A New Approach to the Synthesis of Nanocrystalline Cobalt Boride in the Course of the Thermal Decomposition of Cobalt Complexes [Co(DMF)6]2+ with Boron Cluster Anions
Abstract
1. Introduction
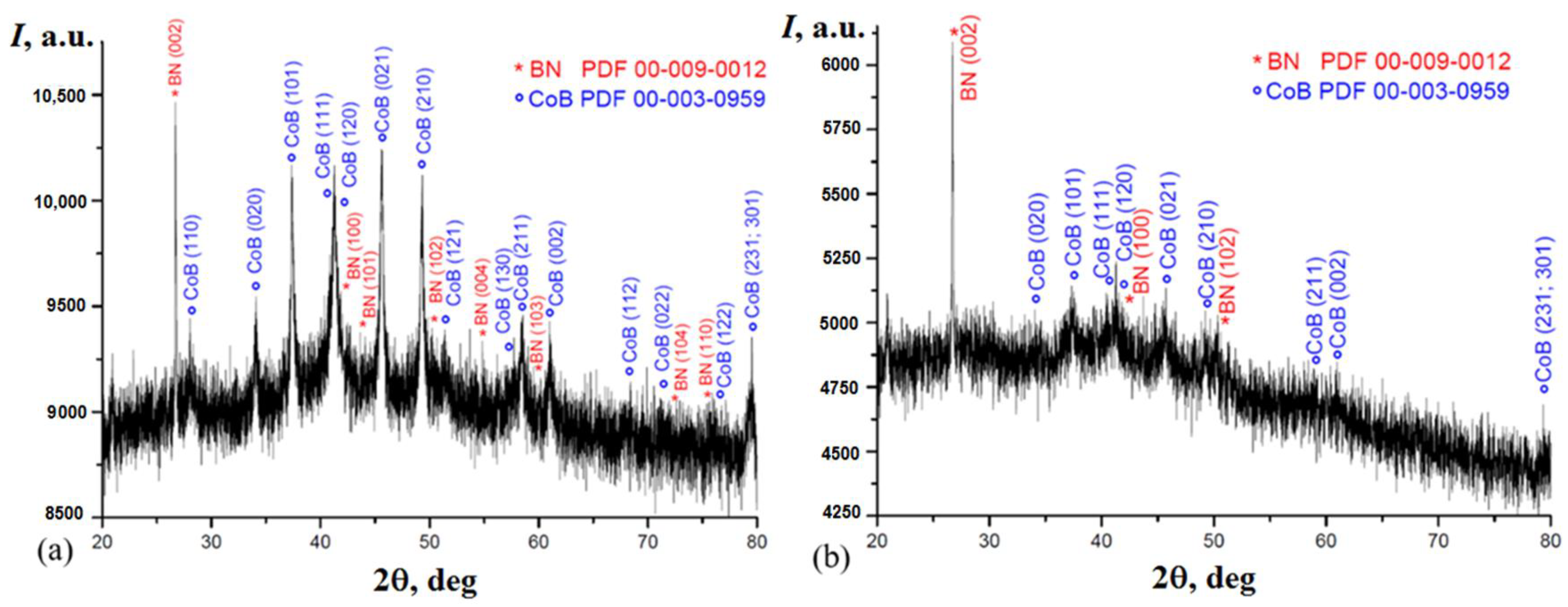
2. Results and Discussion
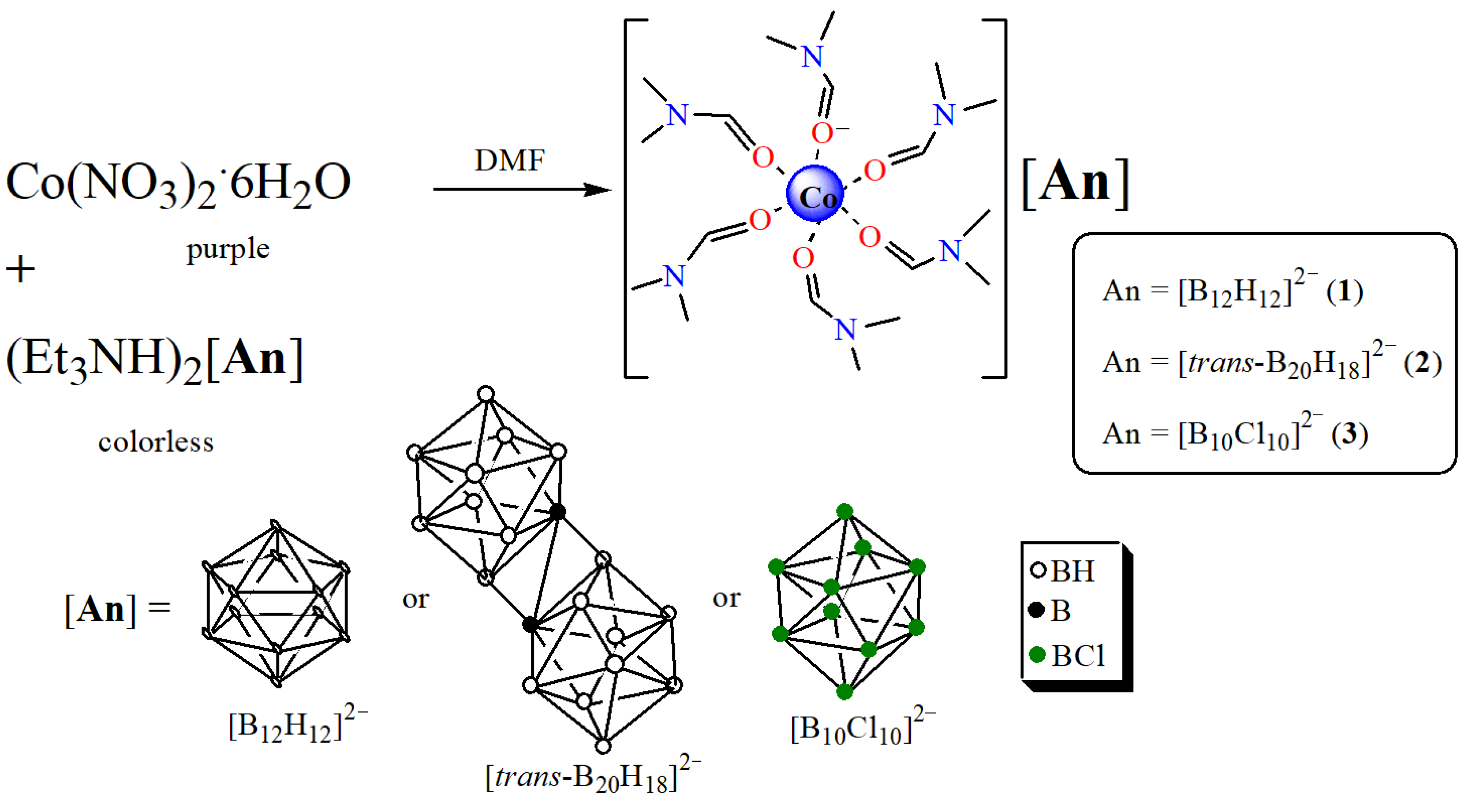
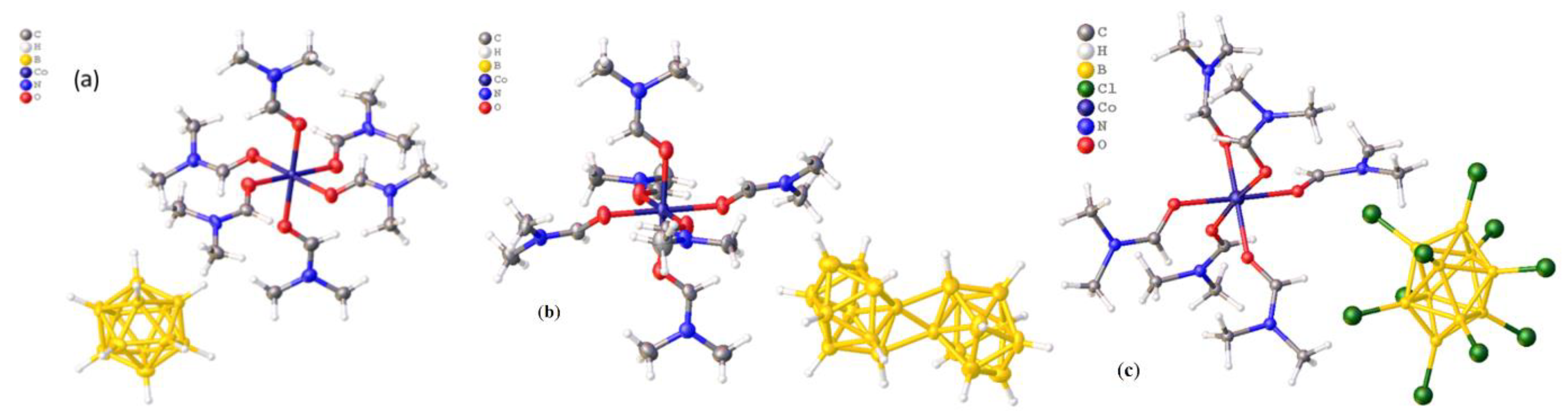
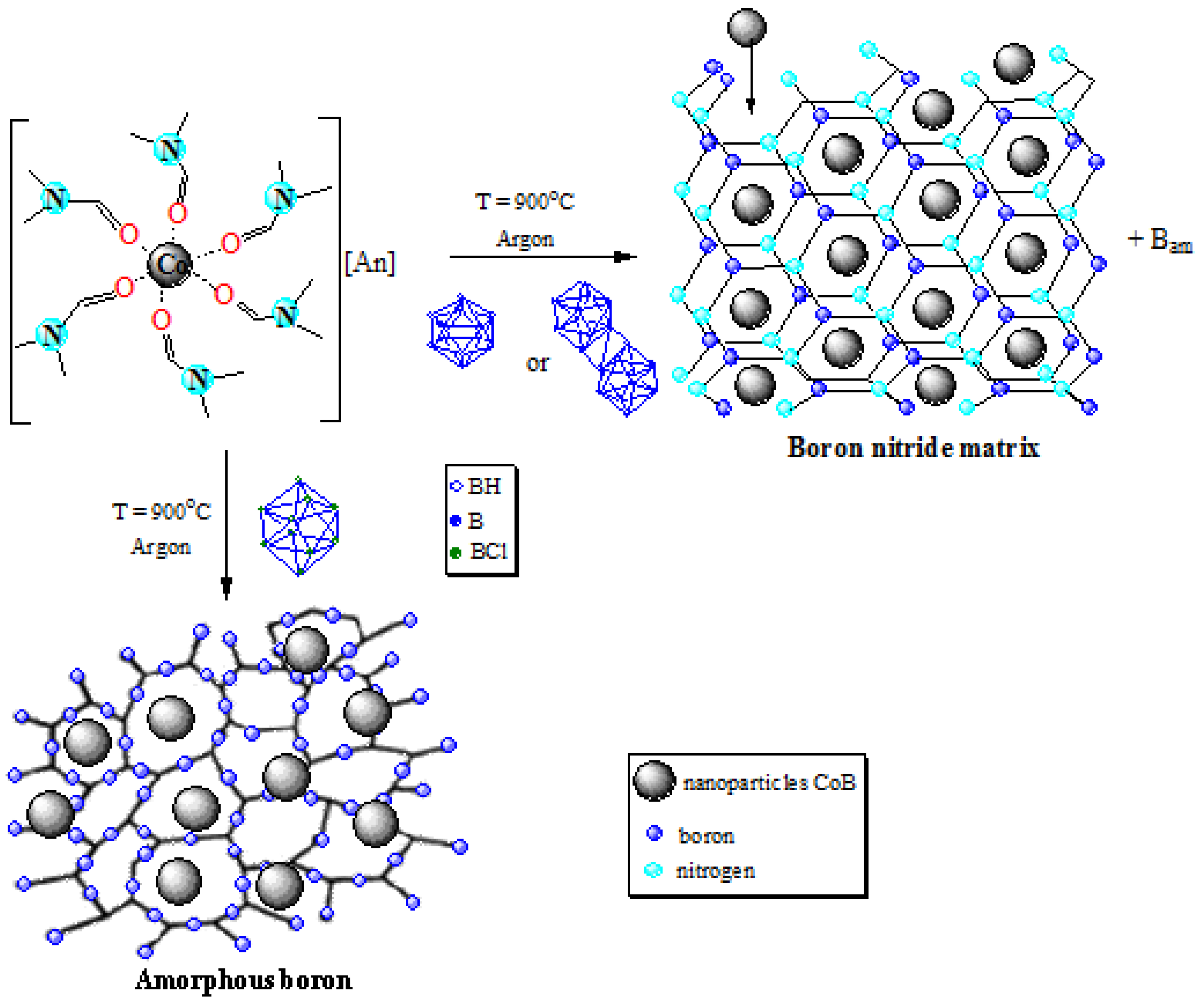
2.1. Synthesis of Precursors [Co(DMF)6][An]
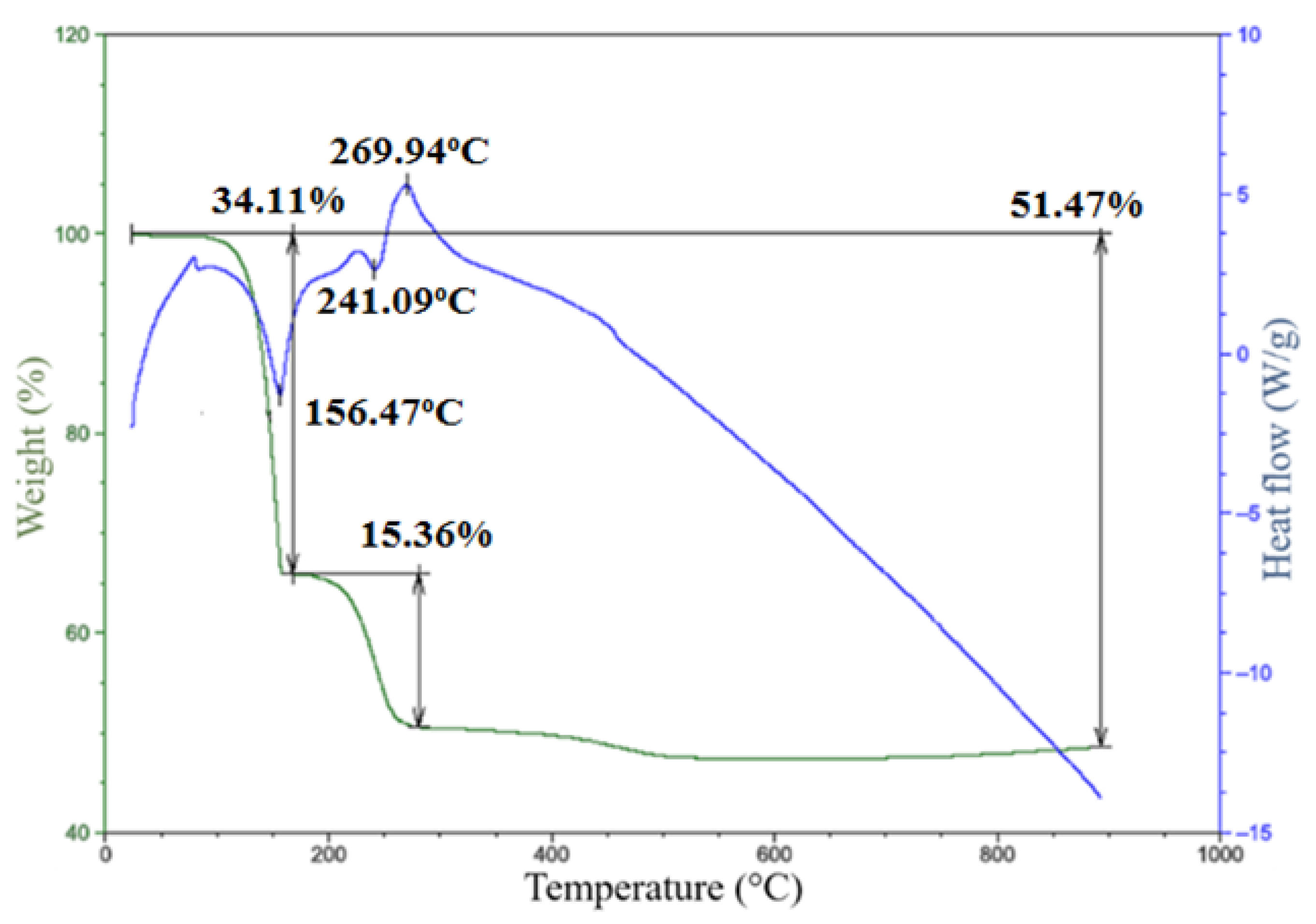
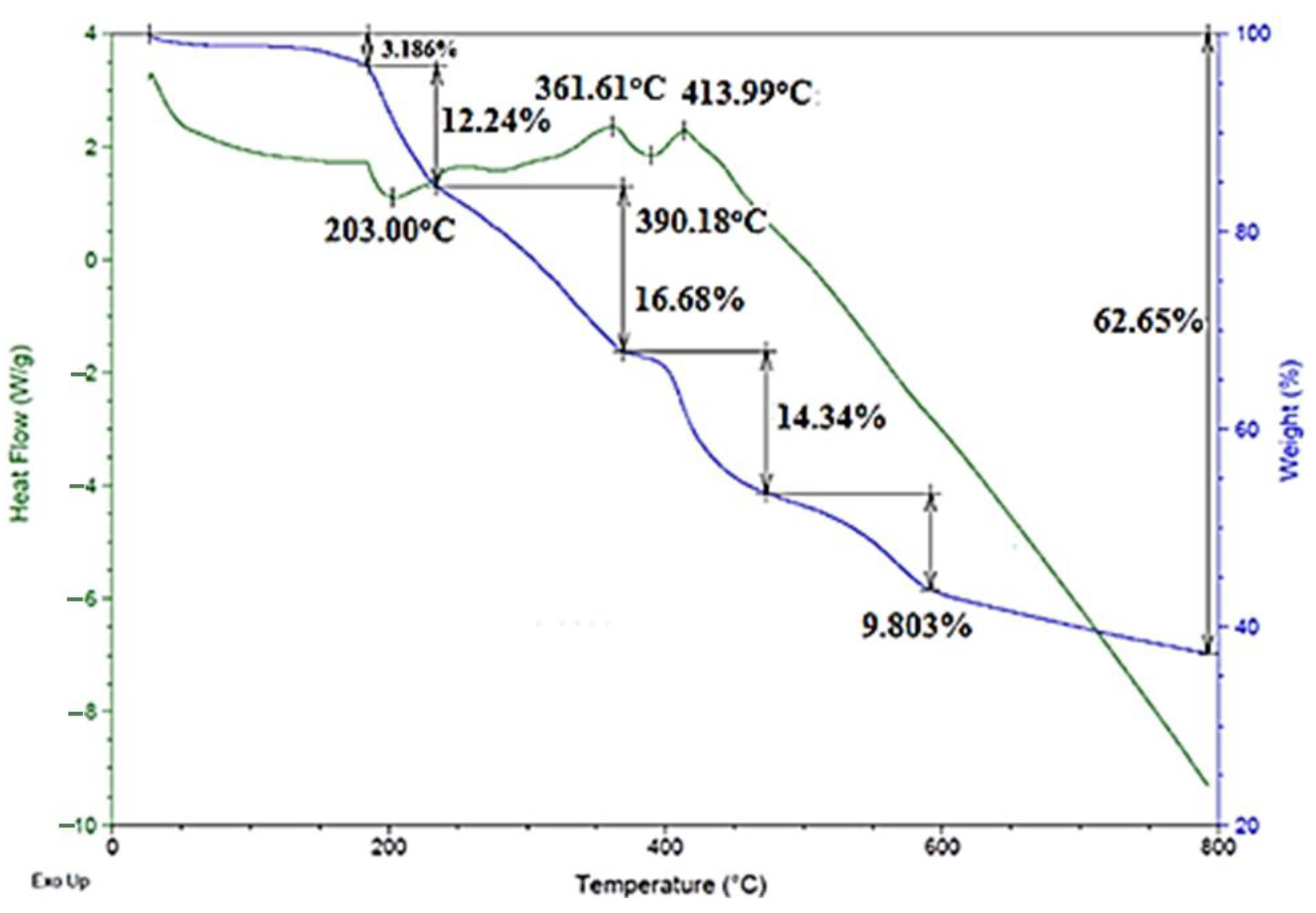
2.2. Thermal Decomposition of 1–3
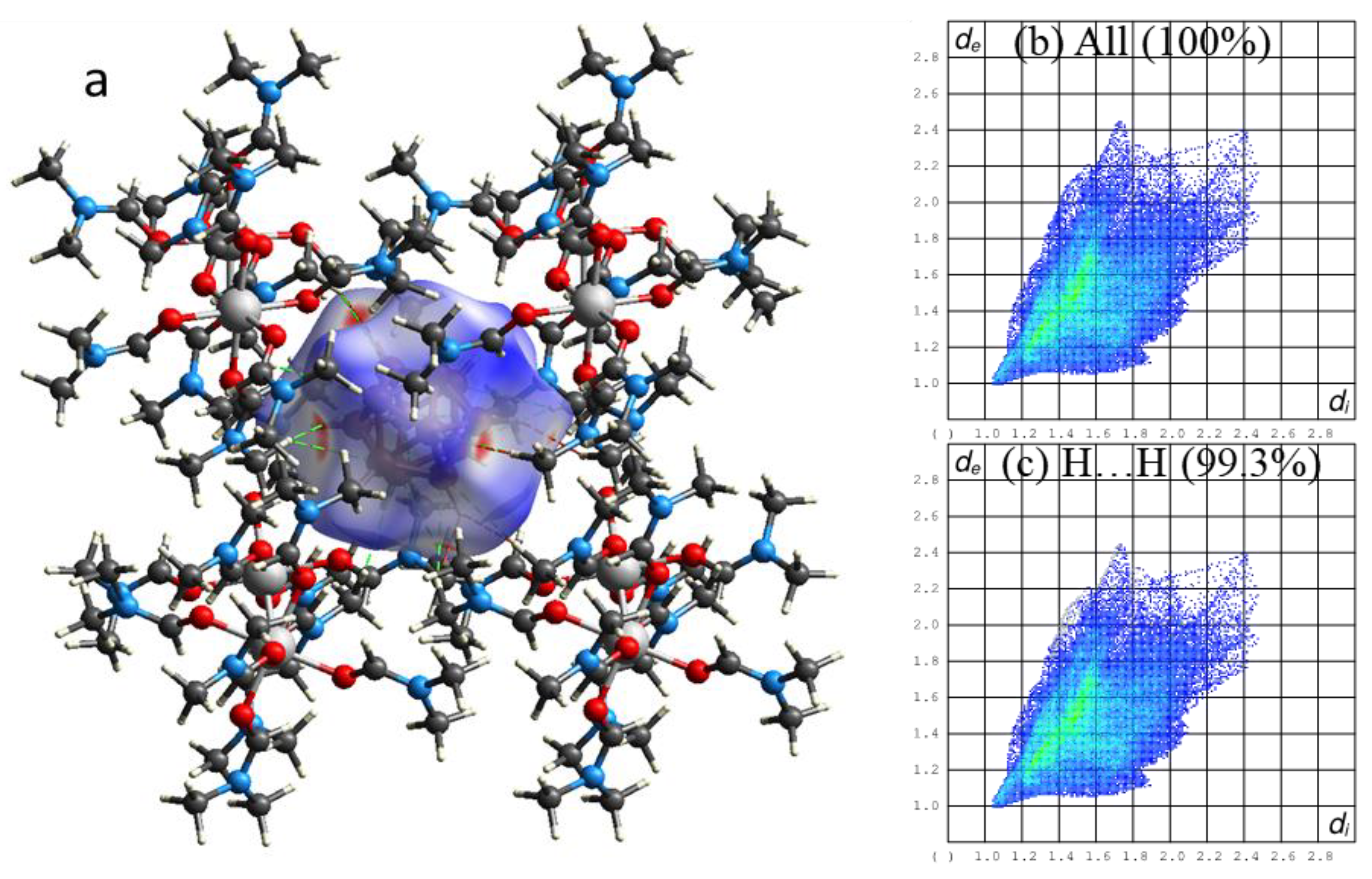
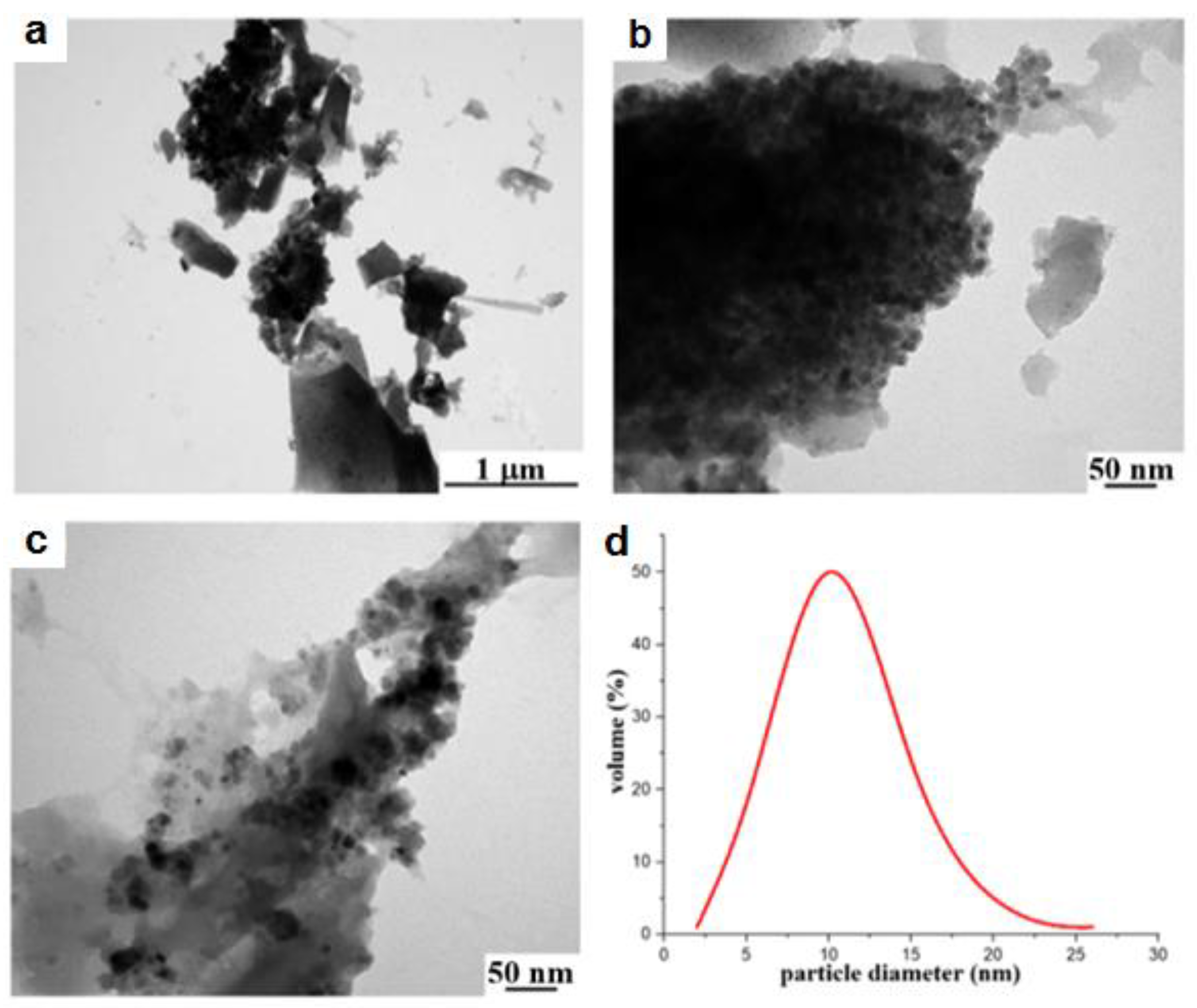
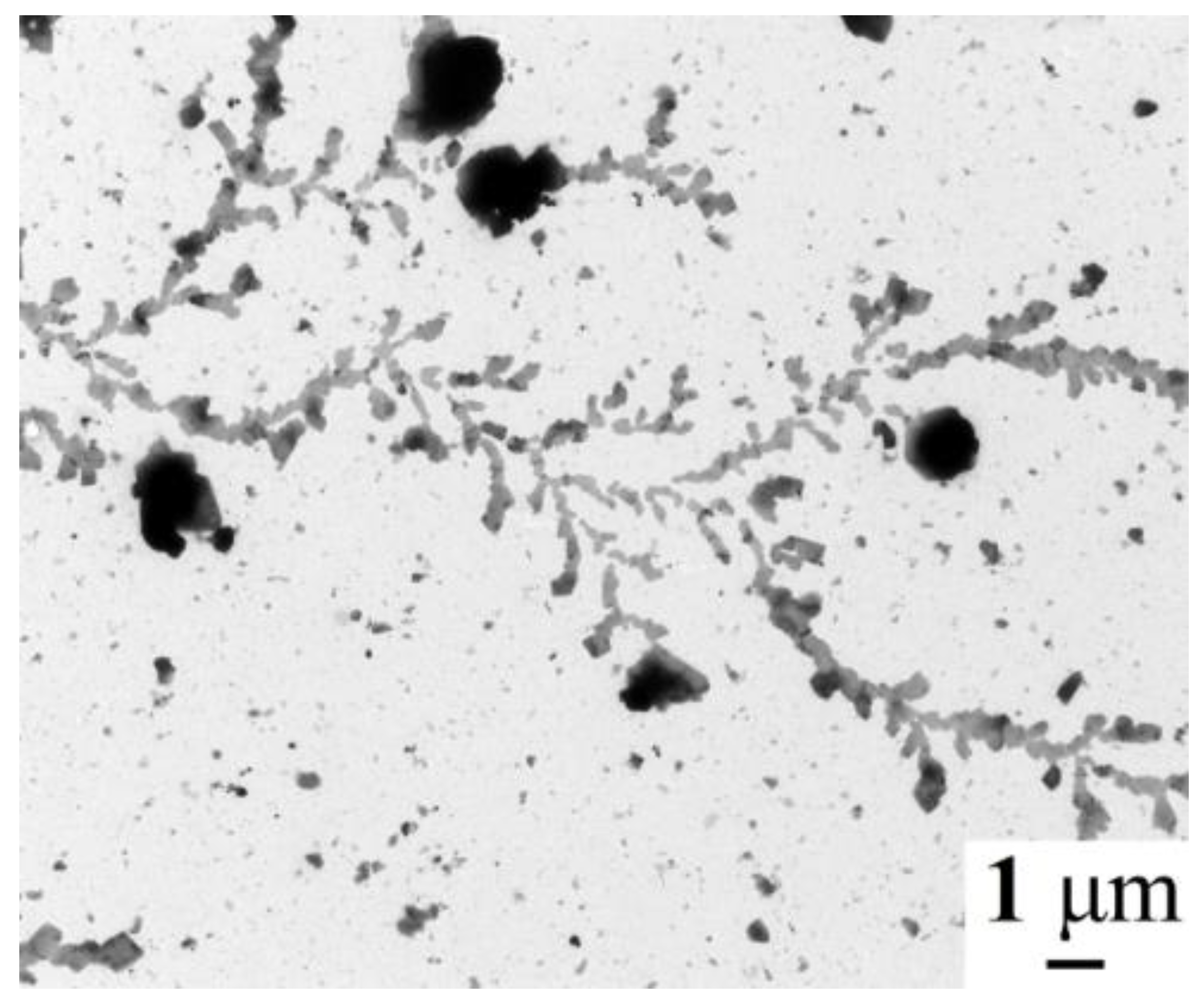
2.3. Morphology of Samples 1a–3a
3. Experimental
3.1. Synthesis of Compounds
3.1.1. Preparation of [Co(DMF)6][An], Where [An] = [B12H12]2− (Compound 1) and [trans-B20H18]2− (Compound 2∙3H2O)
3.1.2. Preparation of [Co(DMF)6][B10Cl10] (3)
3.1.3. Preparation of Compounds 1a–3a
3.2. Methods of Investigation
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Carenco, S.; Portehault, D.; Boissière, C.; Mézailles, N.; Sanchez, C. Nanoscaled Metal Borides and Phosphides: Recent Developments and Perspectives. Chem. Rev. 2013, 113, 7981–8065. [Google Scholar] [CrossRef] [PubMed]
- Paksoy, A.; Kurtoğlu, S.F.; Dizaji, A.K.; Altıntaş, Z.; Khoshsima, S.; Uzun, A.; Balcı, Ö. Nanocrystalline cobalt–nickel–boron (metal boride) catalysts for efficient hydrogen production from the hydrolysis of sodium borohydride. Int. J. Hydrogen Energy 2021, 46, 7974–7988. [Google Scholar] [CrossRef]
