Discriminating between Parallel, Anti-Parallel and Hybrid G-Quadruplexes: Mechanistic Details on Their Binding to Small Molecules
Abstract
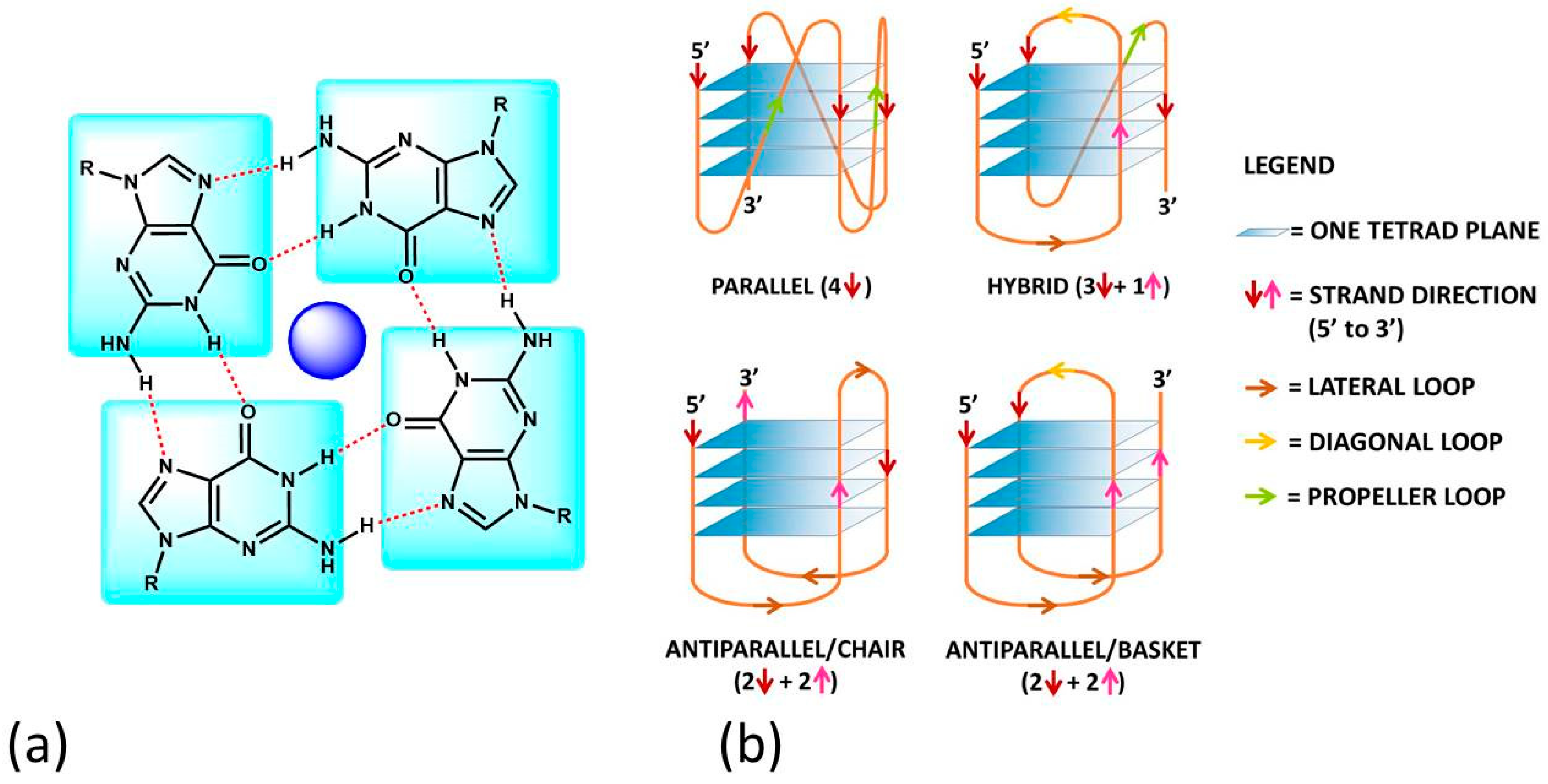
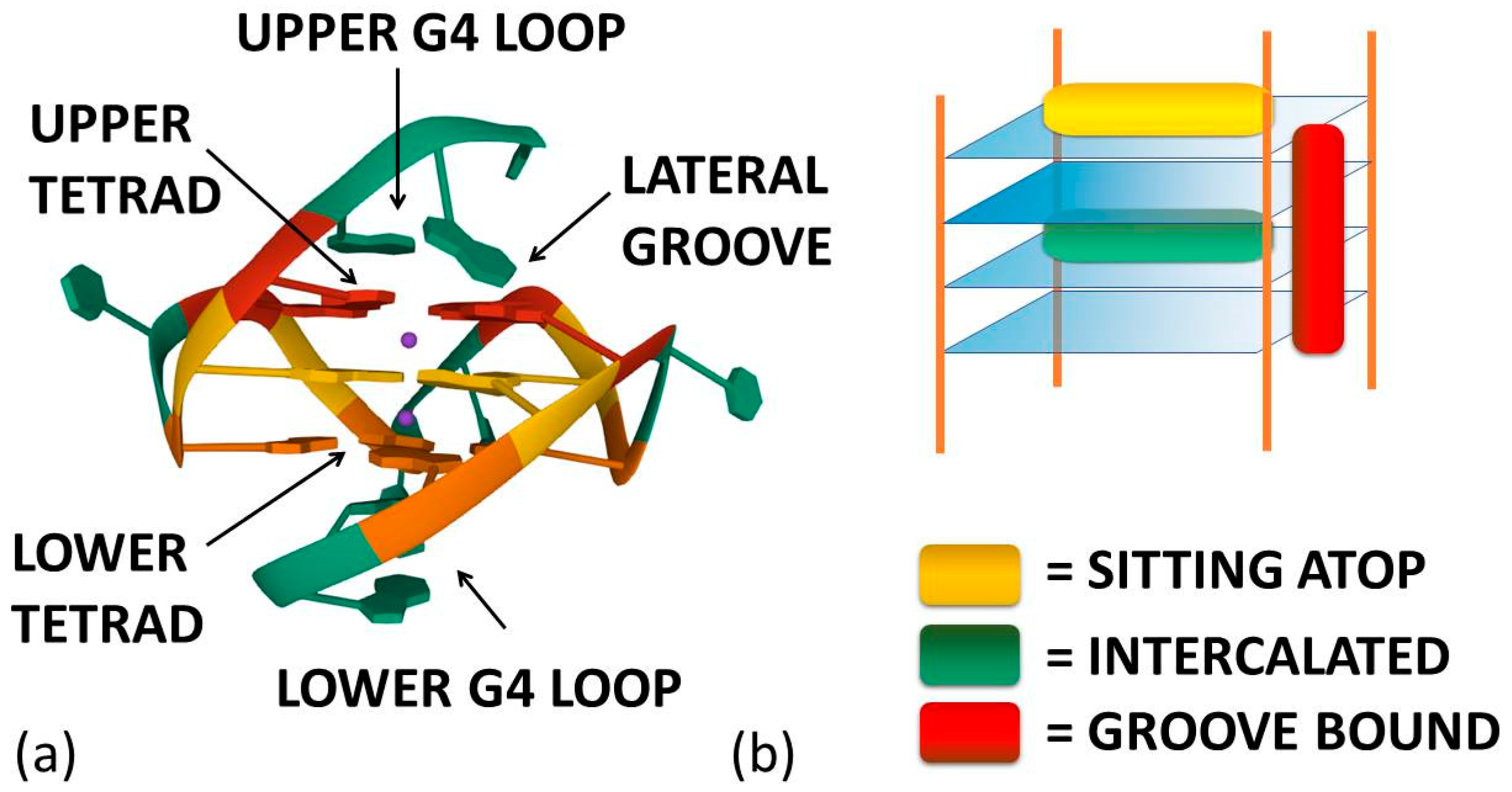
:1. Introduction
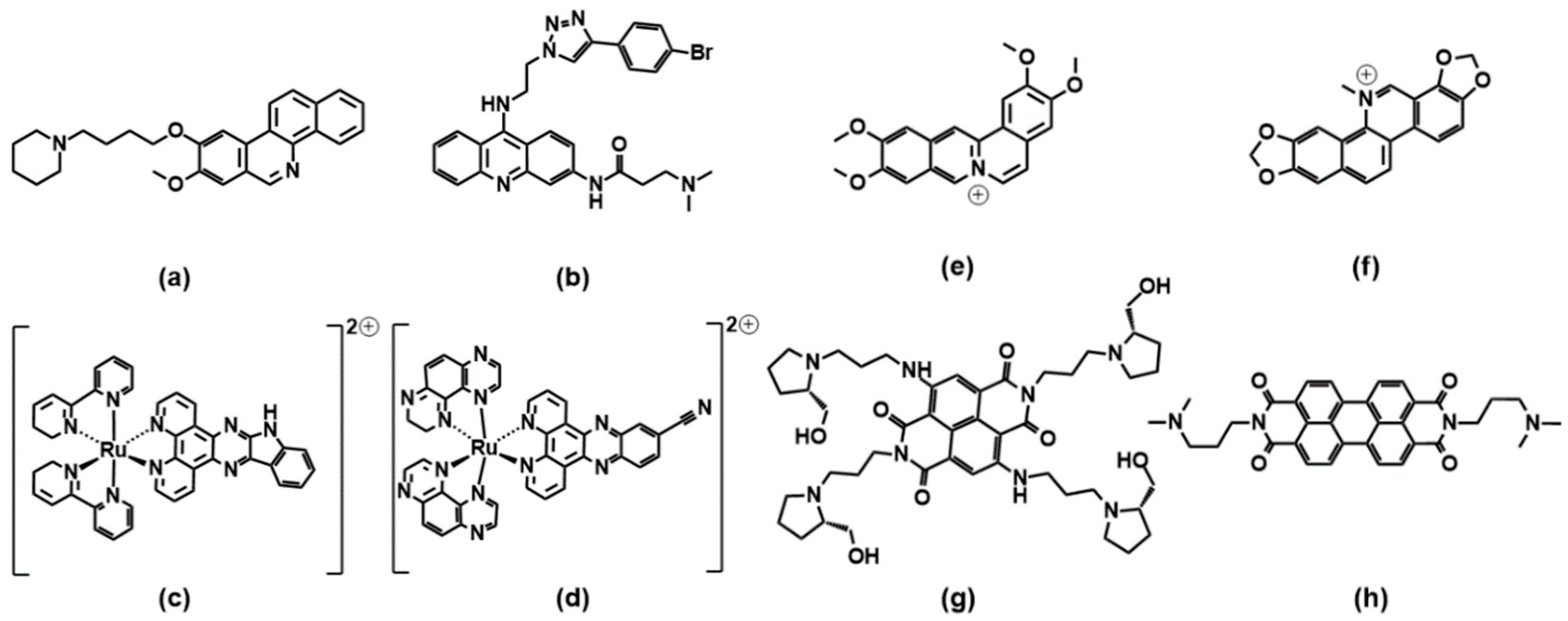
2. Selective Binders
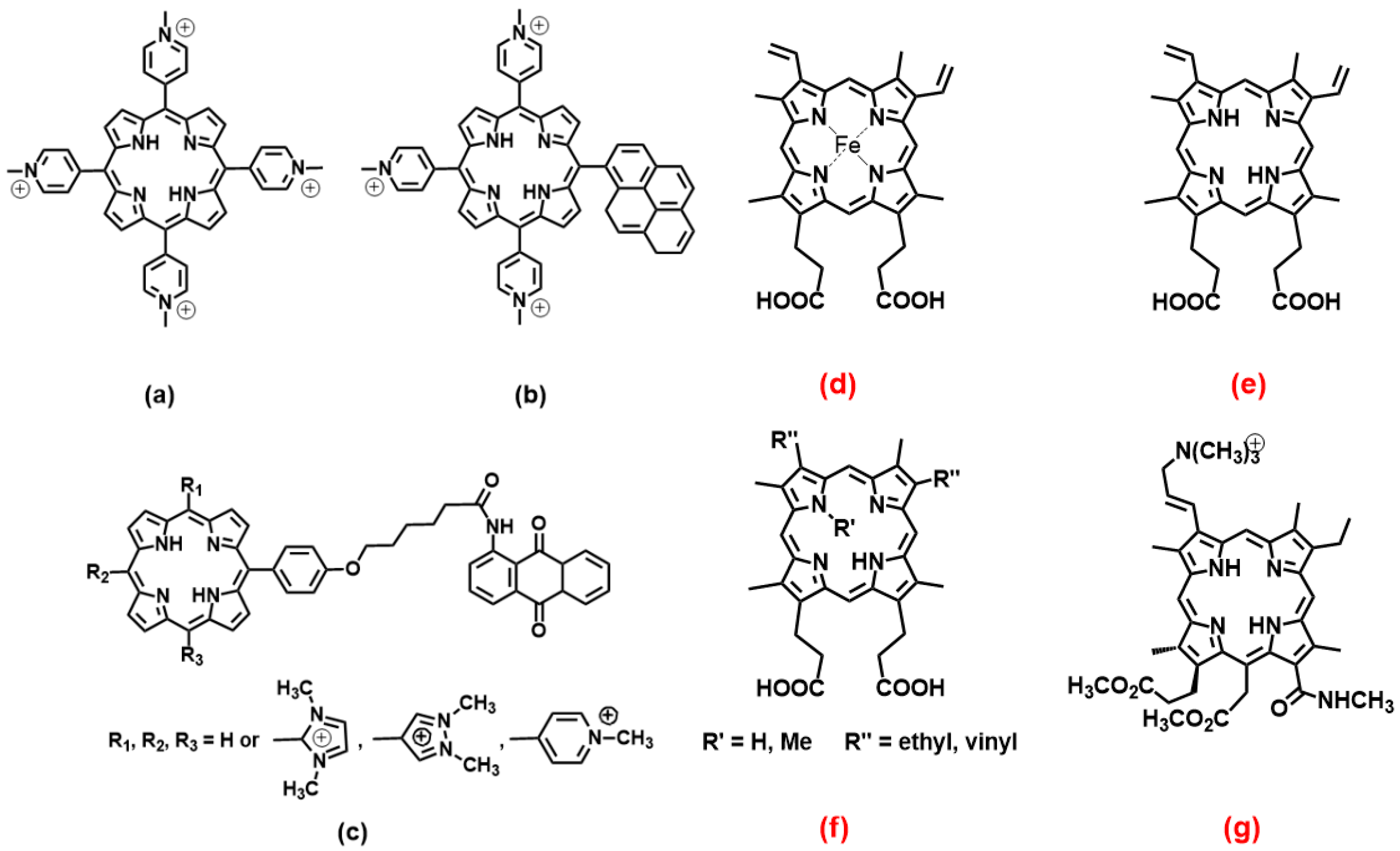
2.1. Porphyrins
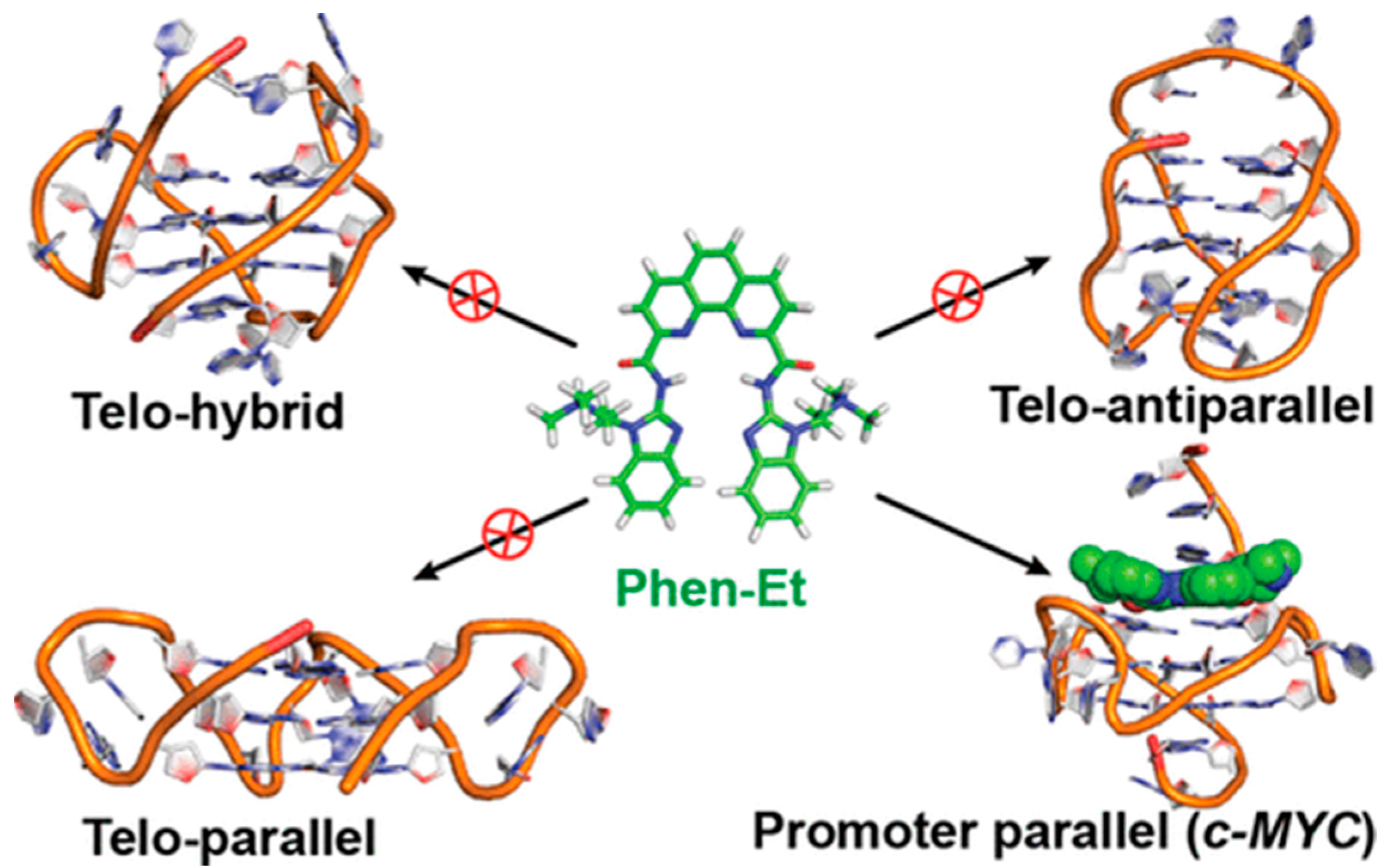
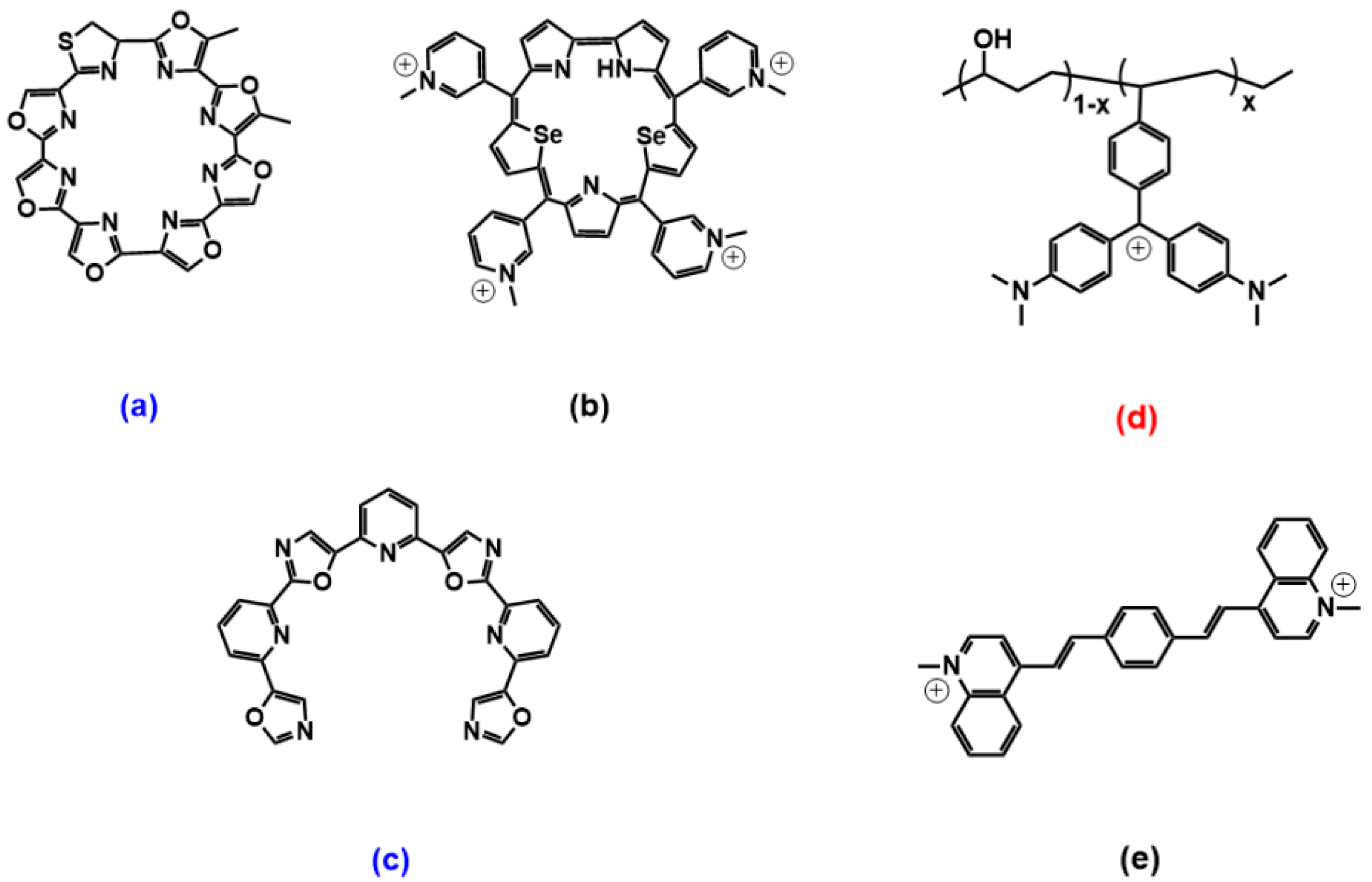
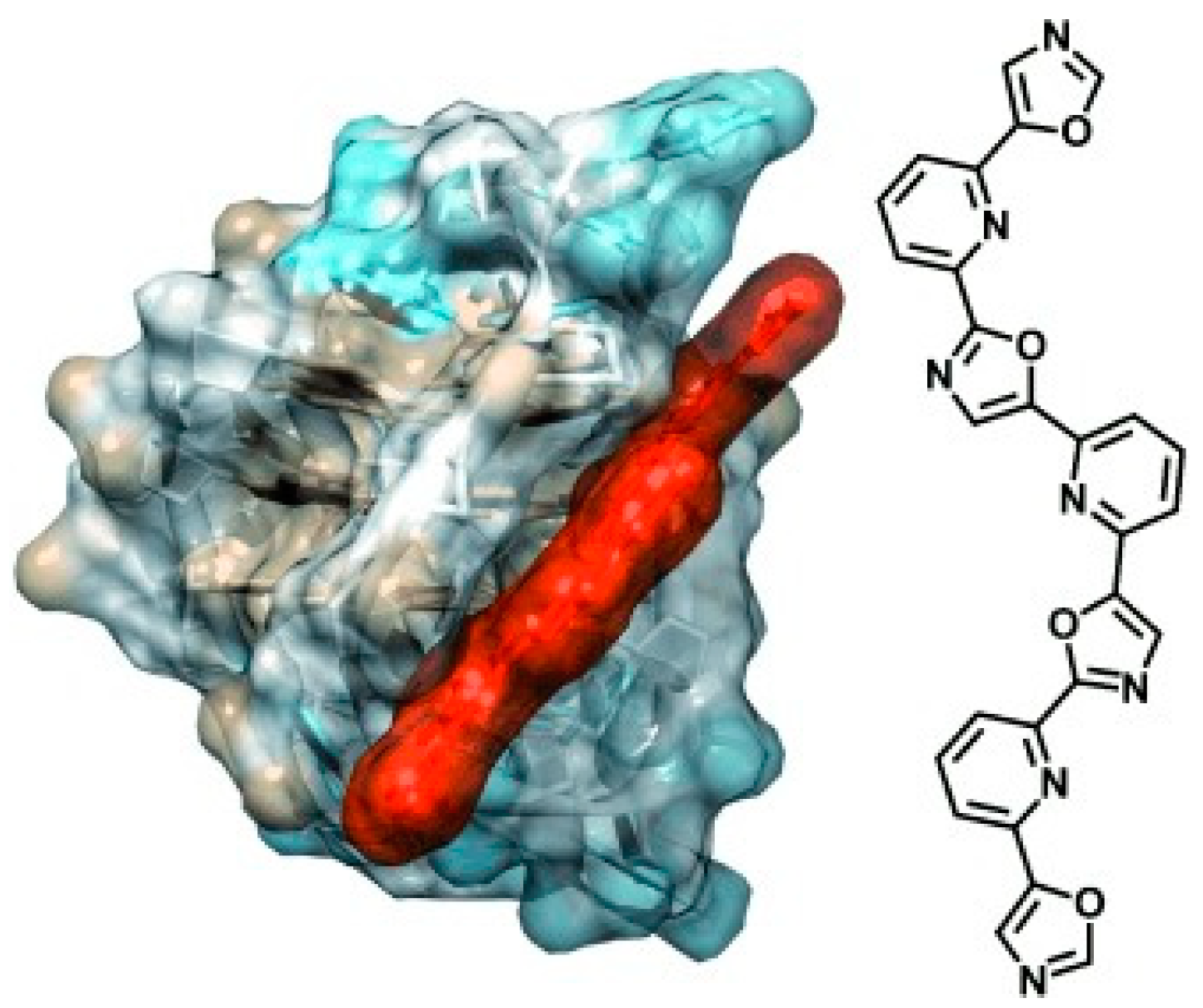
2.2. Phenanthrolines and Their Metal Complexes
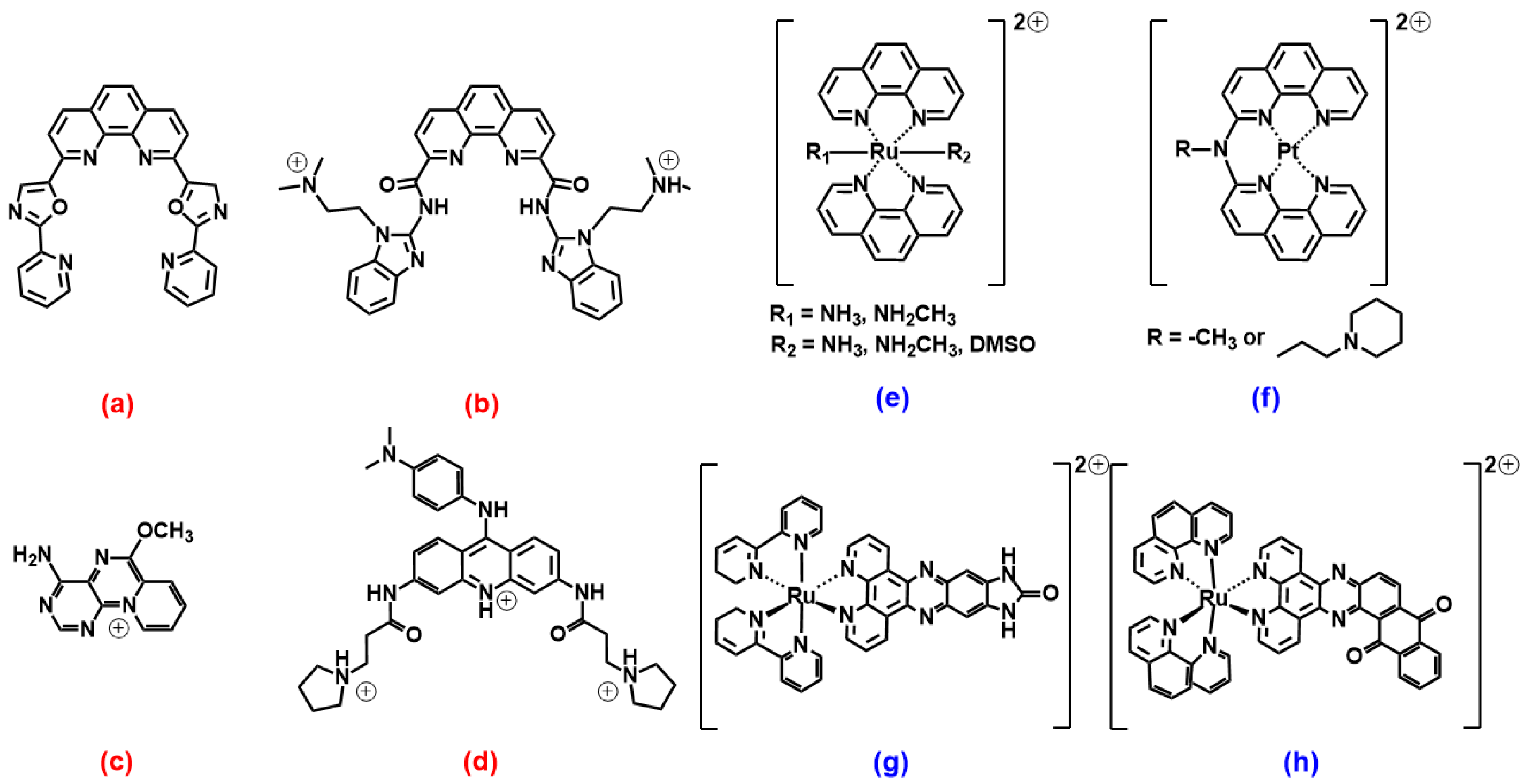
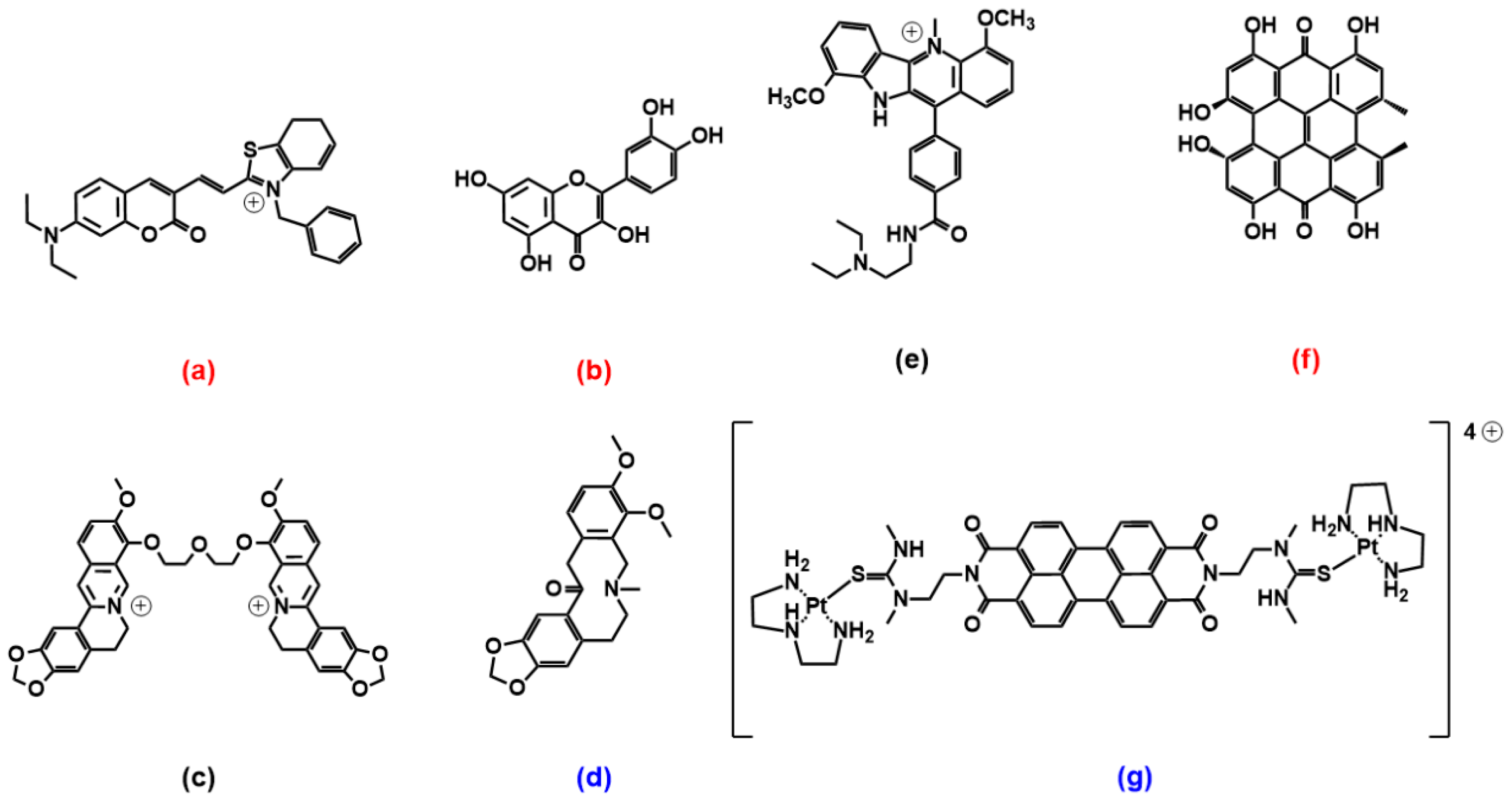
2.3. Coumarin, Quercetin, Berberine and Pericyclic Compounds
2.4. Miscellaneous
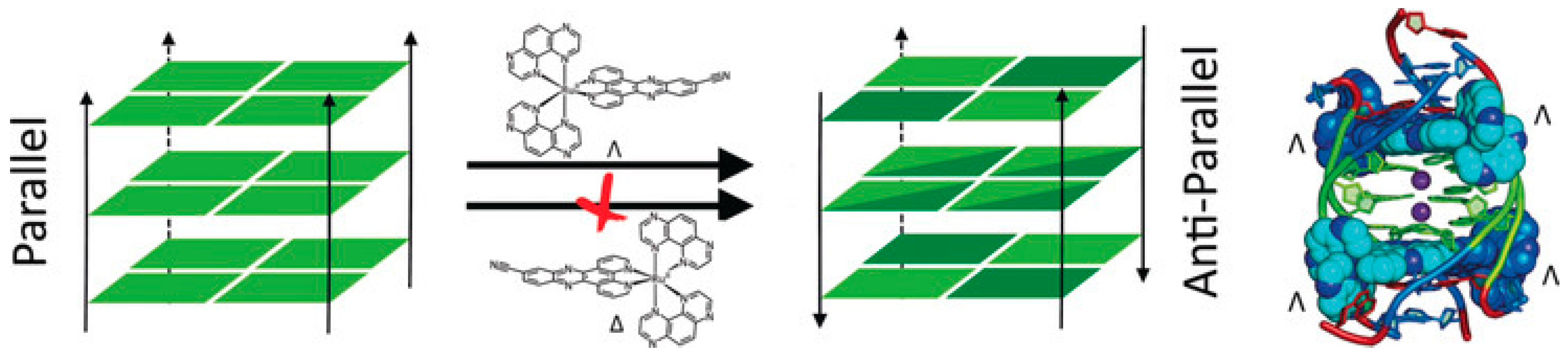
3. Topology Switch Inducers
3.1. Porphyrins
3.2. Phenanthrolines Derivatives and Their Metal Complexes
3.3. Pericyclic Compounds
4. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Burge, S.; Parkinson, G.N.; Hazel, P.; Todd, A.K.; Neidle, S. Quadruplex DNA: Sequence, topology and structure. Nucleic Acids Res. 2006, 34, 5402–5415. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sen, D.; Gilbert, W. Formation of parallel four-stranded complexes by guanine-rich motifs in DNA and its implications for meiosis. Nature 1988, 334, 364–366. [Google Scholar] [CrossRef] [PubMed]
- Kilpatrick, M.W.; Torri, A.; Kang, D.S.; Engler, J.A.; Wells, R.D. Unusual DNA structures in the adenovirus genome. J. Biol. Chem. 1986, 261, 11350–11354. [Google Scholar] [CrossRef]
- Masai, H.; Tanaka, T. G-quadruplex DNA and RNA: Their roles in regulation of DNA replication and other biological functions. Biochem. Biophys. Res. Commun. 2020, 531, 25–38. [Google Scholar] [CrossRef]
- Varshney, D.; Spiegel, J.; Zyner, K.; Tannahill, D.; Balasubramanian, S. The regulation and functions of DNA and RNA G-quadruplexes. Nat. Rev. Mol. Cell Biol. 2020, 21, 459–474. [Google Scholar] [CrossRef]
- Bhattacharyya, D.; Arachchilage, G.M.; Basu, S. Metal Cations in G-Quadruplex Folding and Stability. Front. Chem. 2016, 4, 38. [Google Scholar] [CrossRef] [Green Version]
- Ishiguro, A.; Katayama, A.; Ishihama, A. Different recognition modes of G-quadruplex RNA between two ALS/FTLD-linked proteins TDP-43 and FUS. FEBS Lett. 2021, 595, 310–323. [Google Scholar] [CrossRef]
- Ou, T.-M.; Lu, Y.-J.; Tan, J.-H.; Huang, Z.-S.; Wong, K.-Y.; Gu, L.-Q. G-Quadruplexes: Targets in Anticancer Drug Design. Chem. Med. Chem. 2008, 3, 690–713. [Google Scholar] [CrossRef]
- Waller, Z.A.E.; Howell, L.A.; MacDonald, C.J.; O’Connell, M.A.; Searcey, M. Identification and characterisation of a G-quadruplex forming sequence in the promoter region of nuclear factor (erythroid-derived 2)-like 2 (Nrf2). Biochem. Biophys. Res. Commun. 2014, 447, 128–132. [Google Scholar] [CrossRef]
- Ma, Y.; Iida, K.; Nagasawa, K. Topologies of G-quadruplex: Biological functions and regulation by ligands. Biochem. Biophys. Res. Commun. 2020, 531, 3–17. [Google Scholar] [CrossRef]
- Marchand, A.; Gabelica, V. Folding and misfolding pathways of G-quadruplex DNA. Nucleic Acids Res. 2016, 44, 10999–11012. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chan, M.S.; Leung, H.M.; Wong, S.W.; Lin, Z.; Gao, Q.; Chang, T.J.H.; Lai, K.W.C.; Lo, P.K. Reversible reconfiguration of high-order DNA nanostructures by employing G-quartet toeholds as adhesive units. Nanoscale 2020, 12, 2464–2471. [Google Scholar] [CrossRef] [PubMed]
