Stevia Polyphenols, Their Antimicrobial and Anti-Inflammatory Properties, and Inhibitory Effect on Digestive Enzymes
Abstract
:1. Introduction
2. Results and Discussion
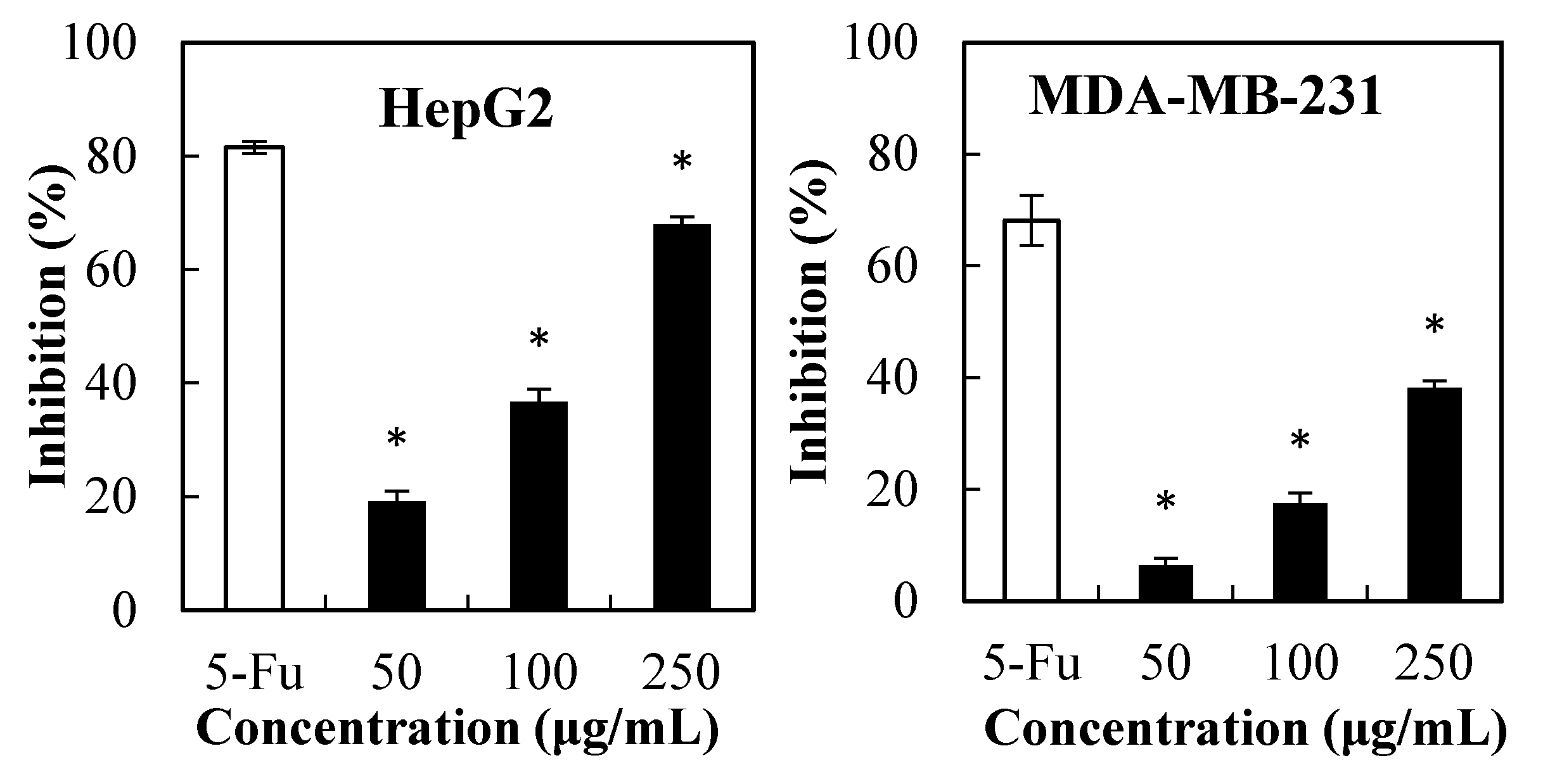
2.1. Cytotoxicity of PPSs towards Cancer Cells
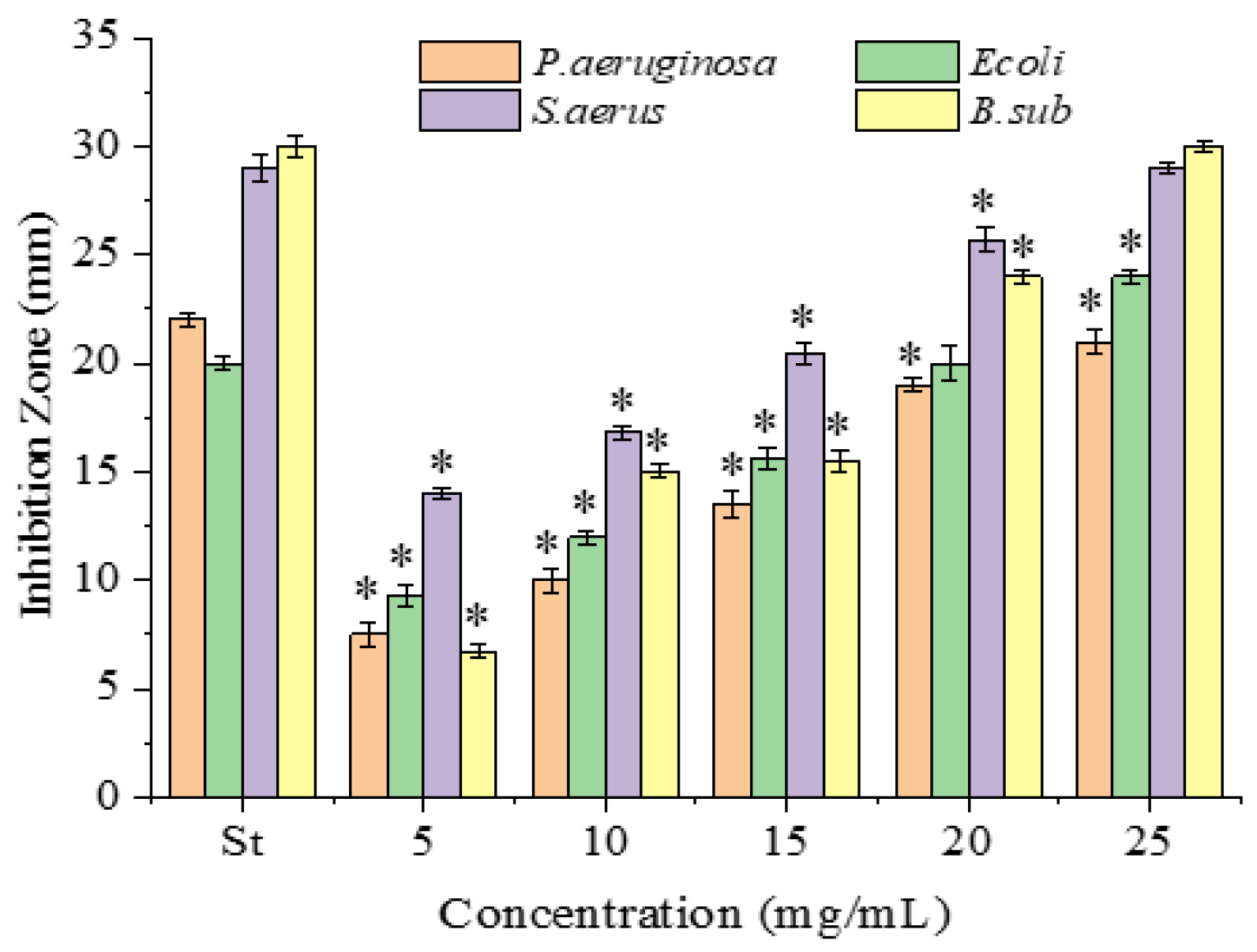
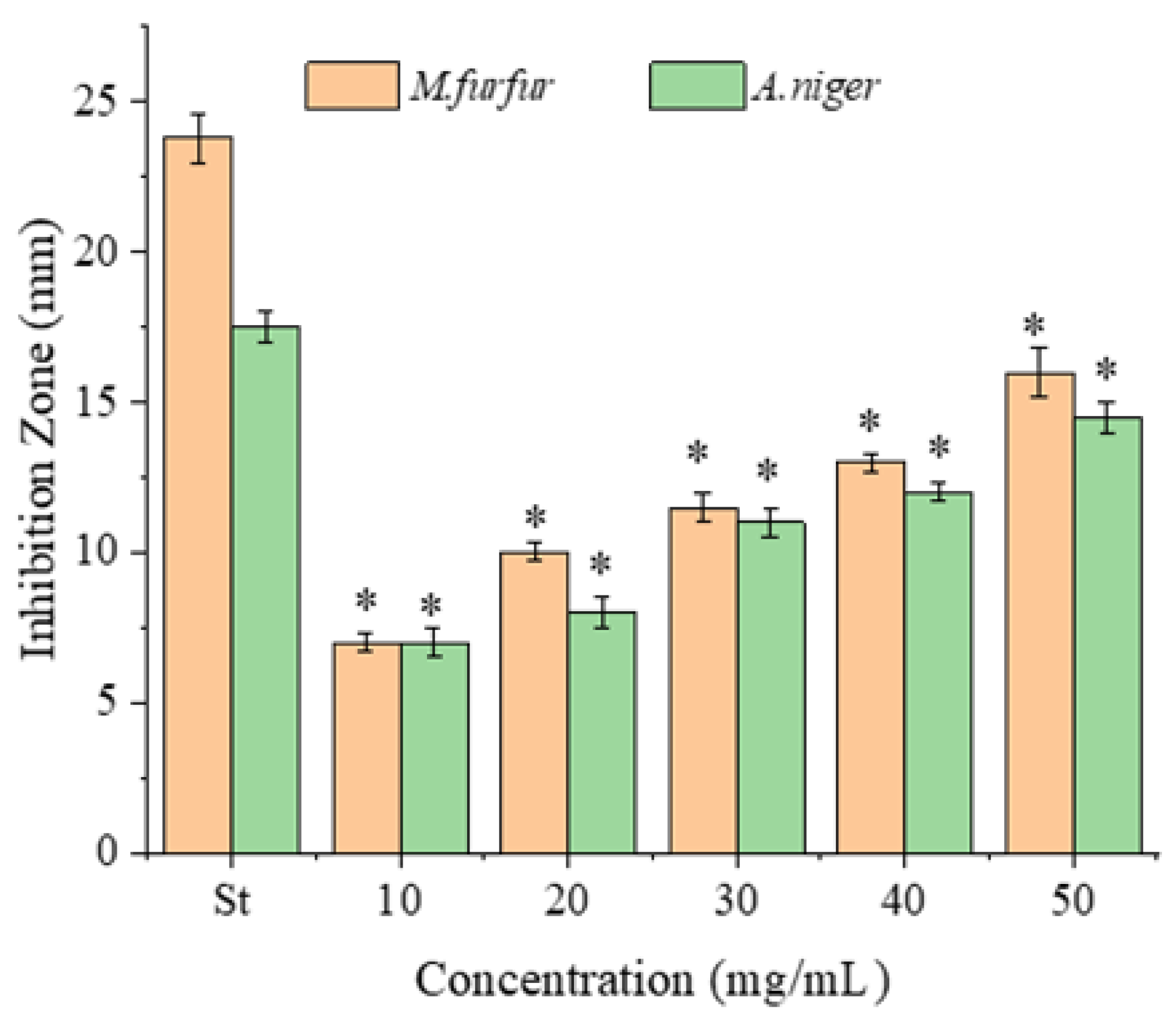
2.2. Antimicrobial Activity
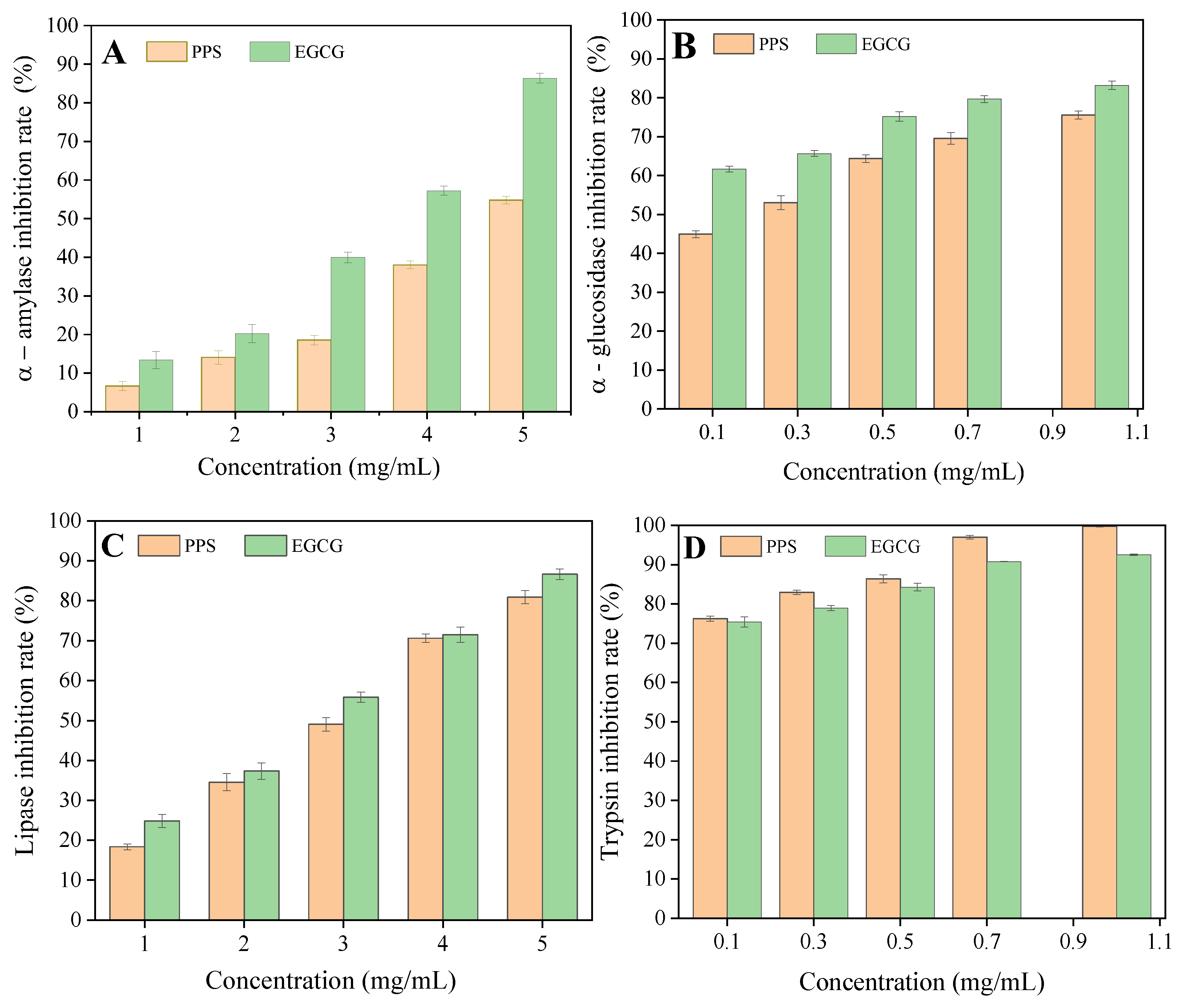
2.3. Inhibitory Effect of PPSs on Digestive Enzymes
2.4. Kinetic Analysis of the Inhibition of Lipase and Trypsin
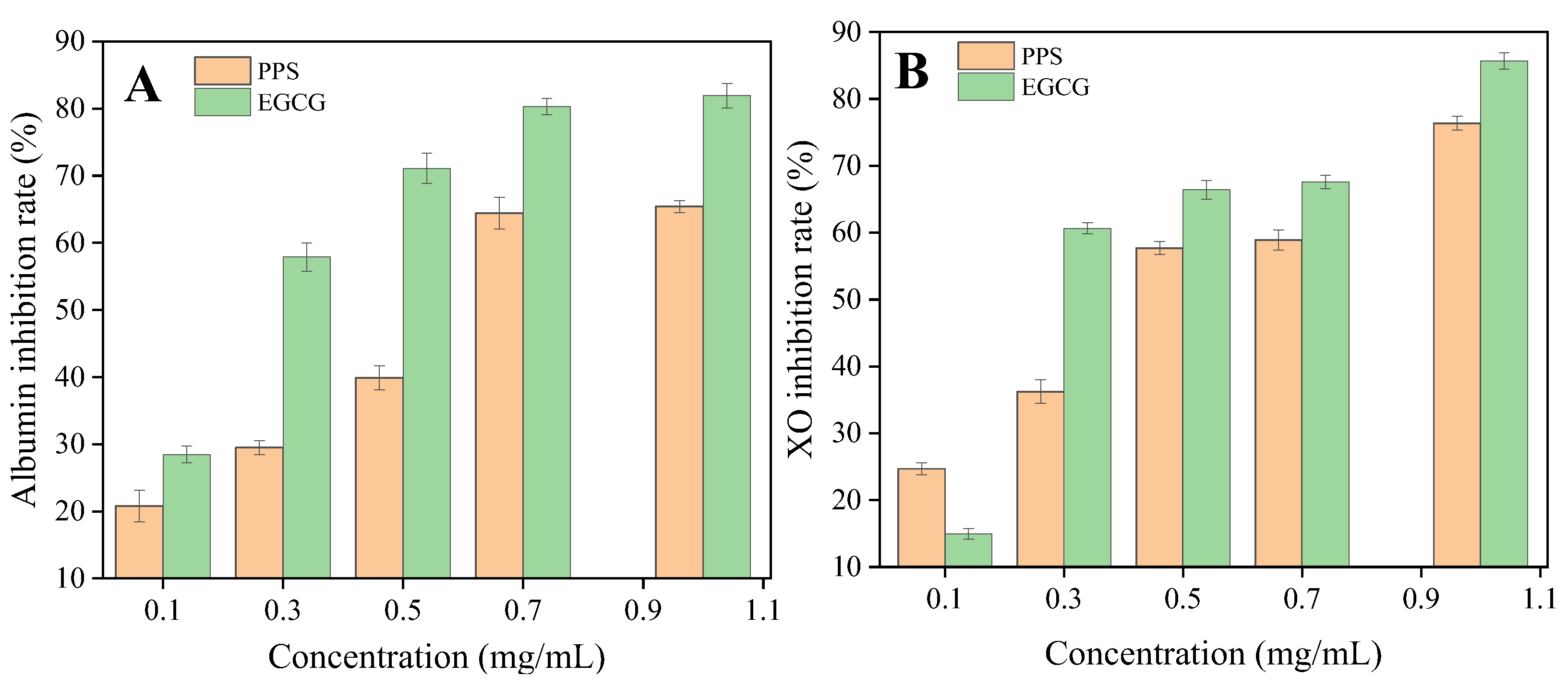
2.5. In Vitro Anti-Inflammatory Activity
3. Materials and Methods
3.1. Materials
3.2. HPLC Analysis of the PPS
3.3. Microbial Strains, Cancer Cell Lines, and Cell Culture
3.4. Cytotoxicity Assay
3.5. Antimicrobial Activity
3.6. Determination of Minimum Inhibitory Concentration (MIC)
3.7. Inhibitory effect of the PPSs on Digestive Enzymes
3.7.1. Pancreatic Lipase Assay
3.7.2. α-Amylase Assay
3.7.3. α-Glucosidase Assay
3.7.4. Trypsin Assay
3.8. Kinetics Analysis
3.9. In Vitro Anti-Inflammation
3.9.1. Inhibition of Albumin
3.9.2. Inhibition of Xanthine Oxidase
3.10. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Myint, K.Z.; Wu, K.; Xia, Y.; Fan, Y.; Shen, J.; Zhang, P.; Gu, J. Polyphenols from Stevia rebaudiana (Bertoni) leaves and their functional properties. J. Food Sci. 2020, 85, 240–248. [Google Scholar] [CrossRef]
- Myint, K.Z.; Yu, Q.; Qing, J.; Zhu, S.; Shen, J.; Xia, Y. Botanic antimicrobial agents, their antioxidant properties, application and safety issue. Food Packag. Shelf Life 2022, 34, 100924. [Google Scholar] [CrossRef]
- Schiatti-Sisó, I.P.; Quintana, S.E.; García-Zapateiro, L.A. Stevia (Stevia rebaudiana) as a common sugar substitute and its application in food matrices: An updated review. J. Food Sci. Technol. 2023, 60, 1483–1492. [Google Scholar] [CrossRef]
