Microgreens—A Comprehensive Review of Bioactive Molecules and Health Benefits
Abstract
1. Introduction
2. Literature Search
3. Sprouts vs. Microgreens vs. Baby Greens vs. Mature Plants
4. Different Varieties of Microgreens
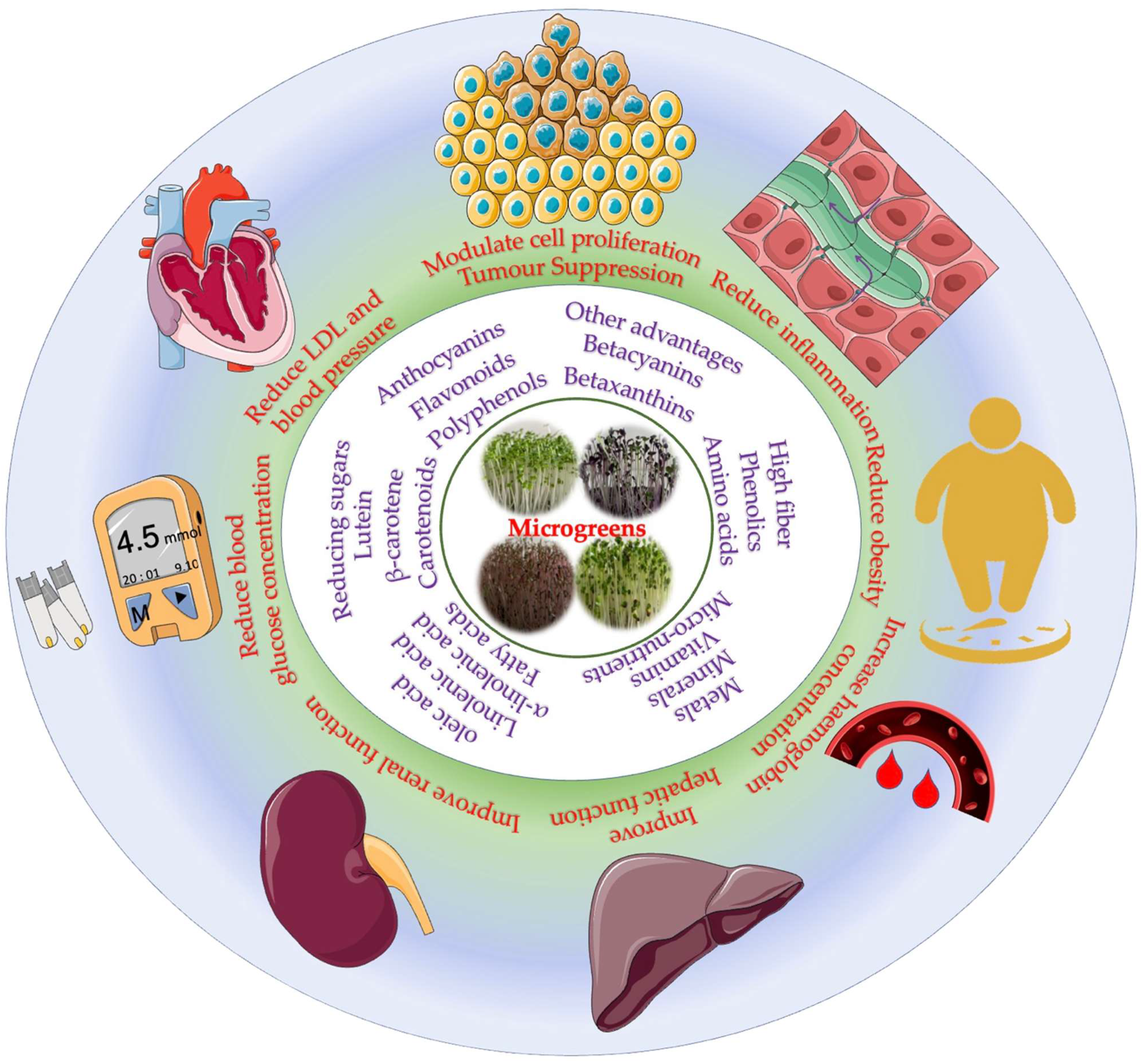
5. Nutrient and Phytochemical Composition of Microgreens
5.1. Ascorbic Acid—Vitamin C
5.2. Phylloquinone—Vitamin K
5.3. α-Tocopherol—Vitamin E
5.4. β-Carotene—Pro Vitamin A
5.5. Phenolic Antioxidants and Sugar Content
5.6. Anthocyanins and Glucosinolates
5.7. Micro and Macroelements
6. Growth Conditions for the Production of Microgreens
6.1. Seed Treatment
6.2. Light
6.3. Growth Medium
6.4. Microbial Colonization and Pests and Diseases
6.5. Harvesting and Post-Harvesting Techniques
7. Effect of Microgreens on Metabolic Health-Promoting Applications
7.1. Diabetes
7.2. Chronic Kidney Disorder
7.3. Cancer
7.4. Cardiovascular Diseases
7.5. Inflammation
7.6. Obesity
7.7. Iron Deficiency
7.8. Biofortification of Essential Nutrients
7.9. Other Applications
8. Conclusions and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ebert, A.W. Sprouts and Microgreens—Novel Food Sources for Healthy Diets. Plants 2022, 11, 571. [Google Scholar] [CrossRef] [PubMed]
- Marchioni, I.; Martinelli, M.; Ascrizzi, R.; Gabbrielli, C.; Flamini, G.; Pistelli, L.; Pistelli, L. Small Functional Foods: Comparative Phytochemical and Nutritional Analyses of Five Microgreens of the Brassicaceae Family. Foods 2021, 10, 427. [Google Scholar] [CrossRef] [PubMed]
- Treadwell, D.; Hochmuth, R.; Landrum, L.; Laughlin, W. Microgreens: A New Specialty Crop: HS1164, rev. 9/2020. Edis 2020, 2020, 1–3. [Google Scholar] [CrossRef]
- Galieni, A.; Falcinelli, B.; Stagnari, F.; Datti, A.; Benincasa, P. Sprouts and microgreens: Trends, opportunities, and horizons for novel research. Agronomy 2020, 10, 1424. [Google Scholar] [CrossRef]
- Samuolienė, G.; Brazaitytė, A.; Viršilė, A.; Miliauskienė, J.; Vaštakaitė-Kairienė, V.; Duchovskis, P. Nutrient Levels in Brassicaceae Microgreens Increase Under Tailored Light-Emitting Diode Spectra. Front. Plant Sci. 2019, 10, 1475. [Google Scholar] [CrossRef]
- Kyriacou, M.C.; Rouphael, Y.; Di Gioia, F.; Kyratzis, A.; Serio, F.; Renna, M.; De Pascale, S.; Santamaria, P. Micro-scale vegetable production and the rise of microgreens. Trends Food Sci. Technol. 2016, 57, 103–115. [Google Scholar] [CrossRef]
- Riggio, G.M.; Wang, Q.; Kniel, K.E.; Gibson, K.E. Microgreens—A review of food safety considerations along the farm to fork continuum. Int. J. Food Microbiol. 2018, 290, 76–85. [Google Scholar] [CrossRef]
- Norman, K.; Haß, U.; Pirlich, M. Malnutrition in Older Adults—Recent Advances and Remaining Challenges. Nutrients 2021, 13, 2764. [Google Scholar] [CrossRef]
- Wells, J.C.; Sawaya, A.L.; Wibaek, R.; Mwangome, M.; Poullas, M.S.; Yajnik, C.S.; Demaio, A. The double burden of malnutrition: Aetiological pathways and consequences for health. Lancet 2019, 395, 75–88. [Google Scholar] [CrossRef]
- Bailey, R.L.; West, K.P., Jr.; Black, R.E. The Epidemiology of Global Micronutrient Deficiencies. Ann. Nutr. Metab. 2015, 66 (Suppl. S2), 22–33. [Google Scholar] [CrossRef]
- Lee-Kwan, S.H.; Moore, L.V.; Blanck, H.M.; Harris, D.M.; Galuska, D. Disparities in State-Specific Adult Fruit and Vegetable Consumption—United States, 2015. MMWR Morb. Mortal. Wkly. Rep. 2017, 66, 1241–1247. [Google Scholar] [CrossRef]
- Szpyrka, E.; Kurdziel, A.; Rupar, J.; Slowik-Borowiec, M. Pesticide residues in fruit and vegetable crops from the central and eastern region of Poland. Rocz Panstw Zakl Hig 2015, 66, 107–113. [Google Scholar]
- Ssemugabo, C.; Guwatudde, D.; Ssempebwa, J.C.; Bradman, A. Pesticide Residue Trends in Fruits and Vegetables from Farm to Fork in Kampala Metropolitan Area, Uganda—A Mixed Methods Study. Int. J. Environ. Res. Public Health 2022, 19, 1350. [Google Scholar] [CrossRef]
- Sandoval-Insausti, H.; Chiu, Y.-H.; Lee, D.H.; Wang, S.; Hart, J.E.; Mínguez-Alarcón, L.; Laden, F.; Korat, A.V.A.; Birmann, B.; Eliassen, A.H.; et al. Intake of fruits and vegetables by pesticide residue status in relation to cancer risk. Environ. Int. 2021, 156, 106744. [Google Scholar] [CrossRef]
