The Medicinal Natural Products of Cannabis sativa Linn.: A Review
Abstract
:1. Introduction
2. Methods
3. Origin and Botanical Description of C. sativa
4. Phytochemistry of C. sativa
4.1. Chemical Profile of C. sativa
4.2. Extraction, Isolation, and Chemical Characterization of C. sativa
4.3. Biological Evaluation/Potentials of C. sativa
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sofowora, A.; Ogunbodede, E.; Onayade, A. The role and place of medicinal plants in the strategies for disease prevention. Afr. J. Tradit. Complement. Altern. Med. 2013, 10, 210–229. [Google Scholar] [CrossRef]
- Choudhary, N.; Siddiqui, M.; Bi, S.; Khatoon, S. Variation in preliminary phytochemicals screening of Cannabis sativa L. leaf, stem and root. Int. J. Pharmacogn. 2014, 1, 516–519. [Google Scholar]
- Bandar, H.; Hijazi, A.; Rammal, H.; Hachem, A.; Saad, Z.; Badran, B. Techniques for the extraction of bioactive compounds from Lebanese Urtica Dioica. Am. J. Phytomed. Clin. Ther. 2013, 1, 507–513. [Google Scholar]
- Andre, C.M.; Hausman, J.F.; Guerriero, G. Cannabis sativa: The plant of the thousand and one molecules. Front. Plant Sci. 2016, 7, 19. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Elsohly, M.A.; Radwan, M.M.; Gul, W.; Chandra, S.; Galal, A. Phytochemistry of Cannabis sativa L. Phytocannabinoids 2017, 103, 1–36. [Google Scholar]
- Baldino, L.; Scognamiglio, M.; Reverchon, E. Supercritical fluid technologies applied to the extraction of compounds of industrial interest from Cannabis sativa L. and to their pharmaceutical formulations: A review. J. Pharm. Fluids 2020, 165, 104960. [Google Scholar] [CrossRef]
- Brunetti, P.; Pichini, S.; Pacifici, R.; Busardò, F.P.; del Rio, A. Herbal preparations of medical cannabis: A vademecum for prescribing doctors. Medicina 2020, 56, 237. [Google Scholar] [CrossRef]
- Lewis, M.M.; Yang, Y.; Wasilewski, E.; Clarke, H.A.; Kotra, L.P. Chemical profiling of medical Cannabis extracts. ACS Omega 2017, 2, 6091–6103. [Google Scholar] [CrossRef] [PubMed]
- Pacifici, R.; Marchei, E.; Salvatore, F.; Guandalini, L.; Busardò, F.P.; Pichini, S. Evaluation of cannabinoids concentration and stability in standardized preparations of cannabis tea and cannabis oil by ultra-high performance liquid chromatography tandem mass spectrometry. Clin. Chem. Lab. Med. 2017, 55, 1555–1563. [Google Scholar] [CrossRef]
- Pacifici, R.; Marchei, E.; Salvatore, F.; Guandalini, L.; Busardò, F.P.; Pichini, S. Evaluation of long-term stability of cannabinoids in standardized preparations of cannabis flowering tops and cannabis oil by ultra-high-performance liquid chromatography tandem mass spectrometry. Clin. Chem. Lab. Med. 2018, 56, 94–96. [Google Scholar] [CrossRef] [PubMed]
- Jin, D.; Dai, K.; Xie, Z.; Chen, J. Secondary metabolites profiled in cannabis inflorescences, leaves, stem barks, and roots for medicinal purposes. Sci. Rep. 2020, 10, 1–14. [Google Scholar]
- Farag, S.; Kayser, O. The Cannabis plant: Botanical aspects. In Handbook of Cannabis and Related Pathologies; Academic Press: Cambridge, MA, USA, 2017; pp. 3–12. [Google Scholar]
- Radwan, M.M.; Chandra, S.; Gul, S.; Elsohly, M.A. Cannabinoids, Phenolics, Terpenes and Alkaloids of Cannabis. Molecules 2021, 26, 2774. [Google Scholar] [CrossRef]
- Stevens, C.J.; Murphy, C.; Roberts, R.; Lucas, L.; Silva, F.; Fuller, D.Q. Between China and South Asia: A Middle Asian corridor of crop dispersal and agricultural innovation in the bronze age. Holocene 2016, 26, 1541–1555. [Google Scholar] [CrossRef] [PubMed]
- Chandra, S.; Lata, H.; Khan, I.A.; Elsohly, M.A. Cannabis sativa L.: Botany and horticulture. In Cannabis sativa L.—Botany and Biotechnology; Springer: Berlin/Heidelberg, Germany, 2017; pp. 79–100. [Google Scholar]
- Duvall, C.S. A brief agricultural history of Cannabis in Africa, from prehistory to canna-colony. Echogéo 2019, 48, 1–25. [Google Scholar] [CrossRef] [Green Version]
- Nsuala, B.N.; Enslin, G.; Viljoen, A. “Wild Cannabis”: A review of the traditional use and phytochemistry of Leonotis leonurus. J. Ethnopharmacol. 2015, 174, 520–539. [Google Scholar] [CrossRef] [PubMed]
- Bonini, S.A.; Premoli, M.; Tambaro, S.; Kumar, A.; Maccarinelli, G.; Memo, M.; Mastinu, A. Cannabis sativa: A comprehensive ethnopharmacological review of a medicinal plant with a long history. J. Ethnopharmacol. 2018, 227, 300–315. [Google Scholar] [CrossRef] [PubMed]
- Dutch Passion. Understanding Cannabis Phenotypes, Genotypes and Chemotypes. 2020. Available online: https://dutch-passion.com/en/blog/understanding-cannabis-phenotypes-genotypes-and-chemotypes-n980 (accessed on 19 February 2022).
- Hanuš, L.O.; Meyer, S.M.; Muñoz, E.; Taglialatela-Scafati, O.; Appendino, G. Phytocannabinoids: A unified critical inventory. Nat. Prod. Rep. 2016, 33, 1357–1392. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Appendino, G.; Chianese, G.; Taglialatela-Scafati, O. Cannabinoids: Occurrence and medicinal chemistry. Curr. Med. Chem. 2011, 18, 1085–1099. [Google Scholar] [CrossRef] [PubMed]
- Hill, A.J.; Williams, C.M.; Whalley, B.J.; Stephens, G.J. Phytocannabinoids as novel therapeutic agents in CNS disorders. Pharmacol. Ther. 2012, 133, 79–97. [Google Scholar] [CrossRef] [Green Version]
- Lewis-Bakker, M.M.; Yang, Y.; Vyawahare, R.; Kotra, L.P. Extractions of medical Cannabis cultivars and the role of decarboxylation in optimal receptor responses. Cannabis Cannabinoid Res. 2019, 4, 183–194. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Elsohly, M.; Gul, W. Constituents of Cannabis sativa. Handb. Cannabis 2014, 3, 1093. [Google Scholar]
- Elzinga, S.; Fischedick, J.; Podkolinski, R.; Raber, J.C. Cannabinoids and terpenes as chemotaxonomic markers in Cannabis. Nat. Prod. Chem. Res. 2015, 3, 1–9. [Google Scholar]
- Hazekamp, A.; Tejkalová, K.; Papadimitriou, S. Cannabis: From cultivar to chemovar II—A metabolomics approach to cannabis classification. Cannabis Cannabinoid Res. 2016, 1, 202–215. [Google Scholar]
- Lynch, R.C.; Vergara, D.; Tittes, S.; White, K.; Schwartz, C.; Gibbs, M.J.; Ruthenburg, T.C.; Decesare, K.; Land, D.P.; Kane, N.C. Genomic and chemical diversity in Cannabis. Crit. Rev. Plant Sci. 2016, 35, 349–363. [Google Scholar] [CrossRef] [Green Version]
- Pollastro, F.; Minassi, A.; Fresu, L.G. Cannabis phenolics and their bioactivities. Curr. Med. Chem. 2018, 25, 1160–1185. [Google Scholar]
- Russo, E.B.; Marcu, J. Cannabis pharmacology: The usual suspects and a few promising leads. Adv. Pharmacol. 2017, 80, 67–134. [Google Scholar]
- Upton, R.; Elsohly, M. (Eds.) Cannabis Inflorescence: Cannabis Spp.; Standards of Identity, Analysis, and Quality Control; American Herbal Pharmacopoeia: Scotts Valley, CA, USA, 2014. [Google Scholar]
- Radwan, M.M.; Elsohly, M.A.; El-Alfy, A.T.; Ahmed, S.A.; Slade, D.; Husni, A.S.; Manly, S.P.; Wilson, L.; Seale, S.; Cutler, S.J.; et al. Isolation and pharmacological evaluation of minor cannabinoids from high-potency Cannabis sativa. J. Nat. Prod. 2015, 78, 1271–1276. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guo, T.; Liu, Q.; Hou, P.; Li, F.; Guo, S.; Song, W.; Zhang, H.; Liu, X.; Zhang, S.; Zhang, J.; et al. Stilbenoids and cannabinoids from the leaves of Cannabis sativa f. sativa with potential reverse cholesterol transport activity. Food Funct. 2018, 9, 6608–6617. [Google Scholar] [CrossRef]
- Guo, T.T.; Zhang, J.C.; Zhang, H.; Liu, Q.C.; Zhao, Y.; Hou, Y.F.; Bai, L.; Zhang, L.; Liu, X.Q.; Liu, X.Y.; et al. Bioactive spirans and other constituents from the leaves of Cannabis sativa f. sativa. J. Asian Nat. Prod. Res. 2017, 19, 793–802. [Google Scholar] [CrossRef] [PubMed]
- Yan, X.; Tang, J.; dos Santos Passos, C.; Nurisso, A.; Simoes-Pires, C.A.; Ji, M.; Lou, H.; Fan, P. Characterization of lignanamides from hemp (Cannabis sativa L.) seed and their antioxidant and acetylcholinesterase inhibitory activities. J. Agric. Food Chem. 2015, 63, 10611–10619. [Google Scholar] [CrossRef]
- Ingallina, C.; Sobolev, A.P.; Circi, S.; Spano, M.; Fraschetti, C.; Filippi, A.; di Sotto, A.; di Giacomo, S.; Mazzoccanti, G.; Gasparrini, F.; et al. Cannabis sativa L. inflorescences from monoecious cultivars grown in central Italy: An untargeted chemical characterization from early flowering to ripening. Molecules 2020, 25, 1908. [Google Scholar] [CrossRef] [PubMed]
- Menghini, L.; Ferrante, C.; Carradori, S.; D’antonio, M.; Orlando, G.; Cairone, F.; Cesa, S.; Filippi, A.; Fraschetti, C.; Zengin, G.; et al. Chemical and bioinformatics analyses of the anti-leishmanial and anti-oxidant activities of hemp essential oil. Biomolecules 2021, 11, 272. [Google Scholar] [CrossRef] [PubMed]
- Di Giacomo, V.; Recinella, L.; Chiavaroli, A.; Orlando, G.; Cataldi, A.; Rapino, M.; di Valerio, V.; Politi, M.; Antolini, M.D.; Acquaviva, A.; et al. Metabolomic profile and antioxidant/anti-inflammatory effects of industrial hemp water extract in fibroblasts, keratinocytes and isolated mouse skin specimens. Antioxidants 2021, 10, 44. [Google Scholar] [CrossRef] [PubMed]
- Weston-Green, K. The united chemicals of Cannabis: Beneficial effects of cannabis phytochemicals on the brain and cognition. In Recent Advances in Cannabinoid Research; IntechOpen: London, UK, 2018. [Google Scholar]
- Smeriglio, A.; Galati, E.M.; Monforte, M.T.; Lanuzza, F.; D’angelo, V.; Circosta, C. Polyphenolic Compounds and antioxidant activity of cold-pressed seed oil from finola cultivar of Cannabis sativa L. Phytother. Res. 2016, 30, 1298–1307. [Google Scholar] [CrossRef]
- Ryz, N.R.; Remillard, D.J.; Russo, E.B. Cannabis roots: A traditional therapy with future potential for treating inflammation and pain. Cannabis Cannabinoid Res. 2017, 2, 210–216. [Google Scholar] [CrossRef]
- Ramirez, C.L.; Fanovich, M.A.; Churio, M.S. Cannabinoids: Extraction methods, analysis, and physicochemical characterization. Stud. Nat. Prod. Chem. 2019, 66, 143–173. [Google Scholar]
- Płotka-Wasylka, J.; Rutkowska, M.; Owczarek, K.; Tobiszewski, M.; Namieśnik, J. Extraction with environmentally friendly solvents. TrAC Trends Anal. Chem. 2017, 91, 12–25. [Google Scholar] [CrossRef]
- International Conference on Harmonisation (ICH). International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use. ICH Harmonised Guideline. Impurities: Guideline for Residual solventsq3c(R6). 2016. Available online: https://database.ich.org/sites/default/files/Q3C-R6_Guideline_ErrorCorrection_2019_0410_0.pdf (accessed on 20 January 2022).