- Wright, W.E.; Tesmer, V.M.; Huffman, K.E.; Levene, S.D.; Shay, J.W. Normal human chromosomes have long G-rich telomeric overhangs at one end. Genes Dev. 1997, 11, 2801–2809. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hsu, S.-T.D.; Varnai, P.; Bugaut, A.; Reszka, A.P.; Neidle, S.; Balasubramanian, S. A G-Rich Sequence within the c-kit Oncogene Promoter Forms a Parallel G-Quadruplex Having Asymmetric G-Tetrad Dynamics. J. Am. Chem. Soc. 2009, 131, 13399–13409. [Google Scholar] [CrossRef] [Green Version]
- Lim, K.W.; Alberti, P.; Guédin, A.; Lacroix, L.; Riou, J.-F.; Royle, N.J.; Mergny, J.-L.; Phan, A.T. Sequence variant (CTAGGG)n in the human telomere favors a G-quadruplex structure containing a G·C·G·C tetrad. Nucleic Acids Res. 2009, 37, 6239–6248. [Google Scholar] [CrossRef]
- Wang, Z.-F.; Li, M.-H.; Hsu, S.-T.D.; Chang, T.-C. Structural basis of sodium–potassium exchange of a human telomeric DNA quadruplex without topological conversion. Nucleic Acids Res. 2014, 42, 4723–4733. [Google Scholar] [CrossRef]
- Ambrus, A.; Chen, D.; Dai, J.; Bialis, T.; Jones, R.A.; Yang, D. Human telomeric sequence forms a hybrid-type intramolecular G-quadruplex structure with mixed parallel/antiparallel strands in potassium solution. Nucleic Acids Res. 2006, 34, 2723–2735. [Google Scholar] [CrossRef] [Green Version]
- Howard, F.B.; Frazier, J.; Miles, H.T. Stable and metastable forms of poly(G). Biopolymers 1977, 16, 791–809. [Google Scholar] [CrossRef]
- Luu, K.N.; Phan, A.T.; Kuryavyi, V.; Lacroix, L.; Patel, D.J. Structure of the Human Telomere in K+ Solution: An Intramolecular (3 + 1) G-Quadruplex Scaffold. J. Am. Chem. Soc. 2006, 128, 9963–9970. [Google Scholar] [CrossRef] [Green Version]
- Miyoshi, D.; Nakao, A.; Sugimoto, N. Structural transition from antiparallel to parallel G-quadruplex of d(G 4 T 4 G 4) induced by Ca2+. Nucleic Acids Res. 2003, 31, 1156–1163. [Google Scholar] [CrossRef] [Green Version]
- Mergny, J.-L.; Hélène, C. G-quadruplex DNA: A target for drug design. Nat. Med. 1998, 4, 1366–1367. [Google Scholar] [CrossRef] [PubMed]
- Wheelhouse, R.T.; Sun, D.; Han, H.; Han, F.X.; Hurley, L.H. Cationic Porphyrins as Telomerase Inhibitors: the Interaction of Tetra-(N-methyl-4-pyridyl)porphine with Quadruplex DNA. J. Am. Chem. Soc. 1998, 120, 3261–3262. [Google Scholar] [CrossRef]
- Yaku, H.; Fujimoto, T.; Murashima, T.; Miyoshi, D.; Sugimoto, N. Phthalocyanines: A new class of G-quadruplex-ligands with many potential applications. Chem. Commun. 2012, 48, 6203–6216. [Google Scholar] [CrossRef] [PubMed]
- Yurenko, Y.P.; Novotný, J.; Marek, R. Weak Supramolecular Interactions Governing Parallel and Antiparallel DNA Quadruplexes: Insights from Large-Scale Quantum Mechanics Analysis of Experimentally Derived Models. Chem. Eur. J. 2017, 23, 5573–5584. [Google Scholar] [CrossRef]
- Haase, L.; Weisz, K. Locked nucleic acid building blocks as versatile tools for advanced G-quadruplex design. Nucleic Acids Res. 2020, 48, 10555–10566. [Google Scholar] [CrossRef]
- Dai, J.; Carver, M.; Punchihewa, C.; Jones, R.A.; Yang, D. Structure of the Hybrid-2 type intramolecular human telomeric G-quadruplex in K+ solution: Insights into structure polymorphism of the human telomeric sequence. Nucleic Acids Res. 2007, 35, 4927–4940. [Google Scholar] [CrossRef] [Green Version]
- Masiero, S.; Trotta, R.; Pieraccini, S.; De Tito, S.; Perone, R.; Randazzo, A.; Spada, G.P. A non-empirical chromophoric interpretation of CD spectra of DNA G-quadruplex structures. Org. Biomol. Chem. 2010, 8, 2683–2692. [Google Scholar] [CrossRef]
- Poudel, L.; Steinmetz, N.F.; French, R.H.; Parsegian, V.A.; Podgornik, R.; Ching, W.-Y. Implication of the solvent effect, metal ions and topology in the electronic structure and hydrogen bonding of human telomeric G-quadruplex DNA. Phys. Chem. Chem. Phys. 2016, 18, 21573–21585. [Google Scholar] [CrossRef]
