Unveiling the Catalytic Pathway of Rh(II)/Silicalite-2 in Propene Carbonylation to Methyl Butyrate: A DFT Study
Abstract
1. Introduction
2. Results and Discussion
2.1. Adsorption Test
2.2. The Reaction Mechanism of Hydroesterification of Propene
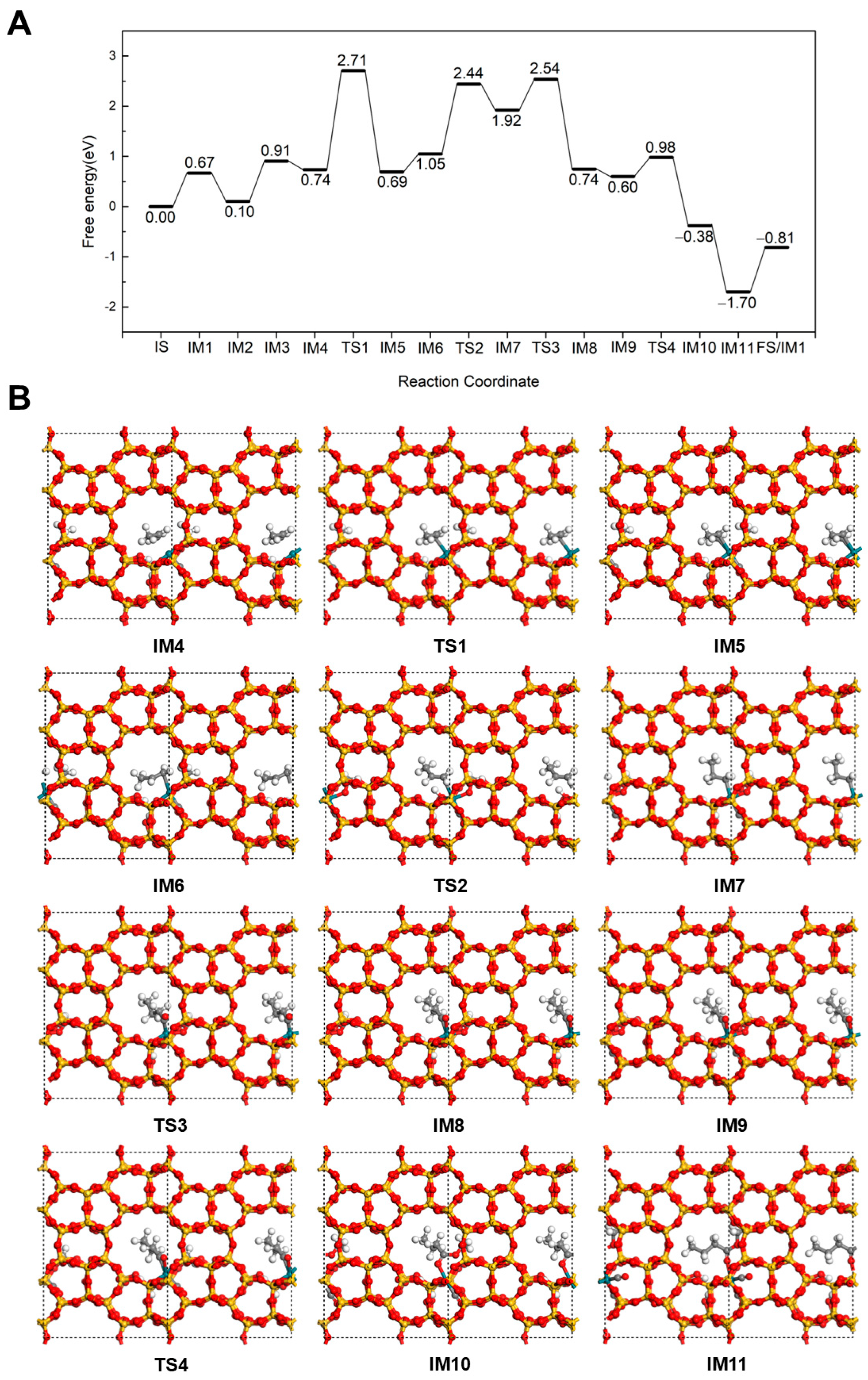
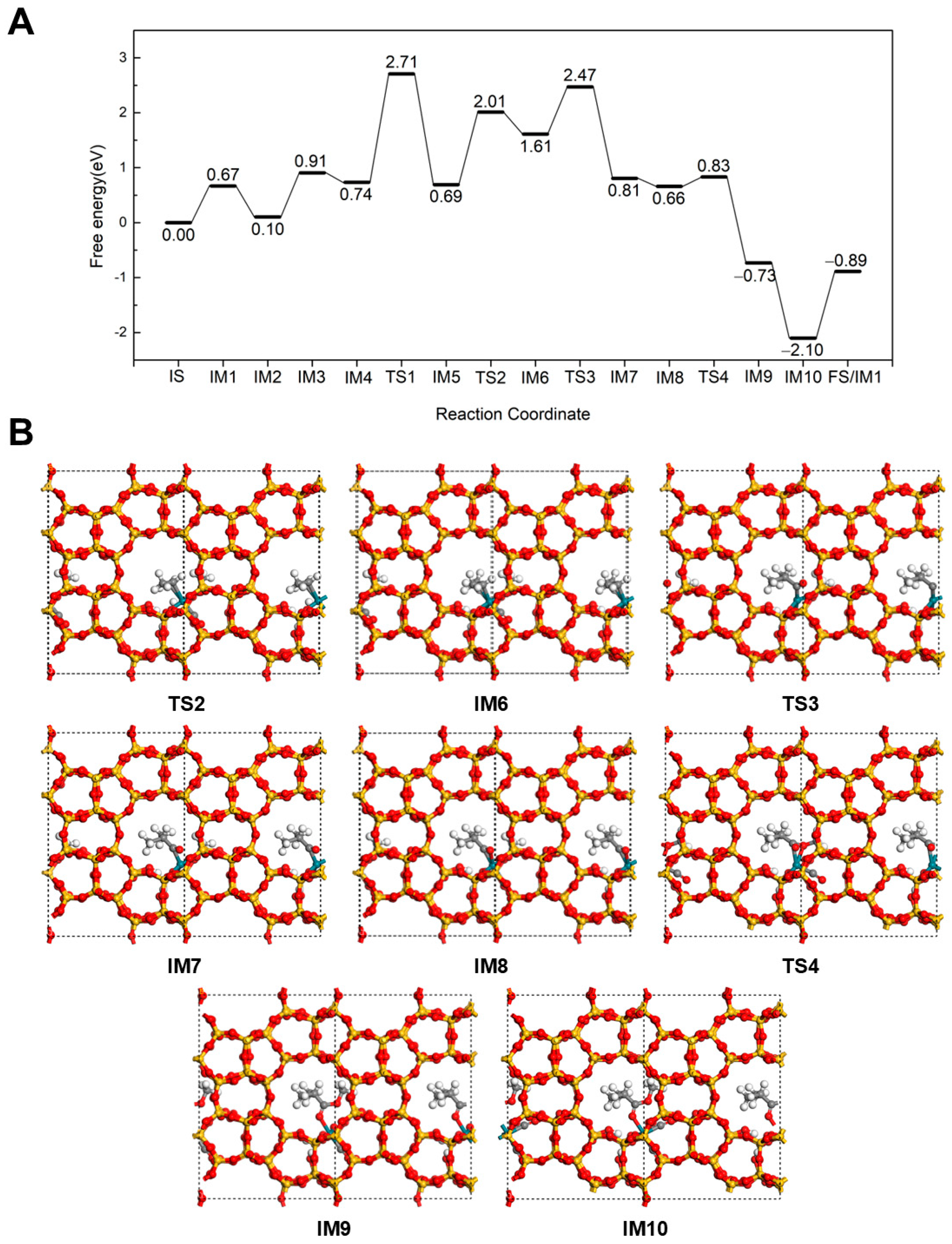
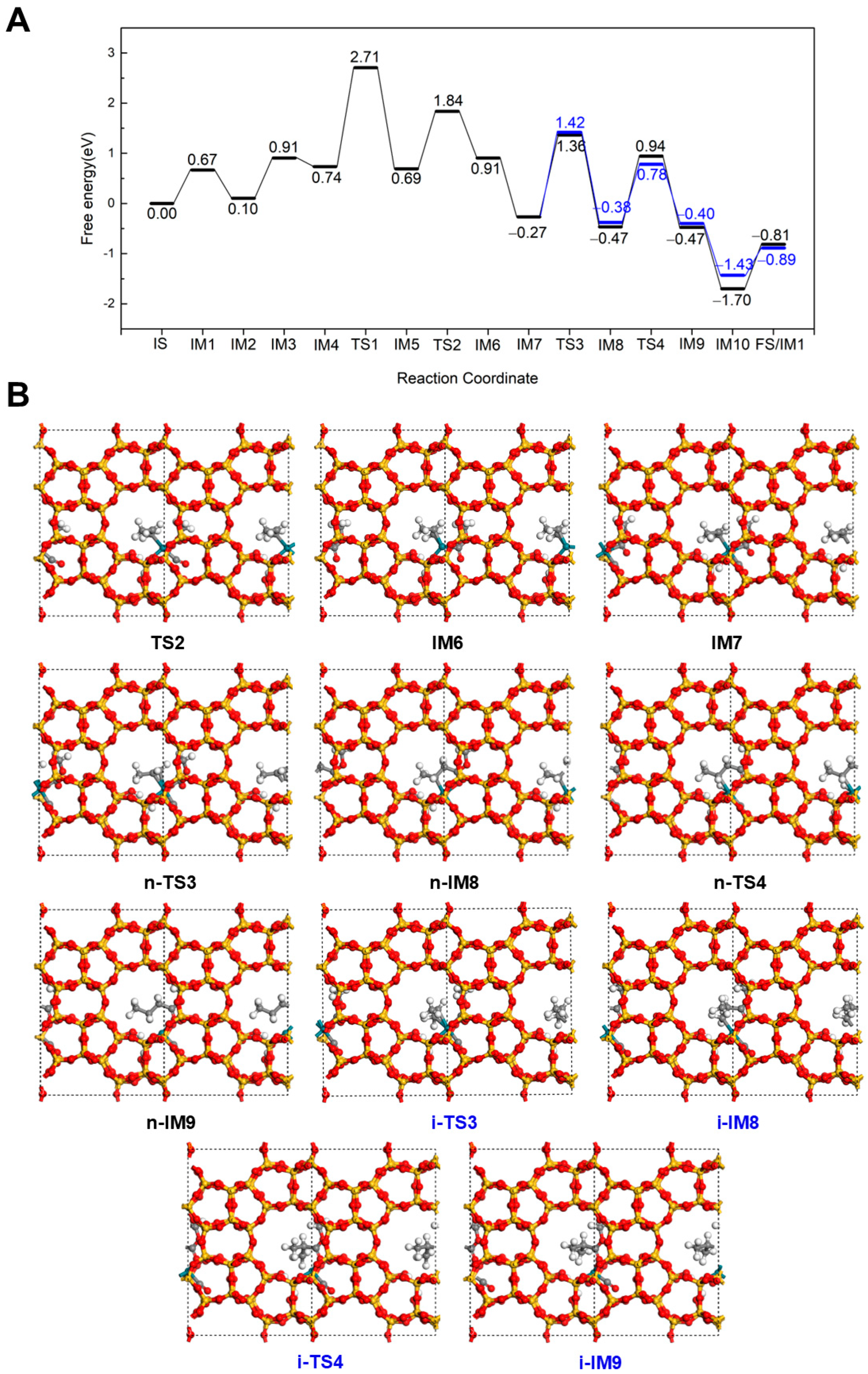
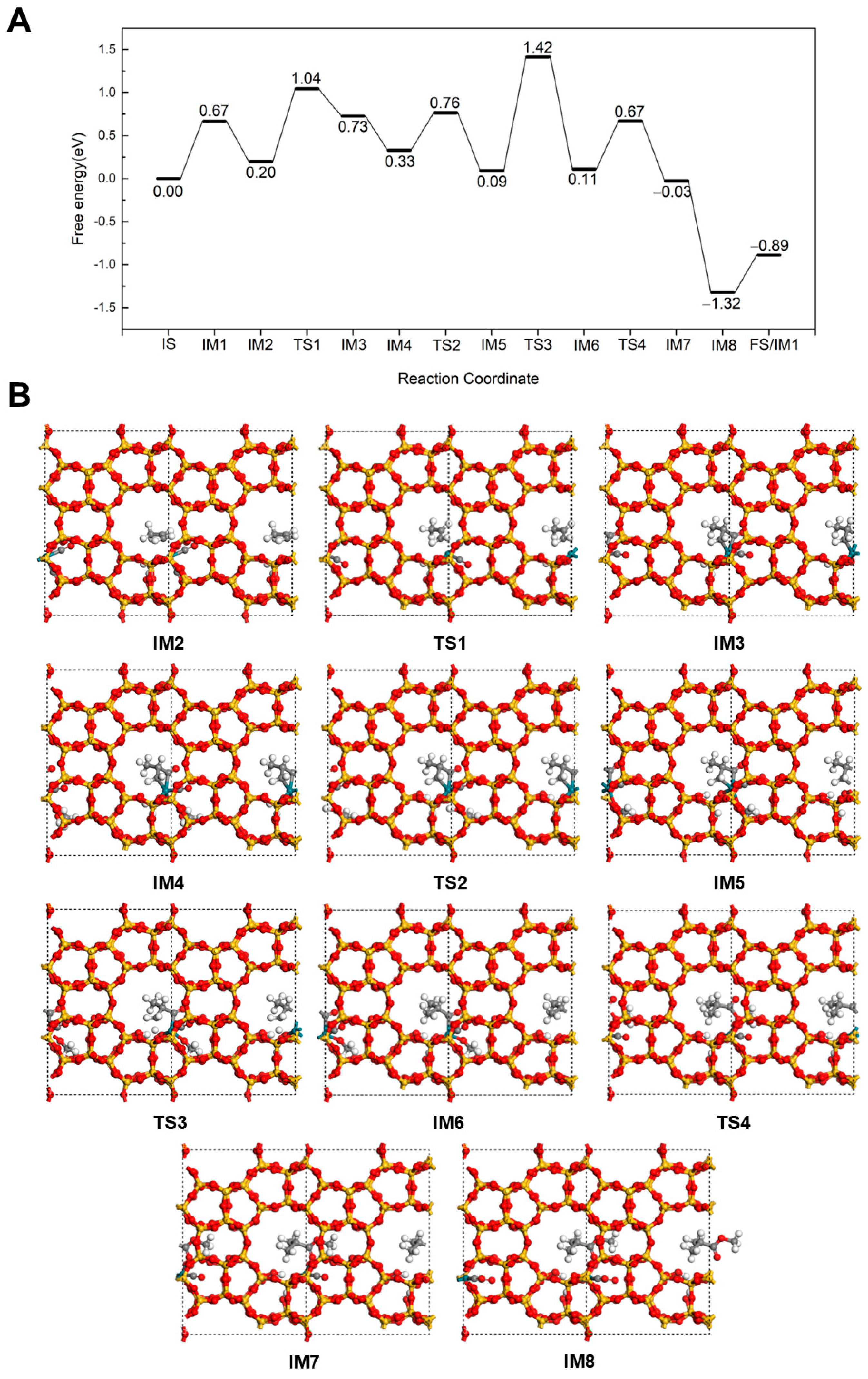
2.2.1. Hydrogenation-First Pathway for Methyl Butyrate and Methyl Isobutyrate Formation
2.2.2. Methoxycarbonyl-Driven Pathway for Methyl Butyrate and Methyl Isobutyrate Formation
2.2.3. Carbonylation-First Pathway for Methyl Butyrate and Methyl Isobutyrate Formation
2.3. Discussion
3. Calculation Method
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Colquhoun, H.M.; Thompson, D.J.; Twigg, M.V. Carbonylation: Direct Synthesis of Carbonyl Compounds; Plenum Press: New York, NY, USA, 1991. [Google Scholar]
- Das, D.; Bhanage, B.M. Double Carbonylation Reactions: Overview and Recent Advances. Adv. Synth. Catal. 2020, 362, 3022–3058. [Google Scholar] [CrossRef]
- Beller, M.; Dixneuf, P.H.; Dupont, J.; Fürstner, A.; Glorius, F.; Gooßen, L.J.; Nolan, S.P.; Okuda, J.; Oro, L.A.; Willis, M.; et al. Hydroformylation. In Topics in Organometallic Chemistry; Springer Nature: Heidelberg, Germany, 2006; pp. 1–33. [Google Scholar]
- Kollár, L. Modern Carbonylation Methods, 1st ed.; John Wiley & Sons: Hoboken, NJ, USA, 2008. [Google Scholar]
- Brennführer, A.; Neumann, H.; Beller, M. Palladium-Catalyzed Carbonylation Reactions of Aryl Halides and Related Compounds. Angew. Chem. Int. Ed. 2009, 48, 4114–4133. [Google Scholar] [CrossRef]
- Wang, L.-C.; Wu, X.-F. Single-Electron-Transfer-Mediated Carbonylation Reactions. Acc. Chem. Res. 2025, 58, 1036–1050. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Neumann, H.; Beller, M. Synthesis of α,β-Unsaturated Carbonyl Compounds by Carbonylation Reactions. Chem. Soc. Rev. 2020, 49, 3187–3210. [Google Scholar] [CrossRef]
