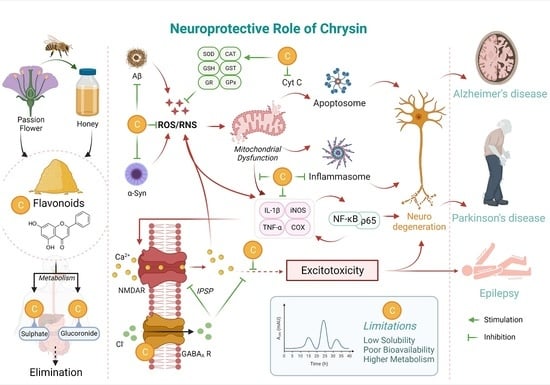
Neuroprotective Potential of Chrysin: Mechanistic Insights and Therapeutic Potential for Neurological Disorders
Abstract
1. Introduction
2. Chemistry and Pharmacokinetics of Chrysin
3. Potential Neuroprotective Mechanisms of Chrysin
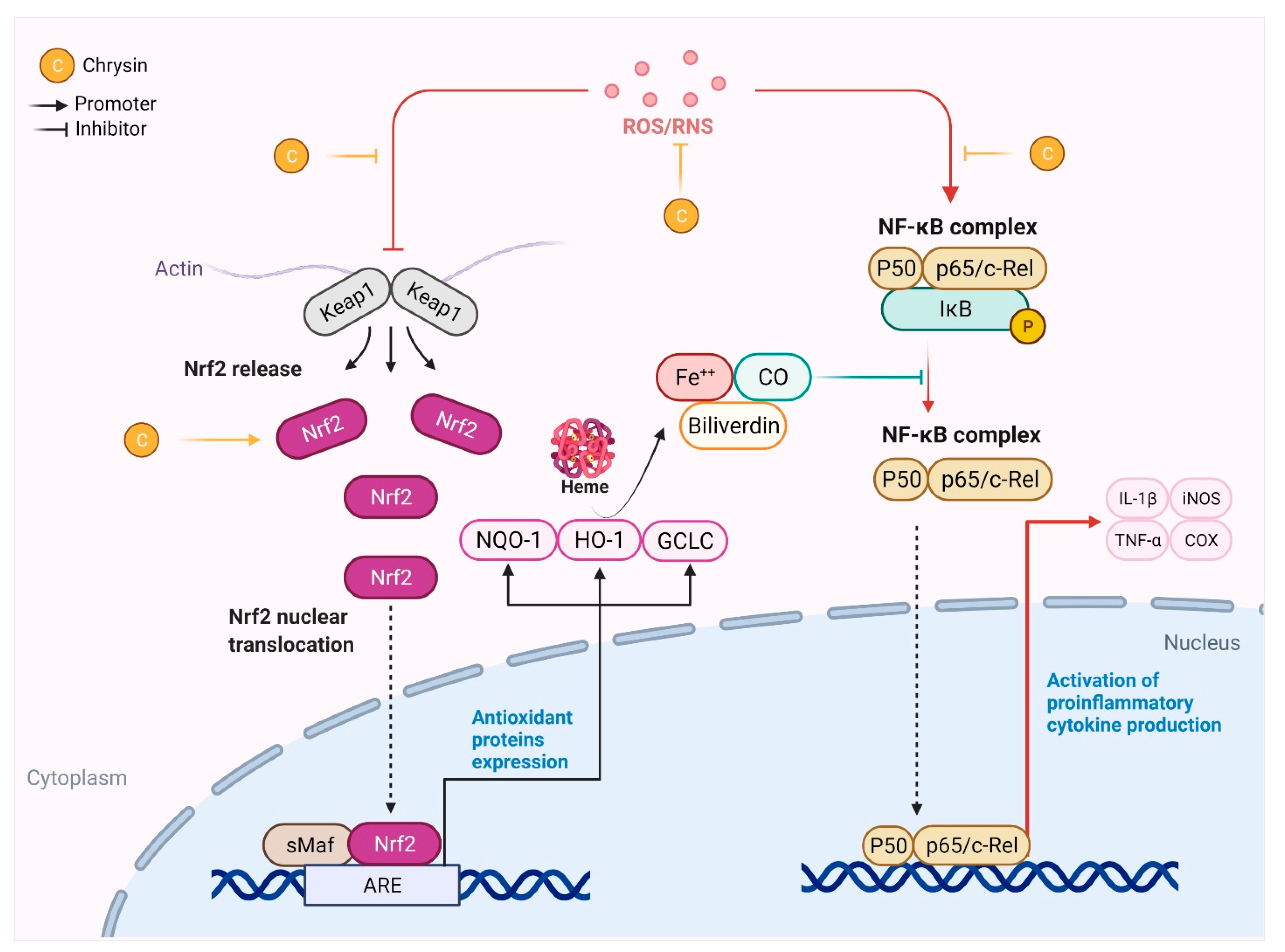
3.1. Chrysin as an Anti-Oxidant Agent
3.2. Chrysin as an Anti-Inflammatory Agent
3.3. Chrysin as an Anti-Apoptotic Agent
3.4. Chrysin as a MAO Inhibitor
3.5. Neuroprotective Role of Chrysin via a GABA Mimetic Action
4. Role of Chrysin in Different Neurological Disorders
4.1. Chrysin in AD
4.2. Chrysin in PD
4.3. Chrysin in Epilepsy
4.4. Chrysin in MS
4.5. Chrysin in Traumatic and Ischemic Brain Injury
4.6. Chrysin in Gliomas
4.7. Possible Limitations of Chrysin and Strategies to Mitigate
4.8. Conclusions and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| 6-OHDA | 6-hydroxydopamine |
| AD | Alzheimer’s diseases |
| ARE | Antioxidant response element |
| BBB | Blood–brain barrier |
| DA | Dopaminergic |
| DAMPs | Damage-associated molecular patterns |
| EAE | Experimental autoimmune encephalomyelitis |
| GPx: | Glutathione peroxidase |
| LPS | lipopolysaccharide |
| MPTP | 1-methyl-4-phenyl-1: 2, 3, 6-tetrahydropyridine; |
| MS | Multiple sclerosis |
| NF-κB | Nuclear factor kappa light chain-enhancer of activated B cells |
| Nrf2 | Nuclear factor erythroid |
| PAMPs | Pathogen-associated molecular patterns |
| PD | Parkinson’s disease |
| PRRs | Pattern recognition receptors |
| PTZ | Pentylenetetrazol |
| ROS | Reactive oxygen species |
| SOD | Superoxide dismutase |
| TBI | Traumatic brain injury |
| TH | Tyrosine hydroxylase |
| TLR | Toll-like receptors |
References
- India State-Level Disease Burden Initiative Neurological Disorders Collaborators. The burden of neurological disorders across the states of India: The Global Burden of Disease Study 1990–2019. Lancet Glob. Health 2021, 9, e1129–e1144. [Google Scholar] [CrossRef]
- Murray, C.J.; Vos, T.; Lozano, R.; Naghavi, M.; Flaxman, A.D.; Michaud, C.; Ezzati, M.; Shibuya, K.; Salomon, J.A.; Abdalla, S.; et al. Disability-adjusted life years (DALYs) for 291 diseases and injuries in 21 regions, 1990–2010: A systematic analysis for the Global Burden of Disease Study 2010. Lancet 2012, 380, 2197–2223. [Google Scholar] [CrossRef]
- Maan, G.; Sikdar, B.; Kumar, A.; Shukla, R.; Mishra, A. Role of Flavonoids in Neurodegenerative Diseases: Limitations and Future Perspectives. Curr. Top. Med. Chem. 2020, 20, 1169–1194. [Google Scholar] [CrossRef]
- Mishra, A.; Bandopadhyay, R.; Singh, P.K.; Mishra, P.S.; Sharma, N.; Khurana, N. Neuroinflammation in neurological disorders: Pharmacotherapeutic targets from bench to bedside. Metab. Brain Dis. 2021, 36, 1591–1626. [Google Scholar] [CrossRef]
- Angelopoulou, E.; Paudel, Y.N.; Piperi, C.; Mishra, A. Neuroprotective potential of cinnamon and its metabolites in Parkinson’s disease: Mechanistic insights, limitations, and novel therapeutic opportunities. J. Biochem. Mol. Toxicol. 2021, 35, e22720. [Google Scholar] [CrossRef]
- Mani, R.; Natesan, V. Chrysin: Sources, beneficial pharmacological activities, and molecular mechanism of action. Phytochemistry 2018, 145, 187–196. [Google Scholar] [CrossRef]
- Ayaz, M.; Sadiq, A.; Junaid, M.; Ullah, F.; Ovais, M.; Ullah, I.; Ahmed, J.; Shahid, M. Flavonoids as Prospective Neuroprotectants and Their Therapeutic Propensity in Aging Associated Neurological Disorders. Front. Aging Neurosci. 2019, 11, 155. [Google Scholar] [CrossRef]
- Choi, Y.T.; Jung, C.H.; Lee, S.R.; Bae, J.H.; Baek, W.K.; Suh, M.H.; Park, J.; Park, C.W.; Suh, S.I. The green tea polyphenol (-)-epigallocatechin gallate attenuates beta-amyloid-induced neurotoxicity in cultured hippocampal neurons. Life Sci. 2001, 70, 603–614. [Google Scholar] [CrossRef]
- Shukitt-Hale, B.; Cheng, V.; Joseph, J.A. Effects of blackberries on motor and cognitive function in aged rats. Nutr Neurosci. 2009, 12, 135–140. [Google Scholar] [CrossRef] [PubMed]
- Manach, C.; Scalbert, A.; Morand, C.; Remesy, C.; Jimenez, L. Polyphenols: Food sources and bioavailability. Am. J. Clin. Nutr 2004, 79, 727–747. [Google Scholar] [CrossRef]
- Spencer, J.P.; Abd-el-Mohsen, M.M.; Rice-Evans, C. Cellular uptake and metabolism of flavonoids and their metabolites: Implications for their bioactivity. Arch. Biochem Biophys 2004, 423, 148–161. [Google Scholar] [CrossRef]
- Yao, W.; Cheng, J.; Kandhare, A.D.; Mukherjee-Kandhare, A.A.; Bodhankar, S.L.; Lu, G. Toxicological evaluation of a flavonoid, chrysin: Morphological, behavioral, biochemical and histopathological assessments in rats. Drug Chem. Toxicol. 2021, 44, 601–612. [Google Scholar] [CrossRef] [PubMed]
- Walle, T.; Otake, Y.; Brubaker, J.A.; Walle, U.K.; Halushka, P.V. Disposition and metabolism of the flavonoid chrysin in normal volunteers. Br. J. Clin. Pharmacol. 2001, 51, 143–146. [Google Scholar] [PubMed]
- Chen, Y.H.; Yang, Z.S.; Wen, C.C.; Chang, Y.S.; Wang, B.C.; Hsiao, C.A.; Shih, T.L. Evaluation of the structure-activity relationship of flavonoids as antioxidants and toxicants of zebrafish larvae. Food Chem 2012, 134, 717–724. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhao, J.; Afzal, O.; Kazmi, I.; Al-Abbasi, F.A.; Altamimi, A.S.A.; Yang, Z. Neuroprotective role of chrysin-loaded poly(lactic-co-glycolic acid) nanoparticle against kindling-induced epilepsy through Nrf2/ARE/HO-1 pathway. J. Biochem Mol. Toxicol 2021, 35, e22634. [Google Scholar] [CrossRef]
- Skulachev, V.P. Membrane-linked systems preventing superoxide formation. Biosci Rep. 1997, 17, 347–366. [Google Scholar] [CrossRef]
- Lin, M.T.; Beal, M.F. Mitochondrial dysfunction and oxidative stress in neurodegenerative diseases. Nature 2006, 443, 787–795. [Google Scholar] [CrossRef] [PubMed]
