Redox Potential and Antioxidant Capacity of Bovine Bone Collagen Peptides towards Stable Free Radicals, and Bovine Meat Lipids and Proteins. Effect of Animal Age, Bone Anatomy and Proteases—A Step Forward towards Collagen-Rich Tissue Valorisation
Abstract
1. Introduction
2. Results
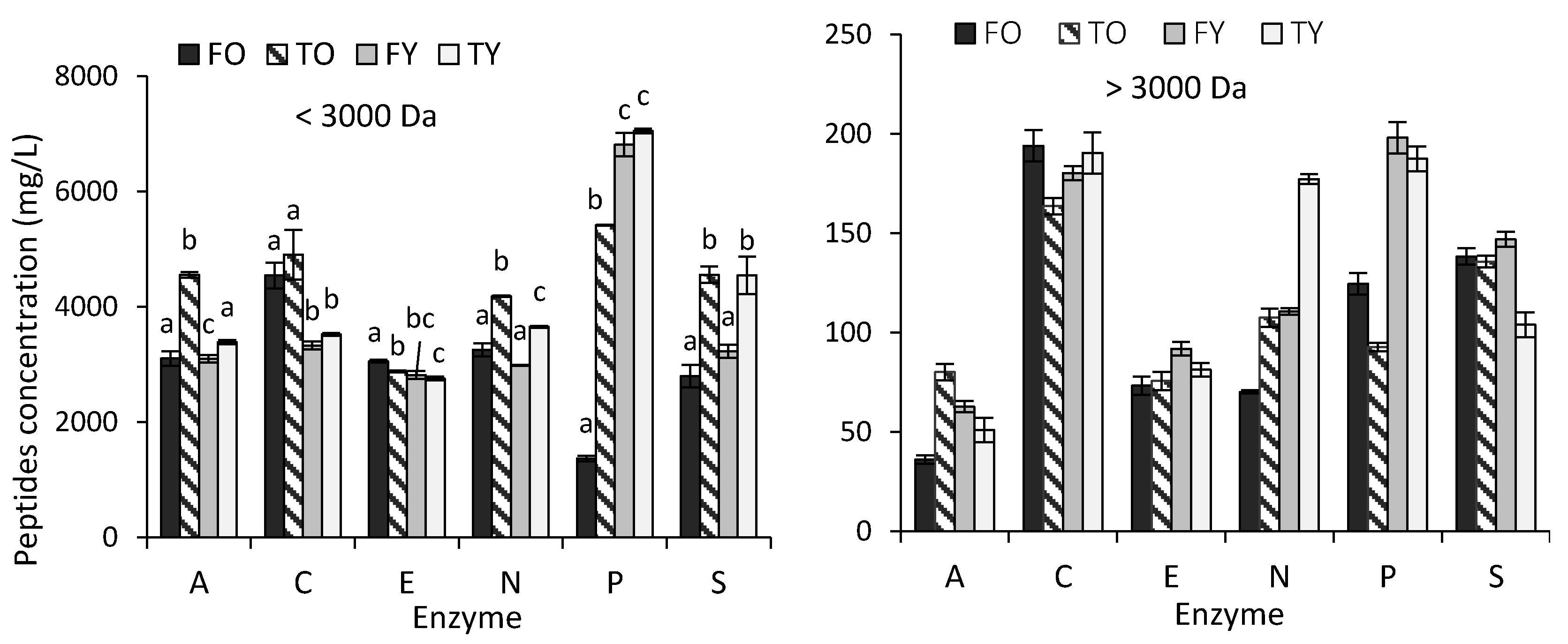
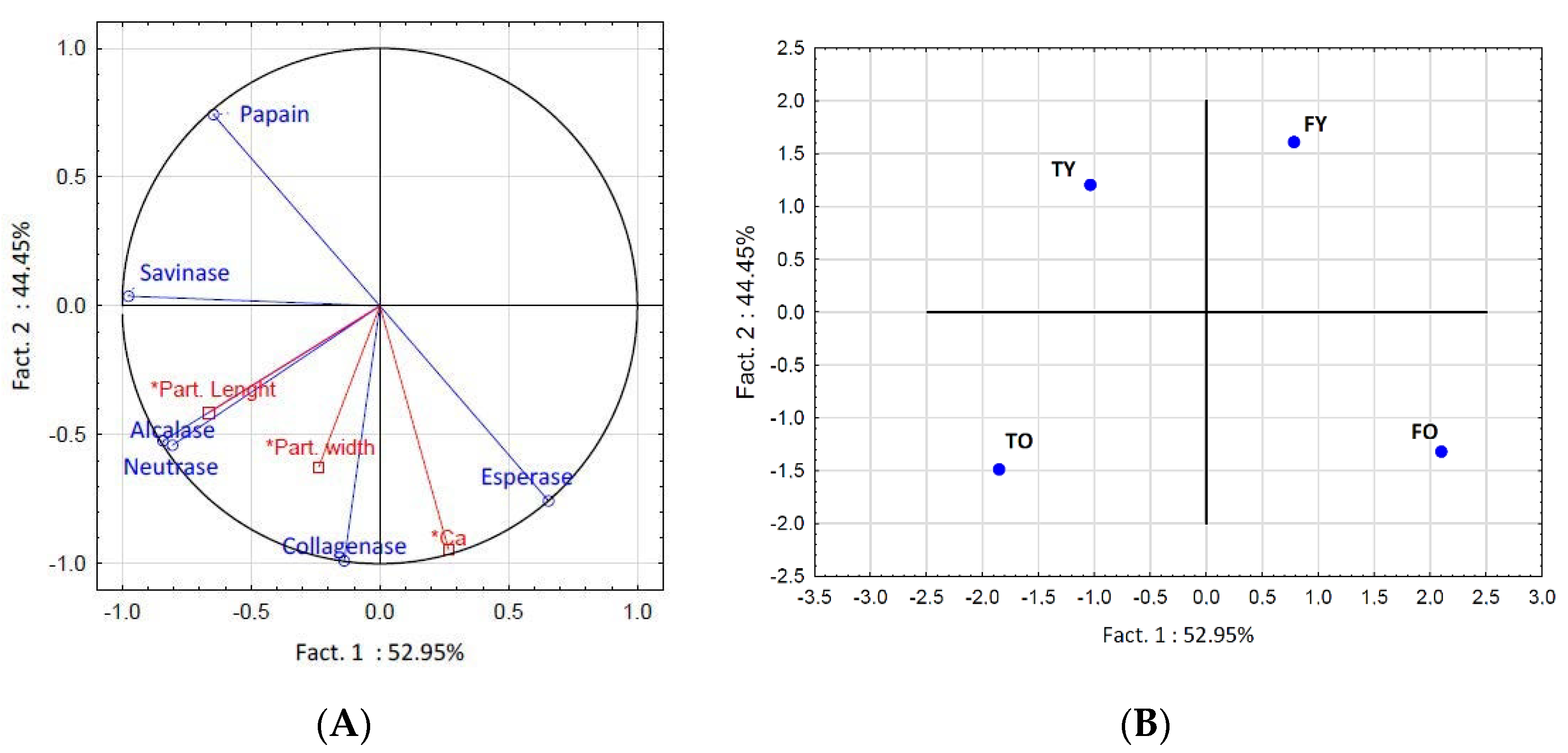
2.1. Effect of Bone Age and Anatomy, and Enzymes, on the Amount of Collagen Peptides Released
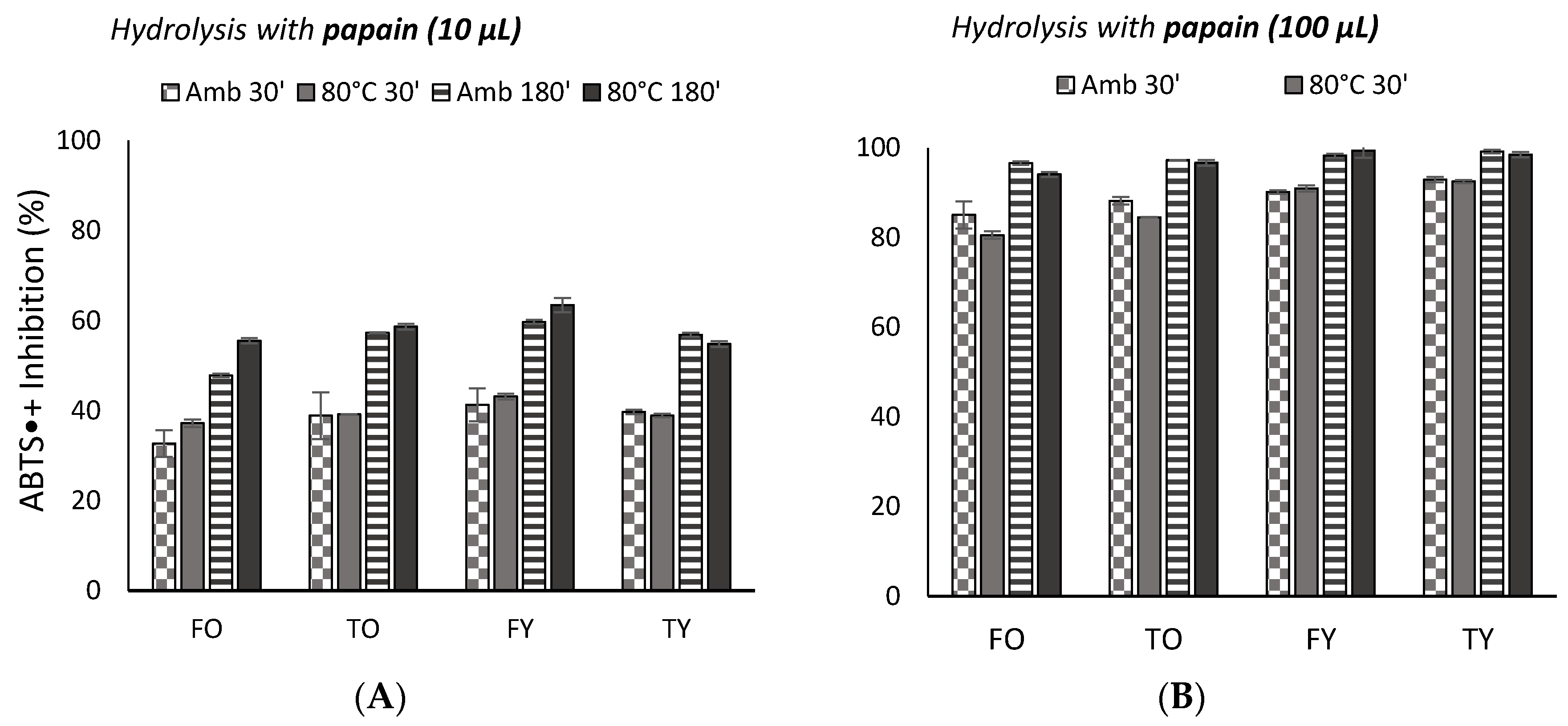
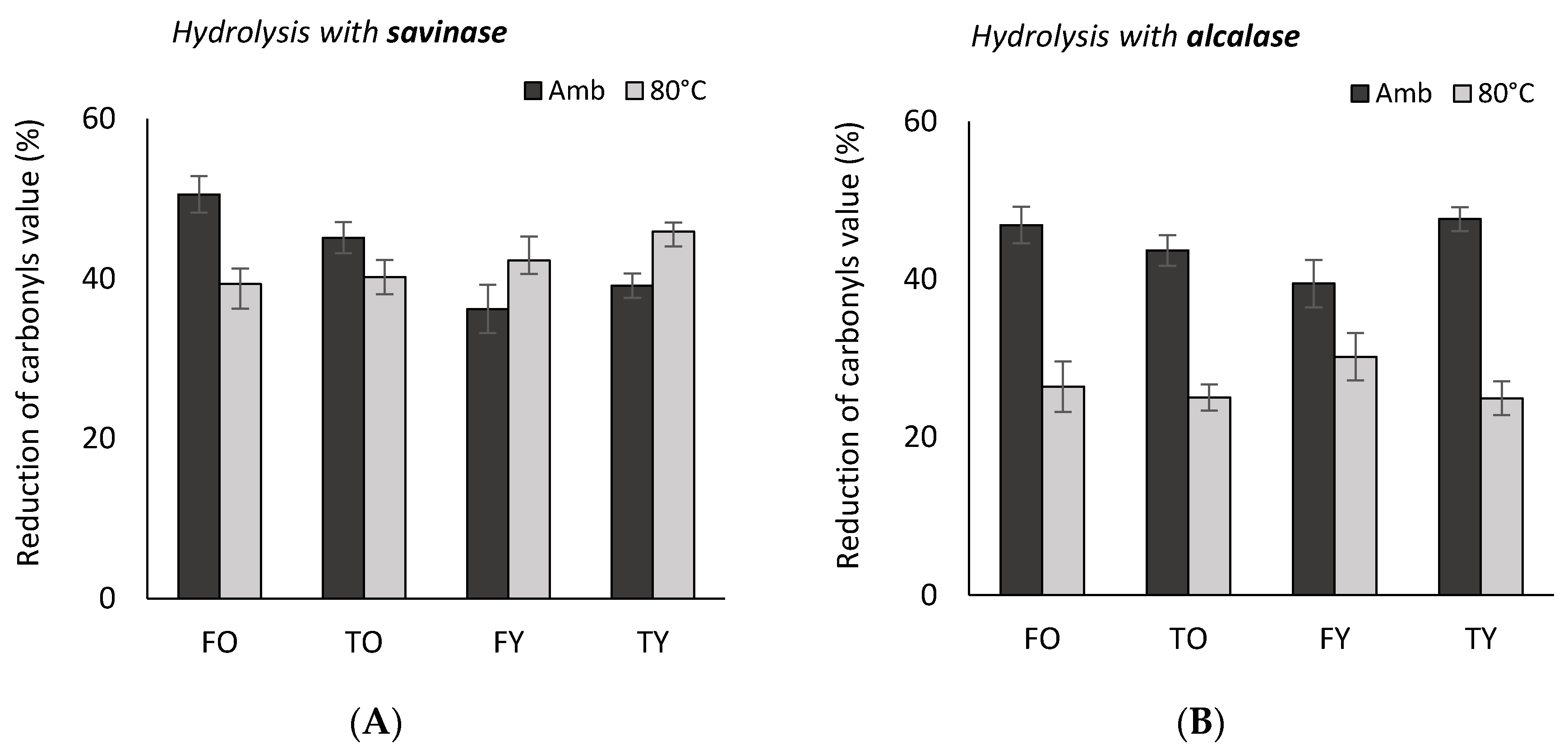
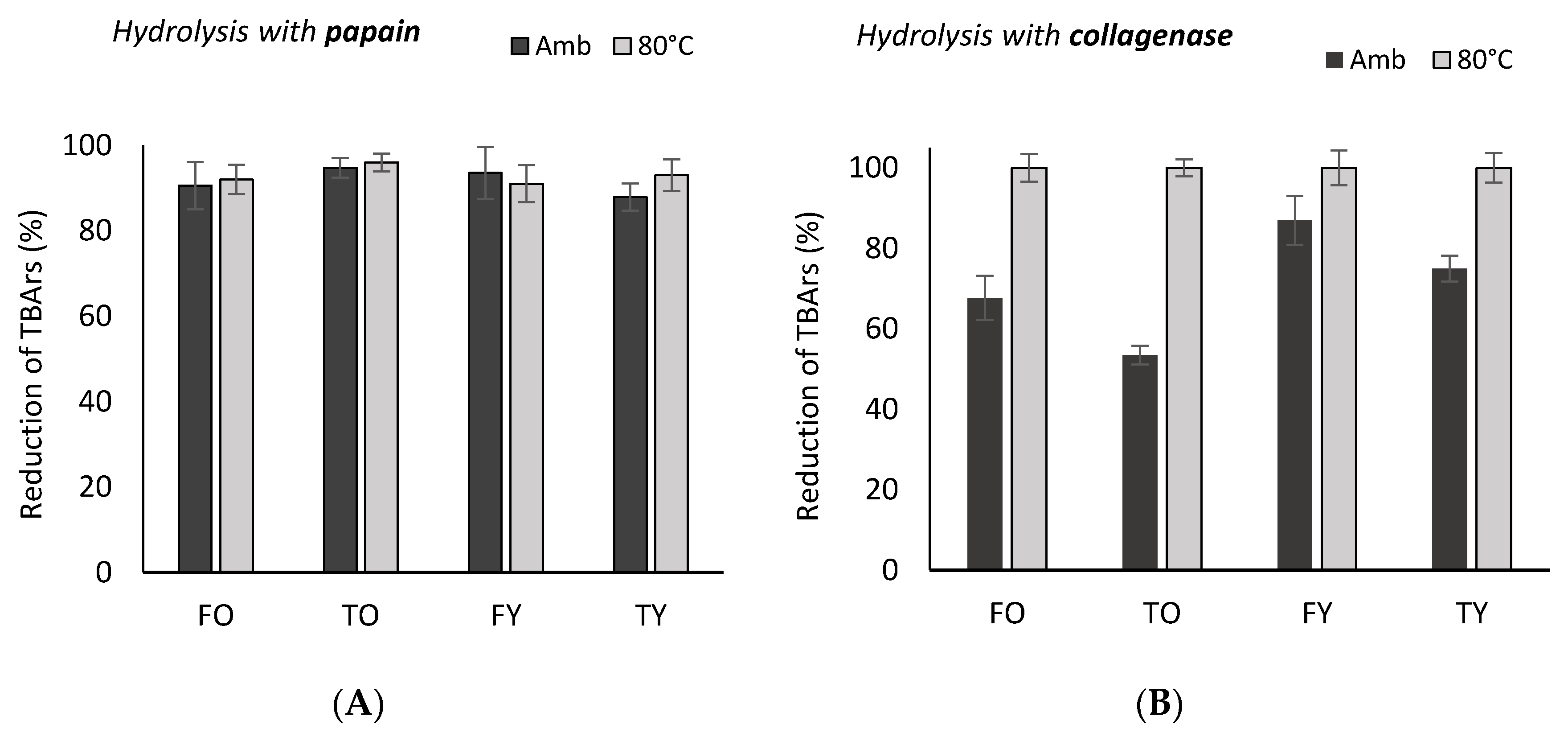
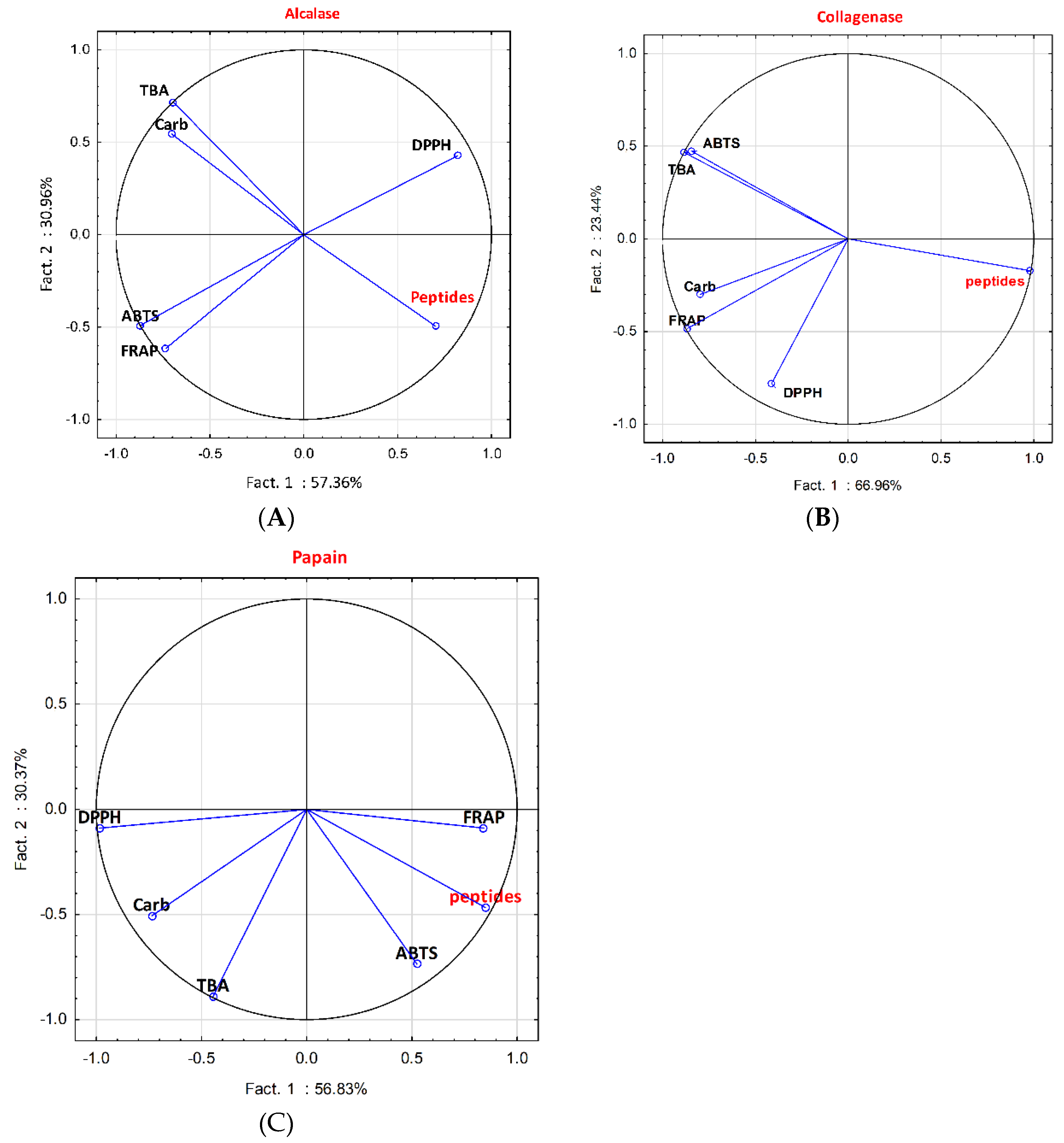
2.2. Antioxidant Activity
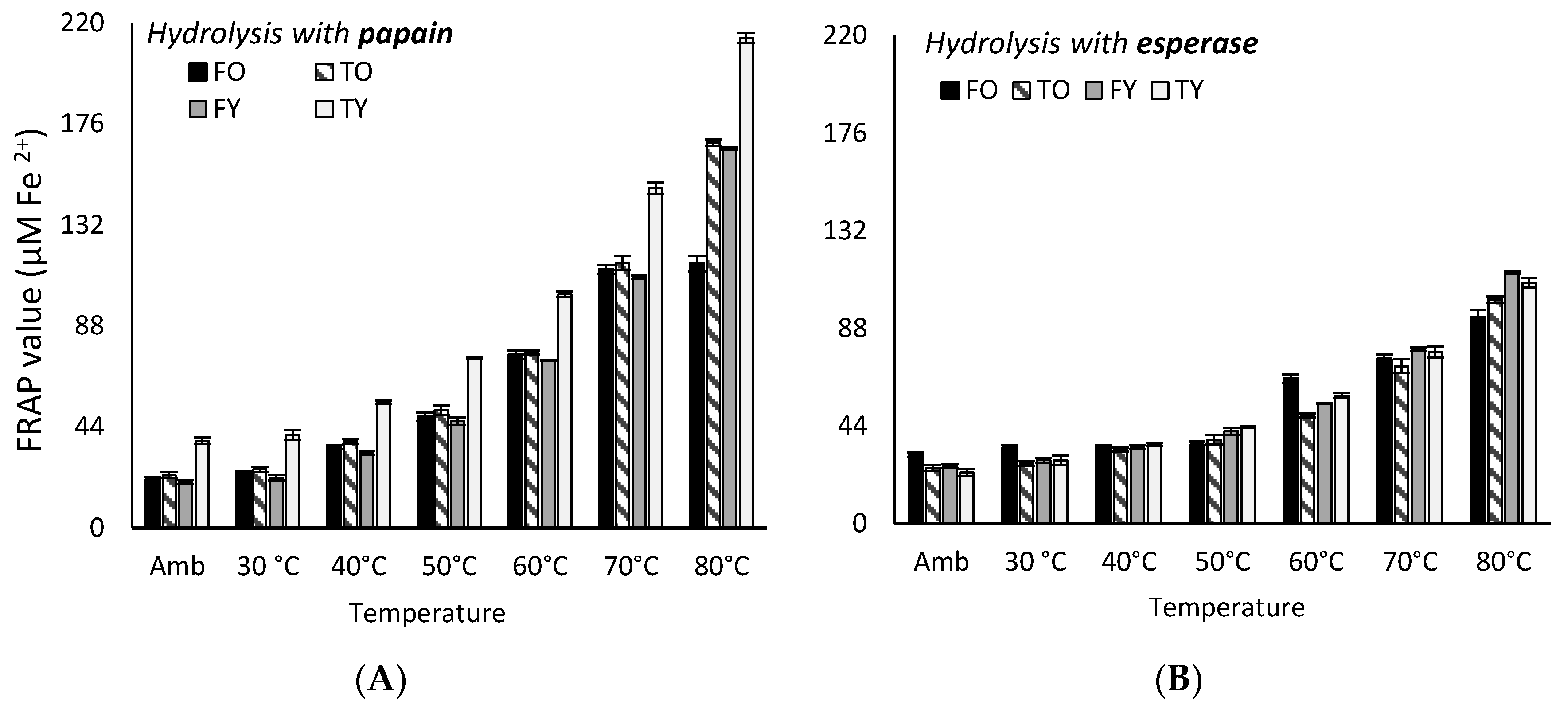
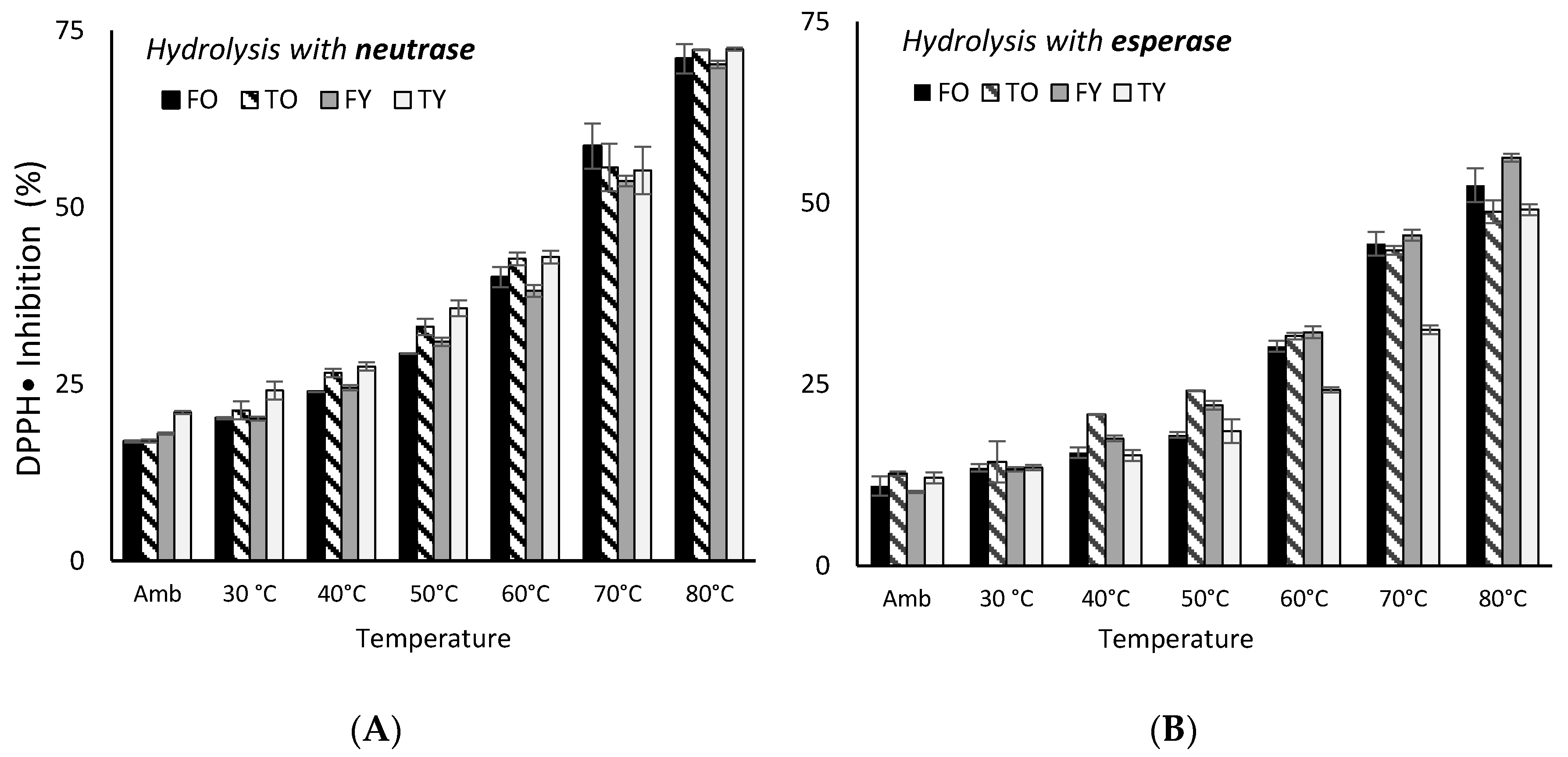
2.3. Kinetics of FRAP and DPPH Assays Antioxidant Activity Under Heating
3. Discussion
3.1. Effect of Bone Age and Anatomy, and Enzymes, on the Amount of Collagen Peptides Released
3.2. Antioxidant Activity
3.3. Kinetics of FRAP and DPPH Antioxidant Activity Under Heating
3.4. Summary and Future Research
4. Materials and Methods
4.1. Bone Samples and Extraction of Collagen
4.2. Scanning Electron Microscopy of Collagen Powders, and Fourier Transform Infrared (FTIR) Spectroscopy and Particle Size Analysis of Collagen in Solution
4.3. Hydrolysis of Collagen
4.4. Collagen Peptides Quantification
4.5. Antioxidant Activity and Effect of Heating
4.5.1. ABTS, DPPH and FRAP Assays
4.5.2. TBArs and Carbonyls Determination
4.6. Statistical Analysis and Experimental Data Modelling
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Gómez-Guillén, M.C.; Giménez, B.; López-Caballero, M.E.; Monteiro, M.P. Functional and bioactive properties of collagen and gelatin from alternative sources: A Review. Food Hydrocoll. 2011, 25, 1813–1827. [Google Scholar] [CrossRef]
