α-Amyrin and β-Amyrin Isolated from Celastrus hindsii Leaves and Their Antioxidant, Anti-Xanthine Oxidase, and Anti-Tyrosinase Potentials
Abstract
:1. Introduction
2. Results
2.1. Isolation of α-Amyrin and β-Amyrin from Leaves of C. hindsii
2.2. Identification of α-Amyrin and β-Amyrin by GC-MS and ESI-MS
2.3. Elucidation of α-Amyrin and β-Amyrin by NMR
2.4. Quantitative Analysis of α-Amyrin and β-Amyrin in C. hindsii Leaves
2.5. Antioxidant, Anti-Xanthine Oxidase, and Anti-Tyrosinase of α-Amyrin and β-Amyrin from C. hindsii
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Preparation of Plant Extract
4.3. Fractionation of Ethyl Acetate Extract
4.4. Identification α-Amyrin and β-Amyrin by Gas Chromatography-Mass Spectrometry (GC-MS)
4.5. Electrospray Ionization-Mass Spectrometry (ESI-MS) Analysis
4.6. Nuclear Magnetic Resonance (NMR) Data of α-Amyrin and β-Amyrin
4.7. Xanthine Oxidase (XO) Inhibitory Activity
4.8. 2,2-Diphenyl-1-picrylhydrazyl (DPPH) Free Radical Scavenging Activity
4.9. 2,20-Azinobis (3-Ethylbenzothiazoline-6-sulfonic Acid) (ABTS) Cation Scavenging Activity
4.10. Tyrosinase Inhibition
4.11. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Ly, T.N.; Shimoyamada, M.; Yamauchi, R. Isolation and characterization of rosmarinic acid oligomers in Celastrus hindsii Benth leaves and their antioxidative activity. J. Agric. Food Chem. 2006, 54, 3786–3793. [Google Scholar] [CrossRef]
- Viet, T.D.; Xuan, T.D.; Van, T.M.; Andriana, Y.; Rayee, R.; Tran, H.D. Comprehensive fractionation of antioxidants and GC-MS and ESI-MS fingerprints of Celastrus hindsii leaves. Medicines 2019, 6, 64. [Google Scholar] [CrossRef] [Green Version]
- Kuo, Y.H.; Chen, C.F.; Kuo, L.M.Y.; King, M.L.; Chen, C.F.; Lee, K.H. Celahinine A, a new sesquiterpene pyridine alkaloid from Celastrus hindsii. J. Nat. Prod. 1995, 58, 1735–1738. [Google Scholar] [CrossRef]
- Pham, D.C.; Nguyen, H.C.; Nguyen, T.H.L.; Ho, H.L.; Trinh, T.K.; Riyaphan, J.; Weng, C.F. Optimization of ultrasound-assisted extraction of flavonoids from Celastrus hindsii leaves using response surface methodology and evaluation of their antioxidant and antitumor activities. BioMed Res. Int. 2020, 2020, 3497107. [Google Scholar] [CrossRef] [Green Version]
- Da Silva Júnior, W.F.; Bezerra de Menezes, D.L.; De Oliveira, L.C.; Koester, L.S.; Oliveira de Almeida, P.D.; Lima, E.S.; Neves de Lima, Á.A. Inclusion complexes of β and HPβ-cyclodextrin with α, β amyrin and in vitro anti-inflammatory activity. Biomolecules 2019, 9, 241. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Barros, F.W.A.; Bandeira, P.N.; Lima, D.J.B.; Meira, A.S.; de Farias, S.S.; Albuquerque, M.R.J.R.; dos Santos, H.S.; Lemos, T.L.G.; de Morais, M.O.; Costa-Lotufo, L.V. Amyrin esters induce cell death by apoptosis in HL-60 leukemia cells. Bioorg. Med. Chem. 2011, 19, 1268–1276. [Google Scholar] [CrossRef]