- Price, D.A.; Wedamulla, P.; Hill, T.D.; Loth, T.M.; Moran, S.D. The polarization dependence of 2D IR cross-peaks distinguishes parallel-stranded and antiparallel-stranded DNA G-quadruplexes. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2022, 267, 120596. [Google Scholar] [CrossRef]
- Georgiades, S.N.; Abd Karim, N.H.; Suntharalingam, K.; Vilar, R. Interaction of Metal Complexes with G-Quadruplex DNA. Angew. Chem. Int. Ed. 2010, 49, 4020–4034. [Google Scholar] [CrossRef]
- Arola, A.; Vilar, R. Stabilisation of G-Quadruplex DNA by Small Molecules. Curr. Top. Med. Chem. 2008, 8, 1405–1415. [Google Scholar] [CrossRef] [PubMed]
- Busto, N.; Calvo, P.; Santolaya, J.; Leal, J.M.; Guédin, A.; Barone, G.; Torroba, T.; Mergny, J.-L.; García, B. Fishing for G-Quadruplexes in Solution with a Perylene Diimide Derivative Labeled with Biotins. Chem. Eur. J. 2018, 24, 11292–11296. [Google Scholar] [CrossRef] [PubMed]
- Neidle, S. Quadruplex nucleic acids as targets for anticancer therapeutics. Nat. Rev. Chem. 2017, 1, 41. [Google Scholar] [CrossRef]
- Monchaud, D.; Granzhan, A.; Saettel, N.; Guédin, A.; Mergny, J.-L.; Teulade-Fichou, M.-P. “One Ring to Bind Them All”—Part I: The Efficiency of the Macrocyclic Scaffold for G-Quadruplex DNA Recognition. J. Nucleic Acids 2010, 2010, 525862. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pérez-Arnaiz, C.; Busto, N.; Santolaya, J.; Leal, J.M.; Barone, G.; García, B. Kinetic evidence for interaction of TMPyP4 with two different G-quadruplex conformations of human telomeric DNA. Biochim. Biophys. Acta (BBA) Gen. Subj. 2018, 1862, 522–531. [Google Scholar] [CrossRef]
- Cao, Q.; Li, Y.; Freisinger, E.; Qin, P.Z.; Sigel, R.K.O.; Mao, Z.-W. G-quadruplex DNA targeted metal complexes acting as potential anticancer drugs. Inorg. Chem. Front. 2017, 4, 10–32. [Google Scholar] [CrossRef] [Green Version]
- Reed, J.E.; Arnal, A.A.; Neidle, S.; Vilar, R. Stabilization of G-Quadruplex DNA and Inhibition of Telomerase Activity by Square-Planar Nickel(II) Complexes. J. Am. Chem. Soc. 2006, 128, 5992–5993. [Google Scholar] [CrossRef]
- Monchaud, D.; Teulade-Fichou, M.-P. A hitchhiker’s guide to G-quadruplex ligands. Org. Biomol. Chem. 2008, 6, 627–636. [Google Scholar] [CrossRef]
- Rajput, C.; Rutkaite, R.; Swanson, L.; Haq, I.; Thomas, J.A. Dinuclear Monointercalating RuII Complexes That Display High Affinity Binding to Duplex and Quadruplex DNA. Chem. Eur. J. 2006, 12, 4611–4619. [Google Scholar] [CrossRef]
- Han, H.; Langley, D.R.; Rangan, A.; Hurley, L.H. Selective Interactions of Cationic Porphyrins with G-Quadruplex Structures. J. Am. Chem. Soc. 2001, 123, 8902–8913. [Google Scholar] [CrossRef]
- Faudale, M.; Cogoi, S.; Xodo, L.E. Photoactivated cationic alkyl-substituted porphyrin binding to g4-RNA in the 5′-UTR of KRAS oncogene represses translation. Chem. Commun. 2012, 48, 874–876. [Google Scholar] [CrossRef] [PubMed]
- Wei, C.; Jia, G.; Zhou, J.; Han, G.; Li, C. Evidence for the binding mode of porphyrins to G-quadruplex DNA. Phys. Chem. Chem. Phys. 2009, 11, 4025–4032. [Google Scholar] [CrossRef] [PubMed]
- Seenisamy, J.; Rezler, E.M.; Powell, T.J.; Tye, D.; Gokhale, V.; Joshi, C.S.; Siddiqui-Jain, A.; Hurley, L.H. The Dynamic Character of the G-Quadruplex Element in the c-MYC Promoter and Modification by TMPyP4. J. Am. Chem. Soc. 2004, 126, 8702–8709. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.-J.; Wang, X.-F.; Wang, P.; Ai, X.-C.; Zhang, J.-P. Spectroscopic study on the binding of a cationic porphyrin to DNA G-quadruplex under different K+ concentrations. Photochem. Photobiol. Sci. 2008, 7, 948–955. [Google Scholar] [CrossRef] [PubMed]