- Liu, D.; Nikoo, M.; Boran, G.; Zhou, P.; Regenstein, J.M. Collagen and Gelatin. Annu. Rev. Food Sci. Technol. 2015, 6, 527–557. [Google Scholar] [CrossRef]
- León-López, A.; Morales-Peñaroza, A.; Martínez-Juárez, V.M.; Vargas-Torres, A.; Zeugolis, D.I.; Aguirre-Alvarez, G. Hydrolized collagen—Sources and Applications. Molecules 2019, 24, 4031. [Google Scholar] [CrossRef] [PubMed]
- Di Bernardini, R.; Harnedy, P.; Bolton, D.; Kerry, J.; O’Neill, E.; Mullen, A.M.; Hayes, M. Antioxidant and antimicrobial peptidic hydrolysates from muscle protein sources and by-product. Food Chem. 2011, 124, 1296–1307. [Google Scholar] [CrossRef]
- Lorenzo, J.M.; Munekata, P.E.S.; Gómez, B.; Barba, F.; Mora, L.; Pérez-Santaescolástica, C.; Toldrá, F. Bioactive peptides as natural antioxidants in food products—A review. Trends Food Sci. Technol. 2018, 79, 136–147. [Google Scholar] [CrossRef]
- European Commission. Best Available Techniques in the Slaughterhouses and Animal By-Products Industries. EIPPCB, Brussels. 2005. Available online: https://eippcb.jrc.ec.europa.eu/sites/default/files/2020-01/sa_bref_0505.pdf (accessed on 20 September 2020).
- Ferraro, V.; Anton, M.; Santé-Lhoutellier, V. The « sisters » α-helices of collagen, elastin and keratin recovered from animal by-products: Functionality, bioactivity and trends of applications. Trends Food Sci. Technol. 2016, 51, 65–75. [Google Scholar] [CrossRef]
- Ben Henda, Y.; Bordenave-Juchereau, S. Using marine cryptides against metabolic syndrome. In Bioactive Compounds from Marine Foods: Plant. and Animal Sources; Hérnandez-Ledesma, B., Herrero, M., Eds.; Wiley: London, UK, 2013; pp. 95–112. [Google Scholar]
- Young, I.S.; Woodside, J.V. Antioxidant in health and disease. J. Clin. Phatol. 2001, 54, 176–186. [Google Scholar] [CrossRef]
- Oliver, C.N.; Ahn, B.W.; Moermant, E.J.; Goldstein, S.; Stadtman, E.R. Age-related changes in oxidized proteins. J. Biol. Chem. 1987, 262, 5488–5491. [Google Scholar]
