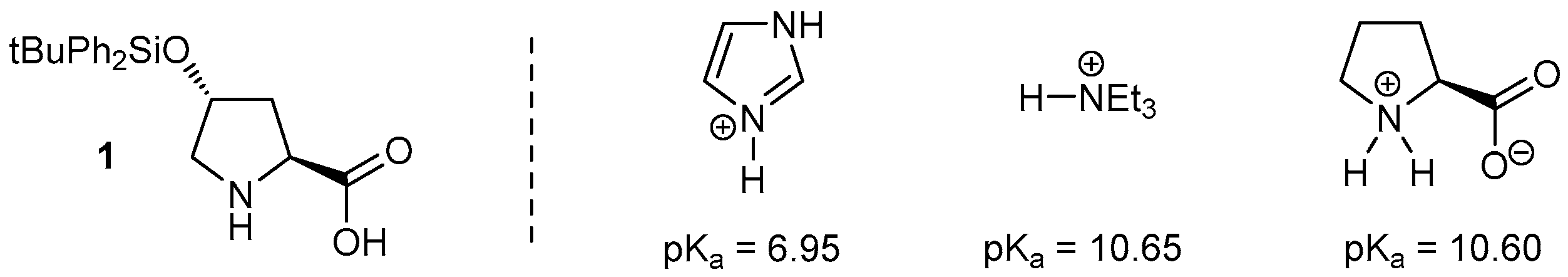
- Lõkov, M.; Tshepelevitsh, S.; Heering, A.; Plieger, P.G.; Vianello, R.; Leito, I. On the Basicity of Conjugated Nitrogen Heterocycles in Different Media. Eur. J. Org. Chem. 2017, 2017, 4475–4489. [Google Scholar] [CrossRef]
- Gallo, G.G.; Pasqualucci, C.R.; Radaelli, P.; Lancini, G.C. The Ionization Constants of Some Imidazoles. J. Org. Chem. 1964, 29, 862–865. [Google Scholar] [CrossRef]
- Hall, H.K. Correlation of the Base Strengths of Amines. J. Am. Chem. Soc. 1957, 79, 5441–5444. [Google Scholar] [CrossRef]
- Jones, F.M.; Arnett, E.M. Thermodynamics of Ionization and Solution of Aliphatic Amines in Water. In Progress in Physical Organic Chemistry; Streitwieser, A., Jr., Taft, R.W., Eds.; Wiley: New York, NY, USA, 1974; Volume 11, Chapter 4; pp. 263–322, specifically p. 292. [Google Scholar]
- Carey, F.A.; Giuliano, R.M. Organic Chemistry, 8th ed.; Chapter 25: Amino Acids, Peptides, and Proteins; McGraw-Hill Education: New York, NY, USA, 2010. [Google Scholar]
- Fazary, A.E.; Mohamed, A.F.; Lebedeva, N.S. Protonation Equilibria Studies of the Standard a-Amino Acids in NaNO3 Solutions in Water and in Mixtures of Water and Dioxane. J. Chem. Thermodyn. 2006, 38, 1467–1473. [Google Scholar] [CrossRef]
- Aratake, S.; Itoh, T.; Okano, T.; Nagae, N.; Sumiya, T.; Shoji, M.; Hayashi, Y. Highly Diastereo- and Enantioselective Direct Aldol Reactions of Aldehydes and Ketones Catalyzed by Siloxyproline in the Presence of Water. Chem. Eur. J. 2007, 13, 10246–10256. [Google Scholar] [CrossRef] [PubMed]
- Kitanosono, T.; Kobayashi, S. Reactions in Water Involving the “On-Water” Mechanism. Chem. Eur. J. 2020, 26, 9408–9429. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Castañeda, Á.; Rodríguez-Solla, H.; Concellón, C.; del Amo, V. TBD/Al2O3: A Novel Catalytic System for Dynamic Intermolecular Aldol Reactions That Exhibit Complex System Behaviour. Org. Biomol. Chem. 2012, 10, 1976–1981. [Google Scholar] [CrossRef]
- Mase, N.; Nakai, Y.; Ohara, N.; Yoda, H.; Takabe, K.; Tanaka, F.; Barbas, C.F., III. Organocatalytic Direct Asymmetric Aldol Reactions in Water. J. Am. Chem. Soc. 2006, 128, 734–735. [Google Scholar] [CrossRef]
- Agarwal, J.; Peddinti, R.K. Glucosamine-based Primary Amines as Organocatalysts for the Asymmetric Aldol Reaction. J. Org. Chem. 2011, 76, 3502–3505. [Google Scholar] [CrossRef]
