Recent Advances and the Potential for Clinical Use of Autofluorescence Detection of Extra-Ophthalmic Tissues
Abstract
1. Introduction
2. Results
2.1. Autofluorescence Imaging (AFI) in Gastrointestinal Diseases
2.1.1. AFI in Gastroesophageal Reflux Disease
2.1.2. AFI in Intestinal Inflammation
2.1.3. AFI in Upper GI Neoplasia
2.1.4. The Accuracy of AFI for the Diagnosis of Colorectal Cancer
2.2. AF Bronchoscopy in Pulmonary Diseases
2.3. AF for Non-Invasive Cardiovascular Risk Prediction
2.3.1. AF of Advanced Glycation End Products
2.3.2. SAF Determination in Diabetes
2.3.3. SAF Determination in Chronic Kidney Disease
2.3.4. SAF Determination in Chronic Obstructive Pulmonary Disease
2.4. AF in Oral Medicine
2.5. AF of the Thyroid and Parathyroid Glands
2.6. Multiphoton Laser Tomography in Dermatology
2.7. Fluorescence Lifetime Imaging Microscopy
2.8. Biomolecules Acting as Endogenous Fluorophores
3. Discussion
3.1. Future Directions
3.2. Further Experimental Trends with Translational Potential
4. Materials and Methods
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| AGE | advanced glycation end products |
| AF | autofluorescence |
| AFB | autofluorescence bronchoscopy |
| AFI | autofluorescence imaging |
| AU | arbitrary units |
| CFOME | confocal fiber-optic micro-endoscopy |
| CI | confidence interval |
| CKD | chronic kidney disease |
| COPD | chronic obstructive pulmonary disease |
| DAFE | diagnostic autofluorescence endoscopy |
| ETMI | endoscopic trimodal imaging |
| ECM | extracellular matrix |
| FAD | flavin adenine dinucleotide |
| FH | functional heartburn |
| FLIM | fluorescence lifetime imaging microscopy |
| GERD | gastroesophageal reflux disease |
| GOLD | global initiative for chronic obstructive lung disease |
| HRE | high resolution endoscopy |
| LDCT | low-dose computed tomography |
| MPT | multiphoton tomography |
| NAD(P)H | reduced nicotinamide adenine dinucleotide phosphate |
| NBI | narrow band imaging |
| NERD | non-erosive reflux disease |
| NILT | near-infrared light transillumination |
| NIR | near-infrared |
| OTIS | overlay tissue imaging system |
| pCLE | probe-based confocal laser endomicroscopy |
| PTG | parathyroid gland |
| UC | ulcerative colitis |
| RGB | red, green and blue |
| SAF | skin autofluorescence |
| SVE | standard white-light video endoscopy |
| SNP | single nucleotide polymorphism |
| WL | white light |
| WLB | white light bronchoscopy |
| WLE | white light endoscopy |
References
- Croce, A.C.; Bottiroli, G. Autofluorescence spectroscopy and imaging: A tool for biomedical research and diagnosis. Eur. J. Histochem. 2014, 58, 2461. [Google Scholar] [CrossRef] [PubMed]
- Wizenty, J.; Ashraf, M.I.; Rohwer, N.; Stockmann, M.; Weiss, S.; Biebl, M.; Pratschke, J.; Aigner, F.; Wuensch, T. Autofluorescence: A potential pitfall in immunofluorescence-based inflammation grading. J. Immunol. Methods 2018, 456, 28–37. [Google Scholar] [CrossRef] [PubMed]
- Wuensch, T.; Wizenty, J.; Quint, J.; Spitz, W.; Bosma, M.; Becker, O.; Adler, A.; Veltzke-Schlieker, W.; Stockmann, M.; Weiss, S.; et al. Expression Analysis of Fibronectin Type III Domain-Containing (FNDC) Genes in Inflammatory Bowel Disease and Colorectal Cancer. Gastroenterol. Res. Pract. 2019, 2019, 3784172. [Google Scholar] [CrossRef] [PubMed]
- Yung, M.; Klufas, M.A.; Sarraf, D. Clinical applications of fundus autofluorescence in retinal disease. Int. J. Retina. Vitreous 2016, 2, 12. [Google Scholar] [CrossRef]
- Ly, A.; Nivison-Smith, L.; Assaad, N.; Kalloniatis, M. Fundus Autofluorescence in Age-related Macular Degeneration. Optom. Vis. Sci. 2017, 94, 246–259. [Google Scholar] [CrossRef]
- Curvers, W.L.; Alvarez Herrero, L.; Wallace, M.B.; Wong Kee Song, L.M.; Ragunath, K.; Wolfsen, H.C.; Prasad, G.A.; Wang, K.K.; Subramanian, V.; Weusten, B.L.; et al. Endoscopic tri-modal imaging is more effective than standard endoscopy in identifying early-stage neoplasia in Barrett’s esophagus. Gastroenterology 2010, 139, 1106–1114. [Google Scholar] [CrossRef]
- Jenvey, C.J.; Stabel, J.R. Autofluorescence and Nonspecific Immunofluorescent Labeling in Frozen Bovine Intestinal Tissue Sections: Solutions for Multicolor Immunofluorescence Experiments. J. Histochem. Cytochem. 2017, 65, 531–541. [Google Scholar] [CrossRef]
- Wang, W.; Uedo, N.; Yang, Y.; Peng, L.; Bai, D.; Lu, Z.; Fan, K.; Wang, J.; Wang, X.; Zhao, Y.; et al. Autofluorescence imaging endoscopy for predicting acid reflux in patients with gastroesophageal reflux disease. J. Gastroenterol. Hepatol. 2014, 29, 1442–1448. [Google Scholar] [CrossRef]
- Luo, X.; Guo, X.X.; Wang, W.F.; Peng, L.H.; Yang, Y.S.; Uedo, N. Autofluorescence imaging endoscopy can distinguish non-erosive reflux disease from functional heartburn: A pilot study. World J. Gastroenterol. 2016, 22, 3845–3851. [Google Scholar] [CrossRef]
- Osada, T.; Arakawa, A.; Sakamoto, N.; Ueyama, H.; Shibuya, T.; Ogihara, T.; Yao, T.; Watanabe, S. Autofluorescence imaging endoscopy for identification and assessment of inflammatory ulcerative colitis. World J. Gastroenterol. 2011, 17, 5110–5116. [Google Scholar] [CrossRef]
- Moriichi, K.; Fujiya, M.; Ijiri, M.; Tanaka, K.; Sakatani, A.; Dokoshi, T.; Fujibayashi, S.; Ando, K.; Nomura, Y.; Ueno, N.; et al. Quantification of autofluorescence imaging can accurately and objectively assess the severity of ulcerative colitis. Int. J. Colorectal. Dis. 2015, 30, 1639–1643. [Google Scholar] [CrossRef] [PubMed]
- Giacchino, M.; Bansal, A.; Kim, R.E.; Singh, V.; Hall, S.B.; Singh, M.; Rastogi, A.; Moloney, B.; Wani, S.B.; Gaddam, S.; et al. Clinical utility and interobserver agreement of autofluorescence imaging and magnification narrow-band imaging for the evaluation of Barrett’s esophagus: A prospective tandem study. Gastrointest. Endosc. 2013, 77, 711–718. [Google Scholar] [CrossRef] [PubMed]
- Mannath, J.; Subramanian, V.; Telakis, E.; Lau, K.; Ramappa, V.; Wireko, M.; Kaye, P.V.; Ragunath, K. An inter-observer agreement study of autofluorescence endoscopy in Barrett’s esophagus among expert and non-expert endoscopists. Dig. Dis. Sci. 2013, 58, 465–470. [Google Scholar] [CrossRef] [PubMed]
- So, J.; Rajnakova, A.; Chan, Y.H.; Tay, A.; Shah, N.; Salto-Tellez, M.; Teh, M.; Uedo, N. Endoscopic tri-modal imaging improves detection of gastric intestinal metaplasia among a high-risk patient population in Singapore. Dig. Dis. Sci. 2013, 58, 3566–3575. [Google Scholar] [CrossRef]
- Lim, L.G.; Yeoh, K.G.; Srivastava, S.; Chan, Y.H.; Teh, M.; Ho, K.Y. Comparison of probe-based confocal endomicroscopy with virtual chromoendoscopy and white-light endoscopy for diagnosis of gastric intestinal metaplasia. Surg. Endosc. 2013, 27, 4649–4655. [Google Scholar] [CrossRef]
- Ishihara, R.; Inoue, T.; Hanaoka, N.; Takeuchi, Y.; Tsujii, Y.; Kanzaki, H.; Oota, T.; Hanafusa, M.; Yamamoto, S.; Nagai, K.; et al. Autofluorescence imaging endoscopy for screening of esophageal squamous mucosal high-grade neoplasia: A phase II study. J. Gastroenterol. Hepatol. 2012, 27, 86–90. [Google Scholar] [CrossRef]
- McGinty, J.; Galletly, N.P.; Dunsby, C.; Munro, I.; Elson, D.S.; Requejo-Isidro, J.; Cohen, P.; Ahmad, R.; Forsyth, A.; Thillainayagam, A.V.; et al. Wide-field fluorescence lifetime imaging of cancer. Biomed. Opt. Express 2010, 1, 627–640. [Google Scholar] [CrossRef]
- Van den Broek, F.J.; Fockens, P.; van Eeden, S.; Reitsma, J.B.; Hardwick, J.C.; Stokkers, P.C.; Dekker, E. Endoscopic tri-modal imaging for surveillance in ulcerative colitis: Randomised comparison of high-resolution endoscopy and autofluorescence imaging for neoplasia detection; and evaluation of narrow-band imaging for classification of lesions. Gut 2008, 57, 1083–1089. [Google Scholar] [CrossRef]
- Rotondano, G.; Bianco, M.A.; Sansone, S.; Prisco, A.; Meucci, C.; Garofano, M.L.; Cipolletta, L. Trimodal endoscopic imaging for the detection and differentiation of colorectal adenomas: A prospective single-centre clinical evaluation. Int. J. Colorectal. Dis. 2012, 27, 331–336. [Google Scholar] [CrossRef]