- Lynch, S.M.; Frei, B. Mechanisms of copper- and iron-dependent oxidative modification of human low-density lipoprotein. J. Lipid Res. 1993, 34, 1745–1753. [Google Scholar]
- Ferraro, V.; Gaillard-Martinie, B.; Sayd, T.; Chambon, C.; Anton, M.; Véronique Santé-Lhoutellier, V. Collagen type I from bovine bone. Effect of animal age, bone anatomy and drying methodology on extraction yield, self-assembly, thermal behaviour and electrokinetic potential. Int. J. Biol. Macrom. 2017, 97, 55–66. [Google Scholar] [CrossRef]
- Boskey, A.L.; Coleman, R. Aging and Bone. J. Dent. Res. 2010, 89, 1333–1348. [Google Scholar] [CrossRef] [PubMed]
- Legros, R.; Balmain, N.; Bonel, G. Age-related changes in mineral of rat and cortical bones. Calcif. Tissue Int. 1987, 41, 137–144. [Google Scholar] [CrossRef] [PubMed]
- Smith, P.K.; Krohn, R.I.; Hermanson, G.T.; Mallia, A.K.; Gartner, F.H.; Provenzano, M.D.; Fujimoto, E.K.; Goeke, N.M.; Olson, B.J.; Klenk, D.C. Measurement of protein using bicinchoninic acid. Anal. Biochem. 1985, 150, 76–85. [Google Scholar] [CrossRef]
- Carlsen, M.H.; Halvorsen, B.L.; Holte, K.; Bøhn, S.K.; Dragland, S.; Sampson, L.; Willey, C.; Senoo, H.; Humezono, Y.; Sanada, C.; et al. The total antioxidant content of more than 3100 foods, beverages, spices, herbs and supplements used worldwide. Nutr. J. 2010, 9, 3. [Google Scholar] [CrossRef]
- Halvorsen, B.L.; Carlsen, M.H.; Phillips, K.M.; Bøhn, S.K.; Holte, K.; Jacobs, D.R., Jr.; Blomhoff, R. Content of redox-active compounds (ie, antioxidants) in food consumed in the United States. Am. J. Clin. Nutr. 2006, 135, 84–95. [Google Scholar]
- Vargas-Bello-Pérez, E.; Màrquez-Hernàndez, R.I.; Henràndez-Castellano, L.E. Bioactive peptides from milk: Animal determinants and their implications in human health. J. Dairy Res. 2019, 86, 136–144. [Google Scholar] [CrossRef]