- Nugent, T.C.; Spiteller, P.; Hussain, I.; Hussein, H.A.E.D.; Najafian, F.T. A Catalyst-Directed Remote Stereogenic Center Switch During the Site-Selective Aldol Desymmetrization of Cyclohexanone-Based Diketones. Adv. Synth. Catal. 2016, 358, 3706–3713. [Google Scholar] [CrossRef]
- Ramasastry, S.S.V.; Albertshofer, K.; Utsumi, N.; Tanaka, F.; Barbas, C.F., III. Mimicking Fructose and Rhamnulose Aldolases: Organocatalytic syn-Aldol Reactions with Unprotected Dihydroxyacetone. Angew. Chem. Int. Ed. 2007, 46, 5572–5575. [Google Scholar] [CrossRef] [PubMed]
- Wheland, G.W.; Farr, J. Acid Strengths of Aliphatic Nitro Compounds. J. Am. Chem. Soc. 1943, 65, 1433. [Google Scholar] [CrossRef]
- Matsui, T.; Hepler, L.G. Acid Ionizations of Nitroalkanes in Aqueous Solution. Can. J. Chem. 1973, 51, 1941–1944. [Google Scholar] [CrossRef]
- Bernasconi, C.F.; Kliner, D.A.V.; Mullin, A.S.; Ni, J.X. Kinetics of Ionization of Nitromethane and Phenylnitromethane by Amines and Carboxylate Ions in Me2SO-Water Mixtures. Evidence of Ammonium Ion-Nitronate Ion Hydrogen Bonded Complex Formation in Me2SO-Rich Solvent Mixtures. J. Org. Chem. 1988, 53, 3342–3351. [Google Scholar] [CrossRef]
- Mofaddel, N.; Bar, N.; Villemin, D.; Desbène, P.L. Determination of Acidity Constants of Enolisable Compounds by Capillary Electrophoresis. Anal. Bioanal. Chem. 2004, 380, 664–668. [Google Scholar] [CrossRef]
- Emma, M.G.; Tamburrini, A.; Martinelli, A.; Lombardo, M.; Quintavalla, A.; Trombini, C. A Simple and Efficient Protocol for Proline-Catalysed Asymmetric Aldol Reaction. Catalysts 2020, 10, 649. [Google Scholar] [CrossRef]
- Nugent, T.C.; Umar, M.N.; Bibi, A. Picolylamine as an Organocatalyst Template for Highly Diastereo- and Enantioselective Aqueous Aldol Reactions. Org. Biomol. Chem. 2010, 8, 4085–4089. [Google Scholar] [CrossRef]
- Haynes, W.M. (Ed.) CRC Handbook of Chemistry and Physics, 95th ed.; CRC Press: Boca Raton, FL, USA, 2014. [Google Scholar]
- Yalkowsky, S.H.; He, Y.; Jain, P. Handbook of Aqueous Solubility Data, 2nd ed.; CRC Press: Boca Raton, FL, USA, 2010. [Google Scholar]
- Barton, D.H.R.; Fernandez, I.; Richard, C.S.; Zard, S.Z. A Mild Procedure for the Reduction of Aliphatic Nitro Compounds to Oximes. Tetrahedron 1987, 43, 551–558. [Google Scholar] [CrossRef]
- Barton, D.H.R.; Motherwell, W.B.B.; Zard, S.Z. Nouveau Réactif de Nitrométhylation, Son Application à la Preparation de Composes Nitrométhylénés et de Certains de Leurs Dérivés et les Composes Nouveaux Obtenus. EP (European Patent) 0087359 A2, 31 August 1983. [Google Scholar]