- Luo, X.; Wang, Y. Analysis of the Effect of Health Management of Chronic Diseases and Metabolic Disorders under the Guidance of B-Ultrasound. J. Health Eng. 2021, 2021, 6648611. [Google Scholar] [CrossRef]
- Sharma, S.; Dhingra, P.; Koranne, S. Microgreens: Exciting new food for 21 st Century. Ecol. Environ. Conserv. 2020, 26, S248–S251. [Google Scholar]
- Aloo, S.O.; Ofosu, F.K.; Kilonzi, S.M.; Shabbir, U.; Oh, D.H. Edible plant sprouts: Health benefits, trends, and opportunities for novel exploration. Nutrients 2021, 13, 2882. [Google Scholar] [CrossRef]
- Wojdyło, A.; Nowicka, P.; Tkacz, K.; Turkiewicz, I.P. Sprouts vs. Microgreens as Novel Functional Foods: Variation of Nutritional and Phytochemical Profiles and Their In Vitro Bioactive Properties. Molecules 2020, 25, 4648. [Google Scholar] [CrossRef]
- Di Bella, M.C.; Niklas, A.; Toscano, S.; Picchi, V.; Romano, D.; Scalzo, R.L.; Branca, F. Morphometric Characteristics, Polyphenols and Ascorbic Acid Variation in Brassica oleracea L. Novel Foods: Sprouts, Microgreens and Baby Leaves. Agronomy 2020, 10, 782. [Google Scholar] [CrossRef]
- Artés-Hernández, F.; Castillejo, N.; Martínez-Zamora, L. UV and visible spectrum led lighting as abiotic elicitors of bioactive compounds in sprouts, microgreens, and baby leaves—A comprehensive review including their mode of action. Foods 2022, 11, 265. [Google Scholar] [CrossRef]
- Martínez-Ispizua, E.; Calatayud, Á.; Marsal, J.I.; Cannata, C.; Basile, F.; Abdelkhalik, A.; Soler, S.; Valcárcel, J.V.; Martínez-Cuenca, M.-R. The Nutritional Quality Potential of Microgreens, Baby Leaves, and Adult Lettuce: An Underexploited Nutraceutical Source. Foods 2022, 11, 423. [Google Scholar] [CrossRef] [PubMed]
- Dimita, R.; Allah, S.M.; Luvisi, A.; Greco, D.; De Bellis, L.; Accogli, R.; Mininni, C.; Negro, C. Volatile Compounds and Total Phenolic Content of Perilla frutescens at Microgreens and Mature Stages. Horticulturae 2022, 8, 71. [Google Scholar] [CrossRef]
- Tan, L.; Nuffer, H.; Feng, J.; Kwan, S.H.; Chen, H.; Tong, X.; Kong, L. Antioxidant properties and sensory evaluation of microgreens from commercial and local farms. Food Sci. Hum. Wellness 2019, 9, 45–51. [Google Scholar] [CrossRef]
- Michell, K.A.; Isweiri, H.; Newman, S.E.; Bunning, M.; Bellows, L.L.; Dinges, M.M.; Grabos, L.E.; Rao, S.; Foster, M.T.; Heuberger, A.L.; et al. Microgreens: Consumer sensory perception and acceptance of an emerging functional food crop. J. Food Sci. 2020, 85, 926–935. [Google Scholar] [CrossRef] [PubMed]
- Turner, E.R.; Luo, Y.; Buchanan, R.L. Microgreen nutrition, food safety, and shelf life: A review. J. Food Sci. 2020, 85, 870–882. [Google Scholar] [CrossRef]
- Choe, U.; Yu, L.; Wang, T.T.Y. The Science behind Microgreens as an Exciting New Food for the 21st Century. J. Agric. Food Chem. 2018, 66, 11519–11530. [Google Scholar] [CrossRef]
- Rocchetti, G.; Tomas, M.; Zhang, L.; Zengin, G.; Lucini, L.; Capanoglu, E. Red beet (Beta vulgaris) and amaranth (Amaranthus sp.) microgreens: Effect of storage and in vitro gastrointestinal digestion on the untargeted metabolomic profile. Food Chem. 2020, 332, 127415. [Google Scholar] [CrossRef]
- Sarker, U.; Oba, S. Protein, dietary fiber, minerals, antioxidant pigments and phytochemicals, and antioxidant activity in selected red morph Amaranthus leafy vegetable. PLoS ONE 2019, 14, e0222517. [Google Scholar] [CrossRef]
- Pathan, S.; Siddiqui, R.A. Nutritional Composition and Bioactive Components in Quinoa (Chenopodium quinoa Willd.) Greens: A Review. Nutrients 2022, 14, 558. [Google Scholar] [CrossRef]
- Petropoulos, S.A.; El-Nakhel, C.; Graziani, G.; Kyriacou, M.C.; Rouphael, Y. The effects of nutrient solution feeding regime on yield, mineral profile, and phytochemical composition of spinach microgreens. Horticulturae 2021, 7, 162. [Google Scholar] [CrossRef]
- Bulgari, R.; Baldi, A.; Ferrante, A.; Lenzi, A. Yield and quality of basil, Swiss chard, and rocket microgreens grown in a hydroponic system. N. Z. J. Crop Hortic. Sci. 2016, 45, 119–129. [Google Scholar] [CrossRef]
- Ghoora, M.D.; Haldipur, A.C.; Srividya, N. Comparative evaluation of phytochemical content, antioxidant capacities and overall antioxidant potential of select culinary microgreens. J. Agric. Food Res. 2020, 2, 100046. [Google Scholar] [CrossRef]