- Vinokurov, A.A.; Dremova, N.N.; Nadkhina, S.E.; Ivanov, A.V.; Shilkin, S.P. Formation of Niobium Diboride Nanoparticles by the Reaction of Niobium Pentachloride with Sodium Borohydride in Ionic Melts of Alkali Metal Halides. Russ. J. Gen. Chem. 2022, 92, 272–275. [Google Scholar] [CrossRef]
- Akopov, G.; Yeung, M.T.; Kaner, R.B. Rediscovering the Crystal Chemistry of Borides. Adv. Mater. 2017, 29, 1604506. [Google Scholar] [CrossRef] [PubMed]
- Demirci, U.B.; Miele, P. Cobalt in NaBH4 Hydrolysis. Phys. Chem. Chem. Phys. 2010, 12, 14651–14665. [Google Scholar] [CrossRef] [PubMed]
- McCrory, C.C.L.; Jung, S.; Ferrer, I.M.; Chatman, S.M.; Peters, J.C.; Jaramillo, T.F. Benchmarking Hydrogen Evolving Reaction and Oxygen Evolving Reaction Electrocatalysts for Solar Water Splitting Devices. Am. J. Chem. Soc. 2015, 137, 4347–4357. [Google Scholar] [CrossRef] [PubMed]
- Masa, J.; Weide, P.; Peeters, D.; Sinev, I.; Xia, W.; Sun, Z.; Somsen, C.; Muhler, M.; Schuhmann, W. Amorphous cobalt boride (Co2B) as a highly efficient nonprecious catalyst for electrochemical water splitting: Oxygen and hydrogen evolution. Adv. Energy Mater. 2016, 6, 1502313. [Google Scholar] [CrossRef]
- Chen, Z.; Kang, Q.; Cao, G.; Xu, N.; Dai, H.; Wang, P. Study of cobalt boride-derived electrocatalysts for overall water splitting. Int. J. Hydrogen Energy 2018, 43, 6076–6087. [Google Scholar] [CrossRef]
