Oyster Fermentation Broth Alleviated Tripterygium-Glycosides-Induced Reproductive Damage in Male Rats
Abstract
1. Introduction
2. Results
2.1. Component Characterization and in Vitro Antioxidant Activity of OFB
2.1.1. Characterization of Major Physicochemical Parameters and Bioactive Components in OFB
2.1.2. Characterization of Phenolic Compounds and Antioxidant Activity of OFB
2.2. Growth Parameters of Rats
2.3. Hematological Parameters
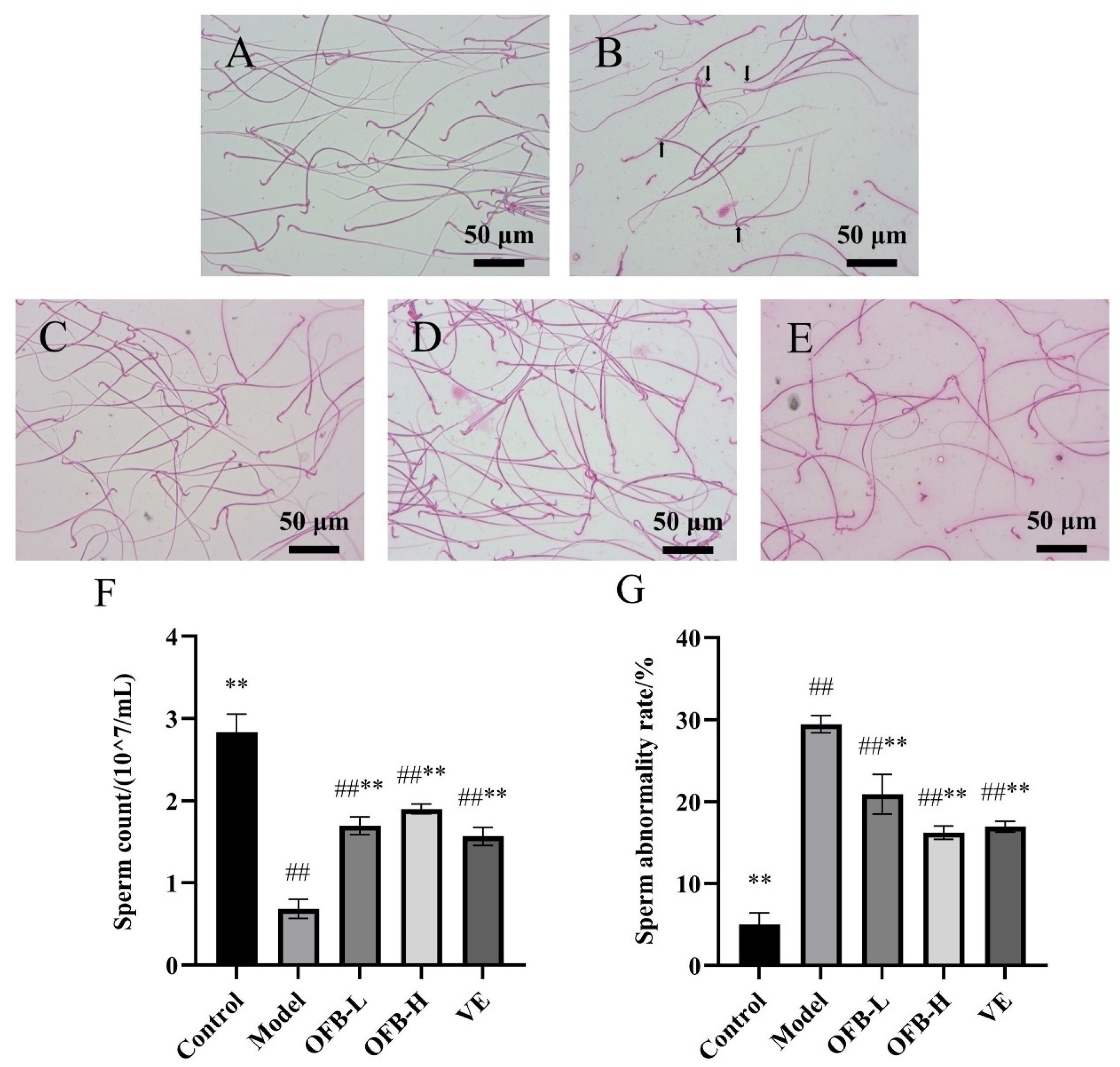
2.4. Sperm Quality Analysis
2.5. Serum Sex Hormone Levels
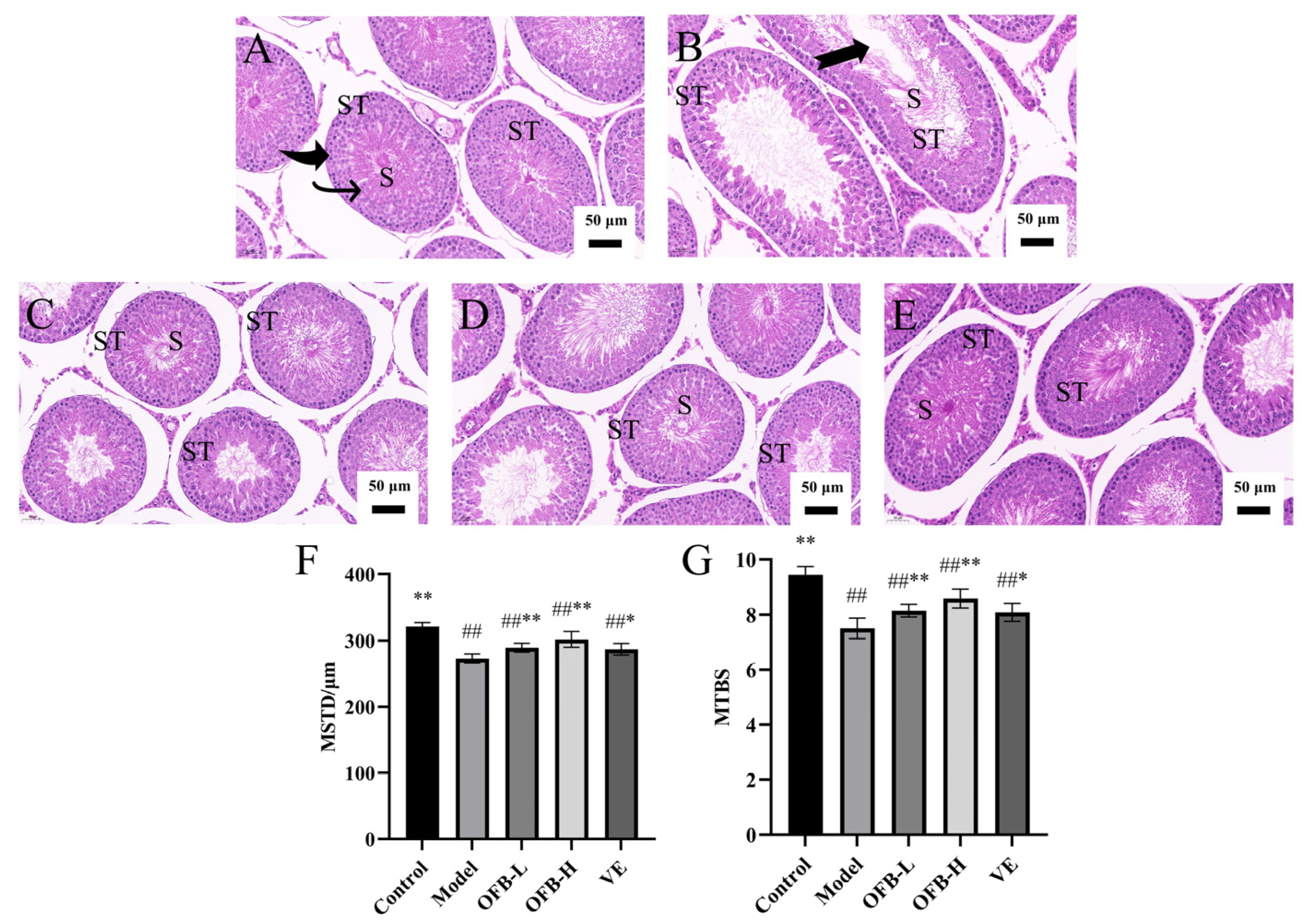
2.6. Histopathological Analysis of Testicular Tissues
2.7. Oxidative Stress Markers in Testicular Tissue
3. Discussion
4. Materials and Methods
4.1. Materials and Reagents
4.2. Instruments and Equipment
4.3. Experimental Animals
4.4. OFB Preparation Process
4.5. Experimental Methods
4.5.1. Determination of Physicochemical Properties, Nutritional Components, and Antioxidant Capacity of OFB
- (1)
- Physicochemical analysis of OFB
- (2)
- Analysis of bioactive components in OFB
- (3)
- In vitro antioxidant activity assay of OFB
- (4)
- Ultra-high-performance liquid chromatography–tandem mass spectrometry (UHPLC-MS/MS) Analysis
4.5.2. Animal Grouping and Administration
4.5.3. Blood Collection and Analysis
4.5.4. Organ Index Determination
4.5.5. Sperm Quality Assessment
4.5.6. Determination of Serum Sex Hormone Levels
4.5.7. Oxidative Stress Index Assays in Testicular Tissue
4.5.8. Histological Observation
4.5.9. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| OFB | Oyster fermentation broth |
| TG | Tripterygium glycosides |
| VE | Vitamin E |
| T | Testosterone |
| FSH | Follicle-stimulating hormone |
| LH | Luteinizing hormone |
| CAT | Catalase |
| SOD | Superoxide dismutase |
| GSH-PX | Glutathione peroxidase |
| MDA | Malondialdehyde |
| OFB-L | Oyster fermentation broth low dose |
| OFB-H | Oyster fermentation broth high dose |
| MSTD | Mean seminiferous tubule diameter |
| MTBS | Mean testicular biopsy score |
| HPLC | High-performance liquid chromatography |
| ICP-MS | Inductively coupled plasma–mass spectrometry |
| WBC | White blood cell |
| RBC | Red blood cell |
| HGB | Hemoglobin |
| LYM | Lymphocyte |
| NEU | Neutrophil |
| HPG | Hypothalamus–pituitary–gonadal |
| GnRH | Gonadotropin-releasing hormone |
| ABP | Androgen-binding protein |
References
- Jiang, S.; Liu, L.; Xu, J.; Zeng, M.; Zhao, Y. Amino Acid Composition and Digestibility of Pacific Oyster (Crassostrea gigas) Proteins Isolated from Different Parts. LWT 2019, 116, 108591. [Google Scholar] [CrossRef]
- Matsuda, Y.; Watanabe, T. Effects of Oyster Extract on the Reproductive Function of Zinc-Deficient Mice: Bioavailability of Zinc Contained in Oyster Extract. Congenit. Anom. 2004, 43, 271–279. [Google Scholar] [CrossRef]
