Antimicrobial Activity of MgB2 Powders Produced via Reactive Liquid Infiltration Method
Abstract
1. Introduction
2. Experimental
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Nagamatsu, J.; Nakagawa, N.; Muranaka, T.; Zenitani, Y.; Akimitsu, J. Superconductivity at 39 K in magnesium diboride. Nature 2001, 410, 63–64. [Google Scholar] [CrossRef]
- Kotegawa, H.; Ishida, K.; Kitaoka, Y.; Muranaka, T.; Nakagawa, N.; Takagiwa, H.; Akimitsu, J. Evidence for high-frequency phonon mediated S-wave superconductivity: B-11 NMR study of Al-doped MgB2. Phys. Rev. B 2002, 66, 064516. [Google Scholar] [CrossRef]
- Kuz′menko, A.B.; Mena, F.P.; Molegraaf, H.J.A.; van der Marel, D.; Gorshunov, B.; Dressel, M.; Mazin, I.I.; Kortus, J.; Dolgov, O.V.; Muranaka, T.; et al. Manifestation of multiband optical properties of MgB2. Solid State Commun. 2002, 121, 479–484. [Google Scholar] [CrossRef]
- Szabo, P.; Samuely, P.; Kacmarcik, J.; Klein, T.; Marcus, J.; Fruchart, D.; Miraglia, S.; Marcenat, C.; Jansen, A.G.M. Evidence for two superconducting energy gaps in MgB2 by point-contact spectroscopy. Phys. Rev. Lett. 2001, 87, 137005. [Google Scholar] [CrossRef]
- Kalsi, S.S. Superconducting Wind Turbine Generator Employing MgB2 Windings Both on Rotor and Stator. IEEE Trans. Appl. Supercond. 2013, 24, 5201907. [Google Scholar]
- Arpaia, P.; Ballarino, A.; Giunchi, G.; Montenero, G. MgB2 cylindrical superconducting shielding for cryogenic measurement applications: A case study on DC current transformers. J. Instrum. 2014, 9, P04020. [Google Scholar] [CrossRef]
- Gozzelino, L.; Gerbaldo, R.; Ghigo, G.; Laviano, F.; Truccato, M. Comparison of the Shielding Properties of Superconducting and Superconducting/Ferromagnetic Bi- and Multi-layer Systems. J. Supercond. Nov. Magn. 2017, 30, 749–756. [Google Scholar] [CrossRef]
- Gozzelino, L.; Minetti, B.; Gerbaldo, R.; Ghigo, G.; Laviano, F.; Lopardo, G.; Plapcianu, C.; Agostino, A.; Cagliero, S.; Truccato, M.; et al. Magnetic Characterization of MgB2 Bulk Superconductor for Magnetic Field Mitigation Solutions. J. Supercond. Nov. Magn. 2011, 24, 307–312. [Google Scholar] [CrossRef]
- Konstantopoulou, K.; Ballarino, A.; Gharib, A.; Stimac, A.; Gonzalez, M.G.; Fontenla, A.T.P.; Sugano, M. Electro-mechanical characterization of MgB2 wires for the Superconducting Link Project at CERN. Supercond. Sci. Technol. 2016, 29, 084005. [Google Scholar] [CrossRef]