- Orellana-Paucar, A.M. Steviol Glycosides from Stevia rebaudiana: An Updated Overview of Their Sweetening Activity, Pharmacological Properties, and Safety Aspects. Molecules 2023, 28, 1258. [Google Scholar] [CrossRef]
- Lopez, V.; Perez, S.; Vinuesa, A.; Zorzetto, C.; Abian, O. Stevia rebaudiana ethanolic extract exerts better antioxidant properties and antiproliferative effects in tumour cells than its diterpene glycoside stevioside. Food Funct. 2016, 7, 2107–2113. [Google Scholar] [CrossRef] [PubMed]
- Myint, K.Z.; Zhou, Z.; Xia, Y.; Fang, Y.; Wu, M.; Zhu, S.; Shen, J. Stevia polyphenols: A stable antioxidant that presents a synergistic effect with vitamin C. J. Food Process. Preserv. 2021, 45, e15317. [Google Scholar] [CrossRef]
- Yılmaz, F.M.; Görgüç, A.; Uygun, Ö.; Bircan, C. Steviol glycosides and polyphenols extraction from Stevia rebaudiana Bertoni leaves using maceration, microwave-, and ultrasound-assisted techniques. Sep. Sci. Technol. 2021, 56, 936–948. [Google Scholar] [CrossRef]
- Muñoz-Fariña, O.; López-Casanova, V.; García-Figueroa, O.; Roman-Benn, A.; Ah-Hen, K.; Bastias-Montes, J.M.; Quevedo-León, R.; Ravanal-Espinosa, M.C. Bioaccessibility of phenolic compounds in fresh and dehydrated blueberries (Vaccinium corymbosum L.). Food Chem. Adv. 2023, 2, 100171. [Google Scholar] [CrossRef]
- Papaefthimiou, M.; Kontou, P.I.; Bagos, P.G.; Braliou, G.G. Antioxidant Activity of Leaf Extracts from Stevia rebaudiana Bertoni Exerts Attenuating Effect on Diseased Experimental Rats: A Systematic Review and Meta-Analysis. Nutrients 2023, 15, 3325. [Google Scholar] [CrossRef] [PubMed]
- Gawel-Beben, K.; Bujak, T.; Niziol-Lukaszewska, Z.; Antosiewicz, B.; Jakubczyk, A.; Karas, M.; Rybczynska, K. Steiva rebaudiana Bert. Leaf extracts as a multifunctional source of natural antioxidants. Molecules 2015, 20, 5468–5486. [Google Scholar] [CrossRef]