- Siregar, A.S.; Nyiramana, M.M.; Kim, E.J.; Cho, S.B.; Woo, M.S.; Lee, D.K.; Hong, S.G.; Han, J.; Kang, S.S.; Kim, D.R.; et al. Oyster-Derived Tyr-Ala (YA) Peptide Prevents Lipopolysaccharide/D-Galactosamine-Induced Acute Liver Failure by Suppressing Inflammatory, Apoptotic, Ferroptotic, and Pyroptotic Signals. Mar. Drugs 2021, 19, 614. [Google Scholar] [CrossRef]
- Cai, B.; Wan, P.; Chen, H.; Chen, D.; Chen, X.; Sun, H.; Pan, J. Composition Characterization of Oyster Polysaccharides from Crassostrea hongkongensis and Their Protective Effect Against H2O2-Induced Oxidative Damage in IEC-6 cells. Int. J. Biol. Macromol. 2019, 124, 246–254. [Google Scholar] [CrossRef]
- Zhang, W.; Wei, Y.; Cao, X.; Guo, K.; Wang, Q.; Xiao, X.; Zhai, X.; Wang, D.; Huang, Z. Enzymatic Preparation of Crassostrea Oyster Peptides and Their Promoting Effect on Male Hormone Production. J. Ethnopharmacol. 2021, 264, 113382. [Google Scholar] [CrossRef]
- Ma, Y.; Jiang, S.; Zeng, M. In vitro Simulated Digestion and Fermentation Characteristics of Polysaccharide from Oyster (Crassostrea gigas), and Its Effects on the Gut Microbiota. Food Res. Int. 2021, 149, 110646. [Google Scholar] [CrossRef]
- Wang, X.; Yu, H.; Xing, R.; Li, P. Hepatoprotective Effect of Oyster Peptide on Alcohol-Induced Liver Disease in Mice. Int. J. Mol. Sci. 2022, 2, 8081. [Google Scholar] [CrossRef]
- Lu, X.; Shi, M.; Liu, L.; Chen, Z.; Xu, X.; Feng, G.; Zeng, M. Enhancement of Flavor Quality in Oyster Hydrolysate Through Fermentation with Oyster-Derived Lactic Acid Bacteria. Food Biosci. 2024, 62, 105231. [Google Scholar] [CrossRef]
- Choudhary, J.; Singh, S.; Nain, L. Thermotolerant Fermenting Yeasts for Simultaneous Saccharification Fermentation of Lignocellulosic Biomass. Electron. J. Biotechnol. 2016, 21, 82–92. [Google Scholar] [CrossRef]
- Rosca, I.; Petrovici, A.R.; Brebu, M.; Stoica, I.; Minea, B.; Marangoci, N. An Original Method for Producing Acetaldehyde and Diacetyl by Yeast Fermentation. Braz. J. Microbiol. 2016, 47, 949–954. [Google Scholar] [CrossRef]
- Maicas, S. The Role of Yeasts in Fermentation Processes. Microorganisms 2020, 8, 1142. [Google Scholar] [CrossRef]
- Hittinger, C.T.; Steele, J.L.; Ryder, D.S. Diverse Yeasts for Diverse Fermented Beverages and Foods. Curr. Opin. Biotechnol. 2018, 49, 199–206. [Google Scholar] [CrossRef]
- Brandão, M.; Marques, D.J.; Sousa, S.; Mateus, M.; Pinheiro, H.M.; Da Fonseca, M.M.R.; Pires, C.; Nunes, M.L.; Marques, A.; Cesário, M.T. Lactic Acid Bacteria and Yeast Fermentation to Improve the Nutritional Value of Ulva rigida. Mar. Drugs 2025, 23, 106. [Google Scholar] [CrossRef]
- Rodríguez Madrera, R.; Pando Bedriñana, R.; Suárez Valles, B. Enhancement of the Nutritional Properties of Apple Pomace by Fermentation with Autochthonous Yeasts. LWT—Food Sci. Technol. 2017, 79, 27–33. [Google Scholar] [CrossRef]
- Chen, H.; Shi, X.Q.; Cen, L.Y.; Zhang, L.; Dai, Y.F.; Qiu, S.Y.; Zeng, X.Y.; Wei, C.Y. Effect of Yeast Fermentation on the Physicochemical Properties and Bioactivities of Polysaccharides of Dendrobium officinale. Foods 2023, 12, 150. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Hwang-Bo, H.; Ji, S.Y.; Kim, M.Y.; Kim, S.Y.; Woo, M.; Keum, Y.S.; Noh, J.S.; Park, J.H.; Lee, B.J.; et al. Effect of Fermented Oyster Extract on Growth Promotion in Sprague-Dawley Rats. Integr. Med. Res. 2020, 9, 100412. [Google Scholar] [CrossRef]