- Lima, E.M.; Nascimento, A.M.; Lenz, D.; Scherer, R.; Meyrelles, S.S.; Boëchat, G.A.P.; Andrade, T.U.; Endringer, D.C. Triterpenes from the Protium heptaphyllum resin-chemical composition and cytotoxicity. Rev. Bras. Farmacogn. 2014, 24, 399–407. [Google Scholar] [CrossRef] [Green Version]
- Aragão, G.F.; Carneiro, L.M.V.; Junior, A.P.F.; Vieira, L.C.; Bandeira, P.N.; Lemos, T.L.G.; Viana, G.S.D.B. A possible mechanism for anxiolytic and antidepressant effects of alpha- and beta-amyrin from Protium heptaphyllum (Aubl.) March. Pharmacol. Biochem. Behav. 2006, 85, 827–834. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, F.A.; Vieira-Júnior, G.M.; Chaves, M.H.; Almeida, F.R.C.; Florêncio, M.G.; Lima, R.C.P.; Silva, R.M.; Santos, F.A.; Rao, V.S.N. Gastroprotective and anti-inflammatory effects of resin from Protium paraphylum in mice and rats. Pharmacal. Res. 2004, 49, 105–111. [Google Scholar] [CrossRef]
- Yasumoto, S.; Seki, H.; Shimizu, Y.; Fukushima, E.O.; Muranaka, T. Functional characterization of CYP716 family P450 enzymes in triterpenoid biosynthesis in tomato. Front. Plant Sci. 2017, 8, 21. [Google Scholar] [CrossRef] [Green Version]
- Jäger, S.; Trojan, H.; Kopp, T.; Laszczyk, M.N.; Scheffler, A. Pentacyclic triterpene distribution in various plants–rich sources for a new group of multi-potent plant extracts. Molecules 2009, 14, 2016–2031. [Google Scholar] [CrossRef] [Green Version]
- Cachofeiro, V.; Goicochea, M.; de Vinuesa, S.D.; Oubina, P.; Lahera, V.; Luno, J. Oxidative stress and inflammation, a link between chronic kidney disease and cardiovascular disease. Kidney Int. Suppl. 2008, 111, S4–S9. [Google Scholar] [CrossRef] [Green Version]
- Tucker, P.S.; Scanlan, A.T.; Dalbo, V.J. Chronic kidney disease influences multiple systems: Describing the relationship between oxidative stress, inflammation, kidney damage, and concomitant disease. Oxidative Med. Cell. Longev. 2015, 2015, 806358. [Google Scholar] [CrossRef] [PubMed]
- Gliozzi, M.; Malara, N.; Muscoli, S.; Mollace, V. The treatment of hyperuricemia. Int. J. Cardiol. 2016, 213, 23–27. [Google Scholar] [CrossRef] [Green Version]
- Abu Bakar, F.I.; Abu Bakar, M.F.; Rahmat, A.; Abdullah, N.; Sabran, S.F.; Endrini, S. Anti-gout potential of Malaysian medicinal plants. Front. Pharmacol. 2018, 9, 261. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, G.; Lu, X.; Zhao, J.; Li, X. Sunflower head enzymatic hydrolysate relives hyperuricemia by inhibiting crucial proteins (xanthine oxidase, adenosine deaminase, uric acid transporter1) and restoring gut microbiota in mice. J. Funct. Foods. 2020, 72, 104055. [Google Scholar] [CrossRef]