- Covarrubias-Cardenas, A.G.; Martinez-Castillo, J.I.; Medina-Torres, N.; Ayora-Talavera, T.; Espinosa-Andrews, H.; Garcia-Cruz, N.U.; Pacheco, N. Antioxidant capacity and UPLC-PDA ESI-MS phenolic profile of Stevia rebaudiana dry powder extracts obtained by Uutrasound assisted extraction. Agronomy 2018, 8, 170. [Google Scholar] [CrossRef]
- Atas, M.; Eruygur, N.; Ucar, E.; Ozyigit, Y.; Turgut, K. The Effects of different nitrogen doses on antioxidant and antimicrobial activity of Stevia (Stevia rebaudiana Bert.). Cell. Mol. Biol. 2018, 64, 39–45. [Google Scholar] [CrossRef] [PubMed]
- Gamboa, F.; Chaves, M. Antimicrobial potential of extracts from Stevia rebaudiana leaves against bacteria of importance in dental caries. Acta Odontol. Latinoam. Aol 2012, 25, 171–175. [Google Scholar] [PubMed]
- Preethi, D.; Sridhar, T.M.; Josthna, P.; Naidu, C.V. Studies on Antibacterial Activity, Phytochemical Analysis of Stevia rebaudiana (Bert.)—An Important Calorie Free Biosweetner. J. Ecobiotechnology 2011, 3, 5–10. [Google Scholar]
- Lemus-Mondaca, R.; Vega-Galvez, A.; Rojas, P.; Stucken, K.; Delporte, C. Antioxidant, antimicrobial and anti-inflammatory potential of Stevia rebaudiana leaves: Effect of different drying methods. J. Appl. Res. Med. Aroma 2018, 11, 37–46. [Google Scholar] [CrossRef]
- Yildiz-Ozturk, E.; Nalbantsoy, A.; Tag, O.; Yesil-Celiktas, O. A comparative study on extraction processes of Stevia rebaudiana leaves with emphasis on antioxidant, cytotoxic and nitric oxide inhibition activities. Ind. Crops Prod. 2015, 77, 961–971. [Google Scholar] [CrossRef]
- Alnuqaydan, A.M.; Rah, B. Comparative assessment of biological activities of different parts of halophytic plant Tamarix articulata (T. articulata) growing in Saudi Arabia. Saudi J. Biol. Sci. 2020, 27, 2586–2592. [Google Scholar] [CrossRef]