- Inhorn, M.C.; Patrizio, P. Infertility around the Globe: New Thinking on Gender, Reproductive Technologies and Global Movements in the 21st Century. Hum. Reprod. Update 2015, 21, 411–426. [Google Scholar] [CrossRef]
- Aitken, R.J. The Male is Significantly Implicated as the Cause of Unexplained Infertility. Semin. Reprod. Med. 2020, 38, 3–20. [Google Scholar] [CrossRef]
- Karna, K.K.; Choi, B.R.; Kim, M.J.; Kim, H.K.; Park, J.K. The Effect of Schisandra Chinensis Baillon on Cross-Talk between Oxidative Stress, Endoplasmic Reticulum Stress, and Mitochondrial Signaling Pathway in Testes of Varicocele-Induced SD Rat. Int. J. Mol. Sci. 2019, 20, 5785. [Google Scholar] [CrossRef] [PubMed]
- Ilgin, S. The Adverse Effects of Psychotropic Drugs as an Endocrine Disrupting Chemicals on the Hypothalamic-Pituitary Regulation in Male. Life Sci. 2020, 253, 117704. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Liu, T.; Wang, H.; Zhao, Y.; Xu, X.; Zeng, M. Identification and Validation of Core Microbes for the Formation of the Characteristic Flavor of Fermented Oysters (Crassostrea gigas). Food Chem. 2024, 449, 138970. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Liu, X.; Liu, D.; Yin, Z.; Yang, X.; Zeng, M. Oyster (Crassostrea gigas) Polysaccharide Ameliorates High-Fat-Diet-Induced Oxidative Stress and Inflammation in the Liver via the Bile Acid-FXR-AMPKα Pathway. J. Agric. Food Chem. 2022, 70, 8662–8671. [Google Scholar] [CrossRef]
- Liu, L.; Liu, X.; Yang, X.; Xu, X.; Zeng, M. The Preparation, Characterization and Gastroprotective Activity of Fermented Oyster Hydrolysate. J. Food Meas. Charact. 2024, 18, 199–215. [Google Scholar] [CrossRef]
- Wang, J.; Bao, B.; Meng, F.; Deng, S.; Feng, J.; Dai, H.; Xu, H.; Zhao, Q.; Li, H.; Wang, B. In Vitro and in Vivo Investigation of the Therapeutic Mechanism of Lycium chinense and Cuscutae semen on Oligoasthenozoospermia. Andrologia 2021, 53, e14014. [Google Scholar] [CrossRef]
- Ma, J.; Tan, H.; Bi, J.; Sun, B.; Zhen, Y.; Lian, W.; Wang, S. Zinc Ameliorates Tripterygium Glycosides-Induced Reproductive Impairment in Male Rats by Regulating Zinc Homeostasis and Expression of Oxidative Stress-Related Genes. Biol. Trace Elem. Res. 2024, 202, 2111–2123. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.; Sun, B.; Te, L.G.; Huang, X.; Zuo, X.; Han, X.K.; Wang, S.S. A Dietary Supplement Jinghuosu Ameliorates Reproductive Damage Induced by Tripterygium Glycosides. Chin. J. Integr. Med. 2024, 30, 330–338. [Google Scholar] [CrossRef]
- Liu, J.; Zuo, X.; Bi, J.; Li, H.; Li, Y.; Ma, J.; Wang, S. Palliative Effect of Combined Application of Zinc and Selenium on Reproductive Injury Induced by Tripterygium Glycosides in Male Rats. Biol. Trace Elem. Res. 2024, 202, 5081–5093. [Google Scholar] [CrossRef]