- Putti, M.; Grasso, G. MgB2, a two-gap superconductor for practical applications. MRS Bull. 2011, 36, 608–613. [Google Scholar] [CrossRef]
- Aldica, G.; Batalu, D.; Popa, S.; Ivan, I.; Nita, P.; Sakka, Y.; Vasylkiv, O.; Miu, L.; Pasuk, I.; Badica, P. Spark plasma sintering of MgB2 in the two-temperature route. Physica C 2012, 477, 43–50. [Google Scholar] [CrossRef]
- Locci, A.M.; Orru, R.; Cao, G.; Sanna, S.; Congiu, F.; Concas, G. Synthesis of bulk MgB2 superconductors by pulsed electric current. Aiche J. 2006, 52, 2618–2626. [Google Scholar] [CrossRef]
- Badica, P.; Agostino, A.; Khan, M.M.R.; Cagliero, S.; Plapcianu, C.; Pastero, L.; Truccato, M.; Hayasaka, Y.; Jakob, G. Bi-2212 and Y123 highly curved single-crystal-like objects: Whiskers, bows and ring-like structures. Supercond. Sci. Technol. 2012, 25, 105003. [Google Scholar] [CrossRef][Green Version]
- Murakami, A.; Iwamoto, A.; Noudem, J.G. Mechanical Properties of Bulk MgB2 Superconductors Processed by Spark Plasma Sintering at Various Temperatures. IEEE Trans. Appl. Supercond. 2018, 28, 8400204. [Google Scholar] [CrossRef]
- Flukiger, R.; Al Hossain, M.S.; Senatore, C.; Buta, F.; Rindfleisch, M. A New Generation of In Situ MgB2 Wires With Improved J(c) and B-irr Values Obtained by Cold Densification (CHPD). IEEE Trans. Appl. Supercond. 2011, 21, 2649–2654. [Google Scholar] [CrossRef]
- Cagliero, S.; Borfecchia, E.; Mino, L.; Calore, L.; Bertolotti, F.; Martinez-Criado, G.; Operti, L.; Agostino, A.; Truccato, M.; Badica, P.; et al. Insight into non-linearly shaped superconducting whiskers via a synchrotron nanoprobe. Supercond. Sci. Technol. 2012, 25, 125002. [Google Scholar] [CrossRef][Green Version]
- Gajda, D.; Morawski, A.; Zaleski, A.; Kurnatowska, M.; Cetner, T.; Gajda, G.; Presz, A.; Rindfleisch, M.; Tomsic, M. The influence of HIP on the homogeneity, J(c), B-irr, T-c and F-p in MgB2 wires. Supercond. Sci. Technol. 2015, 28, 015002. [Google Scholar] [CrossRef]
- Noudem, J.G.; Aburras, M.; Bernstein, P.; Chaud, X.; Muralidhar, M.; Murakami, M. Development in processing of MgB2 cryo-magnet superconductors. J. Appl. Phys. 2014, 116, 163916. [Google Scholar] [CrossRef]
- Sasaki, T.; Naito, T.; Fujishiro, H. Trapped magnetic field of dense MgB2 bulks fabricated under high pressure. Phys. Procedia 2013, 45, 93–96. [Google Scholar] [CrossRef][Green Version]
