Bis(4-methylpiperidine-1-carbodithioato)-lead(II) and Bis(4-benzylpiperidine-1-carbodithioato)-lead(II) as Precursors for Lead Sulphide Nano Photocatalysts for the Degradation of Rhodamine B
Abstract
:1. Introduction
2. Results and Discussion
2.1. Sybtheses
2.2. Molecular Structure of Bis(4-methylpiperidine-1-carbodithioato)-lead(II) and Bis(4-benzylpiperidine-1-carbodithioato)-lead(II)
2.3. FTIR Spectroscopic Studies of the Lead(II) Dithiocarbamate
2.4. Structural and Morphological Studies of the PbS Nanoparticles
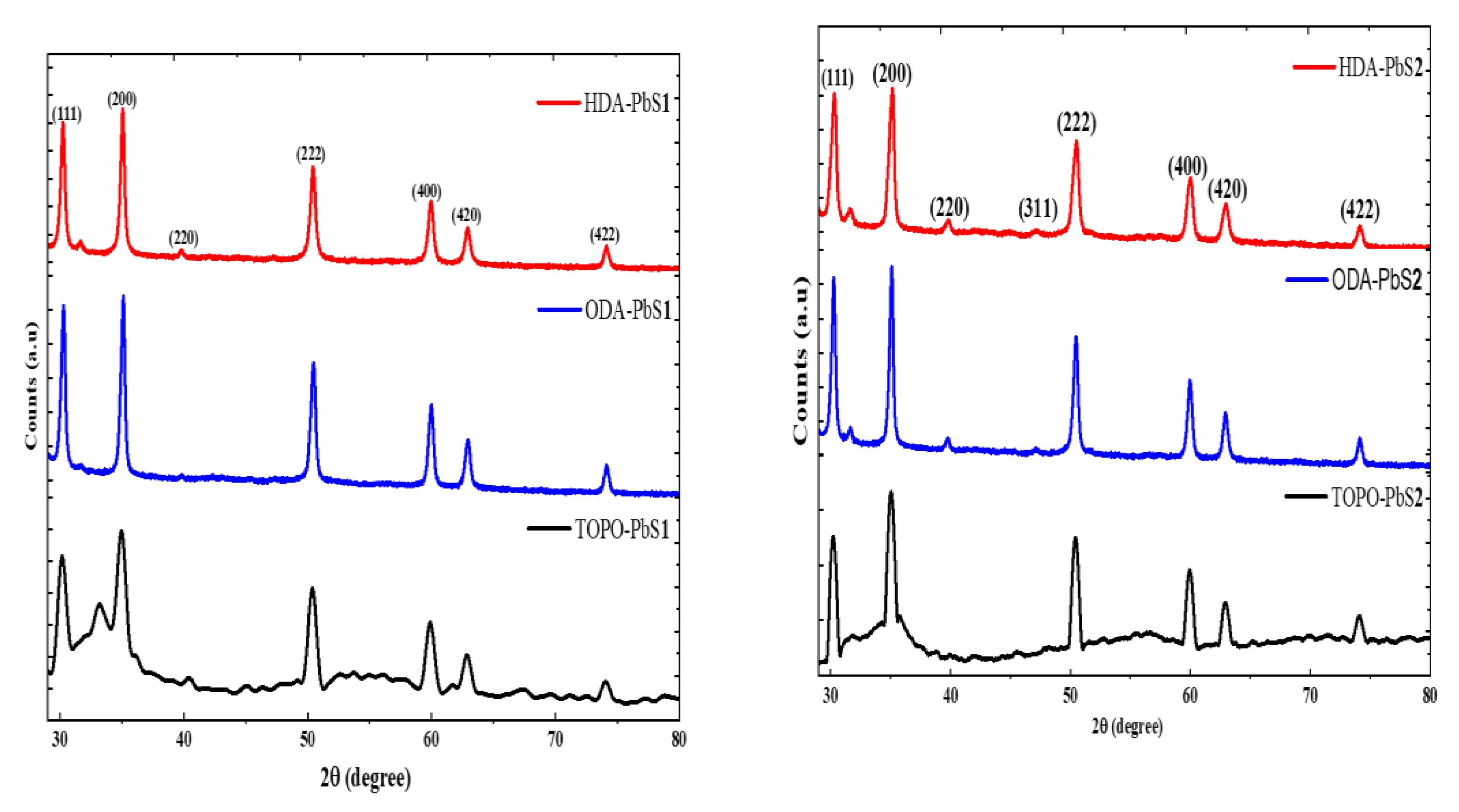
2.4.1. Powder X-ray Diffraction Studies of the PbS Nanoparticles
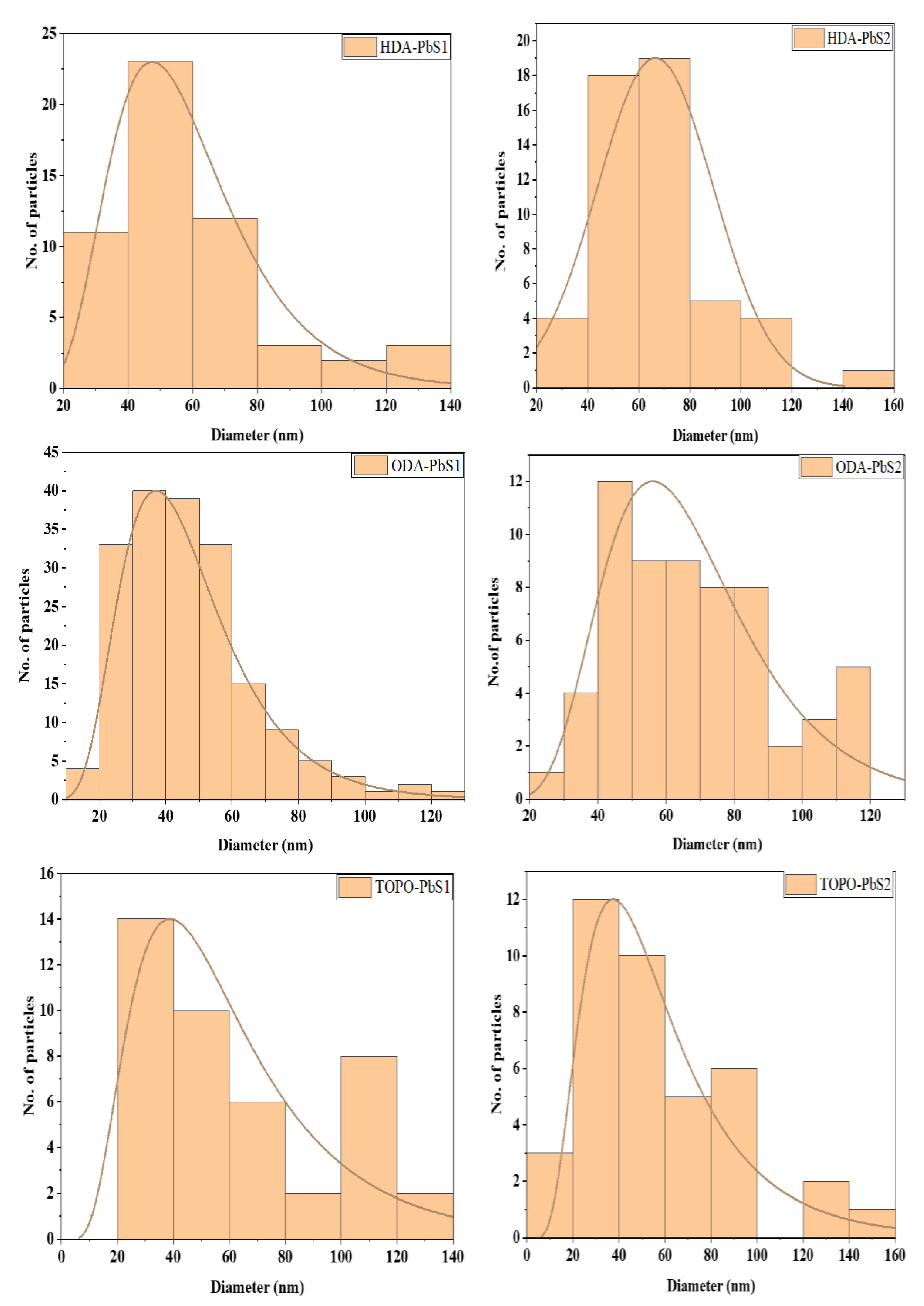
2.4.2. HRTEM Micrographs of the PbS Nanoparticles
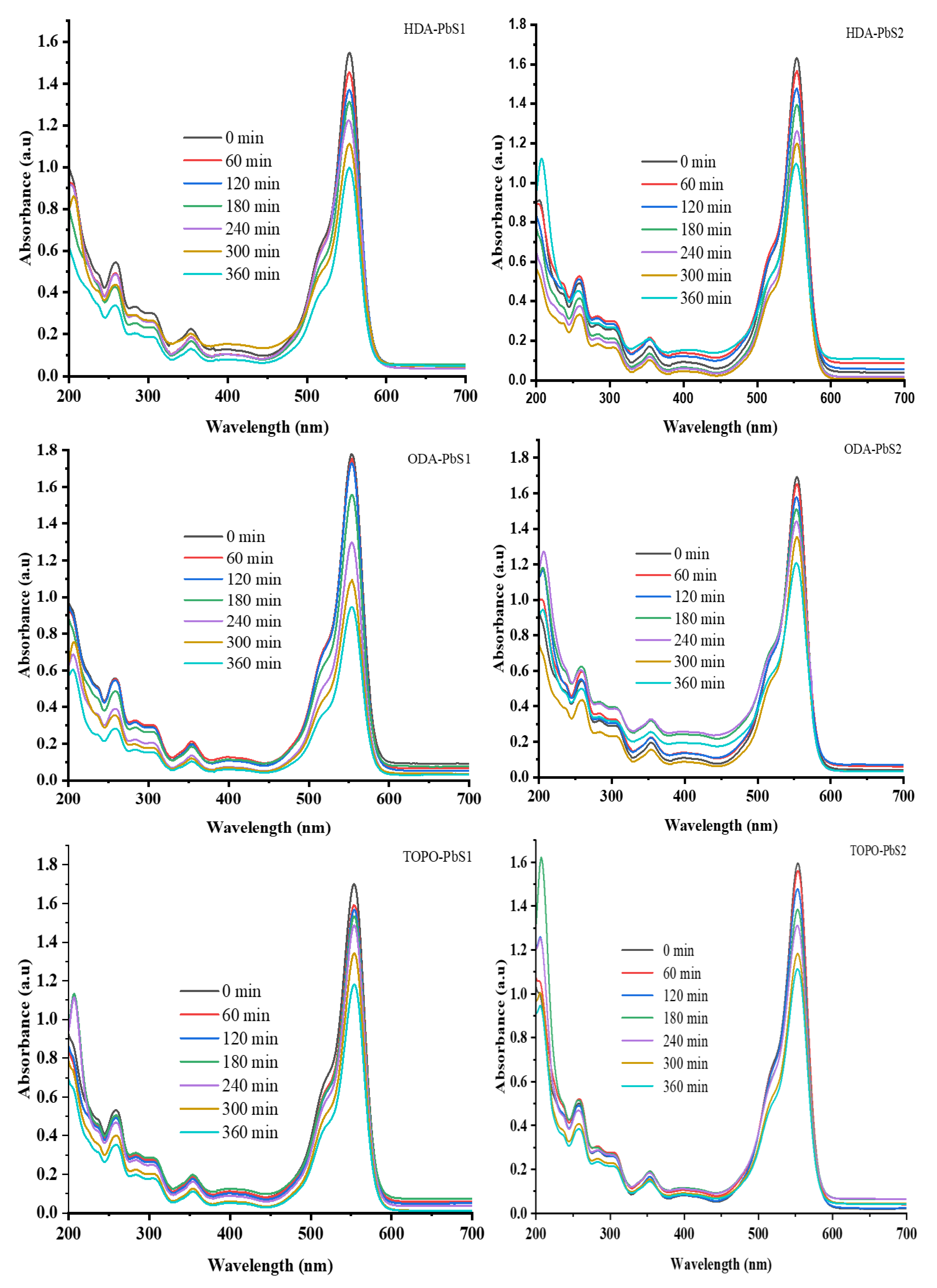
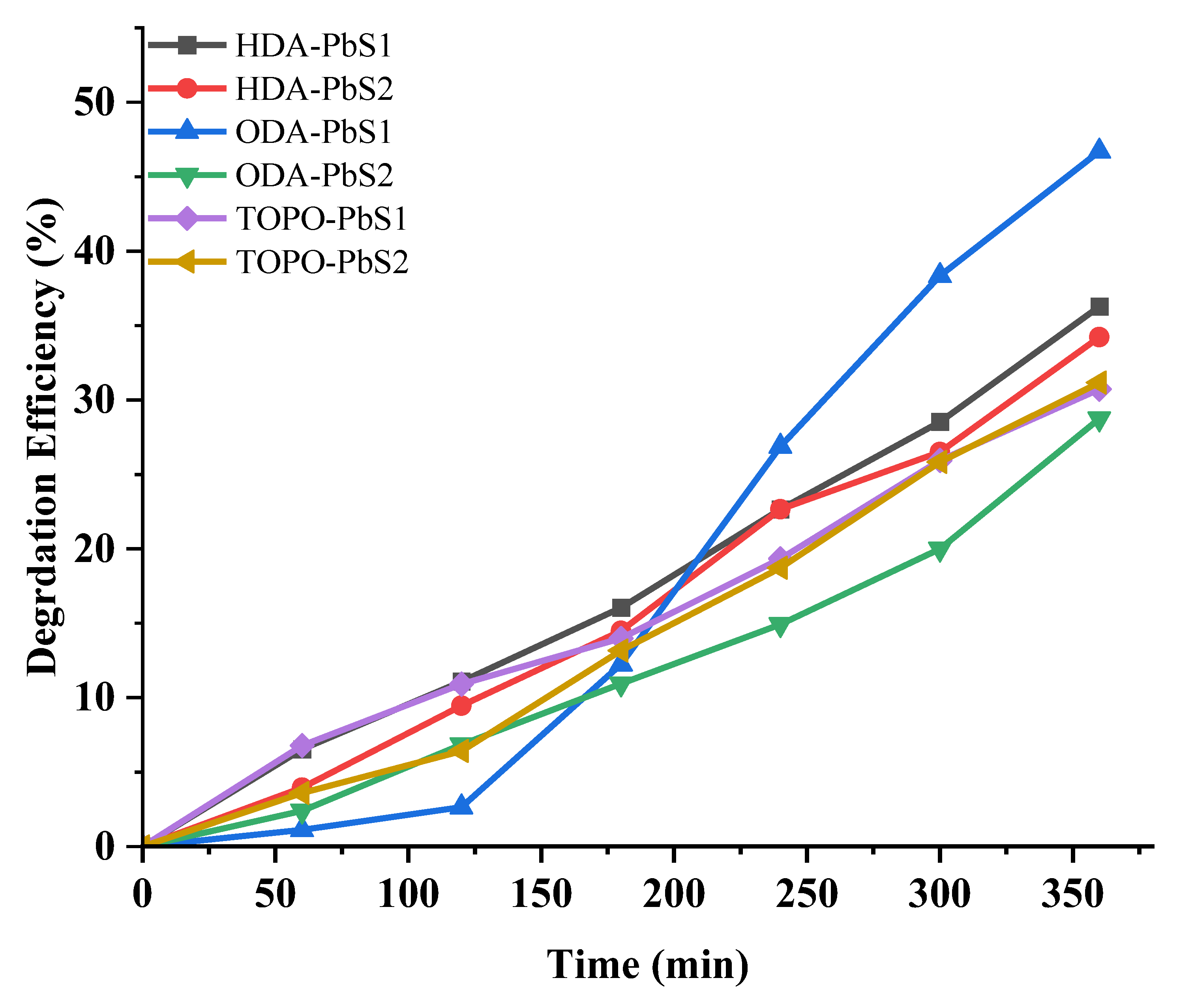
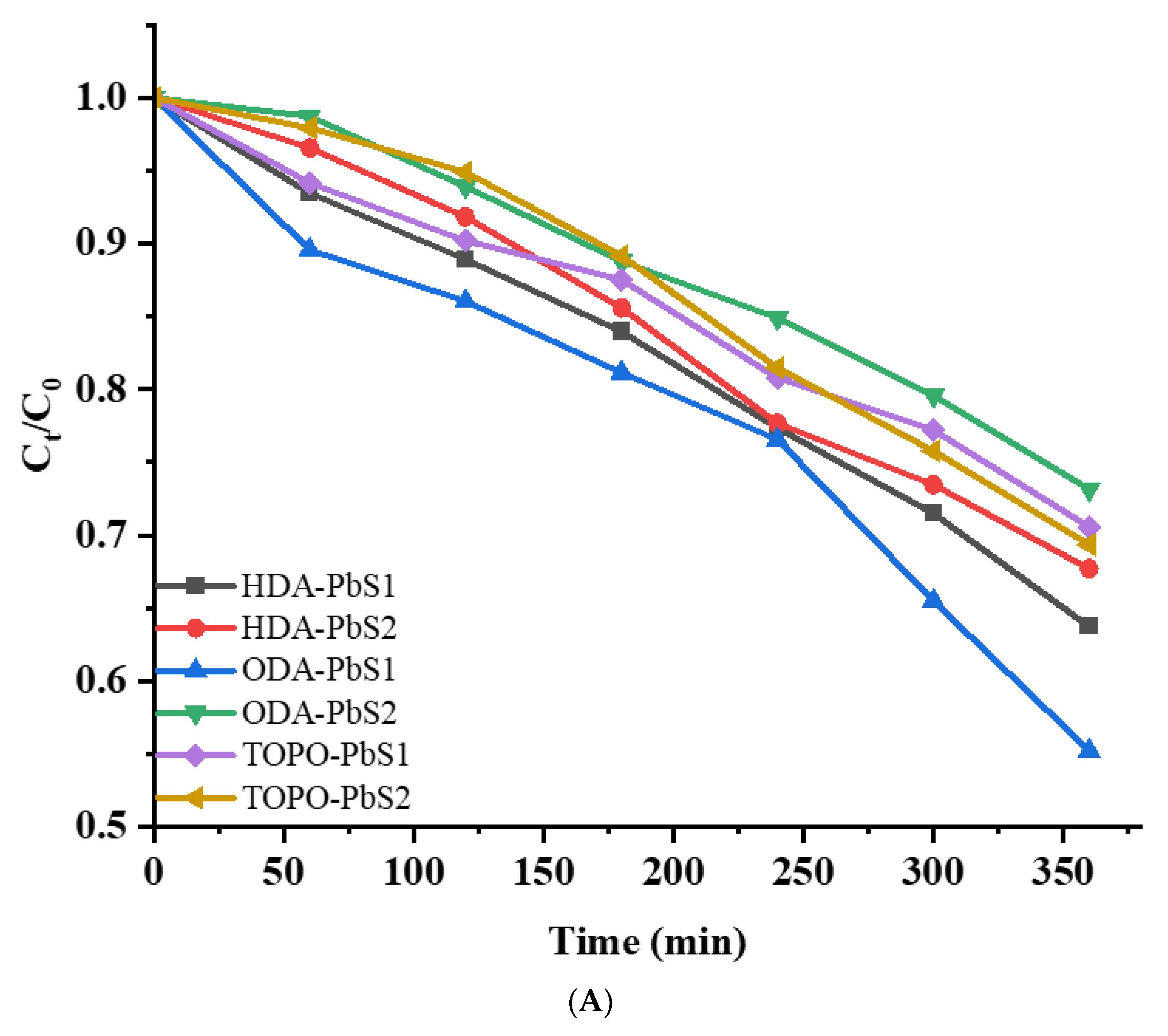
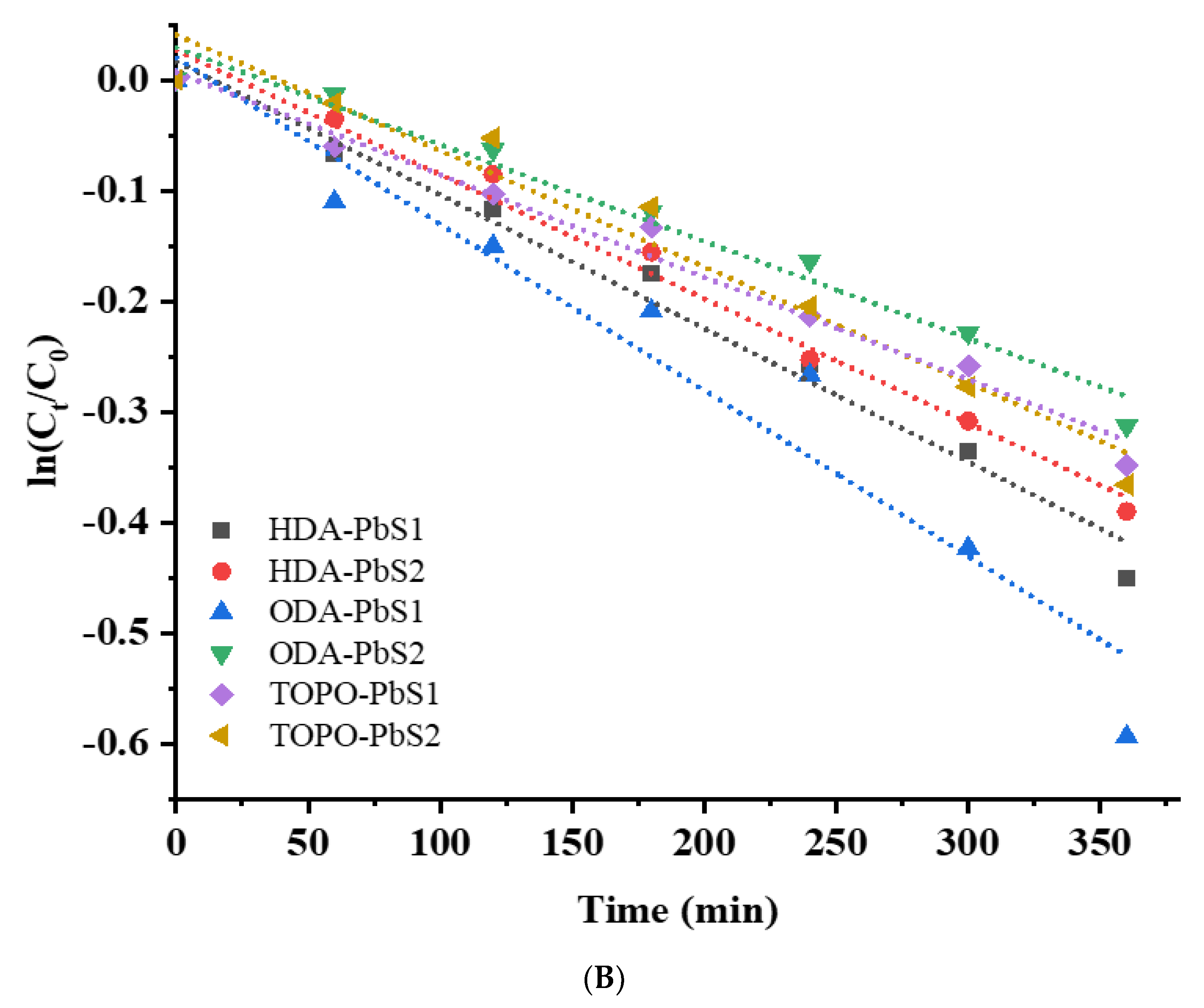
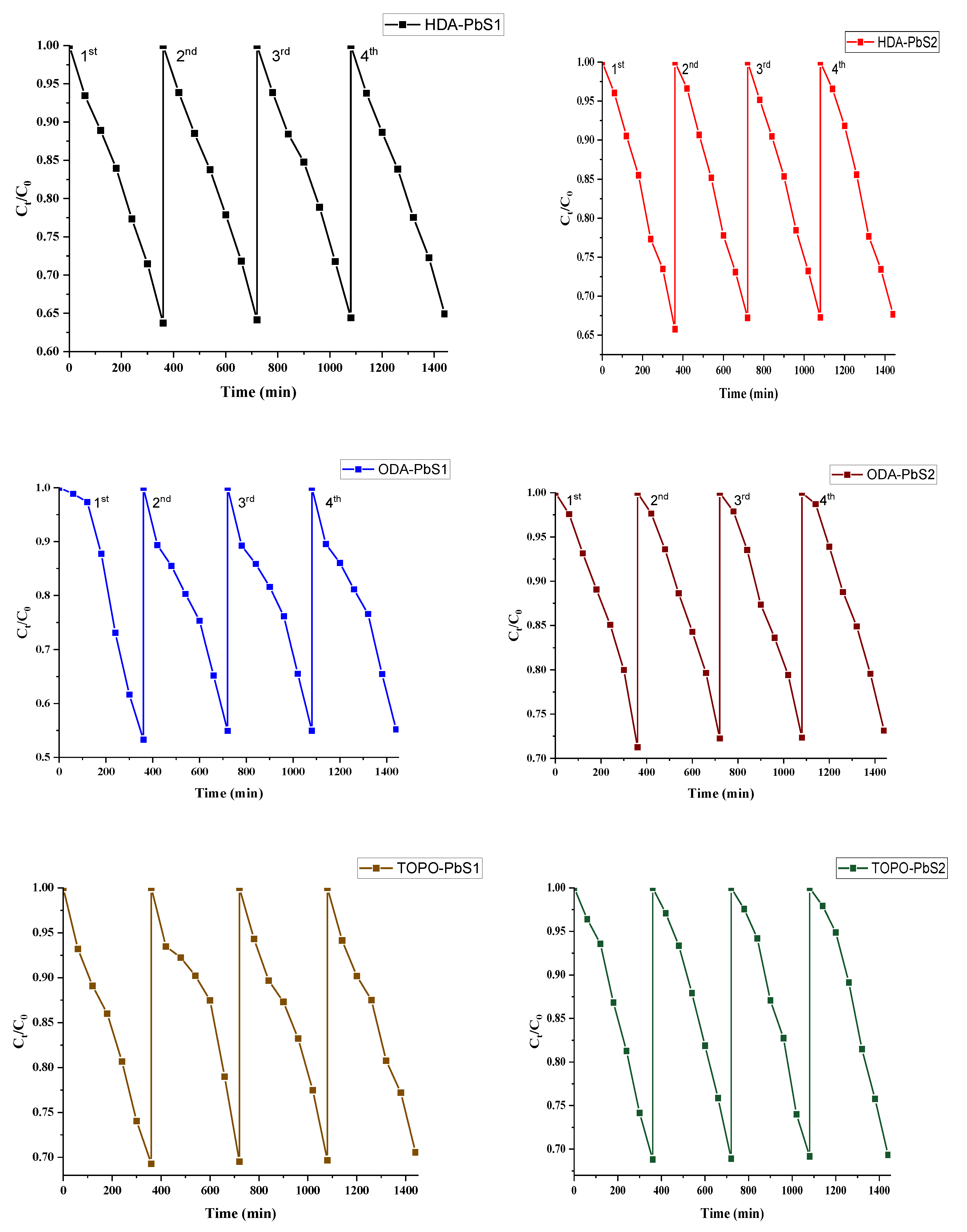
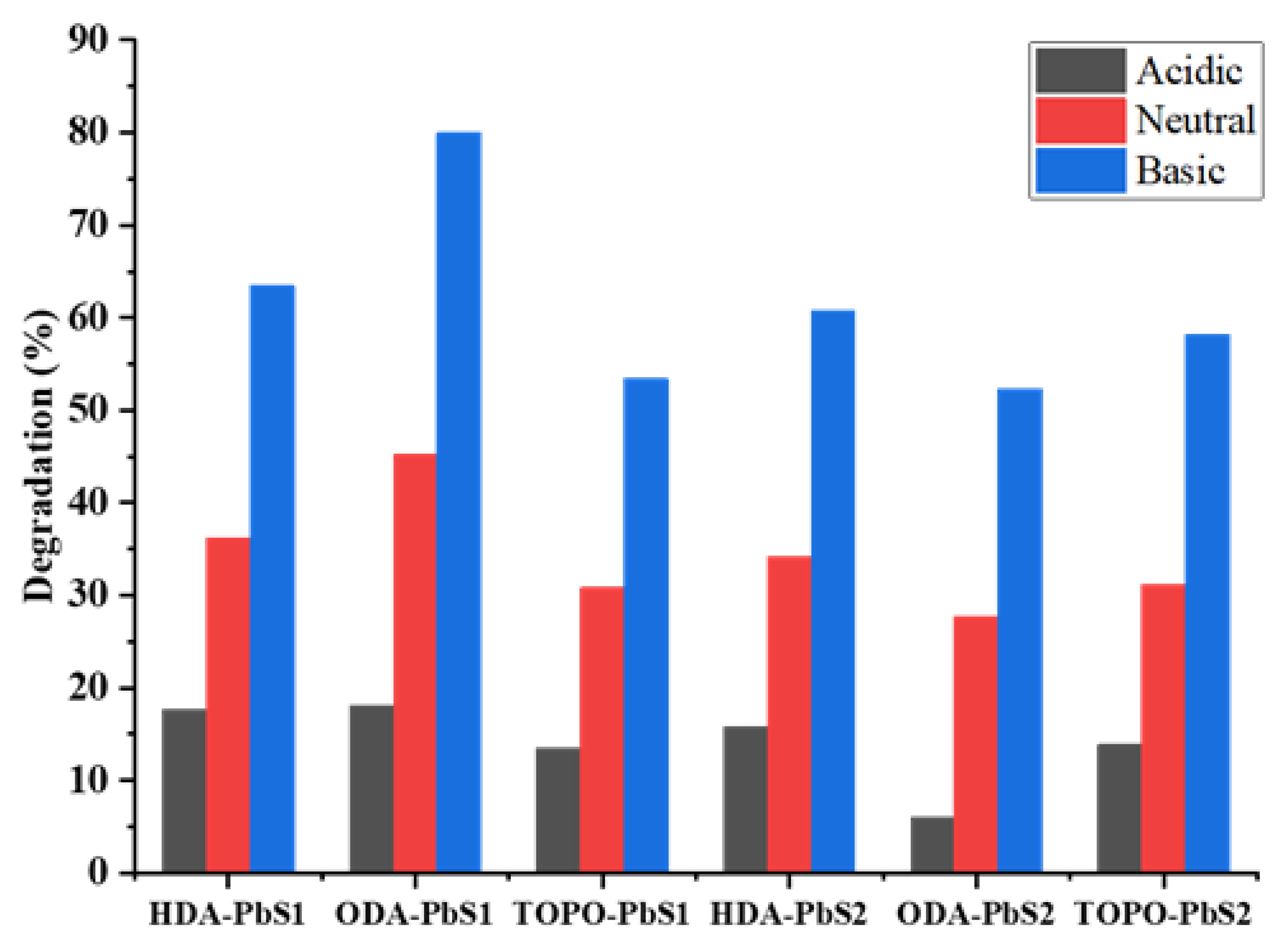
2.5. Photocatalytic Studies
3. Experimental
3.1. Characterization Techniques
3.2. Synthesis of Sodium Salt 4-Methylpiperidine Dithiocarbamate Ligand
3.3. Synthesis of Sodium Salt 4-Benzylpiperidine Dithiocarbamate Ligand
3.4. Synthesis of Pb(II) Dithiocarbamate Complexes
3.5. X-ray Crystallography
3.6. Preparation of PbS Nanoparticles
3.7. Photodegradation of Rhodamine B Dye
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Angeloski, A.; Gentle, A.R.; Scott, J.A.; Cortie, M.B.; Hook, J.M.; Westerhausen, M.T.; Bhadbhade, M.; Baker, A.T.; McDonagh, A.M. From lead(II) dithiocarbamate precursors to a fast response PbS positive temperature coefficient thermistor. Inorg. Chem. 2018, 57, 2132–2140. [Google Scholar] [CrossRef]
- Bederak, D.; Dirin, D.N.; Sukharevska, N.; Momand, J.; Kovalenko, M.V.; Loi, M.A. S-Rich PbS quantum dots: A promising p-type material for optoelectronic devices. Chem. Mater. 2021, 33, 320–326. [Google Scholar] [CrossRef]
- Saraidarov, T.; Reisfeld, R.; Sashchiuk, A.; Lifshitz, E. Synthesis and characterization of PbS nanorods and nanowires. Phys. E Low-Dimens. Syst. Nanostruct. 2007, 37, 173–177. [Google Scholar] [CrossRef]
