Colour Me Blue: The History and the Biotechnological Potential of Pyocyanin
Abstract
1. Introduction
2. History and Chronology of Studies with Pyocyanin
3. Pseudomonas aeruginosa: The Exclusive Natural Pyocyanin Producer
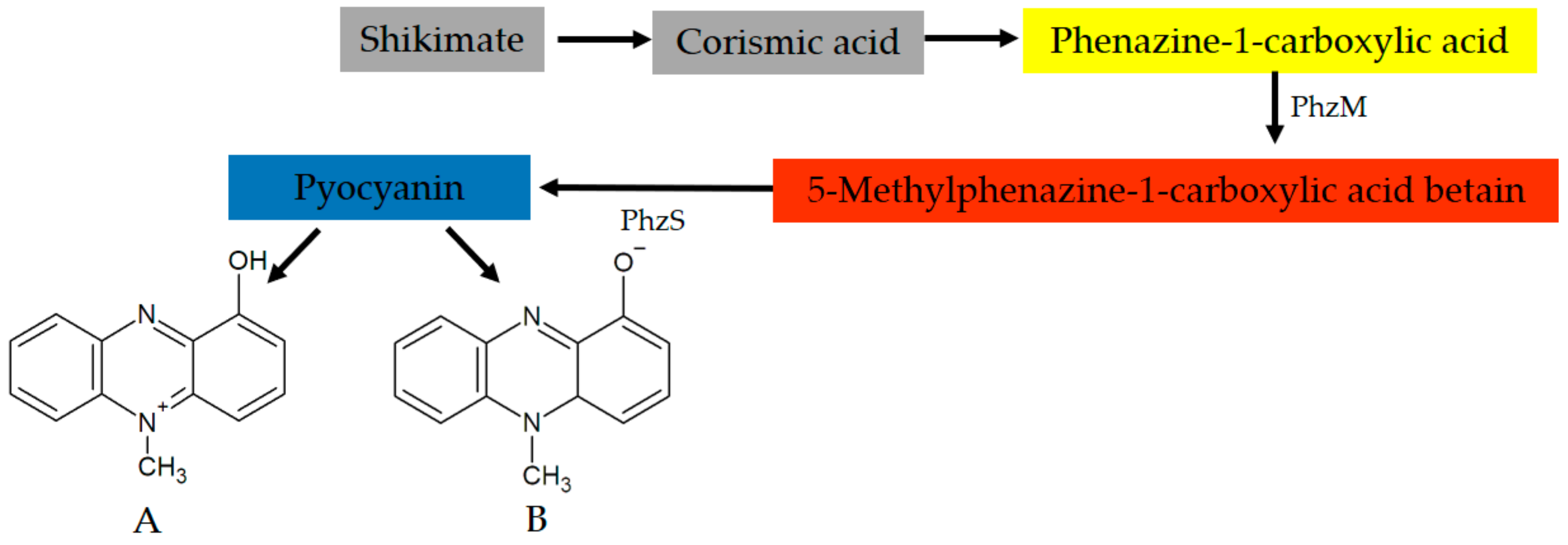
4. Pyocyanin: Fundamentals and General Properties of the Molecule
5. Modulation and Regulation of Pyocyanin Production
6. Mechanism of Action of Pyocyanin
7. Benefits of Pyocyanin for Pseudomonas aeruginosa
8. Mechanisms of Resistance of Pseudomonas aeruginosa to Pyocyanin
9. Production of Pyocyanin on a Laboratory Scale
10. Pyocyanin Applications and Perspectives
11. Conclusions
Funding
Acknowledgments
Conflicts of Interest
References
- Chadni, Z.; Rahaman, M.H.; Jerin, I.; Hoque, K.M.F.; Reza, M.A. Extraction and optimization of red pigment production as secondary metabolites from Talaromyces verruculosus and its potential use in textile industries. Micology 2017, 8, 48–57. [Google Scholar] [CrossRef]
- Kurbanoglu, E.B.; Ozdal, M.; Ozdal, O.G.; Algur, O.F. Enhanced production of prodigiosin by Serratia marcescens MO-1 using ram horn peptone. Braz. J. Microbiol. 2015, 46, 631–637. [Google Scholar] [CrossRef] [PubMed]
- Hajjaj, H.; Blanc, P.J.; Goma, G.; François, J. Sampling techniques and comparative extraction procedures for quantitative determination of intra- and extracellular metabolites in filamentous fungi. FEMS Microbiol. Lett. 1998, 164, 195–200. [Google Scholar] [CrossRef]
- Nowicka, B.; Kruk, J. Powered by light: Phototrophy and photosynthesis in prokariotes and its evolution. Microbiol. Res. 2016, 186–187, 99–118. [Google Scholar] [CrossRef] [PubMed]
- Moeller, R.; Horneck, G.; Facius, R.; Stackebrandt, E. Role of pigmentation in protecting Bacillus sp. endospores against environmental UV radiation. FEMS Microbiol. Ecol. 2005, 51, 231–236. [Google Scholar] [CrossRef] [PubMed]
- Cornelis, P.; Dingemans, J. Pseudomonas aeruginosa adapts its iron uptake strategies in function of the type of infections. Front. Cell Infect. Microbiol. 2013, 7. [Google Scholar] [CrossRef]
- Viana, A.A.G.; Martins, R.X.; Ferreira, G.F.; Zenaide-Neto, H.; Amaral, I.P.G.; Vasconcelos, U. Pseudomonas aeruginosa and pyocyanin negatively act on the establishment of Enterobacteriaceae biofilm on a ceramic surface. IJERA 2017, 7, 23–30. [Google Scholar]
- Liu, G.Y.; Nizet, V. Color me bad: Microbial pigments as virulence factors. Trends Microbiol. 2009, 17, 406–413. [Google Scholar] [CrossRef]
- Pierson, L.S.; Pierson, E.A. Metabolism and function of phenazines in bacteria: Impacts on the behaviour of bacteria in the environment and biotechnological process. Appl. Microbiol. Biotechnolol. 2010, 86, 1659–1670. [Google Scholar] [CrossRef]
- Shanmugaiah, V.; Mathivanan, N.; Varghese, B. Purification, crystal structure and antimicrobial activity of phenazine-1-carboxamide produced by a growth-promoting biocontrol bacterium, Pseudomonas aeruginosa MML2212. J. Appl. Microbiol. 2010, 108, 703–711. [Google Scholar] [CrossRef]
- Mavrodi, D.; Bonsall, R.F.; Delaney, S.M.; Soule, M.J.; Phillips, G.; Thomashow, L.S. Functional analysis of genes for biosynthesis of pyocyanin and phenazine-1-carboxamide from Pseudomonas aeruginosa PAO1. J. Bacteriol. 2001, 183, 6454–6465. [Google Scholar] [CrossRef] [PubMed]
- Usman, H.M.; Abdulkadir, N.; Gani, M.; Maiturare, H.M. Bacterial pigments and its significance. MOJ Bioequiv. Availab. 2017, 4, 285–288. [Google Scholar]
- Venil, C.K.; Zakaria, Z.A.; Ahmad, W.A. Bacterial pigments and their applications. Process. Biochem. 2013, 48, 1065–1079. [Google Scholar] [CrossRef]
- Young, G. Pigment production and antibiotic activity in cultures of Pseudomonas aeruginosa. J. Bacteriol. 1947, 54, 109–117. [Google Scholar] [CrossRef]
- Kurashi, M. Studies on the biosynthesis of pyocyanine (I): On the cultural condition for pyocyanine formation. Bull. Inst. Chem. Res. Kyoto Univ. 1958, 36, 163–173. [Google Scholar]
- Stanley, M.M. Bacillus pyocyaneus infections: A review, report of cases and discussion of newer therapy including streptomycin (concluded). Am. J. Med. 1947, 2, 347–367. [Google Scholar] [CrossRef]
- Turner, J.M.; Messenger, A.J. Occurrence, biochemistry and physiology of phenazine pigment production. Adv. Microbiol. Physiol. 1986, 27, 211–275. [Google Scholar]
- Gaby, W.L. A study of the dissociative bahavior of Pseudomonas aeruginosa. J. Bacteriol. 1946, 51, 217–234. [Google Scholar] [CrossRef]
- Schoental, R. The nature of the antibacterial agents present in Pseudomonas pyocyanea cultures. Br. J. Exp. Pathol. 1941, 22, 137–147. [Google Scholar]
- Waksman, S.A. History of the world “antibiotic”. J. Hist. Med. Allied Sci. 1973, 23, 284–286. [Google Scholar] [CrossRef]
- Waksman, S.A.; Woodruff, H.B. Selective antibiotic action of various substances of microbial origin. J. Bacteriol. 1942, 44, 373–384. [Google Scholar] [CrossRef] [PubMed]
- Tavares, W. Antibióticos e Quimioterápicos para o Clínico, 3rd ed.; Atheneu: São Paulo, Brasil, 2014; pp. 1–10. [Google Scholar]
- Burkholder, P.R. Cooperation and conflict among primitive organisms. Am. Sci. 1952, 40, 601–631. [Google Scholar]
- Gould, K. Antibiotics: From prehistory to the present day. J. Antimicrob. Chemother. 2016, 71, 572–575. [Google Scholar] [CrossRef] [PubMed]
- Leisinger, T.; Margraff, R. Secondary metabolites of the fluorescent pseudomonads. Microbiol. Rev. 1979, 43, 422–442. [Google Scholar] [CrossRef] [PubMed]