- Mino, L.; Bonino, V.; Agostino, A.; Prestipino, C.; Borfecchia, E.; Lamberti, C.; Operti, L.; Fretto, M.; De Leo, N.; Truccato, M. Maskless X-Ray Writing of Electrical Devices on a Superconducting Oxide with Nanometer Resolution and Online Process Monitoring. Sci. Rep. 2017, 7, 9066. [Google Scholar] [CrossRef] [PubMed]
- Susner, M.A.; Bohnenstiehl, S.D.; Dregia, S.A.; Sumption, M.D.; Yang, Y.; Donovan, J.J.; Collings, E.W. Homogeneous carbon doping of magnesium diboride by high-temperature, high-pressure synthesis. Appl. Phys. Lett. 2014, 104, 162603. [Google Scholar] [CrossRef]
- Plapcianu, C.; Agostino, A.; Badica, P.; Aldica, G.V.; Bonometti, E.; Ieluzzi, G.; Popa, S.; Truccato, M.; Cagliero, S.; Sakka, Y.; et al. Microwave Synthesis of Fullerene-Doped MgB2. Ind. Eng. Chem. Res. 2012, 51, 11005–11010. [Google Scholar] [CrossRef]
- Mino, L.; Borfecchia, E.; Agostino, A.; Lamberti, C.; Truccato, M. Oxygen doping tuning in superconducting oxides by thermal annealing and hard X-ray irradiation. J. Electron. Spectrosc. Relat. Phenom. 2017, 220, 69–75. [Google Scholar] [CrossRef]
- Bhagurkar, A.G.; Yamamoto, A.; Babu, N.H.; Durrell, J.H.; Dennis, A.R.; Cardwell, D.A. Synthesis of dense bulk MgB2 by an infiltration and growth process. Supercond. Sci. Technol. 2015, 28, 015012. [Google Scholar] [CrossRef]
- Giunchi, G. High density MgB2 obtained by reactive liquid Mg infiltration. Int. J. Mod. Phys. B 2003, 17, 453–460. [Google Scholar] [CrossRef]
- Giunchi, G.; Ripamonti, G.; Cavallin, T.; Bassani, E. The reactive liquid Mg infiltration process to produce large superconducting bulk MgB2 manufacts. Cryogenics 2006, 46, 237–242. [Google Scholar] [CrossRef]
- Batalu, D.; Stanciuc, A.M.; Moldovan, L.; Aldica, G.; Badica, P. Evaluation of pristine and Eu2O3-added MgB2 ceramics for medical applications: Hardness, corrosion resistance, cytotoxicity and antibacterial activity. Mater. Sci. Eng. C Mater. Biol. Appl. 2014, 42, 350–361. [Google Scholar] [CrossRef]
- Fan, M.J.; Wen, Y.Y.; Ye, D.; Jin, Z.K.; Zhao, P.H.; Chen, D.Y.; Lu, X.F.; He, Q.J. Acid-Responsive H-2-Releasing 2D MgB2 Nanosheet for Therapeutic Synergy and Side Effect Attenuation of Gastric Cancer Chemotherapy. Adv. Healthc. Mater. 2019, 8, 1900157. [Google Scholar] [CrossRef]
- Abhinandan, R.; Pranav Adithya, S.; Saleth Sidharthan, D.; Balagangadharan, K.; Selvamurugan, N. Synthesis and characterization of magnesium diboride nanosheets in alginate/polyvinyl alcohol scaffolds for bone tissue engineering. Colloids Surf. B Biointerfaces 2021, 203, 111771. [Google Scholar] [CrossRef]