- Chinchilla, S.P.; Urionaguena, I.; Perez-Ruiz, F. Febuxostat for the chronic management of hyperuricemia in patients with gout. Expert Rev. Clin. Pharmacol. 2016, 9, 665–673. [Google Scholar] [CrossRef]
- Gupta, A.K.; Mausumi, B.; Ravi, M. Skin cancer concerns in people of color: Risk factors and prevention. Asian Pac. J. Cancer Prev. 2016, 17, 5257. [Google Scholar] [PubMed]
- Mukherjee, P.K.; Biswas, R.; Sharma, A.; Banerjee, S.; Biswas, S.; Katiyar, C.K. Validation of medicinal herbs for anti-tyrosinase potential. J. Herb. Med. 2018, 14, 1–16. [Google Scholar] [CrossRef]
- Anh, L.H.; Quan, N.V.; Lam, V.Q.; Iuchi, Y.; Takami, A.; Teschke, R.; Xuan, T.D. Antioxidant, anti-tyrosinase, anti-α-amylase, and cytotoxic potentials of the invasive weed Andropogon virginicus. Plants 2021, 10, 69. [Google Scholar] [CrossRef]
- Banerjee, S.; Mazumdar, S. Electrospray ionization mass spectrometry: A technique to access the information beyond the molecular weight of the analyte. Int. J. Anal. Chem. 2012, 2012, 282574. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pinto, S.A.H.; Pinto, L.M.S.; Cunha, G.M.A.; Chaves, M.H.; Santos, F.A.; Rao, V.S. Anti-inflammatory effect of α, β-amyrin, a pentacyclic triterpene from Protium heptaphyllum in rat model of acute periodontitis. Inflammopharmacology 2008, 16, 48–52. [Google Scholar] [CrossRef]
- Mahato, S.B.; Kundu, A.P. 13C NMR spectra of pentacyclic triterpenoids—A compilation and some salient features. Phytochemistry 1994, 37, 1517–1575. [Google Scholar] [CrossRef]
- Shantos, K.B.; Raja, R. Quantitative 1H NMR spectroscopy. TrAC-Trends Anal. Chem. 2012, 35, 5–26. [Google Scholar]
- Pham, T.L.; Ha, T.T.T.; Hoang, T.L.T. Comparative proteomic analysis of Celastrus hindsii Benth. phenotypes reveal an intraspecific variation. Plant Biotechnol. J. 2020, 47, 273–282. [Google Scholar]
- Kuo, Y.H.; Kuo, L.M.Y. Antitumor and anti-AIDS triterpenes from Celastrus hindsii. Phytochemistry 1997, 44, 1275–1281. [Google Scholar] [PubMed]
- Hu, X.Q.; Han, W.; Han, Z.Z.; Liu, Q.X.; Xu, X.K.; Fu, P.; Li, H.L. Three new diphenylpropanes from Celastrus hindsii. Arch. Pharm. Res. 2014, 37, 1411–1415. [Google Scholar] [CrossRef]
- Sung, T.V.; Thuy, T.T.; Ninh, P.T.; Nhung, L.T.H. Isolation and structural characterization of phenolic glycoside and triterpenes in Celastrus hindsii Benth. New J. Chem. 2008, 46, 224–228. [Google Scholar]
- Nicholson, J.K.; Foxall, P.J.; Spraul, M.; Farrant, R.D.; Lindon, J.C. 750 MHz 1H and 1H-13C NMR spectroscopy of human blood plasma. Anal. Chem. 1995, 67, 793–811. [Google Scholar] [CrossRef]