- El Feghali, P.A.R.; Nawas, T. Extraction and purification of pyocyanin: A simpler and more reliable method. MOJ Toxicol. 2018, 4, 417–422. [Google Scholar]
- Bodey, G.P.; Bolivar, R.; Fainstein, V.; Jadeja, L. Infections caused by Pseudomonas aeruginosa. Rev. Infect. Dis. 1983, 5, 279–313. [Google Scholar] [CrossRef] [PubMed]
- Hays, E.E.; Wells, I.C.; Katzman, P.A.; Cain, C.K.; Jacobs, F.A.; Thayer, S.A.; Doisy, E.A.; Gaby, W.L.; Roberts, E.C.; Muir, R.D.; et al. Antibiotic substances produced by Pseudomonas aeruginosa. J. Biol. Chem. 1941, 159, 725–750. [Google Scholar]
- Flood, M.E.; Herbert, R.B.; Holliman, F.G. Biosynthesis of pyocyanin, a phenazine microbial metabolite. Chem. Commun. 1970, 22, 1514–1515. [Google Scholar] [CrossRef]
- Whooley, M.A.; McLoughlin, A.J. The regulation of pyocyanin production in Pseudomonas aeruginosa. Appl. Microbiol. Biotechnol. 1982, 15, 161–166. [Google Scholar] [CrossRef]
- Tamagnini, L.M.; Gonzáles, R.D. Bacteriological stability and growth kinetics of Pseudomonas aeruginosa in bottled water. J. Appl. Microbiol. 1997, 83, 91–94. [Google Scholar] [CrossRef][Green Version]
- Hassan, H.; Fridovich, I. Mechanism of the antibiotic action of pyocyanine. J. Bacteriol. 1980, 141, 156–163. [Google Scholar] [CrossRef] [PubMed]
- Tyc, O.; Song, C.; Dickschat, J.S.; Vos, M.; Garbeva, P. The ecological role of volatile and soluble secondary metabolites produced by soil bacteria. Trends Microbiol. 2017, 25, 280–292. [Google Scholar] [CrossRef] [PubMed]
- Barakat, K.M.; Mattarm, M.Z.; Sabae, S.Z.; Darwesh, O.M.; Hassan, S.H. production and characterization of bioactive pyocyanin pigment by marine Pseudomonas aeruginosa OSh1. Res. J. Pharm. Biol. Chem. Sci. 2015, 6, 933–943. [Google Scholar]
- Vasconcelos, U.; Lima, M.A.G.A.; Calazans, G.M.T. Pseudomonas aeruginosa associated with negative interactions on coliform bacteria growth. Can. J. Pure Appl. Sci. 2010, 4, 1133–1139. [Google Scholar]
- Banning, N.; Toze, S.; Mee, B.J. Persistence of biofilm-associated Escherichia coli and Pseudomonas aeruginosa in groundwater and treated effluent in a laboratory model system. Microbiology 2003, 149, 47–55. [Google Scholar] [CrossRef][Green Version]
- Dietrich, L.E.P.; Price-Whelan, A.; Petersen, A.; Whiteley, M.; Nwman, D.K. The phenazine pyocyanin is a terminal signalling factor in the quorum sensing network of Pseudomonas aeruginosa. Mol. Microbiol. 2006, 61, 1308–1321. [Google Scholar] [CrossRef]
- Denyer, S.P. Mechanisms of action of antibacterial biocides. Int. Biodeterior. Biodegrad. 1995, 36, 227–245. [Google Scholar] [CrossRef]
- Kanthakumar, K.; Taylor, G.; Tang, K.H.T.; Cundell, D.R.; Rutman, A.; Smith, S.; Jeffery, P.K.; Cole, P.J.; Wilson, R. Mechanisms of action of Pseudomonas aeruginosa pyocyanin in human ciliary beat in vitro. Infect. Immun. 1993, 61, 2848–2853. [Google Scholar] [CrossRef]
- Huang, J.; Xu, Y.; Zhang, H.; Li, Y.; Huang, X.; Ren, B.; Zhang, X. Temperature-depenent expression of phzM and its regulatory genes lasI and ptsP in rhizosphere isolate Pseudomonas sp. strain M18. Appl. Environ. Mirobiol. 2009, 75, 6568–6580. [Google Scholar] [CrossRef]
- Lopes, L.C.; Davis II, E.W.; Silva, M.C.P.; Weisberg, A.J.; Bresciani, L.; Chang, J.H.; Loper, J.R.; Andreote, F.D. Tropical soils are reservoir for fluorescent Pseudomonas spp. biodiversity. Environ. Microbiol. 2018, 20, 62–74. [Google Scholar] [CrossRef]
- Jabur, M.H. Study of some virulence factors of Pseudomonas aeruginosa isolated from different clinical sources. Mag. Al Kufa Univ. Biol. 2014, 6, 227–233. [Google Scholar]
- Abdull-Hussein, Z.R.; Atia, S.S. Antimicrobial effect of pyocyanin from Pseudomonas aeruginosa. Eur. J. Exp. Biol. 2016, 6, 1–4. [Google Scholar]
- Maia, A.A.; Cantisani, M.L.; Esposto, E.M.; Silva, W.C.P.; Rodrigues, E.C.P.; Rodrigues, D.P.; Lázaro, N.S. Resistência antimicrobiana de Pseudomonas aeruginosa isolados de pescado e de cortes e de miúdos de frango. Ciênc. Tecnol. Aliment. 2009, 29, 114–119. [Google Scholar] [CrossRef][Green Version]
- Pirnay, J.P.; Matthijs, S.; Colak, H.; Chablain, P.; Bilocq, F.; Van Eldere, J.; De Vos, D.; Zizi, M.; Triest, L.; Cornelis, P. Global Pseudomonas aeruginosa biodiversity as reflected in a Belgian river. Environ. Microbiol. 2005, 7, 969–980. [Google Scholar] [CrossRef]
- Moayedi, A.; Nowroozi, J.; Sepahy, A.A. Effect of fetal and adult bovine serum on pyocyanin production in Pseudomonas aeruginosa isolated from clinical and soil samples. Iran. J. Basic Med. Sci. 2017, 20, 1331–1338. [Google Scholar] [CrossRef]
- Scott-Thomas, A.; Syhre, M.; Pattemorel, P.K.; Epton, M.; Laing, R.; Pearson, J.; Chambers, S.T. 2-Aminoacetophenone as a potential breath biomarker for Pseudomonas aeruginosa in the cystic fibrosis lung. BMC Pulm. Med. 2010, 10, 56. [Google Scholar] [CrossRef]
- APHA; AWWA; WEF. Standard Methods for the Examination of Water and Wastewater, 22nd ed.; APHA; AWWA; WEF: Baltimore, MD, USA, 2012; pp. 9–49. [Google Scholar]
- Al-Araji, M.K.; Ali, S. 2-Aminoacetophenone as a virulent factor for Pseudomonas aeruginosa causing sever burn and wound infections in Iraq. Ibn Al Haitham J. Pure Appl. Sci. 2012, 25, 88–97. [Google Scholar]
- Winstanley, C.; O’brien, S.; Brockhurst, M.A. Pseudomonas aeruginosa evolutionary adaptation and diversification in cystic fibrosis chronic lung infections. Trends Microbiol. 2016, 24, 327–337. [Google Scholar] [CrossRef]