- Samuolienė, G.; Brazaitytė, A.; Viršilė, A.; Jankauskienė, J.; Sakalauskienė, S.; Duchovskis, P. Red Light-Dose or Wavelength-Dependent Photoresponse of Antioxidants in Herb Microgreens. PLoS ONE 2016, 11, e0163405. [Google Scholar] [CrossRef] [PubMed]
- Paradiso, V.M.; Castellino, M.; Renna, M.; Gattullo, C.E.; Calasso, M.; Terzano, R.; Allegretta, I.; Leoni, B.; Caponio, F.; Santamaria, P. Nutritional characterization and shelf-life of packaged microgreens. Food Funct. 2018, 9, 5629–5640. [Google Scholar] [CrossRef]
- Kyriacou, M.C.; El-Nakhel, C.; Pannico, A.; Graziani, G.; Soteriou, G.A.; Giordano, M.; Palladino, M.; Ritieni, A.; De Pascale, S.; Rouphael, Y. Phenolic Constitution, Phytochemical and Macronutrient Content in Three Species of Microgreens as Modulated by Natural Fiber and Synthetic Substrates. Antioxidants 2020, 9, 252. [Google Scholar] [CrossRef]
- Yadav, L.P.; Koley, T.K.; Tripathi, A.; Singh, S. Antioxidant Potentiality and Mineral Content of Summer Season Leafy Greens: Comparison at Mature and Microgreen Stages Using Chemometric. Agric. Res. 2018, 8, 165–175. [Google Scholar] [CrossRef]
- Zhang, X.; Bian, Z.; Yuan, X.; Chen, X.; Lu, C. A review on the effects of light-emitting diode (LED) light on the nutrients of sprouts and microgreens. Trends Food Sci. Technol. 2020, 99, 203–216. [Google Scholar] [CrossRef]
- Pannico, A.; Graziani, G.; El-Nakhel, C.; Giordano, M.; Ritieni, A.; Kyriacou, M.; Rouphael, Y. Nutritional stress suppresses nitrate content and positively impacts ascorbic acid concentration and phenolic acids profile of lettuce microgreens. Italus Hortus 2020, 27, 41–52. [Google Scholar] [CrossRef]
- Xiao, Z.; Lester, G.E.; Park, E.; Saftner, R.A.; Luo, Y.; Wang, Q. Evaluation and correlation of sensory attributes and chemical compositions of emerging fresh produce: Microgreens. Postharvest Biol. Technol. 2015, 110, 140–148. [Google Scholar] [CrossRef]
- Szewczyk, K.; Chojnacka, A.; Górnicka, M. Tocopherols and tocotrienols—Bioactive dietary compounds; what is certain, what is doubt? Int. J. Mol. Sci. 2021, 22, 6222. [Google Scholar] [CrossRef] [PubMed]
- Miyazawa, T.; Burdeos, G.C.; Itaya, M.; Nakagawa, K.; Miyazawa, T. Vitamin E: Regulatory redox interactions. IUBMB Life 2019, 71, 430–441. [Google Scholar] [CrossRef] [PubMed]
- Maurya, V.K.; Shakya, A.; Aggarwal, M.; Gothandam, K.M.; Bohn, T.; Pareek, S. Fate of β-carotene within loaded delivery systems in food: State of knowledge. Antioxidants 2021, 10, 426. [Google Scholar] [CrossRef]
- Chew, B.P.; Park, J.S. Carotenoid Action on the Immune Response. J. Nutr. 2004, 134, 257S–261S. [Google Scholar] [CrossRef]
- Kumar, N.; Goel, N. Phenolic acids: Natural versatile molecules with promising therapeutic applications. Biotechnol. Rep. 2019, 24, e00370. [Google Scholar] [CrossRef]
- Gao, M.; He, R.; Shi, R.; Zhang, Y.; Song, S.; Su, W.; Liu, H. Differential Effects of Low Light Intensity on Broccoli Microgreens Growth and Phytochemicals. Agronomy 2021, 11, 537. [Google Scholar] [CrossRef]
- Panchal, S.K.; John, O.D.; Mathai, M.L.; Brown, L. Anthocyanins in Chronic Diseases: The Power of Purple. Nutrients 2022, 14, 2161. [Google Scholar] [CrossRef]
- El-Nakhel, C.; Pannico, A.; Graziani, G.; Kyriacou, M.C.; Gaspari, A.; Ritieni, A.; Rouphael, Y. Nutrient supplementation configures the bioactive profile and production characteristics of three Brassica L. microgreens species grown in peat-based media. Agronomy 2021, 11, 346. [Google Scholar] [CrossRef]
- Tomas, M.; Zhang, L.; Zengin, G.; Rocchetti, G.; Capanoglu, E.; Lucini, L. Metabolomic insight into the profile, in vitro bioaccessibility and bioactive properties of polyphenols and glucosinolates from four Brassicaceae microgreens. Food Res. Int. 2021, 140, 110039. [Google Scholar] [CrossRef]
- Liu, Z.; Shi, J.; Wan, J.; Pham, Q.; Zhang, Z.; Sun, J.; Yu, L.; Luo, Y.; Wang, T.T.; Chen, P. Profiling of Polyphenols and Glucosinolates in Kale and Broccoli Microgreens Grown under Chamber and Windowsill Conditions by Ultrahigh-Performance Liquid Chromatography High-Resolution Mass Spectrometry. ACS Food Sci. Technol. 2021, 2, 101–113. [Google Scholar] [CrossRef]
