Synthesis and Evaluation of New Coumarin Derivatives as Antioxidant, Antimicrobial, and Anti-Inflammatory Agents
Abstract
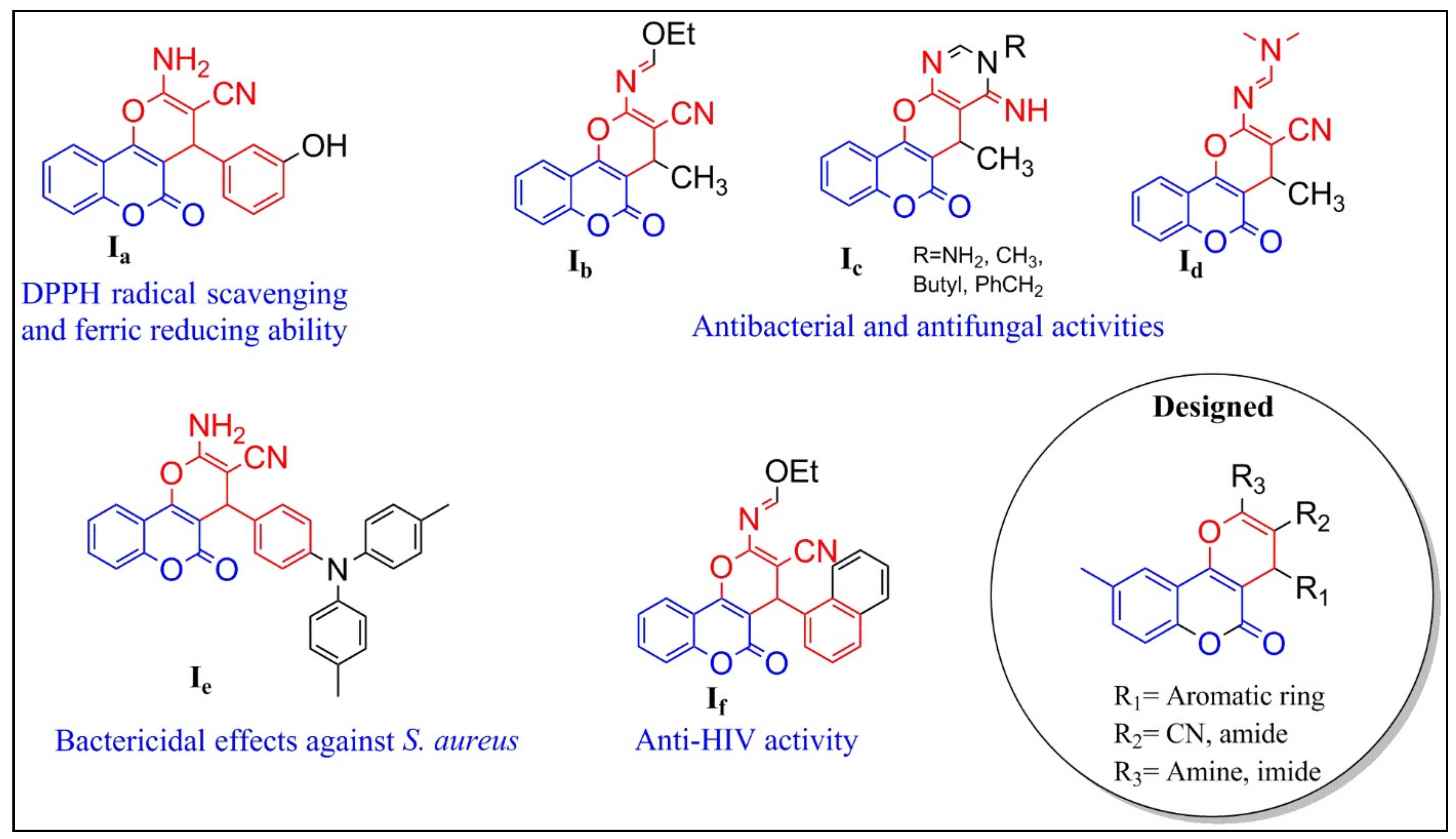
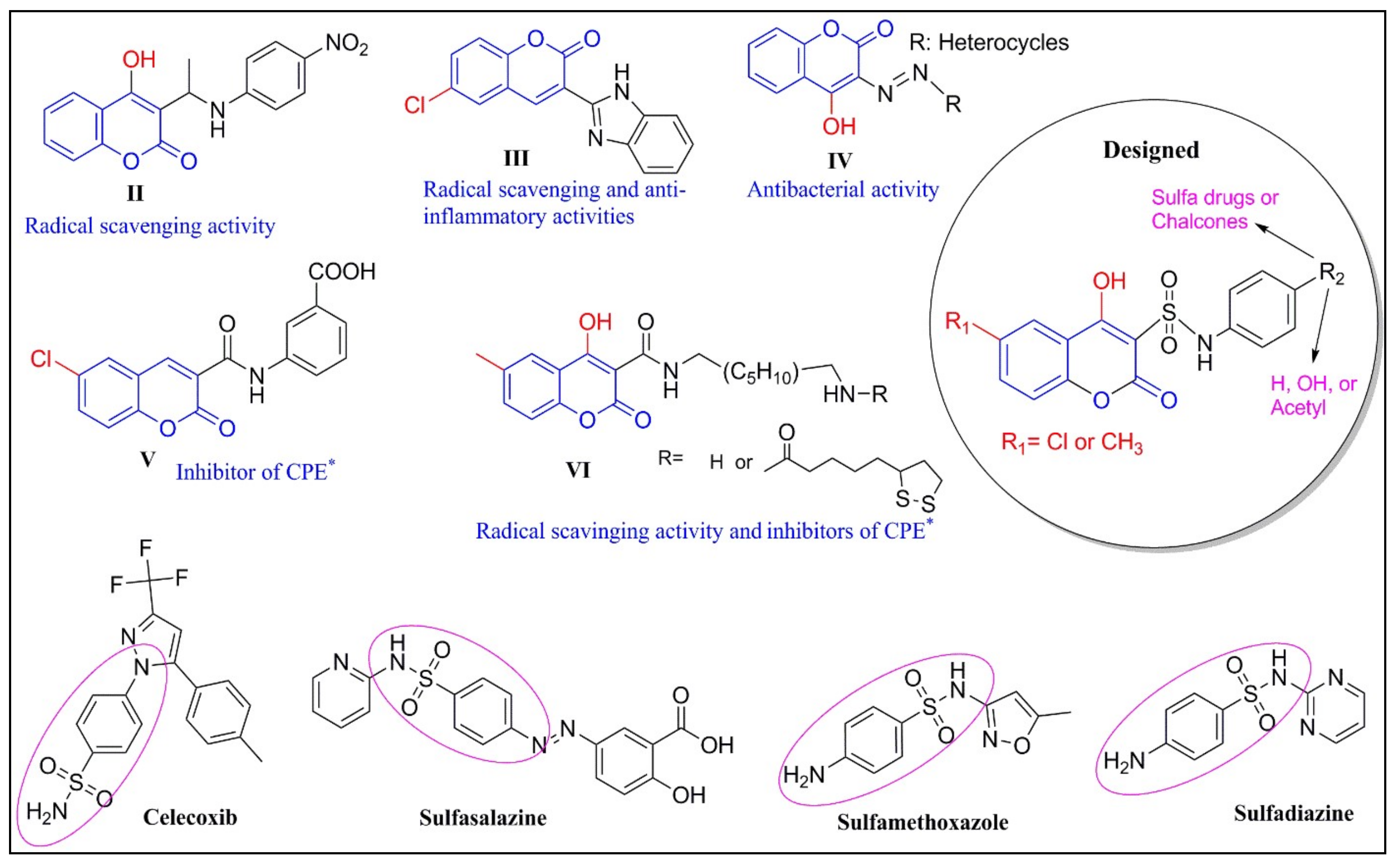
1. Introduction
2. Results and Discussion
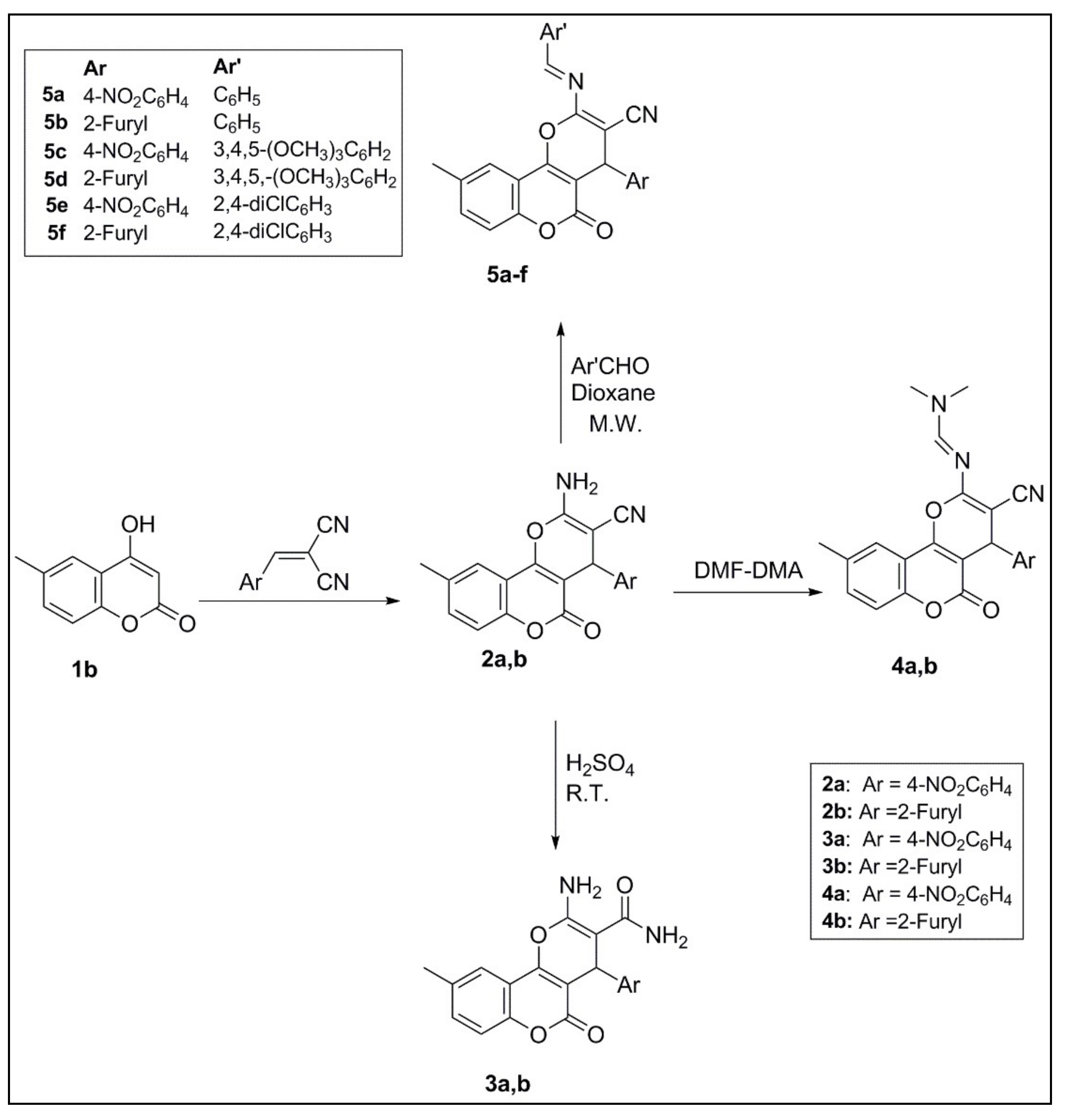
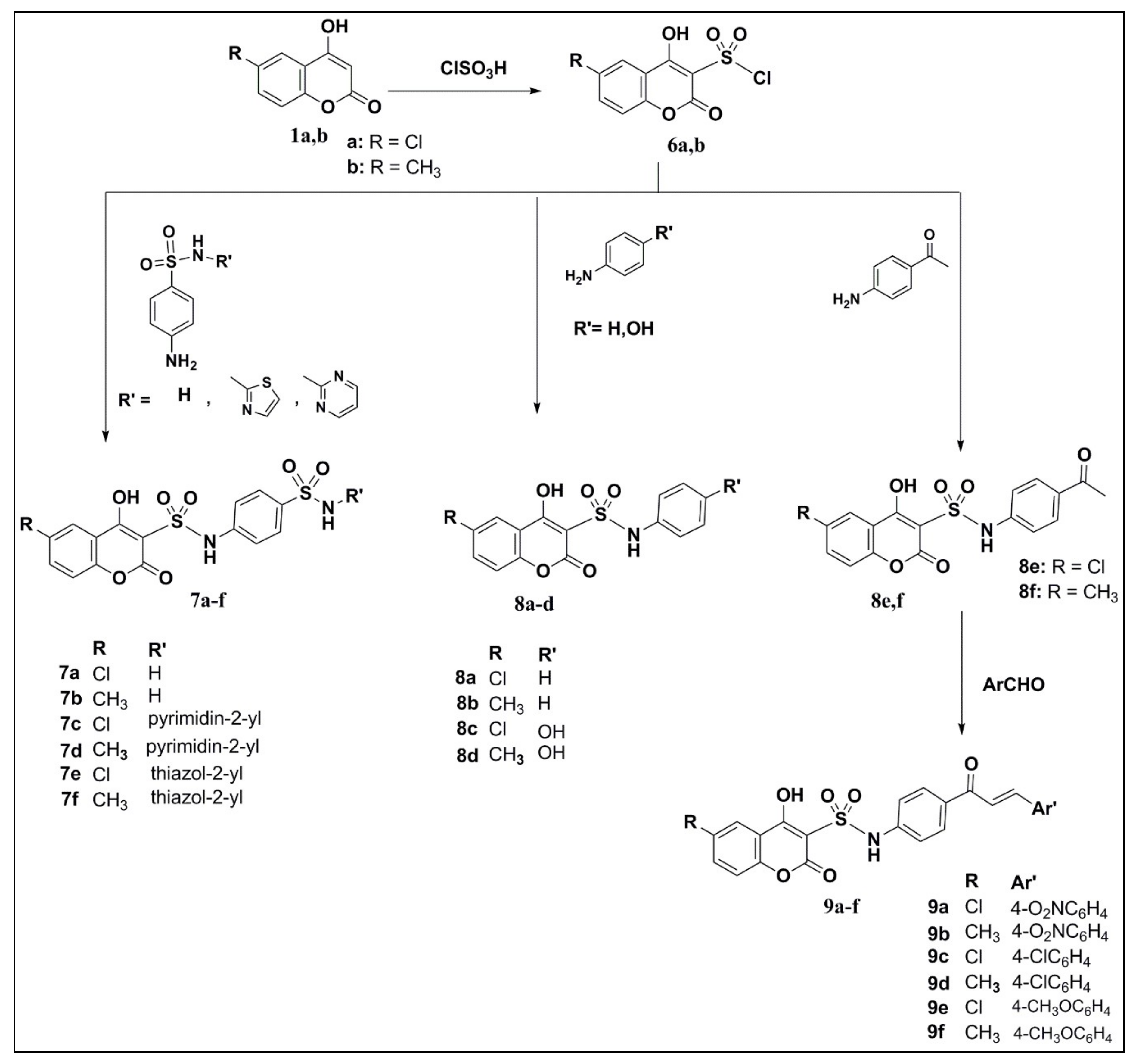
2.1. Chemistry
2.2. Antioxidant Testing
