Gossypitrin, A Naturally Occurring Flavonoid, Attenuates Iron-Induced Neuronal and Mitochondrial Damage
Abstract
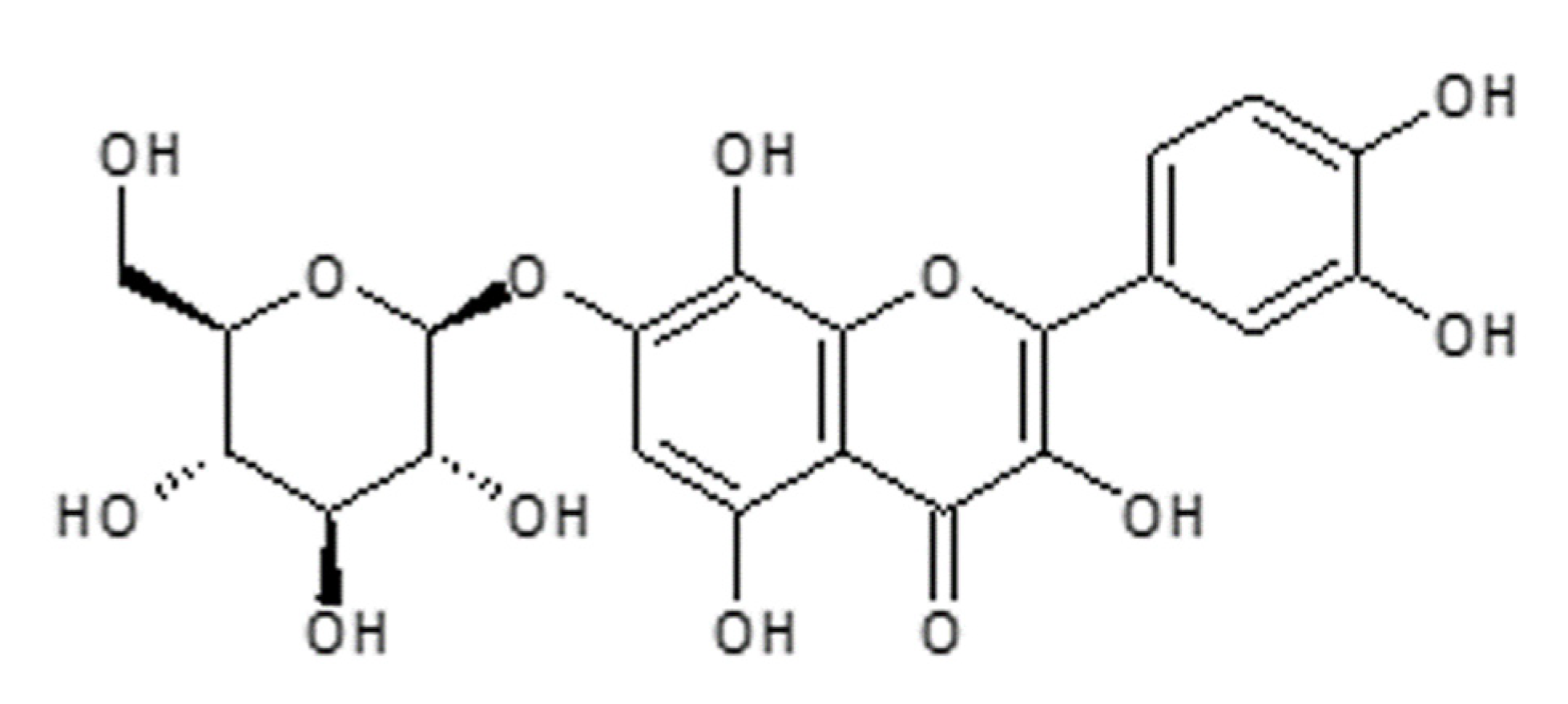
1. Introduction
2. Results
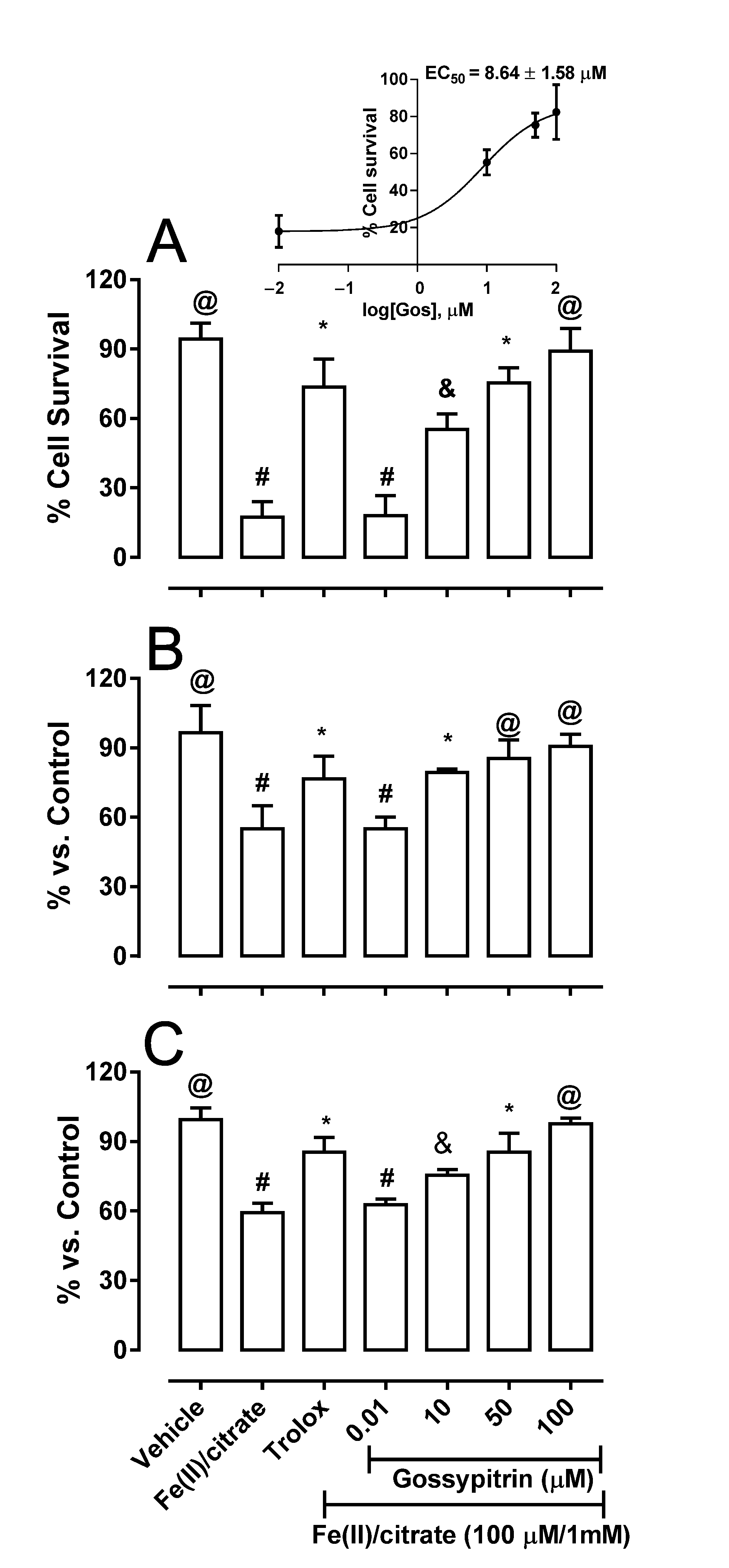
2.1. Gos Rescues HT-22 Cells from Fe(II)-Induced Cell Death
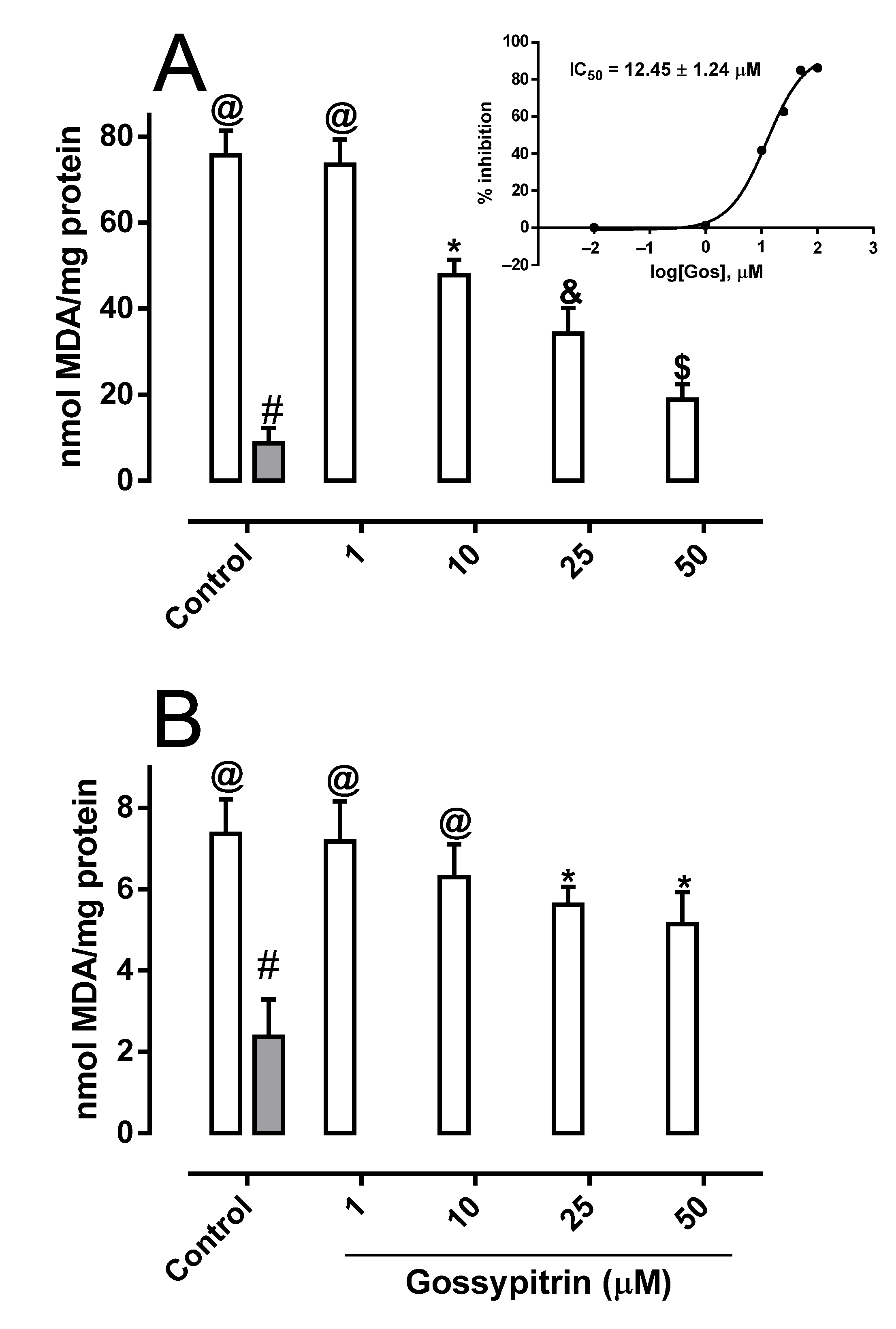
2.2. Gos Rescues Rat-Brain Mitochondria from Iron-Induced Mitochondrial Lipoperoxidation
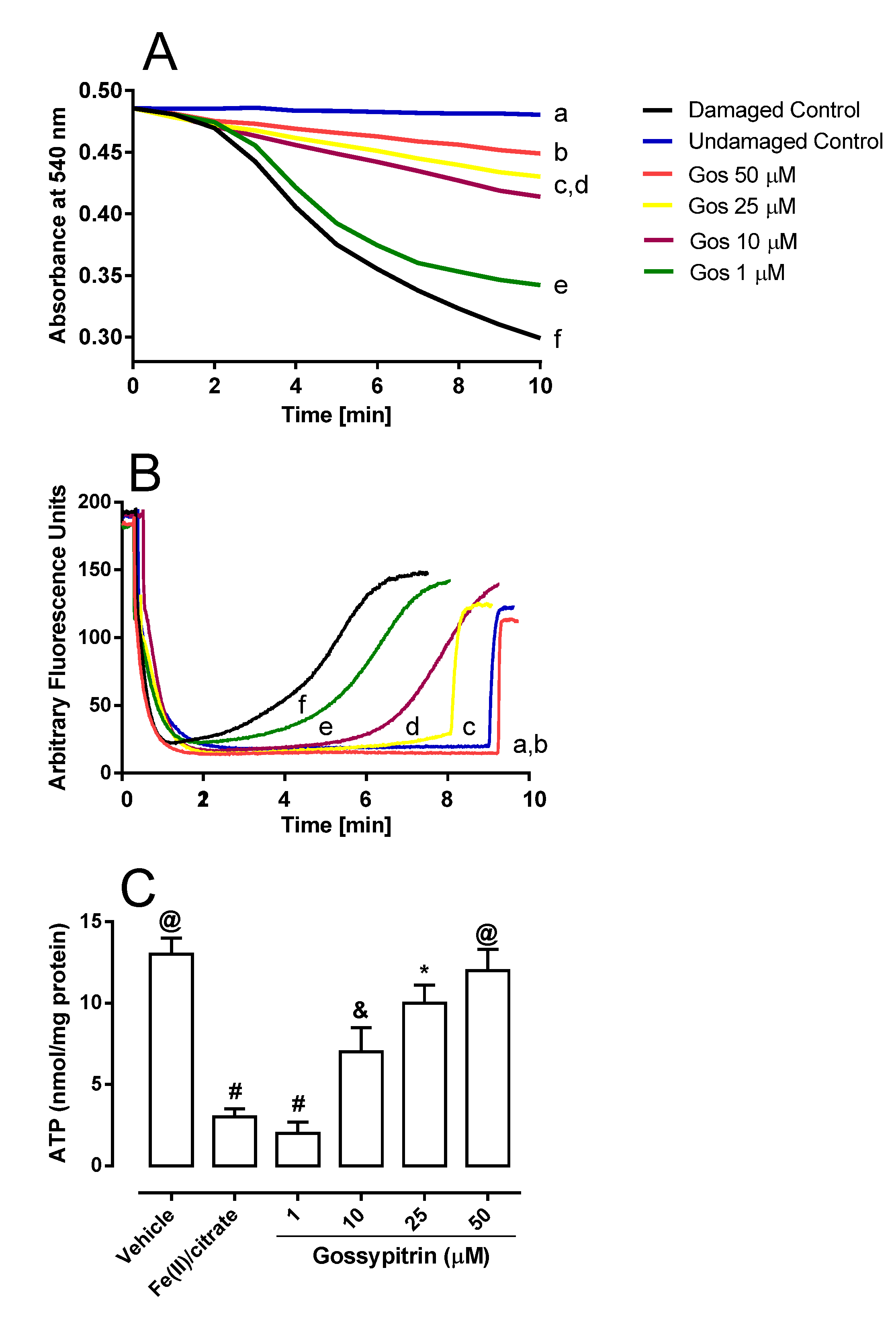
2.3. Gos Prevents Iron-Mediated Mitochondrial Swelling, ΔΨ Dissipation, and ATP Depletion
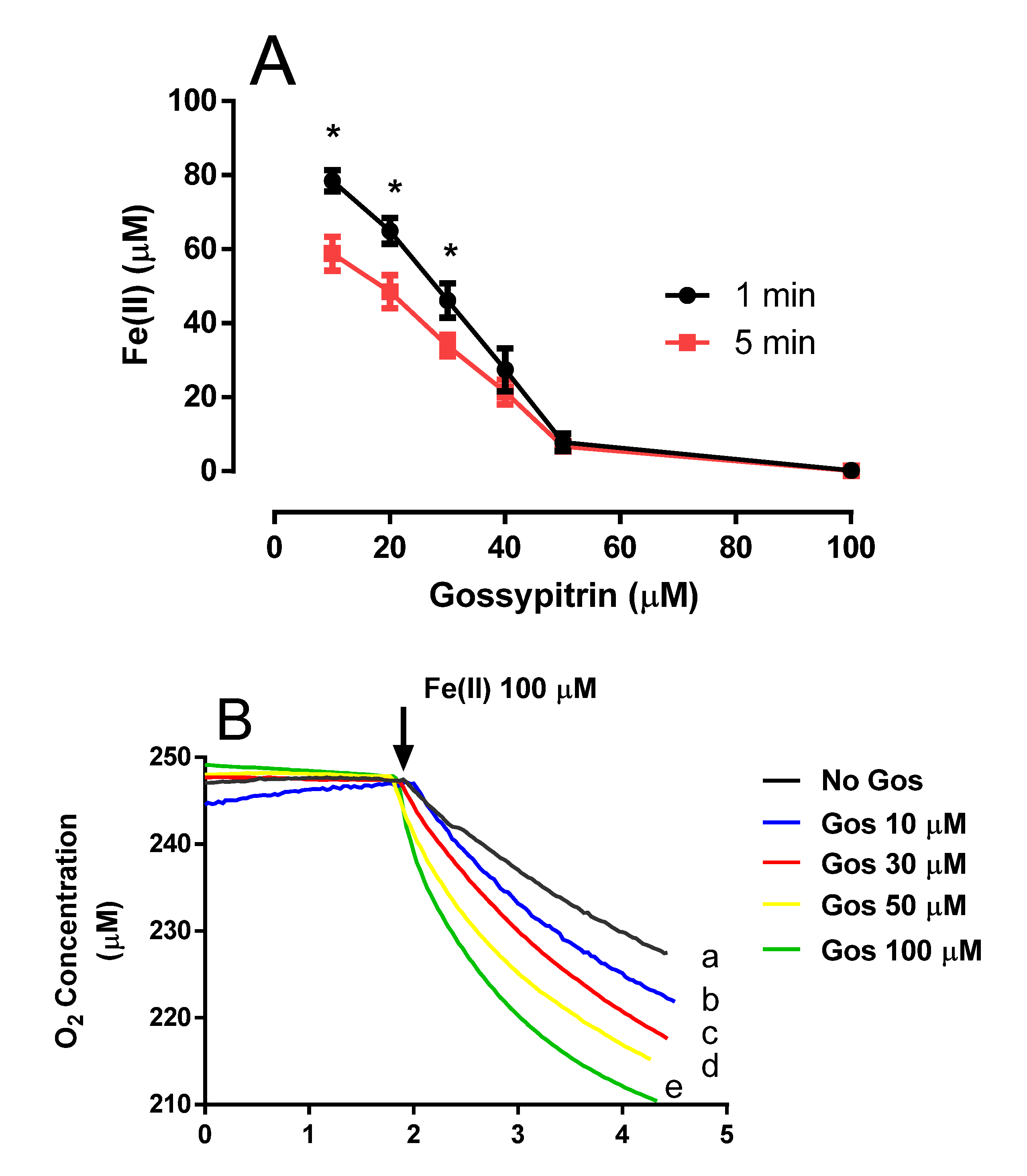
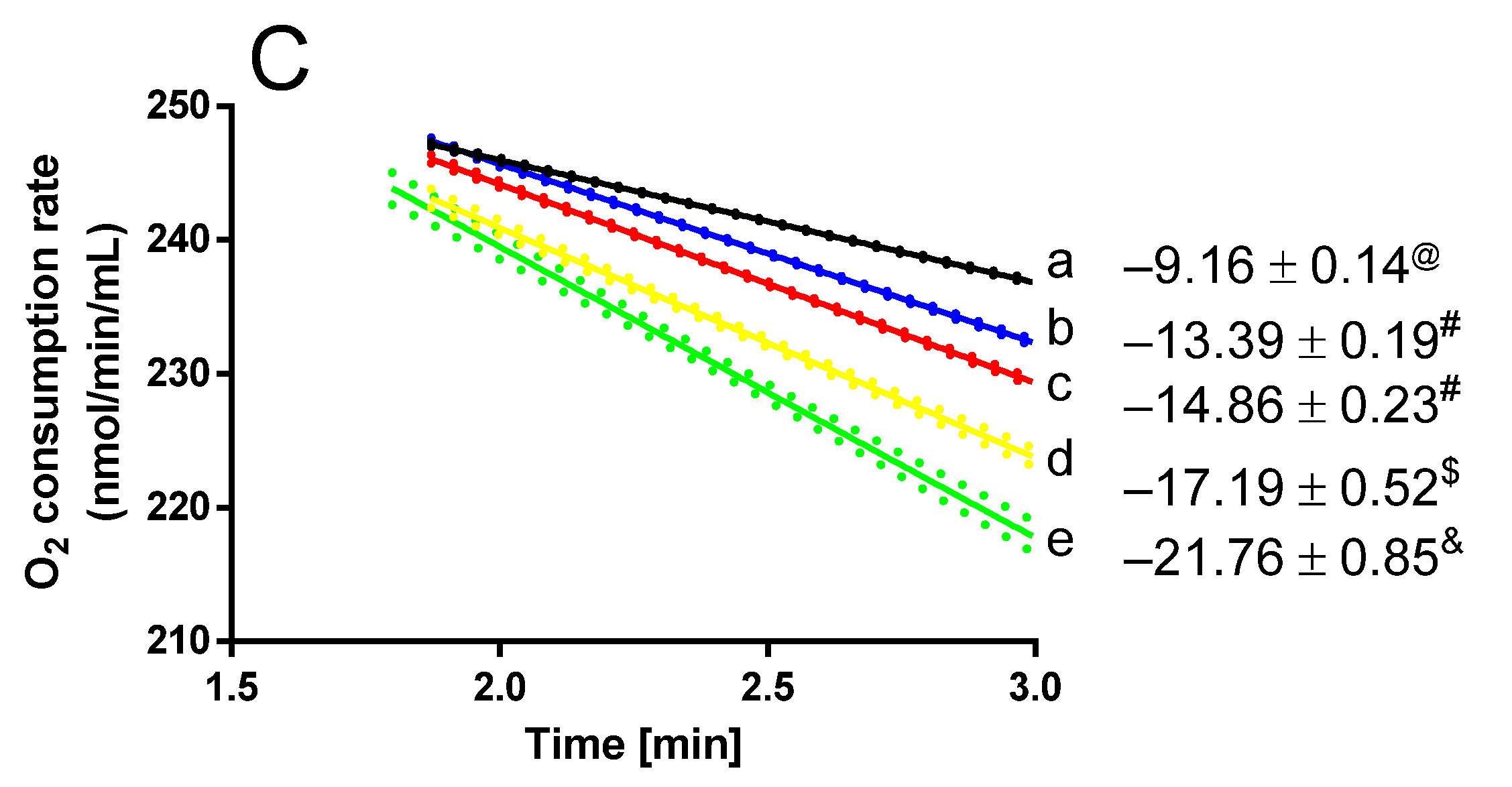
2.4. Gos Induces Fe(II)-Citrate Autoxidation and O2 Consumption
2.5. Gos–Fe(II) Complexes Detection
2.6. Gos Prevented Fe(III) Reduction by Ascorbate
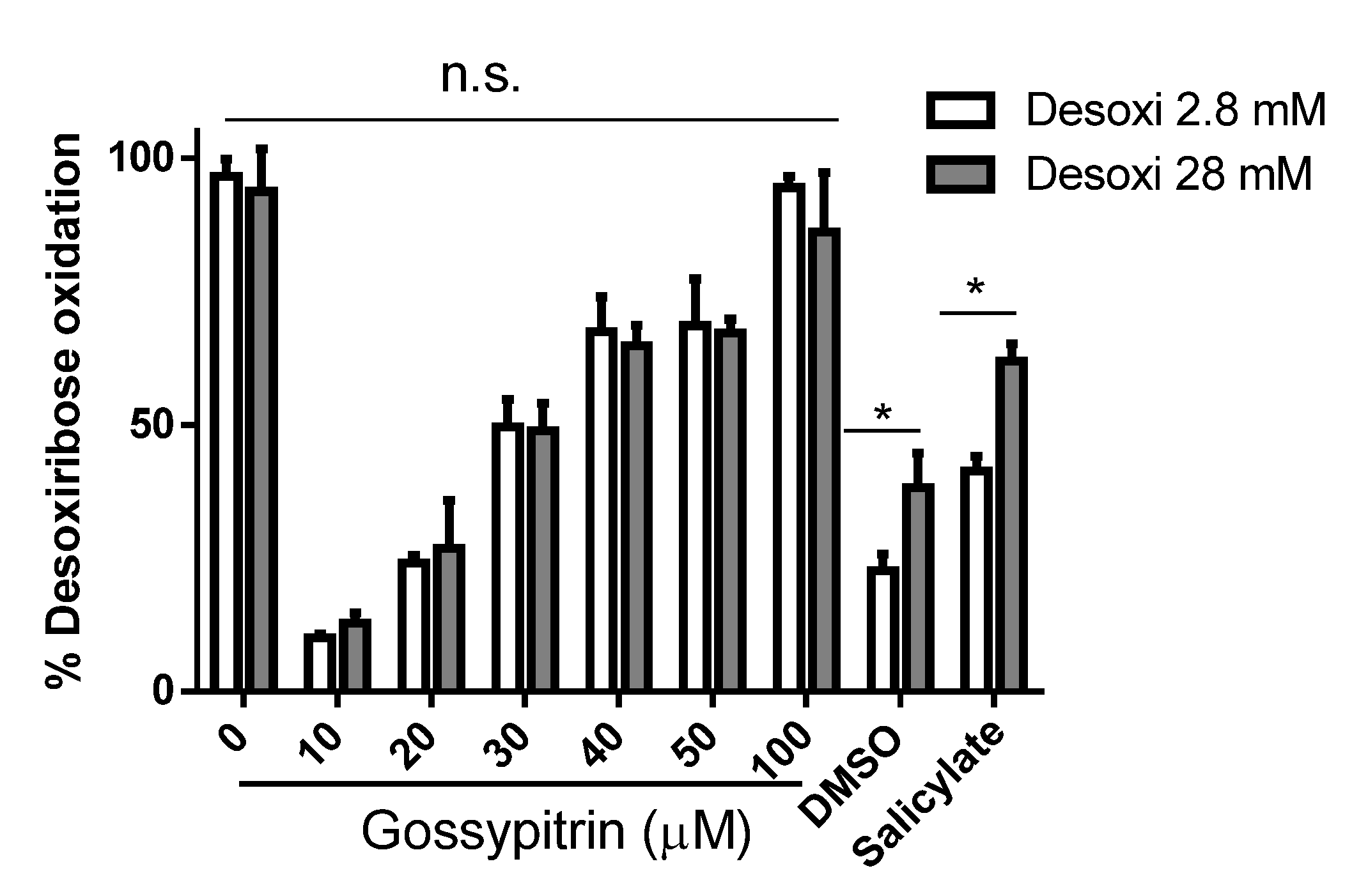
2.7. Gos Protects against 2-Deoxyribose Oxidative Degradation
3. Discussion
4. Materials and Methods
4.1. Reagents
4.2. Plant Material
4.3. Extract and Sample Preparation
4.4. UV/Vis and IR Spectroscopic Studies
4.5. Cell Cultures
4.6. Induction of Cell Oxidative Damage and Treatment with Gos
4.7. Cell Viability Assay
4.8. Mitochondrial Membrane Potential (ΔΨ) Assay in HT-22 Cells
4.9. ATP Assay
4.10. Animals
4.11. Mitochondrial Isolation
4.12. Continuous-Monitoring Mitochondrial Assays
4.13. Lipid Peroxidation Assays
4.14. Determination of Fe(II) Concentration
4.15. Oxygen Consumption Monitoring
4.16. 2-Deoxyribose Oxidation Assay
4.17. Gos-Fe(II) Complexes Detection
4.18. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Gutteridge, J.M.; Halliwell, B. Free radicals and antioxidants in the year 2000. A historical look to the future. Ann. N. Y. Acad. Sci. 2000, 899, 136–147. [Google Scholar] [CrossRef]
- Kruszewski, M. Labile iron pool: The main determinant of cellular response to oxidative stress. Mutat. Res. Mol. Mech. Mutagen. 2003, 531, 81–92. [Google Scholar] [CrossRef]
- Nunez, M.T.; Gallardo, V.; Munoz, P.; Tapia, V.; Esparza, A.; Salazar, J.; Speisky, H. Progressive iron accumulation induces a biphasic change in the glutathione content of neuroblastoma cells. Free Radic. Biol. Med. 2004, 37, 953–960. [Google Scholar] [CrossRef]
