Attenuation of Nrf2/Keap1/ARE in Alzheimer’s Disease by Plant Secondary Metabolites: A Mechanistic Review
Abstract
1. Introduction
2. Study Design
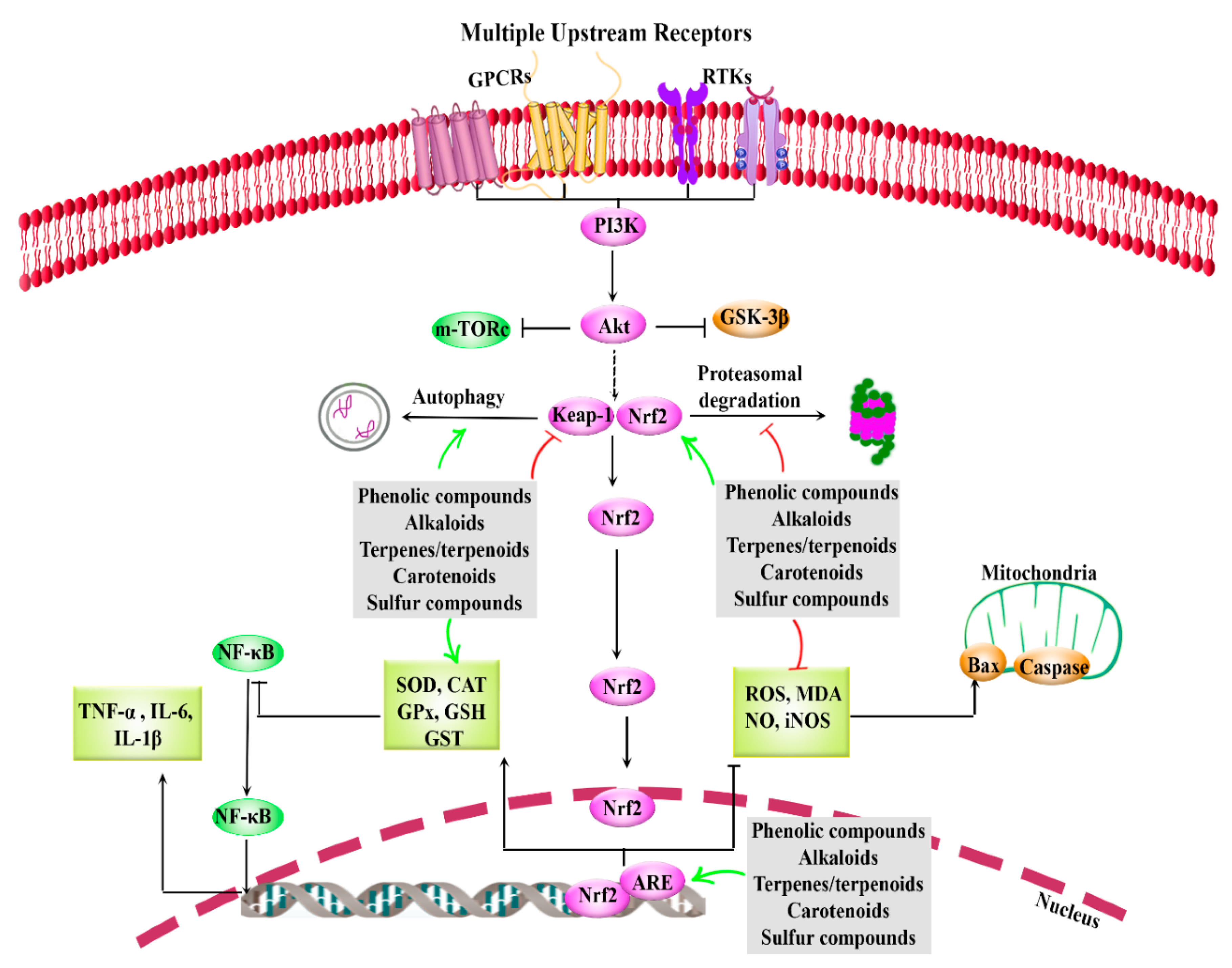
3. Nrf2/Keap1/ARE Pathway and Interconnected Mediators in AD
4. Phytochemicals Affecting Nrf2/Keap1/ARE Pathway to Combat AD
4.1. Phenolic Compounds
4.1.1. Curcumin
4.1.2. Naringenin and Naringin
4.1.3. Quercetin
4.1.4. Chalcones
4.1.5. Other Phenolic Compounds
4.2. Alkaloids
4.3. Terpenes and Terpenoids
4.4. Carotenoids
4.5. Sulfur-Containing Secondary Metabolites
4.5.1. Sulforaphane
4.5.2. S-Allyl Cysteine
4.5.3. Other Sulfur-Containing Secondary Metabolites
4.6. Miscellaneous Compounds
5. Clinical Complementary Uses of Plant Secondary Metabolites in Cognitive Dysfunctions
6. Conclusions and Perspectives
Author Contributions
Funding
Conflicts of Interest
References
- Weuve, J.; Hebert, L.E.; Scherr, P.A.; Evans, D.A. Deaths in the United States among persons with Alzheimer’s disease (2010–2050). Alzheimer’s Dement. 2014, 10, e40–e46. [Google Scholar] [CrossRef] [PubMed]
- Gan, L.; Johnson, J.A. Oxidative damage and the Nrf2-ARE pathway in neurodegenerative diseases. Biochim. Biophys. Acta (BBA)-Mol. Basis Dis. 2014, 1842, 1208–1218. [Google Scholar] [CrossRef] [PubMed]
- Skibinski, G.; Hwang, V.; Ando, D.M.; Daub, A.; Lee, A.K.; Ravisankar, A.; Modan, S.; Finucane, M.M.; Shaby, B.A.; Finkbeiner, S. Nrf2 mitigates LRRK2- and α-synuclein-induced neurodegeneration by modulating proteostasis. Proc. Natl. Acad. Sci. USA 2016, 114, 1165–1170. [Google Scholar] [CrossRef] [PubMed]
- Teixeira, J.P.; De Castro, A.A.; Soares, F.V.; Da Cunha, E.F.F.; Ramalho, T.C. Future Therapeutic Perspectives into the Alzheimer’s Disease Targeting the Oxidative Stress Hypothesis. Molecules 2019, 24, 4410. [Google Scholar] [CrossRef]
- McMahon, M.; Lamont, D.J.; Beattie, K.A.; Hayes, J.D. Keap1 perceives stress via three sensors for the endogenous signaling molecules nitric oxide, zinc, and alkenals. Proc. Natl. Acad. Sci. USA 2010, 107, 18838–18843. [Google Scholar] [CrossRef]
- Kobayashi, M.; Li, L.; Iwamoto, N.; Nakajima-Takagi, Y.; Kaneko, H.; Nakayama, Y.; Eguchi, M.; Wada, Y.; Kumagai, Y.; Yamamoto, M. The Antioxidant Defense System Keap1-Nrf2 Comprises a Multiple Sensing Mechanism for Responding to a Wide Range of Chemical Compounds. Mol. Cell. Biol. 2008, 29, 493–502. [Google Scholar] [CrossRef]
- Bresciani, A.; Missineo, A.; Gallo, M.; Cerretani, M.; Fezzardi, P.; Tomei, L.; Cicero, D.O.; Altamura, S.; Santoprete, A.; Ingenito, R.; et al. Nuclear factor (erythroid-derived 2)-like 2 (NRF2) drug discovery: Biochemical toolbox to develop NRF2 activators by reversible binding of Kelch-like ECH-associated protein 1 (KEAP1). Arch. Biochem. Biophys. 2017, 631, 31–41. [Google Scholar] [CrossRef]
- Prasad, K.N. Simultaneous activation of Nrf2 and elevation of antioxidant compounds for reducing oxidative stress and chronic inflammation in human Alzheimer’s disease. Mech. Ageing Dev. 2016, 153, 41–47. [Google Scholar] [CrossRef]
- Mohandas, E.; Rajmohan, V.; Raghunath, B. Neurobiology ofAlzheimer′s disease. Indian J. Psychiatry 2009, 51, 55–61. [Google Scholar] [CrossRef]
- De Castro, A.A.; Soares, F.V.; Pereira, A.F.; Polisel, D.A.; Caetano, M.S.; Leal, D.H.S.; Da Cunha, E.F.F.; Nepovimova, E.; Kuca, K.; Kuca, K. Non-conventional compounds with potential therapeutic effects against Alzheimer’s disease. Expert Rev. Neurother. 2019, 19, 375–395. [Google Scholar] [CrossRef]
- Andrade, S.; Ramalho, M.J.; Loureiro, J.A.; Pereira, M.C. Natural Compounds for Alzheimer’s Disease Therapy: A Systematic Review of Preclinical and Clinical Studies. Int. J. Mol. Sci. 2019, 20, 2313. [Google Scholar] [CrossRef] [PubMed]
- Cuadrado, A.; Manda, G.; Hassan, A.; Alcaraz, M.J.; Barbas, C.; Daiber, A.; Ghezzi, P.; León, R.; López, M.G.; Oliva, B.; et al. Transcription Factor NRF2 as a Therapeutic Target for Chronic Diseases: A Systems Medicine Approach. Pharmacol. Rev. 2018, 70, 348–383. [Google Scholar] [CrossRef]
- Cuadrado, A.; Rojo, A.I.; Wells, G.; Hayes, J.D.; Cousin, S.P.; Rumsey, W.L.; Attucks, O.C.; Franklin, S.; Levonen, A.-L.; Kensler, T.W.; et al. Therapeutic targeting of the NRF2 and KEAP1 partnership in chronic diseases. Nat. Rev. Drug Discov. 2019, 18, 295–317. [Google Scholar] [CrossRef]
- Bahn, G.; Jo, D.-G. Therapeutic Approaches to Alzheimer’s Disease Through Modulation of NRF2. NeuroMolecular Med. 2019, 21, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Murphy, K.E.; Park, J. Can Co-Activation of Nrf2 and Neurotrophic Signaling Pathway Slow Alzheimer’s Disease? Int. J. Mol. Sci. 2017, 18, 1168. [Google Scholar] [CrossRef] [PubMed]
- Fão, L.; Mota, S.I.; Rego, A.C. Shaping the Nrf2-ARE-related pathways in Alzheimer’s and Parkinson’s diseases. Ageing Res. Rev. 2019, 54, 100942. [Google Scholar] [CrossRef]
- Kerr, F.; Sofola-Adesakin, O.; Ivanov, D.K.; Gatliff, J.; Perez-Nievas, B.G.; Bertrand, H.C.; Martinez, P.; Callard, R.; Snoeren, I.; Cochemé, H.M.; et al. Direct Keap1-Nrf2 disruption as a potential therapeutic target for Alzheimer’s disease. PLoS Genet. 2017, 13, e1006593. [Google Scholar] [CrossRef]
- Müller, W.E.; Eckert, A.; Kurz, C.; Eckert, G.P.; Leuner, K. Mitochondrial Dysfunction: Common Final Pathway in Brain Aging and Alzheimer’s Disease—Therapeutic Aspects. Mol. Neurobiol. 2010, 41, 159–171. [Google Scholar] [CrossRef]
- Thimmulappa, R.K.; Mai, K.H.; Srisuma, S.; Kensler, T.W.; Yamamoto, M.; Biswal, S. Identification of Nrf2-regulated genes induced by the chemopreventive agent sulforaphane by oligonucleotide microarray. Cancer Res. 2002, 62, 5196–5203. [Google Scholar]
- Buendia, I.; Michalska, P.; Navarro, E.; Gameiro, I.; Egea, J.; León, R. Nrf2-ARE pathway: An emerging target against oxidative stress and neuroinflammation in neurodegenerative diseases. Pharmacol. Ther. 2016, 157, 84–104. [Google Scholar] [CrossRef]
- Nakaso, K.; Yano, H.; Fukuhara, Y.; Takeshima, T.; Wada-Isoe, K.; Nakashima, K. PI3K is a key molecule in the Nrf2-mediated regulation of antioxidative proteins by hemin in human neuroblastoma cells. FEBS Lett. 2003, 546, 181–184. [Google Scholar] [CrossRef]
- Keum, Y.-S.; Yu, S.; Chang, P.P.-J.; Yuan, X.; Kim, J.-H.; Xu, C.; Han, J.; Agarwal, A.; Kong, A.-N.T. Mechanism of Action of Sulforaphane: Inhibition of p38 Mitogen-Activated Protein Kinase Isoforms Contributing to the Induction of Antioxidant Response Element-Mediated Heme Oxygenase-1 in Human Hepatoma HepG2 Cells. Cancer Res. 2006, 66, 8804–8813. [Google Scholar] [CrossRef] [PubMed]
- Chowdhry, S.; Zhang, Y.; McMahon, M.; Sutherland, C.; Cuadrado, A.; Hayes, J.D. Nrf2 is controlled by two distinct β-TrCP recognition motifs in its Neh6 domain, one of which can be modulated by GSK-3 activity. Oncogene 2012, 32, 3765–3781. [Google Scholar] [CrossRef] [PubMed]
- Culbreth, M.; Aschner, M. GSK-3β, a double-edged sword in Nrf2 regulation: Implications for neurological dysfunction and disease. F1000Research 2018, 7, 1043. [Google Scholar] [CrossRef]
- Yu, M.; Li, H.; Liu, Q.; Liu, F.; Tang, L.; Li, C.; Yuan, Y.; Zhan, Y.; Xu, W.; Li, W.; et al. Nuclear factor p65 interacts with Keap1 to repress the Nrf2-ARE pathway. Cell. Signal. 2011, 23, 883–892. [Google Scholar] [CrossRef]
- Ramsey, C.P.; Glass, C.A.; Montgomery, M.B.; Lindl, K.A.; Ritson, G.P.; Chia, L.A.; Hamilton, R.L.; Chu, C.T.; Jordan-Sciutto, K.L. Expression of Nrf2 in Neurodegenerative Diseases. J. Neuropathol. Exp. Neurol. 2007, 66, 75–85. [Google Scholar] [CrossRef] [PubMed]
- Tungmunnithum, D.; Thongboonyou, A.; Pholboon, A.; Yangsabai, A. Flavonoids and Other Phenolic Compounds from Medicinal Plants for Pharmaceutical and Medical Aspects: An Overview. Medicines 2018, 5, 93. [Google Scholar] [CrossRef]
- Fakhri, S.; Moradi, S.Z.; Farzaei, M.H.; Bishayee, A. Modulation of dysregulated cancer metabolism by plant secondary metabolites: A mechanistic review. Semin. Cancer Biol. 2020. [Google Scholar] [CrossRef]
- Moradi, S.Z.; Momtaz, S.; Bayrami, Z.; Farzaei, M.H.; Abdollahi, M. Nanoformulations of Herbal Extracts in Treatment of Neurodegenerative Disorders. Front. Bioeng. Biotechnol. 2020, 8, 8. [Google Scholar] [CrossRef]
- Hewlings, S.J.; Kalman, D.S. Curcumin: A Review of Its’ Effects on Human Health. Foods 2017, 6, 92. [Google Scholar] [CrossRef]
- Hamaguchi, T.; Ono, K.; Murase, A.; Yamada, M. Phenolic Compounds Prevent Alzheimer’s Pathology through Different Effects on the Amyloid-β Aggregation Pathway. Am. J. Pathol. 2009, 175, 2557–2565. [Google Scholar] [CrossRef]
- Sarkar, B.; Dhiman, M.; Mittal, S.; Mantha, A.K. Curcumin revitalizes amyloid beta (25–35)-induced and organophosphate pesticides pestered neurotoxicity in SH-SY5Y and IMR-32 cells via activation of APE1 and Nrf2. Metab. Brain Dis. 2017, 32, 2045–2061. [Google Scholar] [CrossRef]
- Xu, J.; Zhou, L.; Weng, Q.; Xiao, L.; Li, Q. Curcumin analogues attenuate Aβ25–35-induced oxidative stress in PC12 cells via Keap1/Nrf2/HO-1 signaling pathways. Chem.-Biol. Interact. 2019, 305, 171–179. [Google Scholar] [CrossRef]
- Ray, B.; Bisht, S.; Maitra, A.; Maitra, A.; Lahiri, D.K. Neuroprotective and Neurorescue Effects of a Novel Polymeric Nanoparticle Formulation of Curcumin (NanoCurc™) in the Neuronal Cell Culture and Animal Model: Implications for Alzheimer’s disease. J. Alzheimer’s Dis. 2011, 23, 61–77. [Google Scholar] [CrossRef]
- Malvajerd, S.S.; Izadi, Z.; Azadi, A.; Kurd, M.; Derakhshankhah, H.; Zadeh, M.S.; Javar, H.A.; Hamidi, M. Neuroprotective Potential of Curcumin-Loaded Nanostructured Lipid Carrier in an Animal Model of Alzheimer’s Disease: Behavioral and Biochemical Evidence. J. Alzheimer’s Dis. 2019, 69, 671–686. [Google Scholar] [CrossRef] [PubMed]
- Huang, N.; Lu, S.; Liu, X.-G.; Zhu, J.; Wang, Y.-J.; Liu, R.-T. PLGA nanoparticles modified with a BBB-penetrating peptide co-delivering Aβ generation inhibitor and curcumin attenuate memory deficits and neuropathology in Alzheimer’s disease mice. Oncotarget 2017, 8, 81001–81013. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.; Li, C.; Zhang, D.; Yuan, M.; Chen, C.; Li, M. Synergic Effects of Berberine and Curcumin on Improving Cognitive Function in an Alzheimer’s Disease Mouse Model. Neurochem. Res. 2020, 1–12. [Google Scholar] [CrossRef]
- Dai, W.; Wang, H.; Fang, J.; Zhu, Y.; Zhou, J.; Wang, X.; Zhou, Y.; Zhou, M. Curcumin provides neuroprotection in model of traumatic brain injury via the Nrf2-ARE signaling pathway. Brain Res. Bull. 2018, 140, 65–71. [Google Scholar] [CrossRef]
- Sahin, K.; Orhan, C.; Tuzcu, Z.; Tuzcu, M.; Sahin, N. Curcumin ameloriates heat stress via inhibition of oxidative stress and modulation of Nrf2/HO-1 pathway in quail. Food Chem. Toxicol. 2012, 50, 4035–4041. [Google Scholar] [CrossRef]
- Pinkaew, D.; Changtam, C.; Tocharus, C.; Govitrapong, P.; Jumnongprakhon, P.; Suksamrarn, A.; Tocharus, J. Association of Neuroprotective Effect of Di-O-Demethylcurcumin on Aβ25–35-Induced Neurotoxicity with Suppression of NF-κB and Activation of Nrf2. Neurotox. Res. 2016, 29, 80–91. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Gao, K.; Li, X.; Shen, X.; Zhang, X.; Ma, C.; Qin, C.; Zhang, L. Long-term naringin consumption reverses a glucose uptake defect and improves cognitive deficits in a mouse model of Alzheimer’s disease. Pharmacol. Biochem. Behav. 2012, 102, 13–20. [Google Scholar] [CrossRef]