- Ali, A.; Bansal, M.; Bhattacharya, S. Ligand 5,10,15,20-Tetra(N-methyl-4-pyridyl)porphine (TMPyP4) Prefers the Parallel Propeller-Type Human Telomeric G-Quadruplex DNA over Its Other Polymorphs. J. Phys. Chem. B 2015, 119, 5–14. [Google Scholar] [CrossRef]
- Arora, A.; Maiti, S. Effect of Loop Orientation on Quadruplex−TMPyP4 Interaction. J. Phys. Chem. B 2008, 112, 8151–8159. [Google Scholar] [CrossRef]
- Lago, S.; Tosoni, E.; Nadai, M.; Palumbo, M.; Richter, S.N. The cellular protein nucleolin preferentially binds long-looped G-quadruplex nucleic acids. Biochim. Biophys. Acta (BBA)—Gen. Subj. 2017, 1861, 1371–1381. [Google Scholar] [CrossRef]
- Cho, H.Y.; Lee, Y.-A.; Oh, Y.S.; Lee, G.J.; Jang, Y.J.; Kim, S.K. Binding mode of a cationic porphyrin to parallel and antiparallel thrombin binding aptamer G-quadruplex. J. Biomol. Struct. Dyn. 2020, 38, 2686–2692. [Google Scholar] [CrossRef]
- Han, J.H.; Cho, H.Y.; Kim, D.Y.; Jang, Y.J.; Lee, Y.-A.; Kim, S.K. Binding properties of pyrene-porphyrin dyad to G-quadruplexes in the presence of K+ and Na+ ion and their effect on stability. J. Mol. Struct. 2020, 1215, 128264. [Google Scholar] [CrossRef]
- Arba, M.; Kartasasmita, R.E.; Tjahjono, D.H. Molecular docking and dynamics simulations on the interaction of cationic porphyrin–anthraquinone hybrids with DNA G-quadruplexes. J. Biomol. Struct. Dyn. 2016, 34, 427–438. [Google Scholar] [CrossRef]
- Okamoto, C.; Momotake, A.; Yamamoto, Y. Structural and functional characterization of complexes between heme and dimeric parallel G-quadruplex DNAs. J. Inorg. Biochem. 2021, 216, 111336. [Google Scholar] [CrossRef] [PubMed]
- Li, T.; Wang, E.; Dong, S. Parallel G-Quadruplex-Specific Fluorescent Probe for Monitoring DNA Structural Changes and Label-Free Detection of Potassium Ion. Anal. Chem. 2010, 82, 7576–7580. [Google Scholar] [CrossRef] [PubMed]
- Nicoludis, J.M.; Barrett, S.P.; Mergny, J.-L.; Yatsunyk, L.A. Interaction of human telomeric DNA with N- methyl mesoporphyrin IX. Nucleic Acids Res. 2012, 40, 5432–5447. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Caterino, M.; D’Aria, F.; Kustov, A.V.; Belykh, D.V.; Khudyaeva, I.S.; Starseva, O.M.; Berezin, D.B.; Pylina, Y.I.; Usacheva, T.; Amato, J.; et al. Selective binding of a bioactive porphyrin-based photosensitizer to the G-quadruplex from the KRAS oncogene promoter. Int. J. Biol. Macromol. 2020, 145, 244–251. [Google Scholar] [CrossRef] [PubMed]
- Medeiros-Silva, J.; Guédin, A.; Salgado, G.F.; Mergny, J.-L.; Queiroz, J.A.; Cabrita, E.J.; Cruz, C. Phenanthroline-bis-oxazole ligands for binding and stabilization of G-quadruplexes. Biochim. Biophys. Acta (BBA)—Gen. Subj. 2017, 1861, 1281–1292. [Google Scholar] [CrossRef] [PubMed]
- Hamon, F.; Largy, E.; Guédin-Beaurepaire, A.; Rouchon-Dagois, M.; Sidibe, A.; Monchaud, D.; Mergny, J.-L.; Riou, J.-F.; Nguyen, C.-H.; Teulade-Fichou, M.-P. An Acyclic Oligoheteroaryle That Discriminates Strongly between Diverse G-Quadruplex Topologies. Angew. Chem. Int. Ed. 2011, 50, 8745–8749. [Google Scholar] [CrossRef]
- Dhamodharan, V.; Harikrishna, S.; Bhasikuttan, A.C.; Pradeepkumar, P.I. Topology Specific Stabilization of Promoter over Telomeric G-Quadruplex DNAs by Bisbenzimidazole Carboxamide Derivatives. ACS Chem. Biol. 2015, 10, 821–833. [Google Scholar] [CrossRef]
- Nowak-Karnowska, J.; Głuszyńska, A.; Kosman, J.; Neunert, G.; Dembska, A. Interaction of 9-Methoxyluminarine with Different G-Quadruplex Topologies: Fluorescence and Circular Dichroism Studies. Int. J. Mol. Sci. 2021, 22, 10399. [Google Scholar] [CrossRef]