- Moldogazieva, N.T.; Lutsenko, S.V.; Terentiev, A.A. Reactive Oxygen and Nitrogen Species-Induced Protein Modifications: Implication in Carcinogenesis and Anticancer Therapy. Cancer Res. 2018, 78, 6040–6047. [Google Scholar] [CrossRef]
- Belli, S.; Rossi, M.; Molasky, N.; Middleton, L.; Caldwell, C.; Bartow-McKenney, C.; Duong, M.; Chiu, J.; Gibbs, E.; Caldwell, A.; et al. Effective and Novel Application of Hydrodynamic Voltammetry to the Study of Superoxide Radical Scavenging by Natural Phenolic Antioxidants. Antioxidants 2019, 8, 14. [Google Scholar] [CrossRef]
- Khezri, S.; Sabzalipour, T.; Jahedsani, A.; Azizian, S.; Atashbar, S.; Salimi, A. Chrysin ameliorates aluminum phosphide-induced oxidative stress and mitochondrial damages in rat cardiomyocytes and isolated mitochondria. Environ. Toxicol 2020, 35, 1114–1124. [Google Scholar] [CrossRef]
- El-Sisi, A.E.; El-Sayad, M.E.; Abdelsalam, N.M. Protective effects of mirtazapine and chrysin on experimentally induced testicular damage in rats. Biomed. Pharmacother. 2017, 95, 1059–1066. [Google Scholar] [CrossRef]
- Vedagiri, A.; Thangarajan, S. Mitigating effect of chrysin loaded solid lipid nanoparticles against Amyloid β25–35 induced oxidative stress in rat hippocampal region: An efficient formulation approach for Alzheimer’s disease. Neuropeptides 2016, 58, 111–125. [Google Scholar] [CrossRef]
- Zhu, Z.Y.; Chen, L.; Liu, F.; Chen, L.J.; Meng, M.; Sun, H.Q.; Zhang, Y.M. Preparation and activity evaluation of chrysin-beta-D-galactopyranoside. Arch. Pharm. Res. 2016, 39, 1433–1440. [Google Scholar] [CrossRef] [PubMed]
- Chovanova, K.; Kamlarova, A.; Maresch, D.; Harichova, J.; Zamocky, M. Expression of extracellular peroxidases and catalases in mesophilic and thermophilic Chaetomia in response to environmental oxidative stress stimuli. Ecotoxicol. Environ. Saf. 2019, 181, 481–490. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Branicky, R.; Noe, A.; Hekimi, S. Superoxide dismutases: Dual roles in controlling ROS damage and regulating ROS signaling. J. Cell Biol. 2018, 217, 1915–1928. [Google Scholar] [CrossRef] [PubMed]
- Souza, L.C.; Antunes, M.S.; Filho, C.B.; Del Fabbro, L.; de Gomes, M.G.; Goes, A.T.; Donato, F.; Prigol, M.; Boeira, S.P.; Jesse, C.R. Flavonoid Chrysin prevents age-related cognitive decline via attenuation of oxidative stress and modulation of BDNF levels in aged mouse brain. Pharmacol. Biochem. Behav. 2015, 134, 22–30. [Google Scholar] [CrossRef]
- Feng, Y.C.; Liao, C.Y.; Xia, W.K.; Jiang, X.Z.; Shang, F.; Yuan, G.R.; Wang, J.J. Regulation of three isoforms of SOD gene by environmental stresses in citrus red mite, Panonychus citri. Exp. Appl. Acarol. 2015, 67, 49–63. [Google Scholar] [CrossRef]
- Blokhina, O.; Virolainen, E.; Fagerstedt, K.V. Antioxidants, oxidative damage and oxygen deprivation stress: A review. Ann. Bot. 2003, 91, 179–194. [Google Scholar] [CrossRef]
- Ayala, A.; Munoz, M.F.; Arguelles, S. Lipid peroxidation: Production, metabolism, and signaling mechanisms of malondialdehyde and 4-hydroxy-2-nonenal. Oxid Med. Cell Longev. 2014, 2014, 360438. [Google Scholar] [CrossRef]
- Feussner, I.; Wasternack, C. The lipoxygenase pathway. Annu. Rev. Plant. Biol. 2002, 53, 275–297. [Google Scholar] [CrossRef] [PubMed]
- Jones, D.P. Redefining oxidative stress. Antioxid Redox Signal. 2006, 8, 1865–1879. [Google Scholar] [CrossRef] [PubMed]
- Ileriturk, M.; Benzer, F.; Aksu, E.H.; Yildirim, S.; Kandemir, F.M.; Dogan, T.; Dortbudak, M.B.; Genc, A. Chrysin protects against testicular toxicity caused by lead acetate in rats with its antioxidant, anti-inflammatory, and antiapoptotic properties. J. Food Biochem. 2021, 45, e13593. [Google Scholar] [CrossRef] [PubMed]
- Carson, M.J.; Doose, J.M.; Melchior, B.; Schmid, C.D.; Ploix, C.C. CNS immune privilege: Hiding in plain sight. Immunol. Rev. 2006, 213, 48–65. [Google Scholar] [CrossRef] [PubMed]
- Carson, M.J.; Thrash, J.C.; Walter, B. The cellular response in neuroinflammation: The role of leukocytes, microglia and astrocytes in neuronal death and survival. Clin. Neurosci. Res. 2006, 6, 237–245. [Google Scholar] [CrossRef]
- Ha, S.K.; Moon, E.; Kim, S.Y. Chrysin suppresses LPS-stimulated proinflammatory responses by blocking NF-kappaB and JNK activations in microglia cells. Neurosci. Lett 2010, 485, 143–147. [Google Scholar] [CrossRef]
- Lyman, M.; Lloyd, D.G.; Ji, X.; Vizcaychipi, M.P.; Ma, D. Neuroinflammation: The role and consequences. Neurosci Res. 2014, 79, 1–12. [Google Scholar] [CrossRef]
- Park, S.E.; Sapkota, K.; Kim, S.; Kim, H.; Kim, S.J. Kaempferol acts through mitogen-activated protein kinases and protein kinase B/AKT to elicit protection in a model of neuroinflammation in BV2 microglial cells. Br. J. Pharmacol. 2011, 164, 1008–1025. [Google Scholar] [CrossRef]
- Glass, C.K.; Saijo, K.; Winner, B.; Marchetto, M.C.; Gage, F.H. Mechanisms underlying inflammation in neurodegeneration. Cell 2010, 140, 918–934. [Google Scholar] [CrossRef]
- Harry, G.J.; Kraft, A.D. Neuroinflammation and microglia: Considerations and approaches for neurotoxicity assessment. Expert Opin. Drug. Metab. Toxicol 2008, 4, 1265–1277. [Google Scholar] [CrossRef]
- Kaushal, V.; Schlichter, L.C. Mechanisms of microglia-mediated neurotoxicity in a new model of the stroke penumbra. J. Neurosci. 2008, 28, 2221–2230. [Google Scholar] [CrossRef]
- Walsh, J.G.; Muruve, D.A.; Power, C. Inflammasomes in the CNS. Nat. Rev. Neurosci. 2014, 15, 84–97. [Google Scholar] [CrossRef] [PubMed]
- Shi, J.; Gao, W.; Shao, F. Pyroptosis: Gasdermin-Mediated Programmed Necrotic Cell Death. Trends Biochem. Sci. 2017, 42, 245–254. [Google Scholar] [CrossRef] [PubMed]
- Lamkanfi, M.; Dixit, V.M. Mechanisms and functions of inflammasomes. Cell 2014, 157, 1013–1022. [Google Scholar] [CrossRef] [PubMed]