2.3. Antimicrobial Testing
2.4. Anti-Inflammatory Activity
2.4.1. Proteinase-Inhibitory Activity
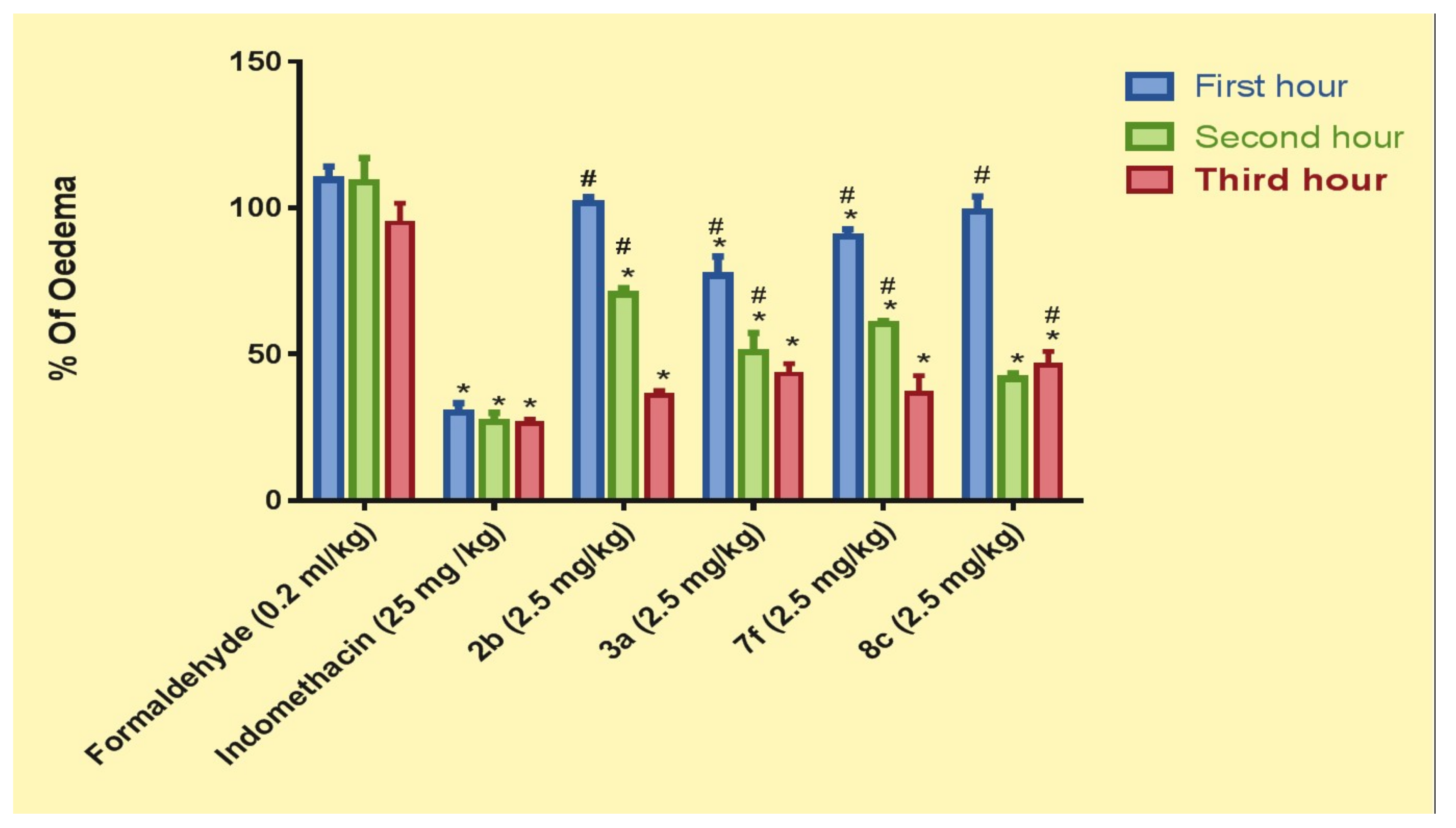
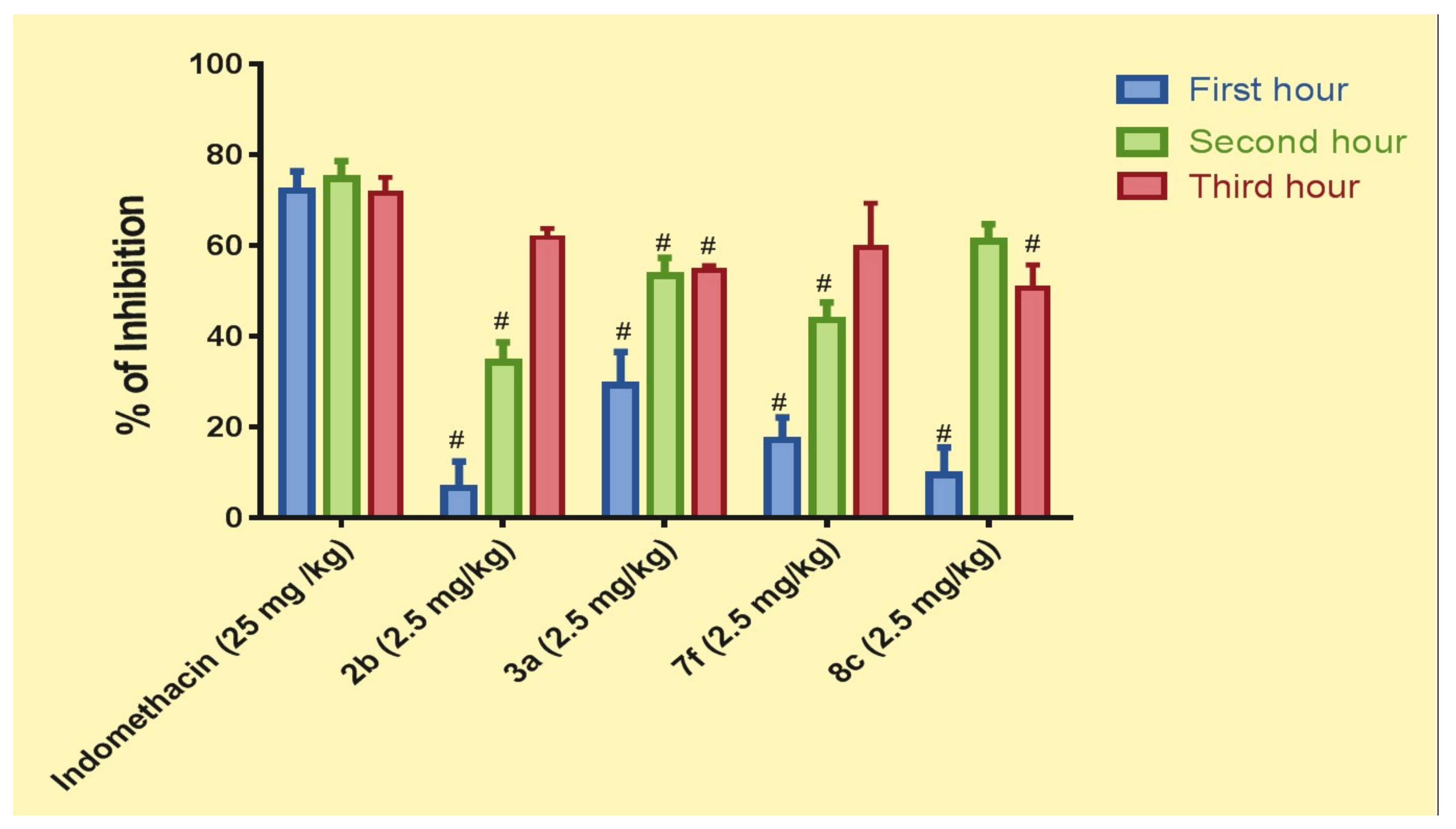
2.4.2. Pharmacological In Vivo Study
2.4.3. COX Inhibition
2.5. In Silico Studies
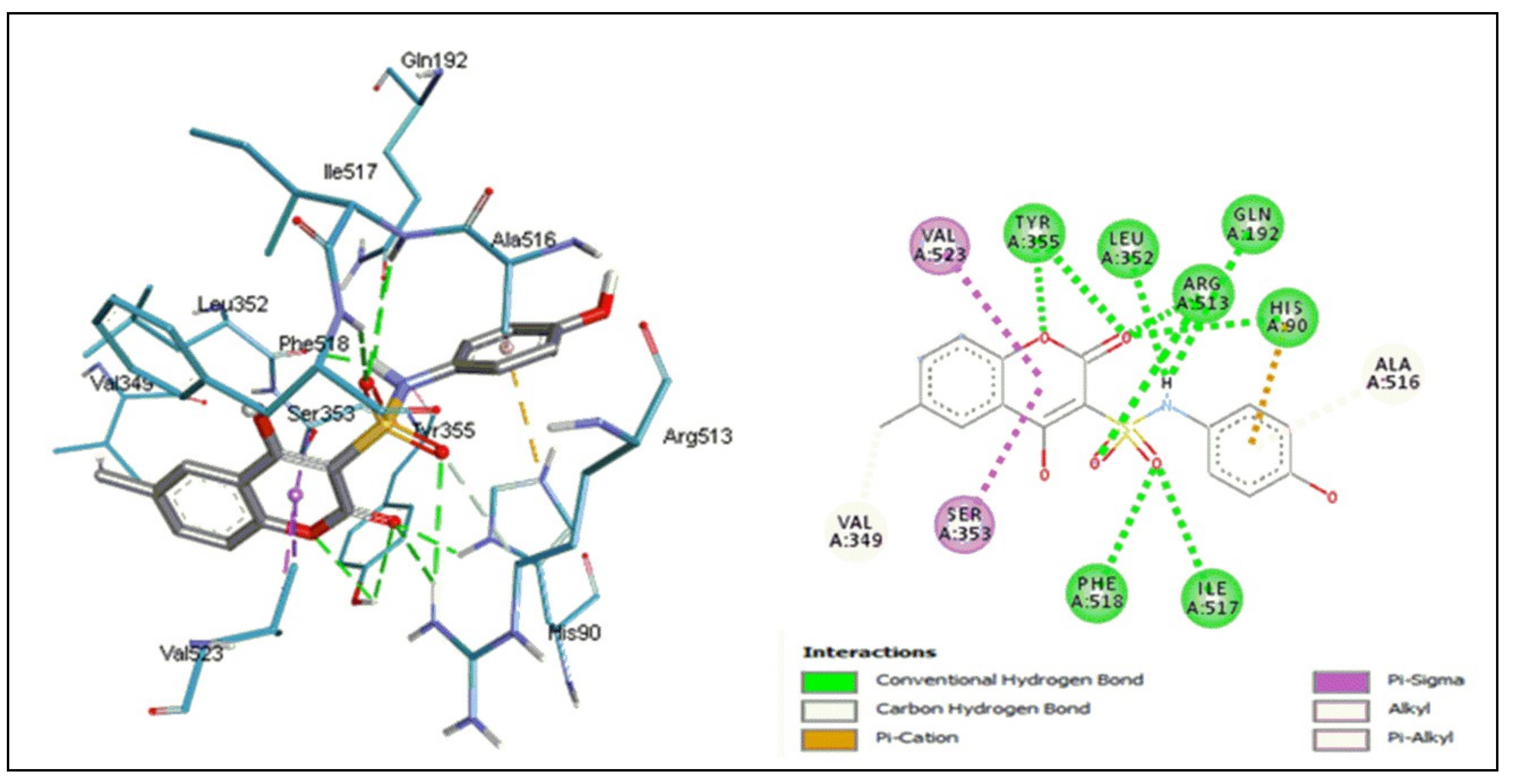
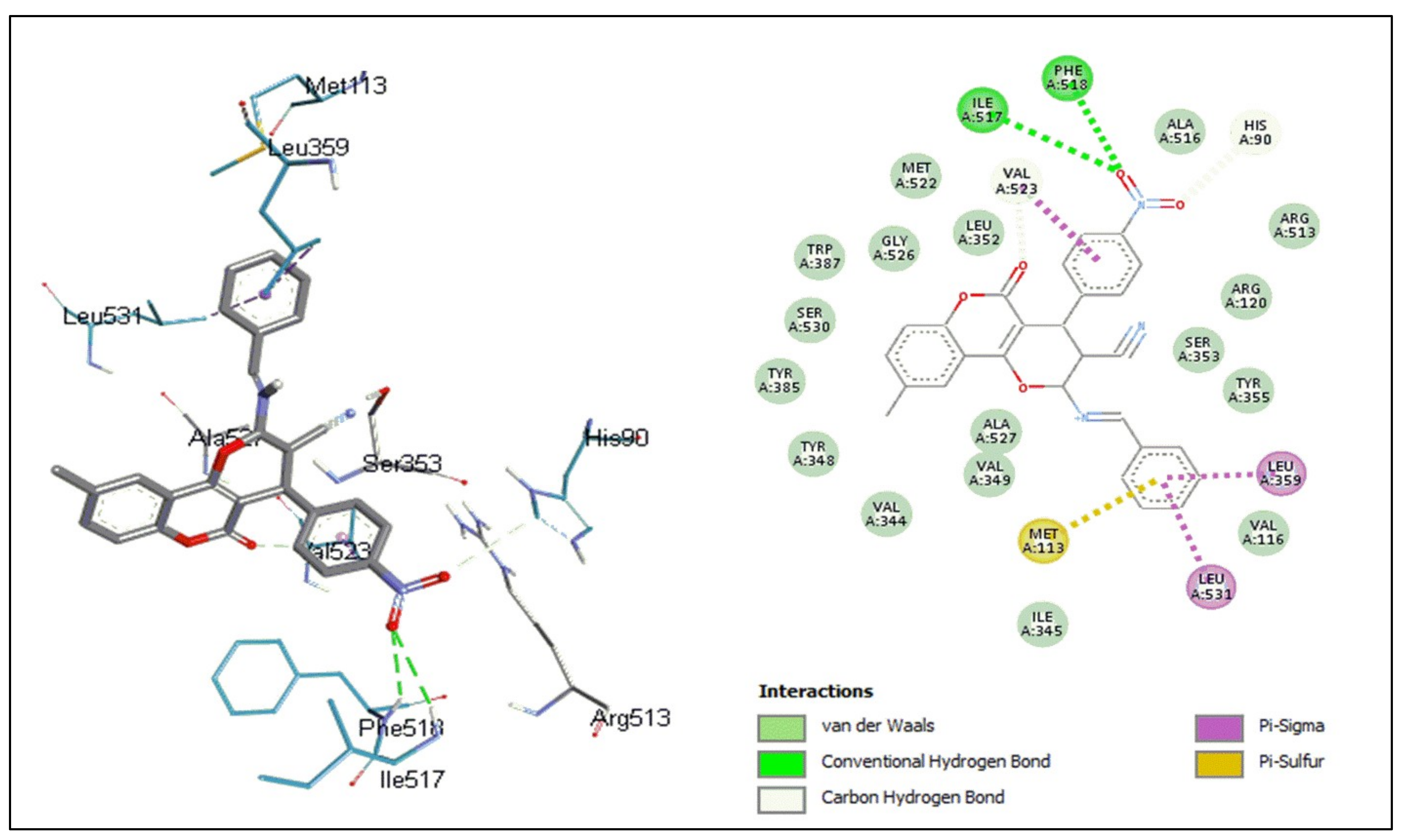
2.5.1. Docking Studies