- Apak, R.; Özyürek, M.; Güçlü, K.; Çapanoğlu, E. Antioxidant activity/capacity measurements. II. Hydrogen atom transfer (HAT)-based, mixed mode (Electron transfer (ET)/HAT), and lipid peroxidation assays. J. Agric. Food Chem. 2016, 64, 1028–1045. [Google Scholar] [CrossRef]
- Mishra, K.; Ojha, H.; Chandhury, N.K. Estimation of antiradical properties of antioxidants using DPPH• assay: A critical review and results. Food Chem. 2012, 130, 1036–1043. [Google Scholar] [CrossRef]
- Tian, X.; Schaich, K.M. Effect of molecular structure on kinetics and dynamics of the trolox equivalent antioxidant capacity assay with ABTS+•. J. Agric. Food Chem. 2013, 61, 5511–5519. [Google Scholar] [CrossRef]
- Piatkivsky, A.; Osburn, S.; Jaderberg, K.; Grzetic, J.; Steill, J.D.; Oomens, J.; Zhao, J.; Kai-Chi Lau, J.K.; Verkerk, U.H.; Hopkinson, A.C.; et al. Structure and reactivity of the distonic and aromatic radical cations of tryptophan. J. Am. Mass Spectrom. 2013, 24, 513–523. [Google Scholar] [CrossRef]
- Reznik, A.Z.; Packer, L. Oxidative damage to proteins: Spectrophotometric method for carbonyl assay. Methods Enzymol. 1994, 233, 357–363. [Google Scholar]
- Lü, J.M.; Lin, P.H.; Yao, Q.; Chen, C. Chemical and molecular mechanisms of antioxidants: Experimental approaches and model systems. J. Cell. Mol. Med. 2010, 14, 840–860. [Google Scholar] [CrossRef] [PubMed]
- Bet, L.; Calabrese, E.; Bava, L.; Magni, E.; Rapuzzi, S.; Pezzoli, G.; Mariani, C. Malondialdheyde production features in platelets of Parkinson and Alzheimer patients and in physiological aging. Neurosci. Res. Commun. 1999, 25, 33–41. [Google Scholar] [CrossRef]
- Barret, A.J.; Rawlings, N.D.; Woessner, J.F. Handbook of Proteolytic Enzymes, 1st ed.; Academic Press: London, UK, 1998; pp. 290–292, 555–556, 1099–1100. [Google Scholar]