- De Melo, K.M.; De Oliveira, F.T.B.; Silva, R.A.C.; Quinderé, A.L.G.; Marinho Filho, J.D.B.; Araújo, A.J.; Santos, F.A. α, β-Amyrin, a pentacyclic triterpenoid from Protium heptaphyllum suppresses adipocyte differentiation accompanied by down regulation of PPARγ and C/EBPα in 3T3-L1 cells. Biomed. Pharmacother. 2019, 109, 1860–1866. [Google Scholar] [CrossRef]
- Ching, J.; Chua, T.K.; Chin, L.C.; Lau, A.J.; Pang, Y.K.; Jaya, J.M.; Koh, H.L. Beta-amyrin from Ardisia elliptica Thunb. is more potent than aspirin in inhibiting collagen-induced platelet aggregation. Indian J. Exp. Biol. 2010, 48, 275–279. [Google Scholar]
- Otuki, M.F.; Ferreira, J.; Lima, F.V.; Meyre-Silva, C.; Malheiros, A.; Muller, L.A.; Calixto, J.B. Antinociceptive properties of mixture of α-amyrin and β-amyrin triterpenes: Evidence for participation of protein kinase C and protein kinase A pathways. J. Pharmacol. Exp. Ther. 2005, 313, 310–318. [Google Scholar] [CrossRef] [Green Version]
- Oliveira, F.A.; Chaves, M.H.; Almeida, F.R.; Lima, R.C., Jr.; Silva, R.M.; Maia, J.L.; Rao, V.S. Protective effect of α-and β-amyrin, a triterpene mixture from Protium heptaphyllum (Aubl.) March. trunk wood resin, against acetaminophen-induced liver injury in mice. J. Ethnopharmacol. 2005, 98, 103–108. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, F.A.; Vieira-Júnior, G.M.; Chaves, M.H.; Almeida, F.R.; Santos, K.A.; Martins, F.S.; Rao, V.S. Gastroprotective effect of the mixture of α-and β-amyrin from Protium paraphylum: Role of capsaicin-sensitive primary afferent neurons. Planta Med. 2004, 70, 780–782. [Google Scholar] [CrossRef]
- Akihisa, T.; Yasukawa, K.; Oinuma, H.; Kasahara, Y.; Yamanouchi, S.; Takido, M.; Tamura, T. Triterpene alcohols from the flowers of compositae and their anti-inflammatory effects. Phytochemistry 1996, 43, 1255–1260. [Google Scholar] [CrossRef]
- Melo, C.M.; Morais, T.C.; Tomé, A.R.; Brito, G.A.C.; Chaves, M.H.; Rao, V.S.; Santos, F.A. Anti-inflammatory effect of α, β-amyrin, a triterpene from Protium heptaphyllum, on cerulein-induced acute pancreatitis in mice. Inflamm. Res. 2011, 60, 673–681. [Google Scholar] [CrossRef]
- Ching, J.; Lin, H.S.; Tan, C.H.; Koh, H.L. Quantification of α- and β-amyrin in rat plasma by gas chromatography–mass spectrometry: Application to preclinical pharmacokinetic study. J. Mass Spectrom. 2011, 46, 457–464. [Google Scholar] [CrossRef] [PubMed]
- Akash, M.S.H.; Rehman, K. Column chromatography. In Essentials of Pharmaceutical Analysis; Springer: Singapore, 2020; pp. 167–174. [Google Scholar]
- Lisec, J.; Schauer, N.; Kopka, J.; Willmitzer, L.; Fernie, A.R. Gas chromatography mass spectrometry–based metabolite profiling in plants. Nat. Protoc. 2006, 1, 387–396. [Google Scholar] [CrossRef] [PubMed]
