Phytochemical Characterization and In Vitro Anti-Inflammatory, Antioxidant and Antimicrobial Activity of Combretum Collinum Fresen Leaves Extracts from Benin
Abstract
1. Introduction
2. Results and Discussion
2.1. Structure Elucidation of Compounds in the 50% Ethanol Extract from C. collinum Leaves and Phytochemical Analysis of the n-Hexane Extract
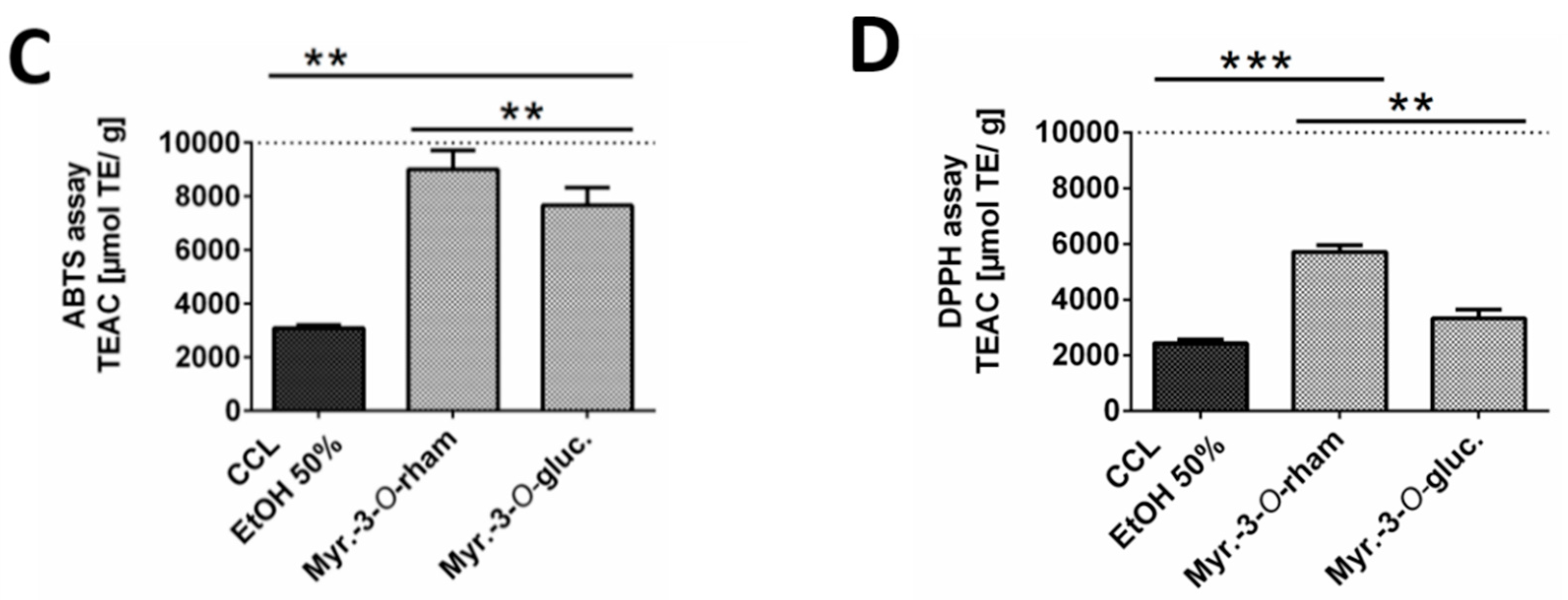
2.2. In-Vitro Antioxidant Activity of C. collinum EtOH 50% Leaves Extract
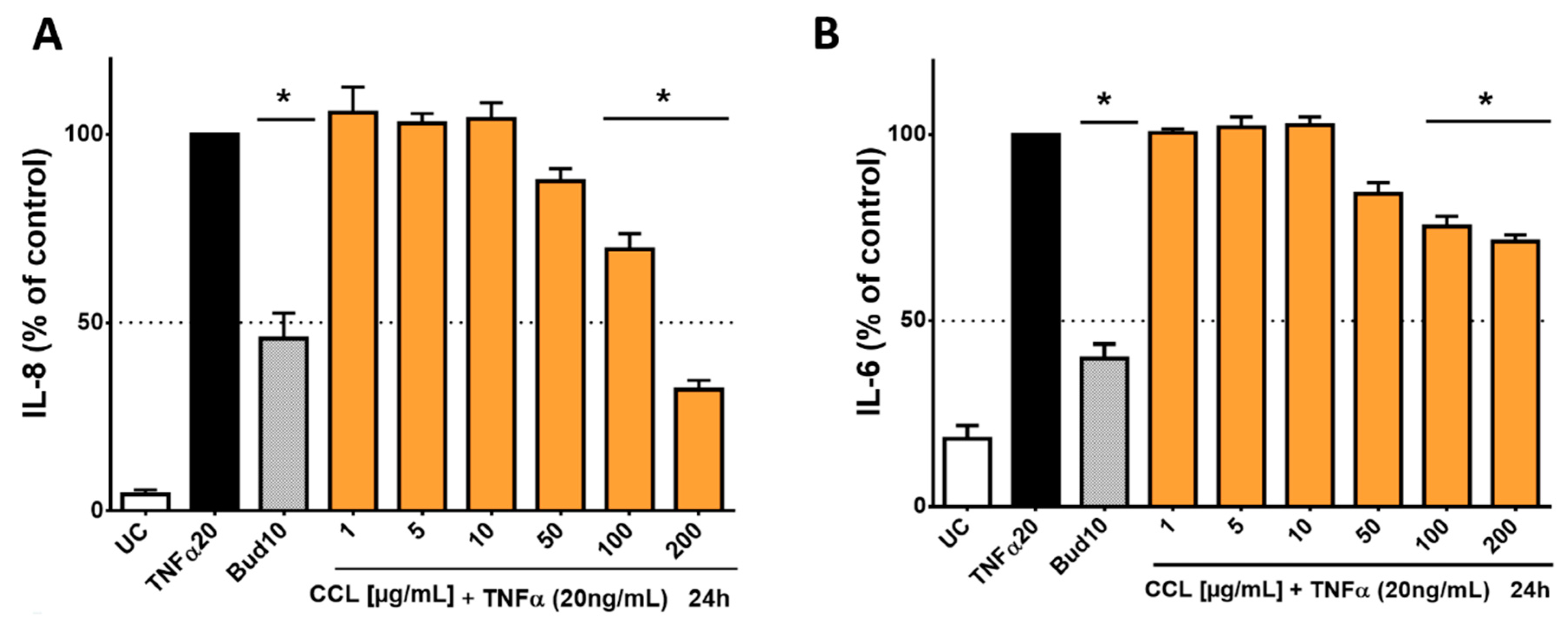
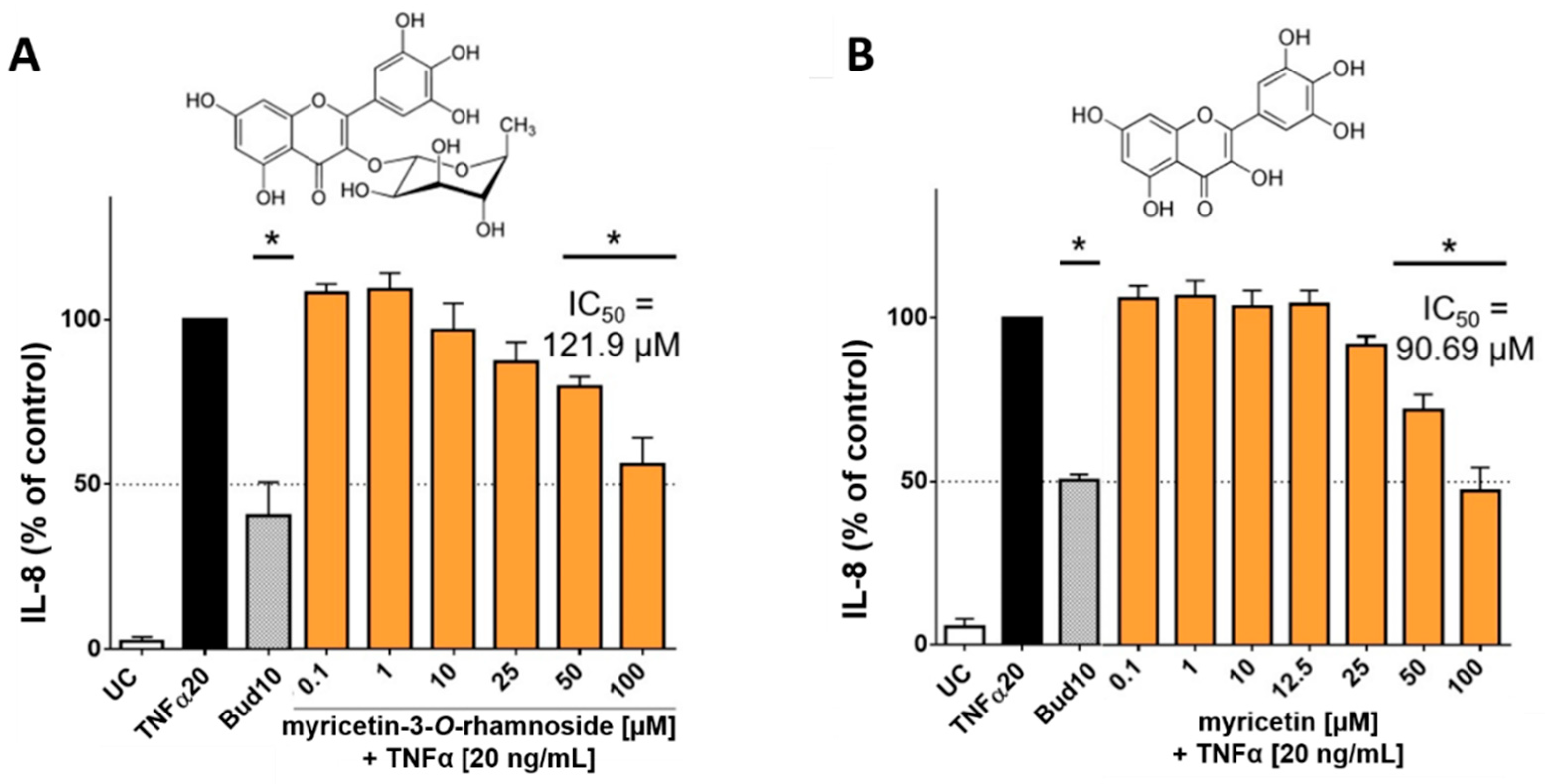
2.3. In-Vitro Anti-Inflammatory Activity of C. collinum Aqueous Leaves Extract, Myricetin-3-O-rhamnoside and Myricetin
2.4. In-Vitro Antibacterial Activity of C. collinum EtOH 50% Leaves Extract
3. Materials and Methods
3.1. Plant Material
3.2. Phytochemical Analysis
3.2.1. GC-MS analysis of Hexane Extract
3.2.2. Chromatographic Separation and Isolation of Flavonoids
3.2.3. HPLC Analysis
3.2.4. Quantitative HPLC Analysis
3.2.5. LC-MS Analysis
3.2.6. NMR Experiments
3.2.7. DPPH Assay
3.2.8. ABTS Assay
3.3. Functional Investigations on HaCaT Cells
3.3.1. Material
3.3.2. Cell Culture
3.3.3. In-Vitro Anti-Inflammatory Assay
3.3.4. Cell Viability Assay
3.4. Antimicrobial Assays
3.4.1. Microbial Strains
3.4.2. Agar Well Diffusion Test
3.4.3. Microdilution Test
3.4.4. Bacterial Growth Curve
3.5. Data Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- James, P.B.; Wardle, J.; Steel, A.; Adams, J. Traditional, complementary and alternative medicine use in Sub-Saharan Africa: A systematic review. BMJ Global Health 2018, 3, e000895. [Google Scholar] [CrossRef]
- Wittig, R.; Dingermann, T.; Sieglstetter, R.; Xie, Y.; Thiombiano, A.; Hahn, K. World-wide every fifth vascular plant species is or was used as medicinal or aromatic plant. Flora Veg. Sudano Sambesica 2013, 16, 3–9. [Google Scholar] [CrossRef]