- Wu, X.-F.; Fang, X.; Wu, L.; Jackstell, R.; Neumann, H.; Beller, M. Transition-Metal-Catalyzed Carbonylation Reactions of Olefins and Alkynes: A Personal Account. Acc. Chem. Res. 2014, 47, 1041–1053. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Kubis, C.; Franke, R. Hydroformylation Catalyzed by Unmodified Cobalt Carbonyl under Mild Conditions. Science 2022, 377, 1223–1227. [Google Scholar] [CrossRef]
- Broussard, M.E.; Juma, B.; Train, S.G.; Peng, W.-J.; Laneman, S.A.; Stanley, G.G. A Bimetallic Hydroformylation Catalyst: High Regioselectivity and Reactivity Through Homobimetallic Cooperativity. Science 1993, 260, 1784–1788. [Google Scholar] [CrossRef] [PubMed]
- Roth, T.F.H.; Spiekermann, M.L.; Lütkenhaus, D.; Niefer, F.; Vogt, D.; Seidensticker, T. The Effect of Polyunsaturation—Insights into the Hydroformylation of Oleochemicals. Catal. Sci. Technol. 2024, 14, 5551–5558. [Google Scholar] [CrossRef]
- Chakrabortty, S.; Almasalma, A.A.; De Vries, J.G. Recent Developments in Asymmetric Hydroformylation. Catal. Sci. Technol. 2021, 11, 5388–5411. [Google Scholar] [CrossRef]
- Pospech, J.; Fleischer, I.; Franke, R.; Buchholz, S.; Beller, M. Alternative Metals for Homogeneous Catalyzed Hydroformylation Reactions. Angew. Chem. Int. Ed. 2013, 52, 2852–2872. [Google Scholar] [CrossRef]
- Breit, B. Synthetic Aspects of Stereoselective Hydroformylation. Acc. Chem. Res. 2003, 36, 264–275. [Google Scholar] [CrossRef]
- Pruett, R.L. Hydroformylation. In Advances in Organometallic Chemistry; Elsevier: Amsterdam, The Netherlands, 1979; Volume 17, pp. 1–60. [Google Scholar]
- Franke, R.; Selent, D.; Börner, A. Applied Hydroformylation. Chem. Rev. 2012, 112, 5675–5732. [Google Scholar] [CrossRef]
- Pourzolfaghar, H.; Abnisa, F.; Daud, W.M.A.W.; Aroua, M.K. A Review of the Enzymatic Hydroesterification Process for Biodiesel Production. Renew. Sustain. Energy Rev. 2016, 61, 245–257. [Google Scholar] [CrossRef]
- Li, J.; Shi, Y. Progress on Transition Metal Catalyzed Asymmetric Hydroesterification, Hydrocarboxylation, and Hydroamidation Reactions of Olefins. Chem. Soc. Rev. 2022, 51, 6757–6773. [Google Scholar] [CrossRef]
- Ko, S.; Na, Y.; Chang, S. A Novel Chelation-Assisted Hydroesterification of Alkenes via Ruthenium Catalysis. J. Am. Chem. Soc. 2002, 124, 750–751. [Google Scholar] [CrossRef]