- Doggui, R.; Al-Jawaldeh, H.; El Ati, J.; Barham, R.; Nasreddine, L.; Alqaoud, N.; Aguenaou, H.; El Ammari, L.; Jabbour, J.; Al-Jawaldeh, A. Meta-Analysis and Systematic Review of Micro- and Macro-Nutrient Intakes and Trajectories of Macro-Nutrient Supply in the Eastern Mediterranean Region. Nutrients 2021, 13, 1515. [Google Scholar] [CrossRef]
- Ziadlou, M.; Hosseini-Esfahani, F.; Khosravi, H.M.; Hosseinpanah, F.; Barzin, M.; Khalaj, A.; Valizadeh, M. Dietary macro- and micro-nutrients intake adequacy at 6th and 12th month post-bariatric surgery. BMC Surg. 2020, 20, 232. [Google Scholar] [CrossRef] [PubMed]
- Rocha, J.; Basra, T.; El Kurdi, B.; Venegas-Borsellino, C. Effects of Potential Micro- and Macro-nutrients in Combatting COVID-19. Curr. Surg. Rep. 2021, 9, 24. [Google Scholar] [CrossRef] [PubMed]
- Renna, M.; Paradiso, V.M. Ongoing research on microgreens: Nutritional properties, shelf-life, sustainable production, innovative growing and processing approaches. Foods 2020, 9, 826. [Google Scholar] [CrossRef] [PubMed]
- Carillo, P.; El-Nakhel, C.; De Micco, V.; Giordano, M.; Pannico, A.; De Pascale, S.; Graziani, G.; Ritieni, A.; Soteriou, G.A.; Kyriacou, M.C.; et al. Morpho-Metric and Specialized Metabolites Modulation of Parsley Microgreens through Selective LED Wavebands. Agronomy 2022, 12, 1502. [Google Scholar] [CrossRef]
- Zeng, W.; Yang, J.; Yan, G.; Zhu, Z. CaSO4 Increases Yield and Alters the Nutritional Contents in Broccoli (Brassica oleracea L. Var. italica) Microgreens under NaCl Stress. Foods 2022, 11, 3485. [Google Scholar] [CrossRef]
- Lee, J.S.; Pill, W.G.; Cobb, B.B.; Olszewski, M. Seed treatments to advance greenhouse establishment of beet and chard microgreens. J. Hortic. Sci. Biotechnol. 2004, 79, 565–570. [Google Scholar] [CrossRef]
- Verlinden, S. Microgreens: Definitions, Product Types, and Production Practices. Hortic. Rev. 2020, 47, 85–124. [Google Scholar]
- Işık, S.; Aytemiş, Z.; Çetin, B.; Topalcengiz, Z. Possible explanation for limited reduction of pathogens on radish microgreens after spray application of chlorinated water during growth with disperse contamination spread of abiotic surrogate on leaves. J. Food Saf. 2022, 42, e12984. [Google Scholar] [CrossRef]
- Weber, C.F. Broccoli microgreens: A mineral-rich crop that can diversify food systems. Front. Nutr. 2017, 4, 7. [Google Scholar] [CrossRef]
- Kou, L.; Luo, Y.; Yang, T.; Xiao, Z.; Turner, E.R.; Lester, G.E.; Wang, Q.; Camp, M.J. Postharvest biology, quality and shelf life of buckwheat microgreens. Lwt 2013, 51, 73–78. [Google Scholar] [CrossRef]
- Bayineni, V.K.; Kavana Herur, N. Natural Synedrella Residues as a Growing Substrate Ingredient: An Eco-friendly Way to Improve Yield and Quality of Beet (Beta vulgaris) Microgreens. Eur. J. Agric. Food Sci. 2022, 4, 1–5. [Google Scholar] [CrossRef]
- Thuong, V.T.; Minh, H.G. Effects of growing substrates and seed density on yield and quality of radish (Raphanus sativus) microgreens. Res. Crops 2020, 21, 579–586. [Google Scholar] [CrossRef]
- Santin, M.; Sciampagna, M.C.; Mannucci, A.; Puccinelli, M.; Angelini, L.G.; Tavarini, S.; Castagna, A. Supplemental UV-B Exposure Influences the Biomass and the Content of Bioactive Compounds in Linum usitatissimum L. Sprouts and Microgreens. Horticulturae 2022, 8, 213. [Google Scholar] [CrossRef]
- Brazaityte, A.; Virsile, A.; Jankauskiene, J.; Sakalauskiene, S.; Samuoliene, G.; Sirtautas, R.; Duchovskis, P. Effect of supplemental UV-A irradiation in solid-state lighting on the growth and phytochemical content of microgreens. Int. Agrophysics 2015, 29, 13–22. [Google Scholar] [CrossRef]
- Samuolienė, G.; Viršilė, A.; Brazaitytė, A.; Jankauskienė, J.; Sakalauskienė, S.; Vaštakaitė-Kairienė, V.; Novičkovas, A.; Viškelienė, A.; Sasnauskas, A.; Duchovskis, P.; et al. Blue light dosage affects carotenoids and tocopherols in microgreens. Food Chem. 2017, 228, 50–56. [Google Scholar] [CrossRef]
- Mohammad, Z.H.; Prado, I.D.; Sirsat, S.A. Comparative microbial analyses of hydroponic versus in-soil grown Romaine lettuce obtained at retail. Heliyon 2022, 8, e11050. [Google Scholar] [CrossRef]
