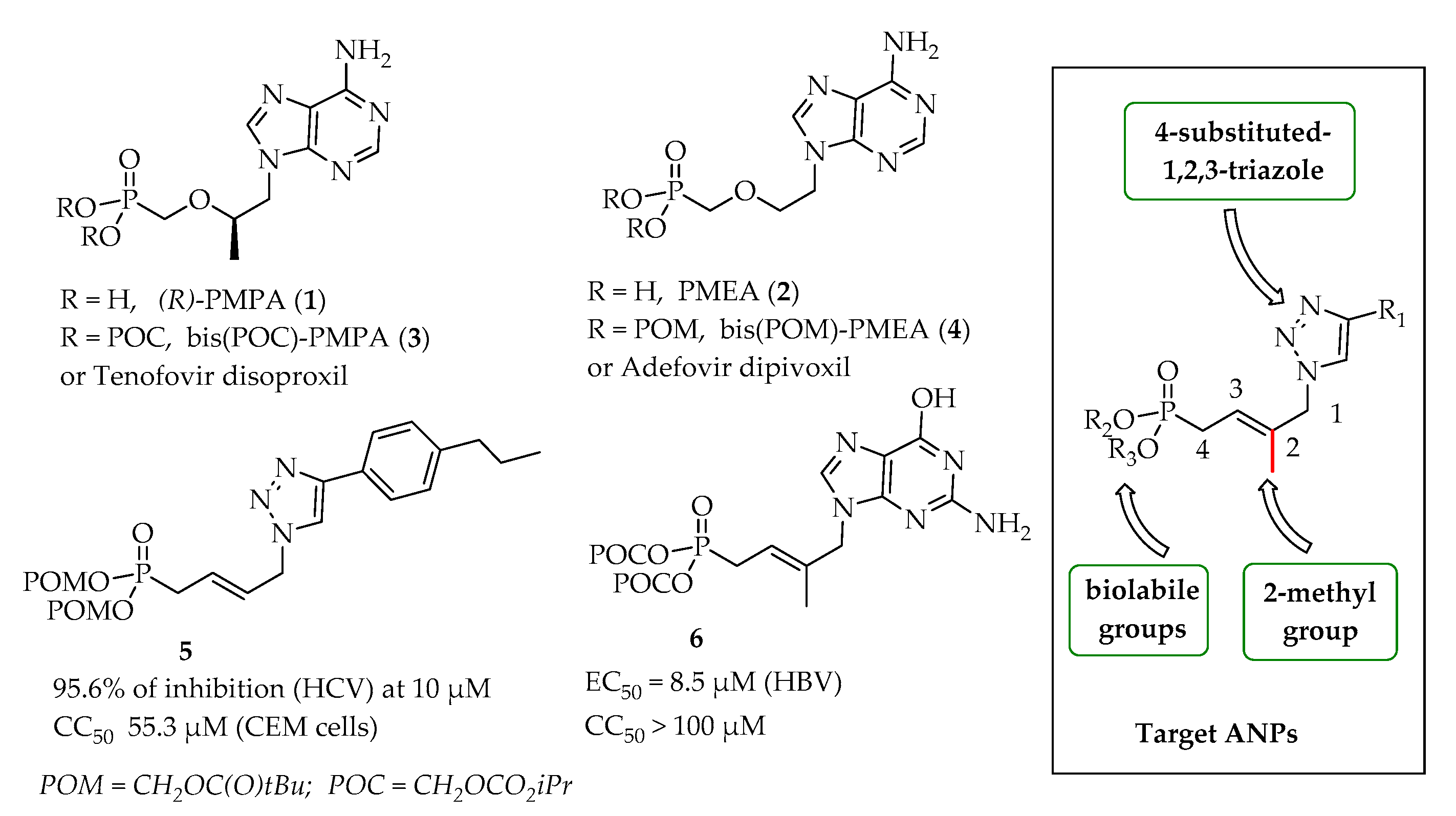
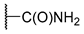3.3. Experimental Section
(E)-4-Hydroxy-3-methyl-but-2-enyl-bis(POC)phosphonate (9)
To a solution of bis(POC) allylphosphonate (7) (500 mg, 2.0 eq., 1.41 mmol) and 2-methyl-2-propen-1-ol (8) (59 μL, 1.0 eq., 0.71 mmol) in dry CH2Cl2 (5 mL), Hoveyda–Grubbs catalyst (66 mg, 15 mol%, 0.11 mmol) was added. The catalyst addition was performed in five equal portions of 3 mol% (13.20 mg, 0.021 mmol) at t = 0, 3, 6, 9 and 21 h over the course of the reaction. The solution was sonicated at 55 °C under nitrogen atmosphere for 24 h. Volatiles were evaporated and the residue was purified by silica gel column chromatography (EtOAc/PE, 7:3) to give the desired phosphonate derivative 9 (264 mg, 94%) as brown oil. 1H NMR (400 MHz, CDCl3) δ 5.65–5.57 (m, 4H, O-CH2-O), 5.41–5.37 (m, 1H, CH=C), 4.87 (sept., J = 6.2 Hz, 2H, CH(CH3)2), 3.98 (d, J = 5.3 Hz, 2H, CH2-OH), 2.69 (dd, J = 22.7, 7.6 Hz, 2H, CH2-P), 2.34 (s, 1H, OH), 1.65 (d, J = 4.5 Hz, 3H, CH3), 1.28 (d, J = 6.3 Hz, 12H, CH(CH3)2). 13C NMR (101 MHz, CDCl3) δ 153.2 (C=O), 141.6, 141.5 (CH=C), 111.6, 111.5 (CH=C), 84.1, 84.1 (O-CH2-O), 73.3 (CH(CH3)2), 68.0, 68.0 (CH2-OH), 27.3, 25.9 (CH2-P), 21.6 (CH(CH3)2), 13.9, 13.9 (CH3). 31P NMR (162 MHz, CDCl3) δ 28.80. HRMS (ESI): m/z [M+H]+ calcd for C15H28O10P: 399.14146, found: 399.14151.
(E)-4-Azido-3-methyl-but-2-enyl-bis(POC)phosphonate (10)
To a solution of (E)-4-hydroxy-3-methyl-but-2-enyl-bis(POC)phosphonate 9 (264 mg, 1.0 equiv., 0.66 mmol) in anhydrous CH2Cl2 (5 mL) were added dropwise methanesulfonyl chloride (56 μL, 1.1 equiv., 0.73 mmol) and anhydrous triethylamine (102 μL, 1.1 equiv., 0.73 mmol) at 0 °C under the argon. After 30 min stirring at room temperature, the mixture was diluted with CH2Cl2 and washed with brine solution (3 × 10 mL). The organic layers were dried over MgSO4, filtrated and concentrated in vacuo. The residue was directly used for the next step without further purification. Thus, it was dissolved in anhydrous DMF (8 mL) and sodium azide (216 mg, 5.0 equiv., 3.32 mmol) was added under nitrogen atmosphere. After 5 h stirring at room temperature, the mixture was diluted with EtOAc, washed with water and then extracted with EtOAc. The combined organic layers were washed with brine solution (5 × 10 mL), dried over MgSO4, and concentrated in vacuo. The residue was purified by silica gel column chromatography (EtOAc/PE, 4:6) to give 10 (261 mg, 93%) as a colorless oil. 1H NMR (400 MHz, CDCl3) δ 5.71–5.61 (m, 4H, O-CH2-O), 5.48–5.42 (m, 1H, CH=C), 4.92 (sept, J = 6.2 Hz, 2H, CH(CH3)2), 3.71 (d, J = 4.0 Hz, 2H, CH2-N3), 2.75 (dd, J = 23.1, 7.9, Hz, 2H, CH2-P), 1.73 (d, J = 4.5 Hz, 3H, CH3), 1.32 (d, J = 6.2 Hz, 12H, CH(CH3)2). 13C NMR (101 MHz, CDCl3) δ 153.2 (C=O), 136.2, 136.1 (C=CH), 116.4, 116.3 (CH=C), 84.2, 84.1 (O-CH2-O), 73.3 (CH(CH3)2), 58.6, 58.6 (CH2-N3), 27.6 (CH2-P), 26.2 (CH2-P), 21.6 (CH(CH3)2), 14.9, 14.9 (CH3). 31P NMR (162 MHz, CDCl3) δ 27.85. IR νmax 2985.61, 2937.37, 2100.12, 1755.01, 1255.06, 1031.52, 984.34, 949.97, 904.26, 832.67, 788.47 cm−1. HRMS (ESI): m/z [M+Na]+ calcd for C15H26N3NaO9P: 446.12988, found: 446.12990.
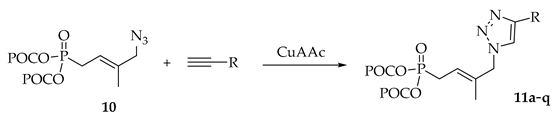
General procedures A for CuAAC reaction
To a solution of (E)-4-Azido-3-methyl-but-2-enyl-bis(POC)phosphonate 10 (1.0 equiv.) and alkyne (1.3 equiv.) in tBuOH/H2O (2:1) were added sodium ascorbate (0.6 equiv.) and CuSO4.5H2O (0.1 equiv.). The resulting suspension was stirred at 40 °C until completion (followed by TLC), then the crude mixture was co-evaporated five times with methanol (10 mL). The residue was purified by silica gel column chromatography using elution gradient of petroleum ether/ethyl acetate to give the desired bis(POC) phosphonate derivatives.