- Khombi Shooshtari, M.; Farbood, Y.; Mansouri, S.M.T.; Badavi, M.; Khorsandi, L.S.; Ghasemi Dehcheshmeh, M.; Sarkaki, A.R. Neuroprotective Effects of Chrysin Mediated by Estrogenic Receptors Following Cerebral Ischemia and Reperfusion in Male Rats. Basic Clin. Neurosci. 2021, 12, 149–162. [Google Scholar] [CrossRef]
- Li, T.F.; Ma, J.; Han, X.W.; Jia, Y.X.; Yuan, H.F.; Shui, S.F.; Guo, D.; Yan, L. Chrysin ameliorates cerebral ischemia/reperfusion (I/R) injury in rats by regulating the PI3K/Akt/mTOR pathway. Neurochem. Int. 2019, 129, 104496. [Google Scholar] [CrossRef] [PubMed]
- Lee, B.K.; Lee, W.J.; Jung, Y.S. Chrysin Attenuates VCAM-1 Expression and Monocyte Adhesion in Lipopolysaccharide-Stimulated Brain Endothelial Cells by Preventing NF-kappaB Signaling. Int. J. Mol. Sci. 2017, 18, 1424. [Google Scholar] [CrossRef]
- Mantawy, E.M.; El-Bakly, W.M.; Esmat, A.; Badr, A.M.; El-Demerdash, E. Chrysin alleviates acute doxorubicin cardiotoxicity in rats via suppression of oxidative stress, inflammation and apoptosis. Eur. J. Pharmacol. 2014, 728, 107–118. [Google Scholar] [CrossRef]
- Chang, Y.H.; Chiang, Y.F.; Chen, H.Y.; Huang, Y.J.; Wang, K.L.; Hong, Y.H.; Ali, M.; Shieh, T.M.; Hsia, S.M. Anti-Inflammatory and Anti-Hyperuricemic Effects of Chrysin on a High Fructose Corn Syrup-Induced Hyperuricemia Rat Model via the Amelioration of Urate Transporters and Inhibition of NLRP3 Inflammasome Signaling Pathway. Antioxidants 2021, 10, 564. [Google Scholar] [CrossRef]
- Liao, T.; Ding, L.; Wu, P.; Zhang, L.; Li, X.; Xu, B.; Zhang, H.; Ma, Z.; Xiao, Y.; Wang, P. Chrysin Attenuates the NLRP3 Inflammasome Cascade to Reduce Synovitis and Pain in KOA Rats. Drug Des. Devel. Ther. 2020, 14, 3015–3027. [Google Scholar] [CrossRef]
- Wang, L.; Kou, M.C.; Weng, C.Y.; Hu, L.W.; Wang, Y.J.; Wu, M.J. Arsenic modulates heme oxygenase-1, interleukin-6, and vascular endothelial growth factor expression in endothelial cells: Roles of ROS, NF-kappaB, and MAPK pathways. Arch. Toxicol. 2012, 86, 879–896. [Google Scholar] [CrossRef]
- Qi, S.M.; Li, Q.; Jiang, Q.; Qi, Z.L.; Zhang, Y. Chrysin inhibits lipopolysaccharide-induced inflammatory responses of macrophages via JAK-STATs signaling pathway. Nan Fang Yi Ke Da Xue Xue Bao 2018, 38, 243–250. [Google Scholar] [PubMed]
- Elmore, S. Apoptosis: A review of programmed cell death. Toxicol. Pathol. 2007, 35, 495–516. [Google Scholar] [CrossRef] [PubMed]
- Wajant, H. The Fas signaling pathway: More than a paradigm. Science 2002, 296, 1635–1636. [Google Scholar] [CrossRef] [PubMed]
- Scaffidi, C.; Fulda, S.; Srinivasan, A.; Friesen, C.; Li, F.; Tomaselli, K.J.; Debatin, K.M.; Krammer, P.H.; Peter, M.E. Two CD95 (APO-1/Fas) signaling pathways. EMBO J. 1998, 17, 1675–1687. [Google Scholar] [CrossRef] [PubMed]
- Hsu, H.; Xiong, J.; Goeddel, D.V. The TNF receptor 1-associated protein TRADD signals cell death and NF-kappa B activation. Cell 1995, 81, 495–504. [Google Scholar] [CrossRef]
- Saelens, X.; Festjens, N.; Vande Walle, L.; van Gurp, M.; van Loo, G.; Vandenabeele, P. Toxic proteins released from mitochondria in cell death. Oncogene 2004, 23, 2861–2874. [Google Scholar] [CrossRef]
- Du, C.; Fang, M.; Li, Y.; Li, L.; Wang, X. Smac, a mitochondrial protein that promotes cytochrome c-dependent caspase activation by eliminating IAP inhibition. Cell 2000, 102, 33–42. [Google Scholar] [CrossRef]
- Schimmer, A.D. Inhibitor of apoptosis proteins: Translating basic knowledge into clinical practice. Cancer Res. 2004, 64, 7183–7190. [Google Scholar] [CrossRef]
- Ayna, A.; Ozbolat, S.N.; Darendelioglu, E. Quercetin, chrysin, caffeic acid and ferulic acid ameliorate cyclophosphamide-induced toxicities in SH-SY5Y cells. Mol. Biol. Rep. 2020, 47, 8535–8543. [Google Scholar] [CrossRef]
- Darendelioglu, E. Neuroprotective Effects of Chrysin on Diclofenac-Induced Apoptosis in SH-SY5Y Cells. Neurochem Res. 2020, 45, 1064–1071. [Google Scholar] [CrossRef]
- Guo, B.; Zheng, C.; Cai, W.; Cheng, J.; Wang, H.; Li, H.; Sun, Y.; Cui, W.; Wang, Y.; Han, Y.; et al. Multifunction of Chrysin in Parkinson’s Model: Anti-Neuronal Apoptosis, Neuroprotection via Activation of MEF2D, and Inhibition of Monoamine Oxidase-B. J. Agric. Food Chem. 2016, 64, 5324–5333. [Google Scholar] [CrossRef] [PubMed]
- Rashno, M.; Sarkaki, A.; Farbood, Y.; Rashno, M.; Khorsandi, L.; Naseri, M.K.G.; Dianat, M. Therapeutic effects of chrysin in a rat model of traumatic brain injury: A behavioral, biochemical, and histological study. Life Sci. 2019, 228, 285–294. [Google Scholar] [CrossRef]
- Thangarajan, S.; Ramachandran, S.; Krishnamurthy, P. Chrysin exerts neuroprotective effects against 3-Nitropropionic acid induced behavioral despair-Mitochondrial dysfunction and striatal apoptosis via upregulating Bcl-2 gene and downregulating Bax-Bad genes in male wistar rats. Biomed. Pharmacother. 2016, 84, 514–525. [Google Scholar] [CrossRef] [PubMed]
- Meiser, J.; Weindl, D.; Hiller, K. Complexity of dopamine metabolism. Cell Commun. Signal. 2013, 11, 34. [Google Scholar] [CrossRef]
- Del Fabbro, L.; Rossito Goes, A.; Jesse, C.R.; de Gomes, M.G.; Cattelan Souza, L.; Lobo Ladd, F.V.; Lobo Ladd, A.A.B.; Nunes Arantes, R.V.; Reis Simionato, A.; Oliveira, M.S.; et al. Chrysin protects against behavioral, cognitive and neurochemical alterations in a 6-hydroxydopamine model of Parkinson’s disease. Neurosci. Lett. 2019, 706, 158–163. [Google Scholar] [CrossRef] [PubMed]
- Bortolotto, V.C.; Pinheiro, F.C.; Araujo, S.M.; Poetini, M.R.; Bertolazi, B.S.; de Paula, M.T.; Meichtry, L.B.; de Almeida, F.P.; de Freitas Couto, S.; Jesse, C.R.; et al. Chrysin reverses the depressive-like behavior induced by hypothyroidism in female mice by regulating hippocampal serotonin and dopamine. Eur. J. Pharmacol. 2018, 822, 78–84. [Google Scholar] [CrossRef]