- Ward, R.J.; Zucca, F.A.; Duyn, J.H.; Crichton, R.; Zecca, L. The role of iron in brain ageing and neurodegenerative disorders. Lancet Neurol. 2014, 13, 1045–1060. [Google Scholar] [CrossRef]
- Zhu, W.Z.; Zhong, W.D.; Wang, W.; Zhan, C.J.; Wang, C.Y.; Qi, J.P.; Wang, J.Z.; Lei, T. Quantitative MR phase-corrected imaging to investigate increased brain iron deposition of patients with Alzheimer disease. Radiology 2009, 253, 497–504. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Righy, C.; Bozza, M.T.; Oliveira, M.F.; Bozza, F.A. Molecular, cellular and clinical aspects of intracerebral hemorrhage: Are the enemies within? Curr. Neuropharmacol. 2016, 14, 392–402. [Google Scholar] [CrossRef]
- DeGregorio-Rocasolano, N.; Martí-Sistac, O.; Gasull, T. Deciphering the iron side of stroke: Neurodegeneration at the crossroads between iron dyshomeostasis, excitotoxicity, and ferroptosis. Front. Neurosci. 2019, 13, 85. [Google Scholar] [CrossRef] [PubMed]
- Zille, M.; Karuppagounder, S.S.; Chen, Y.; Gough, P.J.; Bertin, J.; Finger, J.; Milner, T.A.; Jonas, E.A.; Ratan, R.R. Neuronal death after hemorrhagic stroke in vitro and in vivo shares features of ferroptosis and necroptosis. Stroke 2017, 48, 1033–1043. [Google Scholar] [CrossRef]
- Horowitz, M.P.; Greenamyre, J.T. Mitochondrial iron metabolism and its role in neurodegeneration. J. Alzheimers Dis. 2010, 20, 551–568. [Google Scholar] [CrossRef]
- Wang, Z.B.; Liu, J.Y.; Xu, X.J.; Mao, X.Y.; Zhang, W.; Zhou, H.H.; Liu, Z.Q. Neurodegeneration with brain iron accumulation: Insights into the mitochondria dysregulation. Biomed. Pharmacother. 2019, 118, 109068. [Google Scholar] [CrossRef] [PubMed]
- Enns, G.M. The contribution of mitochondria to common disorders. Mol. Genet. Metab. 2003, 80, 11–26. [Google Scholar] [CrossRef] [PubMed]
- Gogvadze, V.; Orrenius, S.; Zhivotovsky, B. Mitochondria as targets for chemotherapy. Apoptosis 2009, 14, 624–640. [Google Scholar] [CrossRef]
- Jellinger, K.A. Recent advances in our understanding of neurodegeneration. J. Neural. Transm. 2009, 116, 1111–1162. [Google Scholar] [CrossRef]
- Mandemakers, W.; Morais, V.A.; Strooper, B.D. A cell biological perspective on mitochondrial dysfunction in Parkinson disease and other neurodegenerative diseases. J. Cell Sci. 2007, 120, 1707–1716. [Google Scholar] [CrossRef]