- Khan, M.B.; Khan, M.M.; Khan, A.; Ahmed, E.; Ishrat, T.; Tabassum, R.; Vaibhav, K.; Ahmad, A.; Islam, F. Naringenin ameliorates Alzheimer’s disease (AD)-type neurodegeneration with cognitive impairment (AD-TNDCI) caused by the intracerebroventricular-streptozotocin in rat model. Neurochem. Int. 2012, 61, 1081–1093. [Google Scholar] [CrossRef]
- Ghofrani, S.; Joghataei, M.-T.; Mohseni, S.; Baluchnejadmojarad, T.; Bagheri, M.; Khamse, S.; Roghani, M. Naringenin improves learning and memory in an Alzheimer’s disease rat model: Insights into the underlying mechanisms. Eur. J. Pharmacol. 2015, 764, 195–201. [Google Scholar] [CrossRef]
- Lou, H.; Jing, X.; Wei, X.; Shi, H.; Ren, D.; Zhang, X. Naringenin protects against 6-OHDA-induced neurotoxicity via activation of the Nrf2/ARE signaling pathway. Neuropharmacology 2014, 79, 380–388. [Google Scholar] [CrossRef]
- Wang, K.; Chen, Z.; Huang, L.; Meng, B.; Zhou, X.; Wen, X.; Ren, D. Naringenin reduces oxidative stress and improves mitochondrial dysfunction via activation of the Nrf2/ARE signaling pathway in neurons. Int. J. Mol. Med. 2017, 40, 1582–1590. [Google Scholar] [CrossRef]
- Sachdeva, A.K.; Kuhad, A.; Chopra, K. Naringin ameliorates memory deficits in experimental paradigm of Alzheimer’s disease by attenuating mitochondrial dysfunction. Pharmacol. Biochem. Behav. 2014, 127, 101–110. [Google Scholar] [CrossRef]
- Kulasekaran, G.; Ganapasam, S. Neuroprotective efficacy of naringin on 3-nitropropionic acid-induced mitochondrial dysfunction through the modulation of Nrf2 signaling pathway in PC12 cells. Mol. Cell. Biochem. 2015, 409, 199–211. [Google Scholar] [CrossRef] [PubMed]
- Sachdeva, A.K.; Chopra, K. Naringin mitigate okadaic acid-induced cognitive impairment in an experimental paradigm of Alzheimer’s disease. J. Funct. Foods 2015, 19, 110–125. [Google Scholar] [CrossRef]
- Wang, H.; Xu, Y.S.; Wang, M.L.; Cheng, C.; Bian, R.; Yuan, H.; Wang, Y.; Guo, T.; Zhu, L.L.; Zhou, H. Protective effect of naringin against the LPS-induced apoptosis of PC12 cells: Implications for the treatment of neurodegenerative disorders. Int. J. Mol. Med. 2017, 39, 819–830. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Dogra, S.; Prakash, A. Protective Effect of Naringin, a Citrus Flavonoid, Against Colchicine-Induced Cognitive Dysfunction and Oxidative Damage in Rats. J. Med. Food 2010, 13, 976–984. [Google Scholar] [CrossRef]
- Dong, F.; Wang, S.; Wang, Y.; Yang, X.; Jiang, J.; Wu, D.; Qu, X.; Fan, H.; Yao, R. Quercetin ameliorates learning and memory via the Nrf2-ARE signaling pathway in d-galactose-induced neurotoxicity in mice. Biochem. Biophys. Res. Commun. 2017, 491, 636–641. [Google Scholar] [CrossRef] [PubMed]
- Arredondo, F.; Echeverry, C.; Abin-Carriquiry, J.A.; Blasina, F.; Antúnez, K.; Jones, D.P.; Go, Y.-M.; Liang, Y.-L.; Dajas, F. After cellular internalization, quercetin causes Nrf2 nuclear translocation, increases glutathione levels, and prevents neuronal death against an oxidative insult. Free. Radic. Biol. Med. 2010, 49, 738–747. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.-R.; Yu, H.-T.; Yang, Y.; Hang, L.; Yang, X.-W.; Ding, S.-H. Quercetin phospholipid complex significantly protects against oxidative injury in ARPE-19 cells associated with activation of Nrf2 pathway. Eur. J. Pharmacol. 2016, 770, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Bahar, E.; Kim, J.-Y.; Yoon, H. Quercetin Attenuates Manganese-Induced Neuroinflammation by Alleviating Oxidative Stress through Regulation of Apoptosis, iNOS/NF-κB and HO-1/Nrf2 Pathways. Int. J. Mol. Sci. 2017, 18, 1989. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Tian, Q.; Li, Z.; Dang, M.; Lin, Y.; Hou, X. Activation of Nrf2 signaling by sitagliptin and quercetin combination against β-amyloid induced Alzheimer’s disease in rats. Drug Dev. Res. 2019, 80, 837–845. [Google Scholar] [CrossRef]
- Godoy, J.A.; Lindsay, C.B.; Quintanilla, R.A.; Carvajal, F.J.; Cerpa, W.; Inestrosa, N.C. Quercetin Exerts Differential Neuroprotective Effects Against H2O2 and Aβ Aggregates in Hippocampal Neurons: The Role of Mitochondria. Mol. Neurobiol. 2016, 54, 7116–7128. [Google Scholar] [CrossRef]
- Saviranta, N.M.; Veeroos, L.; Granlund, L.J.; Hassinen, V.H.; Kaarniranta, K.; O Karjalainen, R. Plant flavonol quercetin and isoflavone biochanin A differentially induce protection against oxidative stress and inflammation in ARPE-19 cells. Food Res. Int. 2011, 44, 109–113. [Google Scholar] [CrossRef]
- Jiménez-Aliaga, K.; Bermejo-Bescós, P.; Benedí, J.; Martín-Aragón, S. Quercetin and rutin exhibit antiamyloidogenic and fibril-disaggregating effects in vitro and potent antioxidant activity in APPswe cells. Life Sci. 2011, 89, 939–945. [Google Scholar] [CrossRef]
- Mahapatra, D.K.; Bharti, S.K.; Asati, V.; Singh, S.K. Perspectives of medicinally privileged chalcone based metal coordination compounds for biomedical applications. Eur. J. Med. Chem. 2019, 174, 142–158. [Google Scholar] [CrossRef]
- Jung, D.-W.; Lim, J.; Gal, J.; Kang, J.C.; Kim, H.J.; Kang, B.Y.; Choi, H.J. Anti-inflammatory activity of xanthohumol involves heme oxygenase-1 induction via NRF2-ARE signaling in microglial BV2 cells. Neurochem. Int. 2011, 58, 153–160. [Google Scholar] [CrossRef]
- Kim, H.J.; Jang, B.K.; Park, J.-H.; Choi, J.W.; Park, S.J.; Byeon, S.R.; Pae, A.N.; Lee, Y.S.; Cheong, E.; Park, K.D. A novel chalcone derivative as Nrf2 activator attenuates learning and memory impairment in a scopolamine-induced mouse model. Eur. J. Med. Chem. 2019, 185, 111777. [Google Scholar] [CrossRef] [PubMed]
- Iwasaki, M.; Izuo, N.; Izumi, Y.; Takada-Takatori, Y.; Akaike, A.; Kume, T. Protective Effect of Green Perilla-Derived Chalcone Derivative DDC on Amyloid β Protein-Induced Neurotoxicity in Primary Cortical Neurons. Biol. Pharm. Bull. 2019, 42, 1942–1946. [Google Scholar] [CrossRef] [PubMed]
- Ogunsuyi, O.B.; Oboh, G.; Oluokun, O.O.; Ademiluyi, A.O.; Ogunruku, O.O. Gallic acid protects against neurochemical alterations in transgenic Drosophila model of Alzheimer’s disease. Adv. Tradit. Med. 2019, 20, 89–98. [Google Scholar] [CrossRef]
- Ma, X.; Sun, Z.; Han, X.; Li, S.; Jiang, X.; Chen, S.; Zhang, J.; Lu, H. Neuroprotective Effect of Resveratrol via Activation of Sirt1 Signaling in a Rat Model of Combined Diabetes and Alzheimer’s Disease. Front. Neurosci. 2020, 13, 1400. [Google Scholar] [CrossRef]
- Tang, Y.-W.; Shi, C.-J.; Yang, H.-L.; Cai, P.; Liu, Q.-H.; Yang, X.-L.; Kong, L.-Y.; Wang, X.-B. Synthesis and evaluation of isoprenylation-resveratrol dimer derivatives against Alzheimer’s disease. Eur. J. Med. Chem. 2019, 163, 307–319. [Google Scholar] [CrossRef]
- Ding, Y.; Bao, X.; Lao, L.; Ling, Y.; Wang, Q.; Xu, S. p-Hydroxybenzyl Alcohol Prevents Memory Deficits by Increasing Neurotrophic Factors and Decreasing Inflammatory Factors in a Mice Model of Alzheimer’s Disease. J. Alzheimer’s Dis. 2019, 67, 1007–1019. [Google Scholar] [CrossRef]
- Gunesch, S.; Hoffmann, M.; Kiermeier, C.; Fischer, W.; Pinto, A.F.; Maurice, T.; Maher, P.; Decker, M. 7-O-Esters of taxifolin with pronounced and overadditive effects in neuroprotection, anti-neuroinflammation, and amelioration of short-term memory impairment in vivo. Redox Biol. 2020, 29, 101378. [Google Scholar] [CrossRef]
- Ghumatkar, P.J.; Patil, S.P.; Jain, P.D.; Tambe, R.M.; Sathaye, S. Nootropic, neuroprotective and neurotrophic effects of phloretin in scopolamine induced amnesia in mice. Pharmacol. Biochem. Behav. 2015, 135, 182–191. [Google Scholar] [CrossRef]
- Li, Y.-S.; Hong, Y.-F.; He, J.; Lin, J.-X.; Shan, Y.-l.; Fu, D.-Y.; Chen, Z.-P.; Ren, X.-R.; Song, Z.-H.; Tao, L. BPB Regular Article Effects of magnolol on impairment of learning and memory abilities induced by scopolamine in mice. Biol. Pharm. Bull. 2013, 36, b12-00880. [Google Scholar] [CrossRef][Green Version]
- Carmona, V.; Martín-Aragón, S.; Goldberg, J.; Schubert, D.; Bermejo-Bescós, P. Several targets involved in Alzheimer’s disease amyloidogenesis are affected by morin and isoquercitrin. Nutr. Neurosci. 2018, 23, 575–590. [Google Scholar] [CrossRef]
- Wang, Y.; Miao, Y.; Mir, A.Z.; Cheng, L.; Wang, L.; Zhao, L.; Cui, Q.; Zhao, W.; Wang, H. Inhibition of beta-amyloid-induced neurotoxicity by pinocembrin through Nrf2/HO-1 pathway in SH-SY5Y cells. J. Neurol. Sci. 2016, 368, 223–230. [Google Scholar] [CrossRef] [PubMed]
- Wan, T.; Wang, Z.; Luo, Y.; Zhang, Y.; He, W.; Mei, Y.; Xue, J.; Li, M.; Pan, H.; Li, W. FA-97, a New Synthetic Caffeic Acid Phenethyl Ester Derivative, Protects against Oxidative Stress-Mediated Neuronal Cell Apoptosis and Scopolamine-Induced Cognitive Impairment by Activating Nrf2/HO-1 Signaling. Oxidative Med. Cell. Longev. 2019, 2019. [Google Scholar] [CrossRef] [PubMed]
- Morroni, F.; Sita, G.; Graziosi, A.; Turrini, E.; Fimognari, C.; Tarozzi, A.; Hrelia, P. Neuroprotective effect of caffeic acid phenethyl ester in a mouse model of Alzheimer’s disease involves Nrf2/HO-1 pathway. Aging Dis. 2018, 9, 605. [Google Scholar] [CrossRef] [PubMed]
- Wruck, C.J.; Claussen, M.; Fuhrmann, G.; Romer, L.; Schulz, A.; Pufe, T.; Waetzig, V.; Peipp, M.; Herdegen, T.; Götz, M.E. Luteolin protects rat PC 12 and C6 cells against MPP+ induced toxicity via an ERK dependent Keapl-Nrf2-ARE pathway. J. Neural Transm. Suppl. 2007, 72, 57–67. [Google Scholar]
- Cui, B.; Zhang, S.; Wang, Y.; Guo, Y. Farrerol attenuates β-amyloid-induced oxidative stress and inflammation through Nrf2/Keap1 pathway in a microglia cell line. Biomed. Pharmacother. 2019, 109, 112–119. [Google Scholar] [CrossRef]
- Zhao, X.; Zou, Y.; Xu, H.; Fan, L.; Guo, H.; Li, X.; Li, G.; Zhang, X.; Dong, M. Gastrodin protect primary cultured rat hippocampal neurons against amyloid-beta peptide-induced neurotoxicity via ERK1/2-Nrf2 pathway. Brain Res. 2012, 1482, 13–21. [Google Scholar] [CrossRef]
- Zhang, Z.; Cui, W.; Li, G.; Yuan, S.; Xu, D.; Hoi, M.P.; Lin, Z.; Dou, J.; Han, Y.; Lee, S.M. Baicalein protects against 6-OHDA-induced neurotoxicity through activation of Keap1/Nrf2/HO-1 and involving PKCα and PI3K/AKT signaling pathways. J. Agric. Food Chem. 2012, 60, 8171–8182. [Google Scholar] [CrossRef]
- Serafini, M.M.; Catanzaro, M.; Fagiani, F.; Simoni, E.; Caporaso, R.; Dacrema, M.; Romanoni, I.; Govoni, S.; Racchi, M.; Daglia, M. Modulation of Keap1/Nrf2/ARE Signaling Pathway by Curcuma-and Garlic-Derived Hybrids. Front. Pharmacol. 2020, 10, 1597. [Google Scholar] [CrossRef]
- Thawabteh, A.; Juma, S.; Bader, M.; Karaman, D.; Scrano, L.; Bufo, S.A.; Karaman, R. The Biological Activity of Natural Alkaloids against Herbivores, Cancerous Cells and Pathogens. Toxins 2019, 11, 656. [Google Scholar] [CrossRef]
- Chaves, S.K.; Feitosa, C.M.; Araújo, L.D.S. Alkaloids Pharmacological Activities-Prospects for the Development of Phytopharmaceuticals for Neurodegenerative Diseases. Curr. Pharm. Biotechnol. 2016, 17, 629–635. [Google Scholar] [CrossRef]
- Liu, X.; Zhou, J.; Abid, M.D.N.; Yan, H.; Huang, H.; Wan, L.; Feng, Z.; Chen, J. Berberine Attenuates Axonal Transport Impairment and Axonopathy Induced by Calyculin A in N2a Cells. PLoS ONE 2014, 9, e93974. [Google Scholar] [CrossRef] [PubMed]
- Sadeghnia, H.R.; Kolangikhah, M.; Asadpour, E.; Forouzanfar, F.; Hosseinzadeh, H. Berberine protects against glutamate-induced oxidative stress and apoptosis in PC12 and N2a cells. Iran J. Basic Med. Sci. 2017, 20, 594–603. [Google Scholar] [PubMed]