- Larit, F.; Elokely, K.M.; Chaurasiya, N.D.; Benyahia, S.; Nael, M.A.; Leon, F.; Abu-Darwish, M.S.; Efferth, T.; Wang, Y.H.; Belouahem-Abed, D.; et al. Inhibition of human monoamine oxidase A and B by flavonoids isolated from two Algerian medicinal plants. Phytomedicine 2018, 40, 27–36. [Google Scholar] [CrossRef]
- Krishnamoorthy, A.; Sevanan, M.; Mani, S.; Balu, M.; Balaji, S.; Ramajayan, P. Chrysin restores MPTP induced neuroinflammation, oxidative stress and neurotrophic factors in an acute Parkinson’s disease mouse model. Neurosci. Lett. 2019, 709, 134382. [Google Scholar] [CrossRef]
- Ngo, D.H.; Vo, T.S. An Updated Review on Pharmaceutical Properties of Gamma-Aminobutyric Acid. Molecules 2019, 24, 2678. [Google Scholar] [CrossRef]
- Nuss, P. Anxiety disorders and GABA neurotransmission: A disturbance of modulation. Neuropsychiatr. Dis. Treat. 2015, 11, 165–175. [Google Scholar]
- Marder, M.; Paladini, A.C. GABA(A)-receptor ligands of flavonoid structure. Curr. Top. Med. Chem. 2002, 2, 853–867. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Landa, J.F.; Guillen-Ruiz, G.; Hernandez-Lopez, F.; Cueto-Escobedo, J.; Rivadeneyra-Dominguez, E.; Bernal-Morales, B.; Herrera-Huerta, E.V. Chrysin reduces anxiety-like behavior through actions on GABAA receptors during metestrus-diestrus in the rat. Behav. Brain Res. 2021, 397, 112952. [Google Scholar] [CrossRef] [PubMed]
- Cueto-Escobedo, J.; Andrade-Soto, J.; Lima-Maximino, M.; Maximino, C.; Hernandez-Lopez, F.; Rodriguez-Landa, J.F. Involvement of GABAergic system in the antidepressant-like effects of chrysin (5,7-dihydroxyflavone) in ovariectomized rats in the forced swim test: Comparison with neurosteroids. Behav. Brain Res. 2020, 386, 112590. [Google Scholar] [CrossRef] [PubMed]
- Cho, I.; Song, H.O.; Cho, J.H. Flavonoids mitigate neurodegeneration in aged Caenorhabditis elegans by mitochondrial uncoupling. Food Sci. Nutr. 2020, 8, 6633–6642. [Google Scholar] [CrossRef]
- Cho, I.; Song, H.O.; Cho, J.H. Mitochondrial Uncoupling Attenuates Age-Dependent Neurodegeneration in C. elegans. Mol. Cells 2017, 40, 864–870. [Google Scholar]
- Ahmed, M.R.; Shaikh, M.A.; Ul Haq, S.H.I.; Nazir, S. Neuroprotective role of chrysin in attenuating loss of dopaminergic neurons and improving motor, learning and memory functions in rats. Int. J. Health Sci. 2018, 12, 35–43. [Google Scholar]
- Goes, A.T.R.; Jesse, C.R.; Antunes, M.S.; Lobo Ladd, F.V.; Lobo Ladd, A.A.B.; Luchese, C.; Paroul, N.; Boeira, S.P. Protective role of chrysin on 6-hydroxydopamine-induced neurodegeneration a mouse model of Parkinson’s disease: Involvement of neuroinflammation and neurotrophins. Chem. Biol. Interact. 2018, 279, 111–120. [Google Scholar] [CrossRef] [PubMed]
- Zhang, K.; Ge, Z.; Xue, Z.; Huang, W.; Mei, M.; Zhang, Q.; Li, Y.; Li, W.; Zhang, Z.; Zhang, Z.; et al. Chrysin suppresses human CD14(+) monocyte-derived dendritic cells and ameliorates experimental autoimmune encephalomyelitis. J. Neuroimmunol. 2015, 288, 13–20. [Google Scholar] [CrossRef]
- Mercer, L.D.; Kelly, B.L.; Horne, M.K.; Beart, P.M. Dietary polyphenols protect dopamine neurons from oxidative insults and apoptosis: Investigations in primary rat mesencephalic cultures. Biochem. Pharmacol. 2005, 69, 339–345. [Google Scholar] [CrossRef]
- Alkahtane, A.A.; Alghamdi, H.A.; Almutairi, B.; Khan, M.M.; Hasnain, M.S.; Abdel-Daim, M.M.; Alghamdi, W.M.; Alkahtani, S. Inhibition of human amylin aggregation by Flavonoid Chrysin: An in-silico and in-vitro approach. Int. J. Med. Sci. 2021, 18, 199–206. [Google Scholar] [CrossRef]
- Hirsch, E.C.; Standaert, D.G. Ten Unsolved Questions about Neuroinflammation in Parkinson’s Disease. Mov. Disord. 2021, 36, 16–24. [Google Scholar] [CrossRef]
- Giacomeli, R.; de Gomes, M.G.; Reolon, J.B.; Haas, S.E.; Colome, L.M.; Jesse, C.R. Chrysin loaded lipid-core nanocapsules ameliorates neurobehavioral alterations induced by beta-amyloid1-42 in aged female mice. Behav. Brain Res. 2020, 390, 112696. [Google Scholar] [CrossRef] [PubMed]
- Nday, C.M.; Eleftheriadou, D.; Jackson, G. Magnetic chrysin silica nanomaterials behavior in an amyloidogenic environment. Hell. J. Nucl. Med. 2019, 22, 42–50. [Google Scholar]
- Mansour, S.Z.; Moawed, F.S.M.; Elmarkaby, S.M. Protective effect of 5, 7-dihydroxyflavone on brain of rats exposed to acrylamide or gamma-radiation. J. Photochem. Photobiol. B 2017, 175, 149–155. [Google Scholar] [CrossRef]
- Sharma, P.; Kumari, A.; Gulati, A.; Krishnamurthy, S.; Hemalatha, S. Chrysin isolated from Pyrus pashia fruit ameliorates convulsions in experimental animals. Nutr. Neurosci. 2019, 22, 569–577. [Google Scholar] [CrossRef]
- Singh, B.; Singh, D.; Goel, R.K. Dual protective effect of Passiflora incarnata in epilepsy and associated post-ictal depression. J. Ethnopharmacol. 2012, 139, 273–279. [Google Scholar] [CrossRef]
- Singh, B.; Mishra, A.; Goel, R.K. Anticonvulsant activity of Passiflora incarnata: No role of chrysin. J. Pharm. Negat. Results 2011, 2, 51–54. [Google Scholar]
- Medina, J.H.; Paladini, A.C.; Wolfman, C.; Levi de Stein, M.; Calvo, D.; Diaz, L.E.; Pena, C. Chrysin (5,7-di-OH-flavone), a naturally-occurring ligand for benzodiazepine receptors, with anticonvulsant properties. Biochem. Pharmacol. 1990, 40, 2227–2231. [Google Scholar] [CrossRef]
- Rashno, M.; Ghaderi, S.; Nesari, A.; Khorsandi, L.; Farbood, Y.; Sarkaki, A. Chrysin attenuates traumatic brain injury-induced recognition memory decline, and anxiety/depression-like behaviors in rats: Insights into underlying mechanisms. Psychopharmacology 2020, 237, 1607–1619. [Google Scholar] [CrossRef]
- Shooshtari, M.K.; Sarkaki, A.; Mansouri, S.M.T.; Badavi, M.; Khorsandi, L.; Ghasemi Dehcheshmeh, M.; Farbood, Y. Protective effects of Chrysin against memory impairment, cerebral hyperemia and oxidative stress after cerebral hypoperfusion and reperfusion in rats. Metab. Brain Dis. 2020, 35, 401–412. [Google Scholar] [CrossRef] [PubMed]