- Riggio, G.; Jones, S.; Gibson, K. Risk of Human Pathogen Internalization in Leafy Vegetables During Lab-Scale Hydroponic Cultivation. Horticulturae 2019, 5, 25. [Google Scholar] [CrossRef]
- Muchjajib, U.; Suknikom, S.; Butsai, J. Evaluation of organic media alternatives for the production of microgreens in Thailand. Acta Hortic. 2015, 1102, 157–162. [Google Scholar] [CrossRef]
- Hoang, G.M.; Vu, T.T. Selection of suitable growing substrates and quality assessment of Brassica microgreens cultivated in greenhouse. Acad. J. Biol. 2022, 44, 133–142. [Google Scholar] [CrossRef]
- Reed, E.; Ferreira, C.M.; Bell, R.; Brown, E.W.; Zheng, J. Plant-Microbe and Abiotic Factors Influencing Salmonella Survival and Growth on Alfalfa Sprouts and Swiss Chard Microgreens. Appl. Environ. Microbiol. 2018, 84, e02814–e02817. [Google Scholar] [CrossRef]
- Moraru, P.I.; Rusu, T.; Mintas, O.S. Trial Protocol for Evaluating Platforms for Growing Microgreens in Hydroponic Conditions. Foods 2022, 11, 1327. [Google Scholar] [CrossRef] [PubMed]
- Mohanty, A.; Mahahlik, G.; Parida, S. Nutritional Analysis of Few Edible Microgreens in Variable Growth Medium using XRF Technique. Asian J. Biol. Life Sci. 2021, 9, 360–364. [Google Scholar] [CrossRef]
- Wright, K.M.; Holden, N.J. Quantification and colonisation dynamics of Escherichia coli O157:H7 inoculation of microgreens species and plant growth substrates. Int. J. Food Microbiol. 2018, 273, 1–10. [Google Scholar] [CrossRef] [PubMed]
- McGehee, C.S.; Raudales, R.E.; Elmer, W.H.; McAvoy, R.J. Efficacy of biofungicides against root rot and damping-off of microgreens caused by Pythium spp. Crop. Prot. 2019, 121, 96–102. [Google Scholar] [CrossRef]
- Sharma, S.; Shree, B.; Sharma, D.; Kumar, S.; Kumar, V.; Sharma, R.; Saini, R. Vegetable microgreens: The gleam of next generation super foods, their genetic enhancement, health benefits and processing approaches. Food Res. Int. 2022, 155, 111038. [Google Scholar] [CrossRef] [PubMed]
- Yan, H.; Li, W.; Chen, H.; Liao, Q.; Xia, M.; Wu, D.; Liu, C.; Chen, J.; Zou, L.; Peng, L.; et al. Effects of Storage Temperature, Packaging Material and Wash Treatment on Quality and Shelf Life of Tartary Buckwheat Microgreens. Foods 2022, 11, 3630. [Google Scholar] [CrossRef]
- Berba, K.J.; Uchanski, M.E. Post-harvest physiology of microgreens. J. Young Investig. 2012, 24, 5. [Google Scholar]
- Zohoori, F.V. Chapter 1: Nutrition and Diet. Monogr. Oral. Sci. 2020, 28, 1–13. [Google Scholar] [CrossRef]
- Chen, Y.; Michalak, M.; Agellon, L.B. Importance of Nutrients and Nutrient Metabolism on Human Health. Yale J. Biol. Med. 2018, 91, 95–103. [Google Scholar]
- Singh, A.; Banerjee, P.; Anas, M.; Singh, N.; Qamar, I. Traditional Nutritional and Health Practices Targeting Lifestyle Behavioral Changes in Humans. J. Lifestyle Med. 2020, 10, 67–73. [Google Scholar] [CrossRef]
- Witkamp, R.F. Nutrition to optimise human health—How to obtain physiological substantiation? Nutrients 2021, 13, 2155. [Google Scholar] [CrossRef]
- Minich, D.M. A Review of the Science of Colorful, Plant-Based Food and Practical Strategies for “Eating the Rainbow”. J. Nutr. Metab. 2019, 2019, 2125070. [Google Scholar] [CrossRef]
- Cena, H.; Calder, P.C. Defining a Healthy Diet: Evidence for the Role of Contemporary Dietary Patterns in Health and Disease. Nutrients 2020, 12, 334. [Google Scholar] [CrossRef]
- Hotamisligil, G.S. Inflammation and metabolic disorders. Nature 2006, 444, 860–867. [Google Scholar] [CrossRef]
- VanWormer, J.J.; Boucher, J.L.; Sidebottom, A.C.; Sillah, A.; Knickelbine, T. Lifestyle changes and prevention of metabolic syndrome in the Heart of New Ulm Project. Prev. Med. Rep. 2017, 6, 242–245. [Google Scholar] [CrossRef]
- Buscemi, S.; Sprini, D.; Grosso, G.; Galvano, F.; Nicolucci, A.; Lucisano, G.; Massenti, F.M.; Amodio, E.; Rini, G.B. Impact of lifestyle on metabolic syndrome in apparently healthy people. Eat. Weight. Disord.-Stud. Anorex. Bulim. Obes. 2014, 19, 225–232. [Google Scholar] [CrossRef]
- IDF (International Diabetes Federation) Diabetes Atlas 2021-10th Edition. Available online: https://diabetesatlas.org/atlas/tenth-edition/?dlmodal=active&dlsrc=https%3A%2F%2Fdiabetesatlas.org%2Fidfawp%2Fresource-files%2F2021%2F07%2FIDF_Atlas_10th_Edition_2021.pdf (accessed on 19 December 2022).