- El-Sayyad, G.S.; Mosallam, F.M.; El-Batal, A.I. One-pot green synthesis of magnesium oxide nanoparticles using Penicillium chrysogenum melanin pigment and gamma rays with antimicrobial activity against multidrug-resistant microbes. Adv. Powder Technol. 2018, 29, 2616–2625. [Google Scholar] [CrossRef]
- Luque-Agudo, V.; Fernandez-Calderon, M.C.; Pacha-Olivenza, M.A.; Perez-Giraldo, C.; Gallardo-Moreno, A.M.; Gonzalez-Martin, M.L. The role of magnesium in biomaterials related infections. Colloids Surf. B Biointerfaces 2020, 191, 110996. [Google Scholar] [CrossRef]
- Nostro, A.; Cellini, L.; Di Giulio, M.; D′Arrigo, M.; Marino, A.; Blanco, A.R.; Favaloro, A.; Cutroneo, G.; Bisignano, G. Effect of alkaline pH on staphylococcal biofilm formation. Apmis 2012, 120, 733–742. [Google Scholar] [CrossRef] [PubMed]
- Robinson, D.A.; Griffith, R.W.; Shechtman, D.; Evans, R.B.; Conzemius, M.G. In vitro antibacterial properties of magnesium metal against Escherichia coli, Pseudomonas aeruginosa and Staphylococcus aureus. Acta Biomater. 2010, 6, 1869–1877. [Google Scholar] [CrossRef] [PubMed]
- Sawai, J.; Kojima, H.; Igarashi, H.; Hashimoto, A.; Shoji, S.; Takehara, A.; Sawaki, T.; Kokugan, T.; Shimizu, M. Escherichia coli damage by ceramic powder slurries. J. Chem. Eng. Jpn. 1997, 30, 1034–1039. [Google Scholar] [CrossRef]
- Davies, D. Understanding biofilm resistance to antibacterial agents. Nat. Rev. Drug Discov. 2003, 2, 114–122. [Google Scholar] [CrossRef]
- Yilmaz, M.T. Minimum inhibitory and minimum bactericidal concentrations of boron compounds against several bacterial strains. Turk. J. Med. Sci. 2012, 42, 1423–1429. [Google Scholar]
- Badica, P.; Batalu, N.D.; Chifiriuc, M.C.; Burdusel, M.; Grigoroscuta, M.A.; Aldica, G.; Pasuk, I.; Kuncser, A.; Enculescu, M.; Popa, M.; et al. MgB2 powders and bioevaluation of their interaction with planktonic microbes, biofilms, and tumor cells. J. Mater. Res. Technol. 2021, 12, 2168–2184. [Google Scholar] [CrossRef]
- Badica, P.; Batalu, N.D.; Burdusel, M.; Grigoroscuta, M.A.; Aldica, G.; Enculescu, M.; Gradisteanu Pircalabioru, G.; Popa, M.; Marutescu, L.G.; Dumitriu, B.G.; et al. Antibacterial composite coatings of MgB2 powders embedded in PVP matrix. Sci. Rep. 2021, 11, 9591. [Google Scholar] [CrossRef]
- Gheorghe, I.; Avram, I.; Corbu, V.M.; Marutescu, L.; Popa, M.; Balotescu, I.; Blajan, I.; Mateescu, V.; Zaharia, D.; Dumbrava, A.S.; et al. In Vitro Evaluation of MgB2 Powders as Novel Tools to Fight Fungal Biodeterioration of Heritage Buildings and Objects. Front. Mater. 2021, 7, 601059. [Google Scholar] [CrossRef]