- Ge, J.C.; Qian, Q.; Gao, Y.H.; Zhang, Y.F.; Li, Y.X.; Wang, X.; Fu, Y.; Ma, Y.M.; Wang, Q. Toxic Effects of Tripterygium Glycoside Tablets on the Reproductive System of Male Rats by Metabolomics, Cytotoxicity, and Molecular Docking. Phytomedicine 2023, 114, 154813. [Google Scholar] [CrossRef]
- Niu, Z.; Zhang, H.; Cai, C.; Yang, T.; Ma, T.; Xu, D.; Cui, D.; Tang, Y. The Mechanisms of Tripterygium Glycosides-Induced Reproductive Toxicity and Detoxification Strategies. Reprod. Toxicol. 2025, 132, 108830. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.Q.; Wang, B.; Ding, C.F.; Wan, L.Y.; Hu, H.M.; Lv, B.D.; Ma, J.X. In Vivo and in Vitro Protective Effects of the Wuzi Yanzong Pill Against Experimental Spermatogenesis Disorder by Promoting Germ Cell Proliferation and Suppressing Apoptosis. J. Ethnopharmacol. 2021, 280, 114443. [Google Scholar] [CrossRef]
- Li, X.; Dong, L.; Wang, J.S.; Sun, S.; Wang, B.; Li, H.S. Effects of Zuogui Wan on Testis Structure and Expression of C-Kit and Oct4 in Rats with Impaired Spermatogenesis. Pharm. Biol. 2019, 58, 44–50. [Google Scholar] [CrossRef]
- Jing, X.; Cheng, W.; Guo, S.; Zou, Y.; Zhang, T.; He, L. Toxic Effects of Tripterygium wilfordii Hook F on the Reproductive System of Adolescent Male Rats. Biomed. Pharmacother. 2017, 95, 1338–1345. [Google Scholar] [CrossRef] [PubMed]
- Jiao, W.; Sun, J.; Zhang, X.; An, Q.; Fu, L.; Xu, W.; Xie, H.; Tang, X.; Liu, J.; Hu, W.; et al. Improvement of Qilin Pills on Male Reproductive Function in Tripterygium Glycoside-Induced Oligoasthenospermia in Rats. Andrologia 2021, 53, e13923. [Google Scholar] [CrossRef] [PubMed]
- Zhou, S.H.; Ding, N.N.; Han, R.H.; Deng, Y. Metabolic Engineering and Fermentation Optimization Strategies for Producing Organic Acids of the Tricarboxylic Acid Cycle by Microbial Cell Factories. Bioresour. Technol. 2023, 379, 128986. [Google Scholar] [CrossRef]
- Chen, Y.Y.; Zeng, L.T.; Liao, Y.Y.; Li, J.L.; Zhou, B.; Yang, Z.Y.; Tang, J.C. Enzymatic Reaction-Related Protein Degradation and Proteinaceous Amino Acid Metabolism during the Black Tea (Camellia sinensis) Manufacturing Process. Foods 2020, 9, 66. [Google Scholar] [CrossRef] [PubMed]
- Acosta-Estrada, B.A.; Gutiérrez-Uribe, J.A.; Serna-Saldívar, S.O. Bound Phenolics in Foods, a Review. Food Chem. 2014, 152, 46–55. [Google Scholar] [CrossRef]
- Nguyen, N.D.; Le, M.T.; Tran, N.Q.T.; Nguyen, Q.H.V.; Cao, T.N. Micronutrient Supplements as Antioxidants in Improving Sperm Quality and Reducing DNA Fragmentation. Basic Clin. Androl. 2023, 33, 23. [Google Scholar] [CrossRef]
- Ommati, M.M.; Sabouri, S.; Retana-Marquez, S.; Nategh Ahmadi, H.; Arjmand, A.; Alidaee, S.; Mazloomi, S.; Akhlagh, A.; Abdoli, N.; Niknahad, H.; et al. Taurine Improves Sperm Mitochondrial Indices, Blunts Oxidative Stress Parameters, and Enhances Steroidogenesis and Kinematics of Sperm in Lead-Exposed Mice. Reprod. Sci. 2022, 30, 1891–1910. [Google Scholar] [CrossRef]