- Vissiennon, Z.; Ahyi, V.; Koupkaki, E.; Nieber, K. Anti-inflammatory effects of novel gel-formulations with traditional used plants in Benin. Planta Med. 2011, 77, PF67. [Google Scholar] [CrossRef]
- Arbonnier, M. Trees, Shrubs and Lianas of West African Dry Zones; MNHN: Paris, France, 2004; p. 573. [Google Scholar]
- Neuwinger, H.D. African Traditional Medicine: A Dictionary of Plant Use and Applications; Medpharm Scientific: Stuttgart, Germany, 2000; p. 589. [Google Scholar]
- Burkill, H.M. Families A–D. In The Useful Plants of West Tropical Africa, 2nd ed.; Royal Botanic Gardens: Richmond, UK, 1985; p. 960. [Google Scholar]
- Abreu, P.M.; Martins, E.S.; Kayser, O.; Bindseil, K.U.; Siems, K.; Seemann, A.; Frevert, J. Antimicrobial, antitumor and antileishmania screening of medicinal plants from Guinea-Bissau. Phytomedicine 1999, 6, 187–195. [Google Scholar] [CrossRef]
- Eloff, J.N.; Katerere, D.R.; McGaw, L.J. The biological activity and chemistry of the southern African Combretaceae. J. Ethnopharmacol. 2008, 119, 686–699. [Google Scholar] [CrossRef] [PubMed]
- Rogers, C.B.; Coombes, P.H. Acidic triterpene glycosides in trichome secretions differentiate subspecies of Combretum collinum in South Africa. Biochem. Syst. Ecol. 1999, 27, 321–323. [Google Scholar] [CrossRef]
- Welch, C.R. Chemistry and Pharmacology of Kinkéliba (Combretum Micranthum), a West African Medicinal Plant. Ph.D. Thesis, Rutgers University-Graduate School, New Brunswick, NJ, USA, 1 January 2010. [Google Scholar]
- Souza, J.N.; Silva, E.M.; Silva, M.N.D.; Arruda, M.S.; Larondelle, Y.; Rogez, H. Identification and antioxidant activity of several flavonoids of Inga edulis leaves. J. Braz. Chem. Soc. 2007, 18, 1276–1280. [Google Scholar] [CrossRef]