- Vleugels, J.L.A.; Rutter, M.D.; Ragunath, K.; Rees, C.J.; Ponsioen, C.Y.; Lahiff, C.; Ket, S.N.; Wanders, L.K.; Samuel, S.; Butt, F.; et al. Diagnostic Accuracy of Endoscopic Trimodal Imaging and Chromoendoscopy for Lesion Characterization in Ulcerative Colitis. J. Crohns. Colitis. 2018, 12, 1438–1447. [Google Scholar] [CrossRef]
- Vleugels, J.L.A.; Rutter, M.D.; Ragunath, K.; Rees, C.J.; Ponsioen, C.Y.; Lahiff, C.; Ket, S.N.; Wanders, L.K.; Samuel, S.; Butt, F.; et al. Chromoendoscopy versus autofluorescence imaging for neoplasia detection in patients with longstanding ulcerative colitis (FIND-UC): An international, multicentre, randomised controlled trial. Lancet Gastroenterol. Hepatol. 2018, 3, 305–316. [Google Scholar] [CrossRef]
- Kuiper, T.; van den Broek, F.J.; Naber, A.H.; van Soest, E.J.; Scholten, P.; Mallant-Hent, R.; van den Brande, J.; Jansen, J.M.; van Oijen, A.H.; Marsman, W.A.; et al. Endoscopic trimodal imaging detects colonic neoplasia as well as standard video endoscopy. Gastroenterology 2011, 140, 1887–1894. [Google Scholar] [CrossRef] [PubMed]
- Moriichi, K.; Fujiya, M.; Sato, R.; Nata, T.; Nomura, Y.; Ueno, N.; Ishikawa, C.; Inaba, Y.; Ito, T.; Okamoto, K.; et al. Autofluorescence imaging and the quantitative intensity of fluorescence for evaluating the dysplastic grade of colonic neoplasms. Int. J. Colorectal. Dis. 2012, 27, 325–330. [Google Scholar] [CrossRef] [PubMed]
- Takeuchi, Y.; Sawaya, M.; Oka, S.; Tamai, N.; Kawamura, T.; Uraoka, T.; Ikematsu, H.; Moriyama, T.; Arao, M.; Ishikawa, H.; et al. Efficacy of autofluorescence imaging for flat neoplasm detection: A multicenter randomized controlled trial (A-FLAT trial). Gastrointest. Endosc. 2019, 89, 460–469. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, T.; Nakamura, S.; Moriyama, T.; Hirahashi, M.; Iida, M. Autofluorescence imaging colonoscopy for the detection of dysplastic lesions in ulcerative colitis: A pilot study. Colorectal. Dis. 2010, 12, e291–e297. [Google Scholar] [CrossRef] [PubMed]
- Sato, R.; Fujiya, M.; Watari, J.; Ueno, N.; Moriichi, K.; Kashima, S.; Maeda, S.; Ando, K.; Kawabata, H.; Sugiyama, R.; et al. The diagnostic accuracy of high-resolution endoscopy, autofluorescence imaging and narrow-band imaging for differentially diagnosing colon adenoma. Endoscopy 2011, 43, 862–868. [Google Scholar] [CrossRef]
- Takeuchi, Y.; Inoue, T.; Hanaoka, N.; Higashino, K.; Iishi, H.; Chatani, R.; Hanafusa, M.; Kizu, T.; Ishihara, R.; Tatsuta, M.; et al. Autofluorescence imaging with a transparent hood for detection of colorectal neoplasms: A prospective, randomized trial. Gastrointest. Endosc. 2010, 72, 1006–1013. [Google Scholar] [CrossRef]
- Kuiper, T.; Alderlieste, Y.A.; Tytgat, K.M.; Vlug, M.S.; Nabuurs, J.A.; Bastiaansen, B.A.; Lowenberg, M.; Fockens, P.; Dekker, E. Automatic optical diagnosis of small colorectal lesions by laser-induced autofluorescence. Endoscopy 2015, 47, 56–62. [Google Scholar] [CrossRef]
- Van der Heijden, E.H.F.M.; Hoefsloot, W.; van Hees, H.W.H.; Schuurbiers, O.C.J. High definition bronchoscopy: A randomized exploratory study of diagnostic value compared to standard white light bronchoscopy and autofluorescence bronchoscopy. Respir. Res. 2015, 16, 33. [Google Scholar] [CrossRef]
- Zaric, B.; Becker, H.D.; Perin, B.; Stojanovic, G.; Jovelic, A.; Eri, Z.; Panjkovic, M.; Ilic, M.D.; Matijasevic, J.; Antonic, M. Autofluorescence imaging videobronchoscopy improves assessment of tumor margins and affects therapeutic strategy in central lung cancer. Jpn. J. Clin. Oncol. 2010, 40, 139–145. [Google Scholar] [CrossRef]
- Fielding, D.; Agnew, J.; Wright, D.; Hodge, R. Autofluorescence improves pretreatment mucosal assessment in head and neck cancer patients. Otolaryngol. Head Neck Surg. 2010, 142, S20–S26. [Google Scholar] [CrossRef] [PubMed]
- Tremblay, A.; Taghizadeh, N.; McWilliams, A.M.; MacEachern, P.; Stather, D.R.; Soghrati, K.; Puksa, S.; Goffin, J.R.; Yasufuku, K.; Amjadi, K.; et al. Low Prevalence of High-Grade Lesions Detected With Autofluorescence Bronchoscopy in the Setting of Lung Cancer Screening in the Pan-Canadian Lung Cancer Screening Study. Chest 2016, 150, 1015–1022. [Google Scholar] [CrossRef] [PubMed]
- Spiro, S.G.; Shah, P.L.; Rintoul, R.C.; George, J.; Janes, S.; Callister, M.; Novelli, M.; Shaw, P.; Kocjan, G.; Griffiths, C.; et al. Sequential screening for lung cancer in a high-risk group: Randomised controlled trial: LungSEARCH: A randomised controlled trial of Surveillance using sputum and imaging for the EARly detection of lung Cancer in a High-risk group. Eur. Respir. J. 2019, 54. [Google Scholar] [CrossRef] [PubMed]
- Iga, N.; Oto, T.; Okada, M.; Harada, M.; Nishikawa, H.; Miyoshi, K.; Otani, S.; Sugimoto, S.; Yamane, M.; Toyooka, S.; et al. Detection of airway ischaemic damage after lung transplantation by using autofluorescence imaging bronchoscopy. Eur. J. Cardiothorac. Surg. 2014, 45, 509–513. [Google Scholar] [CrossRef] [PubMed]
- Yserbyt, J.; Dooms, C.; Decramer, M.; Verleden, G.M. Acute lung allograft rejection: Diagnostic role of probe-based confocal laser endomicroscopy of the respiratory tract. J. Heart Lung. Transplant. 2014, 33, 492–498. [Google Scholar] [CrossRef] [PubMed]
- Lam, S.; Mandrekar, S.J.; Gesthalter, Y.; Allen Ziegler, K.L.; Seisler, D.K.; Midthun, D.E.; Mao, J.T.; Aubry, M.C.; McWilliams, A.; Sin, D.D.; et al. A Randomized Phase IIb Trial of myo-Inositol in Smokers with Bronchial Dysplasia. Cancer Prev. Res. (Phila.) 2016, 9, 906–914. [Google Scholar] [CrossRef]
- Xie, J.; Mendez, J.D.; Mendez-Valenzuela, V.; Aguilar-Hernandez, M.M. Cellular signalling of the receptor for advanced glycation end products (RAGE). Cell. Signal. 2013, 25, 2185–2197. [Google Scholar] [CrossRef]
- Monnier, V.M.; Kohn, R.R.; Cerami, A. Accelerated age-related browning of human collagen in diabetes mellitus. Proc. Nat. Acad. Sci. USA 1984, 81, 583–587. [Google Scholar] [CrossRef]
- Meerwaldt, R.; Graaff, R.; Oomen, P.H.N.; Links, T.P.; Jager, J.J.; Alderson, N.L.; Thorpe, S.R.; Baynes, J.W.; Gans, R.O.B.; Smit, A.J. Simple non-invasive assessment of advanced glycation endproduct accumulation. Diabetologia 2004, 47, 1324–1330. [Google Scholar] [CrossRef]
- Hofmann, B.; Adam, A.C.; Jacobs, K.; Riemer, M.; Erbs, C.; Bushnaq, H.; Simm, A.; Silber, R.E.; Santos, A.N. Advanced glycation end product associated skin autofluorescence: A mirror of vascular function? Exp. Gerontol. 2013, 48, 38–44. [Google Scholar] [CrossRef]
- Momma, H.; Niu, K.; Kobayashi, Y.; Guan, L.; Sato, M.; Guo, H.; Chujo, M.; Otomo, A.; Yufei, C.; Tadaura, H.; et al. Skin advanced glycation end product accumulation and muscle strength among adult men. Eur. J. Appl. Physiol. 2011, 111, 1545–1552. [Google Scholar] [CrossRef] [PubMed]
- Yozgatli, K.; Lefrandt, J.D.; Noordzij, M.J.; Oomen, P.H.N.; Brouwer, T.; Jager, J.; Castro Cabezas, M.; Smit, A.J. Accumulation of advanced glycation end products is associated with macrovascular events and glycaemic control with microvascular complications in Type 2 diabetes mellitus. Diabet. Med. 2018, 35, 1242–1248. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Xu, L.; Gao, H.; Ye, J.; Huang, Y.; Wu, M.; Xie, T.; Ni, P.; Yu, X.; Cao, Y.; et al. The association between skin autofluorescence and vascular complications in Chinese patients with diabetic foot ulcer: An observational study done in Shanghai. Int. J. Low Extrem. Wounds 2015, 14, 28–36. [Google Scholar] [CrossRef] [PubMed]
- Sanchez, E.; Baena-Fustegueras, J.A.; de la Fuente, M.C.; Gutierrez, L.; Bueno, M.; Ros, S.; Lecube, A. Advanced glycation end-products in morbid obesity and after bariatric surgery: When glycemic memory starts to fail. Endocrinol. Diabetes Nutr. 2017, 64, 4–10. [Google Scholar] [CrossRef]
- Ninomiya, H.; Katakami, N.; Sato, I.; Osawa, S.; Yamamoto, Y.; Takahara, M.; Kawamori, D.; Matsuoka, T.A.; Shimomura, I. Association between Subclinical Atherosclerosis Markers and the Level of Accumulated Advanced Glycation End-Products in the Skin of Patients with Diabetes. J. Atheroscler. Thromb. 2018, 25, 1274–1284. [Google Scholar] [CrossRef]