- Guimarães, C.M.; Gião, M.S.; Martinez, S.S.; Pintado, A.I.; Pintado, E.M.; Bento, L.S.; Malcata, F.X. Antioxidant activity of sugar molasses, including protective effect against oxidative damage. Food Chem. Toxicol. 2007, 72, 39–43. [Google Scholar] [CrossRef] [PubMed]
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C. Use of a free radical method to evaluate antioxidant activity. LWT Food Sci. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Benzie, I.F.F.; Strain, J.J. The ferric reducing ability of plasma (FRAP) as a measure of “Antioxidant Power”: The FRAP assay. Anal. Biochem. 1996, 239, 70–76. [Google Scholar] [CrossRef]
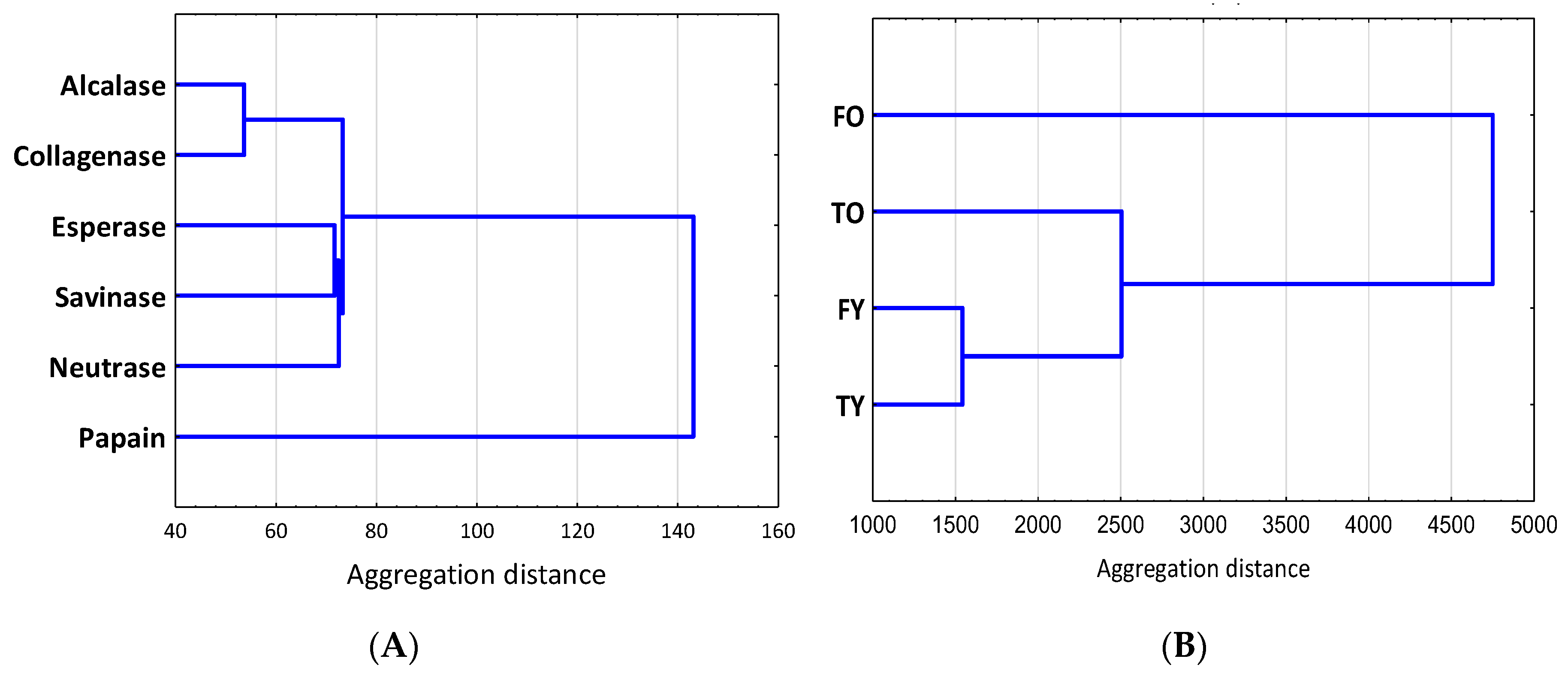
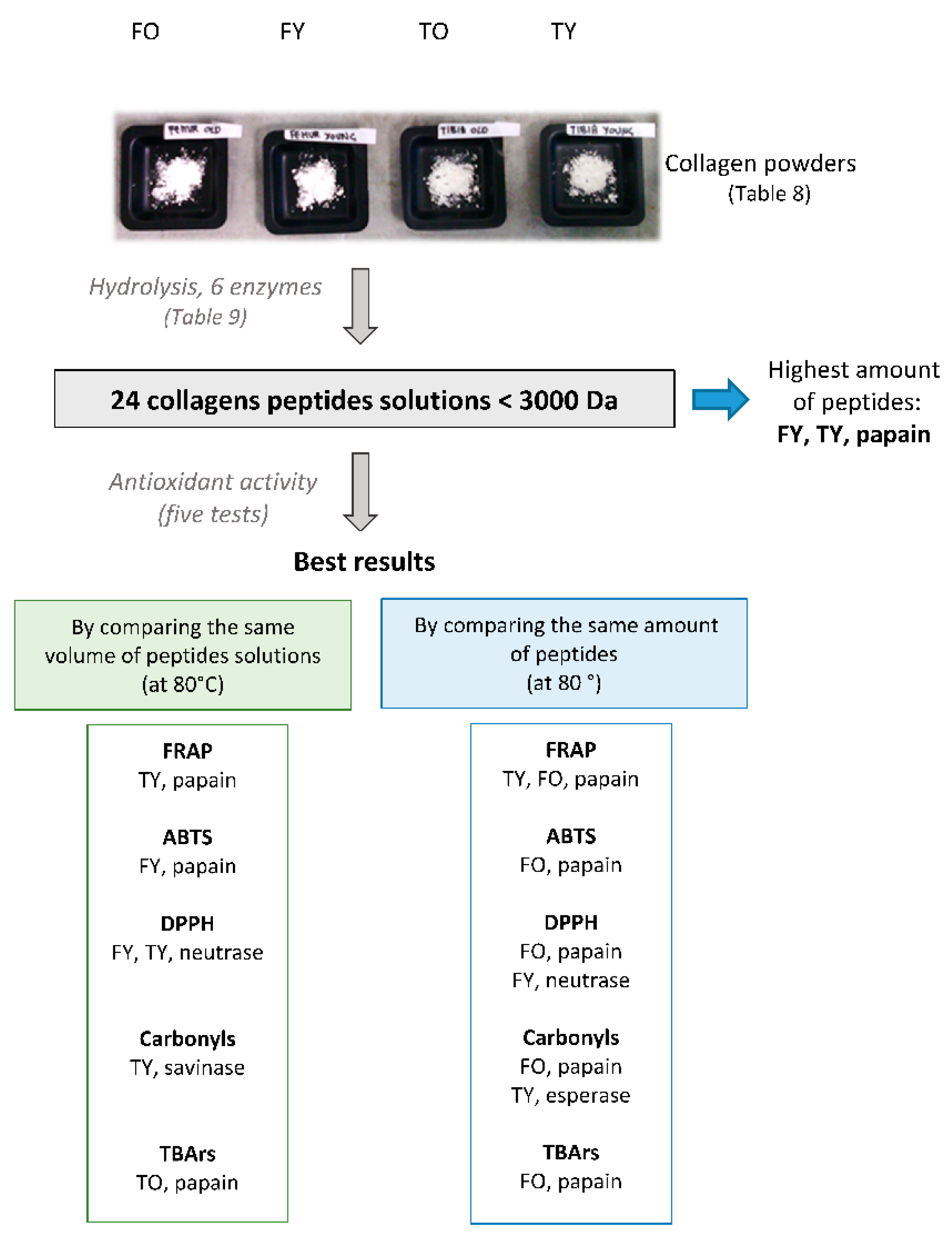
| Alcalase | Collagenase | Esperase | Neutrase | Papain | Savinase | |
|---|---|---|---|---|---|---|
| FO | 0.0050 a,A | 0.0035 a,B | 0.0077 a,C | 0.0076 a,C | 0.0096 a,D | 0.0029 a,E |
| TO | 0.0051 b,A | 0.0073 b,B | 0.0084 b,C | 0.0075 b,D | 0.0140 b,E | 0.0083 b,F |
| FY | 0.0092 c,A | 0.0102 c,B | 0.0094 c,C | 0.0064 c,D | 0.0138 c,E | 0.0076 c,F |
| TY | 0.0136 d,A | 0.0130 d,B | 0.0090 d,C | 0.0110 d,D | 0.0178 d,E | 0.0104 d,F |
| Alcalase | Collagenase | Esperase | Neutrase | Papain | Savinase | |
|---|---|---|---|---|---|---|
| FO | 16.1 ± 0.8 a,A | 7.7 ± 0.5 a,B | 25.3 ± 0.9 a,C | 23.4 ± 2.1 a,D | 40.4 ± 0.9 a,E | 10.5 ± 1.3 a,F |
| TO | 11.2 ± 1.1 b,A | 15.0 ± 0.3 b,B | 29.3 ± 1.5 b,C | 17.9 ± 1.8 b,D | 25.9 ± 0.8 b,E | 18.3 ± 0.9 b,D |
| FY | 29.6 ± 1.3 c,A | 30.8 ± 1.2 c,A | 33.5 ± 1.7 c,B | 21.3 ± 1.6 c,C | 20.2 ± 1.4 c,D | 23.6 ± 1.2 c,E |
| TY | 40.2 ± 0.9 d,A | 37.0 ± 0.9 d,B | 32.9 ± 1.3 c,C | 30.1 ± 2.0 d,D | 25.2 ± 1.6 b,E | 23.0 ± 1.5 c,F |
| Alcalase | Collagenase | Esperase | Neutrase | Papain | Savinase | |
|---|---|---|---|---|---|---|
| FO | 69.0 ± 2.3 a,A | 35.8 ± 0.8 a,B | 57.3 ± 1.1 a,C | 72.8 ± 1.3 a,D | 80.2 ± 0.9 a,E | 60.6 ± 2.2 a,F |