- Barco, A.; Benetti, S.; De Risi, C.; Morelli, C.F.; Pollini, G.P.; Zanirato, V. Generation and Cycloaddition Reactions of Substituted 2-Nitro-1,3-dienes. Tetrahedron 1996, 52, 9275–9288. [Google Scholar] [CrossRef]
- Lund, H.; Nilsson, N.H. Electroorganic Preparations. XXXVII. Electrosynthesis of Cinnolines by Reductive Ring Closure of Dinitrocompounds. Acta Chem. Scand. B 1976, 30, 5–11. [Google Scholar] [CrossRef]
- Mei, H.; Xiao, X.; Zhao, X.; Fang, B.; Liu, X.; Lin, L.; Feng, X. Catalytic Asymmetric Henry Reaction of Nitroalkanes and Aldehydes Catalyzed by a Chiral N,N′-Dioxide/Cu(I) Complex. J. Org. Chem. 2015, 80, 2272–2280. [Google Scholar] [CrossRef]
- Karmakar, A.; Maji, T.; Wittmann, S.; Reiser, O. L-Proline/CoCl2-Catalyzed Highly Diastereo- and Enantioselective Direct Aldol Reactions. Chem. Eur. J. 2011, 17, 11024–11029. [Google Scholar] [CrossRef]
- Pandey, S.; Bansal, D.; Gupta, R. A Metalloligand Appended with Benzimidazole Rings: Tetranuclear [CoZn3] and [CoCd3] Complexes and Their Catalytic Applications. New J. Chem. 2018, 42, 9847–9856. [Google Scholar] [CrossRef]
- Khromova, O.V.; Yashkina, L.V.; Stoletova, N.V.; Maleev, V.I.; Belokon, Y.N.; Larionov, V.A. Selectivity Control in Nitroaldol (Henry) Reaction by Changing the Basic Anion in a Chiral Copper(II) Complex Based on (S)-2-Aminomethylpyrrolidine and 3,5-Di-tert-butylsalicylaldehyde. Molecules 2024, 29, 5207. [Google Scholar] [CrossRef]
- Zhang, S.; Li, X.; Li, W.; Rao, W.; Ge, D.; Shen, Z.; Chu, X. Iron(0)-Mediated Henry-Type Reaction of Bromonitromethane with Aldehydes for the Efficient Synthesis of 2-Nitro-alkan-1-ols. Chin. J. Org. Chem. 2022, 42, 235–241. [Google Scholar] [CrossRef]
- Blay, G.; Domingo, L.R.; Hernández-Olmos, V.; Pedro, J.R. New Highly Asymmetric Henry Reaction Catalyzed by CuII and a C1-Symmetric Aminopyridine Ligand, and Its Application to the Synthesis of Miconazole. Chem. Eur. J. 2008, 14, 4725–4730. [Google Scholar] [CrossRef] [PubMed]
- Nitabaru, T.; Nojiri, A.; Kobayashi, M.; Kumagai, N.; Shibasaki, M. anti-Selective Catalytic Asymmetric Nitroaldol Reaction via a Heterobimetallic Heterogeneous Catalyst. J. Am. Chem. Soc. 2009, 131, 13860–13869. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Deng, P.; Zeng, Y.; Xiong, Y.; Zhou, H. anti-Selective Asymmetric Henry Reaction Catalyzed by a Heterobimetallic Cu−Sm−Aminophenol Sulfonamide Complex. Org. Lett. 2016, 18, 1578–1581. [Google Scholar] [CrossRef] [PubMed]