- Van Eupen, M.G.; Schram, M.T.; van Sloten, T.T.; Scheijen, J.; Sep, S.J.; van der Kallen, C.J.; Dagnelie, P.C.; Koster, A.; Schaper, N.; Henry, R.M.; et al. Skin Autofluorescence and Pentosidine Are Associated With Aortic Stiffening: The Maastricht Study. Hypertension 2016, 68, 956–963. [Google Scholar] [CrossRef]
- Hofmann, B.; Jacobs, K.; Navarrete Santos, A.; Wienke, A.; Silber, R.E.; Simm, A. Relationship between cardiac tissue glycation and skin autofluorescence in patients with coronary artery disease. Diabetes Metab. 2015, 41, 410–415. [Google Scholar] [CrossRef]
- Willemsen, S.; Hartog, J.W.; Hummel, Y.M.; van Ruijven, M.H.; van der Horst, I.C.; van Veldhuisen, D.J.; Voors, A.A. Tissue advanced glycation end products are associated with diastolic function and aerobic exercise capacity in diabetic heart failure patients. Eur. J. Heart Fail. 2011, 13, 76–82. [Google Scholar] [CrossRef]
- Hartog, J.W.; van de Wal, R.M.; Schalkwijk, C.G.; Miyata, T.; Jaarsma, W.; Plokker, H.W.; van Wijk, L.M.; Smit, A.J.; van Veldhuisen, D.J.; Voors, A.A. Advanced glycation end-products, anti-hypertensive treatment and diastolic function in patients with hypertension and diastolic dysfunction. Eur. J. Heart Fail. 2010, 12, 397–403. [Google Scholar] [CrossRef]
- Blanc-Bisson, C.; Velayoudom-Cephise, F.L.; Cougnard-Gregoire, A.; Helmer, C.; Rajaobelina, K.; Delcourt, C.; Alexandre, L.; Blanco, L.; Mohammedi, K.; Monlun, M.; et al. Skin autofluorescence predicts major adverse cardiovascular events in patients with type 1 diabetes: A 7-year follow-up study. Cardiovasc. Diabetol. 2018, 17, 82. [Google Scholar] [CrossRef]
- Uruska, A.; Gandecka, A.; Araszkiewicz, A.; Zozulinska-Ziolkiewicz, D. Accumulation of advanced glycation end products in the skin is accelerated in relation to insulin resistance in people with Type 1 diabetes mellitus. Diabet. Med. 2019, 36, 620–625. [Google Scholar] [CrossRef] [PubMed]
- Banser, A.; Naafs, J.C.; Hoorweg-Nijman, J.J.; van de Garde, E.M.; van der Vorst, M.M. Advanced glycation end products, measured in skin, vs. HbA1c in children with type 1 diabetes mellitus. Pediatr. Diabetes 2016, 17, 426–432. [Google Scholar] [CrossRef] [PubMed]
- Van Waateringe, R.P.; Fokkens, B.T.; Slagter, S.N.; van der Klauw, M.M.; van Vliet-Ostaptchouk, J.V.; Graaff, R.; Paterson, A.D.; Smit, A.J.; Lutgers, H.L.; Wolffenbuttel, B.H.R. Skin autofluorescence predicts incident type 2 diabetes, cardiovascular disease and mortality in the general population. Diabetologia 2019, 62, 269–280. [Google Scholar] [CrossRef] [PubMed]
- Smit, A.J.; Smit, J.M.; Botterblom, G.J.; Mulder, D.J. Skin autofluorescence based decision tree in detection of impaired glucose tolerance and diabetes. PLoS ONE 2013, 8, e65592. [Google Scholar] [CrossRef] [PubMed]
- Verhey, F.R.J.; Spauwen, P.J.J.; Köhler, S.; van Boxtel, M.P.J.; Stehouwer, C.D.A.; Schalkwijk, C.G.; van der Kallen, C.J.H.; van Eupen, M.G.A.; Schram, M.T.; Schaper, N.C.; et al. Associations of Advanced Glycation End-Products With Cognitive Functions in Individuals With and Without Type 2 Diabetes: The Maastricht Study. J. Clin. Endocrinol. Metab. 2015, 100, 951–960. [Google Scholar] [CrossRef]
- Bakker, S.F.; Tushuizen, M.E.; Gozutok, E.; Ciftci, A.; Gelderman, K.A.; Mulder, C.J.; Simsek, S. Advanced glycation end products (AGEs) and the soluble receptor for AGE (sRAGE) in patients with type 1 diabetes and coeliac disease. Nutr. Metab. Cardiovasc. Dis. 2015, 25, 230–235. [Google Scholar] [CrossRef]
- Hartog, J.W.; Willemsen, S.; van Veldhuisen, D.J.; Posma, J.L.; van Wijk, L.M.; Hummel, Y.M.; Hillege, H.L.; Voors, A.A. Effects of alagebrium, an advanced glycation endproduct breaker, on exercise tolerance and cardiac function in patients with chronic heart failure. Eur. J. Heart Fail. 2011, 13, 899–908. [Google Scholar] [CrossRef]
- Stirban, A.; Pop, A.; Fischer, A.; Heckermann, S.; Tschoepe, D. Variability of skin autofluorescence measurement over 6 and 12 weeks and the influence of benfotiamine treatment. Diabetes Technol. Ther. 2013, 15, 733–737. [Google Scholar] [CrossRef]
- Li, Z.; Wang, G.; Zhu, Y.-J.; Li, C.-G.; Tang, Y.-Z.; Jiang, Z.-H.; Yang, M.; Ni, C.-L.; Chen, L.-M.; Niu, W.-Y. The relationship between circulating irisin levels and tissues AGE accumulation in type 2 diabetes patients. Biosci. Rep. 2017, 37, BSR20170213. [Google Scholar] [CrossRef]
- Flaim, C.; Kob, M.; Di Pierro, A.M.; Herrmann, M.; Lucchin, L. Effects of a whey protein supplementation on oxidative stress, body composition and glucose metabolism among overweight people affected by diabetes mellitus or impaired fasting glucose: A pilot study. J. Nutr. Biochem. 2017, 50, 95–102. [Google Scholar] [CrossRef]
- Chen, J.; van der Duin, D.; Campos-Obando, N.; Ikram, M.A.; Nijsten, T.E.C.; Uitterlinden, A.G.; Zillikens, M.C. Serum 25-hydroxyvitamin D3 is associated with advanced glycation end products (AGEs) measured as skin autofluorescence: The Rotterdam Study. Eur. J. Epidemiol. 2019, 34, 67–77. [Google Scholar] [CrossRef] [PubMed]
- Krul-Poel, Y.H.M.; Agca, R.; Lips, P.; van Wijland, H.; Stam, F.; Simsek, S. Vitamin D status is associated with skin autofluorescence in patients with type 2 diabetes mellitus: A preliminary report. Cardiovasc. Diabetol. 2015, 14, 89. [Google Scholar] [CrossRef] [PubMed]
- Sturmer, M.; Sebekova, K.; Fazeli, G.; Bahner, U.; Stab, F.; Heidland, A. 25-hydroxyvitamin d and advanced glycation endproducts in healthy and hypertensive subjects: Are there interactions? J. Ren. Nutr. 2015, 25, 209–216. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, K.; Nakayama, M.; Kanno, M.; Kimura, H.; Watanabe, K.; Tani, Y.; Kusano, Y.; Suzuki, H.; Hayashi, Y.; Asahi, K.; et al. Skin autofluorescence is associated with the progression of chronic kidney disease: A prospective observational study. PLoS ONE 2013, 8, e83799. [Google Scholar] [CrossRef] [PubMed]
- Nongnuch, A.; Davenport, A. Skin autofluorescence advanced glycosylation end products as an independent predictor of mortality in high flux haemodialysis and haemodialysis patients. Nephrology (Carlton) 2015, 20, 862–867. [Google Scholar] [CrossRef] [PubMed]
- Nongnuch, A.; Davenport, A. The effect of vegetarian diet on skin autofluorescence measurements in haemodialysis patients. Br. J. Nutr. 2015, 113, 1040–1043. [Google Scholar] [CrossRef]
- Macsai, E.; Benke, A.; Kiss, I. Skin Autofluorescence and Mortality in Patients on Peritoneal Dialysis. Medicine 2015, 94, e1933. [Google Scholar] [CrossRef]
- Jiang, J.; Chen, P.; Chen, J.; Yu, X.; Xie, D.; Mei, C.; Xiong, F.; Shi, W.; Zhou, W.; Liu, X.; et al. Accumulation of tissue advanced glycation end products correlated with glucose exposure dose and associated with cardiovascular morbidity in patients on peritoneal dialysis. Atherosclerosis 2012, 224, 187–194. [Google Scholar] [CrossRef]
- Crowley, L.E.; Johnson, C.P.; McIntyre, N.; Fluck, R.J.; McIntyre, C.W.; Taal, M.W.; Leung, J.C. Tissue advanced glycation end product deposition after kidney transplantation. Nephron. Clin. Pract. 2013, 124, 54–59. [Google Scholar] [CrossRef]
- Schutte, E.; de Vos, L.C.; Lutgers, H.L.; Lambers Heerspink, H.J.; Wolffenbuttel, B.H.; Vart, P.; Zeebregts, C.J.; Gansevoort, R.T.; Lefrandt, J.D. Association of Skin Autofluorescence Levels With Kidney Function Decline in Patients With Peripheral Artery Disease. Arterioscler. Thromb. Vasc. Biol. 2016, 36, 1709–1714. [Google Scholar] [CrossRef]
- Franca, R.A.; Esteves, A.B.A.; Borges, C.M.; Quadros, K.; Falcao, L.C.N.; Caramori, J.C.T.; Oliveira, R.B. Advanced glycation end-products (AGEs) accumulation in skin: Relations with chronic kidney disease-mineral and bone disorder. J. Bras. Nefrol. 2017, 39, 253–260. [Google Scholar] [CrossRef] [PubMed]
- Hoonhorst, S.J.; Lo Tam Loi, A.T.; Hartman, J.E.; Telenga, E.D.; van den Berge, M.; Koenderman, L.; Lammers, J.W.; Boezen, H.M.; Postma, D.S.; Ten Hacken, N.H. Advanced glycation end products in the skin are enhanced in COPD. Metabolism 2014, 63, 1149–1156. [Google Scholar] [CrossRef] [PubMed]