- Lutterotti, L.; Matthies, S.; Wenk, H.R. MAUD (Material Analysis Using Diffraction): A user friendly Java program for Rietveld Texture Analysis and more. In Proceedings of the Twelfth International Conference on Textures of Materials (ICOTOM-12), Montreal, QC, Canada, 9–13 August 1999; p. 1599. [Google Scholar]
- Lutterotti, L. Total pattern fitting for the combined size-strain-stress-texture determination in thin film diffraction. Nucl. Instrum. Methods Phys. Res. Sect. B Beam Interact. Mater. At. 2010, 268, 334–340. [Google Scholar] [CrossRef]
- Albisetti, A.F.; Saglietti, L.; Perini, E.; Schiavone, C.; Ripamonti, G.; Giunchi, G. The Mg2B25 formation and its role in the preparation of bulk MgB2 superconductors. Solid State Sci. 2012, 14, 1632–1635. [Google Scholar] [CrossRef]
- Giunchi, G.; Malpezzi, L.; Masciocchi, N. A new crystalline phase of the boron-rich metal-boride family: The Mg2B25 species. Solid State Sci. 2006, 8, 1202–1208. [Google Scholar] [CrossRef]
- Chen, W.; Liu, W.; Chen, C.; Wang, R.; Feng, Q. Single-crystal MgB2 hexagonal microprisms via hybrid physical-chemical vapor deposition. CrystEngComm 2011, 13, 3959–3961. [Google Scholar] [CrossRef]
- Song, X. Atomic structure and chemistry of dense nano-precipitates in MgB2 ceramic. Ceram. Int. 2013, 39, 4299–4305. [Google Scholar] [CrossRef]
- Yang, Y.; Sumption, M.D.; Rindfleisch, M.; Tomsic, M.; Collings, E.W. Enhanced higher temperature irreversibility field and critical current density in MgB2 wires with Dy2O3 additions. Supercond. Sci. Technol. 2021, 34, 025010. [Google Scholar] [CrossRef] [PubMed]
- Sinha, B.B.; Kadam, M.B.; Mudgel, M.; Awana, V.P.S.; Kishan, H.; Pawar, S.H. Synthesis and characterization of excess magnesium MgB2 superconductor under inert carbon environment. Phys. C 2010, 470, 25–30. [Google Scholar] [CrossRef]
- Zeng, R.; Lu, L.; Li, W.X.; Wang, J.L.; Shi, D.Q.; Horvat, J.; Dou, S.X.; Bhatia, M.; Sumption, M.; Collings, E.W.; et al. Excess Mg addition MgB2/Fe wires with enhanced critical current density. J. Appl. Phys. 2008, 103, 083911. [Google Scholar] [CrossRef]
- Choel, M.; Deboudt, K.; Osan, J.; Flament, P.; Van Grieken, R. Quantitative determination of low-Z elements in single atmospheric particles on boron substrates by automated scanning electron microscopy-Energy-dispersive X-ray spectrometry. Anal. Chem. 2005, 77, 5686–5692. [Google Scholar] [CrossRef] [PubMed]
