Chemical Proprieties of Biopolymers (Chitin/Chitosan) and Their Synergic Effects with Endophytic Bacillus Species: Unlimited Applications in Agriculture
Abstract
1. Introduction
2. Chitin and Chitosan
2.1. History of the Chitin and Chitosan’s Discovery
2.2. Origin of Biopolymers (Chitin/Chitosan)
2.3. Characteristic and Composition of the Crustacean Shells’ Waste
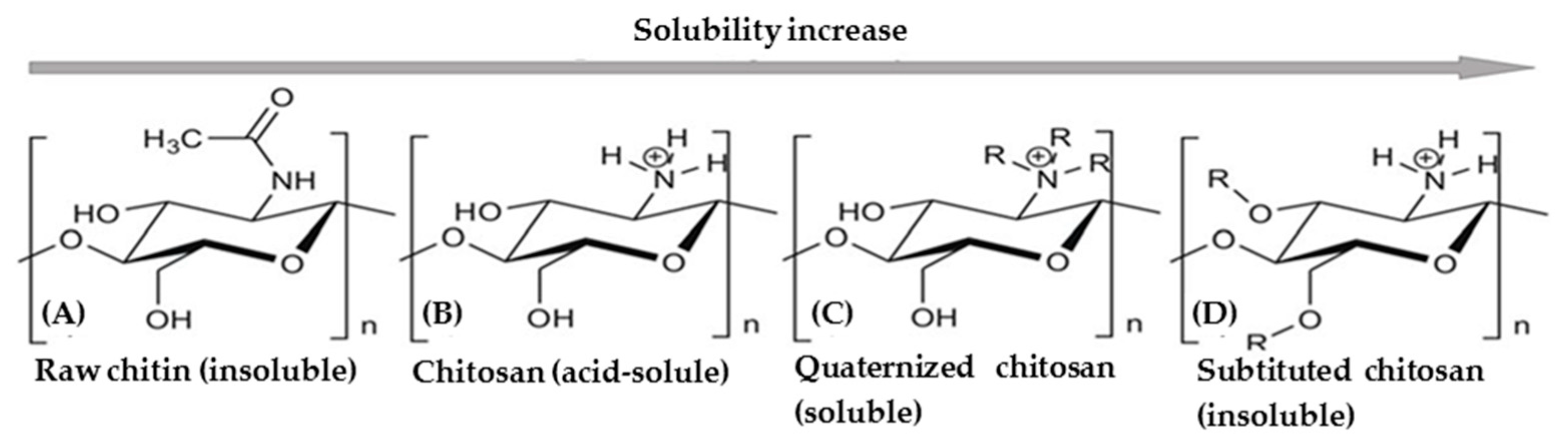
2.4. Structure of Chitin and Chitosan
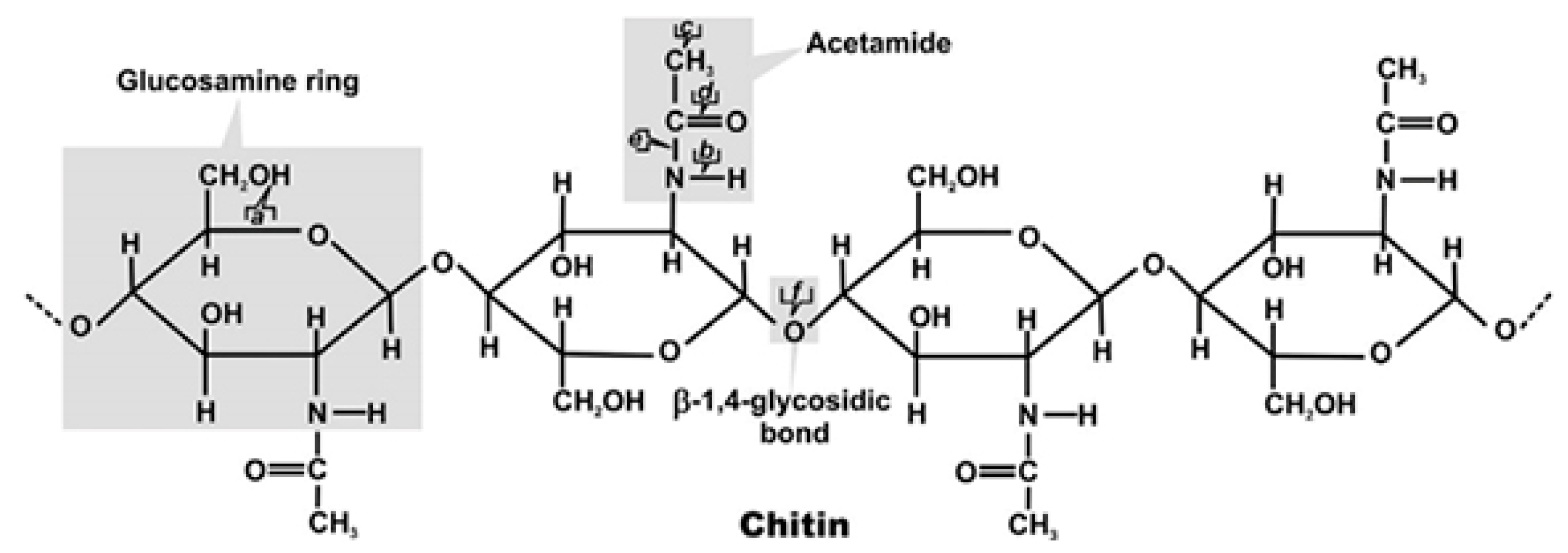
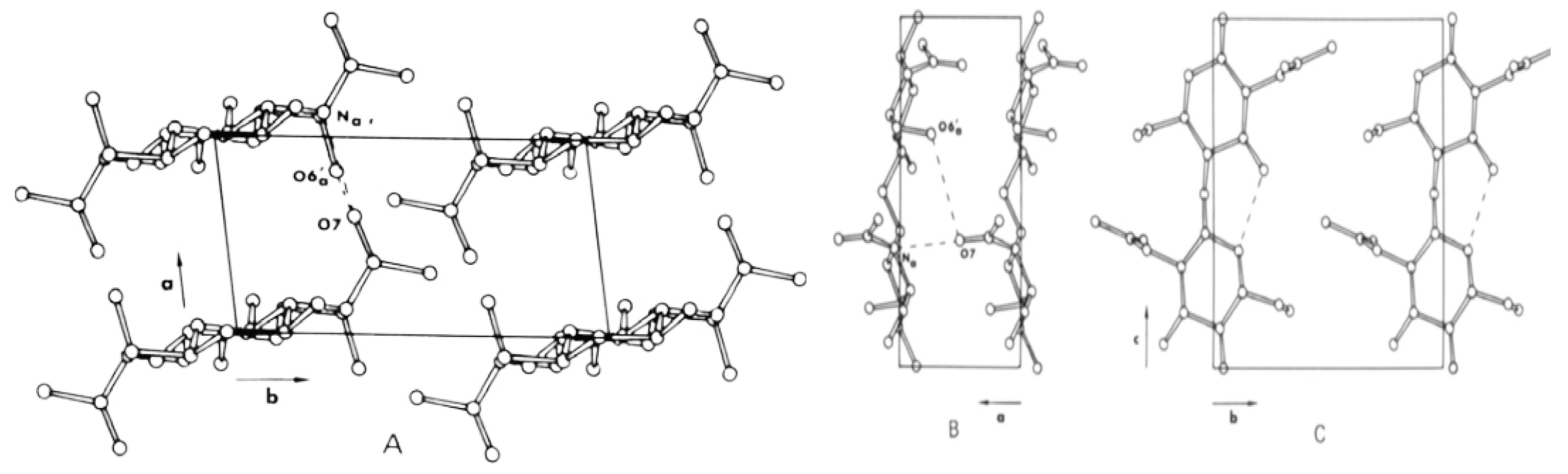
2.4.1. Chitin
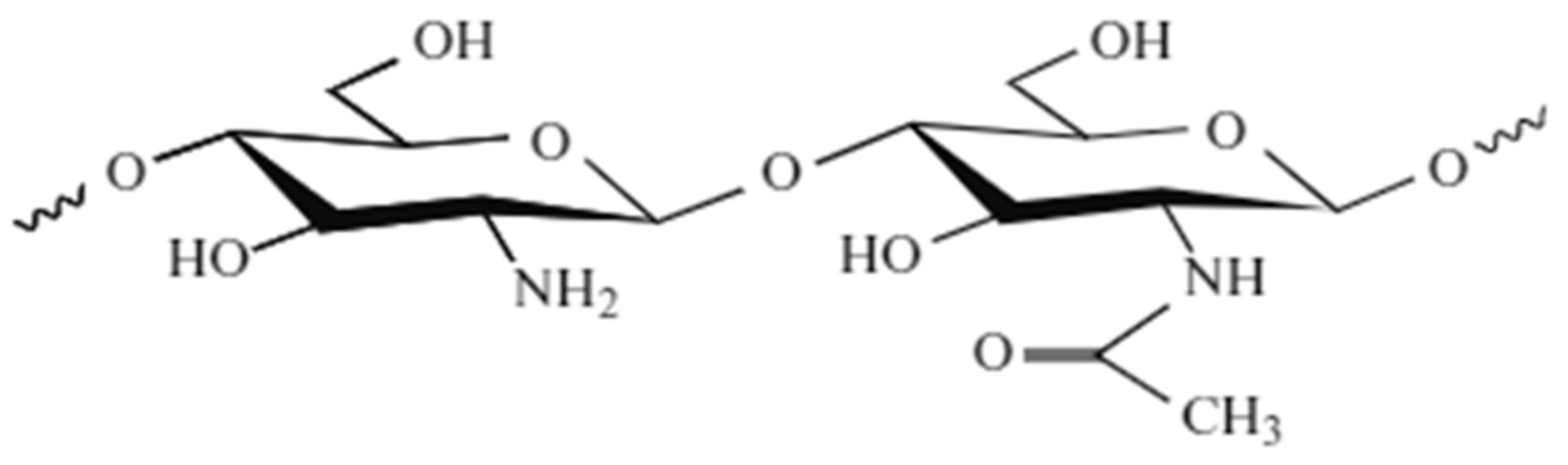
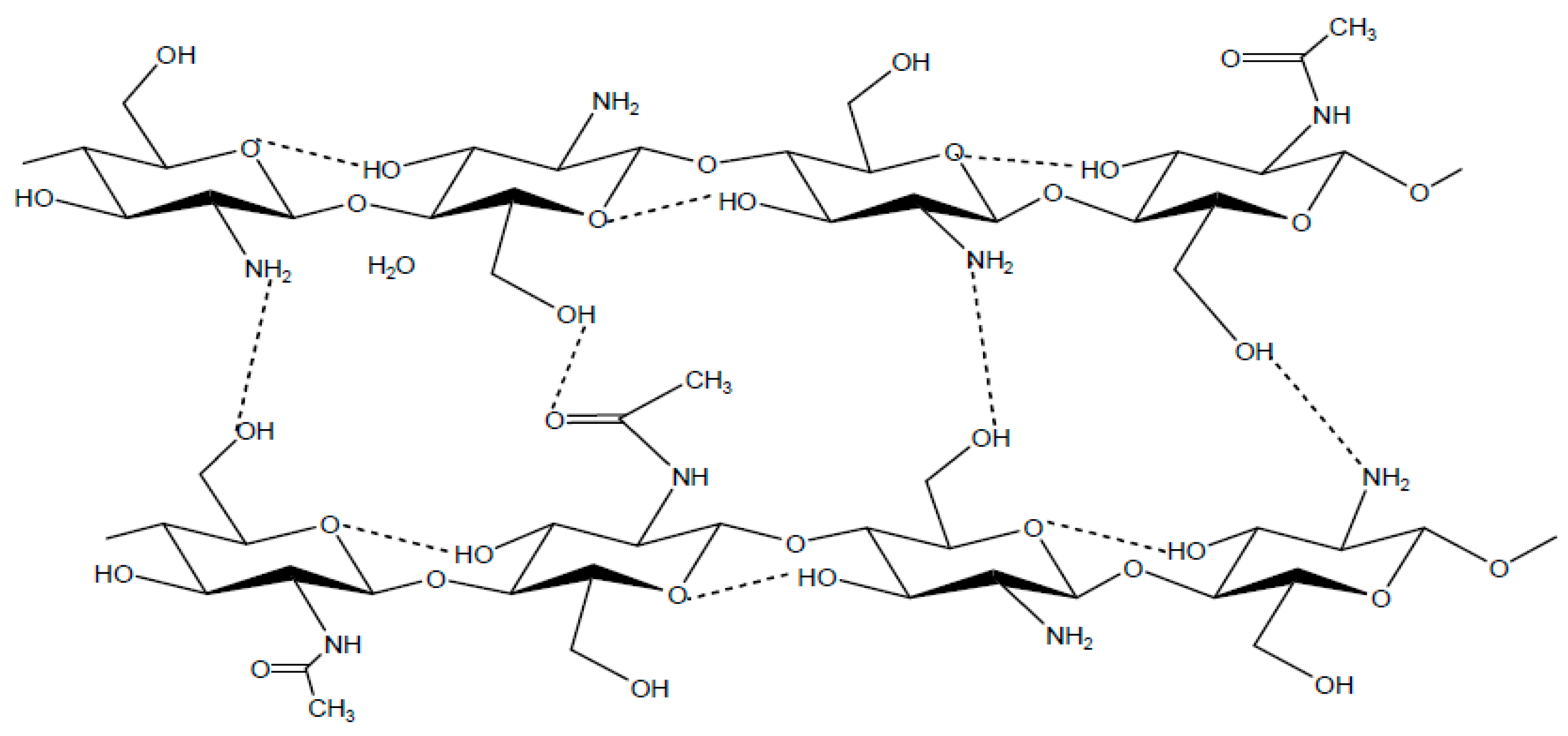
2.4.2. Chitosan:
2.4.3. Chemical Properties of Chitosan
2.5. Global Demand and Production of Chitin and Chitosan
2.6. Methods of Gauging Crustacean Coproducts:
2.6.1. Chemical Method
2.6.2. Biological Method
2.7. Application of Chitin
2.7.1. Applications in the Agricultural Sector
2.7.2. Chitin’s Effect on Crop Protection
2.7.3. Antifungal Activity of Chitin
- Direct action on the molecules of the fungus affecting their growth and development.
- Triggering the defense mechanisms that interfere with or hold back the development of pathogens, which consequently halt or limit the progression of the disease.
2.8. Applications of Chitosan
2.8.1. Antiviral Activity
2.8.2. Nematicidal Activity
2.8.3. Antioxidant Activity
2.8.4. Antifungal Activity
2.8.5. Antimicrobial Activity
2.8.6. Plant Growth Stimulating Activity
2.8.7. Stimulating Activity of the Plant’s Defense
2.8.8. Complex Associations of Metal Ions: Essential Role in Water Purification