- Akkerman, Q.A.; Martín-García, B.; Buha, J.; Almeida, G.; Toso, S.; Marras, S.; Bonaccorso, F.; Petralanda, U.; Infante, I.; Manna, L. Ultrathin orthorhombic PbS nanosheets. Chem. Mater. 2019, 31, 8145–8153. [Google Scholar] [CrossRef]
- Kord, M.; Hedayati, K.; Farhadi, M. Green synthesis and characterization of flower-like PbS and metal-doped nanostructures via hydrothermal method. Main Group Met. Chem. 2017, 40, 35–40. [Google Scholar] [CrossRef]
- Saah, S.A.; Boadi, N.O.; Adu-Poku, D.; Wilkins, C. Lead ethyl dithiocarbamates: Efficient single-source precursors to PbS nanocubes. Roy. Soc. Open Sci. 2019, 6, 190943. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Song, C.; Sun, M.; Yin, Y.; Xiao, J.; Dong, W.; Li, C.; Zhang, L. Synthesis of star-shaped lead sulfide (PbS) nanomaterials and theirs gas-sensing properties. Mater. Res. 2016, 19, 1351–1355. [Google Scholar] [CrossRef] [Green Version]
- Wang, D.; Yu, D.; Shao, M.; Liu, J.; Yu, W.; Qian, Y. Dendritic growth of PbS crystals with different morphologies. J. Cryst. Growth 2003, 257, 384–389. [Google Scholar] [CrossRef]
- Li, F.; Huang, X.; Kong, T.; Liu, X.; Qin, Q.; Li, Z. Synthesis and characterization of PbS crystals via a solvothermal route. J. Alloy. Compd. 2009, 485, 554–560. [Google Scholar] [CrossRef]
- Bozkurt, P.A.; Derkuş, B.; Emregül, K.C.; Canel, M. Sonochemical synthesis and characterisation of lead sulfide nanoparticles using different capping agents. J. Chem. Res. 2015, 39, 649–653. [Google Scholar] [CrossRef]
- Kumar, D.; Agarwal, G.; Tripathi, B.; Vyas, D.; Kulshrestha, V. Characterization of PbS nanoparticles synthesized by chemical bath deposition. J. Alloy. Compd. 2009, 484, 463–466. [Google Scholar] [CrossRef]
- Roffey, A.; Hollingsworth, N.; Hogarth, G. Synthesis of ternary sulfide nanomaterials using dithiocarbamate complexes as single source precursors. Nanoscale Adv. 2019, 1, 3056–3066. [Google Scholar] [CrossRef] [Green Version]
- Garcia-Gutierrez, D.F.; Hernandez-Casillas, L.P.; Cappellari, M.V.; Fungo, F.; Martínez-Guerra, E.; García-Gutiérrez, D.I. Influence of the Capping Ligand on the Band Gap and Electronic Levels of PbS Nanoparticles through Surface Atomistic Arrangement Determination. ACS Omega 2018, 3, 393–405. [Google Scholar] [CrossRef] [PubMed]