- Kisseih, E.; Lechtenberg, M.; Petereit, F.; Sendker, J.; Zacharski, D.; Brandt, S.; Agyare, C.; Hensel, A. Phytochemical characterization and in vitro wound healing activity of leaf extracts from Combretum mucronatum Schum. and Thonn.: Oligomeric procyanidins as strong inductors of cellular differentiation. J. Ethnopharmacol. 2015, 174, 628–636. [Google Scholar] [CrossRef] [PubMed]
- Pietta, P.G. Flavonoids as antioxidants. J. Nat. Prod. 2000, 63, 1035–1042. [Google Scholar] [CrossRef]
- Kim, D.O.; Lee, C.Y. Comprehensive study on vitamin C equivalent antioxidant capacity (VCEAC) of various polyphenolics in scavenging a free radical and its structural relationship. Crit. Rev. Food Sci. Nutr. 2004, 44, 253–273. [Google Scholar] [CrossRef]
- Heim, K.E.; Tagliaferro, A.R.; Bobilya, D.J. Flavonoid antioxidants: Chemistry, metabolism and structure-activity relationships. J. Nutr. Biochem. 2002, 13, 572–584. [Google Scholar] [CrossRef]
- Limasset, B.; Le Doucen, C.; Dore, J.C.; Ojasoo, T.; Damon, M.; De Paulet, A.C. Effects of flavonoids on the release of reactive oxygen species by stimulated human neutrophils: Multivariate analysis of structure-activity relationships (SAR). Biochem. Pharmacol. 1993, 46, 1257–1271. [Google Scholar] [CrossRef]
- Hussain, T.; Tan, B.; Yin, Y.; Blachier, F.; Tossou, M.C.; Rahu, N. Oxidative stress and inflammation: What polyphenols can do for us? Oxid. Med. Cell Longev. 2016, 2016, 7432797. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.L.; Chen, J.P.; Lam, K.Y.C.; Zhan, J.Y.X.; Yao, P.; Dong, T.T.X.; Tsim, K.W.K. Hydrolysis of glycosidic flavonoids during the preparation of Danggui Buxue Tang: An outcome of moderate boiling of chinese herbal mixture. Evid. Based Complement Alternat. Med. 2014, 2014, 608721. [Google Scholar] [CrossRef] [PubMed]
- Hostetler, G.L.; Riedl, K.M.; Schwartz, S.J. Effects of food formulation and thermal processing on flavones in celery and chamomile. Food Chem. 2013, 141, 1406–1411. [Google Scholar] [CrossRef] [PubMed]