- Chen, M.; Tan, M.; Jing, M.; Liu, A.-M.; Liu, Q.; Wen, S.; Chen, Z.; Chao, X.; He, X.-X.; Ramassamy, C.; et al. Berberine protects homocysteic acid-induced HT-22 cell death: Involvement of Akt pathway. Metab. Brain Dis. 2014, 30, 137–142. [Google Scholar] [CrossRef] [PubMed]
- Sadraie, S.; Kiasalari, Z.; Razavian, M.; Azimi, S.; SedighNejad, L.; Afshin-Majd, S.; Baluchnejadmojarad, T.; Roghani, M. Berberine ameliorates lipopolysaccharide-induced learning and memory deficit in the rat: Insights into underlying molecular mechanisms. Metab. Brain Dis. 2018, 34, 245–255. [Google Scholar] [CrossRef]
- Brandes, M.S.; Gray, N.E. NRF2 as a Therapeutic Target in Neurodegenerative Diseases. ASN Neuro 2020, 12, 1759091419899782. [Google Scholar] [CrossRef] [PubMed]
- Mohan, S.; Gupta, D. Crosstalk of toll-like receptors signaling and Nrf2 pathway for regulation of inflammation. Biomed. Pharmacother. 2018, 108, 1866–1878. [Google Scholar] [CrossRef]
- De Oliveira, J.S.; Abdalla, F.H.; Dornelles, G.L.; Palma, T.V.; Signor, C.; Bernardi, J.D.S.; Baldissarelli, J.; Lenz, L.S.; De Oliveira, V.A.; Schetinger, M.R.C.; et al. Neuroprotective effects of berberine on recognition memory impairment, oxidative stress, and damage to the purinergic system in rats submitted to intracerebroventricular injection of streptozotocin. Psychopharmacol 2018, 236, 641–655. [Google Scholar] [CrossRef]
- Fahanik-Babaei, J.; Baluchnejadmojarad, T.; Nikbakht, F.; Roghani, M. Trigonelline protects hippocampus against intracerebral Aβ(1–40) as a model of Alzheimer’s disease in the rat: Insights into underlying mechanisms. Metab. Brain Dis. 2019, 34, 191–201. [Google Scholar] [CrossRef]
- Chowdhury, A.A.; Gawali, N.B.; Munshi, R.; Juvekar, A.R. Trigonelline insulates against oxidative stress, proinflammatory cytokines and restores BDNF levels in lipopolysaccharide induced cognitive impairment in adult mice. Metab. Brain Dis. 2017, 33, 681–691. [Google Scholar] [CrossRef]
- Guan, D.; Su, Y.; Li, Y.; Wu, C.; Meng, Y.; Peng, X.; Cui, Y. Tetramethylpyrazine inhibits CoCl2 -induced neurotoxicity through enhancement of Nrf2/GCLc/GSH and suppression of HIF1alpha/NOX2/ROS pathways. J. Neurochem. 2015, 134, 551–565. [Google Scholar] [CrossRef]
- Shi, J.; Wang, Y.; Luo, G. Ligustrazine Phosphate Ethosomes for Treatment of Alzheimer’s Disease, In Vitro and in Animal Model Studies. AAPS PharmSciTech 2012, 13, 485–492. [Google Scholar] [CrossRef]
- Shi, J.; Liu, Q.; Wang, Y.; Luo, G. Coadministration of huperzine A and ligustrazine phosphate effectively reverses scopolamine-induced amnesia in rats. Pharmacol. Biochem. Behav. 2010, 96, 449–453. [Google Scholar] [CrossRef]
- Yang, X.; Wei, H.; Hu, G.; Zhao, J.; Long, L.; Li, C.; Zhao, Z.; Zeng, H.; Nie, H. Combining antioxidant astaxantin and cholinesterase inhibitor huperzine A boosts neuroprotection. Mol. Med. Rep. 2020, 21, 1043–1050. [Google Scholar] [CrossRef]
- Yang, X.; Ji, J.; Liu, C.; Zhou, M.; Li, H.; Ye, S.; Hu, Q. HJ22, a Novel derivative of piperine, Attenuates ibotenic acid-induced cognitive impairment, oxidativestress, apoptosis and inflammation via inhibiting the protein-protein interaction of Keap1-Nrf2. Int. Immunopharmacol. 2020, 83, 106383. [Google Scholar] [CrossRef]
- Chonpathompikunlert, P.; Yoshitomi, T.; Han, J.; Isoda, H.; Nagasaki, Y. The use of nitroxide radical-containing nanoparticles coupled with piperine to protect neuroblastoma SH-SY5Y cells from Aβ-induced oxidative stress. Biomaterials 2011, 32, 8605–8612. [Google Scholar] [CrossRef]
- Wang, L.; Pu, Z.; Li, M.; Wang, K.; Deng, L.; Chen, W. Antioxidative and antiapoptosis: Neuroprotective effects of dauricine in Alzheimer’s disease models. Life Sci. 2020, 243, 117237. [Google Scholar] [CrossRef] [PubMed]
- Bao, F.; Tao, L.; Zhang, H. Neuroprotective Effect of Natural Alkaloid Fangchinoline Against Oxidative Glutamate Toxicity: Involvement of Keap1-Nrf2 Axis Regulation. Cell. Mol. Neurobiol. 2019, 39, 1177–1186. [Google Scholar] [CrossRef]
- Deng, G.; Wu, C.; Rong, X.; Li, S.; Ju, Z.; Wang, Y.; Ma, C.; Ding, W.; Guan, H.; Cheng, X.; et al. Ameliorative effect of deoxyvasicine on scopolamine-induced cognitive dysfunction by restoration of cholinergic function in mice. Phytomedicine 2019, 63, 153007. [Google Scholar] [CrossRef]
- Nakhate, K.T.; Bharne, A.P.; Verma, V.S.; Aru, D.N.; Kokare, D.M. Plumbagin ameliorates memory dysfunction in streptozotocin induced Alzheimer’s disease via activation of Nrf2/ARE pathway and inhibition of beta-secretase. Biomed. Pharmacother. 2018, 101, 379–390. [Google Scholar] [CrossRef] [PubMed]
- Bhuvanendran, S.; Kumari, Y.; Othman, I.; Shaikh, M.F. Amelioration of Cognitive Deficit by Embelin in a Scopolamine-Induced Alzheimer’s Disease-Like Condition in a Rat Model. Front. Pharmacol. 2018, 9, 665. [Google Scholar] [CrossRef] [PubMed]
- Bruna, B.; Lobos, P.; Herrera-Molina, R.; Hidalgo, C.; Paula-Lima, A.; Adasme, T. The signaling pathways underlying BDNF-induced Nrf2 hippocampal nuclear translocation involve ROS, RyR-Mediated Ca2+ signals, ERK and PI3K. Biochem. Biophys. Res. Commun. 2018, 505, 201–207. [Google Scholar] [CrossRef]
- Li, S.-P.; Wang, Y.-W.; Qi, S.-L.; Zhang, Y.-P.; Deng, G.; Ding, W.-Z.; Ma, C.; Lin, Q.-Y.; Guan, H.-D.; Liu, W.; et al. Analogous β-Carboline Alkaloids Harmaline and Harmine Ameliorate Scopolamine-Induced Cognition Dysfunction by Attenuating Acetylcholinesterase Activity, Oxidative Stress, and Inflammation in Mice. Front. Pharmacol. 2018, 9, 346. [Google Scholar] [CrossRef] [PubMed]
- Xian, Y.-F.; Lin, Z.-X.; Mao, Q.-Q.; Ip, S.-P.; Su, Z.-R.; Lai, X.-P. Protective Effect of Isorhynchophylline Against β-Amyloid-Induced Neurotoxicity in PC12 Cells. Cell. Mol. Neurobiol. 2011, 32, 353–360. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Zhang, G.; Li, M.; Luo, Q.; Leng, Y.; Liu, X. Neuro-protective effects of aloperine in an Alzheimer’s disease cellular model. Biomed. Pharmacother. 2018, 108, 137–143. [Google Scholar] [CrossRef] [PubMed]
- Sun, K.; Bai, Y.; Zhao, R.; Guo, Z.; Su, X.; Li, P.; Yang, P. Neuroprotective effects of matrine on scopolamine-induced amnesia via inhibition of AChE/BuChE and oxidative stress. Metab. Brain Dis. 2018, 34, 173–181. [Google Scholar] [CrossRef] [PubMed]
- Eduviere, A.T.; Umukoro, S.; Aderibigbe, A.O.; Ajayi, A.M.; Adewole, F.A. Methyl jasmonate enhances memory performance through inhibition of oxidative stress and acetylcholinesterase activity in mice. Life Sci. 2015, 132, 20–26. [Google Scholar] [CrossRef]
- Yin, S.; Ran, Q.; Yang, J.; Zhao, Y.; Li, C. Nootropic effect of neferine on aluminium chloride–induced Alzheimer’s disease in experimental models. J. Biochem. Mol. Toxicol. 2019, 34, e22429. [Google Scholar] [CrossRef]
- Jia, X.; Liu, Y.; Li, X.; Huo, C.; Li, D.; Xu, R.; Hou, L.; Wang, X. Norcepharadione B attenuates H2O2-induced neuronal injury by upregulating cellular antioxidants and inhibiting volume-sensitive Cl− channel. Exp. Biol. Med. 2019, 244, 1463–1474. [Google Scholar] [CrossRef]
- Han, J.; Qu, Q.; Qiao, J.; Zhang, J. Vincamine Alleviates Amyloid-β 25–35 Peptides-induced Cytotoxicity in PC12 Cells. Pharmacogn. Mag. 2017, 13, 123–128. [Google Scholar] [CrossRef]
- Liang, S.; Zheng, Y.; Lei, L.; Deng, X.; Ai, J.; Li, Y.; Zhang, T.; Mei, Z.; Ren, Y. Corydalis edulis total alkaloids (CETA) ameliorates cognitive dysfunction in rat model of Alzheimer disease through regulation of the antioxidant stress and MAP2/NF-κB. J. Ethnopharmacol. 2020, 251, 112540. [Google Scholar] [CrossRef]
- Mani, V.; Ramasamy, K.; Ahmad, A.; Wahab, S.N.; Jaafar, S.M.; Kek, T.L.; Salleh, M.Z.; Majeed, A.B.A. Effects of the Total Alkaloidal Extract of Murraya koenigii Leaf on Oxidative Stress and Cholinergic Transmission in Aged Mice. Phytother. Res. 2012, 27, 46–53. [Google Scholar] [CrossRef] [PubMed]
- Koziol, A.; Stryjewska, A.; Librowski, T.; Salat, K.; Gawel, M.; Moniczewski, A.; Lochynski, S. An Overview of the Pharmacological Properties and Potential Applications of Natural Monoterpenes. Mini-Rev. Med. Chem. 2015, 14, 1156–1168. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.; Luo, Q.; Qiao, H.; Ding, H.; Cao, Y.; Yu, J.; Liu, R.; Zhang, Q.; Zhu, H.; Qu, L. The Neuroprotective Effects of Carvacrol on Ethanol-Induced Hippocampal Neurons Impairment via the Antioxidative and Antiapoptotic Pathways. Oxidative Med. Cell. Longev. 2017, 2017, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Porres-Martínez, M.; González-Burgos, E.; Carretero, M.E.; Gómez-Serranillos, M.P. In vitro neuroprotective potential of the monoterpenes α-pinene and 1,8-cineole against H2O2-induced oxidative stress in PC12 cells. Z. Naturforsch. C 2016, 71, 191–199. [Google Scholar] [CrossRef]
- De Oliveira, T.M.; De Carvalho, R.B.F.; Da Costa, I.H.F.; De Oliveira, G.A.L.; De Souza, A.A.; De Lima, S.G.; De Freitas, R.M. Evaluation ofp-cymene, a natural antioxidant. Pharm. Biol. 2014, 53, 423–428. [Google Scholar] [CrossRef]
- Xu, P.; Wang, K.; Lu, C.; Dong, L.; Gao, L.; Yan, M.; Aibai, S.; Yang, Y.; Liu, X. The Protective Effect of Lavender Essential Oil and Its Main Component Linalool against the Cognitive Deficits Induced by d-Galactose and Aluminum Trichloride in Mice. Evid. Based Complementary Altern. Med. 2017, 2017, 1–11. [Google Scholar] [CrossRef]
- Li, H.; Qin, T.; Li, M.; Ma, S. Thymol improves high-fat diet-induced cognitive deficits in mice via ameliorating brain insulin resistance and upregulating NRF2/HO-1 pathway. Metab. Brain Dis. 2017, 32, 385–393. [Google Scholar]
- Pires, L.F.; Costa, L.; De Almeida, A.A.C.; Silva, O.A.; Cerqueira, G.S.; De Sousa, D.P.; De Freitas, R.M. Is There a Correlation Between In Vitro Antioxidant Potential and In Vivo Effect of Carvacryl Acetate Against Oxidative Stress in Mice Hippocampus? Neurochem. Res. 2014, 39, 758–769. [Google Scholar] [CrossRef]
- Hur, J.; Pak, S.C.; Koo, B.-S.; Jeon, S. Borneol alleviates oxidative stress via upregulation of Nrf2 and Bcl-2 in SH-SY5Y cells. Pharm. Biol. 2012, 51, 30–35. [Google Scholar] [CrossRef]
- Prasad, S.N.; Muralidhara, M. Analysis of the antioxidant activity of geraniol employing various in-vitro models: Relevance to neurodegeneration in diabetic neuropathy. Asian J. Pharm. Clin. Res. 2017, 10, 101–105. [Google Scholar] [CrossRef]
- Lee, J.; Song, K.; Huh, E.; Oh, M.S.; Kim, Y.S. Neuroprotection against 6-OHDA toxicity in PC12 cells and mice through the Nrf2 pathway by a sesquiterpenoid from Tussilago farfara. Redox Biol. 2018, 18, 6–15. [Google Scholar] [CrossRef]
- Lim, H.J.; Dong, G.-Z.; Lee, H.J.; Ryu, J.-H. In vitro neuroprotective activity of sesquiterpenoids from the flower buds of Tussilago farfara. J. Enzym. Inhib. Med. Chem. 2014, 30, 852–856. [Google Scholar] [CrossRef] [PubMed]
- Venkatesan, R.; Subedi, L.; Yeo, E.-J.; Do, M.H. Lactucopicrin ameliorates oxidative stress mediated by scopolamine-induced neurotoxicity through activation of the NRF2 pathway. Neurochem. Int. 2016, 99, 133–146. [Google Scholar] [CrossRef] [PubMed]
- Huang, B.; He, D.; Chen, G.; Ran, X.; Guo, W.; Kan, X.; Wang, W.; Liu, D.; Fu, S.; Liu, J. α-Cyperone inhibits LPS-induced inflammation in BV-2 cells through activation of Akt/Nrf2/HO-1 and suppression of the NF-κB pathway. Food Funct. 2018, 9, 2735–2743. [Google Scholar] [CrossRef]
- Li, S.; Zhao, X.; Lazarovici, P.; Zheng, W. Artemether Activation of AMPK/GSK3β (ser9)/Nrf2 Signaling Confers Neuroprotection towards β-Amyloid-Induced Neurotoxicity in 3xTg Alzheimer’s Mouse Model. Oxidative Med. Cell. Longev. 2019, 2019. [Google Scholar] [CrossRef]