- Sarkaki, A.; Farbood, Y.; Mansouri, S.M.T.; Badavi, M.; Khorsandi, L.; Dehcheshmeh, M.G.; Shooshtari, M.K. Chrysin prevents cognitive and hippocampal long-term potentiation deficits and inflammation in rat with cerebral hypoperfusion and reperfusion injury. Life Sci. 2019, 226, 202–209. [Google Scholar] [CrossRef]
- El Khashab, I.H.; Abdelsalam, R.M.; Elbrairy, A.I.; Attia, A.S. Chrysin attenuates global cerebral ischemic reperfusion injury via suppression of oxidative stress, inflammation and apoptosis. Biomed. Pharmacother. 2019, 112, 108619. [Google Scholar] [CrossRef]
- Durak, M.A.; Oztanir, M.N.; Basak Turkmen, N.; Ciftci, O.; Taslidere, A.; Tecellioglu, M.; Onder, A. Chrysin prevents brain damage caused by global cerebralischemia/reperfusion in a C57BL/J6 mouse model. Turk. J. Med. Sci. 2016, 46, 1926–1933. [Google Scholar] [CrossRef] [PubMed]
- Yao, Y.; Chen, L.; Xiao, J.; Wang, C.; Jiang, W.; Zhang, R.; Hao, J. Chrysin protects against focal cerebral ischemia/reperfusion injury in mice through attenuation of oxidative stress and inflammation. Int. J. Mol. Sci. 2014, 15, 20913–20926. [Google Scholar] [CrossRef] [PubMed]
- He, X.L.; Wang, Y.H.; Bi, M.G.; Du, G.H. Chrysin improves cognitive deficits and brain damage induced by chronic cerebral hypoperfusion in rats. Eur.. Pharmacol. 2012, 680, 41–48. [Google Scholar] [CrossRef]
- Wang, J.; Wang, H.; Sun, K.; Wang, X.; Pan, H.; Zhu, J.; Ji, X.; Li, X. Chrysin suppresses proliferation, migration, and invasion in glioblastoma cell lines via mediating the ERK/Nrf2 signaling pathway. Drug Des. Devel. Ther. 2018, 12, 721–733. [Google Scholar] [CrossRef]
- Jia, W.Z.; Zhao, J.C.; Sun, X.L.; Yao, Z.G.; Wu, H.L.; Xi, Z.Q. Additive anticancer effects of chrysin and low dose cisplatin in human malignant glioma cell (U87) proliferation and evaluation of the mechanistic pathway. J. BUON 2015, 20, 1327–1336. [Google Scholar]
- Santos, B.L.; Oliveira, M.N.; Coelho, P.L.; Pitanga, B.P.; da Silva, A.B.; Adelita, T.; Silva, V.D.; Costa Mde, F.; El-Bacha, R.S.; Tardy, M.; et al. Flavonoids suppress human glioblastoma cell growth by inhibiting cell metabolism, migration, and by regulating extracellular matrix proteins and metalloproteinases expression. Chem. Biol. Interact. 2015, 242, 123–138. [Google Scholar] [CrossRef] [PubMed]
- Liao, C.L.; Chen, C.M.; Chang, Y.Z.; Liu, G.Y.; Hung, H.C.; Hsieh, T.Y.; Lin, C.L. Pine (Pinus morrisonicola Hayata) needle extracts sensitize GBM8901 human glioblastoma cells to temozolomide by downregulating autophagy and O(6)-methylguanine-DNA methyltransferase expression. J. Agric. Food Chem. 2014, 62, 10458–10467. [Google Scholar] [CrossRef] [PubMed]
- Weng, M.S.; Ho, Y.S.; Lin, J.K. Chrysin induces G1 phase cell cycle arrest in C6 glioma cells through inducing p21Waf1/Cip1 expression: Involvement of p38 mitogen-activated protein kinase. Biochem. Pharmacol. 2005, 69, 1815–1827. [Google Scholar] [CrossRef]
- Del Fabbro, L.; de Gomes, M.G.; Souza, L.C.; Goes, A.R.; Boeira, S.P.; Oliveira, M.S.; Furian, A.F.; Jesse, C.R. Chrysin suppress immune responses and protects from experimental autoimmune encephalomyelitis in mice. J. Neuroimmunol. 2019, 335, 577007. [Google Scholar] [CrossRef]
- Zhang, Z.; Li, G.; Szeto, S.S.W.; Chong, C.M.; Quan, Q.; Huang, C.; Cui, W.; Guo, B.; Wang, Y.; Han, Y.; et al. Examining the neuroprotective effects of protocatechuic acid and chrysin on in vitro and in vivo models of Parkinson disease. Free Radic. Biol. Med. 2015, 84, 331–343. [Google Scholar] [CrossRef]
- Li, Z.; Chu, S.; He, W.; Zhang, Z.; Liu, J.; Cui, L.; Yan, X.; Li, D.; Chen, N. A20 as a novel target for the anti-neuroinflammatory effect of chrysin via inhibition of NF-kappaB signaling pathway. Brain Behav. Immun. 2019, 79, 228–235. [Google Scholar] [CrossRef]
- Mani, R.; Natesan, V.; Arumugam, R. Neuroprotective effect of chrysin on hyperammonemia mediated neuroinflammatory responses and altered expression of astrocytic protein in the hippocampus. Biomed. Pharmacother. 2017, 88, 762–769. [Google Scholar] [CrossRef]
- Filho, C.B.; Jesse, C.R.; Donato, F.; Giacomeli, R.; Del Fabbro, L.; da Silva Antunes, M.; de Gomes, M.G.; Goes, A.T.; Boeira, S.P.; Prigol, M.; et al. Chronic unpredictable mild stress decreases BDNF and NGF levels and Na(+),K(+)-ATPase activity in the hippocampus and prefrontal cortex of mice: Antidepressant effect of chrysin. Neuroscience 2015, 289, 367–380. [Google Scholar] [CrossRef]
- Jiang, Y.; Gong, F.L.; Zhao, G.B.; Li, J. Chrysin suppressed inflammatory responses and the inducible nitric oxide synthase pathway after spinal cord injury in rats. Int. J. Mol. Sci. 2014, 15, 12270–12279. [Google Scholar] [CrossRef]
- Mehri, S.; Karami, H.V.; Hassani, F.V.; Hosseinzadeh, H. Chrysin reduced acrylamide-induced neurotoxicity in both in vitro and in vivo assessments. Iran. Biomed. J. 2014, 18, 101–106. [Google Scholar]
- Cummings, J. New approaches to symptomatic treatments for Alzheimer’s disease. Mol. Neurodegener. 2021, 16, 2. [Google Scholar] [CrossRef]
- Qi, Y.; Cheng, X.; Jing, H.; Yan, T.; Xiao, F.; Wu, B.; Bi, K.; Jia, Y. Comparative pharmacokinetic study of the components in Alpinia oxyphylla Miq.-Schisandra chinensis (Turcz.) Baill. herb pair and its single herb between normal and Alzheimer’s disease rats by UPLC-MS/MS. J. Pharm. Biomed. Anal. 2020, 177, 112874. [Google Scholar] [CrossRef]
- Bortolotto, V.C.; Araujo, S.M.; Pinheiro, F.C.; Poetini, M.R.; de Paula, M.T.; Meichtry, L.B.; de Almeida, F.P.; Musachio, E.A.S.; Guerra, G.P.; Prigol, M. Modulation of glutamate levels and Na(+),K(+)-ATPase activity contributes to the chrysin memory recovery in hypothyroidism mice. Physiol. Behav. 2020, 222, 112892. [Google Scholar] [CrossRef]