- Chieda, Y.; Iiyama, K.; Lee, J.M.; Kusakabe, T.; Yasunaga-Aoki, C.; Shimizu, S. Inactivation of pyocyanin synthesis genes has no effect on the virulence of Pseudomonas aeruginosa PAO1 toward the silkworm, Bombyx mori. FEMS Microbiol. Lett. 2008, 278, 101–107. [Google Scholar] [CrossRef]
- Allydice-Francis, K.; Brown, P.D. Diversity of antimicrobial resistance and virulence determinants in Pseudomonas aeruginosa associated with fresh vegetables. Int. J. Microbiol. 2012. [Google Scholar] [CrossRef]
- Yin, Y.; Papavasiliou, G.; Zaborina, O.Y.; Alverdy, J.C.; Teymour, F. De novo synthesis and functional analysis of polyphosphate-loaded poly(ethylene) glycol hydrogel nanoparticles targeting pyocyanin and pyoverdin production in Pseudomonas aeruginosa as a model intestinal pathogen. Ann. Biomed. Eng. 2017, 45, 1058–1068. [Google Scholar] [CrossRef] [PubMed]
- Ferguson, D.; Cahill, O.J.; Quilty, B. Phenotypic, molecular and antibiotic resistance profiling of nosocomial Pseudomonas aeruginosa strain isolated from two Irish hospitals. J. Med. Biol. Sci. 2017, 1, 1–15. [Google Scholar]
- Abu, E.A.; Su, S.; Sallans, L.; Boissy, R.E.; Greatens, A.; Heineman, W.R.; Hassett, D.J. Cyclic voltammetric, fluorescence and biological analysis of purified aeruginosin A, a secreted red pigment of Pseudomonas aeruginosa PAO1. Microbiology 2013, 159, 1736–1747. [Google Scholar] [CrossRef] [PubMed]
- Finalayson, E.A.; Brown, P.D. Comparison of antibiotic resistance and virulence factors in pigmented and non-pigmented Pseudomonas aeruginosa. West Indian Med. J. 2011, 60, 24–32. [Google Scholar]
- Guttenberger, N.; Schlatzer, T.; Leypold, M.; Tassoti, S.; Breinbauer, R. Synthesis of novel ligands targeting phenazine biosynthesis proteins as a strategy for antibiotic intervention. Mon. Chem. 2018, 149, 847–856. [Google Scholar] [CrossRef] [PubMed]
- Legani, P.; Leoni, E.; Rapuano, S.; Turin, D.; Valenti, C. Survival and growth of Pseudomonas aeruginosa in natural mineral water: A 5-year study. Int. J. Food Microbiol. 1999, 53, 153–158. [Google Scholar] [CrossRef]
- Emerenini, B.O.; Hense, B.A.; Kuttler, C.; Eberl, H.J. A mathematical model of quorum sensing induced biofilm detachment. PLoS ONE 2015, 10, e0132385. [Google Scholar] [CrossRef]
- Santos, A.L.S.; Galdino, A.C.M.; Mello, T.P.; Ramos, L.S.; Branquinha, M.H.; Bolognese, A.M.; Columbano Neto, J.; Roudbary, M. What are the advantages of living in a community? A microbial biofilm perspective! Mem. Inst. Oswaldo Cruz 2018, 113, 1–7. [Google Scholar] [CrossRef]
- Vasconcelos, U.; Das, P.; Dias, D.S.B.; Bonifácio, T.T.C.; Arruda, R.R.A.; Oliveira, B.T.M.; Cavalcanti, T.G. Microbial Biofilms Current Research and Practical Implications; Caister Academic Press: Wymondham, UK, 2020; pp. 75–98. [Google Scholar] [CrossRef]
- Mendis, N.; Lin, Y.R.; Faucher, S.P. Comparison of virulence properties of Pseudomonas aeruginosa expoewd to water and grown rich broth. Can. J. Microbiol. 2014, 60, 777–781. [Google Scholar] [CrossRef]
- Fux, C.A.; Costerton, W.J.; Stewart, P.S.; Stooley, P. Survival strategies of infectious biofilms. Trends Microbiol. 2015, 13, 34–40. [Google Scholar] [CrossRef]
- Levipan, H.A.; Avendaño-Herrera, R. Different phenotypes of mature biofilm in Flavobacterium psychrophilum share a potential for virulence that differs from planktonic state. Front. Cell Infect. Microbiol. 2017, 15, 76. [Google Scholar] [CrossRef] [PubMed]
- Toyofuko, M.; Inaba, T.; Kiyokawa, T.; Obana, N.; Yawata, Y.; Nomura, N. Environmental factors that shape biofilm formation. Biosci. Biotechnol. Biochem. 2015, 80, 7–12. [Google Scholar] [CrossRef] [PubMed]
- Klausen, M.; Aaes-Jørgensen, A.; Molin, S.; Tolker-Nielsen, T. Involvement of bacterial migration in the development of complex multicellular structures in Pseudomonas aeruginosa biofilms. Mol. Microbiol. 2003, 50, 61–68. [Google Scholar] [CrossRef] [PubMed]
- Muller, M. Glutathione modulates the toxicity of, but is not a biologically relevant reductant for, the Pseudomonas aeruginosa redox toxin pyocyanin. Free Radic. Biol. Med. 2011, 50, 971–977. [Google Scholar] [CrossRef] [PubMed]
- Hall, S.; McDermott, C.; Anoopkumar-Dukie, S.; McFarland, A.J.; Forbes, A.; Perkins, A.V.; Davey, A.K.; Chess-Williams, R.; Kiefel, M.J.; Arora, D.; et al. Cellular effects of pyocyanin, a secreted virulence factor of Pseudomonas aeruginosa. Toxins 2016, 8, 823. [Google Scholar] [CrossRef] [PubMed]
- El-Fouly, M.Z.; Sharaf, A.M.; Shain, A.A.M.; El-Bialy, H.A.; Omara, A.M.A. Biosynthesis of pyocyanin pigment by Pseudomonas aeruginosa. J. Radic. Res. Appl. Sci. 2015, 8, 36–48. [Google Scholar]
- Özcan, D.; Kahraman, H. Pyocyanin production in the presence of calcium ion in Pseudomonas aeruginosa and recombinant bacteria. Turk. J. Sci. Technol. 2015, 10, 13–19. [Google Scholar]
- Hunter, R.C.; Vanja, K.; Magen, M.L.; Hannah, G.; Thomas, R.M.; Dianne, K.N. Phenazine content in the cystic fibrosis respiratory tract negatively correlates with lung function and microbial complexity. Am. J. Respir. Cell Mol. Biol. 2012, 47, 738–745. [Google Scholar] [CrossRef]
- Sterritt, O.W.; Lang, E.J.M.; Kessans, S.A.; Ryan, T.M.; Demeler, B.; Jameson, G.B.; Parker, E.J. Structural and functional characterisation of the entry point to pyocyanin biosynthesis in Pseudomonas aeruginosa defines a new 3-deoxy-d-arabino-heptulosonate 7-phosphate synthase subclass. Biosci. Rep. 2018, 38, BSR20181605. [Google Scholar] [CrossRef]
