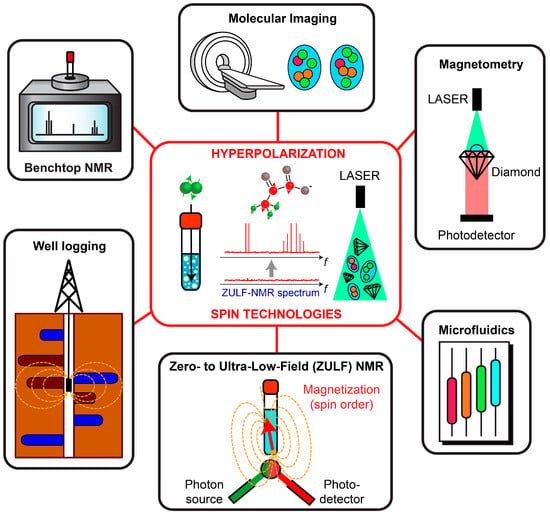
Molecules, Up Your Spins!
- Hyperpolarization Techniques
- Techniques Utilizing Electromagnetic Fields
- Techniques Utilizing Chemistry and Spin Statistics
- Medical Applications of Hyperpolarization
- Comparison of PET and Hyperpolarization-Enhanced MRI
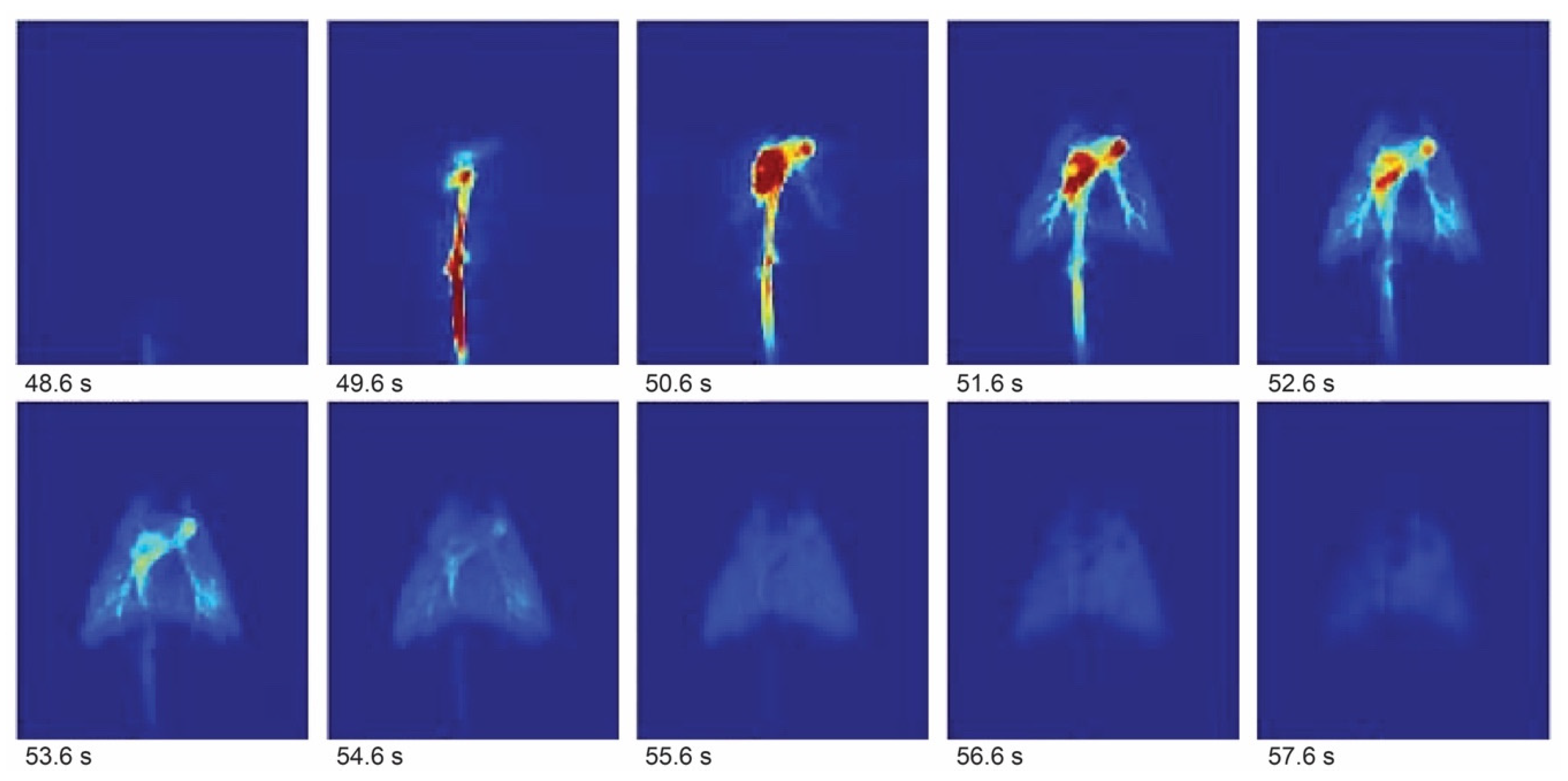
- Challenges of Hyperpolarized Molecular MRI
- Emerging Spin Technologies
- Conclusions
Funding
Acknowledgments
Conflicts of Interest
References
- Nagornov, K.O.; Gorshkov, M.V.; Kozhinov, A.N.; Tsybin, Y.O. High-resolution Fourier transform ion cyclotron resonance mass spectrometry with increased throughput for biomolecular analysis. Anal. Chem. 2014, 86, 9020–9028. [Google Scholar] [CrossRef] [PubMed]
- Vasconcelos Soares Maciel, E.; de Toffoli, A.L.; Sobieski, E.; Domingues Nazário, C.E.; Lanças, F.M. Miniaturized liquid chromatography focusing on analytical columns and mass spectrometry: A review. Anal. Chim. Acta 2020, 1103, 11–31. [Google Scholar] [CrossRef] [PubMed]
- Huffman, R.G.; Leduc, A.; Wichmann, C.; Di Gioia, M.; Borriello, F.; Specht, H.; Derks, J.; Khan, S.; Khoury, L.; Emmott, E.; et al. Prioritized mass spectrometry increases the depth, sensitivity and data completeness of single-cell proteomics. Nat. Methods 2023, 20, 714–722. [Google Scholar] [CrossRef] [PubMed]
- Abragam, A. The Principles of Nuclear Magnetism; Clarendon Press: Oxford, UK, 1961. [Google Scholar]
- Salikhov, K.M.; Molin, Y.N.; Sagdeev, R.Z.; Buchachenko, A.L. Spin Polarization and Magnetic Effects in Chemical Reactions; Elsevier: Amsterdam, The Netherlands, 1984. [Google Scholar]
- Fisher, M.P.A.; Radzihovsky, L. Quantum indistinguishability in chemical reactions. Proc. Natl. Acad. Sci. USA 2018, 115, E4551–E4558. [Google Scholar] [CrossRef] [PubMed]
- Roose, B.W.; Zemerov, S.D.; Dmochowski, I.J. Nanomolar small-molecule detection using a genetically encoded 129Xe NMR contrast agent. Chem. Sci. 2017, 8, 7631–7636. [Google Scholar] [CrossRef] [PubMed]
- Wen, L.; Meng, H.; Gu, S.; Wu, J.; Zhao, Y. Toward Nanomolar Multi-Component Analysis by 19F NMR. Anal. Chem. 2022, 94, 8024–8032. [Google Scholar] [CrossRef] [PubMed]