| TO | 41.3 ± 1.5 b,A | 38.8 ± 1.6 b,B | 56.6 ± 0.6 a,C | 57.6 ± 0.8 b,C | 35.3 ± 2.3 b,D | 36.7 ± 1.4 b,E |
| FY | 48.0 ± 1.8 c,A | 57.3 ± 1.7 c,B | 66.7 ± 1.6 b,C | 78.5 ±1.6 c,D | 25.7 ± 1.4 c,D | 50.5 ± 1.3 c,E |
| TY | 47.3 ± 1.6 c,A | 52.1 ± 2.1 d,B | 59.5 ± 1.5 c,C | 66.1 ±1.4 d,D | 24.2 ± 1.8 d,E | 34.9 ± 1.8 d,F |
| Alcalase | Collagenase | Esperase | Neutrase | Papain | Savinase | |
|---|---|---|---|---|---|---|
| FO | 15.1 ± 2.1 a,A | 10.3 ± 0.7 a,B | 14.1 ± 1.3 a,C | 14.4 ± 0.6 a,C | 20.5 ± 1.1 a,E | 18.1 ± 0.7 a,F |
| TO | 9.6 ± 1.8 b,A | 9.4 ± 1.2 b,A | 15.6 ± 1.8 b,C | 9.5 ± 0.9 b,A | 7.7 ± 1.2 b,B | 9.9 ± 0.9 b,C |
| FY | 12.8 ± 1.6 c,A | 10.5 ± 2.0 a,B | 15.2 ± 0.9 b,C | 12.7 ±1.1 c,A | 6.0 ± 0.3 c,D | 11.2 ± 1.1 c,E |
| TY | 14.1 ± 1.3 d,A | 12.1 ± 0.9 c,B | 18.5 ± 1.3 c,C | 11.1 ±0.4 d,D | 7.1 ± 0.8 b,E | 8.6 ± 1.2 d,F |
| Alcalase | Collagenase | Esperase | Neutrase | Papain | Savinase | |
|---|---|---|---|---|---|---|
| FO | 55.2 ± 1.8 a,A | 30.3 ± 0.9 a,B | 63.1 ± 2.2 a,C | 56.3 ± 2.1 a,A | 76.2 ± 1.2 a,D | 65.1 ± 1.4 a,E |
| TO | 33.4 ± 1.1 b,A | 22.5 ± 1.3 b,B | 66.2 ± 1.7 b,C | 39.4 ± 1.6 b,D | 34.1 ± 0.9 b,A | 38.2 ± 1.3 b,E |
| FY | 58.2 ± 1.6 c,A | 52.4 ± 1.6 a,B | 55.1 ± 1.4 b,C | 58.1 ± 1.4 c,A | 27.2 ± 0.8 c,D | 52.2 ± 1.2 c,B |
| TY | 47.1 ± 1.3 d,A | 43.3 ± 1.5 c,B | 53.2 ± 1.9 c,C | 50.2 ± 0.9 d,D | 25.3 ± 1.1 b,E | 44.1 ± 1.5 d,F |
| Peptides (FRAP) | y0 (µM Fe 2+) | T0 (°C) | A (µM Fe 2+) | t (°C −1) | Adj. χ2 |
|---|---|---|---|---|---|
| Papain FO (ambient–70 °C) (Equation (1a)) | 7.9 | 0 | 4.07 | 21.52 | 0.97 |
| Papain TO (Equation (1a)) | 6.22 | 0 | 5.77 | 23.96 | 0.98 |
| Papain FY (Equation (1a)) | 3.97 | 0 | 5.15 | 23.23 | 0.98 |
| Papain TY (Equation (1b)) | 16.94 | 17.25 | 14.76 | 24.24 | 0.96 |
| Esperase FO (50–80 °C) (Equation (1a)) | 50.37 | 0 | 4.07 | 17.58 | 0.95 |
| Esperase TO (Equation (1a)) | 20.12 | 0 | 1.57 | 20.28 | 0.99 |