- Brighenti, V.; Pellati, F.; Steinbach, M.; Maran, D.; Benvenuti, S. Development of a new extraction technique and hplc method for the analysis of non-psychoactive cannabinoids in fibre-type Cannabis sativa L.(hemp). J. Pharma. Biomed. Anal. 2017, 143, 228–236. [Google Scholar] [CrossRef] [PubMed]
- Recommended Methods for the Identification and Analysis of Cannabis and Cannabis Products. 2009. Available online: http://www.unodc.org/documents/scientific/ST-NAR-40-Ebook.pdf (accessed on 1 November 2021).
- Allwood, J.W.; Ellis, D.I.; Goodacre, R. Metabolomic technologies and their application to the study of plants and plant-host interactions. Physiol. Plant. 2008, 132, 117–135. [Google Scholar] [CrossRef] [PubMed]
- Cardenia, V.; Toschi, T.G.; Scappini, S.; Rubino, R.C.; Rodriguez-Estrada, M.T. Development and validation of a fast gas chromatography/mass spectrometry method for the determination of cannabinoids in Cannabis sativa L. J. Food Drug Anal. 2018, 26, 1283–1292. [Google Scholar] [CrossRef] [PubMed]
- Rochfort, S.; Isbel, A.; Ezernieks, V.; Elkins, A.; Vincent, D.; Deseo, M.A.; Spangenberg, G.C. Utilisation of design of experiments approach to optimise supercritical fluid extraction of medicinal Cannabis. Sci. Rep. 2020, 10, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Patel, B.; Wene, D.; Fan, Z.T. Qualitative and quantitative measurement of cannabinoids in Cannabis using modified hplc/dad method. J. Pharma Biomed. Anal. 2017, 146, 15–23. [Google Scholar] [CrossRef] [PubMed]
- Bercht, C.; Lousberg, R.; Küppers, F.; Salemink, C.; Vree, T.; van Rossum, J. Cannabis: VII. Identification of cannabinol methyl ether from hashish. J. Chromatogr. Anal. 1973, 81, 163–166. [Google Scholar]
- Elsohly, M.; El-Feraly, F.; Turner, C. Isolation and characterization of (+) cannabitriol and () 10 ethoxy 9 hydroxy delta 6a tetrahydrocannabinol: Two new cannabinoids from Cannabis sativa L. extract. Lloydia 1977, 40, 275–280. [Google Scholar] [PubMed]
- Shoyama, Y.; Kuboe, K.; Nishioka, I.; Yamauchi, T. Cannabidiol monomethyl ether. A new neutral cannabinoid. Chem. Pharm. Bull. 1972, 20, 2072. [Google Scholar] [CrossRef] [Green Version]
- Ahmed, S.A.; Ross, S.A.; Slade, D.; Radwan, M.M.; Khan, I.A.; Elsohly, M.A. Minor oxygenated cannabinoids from high potency Cannabis sativa L. Phytochemistry 2015, 117, 194–199. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ahmed, S.A.; Ross, S.A.; Slade, D.; Radwan, M.M.; Zulfiqar, F.; Elsohly, M.A. Cannabinoid ester constituents from high-potency Cannabis sativa. J. Nat. Prod. 2008, 71, 536–542. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Radwan, M.M.; Elsohly, M.A.; Slade, D.; Ahmed, S.A.; Wilson, L.; El-Alfy, A.T.; Khan, I.A.; Ross, S.A. Non-cannabinoid constituents from a high potency Cannabis sativa variety. Phytochemistry 2008, 69, 2627–2633. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chianese, G.; Lopatriello, A.; Schiano-Moriello, A.; Caprioglio, D.; Mattoteia, D.; Benetti, E.; Ciceri, D.; Arnoldi, L.; de Combarieu, E.; Vitale, R.M.; et al. Cannabitwinol, a dimeric phytocannabinoid from hemp, Cannabis sativa L.; is a selective thermo-TRP modulator. J. Nat. Prod. 2020, 83, 2727–2736. [Google Scholar] [CrossRef] [PubMed]
- Citti, C.; Linciano, P.; Russo, F.; Luongo, L.; Iannotta, M.; Maione, S.; Laganà, A.; Capriotti, A.L.; Forni, F.; Vandelli, M.A.; et al. A novel phytocannabinoid isolated from Cannabis sativa L. with an in vivo cannabimimetic activity higher than Δ9-: Δ9-tetrahydrocannabiphorol. Sci. Rep. 2019, 9, 20335. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Elsohly, M.A.; Ross, S.A. Method of Preparing Delta-9-Tetrahydrocannabinol. U.S. Patent US6365416B1, 4 March 2002. [Google Scholar]
- Linciano, P.; Citti, C.; Russo, F.; Tolomeo, F.; Laganà, A.; Capriotti, A.L.; Luongo, L.; Iannotta, M.; Belardo, C.; Maione, S.; et al. Identification of a new cannabidiol n-hexyl homolog in a medicinal cannabis variety with an antinociceptive activity in mice: Cannabidihexol. Sci. Rep. 2020, 10, 22019. [Google Scholar] [CrossRef] [PubMed]
- Zulfiqar, F.; Ross, S.A.; Slade, D.; Ahmed, S.A.; Radwan, M.M.; Ali, Z.; Khan, I.A.; Elsohly, M.A. Cannabisol, a novel Δ9-THC dimer possessing a unique methylene bridge, isolated from Cannabis sativa. Tetrahedron Lett. 2012, 53, 3560–3562. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Harvey, D. Characterization of the butyl homologues of D-1-tetrahydrocannabinol, cannabinol and cannabidiol in samples of cannabis by combined gas chromotography and mass spectrometry. J. Pharm. Pharmacol. 1976, 28, 280–285. [Google Scholar] [CrossRef] [PubMed]
- Archer, R.A.; Boyd, D.B.; Demarco, P.V.; Tyminski, I.J.; Allinger, N. Structural studies of cannabinoids. Theoretical and proton magnetic resonance analysis. J. Am. Chem. Soc. 1970, 92, 5200–5206. [Google Scholar] [CrossRef]
- Elsohly, M.A.; Slade, D. Chemical constituents of marijuana: The complex mixture of natural cannabinoids. Life Sci. 2005, 78, 539–548. [Google Scholar] [CrossRef] [PubMed]
- Gill, E.W. Propyl homologue of tetrahydrocannabinol: Its isolation from Cannabis, properties, and synthesis. J. Chem. Soc. C 1971, 3, 579–582. [Google Scholar] [CrossRef]
- Krejcí, Z.; Šantavý, F. Isolation of two new cannabinoid acids from Cannabis sativa L. of Czechoslovak origin. Acta Univ. Olomuc. Fac. Med. 1975, 74, 161–166. [Google Scholar]
- Shani, A.; Mechoulam, R. Cannabielsoic acids: Isolation and synthesis by a novel oxidative cyclization. Tetrahedron 1974, 30, 2437–2446. [Google Scholar] [CrossRef]
- Vree, T.; Breimer, D.; van Ginneken, C.A.M.; van Rossum, J.M. Identification in hashish of tetrahydrocannabinol, cannabidiol and cannabinol analogues with a methyl side-chain. J. Pharm. Pharmacol. 1972, 24, 7–12. [Google Scholar] [CrossRef] [PubMed]
- Shoyama, Y.; Hirano, H.; Makino, H.; Umekita, N.; Nishioka, I. Cannabis. X. The isolation and structures of four new propyl cannabinoid acids, tetrahydrocannabivarinic acid, cannabidivarinic acid, cannabichromevarinic acid and cannabigerovarinic acid, from Thai Cannabis,’meao variant’. Chem. Pharma. Bull. 1977, 25, 2306–2311. [Google Scholar] [CrossRef] [Green Version]
- Shoyama, Y.; Hirano, H.; Oda, M.; Somehara, T.; Nishioka, I. Cannabichromevarin and cannabigerovarin, two new propyl homologues of cannabichromene and cannabigerol. Chem. Pharm. Bull. 1975, 23, 1894–1895. [Google Scholar] [CrossRef] [Green Version]
- Appendino, G.; Giana, A.; Gibbons, S.; Maffei, M.; Gnavi, G.; Grassi, G.; Sterner, O. A polar cannabinoid from Cannabis sativa Var. Carma. Nat. Prod. Commun. 2008, 3, 1934578X0800301207. [Google Scholar] [CrossRef] [Green Version]
- Pagani, A.; Scala, F.; Chianese, G.; Grassi, G.; Appendino, G.; Taglialatela-Scafati, O. Cannabioxepane, a novel tetracyclic cannabinoid from hemp, Cannabis sativa L. Tetrahedron 2011, 67, 3369–3373. [Google Scholar] [CrossRef]
- Taglialatela-Scafati, O.; Pagani, A.; Scala, F.; de Petrocellis, L.; di Marzo, V.; Grassi, G.; Appendino, G. Cannabimovone, a cannabinoid with a rearranged terpenoid skeleton from hemp. Eur. J. Org. Chem. 2010, 2010, 2023. [Google Scholar] [CrossRef]
- Taura, F.; Morimoto, S.; Shoyama, Y. Cannabinerolic acid, a cannabinoid from Cannabis sativa. Phytochemistry 1995, 39, 457–458. [Google Scholar] [CrossRef]
- Borges, G.R.; Birk, L.; Scheid, C.; Morés, L.; Carasek, E.; Kitamura, R.O.S.; Roveri, F.L.; Eller, S.; de Oliveira Merib, J.; de Oliveira, T.F. Simple and straightforward analysis of cannabinoids in medicinal products by fast-GC–FID. Forensic Toxicol. 2020, 38, 531–535. [Google Scholar] [CrossRef]