2.5.2. Drug-Likeness Assessment
3. Materials and Methods
3.1. Chemistry
3.1.1. 4-Hydroxy-6-methylcoumarin (1b)
3.1.2. 2-Amino-9-methyl-5-oxo-4-aryl-4H,5H-pyrano[3,2-c]chromene-3-carbonitriles (2a–b)
3.1.3. 2-Amino-9-methyl-5-oxo-4-aryl-4H,5H-pyrano[3,2-c]chromene-3-carboxamides (3a,b)
3.1.4. N′-(3-cyano-9-methyl-5-oxo-4-aryl-4H,5H-pyrano[3,2-c]chromen-2-yl)-N,N-dimethyl formimidamides (4a,b)
3.1.5. 2-(Arylideneamino)-9-methyl-4-(4-nitrophenyl)-5-oxo-4H,5H-pyrano[3,2-c]chromene-3-carbonitriles (5a–f)
3.1.6. 6-substituted-4-hydroxyl-2-oxo-2H-chromene-3-sulfonyl chloride (6a,b)
3.1.7. 4-Hydroxy-6-(substituted) coumarin-3-sulfonamides (7a–f)
3.1.8. 4-Hydroxy-6-(substituted)coumarin-3-sulfonamides (8a–f)
3.1.9. Coumarin-Sulfonamide Chalcones (9a–f)
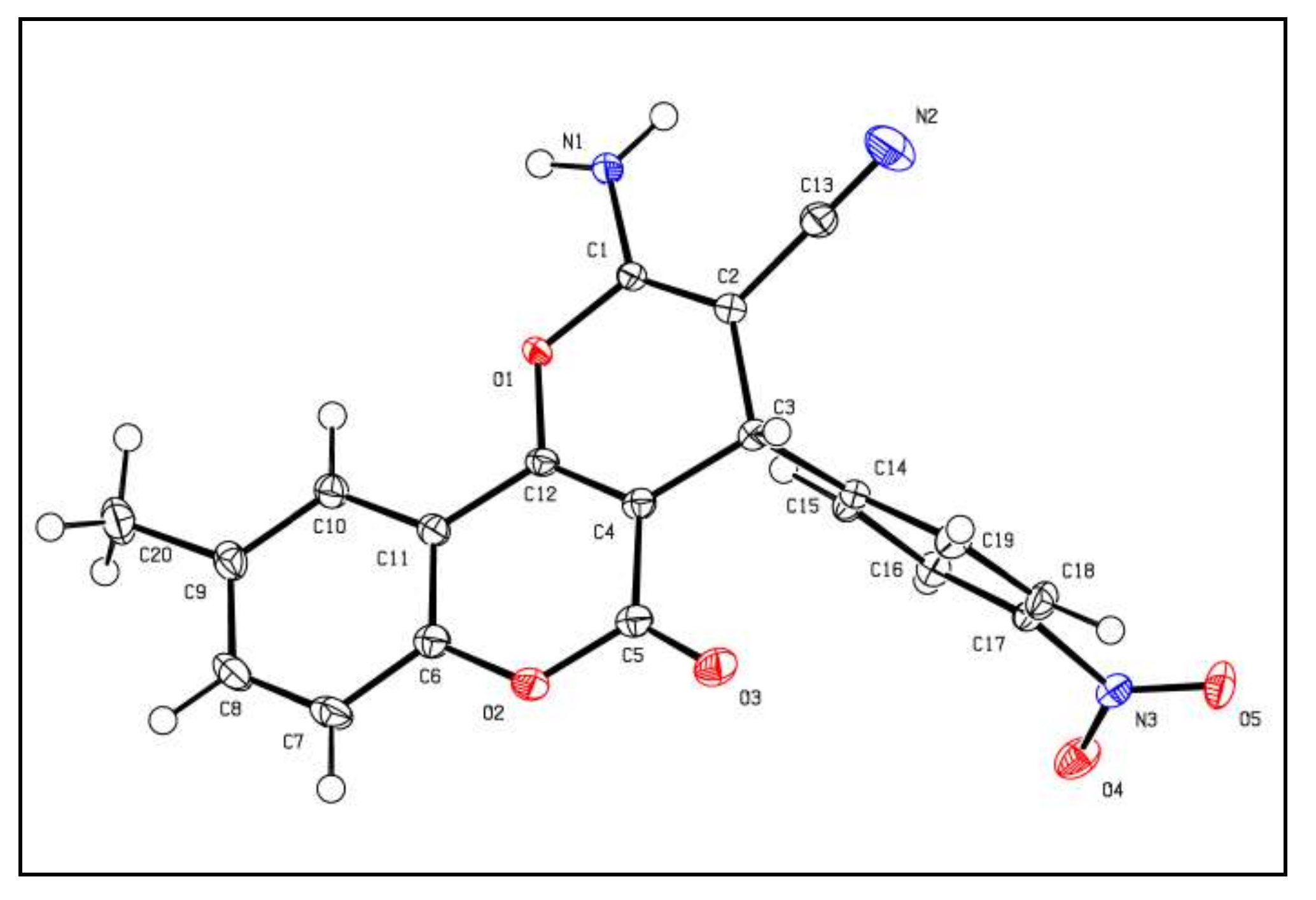
3.1.10. X-Ray Crystallographic Study of Compound 2a
3.2. Antioxidant Testing
3.3. Antimicrobial Activity
3.3.1. Agar Well Diffusion Method
3.3.2. MIC Measurement
3.4. Anti-Inflammatory Activity
3.4.1. Proteinase-Inhibitory Activity
3.4.2. Pharmacological In Vivo Study
- Positive control group: Paw oedema was induced by 0.2 mL (1%, w/v) formaldehyde injected in the subplantar area of the left hind paw of the rat [33].
- Prophylactic groups:
- (a)
- Indomethacin (Reference) group: Rats were given indomethacin orally at 25 mg/kg [44].
- (b)
- Test groups: rats were given coumarin derivative 2b, 3a, 7f, and 8c orally in a dose of 2.5 mg/kg each.