| Esperase FY (Equation (1a)) | 20.28 | 0 | 1.96 | 20.71 | 0.96 |
| Esperase TY (Equation (1a)) | 13.05 | 0 | 4.56 | 26.31 | 1 |
| a (µM Fe 2+) | b (µM Fe 2+/°C) | ||||
| Papain FO (70–80 °C) (Equation (2)) | 99 | 0.2 | |||
| Esperase FO (ambient–50 °C) (Equation (3)) | −1.53 | 6.23 |
| Peptides (DPPH) | y0 (µM Fe 2+) | A (µM Fe 2+) | t (°C −1) | Adj. χ2 |
|---|---|---|---|---|
| Esperase TY (Equation (1a)) | 10.42 | 0.57 | 19.02 | 0.98 |
| Neutrase TO (Equation (1a)) | 1.99 | 8.14 | 36.97 | 0.99 |
| Neutrase FY (Equation (1a)) | 9.86 | 3.42 | 27.70 | 0.96 |
| Neutrase TY (Equation (1a)) | 10.10 | 5.38 | 32.81 | 0.99 |
| Peptides (DPPH) | a | b | c | Adj. χ2 |
| Esperase FO (amb.–50 °C) (Equation (4)) | 0.83 | −4.88 | −16.93 | 0.97 |
| Esperase FO (50–80 °C) (Equation (5)) | 340 | −0.27 | 0.99 | |
| Esperase TO (amb.–50 °C) (Equation (2)) | 0.80 | 0.47 | 1 | |
| Esperase TO (50–80 °C) (Equation (2)) | −17.83 | 0.85 | 0.96 | |
| Esperase FY (amb.–50 °C) (Equation (2)) | −1.58 | 0.48 | 0.98 | |
| Esperase FY (50–80 °C) (Equation (2)) | −39.33 | 1.20 | 0.99 | |
| Neutrase FO (amb.–50 °C) (Equation (2)) | 5.54 | 0.47 | 0.98 | |
| Neutrase FO (50–80 °C) (Equation (5)) | 458.37 | −0.27 | 0.99 |
| Sample | Organic Matter (%) | Collagen (%) | Non-Collagen (%) | Minerals (%) | Calcium (%) |
|---|---|---|---|---|---|
| FO | 35.3 | 34.5 | 0.83 | 64.7 | 20.5 |
| TO | 36 | 35.1 | 0.93 | 64 | 20 |
| FY | 39 | 38.4 | 0.64 | 61 | 19 |
| TY | 40 | 39.2 | 0.74 | 60 | 18 |
| Enzyme | MW (kDa) | Specificity/Cutting Sites | Temperature (°C) | pH |
|---|---|---|---|---|
| Alcalase | 20–45 | non-specific/larger and uncharged AA | 60 | 7 |
| Collagenase B | 68–130 | specific/Gly-AA | 37 | 7 |
| Esperase | 20–30 | non-specific/larger and uncharged AA | 55 | 10 |
| Neutrase | 37 | non-specific/larger and uncharged AA | 55 | 6 |