- Naz, S.; Hanif, M.A.; Bhatti, H.N.; Ansari, T.M. Impact of supercritical fluid extraction and traditional distillation on the isolation of aromatic compounds from Cannabis indica and Cannabis sativa. J. Essent. Oil Bear. Plants 2017, 20, 175–184. [Google Scholar] [CrossRef]
- Ross, S.A.; Elsohly, M.A. The volatile oil composition of fresh and air-dried buds of Cannabis sativa. J. Nat. Prod. 1996, 59, 49–51. [Google Scholar] [CrossRef] [PubMed]
- Strömberg, L. Minor Components of Cannabis Resin: IV. Mass spectrometric data and gas chromatographic retention times of terpenic components with retention times shorter than that of cannabidiol. J. Chromatogr. A 1974, 96, 99–114. [Google Scholar] [CrossRef]
- Turner, C.E.; Elsohly, M.A.; Boeren, E.G. Constituents of Cannabis sativa L. XVII. A review of the natural constituents. J. Nat. Prod. 1980, 43, 169–234. [Google Scholar] [CrossRef]
- Hendriks, H.; Malingré, T.M.; Batterman, S.; Bos, R. Alkanes of the essential oil of Cannabis sativa. Phytochemistry 1977, 16, 719–721. [Google Scholar] [CrossRef]
- El-Feraly, F.S.; El-Sherei, M.M.; Al-Muhtadi, F.J. Spiro-indans from Cannabis sativa. Phytochemistry 1986, 25, 1992–1994. [Google Scholar] [CrossRef]
- Chen, B.; Cai, G.; Yuan, Y.; Li, T.; He, Q.; He, J.F. Chemical constituents in hemp pectin I. Chin. J. Exp. Tradit. Med. Form. 2012, 18, 98–100. [Google Scholar]
- Boeren, E.; Elsohly, M.; Turner, C.; Salemink, C. ß-Cannabispiranol: A new non-cannabinoid phenol from Cannabis sativa L. Experiential 1977, 33, 848. [Google Scholar] [CrossRef] [PubMed]
- Slatkin, D.J.; Doorenbos, N.J.; Harris, L.S.; Masoud, A.N.; Quimby, M.W.; Schiff, P.L. Chemical constituents of Cannabis sativa L. root. J. Pharm. Sci. 1971, 60, 1891–1892. [Google Scholar] [CrossRef]
- Latter, H.L.; Abraham, D.J.; Turner, C.E.; Knapp, J.E.; Schiff, P.L., Jr.; Slatkin, D.J. Cannabisativine, a new alkaloid from Cannabis sativa L. root. Tetrahedron Lett. 1975, 16, 2815–2818. [Google Scholar] [CrossRef]
- Shoyama, Y.; Nishioka, I. Cannabis. XIII. Two new spiro-compounds, cannabispirol and acetyl cannabispirol. Chem. Pharm. Bull. 1978, 26, 3641–3646. [Google Scholar] [CrossRef]
- Sánchez-Duffhues, G.; Calzado, M.A.; de Vinuesa, A.G.; Caballero, F.J.; Ech-Chahad, A.; Appendino, G.; Krohn, K.; Fiebich, B.L.; Muñoz, E. Denbinobin, a naturally occurring 1, 4-phenanthrenequinone, inhibits HIV-1 replication through an NF-κB-dependent pathway. Biochem. Pharmacol. 2008, 76, 1240–1250. [Google Scholar] [CrossRef] [PubMed]
- Nalli, Y.; Arora, P.; Riyaz-Ul-Hassan, S.; Ali, A. Chemical investigation of Cannabis sativa leading to the discovery of a prenylspirodinone with anti-microbial potential. Tetrahedron Lett. 2018, 59, 2470–2472. [Google Scholar] [CrossRef]
- Crombie, L.; Crombie, W.M.L. Natural products of Thailand high Δ 1-THC-strain Cannabis. the bibenzyl-spiran-dihydrophenanthrene group: Relations with cannabinoids and canniflavones. J. Chem. Soc. Perkin Trans. 1982, 1, 1455–1466. [Google Scholar] [CrossRef]
- Ross, S.; Elsohly, M.A. Constituents of Cannabis sativa L. XXVIII. A review of the natural constituents. Zagazig J. Pharm. Sci. 1995, 4, 1–10. [Google Scholar] [CrossRef]
- Elsohly, H.N.; Ma, G.E.; Turner, C.E.; Elsohly, M.A. Constituents of Cannabis sativa, XXV. Isolation of two new dihydrostilbenes from a panamanian variant. J. Nat. Prod. 1984, 47, 445–452. [Google Scholar] [CrossRef] [PubMed]
- Ottersen, T.; Aasen, A.; El-Feraly, F.S.; Turner, C.E. X-ray structure of cannabispiran: A novel Cannabis constituent. Chem. Commun. 1976, 15, 580–581. [Google Scholar] [CrossRef]
- Ross, S.A.; Elsohly, M.A.; Sultana, G.N.; Mehmedic, Z.; Hossain, C.F.; Chandra, S. Flavonoid glycosides and cannabinoids from the pollen of Cannabis sativa L. Phytochem. Anal. Int. J. Plant Chem. Biochem. Tech. 2005, 16, 45–48. [Google Scholar] [CrossRef] [PubMed]
- Cheng, L.; Kong, D.; Hu, G.; Li, H. A new 9, 10-dihydrophenanthrenedione from Cannabis sativa. Chem. Nat. Compd. 2010, 46, 710–712. [Google Scholar] [CrossRef]
- Bercht, C.; van Dongen, J.; Heerma, W.; Lousberg, R.C.; Küppers, F. Cannabispirone and cannabispirenone, two naturally occurring spiro-compounds. Tetrahedron 1976, 32, 2939–2943. [Google Scholar] [CrossRef]
- Hammond, C.T.; Mahlberg, P.G. Phloroglucinol glucoside as a natural constituent of Cannabis sativa. Phytochemistry 1994, 37, 755–756. [Google Scholar] [CrossRef]
- Namdar, D.; Voet, H.; Ajjampura, V.; Nadarajan, S.; Mayzlish-Gati, E.; Mazuz, M.; Shalev, N.; Koltai, H. Terpenoids and phytocannabinoids co-produced in Cannabis sativa strains show specific interaction for ellcytotoxic activity. Molecules 2019, 24, 3031. [Google Scholar] [CrossRef] [Green Version]
- Whalley, B.; Stephens, G.; Williams, C.; Guy, G.; Wright, S.; Kikuchi, T.; GW Pharma Ltd.; Otsuka Pharmaceutical Co Ltd. Use of One or a Combination of Phyto-Cannabinoids in the Treatment of Epilepsy. U.S Patent 9,066,920, 30 June 2015. [Google Scholar]
- Parker, L.; Rock, E.; Sticht, M.; Wills, K.; Limebeer, C.L. Cannabinoids suppress acute and anticipatory nausea in preclinical rat models of conditioned gaping. Pharm. Ther. 2015, 97, 559–561. [Google Scholar] [CrossRef] [PubMed]
- Erridge, S.; Mangal, N.; Salazar, O.; Pacchetti, B.; Sodergren, M.H. Cannflavins—From plant to patient: A scoping review. Fitoterapia 2020, 146, 104712. [Google Scholar] [CrossRef] [PubMed]
- Pacher, P.; Mechoulam, R. Is lipid signaling through cannabinoid 2 receptors part of a protective system? Prog. Lipid Res. 2011, 50, 193–211. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mcguire, P.; Robson, P.; Cubala, W.J.; Vasile, D.; Morrison, P.D.; Barron, R.; Taylor, A.; Wright, S.J. Cannabidiol (CBD) as an adjunctive therapy in schizophrenia: A multicenter randomized controlled trial. Am. J. Psychiatry 2018, 175, 225–231. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Parker, L.A.; Rock, E.M.; Limebeer, C.L. Regulation of nausea and vomiting by cannabinoids. Br. J. Pharmacol. 2011, 163, 1411–1422. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yeshurun, M.; Shpilberg, O.; Herscovici, C.; Shargian, L.; Dreyer, J.; Peck, A.; Israeli, M.; Levy-Assaraf, M.; Gruenewald, T.; Mechoulam, R.; et al. Cannabidiol for the prevention of graft-versus-host-disease after allogeneic hematopoietic cell transplantation: Results of a phase II study. Biol. Blood Marrow Transpl. 2015, 21, 1770–1775. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brierley, D.I.; Samuels, J.; Duncan, M.; Whalley, B.J.; Williams, C.M. Cannabigerol is a novel, well-tolerated appetite stimulant in pre-satiated rats. Psychopharmacology 2016, 233, 3603–3613. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rock, E.; Kopstick, R.L.; Limebeer, C.L.; Parker, L.A. Tetrahydrocannabinolic acid reduces nausea-induced conditioned gaping in rats and vomiting in S uncus murinus. Br. J. Pharmacol. 2013, 170, 641–648. [Google Scholar] [CrossRef] [Green Version]
- Rock, E.M.; Sticht, M.A.; Parker, L.A. Effect of Phytocannabinoids on Nausea and Vomiting; Oxford University Press: Oxford, UK, 2014. [Google Scholar]
- Tsien, R.; Whalley, B.J.; Devinsky, O. Cannabinoids and Epilepsy. Neurotherapeutics 2015, 12, 747–768. [Google Scholar]