- Bender, C.; Graziano, S.; Zimmermann, B.F. Study of Stevia rebaudiana Bertoni antioxidant activities and cellular properties. Int. J. Food Sci. Nutr. 2015, 66, 553–558. [Google Scholar] [CrossRef] [PubMed]
- Dkhil, M.A.; Zreiq, R.; Hafiz, T.A.; Mubaraki, M.A.; Sulaiman, S.; Algahtani, F.; Abdel-Gaber, R.; Al-Shaebi, E.M.; Al-Quraishy, S. Anthelmintic and antimicrobial activity of Indigofera oblongifolia leaf extracts. Saudi J. Biol. Sci. 2020, 27, 594–598. [Google Scholar] [CrossRef]
- Hemeg, H.A.; Moussa, I.M.; Ibrahim, S.; Dawoud, T.M.; Alhaji, J.H.; Mubarak, A.S.; Kabli, S.A.; Alsubki, R.A.; Tawfik, A.M.; Marouf, S.A. Antimicrobial effect of different herbal plant extracts against different microbial population. Saudi J. Biol. Sci. 2020, 12, 3221–3227. [Google Scholar] [CrossRef]
- Guo, N.; Zang, Y.P.; Cui, Q.; Gai, Q.Y.; Jiao, J.; Wang, W.; Zu, Y.G.; Fu, Y.J. The preservative potential of Amomum tsaoko essential oil against E. coil, its antibacterial property and mode of action. Food Control 2017, 75, 236–245. [Google Scholar] [CrossRef]
- Kaeuffer, C.; Baldacini, M.; Ruge, T.; Ruch, Y.; Zhu, Y.J.; De Cian, M.; Philouze, G.; Bachellier, P.; Denis, J.; Lefebvre, N.; et al. Fungal Infections Caused by Kazachstania spp., Strasbourg, France, 2007–2020. Emerg. Infect. Dis. 2022, 28, 29–34. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Li, X.; Hou, L.-X.; Wei, A.-Z. Extraction solvent affects the antioxidant, antimicrobial, cholinesterase and HepG2 human hepatocellular carcinoma cell inhibitory activities of Zanthoxylum bungeanum pericarps and the major chemical components. Ind. Crops Prod. 2019, 142, 111872. [Google Scholar] [CrossRef]
- Tourabi, M.; Metouekel, A.; Ghouizi, A.E.L.; Jeddi, M.; Nouioura, G.; Laaroussi, H.; Hosen, M.E.; Benbrahim, K.F.; Bourhia, M.; Salamatullah, A.M.; et al. Efficacy of various extracting solvents on phytochemical composition, and biological properties of Mentha longifolia L. leaf extracts. Sci. Rep. 2023, 13, 18028. [Google Scholar] [CrossRef]