- Chuang, S.Y.; Lin, Y.K.; Lin, C.F.; Wang, P.W.; Chen, E.L.; Fang, J.Y. Elucidating the skin delivery of aglycone and glycoside flavonoids: How the structures affect cutaneous absorption. Nutrients 2017, 9, 1304. [Google Scholar] [CrossRef]
- Ernst, I.M.A.; Wagner, A.E.; Lipinski, S.; Skrbek, S.; Ruefer, C.E.; Desel, C.; Rimbach, G. Cellular uptake, stability, visualization by ‘Naturstoff reagent A’, and multidrug resistance protein 1 gene-regulatory activity of cyanidin in human keratinocytes. Pharmacol. Res. 2010, 61, 253–258. [Google Scholar] [CrossRef]
- Lee, D.H.; Lee, C.S. Flavonoid myricetin inhibits TNF-α-stimulated production of inflammatory mediators by suppressing the Akt, mTOR and NF-κB pathways in human keratinocytes. Eur. J. Pharmacol. 2016, 784, 164–172. [Google Scholar] [CrossRef]
- Shimosaki, S.; Tsurunaga, Y.; Itamura, H.; Nakamura, M. Anti-allergic effect of the flavonoid myricitrin from Myrica rubra leaf extracts in vitro and in vivo. Nat. Prod. Res. 2011, 25, 374–380. [Google Scholar] [CrossRef]
- Chen, L.; Teng, H.; Jia, Z.; Battino, M.; Miron, A.; Yu, Z.; Cao, H.; Xiao, J. Intracellular signaling pathways of inflammation modulated by dietary flavonoids: The most recent evidence. Crit. Rev. Food Sci. Nutr. 2018, 58, 2908–2924. [Google Scholar] [CrossRef]
- Harada, A.; Sekido, N.; Akahoshi, T.; Wada, T.; Mukaida, N.; Matsushima, K. Essential involvement of interleukin-8 (IL-8) in acute inflammation. J. Leukoc. Biol. 1994, 56, 559–564. [Google Scholar] [CrossRef] [PubMed]
- Arican, O.; Aral, M.; Sasmaz, S.; Ciragil, P. Serum levels of TNF-α, IFN-γ, IL-6, IL-8, IL-12, IL-17, and IL-18 in patients with active psoriasis and correlation with disease severity. Mediators Inflamm. 2005, 2005, 273–279. [Google Scholar] [CrossRef] [PubMed]