- Moreno-Sanz, G. Can you pass the acid test? critical review and novel therapeutic perspectives of Δ9-tetrahydrocannabinolic acid A. Cannabis Cannabinoid Res. 2016, 1, 124–130. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rock, E.; Parker, L.A. Effect of low doses of cannabidiolic acid and ondansetron on licl-induced conditioned gaping (a model of nausea-induced behaviour) in rats. Br. J. Pharmacol. 2013, 169, 685–692. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rock, E.M.; Connolly, C.; Limebeer, C.L.; Parker, L.A. Effect of combined oral doses of Δ 9-tetrahydrocannabinol (THC) and cannabidiolic acid (CBDA) on acute and anticipatory nausea in rat models. Psychopharmacology 2016, 233, 3353–3360. [Google Scholar] [CrossRef] [PubMed]
- Russo, E.B. Taming THC: Potential cannabis synergy and phytocannabinoid-terpenoid entourage effects. Br. J. Pharmacol. 2011, 163, 1344–1364. [Google Scholar] [CrossRef] [PubMed]
- Cascone, P.; Iodice, L.; Maffei, M.E.; Bossi, S.; Arimura, G.I.; Guerrieri, E. Tobacco overexpressing β-ocimene induces direct and indirect responses against aphids in receiver tomato plants. J. Plant Physiol. 2015, 173, 28–32. [Google Scholar] [CrossRef] [PubMed]
- Bomfim, L.M.; Menezes, L.R.; Rodrigues, A.C.; Dias, R.B.; Gurgel Rocha, C.A.; Soares, M.B.; Neto, A.F.; Nascimento, M.P.; Campos, A.F.; Silva, L.C.; et al. Antitumour activity of the microencapsulation of Annona vepretorum essential oil. Basic Clin. Pharmacol. 2016, 118, 208–213. [Google Scholar] [CrossRef] [PubMed]
- De Oliveira Ramalho, T.R.; de Oliveira, M.T.; de Araujo Lima, A.L.; Bezerra-Santos, C.R.; Piuvezam, M.R. Gamma-terpinene modulates acute inflammatory response in mice. Planta Med. 2015, 81, 1248–1254. [Google Scholar]
- Fitsiou, E.; Anestopoulos, I.; Chlichlia, K.; Galanis, A.; Kourkoutas, I.; Panayiotidis, M.I.; Pappa, A. Antioxidant and antiproliferative properties of the essential oils of Satureja thymbra and Satureja parnassica and their major constituents. Anticancer Res. 2016, 36, 5757–5763. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kasuya, H.; Okada, N.; Kubohara, M.; Satou, T.; Masuo, Y.; Koike, K. Expression of BDNF and TH mRNA in the Brain Following Inhaled Administration of A-Pinene. Phytother. Res. 2015, 29, 43–47. [Google Scholar] [CrossRef] [PubMed]
- Han, H.D.; Cho, Y.J.; Cho, S.K.; Byeon, Y.; Jeon, H.N.; Kim, H.S.; Kim, B.G.; Bae, D.S.; Lopez-Berestein, G.; Sood, A.K.; et al. Linalool-incorporated nanoparticles as a novel anticancer agent for epithelial ovarian carcinoma. Mol. Cancer Ther. 2016, 15, 618–627. [Google Scholar] [CrossRef] [Green Version]
- Russo, E.B. Cannabis and epilepsy: An ancient treatment returns to the fore. Epilepsy Behav. 2017, 70, 292–297. [Google Scholar] [CrossRef] [PubMed]
- Sulak, D.; Saneto, R.; Goldstein, B. The current status of artisanal Cannabis for the treatment of epilepsy in the United States. Epilepsy Behav. 2017, 70, 328–333. [Google Scholar] [CrossRef]
- Piccinelli, A.C.; Santos, J.A.; Konkiewitz, E.C.; Oesterreich, S.A.; Formagio, A.S.; Croda, J.; Ziff, E.B.; Kassuya, C. Antihyperalgesic and antidepressive actions of (R)-(+)-Limonene, A-phellandrene, and essential oil from schinus terebinthifolius fruits in a neuropathic pain model. Nutr. Neurosci. 2015, 18, 24. [Google Scholar] [CrossRef]
- Siqueira, H.D.; Neto, B.S.; Sousa, D.P.; Gomes, B.S.; da Silva, F.V.; Cunha, F.V.; Wanderley, C.W.; Pinheiro, G.; Cândido, A.G.; Wong, D.V.; et al. A-Phellandrene, a cyclic monoterpene, attenuates inflammatory response through neutrophil migration inhibition and mast cell degranulation. Life Sci. 2016, 160, 27–33. [Google Scholar] [CrossRef]
- Lima, D.F.; Brandão, M.S.; Moura, J.B.; Leitão, J.M.; Carvalho, F.A.; Miúra, L.M.; Leite, J.R.; Sousa, D.P.; Almeida, F.R. Antinociceptive activity of the monoterpene α-phellandrene in rodents: Possible mechanisms of action. J. Pharm. Pharmacol. 2012, 64, 283–292. [Google Scholar] [CrossRef] [PubMed]
- Aydin, E.; Türkez, H.; Taşdemir, Ş. Anticancer and antioxidant properties of terpinolene in rat brain cells. Arch. Ind. Hyg. Toxicol. 2013, 64, 415–424. [Google Scholar] [CrossRef] [PubMed]
- Turkez, H.; Aydın, E.; Geyikoglu, F.; Cetin, D. Genotoxic and oxidative damage potentials in human lymphocytes after exposure to terpinolene in vitro. Cytotechnology 2015, 67, 409–418. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Paula-Freire, L.I.; Andersen, M.; Gama, V.; Molska, G.; Carlini, E.L. The oral administration of trans-caryophyllene attenuates acute and chronic pain in mice. Phytomedicine 2014, 21, 356–362. [Google Scholar] [CrossRef] [PubMed]
- Varga, Z.V.; Matyas, C.; Erdelyi, K.; Cinar, R.; Nieri, D.; Chicca, A.; Nemeth, B.T.; Paloczi, J.; Lajtos, T.; Corey, L.; et al. Research Paper Themed Issue Β-Caryophyllene protects against alcoholic steatohepatitis by attenuating inflammation and metabolic dysregulation in mice. Br. J. Pharmacol. 2017, 175, 320–334. [Google Scholar] [CrossRef] [PubMed]
- Katsuyama, S.; Mizoguchi, H.; Kuwahata, H.; Komatsu, T.; Nagaoka, K.; Nakamura, H.; Bagetta, G.; Sakurada, T.; Sakurada, S. Involvement of peripheral cannabinoid and opioid receptors in β-caryophyllene-induced antinociception. Eur. J. Pain 2013, 17, 664–675. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.B.; Zheng, L.P.; Li, L.; Xu, L.Z.; Fu, J. Elemene, one ingredient of a chinese herb, against malignant tumors: A literature-based meta-analysis. Cancer Invest. 2013, 31, 156–166. [Google Scholar] [CrossRef] [PubMed]
- Yang, Q.; Wu, J.; Luo, Y.; Huang, N.; Zhen, N.; Zhou, Y.; Sun, F.; Li, Z.; Pan, Q.; Li, Y. (−)-Guaiol Regulates RAD51 stability via autophagy to induce cell apoptosis in non-small cell lung cancer. Oncotarget 2016, 7, 62585. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chinsembu, K.C. Tuberculosis and nature’s pharmacy of putative anti-tuberculosis agents. Acta Tropica 2016, 153, 46–56. [Google Scholar] [CrossRef] [PubMed]
- Semenya, S.; Potgieter, M.; Tshisikhawe, M.; Shava, S.; Maroyi, A. Medicinal utilization of exotic plants by bapedi traditional healers to treat human ailments in Limpopo province, South Africa. J. Ethnopharmacol. 2012, 144, 646–655. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.H.; Son, J.K.; Jung, B.; Zheng, M.; Kim, J.R. Epifriedelanol from the root bark of Ulmus davidiana inhibits cellular senescence in human primary cells. Planta Medica 2011, 77, 441–449. [Google Scholar] [CrossRef] [Green Version]
- Zhou, X.; Wang, F.; Zhou, R.; Song, X.; Xie, M. Apigenin: A current review on its beneficial biological activities. J. Food Biochem. 2017, 41, e12376. [Google Scholar] [CrossRef]
- He, M.; Min, J.W.; Kong, W.L.; He, X.H.; Li, J.X.; Peng, B.W. A review on the pharmacological effects of vitexin and isovitexin. Fitoterapia 2016, 115, 74–85. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A.; Kashyap, D.; Sak, K.; Tuli, H.S.; Sharma, A.K. Therapeutic charm of quercetin and its derivatives: A review of research and patents. Pharm. Pat. Anal. 2018, 7, 15–32. [Google Scholar] [CrossRef] [PubMed]
- Ashaari, Z.; Hassanzadeh, G.; Alizamir, T.; Yousefi, B.; Keshavarzi, Z.; Mokhtari, T. The flavone luteolin improves central nervous system disorders by different mechanisms: A review. J. Mol. Neurosci. 2018, 65, 491–506. [Google Scholar] [CrossRef]
- Rudroff, T.; Sosnoff, J.J. Cannabidiol to improve mobility in people with multiple sclerosis. Front. Neurol. 2018, 9, 183. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wong, K.U.; Baum, C.R. Acute Cannabis Toxicity. Pediatr. Emerg. Care 2019, 35, 799–806. [Google Scholar] [CrossRef] [PubMed]
- International Conference on Harmonisation (ICH). All Guidelines. Available online: https://www.ich.org/page/ich-guidelines (accessed on 19 April 2020).