- Jayaseelan, S.; Ramaswamy, D.; Dharmaraj, S. Pyocyanin: Production, applications, challenges and new insights. World J. Mirobiol. Biotechnol. 2014, 30, 1159–1168. [Google Scholar] [CrossRef]
- Machan, Z.A.; Pitt, T.L.; White, W.; Watson, D.; Taylor, G.W.; Cole, P.J.; Wilson, R. Interaction between Pseudomonas aeruginosa and Staphylococcus aureus: Description of an antistaphylococcal substance. J. Med. Microbiol. 1991, 34, 213–217. [Google Scholar] [CrossRef] [PubMed]
- Denning, G.M.; Iyer, S.S.; Reszka, K.J.; O’Maley, Y.; Rasmussen, G.T.; Britigan, B.E. Phenazine-1-carboxylic acid, a secondary metabolite of Pseudomonas aeruginosa, alters expression of immunomodulatory proteins by human airway epithelial cells. Am. J. Physiol. Lung Cell Mol. Physiol. 2003, 285, L584–L592. [Google Scholar] [CrossRef] [PubMed]
- Greenhagen, B.T.; Shi, K.; Robinson, H.; Gamage, S.; Bera, A.K.; Ladner, J.E.; Parsons, J.F. Crystal structure of the pyocyanin biosynthetic protein PhzS. Biochemistry 2008, 47, 5281–5289. [Google Scholar] [CrossRef] [PubMed]
- Parsons, J.F.; Greenhagen, B.T.; Shi, K.; Calabrese, K.; Robinson, H.; Ladner, J.E. Structural and functional analysis of the pyocyanin biosynthetic protein PhzM from Pseudomonas aeruginosa. Biochemistry 2007, 46, 1821–1828. [Google Scholar] [CrossRef] [PubMed]
- Mavrodi, D.V.; Peever, T.L.; Mavrodi, O.V.; Parejko, J.A.; Raaijmakers, J.M.; Lemanceaus, P.; Mazurier, S.; Heide, L.; Blankenfeldt, W.; Weller, D.M.; et al. Diversity and evolution of the phenazine biosynthesis pathway. Appl. Environ. Microbiol. 2010, 76, 866–879. [Google Scholar] [CrossRef] [PubMed]
- Mazzola, M.; Cook, R.J.; Thomashow, L.S.; Weler, D.M.; Person III, L.S. Conribution of phenazine antibiotic biosynthesis to the ecological competence of fluorescent pseudomonads in soil habitats. Appl. Environ. Microbiol. 1992, 58, 2616–2624. [Google Scholar] [CrossRef]
- Al-Shabib, N.A.; Husain, F.M.; Khan, R.A.; Khan, M.S.; Alam, M.Z.; Ansari, F.A.; Laeeq, S.; Zubair, M.; Shahzad, S.A.; Khan, J.M.; et al. Interference of phosphane copper (I) complexes of b-carboline with quorum sensing regulated virulence functions and biofilm in foodborne pathogenic bacteria: A first report. Saudi J. Biol. Sci. 2018, 26, 308–316. [Google Scholar] [CrossRef]
- Brackman, G.; Coenye, T. Quorum sensing inhibitors as anti-biofilm agents. Curr. Pharm. Des. 2015, 21, 5–11. [Google Scholar] [CrossRef]
- Hense, B.A.; Schuster, M. Core principles of bacterial autoinducer systems. Microbiol. Mol. Biol. Rev. 2015, 79, 153–169. [Google Scholar] [CrossRef]
- Samrot, A.V.; Syed Azeemullah, A.; Suhail Azharudeen, M.; Sree Smanvitha, K.; Shaya Sneha, J. Characterization of acyl homoserine lactone of pigment producing Effects of cultivation media components on biosurfactant and pigment production from Pseudomonas aeruginosa SU-3. Pharma Chem. 2016, 8, 74–79. [Google Scholar]
- Miller, L.C.; O’Loughlin, C.T.; Zhang, Z.; Siryaporn, A.; Silpe, J.E.; Bassler, B.L.; Semmelhack, M.F. Development of potent inhibitors of pyocyanin production in Effects of cultivation media components on biosurfactant and pigment production from Pseudomonas aeruginosa. J. Med. Chem. 2015, 58, 1298–1306. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Lin, W.; Xia, H.; Xu, S.; Li, Y.; Yao, H.; Bai, F.; Zhang, X.; Bai, Y.; Saris, P.; et al. Influence of ptsP gene on pyocyanin production in Pseudomonas aeruginosa. FEMS Microbiol. Lett. 2005, 253, 103–109. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Wally, H.; Miller, S.J.; Lu, C.-D. The multifaceted proteins MvaT and MvaU, members of the H-NS family, control arginine metabolism, pytocyanin synthesis and prophage activation in Pseudomonas aeruginosa PAO1. J. Bacteriol. 2009, 191, 6211–6218. [Google Scholar] [CrossRef] [PubMed]
- Das, T.; Kutty, S.K.; Kumar, N.; Manifield, M. Pyocyanin facilitates extracellular DNA binding to Pseudomonas aeruginosa influencing cell surface properties and aggregation. PLoS ONE 2013, 8, e58299. [Google Scholar] [CrossRef] [PubMed]
- Tjeerd van Rij, E.; Wesselink, M.; Chin-A-Woeng, T.F.C.; Bloebberg, G.V.; Lugtenerg, B.J.J. Influence of environmental conditions on the production of phenazine-1-carboxamide by Pseudomonas aeruginosa PCL 1391. Mol. Plant Microbe Interact. 2004, 17, 557–566. [Google Scholar]
- Mavrodi, D.V.; Blankenfeldt, W.; Thomashow, L.S. Phenazine compounds in fluorescent Pseudomonas spp. biosynthesis and regulation. Annu. Rev. Phytopathol. 2006, 44, 417–445. [Google Scholar] [CrossRef]
- Huang, J.; Sonnleitner, E.; Ren, B.; Xu, Y.; Haas, D. Catabolite repression control of pyocyanin biosynthesis at an intersection of primary and secondary metabolism in Pseudomonas aeruginosa. Appl. Environ. Microbiol. 2012, 78, 5016–5020. [Google Scholar] [CrossRef]
- Price-Whelan, A.; Dietrich, L.E.P.; Newman, D.K. Pyocyanin alters redox homeostasis and carbon flux through central metabolic pathways in Pseudomonas aeruginosa PA14. J. Bacteriol. 2007, 189, 6237–6381. [Google Scholar] [CrossRef]
- Iiyama, K.; Takahashi, E.; Lee, J.M.; Mon, H.; Morishita, M.; Kusakabe, T.; Yasunaga-Aoki, C. Alkaline protease contributes to pyocyanin production in Pseudomonas aeruginosa. FEMS Microbiol. Lett. 2017, 364. [Google Scholar] [CrossRef]
- Gao, L.; Zhang, Y.; Wang, Y.; Qiao, X.; Zi, J.; Chen, C.; Wan, Y. Reduction of PCN biosynthesis by NO in Effects of cultivation media components on biosurfactant and pigment production from Pseudomonas aeruginosa. Redox Biol. 2016, 8, 252–258. [Google Scholar] [CrossRef][Green Version]