- Scheller, J.; Chalaris, A.; Schmidt-Arras, D.; Rose-John, S. The pro-and anti-inflammatory properties of the cytokine interleukin-6. Biochim. Biophys. Acta Mol. Cell Res. 2011, 1813, 878–888. [Google Scholar] [CrossRef] [PubMed]
- Okwu, M.U.; Olley, M.; Akpoka, A.O.; Izevbuwa, O.E. Methicillin-resistant Staphylococcus aureus (MRSA) and anti-MRSA activities of extracts of some medicinal plants: A brief review. AIMS Microbiol. 2019, 5, 117–137. [Google Scholar] [CrossRef]
- Aliyu, A.B.; Musa, A.M.; Abdullahi, M.S.; Oyewale, A.O.; Gwarzo, U.S. Activity of plant extracts used in northern Nigerian traditional medicine against methicillin-resistant Staphylococcus aureus (MRSA). Niger. J. Pharm. Sci. 2008, 7, 1–8. [Google Scholar]
- Akinjogunla, O.J.; Yah, C.S.; Eghafona, N.O.; Ogbemudia, F.O. Antibacterial activity of leave extracts of Nymphaea lotus (Nymphaeaceae) on Methicillin resistant Staphylococcus aureus (MRSA) and Vancomycin resistant Staphylococcus aureus (VRSA) isolated from clinical samples. Ann. Biol. Res. 2010, 1, 174–184. [Google Scholar]
- Cos, P.; Vlietinck, A.J.; Berghe, D.V.; Maes, L. Anti-infective potential of natural products: How to develop a stronger in vitro ‘proof-of-concept’. J. Ethnopharmacol. 2006, 106, 290–302. [Google Scholar] [CrossRef]
- de Morais Lima, G.R.; de Sales, I.R.P.; Caldas Filho, M.R.D.; de Jesus, N.Z.T.; de Sousa Falcão, H.; Barbosa-Filho, J.M.; Cabral, A.G.; Souto, A.L.; Tavares, J.F.; Batista, L.M. Bioactivities of the genus Combretum (Combretaceae): A review. Molecules 2012, 17, 9142–9206. [Google Scholar] [CrossRef]
- Chandra, H.; Bishnoi, P.; Yadav, A.; Patni, B.; Mishra, A.; Nautiyal, A. Antimicrobial resistance and the alternative resources with special emphasis on plant-based antimicrobials—A review. Plants 2017, 6, 16. [Google Scholar] [CrossRef]
- Kachur, K.; Suntres, Z. The antibacterial properties of phenolic isomers, carvacrol and thymol. Crit. Rev. Food Sci. Nutr. 2019, 1–12. [Google Scholar] [CrossRef]