- Vogelmeier, C.F.; Criner, G.J.; Martinez, F.J.; Anzueto, A.; Barnes, P.J.; Bourbeau, J.; Celli, B.R.; Chen, R.; Decramer, M.; Fabbri, L.M.; et al. Global Strategy for the Diagnosis, Management, and Prevention of Chronic Obstructive Lung Disease 2017 Report: GOLD Executive Summary. Arch. Bronconeumol. 2017, 53, 128–149. [Google Scholar] [CrossRef] [PubMed]
- Kubo, A.; Kato, M.; Sugioka, Y.; Mitsui, R.; Fukuhara, N.; Nihei, F.; Takeda, Y. Relationship between advanced glycation end-product accumulation in the skin and pulmonary function. J. Phys. Ther. Sci. 2018, 30, 413–418. [Google Scholar] [CrossRef][Green Version]
- Hoonhorst, S.J.; Lo Tam Loi, A.T.; Pouwels, S.D.; Faiz, A.; Telenga, E.D.; van den Berge, M.; Koenderman, L.; Lammers, J.W.; Boezen, H.M.; van Oosterhout, A.J.; et al. Advanced glycation endproducts and their receptor in different body compartments in COPD. Respir. Res. 2016, 17, 46. [Google Scholar] [CrossRef]
- Kanchwala, N.; Kumar, N.; Gupta, S.; Lokhandwala, H. Fluorescence spectroscopic study on malignant and premalignant oral mucosa of patients undergoing treatment: An observational prospective study. Int. J. Surg. 2018, 55, 87–91. [Google Scholar] [CrossRef]
- Sawan, D.; Mashlah, A. Evaluation of premalignant and malignant lesions by fluorescent light (VELscope). J. Int. Soc. Prev. Community Dent. 2015, 5, 248–254. [Google Scholar] [CrossRef]
- Hanken, H.; Kraatz, J.; Smeets, R.; Heiland, M.; Assaf, A.T.; Blessmann, M.; Eichhorn, W.; Clauditz, T.S.; Grobe, A.; Kolk, A.; et al. The detection of oral pre-malignant lesions with an autofluorescence based imaging system (VELscope)–a single blinded clinical evaluation. Head Face Med. 2013, 9, 23. [Google Scholar] [CrossRef]
- Rana, M.; Zapf, A.; Kuehle, M.; Gellrich, N.C.; Eckardt, A.M. Clinical evaluation of an autofluorescence diagnostic device for oral cancer detection: A prospective randomized diagnostic study. Eur. J. Cancer Prev. 2012, 21, 460–466. [Google Scholar] [CrossRef]
- Simonato, L.E.; Tomo, S.; Scarparo Navarro, R.; Balbin Villaverde, A.G.J. Fluorescence visualization improves the detection of oral, potentially malignant, disorders in population screening. Photodiagn. Photodyn. Ther. 2019, 27, 74–78. [Google Scholar] [CrossRef]
- Koch, F.P.; Kaemmerer, P.W.; Biesterfeld, S.; Kunkel, M.; Wagner, W. Effectiveness of autofluorescence to identify suspicious oral lesions–a prospective, blinded clinical trial. Clin. Oral Investig. 2011, 15, 975–982. [Google Scholar] [CrossRef] [PubMed]
- Mehrotra, R.; Singh, M.; Thomas, S.; Nair, P.; Pandya, S.; Nigam, N.S.; Shukla, P. A cross-sectional study evaluating chemiluminescence and autofluorescence in the detection of clinically innocuous precancerous and cancerous oral lesions. J. Am. Dent. Assoc. 2010, 141, 151–156. [Google Scholar] [CrossRef] [PubMed]
- Cherry, K.D.; Schwarz, R.A.; Yang, E.C.; Vohra, I.S.; Badaoui, H.; Williams, M.D.; Vigneswaran, N.; Gillenwater, A.M.; Richards-Kortum, R. Autofluorescence Imaging to Monitor the Progression of Oral Potentially Malignant Disorders. Cancer Prev. Res. (Phila.) 2019, 12, 791–800. [Google Scholar] [CrossRef] [PubMed]
- Moro, A.; De Waure, C.; Di Nardo, F.; Spadari, F.; Mignogna, M.D.; Giuliani, M.; Califano, L.; Gianni, A.B.; Cardarelli, L.; Celentano, A.; et al. The GOCCLES(R) medical device is effective in detecting oral cancer and dysplasia in dental clinical setting. Results from a multicentre clinical trial. Acta Otorhinolaryngol. Ital. 2015, 35, 449–454. [Google Scholar] [CrossRef] [PubMed]
- Winiarski, P.; Szewczyk-Golec, K.; Orlowski, P.; Kaluzna, E.; Wamka, M.; Mackiewicz-Nartowicz, H.; Sinkiewicz, A.; Fisz, J.J. Autofluorescence spectroscopy in the differentiation of laryngeal epithelial lesions - preliminary results. Acta Otolaryngol. 2016, 136, 580–584. [Google Scholar] [CrossRef]
- Angmar-Mansson, B.; ten Bosch, J.J. Quantitative light-induced fluorescence (QLF): A method for assessment of incipient caries lesions. Dentomaxillofac. Radiol. 2001, 30, 298–307. [Google Scholar] [CrossRef]
- Schwass, D.R.; Leichter, J.W.; Purton, D.G.; Swain, M.V. Evaluating the efficiency of caries removal using an Er:YAG laser driven by fluorescence feedback control. Arch. Oral Biol. 2013, 58, 603–610. [Google Scholar] [CrossRef]
- Thomas, S.S.; Mohanty, S.; Jayanthi, J.L.; Varughese, J.M.; Balan, A.; Subhash, N. Clinical trial for detection of dental caries using laser-induced fluorescence ratio reference standard. J. Biomed. Opt. 2010, 15, 027001. [Google Scholar] [CrossRef]
- Lederer, A.; Kunzelmann, K.H.; Heck, K.; Hickel, R.; Litzenburger, F. In vitro validation of near-infrared transillumination at 780 nm for the detection of caries on proximal surfaces. Clin. Oral Investig. 2019, 23, 3933–3940. [Google Scholar] [CrossRef]
- Tassoker, M.; Sener, S.; Karabekiroglu, S. Occlusal Caries Detection and Diagnosis Using Visual ICDAS Criteria, Laser Fluorescence Measurements and Near-Infrared Light Transillumination Images. Med. Princ. Pract. 2020, 29, 25–31. [Google Scholar] [CrossRef]
- Terrer, E.; Raskin, A.; Koubi, S.; Dionne, A.; Weisrock, G.; Sarraquigne, C.; Mazuir, A.; Tassery, H. A new concept in restorative dentistry: LIFEDT-light-induced fluorescence evaluator for diagnosis and treatment: Part 2–treatment of dentinal caries. J. Contemp. Dent. Pract. 2010, 11, E095–E102. [Google Scholar] [PubMed]
- Banerjee, A.; Cook, R.; Kellow, S.; Shah, K.; Festy, F.; Sherriff, M.; Watson, T. A confocal micro-endoscopic investigation of the relationship between the microhardness of carious dentine and its autofluorescence. Eur. J. Oral Sci. 2010, 118, 75–79. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, A.; Kellow, S.; Mannocci, F.; Cook, R.J.; Watson, T.F. An in vitro evaluation of microtensile bond strengths of two adhesive bonding agents to residual dentine after caries removal using three excavation techniques. J. Dent. 2010, 38, 480–489. [Google Scholar] [CrossRef] [PubMed]
- Bergenfelz, A.O.; Jansson, S.K.; Wallin, G.K.; Martensson, H.G.; Rasmussen, L.; Eriksson, H.L.; Reihner, E.I. Impact of modern techniques on short-term outcome after surgery for primary hyperparathyroidism: A multicenter study comprising 2708 patients. Langenbecks. Arch. Surg. 2009, 394, 851–860. [Google Scholar] [CrossRef]
- Paras, C.; Keller, M.; White, L.; Phay, J.; Mahadevan-Jansen, A. Near-infrared autofluorescence for the detection of parathyroid glands. J. Biomed. Opt. 2011, 16, 067012. [Google Scholar] [CrossRef]
- Falco, J.; Dip, F.; Quadri, P.; de la Fuente, M.; Rosenthal, R. Cutting Edge in Thyroid Surgery: Autofluorescence of Parathyroid Glands. J. Am. Coll. Surg. 2016, 223, 374–380. [Google Scholar] [CrossRef]
- Kim, S.W.; Song, S.H.; Lee, H.S.; Noh, W.J.; Oak, C.; Ahn, Y.C.; Lee, K.D. Intraoperative Real-Time Localization of Normal Parathyroid Glands with Autofluorescence Imaging. J. Clin. Endocrinol. Metab. 2016, 101, 4646–4652. [Google Scholar] [CrossRef]
- McWade, M.A.; Paras, C.; White, L.M.; Phay, J.E.; Solorzano, C.C.; Broome, J.T.; Mahadevan-Jansen, A. Label-free intraoperative parathyroid localization with near-infrared autofluorescence imaging. J. Clin. Endocrinol. Metab. 2014, 99, 4574–4580. [Google Scholar] [CrossRef]
- McWade, M.A.; Sanders, M.E.; Broome, J.T.; Solorzano, C.C.; Mahadevan-Jansen, A. Establishing the clinical utility of autofluorescence spectroscopy for parathyroid detection. Surgery 2016, 159, 193–202. [Google Scholar] [CrossRef]
- Ladurner, R.; Al Arabi, N.; Guendogar, U.; Hallfeldt, K.; Stepp, H.; Gallwas, J. Near-infrared autofluorescence imaging to detect parathyroid glands in thyroid surgery. Ann. R. Coll. Surg. Engl. 2018, 100, 33–36. [Google Scholar] [CrossRef]
- Ladurner, R.; Lerchenberger, M.; Al Arabi, N.; Gallwas, J.K.S.; Stepp, H.; Hallfeldt, K.K.J. Parathyroid Autofluorescence-How Does It Affect Parathyroid and Thyroid Surgery? A 5 Year Experience. Molecules 2019, 24. [Google Scholar] [CrossRef] [PubMed]