3. Endophytic Bacteria
- Obligatory endophytes bank entirely on the host plant for their survival and growth. Their transmission to other hosts is carried out either vertically or by vectors.
- Facultative endophytes have a stage in their life cycle where they exist outside of the host plants. At the extremity, phytopathogenic bacteria could be included as endophytes (facultative or obligatory).
3.1. Endophytic Bacteria of the Genus Bacillus sp.
- Bacilli with nondeforming oval spore;
- Bacilli with oval deforming spore;
- Bacilli with round deforming spore.
3.2. Endophytic Bacteria and Growth Stimulation
3.2.1. Production of Indole-3-Acetic Acid (IAA) and Other Hormones
3.2.2. Improvement of Photosynthetic Activity
3.2.3. Regulation of Ethylene Levels by the Bacteria Producing ACC Deaminase
4. Chitin and Chitosan Degradation Enzymes
4.1. Chitinases
- Exochitinases, that only show activity for the nonreducing end of the chitin chain;
- Endochitinases, hydrolyzing the internal β-1, 4-glycoside;
- β-N-acetylglucosaminidase, that cleaves GlcNAc units sequentially from the nonreducing end of the substrate.
4.2. Families of Chitinases
- Chitinases (EC 3.2.1.14) that cleave the chitin chain at internal sites haphazardly. They are found in four families of glycoside hydrolases (GH) (18, 19, 23, and 48).
- β-N-acetylhexosaminidases (EC 3.2.1.52) that catalyze the respective deletion of GlcNAc (N-Acetylglucosamine) residues from the nonreducing end of the chain, and they are included in GH3, GH18, GH20, and GH84.