- Niu, Z.; Li, Y. Removal and Utilization of Capping Agents in Nanocatalysis. Chem. Mater. 2014, 26, 72–83. [Google Scholar] [CrossRef]
- Olenin, A.Y.; Krutyakov, Y.A.; Kudrinskii, A.A.; Lisichkin, G.V. Formation of surface layers on silver nanoparticles in aqueous and water-organic media. Colloid J. 2008, 70, 71–76. [Google Scholar] [CrossRef]
- Jadhav, S.A.; Brunella, V.; Scalarone, D. Polymerizable ligands as stabilizers for nanoparticles. Part. Part. Syst. Charact. 2015, 32, 417–428. [Google Scholar] [CrossRef]
- Mphahlele, L.L.R.; Ajibade, P.A. CdS quantum dots as photocatalyst for methylene blue and methyl red degradation and its electrochemical properties. Int. J. Electrochem. Sci. 2020, 15, 1206–1217. [Google Scholar] [CrossRef]
- Ajibade, P.A.; Solomane, N. Synthesis and crystal structure of bis(thiomorpholinyldithiocarbamato) Zn(II): Structural, optical and photocatalytic studies of ZnS nanoparticles from the complex. J. Coord. Chem. 2020, 73, 1292–1305. [Google Scholar] [CrossRef]
- Oluwalana, A.E.; Ajibade, P.A. Effect of temperature and capping agents on structural and optical properties of tin sulphide nanocrystals. J. Nanotech. 2019, 8235816, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Claudio, E.S.; Godwin, H.A.; Magyar, J.S. Fundamental Coordination Chemistry, Environmental Chemistry, and Biochemistry of Lead(II). In Progress in Inorganic Chemistry; John Wiley & Sons: Hoboken, NJ, USA, 2002; pp. 1–144. [Google Scholar]
- Wu, X.-S.; Tang, Y.-R.; Liu, J.-L.; Wang, L.; Ren, X.-M. Comprehensively understanding the steric hindrance effect on the coordination sphere of Pb2+ ions and photophysical nature of two luminescent Pb(II)-coordination polymers. Dalton Trans. 2019, 48, 13841–13849. [Google Scholar] [CrossRef]
- Breza, M.; Bučinský, L.; Šoralová, S.; Biskupič, S. On the origin of the hemidirected geometry of tetracoordinated lead(II) compounds. Chem. Phys. 2010, 368, 14–19. [Google Scholar] [CrossRef]
- Iwasaki, H.; Hagihara, H. The crystal structure of lead(II) diethyldithiocarbamate. Acta Crystallogr. B 1972, 28, 507–513. [Google Scholar] [CrossRef]