- Park, S.Y.; Choi, M.H.; Park, G.; Choi, Y.-W. Petasites japonicus bakkenolide B inhibits lipopolysaccharide-induced pro-inflammatory cytokines via AMPK/Nrf2 induction in microglia. Int. J. Mol. Med. 2017, 41, 1683–1692. [Google Scholar] [CrossRef]
- De Oliveira, M.R.; Ferreira, G.C.; Schuck, P.F.; Bosco, S.M.D. Role for the PI3K/Akt/Nrf2 signaling pathway in the protective effects of carnosic acid against methylglyoxal-induced neurotoxicity in SH-SY5Y neuroblastoma cells. Chem. Interactions 2015, 242, 396–406. [Google Scholar] [CrossRef] [PubMed]
- Kosaka, K.; Mimura, J.; Itoh, K.; Satoh, T.; Shimojo, Y.; Kitajima, C.; Maruyama, A.; Yamamoto, M.; Shirasawa, T. Role of Nrf2 and p62/ZIP in the neurite outgrowth by carnosic acid in PC12h cells. J. Biochem. 2010, 147, 73–81. [Google Scholar] [CrossRef]
- Satoh, T.; Kosaka, K.; Itoh, K.; Kobayashi, A.; Yamamoto, M.; Shimojo, Y.; Kitajima, C.; Cui, J.; Kamins, J.; Okamoto, S.I. Carnosic acid, a catechol-type electrophilic compound, protects neurons both in vitro and in vivo through activation of the Keap1/Nrf2 pathway via S-alkylation of targeted cysteines on Keap1. J. Neurochem. 2008, 104, 1116–1131. [Google Scholar] [CrossRef]
- Meng, P.; Yoshida, H.; Matsumiya, T.; Imaizumi, T.; Tanji, K.; Xing, F.; Hayakari, R.; Dempoya, J.; Tatsuta, T.; Aizawa-Yashiro, T. Carnosic acid suppresses the production of amyloid-β 1–42 by inducing the metalloprotease gene TACE/ADAM17 in SH-SY5Y human neuroblastoma cells. Neurosci. Res. 2013, 75, 94–102. [Google Scholar] [CrossRef]
- Seo, J.Y.; Pyo, E.; An, J.-P.; Kim, J.; Sung, S.H.; Oh, W.K. Andrographolide activates Keap1/Nrf2/ARE/HO-1 pathway in HT22 cells and suppresses microglial activation by Aβ42 through Nrf2-related inflammatory response. Mediat. Inflamm. 2017, 2017. [Google Scholar] [CrossRef]
- Gu, L.; Yu, Q.; Li, Q.; Zhang, L.; Lu, H.; Zhang, X. Andrographolide Protects PC12 Cells Against β-Amyloid-Induced Autophagy-Associated Cell Death Through Activation of the Nrf2-Mediated p62 Signaling Pathway. Int. J. Mol. Sci. 2018, 19, 2844. [Google Scholar] [CrossRef]
- Seo, J.Y.; Ju, S.H.; Oh, J.; Lee, S.K.; Kim, J.-S. Neuroprotective and Cognition-Enhancing Effects of Compound K Isolated from Red Ginseng. J. Agric. Food Chem. 2016, 64, 2855–2864. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Bai, X.; Yu, S.; Zhao, W.; Qiao, J.; Liu, Y.; Zhao, D.-Q.; Wang, J.; Wang, S. Ginsenoside Re Inhibits ROS/ASK-1 Dependent Mitochondrial Apoptosis Pathway and Activation of Nrf2-Antioxidant Response in Beta-Amyloid-Challenged SH-SY5Y Cells. Molecules 2019, 24, 2687. [Google Scholar] [CrossRef]
- Tom, S.; Rane, A.; Katewa, A.S.; Chamoli, M.; Matsumoto, R.R.; Andersen, J.K.; Chinta, S.J. Gedunin Inhibits Oligomeric Aβ1–42-Induced Microglia Activation Via Modulation of Nrf2-NF-κB Signaling. Mol. Neurobiol. 2019, 56, 7851–7862. [Google Scholar] [CrossRef]
- Wang, J.; Li, L.; Wang, Z.; Cui, Y.; Tan, X.; Yuan, T.; Liu, Q.; Liu, Z.; Liu, X. Supplementation of lycopene attenuates lipopolysaccharide-induced amyloidogenesis and cognitive impairments via mediating neuroinflammation and oxidative stress. J. Nutr. Biochem. 2018, 56, 16–25. [Google Scholar] [CrossRef]
- Meng, X.; Wang, M.; Sun, G.; Ye, J.; Zhou, Y.; Dong, X.; Wang, T.; Lu, S.; Sun, X. Attenuation of Aβ25–35-induced parallel autophagic and apoptotic cell death by gypenoside XVII through the estrogen receptor-dependent activation of Nrf2/ARE pathways. Toxicol. Appl. Pharmacol. 2014, 279, 63–75. [Google Scholar] [CrossRef]
- Young, A.J.; Lowe, G.M. Carotenoids—Antioxidant Properties. Antioxidants 2018, 7, 28. [Google Scholar] [CrossRef]
- Yabuzaki, J. Carotenoids Database: Structures, chemical fingerprints and distribution among organisms. Database 2017, 2017. [Google Scholar] [CrossRef]
- Britton, G. Carotenoid research: History and new perspectives for chemistry in biological systems. Biochim. et Biophys. Acta (BBA)-Mol. Cell Biol. Lipids 2020, 1865, 158699. [Google Scholar] [CrossRef]
- Fakhri, S.; Aneva, I.Y.; Farzaei, M.H.; Sobarzo-Sánchez, E. The Neuroprotective Effects of Astaxanthin: Therapeutic Targets and Clinical Perspective. Molecules 2019, 24, 2640. [Google Scholar] [CrossRef] [PubMed]
- Fakhri, S.; Abbaszadeh, F.; Dargahi, L.; Jorjani, M. Astaxanthin: A mechanistic review on its biological activities and health benefits. Pharmacol. Res. 2018, 136, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Khoei, H.H.; Fakhri, S.; Parvardeh, S.; Mofarahe, Z.S.; Baninameh, Z.; Vardiani, M. Astaxanthin prevents the methotrexate-induced reproductive toxicity by targeting oxidative stress in male mice. Toxin Rev. 2018, 38, 248–254. [Google Scholar] [CrossRef]
- Wen, X.; Huang, A.; Hu, J.; Zhong, Z.; Liu, Y.; Li, Z.; Pan, X. Neuroprotective effect of astaxanthin against glutamate-induced cytotoxicity in HT22 cells: Involvement of the Akt/GSK-3β pathway. Neuroscience 2015, 303, 558–568. [Google Scholar] [CrossRef]
- Al-Amin, M.; Mahmud, W.; Pervin, M.S.; Islam, S.R.; Rahman, M.A.; Zinchenko, A. Astaxanthin ameliorates scopolamine-induced spatial memory deficit via reduced cortical-striato-hippocampal oxidative stress. Brain Res. 2019, 1710, 74–81. [Google Scholar] [CrossRef]
- Taksima, T.; Chonpathompikunlert, P.; Sroyraya, M.; Hutamekalin, P.; Limpawattana, M.; Klaypradit, W. Effects of Astaxanthin from Shrimp Shell on Oxidative Stress and Behavior in Animal Model of Alzheimer’s Disease. Mar. Drugs 2019, 17, 628. [Google Scholar] [CrossRef]
- Han, J.H.; Lee, Y.S.; Im, J.H.; Ham, Y.W.; Lee, H.P.; Han, S.B.; Hong, J.T. Astaxanthin Ameliorates Lipopolysaccharide-Induced Neuroinflammation, Oxidative Stress and Memory Dysfunction through Inactivation of the Signal Transducer and Activator of Transcription 3 Pathway. Mar. Drugs 2019, 17, 123. [Google Scholar] [CrossRef]
- Fakhri, S.; Dargahi, L.; Abbaszadeh, F.; Jorjani, M. Astaxanthin attenuates neuroinflammation contributed to the neuropathic pain and motor dysfunction following compression spinal cord injury. Brain Res. Bull. 2018, 143, 217–224. [Google Scholar] [CrossRef]
- Fakhri, S.; Dargahi, L.; Abbaszadeh, F.; Jorjani, M. Effects of astaxanthin on sensory-motor function in a compression model of spinal cord injury: Involvement of ERK and AKT signalling pathway. Eur. J. Pain 2018, 23, 750–764. [Google Scholar] [CrossRef]
- Wang, C.; Cai, X.; Hu, W.; Li, Z.; Kong, F.; Chen, X.; Wang, D. Investigation of the neuroprotective effects of crocin via antioxidant activities in HT22 cells and in mice with Alzheimer’s disease. Int. J. Mol. Med. 2018, 43, 956–966. [Google Scholar] [CrossRef]
- Wang, L.; Chen, Y.; Sternberg, P.; Cai, J. Essential Roles of the PI3 Kinase/Akt Pathway in Regulating Nrf2-Dependent Antioxidant Functions in the RPE. Investig. Opthalmology Vis. Sci. 2008, 49, 1671–1678. [Google Scholar] [CrossRef] [PubMed]
- Mohammadzadeh, L.; Abnous, K.; Razavi, B.M.; Hosseinzadeh, H. Crocin-protected malathion-induced spatial memory deficits by inhibiting TAU protein hyperphosphorylation and antiapoptotic effects. Nutr. Neurosci. 2019, 23, 221–236. [Google Scholar] [CrossRef] [PubMed]
- NaghiZadeh, B.; Mansouri, S.M.T.; Ghorbanzadeh, B.; Farbood, Y.; Sarkaki, A. Protective effects of oral crocin against intracerebroventricular streptozotocin-induced spatial memory deficit and oxidative stress in rats. Phytomedicine 2013, 20, 537–542. [Google Scholar] [CrossRef]
- NaghiZadeh, B.; Mansouri, S.M.T.; Ghorbanzadeh, B. Protective effects of crocin against streptozotocin-induced oxidative damage in rat striatum. Acta Medica Iran. 2014, 52, 101–105. [Google Scholar]
- Saini, R.K.; Rengasamy, K.R.; Mahomoodally, F.M.; Keum, Y.-S. Protective effects of lycopene in cancer, cardiovascular, and neurodegenerative diseases: An update on epidemiological and mechanistic perspectives. Pharmacol. Res. 2020, 155, 104730. [Google Scholar] [CrossRef] [PubMed]
- Yu, L.; Wang, W.; Pang, W.; Xiao, Z.; Jiang, Y.; Hong, Y. Dietary Lycopene Supplementation Improves Cognitive Performances in Tau Transgenic Mice Expressing P301L Mutation via Inhibiting Oxidative Stress and Tau Hyperphosphorylation. J. Alzheimer’s Dis. 2017, 57, 475–482. [Google Scholar] [CrossRef]
- Hira, S.; Saleem, U.; Anwar, F.; Sohail, M.F.; Raza, Z.; Ahmad, B. β-Carotene: A Natural Compound Improves Cognitive Impairment and Oxidative Stress in a Mouse Model of Streptozotocin-Induced Alzheimer’s Disease. Biomolecules 2019, 9, 441. [Google Scholar] [CrossRef]
- Al-Amin, M.; Chowdury, I.A.; Saifullah, A.R.M.; Alam, M.N.; Jain, P.; Hossain, M.; Alam, A.; Kazi, M.; Ahmad, A.; Raish, M.; et al. Levocarnitine Improves AlCl3-Induced Spatial Working Memory Impairment in Swiss albino Mice. Front. Neurosci. 2019, 13, 278. [Google Scholar] [CrossRef]
- Stauder, R.; Welsch, R.; Camagna, M.; Kohlen, W.; Balcke, G.U.; Tissier, A.; Walter, M.H. Strigolactone Levels in Dicot Roots Are Determined by an Ancestral Symbiosis-Regulated Clade of the PHYTOENE SYNTHASE Gene Family. Front. Plant Sci. 2018, 9, 255. [Google Scholar] [CrossRef]
- Kurt, B.; Ozleyen, A.; Antika, G.; Yilmaz, Y.B.; Tumer, T.B. Multitarget Profiling of a Strigolactone Analogue for Early Events of Alzheimer’s Disease: In Vitro Therapeutic Activities against Neuroinflammation. ACS Chem. Neurosci. 2020, 11, 501–507. [Google Scholar] [CrossRef]
- Fahey, J.W.; Zalcmann, A.T.; Talalay, P. The chemical diversity and distribution of glucosinolates and isothiocyanates among plants. Phytochemistry 2001, 56, 5–51. [Google Scholar] [CrossRef]
- Venditti, A. Sulfur-containing Secondary Metabolites as Neuroprotective Agents. Curr. Med. Chem. 2020, 27, 4421–4436. [Google Scholar] [CrossRef] [PubMed]
- Uddin, S.; Al Mamun, A.; Jakaria, M.; Thangapandiyan, S.; Ahmad, J.; Rahman, A.; Mathew, B.; Abdel-Daim, M.M.; Aleya, L. Emerging promise of sulforaphane-mediated Nrf2 signaling cascade against neurological disorders. Sci. Total. Environ. 2020, 707, 135624. [Google Scholar] [CrossRef]
- Zhao, F.; Zhang, J.; Chang, N. Epigenetic modification of Nrf2 by sulforaphane increases the antioxidative and anti-inflammatory capacity in a cellular model of Alzheimer’s disease. Eur. J. Pharmacol. 2018, 824, 1–10. [Google Scholar] [CrossRef]
- Tarozzi, A.; Morroni, F.; Merlicco, A.; Hrelia, S.; Angeloni, C.; Hrelia, P.; Cantelli-Forti, G. Sulforaphane as an inducer of glutathione prevents oxidative stress-induced cell death in a dopaminergic-like neuroblastoma cell line. J. Neurochem. 2009, 111, 1161–1171. [Google Scholar] [CrossRef]
- Soane, L.; Dai, W.L.; Fiskum, G.; Bambrick, L.L. Sulforaphane protects immature hippocampal neurons against death caused by exposure to hemin or to oxygen and glucose deprivation. J. Neurosci. Res. 2009, 88, 1355–1363. [Google Scholar] [CrossRef]
- Mizuno, K.; Kume, T.; Muto, C.; Takada-Takatori, Y.; Izumi, Y.; Sugimoto, H.; Akaike, A. Glutathione Biosynthesis via Activation of the Nuclear Factor E2-Related Factor 2 (Nrf2)–Antioxidant-Response Element (ARE) Pathway Is Essential for Neuroprotective Effects of Sulforaphane and 6-(Methylsulfinyl) Hexyl Isothiocyanate. J. Pharmacol. Sci. 2011, 115, 320–328. [Google Scholar] [CrossRef]
- Jin, H.; Deng, C.; Tao, R.; Yu, S.-Z. Sulforaphane protects against 6-hydroxydopamine-induced cytotoxicity by increasing expression of heme oxygenase-1 in a PI3K/Akt-dependent manner. Mol. Med. Rep. 2011, 5, 847–851. [Google Scholar] [CrossRef][Green Version]