- De Lacy Costello, B.P.J.; Evans, P.; Ewen, R.J.; Gunson, H.E.; Jones, P.R.; Ratcliffe, N.M.; Spencer-Phillips, P.T. Gas chromatography–mass spectrometry analyses of volatile organic compounds from potato tubers inoculated with Phytophthora infesting or Fusarium coeruleus. Plant Pathol. J. 2001, 50, 489–496. [Google Scholar] [CrossRef]
- Ho, C.S.; Lam, C.W.K.; Chan, M.H.M.; Cheung, R.C.K.; Law, L.K.; Lit, L.C.W.; Ng, K.F.; Suen, M.W.M.; Tai, H.L. Electrospray ionization mass spectrometry: Principles and clinical applications. Clin. Biochem. Rev. 2003, 24, 3. [Google Scholar]
- Bruins, A.P. Mechanistic aspects of electrospray ionization. J. Chromatogr. A 1998, 794, 345–357. [Google Scholar] [CrossRef]
- Hoult, D.I.; Bhakar, B. NMR signal reception: Virtual photons and coherent spontaneous emission. Concepts Magn. Reson. A Bridg. Educ. Res. 1997, 9, 277–297. [Google Scholar] [CrossRef]
- Sternberg, U.; Witter, R.; Kuprov, I.; Lamley, J.M.; Oss, A.; Lewandowski, J.R.; Samoson, A. 1H line width dependence on MAS speed in solid state NMR–comparison of experiment and simulation. J. Magn. Reson. 2018, 291, 32–39. [Google Scholar] [CrossRef]
- Morita, M.; Shibuya, M.; Kushiro, T.; Masuda, K.; Ebizuka, Y. Molecular cloning and functional expression of triterpene synthases from pea (Pisum sativum) New α-amyrin-producing enzyme is a multifunctional triterpene synthase. Eur. J. Biochem. 2000, 267, 3453–3460. [Google Scholar] [CrossRef]
- Martelanc, M.; Vovk, I.; Simonovska, B. Determination of three major triterpenoids in epicuticular wax of cabbage (Brassica oleracea L.) by high-performance liquid chromatography with UV and mass spectrometric detection. J. Chromatogr. A 2007, 1164, 145–152. [Google Scholar] [CrossRef]
- Sunil, C.; Irudayaraj, S.S.; Duraipandiyan, V.; Al-Dhabi, N.A.; Agastian, P.; Ignacimuthu, S. Antioxidant and free radical scavenging effects of β-amyrin isolated from Symplocos cochinchinensis Moore. leaves. Ind. Crops Prod. 2014, 61, 510–516. [Google Scholar] [CrossRef]
- Okoye, N.N.; Ajaghaku, D.L.; Okeke, H.N.; Ilodigwe, E.E.; Nworu, C.S.; Okoye, F.B.C. Beta-amyrin and alpha-amyrin acetate isolated from the stem bark of Alstonia boonei display profound anti-inflammatory activity. Pharm. Biol. 2014, 52, 1478–1486. [Google Scholar] [CrossRef] [Green Version]
- Sirat, H.M.; Susanti, D.; Ahmad, F.; Takayama, H.; Kitajima, M. Amides, triterpene, and flavonoids from the leaves of Melastoma malabathricum L. J. Nat. Med. 2010, 64, 492–495. [Google Scholar] [CrossRef]
- Saeidnia, S.; Ara, L.; Hajimehdipoor, H.; Read, R.W.; Arshadi, S.; Nikan, M. Chemical constituents of Swertia longifolia Boiss. with α-amylase inhibitory activity. Res. Pharm. Sci. 2016, 11, 23–32. [Google Scholar]