- Soliman, M.M.; Aldhahrani, A.; Ghamry, H.I.; Albogami, S.; Youssef, G.B.A.; Kesba, H.; Shukry, M. Chrysin Abrogates Gibberellic Acid-Induced Testicular Oxidative Stress and Dysfunction via the Regulation of Antioxidants and Steroidogenesis- and Apoptosis-Associated Genes. J. Food Biochem. 2022, 46, e14165. [Google Scholar] [CrossRef]
- Jin, Q.G.; Shi, W.T.; Wang, Y.C.; Li, S.Y.; Xue, C.; Xu, H.R.; Wu, M.T.; Wei, Y. Oyster Peptide Prevents the Occurrence of Exercise-Hypogonadal Male Condition by Improving the Function of Pituitary Gonadal Axis in Male rats. Andrologia 2021, 53, e14005. [Google Scholar] [CrossRef]
- Owumi, S.E.; Popoola, O.; Otunla, M.T.; Okuu, U.A.; Najophe, E.S. Benzo-A-Pyrene-Induced Reproductive Toxicity was Abated in Rats Co-Treated with Taurine. Toxin Rev. 2021, 41, 846–859. [Google Scholar] [CrossRef]
- Zhang, Y.F.; Yang, J.Y.; Meng, X.P.; Qiao, X.L. L-arginine Protects Against T-2 Toxin-Induced Male Reproductive Impairments in Mice. Theriogenology 2019, 126, 249–253. [Google Scholar] [CrossRef]
- Santos, H.O.; Teixeira, F.J. Use of Medicinal Doses of Zinc as a Safe and Efficient Coadjutant in the Treatment of Male Hypogonadism. Aging Male 2019, 23, 669–678. [Google Scholar] [CrossRef] [PubMed]
- Oh, S.; Choi, C.H.; Lee, B.J.; Park, J.H.; Son, K.H.; Byun, K. Fermented Oyster Extract Attenuated Dexamethasone-Induced Muscle Atrophy by Decreasing Oxidative Stress. Molecules 2021, 26, 7128. [Google Scholar] [CrossRef]
- Li, M.; Zhou, M.; Wei, Y.; Jia, F.; Yan, Y.; Zhang, R.; Cai, M.; Gu, R. The Beneficial Effect of Oyster Peptides and Oyster Powder on Cyclophosphamide-Induced Reproductive Impairment in Male Rats: A Comparative Study. J. Food Biochem. 2020, 44, e13468. [Google Scholar] [CrossRef]
- Wang, S.Y.; Yang, X.M.; Cheng, S.Z.; Du, M. The Structural Characteristics of Crassostrea gigas Peptide-Zinc Complexes and the Bioactive Mechanism of their Enhanced Zinc Absorption. Food Biosci. 2024, 58, 103722. [Google Scholar] [CrossRef]
- Zhao, C.M.; Wilkinson, K.J. Biotic Ligand Model Does not Predict the Bioavailability of Rare Earth Elements in the Presence of Organic Ligands. Environ. Sci. Technol. 2015, 49, 2207–2214. [Google Scholar] [CrossRef]
- Ma, B.; Zhang, J.; Zhu, Z.; Bao, X.; Zhang, M.; Ren, C.; Zhang, Q. Aucubin, a Natural Iridoid Glucoside, Attenuates Oxidative Stress-Induced Testis Injury by Inhibiting JNK and CHOP Activation via Nrf2 Up-Regulation. Phytomedicine 2019, 64, 153057. [Google Scholar] [CrossRef]
- Karagüzel, E.; Kutlu, Ö.; Yuluğ, E.; Mungan, S.; Kazaz, İ.O.; Tok, D.S.; Özgür, G.K. Comparison of the Protective Effect of Dipyridamole and Acetylsalicylic Acid on Long-Term Histologic Damage in a Rat Model of Testicular Ischemia-Reperfusion Injury. J. Pediatr. Surg. 2012, 47, 1716–1723. [Google Scholar] [CrossRef]
- Zhang, Y.; Yang, Y.; Tao, Y.; Guo, X.; Cui, Y.; Li, Z. Phthalates (PAEs) and Reproductive Toxicity: Hypothalamic-Pituitary-Gonadal (HPG) Axis Aspects. J. Hazard. Mater. 2023, 459, 132182. [Google Scholar] [CrossRef] [PubMed]