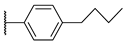
[[(E)-4-[4-(4-Propylphenyl)triazol-1-yl]-3-methyl-but-2-enyl]-(isopropoxycarbonyloxymethoxy)phosphoryl]oxymethyl isopropyl carbonate (11a)
The title compound was prepared from 10 (101 mg, 0.24 mmol) and 1-ethynyl-4-propylbenzene (49 μL mg, 0.31 mmol) following the general procedure A; the resulting suspension was stirred for 2 h at 40 °C. After purification on a silica gel column chromatography (EtOAc/PE, 4:6), the desired pure compound 11a (112 mg, 83%) was obtained as a colorless oil. 1H NMR (250 MHz, CDCl3) δ 7.76 (s, 1H, H5), 7.74 (d, J = 5.7 Hz, 2H, HAr), 7.25–7.17 (d, J = 8.5 Hz, 2H, HAr), 5.77–5.51 (m, 4H, O-CH2-O; 1H, CH=C), 4.98–4.80 (m, 2H, CH2-N; 2H, CH(CH3)2), 2.78 (dd, J = 23.1, 7.8 Hz, 2H, CH2-P), 2.59 (t, J = 7.6 Hz, 2H, Ar-CH2), 1.70–1.58 (m, 3H, CH3; 2H, Ar-CH2-CH2), 1.27 (d, J = 6.3 Hz, 12H, CH(CH3)2), 0.93 (t, J = 7.3 Hz, 3H, Ar-CH2-CH2-CH3). 13C NMR (101 MHz, CDCl3) δ 153.2 (C=O), 148.3 (Cq,Ar), 142.7 (Cq,Ar), 135.9, 135.7 (CH=C), 128.9 (CHAr), 128.1 (Cq,Ar), 125.6 (CHAr), 119.0 (CHAr), 118.1, 118.0 (CH=C), 84.1, 84.1 (O-CH2-O), 73.4 (CH(CH3)2), 57.8, 57.8 (CH2-N), 37.8 (Ar-CH2), 27.8, 26.4 (CH2-P), 24.5 (Ar-CH2-CH2), 21.6, 21.6 (CH(CH3)2), 14.2, 14.2 (CH3), 13.8 (Ar-CH2-CH2-CH3). 31P NMR (162 MHz, CDCl3) δ 27.47. HRMS (ESI): m/z [M+H]+ calcd for C26H39N3O9P: 568.24184, found: 568.24217.
[[(E)-4-[4-(4-Butylphenyl)triazol-1-yl]-3-methyl-but-2-enyl]-(isopropoxycarbonyloxymethoxy)phosphoryl]oxymethyl isopropyl carbonate (11b)
The title compound was prepared from 10 (102 mg, 0.24 mmol) and 1-butyl-4-ethynylbenzene (55 μL, 0.31 mmol) following the general procedure A; the resulting suspension was stirred for 2 h at 40 °C. After purification on a silica gel column chromatography (EtOAc/PE, 4:6), the desired pure compound 11b (123 mg, 88%) was obtained as a colorless oil. 1H NMR (250 MHz, CDCl3) δ 7.76 (d, J = 7.8 Hz, 2H, HAr), 7.75 (s, 1H, H5), 7.22 (d, J = 7.8 Hz, 2H, HAr), 5.73–5.63 (m, 4H, O-CH2-O), 5.60–5.54 (m, 1H, CH=C), 4.94 (d, J = 5.0 Hz, 2H, CH2-N), 4.90 (sept., J = 6.3 Hz, 2H, CH(CH3)2), 2.80 (dd, J = 23.3, 7.9 Hz, 2H, CH2-P), 2.62 (t, J = 7.8 Hz, 2H, Ar-CH2-CH2-CH2-CH3), 1.64–1.58 (m, 3H, CH3; 2H, Ar-CH2-CH2-CH2-CH3), 1.40–1.34 (m, 2H, Ar-CH2-CH2-CH2-CH3), 1.28 (d, J = 6.3 Hz, 12H, CH(CH3)2), 0.92 (t, J = 7.3 Hz, 3H, Ar-CH2-CH2-CH2-CH3). 13C NMR (101 MHz, CDCl3) δ 153.2 (C=O), 148.4 (Cq,Ar), 143.0 (Cq,Ar), 135.9, 135.8 (CH=C), 128.8 (CHAr), 128.0 (Cq,Ar), 125.7 (CHAr), 119.0 (CHAr), 118.1, 118.0 (CH=C), 84.2, 84.1 (O-CH2-O), 73.4 (CH(CH3)2), 57.8, 57.8 (CH2-N), 35.4 (Ar-CH2-CH2-CH2-CH3), 33.5 (Ar-CH2-CH2-CH2-CH3), 27.8 (CH2-P), 26.4 (CH2-P), 22.3 (Ar-CH2-CH2-CH2-CH3), 21.6, 21.6 (CH(CH3)2), 14.2, 14.2 (CH3), 13.9 (Ar-CH2-CH2-CH2-CH3). 31P NMR (162 MHz, CDCl3) δ 27.46. HRMS (ESI): m/z [M+Na]+ calcd for C27H40N3NaO9P: 604.23943, found: 604.23935.
[[(E)-4-[4-(4-Pentylphenyl)triazol-1-yl]-3-methyl-but-2-enyl]-(isopropoxycarbonyloxymethoxy)phosphoryl]oxymethyl isopropyl carbonate (11c)
The title compound was prepared from 10 (96 mg, 0.23 mmol) and 1-ethynyl-4-penthylbenzene (57 μL, 0.29 mmol) following the general procedure A; the resulting suspension was stirred for 2 h at 40 °C. After purification on a silica gel column chromatography (EtOAc/PE, 4:6), the desired pure compound 11c (115 mg, 85%) was obtained as a colorless oil. 1H NMR (400 MHz, CDCl3) δ 7.76 (s, 2H, HAr), 7.74 (s, 1H, H5), 7.22 (d, J = 7.8 Hz, 2H, HAr), 5.72–5.64 (m, 4H, O-CH2-O), 5.61–5.56 (m, 1H, CH=C), 4.94 (d, J = 5.1 Hz, 2H, CH2-N), 4.90 (sept., J = 6.2 Hz, 2H, CH(CH3)2), 2.80 (dd, J = 23.1, 7.8 Hz, 2H, CH2-P), 2.62 (t, J = 7.8 Hz, 2H, Ar-CH2-(CH2)3-CH3), 1.64–1.59 (m, 3H, CH3; 2H, Ar-CH2-CH2-CH2-CH2-CH3), 1.33–1.32 (m, 4H, Ar-CH2-CH2-CH2-CH2-CH3), 1.28 (d, J = 6.3 Hz, 12H, CH(CH3)2), 0.89 (t, J = 7.3 Hz, 3H, Ar-(CH2)4-CH3). 13C NMR (101 MHz, CDCl3) δ 153.2 (C=O), 148.4 (Cq,Ar), 143.0 (Cq,Ar), 135.9, 135.7 (CH=C), 128.8 (CHAr), 128.0 (Cq,Ar), 125.7 (CHAr), 119.0 (CHAr), 118.1, 118.0 (CH=C), 84.2, 84.1 (O-CH2-O), 73.4 (CH(CH3)2), 57.8, 57.8 (CH2-N), 35.7 (Ar-CH2-(CH2)3-CH3), 31.4, 31.1 (Ar-CH2-CH2-CH2-CH2-CH3), 27.8 (CH2-P), 26.4 (CH2-P), 23.9 (Ar-CH2-CH2-CH2-CH2-CH3), 22.5 (Ar-(CH2)3CH2-CH3), 21.6, 21.6 (CH(CH3)2), 14.2, 14.2 (CH3), 14.0 (Ar-(CH2)4-CH3). 31P NMR (162 MHz, CDCl3) δ 27.51. HRMS (ESI): m/z [M+Na]+ calcd for C28H42N3NaO9P: 618.25508, found: 618.25473.
[[(E)-4-[4-(4-Hexylphenyl)triazol-1-yl]-3-methyl-but-2-enyl]-(isopropoxycarbonyloxymethoxy)phosphoryl]oxymethyl isopropyl carbonate (11d)
The title compound was prepared from 10 (93 mg, 0.22 mmol) and 1-ethynyl-4-hexylbenzene (60 μL mg, 0.29 mmol) following the general procedure A; the resulting suspension was stirred for 2 h at 40 °C. After purification on a silica gel column chromatography (EtOAc/PE, 35:65), the desired pure compound 11d (112 mg, 84%) was obtained as a yellow oil. 1H NMR (400 MHz, CDCl3) δ 7.75 (d, J = 5.4 Hz, 2H, HAr), 7.72 (s, 1H, H5), 7.20 (d, J = 7.8 Hz, 2H, HAr), 5.70–5.62 (m, 4H, O-CH2-O), 5.59–5.53 (m, 1H, CH=C), 4.92 (d, J = 3.8 Hz, 2H, CH2-N), 4.88 (sept., J = 6.2 Hz, 2H, CH(CH3)2), 2.78 (dd, J = 23.1, 7.8 Hz, 2H, CH2-P), 2.60 (t, J = 7.8 Hz, 2H, Ar-CH2-(CH2)4-CH3), 1.62–1.56 (m, 3H, CH3; 2H, Ar-CH2-CH2-(CH2)3CH3), 1.33–1.25 (m, 18H, Ar-CH2-CH2-(CH2)3CH3, CH(CH3)2), 0.86 (t, J = 7.0 Hz, 3H, Ar-(CH2)5-CH3). 13C NMR (101 MHz, CDCl3) δ 153.2 (C=O), 148.3 (Cq,Ar), 143.0 (Cq,Ar), 135.9, 135.8 (CH=C), 128.8 (CHAr), 128.0 (Cq,Ar), 125.7 (CHAr), 119.1 (CHAr), 118.1, 117.9 (CH=C), 84.2, 84.1 (O-CH2-O), 73.4 (CH(CH3)2), 57.8, 57.7 (CH2-N), 35.7 (Ar-CH2-(CH2)4-CH3), 31.7, 31.3 (Ar-CH2-CH2-(CH2)3CH3), 28.9 (Ar-CH2-CH2-CH2-CH2-CH2-CH3), 27.8 (CH2-P), 26.4 (CH2-P), 23.9 (Ar-CH2-CH2-CH2-CH2-CH2-CH3), 22.6 (Ar-(CH2)4CH2-CH3), 21.6, 21.6 (CH(CH3)2), 14.2, 14.21 (CH3), 14.1 (Ar-(CH2)5-CH3). 31P NMR (162 MHz, CDCl3) δ 27.50. HRMS (ESI): m/z [M+Na]+ calcd for C29H44N3NaO9P: 632.27073, found: 632.27074.