- Cushnie, T.T.; Lamb, A.J. Antimicrobial activity of flavonoids. Int. J. Antimicrob. Agents 2005, 26, 343–356. [Google Scholar] [CrossRef]
- Silva, L.N.; Da Hora, G.C.A.; Soares, T.A.; Bojer, M.S.; Ingmer, H.; Macedo, A.J.; Trentin, D.S. Myricetin protects Galleria mellonella against Staphylococcus aureus infection and inhibits multiple virulence factors. Sci. Rep. 2017, 7, 2823. [Google Scholar] [CrossRef] [PubMed]
- Madikizela, B.; Aderogba, M.A.; Van Staden, J. Isolation and characterization of antimicrobial constituents of Searsia chirindensis L. (Anacardiaceae) leaf extracts. J. Ethnopharmacol. 2013, 150, 609–613. [Google Scholar] [CrossRef] [PubMed]
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C.L.W.T. Use of a free radical method to evaluate antioxidant activity. LWT Food Sci. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Zou, Y.; Chang, S.K.; Gu, Y.; Qian, S.Y. Antioxidant activity and phenolic compositions of lentil (Lens culinaris var. Morton) extract and its fractions. J. Agric. Food Chem. 2011, 59, 2268–2276. [Google Scholar] [CrossRef]
- Boukamp, P.; Petrussevska, R.T.; Breitkreutz, D.; Hornung, J.; Markham, A.; Fusenig, N.E. Normal keratinization in a spontaneously immortalized aneuploid human keratinocyte cell line. J. Cell Biol. 1988, 106, 761–771. [Google Scholar] [CrossRef]
- Wedler, J.; Daubitz, T.; Schlotterbeck, G.; Butterweck, V. In vitro anti-inflammatory and wound-healing potential of a Phyllostachys edulis leaf extract–identification of isoorientin as an active compound. Planta Med. 2014, 80, 1678–1684. [Google Scholar] [CrossRef]
- EUCAST European Committee on Antimicrobial Susceptibility Testing. Version 7.1. 2017. Available online: http://www.eucast.org/fileadmin/src/media/PDFs/EUCAST_files/General_documents/Publications/Disk_diffusion_paper_printed_version_March_2014.pdf (accessed on 25 October 2019).
