Pressurized Hot Water Extraction and Bio-Hydrogels Formulation with Aristotelia chilensis [Mol.] Stuntz Leaves
Abstract
:1. Introduction
2. Results and Discussion
2.1. Characterization of the Raw Material
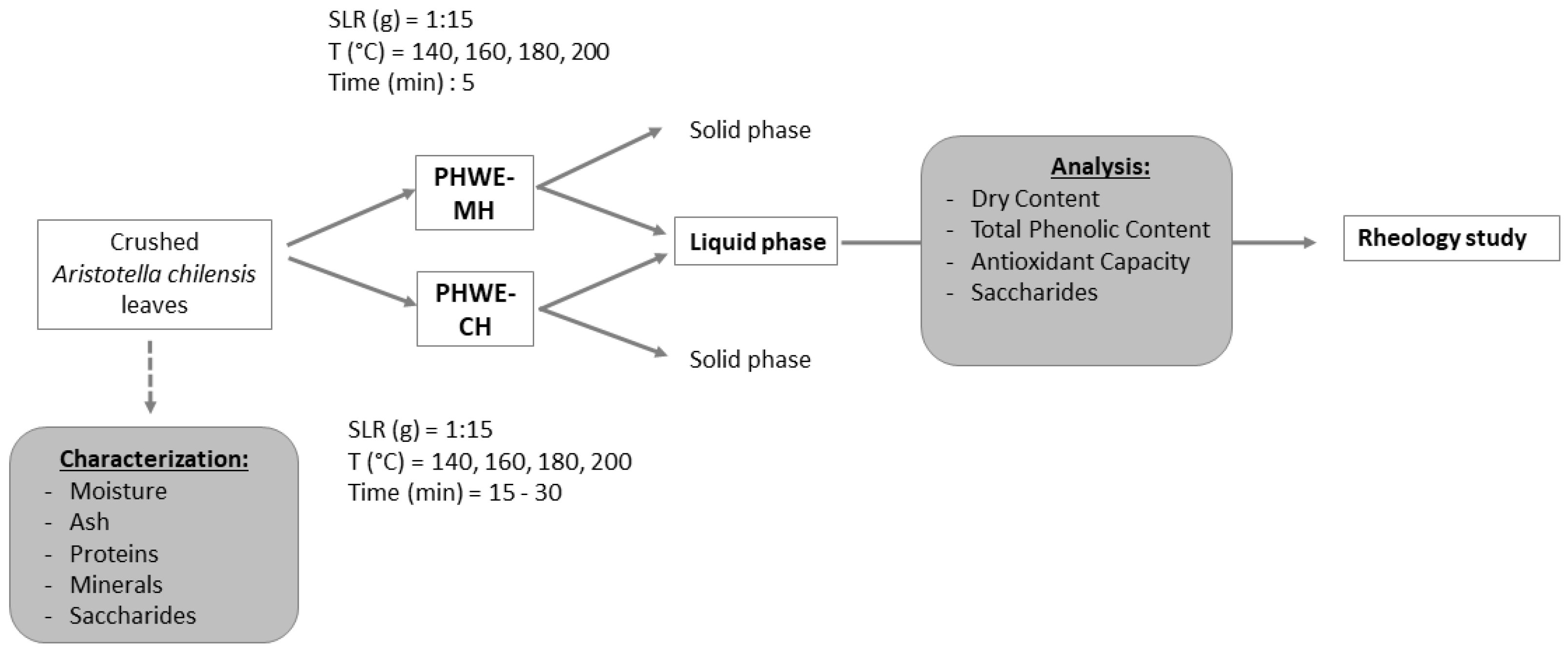
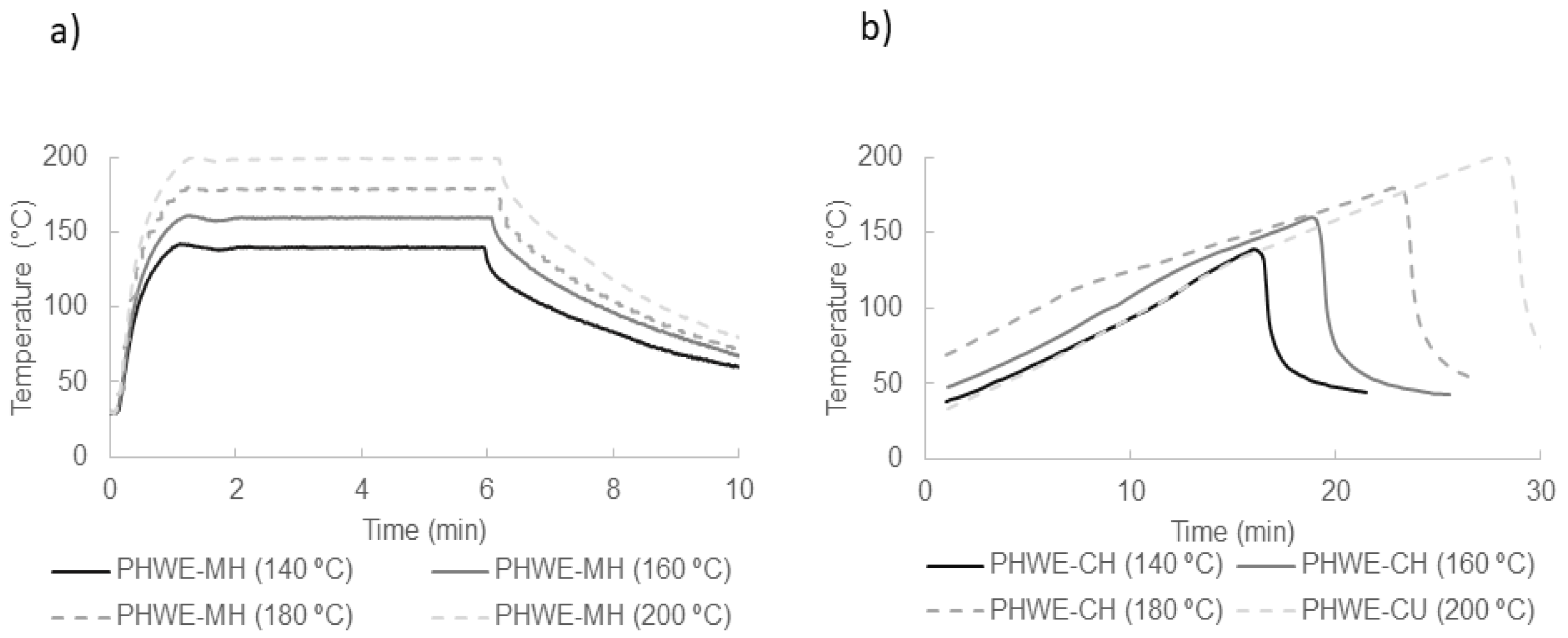
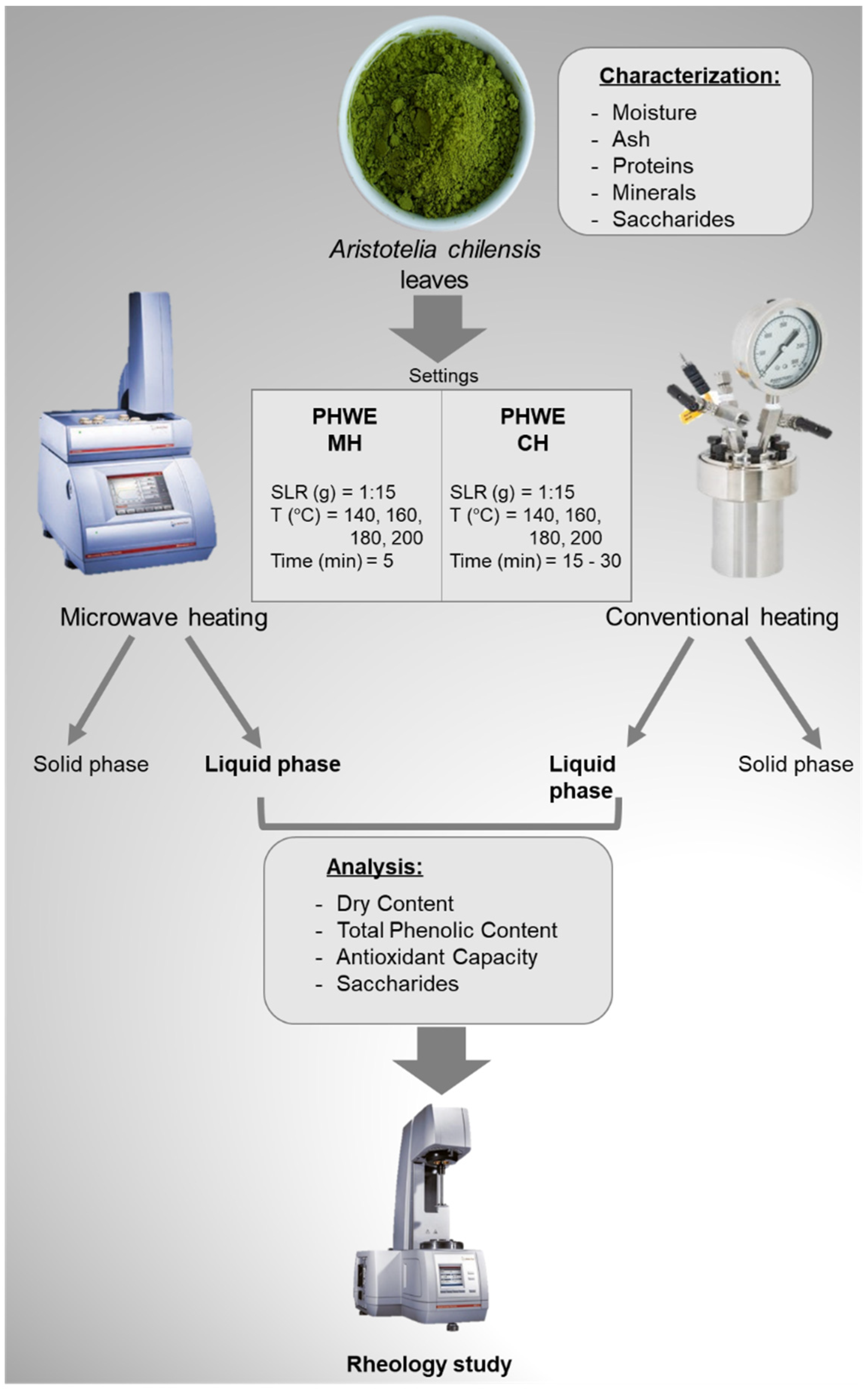
2.2. Extraction
2.3. Liquid Phase Composition
2.3.1. Total Extraction Yield and Phenolic Content
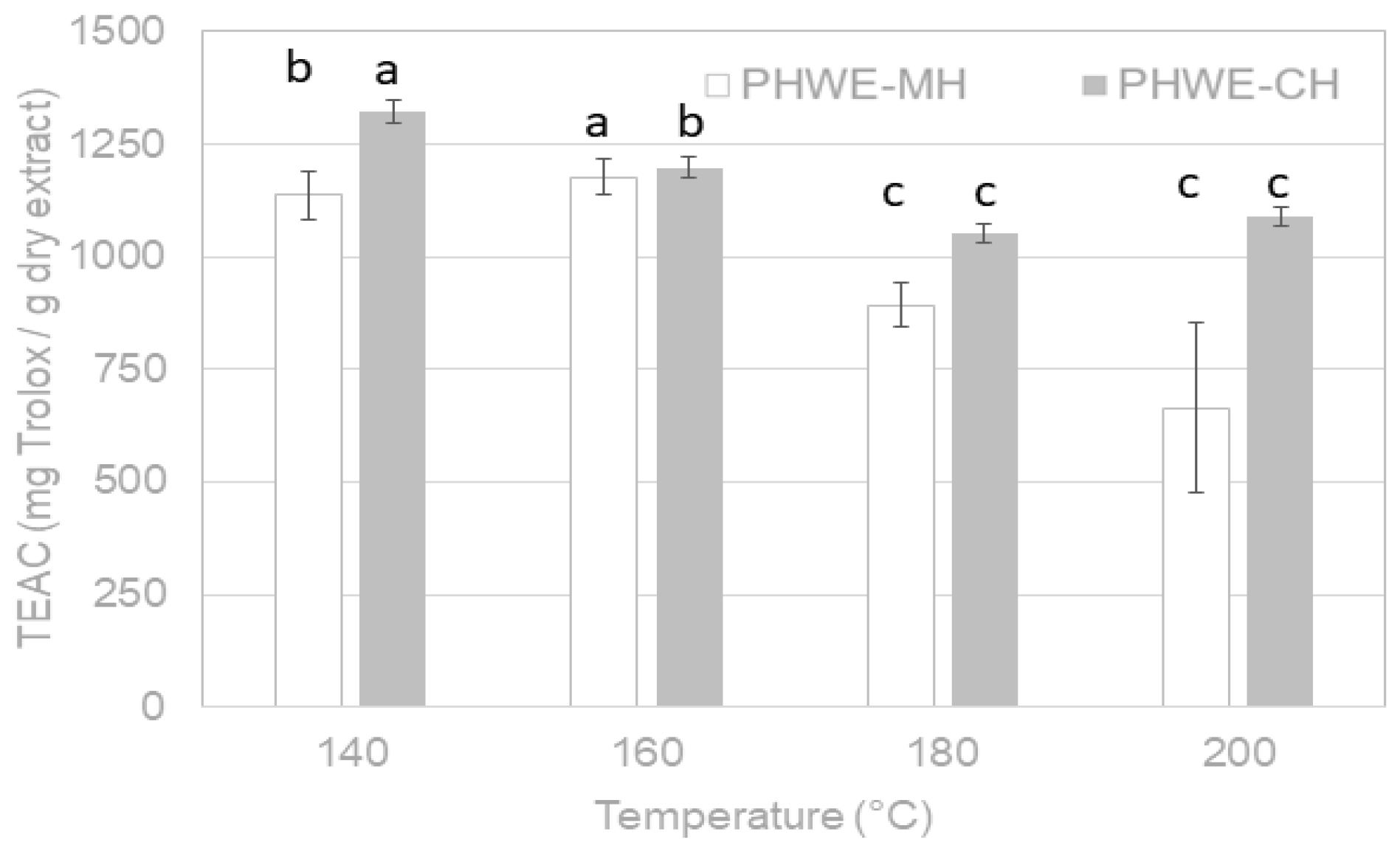
2.3.2. Radical Scavenging Capacity
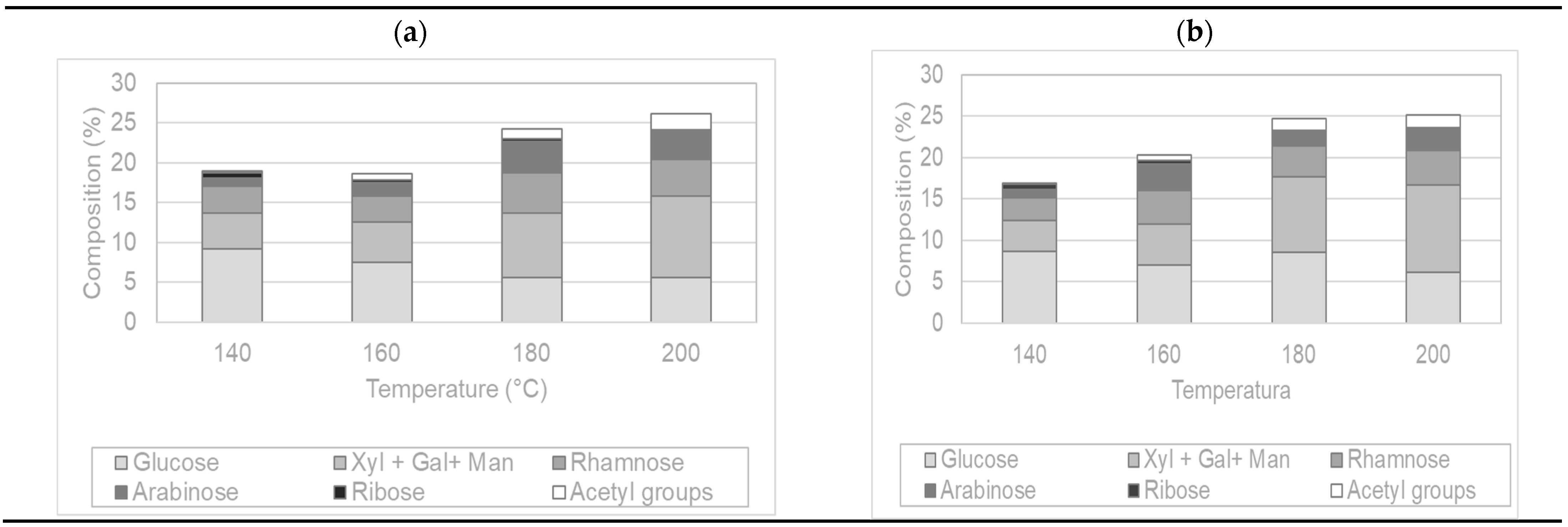
2.3.3. Saccharides and Derivatives Content
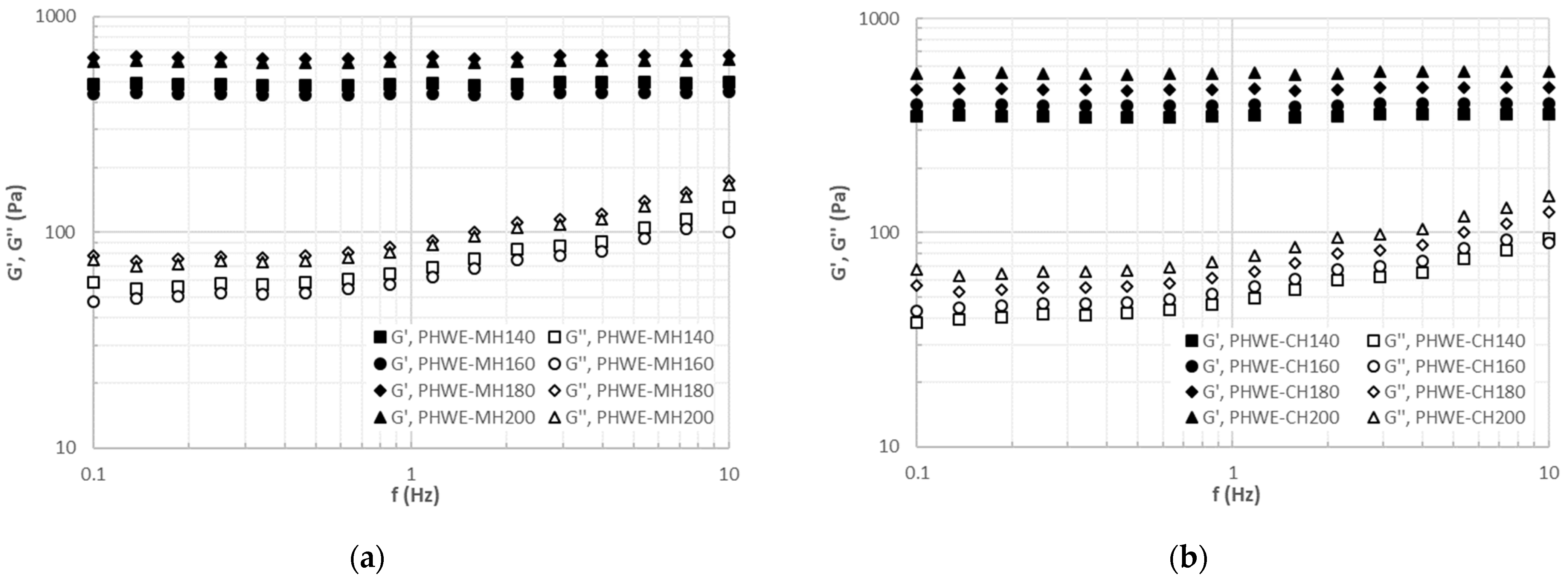