- Ladurner, R.; Sommerey, S.; Arabi, N.A.; Hallfeldt, K.K.J.; Stepp, H.; Gallwas, J.K.S. Intraoperative near-infrared autofluorescence imaging of parathyroid glands. Surg. Endosc. 2017, 31, 3140–3145. [Google Scholar] [CrossRef] [PubMed]
- Alesina, P.F.; Meier, B.; Hinrichs, J.; Mohmand, W.; Walz, M.K. Enhanced visualization of parathyroid glands during video-assisted neck surgery. Langenbecks. Arch. Surg. 2018, 403, 395–401. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.W.; Lee, H.S.; Ahn, Y.C.; Park, C.W.; Jeon, S.W.; Kim, C.H.; Ko, J.B.; Oak, C.; Kim, Y.; Lee, K.D. Near-Infrared Autofluorescence Image-Guided Parathyroid Gland Mapping in Thyroidectomy. J. Am. Coll. Surg. 2018, 226, 165–172. [Google Scholar] [CrossRef]
- Kim, Y.; Kim, S.W.; Lee, K.D.; Ahn, Y.C. Video-assisted parathyroid gland mapping with autofocusing. J. Biophotonics 2019, 12, e201900017. [Google Scholar] [CrossRef]
- De Leeuw, F.; Breuskin, I.; Abbaci, M.; Casiraghi, O.; Mirghani, H.; Ben Lakhdar, A.; Laplace-Builhe, C.; Hartl, D. Intraoperative Near-infrared Imaging for Parathyroid Gland Identification by Auto-fluorescence: A Feasibility Study. World J. Surg. 2016, 40, 2131–2138. [Google Scholar] [CrossRef]
- Kose, E.; Rudin, A.V.; Kahramangil, B.; Moore, E.; Aydin, H.; Donmez, M.; Krishnamurthy, V.; Siperstein, A.; Berber, E. Autofluorescence imaging of parathyroid glands: An assessment of potential indications. Surgery 2019, 167, 173–179. [Google Scholar] [CrossRef]
- Kahramangil, B.; Dip, F.; Benmiloud, F.; Falco, J.; de La Fuente, M.; Verna, S.; Rosenthal, R.; Berber, E. Detection of Parathyroid Autofluorescence Using Near-Infrared Imaging: A Multicenter Analysis of Concordance Between Different Surgeons. Ann. Surg. Oncol. 2018, 25, 957–962. [Google Scholar] [CrossRef]
- Falco, J.; Dip, F.; Quadri, P.; de la Fuente, M.; Prunello, M.; Rosenthal, R.J. Increased identification of parathyroid glands using near infrared light during thyroid and parathyroid surgery. Surg. Endosc. 2017, 31, 3737–3742. [Google Scholar] [CrossRef]
- Kahramangil, B.; Berber, E. Comparison of indocyanine green fluorescence and parathyroid autofluorescence imaging in the identification of parathyroid glands during thyroidectomy. Gland. Surg. 2017, 6, 644–648. [Google Scholar] [CrossRef]
- Benmiloud, F.; Godiris-Petit, G.; Gras, R.; Gillot, J.C.; Turrin, N.; Penaranda, G.; Noullet, S.; Chereau, N.; Gaudart, J.; Chiche, L.; et al. Association of Autofluorescence-Based Detection of the Parathyroid Glands During Total Thyroidectomy With Postoperative Hypocalcemia Risk: Results of the PARAFLUO Multicenter Randomized Clinical Trial. JAMA Surg. 2019, 155, 106–112. [Google Scholar] [CrossRef] [PubMed]
- McWade, M.A.; Thomas, G.; Nguyen, J.Q.; Sanders, M.E.; Solorzano, C.C.; Mahadevan-Jansen, A. Enhancing Parathyroid Gland Visualization Using a Near Infrared Fluorescence-Based Overlay Imaging System. J. Am. Coll. Surg. 2019, 228, 730–743. [Google Scholar] [CrossRef] [PubMed]
- Thomas, G.; McWade, M.A.; Paras, C.; Mannoh, E.A.; Sanders, M.E.; White, L.M.; Broome, J.T.; Phay, J.E.; Baregamian, N.; Solorzano, C.C.; et al. Developing a Clinical Prototype to Guide Surgeons for Intraoperative Label-Free Identification of Parathyroid Glands in Real Time. Thyroid 2018, 28, 1517–1531. [Google Scholar] [CrossRef] [PubMed]
- Thomas, G.; McWade, M.A.; Nguyen, J.Q.; Sanders, M.E.; Broome, J.T.; Baregamian, N.; Solorzano, C.C.; Mahadevan-Jansen, A. Innovative surgical guidance for label-free real-time parathyroid identification. Surgery 2019, 165, 114–123. [Google Scholar] [CrossRef]
- Serra, C.; Silveira, L.; Canudo, A.; Lemos, M.C. Parathyroid identification by autofluorescence - preliminary report on five cases of surgery for primary hyperparathyroidism. BMC Surg. 2019, 19, 120. [Google Scholar] [CrossRef]
- DiMarco, A.; Chotalia, R.; Bloxham, R.; McIntyre, C.; Tolley, N.; Palazzo, F.F. Does fluoroscopy prevent inadvertent parathyroidectomy in thyroid surgery? Ann. R. Coll. Surg. Engl. 2019, 101, 508–513. [Google Scholar] [CrossRef]
- Benmiloud, F.; Rebaudet, S.; Varoquaux, A.; Penaranda, G.; Bannier, M.; Denizot, A. Impact of autofluorescence-based identification of parathyroids during total thyroidectomy on postoperative hypocalcemia: A before and after controlled study. Surgery 2018, 163, 23–30. [Google Scholar] [CrossRef]
- Dip, F.; Falco, J.; Verna, S.; Prunello, M.; Loccisano, M.; Quadri, P.; White, K.; Rosenthal, R. Randomized Controlled Trial Comparing White Light with Near-Infrared Autofluorescence for Parathyroid Gland Identification During Total Thyroidectomy. J. Am. Coll. Surg. 2019, 228, 744–751. [Google Scholar] [CrossRef]
- DiMarco, A.; Chotalia, R.; Bloxham, R.; McIntyre, C.; Tolley, N.; Palazzo, F.F. Autofluorescence in Parathyroidectomy: Signal Intensity Correlates with Serum Calcium and Parathyroid Hormone but Routine Clinical Use is Not Justified. World J. Surg. 2019, 43, 1532–1537. [Google Scholar] [CrossRef]
- Squires, M.H.; Jarvis, R.; Shirley, L.A.; Phay, J.E. Intraoperative Parathyroid Autofluorescence Detection in Patients with Primary Hyperparathyroidism. Ann. Surg. Oncol. 2019, 26, 1142–1148. [Google Scholar] [CrossRef]
- Wolf, H.W.; Grumbeck, B.; Runkel, N. Intraoperative verification of parathyroid glands in primary and secondary hyperparathyroidism using near-infrared autofluorescence (IOPA). Updates Surg. 2019, 71, 579–585. [Google Scholar] [CrossRef] [PubMed]
- Kose, E.; Kahramangil, B.; Aydin, H.; Donmez, M.; Berber, E. Heterogeneous and low-intensity parathyroid autofluorescence: Patterns suggesting hyperfunction at parathyroid exploration. Surgery 2019, 165, 431–437. [Google Scholar] [CrossRef] [PubMed]
- Squires, M.H.; Shirley, L.A.; Shen, C.; Jarvis, R.; Phay, J.E. Intraoperative Autofluorescence Parathyroid Identification in Patients with Multiple Endocrine Neoplasia Type 1. JAMA Otolaryngol. Head Neck Surg. 2019, 145, 897–902. [Google Scholar] [CrossRef] [PubMed]
- Konig, K.; Speicher, M.; Kohler, M.J.; Scharenberg, R.; Kaatz, M. Clinical application of multiphoton tomography in combination with high-frequency ultrasound for evaluation of skin diseases. J. Biophotonics 2010, 3, 759–773. [Google Scholar] [CrossRef]
- Koehler, M.J.; Preller, A.; Elsner, P.; Konig, K.; Hipler, U.C.; Kaatz, M. Non-invasive evaluation of dermal elastosis by in vivo multiphoton tomography with autofluorescence lifetime measurements. Exp. Dermatol. 2012, 21, 48–51. [Google Scholar] [CrossRef] [PubMed]
- Koehler, M.J.; Zimmermann, S.; Springer, S.; Elsner, P.; Konig, K.; Kaatz, M. Keratinocyte morphology of human skin evaluated by in vivo multiphoton laser tomography. Skin Res. Technol. 2011, 17, 479–486. [Google Scholar] [CrossRef]
- Kaatz, M.; Sturm, A.; Elsner, P.; Konig, K.; Buckle, R.; Koehler, M.J. Depth-resolved measurement of the dermal matrix composition by multiphoton laser tomography. Skin Res. Technol. 2010, 16, 131–136. [Google Scholar] [CrossRef]
- Koehler, M.J.; Kellner, K.; Kaatz, M.; Hipler, U.C. Epidermal changes during UVB phototherapy assessed by multiphoton laser tomography. Skin Res. Technol. 2016, 22, 437–442. [Google Scholar] [CrossRef]
- Koehler, M.J.; Kellner, K.; Hipler, U.C.; Kaatz, M. Acute UVB-induced epidermal changes assessed by multiphoton laser tomography. Skin Res. Technol. 2015, 21, 137–143. [Google Scholar] [CrossRef]
- Bazin, R.; Flament, F.; Colonna, A.; Le Harzic, R.; Buckle, R.; Piot, B.; Laize, F.; Kaatz, M.; Konig, K.; Fluhr, J.W. Clinical study on the effects of a cosmetic product on dermal extracellular matrix components using a high-resolution multiphoton tomograph. Skin Res. Technol. 2010, 16, 305–310. [Google Scholar] [CrossRef]
- Wang, S.; Zhao, J.; Lui, H.; He, Q.; Zeng, H. In vivo near-infrared autofluorescence imaging of pigmented skin lesions: Methods, technical improvements and preliminary clinical results. Skin Res. Technol. 2013, 19, 20–26. [Google Scholar] [CrossRef] [PubMed]
- Coda, S.; Siersema, P.D.; Stamp, G.W.; Thillainayagam, A.V. Biophotonic endoscopy: A review of clinical research techniques for optical imaging and sensing of early gastrointestinal cancer. Endosc. Int. Open 2015, 3, E380–E392. [Google Scholar] [CrossRef] [PubMed]