- Morroni, F.; Tarozzi, A.; Sita, G.; Bolondi, C.; Moraga, J.M.Z.; Cantelli-Forti, G.; Hrelia, P. Neuroprotective effect of sulforaphane in 6-hydroxydopamine-lesioned mouse model of Parkinson’s disease. NeuroToxicology 2013, 36, 63–71. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Q.; Chen, B.; Wang, X.; Wu, L.; Yang, Y.; Cheng, X.; Hu, Z.; Cai, X.; Yang, J.; Sun, X.; et al. Sulforaphane protects against rotenone-induced neurotoxicity in vivo: Involvement of the mTOR, Nrf2 and autophagy pathways. Sci. Rep. 2016, 6, 32206. [Google Scholar] [CrossRef]
- An, Y.W.; Jhang, K.A.; Woo, S.-Y.; Kang, J.L.; Chong, Y.H. Sulforaphane exerts its anti-inflammatory effect against amyloid-β peptide via STAT-1 dephosphorylation and activation of Nrf2/HO-1 cascade in human THP-1 macrophages. Neurobiol. Aging 2016, 38, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Pu, D.; Zhao, Y.; Chen, J.; Sun, Y.; Lv, A.; Zhu, S.; Luo, C.; Zhao, K.; Xiao, Q. Protective Effects of Sulforaphane on Cognitive Impairments and AD-like Lesions in Diabetic Mice are Associated with the Upregulation of Nrf2 Transcription Activity. Neuroscience 2018, 381, 35–45. [Google Scholar] [CrossRef] [PubMed]
- Bergström, P.; Andersson, H.C.; Gao, Y.; Karlsson, J.-O.; Nodin, C.; Anderson, M.F.; Nilsson, M.; Hammarsten, O. Repeated transient sulforaphane stimulation in astrocytes leads to prolonged Nrf2-mediated gene expression and protection from superoxide-induced damage. Neuropharmacology 2011, 60, 343–353. [Google Scholar] [CrossRef]
- Jin, H.; Deng, C.; Tao, R.; Yu, S.-Z. Inhibition of 6-hydroxydopamine-induced endoplasmic reticulum stress by sulforaphane through the activation of Nrf2 nuclear translocation. Mol. Med. Rep. 2012, 6, 215–219. [Google Scholar] [CrossRef]
- Javed, H.; Khan, M.M.; Khan, A.; Vaibhav, K.; Ahmad, A.; Khuwaja, G.; Ahmed, E.; Raza, S.S.; Ashafaq, M.; Tabassum, R.; et al. S-allyl cysteine attenuates oxidative stress associated cognitive impairment and neurodegeneration in mouse model of streptozotocin-induced experimental dementia of Alzheimer’s type. Brain Res. 2011, 1389, 133–142. [Google Scholar] [CrossRef]
- Shi, H.; Jing, X.; Wei, X.; Perez, R.G.; Ren, M.; Zhang, X.; Lou, H. S-allyl cysteine activates the Nrf2-dependent antioxidant response and protects neurons against ischemic injuryin vitroandin vivo. J. Neurochem. 2015, 133, 298–308. [Google Scholar] [CrossRef]
- García, E.; Santana-Martínez, R.; Silva-Islas, C.A.; Colín-González, A.L.; Galván-Arzate, S.; Heras, Y.; Maldonado, P.D.; Sotelo, J.; Santamaría, A. S-allyl cysteine protects against MPTP-induced striatal and nigral oxidative neurotoxicity in mice: Participation of Nrf2. Free. Radic. Res. 2013, 48, 159–167. [Google Scholar] [CrossRef]
- Ito, Y.; Kosuge, Y.; Sakikubo, T.; Horie, K.; Ishikawa, N.; Obokata, N.; Yokoyama, E.; Yamashina, K.; Yamamoto, M.; Saito, H.; et al. Protective effect of S-allyl-l-cysteine, a garlic compound, on amyloid β-protein-induced cell death in nerve growth factor-differentiated PC12 cells. Neurosci. Res. 2003, 46, 119–125. [Google Scholar] [CrossRef]
- Denzer, I.; Münch, G.; Pischetsrieder, M.; Friedland, K. S-allyl-l-cysteine and isoliquiritigenin improve mitochondrial function in cellular models of oxidative and nitrosative stress. Food Chem. 2016, 194, 843–848. [Google Scholar] [CrossRef]
- Kosuge, Y.; Koen, Y.; Ishige, K.; Minami, K.; Urasawa, H.; Saito, H.; Ito, Y. S-allyl-l-cysteine selectively protects cultured rat hippocampal neurons from amyloid β-protein- and tunicamycin-induced neuronal death. Neuroscience 2003, 122, 885–895. [Google Scholar] [CrossRef] [PubMed]
- Tsai, S.-J.; Chiu, C.P.; Yang, H.-T.; Yin, M.-C. S-Allyl cysteine, S-ethyl cysteine, and S-propyl cysteine alleviate β-amyloid, glycative, and oxidative injury in brain of mice treated by d-galactose. J. Agric. Food Chem. 2011, 59, 6319–6326. [Google Scholar] [CrossRef] [PubMed]
- Morroni, F.; Sita, G.; Graziosi, A.; Turrini, E.; Fimognari, C.; Tarozzi, A.; Hrelia, P. Protective Effects of 6-(Methylsulfinyl) hexyl isothiocyanate on aβ1–42-induced cognitive deficit, oxidative stress, inflammation, and apoptosis in mice. Int. J. Mol. Sci. 2018, 19, 2083. [Google Scholar] [CrossRef] [PubMed]
- Tu, G.; Zhang, Y.-F.; Wei, W.; Li, L.; Zhang, Y.; Yang, J.; Xing, Y. Allicin attenuates H2O2-induced cytotoxicity in retinal pigmented epithelial cells by regulating the levels of reactive oxygen species. Mol. Med. Rep. 2016, 13, 2320–2326. [Google Scholar] [CrossRef]
- Li, X.-H.; Li, C.-Y.; Lu, J.-M.; Tian, R.-B.; Wei, J. Allicin ameliorates cognitive deficits ageing-induced learning and memory deficits through enhancing of Nrf2 antioxidant signaling pathways. Neurosci. Lett. 2012, 514, 46–50. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.-F.; Li, X.-H.; Yuan, Z.-P.; Li, C.-Y.; Tian, R.-B.; Jia, W.; Xiao, Z.-P. Allicin improves endoplasmic reticulum stress-related cognitive deficits via PERK/Nrf2 antioxidative signaling pathway. Eur. J. Pharmacol. 2015, 762, 239–246. [Google Scholar] [CrossRef] [PubMed]
- Yun, H.-M.; Jin, P.; Park, K.-R.; Hwang, J.; Jeong, H.-S.; Kim, E.-C.; Jung, J.-K.; Oh, K.-W.; Hwang, B.Y.; Han, S.-B.; et al. Thiacremonone Potentiates Anti-Oxidant Effects to Improve Memory Dysfunction in an APP/PS1 Transgenic Mice Model. Mol. Neurobiol. 2015, 53, 2409–2420. [Google Scholar] [CrossRef] [PubMed]
- Cui, Y.; Ma, S.; Zhang, C.; Li, D.; Yang, B.; Lv, P.; Xing, Q.; Huang, T.; Yang, G.L.; Cao, W.; et al. Pharmacological activation of the Nrf2 pathway by 3H-1,2-dithiole-3-thione is neuroprotective in a mouse model of Alzheimer disease. Behav. Brain Res. 2018, 336, 219–226. [Google Scholar] [CrossRef]
- Kimura, Y.; Dargusch, R.; Schubert, D.; Kimura, H. Hydrogen Sulfide Protects HT22 Neuronal Cells from Oxidative Stress. Antioxid. Redox Signal. 2006, 8, 661–670. [Google Scholar] [CrossRef]
- Koriyama, Y.; Nakayama, Y.; Matsugo, S.; Kato, S. Protective effect of lipoic acid against oxidative stress is mediated by Keap1/Nrf2-dependent heme oxygenase-1 induction in the RGC-5 cellline. Brain Res. 2013, 1499, 145–157. [Google Scholar] [CrossRef]
- Yang, Q.; Lin, J.; Zhang, H.; Liu, Y.; Kan, M.; Xiu, Z.; Chen, X.; Lan, X.; Li, X.; Shi, X.; et al. Ginsenoside Compound K Regulates Amyloid beta via the Nrf2/Keap1 Signaling Pathway in Mice with Scopolamine Hydrobromide-Induced Memory Impairments. J. Mol. Neurosci. 2019, 67, 62–71. [Google Scholar] [CrossRef]
- Lu, C.; Dong, L.; Lv, J.; Wang, Y.; Fan, B.; Wang, F.; Liu, X. 20(S)-protopanaxadiol (PPD) alleviates scopolamine-induced memory impairment via regulation of cholinergic and antioxidant systems, and expression of Egr-1, c-Fos and c-Jun in mice. Chem. Interactions 2018, 279, 64–72. [Google Scholar] [CrossRef]
- Du, J.; Cui, C.H.; Park, S.C.; Kim, J.K.; Yu, H.S.; Jin, F.X.; Sun, C.; Kim, S.C.; Im, W.T. Identification and characterization of a ginsenoside-transforming beta-glucosidase from Pseudonocardia sp. Gsoil 1536 and its application for enhanced production of minor ginsenoside Rg2(S). PLoS ONE 2014, 9, e96914. [Google Scholar]
- Liu, J.F.; Yan, X.D.; Qi, L.S.; Li, L.; Hu, G.Y.; Li, P.; Zhao, G. Ginsenoside Rd attenuates Aβ25–35-induced oxidative stress and apoptosis in primary cultured hippocampal neurons. Chem.-Biol. Interact. 2015, 239, 12–18. [Google Scholar] [CrossRef]
- Lu, C.; Lv, J.; Dong, L.; Jiang, N.; Wang, Y.; Wang, Q.; Li, Y.; Chen, S.; Fan, B.; Wang, F.; et al. Neuroprotective effects of 20(S)-protopanaxatriol (PPT) on scopolamine-induced cognitive deficits in mice. Phytotherapy Res. 2018, 32, 1056–1063. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Kan, H.; Yin, Y.; Wu, W.; Hu, W.; Wang, M.; Li, W.; Li, W. Protective effects of ginsenoside Rg1 on chronic restraint stress induced learning and memory impairments in male mice. Pharmacol. Biochem. Behav. 2014, 120, 73–81. [Google Scholar] [CrossRef]
- Kovac, S.; Angelova, P.R.; Holmström, K.M.; Zhang, Y.; Dinkova-Kostova, A.T.; Abramov, A.Y. Nrf2 regulates ROS production by mitochondria and NADPH oxidase. Biochim. Biophys. Acta (BBA)-Gen. Subj. 2015, 1850, 794–801. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.-M.; Liu, M.-Y.; Wang, F.; Wei, Q.; Wang, S.; Wu, C.; Yang, J. Anti-amnesic effect of pseudoginsenoside-F11 in two mouse models of Alzheimer’s disease. Pharmacol. Biochem. Behav. 2013, 106, 57–67. [Google Scholar] [CrossRef]
- Zhao, X.; Liu, C.; Qi, Y.; Fang, L.; Luo, J.; Bi, K.; Jia, Y. Timosaponin B-II ameliorates scopolamine-induced cognition deficits by attenuating acetylcholinesterase activity and brain oxidative damage in mice. Metab. Brain Dis. 2016, 31, 1455–1461. [Google Scholar] [CrossRef]
- Tao, L.; Xie, J.; Wang, Y.; Wang, S.; Wu, S.; Wang, Q.; Ding, H. Protective effects of aloe-emodin on scopolamine-induced memory impairment in mice and H2O2-induced cytotoxicity in PC12 cells. Bioorg. Med. Chem. Lett. 2014, 24, 5385–5389. [Google Scholar] [CrossRef]
- Fragoulis, A.; Siegl, S.; Fendt, M.; Jansen, S.; Soppa, U.; Brandenburg, L.-O.; Pufe, T.; Weis, J.; Wruck, C.J. Oral administration of methysticin improves cognitive deficits in a mouse model of Alzheimer’s disease. Redox Biol. 2017, 12, 843–853. [Google Scholar] [CrossRef]
- Gugliandolo, A.; Chiricosta, L.; Silvestro, S.; Bramanti, P.; Mazzon, E. α-Tocopherol Modulates Non-Amyloidogenic Pathway and Autophagy in an In Vitro Model of Alzheimer’s Disease: A Transcriptional Study. Brain Sci. 2019, 9, 196. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.-W.; Yang, S.-G.; Liu, W.; Zhang, Y.-X.; Xu, P.-X.; Wang, T.; Ling, T.-J.; Liu, R.-T. Alpha-tocopherol quinine ameliorates spatial memory deficits by reducing beta-amyloid oligomers, neuroinflammation and oxidative stress in transgenic mice with Alzheimer’s disease. Behav. Brain Res. 2016, 296, 109–117. [Google Scholar] [CrossRef] [PubMed]
- Lee, A.Y.; Lee, M.H.; Lee, S.; Cho, E.J. Neuroprotective Effect of Alpha-Linolenic Acid against Abeta-Mediated Inflammatory Responses in C6 Glial Cell. J. Agric. Food Chem. 2018, 66, 4853–4861. [Google Scholar] [CrossRef]
- Hashimoto, M.; Tanabe, Y.; Hossain, S.; Matsuzaki, K.; Ohno, M.; Kato, S.; Katakura, M.; Shido, O. Intake of Alpha-Linolenic Acid-Rich Perilla frutescens Leaf Powder Decreases Home Blood Pressure and Serum Oxidized Low-Density Lipoprotein in Japanese Adults. Molecules 2020, 25, 2099. [Google Scholar] [CrossRef] [PubMed]
- Andújar, I.; Ríos, J.L.; Giner, R.M.; Recio, M.C. Pharmacological Properties of Shikonin—A Review of Literature since 2002. Planta Medica 2013, 79, 1685–1697. [Google Scholar] [CrossRef]
- Guo, C.; He, J.; Song, X.; Tan, L.; Wang, M.; Jiang, P.; Li, Y.; Cao, Z.; Peng, C. Pharmacological properties and derivatives of shikonin—A review in recent years. Pharmacol. Res. 2019, 149, 104463. [Google Scholar] [CrossRef]
- Tong, Y.; Bai, L.; Gong, R.; Chuan, J.; Duan, X.; Zhu, Y. Shikonin Protects PC12 Cells Against β-amyloid Peptide-Induced Cell Injury Through Antioxidant and Antiapoptotic Activities. Sci. Rep. 2018, 8, 1–10. [Google Scholar] [CrossRef]
- Gandía-Herrero, F.; Escribano, J.; Garcia-Carmona, F. Biological Activities of Plant Pigments Betalains. Crit. Rev. Food Sci. Nutr. 2014, 56, 937–945. [Google Scholar] [CrossRef]