Sample Availability: Samples of Combretum collinum extracts and myricetin-3-O-rhamnoside are available from the authors. |
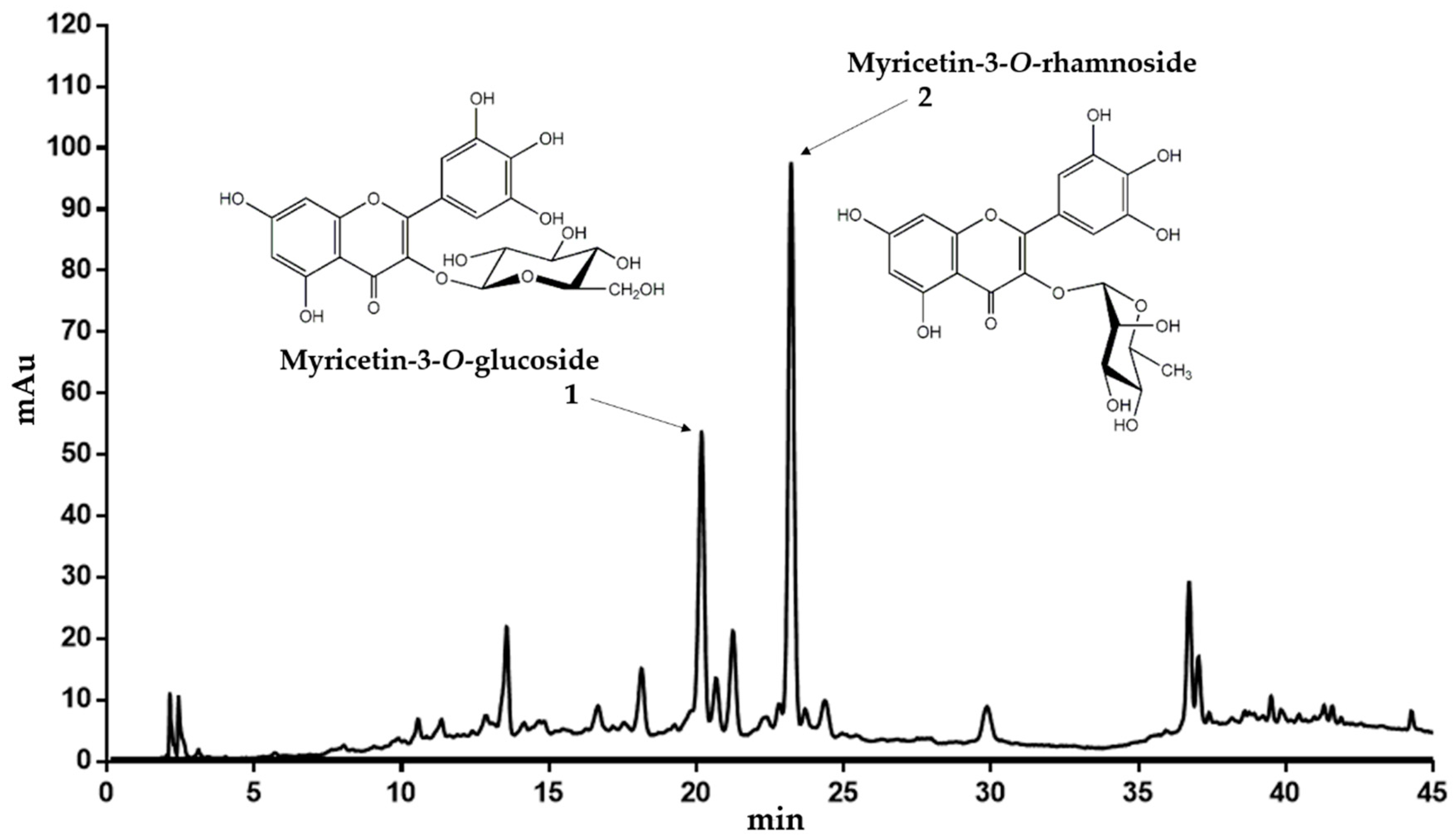



| Peak | RT 1 [min] | λmax [nm] | m/z | Ion | Compound |
|---|---|---|---|---|---|
| 1 | 20.177 | 264 350 | 319.096 481.189 479.193 | [A + H]+ [M + H]+ [M − H]− | Myricetin-3-O-glucoside |
| 2 | 23.219 | 262 350 | 319.094 465.179 463.188 | [A + H]+ [M + H]+ [M − H]− | Myricetin-3-O-rhamnoside |
| Signal | Myricetin-3-O-rhamnoside (F31–F33) | Myricetin-3-O-rhamnoside [10] |
|---|---|---|
| H-3′′-6′′ | 0.95, d, J 6 Hz | 0.94, d, J 5.6 Hz |
| sugar H | 3.3–3.8 | 3.3–3.8 |
| H-2′′ | 4.25, s | 4.23, s |
| H-1′′ | 5.34, brs | 5.31, brs |
| H-6 | 6.22, s | 6.19, s |
| H-8 | 6.39, s | 6.35, s |
| H-2′ and H-6′ | 6.97, s | 6.95, s |
| No. | RT 1 [min] | Compound | NIST Match Quality |
|---|---|---|---|
| 1 | 12.62 | Malic acid | 97 |
| 2 | 12.73 | Tetradecanoic acid (syn. myristic acid) | 98 |
| 3 | 14.39 | Hexadecanoic acid (syn. palmitic acid) | 98 |
| 4 | 15.87 | Oleic acid | 98 |
| 5 | 15.92 | Octadecanoic acid (syn. stearic acid) | 99 |
| 6 | 17.64 | Eicosanoic acid (syn. arachidic acid) | 99 |
| 7 | 19.78 | Docosanoic acid (syn. behenic acid) | 99 |
| 8 | 22.40 | Tetracosanoic acid (syn. lignoceric acid) | 99 |
| 9 | 17.94 | Pentacosane | 96 |
| 10 | 18.99 | Hexacosane | 99 |
| 11 | 20.17 | Heptacosane | 99 |
| 12 | 21.48 | Octacosane | 99 |
| 13 | 22.92 | Nonacosane | 96 |
| 14 | 24.42 | Triacontane | 99 |
| 15 | 26.06 | Nonadecane | 96 |
| 16 | 23.22 | Squalene | 97 |
| 17 | 30.78 | Campestrol | 99 |
| 18 | 31.18 | Stigmasterol | 99 |
| Species | Inhibition Zones (mm) | Inhibition Zones (Positive Controls) | MIC |
|---|---|---|---|
| S. epidermidis | 21.75 ± 0.43 | 28.67 ± 0.47 (G) | 275.0 µg/mL |