- Jayaraman, S.; Manoharan, M.S.; Illanchezian, S. In-vitro Antimicrobial and Antitumor Activities of Stevia Rebaudiana (Asteraceae) Leaf Extracts. Trop. J. Pharm. Res. 2008, 7, 1143–1149. [Google Scholar] [CrossRef]
- Yilmazer-Musa, M.; Griffith, A.M.; Michels, A.J.; Schneider, E.; Frei, B. Grape seed and tea extracts and catechin 3-gallates are potent inhibitors of α-amylase and α-glucosidase activity. J. Agric. Food Chem. 2012, 60, 8924–8929. [Google Scholar] [CrossRef]
- Franco, R.R.; Ribeiro Zabisky, L.F.; Pires de Lima Júnior, J.; Mota Alves, V.H.; Justino, A.B.; Saraiva, A.L.; Goulart, L.R.; Espindola, F.S. Antidiabetic effects of Syzygium cumini leaves: A non-hemolytic plant with potential against process of oxidation, glycation, inflammation and digestive enzymes catalysis. J. Ethnopharmacol. 2020, 261, 113132. [Google Scholar] [CrossRef]
- Fettach, S.; Mrabti, H.N.; Sayah, K.; Bouyahya, A.; Salhi, N.; Cherrah, Y.; El Abbes, F.M. Phenolic content, acute toxicity of Ajuga iva extracts and assessment of their antioxidant and carbohydrate digestive enzyme inhibitory effects. S. Afr. J. Bot. 2019, 125, 381–385. [Google Scholar] [CrossRef]
- Jang, G.; Lee, S.; Hong, J.; Park, B.; Kim, D.; Kim, C. Anti-Inflammatory Effect of 4,5-Dicaffeoylquinic Acid on RAW264.7 Cells and a Rat Model of Inflammation. Nutrients 2021, 13, 3537. [Google Scholar] [CrossRef]
- Angeloni, S.; Freschi, M.; Marrazzo, P.; Hrelia, S.; Beghelli, D.; Juan-Garcia, A.; Juan, C.A.; Caprioli, G.; Sagratini, G.; Angeloni, C. Antioxidant and Anti-Inflammatory Profiles of Spent Coffee Ground Extracts for the Treatment of Neurodegeneration. Oxidative Med. Cell. Longev. 2021, 2021, 6620913. [Google Scholar] [CrossRef]