[[(E)-4-[4-(4-Heptylphenyl)triazol-1-yl]-3-methyl-but-2-enyl]-(isopropoxycarbonyloxymethoxy)phosphoryl]oxymethyl isopropyl carbonate (11e)
The title compound was prepared from 10 (113 mg, 0.27 mmol) and 1-ethynyl-4-heptylbenzene (78 μL, 0.35 mmol) following the general procedure A; the resulting suspension was stirred for 2 h at 40 °C. After purification on a silica gel column chromatography (EtOAc/PE, 3:7), the desired pure compound 11e (151 mg, 91%) was obtained as a yellow oil. 1H NMR (400 MHz, CDCl3) δ 7.76 (d, J = 2.4 Hz, 2H, HAr), 7.74 (s, 1H, H5), 7.22 (d, J = 7.8 Hz, 2H, HAr), 5.72–5.64 (m, 4H, O-CH2-O), 5.62–5.56 (m, 1H, CH=C), 4.94 (d, J = 3.8 Hz, 2H, CH2-N), 4.89 (sept., J = 6.3 Hz, 2H, CH(CH3)2), 2.79 (dd, J = 23.1, 7.8 Hz, 2H, CH2-P), 2.61 (t, J = 7.8 Hz, 2H, Ar-CH2-(CH2)5-CH3), 1.64–1.58 (m, 3H, CH3; 2H, Ar-CH2-CH2-(CH2)4CH3), 1.33–1.25 (m, 20H, Ar-CH2-CH2-(CH2)4CH3, CH(CH3)2), 0.87 (t, J = 7.1 Hz, 3H, Ar-(CH2)6-CH3). 13C NMR (101 MHz, CDCl3) δ 153.2 (C=O), 148.4 (Cq,Ar), 143.0 (Cq,Ar), 135.9, 135.8 (CH=C), 128.8 (CHAr), 128.0 (Cq,Ar), 125.6 (CHAr), 119.0 (CHAr), 118.2, 118.1 (CH=C), 84.2, 84.1 (O-CH2-O), 73.4 (CH(CH3)2), 57.9, 57.8 (CH2-N), 35.7 (Ar-CH2-(CH2)5-CH3), 31.8, 31.4 (Ar-CH2-CH2-(CH2)4CH3), 29.7, 29.2, 29.1 (Ar-CH2-CH2-CH2-CH2-CH2-CH2-CH3), 27.8 (CH2-P), 26.4 (CH2-P), 22.6 (Ar-(CH2)5CH2-CH3), 21.6, 21.5 (CH(CH3)2), 14.2, 14.1 (CH3), 14.1 (Ar-(CH2)6-CH3). 31P NMR (162 MHz, CDCl3) δ 27.46. HRMS (ESI): m/z [M+H]+ calcd for C30H47N3O9P: 624.30444, found: 624.30373.
[[(E)-4-(4-Cyclopropyltriazol-1-yl)-3-methyl-but-2-enyl]-(isopropoxycarbonyloxymethoxy)phosphoryl]oxymethyl isopropyl carbonate (11f)
The title compound was prepared from 10 (94 mg, 0.22 mmol) and cyclopropylacetylene (24 μL, 0.29 mmol) following the general procedure A; the resulting suspension was stirred for 2 h at 40 °C. After purification on a silica gel column chromatography (EtOAc/PE, 4:6), the desired pure compound 11f (86 mg, 79%) was obtained as a colorless oil. 1H NMR (250 MHz, CDCl3) δ 7.23 (s, 1H, H5), 5.72–5.62 (m, 4H, O-CH2-O), 5.54–5.45 (m, 1H, CH=C), 4.92 (sept., J = 6.2 Hz, 2H, CH(CH3)2), 4.83 (d, J = 4.1 Hz, 2H, CH2-N), 2.76 (dd, J = 23.1, 7.8 Hz, 2H, CH2-P), 2.00–1.89 (m, 1H, CH2CHCH2), 1.59 (d, J = 5.5 Hz, 3H, CH3), 1.31 (dd, J = 6.3, 1.3 Hz, 12H, CH(CH3)2), 0.95–0.82 (m, 4H, CH2CH2). 13C NMR (101 MHz, CDCl3) δ 153.2 (C=O), 150.7 (Cq,Ar), 136.01, 135.9 (CH=C), 119.4 (CHAr), 117.8, 117.7 (CH=C), 84.1, 84.1 (O-CH2-O), 73.4 (CH(CH3)2), 57.6, 57.6 (CH2-N), 27.7, 26.4 (CH2-P), 21.7, 21.6 (CH(CH3)2), 14.2, 14.1 (CH3), 7.8 (CH2CH2), 6.7 (CH2CHCH2). 31P NMR (162 MHz, CDCl3) δ 27.6. HRMS (ESI): m/z [M+H]+ calcd for C20H33N3O9P: 490.19489, found: 490.19492.
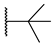
[[(E)-4-(4-tert-Butyltriazol-1-yl)-3-methyl-but-2-enyl]-(isopropoxycarbonyloxymethoxy)phosphoryl]oxymethyl isopropyl carbonate (11g)
The title compound was prepared from 10 (98 mg, 0.23 mmol) and 3,3-dimethyl-1-butyne (37 μL, 0.30 mmol) following the general procedure A; the resulting suspension was stirred for 24 h at 40 °C. After purification on a silica gel column chromatography (EtOAc/PE, 6:4), the desired pure compound 11g (56 mg, 48%) was obtained as a colorless oil. 1H NMR (400 MHz, CDCl3) δ 7.25 (s, 1H, H5), 5.71–5.62 (m, 4H, O-CH2-O), 5.51–5.45 (m, 1H, CH=C), 4.92 (sept., J = 6.2 Hz, 2H, CH(CH3)2), 4.85 (d, J = 3.9 Hz, 2H, CH2-N), 2.76 (dd, J = 23.1, 7.8 Hz, 2H, CH2-P), 1.60 (d, J = 4.5 Hz, 3H, CH3), 1.33 (s, 9H, C(CH)3), 1.31 (d, J = 6.3, Hz, 12H, CH(CH3)2). 13C NMR (101 MHz, CDCl3) δ 158.2 (Cq,Ar), 153.1 (C=O), 136.1, 136.0 (CH=C), 118.4 (CHAr), 117.6, 117.5 (CH=C), 84.2, 84.1 (O-CH2-O), 73.4 (CH(CH3)2), 57.6, 57.5 (CH2-N), 30.8 (C(CH)3), 30.3 (C(CH)3), 27.7, 26.3 (CH2-P), 21.6, 21.5 (CH(CH3)2), 14.3, 14.2 (CH3). 31P NMR (162 MHz, CDCl3) δ 27.62. HRMS (ESI): m/z [M+H]+ calcd for C21H37N3O9P: 506.22619, found: 506.22601.
[[(E)-4-[4-(Hydroxymethyl)triazol-1-yl]-3-methyl-but-2-enyl]-(isopropoxycarbonyloxymethoxy)phosphoryl]oxymethyl isopropyl carbonate (11h)
The title compound was prepared from 10 (105 mg, 0.25 mmol) and propargyl alcohol (19 μL, 0.32 mmol) following the general procedure A; the resulting suspension was stirred for 24 h at 40 °C. After purification on a silica gel column chromatography (MeOH/CH2Cl2, 5/95), the desired pure compound 11h (84 mg, 71%) was obtained as a yellow oil. 1H NMR (400 MHz, CDCl3) δ 7.55 (s, 1H, H5), 5.67–5.59 (m, 4H, O-CH2-O), 5.49–5.44 (m, 1H, CH=C), 4.92–4.86 (m, 4H, CH(CH3)2), CH2-N), 4.75 (s, 2H, CH2OH), 2.74 (dd, J = 23.1, 7.8 Hz, 2H, CH2-P), 1.57 (d, J = 4.6 Hz, 3H, CH3), 1.28 (d, J = 5.6, Hz, 12H, CH(CH3)2). 13C NMR (101 MHz, CDCl3) δ 153.2 (C=O), 135.7, 135.6 (CH=C), 121.5 (CHAr), 118.1, 118.0 (CH=C), 84.1, 84.0 (O-CH2-O), 73.5 (CH(CH3)2), 57.7, 57.6 (CH2-N), 56.5 (CH2OH), 27.7, 26.3 (CH2-P), 21.6, 21.5 (CH(CH3)2), 14.3, 14.2 (CH3). 31P NMR (162 MHz, CDCl3) δ 27.40. HRMS (ESI): m/z [M+Na]+ calcd for C18H30N3NaO10P: 502.156102, found: 502.155885.