5. Synergistic Effect of Chitin/Chitosan and Endophytic Bacteria (Genus Bacillus sp.) on the Germination of Seeds, Growth and Fructification of Plants
6. Effects of Formulation Chitin/Chitosan and Bacillus sp. on Plant Protection
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kean, T.; Thanou, M. Biodegradation, biodistribution and toxicity of chitosan. Adv. Drug Deliv. Rev. 2010, 62, 3–11. [Google Scholar] [CrossRef] [PubMed]
- Dutta, P.K.; Dutta, J.; Tripathi, V.S. Chitin and chitosan: Chemistry, properties and applications. J. Sci. Ind. Res. 2004, 63, 20–31. [Google Scholar]
- Rinaudo, M. Chitin and chitosan: Properties and applications. Prog. Polym. Sci. 2006, 31, 603–632. [Google Scholar] [CrossRef]
- Wattjes, J.; Sreekumar, S.; Richter, C.; Cord-Landwehr, S.; Singh, R.; El Gueddari, N.E.; Moerschbacher, B.M. Patterns matter part 1: Chitosan polymers with non-random patterns of acetylation. React. Funct. Polym. 2020, 151, 104583. [Google Scholar] [CrossRef]
- Hassan, O.; Chang, T. Chitosan for eco-friendly control of plant disease. Asian J. Plant Pathol. 2017, 11, 53–70. [Google Scholar] [CrossRef]
- Orzali, L.; Corsi, B.; Forni, C.; Riccioni, L. Chitosan in agriculture: A new challenge for managing plant disease. In Biological Activities and Application of Marine Polysaccharides; InTechOpen: Croatia, Rijeka, 2017; pp. 17–36. [Google Scholar]
- Zhao, L.; Xu, Y.; Lai, X. Antagonistic endophytic bacteria associated with nodules of soybean (Glycine max L.) and plant growth-promoting properties. Braz. J. Microbiol. 2018, 49, 269–278. [Google Scholar] [CrossRef]
- Khamwan, S.; Boonlue, S.; Riddech, N.; Jogloy, S.; Mongkolthanaruk, W. Characterization of endophytic bacteria and their response to plant growth promotion in Helianthus tuberosus L. Biocatal. Agric. Biotechnol. 2018, 13, 153–159. [Google Scholar] [CrossRef]
- Hashem, A.; Tabassum, B.; Abd_Allah, E.F. Bacillus subtilis: A plant-growth promoting rhizobacterium that also impacts biotic stress. Saudi J. Biol. Sci. 2019, 26, 1291–1297. [Google Scholar] [CrossRef]
- Zhao, Q.; Ran, W.; Wang, H.; Li, X.; Shen, Q.; Shen, S.; Xu, Y. Biocontrol of Fusarium wilt disease in muskmelon with Bacillus subtilis Y-IVI. BioControl 2013, 58, 283–292. [Google Scholar] [CrossRef]
- Qin, Z.; Zhao, L. The History of Chito/Chitin Oligosaccharides and Its Monomer. In Oligosaccharides of Chitin and Chitosan; Springer: Singapore, 2019; pp. 3–14. ISBN 978-981-13-9402-7. [Google Scholar]
- Diario, F.; Rapanà, P.; Tomati, U.; Galli, E. Chitin and chitosan from Basidiomycetes. Int. J. Biol. Macromol. 2008, 43, 8–12. [Google Scholar]
- Tolaimate, A.; Desbrières, J.; Rhazi, M.; Alagui, A. Contribution to the preparation of chitins and chitosans with controlled physic-chemical properties. Polymer 2003, 44, 7939–7952. [Google Scholar] [CrossRef]
- Jones, M.; Kujundzic, M.; John, S.; Bismarck, A. Crab vs. Mushroom: A review of crustacean and fungal chitin in wound treatment. Mar. Drugs 2020, 18, 64. [Google Scholar] [CrossRef] [PubMed]
- Synowiecki, J.; Al-Khateeb, N.A.A.Q. The recovery of protein hydrolysate during enzymatic isolation of chitin from shrimp Crangon crangon processing discards. Food Chem. 2000, 68, 147–152. [Google Scholar] [CrossRef]
- Johnson, E.L.; Peniston, Q.P. Utilization of shellfish waste for chitin and chitosan production. In Chemistry and Biochemistry of Marine Food Products; Avi Publishing Company: Westport, Connecticut, 1979; pp. 415–422. [Google Scholar]
- Shahidi, F.; Synowiecki, J. Isolation and characterization of nutrients and value-added products from snow crab (Chionoecetes opilio) and shrimp (Pandalus borealis) processing discards. J. Agric. Food Chem. 1991, 39, 1527–1532. [Google Scholar] [CrossRef]
- Le Roux, K. Purification de la chitine par hydrolyse enzymatique à partir de coproduits de crevette Penaeus vannamei. Caractérisations des produits et optimisation du procédé. Ph.D. Thesis, Université de Nantes-France Faculté des Sciences et des Techniques, Nantes, France, 2012. [Google Scholar]
- Lage Yusty, M.A.; Vilasoa Martínez, M.; Álvarez Pérez, S.; López Hernández, J. Chemical composition of snow crab shells (Chionoecetes opilio). CyTA J. Food Sci. 2011, 9, 265–270. [Google Scholar] [CrossRef]
- Das, S.; Roy, D.; Sen, R. Utilization of chitinaceous wastes for the production of chitinase. Adv. Food Nutr. Res. 2016, 78, 27–46. [Google Scholar] [PubMed]
- Raabe, D.; Romano, P.; Sachs, C.; Fabritius, H.; Al-Sawalmih, A.; Yi, S.B.; Servos, G.; Hartwig, H.G. Microstructure and crystallographic texture of the chitinprotein network in the biological composite material of the exoskeleton of the lobster Homarus americanus. Mater. Sci. Eng. A 2006, 421, 143–153. [Google Scholar] [CrossRef]
- Poirier, M. Fractionnement et Caractérisation de la Chitine Dans le Système n,n-Dimethylacétamide/Chlorure de Lithium. Ph.D. Thesis, Département de Chimie Faculté des Sciences et de Genie, Université Laval, Québec, QC, Canada, 2000. [Google Scholar]
- Minke, R.; Blackwell, J. The structure of α-chitin. J. Mol. Biol. 1978, 120, 167–181. [Google Scholar] [CrossRef]
- Gardner, K.H.; Blackwell, J. Refinement of the structure of β-chitin. Biopolym. Orig. Res. Biomol. 1975, 14, 1581–1595. [Google Scholar]
- Andonegi, M.; Las Heras, K.; Santos-Vizcaíno, E.; Igartua, M.; Hernandez, R.M.; de la Caba, K.; Guerrero, P. Structure-properties relationship of chitosan/collagen films with potential for biomedical applications. Carbohydr. Polym. 2020, 237, 116–159. [Google Scholar] [CrossRef] [PubMed]
- Chawla, S.P.; Kanatt, S.R.; Sharma, A.K. Chitosan, Polysaccharides; Springer International Publishing: Cham, Switzerland, 2015; pp. 1–24. [Google Scholar]
- Truong, T.O.; Hausler, R.; Monette, F.; Niquette, P. Valorisation des résidus industriels de pêches pour la transformation de chitosane par technique hydrothermo-chimique. Revue Sci. L’eau/J. Water Sci. 2007, 20, 253–262. [Google Scholar] [CrossRef]
- Nwe, N.; Stevens, W.F. Production of fungal chitosan by solid substrate fermentation followed by enzymatic extraction. Biotechnol. Lett. 2002, 24, 131–134. [Google Scholar] [CrossRef]
- Randriamahatody, Z. Valorisation Biotechnologique des Coproduits de Crevette: Utilisation de la Protéolyse Enzymatique Pour des Applications Avicoles à Madagascar. Ph.D. Thesis, Antananarivo University, Antananarivo, Madagascar, 2011. [Google Scholar]
- Ma, P.L. Formation et Caractérisation Physico-Chimique des Complexes adn/Chitosane Pour la Thérapie Génique. Ph.D. Thesis, University of Montreal, Montreal, QC, Canada, 2010. [Google Scholar]