- Magkos, F.; Hjorth, M.F.; Astrup, A. Diet and exercise in the prevention and treatment of type 2 diabetes mellitus. Nat. Rev. Endocrinol. 2020, 16, 545–555. [Google Scholar] [CrossRef]
- Hamdy, O.; Barakatun-Nisak, M.Y. Nutrition in diabetes. Endocrinol. Metab. Clin. 2016, 45, 799–817. [Google Scholar] [CrossRef]
- Schwingshackl, L.; Hoffmann, G.; Lampousi, A.-M.; Knüppel, S.; Iqbal, K.; Schwedhelm, C.; Bechthold, A.; Schlesinger, S.; Boeing, H. Food groups and risk of type 2 diabetes mellitus: A systematic review and meta-analysis of prospective studies. Eur. J. Epidemiol. 2017, 32, 363–375. [Google Scholar] [CrossRef]
- Wadhawan, S.; Tripathi, J.; Gautam, S. In vitro regulation of enzymatic release of glucose and its uptake by Fenugreek microgreen and Mint leaf extract. Int. J. Food Sci. Technol. 2017, 53, 320–326. [Google Scholar] [CrossRef]
- Ma, S.; Tian, S.; Sun, J.; Pang, X.; Hu, Q.; Li, X.; Lu, Y. Broccoli microgreens have hypoglycemic effect by improving blood lipid and inflammatory factors while modulating gut microbiota in mice with type 2 diabetes. J. Food Biochem. 2022, 46, e14145. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, S.M.; Abdel-Rahim, E.A.; Aly, T.A.; Naguib, A.M.; Khattab, M.S. Barley microgreen incorporation in diet-controlled diabetes and counteracted aflatoxicosis in rats. Exp. Biol. Med. 2021, 247, 385–394. [Google Scholar] [CrossRef] [PubMed]
- Khattab, M.S.; Aly, T.A.; Mohamed, S.M.; Naguib, A.M.M.; AL-Farga, A.; Abdel-Rahim, E.A. Hordeum vulgare L. microgreen mitigates reproductive dysfunction and oxidative stress in streptozotocin-induced diabetes and aflatoxicosis in male rats. Food Sci. Nutr. 2022, 10, 3355–3367. [Google Scholar] [CrossRef] [PubMed]
- Kalantar-Zadeh, K.; Jafar, T.H.; Nitsch, D.; Neuen, B.L.; Perkovic, V. Chronic kidney disease. Lancet 2021, 398, 786–802. [Google Scholar] [CrossRef] [PubMed]
- Renna, M.; Castellino, M.; Leoni, B.; Paradiso, V.M.; Santamaria, P. Microgreens Production with Low Potassium Content for Patients with Impaired Kidney Function. Nutrients 2018, 10, 675. [Google Scholar] [CrossRef] [PubMed]
- Siegel, R.L.; Miller, K.D.; Fuchs, H.E.; Jemal, A. Cancer statistics, 2021. CA Cancer J. Clin. 2021, 71, 7–33. [Google Scholar] [CrossRef]
- Vainio, H.; Weiderpass, E. Fruit and Vegetables in Cancer Prevention. Nutr. Cancer 2006, 54, 111–142. [Google Scholar] [CrossRef]
- Lu, Y.-T.; Gunathilake, M.; Kim, J. The influence of dietary vegetables and fruits on endometrial cancer risk: A meta-analysis of observational studies. Eur. J. Clin. Nutr. 2022, 1–13. [Google Scholar] [CrossRef]
- Teng, J.; Liao, P.; Wang, M. The role of emerging micro-scale vegetables in human diet and health benefits—An updated review based on microgreens. Food Funct. 2021, 12, 1914–1932. [Google Scholar] [CrossRef]
- Pierce, J.P.; Natarajan, L.; Caan, B.J.; Parker, B.A.; Greenberg, E.R.; Flatt, S.W.; Rock, C.L.; Kealey, S.; Al-Delaimy, W.K.; Bardwell, W.A.; et al. Influence of a diet very high in vegetables, fruit, and fiber and low in fat on prognosis following treatment for breast cancer: The Women’s Healthy Eating and Living (WHEL) randomized trial. JAMA 2007, 298, 289–298. [Google Scholar] [CrossRef]
- Aune, D.; Giovannucci, E.; Boffetta, P.; Fadnes, L.T.; Keum, N.; Norat, T.; Greenwood, D.C.; Riboli, E.; Vatten, L.J.; Tonstad, S. Fruit and vegetable intake and the risk of cardiovascular disease, total cancer and all-cause mortality-a systematic review and dose-response meta-analysis of prospective studies. Int. J. Epidemiol. 2017, 46, 1029–1056. [Google Scholar] [CrossRef] [PubMed]
- Soundararajan, P.; Kim, J.S. Anti-Carcinogenic Glucosinolates in Cruciferous Vegetables and Their Antagonistic Effects on Prevention of Cancers. Molecules 2018, 23, 2983. [Google Scholar] [CrossRef] [PubMed]
- De La Fuente, B.; López-García, G.; Máñez, V.; Alegría, A.; Barberá, R.; Cilla, A. Antiproliferative Effect of Bioaccessible Fractions of Four Brassicaceae Microgreens on Human Colon Cancer Cells Linked to Their Phytochemical Composition. Antioxidants 2020, 9, 368. [Google Scholar] [CrossRef] [PubMed]
- Luang-In, V.; Saengha, W.; Karirat, T.; Buranrat, B.; Matra, K.; Deeseenthum, S.; Katisart, T. Effect of cold plasma and elicitors on bioactive contents, antioxidant activity and cytotoxicity of Thai rat-tailed radish microgreens. J. Sci. Food Agric. 2021, 101, 1685–1698. [Google Scholar] [CrossRef] [PubMed]