- Eshuis, N.; Hermkens, N.; van Weerdenburg, B.J.A.; Feiters, M.C.; Rutjes, F.P.J.T.; Wijmenga, S.S.; Tessari, M. Toward Nanomolar Detection by NMR Through SABRE Hyperpolarization. J. Am. Chem. Soc. 2014, 136, 2695–2698. [Google Scholar] [CrossRef] [PubMed]
- Levitt, M.H.; Bengs, C. Hyperpolarization and the physical boundary of Liouville space. Magn. Reson. 2021, 2, 395–407. [Google Scholar] [CrossRef] [PubMed]
- Barskiy, D.A.; Kovtunov, K.V.; Koptyug, I.V.; He, P.; Groome, K.A.; Best, Q.A.; Shi, F.; Goodson, B.M.; Shchepin, R.V.; Truong, M.L.; et al. In Situ and Ex Situ Low-Field NMR Spectroscopy and MRI Endowed by SABRE Hyperpolarization. ChemPhysChem 2014, 15, 4100–4107. [Google Scholar] [CrossRef]
- Kircher, R.; Xu, J.; Barskiy, D.A. In Situ Hyperpolarization Enables 15N and 13C Benchtop NMR at Natural Isotopic Abundance. J. Am. Chem. Soc. 2024, 146, 514–520. [Google Scholar] [CrossRef]
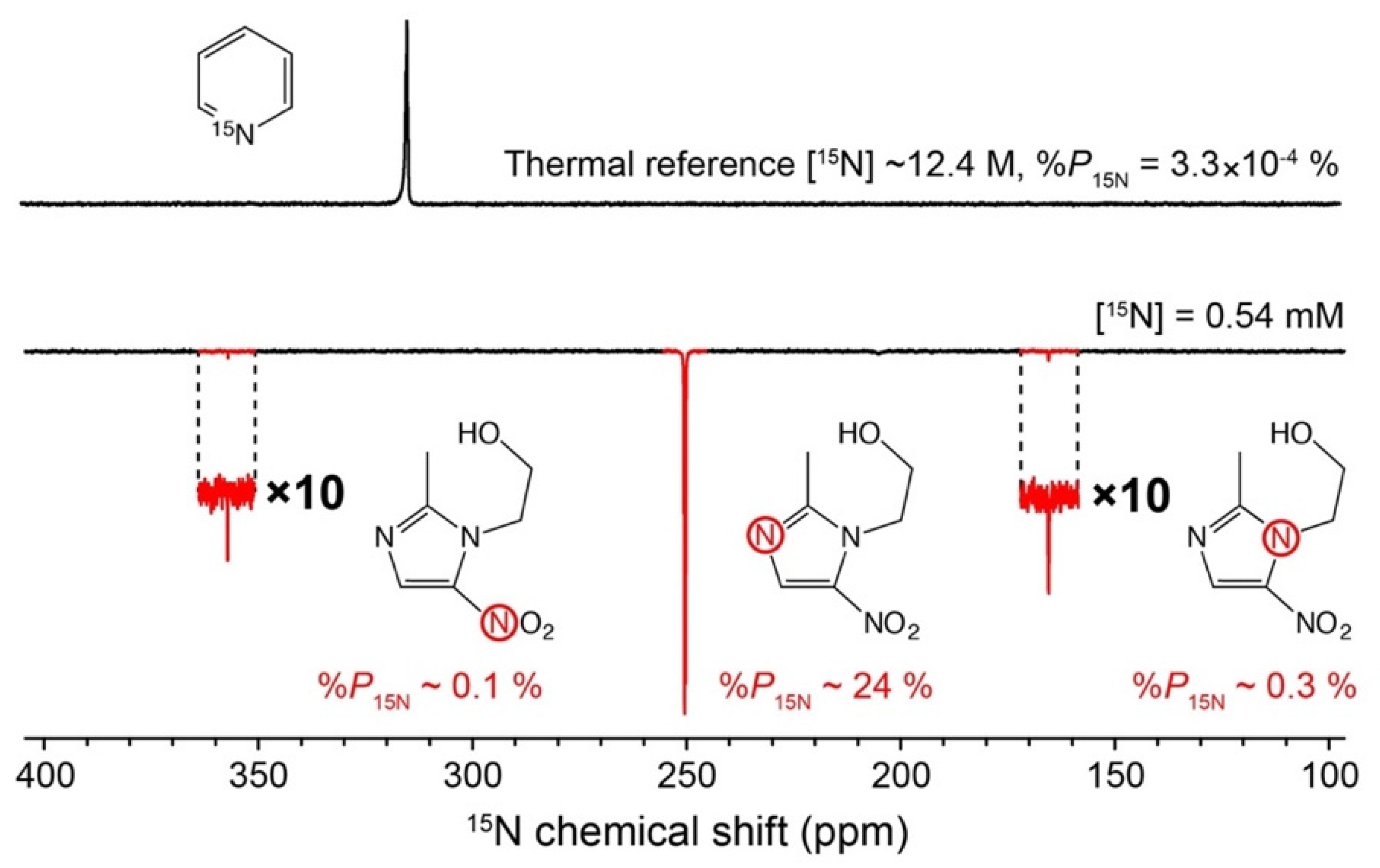
- Barskiy, D.A.; Shchepin, R.V.; Coffey, A.M.; Theis, T.; Warren, W.S.; Goodson, B.M.; Chekmenev, E.Y. Over 20% N-15 Hyperpolarization in Under One Minute for Metronidazole, an Antibiotic and Hypoxia Probe. J. Am. Chem. Soc. 2016, 138, 8080–8083. [Google Scholar] [CrossRef]
- Kovtunov, K.V.; Pokochueva, E.V.; Salnikov, O.G.; Cousin, S.F.; Kurzbach, D.; Vuichoud, B.; Jannin, S.; Chekmenev, E.Y.; Goodson, B.M.; Barskiy, D.A.; et al. Hyperpolarized NMR spectroscopy: d-DNP, PHIP, and SABRE techniques. Chem. Asian J. 2018, 13, 1857–1871. [Google Scholar] [CrossRef] [PubMed]
- Ardenkjaer-Larsen, J.H. On the present and future of dissolution-DNP. J. Magn. Reson. 2016, 264, 3–12. [Google Scholar] [CrossRef] [PubMed]
- Jähnig, F.; Kwiatkowski, G.; Ernst, M. Conceptual and instrumental progress in dissolution DNP. J. Magn. Reson. 2016, 264, 22–29. [Google Scholar] [CrossRef] [PubMed]
- Buratto, R.; Bornet, A.; Milani, J.; Mammoli, D.; Vuichoud, B.; Salvi, N.; Singh, M.; Laguerre, A.; Passemard, S.; Gerber-Lemaire, S.; et al. Drug Screening Boosted by Hyperpolarized Long-Lived States in NMR. ChemMedChem 2014, 9, 2509–2515. [Google Scholar] [CrossRef] [PubMed]