2.4. Rheological Properties of Enriched Hydrogels
3. Materials and Methods
3.1. Raw Material
3.2. Physicochemical Characterization of the Raw Material
3.3. Extraction Technologies
3.3.1. Hot Pressurized Liquid Extraction with Microwave Heating (PHWE-MH)
3.3.2. Hot Pressurized Liquid Extraction with Conventional Heating (PHWE-CH)
3.4. Aqueous Extracts Analysis
3.4.1. Dry Weight Content
3.4.2. Total Phenolic Content
3.4.3. Antiradical Capacity (TEAC Value)
3.4.4. Saccharides and Derivate Compounds Content
3.5. Gelling Matrices
3.6. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Misle, E.; Garrido, E.; Contardo, H.; González, W. Maqui [Aristotelia chilensis (Mol.) Stuntz]-the Amazing Chilean Tree: A Review. J. Agric. Sci. Technol. B 2011, 1, 473–482. [Google Scholar]
- Rodríguez, K.; Ah-Hen, K.S.; Vega-Gálvez, A.; Vásquez, V.; Quispe-Fuentes, I.; Rojas, P.; Lemus-Mondaca, R. Changes in bioactive components and antioxidant capacity of maqui, Aristotelia chilensis [Mol] Stuntz, berries during drying. LWT-Food Sci. Technol. 2016, 65, 537–542. [Google Scholar] [CrossRef]
- Zúñiga, G.E.; Tapia, A.; Arenas, A.; Contreras, R.A.; Zúñiga-Libano, G. Phytochemistry and biological properties of Aristotelia chilensis a Chilean blackberry: A review. Phytochem. Rev. 2017, 16, 1081–1094. [Google Scholar] [CrossRef]