- Guerrero-Rubio, M.A.; Hernández-García, S.; Escribano, J.; Jiménez-Atiénzar, M.; Cabanes, J.; García-Carmona, F.; Gandía-Herrero, F. Betalain health-promoting effects after ingestion in Caenorhabditis elegans are mediated by DAF-16/FOXO and SKN-1/Nrf2 transcription factors. Food Chem. 2020, 330, 127228. [Google Scholar] [CrossRef]
- Shah, S.A.; Khan, M.; Jo, M.H.; Jo, M.G.; Amin, F.U.; Kim, M.O. Melatonin stimulates the SIRT1/Nrf2 signaling pathway counteracting lipopolysaccharide (LPS)-induced oxidative stress to rescue postnatal rat brain. CNS Neurosci. Ther. 2017, 23, 33–44. [Google Scholar] [CrossRef]
- Khodagholi, F.; Eftekharzadeh, B.; Maghsoudi, N.; Rezaei, P.F. Chitosan prevents oxidative stress-induced amyloid beta formation and cytotoxicity in NT2 neurons: Involvement of transcription factors Nrf2 and NF-kappaB. Mol. Cell Biochem. 2010, 337, 39–51. [Google Scholar] [CrossRef] [PubMed]
- Cao, S.; Du, J.; Hei, Q. Lycium barbarum polysaccharide protects against neurotoxicity via the Nrf2-HO-1 pathway. Exp. Ther. Med. 2017, 14, 4919–4927. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Chen, X.; Zhang, Y.; Liu, X.; Wang, C.; Teng, L.; Wang, D. Protective roles of Amanita caesarea polysaccharides against Alzheimer’s disease via Nrf2 pathway. Int. J. Biol. Macromol. 2019, 121, 29–37. [Google Scholar] [CrossRef] [PubMed]
- Han, Y.; Nan, S.; Fan, J.; Chen, Q.; Zhang, Y. Inonotus obliquus polysaccharides protect against Alzheimer’s disease by regulating Nrf2 signaling and exerting antioxidative and antiapoptotic effects. Int. J. Biol. Macromol. 2019, 131, 769–778. [Google Scholar] [CrossRef]
- Han, Y.; Yang, H.; Li, L.; Du, X.; Sun, C. Schisanhenol improves learning and memory in scopolamine-treated mice by reducing acetylcholinesterase activity and attenuating oxidative damage through SIRT1-PGC-1α-Tau signaling pathway. Int. J. Neurosci. 2018, 129, 110–118. [Google Scholar] [CrossRef]
- Rutledge, G.A.; Fisher, D.R.; Miller, M.G.; Kelly, M.E.; Bielinski, D.F.; Shukitt-Hale, B. The effects of blueberry and strawberry serum metabolites on age-related oxidative and inflammatory signaling in vitro. Food Funct. 2019, 10, 7707–7713. [Google Scholar] [CrossRef]
- Bowtell, J.L.; Aboo-Bakkar, Z.; Conway, M.E.; Adlam, A.-L.R.; Fulford, J. Enhanced task-related brain activation and resting perfusion in healthy older adults after chronic blueberry supplementation. Appl. Physiol. Nutr. Metab. 2017, 42, 773–779. [Google Scholar] [CrossRef]
- Nilsson, A.; Salo, I.; Plaza, M.; Björck, I. Effects of a mixed berry beverage on cognitive functions and cardiometabolic risk markers; A randomized cross-over study in healthy older adults. PLoS ONE 2017, 12, e0188173. [Google Scholar] [CrossRef]
- Lee, J.; Torosyan, N.; Silverman, D.H. Examining the impact of grape consumption on brain metabolism and cognitive function in patients with mild decline in cognition: A double-blinded placebo controlled pilot study. Exp. Gerontol. 2017, 87, 121–128. [Google Scholar] [CrossRef]
- Kennedy, D.O.; Wightmann, E.L.; Reay, J.L.; Lietz, G.; Okello, E.J.; Wilde, A.; Haskell, C.F. Effects of resveratrol on cerebral blood flow variables and cognitive performance in humans: A double-blind, placebo-controlled, crossover investigation. Am. J. Clin. Nutr. 2010, 91, 1590–1597. [Google Scholar] [CrossRef]
- Power, R.; Coen, R.F.; Beatty, S.; Mulcahy, R.; Moran, R.; Stack, J.; Howard, A.; Nolan, J.M. Supplemental Retinal Carotenoids Enhance Memory in Healthy Individuals with Low Levels of Macular Pigment in A Randomized, Double-Blind, Placebo-Controlled Clinical Trial. J. Alzheimer’s Dis. 2018, 61, 947–961. [Google Scholar] [CrossRef] [PubMed]
- Rinaldi, P.; Polidori, M.; Metastasio, A.; Mariani, E.; Mattioli, P.; Cherubini, A.; Catani, M.; Cecchetti, R.; Senin, U.; Mecocci, P. Plasma antioxidants are similarly depleted in mild cognitive impairment and in Alzheimer’s disease. Neurobiol. Aging 2003, 24, 915–919. [Google Scholar] [CrossRef]
- Abbaszadeh, F.; Fakhri, S.; Khan, H. Targeting apoptosis and autophagy following spinal cord injury: Therapeutic approaches to polyphenols and candidate phytochemicals. Pharmacol. Res. 2020, 160, 105069. [Google Scholar] [CrossRef]
| Compounds | Types of Study | Cell Line(s)/Animal Model(s) | Mechanisms of Action | References |
|---|---|---|---|---|
| Curcumin | In vitro | human neuroblastoma cell lines (SH-SY5Y, IMR-32) | ↑APE1 and Nrf2 | [32] |
| In vitro, In vivo | human neuroblastoma cell lines (SH-SY5Y), CD1 athymic mice | ↓caspase-3 and caspase 7 ↓H2O2 ↑GSH ↑the ratio of free to oxidized GSH | [34] | |
| In vivo | male Sprague-Dawley rats | ↓mitochondrial dysfunction ↓MDA ↓ROS ↑Thiol | [35] | |
| male AD model (APP/PS1dE9) mice | ↓IL-6 ↓ROS ↓Aβ level ↓TNF-α ↑SOD activities | [36] | ||
| male ICR mice model of traumatic brain injury | ↓neuronal apoptosis ↓MDA ↑SOD ↑GPx | [38] | ||
| Japanese quails (Coturnix coturnix japonica) | ↓MDA levels ↑SOD ↑GPx ↑CAT ↑Nrf2 and HO-1 levels | [39] | ||
| Curcumin and berberine | In vivo | B6C3-Tg (APPswePSEN1dE9)/Nju double transgenic mice model of AD | ↓IL-1β ↓TNF-α ↓IL-6 ↓GFAP and IBA1 | [37] |
| In vitro | human neuroblastoma cell line (SH-SY5Y) | ↓ROS ↓iNOS, ↓NO ↓NF-κB ↑Nrf2 | [40] | |
| In vitro | rat adrenal pheochromocytoma cell line (PC12) | ↑Nrf2/HO-1 protein expression ↓Keap1 ↑translation of Nrf2 into nuclear ↑SOD/CAT ↑Bcl-2 ↓Bax and cytochrome c | [33] | |
| Naringenin | In vivo | male Wistar rats’ model of AD | ↑SOD ↑GPx ↑GST ↑CAT ↑GR ↑Na+/K+ ATPase | [42] |
| ↓MDA levels | [43] | |||
| In vivo, In vitro | male C57BL/6 mice model of PD, human neuroblastoma cell line (SH-SY5Y) | ↑GSH levels ↑Nrf2/ARE pathway ↑ARE pathway genes ↓ROS formation ↑Nrf2 protein levels | [44] | |
| In vitro | neurons isolated from the brains of Sprague-Dawley rats | ↓ROS ↑high-energy phosphates level ↑mitochondrial ANT transport activity ↑mitochondrial membrane potential ↑expression of Nrf2 ↑activation of the Nrf2/ARE | [45] | |
| Naringin | In vivo | male Wistar rats’ model of AD | ↑CAT ↑SOD ↑GSH ↓TNF-α ↓IL-1β | [46] |
| In vitro | pheochromocytoma cell line (PC12) | ↓LDH ↓lipid peroxidation ↓ROS generation ↑GPx ↑CAT ↑GR ↑SOD ↑GSH levels ↑Nrf2 activation ↑HO-1 and NQO-1 | [47] | |
| In vivo | male Wistar rats’ model of cognitive impairment | ↑CAT ↑SOD ↑GSH levels ↓TNF-α, ↓TGF-β, ↓IL-1β ↓NF-κβ p65 subunit, ↓caspase-3 | [48] | |
| In vitro | pheochromocytoma cell line (PC12) | ↓CYP2E1 ↓ROS rectify the antioxidant protein contents of Nrf2, HO-1, SOD2, and GSS | [49] | |
| In vivo | male Wistar rats’ model of cognitive impairment | ↓MDA ↓nitrite ↑CAT ↑SOD ↑GST ↑GSH levels | [50] | |
| Quercetin | In vivo | male Kunming strain mice model of brain aging process and learning and memory defect | ↑Nrf2/ARE ↑Nrf2 ↑HO-1 ↑SOD | [51] |
| In vitro | cerebellar granule neurons isolated from Sprague-Dawley rats | ↑GCLC gene expression ↑activation of the Nrf2 ↑GSH | [52] | |
| In vitro | human RPE cell line (ARPE-19) | ↑GPx ↓ROS and MDA ↑HO-1, NQO-1, and GCL ↑CAT ↑SOD ↑Nrf2 | [53] | |
| In vitro, In vivo | Sprague-Dawley male rats, Neuroepithelioma cell line (SK-N-MC) | ↑CAT ↑SOD ↓MDA ↑GSH levels ↓ROS | [54] | |
| In vivo | male Sprague-Dawley rats | ↑CAT ↑SOD ↓MDA ↑GSH levels ↑Nrf2/HO-1 pathway | [55] | |
| In vitro, In vivo | homozygotic transgenic mouse line B6.129S7-Sod2tm1Leb/J, hippocampal neurons isolated from Sprague-Dawley rat embryos | ↓ROS | [56] | |
| In vitro | human RPE cell line (ARPE-19) | ↑Nrf2 ↑HO-1 | [57] | |
| APP695-transfected SH-SY5Y cell line (APPswe), human neuroblastoma cell lines (SH-SY5Y) | ↓lipid peroxidation ↓MDA ↑GSH levels ↓ROS | [58] | ||
| Xanthohumol | In vitro | mouse microglia BV2 cells | ↑Nrf2-ARE signaling pathway activation ↑Nrf2 expression ↑HO-1 expression ↑GSH ↓NO, IL-1β, and TNF-α ↓NF-κB | [60] |
| Chalcone derivative | In vivo | scopolamine-induced mice model | ↑Nrf2/HO-1 protein expression ↓ROS ↑Superoxide dismutase activity | [61] |
| 2′,3′-Dihydroxy-4′,6′-dimethoxy-chalcone (DDC) | In vitro | cerebellar cortex neurons isolated from embryonic 17–19-day-old Wistar/ST rat fetuses | ↑Nrf2-ARE signaling pathway activation ↑Nrf2 expression ↑HO-1 expression | [62] |
| Rutin | In vitro | APP695-transfected SH-SY5Y cell line (APPswe), human neuroblastoma cell lines (SH-SY5Y) | ↓lipid peroxidation ↓MDA ↑GSH levels ↓ROS | [58] |
| Phloretin | In vivo | scopolamine-induced mice model | ↑CAT ↑SOD ↓MDA | [68] |
| Magnolol | ↑total nitric oxide synthase ↑AChE activity ↑SOD ↓methane dicarboxylic aldehyde | [69] | ||
| Morin | In vitro | mouse hippocampal nerve cells (HT22), Swedish mutant APP stable cell line (APP695-transfected SH-SY5Y) | ↓ROS levels ↓caspase-9, and -3 ↓β- and γ-secretase | [70] |
| Isoquercitrin | ↓ROS levels ↓caspase-9, and -3 ↓β- and γ-secretase | [70] | ||
| Pinocembrin | In vitro | human neuroblastoma cell lines (SH-SY5Y) | ↑Nrf2 protein levels ↑Nrf2/HO-1 pathway ↓ROS levels | [71] |
| Caffeic acid derivative | In vivo, In vitro | male Kunming mice model of learning and memory impairment human neuroblastoma cell lines (SH-SY5Y) | ↑GSH ↑SOD ↑HO-1 and NQO-1 ↑Nrf2 ↓protein carbonylation level ↓MDA | [72] |
| Caffeic acid derivative | In vivo | male C57Bl/6 mice model of AD | ↓ROS ↑Nrf2 mRNA ↑activation of the Nrf2 signal ↑HO-1 protein | [73] |
| Gallic acid | In vivo | Drosophila melanogaster model of AD | ↓activity of cholinesterases ↓MDA and ROS | [63] |
| Resveratrol | In vivo | Wistar rats’ model of combined AD and diabetes | ↑GSH levels ↑SOD ↓MDA levels | [64] |
| Resveratrol derivative | In vitro | pheochromocytoma cell line (PC12), mouse microglia BV2 cells | ↓NO ↓ROS | [65] |
| p-hydroxybenzyl alcohol | In vitro, In vivo | ICR mice model of AD, human neuroblastoma cell line (SH-SY5Y) | ↑Nrf2 protein levels ↑BDNF ↑GDNF | [66] |
| Taxifolin derivative | In vitro, In vivo | male Swiss mice model of AD, mouse hippocampal nerve cell (HT22) | ↑Nrf2 ↑GSH | [67] |
| Luteolin | In vitro | pheochromocytoma cell line (PC12), rat glioblastoma cell line (C6) | ↑Nrf2 ↑NQO1-ARE-response interfering ERK1/2 pathway | [74] |
| Farrerol | In vitro | mouse microglial BV-2 cells | ↑Nrf2/Keap1 pathway ↓ROS and MDA ↑SOD ↑SOD1 and SOD2 mRNA | [75] |
| Gastrodin | In vitro | hippocampal neurons isolated from Sprague-Dawley rats | ↑ SOD ↑mRNA expression of CAT ↑CAT ↑gene expression of Nrf2 ↑ERK1/2 phosphorylation | [76] |
| Baicalein | In vitro | pheochromocytoma cell line (PC12) | ↑Nrf2/HO-1 | [77] |
| Curcuma & garlic-derived hybrids | In vitro | human neuroblastoma cell line (SH-SY5Y) | ↑Nrf2 | [78] |
| Compounds | Types of Study | Cell Line(s)/Animal Model(s) | Mechanisms of Action | References |
|---|---|---|---|---|
| Berberine | In vitro | axonal transport impairment induced by calyculin A in wild-type mouse neuroblastoma-2a cell line (N2a) | ↑SOD ↓MDA ↓tau and NFs hyperphosphorylation, ↑cell metabolism, ↑cell viability ↑PP-2A activity ↑NF axonal transport | [81] |
| glutamate-induced oxidative stress and apoptosis in pheochromocytoma cells (PC12) and neuroblastoma-2a (N2a) cell lines | ↓ROS ↓MDA ↑SOD ↑GSH ↓Bax/Bcl-2 ↓caspase-3 ↓DNA fragmentation ↑cell viability | [82] | ||
| homocysteic acid-induced neuronal cell death in murine hippocampal neuronal cell line (HT-22) | ↓ROS, ↓LDH, ↓nuclear condensation, ↓necrotic death, ↓cell apoptosis, ↑cell survival, ↑phosphorylated Akt | [83] | ||