- Xu, K.; Lai, G.; Zha, Z.; Pan, S.; Chen, H.; Wang, Z. A Highly anti-Selective Asymmetric Henry Reaction Catalyzed by a Chiral Copper Complex: Applications to the Syntheses of (+)-Spisulosine and a Pyrroloisoquinoline Derivative. Chem. Eur. J. 2012, 18, 12357–12362. [Google Scholar] [CrossRef]
- Qian, Y.; Zheng, X.; Wang, Y. A Green and Efficient Asymmetric Aldol Reaction Catalyzed by a Chiral Anion Modified Ionic Liquid. Eur. J. Org. Chem. 2010, 2010, 3672–3677. [Google Scholar] [CrossRef]
- Ma, G.; Bartoszewicz, A.; Ibrahem, I.; Córdova, A. Highly Enantioselective Co-Catalytic Direct Aldol Reactions by Combination of Hydrogen-Bond Donating and Acyclic Amino Acid Catalysts. Adv. Synth. Catal. 2011, 353, 3114–3122. [Google Scholar] [CrossRef]
- Toussaint, A.; Pfaltz, A. Asymmetric Henry Reactions Catalyzed by Metal Complexes of Chiral Boron-Bridged Bisoxazoline (Borabox) Ligands. Eur. J. Org. Chem. 2008, 27, 4591–4597. [Google Scholar]
- Blay, G.; Hernández-Olmos, V.; Pedro, J.R. Enantioselective Henry Addition of Methyl 4-Nitrobutyrate to Aldehydes. Chiral Building Blocks for 2-Pyrrolidinones and Other Derivatives. Org. Lett. 2010, 12, 3058–3061. [Google Scholar] [CrossRef]
- Gao, J.; Bai, S.; Gao, Q.; Liu, Y.; Yang, Q. Acid Controlled Diastereoselectivity in Asymmetric Aldol Reaction of Cycloketones with Aldehydes using Enamine-Based Organocatalysts. Chem. Commun. 2011, 47, 6716–6718. [Google Scholar] [CrossRef]
- Wang, G.-W. Mechanochemical Organic Synthesis. Chem. Soc. Rev. 2013, 42, 7668–7700. [Google Scholar] [CrossRef] [PubMed]
- Egorov, I.N.; Santra, S.; Kopchuk, D.S.; Markevich, A.V.; Gordeev, E.G.; Ananikov, V.P. Ball Milling: An Efficient and Green Approach for Asymmetric Organic Syntheses. Green Chem. 2020, 22, 3026–3050. [Google Scholar] [CrossRef]


| Entry | MeNO2 (Equiv) | Additive | Solvent (Molarity) | Time | Yield b (%) |
|---|---|---|---|---|---|
| 1 [25] | 5.0 | 4 Å MS c | DMSO (0.15) | 24 h | 91 |
| 2 [26] | 3.0 | phosphate buffer (pH = 7.0) | H2O (0.25) | 18 h | 78 |
| 3 [27] | 1.4 | NaOAc (33 mol%) | EtOH (1.35) | 18 h | 64 |
| 4 [28] | 3.0 | imidazole (25 mol%) | H2O (0.50) | 15 min | 90 |
| 5 [29] d | 4.0 | DABCO (50 mol%) | CH3CN e | 24 h | 95 |
| 6 [30] | 2.0 | cyclen (5 mol%) | THF (1.0) | 24 h | 80 |
| Entry c | Henry Nucleophile | Major Competition Product | Time (h) | anti-Aldol (3) Yield (%) d | dr e | ee f |
|---|---|---|---|---|---|---|
| 1 [43] | MeNO2 | 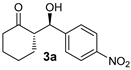 | 24 | 91 | >19:1 | 99 |
| 2 [44] | MeNO2 |  | 36 | 81 | >19:1 | 99 |