- Schlag, A.K.; O’sullivan, S.E.; Zafar, R.R.; Nutt, D.J. Current controversies in medical cannabis: Recent developments in human clinical applications and potential therapeutics. Neuropharmacology 2021, 191, 108586. [Google Scholar] [CrossRef]
- Hill, K.P. Cannabinoids and the coronavirus. Cannabis Cannabinoid Res. 2020, 5, 118–120. [Google Scholar] [CrossRef]
- Costiniuk, C.T.; Jenabian, M.A. Acute inflammation and pathogenesis of SARS-CoV-2 infection: Cannabidiol as a potential anti-inflammatory treatment? Cytokine Growth Factor Rev. 2020, 53, 63. [Google Scholar] [CrossRef] [PubMed]
- Byrareddy, S.N.; Mohan, M. SARS-CoV2 induced respiratory distress: Can cannabinoids be added to anti-viral therapies to reduce lung inflammation? Brain Behav. Immun. 2020, 87, 120. [Google Scholar] [CrossRef] [PubMed]
- Esposito, G.; Pesce, M.; Seguella, L.; Sanseverino, W.; Lu, J.; Corpetti, C.; Sarnelli, G. The potential of cannabidiol in the COVID-19 pandemic. Br. J. Pharmacol. 2020, 177, 4967–4970. [Google Scholar] [CrossRef] [PubMed]
- O’sullivan, S.E.; Stevenson, C.W.; Laviolette, S.R. Could cannabidiol be a treatment for coronavirus disease-19-related anxiety disorders? Cannabis Cannabinoid Res. 2021, 6, 7–18. [Google Scholar] [CrossRef] [PubMed]


| S/N * | Class of Compounds | Plant Part(s) | Isolated Compounds | References |
|---|---|---|---|---|
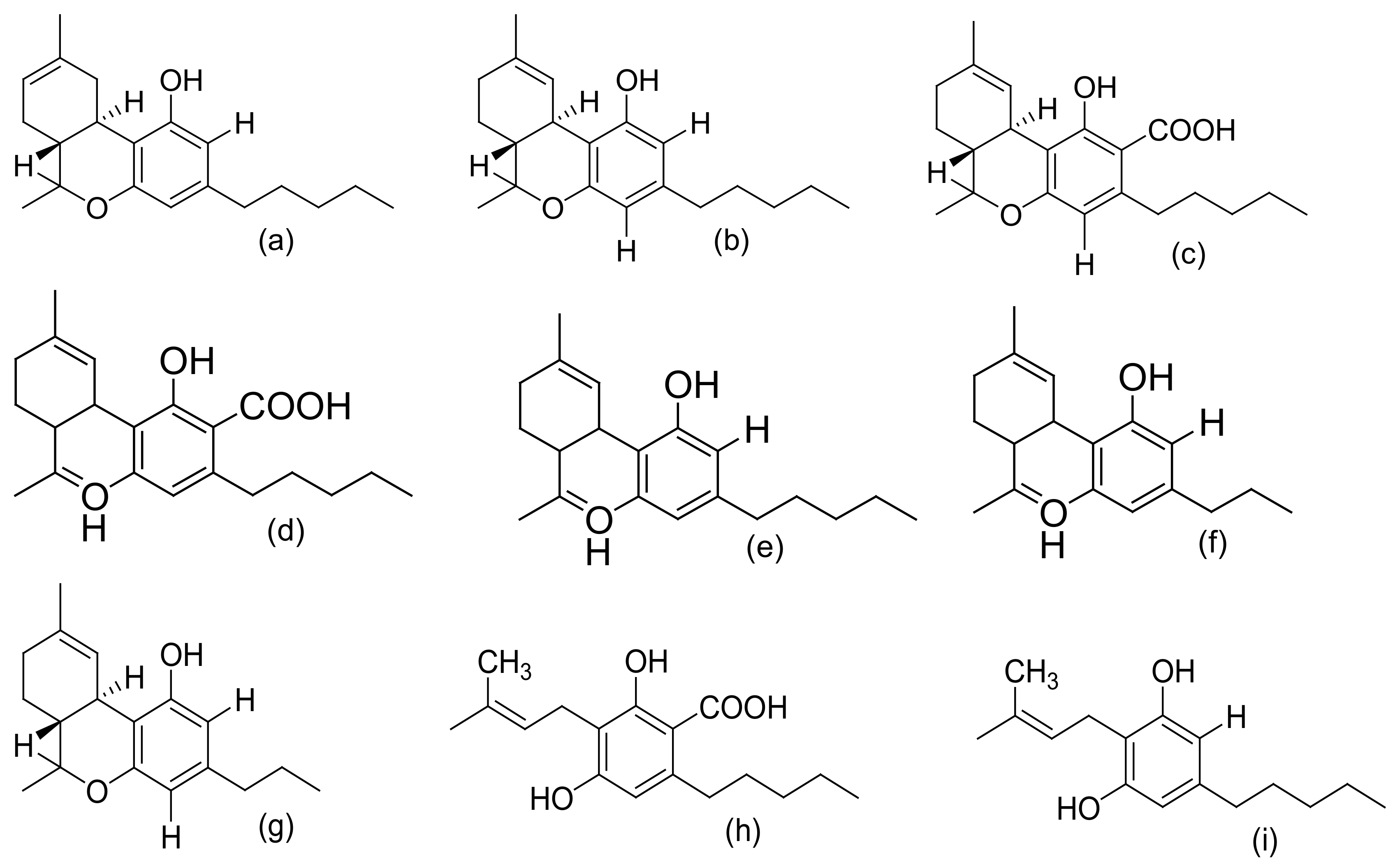
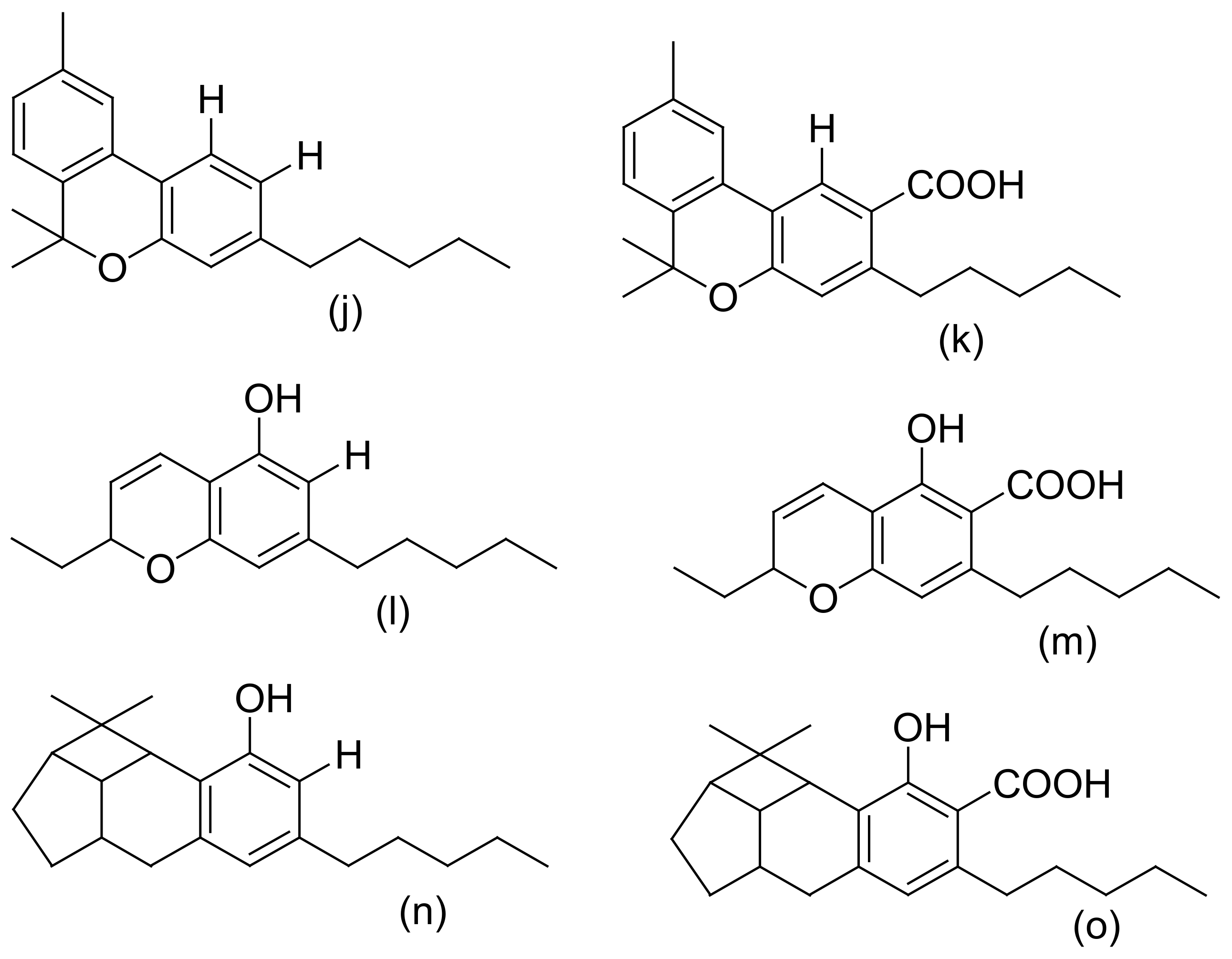
| 1 | Cannabinoids: -Cannabinoid acids -Neutral cannabinoids -Cannabinoid derivatives -Cannabinoid acid esters | Leaves, flowers, resin, stembarks, and roots | Δ9-tetrahydrocannabivarin, α/β-fenchyl Δ9-tetrahydrocannabinolate, Δ9-tetrahydrocannabinol, α-terpenyl (−)-Δ9-trans-tetrahydrocannabinolate, γ-eudesmyl (−)Δ9-trans-tetrahydrocannabinolate, 8α-hydroxy-(−)-Δ9-trans-tetrahydrocannabinol, 8b-hydroxy-(−)- Δ9-trans-tetrahydro cannabinol, 8-oxo-(−)-Δ9-trans-tetrahydrocannabinol, Cannabisol, (−)-Δ9-trans-tetrahydrocannabiphorol, (−)-Δ9-trans-tetrahydrocannabihexol, (−)-Δ8-trans-tetrahydrocannabinol, Δ8- trans-tetrahydrocannabinolic acid, Cannabigerol, 6,7-trans/cis-epoxycannabigerolic acid, Sesquicannabigerol, Cannabigerolic acid, Cannnabigerovarin, Cannabidiol, C4-Cannabidiol, Cannabidivarin, C4-tetrahydrocannabinol, Cannabichromene, Cannabichromevarin, Cannabichromanone, -D4-acetoxycannabichromene, Cannabicitran, Cannabiripsol, Cannabicoumaronone, Cannabifuran, Cannabielsoin, Cannabielsoic acid, Cannabicyclol, Cannabinodiol, bornyl/epi-bornyl-Δ9-tetrahydrocannabinlate Cannabinol, Cannabitriol, Cannabimovone, and Cannabioxepane | [8,11,13,24,25,26,27,28,29,30,31] |
| Non-Cannabinoid Constituents | ||||
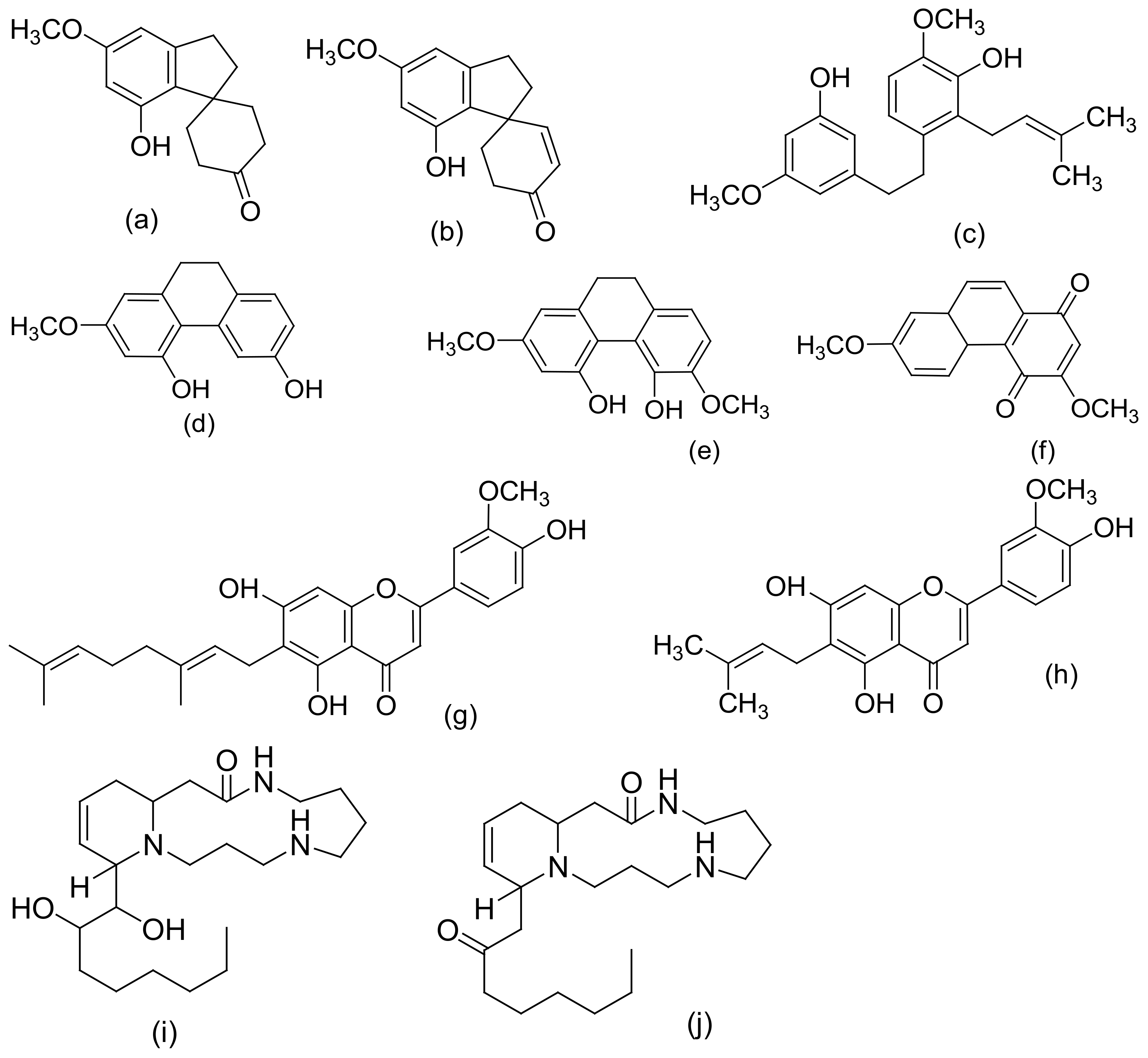
| 2 | Non-cannabinoid phenol: -Stilbenoids -Spiro-indans -Phenanthrenes -Lignans, lignanamides, and phenolic amides | Leaves, flowers, stem, hemp pectin, resin, fruit, seed, and root | Dihydrostilbenes, Dihydrophenathrenes, Cannabistilbene, Canniprene, Cannithrene, Denbinobin, Phloroglucinol β-D-glucoside, Cannabispiran, Cannabispirone, Cannabispirenone, Cannabispirol, Cannabispirketal, a-cannabispiranol-4′-O-β-glucopyranose, prenylspirodienone, 4,5-dihydroxy-2,3,6-trimethoxy-9,10-dihydrophenanthrene, Cannabisin A–O 4,7-dimethoxy-1,2,5-trihydroxyphenathrene, 5-methyl-4-pentyl-2,6,2-trihydroxybiphenyl, 5-methyl-4-pentylbiphenyl-2,2,6-triol, N-trans-coumaroyltyramine, N-trans-feruloyltyramine, Ntrans-caffeoyltyramine, 3,3′-demethylheliotropamide, and Grossamide. | [8,13,28,32,33,34] |
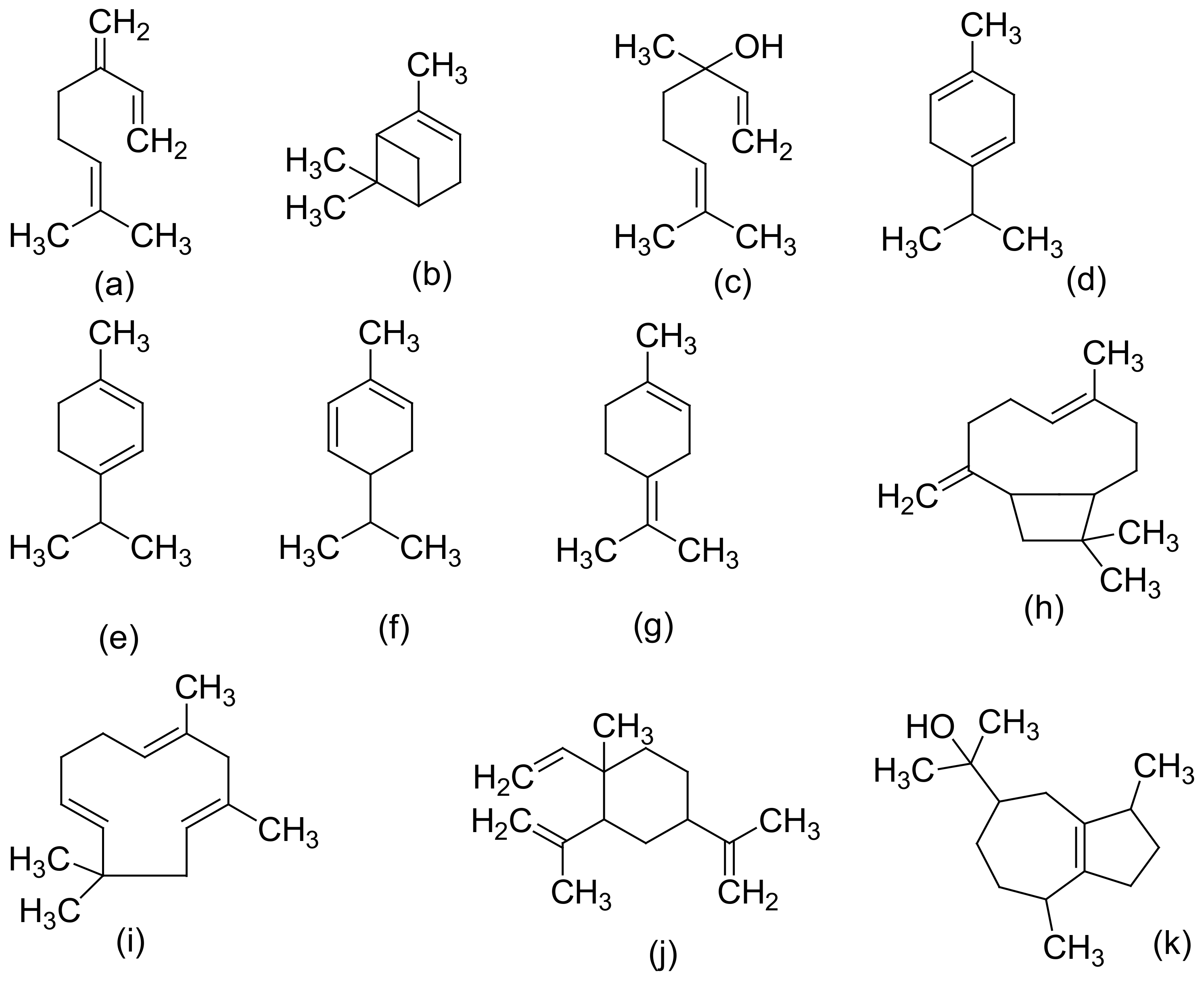
| 3 | Terpenoids (Terpenes): -Monoterpenes -Sesquiterpenes -Diterpenes -Triterpene, | Essential oils of fresh and dried leaves, flowers, stembarks, and roots | α-pinene, β-pinene, linalool, linalool oxide, myrcene, limonene, camphene, α-terpinene, γ-terpinene, α-terpinolene, α-terpineol, terpinene-4-ol, sabinene, sabinene hydrate, cis-sabinene hydrate, α-phellandrene, β-phellandrene, 2-methyl-2-heptene-6-one, borneol, piperitenone, geraniol, carvacrol, carvone, cis-carveol, citronellol, bornyl acetate, ipsdienol, germacrene-B, clovandiol, α-bisabolol, β-eudesmol, γ-eudesmol, α-caryophyllene, β-caryophyllene oxide, α-Humulene, Phytol, neophytadiene, friedelin (friedelan-3-one), epifriedelanol, β-amyrin, Vomifoliol, dihydrovomifoliol, and dihydroactinidiolide β-ionone | [8,11,13,28,29,35,36] |
| 4 | Flavonoids: -Methylated -Glycosylated (C or O glycosides) -Prenylated -Geranylated | Leaves, flowers, seed, and fruit | Orientin, Vitexin, Isovitexin, Apigenin, Luteolin, Kaempferol, Quercetin, Cytisoside, Cytisoside glucoside, Canniflavone (Cannflavin), Naringenin, and Naringin | [8,11,13,28,29,35,37,38,39] |
| 5 | Sterols | Stembarks, roots, and leaves | Campsterol, Stigmasterol, and β- Sitosterol | [11,40] |
| 7 | Alkaloids | Roots, leaves, stembark | Cannabisativine and Anhydrocannabisativine | [13,40] |
| 8 | Fatty acids: -Saturated and unsaturated fatty acids and their esters | Seeds | Roughanic acid, Stearidonic acid, α-linolenic acid, and oxylipins. | [8,28] |
| 9 | Hydrocarbons (n-alkane) | - | Δ9-Tetrahydrocannabiorcolic acid | [8] |
| Extraction Solvent(s) | Matrix and Species | Identification, Isolation, and Purification Methods | Elucidation/Analytical Techniques | Analytes | References |
|---|---|---|---|---|---|
| Phenolic Cannabinoids | |||||
| Methanol/chloroform mixture. | C. sativa inflorescence | Qualitative, quantitative, and comparative derivatization study of cannabinoids | Fast GC–MS | CBDA, CBGA, CBG, CBD, THC, Δ8-THC, CBC, THCA, THC | [47] |
| Supercritical fluid extraction | Plant biomass and medicinal Cannabis resin | Quantitative and qualitative analysis of cannabinoids | UHPLC–DAD and statistical analysis | CBDA, THCA, CBD, CBN, CBC, THC | [48] |