- Tian, D.; Xu, R.; Zhu, J.; Huang, J.; Dong, W.; Claverie, J.; Tang, W. Asymmetric Hydroesterification of Diarylmethyl Carbinols. Angew. Chem. 2021, 133, 6375–6379. [Google Scholar] [CrossRef]
- Mowla, O.; Kennedy, E.; Stockenhuber, M. Hydroesterification of Bio-Oils over HZSM-5, BETA and Y Zeolites. Clean. Techn Env. Environ. Policy 2018, 20, 727–738. [Google Scholar] [CrossRef]
- Ketzer, F.; Wancura, J.H.C.; Tres, M.V.; De Oliveira, J.V. Kinetic and Thermodynamic Study of Enzymatic Hydroesterification Mechanism to Fatty Acid Methyl Esters Synthesis. Bioresour. Technol. 2022, 356, 127335. [Google Scholar] [CrossRef]
- Leng, L.; Ready, J.M. Hydroesterification and Difunctionalization of Olefins with N-Hydroxyphthalimide Esters. ACS Catal. 2021, 11, 13714–13720. [Google Scholar] [CrossRef]
- Wang, M.-M.; Lu, S.-M.; Li, C. Regioselective Hydroesterification of Alkenes and Alkenylphenols Utilizing CO2 and Hydrosilane. Chem. Sci. 2023, 14, 5483–5489. [Google Scholar] [CrossRef]
- Liu, D.; Zhang, L.; Cheng, J.; Wei, Q.; Jia, Z.; Chen, F.-E. Recyclable Picolinamide-Derived Ligand-Controlled Branched-Selective Hydroesterification of Alkynes with Alcohols and Phenols. Green Chem. 2024, 26, 9690–9696. [Google Scholar] [CrossRef]
- Folster, C.P.; Harkins, R.P.; Lo, S.-Y.; Sachs, J.D.; Tonks, I.A. Development and Applications of Selective Hydroesterification Reactions. Trends Chem. 2021, 3, 469–484. [Google Scholar] [CrossRef]
- Mirbach, M.F.; Mirbach, M.J. Reactions of Cobalt Carbonyls in Methanol under High Pressure of Carbon Monoxide: A Reexamination of the Hydroesterification Mechanism. J. Mol. Catal. 1985, 32, 59–75. [Google Scholar] [CrossRef]
- Jin, H.; Cao, Q.; Wu, D. A RuCl3 Complex Catalytic System for Hydroesterification of Ethylene, Carbon Monoxide, and Ethanol. Appl. Organom. Chemis 2024, 38, e7351. [Google Scholar] [CrossRef]
- Katafuchi, Y.; Fujihara, T.; Iwai, T.; Terao, J.; Tsuji, Y. Palladium-Catalyzed Hydroesterification of Alkynes Employing Aryl Formates without the Use of External Carbon Monoxide. Adv. Synth. Catal. 2011, 353, 475–482. [Google Scholar] [CrossRef]