[[(E)-4-(4-Formyltriazol-1-yl)-3-methyl-but-2-enyl]-(isopropoxycarbonyloxymethoxy)phosphoryl]oxymethyl isopropyl carbonate (11i)
The title compound was prepared from 10 (82 mg, 0.19 mmol) and 3,3-diethoxy-1-propyne (36 μL, 0.25 mmol) following the general procedure A; the resulting suspension was stirred for 2 h at 40 °C. After purification on a silica gel column chromatography (EtOAc/PE, 6/4), the desired pure compound 11i (19 mg, 21%) was obtained as a colorless oil. 1H NMR (400 MHz, CDCl3) δ 10.14 (s, 1H, CHO), 8.14 (s, 1H, H5), 5.71–5.63 (m, 4H, O-CH2-O), 5.60–5.57 (m, 1H, CH=C), 4.98 (d, J = 3.9 Hz, 2H, CH2-N), 4.92 (sept., J = 6.2 Hz, 2H, CH(CH3)2), 2.78 (dd, J = 23.3, 7.8 Hz, 2H, CH2-P), 1.63 (d, J = 4.8 Hz, 3H, CH3), 1.31 (d, J = 6.2 Hz, 12H, CH(CH3)2). 13C NMR (101 MHz, CDCl3) δ 184.9 (CHO), 153.1 (C=O), 148.0 (Cq,Ar), 134.8, 134.6 (CH=C), 125.2 (CHAr), 119.5, 119.4 (CH=C), 84.2, 84.1 (O-CH2-O), 73.5 (CH(CH3)2), 58.1, 58.0 (CH2-N), 27.8, 26.4 (CH2-P), 21.6, 21.5 (CH(CH3)2), 14.3, 14.2 (CH3). 31P NMR (162 MHz, CDCl3) δ 27.04. HRMS (ESI): m/z [M+H]+ calcd for C18H29N3O10P: 478.158507, found: 478.158468.
[[(E)-4-(4-Carbamoyltriazol-1-yl)-3-methyl-but-2-enyl]-(isopropoxycarbonyloxymethoxy)phosphoryl]oxymethyl isopropyl carbonate (11j)
The title compound was prepared from 10 (106 mg, 0.25 mmol) and propiolamide (22 mg, 0.33 mmol) following the general procedure A; the resulting suspension was stirred for 14 h at 40 °C. After purification on a silica gel column chromatography (MeOH/CH2Cl2, 3/97), the desired pure compound 11j (96 mg, 78%) was obtained as a colorless oil. 1H NMR (400 MHz, Acetone-d6) δ 8.31 (s, 1H, H5), 7.38 (s, 1H, NH), 6.78 (s, 1H, NH), 5.70–5.63 (m, 4H, O-CH2-O), 5.59–5.57 (m, 1H, CH=C), 5.08 (d, J = 4.0 Hz, 2H, CH2-N), 4.91 (sept., J = 6.2 Hz, 2H, CH(CH3)2), 2.84 (dd, J = 22.4, 7.9 Hz, 2H, CH2-P), 1.66 (d, J = 3.9 Hz, 3H, CH3), 1.29 (d, J = 6.3 Hz, 12H, CH(CH3)2). 13C NMR (101 MHz, Acetone-d6) δ 161.6 (NH2C=O), 153.1 (C=O), 143.4 (Cq,Ar), 135.4, 135.3 (CH=C), 126.0 (CHAr), 118.4, 118.3 (CH=C), 84.2, 84.1 (O-CH2-O), 72.8 (CH(CH3)2), 57.2, 57.1 (CH2-N), 27.3, 25.9 (CH2-P), 20.9 (CH(CH3)2), 13.6, 13.5 (CH3). 31P NMR (162 MHz, CDCl3) δ 27.05. HRMS (ESI): m/z [M+H]+ calcd for C18H30N4O10P: 493.169406, found: 493.169337.
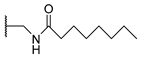
[[(E)-4-(4-Octanoylaminomethyltriazol-1-yl)-3-methyl-but-2-enyl]-(isopropoxycarbonyloxymethoxy)phosphoryl]oxymethyl isopropyl carbonate (11k)
The title compound was prepared from 10 (109 mg, 0.26 mmol) and N-N(prop-2-yn-1-yl)octanamide (56 mg, 0.33 mmol) following the general procedure A; the resulting suspension was stirred for 24 h at 40 °C. After purification on a silica gel column chromatography (MeOH/CH2Cl2, 3/97), the desired pure compound 11k (147 mg, 94%) was obtained as a yellowish oil. 1H NMR (400 MHz, CDCl3) δ 7.51 (s, 1H, H5), 6.20 (s, 1H, NH), 5.70–5.62 (m, 4H, O-CH2-O), 5.52–5.47 (m, 1H, CH=C), 4.92 (sept., J = 6.3 Hz, 2H, CH(CH3)2), 4.87 (d, J = 4.0 Hz, 2H, CH2-N), 4.50 (d, J = 5.5 Hz, 2H, ArCH2NH), 2.76 (dd, J = 23.3, 7.9 Hz, 2H, CH2-P), 2.18 (t, J = 7.7 Hz, 2H, O=CCH2(CH2)5CH3), 1.65–1.59 (m, 5H, CH3; O=CCH2CH2(CH2)4CH3), 1.31 (d, J = 6.4 Hz, 12H, CH(CH3)2), 1.24–1.28 (m, 8H, O=CCH2CH2(CH2)4CH3), 0.86 (t, J = 6.9 Hz, 3H, O=C(CH2)6CH3). 13C NMR (101 MHz, CDCl3) δ 173.2 (NHC=O), 153.2 (C=O), 145.1 (Cq,Ar), 135.6, 135.5 (CH=C), 121.8 (CHAr), 118.2, 118.1 (CH=C), 84.6, 84.1 (O-CH2-O), 73.4 (CH(CH3)2), 57.8, 57.7 (CH2-N), 36.6 (ArCH2NH), 35.0 (O=CCH2(CH2)5CH3), 31.7 (O=CCH2CH2(CH2)4CH3, 29.3 (O=CCH2CH2CH2(CH2)3CH3), 29.0 (O=C(CH2)3CH2CH2CH2CH3), 27.8 (CH2-P), 26.4 (CH2-P), 25.6 (O=C(CH2)4CH2CH2CH3), 22.6 (O=C(CH2)5CH2CH3), 21.6, 21.6 (CH(CH3)2, 14.3, 14.2 (CH3), 14.1 (O=C(CH2)6CH3). 31P NMR (162 MHz, CDCl3) δ 27.31. HRMS (ESI): m/z [M+Na]+ calcd for C26H45N4NaO10P: 627.276551, found: 627.275980.
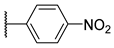
[[(E)-4-(4-Nitrophenyltriazol-1-yl)-3-methyl-but-2-enyl]-(isopropoxycarbonyloxymethoxy)phosphoryl]oxymethyl isopropyl carbonate (11l)
The title compound was prepared from 10 (100 mg, 0.24 mmol) and 1-ethynyl-4-nitrobenzene (45 mg, 0.31 mmol) following the general procedure A; the resulting suspension was stirred for 2 h at 40 °C. After purification on a silica gel column chromatography (EtOAc/PE, 5/5), the desired pure compound 11l (93 mg, 69%) was obtained as a yellowish oil. 1H NMR (400 MHz, CDCl3) δ 8.28 (d, J = 8.5 Hz, 2H, HAr), 8.04 (d, J = 8.5 Hz, 2H, HAr), 8.00 (s, 1H, H5), 5.73–5.60 (m, 5H, O-CH2-O, CH=C), 4.98 (d, J = 3.7 Hz, 2H, CH2-N), 4.89 (sept., J = 6.4 Hz, 2H, CH(CH3)2), 2.81 (dd, J = 23.2, 7.9 Hz, 2H, CH2-P), 1.66 (d, J = 4.9 Hz, 3H, CH3), 1.28 (d, J = 6.2 Hz, 12H, CH(CH3)2). 13C NMR (101 MHz, CDCl3) δ 153.2 (C=O), 147.3 (Cq,Ar), 146.1 (Cq,Ar), 137.0 (Cq,Ar), 135.3, 135.2 (CH=C), 126.2 (CHAr), 124.2 (CHAr), 121.0 (CHAr), 119.1, 119.0 (CH=C), 84.2, 84.1 (O-CH2-O), 73.5 (CH(CH3)2), 58.2, 58.1 (CH2-N), 27.8, (CH2-P), 26.4 (CH2-P), 21.6, 21.5 (CH(CH3)2), 14.3, 14.3 (CH3). 31P NMR (162 MHz, CDCl3) δ 27.26. HRMS (ESI): m/z [M+H]+ calcd for C23H32N4O11P: 571.179971, found: 571.179797.