- Anand, N.; Holcom, A.; Broussalian, M.; Schmidt, M.; Chinta, S.J.; Lithgow, G.J.; Andersen, J.K.; Chamoli, M. Dysregulated iron metabolism in C. Elegans catp-6/ATP13A2 mutant impairs mitochondrial function. Neurobiol. Dis. 2020, 139, 104786. [Google Scholar] [CrossRef]
- Hanafy, K.A.; Gomes, J.A.; Selim, M. Rationale and current evidence for testing iron chelators for treating stroke. Curr. Cardiol. Rep. 2019, 21, 20. [Google Scholar] [CrossRef] [PubMed]
- Forni, G.L.; Balocco, M.; Cremonesi, L.; Abbruzzese, G.; Parodi, R.C.; Marchese, R. Regression of symptoms after selective iron chelation therapy in a case of neurodegeneration with brain iron accumulation. Mov. Disord. 2008, 23, 904–907. [Google Scholar] [CrossRef]
- Mena, N.P.; Garcia-Beltran, O.; Lourido, F.; Urrutia, P.J.; Mena, R.; Castro-Castillo, V.; Cassels, B.K.; Nunez, M.T. The novel mitochondrial iron chelator 5-((methylamino) methyl)-8-hydroxyquinoline protects against mitochondrial-induced oxidative damage and neuronal death. Biochem. Biophys. Res. Commun. 2015, 463, 787–792. [Google Scholar] [CrossRef]
- Hanson, L.R.; Roeytenberg, A.; Martinez, P.M.; Coppes, V.G.; Sweet, D.C.; Rao, R.J.; Marti, D.L.; Hoekman, J.D.; Matthews, R.B.; Frey, W.H.; et al. Intranasal deferoxamine provides increased brain exposure and significant protection in rat ischemic stroke. J. Pharmacol. Exp. Ther. 2009, 330, 679–686. [Google Scholar] [CrossRef]
- Di Meo, F.; Valentino, A.; Petillo, O.; Peluso, G.; Filosa, S.; Crispi, S. Bioactive polyphenols and neuromodulation: Molecular mechanisms in neurodegeneration. Int. J. Mol. Sci. 2020, 21, 2564. [Google Scholar] [CrossRef] [PubMed]
- Renaud, J.; Martinoli, M.G. Considerations for the use of polyphenols as therapies in neurodegenerative diseases. Int. J. Mol. Sci. 2019, 20, 1883. [Google Scholar] [CrossRef]
- de la Vega-Hernández, K.; Antuch, M.; Cuesta-Rubio, O.; Núñez-Figueredo, Y.; Pardo-Andreu, G.L. Discerning the antioxidant mechanism of rapanone: A naturally occurring benzoquinone with iron complexing and radical scavenging activities. J. Inorg. Biochem. 2017, 170, 134–147. [Google Scholar] [CrossRef]