- Aonofriesei, F.; Crâşmaru, M. Antibacterial activity of pyocyanin produced by some Pseudomonas strains isolated from seawater. In Annals of the University of Craiova, 2004, IX (XLV); University of Craiova: Craiova, Romania, 2004; pp. 121–126. [Google Scholar]
- Salih, S.S.; Mohammed, L.M.A. Antibbiotic action of pyocyanin on some pathogenic bacteria isolated from wond infection. Int. J. Adv. Res. 2017, 5, 1197–1201. [Google Scholar] [CrossRef]
- Karpagam, S.; Sudhakar, T.; Lakshmipathy, M. Microbiocidal response of pyocyanin produced by P. aeruginosa toward clinical isolates of fungi. Int. J. Pharm. Pharm. Sci. 2013, 5, 870–873. [Google Scholar]
- Lau, G.W.; Hassett, D.J.; Ran, H.; Kong, F. The role of pyocyanin in Pseudomonas aeruginosa infection. TRENS Mol. Med. 2004, 10, 599–606. [Google Scholar] [CrossRef] [PubMed]
- Baron, S.S.; Rowe, J.J. Antibiotic action of pyocyanin. Antimicrob. Agents Chemother. 1981, 20, 814–820. [Google Scholar] [CrossRef]
- Muller, M. Premature cellular senescence induced by pyocyanin, a redox-active Pseudomonas aeruginosa toxin. Free Radic. Biol. Med. 2006, 41, 1670–1677. [Google Scholar] [CrossRef]
- Reszka, K.J.; O’Malley, Y.; McCormick, M.L.; Denning, G.M.; Britigan, B.E. Oxidation of pyocyanin, a cytotoxic product from Pseudomonas aeruginosa, by microperoxidase 11 and hydrogen peroxide. Free Radic. Biol. Med. 2004, 36, 1148–1159. [Google Scholar] [CrossRef]
- Baron, S.S.; Terranova, G.; Rowe, J.J. Molecular mechanism of the antimicrobial action of pyocyanin. Curr. Microbiol. 1989, 18, 223–230. [Google Scholar] [CrossRef]
- Bonifácio, T.T.C.; Arruda, R.R.A.; Oliveira, B.T.M.; Silva, J.E.G.; Vasconcelos, U. Exposure to pyocyanin promotes cellular changes in Candida spp. Res. J. Pharm. Biol. Chem. Sci. 2020, 11, 111–119. [Google Scholar]
- Managò, A.; Becker, K.A.; Carpinteiro, A.; Wilker, B.; Soddemann, M.; Seitz, A.P.; Edwards, M.J.; Grassmé, H.; Szabò, I.; Gulbins, E. Pseudomonas aeruginosa pyocyanin induces eutrophil death via mitochondrial reactive oxygen species and mitochondrial acid sphingomyelinase. Antoxidants Redox Signal. 2015, 22, 1097–1110. [Google Scholar] [CrossRef]
- Kwon, D.H.; Cha, H.-J.; Lee, H.; Homg, S.-H.; Park, C.; Park, S.-H.; Kim, G.-Y.; Kim, S.; Kim, H.-S.; Hwang, H.-J.; et al. Protective effect of glutathione against oxidative stress-induced cytotoxicity in raw 264.7 macrophages through activating the nuclear factor erythroid 2-related factor-2/heme oxygenase-1 pathway. Antioxidants 2019, 8, 82. [Google Scholar] [CrossRef]
- Sudhakar, T.; Karpagam, S.; Jayavarthanan, R. Pyocyanin and its bacteriostatic effect towards common clinical pathogens. Int. J. Pharm. Tech. Res. 2013, 5, 1487–1492. [Google Scholar]
- Sudhakar, T.; Karpagam, S.; Premkumar, J. Biosynthesis, antibacterial activity of pyocyanin pigment produced by Pseudomonas aeruginosa SU1. J. Chem. Pharm. Res. 2015, 7, 921–924. [Google Scholar]
- Özyürek, S.B.; Gür, S.D.; Bilkay, I.S. Investigation of antimicrobial activity of pyocyanin produced by Pseudomonas aeruginosa strains isolated from different clinical aspecimens. Hacet. J. Biol. Chem. 2016, 44, 1–6. [Google Scholar]
- Agrawal, A.H.; Chauhan, P.B. Effect of cultivation media components on pyocyanin production and its application in antimicrobial property. Int. J. Curr. Adv. Res. 2016, 5, 829–833. [Google Scholar]
- Arruda, R.R.A.; Oliveira, B.T.M.; Bonifácio, T.T.C.; Morais, V.C.; Amaral, I.P.G.; Vasconcelos, U. Activity of two exometabolites produced by Escherichia coli on the synthesis of pyocyanin. Int. J. Adv. Eng. Res. Sci. 2019, 6, 267–271. [Google Scholar] [CrossRef]
- Jameel, Z.J.; Hussain, A.F.; Al-Mahdawi, M.A.; Alkerim, N.F.A.; Alrahman, E.S.A. Bioactivity of pyocyanin of Pseudomonas aeruginosa clinical isolates against a variety of human pathogenic bacteria and fungi species. Int. Arab. J. Antimicrob. Agents 2017, 7, 2. [Google Scholar] [CrossRef]
- Priyaja, P.; Jayesh, P.; Correya, N.S.; Sreelakshmi, B.; Sudheer, N.S.; Phillip, R.; Singh, I.S.B. Antagonistic effect of Pseudomonas aeruginosa isolates from various ecological niches in Vibrio species pathogenic to crustaceans. J. Coast. Life Med. 2014, 2, 76–84. [Google Scholar]
- Kerr, J.R.; Taylor, G.W.; Rutman, A.; Høiby, N.; Cole, P.J.; Wilson, R. Pseudomonas aeruginosa pyocyanin and 1-hydroxyphenazine inhibit fungal growth. J. Clin. Pathol. 1999, 52, 385–387. [Google Scholar] [CrossRef]
- Silva, J.E.G.; Bonifácio, T.T.C.; Arrruda, R.R.A.; Kretzschmar, E.A.M.; Vasconcelos, U. Atividade bioestática da piocianina sobre fungos filamentosos isolados de parede de concreto. Int. J. Dev. Res. 2020, 10, 35230–35264. [Google Scholar]
- Hassani, H.H.; Hasan, H.M.; Al-Saadi, A.; Ali, A.M.; Muhammad, M.H. A comparative study on cytotoxicity and apoptotic activity of pyocyanin produced by wild type and mutant strains of Pseudomonas aeruginosa. Eur. J. Exp. Biol. 2012, 2, 1389–1394. [Google Scholar]
- Cezaitliyan, B.; Vinayavekhin, N.; Grefell-Lee, D.; Yuen, G.J.; Saghatelian, A.; Ausubel, F.M. Identification of Pseudomonas aeruginosa phenazines that kill Caenorhabditis elegans. PLOS Pathog. 2013, 9, e1003101. [Google Scholar] [CrossRef]
- Ghoul, M.; Mitri, S. The ecology and evolution of microbial competition. Trends Microbiol. 2016, 24, 833–845. [Google Scholar] [CrossRef] [PubMed]