- Gibson, E.A.; Masihzadeh, O.; Lei, T.C.; Ammar, D.A.; Kahook, M.Y. Multiphoton microscopy for ophthalmic imaging. J. Ophthalmol. 2011, 2011, 870879. [Google Scholar] [CrossRef]
- Coda, S.; Thompson, A.J.; Kennedy, G.T.; Roche, K.L.; Ayaru, L.; Bansi, D.S.; Stamp, G.W.; Thillainayagam, A.V.; French, P.M.; Dunsby, C. Fluorescence lifetime spectroscopy of tissue autofluorescence in normal and diseased colon measured ex vivo using a fiber-optic probe. Biomed. Opt. Express 2014, 5, 515–538. [Google Scholar] [CrossRef]
- Duran-Sierra, E.; Cheng, S.; Cuenca-Martinez, R.; Malik, B.; Maitland, K.C.; Lisa Cheng, Y.S.; Wright, J.; Ahmed, B.; Ji, J.; Martinez, M.; et al. Clinical label-free biochemical and metabolic fluorescence lifetime endoscopic imaging of precancerous and cancerous oral lesions. Oral Oncol. 2020, 105, 104635. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Reilley, M.; Li, R.; Xu, M. Mapping absolute tissue endogenous fluorophore concentrations with chemometric wide-field fluorescence microscopy. J. Biomed. Opt. 2017, 22, 66009. [Google Scholar] [CrossRef]
- Chaturvedi, P.; Majumder, S.K.; Krishna, H.; Muttagi, S.; Gupta, P.K. Fluorescence spectroscopy for noninvasive early diagnosis of oral mucosal malignant and potentially malignant lesions. J. Cancer Res. Ther. 2010, 6, 497–502. [Google Scholar] [CrossRef]
- DaCosta, R.S.; Andersson, H.; Cirocco, M.; Marcon, N.E.; Wilson, B.C. Autofluorescence characterisation of isolated whole crypts and primary cultured human epithelial cells from normal, hyperplastic, and adenomatous colonic mucosa. J. Clin. Pathol. 2005, 58, 766–774. [Google Scholar] [CrossRef]
- Vida, C.; de Toda, I.M.; Cruces, J.; Garrido, A.; Gonzalez-Sanchez, M.; De la Fuente, M. Role of macrophages in age-related oxidative stress and lipofuscin accumulation in mice. Redox Biol. 2017, 12, 423–437. [Google Scholar] [CrossRef]
- Izuishi, K.; Tajiri, H.; Fujii, T.; Boku, N.; Ohtsu, A.; Ohnishi, T.; Ryu, M.; Kinoshita, T.; Yoshida, S. The histological basis of detection of adenoma and cancer in the colon by autofluorescence endoscopic imaging. Endoscopy 1999, 31, 511–516. [Google Scholar] [CrossRef]
- Wu, X.; Cai, J.; Zuo, Z.; Li, J. Collagen facilitates the colorectal cancer stemness and metastasis through an integrin/PI3K/AKT/Snail signaling pathway. Biomed. Pharmacother. 2019, 114, 108708. [Google Scholar] [CrossRef] [PubMed]
- De Veld, D.C.; Skurichina, M.; Witjes, M.J.; Duin, R.P.; Sterenborg, D.J.; Star, W.M.; Roodenburg, J.L. Autofluorescence characteristics of healthy oral mucosa at different anatomical sites. Lasers Surg. Med. 2003, 32, 367–376. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.; Kim, S.W.; Lee, K.D.; Ahn, Y.C. Phase-sensitive fluorescence detector for parathyroid glands during thyroidectomy: A preliminary report. J. Biophotonics 2020, 13, e201960078. [Google Scholar] [CrossRef] [PubMed]
- DaCosta, R.S.; Kulbatski, I.; Lindvere-Teene, L.; Starr, D.; Blackmore, K.; Silver, J.I.; Opoku, J.; Wu, Y.C.; Medeiros, P.J.; Xu, W.; et al. Point-of-Care Autofluorescence Imaging for Real-Time Sampling and Treatment Guidance of Bioburden in Chronic Wounds: First-in-Human Results. PLoS ONE 2015, 10, e0116623. [Google Scholar] [CrossRef] [PubMed]
- Richards-Kortum, R.; Sevick-Muraca, E. Quantitative optical spectroscopy for tissue diagnosis. Annu. Rev. Phys. Chem. 1996, 47, 555–606. [Google Scholar] [CrossRef]
- Fujimoto, D.; Akiba, K.-y.; Nakamura, N. Isolation and characterization of a fluorescent material in bovine achilles tendon collagen. Biochem. Biophys. Res. Commun. 1977, 76, 1124–1129. [Google Scholar] [CrossRef]
- Grimaud, J.A.; Druguet, M.; Peyrol, S.; Chevalier, O.; Herbage, D.; El Badrawy, N. Collagen immunotyping in human liver: Light and electron microscope study. J. Histochem. Cytochem. 1980, 28, 1145–1156. [Google Scholar] [CrossRef]
- Romer, T.J.; Fitzmaurice, M.; Cothren, R.M.; Richards-Kortum, R.; Petras, R.; Sivak, M.V., Jr.; Kramer, J.R., Jr. Laser-induced fluorescence microscopy of normal colon and dysplasia in colonic adenomas: Implications for spectroscopic diagnosis. Am. J. Gastroenterol. 1995, 90, 81–87. [Google Scholar]
- Georgakoudi, I.; Jacobson, B.C.; Muller, M.G.; Sheets, E.E.; Badizadegan, K.; Carr-Locke, D.L.; Crum, C.P.; Boone, C.W.; Dasari, R.R.; Van Dam, J.; et al. NAD(P)H and collagen as in vivo quantitative fluorescent biomarkers of epithelial precancerous changes. Cancer Res. 2002, 62, 682–687. [Google Scholar]
- Lin, B.; Urayama, S.; Saroufeem, R.M.; Matthews, D.L.; Demos, S.G. Characterizing the origin of autofluorescence in human esophageal epithelium under ultraviolet excitation. Opt. Express 2010, 18, 21074–21082. [Google Scholar] [CrossRef]
- Blomfield, J.; Farrar, J.F. The Fluorescent Properties of Maturing Arterial Elastin. Cardiovasc. Res. 1969, 3, 161–170. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Qu, J.Y. Autofluorescence spectroscopy of epithelial tissues. J. Biomed. Opt. 2006, 11, 054023. [Google Scholar] [CrossRef] [PubMed]
- Jayanthi, J.L.; Mallia, R.J.; Shiny, S.T.; Baiju, K.V.; Mathews, A.; Kumar, R.; Sebastian, P.; Madhavan, J.; Aparna, G.N.; Subhash, N. Discriminant analysis of autofluorescence spectra for classification of oral lesions in vivo. Lasers Surg. Med. 2009, 41, 345–352. [Google Scholar] [CrossRef] [PubMed]
- Chance, B.; Baltscheffsky, H. Respiratory enzymes in oxidative phosphorylation. VII. Binding of intramitochondrial reduced pyridine nucleotide. J. Biol. Chem. 1958, 233, 736–739. [Google Scholar]
- Villette, S.; Pigaglio-Deshayes, S.; Vever-Bizet, C.; Validire, P.; Bourg-Heckly, G. Ultraviolet-induced autofluorescence characterization of normal and tumoral esophageal epithelium cells with quantitation of NAD(P)H. Photochem. Photobiol. Sci.: Off. J. Eur. Photochem. Assoc. Eur. Soc. Photobiol. 2006, 5, 483–492. [Google Scholar] [CrossRef]
- Barile, M.; Giancaspero, T.A.; Brizio, C.; Panebianco, C.; Indiveri, C.; Galluccio, M.; Vergani, L.; Eberini, I.; Gianazza, E. Biosynthesis of flavin cofactors in man: Implications in health and disease. Curr. Pharm. Des. 2013, 19, 2649–2675. [Google Scholar] [CrossRef]
- Galbán, J.; Sanz-Vicente, I.; Navarro, J.; de Marcos, S. The intrinsic fluorescence of FAD and its application in analytical chemistry: A review. Methods Appl. Fluoresc. 2016, 4, 042005. [Google Scholar] [CrossRef]
- Konig, K.; Flemming, G.; Hibst, R. Laser-induced autofluorescence spectroscopy of dental caries. Cell Mol. Biol. (Noisy-le-grand) 1998, 44, 1293–1300. [Google Scholar]
- Moesta, K.T.; Ebert, B.; Handke, T.; Nolte, D.; Nowak, C.; Haensch, W.E.; Pandey, R.K.; Dougherty, T.J.; Rinneberg, H.; Schlag, P.M. Protoporphyrin IX occurs naturally in colorectal cancers and their metastases. Cancer Res. 2001, 61, 991–999. [Google Scholar]
- Wolman, M. Lipid pigments (chromolipids): Their origin, nature, and significance. Pathobiol. Annu. 1980, 10, 253–267. [Google Scholar]
- Monnier, V.M.; Vishwanath, V.; Frank, K.E.; Elmets, C.A.; Dauchot, P.; Kohn, R.R. Relation between complications of type I diabetes mellitus and collagen-linked fluorescence. N. Engl. J. Med. 1986, 314, 403–408. [Google Scholar] [CrossRef] [PubMed]
- Tuuminen, R.; Tuulonen, A.; Kaarniranta, K. The Finnish national guideline for diagnosis, treatment and follow-up of patients with wet age-related macular degeneration. Acta Ophthalmol. 2017, 95, 649–650. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Hart, K.M.; Abbott, C.; Ly, A.; Kalff, S.; Lek, J.J.; Milston, R.; Page, G.; Robertson, B.; Ayton, L. Optometry Australia’s chairside reference for the diagnosis and management of age-related macular degeneration. Clin. Exp. Optom. 2019, 103, 254–264. [Google Scholar] [CrossRef] [PubMed]
- S3-Leitlinie Diagnostik und Therapie des Mundhöhlenkarzinoms, Langversion 3.01 (Konsultationsfassung). Available online: https://www.leitlinienprogramm-onkologie.de/leitlinien/mundhoehlenkarzinom/ (accessed on 5 February 2020).