- Gupta, S.; Patel, N.; Miotello, A.; Kothari, D.C. Cobalt-Boride: An efficient and robust electrocatalyst for hydrogen evolution reaction. J. Power Sources 2015, 279, 620–625. [Google Scholar] [CrossRef]
- Chen, S.; Li, Y.; Zhang, Z.; Fu, Q.; Bao, X. The synergetic effect of h-BN shells and subsurface B in CoB: X@h-BN nanocatalysts for enhanced oxygen evolution reactions. J. Mater. Chem. A 2018, 6, 10644–10648. [Google Scholar] [CrossRef]
- Klemenz, S.; Schuch, J.; Hawel, S.; Zieschang, A.M.; Kaiser, B.; Jaegermann, W.; Albert, B. Synthesis of a Highly Efficient Oxygen- Evolution Electrocatalyst by Incorporation of Iron into Nanoscale Cobalt Borides. ChemSusChem 2018, 11, 3150–3156. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.; Wen, J.; Zhang, S.; Yuan, H.; Li, K.; Yan, F.; Zhang, X.; Chen, Y. Crystal CoxB (x = 1−3) Synthesized by a Ball-Milling Method as High-Performance Electrocatalysts for the Oxygen Evolution Reaction. ACS Sustain. Chem. Eng. 2017, 5, 10266–10274. [Google Scholar] [CrossRef]
- Gupta, S.; Jadhav, H.; Sinha, S.; Miotello, A.; Patel, M.K.; Sarkar, A.; Patel, N. Cobalt-Boride Nanostructured Thin Films with High Performance and Stability for Alkaline Water Oxidation. ACS Sustain. Chem. Eng. 2019, 7, 16651–16658. [Google Scholar] [CrossRef]
- Kalyon, N.; Hofmann, K.; Malter, J.; Lucas, M.; Claus, P.; Albert, B. Catalytic Activity of Nanoscale Borides: Co2B and Ni7B3 in the Liquid-phase Hydrogenation of Citral. J. Catal. 2017, 352, 436–441. [Google Scholar] [CrossRef]