- Martins, V.V.; Macarenco, A.C.; Gradella, D.G.; Stehling, E.G. Antagonism between clinical and environmental isolates of Pseudomonas aeruginosa against coliforms. Water Supply 2014, 14, 99–106. [Google Scholar] [CrossRef]
- Faust, K.; Raes, J. Microbial interactions: From networks to models. Nat. Rev. Microbiol. 2012, 10. [Google Scholar] [CrossRef]
- Hotterbeekx, A.; Kumar-Singh, S.; Goossens, H.; Malhotra-Kumar, S. In vivo and in vitro interactions between Pseudomonas aeruginosa and Staphylococcus spp. Front. Cell Infect. Microbiol. 2017, 106. [Google Scholar] [CrossRef]
- Cox, C.D. Role of pyocyanin in the acquisition of iron from transferrin. Infect. Immun. 1986, 52, 263–270. [Google Scholar] [CrossRef]
- Nagler, M.; Insam, H.; Pietramellara, G.; Ascher-Jenull, J. Extracellular DNA in natural environments: Features, relevance and applications. Appl. Microbiol. Biotechnol. 2018, 102, 6343–6356. [Google Scholar] [CrossRef]
- Das, T.; Manefield, M. Pyocyanin promotes extracellular DNA release in Pseudomonas aeruginosa. PLoS ONE 2012, 7, e0046718. [Google Scholar] [CrossRef]
- Sismaet, H.J.; Webster, T.A.; Goluch, E.D. Up-regulating pyocyanin production by amino acid addition for early electrochemical identification of Pseudomonas aeruginosa. Analyst 2014, 139, 4241–4246. [Google Scholar] [CrossRef]
- Whitchurch, C.B.; Tolker-Nielsen, T.; Ragas, P.C.; Mattick, J.S. Extracellular DNA required for bacterial biofilm formation. Science 2002, 295, 1487. [Google Scholar] [CrossRef]
- Hussein, K.A.; Joo, J.H. heavy metal resistance of bacteria and its impact on the production of antioxidants enzymes. Afr. J. Microbiol. 2013, 7, 2288–2296. [Google Scholar]
- Wang, Y.; Wilks, J.C.; Danhorn, T.; Ramos, I.; Croal, L.; Newman, D.K. Phenazine-1-carboxylic acid promotes bacterial biofilm development via ferrous iron acquisition. J. Bacteriol. 2011, 193, 3606–3617. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.-H.; Yet-Pole, I.; Chiu, Y.-H.; Lin, C.-W. Enhancement of power generation by toluene biodegradation in a microbial fuel cell in the presence of pyocyanin. J. Taiwan Inst. Chem. Eng. 2014, 45, 2319–2324. [Google Scholar] [CrossRef]
- Muller, M.; Merrett, N.D. Pyocyanin production by Pseudomonas aeruginosa confers resistance to ionic silver. Antimicrob. Agents Chemother. 2014, 58, 5492–5499. [Google Scholar] [CrossRef]
- Sales-Neto, J.M.; Lima, E.A.; Cavalcante-Silva, L.H.A.; Vasconcelos, U.; Rodrigues-Mascarenhas, S. Anti-inflammatory potential of pyocyanin in LPS-stimulated murine macroiphages. Immunopharmacol. Immunotoxicol. 2019. [Google Scholar] [CrossRef]
- Rada, B.; Leto, T.L. Pyocyanin effects on respiratory epithelium: Relevance in Pseudomonas aeruginosa airway infections. Trends Microbiol. 2013, 21, 73–81. [Google Scholar] [CrossRef]
- Nowroozi, J.; Sepahi, A.A.; Rashnonejad, A. Pyocyanine biosynthetic genes in clinical and environmental isolates of Pseudomonas aeruginosa and detection of pyocyanine’s antimicrobial effects with and without colloidal silver nanoparticles. Cell J. 2012, 14, 7–18. [Google Scholar]
- Kamal, M.A.; Pambuk, C.I.A.; Husein, A.S. The virulence of pigmented and non-pigmented Pseudomonas aeruginosa in mice with antibiotics susceptibility. Int. J. Vaccines Vaccin 2016, 3. [Google Scholar] [CrossRef]
- Castañeda-Tamez, P.; Ramirez-Peris, J.; Pérez-Velázquez, J.; Kuttler, C.; Jalalimanesh, A.; Saucedo-Mora, M.A.; Jiménez-Cortés, J.G.; Maeda, T.; Gonzáles, Y.; Tomás, M.; et al. Pyocyanin restricts social cheating in Pseudomonas aeruginosa. Front. Microbiol. 2018, 9, 1348. [Google Scholar] [CrossRef]
- Hassett, D.J.; Charniga, L.; Bean, K.; Ohman, D.E.; Cohen, M.S. Response of Pseudomonas aeruginosa to pyocyanin: Mechanisms of resistence, antioxidant defences, and demonstration of a magnese-cofactored superoxide dismutase. Infect. Immun. 1992, 60, 328–336. [Google Scholar] [CrossRef]
- Cunningham, L.; Williams, H.D. Isolation and characterization of mutants defective in the cyanide-insensitive respiratory pathway of Pseudomonas aeruginosa. J. Bacteriol. 1995, 117, 432–438. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Pinhal, S.; Ropers, D.; Geiselmann, J.; De Jong, H. Acetate metabolism and the inhibition of bacterial growth by acetate. J. Bacteriol. 2019, 201. [Google Scholar] [CrossRef] [PubMed]
- Das, S.; Das, P. Effects of cultivation media components on biosurfactant and pigment production from Pseudomonas aeruginosa. Braz. J. Chem. Eng. 2015, 32, 317–324. [Google Scholar] [CrossRef][Green Version]
- Dantas, P.V.; Peres, S.; Campos-Takashi, G.M.; La Rotta, C.E. Utilization of raw glycerol for pyocyanin production from Pseudomonas aeruginosa in half-microbial fuel cells: Evaluation of two electrochemical approaches. J. Electrochem. Soc. 2013, 106, G142–G148. [Google Scholar] [CrossRef]
- Lundgren, B.R.; Thornton, W.; Dornan, M.H.; Villegas-Peñaranda, L.R.; Boddy, C.N.; Nomura, C.T. Gene PA2449 is essential for glycine metabolism and pyocyanin biosynthesis in Pseudomonas aeruginosa PAO1. J. Bacteriol. 2013, 195, 2087–2100. [Google Scholar] [CrossRef] [PubMed]
- Lenney, W.; Gilchrist, F.J. Pseudomonas aeruginosa and cyanie production. Eur. Respir. J. 2011, 37, 482–483. [Google Scholar] [CrossRef]
- Zlosnik, J.E.A.; Tavankar, G.R.; Bundy, J.G.; Mossialos, D.; O’toole, R.; Williams, H.D. Investigation of the physiological relationship between the cyanide-insensitive oxidase and cyanide production in Pseudomonas aeruginosa. Microbiology 2006, 125, 1407–1415. [Google Scholar] [CrossRef]