- Akyuz, E.; Polat, A.K.; Eroglu, E.; Kullu, I.; Angelopoulou, E.; Paudel, Y.N. Revisiting the role of neurotransmitters in epilepsy: An updated review. Life Sci. 2021, 265, 118826. [Google Scholar] [CrossRef]
- Mishra, A.; Goel, R.K. Modulatory Effect of Serotonergic System in Pentylenetetrazole-Induced Seizures and Associated Memory Deficit: Role of 5-HT1A and 5-HT2A/2C. J. Epilepsy Res. 2019, 9, 119–125. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Mishra, A.; Goel, R.K. Chronic 5-HT3 receptor antagonism ameliorates seizures and associated memory deficit in pentylenetetrazole-kindled mice. Neuroscience 2016, 339, 319–328. [Google Scholar] [CrossRef]
- Stompor-Gorący, M.; Bajek-Bil, A.; Machaczka, M. Chrysin: Perspectives on Contemporary Status and Future Possibilities as Pro-Health Agent. Nutrients 2021, 13, 2038. [Google Scholar] [CrossRef] [PubMed]
- Markiewicz-Zukowska, R.; Borawska, M.H.; Fiedorowicz, A.; Naliwajko, S.K.; Sawicka, D.; Car, H. Propolis changes the anticancer activity of temozolomide in U87MG human glioblastoma cell line. BMC Complement. Altern. Med. 2013, 13, 50. [Google Scholar] [CrossRef]
- Balakrishnan, R.; Azam, S.; Cho, D.Y.; Su-Kim, I.; Choi, D.K. Natural Phytochemicals as Novel Therapeutic Strategies to Prevent and Treat Parkinson’s Disease: Current Knowledge and Future Perspectives. Oxid Med. Cell Longev. 2021, 2021, 6680935. [Google Scholar]
- Gao, S.; Siddiqui, N.; Etim, I.; Du, T.; Zhang, Y.; Liang, D. Developing nutritional component chrysin as a therapeutic agent: Bioavailability and pharmacokinetics consideration, and ADME mechanisms. Biomed. Pharmacother. 2021, 142, 112080. [Google Scholar] [CrossRef]
- Hofer, S.J.; Davinelli, S.; Bergmann, M.; Scapagnini, G.; Madeo, F. Caloric Restriction Mimetics in Nutrition and Clinical Trials. Front. Nutr. 2021, 8, 717343. [Google Scholar] [CrossRef]
- Dong, D.; Quan, E.; Yuan, X.; Xie, Q.; Li, Z.; Wu, B. Sodium Oleate-Based Nanoemulsion Enhances Oral Absorption of Chrysin through Inhibition of UGT-Mediated Metabolism. Mol. Pharm. 2017, 14, 2864–2874. [Google Scholar] [CrossRef]
- Ge, S.; Gao, S.; Yin, T.; Hu, M. Determination of pharmacokinetics of chrysin and its conjugates in wild-type FVB and Bcrp1 knockout mice using a validated LC-MS/MS method. J. Agric. Food Chem. 2015, 63, 2902–2910. [Google Scholar] [CrossRef]
- Mohos, V.; Fliszár-Nyúl, E.; Schilli, G.; Hetényi, C.; Lemli, B.; Kunsági-Máté, S.; Bognár, B.; Poór, M. Interaction of Chrysin and Its Main Conjugated Metabolites Chrysin-7-Sulfate and Chrysin-7-Glucuronide with Serum Albumin. Int. J. Mol. Sci. 2018, 19, 4073. [Google Scholar] [CrossRef]
- Mohos, V.; Fliszár-Nyúl, E.; Ungvári, O.; Bakos, É.; Kuffa, K.; Bencsik, T.; Zsidó, B.Z.; Hetényi, C.; Telbisz, Á.; Özvegy-Laczka, C.; et al. Effects of Chrysin and Its Major Conjugated Metabolites Chrysin-7-Sulfate and Chrysin-7-Glucuronide on Cytochrome P450 Enzymes and on OATP, P-gp, BCRP, and MRP2 Transporters. Drug Metab. Dispos. 2020, 48, 1064–1073. [Google Scholar] [CrossRef]
- Anari, E.; Akbarzadeh, A.; Zarghami, N. Chrysin-loaded PLGA-PEG nanoparticles designed for enhanced effect on the breast cancer cell line. Artif Cells Nanomed. Biotechnol. 2016, 44, 1410–1416. [Google Scholar] [CrossRef] [PubMed]
- Chadha, R.; Bhalla, Y.; Nandan, A.; Chadha, K.; Karan, M. Chrysin cocrystals: Characterization and evaluation. J. Pharm. Biomed. 2017, 134, 361–371. [Google Scholar] [CrossRef] [PubMed]
- Abbas, M. Potential Role of Nanoparticles in Treating the Accumulation of Amyloid-Beta Peptide in Alzheimer’s Patients. Polymers 2021, 13, 1051. [Google Scholar] [CrossRef]
- Sharma, T.; Katare, O.P.; Jain, A.; Jain, S.; Chaudhari, D.; Borges, B.; Singh, B. QbD-Steered Development of Biotin-Conjugated Nanostructured Lipid Carriers for Oral Delivery of Chrysin: Role of Surface Modification for Improving Biopharmaceutical Performance. Colloids Surf. B Biointerfaces. 2021, 197, 111429. [Google Scholar] [CrossRef]

| # | Dose and Route | Experimental Model | Animal/Cell Lines | Outcome | Ref. |
|---|---|---|---|---|---|
| In Parkinson’s Disease | |||||
| 1. | Chrysin (100 µM) | 2,4-dinitrophenol-induced mitophagy | Caenorhabditis elegans (Bristol N2 wild-type, ucp-4 deletion mutant, pdr-1, and zdIs5) | Chrysin served as mitochondrial uncoupler and mitigated neurodegeneration possibly via PINK1/Parkin mitophagy | [75] |
| 2. | Chrysin (50, 100, and 200 mg/kg, p.o.) for 5 days | MPTP-induced rodent model of PD | Male C57BL/6 J mice (18–22 g) | Improvement in motor dysfunction and dopaminergic neuroprotection in nigro-striatal region, possibly by mitigating oxidative stress and neuroinflammation | [68] |
| 3. | Chrysin (10 mg/kg, i.g.) for 28 days | 6-OHDA-induced rodent model of PD | Female C57B/6 J mice (30–40 g, 20 months) | Improvement in motor and cognitive functions along with reduction in oxidative stress, neuroinflammation | [65] |
| 4. | Chrysin (50 mg/Kg, i.p.) for 4 weeks | Rotenone-induced rodent model of PD | Sprague–Dawley rats | Improvement in motor impairments and attenuation of nigrostriatal dopaminergic neurodegeneration | [76] |
| 5. | Chrysin (10 mg/kg, p.o.) for 28 days | 6-OHDA-induced rodent model of PD | Male C57B/6 J mice (20–30g, 90 days) | Improvement in motor functions and restoration of dopaminergic neurons, inflammatory cytokines, and neurotrophic factors levels | [77] |
| 6 | Chrysin (50 mM) | MPP+-induced neurotoxicity | Primary Cerebellar Granule Neuron Culture | Neuroprotection via inhibition of apoptosis by activating MEF2D via AKT-GSK3β signaling | [61] |
| 7 | Chrysin (10, 100 mg/kg, p.o.) | MPTP-induced rodent model of PD | Male C57BL/6 mice (28 ± 2 g, 8–9 weeks) | Restoration of dopaminergic neuronal loss via anti-apoptotic, activation of the AKT-GSK3β/MEF2D pathway, and inhibition of MAO-B activity | [61] |