- Llovet, X.; Powell, C.J.; Salvat, F.; Jablonski, A. Cross Sections for Inner-Shell Ionization by Electron Impact. J. Phys. Chem. Ref. Data 2014, 43, 013102. [Google Scholar] [CrossRef]
- Elam, W.T.; Ravel, B.D.; Sieber, J.R. A new atomic database for X-ray spectroscopic calculations. Radiat. Phys. Chem. 2002, 63, 121–128. [Google Scholar] [CrossRef]
- Arvapalli, S.S.; Muralidhar, M.; Murakami, M. High-Performance Bulk MgB2 Superconductor Using Amorphous Nano-boron. J. Supercond. Nov. Magn. 2019, 32, 1891–1895. [Google Scholar] [CrossRef]
- Guo, Y.; Zhang, W.; Yang, D.; Yao, R.L. Decomposition and Oxidation of Magnesium Diboride. J. Am. Ceram. Soc. 2012, 95, 754–759. [Google Scholar] [CrossRef]
- Rosenband, V.; Gany, A. Thermal explosion synthesis of a magnesium diboride powder. Combust. Explos. 2014, 50, 653–657. [Google Scholar] [CrossRef]
- Liang, D.L.; Xiao, R.; Li, H.P.; Liu, J.Z. Heterogeneous decomposition and oxidation during combustion of magnesium diboride particles. Acta Astronaut. 2018, 153, 159–165. [Google Scholar] [CrossRef]
- Hosseini, S.G.; Sheikhpour, A.; Keshavarz, M.H.; Tavangar, S. The effect of metal oxide particle size on the thermal behavior and ignition kinetic of Mg-CuO thermite mixture. Thermochim. Acta 2016, 626, 1–8. [Google Scholar] [CrossRef]
- Song, M.R.; Chen, M.; Zhang, Z.J. Preparation and characterization of Mg nanoparticles. Mater. Charact. 2008, 59, 514–518. [Google Scholar] [CrossRef]
- Gunda, H.; Ghoroi, C.; Jasuja, K. Layered magnesium diboride and its derivatives as potential catalytic and energetic additives for tuning the exothermicity of ammonium perchlorate. Thermochim. Acta 2020, 690, 178674. [Google Scholar] [CrossRef]
- Gunda, H.; Klebanoff, L.E.; Sharma, P.A.; Varma, A.K.; Dolia, V.; Jasuja, K.; Stavila, V. Progress, Challenges, and Opportunities in the Synthesis, Characterization, and Application of Metal-Boride-Derived Two-Dimensional Nanostructures. ACS Mater. Lett. 2021, 3, 535–556. [Google Scholar] [CrossRef]
- Ashour, A.H.; El-Batal, A.I.; Maksoud, M.; El-Sayyad, G.S.; Labib, S.; Abdeltwab, E.; El-Okr, M.M. Antimicrobial activity of metal-substituted cobalt ferrite nanoparticles synthesized by sol-gel technique. Particuology 2018, 40, 141–151. [Google Scholar] [CrossRef]