- King, E.O.; Ward, M.K.; Raney, D.E. Two simple media for the demonstration of pyocyanin and fluorescein. J. Lab. Clin. Med. 1954, 44, 301–307. [Google Scholar]
- Devnath, P.; Uddin, M.K.; Ahmed, F.; Hossain, M.T.; Manchur, M.A. Extraction, purification and characterization of pyocyanin produced by Pseudomonas aeruginosa and evaluation for its antimicrobial activity. Int. Res. J. Biol. Sci. 2017, 6, 1–7. [Google Scholar]
- Atzél, B.; Szoboszlay, S.; Mikuska, Z.; Kriszt, B. Comparison of phenotypic and genotypic methods for the detection of environmental isolates of Pseudomonas aeruginosa. Int. J. Hyg. Environ. Health 2008, 21, 143–155. [Google Scholar] [CrossRef]
- Brodsky, M.H.; Nixon, M.C. Rapid method for detection of Pseudomonas aeruginosa on MacConkey agar under ultraviolet light. Appl. Microbiol. 1973, 26, 219–220. [Google Scholar] [CrossRef] [PubMed]
- Mossel, D.A.A.; Indacochea, L. A new cetrimide medium for the detection of Pseudomonas aeruginosa. J. Med. Microbiol. 1971, 4, 380–382. [Google Scholar] [CrossRef] [PubMed]
- Viana, A.A.G.; Oliveira, B.T.M.; Cavalcanti, T.G.; Sousa, K.A.; Mendonça, E.A.M.; Amaral, I.P.G.; Vasconcelos, U. Correlation between pyocyanin production and hydrocabonoclactic activity in nine strains of Pseudomonas aeruginosa. Int. J. Adv. Eng. Res. Sci. 2018, 5, 212–223. [Google Scholar] [CrossRef]
- Elbargisy, R.M. Optimization of nutritional and environmental conditions for pyocyanin production by urine isolates of Pseudomonas aeruginosa. Saudi J. Biol. Sci. 2020. [Google Scholar] [CrossRef]
- Raina, S.; Murphy, T.; Vizio, D.; Reffatti, P.; Keshavarz, T. Novel strategies for over-production of microbial products. Chem. Eng. Trans. 2011, 24, 847–852. [Google Scholar]
- Thomson, N.R.; Crow, M.A.; McGowan, S.J.; Cox, A.; Salmond, G.P.C. Biosynthesis of carbapenem antibiotic and prodigiosin pigment in Serratia is under quorum sensing control. Mol. Microbiol. 2000, 36, 539–556. [Google Scholar] [CrossRef]
- McClean, K.H.; Winson, M.K.; Fish, L.; Taylor, A.; Chhabra, S.R.; Camara, M.; Daykin, M.; Lamb, J.H.; Swift, S.; Bycroft, B.W.; et al. Quorum sensing and Chromobacterium violaceum: Exploitation of violacein production and inhibition for the detection of N-acyl homoserine lactones. Microbiology 1997, 143, 3703–3711. [Google Scholar] [CrossRef]
- Van der Berg, J.P.; Velema, W.A.; Szymanski, W.; Driessen, J.M.; Feringa, B.L. Controlling the activity of quorum sensing autoinducers with light. Chem. Sci. 2015, 6, 3593–3598. [Google Scholar] [CrossRef]
- Moradali, M.F.; Ghods, S.; Rehm, B.H.A. Pseudomonas aeruginosa lifestyle: A paradigm for adaptation, survival, and persistence. Front. Cell Infect. Microbiol. 2017, 7. [Google Scholar] [CrossRef]
- Vipin, C.; Ashwini, P.; Kavya, A.V.; Rekha, P.D. Overproduction of pyocyanin in Pseudomonas aeruginosa by supplementation of pathway precursor shikimic acid and evaluation of its activity. Res. J. Pharm. Technol. 2017, 10, 533–536. [Google Scholar] [CrossRef]
- Oliveira, B.T.M.; Barbosa, P.S.Z.; Cavalcanti, T.G.; Amaral, I.P.G.; Vasconcelos, U. Craft beer waste as substrate for pyocyanin synthesis. J. Pharm. Biol. Sci. 2019, 14, 21–25. [Google Scholar]
- Cavalcanti, T.G.; Souza, A.F.; Ferreira, G.F.; Dias, D.S.B.; Severino, L.S.; Morais, J.P.S.; Sousa, K.A.; Vasconcelos, U. Use of agro-industrial waste in the removal os phenanthrene and pyrene by microbial consortia in soil. Waste Biomass Valorization 2017. [Google Scholar] [CrossRef]
- Das, P.; Ma, L.Z. Pyocyanin pigment assisting biosurfactant-mediated hydrocarbon emulsification. Int. Biodegrad. Biodeterior. 2013, 85, 278–283. [Google Scholar] [CrossRef]
- Pragana, L.G.; Silva, E.S.; Vasconcelos, U. Exploração do potencial da cafeína como ativo antibiofilme em Pseudomonas aeruginosa na década de 2010. Int. J. Dev. Res. 2020, 10, 37869–37874. [Google Scholar]
- Angell, S.; Bench, B.J.; Williams, H.; Wartanabe, C.M.H. Pyocyanin isolated from a marine microbial population: Synergic production between two distinct bacterial species and mode of action. Chem. Biol. 2006, 13, 1349–1359. [Google Scholar] [CrossRef] [PubMed]
- Méndez, D.A.C.; Gutierrez, E.; Lamarque, G.C.C.; Rizzato, V.L.; Buzalaf, M.A.R.; Machado, M.A.A.M.; Cruvinel, T. A eficácia da terapia fotodinâmica antimicrobiana mediada pela curcumina depende dos tempos de pré-irradiação e crescimento do biofilme. Fotodiagn. Ter. Fotodin. 2019, 27, 474–480. [Google Scholar]
- Norman, R.S.; Moeller, P.; McDonald, T.J.; Morris, P.J. Effect of pyocyanin on a crude-oil-degrading microbial community. Appl. Environ. Microbiol. 2004, 70, 4004–4011. [Google Scholar] [CrossRef]
- Rabaey, K.; Boon, N.; Höfte, M.; Verstrate, W. Microbial phenazine production enhances electron transfer in biofuels cells. Environ. Sci. Technol. 2005, 39, 3401–3408. [Google Scholar] [CrossRef]
- Fuse, K.; Fujimura, S.; Kikuchi, T.; Gomi, K.; Iida, Y.; Nukiwa, T.; Watanabe, A. Reduction of virulence factor pyocyanin production in multidrug-resistant Effects of cultivation media components on biosurfactant and pigment production from Pseudomonas aeruginosa. J. Infect. Chemother. 2013, 19, 82–88. [Google Scholar] [CrossRef]
- Gharieb, M.M.; El-Sheekh, M.M.; El-Sabbagh, S.; Hamza, W.T. Efficacy of pyocyanin produced by Pseudomonas aeruginosa as a topical treatment of infected skin of rabbits. Biotechnol. Indian J. 2013, 7, 184–193. [Google Scholar]