| MRSA | 16.25 ± 0.43 | 18.67 ± 0.47 (V) | 385.5 µg/mL |
| S. aureus | 15.75 ± 0.43 | 24.33 ± 0.47 (G) | n.d. |
| K. pneumoniae | 14.00 ± 0.00 | 25.33 ± 0.94 (G) | n.d. |
| P. aeruginosa | 11.00 ± 0.00 | 12.00 ± 0.00 (G) | n.d. |
| E. coli | 11.25 ± 0.43 | 26.67 ± 0.47 (G) | n.d. |
| Enterococcus | 0 | 16.67 ± 0.47 (G) | n.d. |
| C. albicans | 0 | 26.67 ± 0.47 (N) | n.d. |
| DMSO 10% | 0 | - | n.d. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Marquardt, P.; Seide, R.; Vissiennon, C.; Schubert, A.; Birkemeyer, C.; Ahyi, V.; Fester, K. Phytochemical Characterization and In Vitro Anti-Inflammatory, Antioxidant and Antimicrobial Activity of Combretum Collinum Fresen Leaves Extracts from Benin. Molecules 2020, 25, 288. https://doi.org/10.3390/molecules25020288
Marquardt P, Seide R, Vissiennon C, Schubert A, Birkemeyer C, Ahyi V, Fester K. Phytochemical Characterization and In Vitro Anti-Inflammatory, Antioxidant and Antimicrobial Activity of Combretum Collinum Fresen Leaves Extracts from Benin. Molecules. 2020; 25(2):288. https://doi.org/10.3390/molecules25020288
Chicago/Turabian StyleMarquardt, Peter, Rick Seide, Cica Vissiennon, Andreas Schubert, Claudia Birkemeyer, Virgile Ahyi, and Karin Fester. 2020. "Phytochemical Characterization and In Vitro Anti-Inflammatory, Antioxidant and Antimicrobial Activity of Combretum Collinum Fresen Leaves Extracts from Benin" Molecules 25, no. 2: 288. https://doi.org/10.3390/molecules25020288
APA StyleMarquardt, P., Seide, R., Vissiennon, C., Schubert, A., Birkemeyer, C., Ahyi, V., & Fester, K. (2020). Phytochemical Characterization and In Vitro Anti-Inflammatory, Antioxidant and Antimicrobial Activity of Combretum Collinum Fresen Leaves Extracts from Benin. Molecules, 25(2), 288. https://doi.org/10.3390/molecules25020288