- Rudnick, L.R. Synthetics, Mineral Oils, and Bio-Based Lubricants: Chemistry and Technology, 3rd ed.; CRC Press: Boca Raton, FL, USA, 2020. [Google Scholar]
- Stratford, M.; Eklund, T. Organic Acids and Esters. In Food Preservatives; Springer: New York, NY, USA, 2003; pp. 48–84. [Google Scholar]
- Zheng, X.; Bao, Y.; Qu, D.; Liu, Y.; Qin, G. Measurement and Modeling of Thermal Conductivity for Short Chain Methyl Esters: Methyl Butyrate and Methyl Caproate. J. Chem. Thermodyn. 2021, 159, 106486. [Google Scholar] [CrossRef]
- Dange, P.N.; Sharma, A.; Rathod, V.K. Synthesis of Methyl Butyrate Using Heterogeneous Catalyst: Kinetic Studies. Catal. Lett. 2014, 144, 1537–1546. [Google Scholar] [CrossRef]
- Colombo, R.; Moroni, G.; Negri, C.; Delen, G.; Monai, M.; Donazzi, A.; Weckhuysen, B.M.; Maestri, M. Surface Carbon Formation and Its Impact on Methane Dry Reforming Kinetics on Rhodium-Based Catalysts by Operando Raman Spectroscopy. Angew. Chem. Int. Ed. 2024, 63, e202408668. [Google Scholar] [CrossRef]
- Galletti, C.; Specchia, S.; Saracco, G.; Specchia, V. Catalytic Performance of Rhodium-Based Catalysts for CO Preferential Oxidation in H2-Rich Gases. Ind. Eng. Chem. Res. 2008, 47, 5304–5312. [Google Scholar] [CrossRef]
- Suvarna, M.; Preikschas, P.; Pérez-Ramírez, J. Identifying Descriptors for Promoted Rhodium-Based Catalysts for Higher Alcohol Synthesis via Machine Learning. ACS Catal. 2022, 12, 15373–15385. [Google Scholar] [CrossRef] [PubMed]
- Chambers, M.B.; Wang, X.; Elgrishi, N.; Hendon, C.H.; Walsh, A.; Bonnefoy, J.; Canivet, J.; Quadrelli, E.A.; Farrusseng, D.; Mellot-Draznieks, C.; et al. Photocatalytic Carbon Dioxide Reduction with Rhodium-based Catalysts in Solution and Heterogenized within Metal–Organic Frameworks. Chem. Sus. Chem. 2015, 8, 603–608. [Google Scholar] [CrossRef]
- Azpíroz, R.; Di Giuseppe, A.; Urriolabeitia, A.; Passarelli, V.; Polo, V.; Pérez-Torrente, J.J.; Oro, L.A.; Castarlenas, R. Hydride–Rhodium(III)-N-Heterocyclic Carbene Catalyst for Tandem Alkylation/Alkenylation via C–H Activation. ACS Catal. 2019, 9, 9372–9386. [Google Scholar] [CrossRef]