- Crini, G.; Badot, P.M.; Roberts, G.A.; Guibal, E. Chitine et Chitosane: Du Biopolymère à L’application; Presses Universitaires: Franche-Comté, France, 2009; pp. 27–28. [Google Scholar]
- Rout, S.K. Physicochemical, Functional and Sprctroscopic Analysis of Crawfish Chitin and Chitosan as Affected by Process Modification. Ph.D. Thesis, Agricultural Center, Lousiana State University, Baton Rouge, LA, USA, 2001; pp. 1–174. [Google Scholar]
- Knaul, J.Z.; Hudson, S.M.; Creber, K.A. Crosslinking of chitosan fibers with dialdehydes: Proposal of a new reaction mechanism. J. Polym. Sci. Part B Polym. Phys. 1999, 37, 1079–1094. [Google Scholar] [CrossRef]
- He, X.; Li, K.; Xing, R.; Liu, S.; Hu, L.; Li, P. The production of fully deacetylated chitosan by compression method. Egypt. J. Aquat. Res. 2016, 42, 75–81. [Google Scholar] [CrossRef]
- No, H.K.; Meyers, S.P.; Lee, K.S. Isolation and Characterization of Chitin from Crawfish Shell Waste. J. Agric. Food Chem. 1989, 37, 575–579. [Google Scholar] [CrossRef]
- Khan, T.A.; Peh, K.K.; Ch’ng, H.S. Reporting degree of deacetylation values of chitosan: The influence of analytical methods. J. Pharm. Pharm. Sci. 2002, 5, 205–212. [Google Scholar]
- Sabnis, S.; Block, L.H. Improved infrared spectroscopic method for the analysis of degree of N-deacetylation of chitosan. Polym. Bull. 1997, 39, 67–71. [Google Scholar] [CrossRef]
- Baxter, A.; Dillon, M.; Taylor, K.A.; Roberts, G.A. Improved method for ir determination of the degree of N-acetylation of chitosan. Int. J. Biol. Macromol. 1992, 14, 166–169. [Google Scholar] [CrossRef]
- Muzzarelli, R.A.A. Human enzymatic activities related to the therapeutic administration of chitin derivatives. Cell. Mol. Life Sci. 1997, 53, 131–140. [Google Scholar] [CrossRef]
- Fukamizo, T.; Ohkawa, T.; Ikeda, Y.; Goto, S. Specificity of chitosanase from Bacillus pumilus. Biochim. Biophys. Acta (BBA)-Protein Struct. Mol. Enzymol. 1994, 1205, 183–188. [Google Scholar] [CrossRef]
- Hunt, S.; Huckerby, T.N. Comparative study of molluscan and crustacean chitin proteoglycans by carbon-13 NMR spectroscopy. Identification of carbohydrate and amino acid contributions and the determination of amino acid chemical shifts in anhydrous formic acid. Comp. Biochem. Physiol. Part B Comp. Biochem. 1987, 88, 1107–1116. [Google Scholar] [CrossRef]
- Gatto, M.; Ochi, D.; Yoshida CM, P.; da Silva, C.F. Study of chitosan with different degrees of acetylation as cardboard paper coating. Carbohydr. Polym. 2019, 210, 56–63. [Google Scholar] [CrossRef] [PubMed]
- Kumirska, J.; Weinhold, M.X.; Thöming, J.; Stepnowski, P. Biomedical Activity of Chitin/Chitosan Based Materials—Influence of Physicochemical Properties Apart from Molecular Weight and Degree of N-Acetylation. Polymers 2011, 3, 1875–1901. [Google Scholar] [CrossRef]
- Al Hoqani, H.A.S.; Noura, A.S.; Hossain, M.A.; Al Sibani, M.A. Isolation and optimization of the method for industrial production of chitin and chitosan from Omani shrimp shell. Carbohydr. Res. 2020, 492, 108001. [Google Scholar] [CrossRef] [PubMed]
- Kumar, M.N.R. A review of chitin and chitosan applications. React. Funct. Polym. 2000, 46, 1–27. [Google Scholar] [CrossRef]
- Madhavan, P.; Ramachandran Nair, K.G. Utilization of prawn waste: Isolation of chitin and its conversion to chitosan. Fish. Technol. 1974, 11, 50–53. [Google Scholar]
- Percot, A.; Viton, C.; Domard, A. Characterization of shrimp shell deproteinization. Biomacromolecules 2003, 4, 1380–1385. [Google Scholar] [CrossRef]
- Synowiecki, J.; Al-Khateeb, N.A. Production, Properties, and Some New Applications of Chitin and Its Derivatives. Crit. Rev. Food Sci. Nutr. 2003, 43, 145–171. [Google Scholar] [CrossRef]
- Charoenvuttitham, P.; John, S.; Gauri Mittal, S. Chitin Extraction from Black Tiger Shrimp (Penaeus monodon) Waste using Organic Acids. Sep. Sci. Technol. 2006, 41, 1135–1153. [Google Scholar] [CrossRef]
- Mukherjee, D.P. Method for Producing Chitin or Chitosan. U.S. Patent 6,310,188, 10 October 2002. [Google Scholar]
- Kurita, K.; Tomita, K.; Tada, T.; Ishii, S.; Nishimura, S.I.; Shimoda, K. Squid chitin as a potential alternative chitin source: Deacetylation behavior and characteristic. J. Polym. Sci. Part A Polym. Chem. 1993, 31, 485–491. [Google Scholar] [CrossRef]
- Khan, W.; Prithiviraj, B.; Smith, D.L. Chitosan and chitin oligomers increase phenylalanine ammonia-lyase and tyrosine ammonia-lyase activities in soybean leaves. J. Plant Physiol. 2003, 160, 859–863. [Google Scholar] [CrossRef]
- Dörnenburg, H.; Knorr, D. Elicitation of chitinases and anthraquinones in Morinda citrifolia cell cultures. J. Food Biotechnol. 1994, 8, 57–65. [Google Scholar] [CrossRef]
- Cottrell, M.T.; Wood, D.N.; Yu, L.; Kirchman, D.L. Selected chitinase genes in cultured and uncultured marine bacteria in the α-and γ-subclasses of the proteobacteria. Appl. Environ. Microbiol. 2000, 66, 1195–1201. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Muzzarelli, R.A.A. Chitosan-based dietary foods. Carbohydr. Polymers 1996, 29, 309–316. [Google Scholar] [CrossRef]
- Piron, E.; Accominotti, M.; Domard, A. Interaction between chitosan and uranyl ions. Role of physical and physic-chemical parameters on the kinetics sorption. Langmuir 1997, 13, 1653–1658. [Google Scholar] [CrossRef]
- Singh, D.K.; Ray, A.R. Biomedical applications of chitin, chitosan, and their derivatives. J. Macromol. Sci. Part C Polym. Rev. 2000, 40, 69–83. [Google Scholar] [CrossRef]
- Bacon, A.; Markin, J.; Sizer, P.J.; Jabbal-Gill, I.; HincHCliffe, M.; Illum, L.; Chatfield, S.; Roberts, M. Carbohydrate biopolymers enhance antibody responses to mucosally delivered vaccine antigens. Infect. Immun. 2000, 68, 5764–5770. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Min, M.; Du, N.; Gu, Y.; Hode, T.; Naylor, M.; Chen, D.; Nordquist, R.E.; Chen, W.R. Chitin, chitosan, and glycated chitosan regulate immune responses: The novel adjuvants for cancer vaccine. Clin. Dev. Immunol. 2013, 2013, 1–8. [Google Scholar] [CrossRef]
- Devlieghere, F.; Vermeulen, A.; Debevere, J. Chitosan: Antimicrobial activity, interactions with food components and applicability as coating on fruit and vegetables. Food Microbiol. 2004, 21, 703–714. [Google Scholar] [CrossRef]
- Chirkov, S.N.; Il’ina, A.V.; Surgucheva, N.A.; Letunova, E.V.; Varitsev, Y.A.; Tatarinova, N.Y.; Varlamov, V.P. Effect of chitosan on systemic viral infection and some defense responses in potato plants. Russ. J. Plant Physiol. 2001, 48, 774–779. [Google Scholar] [CrossRef]
- Khalil, M.S.; Badawy, M.E.I. Nematicidal activity of a biopolymer chitosan at different molecular weights against root-knot nematode, Meloidogyne incognita. J. Plant Protec. Sci. 2012, 48, 170–178. [Google Scholar] [CrossRef]