- Banihani, S.A. Ginger and Testosterone. Biomolecules 2018, 8, 119. [Google Scholar] [CrossRef] [PubMed]
- Lafuente, A.; Cabaleiro, T.; Caride, A.; Esquifino, A.I. Toxic Effects of Methoxychlor Administered Subcutaneously on the Hypothalamic-Pituitary-Testicular Axis in Adult rats. Food Chem. Toxicol. 2008, 46, 1570–1575. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.Y.; Yan, Z.; Liu, S.; Liu, A.Q.; Sun, J.S. Effects of Enzymatic Hydrolysate of Oyster on Erectile Function in Male Hemicastrated Rats. Int. J. Pept. Res. Ther. 2020, 26, 2001–2007. [Google Scholar] [CrossRef]
- He, Z.B.; Yin, G.C.; Li, Q.Q.; Zeng, Q.F.; Duan, J.L. Diabetes Mellitus Causes Male Reproductive Dysfunction: A Review of the Evidence and Mechanisms. In Vivo 2021, 35, 2503–2511. [Google Scholar] [CrossRef]
- Xie, Q.; Kang, Y.; Zhang, C.; Xie, Y.; Wang, C.; Liu, J.; Yu, C.; Zhao, H.; Huang, D. The Role of Kisspeptin in the Control of the Hypothalamic-Pituitary-Gonadal Axis and Reproduction. Front. Endocrinol. 2022, 13, 925206. [Google Scholar] [CrossRef]
- Chiang, Y.F.; Chen, Y.T.; Huang, K.C.; Hung, W.L.; Chung, C.P.; Shieh, T.M.; Huang, Y.J.; Ali, M.; Hsia, S.M. Protective Effects of L-Cysteine Against Cisplatin-Induced Oxidative Stress-Mediated Reproductive Damage. Antioxidants 2024, 13, 1443. [Google Scholar] [CrossRef]
- Shafie, N.F.S.; Kabir, N.B.N.; Azlina, A.A.N.; Razif, D.; Hashim, N.H. Protective Role of Cocos nucifera L. Water on BPA-Mediated Oxidative Stress and Reproductive Damage in Male Rats. Int. J. Environ. Health Res. 2025, 28, 1–13. [Google Scholar] [CrossRef]
- Fu, X.; Jing, Y.; Yang, R.; Zhu, C.; Tu, Y.; Hu, Z.; Sheng, W.; Jiao, Q.; Liu, L.; Zhang, W.; et al. Guhan Yangsheng Jing Mitigates Oxidative Stress and Ferroptosis to Improve Reproductive Damage in Diabetic Male Rats. J. Ethnopharmacol. 2025, 347, 119746. [Google Scholar] [CrossRef]
- Chen, H.W.; Liu, M.Q.; Zhang, G.Z.; Zhang, C.Y.; Wang, Z.H.; Lin, A.X.; Kang, J.H.; Liu, W.Z.; Guo, X.D.; Wang, Y.D.; et al. Proanthocyanidins Inhibit the Apoptosis and Aging of Nucleus Pulposus Cells Through the PI3K/Akt Pathway Delaying Intervertebral Disc Degeneration. Connect. Tissue Res. 2022, 63, 650–662. [Google Scholar] [CrossRef]
- Liu, H.; Ni, Y.; Yu, Q.; Fan, L. Evaluation of Co-Fermentation of L. Plantarum and P. kluyveri of a Plant-Based Fermented Beverage: Physicochemical, Functional, and Sensory Properties. Food Res. Int. 2023, 172, 113060. [Google Scholar] [CrossRef] [PubMed]
- Neupane, P.; Lamichhane, J. Estimation of Total Phenolic Content, Total Flavonoid Content and Antioxidant Capacities of Five Medicinal Plants from Nepal. Vegetos 2020, 33, 360–366. [Google Scholar] [CrossRef]
- Bibi, N.; Shah, M.H.; Khan, N.; Al-Hashimi, A.; Elshikh, M.S.; Iqbal, A.; Ahmad, S.; Abbasi, A.M. Variations in Total Phenolic, Total Flavonoid Contents, and Free Radicals’ Scavenging Potential of Onion Varieties Planted Under Diverse Environmental Conditions. Plants 2022, 11, 950. [Google Scholar] [CrossRef]