- Figueredo, Y.N.; García-Pupo, L.; Cuesta Rubio, O.; Delgado Hernández, R.; Naal, Z.; Curti, C.; Pardo Andreu, G.L. A strong protective action of guttiferone-A, a naturally occurring prenylated benzophenone, against iron-induced neuronal cell damage. J. Pharmacol. Sci. 2011, 116, 36–46. [Google Scholar] [CrossRef]
- Pardo Andreu, G.; Delgado, R.; Velho, J.; Inada, N.M.; Curti, C.; Vercesi, A.E. Mangifera indica L. extract (Vimang) inhibits Fe2+-citrate induced lipoperoxidation in isolated rat liver mitochondria. Pharmacol. Res. 2005, 51, 427–435. [Google Scholar] [CrossRef] [PubMed]
- Pardo-Andreu, G.L.; Barrios, M.F.; Curti, C.; Hernández, I.; Merino, N.; Lemus, Y.; Martínez, I.; Riaño, A.; Delgado, R. Protective effects of Mangifera indica L. extract (Vimang), and its major component mangiferin, on iron-induced oxidative damage to rat serum and liver. Pharmacol. Res. 2008, 57, 79–86. [Google Scholar] [CrossRef] [PubMed]
- Pardo-Andreu, G.L.; Delgado, R.; Núñez-Sellés, A.J.; Vercesi, A.E. Dual mechanism of mangiferin protection against iron-induced damage to 2-deoxyribose and ascorbate oxidation. Pharmacol. Res. 2006, 53, 253–260. [Google Scholar] [CrossRef] [PubMed]
- Pardo-Andreu, G.L.; Delgado, R.; Núñez-Sellés, A.J.; Vercesi, A.E. Mangifera indica L. extract (Vimang) inhibits 2-deoxyribose damage induced by Fe (III) plus ascorbate. Phytother. Res. 2006, 20, 120–124. [Google Scholar] [CrossRef]
- Andreu, G.P.; Delgado, R.; Velho, J.A.; Curti, C.; Vercesi, A.E. Iron complexing activity of mangiferin, a naturally occurring glucosylxanthone, inhibits mitochondrial lipid peroxidation induced by Fe2+-citrate. Eur. J. Pharmacol. 2005, 513, 47–55. [Google Scholar] [CrossRef]
- Bécquer-Viart, M.A.; González-Yaque, J.; Fonseca-Fonseca, L.A.; Núñez-Figueredo, Y.; Pardo Andreu, G.L. Antioxidant and neuroprotective effects of gossypitrin, a flavonoid from Taliparity elatum, against chemical hypoxia-induced PC12 cell death. J. Pharm. Pharmacogn. Res. 2018, 6, 72–80. [Google Scholar]
- Mira, L.; Fernandez, M.T.; Santos, M.; Rocha, R.; Florencio, M.H.; Jennings, K.R. Interactions of flavonoids with iron and copper ions: A mechanism for their antioxidant activity. Free Radic. Res. 2002, 36, 1199–1208. [Google Scholar] [CrossRef] [PubMed]