| In vivo | LPS-induced learning and memory dysfunctions in the male albino Wistar rats | ↑GSH, ↑GPx, ↑SOD, ↑CAT, ↓MDA, ↓3-NT, ↓NF-κB, ↓TLR4, ↓TNF-α, ↓IL-6, ↓COX-2, ↓AChE, ↓DNA fragmentation, ↓caspase-3 ↓protein carbonyl, ↓GFAP, ↑sirtuin 1, ↓p38 MAPK, ↑spatial recognition memory | [84] | |
| streptozotocin-induced dementia in male Wistar rats | ↓ROS, ↑GSH, ↑T-SHs, ↑GST activity, ↓TBARS, ↓protein carbonyl levels, ↑δ-ALA-D, ↑NTPDase, ↑5ʹ-nucleotidase activity, ↑ADA activity, ↑recognition index | [87] | ||
| Trigonelline | In vivo | Aβ1–40 induced AD in adult male Swiss albino mice | ↑SOD, ↑GSH, ↑MMP, ↓MDA, ↓protein carbonyl levels, ↓LDH, ↓COX-2, ↓GFAP, ↓TNF-α, ↓IL-6, ↑spatial recognition memory | [88] |
| LPS-induced cognitive impairment in the male albino Wistar rats | ↑SOD, ↑GSH, ↓MDA, ↓AChE, ↓TNF-α, ↓IL-6, ↑BDNF, ↑spatial learning, and memory | [89] | ||
| Tetramethylpyrazine (Ligustrazine) | In vitro | cobalt chloride-induced neurotoxicity in PC12 cells | ↓mitochondrial and intracellular superoxide, ↓ROS, ↑GSH, ↓GSSG, ↑nuclear Nrf2 expression, ↑GCLc expression, ↑Nrf2 transcription activity, ↑ARE-luciferase, ↑cell viability, ↓Bax, ↓cleavage of caspase-3 and -9, ↓PARP, ↑Bcl-2, ↓cytochrome c, ↓cell apoptosis, ↓HIF-1α/NOX2 pathway | [90] |
| In vivo | cobalt chloride-induced neurotoxicity in adult male Wistar rats | ↑Nrf2, ↓HIF-1α, ↓NOX2 protein expression, ↓cell apoptosis, ↑spatial learning and memory | ||
| Ligustrazine phosphate | In vivo | scopolamine-induced amnesia in male Sprague-Dawley rats | ↑SOD, ↑GPx, ↓MDA, ↑behavioral performance | [91] |
| In vitro | abdominal skins of male Sprague-Dawley rats | ↑penetration ability, ↑drug deposition in skin | ||
| Ligustrazine phosphate and huperzine A | In vivo | scopolamine-induced amnesia in male Sprague-Dawley rats | ↑SOD, ↑GPx, ↓MDA, ↑spatial memory | [92] |
| Huperzine A | In vitro | tert-butyl hydroperoxide-induced oxidative stress in pheochromocytoma cells (PC12) Aβ25–35-induced neurotoxicity in PC12 cells | ↓ROS, ↑SOD, ↓LDH, ↑cell viability, ↓morphological damage ↑cell viability | [93] |
| Deoxyvasicine | In vivo | scopolamine-induced cognitive dysfunction in male C57BL/6J mice | ↑GPx, ↓TNF-α, ↓AChE, ↑ChAT, ↑BDNF, ↑ACh, ↑spatial learning and memory | [98] |
| HJ22 (a novel derivative of piperine) | In vivo | ibotenic acid-induced cognitive impairment in Sprague-Dawley rats | ↓PPI of Keap1-Nrf2, ↑nuclear Nrf2 expression, ↑SOD, ↑CAT activities, ↑GR, ↓MDA, ↑ARE, ↓IL-1β, ↓TXNIP, ↓NLRP3, ↓apoptotic cell death, ↓AChE, ↑ChAT, ↑ACh, ↑Bcl-2/Bax ratio, ↑Nissl body, ↓ASC, ↓caspase-1 | [94] |
| Radical-containing nanoparticles coupled with piperine | In vitro | Aβ1–42-induced damage in human neuroblastoma SH-SY5Y cells | ↓ROS, ↓hydroxyl radical production, ↑GPx, ↑CAT, ↓MDA, ↓protein carbonyl levels, ↓8-OHdG, ↓DNA fragments, ↑cell viability | [95] |
| Fangchinoline | In vitro | glutamate-induced oxidative neuronal damage in mouse neuronal cells (HT-22) | ↓ROS overproduction, ↑SOD activity, ↑Nrf2 protein level, ↓Keap1 expression, ↑HO-1 protein level, ┴cell morphological damages, ↑cell viability, and regulating Keap1/Nrf-2 antioxidation signaling pathway | [97] |
| Dauricine | In vitro | Cu2+ induced oxidative damage on APPsw cells | ↑nuclear Nrf2, ↓Keap1 expression, ↑cell viability, ↓ROS levels, ↑SOD activity, ↑MMP level, ↓Aβ1–42 secretion, ↓Bax/Bcl-2 ratio, ↓caspase-3 activity, ↓apoptotic rate | [96] |
| Aβ1–42-transgenic Caenorhabditis elegans GMC101 | ↓oxidative toxicity of Aβ, ↑survival rates | |||
| Plumbagin | In vivo | streptozotocin-induced AD in adult male Swiss-albino mice | ↑activation of Nrf2/ARE pathway, ↓astrogliosis, ↓GFAP expression, ↑spatial learning and memory | [99] |
| In silico | ↓β-secretase enzyme | |||
| Embelin | In vivo | scopolamine-induced amnesia in Sprague-Dawley rats | ↑SOD1, ↑CAT, ↓4-HNE, ↑immature neurons in the SGZ, ↑BDNF expression, ↑CREB1, ↑ACh, ↓Glu, ↓Dopamine, ↓NE, ↑recognition index, ↑memory retention, | [100] |
| Harmaline | In vivo | scopolamine-induced memory impairments in male C57BL/6 mice | ↑SOD, ↑GPx, ↓MDA level, ↓MPO, ↓NO, ↓TNF-α, ↓AChE activity, ↑ChAT activity, ↑ACh, ↑L-Trp, ↑5-HT, ↑L-Glu, ↓γ-GABA, ↑spatial learning and memory,↓MDA level, ↓TNF-α, ↑ChAT activity, ↑ACh, ↑L-Trp, | [102] |
| Isorhynchophylline | In vitro | Aβ25–35-induced neurotoxicity cells (PC12) | ↑cell viability, ↓ROS levels, ↑GSH, ↓MDA levels, ↑MMP level, ↓DNA fragmentation, ↓caspase-3 activity, ↑Bcl-2/Bax ratio | [103] |
| Aloperine | In vitro | neuroblastoma N2a cells co-transfected with Swedish mutant APP and ΔE9 deleted presenilin-1 (N2a/Swe.D9) H2O2-induced secondary insults in N2a/Swe.D9 cells | ↑intracellular GSH levels, ↑GPx activity, ↓generation of ROS, ↓4-HNE, ↑MMP level, ↑intracellular ATP level ↑cell viability, ↓apoptosis, ┴LDH release, ↓translocation of cytochrome c, ↓Bax/Bcl-2 ratio, ↓caspase-3 activity, ┴p38-JNK pathway | [104] |
| Matrine | In vivo | scopolamine-induced amnesia in male ICR mice | ↑T-AOC, ↑SOD, ↑CAT, ↓MDA, ↓AChE activity, ↓BuChE activity, improve learning and memory | [105] |
| Methyl jasmonate | In vivo | scopolamine-induced cognitive impairment in male Swiss mice | ↑SOD, ↑CAT, ↑GSH, ↓MDA, ↓AChE activity, ↑spatial working memory, ↑recognition memory, ↑alternation behaviors, | [106] |
| Neferine | In vivo | aluminium chloride-induced AD in Wistar rats | ↓ROS formation, ↑SOD, ↑CAT, ↑GSH, ↓MDA, ↓LDH, ↓NO, ↓AChE activity, ↓Na+K+ATPase activity, ↓TNF-α, ↓IL-1β, ↓IL-6, ↓iNOS, ↓COX-2, ↓NF-κB, ↑IKBα, ↑memory and learning ability | [107] |
| Norcepharadione B | In vitro | hydrogen peroxide (H2O2)-induced neuronal injury in HT-22 mouse neuronal cells | ↑SOD, ↑GSH, ↓MDA, ↓LDH activity, ↑HO-1, ↑Bcl-2/Bax ratio, ↓VSOR Cl− currents, ↓cell apoptosis, ↓cell volume change, ↑phosphorylated Akt | [108] |
| Vincamine | In vitro | Aβ25–35 induced cytotoxicity in PC12 cells | ↓ROS levels, ↑SOD, ↑GSH, ↓MDA, ↑Bcl-2/Bax ratio, ↑phospho-Akt/Akt ratio, ↑cell viability, ↓cell apoptosis | [109] |
| Corydalis edulis total alkaloids | In vivo | d-gal induced AD in Sprague-Dawley male rat | ↓ROS, ↑SOD, ↑CAT, ↓MDA, ↓TNF-α, ↓IL-1β, ↓Aβ accumulation, ↓NF-κBp65 expression, ↑MAP2, ↑memory and learning ability | [110] |
| girinimbine, mahanimbine and murrayanine | In vivo | Ageing-induced oxidative stress in male Swiss albino mice | ↑GPx, ↑GSH, ↑GRD, ↑SOD, ↑CAT, ↓LPO level, ↓NO levels, ↑ACh, ↓AChE activity | [111] |
| Compounds | Classification | Types of Study | Cell Line(s)/Animal Model(s) | Mechanisms of Action | References |
|---|---|---|---|---|---|
| Carvacrol | monoterpene | In vivo, In vitro | male C57BL/6 mice, hippocampal neurons isolated from neonatal C57BL/6 mice | ↑GPx ↑CAT ↑SOD ↓MDA ↑GSH ↓ROS | [113] |
| α-Pinene | ↓ROS | [114] | |||
| 1,8-Cineole | monoterpene | In vitro | pheochromocytoma cell line (PC12) | ↑CAT ↑SOD ↑GPx ↑GR ↑HO-1 | |
| p-Cymene | monoterpene | In vivo | male Swiss mice | ↓nitrite ↓lipid peroxidation ↑CAT ↑SOD | [115] |
| Linalool | monoterpenoid | In vivo | male C57BL/6 J mice | ↑GPx ↑SOD ↑Nrf2/HO-1 | [116] |
| Thymol | ↑Nrf2/HO-1 signaling | [117] | |||
| Carvacryl Acetate | monoterpenoid | In vivo, In vitro | male Swiss albino mice, hippocampal neurons isolated from Swiss albino mice | ↓lipid peroxidation ↓nitrite contents ↓hydroxyl radical contents ↑GSH ↑CAT ↑GPx ↑ SOD | [118] |
| Borneol | monoterpenoid | In vitro | human neuroblastoma cell line (SH-SY5Y) | ↓ROS ↑Nrf2 ↑HO-1 | [119] |
| Geraniol | ↑GSH ↓ROS | [120] | |||
| ECN | sesquiterpenoid | In vitro, In vivo | pheochromocytoma cell line (PC12) Male ICR mice | ↑Nrf2 ↑HO-1 ↑Nrf2/ARE signaling | [121] |
| In vitro | murine microglial cell line (BV-2) | ↓ROS production | [122] | ||
| Lactucopicrin | sesquiterpenoid | In vitro | neuroblastoma cell lines (N2a), Rat glioblastoma cell line (C6) | ↑Nrf2 ↑nerve growth factor ↓ROS ↑mAChR, p-Akt, and Bcl-2 | [123] |
| α-Cyperone | sesquiterpenoid | In vitro | murine microglial cell line (BV-2), human neuroblastoma cell line (SH-SY5Y), mouse hippocampal nerve cells (HT22) | ↑Akt/Nrf2/HO-1 ↑nuclear tanslocation of Nrf2 | [124] |
| Artemether | sesquiterpenoid | In vitro, In vivo | homozygous 3xTg-AD mouse (34,830-JAX) model of AD, human neuroblastoma cell line (SH-SY5Y), pheochromocytoma cell line (PC12) | ↑HO-1 ↑SOD ↑Nrf2 ↓MDA | [125] |
| Bakkenolide B | sesquiterpenes | In vitro | LPS-induced neuroinflammation in mouse BV2 microglial cells | ↑Nrf2, ↑HO-1, ↑NQO1, ↓ROS production, ↑AMPK phosphorylation, ↓IL-1β, ↓IL-6, ↓IL-12, ↓TNF-α, ↓NO, ↓iNOS, ↑cell viability | [126] |
| Carnosic acid | diterpene | In vitro | human neuroblastoma cell line (SH-SY5Y) | ↑PI3K/Akt ↑Nrf2 | [127] |
| In vitro | rat pheochromocytoma subclone cell line (PC12h) | ↑Nrf2 | [128] | ||
| In vitro, In vivo | male C57BL/6 mice, Rat pheochromocytoma subclone cell line (PC12h) | ↑Nrf2/ARE pathway | [129] | ||
| In vitro | human neuroblastoma cell line (SH-SY5Y) | inducing the metalloprotease gene TACE/ADAM17 | [130] | ||
| Andrographolide | diterpenoid | In vitro | mouse hippocampal nerve cells (HT22) | ↑Nrf2/ARE/HO-1 pathway | [131] |
| pheochromocytoma cell line (PC12) | ↑Nrf2-mediated p62 signaling pathway | [132] | |||
| Compound K | triterpenoid | In vitro, In vivo | male C57BL/6 mice, mouse hippocampal nerve cells (HT22) | ↑Nrf2 ↑HO-1 ↑quinone oxidoreductase 1 | [133] |
| Ginsenoside | triterpenoi | In vitro | human neuroblastoma cell line (SH-SY5Y) | ↑activation Nrf2 | [134] |
| Gedunin | triterpenoid | In vitro | immortalized microglial cell line (IMG), human neuroblastoma cell line (SH-SY5Y) | ↓NO ↓NF-κB ↑Nrf2 | [135] |
| Lycopene | tetraterpene | In vitro, In vivo | male C57BL/6J mice, murine microglial cell line (BV-2) | ↑Nrf2 ↑HO-1 ↑NQO-1 | [136] |
| Gypenoside xvii | tetraterpenoid | In vitro | pheochromocytoma cell line (PC12) | ↑Nrf2/ARE/HO-1 pathways | [137] |
| Compounds | Types of Study | Cell Line(s)/Animal Model(s) | Mechanisms of Action | References |
|---|---|---|---|---|
| Astaxanthin | In vitro | glutamate-induced neurotoxicity in mouse neuronal cell line (HT-22) | ↓intracellular ROS accumulation, ↑ARE, ↑nuclear Nrf2, ↑HO-1, ↑Bcl-2/Bax ratio, ↓PARP, ↓caspase-3/8/9 activity, ↓cytochrome c, ↓LDH, ↓AIF, ↑p-Akt, ↑p-GSK-3β (Ser9), ↑cell viability | [144] |
| In vivo | scopolamine-induced spatial learning deficits in Swiss albino male mice | ↑SOD, ↑CAT, ↓NO, ↑spatial learning and memory | [145] | |
| In vivo | Aβ1–42-induced AD in adult male Wistar rats | ↑GPx, ↓MDA, ↓superoxide anion, ↓protein carbonyl levels, ↓neuronal degeneration, ↓positive staining of Aβ, ↑spatial learning and memory | [146] | |
| In vivo | LPS-induced mice AD model | ↓ROS, ↑GSH, ↑GSH/GSSG ratio, ↓thiobarbituric acid, ↓NO, ↓β-secretase activity, ↓APP level, ↓BACE1, ↓Aβ1–42, ↓COX-2, ↓GFAP, ↓IBA-1, ↓iNOS, ↓TNF-α, ↓IL-1β, ↓IL-6, ↓MCP-1, ↓MIP-1α, ↓MIP-1β, ↓STAT3, ↑spatial learning and memory | [147] | |
| In vitro | BV-2 microglial cells | ↓NO, ↓TBARS, ↓β-secretase, ↓APP level, ↓BACE1, ↓COX-2, ↓IBA-1, ↓iNOS, ↓TNF-α, ↓IL-1β, ↓IL-6, ↓MCP-1, ↓MIP-1α, ↓MIP-1β, ↓STAT3 | ||
| In vitro | tert-butyl hydroperoxide- induced oxidative stress in pheochromocytoma cell line (PC12) | ↓ROS, ↑SOD, ↓MDA, ↓LDH release, ↑cell viability, ↓morphological damage | [93] | |
| Aβ25–35-induced neurotoxicity in PC12 cells | ↑cell viability | |||
| Astaxanthin and Huperzine A | In vitro | tert-butyl hydroperoxide- induced oxidative stress in PC12 cells Aβ25–35-induced neurotoxicity in PC12 cells | ↓ROS, ↑SOD, ↓MDA, ↓LDH, ↑cell viability, ↓morphological damage ↑cell viability | [93] |