- Asem, N.; Abdul Gapar, N.A.; Abd Hapit, N.H.; Omar, E.A. Correlation Between Total Phenolic and Flavonoid Contents with Antioxidant Activity of Malaysian Stingless Bee Propolis Extract. J. Apic. Res. 2019, 29, 103403. [Google Scholar] [CrossRef]
- Lan, Y.; Wu, J.; Wang, X.; Sun, X.; Hackman, R.M.; Li, Z.; Feng, X. Evaluation of Antioxidant Capacity and Flavor Profile Change of Pomegranate Wine During Fermentation and Aging Process. Food Chem. 2017, 232, 777–787. [Google Scholar] [CrossRef]
- Wang, T.; Wang, Z.; Yang, Z.P.; Cui, X.; Yan, L.; Xu, Z.S.; Liu, X.L. Effect of the Fermentation Broth of the Mixture of Pueraria lobata, Lonicera japonica, and Crataegus pinnatifida by Lactobacillus rhamnosus 217-1 on Liver Health and Intestinal Flora in Mice with Alcoholic Liver Disease Induced by Liquor. Front. Microbiol. 2021, 12, 722171. [Google Scholar] [CrossRef] [PubMed]
- Yuan, D.X.; Shen, K.N.; Dong, X.X.; Yang, Y.; Wang, B.H.; Feng, X.J.; Li, F.; Zhao, X.R.; Tian, F.Z.; Quan, J.R.; et al. Selenium Improves Arsenic-Induced Male Reproductive Dysfunction by Regulating H3K14ac Level. Biol. Trace Elem. Res. 2025, 203, 3159–3170. [Google Scholar] [CrossRef]
- Johnsen, S.G. Testicular Biopsy Score Count-A Method for Registration of Spermatogenesis in Human Testes: Normal Values and Results in 335 Hypogonadal Males. Hormones 2008, 1, 2–25. [Google Scholar] [CrossRef] [PubMed]



| Test Parameters | Test Indicators | Pre-Fermentation | OFB |
|---|---|---|---|
| Physicochemical properties | Total acidity/(g/L) | 1.36 ± 0.07 b | 4.27 ± 0.19 a |
| Reducing sugars/(g/L) | 60.64 ± 0.81 a | 2.54 ± 0.14 b | |
| Protein content/(g/L) | 1.59 ± 1.86 a | 1.19 ± 1.15 b | |
| Taurine/(g/L) | 0.74 ± 0.03 a | 0.76 ± 0.03 a | |
| Reducing sugar composition | Glucose/(g/L) | 19.73 | 0.28 |
| Fructose/(g/L) | 34.17 | 2.12 | |
| Hydrolyzed glucose/(g/L) | 9.13 | 0.93 | |
| Mineral element content/(mg/L) | Zn2+ | 0.70 | 1.10 |
| K+ | 60.00 | 71.00 | |
| Na+ | 25.00 | 31.00 | |
| Ca2+ | 7.00 | 9.00 | |
| P5+ | 17.00 | 9.00 | |
| Total | 109.70 | 121.10 | |
| Organic acid content/(μg/mL) | Oxalic acid | 13.19 | 0.23 |
| Malic acid | 4.22 | 445.86 | |
| Lactic acid | 11.96 | 272.33 | |
| Fumaric acid | 0.13 | 4.37 | |
| Succinic acid | 100.82 | 1528.30 | |
| Citric acid | 13.02 | 52.56 | |
| Total | 143.34 | 2303.66 | |
| Amino acid content/(mg/L) | Lysine * | 6.11 | 20.72 |
| Phenylalanine * | 5.39 | 7.80 | |
| Methionine * | 1.45 | 1.64 | |
| Threonine * | 17.12 | 6.30 | |
| Isoleucine * | 15.13 | 29.30 | |
| Leucine * | 32.31 | 53.50 | |
| Valine * | 8.73 | 18.00 | |
| Histidine * | 4.16 | 7.80 | |
| Arginine * | 39.25 | 41.92 | |
| Serine | 5.53 | 14.20 | |
| Glycine | 14.54 | 18.10 | |
| Aspartic acid | 60.16 | 94.54 | |
| Glutamic acid | 76.94 | 89.70 | |
| Alanine | 14.35 | 13.20 | |
| Proline | 20.35 | 5.20 | |
| Tyrosine | 2.55 | 5.41 | |