| Methanol/chloroform solvent mixture | Cannabis flower samples | Qualitative and quantitative measurement of cannabinoids | HPLC–DAD | Δ9-THC, CBD, CBDA, THCA, CBN, CBG, CBGA, Δ8-THC | [49] |
| Ethanol/ethanolic extracts | (i) Lebanese C. sativa (ii) Cannabis (iii) Decarbo-xylated hemp leaves | (i) Purified by counter-current distribution and silica gel chromatography (ii) Florisil and silica gel column chromatography | GCMS, IR, and 1H NMR comparison with an authentic sample | Cannabielsoin, (+)-trans-CBT and (−)-trans -CBT-OEt-C5(Cannibitriol), Cannabicitran, and Monomethylether of CBD | [13,50,51,52] |
| Sequential extraction (hexanes, CH2Cl2, EtOAc, EtOH, EtOH/H2O, and H2O) | (i) Bud and leaves of high-potency variety of C. sativa | (i) Silica gel VLC, C18-solid phase extraction (SPE), and HPLC (ii) VLC chromatography of hexane extract, TLC, flash silica gel, Sephadex LH-20 chromatography, and semipreparative reversed-phase (RP) and chiral HPLC | HRESIMS, 1D and 2D NMR, GC–MS | epi-bornyl Δ9-tetrahydrocannabinolate, α-terpenyl Δ9-tetrahydrocannabinolate, 4-terpenyl Δ9-tetrahydro-cannabinolate, α-cadinyl Δ9-tetrahydrocannabinolate, γ-eudesmyl Δ9-tetrahydro-cannabinolate, γ-eudesmyl cannabigerolate, 4-terpenyl cannabinolate, bornyl Δ9-tetrahydrocannabinolate, α-fenchyl Δ9-tetrahydro-cannabinolate, α-cadinyl cannabigerolate, Δ9-tetrahydro-cannabinol (Δ9-THC), Δ9-tetrahydrocannabinolic acid A (Δ9-THCA), Cannabinolic acid A (CBNA), and Cannabigerolic acid (CBGA), (±)-6,7-trans-epoxycannabigerolic acid, (+)-6,7-cis-epoxycannabigerolic acid, (±)-6,7-cis-epoxycannabigerol, 5′-Methoxy-cannabigerolic acid, 5′-methyl-4-pentylbiphenyl-2, 2′, 6-triol,7-methoxy-cannabispirone, (±)-6,7- trans-epoxycannabigerol, 8α-hydroxy-(−)-Δ9-trans-tetrahydrocannabinol, 8b-hydroxy-(−)- Δ9-trans-tetrahydro cannabinol, 8-oxo-(−)-Δ9-trans-tetrahydrocannabinol, 10α-hydroxy-Δ8-tetra-hydrocannabinol, 10β-hydroxy- Δ8-tetra-hydrocannabinol, 10a-α-hydroxy-10-oxo-Δ8-tetrahydrocannabinol, (±)-4-acetoxycannabichromene, (±)-3″-hydroxy-Δ(4″, 5″)-cannabichromene,(−)-7-hydroxycannabichromane,(−)-7 R-cannabicoumarononic acid A, 5-acetyl-4-hydroxycannabigerol, 4-acetoxy-2-geranyl-5-hydroxy-3-n-pentylphenol, 8-hydroxycannabinol, 8-hydroxycannabinolic acid A, and 2-geranyl-5-hydroxy-3-n-pentyl-1, 4-benzoquinone, (±)-4-acetoxycannabichromene, (±)-3”-hydroxy- Δ4”-cannabichromene, (–)-7-hydroxycannabichromane, 8-hydroxycannabinol, 8-hydroxy cannabinolic acid, 10S-hydroxy-cannabinol, 9b,10b-epoxyhexahydrocannabinol, 9a-hydroxyhexahydrocannabinol, 10a-hydroxyhexahydrocannabinol, and 10aRhydroxyhexahydrocannabinol | [31,53,54,55] |
| Hexane extract | (i) C. sativa (air-dried and powdered buds) (ii) high-potency variety of C. sativa (iii) Illicit Cannabis samples (iv) C. sativa inflorescence (strain CINRO) (v) Lebanese C. sativa (vi) Hemp (vii) Nepalese and Brazilian C. Sativa. | (i) Column chromatography using silica or alumina, TLC, then fractional distillation and preparative C18 HPLC (ii) VLC (vacuum liquid chromatography) silica gel column chromatography, C18 HPLC and chiral HPLC (iii) Flash silica gel chromatography (iv) Florisil column chromatography | 1H NMR, 13C NMR (2D NMR) HRESIMS, circular dichroism (CD), UV, LC-HRMS, MS/MS, GC–MS, and confirmation by phytochemical transformations. | Δ9-THC, Δ9-THC aldehyde, Cannabinoid esters, Cannabisol, Δ9-trans-tetrahydrocannabiphorol, Δ9-trans-tetrahydrocannabihexol, Cannabidiorcol, Cannabidihexol (CBDH) and Cannabidiphorol (CBDP), Cannabitwinol, Cannabinodivirin and cannabinodiol (CBND), Cannabichromene (CBC), 9a-hydroxyhexahydrocannabinol, 7-oxo-9a-hydroxyhexa-hydrocannabinol, 10a-hydroxyhexahydrocannabinol, and 10a-R-hydroxyhexahydrocannabinol | [53,56,57,58,59,60] |
| Ethyl acetate extracts | Cannabis resins, tinctures, and leaves | - | GC–MS and GC–FID analysis | Δ9-THC and Δ9-THCA | [61] |
| Petroleum ether | (i) Cannabis tincture of Pakistani origin (ii) Brazilian C. sativa (iii) Cannabis leaves and flowers (Maryland and Czechoslovakian origin) (iv) Congo C. sativa (v) Hashish and Cannabis sativa | (i) Silicic acid column chromatography (ii) Silica gel and Florisil chromatography, preparative TLC | IR, NMR, MS, GC–MS confirmed by synthesis | Δ9-THCV, Δ9-THCO or Δ9-THC, Δ8-THC (Δ8-THCA), Cannabielsoin acid A (CBEAA,), Cannabielsoin acid B, and Cannabicyclovarin (CBLV) | [61,62,63,64,65,66,67] |
| Benzene | (i) Fresh C. sativa leaves from Thailand (ii) Fresh tops and leaves of C. sativa (iii) Hemp | Polyamide and silica gel column chromatography | IR, UV, NMR, and comparing UV spectrum with that of derivatives | Δ9-THCVA, CBDV, THCV, CBCV, Cannabigerovarin CBGV, cannabigerovarinic acid (CBGVA), CBDA, cannabidivarinic acid (CBDVA), Cannabicyclolic acid (CBLA), and Cannabichromenic acid (CBCA) | [49,68,69] |
| Acetone extract | (i) Leaves of C. sativa (Mexican strain) (ii) Wax of decarboxylated aerial parts of C. sativa (Carma strain) (iii) Cannabis variety (carmagnola) | (i) Silica gel column chromatography (ii) Silica and alumina column chromatography, followed by normal phase (NP)-HPLC. (iii). Flash chromatography, over reverse-phased C18 silica gel followed by normal-phase HPLC | FAB–MS, 1H-NMR, 13C-NMR), and ESI–MS semisyn-thesis. | Cannabigerolic acid (CBGA), dihydroxycannabigerol derivative (camagerol), Sesquicannabi-gerol, Cannabimovone, and Cannabioxepane | [70,71,72,73] |
| Essential/volatile oils | |||||
| Methanol dilutions | Cannabis sativa oil samples | Separation/quantitation of cannabinoids | Fast-GC–FID | CBD, CBN, and THC | [74] |
| Essential oil | Fresh C. sativa L. from India | Fractional distillation and chromatography over alumina. | GC–MS and physico–chemical analyses | α-terpinene, β-phellandrene, γ-terpinene, α-terpinolene, α-pinene, β-pinene, camphene, linalool, α-terpineol, terpinene-4-ol, linalool oxide, and sabinene hydrate | [13] |