- Bratz, K.; Wörz, N.; Brandner, A.; Hofmann, K.; Claus, P.; Albert, B. Co2B als interessanter Katalysator in der Citralhydrierung. Anorg. Allg. Chem. 2010, 636, 2098. [Google Scholar] [CrossRef]
- Wang, Y.D.; Ai, X.P.; Cao, Y.L.; Yang, H.X. Exceptional electrochemical activities of amorphous Fe–B and Co–B alloy powders used as high capacity anode materials. Electrochem. Commun. 2004, 6, 780–784. [Google Scholar] [CrossRef]
- Krishnan, P.; Advani, S.G.; Prasad, A.K. Cobalt oxides as Co2B catalyst precursors for the hydrolysis of sodium borohydride solutions to generate hydrogen for PEM fuel cells. Int. J. Hydrogen Energy 2008, 33, 7095–7102. [Google Scholar] [CrossRef]
- Wu, C.; Wu, F.; Bai, Y.; Yi, B.; Zhang, H. Cobalt boride catalysts for hydrogen generation from alkaline NaBH4 solution. Mater. Lett. 2005, 59, 1748–1751. [Google Scholar] [CrossRef]
- Gouget, G.; Debecker, D.P.; Kim, A.; Olivieri, G.; Gallet, J.J.; Bournel, F.; Thomas, C.; Ersen, O.; Moldovan, S.; Sanchez, C.; et al. In Situ Solid-Gas Reactivity of Nanoscaled Metal Borides from Molten Salt Synthesis. Inorg. Chem. 2017, 56, 9225–9234. [Google Scholar] [CrossRef] [PubMed]
- Jiang, B.; Song, H.; Kang, Y.; Wang, S.; Wang, Q.; Zhou, X.; Kani, K.; Guo, Y.; Ye, J.; Li, H.; et al. A mesoporous non-precious metal boride system: Synthesis of mesoporous cobalt boride by strictly controlled chemical reduction. Chem. Sci. 2020, 11, 791–796. [Google Scholar] [CrossRef]