- Machireddy, B.; Sullivan, H.-J.; Wu, C. Binding of BRACO19 to a Telomeric G-Quadruplex DNA Probed by All-Atom Molecular Dynamics Simulations with Explicit Solvent. Molecules 2019, 24, 1010. [Google Scholar] [CrossRef] [Green Version]
- Long, W.; Zheng, B.-X.; Li, Y.; Huang, X.-H.; Lin, D.-M.; Chen, C.-C.; Hou, J.-Q.; Ou, T.-M.; Wong, W.-L.; Zhang, K.; et al. Rational design of small-molecules to recognize G-quadruplexes of c-MYC promoter and telomere and the evaluation of their In Vivo antitumor activity against breast cancer. Nucleic Acids Res. 2022, 50, 1829–1848. [Google Scholar] [CrossRef]
- Machireddy, B.; Kalra, G.; Jonnalagadda, S.; Ramanujachary, K.; Wu, C. Probing the Binding Pathway of BRACO19 to a Parallel-Stranded Human Telomeric G-Quadruplex Using Molecular Dynamics Binding Simulation with AMBER DNA OL15 and Ligand GAFF2 Force Fields. J. Chem. Inf. Modeling 2017, 57, 2846–2864. [Google Scholar] [CrossRef] [PubMed]
- Campbell, N.H.; Parkinson, G.N.; Reszka, A.P.; Neidle, S. Structural Basis of DNA Quadruplex Recognition by an Acridine Drug. J. Am. Chem. Soc. 2008, 130, 6722–6724. [Google Scholar] [CrossRef]
- He, L.; Chen, X.; Meng, Z.; Wang, J.; Tian, K.; Li, T.; Shao, F. Octahedral ruthenium complexes selectively stabilize G-quadruplexes. Chem. Commun. 2016, 52, 8095–8098. [Google Scholar] [CrossRef] [PubMed]
- He, L.; Meng, Z.; Xie, Y.-Q.; Chen, X.; Li, T.; Shao, F. Aza-bridged bisphenanthrolinyl Pt(II) complexes: Efficient stabilization and topological selectivity on telomeric G-quadruplexes. J. Inorg. Biochem. 2017, 166, 135–140. [Google Scholar] [CrossRef] [PubMed]
- Shi, S.; Xu, J.-H.; Gao, X.; Huang, H.-L.; Yao, T.-M. Binding Behaviors for Different Types of DNA G-Quadruplexes: Enantiomers of [Ru(bpy)2(L)]2+ (L = dppz, dppz-idzo). Chem. Eur. J. 2015, 21, 11435–11445. [Google Scholar] [CrossRef] [PubMed]
- McQuaid, K.T.; Takahashi, S.; Baumgaertner, L.; Cardin, D.J.; Paterson, N.G.; Hall, J.P.; Sugimoto, N.; Cardin, C.J. Ruthenium Polypyridyl Complex Bound to a Unimolecular Chair-Form G-Quadruplex. J. Am. Chem. Soc. 2022, 144, 5956–5964. [Google Scholar] [CrossRef] [PubMed]
- Deore, P.S.; Manderville, R.A. Ratiometric fluorescent sensing of the parallel G-quadruplex produced by PS2.M: Implications for K+ detection. Analyst 2020, 145, 1288–1293. [Google Scholar] [CrossRef] [PubMed]
- Tyagi, S.; Saxena, S.; Srivastava, P.; Sharma, T.; Kundu, N.; Kaur, S.; Shankaraswamy, J. Screening the binding potential of quercetin with parallel, antiparallel and mixed G-quadruplexes of human telomere and cancer protooncogenes using molecular docking approach. SN Appl. Sci. 2020, 2, 490. [Google Scholar] [CrossRef] [Green Version]
- Li, Z.-Q.; Liao, T.-C.; Dong, C.; Yang, J.-W.; Chen, X.-J.; Liu, L.; Luo, Y.; Liang, Y.-Y.; Chen, W.-H.; Zhou, C.-Q. Specifically targeting mixed-type dimeric G-quadruplexes using berberine dimers. Org. Biomol. Chem. 2017, 15, 10221–10229. [Google Scholar] [CrossRef]
- Mandal, P.; Sahoo, D.; Saha, S.; Chowdhury, J. Sensing of Different Human Telomeric G-Quadruplex DNA Topologies by Natural Alkaloid Allocryptopine Using Spectroscopic Techniques. J. Phys. Chem. B 2018, 122, 10279–10290. [Google Scholar] [CrossRef]
- Funke, A.; Dickerhoff, J.; Weisz, K. Towards the Development of Structure-Selective G-Quadruplex-Binding Indolo[3,2-b]quinolines. Chemistry 2016, 22, 3170–3181. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Jin, B.; Zheng, W.; Zhang, N.; Liu, X.; Bing, T.; Wei, Y.; Wang, F.; Shangguan, D. Interaction of hypericin with guanine-rich DNA: Preferential binding to parallel G-Quadruplexes. Dye. Pigment. 2016, 132, 405–411. [Google Scholar] [CrossRef]