1-[(E)-4-[Bis(isopropoxycarbonyloxymethoxy)phosphoryl]-2-methyl-but-2-enyl]triazole-4-carboxylic acid (11m)
To a solution of propiolic acid (49 μL, 1.1 equiv., 0.80mmol) and (E)-4-Azido-3-methyl-but-2-enyl-bis(POC)phosphonate 10 (307 mg, 1.0 equiv., 0.73 mmol) in anhydrous CH2Cl2 (10 mL) was added 2-nitrophenylboronic acid (25 mg, 0.21 equiv., 0.15 mmol) at room temperature. The solution was stirred for 42 h at room temperature. After evaporation of the solvent, the residue was purified by silica gel chromatography (MeOH/EtOAc, 1/9) to give compound 11m (203 mg, 57%) as a colorless oil. 1H NMR (400 MHz, CD3OD) δ 8.28 (s, 1H, H5), 5.72–5.64 (m, 4H, O-CH2-O), 5.57–5.51 (m, 1H, CH=C), 5.06 (d, J = 4.4 Hz, 2H, CH2-N), 4.94 (sept., J = 6.4 Hz, 2H, CH(CH3)2), 2.91 (dd, J = 23.2, 7.9 Hz, 2H, CH2-P), 1.67 (d, J = 4.8 Hz, 3H, CH3), 1.33 (d, J = 6.2 Hz, 12H, CH(CH3)2). 13C NMR (101 MHz, CD3OD) δ 153.2 (C=O), 136.0, 135.8 (CH=C), 126.7 (CHAr), 117.5, 117.3 (CH=C), 84.4, 84.3 (O-CH2-O), 73.1 (CH(CH3)2), 57.2, 57.1 (CH2-N), 26.8, 25.4 (CH2-P), 20.5 (CH(CH3)2), 13.6, 13.1 (CH3). 31P NMR (162 MHz, CD3OD) δ 27.94. HRMS (ESI): m/z [M+H]+ calcd for C18H29N3O11P: 494.153422, found: 494.153404.
Methyl-1-[(E)-4-[bis(isopropoxycarbonyloxymethoxy)phosphoryl]-2-methyl-but-2-enyl]triazole-4-carboxylate (11n)
The title compound was prepared from 10 (124 mg, 0.29 mmol) and methyl propiolate (34 μL, 0.38 mmol) using the general procedure A; the resulting suspension was stirred for 2 h at 40 °C. After purification on a silica gel column chromatography (EtOAc/PE, 6/4), the desired pure compound 11n (92 mg, 63%) was obtained as a colorless oil. 1H NMR (250 MHz, CD3OD) δ 8.57 (s, 1H, H5), 5.79–5.69 (m, 4H, O-CH2-O), 5.65–5.56 (m, 1H, CH=C), 5.14 (d, J = 4.2 Hz, 2H, CH2-N), 4.99 (sept., J = 6.4 Hz, 2H, CH(CH3)2), 4.00 (s, 3H, OCH3), 2.97 (dd, J = 23.2, 7.9 Hz, 2H, CH2-P), 1.74 (d, J = 4.7 Hz, 3H, CH3), 1.39 (d, J = 6.2 Hz, 12H, CH(CH3)2). 13C NMR (101 MHz, CDCl3) δ 161.0 (O=C-OMe), 153.1 (C=O), 140.3 (Cq,Ar), 135.1, 134.9 (CH=C), 127.4 (CHAr), 119.1, 118.9 (CH=C), 84.2, 84.1 (O-CH2-O), 73.4 (CH(CH3)2), 58.0, 57.9 (CH2-N), 52.1 (OCH3), 27.8, 26.4 (CH2-P), 21.6, 21.5 (CH(CH3)2), 14.3, 14.2 (CH3). 31P NMR (162 MHz, CDCl3) δ 27.19. HRMS (ESI): m/z [M+Na]+ calcd for C19H30N3NaO11P: 530.151016, found: 530.151002.
Ethyl-1-[(E)-4-[bis(isopropoxycarbonyloxymethoxy)phosphoryl]-2-methyl-but-2-enyl]triazole-4-carboxylate (11o)
The title compound was prepared from 10 (120 mg, 0.28 mmol) and methyl propiolate (37 μL, 0.37 mmol) using the general procedure A; the resulting suspension was stirred for 2 h at 40 °C. After purification on a silica gel column chromatography (EtOAc/PE, 5/5), the desired pure compound 11o (94 mg, 64%) was obtained as a colorless oil. 1H NMR (400 MHz, CD3OD) δ 8.53 (s, 1H, H5), 5.74–5.66 (m, 4H, O-CH2-O), 5.59–5.54 (m, 1H, CH=C), 5.10 (d, J = 4.2 Hz, 2H, CH2-N), 4.95 (sept., J = 6.3 Hz, 2H, CH(CH3)2), 4.43 (q, J = 7.1 Hz, 2H, OCH2CH3), 2.93 (dd, J = 23.2, 7.9 Hz, 2H, CH2-P), 1.70 (d, J = 4.8 Hz, 3H, CH3), 1.42 (t, J = 7.1 Hz, 3H, OCH2CH3), 1.34 (d, J = 6.7 Hz, 12H, CH(CH3)2). 13C NMR (101 MHz, CD3OD) δ 161.5 (O=C-OEt), 153.2 (C=O), 139.7 (Cq,Ar), 135.8, 135.6 (CH=C), 128.2 (CHAr), 117.9, 117.8 (CH=C), 84.3, 84.2 (O-CH2-O), 73.1 (CH(CH3)2), 60.8 (OCH2CH3), 57.2, 57.1 (CH2-N), 26.8, 25.4 (CH2-P), 20.5 (CH(CH3)2), 13.2 (OCH2CH3), 13.1, 13.0 (CH3). 31P NMR (162 MHz, CD3OD) δ 27.92. HRMS (ESI): m/z [M+Na]+ calcd for C20H32N3NaO11P: 544.166666, found: 544.166837.
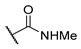
[[(E)-4-(4-Methylcarbamoyltriazol-1-yl)-3-methyl-but-2-enyl]-(isopropoxycarbonyloxymethoxy)phosphoryl]oxymethyl isopropyl carbonate (11p)
The title compound was prepared from 10 (121 mg, 0.29 mmol) and N-methylprop-2-ynamide (31 mg, 0.37 mmol) following the general procedure A; the resulting suspension was stirred for 2 h at 40 °C. After purification on a silica gel column chromatography (MeOH/CH2Cl2, 1/99), the desired pure compound 11p (118 mg, 82%) was obtained as a colorless oil. 1H NMR (400 MHz, CD3OD) δ 8.34 (s, 1H, H5), 5.74–5.66 (m, 4H, O-CH2-O), 5.58–5.52 (m, 1H, CH=C), 5.09 (d, J = 4.0 Hz, 2H, CH2-N), 4.96 (sept., J = 6.2 Hz, 2H, CH(CH3)2), 2.98 (s, 3H, NHCH3), 2.92 (dd, J = 23.2, 7.9 Hz, 2H, CH2-P), 1.69 (d, J = 4.6 Hz, 3H, CH3), 1.35 (d, J = 6.3 Hz, 12H, CH(CH3)2). 13C NMR (101 MHz, CD3OD) δ 161.7 (O=C-NHCH3), 153.2 (C=O), 142.9 (Cq,Ar), 136.0, 135.8 (CH=C), 125.7 (CHAr), 117.5, 117.4 (CH=C), 84.4, 84.3 (O-CH2-O), 73.1 (CH(CH3)2), 57.1, 57.0 (CH2-N), 26.8, 25.4 (CH2-P), 24.7 (NHCH3), 20.5 (CH(CH3)2), 13.1, 13.1 (CH3). 31P NMR (162 MHz, CD3OD) δ 27.91. HRMS (ESI): m/z [M+Na]+ calcd for C19H31N4NaO10P: 529.167001, found: 529.166551.
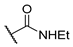
[[(E)-4-[4-(Ethylcarbamoyl)triazol-1-yl]-3-methyl-but-2-enyl]-(isopropoxycarbonyloxymethoxy)phosphoryl]oxymethyl isopropyl carbonate (11q)
The title compound was prepared from 10 (115 mg, 0.27 mmol) and N-ethylprop-2-ynamide (34 mg, 0.35 mmol) following the general procedure A; the resulting suspension was stirred for 2 h at 40 °C. After purification on a silica gel column chromatography (MeOH/CH2Cl2, 1/99), the desired pure compound 11q (128 mg, 91%) was obtained as a colorless oil. 1H NMR (400 MHz, CD3OD) δ 8.35 (s, 1H, H5), 5.75–5.67 (m, 4H, O-CH2-O), 5.59–5.53 (m, 1H, CH=C), 5.09 (d, J = 4.0 Hz, 2H, CH2-N), 4.96 (sept., J = 6.2 Hz, 2H, CH(CH3)2), 3.47 (q, J = 7.2 Hz, 2H, NHCH2CH3), 2.93 (dd, J = 23.1, 7.9 Hz, 2H, CH2-P), 1.70 (d, J = 4.7 Hz, 3H, CH3), 1.35 (d, J = 6.2 Hz, 12H, CH(CH3)2), 1.28 (t, J = 7.2 Hz, 3H, NHCH2CH3). 13C NMR (101 MHz, CD3OD) δ 160.8 (O=C-NH), 153.2 (C=O), 142.99 (Cq,Ar), 136.0, 135.8 (CH=C), 125.7 (CHAr), 117.5, 117.4 (CH=C), 84.4, 84.3 (O-CH2-O), 73.1 (CH(CH3)2), 57.1, 57.0 (CH2-N), 33.7 (NHCH2CH3), 26.8, 25.4 (CH2-P), 20.5 (CH(CH3)2), 13.6 (NHCH2CH3), 13.1, 13.0 (CH3). 31P NMR (162 MHz, CD3OD) δ 27.91. HRMS (ESI): m/z [M+Na]+ calcd for C20H33N4NaO10P: 543.182651, found: 543.182603.