3.4.3. COX Inhibition
3.5. In Silico Studies
3.5.1. Molecular Docking Study
3.5.2. Drug-Likeness Assessment
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Liguori, I.; Russo, G.; Curcio, F.; Bulli, G.; Aran, L.; Della-Morte, D.; Gargiulo, G.; Testa, G.; Cacciatore, F.; Bonaduce, D.; et al. Oxidative Stress, Aging, and Diseases. Clin. Interv. Aging 2018, 13, 757–772. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Deng, H.; Cui, H.; Fang, J.; Zuo, Z.; Deng, J.; Li, Y.; Wang, X.; Zhao, L. Inflammatory Responses and Inflammation-Associated Diseases in Organs. Oncotarget 2017, 9, 7204–7218. [Google Scholar] [CrossRef] [PubMed]
- Pal, C.; Bengtsson-Palme, J.; Kristiansson, E.; Larsson, D.G.J. The Structure and Diversity of Human, Animal and Environmental Resistomes. Microbiome 2016, 4, 54. [Google Scholar] [CrossRef] [PubMed]
- Laxminarayan, R.; Duse, A.; Wattal, C.; Zaidi, A.K.M.; Wertheim, H.F.L.; Sumpradit, N.; Vlieghe, E.; Hara, G.L.; Gould, I.M.; Goossens, H.; et al. Antibiotic Resistance—the Need for Global Solutions. Lancet Infect. Dis. 2013, 13, 1057–1098. [Google Scholar] [CrossRef]
- Pereira, T.M.; Franco, D.P.; Vitorio, F.; Kummerle, A.E. Coumarin Compounds in Medicinal Chemistry: Some Important Examples from the Last Years. Curr. Top. Med. Chem. 2018, 18, 124–148. [Google Scholar] [CrossRef]
- Kontogiorgis, C.; Detsi, A.; Hadjipavlou-Litina, D. Coumarin-Based Drugs: A Patent Review (2008–Present). Expert Opin. Ther. Pat. 2012, 22, 437–454. [Google Scholar] [CrossRef]
- Dehkordi, M.F.; Dehghan, G.; Mahdavi, M.; Hosseinpour Feizi, M.A. Multispectral Studies of DNA Binding, Antioxidant and Cytotoxic Activities of a New Pyranochromene Derivative. Spectrochim. Acta. A. Mol. Biomol. Spectrosc. 2015, 145, 353–359. [Google Scholar] [CrossRef]
- Vukovic, N.; Sukdolak, S.; Solujic, S.; Niciforovic, N. Substituted Imino and Amino Derivatives of 4-Hydroxycoumarins as Novel Antioxidant, Antibacterial and Antifungal Agents: Synthesis and in Vitro Assessments. Food Chem. 2010, 120, 1011–1018. [Google Scholar] [CrossRef]
- Arora, R.K.; Kaur, N.; Bansal, Y.; Bansal, G. Novel Coumarin–Benzimidazole Derivatives as Antioxidants and Safer Anti-Inflammatory Agents. Acta Pharm. Sin. B 2014, 4, 368–375. [Google Scholar] [CrossRef]
- Bylov, I.E.; Vasylyev, M.V.; Bilokin, Y.V. Synthesis and Anti-Inflammatory Activity of N-Substituted 2-Oxo-2H-1-Benzopyran-3-Carboxamides and Their 2-Iminoanalogues. Eur. J. Med. Chem. 1999, 34, 997–1001. [Google Scholar] [CrossRef]
- Sashidhara, K.V.; Kumar, M.; Modukuri, R.K.; Sonkar, R.; Bhatia, G.; Khanna, A.K.; Rai, S.; Shukla, R. Synthesis and Anti-Inflammatory Activity of Novel Biscoumarin–Chalcone Hybrids. Bioorg. Med. Chem. Lett. 2011, 21, 4480–4484. [Google Scholar] [CrossRef] [PubMed]
- Bedair, A.H.; El-Hady, N.A.; El-Latif, M.S.A.; Fakery, A.H.; El-Agrody, A.M. 4-Hydroxycoumarin in Heterocyclic Synthesis: Part III. Synthesis of Some New Pyrano[2,3-d]Pyrimidine, 2-Substituted[1,2,4]Triazolo[1,5-c]Pyrimidine and Pyrimido[1,6-b][1,2,4]Triazine Derivatives. Il Farm. 2000, 55, 708–714. [Google Scholar] [CrossRef]
- Shi, X.; Lv, C.; Li, J.; Hou, Z.; Yang, X.; Zhang, Z.; Luo, X.; Yuan, Z.; Li, M. Synthesis, Photoluminescent, Antibacterial Activities and Theoretical Studies of Three Novel Coumarin and Dihydropyran Derivatives Containing a Triphenylamine Group. Res. Chem. Intermed. 2015, 41, 8965–8974. [Google Scholar] [CrossRef]
- Al-Masoudi, N.A.; Mohammed, H.H.; Hamdy, A.M.; Akrawi, O.A.; Eleya, N.; Spannenberg, A.; Pannecouque, C.; Langer, P. Synthesis and Anti-HIV Activity of New Fused Chromene Derivatives Derived from 2-Amino-4-(1-Naphthyl)-5-Oxo-4H,5H-Pyrano[3,2- c]Chromene-3-Carbonitrile. Z. Für Naturforschung B 2014, 68, 229–238. [Google Scholar] [CrossRef]
- Sahoo, J.; Kumar Mekap, S.; Sudhir Kumar, P. Synthesis, Spectral Characterization of Some New 3-Heteroaryl Azo 4-Hydroxy Coumarin Derivatives and Their Antimicrobial Evaluation. J. Taibah Univ. Sci. 2015, 9, 187–195. [Google Scholar] [CrossRef]
- Alshibl, H.M.; Al-Abdullah, E.S.; Alkahtani, H.M. Coumarin: A Promising Scaffold for Design and Development of Bioactive Agents. Curr. Bioact. Compd. 2019, 15, 1–15. [Google Scholar] [CrossRef]
- Silverstein, F.E.; Faich, G.; Goldstein, J.L.; Simon, L.S.; Pincus, T.; Whelton, A.; Makuch, R.; Eisen, G.; Agrawal, N.M.; Stenson, W.F.; et al. Gastrointestinal Toxicity With Celecoxib vs. Nonsteroidal Anti-Inflammatory Drugs for Osteoarthritis and Rheumatoid Arthritis The CLASS Study: A Randomized Controlled Trial. JAMA 2000, 284, 1247–1255. [Google Scholar] [CrossRef]
- Peppercorn, M.A. Sulfasalazine. Pharmacology, Clinical Use, Toxicity, and Related New Drug Development. Ann. Intern. Med. 1984, 101, 377–386. [Google Scholar] [CrossRef]