- Radwan, M.A.; Farrag, S.A.; Abu-Elamayem, M.M.; Ahmed, N.S. Extraction, characterization, and nematicidal activity of chitin and chitosan derived from shrimp shell wastes. Biol. Fertil. Soils 2012, 48, 463–468. [Google Scholar] [CrossRef]
- Rajalakshmi, A.; Krithiga, N.; Jayachitra, A. Antioxidant Activity of the Chitosan Extracted from Shrimp Exoskeleton. Middle East J. Sci. Res. 2013, 16, 1446–1451. [Google Scholar]
- Hewajulige, I.G.N.; Sultanbawa, Y.; Shanthi Wilson Wijeratnam, R.; Ravindra Wijesundara, L.C. Mode of action of chitosan coating on anthracnose disease control in papaya. Phytoparasitica 2009, 37, 437–444. [Google Scholar] [CrossRef]
- Liu, X.; Feng, X.Q.; Yang, S.; Wang, T.P.; Su, Z.X. Effects of MolecularWeight and Concentration of Chitosan on Antifungal Activity Against Aspergillus Niger. Iran. Polym. J. 2008, 17, 843–852. [Google Scholar]
- El-Mougy, N.S.; El-Gamal, N.G.; Fotouh, Y.O.; Abd-El-Kareem, F. Evaluation of Different Application Methods of Chitin and Chitosan for Controlling Tomato Root Rot Disease under Greenhouse and Field Conditions. Res. J. Agric. Biol. Sci. 2006, 2, 190–195. [Google Scholar]
- Benhamou, N. Potential of the Mycoparasite, Verticillium lecanii, to Protect Citrus Fruit against Penicillium digitatum, the Causal Agent of Green Mold: A Comparison with the Effect of Chitosan. Am. Phytopathol. Soc. 2004, 94, 693–705. [Google Scholar] [CrossRef]
- Rodríguez, A.T.; Ramírez, M.A.; María, C.N.; Ramona, M.; Regla, M.C. Antifungal activity of chitosan and one of its hydrolysates on pyricularia grisea, sacc. Fungus. Cultiv. Trop. 2003, 24, 85–88. [Google Scholar]
- No, H.K.; Park, N.Y.; Lee, S.H.; Meyers, S.P. Antibacterial activity of chitosans and chitosan oligomers with different molecular weights. Int. J. Food Microbiol. 2002, 74, 65–72. [Google Scholar] [CrossRef]
- Tsai, G.J.; Su, W.H. Antibacterial activity of shrimp chitosan against Escherichia coli. J. Food Prot. 1999, 62, 239–243. [Google Scholar] [CrossRef]
- Limam, Z.; Sadok, S.; El Abed, O. Étude de la composition biochimique de la chair et des coproduits de la crevette royale Penaeus kerathurus du nord et sud de la tunisie. J. Bull. l’Institut Natl. Sci. Technol. Mer Tech. Mer Salammbô 2010, 37, 75–81. [Google Scholar]
- Nge, K.L.; Nwe, N.; Chandrkrachang, S.; Stevens, W.F. Chitosan as a growth stimulator in orchid tissue culture. Plant Sci. 2006, 170, 1185–1190. [Google Scholar] [CrossRef]
- Boonlertnirun, S.; Boonraung, C.; Suvanasara, R. Application of Chitosan in Rice Production. J. Met. Mater. Miner. 2008, 18, 47–52. [Google Scholar]
- El-Tantawy, E.M. Behavior of Tomato Plants as Affected by Spraying with Chitosan and Aminofort as Natural Stimulator Substances under Application of Soil Organic Amendments. Pak. J. Biol. Sci. 2009, 12, 1164–1173. [Google Scholar] [CrossRef] [PubMed]
- Mondal, M.M.A.; Malek, M.A.; Puteh, A.B.; Ismail, M.R.; Ashrafuzzaman, M.; Naher, L. Effect of foliar application of chitosan on growth and yield in okra. Aust. J. Crop Sci. 2012, 6, 918–921. [Google Scholar]
- Kim, H.J.; Chen, F.; Wang, X.; Rajapakse, N.C. Effect of Chitosan on the Biological Properties of Sweet Basil (Ocimum basilicum, L.). J. Agric. Food Chem. 2005, 53, 3696–3701. [Google Scholar] [CrossRef]
- Liopa-Tsakalidi, A.; Chalikiopoulos, D.; Papasavvas, A. Effect of chitin on growth and chlorophyll content of two medicinal plants. J. Med. Plants Res 2010, 4, 499–508. [Google Scholar]
- Dzung, N.A.; Khanh, V.T.P.; Dung, T.T. Research on impact of chitosan oligomeron biophysical characteristics, growth, development and drought resistance of coffee. Carbohydr. Polym. 2011, 84, 751–755. [Google Scholar] [CrossRef]
- Bittelli, M.; Flury, M.; Campbell, G.S.; Nichols, E.J. Reduction of transpiration through foliar application of chitosan. Agric. For. Meteorol. 2001, 107, 167–175. [Google Scholar] [CrossRef]
- Van, S.N.; Minh, H.D.; Anh, D.N. Study on chitosan nanoparticles on biophysical characteristics and growth of Robusta coffee in green house. Biocatal. Agric. Biotechnol. 2013, 2, 289–294. [Google Scholar]
- Sharp, R.G. A review of the applications of chitin and its derivatives in agriculture to modify plant-microbial interactions and improve crop yields. Agronomy 2013, 3, 757–793. [Google Scholar] [CrossRef]
- Saharan, V.; Pal, A. Chitosan Based Nanomaterials in Plant Growth and Protection; Springer: New Delhi, India, 2016; pp. 33–41. [Google Scholar]
- Rkhaila, A.; Abla, E.H.; Mohammed, B.I.; Ounine, K. The Amendment with Chitin and/or Chitosan Improves the Germination and Growth of Lycopersicon esculentum, L.; Capsicum annuum, L. and Solanum melongena, L. Indian J. Agric. Res. 2020, 54, 420–428. [Google Scholar]
- Benhamou, N.; Thériault, G. Treatment with chitosan enhances resistance of tomato plants to the crown and root rot pathogen Fusarium oxysporum f. sp. radicis-lycopersici. Physiol. Mol. Plant Pathol. 1992, 41, 33–52. [Google Scholar] [CrossRef]
- Benhamou, N.; Picard, K. La résistance induite: Une nouvelle stratégie de défense des plantes contre les agents pathogènes. J. Phytoprotection 2000, 80, 137–168. [Google Scholar] [CrossRef]
- Amine, R.; Khadija, O. Shrimp shells, Chitin and chitosan powders effect on growth of Lycopersicon esculentum and their ability to induce resistance against Fusarium oxysporum f. sp. radicis-lycopersici attack. Indian J. Agric. Res. 2018, 52, 512–517. [Google Scholar] [CrossRef]
- Rhazi, M.; Desbrières, J.; Tolaimate, A.; Rinaudo, M.; Vottero, P.; Alagui, A.; EL Meray, M. Influence of the nature of the metal ions on the complexation with chitosan. Application to the treatment of liquid waste. Eur. Polym. J. 2002, 38, 1523–1530. [Google Scholar] [CrossRef]
- Dobereiner, J. History and new perspectives of diazotrophs in association with non leguminous plants. Symbiosis 1992, 13, 1–13. [Google Scholar]
- Mahaffee, W.F.; Kloepper, J.W. Temporal changes in the bacterial communities of soil, rhizosphere, and endorhiza associated with field-grown cucumber (Cucumis sativus, L.). Microb. Ecol. 1997, 34, 210–223. [Google Scholar] [CrossRef] [PubMed]
- Hardoim, P.R.; van Overbeek, L.S.; Berg, G.; Pirttilä, A.M.; Compant, S.; Campisano, A.; Döring, M.; Sessitsch, A. The hidden world within Plants: Ecological and evolutionary considerations for defining functioning of microbial endophytes. Microbiol. Mol. Biol. Rev 2015, 79, 293–320. [Google Scholar] [CrossRef] [PubMed]
- Van Overbeek, L.S.; Bergervoet, J.H.; Jacobs, F.H.; van Elsas, J.D. The low-temperature-induced viable-but-nonculturable state affects the virulence of Ralstonia solanacearum biovar 2. Phytopathology 2004, 94, 463–469. [Google Scholar] [CrossRef]