- Ito, M.; Iwasaki, H. Structure of lead(II) N,N-diisopropyldithiocarbamate [bis(N,N-diisopropyldithiocarbamato)lead(II)]. Acta Crystallogr. B 1980, 36, 443–444. [Google Scholar] [CrossRef]
- Akhtar, J.; Malik, M.A.; O’Brien, P.; Helliwell, M. Controlled synthesis of PbS nanoparticles and the deposition of thin films by Aerosol-Assisted Chemical Vapour Deposition (AACVD). J. Mater. Chem. 2010, 20, 6116–6124. [Google Scholar] [CrossRef]
- Afzaal, M.; Ellwood, K.; Pickett, N.L.; O’Brien, P.; Raftery, J.; Waters, J. Growth of lead chalcogenide thin films using single-source precursors. J. Mater. Chem. 2004, 14, 1310–1315. [Google Scholar] [CrossRef]
- Ajibade, P.A.; Oluwalana, A.E.; Andrew, F.P. Morphological studies, photocatalytic activity, and electrochemistry of platinum disulfide nanoparticles from bis(morpholinyl-4-carbodithioato)-platinum(II). ACS Omega 2020, 5, 27142–27153. [Google Scholar] [CrossRef]
- Sathiyaraj, E.; Thirumaran, S. Synthesis and spectral studies on Pb(II) dithiocarbamate complexes containing benzyl and furfuryl groups and their use as precursors for PbS nanoparticles. Spectrochim. Acta A 2012, 97, 575–581. [Google Scholar] [CrossRef]
- Oluwalana, A.E.; Ajibade, P.A. Synthesis and crystal structures of Pb(II) dithiocarbamates complexes: Precursors for PbS nanophotocatalyst. J. Sulfur Chem. 2020, 41, 182–199. [Google Scholar] [CrossRef]
- Ajibade, P.; Mbese, J.; Omondi, B. Group 12 dithiocarbamate complexes: Synthesis, characterization and X-ray crystal structures of Zn(II) and Hg(II) complexes and their use as precursors for metal sulfide nanoparticles. Synth. React. Inorg. Met. 2016, 47, 202–212. [Google Scholar] [CrossRef]
- Singh, H.L.; Singh, J.B.; Bhanuka, S. Synthesis, spectral, DFT, and antimicrobial studies of tin(II) and lead(II) complexes with semicarbazone and thiosemicarbazones derived from (2-hydroxyphenyl)(pyrrolidin-1-yl)methanone. J. Coord. Chem. 2016, 69, 343–353. [Google Scholar] [CrossRef]
- Meng, W.; Yuan, W.; Wu, Z.; Wang, X.; Xu, W.; Wang, L.; Zhang, Q.; Zhang, C.; Wang, J.; Song, Q. Mechanochemical synthesis of lead sulfide (PbS) nanocrystals from lead oxide. Powder Technol. 2019, 347, 130–135. [Google Scholar] [CrossRef]