- Chandra, S.; Chatterjee, P.; Dey, P.; Bhattacharya, S. Evaluation of in vitro anti-inflammatory activity of coffee against the denaturation of protein. Asian Pac. J. Trop. Biomed. 2012, 2 (Suppl. S1), S178–S180. [Google Scholar] [CrossRef]
- Yan, W.; Yang, J.; Tang, H.; Xue, L.; Chen, K.; Wang, L.; Zhao, M.; Tang, M.; Peng, A.; Long, C.; et al. Flavonoids from the stems of Millettia pachyloba Drake mediate cytotoxic activity through apoptosis and autophagy in cancer cells. J. Adv. Res. 2019, 20, 117–127. [Google Scholar] [CrossRef] [PubMed]
- Arriola, N.D.A.; Chater, P.I.; Wilcox, M.; Lucini, L.; Rocchetti, G.; Dalmina, M.; Pearson, J.P.; Amboni, R.D.D.C. Encapsulation of Stevia rebaudiana Bertoni aqueous crude extracts by ionic gelation—Effects of alginate blends and gelling solutions on the polyphenolic profile. Food Chem. 2019, 275, 123–134. [Google Scholar] [CrossRef] [PubMed]
- Yin, L.J.; Han, H.Y.; Zheng, X.; Wang, G.; Li, Y.; Wang, W. Flavonoids analysis and antioxidant, antimicrobial, and anti-inflammatory activities of crude and purified extracts from Veronicastrum latifolium. Ind. Crops Prod. 2019, 137, 652–661. [Google Scholar] [CrossRef]
- Ju, J.; Xu, X.M.; Xie, Y.F.; Guo, Y.H.; Cheng, Y.L.; Qian, H.; Yao, W.R. Inhibitory effects of cinnamon and clove essential oils on mold growth on baked foods. Food Chem. 2018, 240, 850–855. [Google Scholar] [CrossRef] [PubMed]
- Bustanji, Y.; Al-Masri, I.M.; Mohammad, M.; Hudaib, M.; Tawaha, K.; Tarazi, H.; AlKhatib, H.S. Pancreatic lipase inhibition activity of trilactone terpenes of Ginkgo biloba. J. Enzym. Inhib. Med. Chem. 2011, 26, 453–459. [Google Scholar] [CrossRef] [PubMed]
- Gutiérrez-Grijalva, E.P.; Antunes-Ricardo, M.; Acosta-Estrada, B.A.; Gutiérrez-Uribe, J.A.; Basilio Heredia, J. Cellular antioxidant activity and in vitro inhibition of α-glucosidase, α-amylase and pancreatic lipase of oregano polyphenols under simulated gastrointestinal digestion. Food Res. Int. 2019, 116, 676–686. [Google Scholar] [CrossRef]