- Rao, L.; Dworkin, J.D.; Nell, W.E.; Bierbach, U. Interactions of a Platinum-Modified Perylene Derivative with the Human Telomeric G-Quadruplex. J. Phys. Chem. B 2011, 115, 13701–13712. [Google Scholar] [CrossRef] [Green Version]
- Macii, F.; Cupellini, L.; Stifano, M.; Santolaya, J.; Pérez-Arnaiz, C.; Pucci, A.; Barone, G.; García, B.; Busto, N.; Biver, T. Combined spectroscopic and theoretical analysis of the binding of a water-soluble perylene diimide to DNA/RNA polynucleotides and G-quadruplexes. Spectrochim. Acta Part. A Mol. Biomol. Spectrosc. 2021, 260, 119914. [Google Scholar] [CrossRef] [PubMed]
- Buchholz, I.; Karg, B.; Dickerhoff, J.; Sievers-Engler, A.; Lämmerhofer, M.; Weisz, K. Selective Targeting of G-Quadruplex Structures by a Benzothiazole-Based Binding Motif. Chem. Eur. J. 2017, 23, 5814–5823. [Google Scholar] [CrossRef]
- Biswas, A.; Singh, S.B.; Todankar, C.S.; Sudhakar, S.; Pany, S.P.P.; Pradeepkumar, P.I. Stabilization and fluorescence light-up of G-quadruplex nucleic acids using indolyl-quinolinium based probes. Phys. Chem. Chem. Phys. 2022, 24, 6238–6255. [Google Scholar] [CrossRef]
- Amato, J.; Pagano, A.; Cosconati, S.; Amendola, G.; Fotticchia, I.; Iaccarino, N.; Marinello, J.; De Magis, A.; Capranico, G.; Novellino, E.; et al. Discovery of the first dual G-triplex/G-quadruplex stabilizing compound: A new opportunity in the targeting of G-rich DNA structures? Biochim. Biophys. Acta (BBA)—Gen. Subj. 2017, 1861, 1271–1280. [Google Scholar] [CrossRef]
- Nikan, M.; Di Antonio, M.; Abecassis, K.; McLuckie, K.; Balasubramanian, S. An Acetylene-Bridged 6,8-Purine Dimer as a Fluorescent Switch-On Probe for Parallel G-Quadruplexes. Angew. Chem. Int. Ed. 2013, 52, 1428–1431. [Google Scholar] [CrossRef] [Green Version]
- Rezler, E.M.; Seenisamy, J.; Bashyam, S.; Kim, M.-Y.; White, E.; Wilson, W.D.; Hurley, L.H. Telomestatin and Diseleno Sapphyrin Bind Selectively to Two Different Forms of the Human Telomeric G-Quadruplex Structure. J. Am. Chem. Soc. 2005, 127, 9439–9447. [Google Scholar] [CrossRef]
- Seenisamy, J.; Bashyam, S.; Gokhale, V.; Vankayalapati, H.; Sun, D.; Siddiqui-Jain, A.; Streiner, N.; Shin-ya, K.; White, E.; Wilson, W.D.; et al. Design and Synthesis of an Expanded Porphyrin That Has Selectivity for the c-MYC G-Quadruplex Structure. J. Am. Chem. Soc. 2005, 127, 2944–2959. [Google Scholar] [CrossRef]
- Benito, S.; Ferrer, A.; Benabou, S.; Aviñó, A.; Eritja, R.; Gargallo, R. Evaluation of the effect of polymorphism on G-quadruplex-ligand interaction by means of spectroscopic and chromatographic techniques. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2018, 196, 185–195. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Uda, R.M.; Nishimoto, N.; Matsui, T.; Takagi, S. Photoinduced binding of malachite green copolymer to parallel G-quadruplex DNA. Soft Matter 2019, 15, 4454–4459. [Google Scholar] [CrossRef] [PubMed]
- Czerwinska, I.; Juskowiak, B. Photoisomerizable arylstilbazolium ligands recognize parallel and antiparallel structures of G-quadruplexes. Int. J. Biol. Macromol. 2012, 51, 576–582. [Google Scholar] [CrossRef] [PubMed]
- Mohanty, J.; Barooah, N.; Dhamodharan, V.; Harikrishna, S.; Pradeepkumar, P.I.; Bhasikuttan, A.C. Thioflavin T as an Efficient Inducer and Selective Fluorescent Sensor for the Human Telomeric G-Quadruplex DNA. J. Am. Chem. Soc. 2013, 135, 367–376. [Google Scholar] [CrossRef]