- Raza, A.; Xu, X.; Xia, L.; Xia, C.; Tang, J.; Ouyang, Z. Quercetin-iron complex: Synthesis, characterization, antioxidant, DNA binding, DNA cleavage, and antibacterial activity studies. J. Fluoresc. 2016, 26, 2023–2031. [Google Scholar] [CrossRef] [PubMed]
- Uranga, R.M.; Salvador, G.A. Unraveling the burden of iron in neurodegeneration: Intersections with amyloid beta peptide pathology. Oxid. Med. Cell. Longev. 2018, 2018, 2850341. [Google Scholar] [CrossRef]
- Gerlach, M.; Ben-Shachar, D.; Riederer, P.; Youdim, M.B. Altered brain metabolism of iron as a cause of neurodegenerative diseases? J. Neurochem. 1994, 63, 793–807. [Google Scholar] [CrossRef]
- Carbonell, T.; Rama, M. Iron, oxidative stress and early neurological deterioration in ischemic stroke. Curr. Med. Chem. 2007, 14, 857–874. [Google Scholar] [CrossRef] [PubMed]
- Niizuma, K.; Endo, H.; Chan, P.H. Oxidative stress and mitochondrial dysfunction as determinants of ischemic neuronal death and survival. J. Neurochem. 2009, 109, 133–138. [Google Scholar] [CrossRef] [PubMed]
- Pelizzoni, I.; Macco, R.; Morini, M.F.; Zacchetti, D.; Grohovaz, F.; Codazzi, F. Iron handling in hippocampal neurons: Activity-dependent iron entry and mitochondria-mediated neurotoxicity. Aging Cell 2011, 10, 172–183. [Google Scholar] [CrossRef]
- Sims, N.R.; Anderson, M.F. Mitochondrial contributions to tissue damage in stroke. Neurochem. Int. 2002, 40, 511–526. [Google Scholar] [CrossRef]
- Sims, N.R.; Muyderman, H. Mitochondria, oxidative metabolism and cell death in stroke. Biochim. Biophys. Acta 2010, 1802, 80–91. [Google Scholar] [CrossRef]
- Richardson, D.R.; Ponka, P. Pyridoxal isonicotinoyl hydrazone and its analogues: Potential orally effective iron chelating agents for the treatment of iron overload disease. J. Lab. Clin. Med 1998, 13, 306–315. [Google Scholar] [CrossRef]
- Orisakwe, O.E.; Amadi, C.N.; Frazzoli, C. Management of iron overload in resource poor nations: A systematic review of phlebotomy and natural chelators. J. Toxicol. 2020, 2020, 4084538. [Google Scholar] [CrossRef]
- Lee, D.G.; Kam, M.K.; Kim, K.M.; Kim, H.S.; Kwon, O.S.; Lee, H.S.; Lee, D.S. Peroxiredoxin 5 prevents iron overload-induced neuronal death by inhibiting mitochondrial fragmentation and endoplasmic reticulum stress in mouse hippocampal HT-22 cells. Int. J. Biochem. Cell Biol. 2018, 102, 10–19. [Google Scholar] [CrossRef]