- Attar, U.A.; Ghane, S.G. In vitro antioxidant, antidiabetic, antiacetylcholine esterase, anticancer activities and RP-HPLC analysis of phenolics from the wild bottle gourd (Lagenaria siceraria (Molina) Standl.). S. Afr. J. Bot. 2019, 125, 360–370. [Google Scholar] [CrossRef]
- Ghane, S.G.; Attar, U.A.; Yadav, P.B.; Lekhak, M.M. Antioxidant, anti-diabetic, acetylcholinesterase inhibitory potential and estimation of alkaloids (lycorine and galanthamine) from Crinum species: An important source of anticancer and anti-Alzheimer drug. Ind. Crops Prod. 2018, 125, 168–177. [Google Scholar] [CrossRef]
- Call, L.; Reiter, E.V.; Wenger-Oehn, G.; Strnad, I.; Grausgruber, H.; Schoenlechner, R.; D’Amico, S. Development of an enzymatic assay for the quantitative determination of trypsin inhibitory activity in wheat. Food Chem. 2019, 299, 125038. [Google Scholar] [CrossRef]
- Gu, Y.; Hurst, W.J.; Stuart, D.A.; Lambert, J.D. Inhibition of Key Digestive Enzymes by Cocoa Extracts and Procyanidins. J. Agric. Food Chem. 2011, 59, 5305–5311. [Google Scholar] [CrossRef] [PubMed]
- Elisha, I.L.; Dzoyem, J.-P.; McGaw, L.J.; Botha, F.S.; Eloff, J.N. The anti-arthritic, anti-inflammatory, antioxidant activity and relationships with total phenolics and total flavonoids of nine South African plants used traditionally to treat arthritis. BMC Complement. Altern. Med. 2016, 16, 307–317. [Google Scholar] [CrossRef]
- An, B.J.; Kwak, J.H.; Son, J.H.; Park, J.M.; Lee, J.Y.; Jo, C.; Byun, M.W. Biological and anti-microbial activity of irradiated green tea polyphenols. Food Chem. 2004, 88, 549–555. [Google Scholar] [CrossRef]


| PPS | Positive Control * | |||
|---|---|---|---|---|