- Insunza, V.; Aballay, E.; Macaya, J. In vitro nematicial activity of aqueous plant extracts on chilean populations of xiphinema americanum sense lato. Nematropica 2001, 31, 47–54. [Google Scholar]
- Essien, S.O.; Young, B.; Baroutian, S. Recent advances in subcritical water and supercritical carbon dioxide extraction of bioactive compounds from plant materials. Trends Food Sci. Technol. 2020, 97, 156–169. [Google Scholar] [CrossRef]
- Flórez, N.; Conde, E.; Domínguez, H. Microwave assisted water extraction of plant compounds. J. Chem. Technol. Biotechnol. 2014, 90, 590–607. [Google Scholar] [CrossRef]
- Samadian, H.; Maleki, H.; Allahyari, Z.; Jaymand, M. Natural polymers-based light-induced hydrogels: Promising biomaterials for biomedical applications. Co-Ord. Chem. Rev. 2020, 420, 213432. [Google Scholar] [CrossRef]
- Anamica; Pande, P.P. Polymer Hydrogels and Their Applications. Int. J. Mater. Sci. 2017, 12, 11–14. [Google Scholar]
- Rodríguez-Seoane, P.; Domínguez, H.; Torres, M.D. Mechanical Characterization of Biopolymer-Based Hydrogels Enriched with Paulownia Extracts Recovered Using a Green Technique. Appl. Sci. 2020, 10, 8439. [Google Scholar] [CrossRef]
- Rivera-Tovar, P.R.; Torres, M.D.; Camilo, C.; Mariotti-Celis, M.S.; Domínguez, H.; Pérez-Correa, J.R. Multi-response optimal hot pressurized liquid recovery of extractable polyphenols from leaves of maqui (Aristotelia chilensis [Mol.] Stuntz). Food Chem. 2021, 357, 129729. [Google Scholar] [CrossRef] [PubMed]
- Damascos, M.A.; Arribere, M. Mineral content from Aristotelia chilensis (Molina) Stuntz leaves used in phytotherapy. Rev. Cuba. Plantas Med. 2009, 14. Available online: http://scielo.sld.cu/scielo.php?script=sci_arttext&pid=S1028-47962009000400003&lng=es (accessed on 1 August 2021).