- Rosenblueth, M.; Martínez-Romero, E. Bacterial endophytes and their interactions with hosts. Mol. Plant-Microbe Interact. 2006, 19, 827–837. [Google Scholar] [CrossRef]
- Chi, F.; Shen, S.; Cheng, H.; Jing, Y.; Yanni, Y.; Dazzo, F. Ascending migration of endophytic rhizobia, from roots to leaves, inside rice plants and assessment of benefits to rice growth physiology. Appl. Environ. Microbiol. 2005, 71, 7271–7278. [Google Scholar] [CrossRef] [PubMed]
- McCully, M.E. Niches for bacterial endophytes in crop plants: A plant biologist’s view. Aust. J. Plant Physiol. 2001, 28, 983–990. [Google Scholar] [CrossRef]
- Liu, H.; Carvalhais, L.C.; Crawford, M.; Singh, E.; Dennis, P.G.; Pieterse, C.M.; Schenk, P.M. Inner plant values: Diversity, colonization and benefits from endophytic bacteria. Front. Microbiol. 2017, 8, 2552. [Google Scholar] [CrossRef]
- Tiwari, S.; Prasad, V.; Lata, C. Bacillus: Plant growth promoting bacteria for sustainable agriculture and environment. In New and Future Developments in Microbial Biotechnology and Bioengineering; Elsevier: Amsterdam, The Netherlands, 2019; pp. 43–55. [Google Scholar]
- Tifrit, A. Isolement et Caractérisation des Bactéries à Intérêts Biotechnologiques à Partir de Niches Écologiques Algériennes. Ph.D. Thesis, University Hassiba Benbouali de Chlef, Algeria, 2016. [Google Scholar]
- BORRISS, Rainer. Use of plant-associated Bacillus strains as biofertilizers and biocontrol agents in agriculture. In Bacteria in Agrobiology: Plant Growth Responses; Springer: Berlin/Heidelberg, Germany, 2011; pp. 41–76. [Google Scholar]
- Xie, G.; Su, B.; Cui, Z. Isolation and identification of N2-fixing strains of Bacillus in rice rhizosphere of the Yangtze River valley. Wei Sheng Wu Xue Bao = Acta Microbiol. Sin. 1998, 38, 480–483. [Google Scholar]
- Li, C.Y.; Massicote, H.B.; Moore, L.V.H. Nitrogen-fixing Bacillus sp. Associated with Douglas-fir tuberculate ectomycorrhizae. Plant Soil 1992, 140, 35–40. [Google Scholar] [CrossRef]
- Ahmad, F.; Ahmad, I.; Khan, M.S. Screening of free-living rhizospheric bacteria for their multiple plant growth promoting activities. Microbiol. Res. 2008, 163, 173–181. [Google Scholar] [CrossRef]
- Różycki, H.; Dahm, H.; Strzelczyk, E.; Li, C.Y. Diazotrophic bacteria in root-free soil and in the root zone of pine (Pinus sylvestris, L.) and oak (Quercus robur, L.). Appl. Soil Ecol. 1999, 12, 239–250. [Google Scholar] [CrossRef]
- Ding, Y.; Wang, J.; Liu, Y.; Chen, S. Isolation and identification of nitrogen-fixing bacilli from plant rhizospheres in Beijing region. J. Appl. Microbiol. 2005, 99, 1271–1281. [Google Scholar] [CrossRef]
- Sorokin, I.D.; Kravchenko, I.K.; Tourova, T.P.; Kolganova, T.V.; Boulygina, E.S.; Sorokin, D.Y. Bacillus alkalidiazotrophicus sp. nov.; a diazotrophic, low salt-tolerant alkaliphile isolated from Mongolian soda soil. Int. J. Syst. Evol. Microbiol. 2008, 58, 2459–2464. [Google Scholar] [CrossRef] [PubMed]
- Sorokin, I.D.; Zadorina, E.V.; Kravchenko, I.K.; Boulygina, E.S.; Tourova, T.P.; Sorokin, D.Y. Natronobacillus azotifigens gen. nov.; sp. Nov.; an anaerobic diazotrophic haloalkaliphile from soda-rich habitats. Extremophiles 2008, 12, 819–827. [Google Scholar] [CrossRef]
- Seldin, L.; Rosado, A.S.; da Cruz, D.W.; Nobrega, A.; van Elsas, J.D.; Paiva, E. Comparison of Paenibacillus azotofixans strains isolated from rhizoplane, rhizosphere, and non-root-associated soil from maize planted in two different Brazilian soils. Appl. Environ. Microbiol. 1998, 64, 3860–3868. [Google Scholar] [CrossRef] [PubMed]
- Beneduzi, A.; Peres, D.; Vargas, L.K.; Bodanese-Zanettini, M.H.; Passaglia, L.M.P. Evaluation of genetic diversity and plant growth promoting activities of nitrogen-fixing bacilli isolated from rice fields in South Brazil. Appl. Soil Ecol. 2008, 39, 311–320. [Google Scholar] [CrossRef]
- Beneduzi, A.; Peres, D.; Costa, P.B.; Bodanese-Zanettini, M.H.; Passaglia, L.M.P. Genetic and phenotypic diversity of plant-growth-promoting bacilli isolated from wheat fields in southern Brazil. Res. Microbiol. 2008, 159, 244–250. [Google Scholar] [CrossRef] [PubMed]
- Alagawadi, A.R.; Gaur, A.C. Associative effect of Rhizobium and phosphate-solubilizing bacteria on the yield and nutrient uptake of chickpea. Plant Soil 1988, 105, 241–246. [Google Scholar] [CrossRef]
- Li, D.; Alexander, M. Co-inoculation with antibiotic-producing bacteria to increase colonization and nodulation by rhizobia. Plant Soil 1988, 108, 211–219. [Google Scholar] [CrossRef]
- Mehboob, I.; Naveed, M.; Zahir, Z.A. Rhizobial association with non-legumes: Mechanisms and applications. Crit. Rev. Plant Sci. 2009, 28, 432–456. [Google Scholar] [CrossRef]
- Gaiero, J.R.; McCall, C.A.; Thompson, K.A.; Day, N.J.; Best, A.S.; Dunfield, K.E. Inside the root microbiome: Bacterial root endophytes and plant growth promotion. Am. J. Bot. 2013, 100, 1738–1750. [Google Scholar] [CrossRef]
- Ait Barka, E.; Nowak, J.; Clément, C. Enhancement of chilling resistance of inoculated grapevine plantlets with a plant growth-promoting rhizobacterium, Burkholderia phytofirmans strain PsJN. Appl. Environ. Microbiol. 2006, 72, 7246–7252. [Google Scholar] [CrossRef]
- Naveed, M.; Hussain, M.B.; Zahir, Z.A.; Mitter, B.; Sessitsch, A. Drought stress amelioration in wheat through inoculation with Burkholderia phytofirmans strain PsJN. Plant Growth Regul. 2014, 73, 121–131. [Google Scholar] [CrossRef]
- Jha, Y.; Subramanian, R.B.; Patel, S. Combination of endophytic and rhizospheric plant growth promoting rhizobacteria in Oryza sativa shows higher accumulation of osmoprotectant against saline stress. Acta Physiol. Plant. 2011, 33, 797–802. [Google Scholar] [CrossRef]
- Cohen, A.C.; Travaglia, C.; Bottini, R.; Piccoli, P. Participation of abscisic acid and gibberellins produced by entophytic Azospirillum in the alleviation of drought effects in maize. Botany 2009, 87, 455–462. [Google Scholar] [CrossRef]
- Tuteja, N. Mechanisms of high salinity tolerance in plants. In Methods in Enzymology; Academic Press: Cambridge, MA, USA, 2007; Volume 428, pp. 419–438. [Google Scholar]
- Qin, S.; Zhang, Y.J.; Yuan, B.; Xu, P.Y.; Xing, K.; Wang, J.; Jiang, J.H. Isolation of ACC deaminase-producing habitat-adapted symbiotic bacteria associated with halophyte Limonium sinense (Girard) Kuntze and evaluating their plant growth-promoting activity under salt stress. Plant Soil 2014, 374, 753–766. [Google Scholar] [CrossRef]