- Mamiyev, Z.; Balayeva, N. Preparation and optical studies of PbS nanoparticles. Opt. Mater. 2015, 46, 522–525. [Google Scholar] [CrossRef]
- Nanda, K.K.; Maisels, A.; Kruis, F.E.; Fissan, H.; Stappert, S. Higher surface energy of free nanoparticles. Phys. Rev. Lett. 2003, 91, 106102. [Google Scholar] [CrossRef]
- Tang, E.; Cheng, G.; Ma, X.; Pang, X.; Zhao, Q. Surface modification of zinc oxide nanoparticle by PMAA and its dispersion in aqueous system. Appl. Surf. Sci. 2006, 252, 5227–5232. [Google Scholar] [CrossRef]
- Gupta, D.; Chauhan, R.; Kumar, N.; Singh, V.; Srivastava, V.C.; Mohanty, P.; Mandal, T.K. Enhancing photocatalytic degradation of quinoline by ZnO:TiO2 mixed oxide: Optimization of operating parameters and mechanistic study. J. Environ. Manag. 2020, 258, 110032. [Google Scholar] [CrossRef] [PubMed]
- Arularasu, M. Effect of organic capping agents on the optical and photocatalytic activity of mesoporous TiO2 nanoparticles by sol–gel method. JSN Appl. Sci. 2019, 1, 393. [Google Scholar] [CrossRef] [Green Version]
- Gao, H.; Lu, B.; Liu, F.; Liu, Y.; Zhao, X. Photocatalytical properties and theoretical analysis of N, Cd-Codoped TiO2 synthesized by thermal decomposition method. Inter. J. Photoenergy 2012, 2012, 453018. [Google Scholar] [CrossRef]
- Estrada-Flores, S.; Martínez-Luévanos, A.; Perez-Berumen, C.M.; García-Cerda, L.A.; Flores-Guia, T.E. Relationship between morphology, porosity, and the photocatalytic activity of TiO2 obtained by sol–gel method assisted with ionic and nonionic surfactants. Bol. Soc. Esp. Ceram. V. 2020, 59, 209–218. [Google Scholar] [CrossRef]
- Ajibade, P.A.; Mbuyazi, T.B.; Oluwalana, A.E. Lead sulphide nanoparticles as photocatalyst for the degradation of methylene blue: Effects of ph, time, adsorption kinetics and recyclability studies. J. Inorg. Organomet. Polym. Mater. 2021, 31, 2197–2208. [Google Scholar] [CrossRef]
- Chen, S.; Zhang, X.; Zhang, Q.; Tan, W. Trioctylphosphine as both solvent and stabilizer to synthesize CdS nanorods. Nanoscale Res. Lett. 2009, 4, 1159. [Google Scholar] [CrossRef] [Green Version]