| Papain | 23.4 | specific/Ala, Val, Leu, Ile, Phe, Tyr | 65 | 6.5 |
| Savinase | 20–30 | non-specific/larger and uncharged AA | 55 | 10 |
Sample Availability: Samples of bones are available from the authors. | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aubry, L.; De-Oliveira-Ferreira, C.; Santé-Lhoutellier, V.; Ferraro, V. Redox Potential and Antioxidant Capacity of Bovine Bone Collagen Peptides towards Stable Free Radicals, and Bovine Meat Lipids and Proteins. Effect of Animal Age, Bone Anatomy and Proteases—A Step Forward towards Collagen-Rich Tissue Valorisation. Molecules 2020, 25, 5422. https://doi.org/10.3390/molecules25225422
Aubry L, De-Oliveira-Ferreira C, Santé-Lhoutellier V, Ferraro V. Redox Potential and Antioxidant Capacity of Bovine Bone Collagen Peptides towards Stable Free Radicals, and Bovine Meat Lipids and Proteins. Effect of Animal Age, Bone Anatomy and Proteases—A Step Forward towards Collagen-Rich Tissue Valorisation. Molecules. 2020; 25(22):5422. https://doi.org/10.3390/molecules25225422
Chicago/Turabian StyleAubry, Laurent, Claude De-Oliveira-Ferreira, Véronique Santé-Lhoutellier, and Vincenza Ferraro. 2020. "Redox Potential and Antioxidant Capacity of Bovine Bone Collagen Peptides towards Stable Free Radicals, and Bovine Meat Lipids and Proteins. Effect of Animal Age, Bone Anatomy and Proteases—A Step Forward towards Collagen-Rich Tissue Valorisation" Molecules 25, no. 22: 5422. https://doi.org/10.3390/molecules25225422
APA StyleAubry, L., De-Oliveira-Ferreira, C., Santé-Lhoutellier, V., & Ferraro, V. (2020). Redox Potential and Antioxidant Capacity of Bovine Bone Collagen Peptides towards Stable Free Radicals, and Bovine Meat Lipids and Proteins. Effect of Animal Age, Bone Anatomy and Proteases—A Step Forward towards Collagen-Rich Tissue Valorisation. Molecules, 25(22), 5422. https://doi.org/10.3390/molecules25225422