| 3 [44] | MeNO2 | 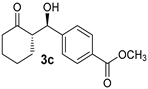 | 40 | 89 | 14:1 | 98 |
| 4 [45] | MeNO2 | 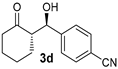 | 40 | 89 | >19:1 | 99 |
| 5 [43] |  |  | 24 | 91 | 14:1 | 99 |
| 6 [44] |  | 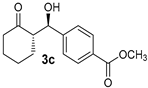 | 40 | 84 | 10:1 | 99 |
| 7 [43] |  | 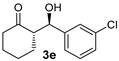 | 49 | 86 | 13:1 | 99 |
| 8 [46] g |  | 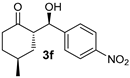 | 24 | 65 | 13:1 >19:1 h | 99 |
| 9 [47] g,i |  | 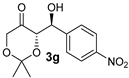 | 24 | 80 | >19:1 | 99 |
| 10 [43] j | 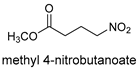 |  | 16 | 86 | >19:1 | 99 |
| Entry | Henry Nucleophile | Major Competition Product b | Chemoselectivity Aldol/ Henry c | Time (h) | Anti-Aldol Yield (%) d | dr c | ee e |
|---|---|---|---|---|---|---|---|
| 1 | 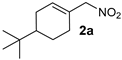 |  | >19:1 | 32 | 84 | 13:1 | 97 |
| 2 |  | 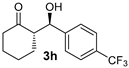 | >19:1 | 32 | 82 | 14:1 | 98 |
| 3 |  |  | >19:1 | 32 | 68 | 16:1 | 98 |
| 4 | 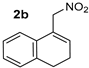 | 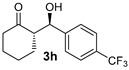 | >19:1 | 32 | 74 | 13:1 | 99 |
| 5 |  | 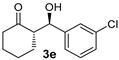 | 6.4:1 | 32 | 76 | 13:1 | 98 |
| 6 |  |  | 6.8:1 | 32 | 74 | 11:1 | 99 |
| 7 f | 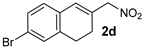 | 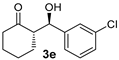 | 4.9:1 | 32 | 66 | 14:1 | 99 |
| 8 g | 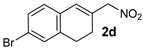 | 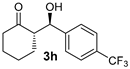 | - | - | - | - | - |
| 9 g |  | 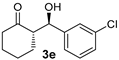 | - | - | - | - | - |
| 10 | 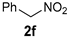 | 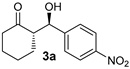 | 5.4:1 5.0:1 i | 24 9 i | 75 h 43 i | 14:1 7:1 i | 99 99 i |
| 11 |  | 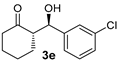 | - j | 32 | 60 | >11:1 k | 99 |
| Entry | Description | Catalyst (mol%) | Solvent | Molarity (Equiv) | Time (h) | Chemoselectivity b | 3e (Yield %) c | dr b | ee d |
|---|---|---|---|---|---|---|---|---|---|
| 1 | unfair e | 2.5 | water | 3.7 (15) | 32 | 4.9:1 | 67 | 14:1 | 98 |
| 2 | unfair e | 2.5 | neat | - | 32 | 5.62:1 | - f | 5.6:1 | - f |
| 3 | homogeneous g | 25 | DMSO | 0.5 | 8 | 2.83:1 | 45 h,i | 1.9:1 | - j |
| 4 | unfair k | 15 | THF | 0.5 | - | - | - | - | - |
| 5 | homogenous g | 20 | EtOH | 0.5 | 21 | 3.4:1 | 54 h | 2.4:1 | 86 |
| 6 | homogenous g | 5 | EtOH | 0.5 | 32 | 5.54:1 | 36 h | 4.5:1 | 94 |
| 7 | unfair e | 2.5 | EtOH | 1.14 (15) | 32 | 6.1:1 | - f | 4.5:1 | - f |