- Lee, D.G.; Park, J.; Lee, H.S.; Lee, S.R.; Lee, D.S. Iron overload-induced calcium signals modulate mitochondrial fragmentation in HT-22 hippocampal neuron cells. Toxicology 2016, 365, 17–24. [Google Scholar] [CrossRef]
- Huang, H.; Chen, J.; Lu, H.; Zhou, M.; Chai, Z.; Hu, Y. Iron-induced generation of mitochondrial ROS depends on AMPK activity. BioMetals 2017, 30, 623–628. [Google Scholar] [CrossRef] [PubMed]
- Gáll, J.; Škrha, J.; Buchal, R.; Sedláčková, E.; Verébová, K.; Pláteník, J. Induction of the mitochondrial permeability transition (MPT) by micromolar iron: Liberation of calcium is more important than NAD(P)H oxidation. Biochim. Biophys. Acta Bioenerg. 2012, 1817, 1537–1549. [Google Scholar] [CrossRef] [PubMed]
- González, J.; Cuellar, A.; Nossin, E.; Monan, M. Iron chelating activity of gossypitrin isolated from the petals of Talipariti Elatum Sw. (Fryxell ) Malvaceae. J. Agric. Stud. 2017, 5, 1–12. [Google Scholar] [CrossRef][Green Version]
- Pardo-Andreu, G.; Sánchez-Baldoquín, C.; Ávila-González, R.; Delgado, R.; Naal, Z.; Curti, C. Fe(III) improves antioxidant and cytoprotecting activities of mangiferin. Eur. J. Pharmacol. 2006, 547, 31–36. [Google Scholar] [CrossRef]
- Pardo-Andreu, G.L.; Sánchez-Baldoquín, C.; Avila-González, R.; Yamamoto, E.T.S.; Revilla, A.; Uyemura, S.A.; Naal, Z.; Delgado, R.; Curti, C. Interaction of Vimang (Mangifera indica L. extract) with Fe(III) improves its antioxidant and cytoprotecting activity. Pharmacol. Res. 2006, 54, 389–395. [Google Scholar] [CrossRef]
- Pardo-Andreu, G.L.; Cavalheiro, R.A.; Dorta, D.J.; Naal, Z.; Delgado, R.; Vercesi, A.E.; Curti, C. Fe(III) shifts the mitochondria permeability transition-eliciting capacity of mangiferin to protection of organelle. J. Pharmacol. Exp. Ther. 2007, 320, 646–653. [Google Scholar] [CrossRef]
- Welch, K.D.; Davies, T.Z.; Aust, S.D. Iron autoxidation and free radical generation: Effects of buffer, ligands and chelators. Arch. Biochem. Biophys. 2002, 397, 360–369. [Google Scholar] [CrossRef] [PubMed]
- Speer, R.E.; Karuppagounder, S.S.; Basso, M.; Sleiman, S.; Kumar, A.; Brand, D.; Smirnova, N.; Gazaryan, I.; Khim, S.J.; Ratan, R.R. Hypoxia inducible factor prolyl hydroxylases as targets for neuroprotection by “antioxidant” metal chelators: From ferroptosis to stroke. Free Radic. Biol. Med. 2013, 62, 26–36. [Google Scholar] [CrossRef]