| Crocin | In vitro | L-glutamate-damaged HT-22 mouse neuronal cells | ↓intracellular ROS, ↓MMP dissipation, ↓overload of Ca2+, ↑Bcl-xL, ↓Bax, ↓Bad, ↓cleaved caspase-3, ↓apoptosis rate, ↑cell viability, ↑phosphorylation of Akt and mTOR | [150] |
| In vivo | AlCl3/d-gal-induced AD in BALB/c mice | ↓ROS, ↑GPx, ↑SOD, ↓Aβ1–42 deposition, ↓AChE, ↑ChAT, ↑ACh, ↑memory abilities and cognitive functions | ||
| In vivo | malathion-induced spatial memory deficits in adult male Wistar rats | ↑GSH, ↓MDA, ↓TNF-α, ↓IL-6, ↓tau hyperphosphorylation, ↑PSD93 protein level, ↓caspase-3/8/9 activity, ↓Bax/Bcl-2 ratio, ↓cell apoptosis, ↑spatial learning and memory | [152] | |
| streptozotocin-induced spatial memory deficit and oxidative stress in adult male Wistar Albino rats | ↑GPx activity, ↑total thiol concentration, ↓MDA | [153] | ||
| [154] | ||||
| Lycopene | In vitro | LPS-treated BV2 microglial cells | ↓intracellular ROS generation, ↑MMP, ↑Nrf2, ↑HO-1 expression, ↑NQO-1 expression, ↓p-ERK, ↓p-JNK, ↓p-p38, ↓p-AKT, ↓NF-κB nuclear translocation, ↓p-IκB, ↑Nrf2 | [136] |
| In vivo | LPS-induced learning and memory loss in male C57BL/6J mice | ↑GSH, ↑SOD, ↑CAT, ↓Aβ1–42 accumulation, ↓APP level, ↓BACE1 expression, ↑ADAM10, ↓IBA-1, ↓COX-2, ↓iNOS, ↓IL-1β, ↑IL-10, ↓MMP-9 expression, ↑spatial learning and memory | ||
| In vivo | Tau transgenic mice expressing P301L mutation | ↑GPx activities, ↓MDA levels, ↓tau hyperphosphorylation, ↑spatial and passive memory | [156] | |
| Strigolactone analogue (GR24rac) | In vitro | LPS-treated SIM-A9 mouse microglial cells | ↑Nrf2 nuclear level, ↑HO-1, ↑NQO-1, ↓NO, ↓iNOS, ↓TNF-α, ↓IL-1β, ↓COX-2, ↓NF-κB, ↑PPARγ expression | [160] |
| LPS-treated BBB bEnd.3 mouse brain endothelial cells | ↓TNF-α, ↓IL-1β, ↑NQO-1 | |||
| β-carotene | In vivo | streptozotocin-induced AD in adult male Swiss albino mice | ↑GSH, ↑SOD, ↑CAT, ↓GSSG/GSH ratio, ↓AChE activity, ↓Aβ1–40 and Aβ1–42 levels, ↑cognitive performance | [157] |
| In silico | ↓AChE activity | |||
| Levocarnitine | In vivo | AlCl3-induced spatial working memory deficits in adult male Swiss albino mice | ↓GSH, ↓MDA, ↓NO, ↓AOPP levels, ↑spatial working memory performance | [158] |
| Compounds | Types of Study | Cell Line(s)/Animal Model(s) | Mechanisms of Action | References |
|---|---|---|---|---|
| Sulforaphane | In vitro | mouse neuroblastoma cell line (N2a) | ↓ROS ↓MDA ↑SOD ↑Nrf2 | [164] |
| human neuroblastoma cell line (SH-SY5Y) | ↑GSH ↑GR ↑glutathione transferase | [165] | ||
| hippocampal neuron isolated from C57Bl6J mice | ↑activation of Nrf2/ARE pathway | [166] | ||
| neuronal cell isolated from the Wistar rat’s striatum | ↑HO-1 ↑GSH ↑Nrf2/ARE pathway | [167] | ||
| pheochromocytoma cell line (PC12) | ↑HO-1 ↑translocation of Nrf2 ↑PI3K/Akt | [168] | ||
| In vivo | male C57Bl/6 mice model of PD | ↑GSH ↑GST ↑GR | [169] | |
| In vitro, In vivo | male C57Bl/6 mice model of PD, human neuroblastoma cell line (SH-SY5Y) | ↓ROS ↓MDA ↑GSH ↑Nrf2 ↑HO-1 | [170] | |
| In vitro | human microglia-like THP-1 cells | ↑Nrf2/HO-1 | [171] | |
| astrocyte isolated from (P1eP2) Sprague-Dawley rats | [173] | |||
| pheochromocytoma cell line (PC12) | [174] | |||
| S-allyl cysteine | In vivo | Swiss albino mice model of experimental dementia of Alzheimer’s type | ↑GSH ↑GPx | [175] |
| In vitro, In vivo | Nrf2 heterozygous mice, Nrf2 knockout mice, neuronal cell isolated from the Sprague-Dawley rat embryos | ↑Nrf2-dependent antioxidant responses | [176] | |
| In vivo | male C57BL/6 mice | ↑Nrf2 transcription factor | [177] | |
| In vitro | pheochromocytoma cell line (PC12) | ↓apoptosis | [178] | |
| In vitro | pheochromocytoma cell line (PC12) | ↑mitochondrial membrane potential | [179] | |
| In vitro | hippocampal and cerebellar granule neurons isolated from embryos of Wistar rats | ↓ROS | [180] | |
| S-ethyl cysteine, S-propyl cysteine | In vivo | male C57BL/6 mice | ↓MDA ↑GSH ↓ROS ↑GPx ↑SOD ↑CAT | [181] |
| 6-(Methylsulfinyl) hexyl isothiocyanate | In vitro | neuronal cell isolated from the Wistar rat’s striatum | ↑HO-1 ↑GSH ↑Nrf2/ARE pathway | [167] |
| In vivo | male C57Bl/6 mice model of AD | ↓ROS ↑GSH ↑Nrf2/ARE pathway | [182] | |
| Allicin | In vitro | human RPE cell line (ARPE-19) | ↓ROS ↓MDA ↑GSH/glutathione disulfide ratio | [183] |
| In vivo | male C57BL/6 mice | ↑Nrf2/ARE ↑GSH levels ↓ROS levels ↑GPx | [184] | |
| In vivo | male Sprague-Dawley rats | ↑PERK and Nrf2 ↓ROS levels ↑GSH level ↓lipid peroxidation | [185] | |
| Thiacremonone | In vitro, In vivo | APP/PS1 transgenic mice model, neuronal cells isolated from the Sprague–Dawley (SD) rats | ↑GSH ↓NF-κB | [186] |
| 3H-1,2-Dithiole-3- thione | In vivo | Tg2576 AD mouse model | ↑Nrf2 ↑HO-1 ↑Sirt1/Nrf2 | [187] |
| Hydrogen sulfide | In vitro | mouse hippocampal nerve cell line (HT22) | ↑GSH ↑cysteine ↑KATP channels ↑Cl− channels | [188] |
| Lipoic acid | In vitro, In vivo | female C57BL/6 mice, retinal neuronal cell line (RGC-5) | ↑Nrf2 ↑HO-1 ↑Keap1/Nrf2 ↓ROS | [189] |
| Compounds | Classification | Types of Study | Cell Line(s)/Animal Model(s) | Mechanisms of Action | Reference |
|---|---|---|---|---|---|
| Compound K | ginsenoside | In vivo | scopolamine hydrobromide-induced memory impaired in ICR mice | ↑Nrf2, ↓Keap1, ↑HO-1, ↑SOD, ↑GPx, ↓MDA, ↓Aβ expression, ↓neuronal apoptosis, ↓Bax, ↑Bcl-2, ↓caspase-3 activity, ↓APP expression, ↓BACE1, ↓PS1 expression, ↑spatial cognition and memory function, normalize neuronal morphology | [190] |
| In vitro | glutamate-induced cytotoxicity in mouse hippocampal cells (HT22), | ↑Nrf2, ↑HO-1, ↑NQO1, ↑GR, ↓apoptotic cells | [133] | ||
| In vivo | scopolamine-induced memory impaired in male C57BL/6 mice | ↑Nrf2-mediated antioxidant enzyme, ↑spatial learning and memory | |||
| 20(S)-Protopanaxadiol | ginsenoside | In vivo | scopolamine-induced memory deficit in ICR male mice | ↑SOD, ↓MDA, ↓AChE, ↑ACh, ↑spatial learning and memory | [191] |
| Ginsenoside Rd | ginsenoside | In vitro | Aβ25–35-induced neuronal damage in primary cultured hippocampal neurons | ↓ROS, ↑SOD, ↑GPx, ↓MDA, ↓loss of hippocampal neurons, ↓cytochrome c, ↓Bax, ↑Bcl-2, ↑neuronal survival | [193] |
| 20(S)-Protopanaxatriol | ginsenoside | In vivo | scopolamine-induced cognitive deficits in male mice | ↑SOD, ↓MDA, ↓AChE, ↑ACh content, ↑ChAT activity, ↑spatial memory | [194] |
| Ginsenoside Rg1 | ginsenoside | In vivo | chronic restraint stress-induced learning and memory impairments in adult male Kunming mice | ↓ROS, ↑SOD, ↓MDA, ↓8-OHdG, ↓NOX2 expression, ↑learning and memory function | [195] |
| Pseudoginsenoside- F11 | saponin | In vivo | Tg-APPswe/PS1dE9 (APP/PS1) mice, | ↑SOD, ↑GPx, ↓MDA, ↓APP, ↓Aβ1–40, ↓caspase-3 activity, ↓JNK 2, ↓p53, ↓learning and memory impairment | [197] |
| Aβ1–42-induced AD in male Kunming mice | ↑spatial learning and memory | ||||
| Timosaponin B-II | saponin | In vivo | scopolamine-induced AD in male Kunming mice | ↑SOD, ↑GPx, ↓MDA, ↓AChE, ↑spatial learning and memory | [198] |
| Aloe-emodin | anthraquinone | In vitro | hydrogen peroxide (H2O2)-induced cytotoxicity in PC12 cells, | ↓intracellular ROS accumulation, ↓NO, ↓LDH, ↑cell viability | [199] |
| In vivo | scopolamine-induced memory impairment in Kunming mice | ↑SOD, ↑GPx, ↓MDA, ↓AChE, ↑ACh content, ↑spatial learning and memory | |||
| Methysticin | kavalacton | In vivo | 52-weeks old transgenic APP/Psen1 mice | ↑HO-1, ↑GCLC expression, ↑Nrf2/ARE pathway, ↓microglia activation, ↓astrogliosis, ↓GFAP, ↓IBA-1, ↓TNF-α, ↓IL-17A, ↓memory loss | [200] |
| α-Tocopherol | vitamins | In vitro | Aβ1–42-induced neurotoxicity in SH-SY5Y neuroblastoma cells | ↑Nrf2, ↓iNOS, ↓APP processing, ↑cell viability | [201] |
| α-Tocopherol quinine | vitamins | In vivo | Memory impairment in APPswe/PS1dE9 transgenic mice (transgenic mice with AD) | ↓ROS, ↑SOD, ↓MDA, ↓NF-κB, ↓IBA-1 protein levels, ↓iNOS, ↓IL-1β, ↓IL-6, ↓Aβ oligomer levels, ↓microglia activation, ↑spatial cognitive performance | [202] |
| In vitro | microglial cells (BV-2) | ↓NF-κB, ↓IBA-1 | |||
| α-Linolenic acid | fatty acid | In vitro | Aβ25–35-induced neurotoxicity in C6 glial cells | ↑Nrf2, ↑HO-1, ↓ROS, ↑neprilysin, ↑IDE expression, ↓NO, ↓TNF-α, ↓IL-6, ↓iNOS, ↓COX-2, ↓Aβ accumulation, ↑cell viability | [203] |
| Chitosan | polysaccharide | In vitro | H2O2/FeSO4- induced cell death in the NT2 neural cells | ↑Nrf2, ↑HO-1, ↑GSH, ↑γ-GCS, ↑Hsp-70, ↓NF-κB, ↓caspase-3, ↓Aβ formation, ↑cell viability | [211] |
| Shikonin | naphthoquinone | In vitro | Aβ1–42-induced neurotoxicity in PC12 cells | ↓ROS, ↑SOD, ↑GPx, ↑CAT, ↓MDA, ↑MMP, ↓LDH, ↓caspase-3, ↑Bcl-2/Bax ratio, ↑cell viability | [207] |
| Lycium barbarum polysaccharide | polysaccharide | In vitro | H2O2- induced neurotoxicity in PC12 cell | ↑Nrf2/HO-1, ↑ARE-luciferase activity, ↓ROS, ↓mitochondrial apoptosis, ↓caspase-3 and -9 activity, ↑cell viability | [212] |
| In vivo | CoCl2-induced neurotoxicity in male Wistar rats | ↑Nrf2/HO-1 expression, ↓apoptosis, ↑spatial learning and memory abilities | |||
| Amanita caesarea polysaccharides | polysaccharide | In vitro | glutamate-induced cytotoxicity in HT22 mouse hippocampal neuronal cells, | ↓intracellular ROS accumulation, ↑Nrf2, ↓Keap1, ↑HO-1, ↑GCLC expression, ↓cytochrome c, ↑MMP, ↓Bax, ↑Bcl-2, ↓caspase-3, ↑cell viability, ↓apoptotic rate, | [213] |
| In vivo | AlCl3/d-gal-induced AD in BALB/c male mice | ↓ROS, ↑SOD, ↑GPx content, ↓Aβ1–42 deposition, ↓AChE, ↑ACh content, ↑ChAT activity, ↓memory impairment | |||
| Inonotus obliquus polysaccharides | polysaccharide | In vitro | l-glutamic acid-induced cytotoxicity in HT22 mouse hippocampal neuronal cells, | ↑Nrf2, ↓Keap1, ↑HO-1, ↑SOD-1, ↑GCLC, ↓intracellular ROS accumulation, ↓LDH, ↑MMP, ↓Bax, ↑Bcl-2, ↓caspase-3 activity, ↑cell viability, ↓apoptotic rate | [214] |
| In vivo | APP/PS1 transgenic male mice | ↓ROS, ↑SOD, ↑GPx content, ↓MDA, ↑Nrf2, ↓Keap1, ↑HO-1, ↑SOD-1, ↑GCLC levels, ↓Aβ1–42 deposition, ↓neuronal fiber tangles deposition, ↓4-HNE, ↑memory and cognition function | |||
| Schisanhenol | tannin | In vivo | scopolamine-induced cognitive impairment in male Kunming mice | ↑SOD, ↑GPx, ↓MDA, ↓AChE activity, ↓phosphorylated Tau protein, ↑Sirtuin 1 expression, ↑PGC-1α, ↑learning and memorial ability | [215] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fakhri, S.; Pesce, M.; Patruno, A.; Moradi, S.Z.; Iranpanah, A.; Farzaei, M.H.; Sobarzo-Sánchez, E. Attenuation of Nrf2/Keap1/ARE in Alzheimer’s Disease by Plant Secondary Metabolites: A Mechanistic Review. Molecules 2020, 25, 4926. https://doi.org/10.3390/molecules25214926
Fakhri S, Pesce M, Patruno A, Moradi SZ, Iranpanah A, Farzaei MH, Sobarzo-Sánchez E. Attenuation of Nrf2/Keap1/ARE in Alzheimer’s Disease by Plant Secondary Metabolites: A Mechanistic Review. Molecules. 2020; 25(21):4926. https://doi.org/10.3390/molecules25214926
Chicago/Turabian StyleFakhri, Sajad, Mirko Pesce, Antonia Patruno, Seyed Zachariah Moradi, Amin Iranpanah, Mohammad Hosein Farzaei, and Eduardo Sobarzo-Sánchez. 2020. "Attenuation of Nrf2/Keap1/ARE in Alzheimer’s Disease by Plant Secondary Metabolites: A Mechanistic Review" Molecules 25, no. 21: 4926. https://doi.org/10.3390/molecules25214926
APA StyleFakhri, S., Pesce, M., Patruno, A., Moradi, S. Z., Iranpanah, A., Farzaei, M. H., & Sobarzo-Sánchez, E. (2020). Attenuation of Nrf2/Keap1/ARE in Alzheimer’s Disease by Plant Secondary Metabolites: A Mechanistic Review. Molecules, 25(21), 4926. https://doi.org/10.3390/molecules25214926