| Entry | Solvent | Molarity (Equiv) | Time (h) | Chemoselectivity b | 3e (Yield %) c | dr b | ee d |
|---|---|---|---|---|---|---|---|
| 1 e | water | 3.7 (15) | 32 | 6.8:1 | 76 | 13:1 | 98 |
| 2 | water | 3.7 (15) | 24 | 5.7:1 | 68 f | 11:1 | 99 |
| 3 | neat | - | 32 | 13:1 | 75 | 5.3:1 | 93 |
| 4 g | EtOH | 0.5 (34) | 22 | 6.1:1 | 55 f | 3.5:1 | 87 |
| 5 h | EtOH | 1.0 (17) | 40 | 8.1:1 | 40 f | 4.8:1 | 94 |
| 6 | EtOH | 1.14 (15) | 72 | 6.2:1 | 62 i | 7.7:1 | 98 |
| 7 | MeOH | 1.65 (15) | 72 | 6.2:1 | 69 i | 9.1:1 | 99 |
| 8 | MeOH/H2O | (3:2) | 60 | 7.7:1 | 77 | 13:1 | 99 |
| 9 | MeOH/H2O | (3:2) | 32 | 7.7:1 | 55 f | 13:1 | 98 |
| Entry | Solvent (Equiv) | Time (h) | Chemoselectivity b | (Yield %) c | dr b | ee 4 d |
|---|---|---|---|---|---|---|
| 1 | Water (15 equiv) | 40 | >19:1 | 3c, 84 | 3c, 10:1 | 3c, 99 |
| 2 | MeOH/H2O (3:2 equiv) | 40 | >19:1 | 3c, 76 | 3c, >19:1 | 3c, 99 |
| 3 | Water (15 equiv) | 49 | >19:1 | 3e, 86 | 3e, 13:1 | 3e, 99 |
| 4 | Water (15 equiv) | 32 | >19:1 | 3e, 74 | 3e, 13:1 | 3e, 98 |
| 5 | MeOH/H2O (3.2 equiv) | 49 | >19:1 | 3e, 82 | 3e, >19:1 | 3e, 98 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dwivedi, K.B.; Knäbe, P.; Shitole, N.N.; Lakew, A.H.; Levochkin, R.; Paredes-Soler, L.; Zhylinska, S.-S.; Kochubei, D.; Guillena, G.; Chinchilla, R.; et al. Defining Aldol Chemoselectivity in the Presence of Henry Nucleophiles (Nitroalkanes). Molecules 2025, 30, 4688. https://doi.org/10.3390/molecules30244688
Dwivedi KB, Knäbe P, Shitole NN, Lakew AH, Levochkin R, Paredes-Soler L, Zhylinska S-S, Kochubei D, Guillena G, Chinchilla R, et al. Defining Aldol Chemoselectivity in the Presence of Henry Nucleophiles (Nitroalkanes). Molecules. 2025; 30(24):4688. https://doi.org/10.3390/molecules30244688
Chicago/Turabian StyleDwivedi, Kritika B., Patrick Knäbe, Nilesh N. Shitole, Aida H. Lakew, Ruslan Levochkin, Luis Paredes-Soler, Sofiia-Stefaniia Zhylinska, Diana Kochubei, Gabriela Guillena, Rafael Chinchilla, and et al. 2025. "Defining Aldol Chemoselectivity in the Presence of Henry Nucleophiles (Nitroalkanes)" Molecules 30, no. 24: 4688. https://doi.org/10.3390/molecules30244688
APA StyleDwivedi, K. B., Knäbe, P., Shitole, N. N., Lakew, A. H., Levochkin, R., Paredes-Soler, L., Zhylinska, S.-S., Kochubei, D., Guillena, G., Chinchilla, R., Alonso, D. A., & Nugent, T. C. (2025). Defining Aldol Chemoselectivity in the Presence of Henry Nucleophiles (Nitroalkanes). Molecules, 30(24), 4688. https://doi.org/10.3390/molecules30244688