- Siddiq, A.; Ayoub, I.A.; Chavez, J.C.; Aminova, L.; Shah, S.; LaManna, J.C.; Patton, S.M.; Connor, J.R.; Cherny, R.A.; Volitakis, I.; et al. Hypoxia-inducible factor prolyl 4-hydroxylase inhibition. A target for neuroprotection in the central nervous system. J. Biol. Chem. 2005, 280, 41732–41743. [Google Scholar] [CrossRef] [PubMed]
- Hirsila, M.; Koivunen, P.; Gunzler, V.; Kivirikko, K.I.; Myllyharju, J. Characterization of the human prolyl 4-hydroxylases that modify the hypoxia-inducible factor. J. Biol. Chem. 2003, 278, 30772–30780. [Google Scholar] [CrossRef]
- Semenza, G.L. Oxygen-dependent regulation of mitochondrial respiration by hypoxia-inducible factor 1. Biochem. J. 2007, 405, 1–9. [Google Scholar] [CrossRef]
- Li, H.-S.; Zhou, Y.-N.; Li, L.; Li, S.-F.; Long, D.; Chen, X.-L.; Zhang, J.-B.; Feng, L.; Li, Y.-P. HIF-1 α protects against oxidative stress by directly targeting mitochondria. Redox Biol. 2019, 25, 101109. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.; Zhao, W.; Yu, J.; Li, S.; Lin, L.; Chen, X. Induction of ferroptosis and mitochondrial dysfunction by oxidative stress in PC12 cells. Sci. Rep. 2018, 8, 574. [Google Scholar] [CrossRef] [PubMed]
- Schliemann, W.; Schneider, B.; Wray, V.; Schmidt, J.; Nimtz, M.; Porzel, A.; Böhm, H. Flavonols and an indole alkaloid skeleton bearing identical acylated glycosidic groups from yellow petals of Papaver nudicaule. Phytochemistry 2006, 67, 191–201. [Google Scholar] [CrossRef] [PubMed]
- Nakagawa, Y.; Moore, G. Role of mitochondrial membrane permeability transition in p-hydroxybenzoate ester-induced cytotoxicity in rat hepatocytes. Biochem. Pharmacol. 1999, 58, 811–816. [Google Scholar] [CrossRef]
- Pardo-Andreu, G.L.; Nuñez-Figueredo, Y.; Tudella, V.G.; Cuesta-Rubio, O.; Rodrigues, F.P.; Pestana, C.R.; Uyemura, S.A.; Leopoldino, A.M.; Alberici, L.C.; Curti, C. The anti-cancer agent nemorosone is a new potent protonophoric mitochondrial uncoupler. Mitochondrion 2011, 11, 255–263. [Google Scholar] [CrossRef]