| 8 | Chrysin (25 µM) | 6-OHDA-induced neurotoxicity | Rat pheochromocytoma (PC12) cells | Neuroprotection by attenuating oxidative stress (NRF-2/HO-1 pathway), neuroinflammation (NF-κB/iNOS pathway) | [78] |
| 9 | Chrysin (3, 6, and 12 µM) | 6-OHDA-induced dopaminergic neurotoxicity | Zebrafish larvae (AB strain) | Protection of dopaminergic neurons | [78] |
| 10 | Chrysin (40 µM) | MPP+-induced neurotoxicity | Primary mesencephalic neurons | Neuroprotection of mesencephalic dopaminergic neurons via attenuation of oxidative stress and apoptosis | [79] |
| In Alzheimer’s Disease | |||||
| 1 | Chrysin (50 µM) | Amylin-induced amylin amyloidosis | C6 Rat Glioma Cell lines | Reduction in amylin amyloidosis | [80] |
| 2 | Chrysin (10 mg/kg, p.o.) for 3 months | Zinc-induced cognitive impairment and amyloidosis | Male Swiss mice | Improvement in cognitive functions and neuroprotection of hippocampal neurons | [81] |
| 3 | Chrysin loaded lipid-core nano capsules (1 and 5 mg/kg., p.o.) for 14 days | Aβ1–42-induced animal model of AD | Female Swiss mice (30–35 g, 18–22 months) | Improvement in learning and memory impairment via attenuation of oxidative stress and neuroinflammation | [82] |
| 4 | Chrysin loaded magnetic PEGylated silica nanospheres (30 µM) | Aβ-induced amyloidosis in hippocampal culture model | Sprague–Dawley rat (neonates) | Improved antioxidant profile and protection against Aβ-induced oxidative stress | [83] |
| 5 | Chrysin (50 mg/kg) for 21 days | Acrylamide or gamma-irradiation induced neurotoxicity | Male Wister rats (120–150 g) | Neuroprotective effect via attenuation of oxidative stress, amyloidosis, and apoptosis | [84] |
| 6 | Chrysin loaded sloid lipid nanoparticles (5, 10, 50, 100 mg/kg, p.o.) for 21 days | Aβ25–35-induced rodent model of AD | Male Sprague–Dawley rats (250–300 g) | Improved memory impairment and amelioration of hippocampal neuronal loss possibly via mitigation of neuronal loss | [22] |
| In Epilepsy | |||||
| 1 | Chrysin-loaded PLGA nanoparticle (5 and 10 µg/mL) | PTZ-induced kindling | Wistar rats | Anticonvulsant effect through mitigating oxidative stress via the NRF2/HO-1 pathway | [15] |
| 2 | Chrysin (2.5, 5, and 10 mg/kg; p.o.) | PTZ-induced convulsions | Male Charles Foster rats (180–220 g) | Anticonvulsant effect possibly via alleviation of oxidative stress in hippocampus and cortex | [85] |
| 3 | Hydroethanolic extract of Passiflora incarnata (150, 300, and 600 mg/kg, i.p.) for 11 days | PTZ-induced kindling | Male Swiss mice (20–30 g) | Presence of chrysin in the extract and anticonvulsant, antidepressant effects | [86] |
| 4 | Extract of Passiflora incarnata (150, 300, and 600 mg/kg; i.p.) | PTZ-induced convulsions | Male Swiss mice (25–35 g) | Presence of chrysin in the extracts and anticonvulsant effect of the extracts | [87] |
| 5 | Chrysin (40 µg, i.c.v.) | PTZ-induced convulsions | Swiss mice (22–28 g) | Anticonvulsant effect via activation of benzodiazepine receptors | [88] |
| Chrysin in Traumatic Brain Injury | |||||
| 1 | Chrysin (25, 50 and 100 mg/kg, p.o.) | Closed head weight-drop-induced rodent model TBI | Male Wistar rats (250–300 g) | Improved neurobehavioral impairments possibly via modulation of inflammation and apoptosis | [89] |
| 2 | Chrysin (25, 50 and 100 mg/kg, p.o.) | Closed head weight-drop-induced rodent model TBI | Adult male Wistar rats (250–300 g) | Improved motor coordination and memory impairment possibly via anti-oxidant and anti-apoptotic effects | [62] |
| Chrysin in Ischemic Brain Injury | |||||
| 1 | Chrysin (10, and 20 mg/kg) for 7 days | Middle cerebral artery occlusion-induced cerebral ischemia/reperfusion injury model | Male Sprague-Dawley rats (250–280 g) | Reduction in ischemia/reperfusion injury in brain, possibly via alleviation of proinflammatory cytokine release and improvement of antioxidant defense by activating the PI3K/Akt/mTOR pathway | [45] |
| 2 | Chrysin (10, 30, and 100 mg/kg, p.o.) for 21 days | Bilateral common carotid arteries occlusion model of cerebral ischemia reperfusion injury | Male Wistar rats (250–300 g, 6 month) | Improvement in cognitive impairment and restoration of hippocampal neurons possibly by reducing oxidative stress and PGE2 levels | [90] |
| 3 | Chrysin (10, 30, and 100 mg/kg, p.o.) for 21 days | Bilateral common carotid arteries occlusion model of cerebral ischemia reperfusion injury | Male Wistar rats (250–300 g) | Improvement in cognitive impairment possibly by alleviating neuroinflammation | [91] |
| 4 | Chrysin (30 mg/kg, p.o.) for 14 days | Bilateral common carotid arteries occlusion model of cerebral ischemia reperfusion injury | Male Wistar rats (200–250 g) | Neuroprotection against ischemia reperfusion injury possibly by attenuating oxidative stress | [92] |
| 5 | Chrysin (50 mg/kg, p.o.) for 10 days | Bilateral common carotid arteries occlusion model of cerebral ischemia reperfusion injury | Male C57BL/6 J mice (18–22 g) | Reduction in degenerative changes in neurons possibly by mitigating oxidative stress | [93] |
| 6 | Chrysin (75 mg/kg, p.o.) for 7 days | Middle cerebral artery occlusion-induced ischemia reperfusion injury | Male C57/BL6 mice (10–12 weeks) | Reduction in neurological deficit scores and infarct volumes, possibly via inhibition of neuroinflammation (by suppression of NF-κB, COX-2, and iNOS expression) | [94] |
| 7 | Chrysin (30, and 100 mg/kg; i.g.) for 26 days | Bilateral common carotid arteries occlusion model of cerebral ischemia reperfusion injury | Male Wistar rats, (330–350 g) | Improvement in dementia and neurodegeneration possibly via attenuation of oxidative stress and neuroinflammation | [95] |
| Chrysin in gliomas | |||||