| Volatile/essential oils | (i) Cannabis (Dutch and Turkish) (ii) Fresh leaves of Cannabis sativa and Cannabis indica | (i) Hydrodistillation or through nitrogen extraction (ii) Hydrodistillation, steam distillation, and supercritical fluid extraction | Capillary gas chromato-graphy, GC–MS analysis | cis-β-ocimene, trans-β-ocimene, α-phellandrene, D3-carene, Δ4-carene, sabinene and α-thujene, caryophyllene, humulene, trans-β-bergamotene, cis-β–farnesene, δ–limonene, carophyllene oxide, linalool, trans-α- bergamotene, cis- β -farnesene, menthol, eucalyptol, and Carvone. | [13,75] |
| Essential oil | Cannabis (marijuana fresh and dried buds) | Steamdistillation | GC–MS and GC–FID | Ipsdienol, cis-carveol, and cis-sabinene hydrate | [76] |
| Essential oil | C. sativa resin | Minor terpenic component analysis | GC–MS and GC retention time | α-gurjunene, α-bisabolol, α-cedrene, α-cubebene, δ-cadinene, epi-β-santalene, farnesol, γ-cadinene, γ-elemene, γ-eudesmol, guaiol, (E,E)-α-farnesene, (Z)-β-farnesene, and farnesyl acetone | [77] |
| Essential oil | Cannabis | Steam distillation and silica gel chromatography | GC, GC–MS | eugenol, methyleugenol, iso-eugenol, trans-anethol, and cis-anethol (simple phenols) | [78] |
| Essential oil | C. sativa | Column chromatography of the essential oil | GC and GC–MS analyses | Iso-caryophyllene, β-selinene, selina-3,7(11)-diene, and selina-4(14),7(11)-diene | [13,79] |
| Non-cannabinoid phenols | |||||
| Ethanol/ethanolic extract | (i) South African Cannabis variant (ii) Saudi Arabia hashish (iii) Leaves of C. sativa (iv) High-potency Cannabis variety grown in Mississippi (v) Hemp pectin (vi). Cannabis roots (vii) Roots, stem, and leaves of a Mexican variant of Cannabis sativa | (i) Partitioning and chromatography on silica and polyamide columns (ii) Normal and reversed phase chromatographic techniques (iii) Purification by macro reticular resin, silica gel column chromatography, and Sephadex-LH-20 (iv) TLC, chromatography over alumina, and recrystallization (v) Partitioning and TLC eluted with chloroform:acetone:ammonia (1:1:1) (vi) Series of acid–base extractions and silica-gel chromatography followed by crystallization of the alkaloid from acetone | IR, GCMS, UV, 1D NMR (1H NMR, 13C NMR) and 2DNMR (COSY, HSQC, HMBC, and ROESY), ESI–MS, comparison with authentic samples, X-ray crystallography, and semi-synthesis | β-cannabispiranol, b-cannabispirol, 5-hydroxy-7-methoxyindan-1-spiro-cyclohexane, 7-hydroxy-5-methoxyindan-1-spiro-cyclohexane, and 5,7-dihydroxyindan-1-spiro-cyclohexane, Cannabispirketal and the glycoside, a-cannabispiranol-4′-O-β-glucopyranose, 3,4′,5-trihydroxy-dihydrostilbene, 4,5-dihydroxy-2,3,7-trimethoxy-9,10-dihydrophenanthrene, 4-hydroxy-2,3,6,7-tetramethoxy-9,10-dihydrophenanthrene and 4,7-dimethoxy-1,2,5-trihydroxyphenanthrene, Rutin, friedelin (friedelan-3-one) and epifriedelanol, Anhydrocannabisativine and cannabisativine, α,α′-dihydro-3′,4,5′-trihydroxy-4′-methoxy-3-isopentenylstilbene, α,α′-dihydro-3,4′,5-trihydroxy-4-methoxy-2,6-diisopentenylstilbene, α,α′-dihydro-3′,4,5′-trihydroxy-4′-methoxy-2′,3-diisopentenylstilbene, α,α′-dihydro-3,4′,5-trihydroxy-4,5′-diisopentenylstilbene, and combretastatin B-2 | [32,33,55,80,81,82,83,84] |
| Benzene | Dried leaves of Japanese cannabis | Chromatographed on a polyamide column followed by silica gel chromatography | IR, 1H NMR, MS, UV. | Cannabispirol and acetyl Cannabispirol | [85] |
| Acetone | C. sativa (CARMA) | Gravity column chromatography on silica gel and purified by crystallization from ether and HPLC | Identified according to its physical and spectroscopic properties and synthesis | Debinoben (1,4-phenanthrenequinone) | [86] |
| Sequential extraction (Hexane, EtOAc, CH2Cl2, EtOH, EtOH/H2O, and H2O | High-potency variety of C. sativa (Mississippi) | VLC, silica gel column chromatography, and RP–HPLC | 1D and 2D NMR, IR analysis | acetoxy-6-geranyl-3-n-pentyl-1,4-benzoquinone, 4,5-dihydroxy-2,3,6-trimethoxy-9,10-dihydrophenanthrene, 4-hydroxy-2,3,6,7-tetramethoxy-9,10-dihydrophenanthrene, 4,7-dimethoxy-1,2,5-trihydroxyphenanthrene, Cannflavin C and β-sitosteryl-3-O-β-d-glucopyranoside-2′-O-palmitate, α-cannabispiranol, Chrysoeriol, 6-prenylapigenin, and Cannflavin A and β-acetyl cannabispiranol | [55] |
| Hexane extract | Leaves of Cannabis sativa | Isolation by normal-phase chromatography followed by C18-HPLC | NMR and ESI–MS analysis | Prenylspirodinone and 7-O-methyl-cannabispirone | [55,87] |
| Dichloro-methane extract | (i) Decarboxy-lated C. sativa hemp (ii). Thai Cannabis sativa leaves (iii) Panamanian variety of cannabis | C18 flash chromatography, followed by silica gel gravity column chromatography and HPLC | HR–ESIMS and NMR (1H, 13C, HSQC, and HMBC) data, X-ray crystallography, and confirmation by hydrogenation | Isocannabispiradienone and a-Cannabispiranol, Cannabispira-dienone, and Cannabidihydro-phenanthrene (Cannithrene1 and Cannithrene2) | [88,89] |
| Methanol/methanolic extract | (i) Branches and leaves of hemp (ii) Pollen grains of Mexican variety of C. sativa (iii) Dried leaves of South African and Indian cannabis sativa (iv) Panamanian variant of C. sativa (v) Leaves and branches of C. sativa | (i) TLC, silica gel column chromatography, normal-phase preparative HPLC, and Sephadex LH-20 column chromatography (ii) Partitioning, silica gel chromatography, Sephadex LH-20 chromatography, semi preparative LC | MS, 1D and 2D NMR, UV experiments, IR, X-ray crystallography and confirmation by total synthesis | Rutin, Quercetin-3-O-α-L-rhamnoside, kaempferol-3-O-α-L-rhamnoside, apigenin-7-O-α-L-rhamnoside, apigenin-7-O-β-D-glucopyranoside, luteolin-7-O-β-D-glucopyranoside, 1,3,6,7-tetrahydroxyl-2-C-β-D-glucopyranosyl xanthone, vitexin, orientin, apigenin-6,8-di-C-β-D-glucopyranoside, vitexin-2″-O-α-L-rhamnoside, orientin-2″-O-β-D-glucopyranoside, quebrachitol, inositol and uracil, kaempferol-3-O-sophoroside (196) and quercetin-3-O-sophoroside, cannabispirone; cannabispirenone, Cannabispiran, Isocannabispiran Canniprene, Cannabistilbene I and Cannabistibene II, and 2,3,5,6-tetramethoxy 9,10-dihydrophenanthrenedione | [90,91,92,93,94] |