- Pardo-Andreu, G.L.; Nuñez-Figueredo, Y.; Tudella, V.G.; Cuesta-Rubio, O.; Rodrigues, F.P.; Pestana, C.R.; Uyemura, S.A.; Leopoldino, A.M.; Alberici, L.C.; Curti, C. The anti-cancer agent guttiferone-A permeabilizes mitochondrial membrane: Ensuing energetic and oxidative stress implications. Toxicol. Appl. Pharmacol. 2011, 253, 282–289. [Google Scholar] [CrossRef]
- Mirandola, S.R.; Melo, D.R.; Saito, A.; Castilho, R.F. 3-nitropropionic acid-induced mitochondrial permeability transition: Comparative study of mitochondria from different tissues and brain regions. J. Neurosci. Res. 2010, 88, 630–639. [Google Scholar] [CrossRef]
- Zanotti, A.; Azzone, G.F. Safranine as membrane potential probe in rat liver mitochondria. Arch. Biochem. Biophys. 1980, 201, 255–265. [Google Scholar] [CrossRef]
- Buege, J.A.; Aust, S.D. Microsomal lipid peroxidation. Methods Enzymol. 1978, 52, 302–310. [Google Scholar] [CrossRef] [PubMed]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bécquer-Viart, M.Á.; Armentero-López, A.; Alvarez-Almiñaque, D.; Fernández-Acosta, R.; Matos-Peralta, Y.; D’Vries, R.F.; Marín-Prida, J.; Pardo-Andreu, G.L. Gossypitrin, A Naturally Occurring Flavonoid, Attenuates Iron-Induced Neuronal and Mitochondrial Damage. Molecules 2021, 26, 3364. https://doi.org/10.3390/molecules26113364
Bécquer-Viart MÁ, Armentero-López A, Alvarez-Almiñaque D, Fernández-Acosta R, Matos-Peralta Y, D’Vries RF, Marín-Prida J, Pardo-Andreu GL. Gossypitrin, A Naturally Occurring Flavonoid, Attenuates Iron-Induced Neuronal and Mitochondrial Damage. Molecules. 2021; 26(11):3364. https://doi.org/10.3390/molecules26113364
Chicago/Turabian StyleBécquer-Viart, María Ángeles, Adonis Armentero-López, Daniel Alvarez-Almiñaque, Roberto Fernández-Acosta, Yasser Matos-Peralta, Richard F. D’Vries, Javier Marín-Prida, and Gilberto L. Pardo-Andreu. 2021. "Gossypitrin, A Naturally Occurring Flavonoid, Attenuates Iron-Induced Neuronal and Mitochondrial Damage" Molecules 26, no. 11: 3364. https://doi.org/10.3390/molecules26113364
APA StyleBécquer-Viart, M. Á., Armentero-López, A., Alvarez-Almiñaque, D., Fernández-Acosta, R., Matos-Peralta, Y., D’Vries, R. F., Marín-Prida, J., & Pardo-Andreu, G. L. (2021). Gossypitrin, A Naturally Occurring Flavonoid, Attenuates Iron-Induced Neuronal and Mitochondrial Damage. Molecules, 26(11), 3364. https://doi.org/10.3390/molecules26113364