| MIC (mg/mL) | MBC/MFC (mg/mL) | MIC (µg/mL) | MBC/MFC (µg/mL) | |
| B. subtilis | 1.67 | 3.33 | 4 | 8 |
| S. aureus | 1.67 | 3.33 | 4 | 8 |
| P. aeruginosa | 3.33 | ≥3.33 | 1.6 | 3.2 |
| E. coli | 3.33 | ≥3.33 | 1.6 | 3.2 |
| M. furfur | 6.67 | 13.33 | 33 | 67 |
| A. niger | 6.67 | 13.33 | 33 | 67 |
| Sample | α-Amylase | α-Glucosidase | Lipase | Trypsin |
|---|---|---|---|---|
| PPS | 4.96 ± 0.09 | 0.19 ± 0.04 | 3.25 ± 0.06 | 0.065 ± 0.001 |
| EGCG | 4.26 ± 0.07 | 0.07 ± 0.00 | 3.72 ± 0.14 | 0.066 ± 0.001 |
| Parameters | Lipase | Trypsin | ||||
|---|---|---|---|---|---|---|
| 0 | 0.5 | 1.5 | 0 | 0.02 | 0.05 | |
| Vmax | 1.67 | 1.59 | 1.68 | 1.07 | 0.32 | 0.19 |
| Km | 30.77 | 36.8 | 83.9 | 0.03 | 0.007 | 0.009 |
| Inhibition type | competitive | uncompetitive |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Myint, K.Z.; Zhou, Z.; Shi, Q.; Chen, J.; Dong, X.; Xia, Y. Stevia Polyphenols, Their Antimicrobial and Anti-Inflammatory Properties, and Inhibitory Effect on Digestive Enzymes. Molecules 2023, 28, 7572. https://doi.org/10.3390/molecules28227572
Myint KZ, Zhou Z, Shi Q, Chen J, Dong X, Xia Y. Stevia Polyphenols, Their Antimicrobial and Anti-Inflammatory Properties, and Inhibitory Effect on Digestive Enzymes. Molecules. 2023; 28(22):7572. https://doi.org/10.3390/molecules28227572
Chicago/Turabian StyleMyint, Khaing Zar, Zhuoyu Zhou, Qiandai Shi, Junming Chen, Xinyu Dong, and Yongmei Xia. 2023. "Stevia Polyphenols, Their Antimicrobial and Anti-Inflammatory Properties, and Inhibitory Effect on Digestive Enzymes" Molecules 28, no. 22: 7572. https://doi.org/10.3390/molecules28227572
APA StyleMyint, K. Z., Zhou, Z., Shi, Q., Chen, J., Dong, X., & Xia, Y. (2023). Stevia Polyphenols, Their Antimicrobial and Anti-Inflammatory Properties, and Inhibitory Effect on Digestive Enzymes. Molecules, 28(22), 7572. https://doi.org/10.3390/molecules28227572