- Li, Y.; Xu, S.; Wu, X.; Xu, Q.; Zhao, Y.; Lou, X.; Yang, X. Thioflavin T as a fluorescence light-up probe for both parallel and antiparallel G-quadruplexes of 29-mer thrombin binding aptamer. Anal. Bioanal. Chem. 2016, 408, 8025–8036. [Google Scholar] [CrossRef]
- Han, H.; Cliff, C.L.; Hurley, L.H. Accelerated Assembly of G-Quadruplex Structures by a Small Molecule. Biochemistry 1999, 38, 6981–6986. [Google Scholar] [CrossRef]
- Shi, S.; Geng, X.; Zhao, J.; Yao, T.; Wang, C.; Yang, D.; Zheng, L.; Ji, L. Interaction of [Ru(bpy)2(dppz)]2+ with human telomeric DNA: Preferential binding to G-quadruplexes over i-motif. Biochimie 2010, 92, 370–377. [Google Scholar] [CrossRef]
- Ou, Z.; Wang, Y.; Gao, Y.; Wang, X.; Qian, Y.; Li, Y.; Wang, X. Targeting human telomeric and c-myc G-quadruplexes with alkynylplatinum(II) terpyridine complexes under molecular crowding conditions. J. Inorg. Biochem. 2017, 166, 126–134. [Google Scholar] [CrossRef]
- Martino, L.; Pagano, B.; Fotticchia, I.; Neidle, S.; Giancola, C. Shedding Light on the Interaction between TMPyP4 and Human Telomeric Quadruplexes. J. Phys. Chem. B 2009, 113, 14779–14786. [Google Scholar] [CrossRef]
- Rodriguez, R.; Pantoş, G.D.; Gonçalves, D.P.N.; Sanders, J.K.M.; Balasubramanian, S. Ligand-driven G-quadruplex conformational switching by using an unusual mode of interaction. Angew. Chem. Int. Ed. Engl. 2007, 46, 5405–5407. [Google Scholar] [CrossRef]
- Deore, P.S.; Gray, M.D.; Chung, A.J.; Manderville, R.A. Ligand-Induced G-Quadruplex Polymorphism: A DNA Nanodevice for Label-Free Aptasensor Platforms. J. Am. Chem. Soc. 2019, 141, 14288–14297. [Google Scholar] [CrossRef] [PubMed]
- Brooks, T.A.; Hurley, L.H. The role of supercoiling in transcriptional control of MYC and its importance in molecular therapeutics. Nat. Rev. Cancer 2009, 9, 849–861. [Google Scholar] [CrossRef] [PubMed]
- Kong, D.-M.; Yang, W.; Wu, J.; Li, C.-X.; Shen, H.-X. Structure–function study of peroxidase-like G-quadruplex-hemin complexes. Analyst 2010, 135, 321–326. [Google Scholar] [CrossRef] [PubMed]
- Rajczak, E.; Juskowiak, B. Conformational rearrangements of G-quadruplex topology promoted by Cu(II) 12-MCCu(II)PyrAcHA-4 metallacrown. Int. J. Biol. Macromol. 2020, 156, 1258–1269. [Google Scholar] [CrossRef]
- Chen, H.; Long, H.; Cui, X.; Zhou, J.; Xu, M.; Yuan, G. Exploring the Formation and Recognition of an Important G-Quadruplex in a HIF1α Promoter and Its Transcriptional Inhibition by a Benzo[c]phenanthridine Derivative. J. Am. Chem. Soc. 2014, 136, 2583–2591. [Google Scholar] [CrossRef]
- Yu, H.-J.; Zhao, Y.; Mo, W.-J.; Hao, Z.-F.; Yu, L. Ru-indoloquinoline complex as a selective and effective human telomeric G-quadruplex binder. Spectrochim. Acta Part. A Mol. Biomol. Spectrosc. 2014, 132, 84–90. [Google Scholar] [CrossRef]
- Yu, H.-J.; Yu, L.; Hao, Z.-F.; Zhao, Y. Interactions of ruthenium complexes containing indoloquinoline moiety with human telomeric G-quadruplex DNA. Spectrochim. Acta Part. A Mol. Biomol. Spectrosc. 2014, 124, 187–193. [Google Scholar] [CrossRef]
- McQuaid, K.; Abell, H.; Gurung, S.P.; Allan, D.R.; Winter, G.; Sorensen, T.; Cardin, D.J.; Brazier, J.A.; Cardin, C.J.; Hall, J.P. Structural Studies Reveal Enantiospecific Recognition of a DNA G-Quadruplex by a Ruthenium Polypyridyl Complex. Angew. Chem. Int. Ed. 2019, 58, 9881–9885. [Google Scholar] [CrossRef]
- Padmapriya, K.; Barthwal, R. NMR based structural studies decipher stacking of the alkaloid coralyne to terminal guanines at two different sites in parallel G-quadruplex DNA, [d(TTGGGGT)]4 and [d(TTAGGGT)]4. Biochim. Biophys. Acta (BBA)—Gen. Subj. 2017, 1861, 37–48. [Google Scholar] [CrossRef]