- Lingen, M.W.; Abt, E.; Agrawal, N.; Chaturvedi, A.K.; Cohen, E.; D’Souza, G.; Gurenlian, J.; Kalmar, J.R.; Kerr, A.R.; Lambert, P.M.; et al. Evidence-based clinical practice guideline for the evaluation of potentially malignant disorders in the oral cavity: A report of the American Dental Association. J. Am. Dent. Assoc. 2017, 148, 712–727. [Google Scholar] [CrossRef] [PubMed]
- Macey, R.; Walsh, T.; Brocklehurst, P.; Kerr, A.R.; Liu, J.L.; Lingen, M.W.; Ogden, G.R.; Warnakulasuriya, S.; Scully, C. Diagnostic tests for oral cancer and potentially malignant disorders in patients presenting with clinically evident lesions. Cochrane Database Syst. Rev. 2015, 5, 1465–1858. [Google Scholar] [CrossRef]
- Haugen, B.R.; Alexander, E.K.; Bible, K.C.; Doherty, G.M.; Mandel, S.J.; Nikiforov, Y.E.; Pacini, F.; Randolph, G.W.; Sawka, A.M.; Schlumberger, M.; et al. 2015 American Thyroid Association Management Guidelines for Adult Patients with Thyroid Nodules and Differentiated Thyroid Cancer: The American Thyroid Association Guidelines Task Force on Thyroid Nodules and Differentiated Thyroid Cancer. Thyroid 2016, 26, 1–133. [Google Scholar] [CrossRef]
- Updated S3-Guideline Ulcerative Colitis. German Society for Digestive and Metabolic Diseases (DGVS). Z. Gastroenterol. 2019, 57, 162–241. [Google Scholar] [CrossRef]
- Matsuoka, K.; Kobayashi, T.; Ueno, F.; Matsui, T.; Hirai, F.; Inoue, N.; Kato, J.; Kobayashi, K.; Kobayashi, K.; Koganei, K.; et al. Evidence-based clinical practice guidelines for inflammatory bowel disease. J. Gastroenterol. 2018, 53, 305–353. [Google Scholar] [CrossRef]
- S3-Leitlinie Kolorektales Karzinom. Available online: https://www.awmf.org/uploads/tx_szleitlinien/021-007OLl_S3_Kolorektales-Karzinom-KRK_2019-01.pdf (accessed on 11 February 2020).
- Croce, A.C.; Bottiroli, G.; Di Pasqua, L.G.; Berardo, C.; Siciliano, V.; Rizzo, V.; Vairetti, M.; Ferrigno, A. Serum and Hepatic Autofluorescence as a Real-Time Diagnostic Tool for Early Cholestasis Assessment. Int. J. Mol. Sci. 2018, 19. [Google Scholar] [CrossRef]
- Croce, A.C.; Ferrigno, A.; Bottiroli, G.; Vairetti, M. Autofluorescence-based optical biopsy: An effective diagnostic tool in hepatology. Liver Int. 2018, 38, 1160–1174. [Google Scholar] [CrossRef]
- Butler, D. Translational research: Crossing the valley of death. Nature 2008, 453, 840–842. [Google Scholar] [CrossRef] [PubMed]
- Krauss, E.; Agaimy, A.; Douplik, A.; Albrecht, H.; Neumann, H.; Hartmann, A.; Hohenstein, R.; Raithel, M.; Hahn, E.G.; Neurath, M.F.; et al. Normalized autofluorescence imaging diagnostics in upper GI tract: A new method to improve specificity in neoplasia detection. Int. J. Clin. Exp. Pathol. 2012, 5, 956–964. [Google Scholar] [CrossRef] [PubMed]
- Quang, T.; Tran, E.Q.; Schwarz, R.A.; Williams, M.D.; Vigneswaran, N.; Gillenwater, A.M.; Richards-Kortum, R. Prospective Evaluation of Multimodal Optical Imaging with Automated Image Analysis to Detect Oral Neoplasia In Vivo. Cancer Prev. Res. (Phila.) 2017, 10, 563–570. [Google Scholar] [CrossRef] [PubMed]
- Schwendicke, F.; Elhennawy, K.; Paris, S.; Friebertshauser, P.; Krois, J. Deep learning for caries lesion detection in near-infrared light transillumination images: A pilot study. J. Dent. 2020, 92, 103260. [Google Scholar] [CrossRef] [PubMed]
- Aihara, H.; Saito, S.; Inomata, H.; Ide, D.; Tamai, N.; Ohya, T.R.; Kato, T.; Amitani, S.; Tajiri, H. Computer-aided diagnosis of neoplastic colorectal lesions using ‘real-time’ numerical color analysis during autofluorescence endoscopy. Eur. J. Gastroenterol. Hepatol. 2013, 25, 488–494. [Google Scholar] [CrossRef]
- Inomata, H.; Tamai, N.; Aihara, H.; Sumiyama, K.; Saito, S.; Kato, T.; Tajiri, H. Efficacy of a novel auto-fluorescence imaging system with computer-assisted color analysis for assessment of colorectal lesions. World J. Gastroenterol. 2013, 19, 7146–7153. [Google Scholar] [CrossRef]
- Ahdi, M.; Gerdes, V.E.; Graaff, R.; Kuipers, S.; Smit, A.J.; Meesters, E.W. Skin autofluorescence and complications of diabetes: Does ethnic background or skin color matter? Diabetes Technol. Ther. 2015, 17, 88–95. [Google Scholar] [CrossRef]
- Ahmad, M.S.; Kimhofer, T.; Ahmad, S.; AlAma, M.N.; Mosli, H.H.; Hindawi, S.I.; Mook-Kanamori, D.O.; Sebekova, K.; Damanhouri, Z.A.; Holmes, E. Ethnicity and skin autofluorescence-based risk-engines for cardiovascular disease and diabetes mellitus. PLoS ONE 2017, 12, e0185175. [Google Scholar] [CrossRef]
- Pol, H.W.; Sibma, E.; Zeebregts, C.J.; Pierik, E.G.; Meerwaldt, R. Increased skin autofluorescence after colorectal operation reflects surgical stress and postoperative outcome. Am. J. Surg. 2011, 202, 583–589. [Google Scholar] [CrossRef]
- Sturm, D.; Marselli, L.; Ehehalt, F.; Richter, D.; Distler, M.; Kersting, S.; Grutzmann, R.; Bokvist, K.; Froguel, P.; Liechti, R.; et al. Improved protocol for laser microdissection of human pancreatic islets from surgical specimens. J. Vis. Exp. 2013, 71, e50231. [Google Scholar] [CrossRef]
- Kamath, S.D.; Bhat, R.A.; Ray, S.; Mahato, K.K. Autofluorescence of normal, benign, and malignant ovarian tissues: A pilot study. Photomed. Laser Surg. 2009, 27, 325–335. [Google Scholar] [CrossRef] [PubMed]