- Kaimuangpak, K.; Tamprasit, K.; Thumanu, K.; Weerapreeyakul, N. Extracellular vesicles derived from microgreens of Raphanus sativus L. var. caudatus Alef contain bioactive macromolecules and inhibit HCT116 cells proliferation. Sci. Rep. 2022, 12, 1–16. [Google Scholar] [CrossRef]
- Roth, G.A.; Mensah, G.A.; Johnson, C.O.; Addolorato, G.; Ammirati, E.; Baddour, L.M.; Barengo, N.C.; Beaton, A.Z.; Benjamin, E.J.; Benziger, C.P.; et al. Global Burden of Cardiovascular Diseases and Risk Factors, 1990-2019: Update From the GBD 2019 Study. J. Am. Coll. Cardiol. 2020, 76, 2982–3021. [Google Scholar] [CrossRef]
- Mc Namara, K.; Alzubaidi, H.; Jackson, J.K. Cardiovascular disease as a leading cause of death: How are pharmacists getting involved? Integr. Pharm. Res. Pract. 2019, 8, 1. [Google Scholar] [CrossRef]
- Ibrahim, M.A.; Asuka, E.; Jialal, I. Hypercholesterolemia; StatPearls: Treasure Island, FL, USA, 2022. [Google Scholar]
- Tayyem, R.F.; Al-Bakheit, A.; Hammad, S.S.; Al-Shudifat, A.-E.; Azab, M.; Bawadi, H. Fruit and vegetable consumption and cardiovascular diseases among Jordanians: A case-control study. Central Eur. J. Public Health 2020, 28, 208–218. [Google Scholar] [CrossRef]
- Alissa, E.M.; Ferns, G.A. Dietary Fruits and Vegetables and Cardiovascular Diseases Risk. Crit. Rev. Food Sci. Nutr. 2017, 57, 1950–1962. [Google Scholar] [CrossRef]
- Huang, H.; Jiang, X.; Xiao, Z.; Yu, L.; Pham, Q.; Sun, J.; Chen, P.; Yokoyama, W.; Yu, L.L.; Luo, Y.S.; et al. Red Cabbage Microgreens Lower Circulating Low-Density Lipoprotein (LDL), Liver Cholesterol, and Inflammatory Cytokines in Mice Fed a High-Fat Diet. J. Agric. Food. Chem. 2016, 64, 9161–9171. [Google Scholar] [CrossRef]
- Jiang, X.; Huang, H.; Xiao, Z.; Yu, L.; Pham, Q.; Yu, L.; Luo, Y.; Wang, T.T.Y. Lipids and Cholesterol-Lowering Activity of Red Cabbage Microgreens. FASEB J. 2016, 30, 431.8. [Google Scholar] [CrossRef]
- Furman, D.; Campisi, J.; Verdin, E.; Carrera-Bastos, P.; Targ, S.; Franceschi, C.; Ferrucci, L.; Gilroy, D.W.; Fasano, A.; Miller, G.W.; et al. Chronic inflammation in the etiology of disease across the life span. Nat. Med. 2019, 25, 1822–1832. [Google Scholar] [CrossRef]
- Marotti, I.; Truzzi, F.; Tibaldi, C.; Negri, L.; Dinelli, G. Evaluation of licorice (Glycyrrhiza glabra L.) as a novel microgreen from the anti-inflammatory potential of polyphenols. AIMS Agric. Food 2021, 6, 1–13. [Google Scholar] [CrossRef]
- Li, X.; Tian, S.; Wang, Y.; Liu, J.; Wang, J.; Lu, Y. Broccoli microgreens juice reduces body weight by enhancing insulin sensitivity and modulating gut microbiota in high-fat diet-induced C57BL/6J obese mice. Eur. J. Nutr. 2021, 60, 3829–3839. [Google Scholar] [CrossRef]
- Phillips, K.M.; Tarragó-Trani, M.T.; Gebhardt, S.E.; Exler, J.; Patterson, K.Y.; Haytowitz, D.B.; Pehrsson, P.R.; Holden, J.M. Stability of vitamin C in frozen raw fruit and vegetable homogenates. J. Food Compos. Anal. 2010, 23, 253–259. [Google Scholar] [CrossRef]
- Kathi, S.; Laza, H.; Singh, S.; Thompson, L.; Li, W.; Simpson, C. Increasing vitamin C through agronomic biofortification of arugula microgreens. Sci. Rep. 2022, 12, 1–10. [Google Scholar] [CrossRef]
- Frąszczak, B.; Kleiber, T. Microgreens Biometric and Fluorescence Response to Iron (Fe) Biofortification. Int. J. Mol. Sci. 2022, 23, 14553. [Google Scholar] [CrossRef]
- Singh, N.; Aditika; Rani, S.; Chaurasia, O.P. Vegetable Microgreens Farming in High-Altitude Region of Trans-Himalayas to Maintain Nutritional Diet of Indian Troops. Proc. Natl. Acad. Sci. India Sect. B Boil. Sci. 2019, 90, 743–752. [Google Scholar] [CrossRef]
- Kyriacou, M.C.; De Pascale, S.; Kyratzis, A.; Rouphael, Y. Microgreens as a Component of Space Life Support Systems: A Cornucopia of Functional Food. Front. Plant Sci. 2017, 8, 1587. [Google Scholar] [CrossRef]
- Vergari, F.; Tibuzzi, A.; Basile, G. An overview of the functional food market: From marketing issues and commercial players to future demand from life in space. Adv. Exp. Med. Biol. 2010, 698, 308–321. [Google Scholar] [CrossRef]
- Cohu, C.M.; Lombardi, E.; Adams, W.W.; Demmig-Adams, B. Increased nutritional quality of plants for long-duration spaceflight missions through choice of plant variety and manipulation of growth conditions. Acta Astronaut. 2014, 94, 799–806. [Google Scholar] [CrossRef]
- Available online: https://science.nasa.gov/technology/technology-highlights/how-do-you-harvest-microgreens-in-microgravity (accessed on 11 January 2023).