- A-Ali, Z.O.; Al-Masoudi, S.R. Nano-Metal Borides of Cobalt, Nickel and Copper. Nanomed. Nanotechnol. 2017, 8, 6. [Google Scholar] [CrossRef]
- Oh, J.-H.; Kim, M.; Lee, Y.H.; Hong, S.-H.; Park, S.S.; Kim, T.-H.; Choi, S. Synthesis of cobalt boride nanoparticles and h-BN nanocage encapsulation by thermal plasma. Ceram. Int. 2020, 46, 28792–28799. [Google Scholar] [CrossRef]
- Oh, J.-H.; Choi, S.; Kim, T.-H. Synthesis of metal boride nanoparticles by using thermal plasmas. Korean J. Phys. Soc. 2022, 80, 808–816. [Google Scholar] [CrossRef]
- Choi, S.; Lapitan, L.D.S.; Cheng, Y.; Watanabe, T. Synthesis of cobalt boride nanoparticles using RF thermal plasma. Adv. Powder Technol. 2014, 25, 365–371. [Google Scholar] [CrossRef]
- Khusnuriyalova, A.F.; Caporali, M.; Hey-Hawkins, E.; Sinyashin, O.G.; Yakhvarov, D.G. Preparation of Cobalt Nanoparticles. Eur. J. Inorg. Chem. 2021, 13, 3023–3047. [Google Scholar] [CrossRef]
- Hosmane, N.S.; Eagling, R. (Eds.) Handbook of Boron Science with Applications in Organometallics, Catalysis, Materials and Medicine; World Scientific: Singapore, 2018; Volume 4. [Google Scholar] [CrossRef]
- Korolenko, S.E.; Avdeeva, V.V.; Malinina, E.A.; Kuznetsov, N.T. Zinc(II) and Cadmium(II) Coordination Compounds with Boron Cluster Anions: Classification of Compounds Depending on Strength of Metal–Boron Cage Interaction and Analysis of Structures (Review). Russ. J. Inorg. Chem. 2021, 66, 1350–1373. [Google Scholar] [CrossRef]