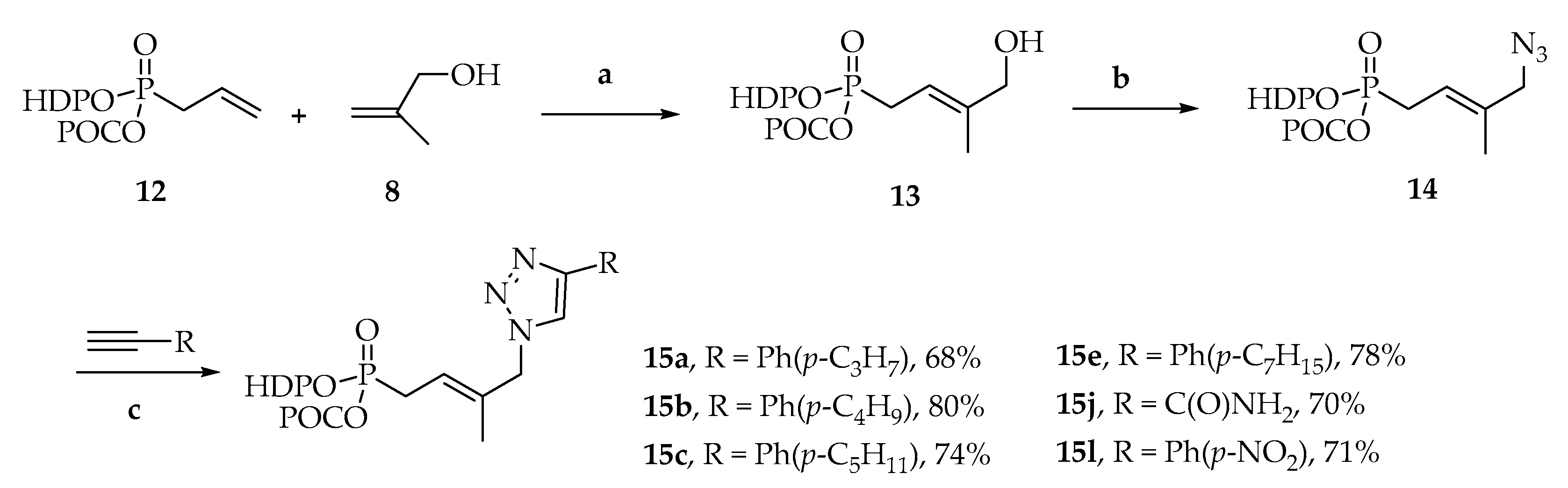
(E)-4-Hydroxy-3-methyl-but-2-enyl-(HDP/POC) phosphonate (13)
To a solution of (HDP/POC) allylphosphonate (12) (1.00 g, 2.0 equiv., 1.94 mmol) and 2-methyl-2-propen-1-ol (8) (81 μL, 1.0 equiv., 0.97 mmol) in dry CH2Cl2 (10 mL), Hoveyda–Grubbs catalyst (91 mg, 15 mol%, 0.15 mmol) was added. The catalyst addition was performed in five equal portions of 3 mol% (18.20 mg, 0.029 mmol) at t = 0, 3, 6, 9 and 21 h over the course of the reaction. The solution was sonicated at 55 °C under nitrogen atmosphere for 24 h. Volatiles were evaporated and the residue was purified by silica gel column chromatography (EtOAc/PE, 5/5) to give the desired phosphonate derivative 13 (480 mg, 88 %) as brown oil. 1H NMR (400 MHz, CDCl3) δ 5.67–5.59 (m, 2H, O-CH2-O), 5.47–5.41 (m, 1H, CH=C), 4.92 (sept., J = 6.3 Hz, 1H, CH(CH3)2), 4.21–4.08 (m, 2H, Ha), 4.02 (d, J = 5.0 Hz, 2H, CH2-OH), 3.47 (t, J = 6.2 Hz, 2H, Hc), 3.38 (t, J = 6.7 Hz, 2H, CH3(CH2)14CH2-O), 2.67 (dd, J = 22.5, 7.8 Hz, 2H, CH2-P), 1.90 (quint., J = 6.3 Hz, 2H, Hb), 1.69 (dd, J = 4.3, 1.3 Hz, 3H, CH3), 1.54 (quint., J = 7.0 Hz, 2H, CH3(CH2)13CH2CH2-O), 1.32–1.24 (m, 32H, CH3(CH2)13CH2CH2-O, CH(CH3)2), 0.87 (t, J = 6.7 Hz, 3H, CH3(CH2)15O). 13C NMR (101 MHz, CDCl3) δ 153.3 (C=O), 140.8, 140.7 (CH=C), 112.6, 112.5 (CH=C), 84.4, 84.3 (O-CH2-O), 73.1 (CH(CH3)2), 71.2 (CH3(CH2)14CH2-O), 68.1, 68.0 (CH2-OH), 66.5, 66.4 (Cc), 63.3, 63.2 (Ca), 31.9, 30.7, 30.6 (Cb), 29.7, 29.5, 29.4, 27.1 (CH2-P), 26.2, 25.7 (CH2-P), 22.7, 21.6 (CH(CH3)2), 14.1 (CH3(CH2)15O), 13.9, 13.8 (CH3). 31P NMR (162 MHz, CDCl3) δ 26.71. HRMS (ESI): m/z [M+H]+ calcd for C29H58O8P: 565.386382, found: 565.385868.
(E)-4-Azido-3-methyl-but-2-enyl-(HDP/POC) phosphonate (14)
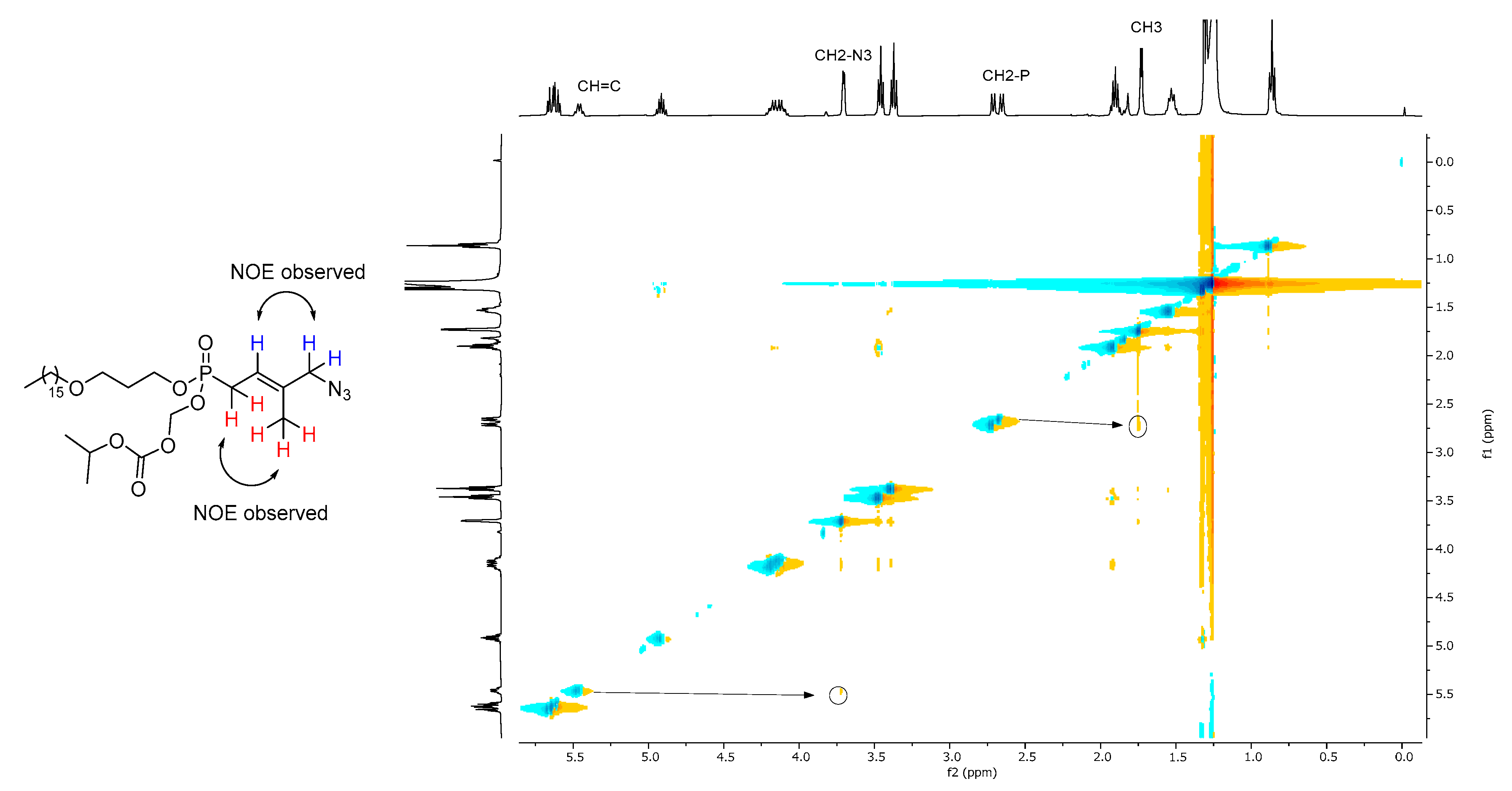
To a solution of (E)-4-hydroxy-3-methyl-but-2-enyl-(HDP/POC)phosphonate 13 (277 mg, 1.0 equiv., 0.49 mmol) in anhydrous CH2Cl2 (5 mL) were added dropwise methansulfonyl chloride (42 μL, 1.1 equiv., 0.54 mmol) and anhydrous triethylamine (75 μL, 1.1 equiv., 0.54 mmol) at 0 °C under the argon. After 40 min stirring at room temperature, the mixture was diluted with CH2Cl2 and washed with water (3 × 10 mL) and brine. The organic layer was dried over MgSO4, filtrated and concentrated in vacuo. The residue was directly used for the next step without further purification. To a solution of the above product in anhydrous DMF (8 mL) was added sodium azide (159 mg, 5.0 equiv., 2.45 mmol) under nitrogen atmosphere. After 5 h stirring at room temperature, the mixture was diluted with EtOAc and water and then extracted with EtOAc. The combined organic layer was washed with water (5 × 10 mL), brine, dried over MgSO4, and concentrated in vacuo. The residue was purified by silica gel column chromatography (EtOAc/PE, 4/6) to give (E)-4-Azido-3-methyl-but-2-enyl-(HDP/POC) phosphonate 14 (242 mg, 84%) as a colorless oil. 1H NMR (400 MHz, CDCl3) δ 5.69–5.58 (m, 2H, O-CH2-O), 5.51–5.41 (m, 1H, CH=C), 4.91 (sept., J = 6.3 Hz, 1H, CH(CH3)2), 4.22–4.07 (m, 2H, Ha), 3.70 (d, J = 3.9 Hz, 2H, CH2-N3), 3.46 (t, J = 6.2 Hz, 2H, Hc), 3.37 (t, J = 6.7 Hz, 2H, CH3(CH2)14CH2-O), 2.68 (dd, J = 22.7, 7.7 Hz, 2H, CH2-P), 1.90 (quint., J = 6.3 Hz, 2H, Hb), 1.73 (d, J = 4.3 Hz, 3H, CH3), 1.53 (quint., J = 7.0 Hz, 2H, CH3(CH2)13CH2CH2-O), 1.32–1.22 (m, 32H, CH3(CH2)13CH2CH2-O, CH(CH3)2), 0.86 (t, J = 6.7 Hz, 3H, CH3(CH2)15O). 13C NMR (100 MHz, CDCl3) δ 153.3 (C=O), 135.4, 135.3 (CH=C), 117.3, 117.2 (CH=C), 84.5, 84.4 (O-CH2-O), 73.1 (CH(CH3)2), 71.2 (CH3(CH2)14CH2-O), 66.5 (Cc), 63.5, 63.4 (Ca), 58.7, 58.6 (CH2-N3), 32.0, 30.8, 30.7 (Cb), 29.6 (CH2-P), 26.1, 26.0 (CH2-P), 22.7, 21.6 (CH(CH3)2), 14.8, 14.7 (CH=CCH3), 14.1 (CH3(CH2)15O). 31P NMR (162 MHz, CDCl3) δ 27.80. IR νmax (neat, cm−1): 2922.60, 2851.80, 2094.55, 1755.94, 1460.42, 1257.26, 1100.26, 1047.93, 992.52, 949.43 cm−1. HRMS (ESI): m/z [M+H]+ calcd for C29H57N3O7P: 590.392864, found: 590.393411.