- Ozawa, H.; Miyazawa, T.; Miyazawa, T. Effects of Dietary Food Components on Cognitive Functions in Older Adults. Nutrients 2021, 13, 2804. [Google Scholar] [CrossRef]

| Conditions | Sprouts | Microgreens | Baby Greens | Mature Plants |
|---|---|---|---|---|
| Height | 5–8 cm | 3–10 cm | 10–15 cm | Several cm |
| Production time | 3–10 days | 7–21 days | 20–40 days | Several months |
| Cultivation system | Do not require soil or medium to grow. Grow solely in water or in moisture. | Can be grown in soil or entirely in medium. | May or may not be grown in soil fields. Require medium to grow. | Grown in soil fields. Require medium to grow. |
| Light requirements | No, do not require light source. | Yes, requires light source. | Yes, requires light source. | Yes, requires light source. |
| Root appearance | Very tiny root without root hairs. | Small roots with root hairs. | Roots with root hairs. | Mature root system. |
| Agrochemicals use | No use of chemicals required. | No use of chemicals required. | Use of chemicals required. | Use of chemicals required. |
| Moisture/water use | Can be grown in little amount of water or even in little moisture content. | Small amount of water is required. | Water is required in large amount. | Abundant of water is needed. |
| Land space | Very small space is required for large scale production also. | Very small space is required for large scale production also. | Require a large area for their growth. | Grown over acres of free and open-spaced lands |
| Plant growth level at harvest time | Partial development of cotyledons with just the germinated seeds. | Full development of cotyledons with one or two true leaves. | Full development of young plant with true leaves. | Full development of mature plant that may bear fruits or vegetables. |
| Harvest type | No harvesting. Wholly edible. | Harvesting is done by removing the roots. | Removing the roots by cutting. | Harvesting is done by cutting the roots either manually or mechanically. |
| Variety | Duration of Growth | Nutrients Reported | Health Benefits | Reference |
|---|---|---|---|---|
| Amaranth (Amaranthaceae) | 10 days |
| antioxidant activity | [27,28] |
| Red Beets (Amaranthaceae) | 10 days |
| antioxidant activity, gastrointestinal activity | [27] |
| Quinoa (Amaranthaceae) | – |
| antioxidant activity | [29] |
| Spinach (Amaranthaceae) | 20 days |
| antioxidant activity | [30] |
| Swiss Chard (Amaranthaceae) | 17 days |
| – | [31] |
| Onion (Amaryllidaceae) | 10–12 days |
| – | [32] |
| Parsley (Apiaceae) | 19 days |
| antioxidant activity | [33] |
| Carrot (Apiaceae) | 7–14 days |
| antioxidant activity | [34] |
| Coriander (Apiaceae) | 3–4 days |
| – | [35] |
| Wild Rocket (Brassicaceae) | 17 days |
| – | [31] |
| Radish (Brassicaceae) | 9 days |
| – | [36] |
| Soybean (Fabaceae) | 8 days |
| antioxidant activity | [37] |
| Cucumber (Cucurbitaceae) | 9 days |
| antioxidant activity | [36] |
| Jute (Malvaceae) | 9 days |
| antioxidant activity | [36] |
| Leek (Amaryllidaceae) | – |
| antioxidant activity, anti-obesity activity, anti-diabetic activity, and anti-cholinergic activity | [18] |
| Green peas (Leguminaceae) | – |
| antioxidant activity, anti-obesity activity, anti-diabetic activity, and anti-cholinergic activity | [18] |
| Variables | Conditions/Requirements | Reference |
|---|---|---|
| Seeds |
| [56] |
| Light |
| [37,57] |
| Growing medium |
| [6] |
| Pathogen treatment |
| [58,59] |
| Harvesting |
| [6] |
| Post-harvesting |
| [60] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bhaswant, M.; Shanmugam, D.K.; Miyazawa, T.; Abe, C.; Miyazawa, T. Microgreens—A Comprehensive Review of Bioactive Molecules and Health Benefits. Molecules 2023, 28, 867. https://doi.org/10.3390/molecules28020867
Bhaswant M, Shanmugam DK, Miyazawa T, Abe C, Miyazawa T. Microgreens—A Comprehensive Review of Bioactive Molecules and Health Benefits. Molecules. 2023; 28(2):867. https://doi.org/10.3390/molecules28020867
Chicago/Turabian StyleBhaswant, Maharshi, Dilip Kumar Shanmugam, Taiki Miyazawa, Chizumi Abe, and Teruo Miyazawa. 2023. "Microgreens—A Comprehensive Review of Bioactive Molecules and Health Benefits" Molecules 28, no. 2: 867. https://doi.org/10.3390/molecules28020867
APA StyleBhaswant, M., Shanmugam, D. K., Miyazawa, T., Abe, C., & Miyazawa, T. (2023). Microgreens—A Comprehensive Review of Bioactive Molecules and Health Benefits. Molecules, 28(2), 867. https://doi.org/10.3390/molecules28020867