- Dingwall, P.; Fuentes, J.A.; Crawford, L.E.; Slawin, A.M.Z.; Bühl, M.; Clarke, M.L. Understanding a Hydroformylation Catalyst That Produces Branched Aldehydes from Alkyl Alkenes. J. Am. Chem. Soc. 2017, 139, 15921–15932. [Google Scholar] [CrossRef] [PubMed]
- Dangat, Y.; Popli, S.; Sunoj, R.B. Unraveling the Importance of Noncovalent Interactions in Asymmetric Hydroformylation Reactions. J. Am. Chem. Soc. 2020, 142, 17079–17092. [Google Scholar] [CrossRef]
- McNamara, B.K.; Yeston, J.S.; Bergman, R.G.; Moore, C.B. The Effect of Alkane Structure on Rates of Photoinduced C−H Bond Activation by Cp*Rh(CO)2 in Liquid Rare Gas Media: An Infrared Flash Kinetics Study. J. Am. Chem. Soc. 1999, 121, 6437–6443. [Google Scholar] [CrossRef]
- Brezny, A.C.; Landis, C.R. Development of a Comprehensive Microkinetic Model for Rh(Bis(Diazaphospholane))-Catalyzed Hydroformylation. ACS Catal. 2019, 9, 2501–2513. [Google Scholar] [CrossRef]
- Hou, S.-H.; Yu, X.; Zhang, R.; Wagner, C.; Dong, G. Rhodium-Catalyzed Diastereo- and Enantioselective Divergent Annulations between Cyclobutanones and 1,5-Enynes: Rapid Construction of Complex C(Sp3)-Rich Scaffolds. J. Am. Chem. Soc. 2022, 144, 22159–22169. [Google Scholar] [CrossRef]
- Suhr, S.; Walter, R.; Beerhues, J.; Albold, U.; Sarkar, B. Rhodium Diamidobenzene Complexes: A Tale of Different Substituents on the Diamidobenzene Ligand. Chem. Sci. 2022, 13, 10532–10545. [Google Scholar] [CrossRef] [PubMed]
- Bens, T.; Walter, R.R.M.; Beerhues, J.; Lücke, C.; Gabler, J.; Sarkar, B. Isolation, Characterization and Reactivity of Key Intermediates Relevant to Reductive (Electro)Catalysis with Cp*Rh Complexes Containing Pyridyl-MIC (MIC=Mesoionic Carbene) Ligands. Chemistry 2024, 30, e202302354. [Google Scholar] [CrossRef]
- Lei, X.; Feng, J.; Guo, Q.; Li, Y.; Shi, J. Synthesis of Polysubstituted Furans via Rh(II)-Catalyzed [2 + 3] Annulation of N-Sulfonyl-1,2,3-Triazoles with Enaminones. Org. Lett. 2023, 25, 7338–7343. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Wei, Q.; Hu, C.; Su, Z. DFT Study on Rh(ii)/Guanidine-Catalyzed Asymmetric N–H Bond Insertion of Benzophenone Imines. Org. Biomol. Chem. 2025, 23, 2896–2903. [Google Scholar] [CrossRef]