- Shao, Y.; Zeng, R.C.; Li, S.Q.; Cui, L.Y.; Zou, Y.H.; Guan, S.K.; Zheng, Y.F. Advance in Antibacterial Magnesium Alloys and Surface Coatings on Magnesium Alloys: A Review. Acta Metall. Sin. 2020, 33, 615–629. [Google Scholar] [CrossRef]
- Hoseinzadeh, E.; Makhdoumi, P.; Taha, P.; Hossini, H.; Stelling, J.; Kamal, M.A.; Ashraf, G.M. A Review on Nano-Antimicrobials: Metal Nanoparticles, Methods and Mechanisms. Curr. Drug Metab. 2017, 18, 120–128. [Google Scholar] [CrossRef] [PubMed]
- Lora-Tamayo, J.; Murillo, O.; Ariza, J. Clinical Use of Colistin in Biofilm-Associated Infections. In Polymyxin Antibiotics: From Laboratory Bench to Bedside; Li, J., Nation, R.L., Kaye, K.S., Eds.; Springer International Publishing AG: Cham, Switzerland, 2019; Volume 1145, pp. 181–195. [Google Scholar]
- Mandell, J.B.; Orr, S.; Koch, J.; Nourie, B.; Ma, D.Z.; Bonar, D.D.; Shah, N.; Urish, K.L. Large variations in clinical antibiotic activity against Staphylococcus aureus biofilms of periprosthetic joint infection isolates. J. Orthop. Res. 2019, 37, 1604–1609. [Google Scholar] [CrossRef] [PubMed]
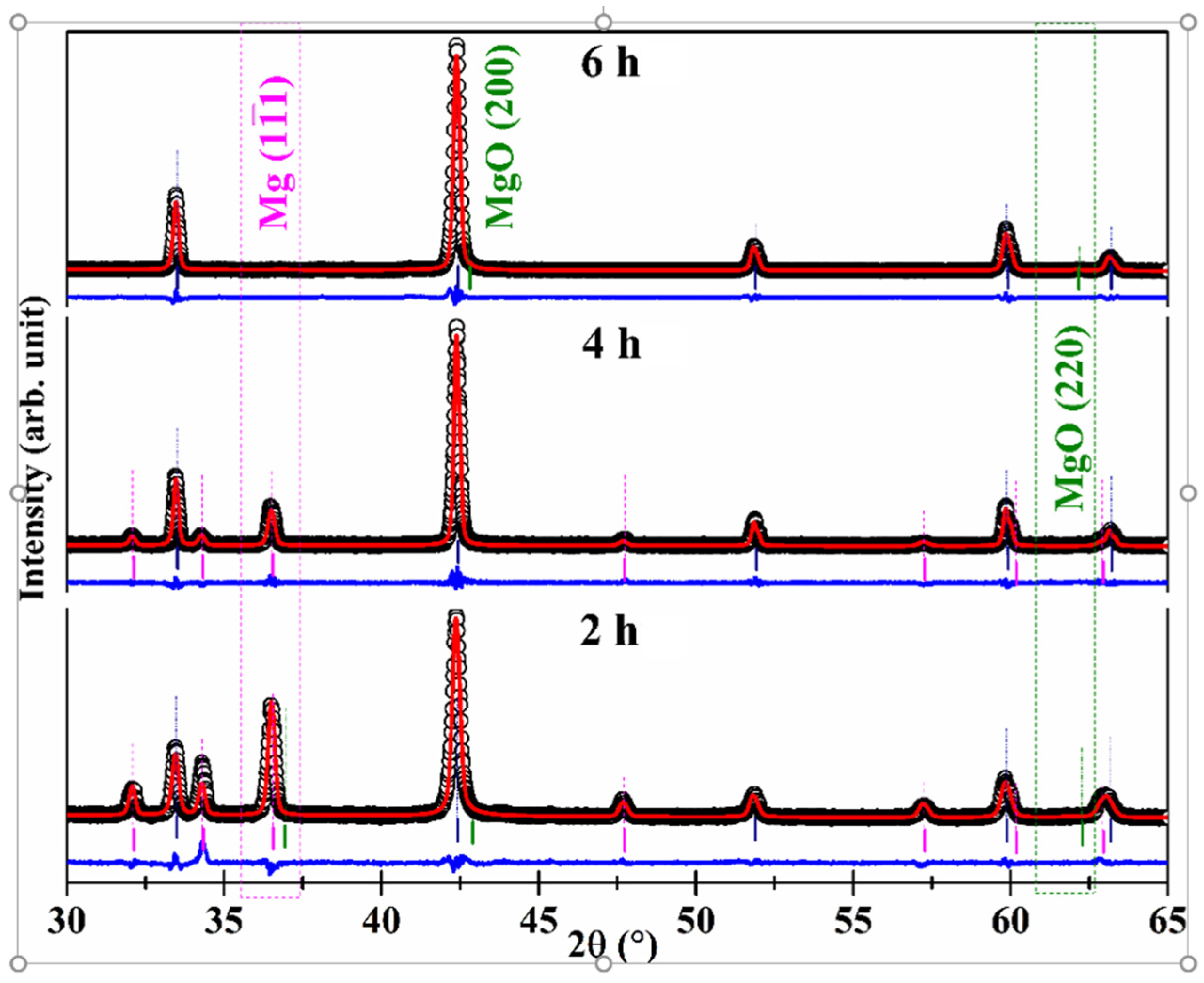
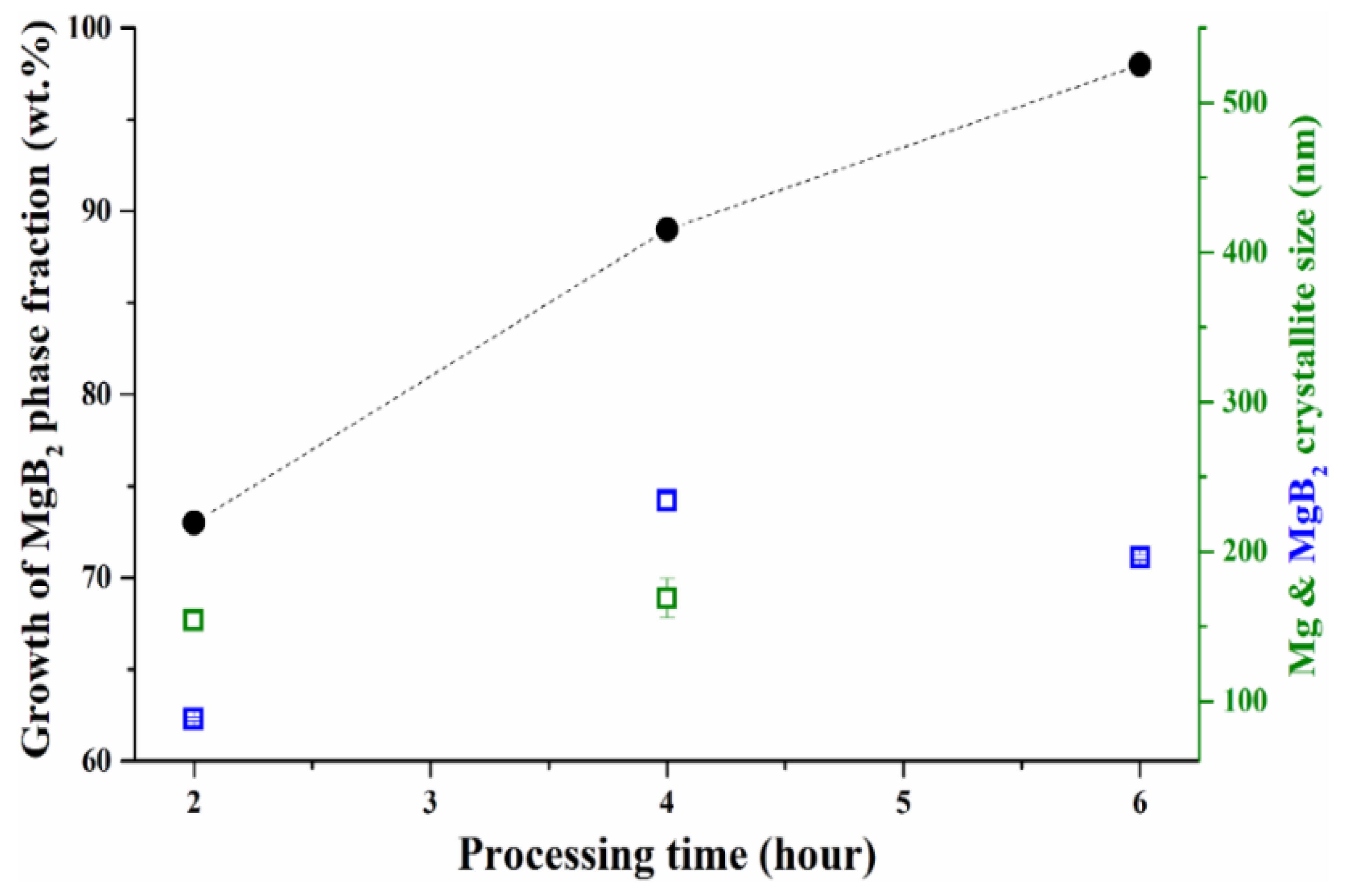

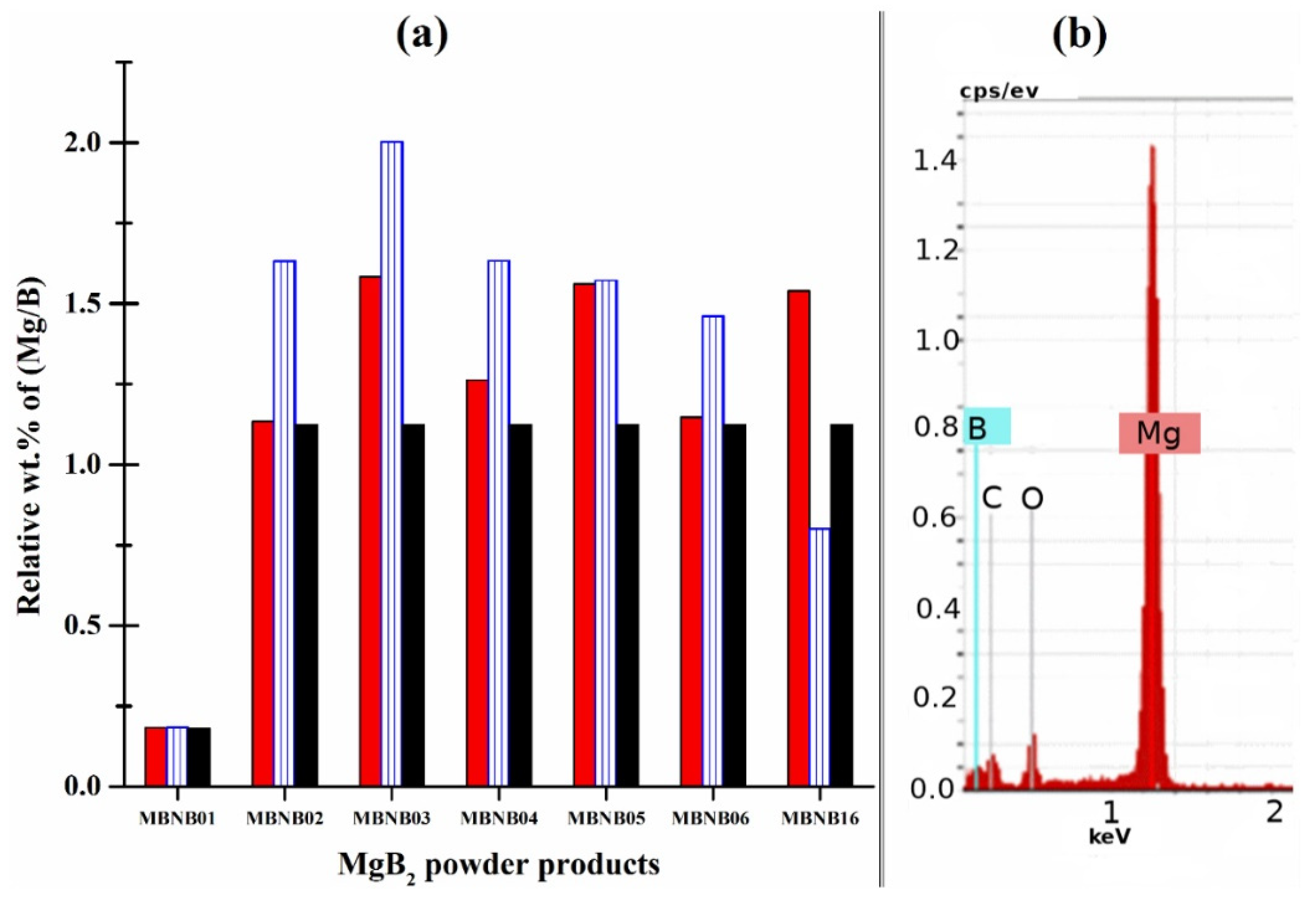
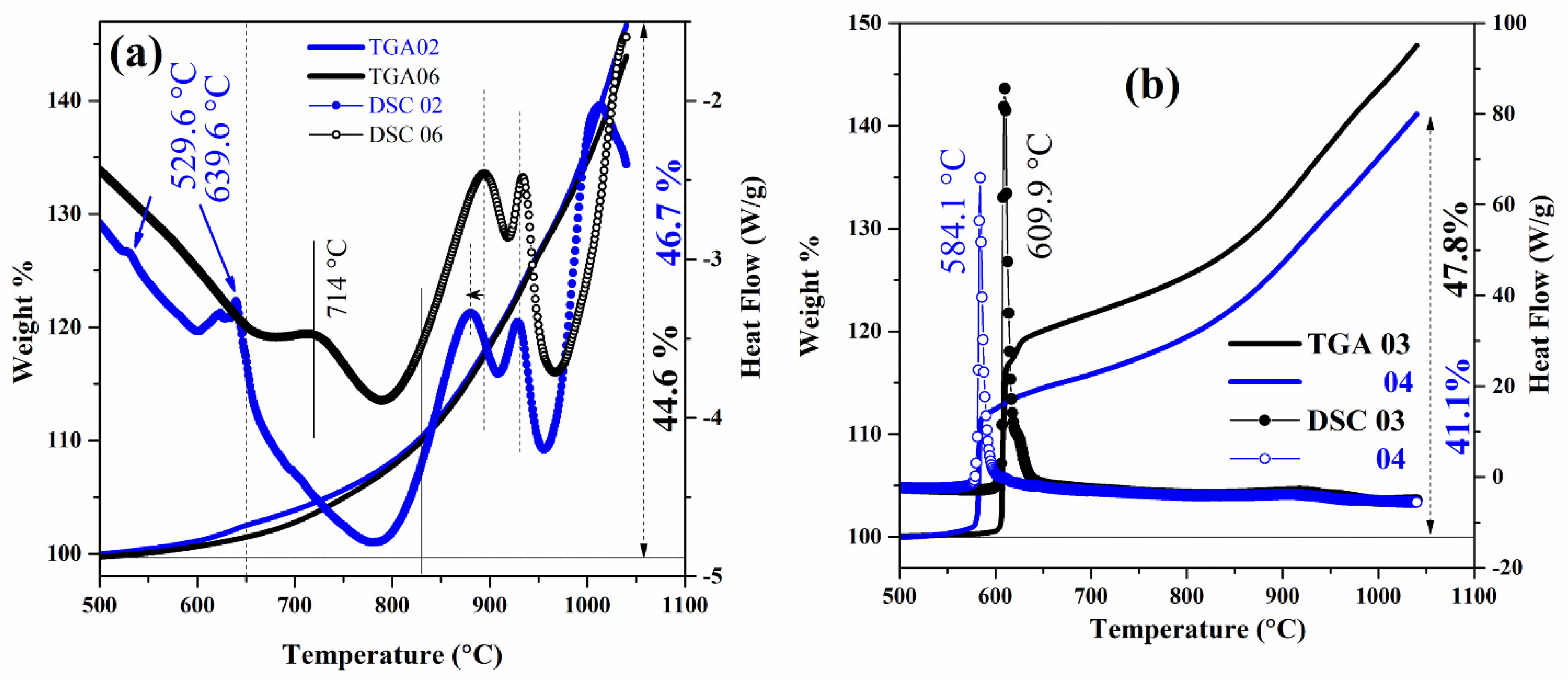
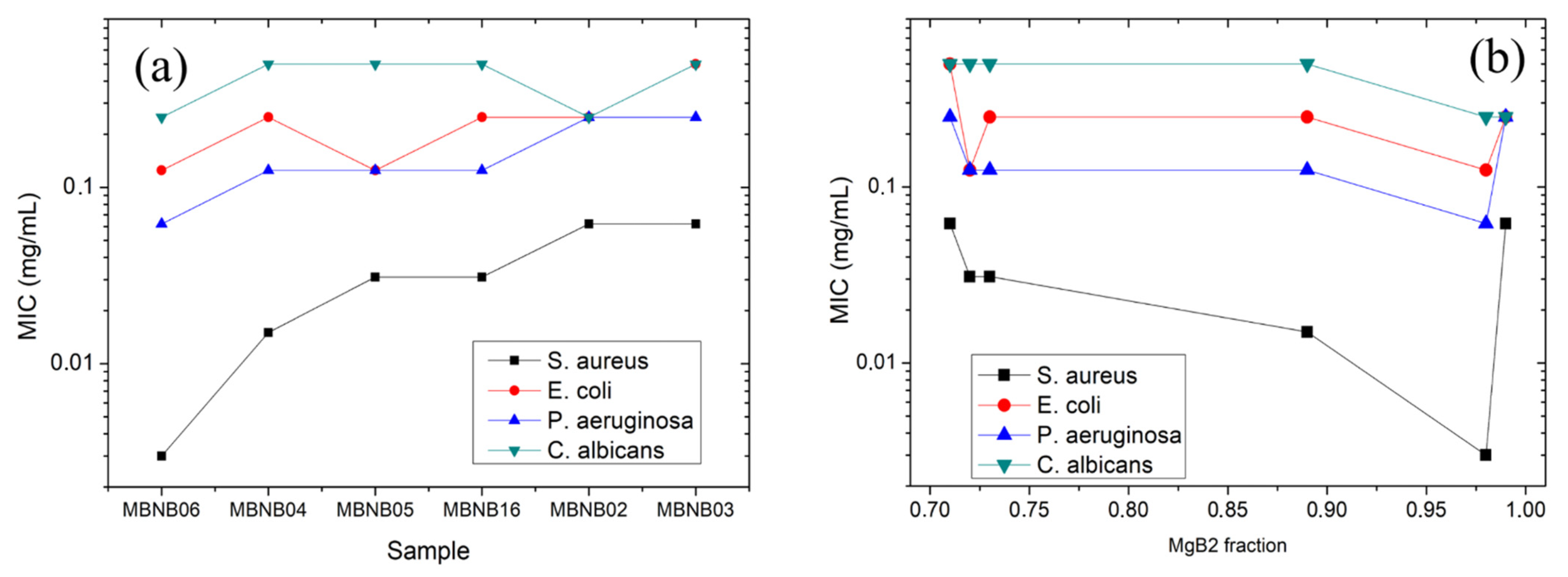
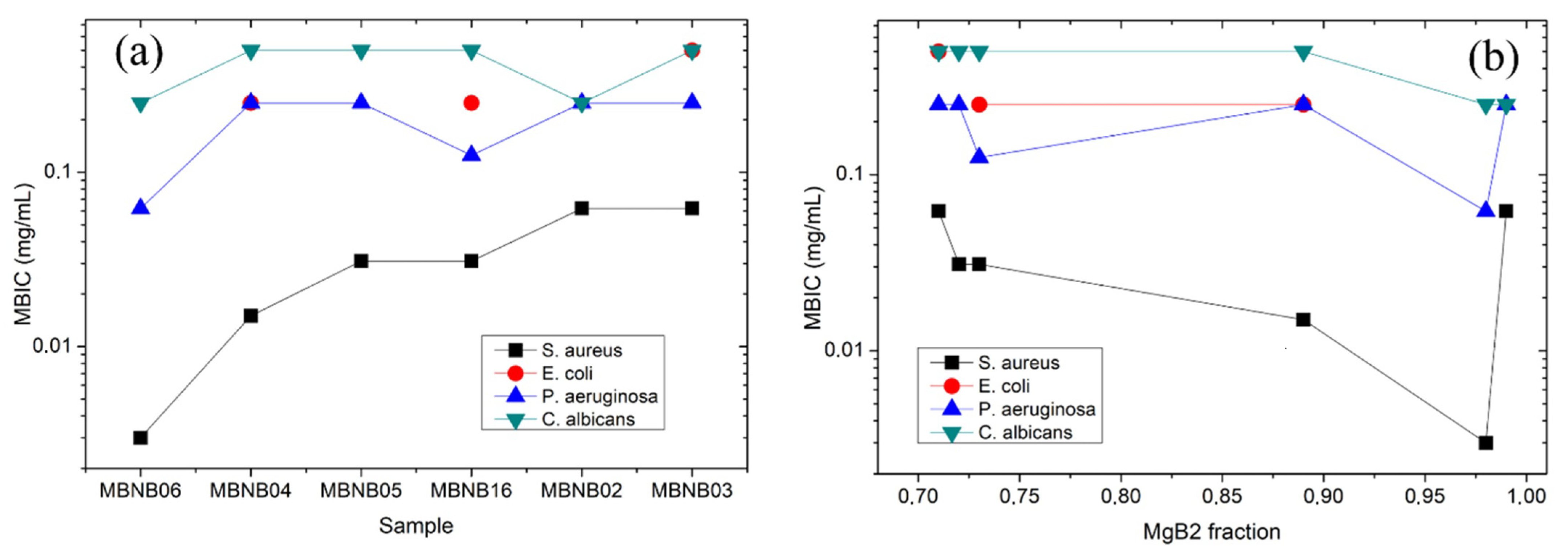
| MBNB01 | MBNB02 | MBNB03 | MBNB04 | MBNB05 | MBNB06 | MBNB16 | |
|---|---|---|---|---|---|---|---|
| Mg:B | 1.4:2 | 1.4:2 | 1.8:2 | 1.4:2 | 1.4:2 | 1.4:2 | 1.4:2 |
| T (°C) | 670 | 800 | 800 | 800 | 760 | 800 | 800 |
| td (h) | 2 | 2 | 4 | 4 | 6 | 6 | 2 |
| Mg2B25 | 0.97 | - | - | - | - | - | - |
| MgB2 | 0.02 | 0.99 | 0.71 | 0.89 | 0.72 | 0.98 | 0.73 |
| Mg | 0.01 | 0.01 | 0.29 | 0.11 | 0.28 | - | 0.22 |
| MgO | - | - | - | - | - | 0.02 | 0.05 |
| Rwp | 6.48 | 9.45 | 9.58 | 19.18 | 17.51 | 9.13 | 9.97 |
| Rexp | 1.79 | 5.67 | 5.72 | 15.81 | 13.98 | 5.44 | 6.75 |
| GoF χ2 | 3.6 | 1.66 | 1.67 | 1.21 | 1.25 | 1.68 | 1.47 |
| MBNB01 | MBNB02 | MBNB03 | MBNB04 | MBNB05 | MBNB06 | MBNB16 | DMSO | |
|---|---|---|---|---|---|---|---|---|
| S. aureus | >0.5 | 0.062 ± 0.002 | 0.062 ± 0.005 | 0.015 ± 0.001 | 0.031 ± 0.001 | 0.003 ± 0.001 | 0.031 ± 0.001 | >50% |
| E. faecalis | >0.5 | >0.5 | 0.50 ± 0.02 | 0.5 ± 0.1 | 0.50 ± 0.05 | >0.5 | >0.5 | >50% |
| E. coli | >0.5 | 0.25 ± 0.05 | 0.50 ± 0.01 | 0.25 ± 0.04 | 0.125 ± 0.005 | 0.125 ± 0.003 | 0.25 ± 0.05 | 50% |
| Ps. aeruginosa | >0.5 | 0.25 ± 0.02 | 0.25 ± 0.05 | 0.125 ± 0.005 | 0.13 ± 0.05 | 0.062 ± 0.002 | 0.125 ± 0.005 | 50% |