- Avdeeva, V.V.; Korolenko, S.E.; Malinina, E.A.; Kuznetsov, N.T. Solvent Molecules as Ligands in Coordination Compounds of Metals with Boron Cluster Anions and Their Derivatives (A Review). Russ. J. Gen. Chem. 2022, 92, 393–417. [Google Scholar] [CrossRef]
- Avdeeva, V.V.; Malinina, E.A.; Kuznetsov, N.T. Boron cluster anions and their derivatives in complexation reactions. Coord. Chem. Rev. 2022, 469, 214636. [Google Scholar] [CrossRef]
- Sivaev, I.B.; Prikaznov, A.V.; Naoufal, D. Fifty years of the closo-decaborate anion chemistry. Collect. Czech. Chem. Commun. 2010, 75, 1149. [Google Scholar] [CrossRef]
- Sivaev, I.B.; Bregadze, V.I.; Sjöberg, S. Chemistry of closo-Dodecaborate Anion [B12H12]2-: A Review. Collect. Czech. Chem. Commun. 2002, 67, 67. [Google Scholar] [CrossRef]
- Avdeeva, V.V.; Polyakova, I.N.; Vologzhanina, A.V.; Goeva, L.V.; Buzanov, G.A.; Generalova, N.B.; Malinina, E.A.; Zhizhin, K.Y.; Kuznetsov, N.T. [Co(solv)6][B10H10] (solv = DMF and DMSO) for low-temperature synthesis of borides. Russ. J. Inorg. Chem. 2016, 61, 1125–1134. [Google Scholar] [CrossRef]
- Malinina, E.A.; Goeva, L.V.; Buzanov, G.A.; Avdeeva, V.V.; Kuznetsov, N.T.; Retivov, V.M. Synthesis and Thermal Reduction of Complexes [NiLn][B10H10] (L = DMF, H2O, n = 6; L = N2H4, n = 3): Formation of Solid Solutions Ni3C1 –xBx. Russ. J. Inorg. Chem. 2020, 65, 126–132. [Google Scholar] [CrossRef]