General Procedure B for Huisgen 1,3-dipolar cycloaddition
To a solution of terminal alkyne (1.3 equiv.) and (E)-4-Azido-3-methyl-but-2-enyl-(HDP/POC) phosphonate 14 (1.0 equiv.) in tBuOH/H2O (2:1) were added sodium ascorbate (0.6 equiv.) and CuSO4.5H2O (0.1 equiv.). The resulting suspension was stirred at 40 °C until completion (followed by TLC), then the crude mixture was co-evaporated five times with methanol. The residue was purified by silica gel column chromatography using an elution gradient of petroleum ether/ethyl acetate to give the desired HDP/POC phosphonate derivatives.
[3-Hexadecyloxypropoxy-[(E)-3-methyl-4-[4-(4-propylphenyl]triazol-1-yl]but-2-enyl]phosphoryl]oxymethyl isopropyl carbonate (15a)
The title compound was prepared from 14 (123 mg, 0.21 mmol) and 1-ethynyl-4-propylbenzene (43 μL mg, 0.27 mmol) following the general procedure B; the resulting suspension was stirred for 2 h at 40 °C. After purification on a silica gel column chromatography (EtOAc/PE, 4/6), the desired pure compound 15a (104 mg, 68%) was obtained as a colorless oil. 1H NMR (400 MHz, CDCl3) δ 7.75 (s, 1H, H5), 7.73 (s, 2H, HAr), 7.22 (d, J = 8.0 Hz, 2H, HAr), 5.70 ‒ 5.56 (m, 3H, O-CH2-O; 1H, CH=C), 4.94 (s, 2H, CH2-N), 4.89 (sept., J = 6.3 Hz, 1H, CH(CH3)2), 4.22 ‒ 4.11 (m, 2H, Ha), 3.44 (t, J = 6.1 Hz, 2H, Hc), 3.35 (t, J = 6.7 Hz, 2H, CH3(CH2)14CH2-O), 2.73 (dd, J = 22.8, 7.8 Hz, 2H, CH2-P), 2.60 (t, J = 7.5 Hz, 2H, Ar-CH2CH2CH3), 1.90 (quint., J = 6.3 Hz, 2H, Hb), 1.68–1.63 (m, 3H, CH3; 2H, CH2CH2CH3), 1.52 (quint., J = 7.0 Hz, 2H, CH3(CH2)13CH2CH2-O), 1.29–1.25 (m, 32H, CH3(CH2)13CH2CH2-O, CH(CH3)2), 0.95 (t, J = 7.3 Hz, 3H, CH2CH2CH3), 0.88 (t, J = 7.0 Hz, 3H, CH3(CH2)15O). 13C NMR (101 MHz, CDCl3) δ 153.3 (C=O), 148.3 (Cq,Ar), 142.8 (Cq,Ar), 128.9 (CHAr), 128.0 (CH=C), 125.6 (CHAr), 118.9 (CH=C), 118.8 (CHAr), 84.4 (O-CH2-O), 73.2 (CH(CH3)2), 71.2 (CH3(CH2)14CH2-O), 66.4 (Cc), 63.5 (Ca), 57.9 (CH2-N), 37.8 (ArCH2CH2CH3), 31.9, 30.7 (Cb), 29.6 (CH2-P), 24.5 (ArCH2CH2CH3), 22.7, 21.6, 21.5 (CH(CH3)2), 14.3 (C=CCH3), 14.1 (CH3(CH2)15O), 13.7 (ArCH2CH2CH3). 31P NMR (162 MHz, CDCl3) δ 27.46. HRMS (ESI): m/z [M+H]+ calcd for C40H69N3NaO7P: 734.486765, found: 734.486600.
[3-Hexadecyloxypropoxy-[(E)-3-methyl-4-[4-(4-pentylphenyl)triazol-1-yl]but-2-enyl]phosphoryl]oxymethyl isopropyl carbonate (15c)
The title compound was prepared from 14 (93 mg, 0.16mmol) and 1-ethynyl-4-penthylbenzene (40 μL, 0.21 mmol) following the general procedure B; the resulting suspension was stirred for 2 h at 40 °C. After purification on a silica gel column chromatography (EtOAc/PE, 4/6), the desired pure compound 15c (89 mg, 74%) was obtained as a colorless oil. 1H NMR (400 MHz, CDCl3) δ 7.74 (d, J = 3.8 Hz, 2H, HAr), 7.73 (s, 1H, H5), 7.22 (d, J = 8.0 Hz, 2H, HAr), 5.70 ‒ 5.56 (m, 3H, O-CH2-O; 1H, CH=C), 4.94 (s, 2H, CH2-N), 4.90 (sept., J = 6.2 Hz, 1H, CH(CH3)2), 4.22–4.11 (m, 2H, Ha), 3.44 (t, J = 6.1 Hz, 2H, Hc), 3.35 (t, J = 6.7 Hz, 2H, CH3(CH2)14CH2-O), 2.73 (dd, J = 22.8, 7.8 Hz, 2H, CH2-P), 2.62 (t, J = 7.8 Hz, 2H, Ar-CH2(CH2)3CH3), 1.90 (quint., J = 6.3 Hz, 2H, Hb), 1.65–1.61 (m, 5H, CH3, Ar-CH2CH2CH2CH2CH3), 1.52 (quint., J = 7.0 Hz, 2H, CH3(CH2)13CH2CH2-O), 1.29–1.25 (m, 36H, CH3(CH2)13CH2CH2-O, CH(CH3)2, Ar-CH2CH2CH2CH2CH3), 0.88 (t, J = 6.9 Hz, 6H, Ar-(CH2)4CH3, CH3(CH2)15O). 13C NMR (101 MHz, CDCl3) δ 153.2 (C=O), 148.4, 148.3 (Cq,Ar), 143.1 (Cq,Ar), 135.2, 135.0 (Cq,Ar), 128.8 (CHAr), 127.9 (CH=C), 125.6 (CHAr), 118.9 (CH=C), 118.8 (CHAr), 84.4 (O-CH2-O), 73.2 (CH(CH3)2), 71.2 (CH3(CH2)14CH2-O), 66.4 (Cc), 63.6 (Ca), 57.8 (CH2-N), 35.7 (Ar-CH2(CH2)3CH3), 31.9, 31.5, 31.1, 30.8, 30.7 (Cb), 29.8, 29.7, 29.6, 29.5, 29.4, 26.2 (CH2-P), 22.7, 22.5, 21.6, 21.5 (CH(CH3)2), 14.3, 14.2 (C=CCH3), 14.1 (CH3(CH2)15O), 14.0 (Ar-(CH2)4CH3). 31P NMR (162 MHz, CDCl3) δ 27.56. HRMS (ESI): m/z [M+Na]+ calcd for C42H72N3NaO7P: 784.500009, found: 784.499917.