- Comment, A.; Rentsch, J.; Kurdzesau, F.; Jannin, S.; Uffmann, K.; van Heeswijk, R.B.; Hautle, P.; Konter, J.A.; van den Brandt, B.; van der Klink, J.J. Producing over 100 mL of highly concentrated hyperpolarized solution by means of dissolution DNP. J. Magn. Reson. 2008, 194, 152–155. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ardenkjaer-Larsen, J.H.; Fridlund, B.; Gram, A.; Hansson, G.; Hansson, L.; Lerche, M.H.; Servin, R.; Thaning, M.; Golman, K. Increase in signal-to-noise ratio of > 10,000 times in liquid-state NMR. Proc. Natl. Acad. Sci. USA 2003, 100, 10158–10163. [Google Scholar] [CrossRef]
- Kouřil, K.; Kouřilová, H.; Bartram, S.; Levitt, M.H.; Meier, B. Scalable dissolution-dynamic nuclear polarization with rapid transfer of a polarized solid. Nat. Commun. 2019, 10, 1733. [Google Scholar] [CrossRef] [PubMed]
- Barskiy, D.A.; Coffey, A.M.; Nikolaou, P.; Mikhaylov, D.M.; Goodson, B.M.; Branca, R.T.; Lu, G.J.; Shapiro, M.G.; Telkki, V.V.; Zhivonitko, V.V.; et al. NMR Hyperpolarization Techniques of Gases. Chem. Eur. J. 2017, 23, 725–751. [Google Scholar] [CrossRef] [PubMed]
- Kehayias, P.; Jarmola, A.; Mosavian, N.; Fescenko, I.; Benito, F.M.; Laraoui, A.; Smits, J.; Bougas, L.; Budker, D.; Neumann, A.; et al. Solution nuclear magnetic resonance spectroscopy on a nanostructured diamond chip. Nat. Commun. 2017, 8, 188. [Google Scholar] [CrossRef]
- Budker, D.; Barskiy, D.; Lenz, T. Kernspinresonanz ohne Magnetfeld: Neue Methoden der Kernspinresonanz. Physik Unserer Zeit 2023, 54, 294–301. [Google Scholar] [CrossRef]
- King, J.P.; Jeong, K.; Vassiliou, C.C.; Shin, C.S.; Page, R.H.; Avalos, C.E.; Wang, H.-J.; Pines, A. Room-temperature in situ nuclear spin hyperpolarization from optically pumped nitrogen vacancy centres in diamond. Nat. Commun. 2015, 6, 8965. [Google Scholar] [CrossRef] [PubMed]
- Goez, M. Photo-CIDNP spectroscopy. Annu. Rep. NMR Spectrosc. 2009, 66, 77–147. [Google Scholar]