- Cespedes, C.; Jakupovic, J.; Silva, M.; Watson, W. Indole alkaloids from Aristotelia chilensis. Phytochemistry 1990, 29, 1354–1356. [Google Scholar] [CrossRef]
- He, K.; Valcic, S.; Timmermann, B.N.; Montenegro, G.; Lkaloids, I.N.A.; Ristotelia, F.A.; Ol, C.M. Indole Alkaloids from Aristotelia chilensis (Mol.) Stuntz. Int. J. Pharmacogn. 1997, 35, 215–217. [Google Scholar] [CrossRef]
- Fredes, C.; Paz, R. Maqui (Aristotela chilensis (Mol.) Stuntz). In Fruit and Vegetable Phytochemicals: Chemistry and Human Health, 2nd ed.; Wiley-Blackwell: Hoboken, NJ, USA, 2018; Volume II, pp. 1127–1136. [Google Scholar]
- Muñoz, O.; Christen, P.; Cretton, S.; Backhouse, N.; Torres, V.; Correa, O.; Costa, E.; Miranda, H.; Delporte, C. Chemical study and anti-inflamatory, analgesic and antioxidant activities of the leaves of Aristotelia chilensis (Mol.) Stuntz, Elaeocarpaceae. J. Pharm. Pharmacol. 2011, 63, 849–859. [Google Scholar] [CrossRef] [PubMed]
- Bacanli, M.; Bas, A.A.; Basaran, N. A Triterpenoid Commonly Found in Human Diet: Ursolic Acid. In Polyphenols: Prevention and Treatment of Human Disease, 2nd ed.; Watson, R., Preedy, V., Zibadi, S., Eds.; Academic Press: London, UK, 2018; pp. 31–36. [Google Scholar]
- Vázquez-Espinosa, M.; Espada-Bellido, E.; de Peredo, A.V.G.; Ferreiro-González, M.; Carrera, C.; Palma, M.; Barroso, C.G.; Barbero, G.F. Optimization of Microwave-Assisted Extraction for the Recovery of Bioactive Compounds from the Chilean Superfruit (Aristotelia chilensis (Mol.) Stuntz). Agronomy 2018, 8, 240. [Google Scholar] [CrossRef] [Green Version]
- Rubilar, M.; Jara, C.; Poo, Y.; Acevedo, F.; Gutiérrez, C.; Sineiro, J.; Shene, C. Extracts of Maqui (Aristotelia chilensis) and Murta (Ugni molinae Turcz): Sources of Antioxidant Compounds and α-Glucosidase/α-Amylase Inhibitors. J. Agric. Food Chem. 2011, 59, 1630–1637. [Google Scholar] [CrossRef]
- Devi, R.A.; Arumugam, S.; Thenmozhi, K.; Veena, B. Phytochemical and in vitro antioxidant of an endemic medicinal plant species, Elaeocarpus munronii (WT.) Mast and Elaeocarpus tuberculatus Roxb. (Elaeocarpaceae). J. Pharmacogn. Phytochem. 2018, 7, 159–164. [Google Scholar]
- Kumar, T.S.; Shanmugam, S.; Palvannan, T.; Kumar, V.B. Evaluation of Antioxidant Properties of Elaeocarpus ganitrus Roxb. Leaves. Iran. J. Pharm. Res. 2008, 7, 211–215. [Google Scholar]
- Huaman-Castilla, N.L.; Martínez-Cifuentes, M.; Camilo, C.; Pedreschi, F.; Mariotti-Celis, M.; Pérez-Correa, J.R. The Impact of Temperature and Ethanol Concentration on the Global Recovery of Specific Polyphenols in an Integrated HPLE / RP Process on Carménère Pomace Extracts. Molecules 2019, 24, 3145. [Google Scholar] [CrossRef] [Green Version]
- Mariotti-Celis, M.S.; Martínez-Cifuentes, M.; Huamán-Castilla, N.; Pedreschi, F.; Iglesias-Rebolledo, N.; Pérez-Correa, J.R. Impact of an integrated process of hot pressurised liquid extraction–macroporous resin purification over the polyphenols, hydroxymethylfurfural and reducing sugars content of Vitis vinifera ‘ Carménère ’ pomace extracts. Int. J. Food Sci. Technol. 2018, 53, 1072–1078. [Google Scholar] [CrossRef]
- Sanz, V.; Flórez-Fernández, N.; Domínguez, H.; Torres, D. Clean technologies applied to the recovery of bioactive extracts from Camellia sinensis leaves agricultural wastes. Food Bioprod. Process. 2020, 122, 214–221. [Google Scholar] [CrossRef]