- Ahmad, D.N.; Chieng, B.W.; Ibrahim, N.A.; Talib, Z.A.; Muhamad, E.N.; Abidin, Z.Z. Functionalizing graphene oxide with alkylamine by gamma-ray irradiation method. Nanomaterials 2017, 7, 135. [Google Scholar] [CrossRef] [Green Version]
- Ma, J. Preparation and characterization of ZrO2 nanoparticles capped by trioctylphosphine oxide (TOPO). J. Wuhan Univ. Technol. 2011, 26, 611–614. [Google Scholar] [CrossRef]
- Ajibade, P.A.; Oluwalana, A.E. Structural, optical, photocatalytic and electrochemical studies of PbS nanoparticles. J. Nano Res. 2020, 61, 18–31. [Google Scholar] [CrossRef]
- Oluwalana, A.E.; Ajibade, P.A. Structural, optical, and photocatalytic studies of hexadecylamine-capped lead sulfide nanoparticles. Inter. J. Industr. Chem. 2020, 11, 249–260. [Google Scholar] [CrossRef]
- Oluwalana, A.E.; Ajibade, P.A. Structural, optical, and photocatalytic studies of oleylamine capped PbS nanoparticles. Opt. Quant. Electr. 2021, 53, 1–13. [Google Scholar] [CrossRef]
- Suganya, M.; Balu, A.R.; Balamurugan, S.; Srivind, J.; Narasimman, V.; Manjula, N.; Rajashree, C.; Nagarethinam, V.S. Photoconductive, photocatalytic and antifungal properties of PbS:Mo nanoparticles synthesized via precipitation method. Surf. Inter. 2018, 13, 148–156. [Google Scholar] [CrossRef]
- Chiu, Y.-H.; Chang, T.-F.M.; Chen, C.-Y.; Sone, M.; Hsu, Y.-J. Mechanistic insights into photodegradation of organic dyes using heterostructure photocatalysts. Catalysts 2019, 9, 430. [Google Scholar] [CrossRef] [Green Version]
- Solomane, N.; Ajibade, P.A. Synthesis and crystal structure of bis(thiomorpholinyldithiocarbamato)Cu(II) complex and its use as precursor for CuS nanoparticles photocatalyst for the degradation of organic dyes. J. Sulfur Chem. 2021, 42, 167–179. [Google Scholar] [CrossRef]
- Antoniou, M.; Dionysiou, D. Application of immobilized titanium dioxide photocatalysts for the degradation of creatinine and phenol, model organic contaminants found in NASA’s spacecrafts wastewater streams. Catal. Today 2007, 124, 215–223. [Google Scholar] [CrossRef]