| Mixture of hydro-alcoholic and organic solvents | C. sativa inflorescence (Ferimon, Uso-31, Felina 32, and Fedora 17 cultivars) | Metabolic and chemical profiling to identify and quantify compounds of different classes | NMR, GC–MS, UHPLC, and HPLC–PDA | Sugars, organic acids, amino acids, cannabinoids, terpenoids, phenols, tannins, flavonoids (Quercetin, Naringenin, and Naringin) and biogenic amines | [35,37] |
| Diethyl ether | Stem exudate (greenhouse-grown C. sativa) | TLC and acid hydrolysis of the exudate | 1H NMR and GC–MS | Phloroglucinol β-D-glucoside | [95] |
| Isolated Bioactive Compound | Bioactivity/Uses | References |
|---|---|---|
| Tetrahydrocannabinol THC | Antioxidant, anti-pruritic, and anti-inflammatory effects | [29,100] |
| Cannabidiol CBD | Anti-convulsive, anti-inflammatory, immunosuppressive properties, antioxidant, and anti-psychotic effects | [101,102,103] |
| Cannabigerol CBG | Anti-fungal effects, anti-cancer, anti-depressant, mild anti-hypertensive agent, analgesic, and anti-erythemic effects | [29,104] |
| Cannabichromene CBC | Anti-inflammatory and analgesic | [29] |
| Cannabinol CBN | Sedative, anti-convulsant, anti-inflammatory, antibiotic, and anti-MRSA activity | [29] |
| Tetrahydrocannabivarin THCV | Anti-convulsant | [29] |
| Tetrahydrocannabinolic acid THCA | Immunomodulatory, anti-inflammatory, neuroprotective, anti-neoplastic activity, and antiemetic effects | [29,105] |
| Cannabidavarin CBDV | Anti-convulsant (anti-epileptic) properties and anti-emetic properties | [106,107] |
| Cannabidiolic acid CBDA | Anti-emetic effects | [104,108,109,110] |
| β-Myrcene | Anti-inflammatory and analgesic sedative agent | [7,29,111] |
| D-Limonene | Strongly anxiolytic, anti-depressant, antibiotic, and anti-cancer agent | [7,29] |
| β-Ocimene | Anti-convulsant activity, anti-fungal activity, anti-tumor activity, and pest resistance | [112,113] |
| γ-Terpinene | Anti-inflammatory activity, antioxidant, and anti-proliferative activity | [114,115] |
| α-Terpinene | Antioxidant | [29] |
| α-Pinene | Anti-inflammatory, bronchodilator, anti-microbial, and anxiolytic effects | [7,11,29,116] |
| Linalool | Analgesic and anticonvulsant, anxiolytic, anti-depressant, anti-glutamatergic, anti-leishmanial activity, anticancer agent, anti-nociceptive, and anti-depressant effects | [111,117,118,119] |
| α-Phellandrene | Anti-nociceptive, anti-depressant, anti-arthritic and allergic, and anti-hyperalgesic effects | [120,121,122] |
| Terpinolene | Anti-fungal and larvicidal, anti-nociceptive, anti-inflammatory antioxidant, and anti-cancer effects | [123,124] |
| β-Caryophyllene | Cardio-protective, hepato-protective, gastro-protective, neuro-protective, nephro-protective, antioxidant, anti-inflammatory, anti-microbial, anti-pruritic, and immunomodulatory activities | [7,125,126,127] |
| Caryophyllene Oxide | Anti-fungal, insecticidal/anti-feedant, and anti-platelet effects | [29] |
| β-Elemene | Anti-cancer and anti-tumor | [128] |
| Guaiol | Anti-inflammatory, antioxidant, anti-cancer anti-rheumatic, antiseptic, diaphoretic, diuretic, and laxative effects | [29,129] |
| Friedelin | Anti-inflammatory, anti-pyretic, and anti-tuberculosis agent | [130,131] |
| Epifriedelanol | Antioxidant | [132] |
| Cannflavin A and B | Anti-inflammatory, neoplastic, antioxidant, neuroprotective, anti-parasitic, and anti-viral agent | [4,7,29,99] |
| Apigenin | Anxiolytic and estrogenic properties, anti-tumor, antioxidant, anti-inflammatory, anti-osteoporosis, and immune regulation effects | [4,133] |
| Vitexin and Isovitexin | Antioxidant, anti-cancer, anti-inflammatory, anti-diabetic, anti-microbial, anti-viral, anti-hyperalgesic, and neuroprotective effects | [134] |
| Quercetin | Anti-cancer/anti-proliferator, antioxidative/anti-aging, anti-viral, anti-inflammatory, cardio-protective, skin-protective, anti-coagulant, and anti-platelet effects | [135] |
| Luteolin | Neuroprotective effects, anti-inflammatory, and antioxidant effects | [136] |
| Lignans | Antioxidant, anti-viral, anti-diabetic, anti-tumorigenic, and anti-obesity activities | [4] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Odieka, A.E.; Obuzor, G.U.; Oyedeji, O.O.; Gondwe, M.; Hosu, Y.S.; Oyedeji, A.O. The Medicinal Natural Products of Cannabis sativa Linn.: A Review. Molecules 2022, 27, 1689. https://doi.org/10.3390/molecules27051689
Odieka AE, Obuzor GU, Oyedeji OO, Gondwe M, Hosu YS, Oyedeji AO. The Medicinal Natural Products of Cannabis sativa Linn.: A Review. Molecules. 2022; 27(5):1689. https://doi.org/10.3390/molecules27051689
Chicago/Turabian StyleOdieka, Anwuli Endurance, Gloria Ukalina Obuzor, Opeoluwa Oyehan Oyedeji, Mavuto Gondwe, Yiseyon Sunday Hosu, and Adebola Omowunmi Oyedeji. 2022. "The Medicinal Natural Products of Cannabis sativa Linn.: A Review" Molecules 27, no. 5: 1689. https://doi.org/10.3390/molecules27051689
APA StyleOdieka, A. E., Obuzor, G. U., Oyedeji, O. O., Gondwe, M., Hosu, Y. S., & Oyedeji, A. O. (2022). The Medicinal Natural Products of Cannabis sativa Linn.: A Review. Molecules, 27(5), 1689. https://doi.org/10.3390/molecules27051689