- Sinha, S.K.; Ghosh, P.; Jain, S.; Maiti, S.; Al-Thabati, S.A.; Alshehri, A.A.; Mokhtar, M.; Maiti, D. Transition-Metal Catalyzed C–H Activation as a Means of Synthesizing Complex Natural Products. Chem. Soc. Rev. 2023, 52, 7461–7503. [Google Scholar] [CrossRef] [PubMed]
- Bhatia, S. Zeolite Catalysis: Principles and Applications, 1st ed.; CRC Press: Boca Raton, FL, USA, 1989. [Google Scholar]
- Vogt, E.T.C.; Weckhuysen, B.M. Fluid Catalytic Cracking: Recent Developments on the Grand Old Lady of Zeolite Catalysis. Chem. Soc. Rev. 2015, 44, 7342–7370. [Google Scholar] [CrossRef]
- Jae, J.; Tompsett, G.A.; Foster, A.J.; Hammond, K.D.; Auerbach, S.M.; Lobo, R.F.; Huber, G.W. Investigation into the Shape Selectivity of Zeolite Catalysts for Biomass Conversion. J. Catal. 2011, 279, 257–268. [Google Scholar] [CrossRef]
- Verboekend, D.; Pérez-Ramírez, J. Design of Hierarchical Zeolite Catalysts by Desilication. Catal. Sci. Technol. 2011, 1, 879. [Google Scholar] [CrossRef]
- Tarach, K.A.; Martinez-Triguero, J.; Rey, F.; Góra-Marek, K. Hydrothermal Stability and Catalytic Performance of Desilicated Highly Siliceous Zeolites ZSM-5. J. Catal. 2016, 339, 256–269. [Google Scholar] [CrossRef]
- Ho, P.H.; Yao, D.; Creaser, D.; Olsson, L. Advantages of High-Siliceous Zeolites in the Reactivity and Stability of Diesel Oxidation Catalysts. ACS Eng. Au 2022, 2, 219–235. [Google Scholar] [CrossRef]
- Wang, J.; Fu, Y.; Kong, W.; Jin, F.; Bai, J.; Zhang, J.; Sun, Y. Design of a Carbon-Resistant Ni@S-2 Reforming Catalyst: Controllable Ni Nanoparticles Sandwiched in a Peasecod-like Structure. Appl. Catal. B Environ. 2021, 282, 119546. [Google Scholar] [CrossRef]
- Bibby, D.M.; Milestone, N.B.; Aldridge, L.P. Silicalite-2, a Silica Analogue of the Aluminosilicate Zeolite ZSM-11. Nature 1979, 280, 664–665. [Google Scholar] [CrossRef]
- Zhang, X.; Yan, T.; Hou, H.; Yin, J.; Wan, H.; Sun, X.; Zhang, Q.; Sun, F.; Wei, Y.; Dong, M.; et al. Regioselective Hydroformylation of Propene Catalysed by Rhodium-Zeolite. Nature 2024, 629, 597–602. [Google Scholar] [CrossRef]