| C. albicans | >0.5 | 0.25 ± 0.02 | 0.50 ± 0.03 | 0.50 ± 0.01 | 0.50 ± 0.01 | 0.25 ± 0.02 | 0.50 ± 0.01 | 50% |
| MBNB01 | MBNB02 | MBNB03 | MBNB04 | MBNB05 | MBNB06 | MBNB16 | DMSO | |
|---|---|---|---|---|---|---|---|---|
| S. aureus | 0.5 | 0.50 ± 0.05 | 0.062 ± 0.003 | 0.062 ± 0.005 | 0.015 ± 0.0001 | 0.031 ± 0.002 | 0.003 ± 0.002 | >50% |
| E. faecalis | >0.5 | >0.5 | >0.5 | >0.5 | >0.5 | >0.5 | >0.5 | >50% |
| E. coli | >0.5 | >0.5 | >0.5 | 0.5 ± 0.1 | 0.25 ± 0.05 | >0.5 | >0.5 | 50% |
| Ps. aeruginosa | 0.5 | 0.50 ± 0.03 | 0.25 ± 0.05 | 0.25 ± 0.04 | 0.25 ± 0.10 | 0.25 ± 0.05 | 0.06 ± 0.04 | 25% |
| C. albicans | 0.5 | 0.50 ± 0.05 | 0.25 ± 0.01 | 0.50 ± 0.06 | 0.50 ± 0.06 | 0.5 ± 0.1 | 0.25 ± 0.02 | 50% |
| MBNB01 | MBNB02 | MBNB03 | MBNB04 | MBNB05 | MBNB06 | MBNB16 | |
|---|---|---|---|---|---|---|---|
| MIC | 0.50 ± 0.06 | 0.26 ± 0.15 | 0.36 ± 0.22 | 0.28 ± 0.06 | 0.26 ± 0.10 | 0.19 ± 0.03 | 0.28 ± 0.06 |
| MBIC | 0.5 ± 0.1 | 0.31 ± 0.05 | 0.36 ± 0.15 | 0.30 ± 0.07 | 0.36 ± 0.02 | 0.26 ± 0.05 | 0.28 ± 0.10 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Padhi, S.K.; Baglieri, N.; Bonino, V.; Agostino, A.; Operti, L.; Batalu, N.D.; Chifiriuc, M.C.; Popa, M.; Burdusel, M.; Grigoroscuta, M.A.; et al. Antimicrobial Activity of MgB2 Powders Produced via Reactive Liquid Infiltration Method. Molecules 2021, 26, 4966. https://doi.org/10.3390/molecules26164966
Padhi SK, Baglieri N, Bonino V, Agostino A, Operti L, Batalu ND, Chifiriuc MC, Popa M, Burdusel M, Grigoroscuta MA, et al. Antimicrobial Activity of MgB2 Powders Produced via Reactive Liquid Infiltration Method. Molecules. 2021; 26(16):4966. https://doi.org/10.3390/molecules26164966
Chicago/Turabian StylePadhi, Santanu Kumar, Nicoletta Baglieri, Valentina Bonino, Angelo Agostino, Lorenza Operti, Nicolae Dan Batalu, Mariana Carmen Chifiriuc, Marcela Popa, Mihail Burdusel, Mihai Alexandru Grigoroscuta, and et al. 2021. "Antimicrobial Activity of MgB2 Powders Produced via Reactive Liquid Infiltration Method" Molecules 26, no. 16: 4966. https://doi.org/10.3390/molecules26164966
APA StylePadhi, S. K., Baglieri, N., Bonino, V., Agostino, A., Operti, L., Batalu, N. D., Chifiriuc, M. C., Popa, M., Burdusel, M., Grigoroscuta, M. A., Aldica, G. V., Radu, D., Badica, P., & Truccato, M. (2021). Antimicrobial Activity of MgB2 Powders Produced via Reactive Liquid Infiltration Method. Molecules, 26(16), 4966. https://doi.org/10.3390/molecules26164966