- Malinina, E.A.; Goeva, L.V.; Buzanov, G.A.; Avdeeva, V.V.; Efimov, N.N.; Kozerozhets, I.V.; Kuznetsov, N.T. Synthesis and Physicochemical Properties of Binary Cobalt(II) Borides. Thermal Reduction of Precursor Complexes [CoLn][B10H10] (L = H2O, n = 6; N2H4, n = 3). Russ. J. Inorg. Chem. 2019, 64, 1325–1334. [Google Scholar] [CrossRef]
- Malinina, E.A.; Goeva, L.V.; Buzanov, G.A.; Avdeeva, V.V.; Efimov, N.N.; Kozerozhets, I.V.; Kuznetsov, N.T. A New Method for Synthesis of Binary Borides with Desired Properties. Dokl. Chem. 2019, 487, 180–183. [Google Scholar] [CrossRef]
- Avdeeva, V.V.; Malinina, E.A.; Vologzhanina, A.V.; Sivaev, I.B.; Kuznetsov, N.T. Formation of oxidopolyborates in destruction of the [B11H14]– anion promoted by transition metals. Inorg. Chim. Acta 2020, 509, 119693. [Google Scholar] [CrossRef]
- Avdeeva, V.V.; Vologzhanina, A.V.; Ugolkova, E.A.; Minin, V.V.; Malinina, E.A.; Kuznetsov, N.T. Synthesis and structures of compounds [ML6][B10Cl10] (M = Co, Ni; L = CH3CN, DMF, DMSO) as precursors for synthesis of cobalt(II) and nickel(II) complexes with organic L ligands. J. Solid State 2021, 296, 121989. [Google Scholar] [CrossRef]
- Avdeeva, V.V.; Privalov, V.I.; Kubasov, A.S.; Nikiforova, S.E.; Malinina, E.A.; Kuznetsov, N.T. 2D COSY 11B NMR spectroscopy in the interpretation of the structures of iso and trans isomers of the macropolyhedral boron cluster [B20H18]2–. Inorg. Chim. Acta, 2023. (in press) CCDC 2159666.