- Taghavi, S.; Garafola, C.; Monchy, S.; Newman, L.; Hoffman, A.; Weyens, N.; Barac, T.; Vangronsveld, J.; van der Lelie, D. Genome survey and characterization of endophytic bacteria exhibiting a beneficial effect on growth and development of poplar trees. Appl. Environ. Microbiol 2009, 75, 748–757. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.; Lou, K.; Li, C. Growth and photosynthetic efficiency promotion of sugar beet (Beta vulgaris, L.) by endophytic bacteria. Photosynth. Res. 2010, 105, 5–13. [Google Scholar] [CrossRef]
- Strader, L.C.; Chen, G.L.; Bartel, B. Ethylene directs auxin to control root cell expansion. Plant J. 2010, 64, 874–884. [Google Scholar] [CrossRef] [PubMed]
- Glick, B.R.; Todorovic, B.; Czarny, J.; Cheng, Z.; Duan, J.; McConkey, B. Promotion of plant growth by bacterial ACC deaminase. Crit. Rev. Plant Sci. 2007, 26, 227–242. [Google Scholar] [CrossRef]
- Nadeem, S.M.; Zahir, Z.A.; Naveed, M.; Ashraf, M. Microbial ACC-deaminase: Prospects and applications for inducing salt tolerance in plants. Crit. Rev. Plant Sci. 2010, 29, 360–393. [Google Scholar] [CrossRef]
- Cody, R.M.; Davis, N.D.; Lin, J.; Shaw, D. Screening microorganisms for chitin hydrolysis and production of ethanol from amino sugars. Biomass 1990, 21, 285–295. [Google Scholar] [CrossRef]
- Duo-Chuan, L. Review of fungal chitinases. Mycopathologia 2006, 161, 345–360. [Google Scholar] [CrossRef]
- Kuddus, M.; Ahmad, I.Z. Isolation of novel chitinolytic bacteria and production optimization of extracellular chitinase. J. Genet. Eng. Biotechnol. 2013, 11, 39–46. [Google Scholar]
- Bhattacharya, D.; Nagpure, A.; Gupta, R.K. Bacterial chitinases: Properties and potential. Crit. Rev. Biotechnol. 2007, 27, 21–28. [Google Scholar] [CrossRef] [PubMed]
- McCreath, K.J.; Gooday, G.W. A rapid and sensitive microassay for determination of chitinolytic activity. J. Microbiol. Methods 1992, 14, 229–237. [Google Scholar] [CrossRef]
- Reynolds, S.E.; Samuels, R.I. Physiology and biochemistry of insect moulting fluid. In Advances in Insect Physiology; Academic Press: Cambridge, MA, USA, 1996; Volume 26, pp. 157–232. [Google Scholar]
- Dahiya, N.; Tewari, R.; Hoondal, G.S. Biotechnological aspects of chitinolytic enzymes: A review. Appl. Microbiol. Biotechnol. 2006, 71, 773–782. [Google Scholar] [CrossRef]
- Henrissat, B.; Bairoch, A. New families in the classification of glycosyl hydrolases based on amino acid sequence similarities. Biochem. J. 1993, 293, 781–788. [Google Scholar] [CrossRef]
- Hart, P.J.; Pfluger, H.D.; Monzingo, A.F.; Hollis, T.; Robertus, J.D. The refined crystal structure of an endochitinase from Hordeum vulgare L. seeds at 1.8 Å resolution. J. Mol. Biol. 1995, 248, 402–413. [Google Scholar] [PubMed]
- Suzuki, K.; Taiyoji, M.; Sugawara, N.; Nikaidou, N.; Henrissat, B.; Watanabe, T. The third chitinase gene (chiC) of Serratia marcescens 2170 and the relationship of its product to other bacterial chitinases. Biochem. J. 1999, 343, 587–596. [Google Scholar] [CrossRef] [PubMed]
- Vervaet, A. Assessing the Presence of Chitinases in the Digestive Tract and their Relationship to Diet and Morphology in Freshwater Fish. Ph.D. Thesis, Ghent University, Ghent, Belgium, 2019. [Google Scholar]
- Zou, E. Invisible endocrine disruption and its mechanisms: A current review. Gen. Comp. Endocrinol. 2020, 293, 113470. [Google Scholar] [CrossRef]
- Collinge, D.B.; Kragh, K.M.; Mikkelsen, J.D.; Nielsen, K.K.; Rasmussen, U.; Vad1, K. Plant chitinases. Plant J. 1993, 3, 31–40. [Google Scholar] [CrossRef]
- Cabib, E.; Silverman, S.J.; Shaw, J.A. Chitinase and chitin synthase 1: Counterbalancing activities in cell separation of Saccharomyces cerevisiae. Microbiology 1992, 138, 97–102. [Google Scholar] [CrossRef]
- Adrangi, S.; Faramarzi, M.A.; Shahverdi, A.R.; Sepehrizadeh, Z. Purification and characterization of two extracellular endochitinases from Massilia timonae. Carbohydr. Res. 2010, 345, 402–407. [Google Scholar] [CrossRef]
- Adrangi, S.; Faramarzi, M.A. From bacteria to human: A journey into the world of chitinases. Biotechnol. Adv. 2013, 31, 1786–1795. [Google Scholar] [CrossRef] [PubMed]
- Shapira, R.; Ordentlich, A.; Chet, I.; Oppenheim, A.B. Control of plant diseases by chitinase expressed from cloned DNA in Escherichia coli. Phytopathology 1989, 79, 1246–1249. [Google Scholar] [CrossRef]
- Wen, Z.; Bai, J.; Wang, L.; Yao, L.; Ahmad, B.; Hanif, M.; Chen, Q. Over expression of a Chitinase 2 gene from Chinese Wild Strawberry improves resistance to anthracnose disease in transgenic Arabidopsis thaliana. Plant Biotechnol. Rep. 2020, 14, 725–736. [Google Scholar] [CrossRef]
- Prakash Udaya, N.; Jayanthi, M.; Sabarinathan, R.; Kangueane, P.; Mathew, L.; Sekar, K. Evolution, homology conservation, and identification of unique sequence signatures in GH19 family chitinases. J. Mol. Evol. 2010, 70, 466–478. [Google Scholar] [CrossRef] [PubMed]
- Larsen, T.; Petersen, B.O.; Storgaard, B.G.; Duus, J.Ø.; Palcic, M.M.; Leisner, J.J. Characterization of a novel Salmonella Typhimurium chitinase which hydrolyzes chitin, chitooligosaccharides and an N-acetyllactosamine conjugate. Glycobiology 2010, 21, 426–436. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Greene, L.H. Sequence and structural analysis of the chitinase insertion domain reveals two conserved motifs involved in chitin-binding. PLoS ONE 2010, 5, e8654. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, T.; Kanai, R.; Kawase, T.; Tanabe, T.; Mitsutomi, M.; Sakuda, S.; Miyashita, K. Family 19 chitinases of Streptomyces species: Characterization and distribution. Microbiology 1999, 145, 3353–3363. [Google Scholar] [CrossRef] [PubMed]
- Horn, S.J.; Sikorski, P.; Cederkvist, J.B.; Vaaje-Kolstad, G.; Sørlie, M.; Synstad, B.; Eijsink, V.G. Costs and benefits of processivity in enzymatic degradation of recalcitrant polysaccharides. Proc. Natl. Acad. Sci. USA 2006, 103, 18089–18094. [Google Scholar] [CrossRef] [PubMed]
- Ueda, M.; Ohata, K.; Konishi, T.; Sutrisno, A.; Okada, H.; Nakazawa, M.; Miyatake, K. A novel goose-type lysozyme gene with chitinolytic activity from the moderately thermophilic bacterium Ralstonia sp. A-471: Cloning, sequencing, and expression. Appl. Microbiol. Biotechnol. 2009, 81, 1077–1085. [Google Scholar] [CrossRef] [PubMed]
- Rhodes, R.G.; Atoyan, J.A.; Nelson, D.R. The chitobiose transporter, chbC, is required for chitin utilization in Borrelia burgdorferi. BMC Microbiol. 2010, 10, 21. [Google Scholar] [CrossRef]
- Delpin, M.W.; Goodman, A.E. Nitrogen regulates chitinase gene expression in a marine bacterium. ISME J. 2009, 3, 1064–1069. [Google Scholar] [CrossRef]
- Prasanna, R.; Gupta, V.; Natarajan, C.; Chaudhary, V. Bioprospecting for genes involved in the production of chitosanases and microcystin-like compounds in Anabaena strains. World J. Microbiol. Biotechnol. 2010, 26, 717–724. [Google Scholar] [CrossRef]