| Cystine | 5.54 | 6.87 | |
| Essential amino acids | 129.65 | 186.98 | |
| Non-essential amino acids | 199.96 | 247.22 | |
| Total | 329.61 | 434.20 |
| Test Parameters | Pre-Fermentation | OFB |
|---|---|---|
| Polyphenol/(mg/L GAE) | 89.42 ± 5.91 b | 123.00 ± 1.46 a |
| Flavone/(mg/L RE) | 1.19 ± 0.14 b | 1.97 ± 0.17 a |
| Total antioxidant capacity/(U/mL) | 0.86 ± 0.13 b | 1.28 ± 0.09 a |
| DPPH scavenging activity/% | 41.20 ± 3.72 b | 55.80 ± 0.68 a |
| ABTS scavenging activity/% | 54.95 ± 6.60 b | 69.54 ± 2.35 a |
| OH- scavenging activity/% | 14.77 ± 1.74 b | 48.36 ± 1.20 a |
| Phenolic Compounds | RT | Formula | CAS | Content/(mg/L) | |
|---|---|---|---|---|---|
| Pre-Fermentation | OFB | ||||
| 3,4-dihydroxyphenylpyruvic acid | 0.756 | C9H8O5 | 4228-66-4 | ND | 0.98 |
| Caffeic acid | 0.803 | C9H8O4 | 331-39-5 | 1.74 | 0.18 |
| Pyrogallol | 1.344 | C6H6O3 | 87-66-1 | ND | 1.44 |
| Xanthurenic acid | 4.722 | C10H7NO4 | 59-00-7 | 3.20 | 1.16 |
| Clove3 | 5.054 | C16H18O9 | 152041-16-2 | 0.39 | ND |
| Salicylic acid | 5.216 | C7H6O3 | 69-72-7 | ND | 1.44 |
| Isoliquiritigenin | 5.624 | C15H12O4 | 961-29-5 | ND | 0.03 |
| Robinin | 6.094 | C33H40O19 | 301-19-9 | 0.04 | 0.02 |
| 4-Hydroxybenzaldehyde | 6.121 | C7H6O2 | 123-08-0 | ND | 0.40 |
| Quercetin | 9.104 | C15H10O7 | 117-39-5 | 7.07 | 4.15 |
| Naringenin | 9.988 | C15H12O5 | 480-41-1 | ND | 0.32 |
| Groups | WBC/(109/L) | RBC/(1012/L) | HGB/(g/L) | LYM/% | NEU/% |
|---|---|---|---|---|---|
| Control | 7.45 ± 0.34 | 7.44 ± 0.61 | 151.80 ± 5.89 | 70.62 ± 2.99 ** | 22.67 ± 1.10 * |
| Model | 7.41 ± 0.42 | 7.23 ± 0.53 | 150.20 ± 5.85 | 66.87 ± 2.16 ## | 19.80 ± 1.87 # |
| OFB-L | 7.68 ± 0.34 | 8.05 ± 0.49 #** | 154.00 ± 3.46 | 71.46 ± 1.23 ** | 19.43 ± 0.98 ## |
| OFB-M | 7.81 ± 0.30 | 8.32 ± 0.32 ##** | 164.60 ± 5.77 #** | 72.12 ± 0.85 ** | 22.73 ± 1.18 * |
| VE | 8.24 ± 0.35 ##** | 7.86 ± 0.30 * | 149.20 ± 4.82 | 70.42 ± 1.24 ** | 22.57 ± 1.18 * |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yin, J.; Zhu, H.; Tian, Y.; Ma, T.; Yan, W.; Sun, H. Oyster Fermentation Broth Alleviated Tripterygium-Glycosides-Induced Reproductive Damage in Male Rats. Molecules 2025, 30, 3550. https://doi.org/10.3390/molecules30173550
Yin J, Zhu H, Tian Y, Ma T, Yan W, Sun H. Oyster Fermentation Broth Alleviated Tripterygium-Glycosides-Induced Reproductive Damage in Male Rats. Molecules. 2025; 30(17):3550. https://doi.org/10.3390/molecules30173550
Chicago/Turabian StyleYin, Jiajia, Hongguang Zhu, Yu Tian, Tengyu Ma, Wenjing Yan, and Haixin Sun. 2025. "Oyster Fermentation Broth Alleviated Tripterygium-Glycosides-Induced Reproductive Damage in Male Rats" Molecules 30, no. 17: 3550. https://doi.org/10.3390/molecules30173550
APA StyleYin, J., Zhu, H., Tian, Y., Ma, T., Yan, W., & Sun, H. (2025). Oyster Fermentation Broth Alleviated Tripterygium-Glycosides-Induced Reproductive Damage in Male Rats. Molecules, 30(17), 3550. https://doi.org/10.3390/molecules30173550