- Morozova, O.B.; Ivanov, K.L. Time-Resolved Chemically Induced Dynamic Nuclear Polarization of Biologically Important Molecules. ChemPhysChem 2019, 20, 197–215. [Google Scholar] [CrossRef] [PubMed]
- Maul, A.; Blümler, P.; Nacher, P.J.; Otten, E.; Tastevin, G.; Heil, W. Nuclear hyperpolarization of 3He by magnetized plasmas. Phys. Rev. A 2018, 98, 063405. [Google Scholar] [CrossRef]
- Rayner, P.J.; Burns, M.J.; Olaru, A.M.; Norcott, P.; Fekete, M.; Green, G.G.R.; Highton, L.A.R.; Mewis, R.E.; Duckett, S.B. Delivering strong 1H nuclear hyperpolarization levels and long magnetic lifetimes through signal amplification by reversible exchange. Proc. Natl. Acad. Sci. USA 2017, 114, E3188–E3194. [Google Scholar] [CrossRef] [PubMed]
- Knecht, S.; Blanchard, J.W.; Barskiy, D.; Cavallari, E.; Dagys, L.; Van Dyke, E.; Tsukanov, M.; Bliemel, B.; Münnemann, K.; Aime, S.; et al. Rapid hyperpolarization and purification of the metabolite fumarate in aqueous solution. Proc. Natl. Acad. Sci. USA 2021, 118, e2025383118. [Google Scholar] [CrossRef] [PubMed]
- Ellermann, F.; Sirbu, A.; Brahms, A.; Assaf, C.; Herges, R.; Hövener, J.-B.; Pravdivtsev, A.N. Spying on parahydrogen-induced polarization transfer using a half-tesla benchtop MRI and hyperpolarized imaging enabled by automation. Nat. Commun. 2023, 14, 4774. [Google Scholar] [CrossRef] [PubMed]
- De Maissin, H.; Groß, P.R.; Mohiuddin, O.; Weigt, M.; Nagel, L.; Herzog, M.; Wang, Z.; Willing, R.; Reichardt, W.; Pichotka, M.; et al. In Vivo Metabolic Imaging of [1-13C]Pyruvate-d3 Hyperpolarized by Reversible Exchange with Parahydrogen. Angew. Chem. Int. Ed. 2023, 62, e202306654. [Google Scholar] [CrossRef]
- Eills, J.; Budker, D.; Cavagnero, S.; Chekmenev, E.Y.; Elliott, S.J.; Jannin, S.; Lesage, A.; Matysik, J.; Meersmann, T.; Prisner, T. Spin hyperpolarization in modern magnetic resonance. Chem. Rev. 2023, 123, 1417–1551. [Google Scholar] [CrossRef]
- Xu, J.; Budker, D.; Barskiy, D.A. Visualization of dynamics in coupled multi-spin systems. Magn. Reson. 2022, 3, 145–160. [Google Scholar] [CrossRef] [PubMed]
- Barskiy, D.A.; Pravdivtsev, A. Magnetization and Polarization of Coupled Nuclear Spin Ensembles. arXiv 2023, arXiv:2308.15837. [Google Scholar]