- Busby, J.N.; Landsberg, M.J.; Simpson, R.M.; Jones, S.A.; Hankamer, B.; Hurst, M.R.; Lott, J.S. Structural analysis of Chi1 chitinase from Yen-Tc: The multisubunit insecticidal ABC toxin complex of Yersinia entomophaga. J. Mol. Biol. 2012, 415, 359–371. [Google Scholar] [CrossRef]
- Chandler, J.C.; Molins, C.R.; Petersen, J.M.; Belisle, J.T. Differential chitinase activity and production within Francisella species, subspecies, and subpopulations. J. Bacteriol. 2011, 193, 3265–3275. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Davis, B.; Eveleigh, D.E. Chitosanases: Occurrence, production and immobilization. In Chitin, Chitosan and Related Enzymes; Zikakis, J.P., Ed.; Academic Press: New York, NY, USA, 1984; pp. 161–179. [Google Scholar]
- Hedges, A.; Wolfe, R.S. Extracellular enzyme from Myxobacter AL-1 that exhibits both β-1, 4-glucanase and chitosanase activities. J. Bacteriol. 1974, 120, 844–853. [Google Scholar] [CrossRef] [PubMed]
- Buswell, J.A.; Cai, Y.J.; Chang, S.T.; Peberdy, J.F.; Fu, S.Y.; Yu, H.S. Lignocellulolytic enzyme profiles of edible mushroom fungi. World J. Microbiol. Biotechnol. 1996, 12, 537–542. [Google Scholar] [CrossRef]
- Pelletier, A.; Sygusch, J. Purification and characterization of three chitosanase activities from Bacillus megaterium P1. Appl. Environ. Microbiol. 1990, 56, 844–848. [Google Scholar] [CrossRef]
- Kendra, D.F.; Hadwiger, L.A. Characterization of the smallest chitosan oligomer that is maximally antifungal toFusarium solani and elicits pisatin formation inPisum sativum. Exp. Mycol. 1984, 8, 276–281. [Google Scholar] [CrossRef]
- Ortiz-Rodríguez, T.; De La Fuente-Salcido, N.; Bideshi, D.K.; Salcedo-Hernández, R.; Barboza-Corona, J.E. Generation of chitin-derived oligosaccharides toxic to pathogenic bacteria using ChiA74, an endochitinase native to Bacillus thuringiensis. Lett. Appl. Microbiol. 2010, 51, 184–190. [Google Scholar] [CrossRef]
- Das, S.N.; Dutta, S.; Kondreddy, A.; Chilukoti, N.; Pullabhotla, S.V.; Vadlamudi, S.; Podile, A.R. Plant growth-promoting chitinolytic Paenibacillus elgii responds positively to tobacco root exudates. J. Plant Growth Regul. 2010, 29, 409–418. [Google Scholar] [CrossRef]
- Arora, N.; Sachdev, B.; Gupta, R.; Vimala, Y.; Bhatnagar, R.K. Characterization of a chitin-binding protein from Bacillus thuringiensis HD-1. PLoS ONE 2013, 8, e66603. [Google Scholar] [CrossRef]
- Agbodjato, N.A.; Noumavo, P.A.; Adjanohoun, A.; Agbessi, L.; Baba-Moussa, L. Synergistic effects of plant growth promoting rhizobacteria and chitosan on in vitro seeds germination, greenhouse growth, and nutrient uptake of maize (Zea mays L.). Biotechnol. Res. Int. 2016, 2016, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Lo Scrudato, M.; Blokesch, M.A. Transcriptional regulator linking quorum sensing and chitin induction to render Vibrio cholerae naturally transformable. Nucleic Acids Res. 2013, 41, 3644–3658. [Google Scholar] [CrossRef]
- Kumar, G.P.; Desai, S.; Moerschbacher, B.M.; Eddine-El Gueddari, N. Seed treatment with chitosan synergizes plant growth promoting ability of Pseudomonas aeruginosa-P17 in sorghum (Sorhum bicolor L.). bioRxiv 2019, 601328. [Google Scholar] [CrossRef]
- Narandelger, T.; Delgermaa, B.; Odonchomeg, B.; Baigalmaa, J.; Sunjidmaa, O. Study of plant pathogen suppression the synergistic effect between biofertilizer and irradiated oligochitosan of tomato. J. Agric. Sci. 2015, 15, 80–85. [Google Scholar] [CrossRef][Green Version]
- Koch, E.; Löffler, I. Pratical characterization of the antimicrobial activity of Streptomyces antimycoticus FZB53. J. Phytopathol. 2009, 157, 235–242. [Google Scholar] [CrossRef]
- Kishore, G.K.; Pande, S. Chitin-supplemented foliar application of chitinolytic Bacillus cereus reduces severity of Botrytis gray mold disease in chickpea under controlled conditions. Lett. Appl. Microbiol. 2007, 44, 98–105. [Google Scholar] [CrossRef]
- Ahmed, A.S.; Ezziyyani, M.; Sánchez, C.P.; Candela, M.E. Effect of chitin on biological control activity of Bacillus spp. and Trichoderma harzianum against root rot disease in pepper (Capsicum annuum) plants. Eur. J. Plant Pathol. 2003, 109, 633–637. [Google Scholar] [CrossRef]
- Manjula, K.; Podile, A.R. Increase in seedling emergence and dry weight of pigeon pea in the field with chitin supplemented formulations of Bacillus subtilis AF1. World J. Microbiol. Biotechnol. 2005, 21, 1057–1062. [Google Scholar] [CrossRef]
- Brzezinska, M.S.; Kalwasińska, A.; Świątczak, J.; Żero, K.; Jankiewicz, U. Exploring the properties of chitinolytic Bacillus isolates for the pathogens biological control. Microb. Pathog. 2020, 148, 104462. [Google Scholar] [CrossRef] [PubMed]


| Source | Chitin Percentage | |
|---|---|---|
| Chitin | Crab | 10 |
| Majoidea (Spider crab) | 16 | |
| Lobster | 17 | |
| Cuttlefish | 20 | |
| Shrimp | 22 | |
| Sea mantis | 24 | |
| Lobster | 32 | |
| Crayfish | 36 | |
| Squid | 40 | |
| Chitosan | Marbled crab (Grapsus marmoratus) | 10 |
| Edible crab | 70 | |
| Grasshopping Lobster (Scyllarus arctus) | 25 | |
| Crawfish (Palinurus vulgaris) | 32 | |
| Prawns (Palaemon fabricius) | 44 | |
| Squid (Loligo vulgaris) | 40 | |
| Mushrooms | ||
| Mucor rouxii | 9.4 | |
| Aspergillus niger | 42 | |
| Aspergillus phoenicis | 23.7 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rkhaila, A.; Chtouki, T.; Erguig, H.; El Haloui, N.; Ounine, K. Chemical Proprieties of Biopolymers (Chitin/Chitosan) and Their Synergic Effects with Endophytic Bacillus Species: Unlimited Applications in Agriculture. Molecules 2021, 26, 1117. https://doi.org/10.3390/molecules26041117
Rkhaila A, Chtouki T, Erguig H, El Haloui N, Ounine K. Chemical Proprieties of Biopolymers (Chitin/Chitosan) and Their Synergic Effects with Endophytic Bacillus Species: Unlimited Applications in Agriculture. Molecules. 2021; 26(4):1117. https://doi.org/10.3390/molecules26041117
Chicago/Turabian StyleRkhaila, Amine, Tarek Chtouki, Hassane Erguig, Noureddine El Haloui, and Khadija Ounine. 2021. "Chemical Proprieties of Biopolymers (Chitin/Chitosan) and Their Synergic Effects with Endophytic Bacillus Species: Unlimited Applications in Agriculture" Molecules 26, no. 4: 1117. https://doi.org/10.3390/molecules26041117
APA StyleRkhaila, A., Chtouki, T., Erguig, H., El Haloui, N., & Ounine, K. (2021). Chemical Proprieties of Biopolymers (Chitin/Chitosan) and Their Synergic Effects with Endophytic Bacillus Species: Unlimited Applications in Agriculture. Molecules, 26(4), 1117. https://doi.org/10.3390/molecules26041117