- Pradhan, S.K.; Dasgupta, D.; Basu, G. Human telomere d[(TTAGGG)4] undergoes a conformational transition to the Na+-form upon binding with sanguinarine in presence of K+. Biochem. Biophys. Res. Commun. 2011, 404, 139–142. [Google Scholar] [CrossRef]
- Hao, X.; Wang, C.; Wang, Y.; Li, C.; Hou, J.; Zhang, F.; Kang, C.; Gao, L. Topological conversion of human telomeric G-quadruplexes from hybrid to parallel form induced by naphthalene diimide ligands. Int. J. Biol. Macromol. 2021, 167, 1048–1058. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Yuan, G. Specific Recognition of Human Telomeric G-Quadruplex DNA with Small Molecules and the Conformational Analysis by ESI Mass Spectrometry and Circular Dichroism Spectropolarimetry. Chemistry 2007, 13, 5018–5023. [Google Scholar] [CrossRef] [PubMed]
- Xiao, C.-D.; Shibata, T.; Yamamoto, Y.; Xu, Y. An intramolecular antiparallel G-quadruplex formed by human telomere RNA. Chem. Commun. 2018, 54, 3944–3946. [Google Scholar] [CrossRef] [PubMed]
- Zhang, A.Y.Q.; Bugaut, A.; Balasubramanian, S. A Sequence-Independent Analysis of the Loop Length Dependence of Intramolecular RNA G-Quadruplex Stability and Topology. Biochemistry 2011, 50, 7251–7258. [Google Scholar] [CrossRef] [Green Version]
- Bai, L.-P.; Liu, J.; Han, L.; Ho, H.-M.; Wang, R.; Jiang, Z.-H. Mass spectrometric studies on effects of counter ions of TMPyP4 on binding to human telomeric DNA and RNA G-quadruplexes. Anal. Bioanal. Chem. 2014, 406, 5455–5463. [Google Scholar] [CrossRef] [Green Version]
- Bai, L.-P.; Hagihara, M.; Nakatani, K.; Jiang, Z.-H. Recognition of Chelerythrine to Human Telomeric DNA and RNA G-quadruplexes. Sci. Rep. 2014, 4, 6767. [Google Scholar] [CrossRef] [Green Version]
- Binacchi, F.; Guarra, F.; Cirri, D.; Marzo, T.; Pratesi, A.; Messori, L.; Gabbiani, C.; Biver, T. On the Different Mode of Action of Au(I)/Ag(I)-NHC Bis-Anthracenyl Complexes Towards Selected Target Biomolecules. Molecules 2020, 25, 5446. [Google Scholar] [CrossRef]
- Vianney, Y.M.; Weisz, K. Indoloquinoline Ligands Favor Intercalation at Quadruplex-Duplex Interfaces. Chem. Eur. J. 2022, 28, e202103718. [Google Scholar] [CrossRef]
- Funke, A.; Weisz, K. Comprehensive Thermodynamic Profiling for the Binding of a G-Quadruplex Selective Indoloquinoline. J. Phys. Chem. B 2017, 121, 5735–5743. [Google Scholar] [CrossRef]
- Beccia, M.R.; Biver, T.; Pardini, A.; Spinelli, J.; Secco, F.; Venturini, M.; Busto Vázquez, N.; Lopez Cornejo, M.P.; Martin Herrera, V.I.; Prado Gotor, R. The Fluorophore 4′,6-Diamidino-2-phenylindole (DAPI) Induces DNA Folding in Long Double-Stranded DNA. Chem. Asian J. 2012, 7, 1803–1810. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Biver, T. Discriminating between Parallel, Anti-Parallel and Hybrid G-Quadruplexes: Mechanistic Details on Their Binding to Small Molecules. Molecules 2022, 27, 4165. https://doi.org/10.3390/molecules27134165
Biver T. Discriminating between Parallel, Anti-Parallel and Hybrid G-Quadruplexes: Mechanistic Details on Their Binding to Small Molecules. Molecules. 2022; 27(13):4165. https://doi.org/10.3390/molecules27134165
Chicago/Turabian StyleBiver, Tarita. 2022. "Discriminating between Parallel, Anti-Parallel and Hybrid G-Quadruplexes: Mechanistic Details on Their Binding to Small Molecules" Molecules 27, no. 13: 4165. https://doi.org/10.3390/molecules27134165
APA StyleBiver, T. (2022). Discriminating between Parallel, Anti-Parallel and Hybrid G-Quadruplexes: Mechanistic Details on Their Binding to Small Molecules. Molecules, 27(13), 4165. https://doi.org/10.3390/molecules27134165