- Das, K.; Stone, N.; Kendall, C.; Fowler, C.; Christie-Brown, J. Raman spectroscopy of parathyroid tissue pathology. Lasers Med. Sci. 2006, 21, 192–197. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.V.; Tsao-Wei, D.D.; Xiong, S.; Groshen, S.; Dorff, T.B.; Quinn, D.I.; Tai, Y.C.; Engel, J.; Hawes, D.; Schally, A.V.; et al. Phase I, dose-escalation study of the targeted cytotoxic LHRH analog AEZS-108 in patients with castration-and taxane-resistant prostate cancer. Clin. Cancer Res. 2014, 20, 6277–6283. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.S.; Athreya, K.; Liu, S.V.; Schally, A.V.; Tsao-Wei, D.; Groshen, S.; Quinn, D.I.; Dorff, T.B.; Xiong, S.; Engel, J.; et al. A Phase II Trial of AEZS-108 in Castration- and Taxane-Resistant Prostate Cancer. Clin. Genitourin. Cancer 2017, 15, 742–749. [Google Scholar] [CrossRef]
- Keller, M.D.; Majumder, S.K.; Kelley, M.C.; Meszoely, I.M.; Boulos, F.I.; Olivares, G.M.; Mahadevan-Jansen, A. Autofluorescence and diffuse reflectance spectroscopy and spectral imaging for breast surgical margin analysis. Lasers Surg. Med. 2010, 42, 15–23. [Google Scholar] [CrossRef]
- Bochenek, K.; Aebisher, D.; Miedzybrodzka, A.; Cieslar, G.; Kawczyk-Krupka, A. Methods for bladder cancer diagnosis—The role of autofluorescence and photodynamic diagnosis. Photodiagn. Photodyn. Ther. 2019, 27, 141–148. [Google Scholar] [CrossRef]
- Varanius, D.; Terbetas, G.; Vaitkus, J.V.; Vaitkuviene, A. Spinal hernia tissue autofluorescence spectrum. Lasers Med. Sci. 2013, 28, 423–430. [Google Scholar] [CrossRef]
- Najari, B.B.; Ramasamy, R.; Sterling, J.; Aggarwal, A.; Sheth, S.; Li, P.S.; Dubin, J.M.; Goldenberg, S.; Jain, M.; Robinson, B.D.; et al. Pilot study of the correlation of multiphoton tomography of ex vivo human testis with histology. J. Urol. 2012, 188, 538–543. [Google Scholar] [CrossRef]
- Liberati, A.; Altman, D.G.; Tetzlaff, J.; Mulrow, C.; Gotzsche, P.C.; Ioannidis, J.P.; Clarke, M.; Devereaux, P.J.; Kleijnen, J.; Moher, D. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: Explanation and elaboration. J. Clin. Epidemiol. 2009, 62, e1–e34. [Google Scholar] [CrossRef]
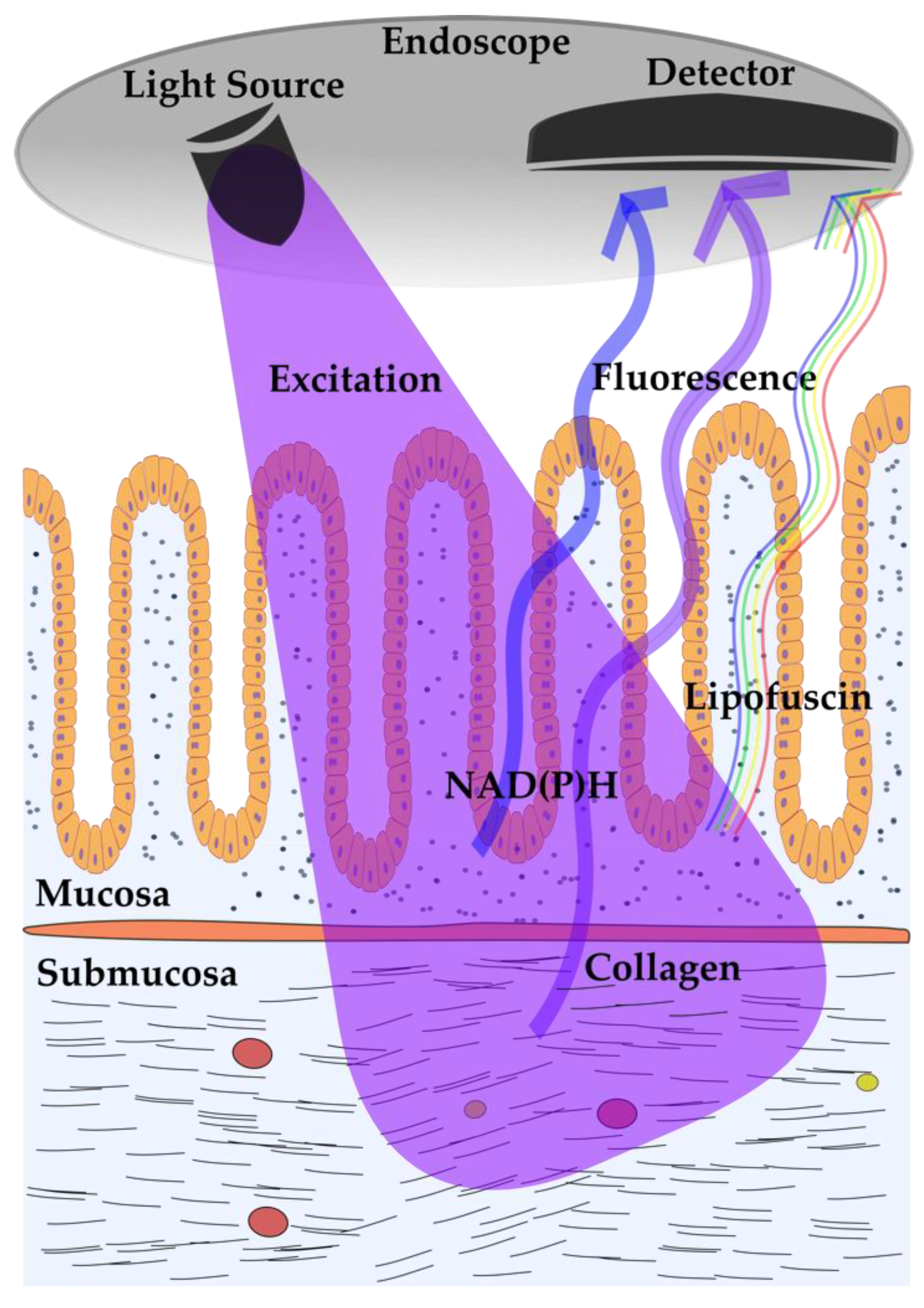




| Clinical Specialty | Excitation Wavelength Ranges | Clinical Imaging Procedure |
|---|---|---|
| Diseases with impaired redox homeostasis (e.g., diabetology, nephrology) | 300−420 nm (ultraviolet light, peak at 370 nm) | Non-invasive skin tissue imaging |
| Oral medicine | 400−460 nm (blue light) | Non-invasive oral imaging |
| Gastroenterology | 390−470 nm (blue light) | Endoscopy |
| Pulmonology | 488 nm (green light) | Endomicroscopy |
| Dentistry | 655 or 780 nm (red or near-infrared light) | Caries screening |
| Thyroid surgery | 690−770 nm (near-infrared light) | Intraoperative imaging |
| Dermatology | 750−850 nm (multiphoton principle) | Multiphoton imaging |
| Fluorophore | Function | Excitation (nm) | Emission (nm) | Diagnostic Value, Esp. Changes Depending on Progress of Disease | References |
|---|---|---|---|---|---|
| Collagen | Structural protein in ECM | 330–340 | 400–410 | Altered in neoplastic lesions or fibrotic states, e.g., in liver fibrosis | [146,147,148,149,150] |
| Elastin | Structural protein in ECM | 350–420 | 420–510 | Altered expression in invasive tumors | [151] |
| Keratin | Structural protein in ECM | 355–405 | 420–480 | Altered expression in invasive tumors, delineate tumor borders | [152,153] |
| NAD(P)H | Cofactor in redox reactions | 330–380 | 440 (bound); 462 (unbound) | Biomarkers of energy metabolism and redox state | [149,150,154,155] |
| FAD | Cofactor in redox reactions | 440–450 | 525 | Biomarker of energy metabolism and redox state e.g., in cancer formation | [85,156,157] |
| Porphyrin | Formation of heme | 405 | 630–700 | Altered in dental caries and neoplastic lesions | [158,159] |
| Lipofuscin | End product of lysosomal digestion | 400–500 | 480–700 | Biomarker of degenerative diseases | [160] |
| AGEs | Metabolic by-products | 300–420 | 420–600 | Accumulate with age and progressive degenerative diseases | [38,39,161] |
| Amino acids, e.g., Tyrosin, Tryptophan | Protein metabolism | <310 | >500 | Altered abundance in invasive tumors | [81,150] |
| Calcium-sensing receptor | Regulation of parathyroid hormone (PTH) secretion | 785 | 822 | Postulated candidate fluorophore for parathyroid AF, distinguishing between parathyroid and surrounding tissues | [98] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wizenty, J.; Schumann, T.; Theil, D.; Stockmann, M.; Pratschke, J.; Tacke, F.; Aigner, F.; Wuensch, T. Recent Advances and the Potential for Clinical Use of Autofluorescence Detection of Extra-Ophthalmic Tissues. Molecules 2020, 25, 2095. https://doi.org/10.3390/molecules25092095
Wizenty J, Schumann T, Theil D, Stockmann M, Pratschke J, Tacke F, Aigner F, Wuensch T. Recent Advances and the Potential for Clinical Use of Autofluorescence Detection of Extra-Ophthalmic Tissues. Molecules. 2020; 25(9):2095. https://doi.org/10.3390/molecules25092095
Chicago/Turabian StyleWizenty, Jonas, Teresa Schumann, Donna Theil, Martin Stockmann, Johann Pratschke, Frank Tacke, Felix Aigner, and Tilo Wuensch. 2020. "Recent Advances and the Potential for Clinical Use of Autofluorescence Detection of Extra-Ophthalmic Tissues" Molecules 25, no. 9: 2095. https://doi.org/10.3390/molecules25092095
APA StyleWizenty, J., Schumann, T., Theil, D., Stockmann, M., Pratschke, J., Tacke, F., Aigner, F., & Wuensch, T. (2020). Recent Advances and the Potential for Clinical Use of Autofluorescence Detection of Extra-Ophthalmic Tissues. Molecules, 25(9), 2095. https://doi.org/10.3390/molecules25092095