| 1 | Chrysin (5, 30, 60, 120, and 240 µM) | Human glioblastoma cell lines | T98, U251, U87 cells | Anticancer activity in glioblastoma cell lines possibly via the ERK/Nrf2 signaling pathway | [96] |
| 2 | Chrysin (10, 20, 40, 80 and 120 µM) and Cisplatin (0.5, 1.0 and 2.0 µM) combination | Human glioma cell lines | U87 cells | Potentiation of antiproliferative effect of Cisplatin | [97] |
| 3 | Chrysin (50 µM) | Human glioblastoma cell lines | GL-15 and U251 cells | Damaged mitochondria, and rough endoplasmic reticulum, apoptosis, and reduction in MMP-2 expression | [98] |
| 4 | Chrysin (100 µM) | Human glioblastoma cell lines | GBM8901 glioblastoma cells | Induction of apoptosis and suppression of migration and invasion. Inhibition of temozolomide-induced autophagy and O6-methylguanine-DNA | [99] |
| 5 | Chrysin (10, 30, and 50 µM) | Rat glioma cell line | C6 glioma cells | Induction of G1 phase cell cycle arrest through induction of p21Waf1/Cip1 and inhibition of proteasome activity | [100] |
| Chrysin in MS | |||||
| 1 | Chrysin (20 mg/kg, i.g.) for 25 days | Myelin oligodendrocyte glycoprotein-induced EAE | Male C57BL/6 mice (20–25 g) | Reduction in HDAC activity, GSK-3β and proinflammatory cytokine release | [101] |
| 2 | Chrysin (100 mg/kg, i.g.) for 3 days | Myelin oligodendrocyte glycoprotein-induced EAE | Female C57BL/6 mice (6–8 weeks) | Amelioration of EAE and anti-inflammatory and immune suppressive effects via suppression of dendritic cells and Th1 cells | [102] |
| Miscellaneous | |||||
| 1 | Chrysin (0.05 mM) | Diclofenac-induced neurotoxicity | SH-SY5Y cells | Neuroprotective effect of mitigating oxidative stress and apoptosis | [60] |
| 2 | Chrysin (400 µM) | Cyclophosphamide-induced neurotoxicity | SH-SY5Y cells | Neuroprotection via suppression of oxidative stress and apoptotic cell death | [59] |
| 3 | Chrysin (5, and 10 μM) | LPS-induced inflammation | BV2 microglia cells and Primary mouse microglia cells | Suppression of neuroinflammation by downregulating NF-κB/TRAF6 pathway and upregulating zinc figure protein A20 | [103] |
| Chrysin (25 and 50 mg/kg, p.o.) for 4 days | LPS-induced neuroinflammation | Male Balb/c mice (20 ± 2 g, 8 weeks) | |||
| 4 | Chrysin (20 mg/kg, i.g.) for 28 days | Methimazole induced hypothyroidism and associated neurobehavioral impairments | Female C57BL/6 mice (3–4 months) | Improvement in depression-like behavior via improvement of cortical and hippocampal serotonin levels and hippocampal dopamine levels | [66] |
| 5 | Chrysin (60, 80, 100, 150 and 200 µg/mL) | LPS-induced neuroinflammation | RAW264.7 macrophage cells | Reduction in inflammatory response by blocking the JAK-STAT pathway mediated by ROS | [51] |
| 6 | Chrysin (10–100 μM) | LPS-induced in vitro study | Mouse cerebral vascular endothelial (bEnd.3) cells | Reduction in VCAM-1 expression by inhibition of NF-κB/MAPK pathway resulted in anti-inflammatory effect | [46] |
| 7 | Chrysin (100 mg/kg, p.o.) | Ammonium chloride-induced neuroinflammation | Male Wistar rats | Attenuation of neuroinflammation by reducing expression of pro-inflammatory markers (TNF-α, IL-1β, IL-6, NF-κB, iNOS and COX-2) in the brain | [104] |
| 8 | Chrysin (50 mg/kg, p.o.) for 14 days | 3-nitro propionic acid-induced neurotoxicity | Male Wistar rats (250–300 g) | Improvement in neurobehavioral impairments, mitochondrial dysfunction, oxidative stress, and apoptosis | [63] |
| 9 | Chrysin (10 mg/kg, p.o.) for 60 days | Age-related memory decline in mice | Male Swiss Mice (3 and 20 months) | Improvement in age-related memory decline by attenuating oxidative stress and Na+/K+ ATPase activity in prefrontal cortex and hippocampus | [26] |
| 10 | Chrysin (5 and 20 mg/kg, p.o.) for 28 days | Chronic unpredictable mild stress | Female C57B/6 J mice (20–25 g, 90 days) | Alleviation of depression-like symptoms possibly by upregulation of BDNF, NGF levels, antioxidant defense factor (GPx, GR, Catalase) and reduction in ROS level and Na+/K+ ATPase activity | [105] |
| 11 | Chrysin (30 and 100 mg/kg, i.g.) for 26 days | Weight-drop method-induced spinal cord injury model | Wistar rats (230–250 g) | Augmentation in neuronal recovery and reduction in pro-inflammatory markers and iNOS expression | [106] |
| 12 | Chrysin in vitro (0.5–5 µM) for 12 and 24 h | Acrylamide-induced neurotoxicity | PC12 cells | Neuroprotection | [107] |
| 13 | Chrysin in vivo (12.5, 25, and 50 mg/kg) | Acrylamide-induced toxicity in vivo | Male Wistar rats (230–250 g) | Reduction in gait abnormality | [107] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mishra, A.; Mishra, P.S.; Bandopadhyay, R.; Khurana, N.; Angelopoulou, E.; Paudel, Y.N.; Piperi, C. Neuroprotective Potential of Chrysin: Mechanistic Insights and Therapeutic Potential for Neurological Disorders. Molecules 2021, 26, 6456. https://doi.org/10.3390/molecules26216456
Mishra A, Mishra PS, Bandopadhyay R, Khurana N, Angelopoulou E, Paudel YN, Piperi C. Neuroprotective Potential of Chrysin: Mechanistic Insights and Therapeutic Potential for Neurological Disorders. Molecules. 2021; 26(21):6456. https://doi.org/10.3390/molecules26216456
Chicago/Turabian StyleMishra, Awanish, Pragya Shakti Mishra, Ritam Bandopadhyay, Navneet Khurana, Efthalia Angelopoulou, Yam Nath Paudel, and Christina Piperi. 2021. "Neuroprotective Potential of Chrysin: Mechanistic Insights and Therapeutic Potential for Neurological Disorders" Molecules 26, no. 21: 6456. https://doi.org/10.3390/molecules26216456
APA StyleMishra, A., Mishra, P. S., Bandopadhyay, R., Khurana, N., Angelopoulou, E., Paudel, Y. N., & Piperi, C. (2021). Neuroprotective Potential of Chrysin: Mechanistic Insights and Therapeutic Potential for Neurological Disorders. Molecules, 26(21), 6456. https://doi.org/10.3390/molecules26216456