- Hariprasad, N.; Anju, S.G.; Yesodharan, E.P. Sunlight induced removal of rhodamine B from water through semiconductor photocatalysis: Effects of adsorption, reaction conditions and additives. Res. J. Mater. Sci. 2013, 1, 9–17. [Google Scholar]
- Dolomanov, O.V.; Bourhis, L.J.; Gildea, R.J.; Howard, J.A.K.; Puschmann, H. OLEX2: A complete structure solution, refinement and analysis program. J. Appl. Crystallogr. 2009, 42, 339–341. [Google Scholar] [CrossRef]
- Sheldrick, G. A short history of SHELX. Acta Crystallogr. A 2008, 64, 112–122. [Google Scholar] [CrossRef] [Green Version]
- Sheldrick, G. Crystal structure refinement with SHELXL. Acta Crystallogr. C 2015, 71, 3–8. [Google Scholar] [CrossRef]
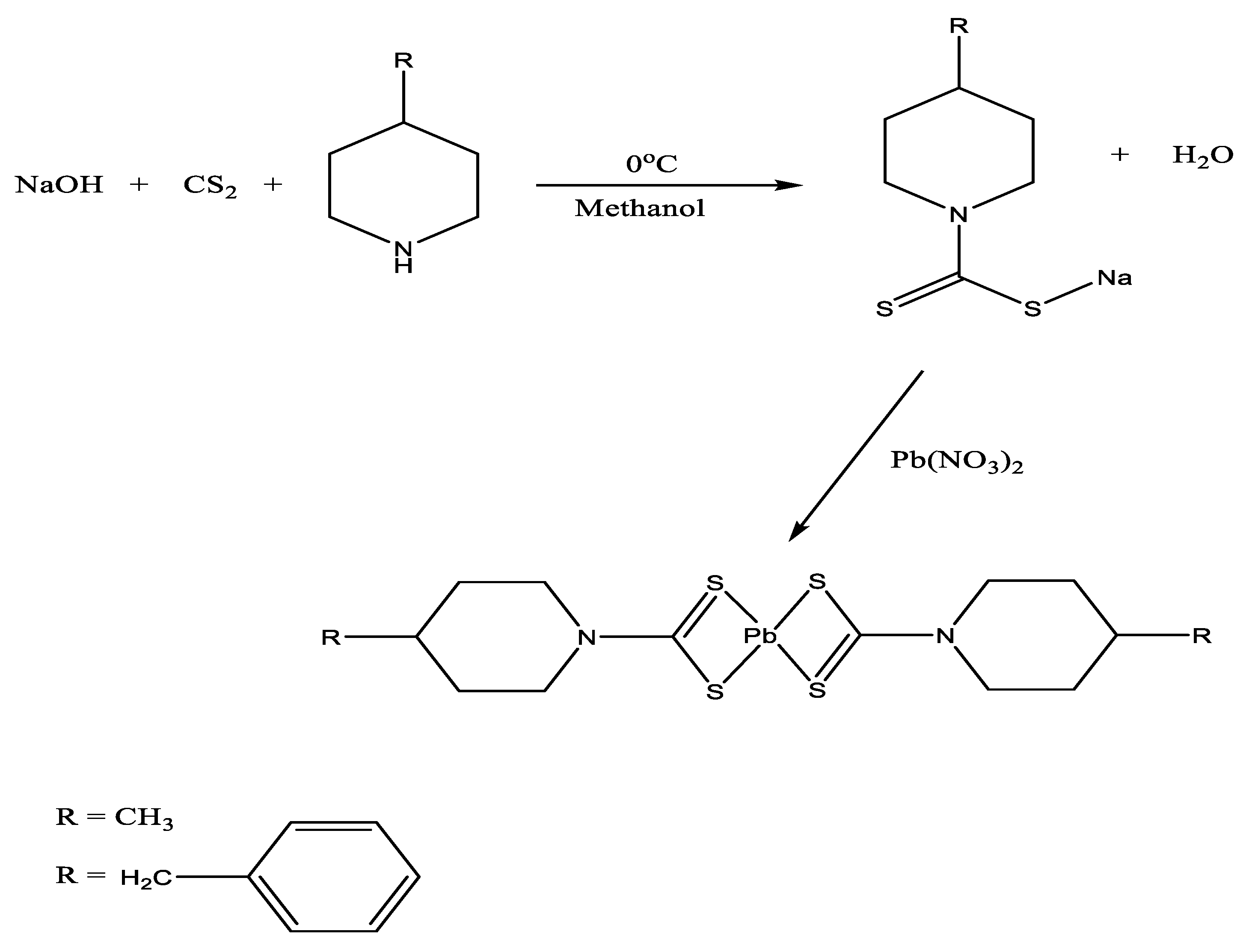
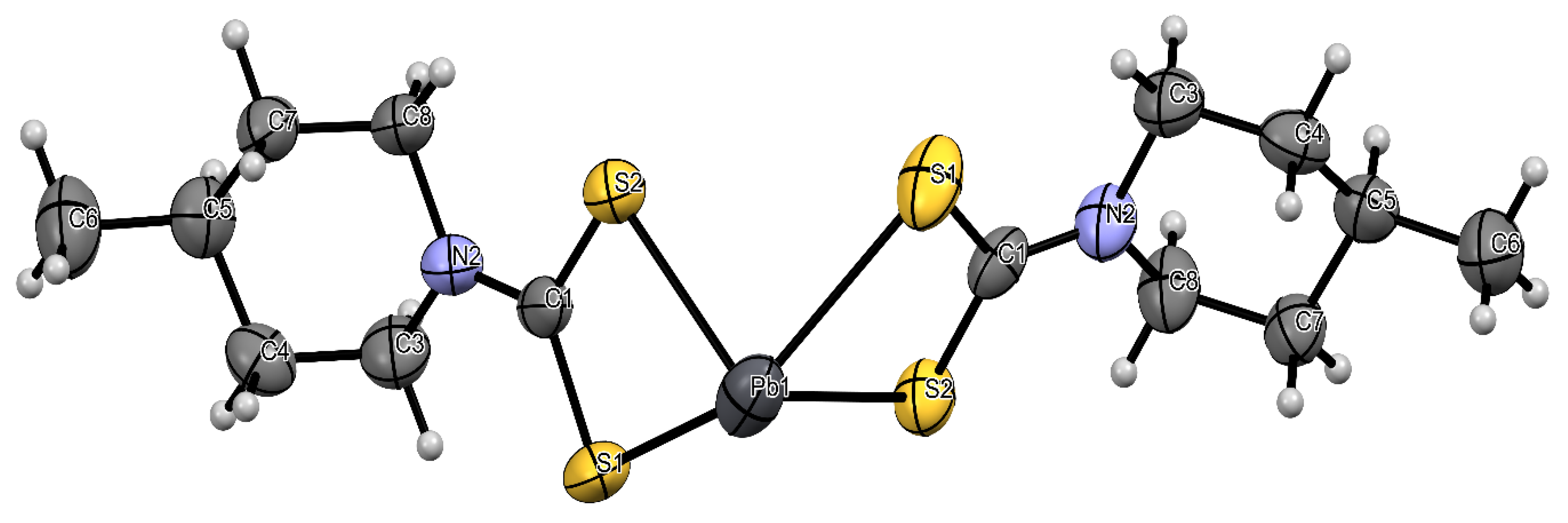
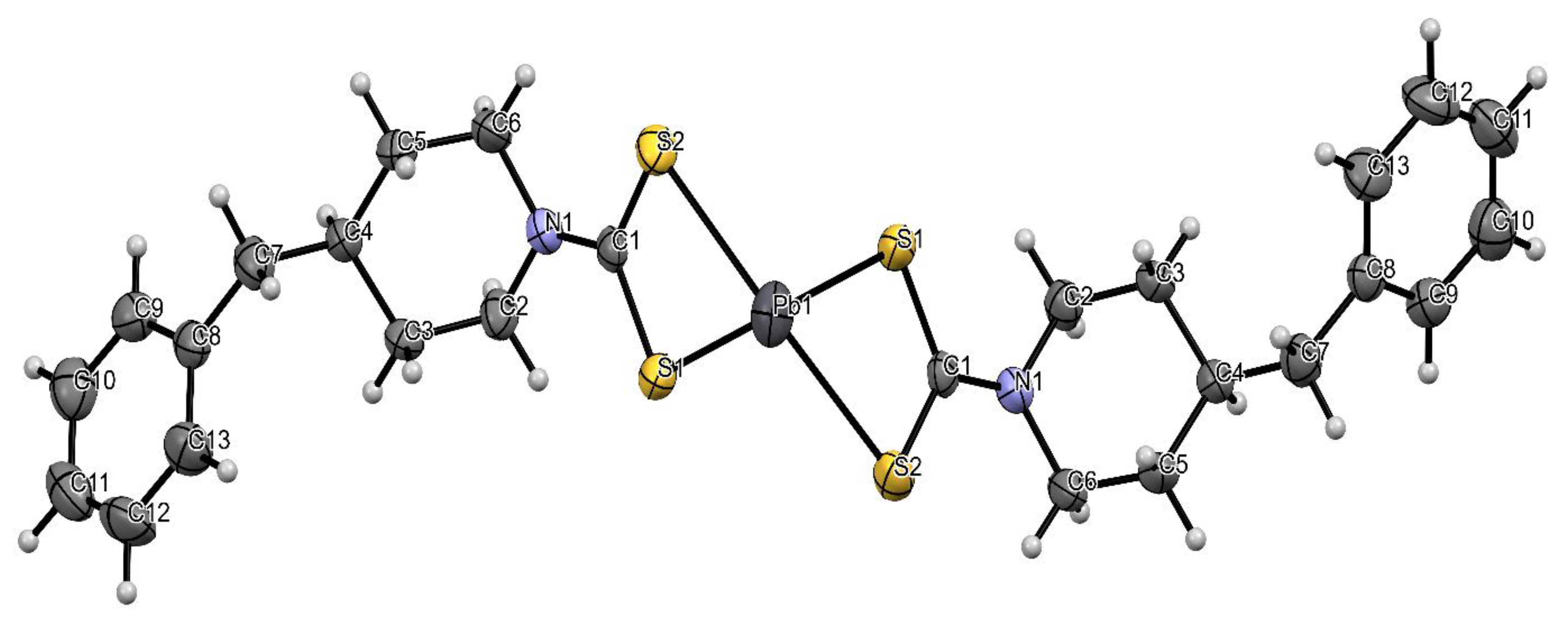
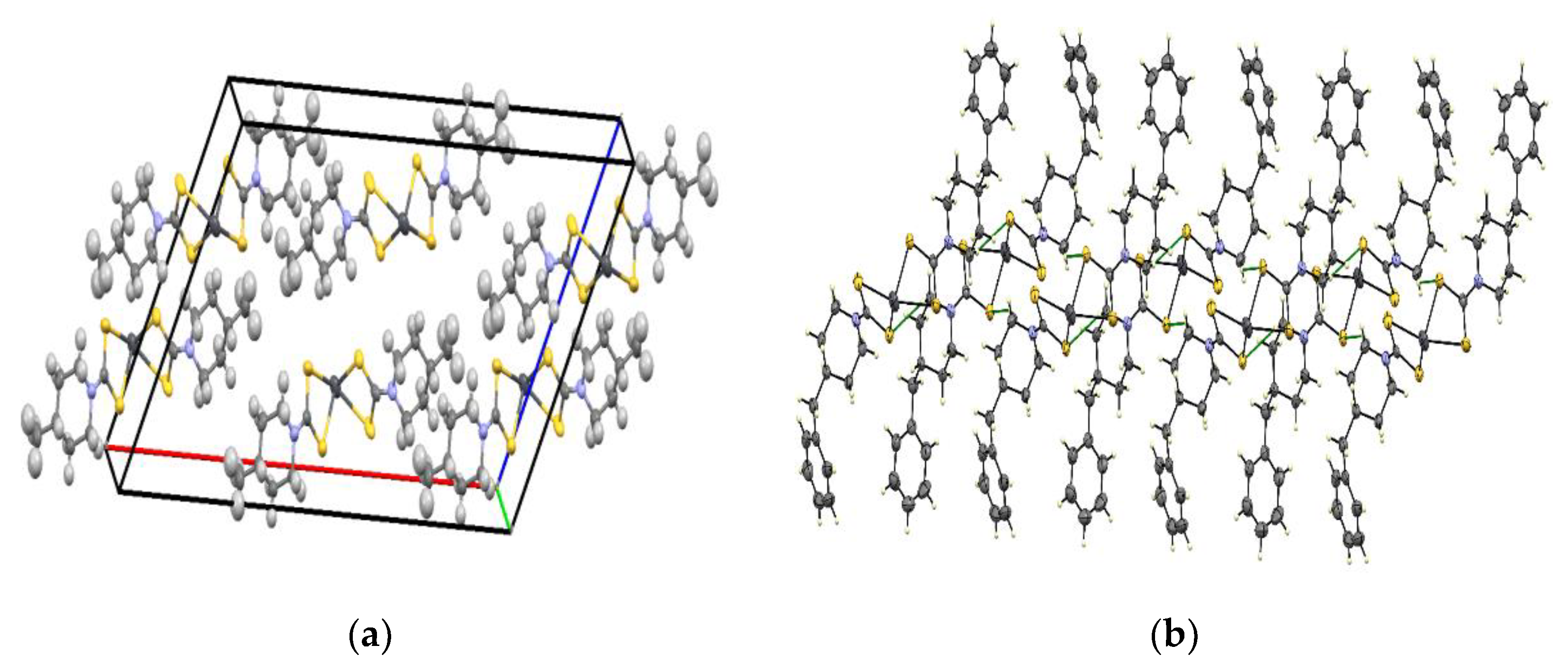

| Compound | [Pb(4-Mpipdtc)2] | [Pb(4-Bpipdtc)2] |
|---|---|---|
| SFormula | C14H24N2PbS4 | C26H32N2PbS4 |
| Dcalc./g cm−3 | 1.951 | 1.724 |
| μ/mm−1 | 21.417 | 15.012 |
| Formula Weight | 555.78 | 707.96 |
| Size/mm3 | 0.505 × 0.190 × 0.055 | 0.335 × 0.270 × 0.120 |
| T/K | 102(2) | 102(2) |
| Crystal System | Monoclinic | Monoclinic |
| Space Group | C2/c | C2/n |
| a/Å | 29.5086(10) | 25.5177(17) |
| b/Å | 4.6830(2) | 9.1660(5) |
| c/Å | 15.6621(5) | 12.0217(7) |
| α/° | 90 | 90 |
| β/° | 119.0530(10) | 104.015(4) |
| γ/° | 90 | 90 |
| V/Å3 | 1891.99(12) | 2728.1(3) |
| Z | 4 | 4 |
| Z’ | ||
| Θmin/° | 3.427 | 0.335 |
| Θmax/° | 72.650 | 72.234 |
| Measured Refl. | 16061 | 26753 |
| Independent Refl. | 1838 | 2679 |
| Reflections Used | 100 | 123 |
| Rint | 0.0547 | 0.0461 |
| Parameters | 97 | 150 |
| Largest Peak | 0.452 | 0.399 |
| Deepest Hole | −2.382 | −1.393 |
| GooF | 1.108 | 1.138 |
| wR2 (all data) | 0.0870 | 0.0563 |
| wR2 | 0.0856 | 0.561 |
| R1 (all data) | 0.0349 | 0.0229 |
| R1 | 0.0338 | 0.0227 |
| [Pb(4-Mpipdtc)2] | [Pb(4-Bpipdtc)2] | ||
|---|---|---|---|
| Bond | Length(Å) | Bonds | Length(Å) |
| Pb1—S2 | 2.7097(10) | Pb1—S1 | 2.6614(7) |
| Pb1—S2 | 2.7097(10) | Pb1—S1 | 2.6614(7) |
| Pb1—S1 | 2.8831(13) | Pb1—S2 | 2.8778(8) |
| Pb1—S1 | 2.8831(13) | Pb1—S2 | 2.8779(8) |