- Nikolaou, P.; Goodson, B.M.; Chekmenev, E.Y. NMR Hyperpolarization Techniques for Biomedicine. Chem. Eur. J. 2015, 21, 3156–3166. [Google Scholar] [CrossRef] [PubMed]
- Meier, S.; Jensen, P.R.; Karlsson, M.; Lerche, M.H. Hyperpolarized NMR Probes for Biological Assays. Sensors 2014, 14, 1576–1597. [Google Scholar] [CrossRef]
- Kurhanewicz, J.; Vigneron, D.B.; Brindle, K.; Chekmenev, E.Y.; Comment, A.; Cunningham, C.H.; DeBerardinis, R.J.; Green, G.G.; Leach, M.O.; Rajan, S.S.; et al. Analysis of Cancer Metabolism by Imaging Hyperpolarized Nuclei: Prospects for Translation to Clinical Research. Neoplasia 2011, 13, 81–97. [Google Scholar] [CrossRef] [PubMed]
- Nagana Gowda, G.A.; Raftery, D. NMR Metabolomics Methods for Investigating Disease. Anal. Chem. 2023, 95, 83–99. [Google Scholar] [CrossRef] [PubMed]
- Shchepin, R.V.; Birchall, J.R.; Chukanov, N.V.; Kovtunov, K.V.; Koptyug, I.V.; Theis, T.; Warren, W.S.; Gelovani, J.G.; Goodson, B.M.; Shokouhi, S.; et al. Hyperpolarizing Concentrated Metronidazole 15NO2 Group over Six Chemical Bonds with More than 15% Polarization and a 20 Minute Lifetime. Chem. Eur. J. 2019, 25, 8829–8836. [Google Scholar] [CrossRef]
- Svyatova, A.; Skovpin, I.V.; Chukanov, N.V.; Kovtunov, K.V.; Chekmenev, E.Y.; Pravdivtsev, A.N.; Hövener, J.B.; Koptyug, I.V. 15N MRI of SLIC-SABRE hyperpolarized 15N-labelled pyridine and nicotinamide. Chem. Eur. J 2019, 25, 8465–8470. [Google Scholar] [CrossRef] [PubMed]
- Budinger, T.F.; Van Brocklin, H.F. Positron Emission Tomography; CRC Press: Boca Raton, FL, USA, 1985. [Google Scholar]
- Moses, W.W. Fundamental Limits of Spatial Resolution in PET. Nucl. Instrum. Methods Phy.s Res. A 2011, 648 (Suppl. 1), S236–S240. [Google Scholar] [CrossRef]
- Angelovski, G. What We Can Really Do with Bioresponsive MRI Contrast Agents. Angew. Chem. Int. Ed. 2016, 55, 7038–7046. [Google Scholar] [CrossRef]
- Lane, N. Transformer: The Deep Chemistry of Life and Death; W. W. Norton & Company: New York, NY, USA, 2022. [Google Scholar]