- Dou, X.; Yan, T.; Qian, L.; Hou, H.; Lopez-Haro, M.; Marini, C.; Agostini, G.; Meira, D.M.; Zhang, X.; Zhang, L.; et al. Regioselective Hydroformylation with Subnanometre Rh Clusters in MFI Zeolite. Nat. Catal. 2024, 7, 666–677. [Google Scholar] [CrossRef]
- Kresse, G.; Hafner, J. Ab Initio Molecular-Dynamics Simulation of the Liquid-Metal–Amorphous-Semiconductor Transition in Germanium. Phys. Rev. B 1994, 49, 14251–14269. [Google Scholar] [CrossRef] [PubMed]
- Kresse, G.; Furthmüller, J. Efficient Iterative Schemes for Ab Initio Total-Energy Calculations Using a Plane-Wave Basis Set. Phys. Rev. B. 1996, 54, 11169–11186. [Google Scholar] [CrossRef]
- Perdew, J.P.; Burke, K.; Ernzerhof, M. Generalized Gradient Approximation Made Simple. Phys. Rev. Lett. 1996, 77, 3865–3868. [Google Scholar] [CrossRef] [PubMed]
- Henkelman, G.; Jónsson, H. Improved Tangent Estimate in the Nudged Elastic Band Method for Finding Minimum Energy Paths and Saddle Points. J. Chem. Phys. 2000, 113, 9978–9985. [Google Scholar] [CrossRef]
- Henkelman, G.; Uberuaga, B.P.; Jónsson, H. A Climbing Image Nudged Elastic Band Method for Finding Saddle Points and Minimum Energy Paths. J. Chem. Phys. 2000, 113, 9901–9904. [Google Scholar] [CrossRef]
- Szécsényi, Á.; Khramenkova, E.; Chernyshov, I.Y.; Li, G.; Gascon, J.; Pidko, E.A. Breaking Linear Scaling Relationships with Secondary Interactions in Confined Space: A Case Study of Methane Oxidation by Fe/ZSM-5 Zeolite. ACS Catal. 2019, 9, 9276–9284. [Google Scholar] [CrossRef]


| State | Adsorption Molecular | Total Adsorption Energy | Adsorption Energy |
|---|---|---|---|
| 1st molecular adsorption | CO | −2.12 | −2.12 |
| CH3OH | −1.50 | −1.50 | |
| C3H6 | −1.64 | −1.64 | |
| 2nd molecular adsorption based on CO adsorption | CO + C3H6 | −3.53 | −1.42 |
| 2CO | −3.77 | −1.66 | |
| CO + CH3OH | −3.34 | −1.22 | |
| 3rd molecular adsorption based on 2CO adsorption | 3CO | −4.40 | −0.67 |
| 2CO + CH3OH | −4.74 | −0.96 | |
| 2CO + C3H6 | −4.35 | −0.57 |
| Pathway | Methyl Butyrate | The Dissociation of H from CH3OH | H Combines with Propylene Derivatives | CO Insertion | OCH3/COOCH3 Combines with Propylene Derivatives |