- Liu, H.; Jiang, N.; Liu, L.; Sheng, X.; Shi, A.; Hu, H.; Yang, Y.; Wang, Q. Extraction, Purification and Primary Characterization of Polysaccharides from Defatted Peanut (Arachis hypogaea) Cakes. Molecules 2016, 21, 716. [Google Scholar] [CrossRef] [Green Version]
- Nooeaid, P.; Chuysinuan, P.; Techasakul, S. Alginate/gelatine hydrogels: Characterisation and application of antioxidant release. Green Mater. 2017, 5, 153–164. [Google Scholar] [CrossRef]
- Overend, R. Fractionation of Lignocellulosics by Steam-Aqueous Pretreatments (and Discussion). Philos. Trans. R. Soc. Lond. 1987, 321, 523–536. [Google Scholar]
- Singleton, V.; Rossi, J.A. Colorimetry of Total Phenolics with Phosphomolybdic-Phosphotungstic Acid Reagents. Am. J. Enol. Vitic. 1965, 16, 144–158. [Google Scholar]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant activity applying an improved ABTS radical. Free Radic. Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef]
- Larotonda, F.D.S.; Torres, M.D.; Gonçalves, M.P.; Sereno, A.M.; Hilliou, L. Hybrid carrageenan-based formulations for edible film preparation: Benchmarking with kappa carrageenan. J. Appl. Polym. Sci. 2016, 133, 42263. [Google Scholar] [CrossRef]

| Moisture (%) | 9.68 ± 0.12 | |
| Ash (%) | 6.43 ± 0.18 | |
| Proteins (%) | 15.15 ± 0.18 1 | |
| Minerals (mg/kg) | Potassium | 10,500 ± 380 1 |
| Calcium | 21,230 ± 300 1 | |
| Magnesium | 1980 ± 10 1 | |
| Sodium | 20.14 ± 0.99 1 | |
| Iron | 237.9 ± 4.5 1 | |
| Zinc | 12.33 ± 0.14 1 | |
| Copper | <7.0 1 | |
| Cadmium | <5.0 1 | |
| Lead | <10.0 1 | |
| Oligomeric carbohydrates and other groups (%) | Total | 37.64 |
| Glucose | 15.68 ± 0.02 | |
| Xylose | 10.39 ± 0.00 | |
| Rhamnose | 2.31 ± 0.01 | |
| Arabinose | 2.44 ± 0.06 | |
| Fucose | 0.22 ± 0.04 | |
| Ribose | 0.79 ± 0.13 | |
| Formic acid | 3.94 ± 0.27 | |
| Acetic acid | 1.89 ± 0.20 |
| Temperature (°C) | Microwave Heating | Conventional Heating |
|---|---|---|
| 140 | 1.36 ± 0.06 a | 1.52 ± 0.08 a |
| 160 | 1.90 ± 0.02 b | 2.22 ± 0.05 a |
| 180 | 2.05 ± 0.01 b | 2.92 ± 0.06 a |
| 200 | 2.56 ± 0.03 b | 3.48 ± 0.09 a |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bianchi, A.; R. Rivera-Tovar, P.; Sanz, V.; Ferreira-Anta, T.; Torres, M.D.; Pérez-Correa, J.R.; Domínguez, H. Pressurized Hot Water Extraction and Bio-Hydrogels Formulation with Aristotelia chilensis [Mol.] Stuntz Leaves. Molecules 2021, 26, 6402. https://doi.org/10.3390/molecules26216402
Bianchi A, R. Rivera-Tovar P, Sanz V, Ferreira-Anta T, Torres MD, Pérez-Correa JR, Domínguez H. Pressurized Hot Water Extraction and Bio-Hydrogels Formulation with Aristotelia chilensis [Mol.] Stuntz Leaves. Molecules. 2021; 26(21):6402. https://doi.org/10.3390/molecules26216402
Chicago/Turabian StyleBianchi, Audrey, Pamela R. Rivera-Tovar, Vanesa Sanz, Tania Ferreira-Anta, María Dolores Torres, José Ricardo Pérez-Correa, and Herminia Domínguez. 2021. "Pressurized Hot Water Extraction and Bio-Hydrogels Formulation with Aristotelia chilensis [Mol.] Stuntz Leaves" Molecules 26, no. 21: 6402. https://doi.org/10.3390/molecules26216402
APA StyleBianchi, A., R. Rivera-Tovar, P., Sanz, V., Ferreira-Anta, T., Torres, M. D., Pérez-Correa, J. R., & Domínguez, H. (2021). Pressurized Hot Water Extraction and Bio-Hydrogels Formulation with Aristotelia chilensis [Mol.] Stuntz Leaves. Molecules, 26(21), 6402. https://doi.org/10.3390/molecules26216402