- Priyaja, P.; Jayesh, P.; Haseeb, M.; Jose, B.; Phillip, R.; Bright Singh, I.S. Evaluation of pyocyanin toxicity in various life stages of Penaeus monodon and in nitrifying bacterial consortia for their safe application in recirculating aquaculture systems (RAS) to abrogate pathogenic vibrios. Aquac. Int. 2016, 25, 743–753. [Google Scholar] [CrossRef]
- De Britto, S.; Gajbar, T.D.; Satapute, P.; Sundaram, L.; Lakshmikantha, R.Y.; Jogaiah, S.; Ito, S.-I. Isolation and characterization of nutrient dependent pyocyanin from Pseudomonas aeruginosa and its dye and agrochemical properties. Sci. Rep. 2020, 10, 1542. [Google Scholar] [CrossRef] [PubMed]
- Khare, E.; Arora, N.K. Dual activity of pyocyanin from Pseudomonas aeruginosa—Antibiotic against phytopathogen and signal molecule for biofilm development by rhizobia. Can. J. Microbiol. 2011, 57, 708–713. [Google Scholar] [CrossRef] [PubMed]
- Yu, D.; Yong, Y.-C.; Liu, C.; Fang, Y.; Bai, L.; Dong, S. New applications of genetically modified Pseudomonas aeruginosa for toxicity detection in water. Chemosphere 2017, 184, 106–111. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Xiao-Chang, C. Organic Light-Emitting Device Having Phenanthroline-Fused Phenazine. U.S. Patent 6,713,781, 2004. [Google Scholar]
- Rabaey, K.; Verstraete, W. Microbial fuel cells: Novel biotechnology for energy generation. Trends Biotechnol. 2005, 23, 291–298. [Google Scholar] [CrossRef]
- Ali, N.; Anam, M.; Yousaf, S.; Maleeha, S.; Bangash, Z. Characterization of the electric current generation potential of the Pseudomonas aeruginosa using glucose, fructose, and sucrose in double chamber Microbial Fuel Cell. Iran. J. Biotechnol. 2017, 15, 216–223. [Google Scholar] [CrossRef]
- Kumar, R.; Singh, L.; Zularisam, A.W. Exoelectrongens: Recent advances in molecular drivers involved in extracellular electron transfer and strategies used to improve it for microbial full cell applications. Renew. Sustain. Energy Rev. 2016, 56, 1322–1336. [Google Scholar] [CrossRef]
- Sismaet, H.J.; Pinto, A.J.; Goluch, E.D. Electrochemical sensors for identifying pyocyanin production in clinical Pseudomonas aeruginosa isolates. Biosens. Bioelectron. 2017, 97, 65–69. [Google Scholar] [CrossRef]
- Das, D.; Baruah, R.; Roy, A.S.; Singh, A.K.; Boruah, H.P.D.; Kalita, J.; Bora, T.C. Complete genome sequence analysis of Pseudomonas aeruginosa N002 reveals its genetic adaptation for crude oil degradation. Genomics 2014, 105, 182–190. [Google Scholar] [CrossRef]
- Vasconcelos, U.; de França, F.P.; Oliveria, F.J.S. Removal of high-molecular weight polycyclic aromatic hydrocarbons. Quim. Nova 2011, 34, 218–221. [Google Scholar] [CrossRef]
- Mortzfeld, F.B.; Pietruszka, J.; Baxendale, I.R. A simple and efficient flow preparation of pyocyanin a virulence factor of Pseudomonas aeruginosa. Eur. J. Org. Chem. 2019, 2019, 5424–5433. [Google Scholar] [CrossRef]
- Priyaja, P.; Jayesh, P.; Philip, R.; Singh, I.S.B. Pyocyanin induced in vitro oxidative damage and its toxicity level in human, fish and insect cell lines for its selective biological application. Cytotechnology 2016, 68, 143–155. [Google Scholar] [CrossRef] [PubMed]
- Chin-A-Woeng, T.F.C.; Bloemberg, G.V.; Lugtenberg, B.J.J. Phenazines and their role in biocontrol by Pseudomonas bacteria. New Phytol. 2003, 157, 503–523. [Google Scholar] [CrossRef]
| Organisms | References |
|---|---|
| Prokaryotes | |
| Bacillus sp. | [43,105,106,107] |
| Bacillus cereus | [108] |
| Bacillus megaterium | [108] |
| Enterobacter aerogenes | [35] |
| Escherichia coli | [7,34,35,43,95,105,106,107,108,109] |
| Klebsiella oxytoca | [95] |
| Proteus mirabilis | [95,105,106] |
| Pseudomonas sp. | [34] |
| Salmonella typhi | [108] |
| Shigella sp. | [110] |
| Staphylococcus aureus | [43,95,105,106,108] |
| Staphylococcus epidermidis | [110] |
| Xanthomonas oryzae | [10] |
| Vibrio sp. | [111] |
| Eukaryotes | |
| Alternaria sp. | [110] |
| Amoeba sp. | [94] |
| Aspergillus fumigatus | [112,113] |
| Aspergillus niger | [34,43,110,113] |
| Candida spp. | [34,43,102,107,110,114] |
| Candida albicans | [112] |
| Candida neoformans | [114] |
| Caenorhabditis elegans | [115] |
| Fusarium sp. | [34] |
| Penicillium sp. | [110,113] |
| Rhizoctonia solani | [10] |
| Rhizopus sp. | [110] |
| Rhodutorula sp. | [110] |
| Trichophyton sp. | [110] |
| Application | Reference |
|---|---|
| Clinical diagnosis | [123] |
| Bioremediation | [147,161] |
| Microbial fuel cells (MFCs) | [9,57,138,162,163] |
| Antibiotic | [164,165] |
| Agrochemical | [73,166,167] |
| Biosensor | [168] |
| Use in Organic Light Emitting Devices (OLED) | [169] |
| Probiotics | [111] |
| Antitumour | [154] |
| Biocolourant | [166] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gonçalves, T.; Vasconcelos, U. Colour Me Blue: The History and the Biotechnological Potential of Pyocyanin. Molecules 2021, 26, 927. https://doi.org/10.3390/molecules26040927
Gonçalves T, Vasconcelos U. Colour Me Blue: The History and the Biotechnological Potential of Pyocyanin. Molecules. 2021; 26(4):927. https://doi.org/10.3390/molecules26040927
Chicago/Turabian StyleGonçalves, Thiago, and Ulrich Vasconcelos. 2021. "Colour Me Blue: The History and the Biotechnological Potential of Pyocyanin" Molecules 26, no. 4: 927. https://doi.org/10.3390/molecules26040927
APA StyleGonçalves, T., & Vasconcelos, U. (2021). Colour Me Blue: The History and the Biotechnological Potential of Pyocyanin. Molecules, 26(4), 927. https://doi.org/10.3390/molecules26040927