- Coffey, A.M.; Kovtunov, K.V.; Barskiy, D.A.; Koptyug, I.V.; Shchepin, R.V.; Waddell, K.W.; He, P.; Groome, K.A.; Best, Q.A.; Shi, F.; et al. High-Resolution Low-Field Molecular Magnetic Resonance Imaging of Hyperpolarized Liquids. Anal. Chem. 2014, 86, 9042–9049. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.; Ploessl, K.; Kung, H.F. PET/SPECT imaging agents for neurodegenerative diseases. Chem. Soc. Rev. 2014, 43, 6683–6691. [Google Scholar] [CrossRef] [PubMed]
- Golman, K.; Olsson, L.E.; Axelsson, O.; Mansson, S.; Karlsson, M.; Petersson, J.S. Molecular imaging using hyperpolarized 13C. Br. J. Radiol. 2003, 76, S118–S127. [Google Scholar] [CrossRef] [PubMed]
- De Feyter, H.M.; Behar, K.L.; Corbin, Z.A.; Fulbright, R.K.; Brown, P.B.; Mcintyre, S.; Nixon, T.W.; Rothman, D.L.; De Graaf, R.A. Deuterium metabolic imaging (DMI) for MRI-based 3D mapping of metabolism in vivo. Sci. Adv. 2018, 4, eaat7314. [Google Scholar] [CrossRef]
- Montrazi, E.T.; Sasson, K.; Lilach, A.; Scherz, A.; Frydman, L. Molecular imaging of tumor metabolism: Insight from pyruvate- and glucose-based deuterium MRI studies. Sci. Adv. 2024, 10, eadm8600. [Google Scholar] [CrossRef] [PubMed]
- Salnikov, O.G.; Trofimov, I.A.; Bender, Z.T.; Trepakova, A.I.; Xu, J.; Wibbels, G.L.; Shchepin, R.V.; Koptyug, I.V.; Barskiy, D.A. Parahydrogen-Induced Polarization of 14N Nuclei. Angew Chem. Int. Ed. 2024, e202402877. [Google Scholar]
- Zimmer, M. Green fluorescent protein (GFP): Applications, structure, and related photophysical behavior. Chem. Rev. 2002, 102, 759–782. [Google Scholar] [CrossRef]
- Shapiro, M.G.; Ramirez, R.M.; Sperling, L.J.; Sun, G.; Sun, J.; Pines, A.; Schaffer, D.V.; Bajaj, V.S. Genetically encoded reporters for hyperpolarized xenon magnetic resonance imaging. Nat. Chem. 2014, 6, 629–634. [Google Scholar] [CrossRef] [PubMed]
- Jayapaul, J.; Schröder, L. Molecular Sensing with Host Systems for Hyperpolarized 129Xe. Molecules 2020, 25, 4627. [Google Scholar] [CrossRef]
- Hövener, J.B.; Schwaderlapp, N.; Lickert, T.; Duckett, S.B.; Mewis, R.E.; Highton, L.A.R.; Kenny, S.M.; Green, G.G.R.; Leibfritz, D.; Korvink, J.G.; et al. A hyperpolarized equilibrium for magnetic resonance. Nat. Commun. 2013, 4, 2946. [Google Scholar] [CrossRef]