|---|---|---|---|---|---|
| Hydrogenation-First | n | 1.97 | 0.62 | 1.39 | 0.38 |
| iso | 1.97 | 0.86 | 1.32 | 0.17 | |
| Methoxycarbonyl-Driven | n | 1.97 | 1.63 | 1.15 | 1.41 |
| iso | 1.97 | 1.69 | 1.15 | 1.16 | |
| Carbonylation-First | n | 1.27 | 0.58 | 0.87 | 0.58 |
| iso | 0.43 | 1.33 | 0.84 | 0.56 |
| Transition State | dmin/Å | ⟨d3⟩/Å |
|---|---|---|
| n-TS1 | 2.31 | 2.45 |
| n-TS2 | 2.56 | 2.65 |
| n-TS3 | 2.47 | 2.55 |
| n-TS4 | 2.28 | 2.48 |
| iso-TS1 | 1.96 | 2.01 |
| iso-TS2 | 2.09 | 2.22 |
| iso-TS3 | 1.86 | 2.09 |
| iso-TS4 | 2.14 | 2.29 |
| Reaction Pathway | Line or Branch Routs | The Combination of H | CO Insertion |
|---|---|---|---|
| Carbonylation-First in Rh(II) | n | 0.58 | 0.87 |
| iso | 1.33 | 0.84 | |
| Hydrogenation-First in Rh(II) | n | 0.62 | 1.39 |
| iso | 0.86 | 1.32 | |
| Hydroformylation in Rh(I) | n | 0.15 | 1.38 |
| iso | 0.49 | 1.80 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, L.; Wang, X.; Li, H.; Chen, H.; Feng, W.; Zhao, Z.; Zhao, F.; Lei, S.; Hou, Z.; Fu, S. Unveiling the Catalytic Pathway of Rh(II)/Silicalite-2 in Propene Carbonylation to Methyl Butyrate: A DFT Study. Molecules 2025, 30, 3549. https://doi.org/10.3390/molecules30173549
Wang L, Wang X, Li H, Chen H, Feng W, Zhao Z, Zhao F, Lei S, Hou Z, Fu S. Unveiling the Catalytic Pathway of Rh(II)/Silicalite-2 in Propene Carbonylation to Methyl Butyrate: A DFT Study. Molecules. 2025; 30(17):3549. https://doi.org/10.3390/molecules30173549
Chicago/Turabian StyleWang, Lu, Xingyong Wang, Hongchen Li, He Chen, Wanru Feng, Zerun Zhao, Fujun Zhao, Shuai Lei, Zhanggui Hou, and Songbao Fu. 2025. "Unveiling the Catalytic Pathway of Rh(II)/Silicalite-2 in Propene Carbonylation to Methyl Butyrate: A DFT Study" Molecules 30, no. 17: 3549. https://doi.org/10.3390/molecules30173549
APA StyleWang, L., Wang, X., Li, H., Chen, H., Feng, W., Zhao, Z., Zhao, F., Lei, S., Hou, Z., & Fu, S. (2025). Unveiling the Catalytic Pathway of Rh(II)/Silicalite-2 in Propene Carbonylation to Methyl Butyrate: A DFT Study. Molecules, 30(17), 3549. https://doi.org/10.3390/molecules30173549