- Pokropivny, V.V.; Smolyar, A.S.; Ovsiannikova, L.I.; Pokropivny, A.V.; Kuts, V.A.; Lyashenko, V.I.; Nesterenko, Y.V. Fluid Synthesis and Structure of a New Polymorphic Modification of Boron Nitride. Phys. Solid State 2013, 55, 878–884. [Google Scholar] [CrossRef]
- Miller, H.C.; Miller, N.E.; Muetterties, E.L. Synthesis of Polyhedral Boranes. Am. Chem. Soc. 1963, 85, 3885. [Google Scholar] [CrossRef]
- Greenwood, N.N.; Morris, J.H. Synthesis of the B12H122- anion. Proc. Chem. Soc. 1963, 11, 338. [Google Scholar]
- Kaczmarczyk, A.; Dobrott, R.D.; Lipscomb, W.N. Reactions of B10H10-2 ion. Proc. Nat. Acad. Sci. USA 1962, 48, 729–733. [Google Scholar] [CrossRef]
- Kravchenko, E.A.; Gippius, A.A.; Korlyukov, A.A.; Vologzhanina, A.V.; Avdeeva, V.V.; Malinina, E.A.; Ulitin, E.O.; Kuznetsov, N.T. Secondary interactions in decachloro-closo-decaborates R2[B10Cl10] (R = Et3NH+, Ph4P+, and [Ag(NH3)2]+): 35Cl NQR, PW-DFT, and X-ray studies. Inorg. Chim. Acta 2016, 447, 22–31. [Google Scholar] [CrossRef]
- Apex3 v2019.1-0; Saint, B. Bruker AXS Inc.: Madison, WI, USA, 2018.
- Krause, L.; Herbst-Irmer, R.; Sheldrick, G.M.; Stalke, D. Comparison of Silver and Molybdenum Microfocus X-Ray Sources for Single-Crystal Structure Determination. J. Appl. Crystallogr. 2015, 48, 3–10. [Google Scholar] [CrossRef] [PubMed]
- Sheldrick, G.M. Crystal Structure Refinement with SHELXL. Acta Crystallogr. Sect. C Struct. Chem. 2015, 71, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Dolomanov, O.V.; Bourhis, L.J.; Gildea, R.J.; Howard, J.A.K.; Puschmann, H. OLEX2: A complete structure solution, refinement and analysis program. J. Appl. Cryst. 2009, 42, 339–341. [Google Scholar] [CrossRef]
- Turner, M.J.; McKinnon, J.J.; Wolff, S.K.; Grimwood, D.J.; Spackman, P.R.; Jayatilaka, D.; Spackman, M.A. CrystalExplorer17.5; University of Western Australia: Perth, Australia, 2017. [Google Scholar]




Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Malinina, E.A.; Myshletsov, I.I.; Buzanov, G.A.; Kubasov, A.S.; Kozerozhets, I.V.; Goeva, L.V.; Nikiforova, S.E.; Avdeeva, V.V.; Zhizhin, K.Y.; Kuznetsov, N.T. A New Approach to the Synthesis of Nanocrystalline Cobalt Boride in the Course of the Thermal Decomposition of Cobalt Complexes [Co(DMF)6]2+ with Boron Cluster Anions. Molecules 2023, 28, 453. https://doi.org/10.3390/molecules28010453
Malinina EA, Myshletsov II, Buzanov GA, Kubasov AS, Kozerozhets IV, Goeva LV, Nikiforova SE, Avdeeva VV, Zhizhin KY, Kuznetsov NT. A New Approach to the Synthesis of Nanocrystalline Cobalt Boride in the Course of the Thermal Decomposition of Cobalt Complexes [Co(DMF)6]2+ with Boron Cluster Anions. Molecules. 2023; 28(1):453. https://doi.org/10.3390/molecules28010453
Chicago/Turabian StyleMalinina, Elena A., Ivan I. Myshletsov, Grigorii A. Buzanov, Alexey S. Kubasov, Irina V. Kozerozhets, Lyudmila V. Goeva, Svetlana E. Nikiforova, Varvara V. Avdeeva, Konstantin Yu. Zhizhin, and Nikolay T. Kuznetsov. 2023. "A New Approach to the Synthesis of Nanocrystalline Cobalt Boride in the Course of the Thermal Decomposition of Cobalt Complexes [Co(DMF)6]2+ with Boron Cluster Anions" Molecules 28, no. 1: 453. https://doi.org/10.3390/molecules28010453
APA StyleMalinina, E. A., Myshletsov, I. I., Buzanov, G. A., Kubasov, A. S., Kozerozhets, I. V., Goeva, L. V., Nikiforova, S. E., Avdeeva, V. V., Zhizhin, K. Y., & Kuznetsov, N. T. (2023). A New Approach to the Synthesis of Nanocrystalline Cobalt Boride in the Course of the Thermal Decomposition of Cobalt Complexes [Co(DMF)6]2+ with Boron Cluster Anions. Molecules, 28(1), 453. https://doi.org/10.3390/molecules28010453