- Bricco, A.R.; Miralavy, I.; Bo, S.; Perlman, O.; Korenchan, D.E.; Farrar, C.T.; McMahon, M.T.; Banzhaf, W.; Gilad, A.A. A Genetic Programming Approach to Engineering MRI Reporter Genes. ACS Synth. Biol. 2023, 12, 1154–1163. [Google Scholar] [CrossRef] [PubMed]
- Chaumeil, M.M.; Bankson, J.A.; Brindle, K.M.; Epstein, S.; Gallagher, F.A.; Grashei, M.; Guglielmetti, C.; Kaggie, J.D.; Keshari, K.R.; Knecht, S.; et al. New Horizons in Hyperpolarized 13C MRI. Mol. Imaging Biol. 2023, 26, 222–232. [Google Scholar] [CrossRef]
- Pearl, R. Breaking the Rules of Healthcare: Selecting the Best Technology. Forbes 2022. [Google Scholar]
- Nelson, S.J.; Kurhanewicz, J.; Vigneron, D.B.; Larson, P.E.; Harzstark, A.L.; Ferrone, M.; van Criekinge, M.; Chang, J.W.; Bok, R.; Park, I. Metabolic imaging of patients with prostate cancer using hyperpolarized [1-13C] pyruvate. Sci. Transl. Med. 2013, 5, ra108–ra198. [Google Scholar] [CrossRef] [PubMed]
- Pezze, L.; Smerzi, A.; Oberthaler, M.K.; Schmied, R.; Treutlein, P. Quantum metrology with nonclassical states of atomic ensembles. Rev. Mod. Phys. 2018, 90, 035005. [Google Scholar] [CrossRef]
- Budker, D. Extreme nuclear magnetic resonance: Zero field, single spins, dark matter…. J. Magn. Reson. 2019, 306, 66–68. [Google Scholar] [CrossRef]
- Sonnefeld, A.; Razanahoera, A.; Pelupessy, P.; Bodenhausen, G.; Sheberstov, K. Long-lived states of methylene protons in achiral molecules. Sci. Adv. 2022, 8, eade2113. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Barskiy, D.A. Molecules, Up Your Spins! Molecules 2024, 29, 1821. https://doi.org/10.3390/molecules29081821
Barskiy DA. Molecules, Up Your Spins! Molecules. 2024; 29(8):1821. https://doi.org/10.3390/molecules29081821
Chicago/Turabian StyleBarskiy, Danila A. 2024. "Molecules, Up Your Spins!" Molecules 29, no. 8: 1821. https://doi.org/10.3390/molecules29081821
APA StyleBarskiy, D. A. (2024). Molecules, Up Your Spins! Molecules, 29(8), 1821. https://doi.org/10.3390/molecules29081821