[3-Hexadecyloxypropoxy-[(E)-3-methyl-4-[4-(heptylphenyl)triazol-1-yl]but-2-enyl]phosphoryl]oxymethyl isopropyl carbonate (15e)
The title compound was prepared from 14 (113 mg, 0.19 mmol) and 1-ethynyl-4-heptylbenzene (56 μL, 0.25 mmol) following the general procedure B; the resulting suspension was stirred for 2 h at 40 °C. After purification on a silica gel column chromatography (EtOAc/PE, 4/6), the desired pure compound 15e (118 mg, 78%) was obtained as a colorless oil. 1H NMR (400 MHz, CDCl3) δ 7.73 (t, J = 3.9 Hz, 3H, HAr, H5), 7.22 (d, J = 8.0 Hz, 2H, HAr), 5.70–5.60 (m, 3H, O-CH2-O; 1H, CH=C), 4.94 (s, 2H, CH2-N), 4.89 (sept., J = 6.3 Hz, 1H, CH(CH3)2), 4.20–4.11 (m, 2H, Ha), 3.43 (t, J = 6.1 Hz, 2H, Hc), 3.35 (t, J = 6.7 Hz, 2H, CH3(CH2)14CH2-O), 2.73 (dd, J = 22.8, 7.8 Hz, 2H, CH2-P), 2.63 (t, J = 7.8 Hz, 2H, Ar-CH2(CH2)5CH3), 1.90 (quint., J = 6.3 Hz, 2H, Hb), 1.65 (d, J = 4.1 Hz, 3H, C=CCH3), 1.60 (quint., J = 7.2, Ar-CH2CH2(CH2)4CH3), 1.52 (quint., J = 7.0 Hz, 2H, CH3(CH2)13CH2CH2-O), 1.29–1.25 (m, 40H, CH3(CH2)13CH2CH2-O, CH(CH3)2, Ar-CH2CH2(CH2)4CH3), 0.93 (t, J = 7.3 Hz, 3H, Ar-(CH2)6CH3), 0.88 (t, J = 6.7 Hz, 3H, CH3(CH2)15O). 13C NMR (101 MHz, CDCl3) δ 153.3 (C=O), 143.5 (Cq,Ar), 135.2 (Cq,Ar), 135.1 (Cq,Ar), 129.5, 129.3 ((CHAr), 128.8 (CHAr), 127.9 (CH=C), 125.6 (CHAr), 118.9 (CH=C), 84.5, 84.4 (O-CH2-O), 73.3 (CH(CH3)2), 71.2 (CH3(CH2)14CH2-O), 66.4 (Cc), 63.6, 63.5 (Ca), 57.8 (CH2-N), 35.4 (Ar-CH2(CH2)5CH3), 33.5, 32.0, 31.4, 30.8, 30.7 (Cb), 30.2, 29.7, 29.7, 29.6, 29.6, 29.5, 29.4, 27.6, 26.2 (CH2-P), 22.7, 22.3, 21.6, 21.6 (CH(CH3)2), 14.3 (C=CCH3), 14.1 (CH3(CH2)15O), 13.9 (Ar-(CH2)6CH3). 31P NMR (162 MHz, CDCl3) δ 27.49. HRMS (ESI): m/z [M+H]+ calcd for C44H77N3O7P: 790.549365, found: 790.549322.
[[(E)-4-(4-Carbamoyltriazol-1-yl)-3-methyl-but-2-enyl]-(3-hexadecyloxypropoxy)phosphoryl]oxymethyl isopropyl carbonate (15j)
The title compound was prepared from 14 (166 mg, 0.28 mmol) and propiolamide (25 mg, 0.37 mmol) following the general procedure B; the resulting suspension was stirred for 18 h at 40 °C. After purification on a silica gel column chromatography (MeOH/CH2Cl2, 3/97), the desired pure compound 15j (130 mg, 70%) was obtained as a colorless oil. 1H NMR (400 MHz, CDCl3) δ 8.07 (s, 1H, H5), 7.05 (s, 1H, NH2), 5.76 (s, 1H, NH2), 5.69–5.58 (m, 2H, O-CH2-O; 1H, CH=C), 4.94 ‒ 4.91 (m, 2H, CH2-N; 1H, CH(CH3)2), 4.22–4.12 (m, 2H, Ha), 3.46 (t, J = 6.1 Hz, 2H, Hc), 3.37 (t, J = 6.7 Hz, 2H, CH3(CH2)14CH2-O), 2.71 (dd, J = 22.9, 7.8 Hz, 2H, CH2-P), 1.90 (quint., J = 6.3 Hz, 2H, Hb), 1.62 (d, J = 4.4 Hz, 3H, C=CCH3), 1.52 (quint., J = 7.0 Hz, 2H, CH3(CH2)13CH2CH2-O), 1.31–1.24 (m, 32H, CH3(CH2)13CH2CH2-O, CH(CH3)2), 0.87 (t, J = 7.0 Hz, 3H, CH3(CH2)15O). 13C NMR (101 MHz, CDCl3) δ 161.8 (NH2C=O), 153.2 (C=O), 143.1 (Cq,Ar), 134.2, 134.1 (CH=C), 125.6 (CHAr), 120.1, 120.0 (CH=C), 84.5, 84.4 (O-CH2-O), 73.2 (CH(CH3)2), 71.3 (CH3(CH2)14CH2-O), 66.4 (Cc), 63.6, 63.5 (Ca), 58.2, 58.1 (CH2-N), 31.9, 30.8, 30.7 (Cb), 29.7, 29.6, 29.6, 29.6, 29.5, 29.3, 27.6, 26.2, 26.1 (CH2-P), 22.7, 21.6 (CH(CH3)2), 14.3, 14.2 (C=CCH3), 14.1 (CH3(CH2)15O). 31P NMR (162 MHz, CDCl3) δ 27.11. HRMS (ESI): m/z [M+H]+ calcd for C32H60N4O8P: 659.414328, found: 659.414346.
[3-Hexadecyloxypropoxy-[(E)-3-methyl-4-[4-(nitrophenyl)triazol-1-yl]but-2-enyl]phosphoryl]oxymethyl isopropyl carbonate (15l)
The title compound was prepared from 14 (115 mg, 0.20 mmol) and 1-ethynyl-4-nitrobenzene (37 mg, 0.25 mmol) following the general procedure B; the resulting suspension was stirred for 3 h at 40 °C. After purification on a silica gel column chromatography (EtOAc/PE, 5/5), the desired pure compound 15l (102 mg, 71%) was obtained as a colorless oil. 1H NMR (400 MHz, CDCl3) δ 8.27 (d, J = 8.8 Hz, 2H, HAr), 8.01 (d, J = 8.9 Hz, 2H, HAr), 7.98 (s, 1H, H5), 5.70 ‒ 5.58 (m, 3H, O-CH2-O; 1H, CH=C), 4.97 (s, 2H, CH2-N), 4.89 (sept., J = 6.3 Hz, 1H, CH(CH3)2), 4.22–4.11 (m, 2H, Ha), 3.44 (t, J = 6.2 Hz, 2H, Hc), 3.35 (t, J = 6.6 Hz, 2H, CH3(CH2)14CH2-O), 2.73 (dd, J = 22.8, 7.8 Hz, 2H, CH2-P), 1.90 (quint., J = 6.2 Hz, 2H, Hb), 1.66 (d, J = 4.4 Hz, 3H, C=CCH3), 1.52 (quint., J = 7.0 Hz, 2H, CH3(CH2)13CH2CH2-O), 1.27–1.23 (m, 32H, CH3(CH2)13CH2CH2-O, CH(CH3)2), 0.86 (t, J = 7.0 Hz, 3H, CH3(CH2)15O). 13C NMR (101 MHz, CDCl3) δ 153.2 (C=O), 147.3 (Cq,Ar), 146.0 (Cq,Ar), 136.9 (Cq,Ar), 134.7, 134.5 (CH=C), 126.1 (CHAr), 124.3 (CHAr), 121.0 (CHAr), 119.8, 119.7 (CH=C), 84.4, 84.3 (O-CH2-O), 73.3, 73.1 (CH(CH3)2), 71.2 (CH3(CH2)14CH2-O), 66.5, 66.3 (Cc), 63.6, 63.5 (Ca), 58.1, 58.0 (CH2-N), 31.9, 30.8, 30.7 (Cb), 29.7, 29.6, 29.6, 29.6, 29.5, 29.5, 29.3, 27.5, 26.1, 26.1 (CH2-P), 22.7, 21.6, 21.6, 21.6 (CH(CH3)2), 14.4, 14.3 (C=CCH3), 14.1 (CH3(CH2)15O). 31P NMR (162 MHz, CDCl3) δ 27.31. HRMS (ESI): m/z [M+Na]+ calcd for C37H61N4NaO9P: 759.406837, found: 759.406817.