- Quan, N.V.; Xuan, T.D.; Tran, H.D.; Thuy, N.T.D.; Trang, L.T.; Huong, C.T.T.; Tuyen, P.T. Antioxidant, α-amylase and α-glucosidase inhibitory activities and potential constituents of Canarium tramdenum bark. Molecules 2019, 24, 605. [Google Scholar] [CrossRef] [Green Version]
- Higuchi, C.T.; Pavan, F.R.; Leite, C.Q.F.; Sannomiya, M.; Vilegas, W.; Leite, S.R.D.A.; Sato, D.N. Triterpenes and antitubercular activity of Byrsonima crassa. Quim. Nova 2008, 31, 1719–1721. [Google Scholar] [CrossRef]
- Hernandez-Vazquez, L.; Bonfill, M.; Moyano, E.; Cusido, R.M.; Navarro-Ocana, A.; Palazon, J. Conversion of α-amyrin into centellosides by plant cell cultures of Centella asiatica. Biotechnol. Lett. 2010, 32, 315–319. [Google Scholar] [CrossRef]
- Persson, T.; Popescu, B.O.; Cedazo-Minguez, A. Oxidative stress in Alzheimer’s disease: Why did antioxidant therapy fail? Oxidative Med. Cell. Longev. 2014, 427318. [Google Scholar] [CrossRef] [Green Version]
- López-Alarcón, C.; Denicola, A. Evaluating the antioxidant capacity of natural products: A review on chemical and cellular-based assays. Anal. Chim. Acta 2013, 763, 1–10. [Google Scholar] [CrossRef]
- Gutteridge, J.M.; Halliwell, B. Invited review free radicals in disease processes: A compilation of cause and consequence. Free Radic. Res. Commun. 1993, 19, 141–158. [Google Scholar] [CrossRef] [PubMed]
- Halliwell, B.A.R.R.Y.; Gutteridge, J.M.; Cross, C.E. Free radicals, antioxidants, and human disease: Where are we now? J. Lab. Clin. Med. 1992, 119, 598–620. [Google Scholar] [PubMed]
- Karen Cardoso, B.; Line Marko de Oliveira, H.; Zonta Melo, U.; Mariano Fernandez, C.M.; Franco de Araújo Almeida Campo, C.; Gonçalves, J.E.; Cristiani Gazim, Z. Antioxidant activity of α and β-amyrin isolated from Myrcianthes pungens leaves. Nat. Prod. Res. 2020, 34, 1777–1781. [Google Scholar] [CrossRef] [PubMed]
- Melo, C.M.; Carvalho, K.M.M.B.; De Sousa Neves, J.C.; Morais, T.C.; Rao, V.S.; Santos, F.A.; De Castro, G.A.; Chaves, M.H. α, β-amyrin, a natural triterpenoid ameliorates L-arginine-induced acute pancreatitis in rats. World J. Gastroenterol. 2010, 16, 4272–4280. [Google Scholar] [CrossRef]
- Shoji, A.; Yamanaka, H.; Kamatani, N. A retrospective study of the relationship between serum urate level and recurrent attacks of gouty arthritis: Evidence for reduction of recurrent gouty arthritis with antihyperuricemic therapy. Arthritis Care Res. 2004, 51, 321–325. [Google Scholar] [CrossRef]
- Yamanaka, H.; Metabolism, T.G. Essence of the revised guideline for the management of hyperuricemia and gout. Jpn. Med. Assoc. J. 2012, 55, 324–329. [Google Scholar]
- Koto, R.; Nakajima, A.; Horiuchi, H.; Yamanaka, H. Real-world treatment of gout and asymptomatic hyperuricemia: A cross-sectional study of Japanese health insurance claims data. Mod. Rheumatol. 2021, 31, 261–269. [Google Scholar] [CrossRef] [PubMed]