| S2—C1 | 1.736(4) | S2—C1 | 1.710(3) |
| S1—C1 | 1.705(4) | S1—C1 | 1.738(3) |
| C1—N2 | 1.338(6) | N1—C1 | 1.323(4) |
| Bond | Angle(°) | Bonds | Angle(°) |
| S2—Pb1—S2 | 97.63(5) | S1—Pb1—S1 | 91.56(3) |
| S2—Pb1—S1 | 64.02(3) | S1—Pb1—S2 | 84.75(2) |
| S21—Pb1—S1 | 83.57(3) | S11—Pb1—S2 | 64.68(2) |
| S2—Pb1—S1 | 83.57(3) | S1—Pb1—S2 | 64.68(2) |
| S21—Pb1—S1 | 64.02(3) | S11—Pb1—S2 | 84.76(2) |
| S1—Pb1—S1 | 130.63(5) | S21—Pb1—S2 | 136.31(3) |
| C1—S2—Pb1 | 90.67(14) | C1—S1—Pb1 | 91.46(9) |
| C1—S1—Pb1 | 85.60(15) | C1—S2—Pb1 | 84.94(9) |
| Compound | Degradation (%) | Rate Constant (min−1) | R2 |
|---|---|---|---|
| HDA-PbS1 | 36.18 | 0.00121 | 0.9816 |
| HDA-PbS2 | 34.22 | 0.00116 | 0.9817 |
| ODA-PbS1 | 45.28 | 0.01981 | 0.9413 |
| ODA-PbS2 | 27.75 | 8.85 × 10−4 | 0.9695 |
| TOPO-PbS1 | 30.81 | 9.53 × 10−4 | 0.9805 |
| TOPO-PbS2 | 31.17. | 0.00106 | 0.9731 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mbuyazi, T.B.; Ajibade, P.A. Bis(4-methylpiperidine-1-carbodithioato)-lead(II) and Bis(4-benzylpiperidine-1-carbodithioato)-lead(II) as Precursors for Lead Sulphide Nano Photocatalysts for the Degradation of Rhodamine B. Molecules 2021, 26, 7251. https://doi.org/10.3390/molecules26237251
Mbuyazi TB, Ajibade PA. Bis(4-methylpiperidine-1-carbodithioato)-lead(II) and Bis(4-benzylpiperidine-1-carbodithioato)-lead(II) as Precursors for Lead Sulphide Nano Photocatalysts for the Degradation of Rhodamine B. Molecules. 2021; 26(23):7251. https://doi.org/10.3390/molecules26237251
Chicago/Turabian StyleMbuyazi, Thandi B., and Peter A. Ajibade. 2021. "Bis(4-methylpiperidine-1-carbodithioato)-lead(II) and Bis(4-benzylpiperidine-1-carbodithioato)-lead(II) as Precursors for Lead Sulphide Nano Photocatalysts for the Degradation of Rhodamine B" Molecules 26, no. 23: 7251. https://doi.org/10.3390/molecules26237251
APA StyleMbuyazi, T. B., & Ajibade, P. A. (2021). Bis(4-methylpiperidine-1-carbodithioato)-lead(II) and Bis(4-benzylpiperidine-1-carbodithioato)-lead(II) as Precursors for Lead Sulphide Nano Photocatalysts for the Degradation of Rhodamine B. Molecules, 26(23), 7251. https://doi.org/10.3390/molecules26237251