- Dalbeth, N.; Haskard, D.O. Mechanisms of inflammation in gout. Rheumatology 2005, 44, 1090–1096. [Google Scholar] [CrossRef] [Green Version]
- Kuo, C.F.; Grainge, M.J.; Mallen, C.; Zhang, W.; Doherty, M. Rising burden of gout in the UK but continuing suboptimal management: A nationwide population study. Ann. Rheum. Dis. 2015, 74, 661–667. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nguyen, T.D.; Thuong, P.T.; Hwang, I.H.; Hoang, T.K.H.; Nguyen, M.K.; Nguyen, H.A.; Na, M. Anti-hyperuricemic, anti-inflammatory and analgesic effects of Siegesbeckia orientalis L. Resulting from the fraction with high phenolic content. BMC Complement. Med. Ther. 2017, 17, 191. [Google Scholar] [CrossRef]
- Korner, A.; Pawelek, J. Mammalian tyrosinase catalyzes three reactions in the biosynthesis of melanin. Science 1982, 217, 1163–1165. [Google Scholar] [CrossRef] [PubMed]
- Boyle, J.L.; Haupt, H.M.; Stern, J.B.; Multhaupt, H.A. Tyrosinase expression in malignant melanoma, desmoplastic melanoma, and peripheral nerve tumors: An immunohistochemical study. Arch. Pathol. Lab. Med. 2002, 126, 816–822. [Google Scholar] [CrossRef] [PubMed]
- Baharav, E.; Merimski, O.; Shoenfeld, Y.; Zigelman, R.; Gilbrud, B.; Yecheskel, G.; Fishman, P. Tyrosinase as an autoantigen in patients with vitiligo. Clin. Exp. Immunol. 1996, 105, 84–88. [Google Scholar] [CrossRef]
- Patel, A.R.; Sun, W.Q.; Payne, G.F. Tyrosinase reaction/chitosan adsorption: Potential for removing polymerization storage inhibitors. Ind. Eng. Chem. Res. 1994, 33, 2168–2173. [Google Scholar] [CrossRef]
- Nichols, L.A.; Chen, Y.; Colella, T.A.; Bennett, C.L.; Clausen, B.E.; Engelhard, V.H. Deletional self-tolerance to a melanocyte/melanoma antigen derived from tyrosinase is mediated by a radio-resistant cell in peripheral and mesenteric lymph nodes. J. Immunol. 2007, 179, 993–1003. [Google Scholar] [CrossRef] [Green Version]
- Högnadóttir, Á.; Rouseff, R.L. Identification of aroma active compounds in orange essence oil using gas chromatography olfactometry and gas chromatography mass spectrometry. J. Chromatogr. A 2003, 998, 201–211. [Google Scholar] [CrossRef]
- Andriana, Y.; Xuan, T.D.; Quy, T.N.; Minh, T.N.; Van, T.M.; Viet, T.D. Antihyperuricemia, antioxidant, and antibacterial activities of Tridax procumbens L. Foods 2019, 8, 21. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Quy, T.N.; Xuan, T.D. Xanthine oxidase inhibitory potential, antioxidant and antibacterial activities of Cordyceps militaris (L.) Link fruiting body. Medicines 2019, 6, 20. [Google Scholar] [CrossRef] [PubMed] [Green Version]

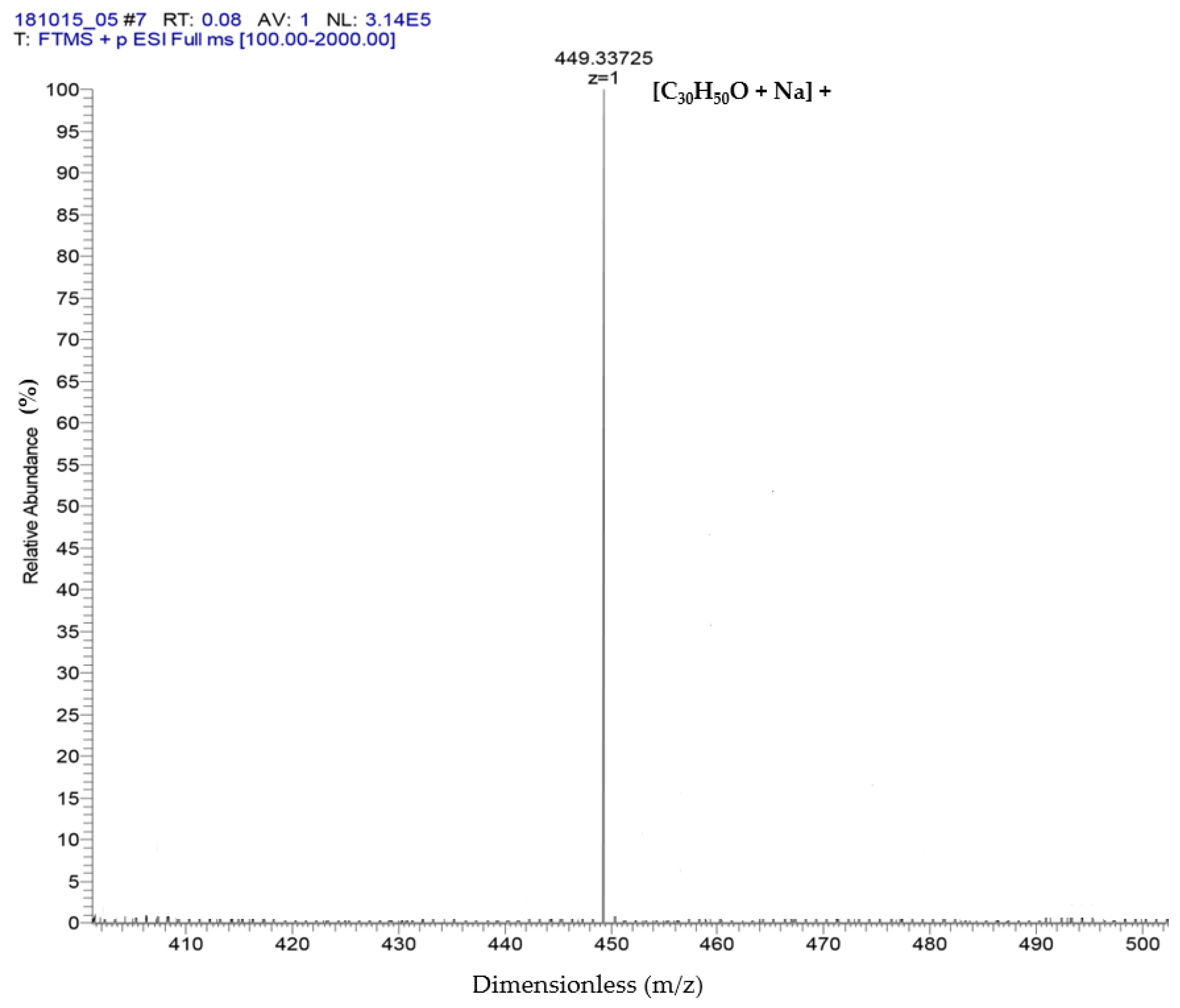
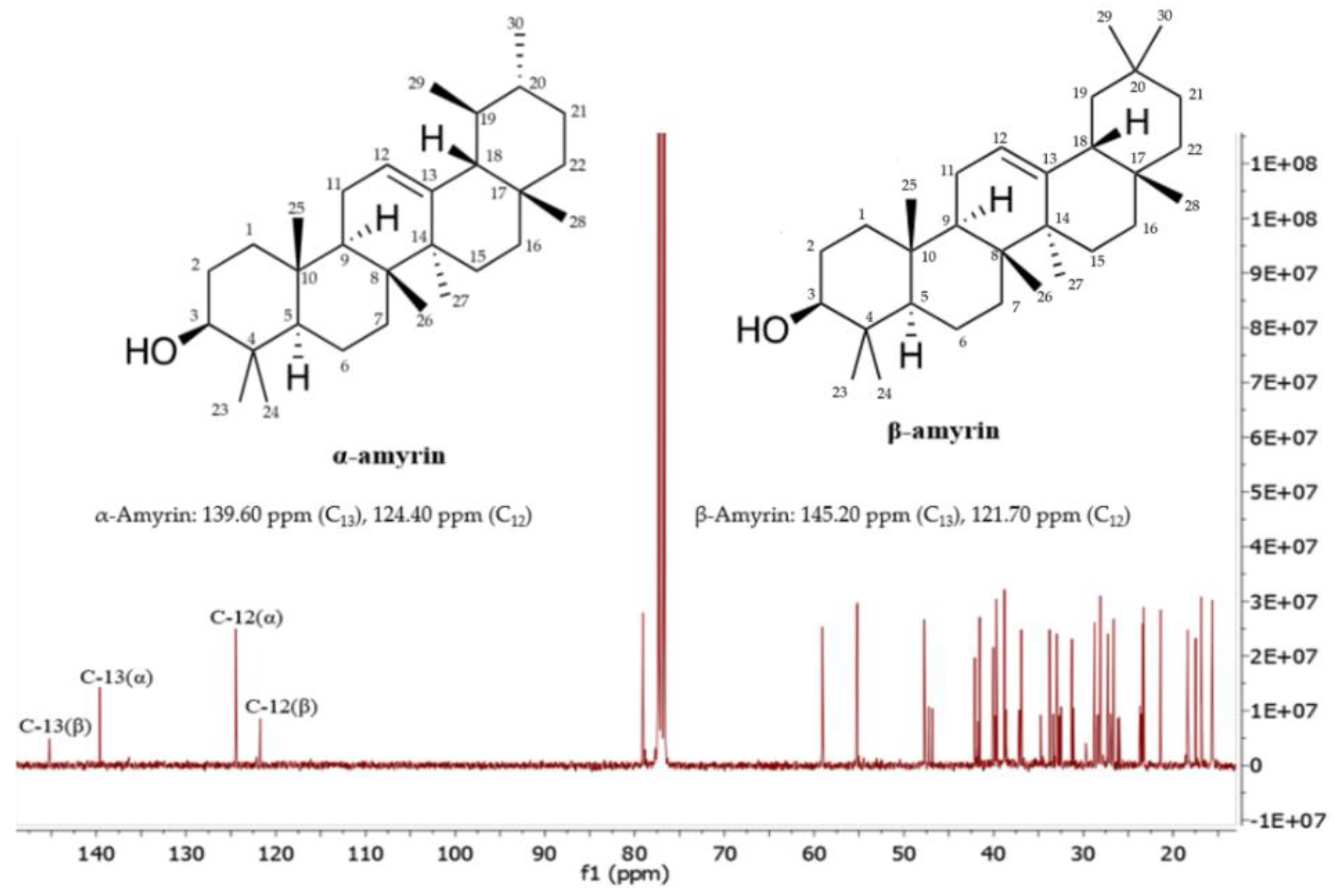

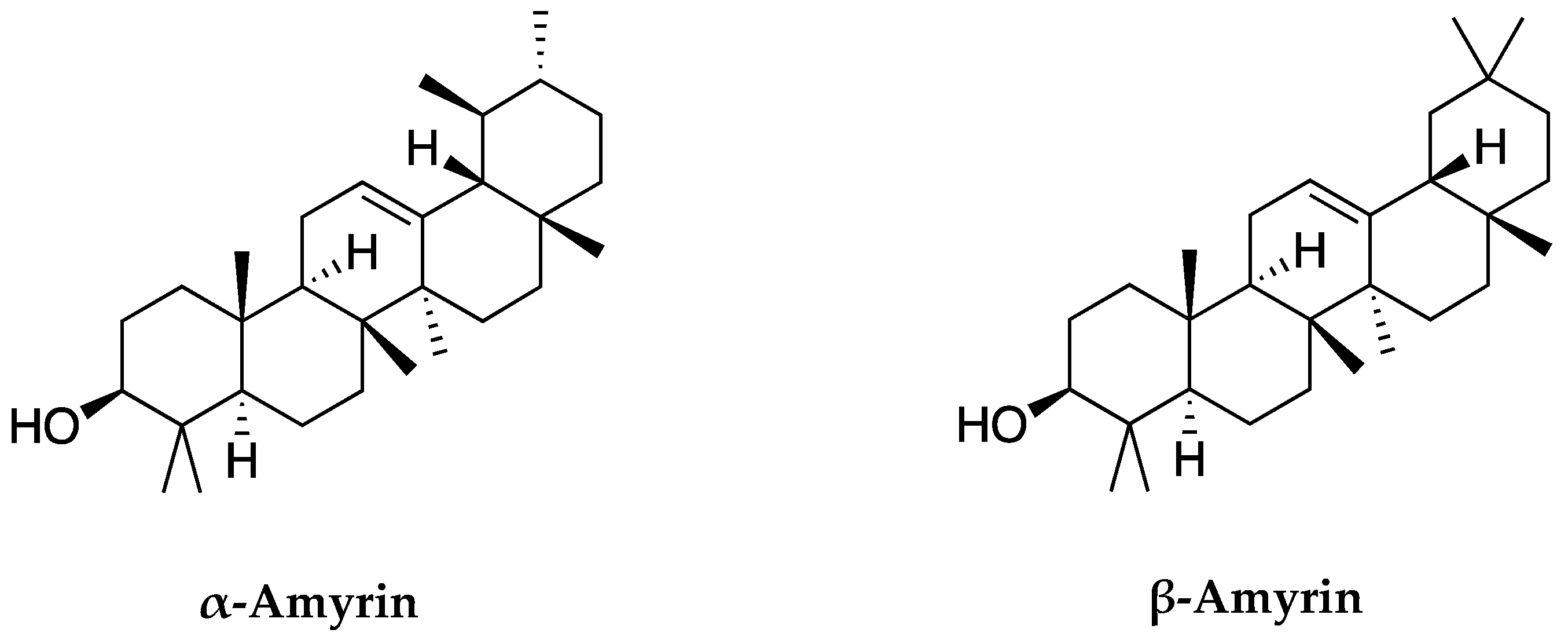
| Time (min) | Peak Area (% of Total) | Compounds | Formula | Molecular Weight (g/mol) | Chemical Class |
|---|---|---|---|---|---|
| 28.92 | 40.61 | β-Amyrin | C30H50O | 426.70 | Triterpene |
| 29.53 | 59.10 | α-Amyrin | C30H50O | 426.70 | Triterpene |
| Position | β-Amyrin | α-Amyrin | Position | β-Amyrin | α-Amyrin |
|---|---|---|---|---|---|
| 1 | 38.60 | 38.80 | 16 | 26.20 | 26.60 |
| 2 | 27.20 | 27.30 | 17 | 32.70 | 33.80 |
| 3 | 79.00 | 79.10 | 18 | 47.20 | 59.10 |
| 4 | 39.80 | 38.80 | 19 | 46.80 | 39.70 |
| 5 | 55.20 | 55.20 | 20 | 31.10 | 39.60 |
| 6 | 18.40 | 18.40 | 21 | 34.70 | 31.30 |
| 7 | 32.50 | 32.90 | 22 | 37.10 | 40.00 |
| 8 | 41.70 | 40.00 | 23 | 28.10 | 28.10 |
| 9 | 47.60 | 47.70 | 24 | 15.60 | 15.70 |
| 10 | 37.00 | 36.90 | 25 | 15.50 | 15.60 |
| 11 | 23.70 | 23.30 | 26 | 16.80 | 17.40 |
| 12 | 121.70 | 124.40 | 27 | 26.00 | 23.40 |
| 13 | 145.20 | 139.60 | 28 | 28.40 | 28.80 |
| 14 | 42.80 | 41.50 | 29 | 33.30 | 16.90 |
| 15 | 26.90 | 28.10 | 30 | 23.50 | 21.40 |
| Content of Mixture (g/kg Dry Weight) | Ratio (%) | Contents (g/kg Dry Weight) | ||
|---|---|---|---|---|
| 10.75 | α-Amyrin | β-Amyrin | α-Amyrin | β-Amyrin |
| 69.60 | 30.40 | 7.48 | 3.27 | |
| Sample | Antioxidant Activity | XO IC50 (µg/mL) | Tyrosinase IC50 (µg/mL) | |
|---|---|---|---|---|
| DPPH IC50 (µg/mL) | ABTS IC50 (µg/mL) | |||
| α-Amyrin and β-Amyrin | 125.55 ± 0.98 a | 155.28 ± 1.01 a | 258.22 ± 2.28 a | 178.85 ± 3.28 a |
| Allopurinol | - | 7.58 ± 1.29 b | ||
| Kojic acid | - | - | 15.55 ± 2.28 b | |
| BHT | 8.22 ± 0.89 b | 53.40 ± 1.52 b | - | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Viet, T.D.; Xuan, T.D.; Anh, L.H. α-Amyrin and β-Amyrin Isolated from Celastrus hindsii Leaves and Their Antioxidant, Anti-Xanthine Oxidase, and Anti-Tyrosinase Potentials. Molecules 2021, 26, 7248. https://doi.org/10.3390/molecules26237248
Viet TD, Xuan TD, Anh LH. α-Amyrin and β-Amyrin Isolated from Celastrus hindsii Leaves and Their Antioxidant, Anti-Xanthine Oxidase, and Anti-Tyrosinase Potentials. Molecules. 2021; 26(23):7248. https://doi.org/10.3390/molecules26237248
Chicago/Turabian StyleViet, Tran Duc, Tran Dang Xuan, and La Hoang Anh. 2021. "α-Amyrin and β-Amyrin Isolated from Celastrus hindsii Leaves and Their Antioxidant, Anti-Xanthine Oxidase, and Anti-Tyrosinase Potentials" Molecules 26, no. 23: 7248. https://doi.org/10.3390/molecules26237248
APA StyleViet, T. D., Xuan, T. D., & Anh, L. H. (2021). α-Amyrin and β-Amyrin Isolated from Celastrus hindsii Leaves and Their Antioxidant, Anti-Xanthine Oxidase, and Anti-Tyrosinase Potentials. Molecules, 26(23), 7248. https://doi.org/10.3390/molecules26237248