- Fischi, M.A.; Dickinson, G.M.; Vole, L.L. Safety and Efficacy of Sulfamethoxazole and Trimethoprim Chemoprophylaxis for Pneumocystis Carinii Pneumonia in AIDS. JAMA 1988, 259, 1185–1189. [Google Scholar] [CrossRef]
- Fox, C.L. Pharmacology and Clinical Use of Silver Sulfadiazine and Related Topical Antimicrobial Agents. Pahlavi Med. J. 1977, 8, 45–64. [Google Scholar]
- Alshibl, H.M.; Al-Abdullah, E.S.; Haiba, M.E. Coumarin Derivatives. U.S. Patent Application Pending no. 16/399,950, 30 April 2019. [Google Scholar]
- Kiyani, H.; Ghorbani, F. Efficient Tandem Synthesis of a Variety of Pyran-Annulated Heterocycles, 3,4-Disubstituted Isoxazol-5(4H)-Ones, and α,β-Unsaturated Nitriles Catalyzed by Potassium Hydrogen Phthalate in Water. Res. Chem. Intermed. 2015, 41, 7847–7882. [Google Scholar] [CrossRef]
- Reddy, N.S.; Mallireddigari, M.R.; Cosenza, S.; Gumireddy, K.; Bell, S.C.; Reddy, E.P.; Reddy, M.V.R. Synthesis of New Coumarin 3-(N-Aryl) Sulfonamides and Their Anticancer Activity. Bioorg. Med. Chem. Lett. 2004, 14, 4093–4097. [Google Scholar] [CrossRef] [PubMed]
- Behrami, A. Antibacterial Activity of Coumarine Derivatives Synthesized from 4-Chloro-Chromen-2-One. The Comparison with Standard Drug. Orient. J. Chem. 2014, 30, 1747–1752. [Google Scholar] [CrossRef]
- Yu, L.; Haley, S.; Perret, J.; Harris, M.; Wilson, J.; Qian, M. Free Radical Scavenging Properties of Wheat Extracts. J. Agric. Food Chem. 2002, 50, 1619–1624. [Google Scholar] [CrossRef] [PubMed]
- Qazi, S.S.; Li, D.; Briens, C.; Berruti, F.; Abou-Zaid, M.M. Antioxidant Activity of the Lignins Derived from Fluidized-Bed Fast Pyrolysis. Mol. Basel Switz. 2017, 22. [Google Scholar] [CrossRef] [PubMed]
- Perez-Eid, C.; Pauli, W.M.; Bazerque, P. An Antibiotic Assay by Agar-Well Diffusion Method. Acta Biologiae et Med. Experimentalis. 1990, 15, 113–115. [Google Scholar]
- Scott, A.C. Laboratory Control of Antimicrobial Therapy. In Practical Medical Microbiology; Collee, J., Duguid, J., Fraser, A., Marmion, B., Eds.; Churchill Livingstone: Edinburgh, UK, 1989; Volume 2, pp. 161–181. [Google Scholar]
- Rutkowska, E.; Pajak, K.; Jóźwiak, K. Lipophilicity-Methods of Determination and Its Role in Medicinal Chemistry. Acta Pol. Pharm. 2013, 70, 3–18. [Google Scholar]
- Kalinowska, M.; Bajko, E.; Matejczyk, M.; Kaczyński, P.; Łozowicka, B.; Lewandowski, W. The Study of Anti-/Pro-Oxidant, Lipophilic, Microbial and Spectroscopic Properties of New Alkali Metal Salts of 5-O-Caffeoylquinic Acid. Int. J. Mol. Sci. 2018, 19, 463. [Google Scholar] [CrossRef]
- Kocabalkanli, A.; Ates, Ö.; Ötük, G. Synthesis of Mannich Bases of Some 2,5-Disubstituted 4-Thiazolidinones and Evaluation of Their Antimicrobial Activities. Arch. Pharm. (Weinheim) 2001, 334, 35–39. [Google Scholar] [CrossRef]
- El-Serwy, W.S.; Mohamed, N.A.; El-Serwy, W.S.; Mahmoud, A.H. Synthesis, biological evaluation and molecular modeling studies of some 5-methylisoxazole derivatives as anti-Inflammatory agents. J. Chem. Pharmaceut. Res. 2017, 9, 117–127. [Google Scholar]
- Ibrahim, B.M.M.; Yassin, N.A.Z.; Hetta, M.H.; Ta, K.F.; Mohammed, W.I.; Hassan, M.E.S. Phytochemical and pharmacological studies on newly-suggested herbal formulations for potential protection against inflammatory conditions. Int. J. Pharmacogn. Phytochem. Res. 2016, 8, 2044–2055. [Google Scholar]
- Owen, C.A.; Campbell, E.J. Neutrophil Proteinases and Matrix Degradation. The Cell Biology of Pericellular Proteolysis. Semin. Cell Biol. 1995, 6, 367–376. [Google Scholar] [CrossRef]
- Leelaprakash, G.; Dass, S.M. In vitro anti-inflammatory activity of methanol extract of Enicostemma axillare. Int. J. Drug Dev. Res. 2011, 3, 189–196. [Google Scholar]
- Ruiz-Ruiz, J.C.; Matus-Basto, A.J.; Acereto-Escoffié, P.; Segura-Campos, M.R. Antioxidant and Anti-Inflammatory Activities of Phenolic Compounds Isolated from Melipona Beecheii Honey. Food Agric. Immunol. 2017, 28, 1424–1437. [Google Scholar] [CrossRef]
- Garavito, R.M.; DeWitt, D.L. The Cyclooxygenase Isoforms: Structural Insights into the Conversion of Arachidonic Acid to Prostaglandins. Biochim. Biophys. Acta Mol. Cell Biol. Lipids 1999, 1441, 278–287. [Google Scholar] [CrossRef]
- Llorens, O.; Perez, J.J.; Palomer, A.; Mauleon, D. Structural Basis of the Dynamic Mechanism of Ligand Binding to Cyclooxygenase. Bioorg. Med. Chem. Lett. 1999, 9, 2779–2784. [Google Scholar] [CrossRef]
- Molinspiration Cheminformatics. Available online: http://www.molinspiration.com (accessed on 24 February 2018).
- Lipinski, C.A.; Lombardo, F.; Dominy, B.W.; Feeney, P.J. Experimental and Computational Approaches to Estimate Solubility and Permeability in Drug Discovery and Development Settings. Adv. Drug Deliv. Rev. 2001, 46, 3–26. [Google Scholar] [CrossRef]
- Clark, D.E. Rapid Calculation of Polar Molecular Surface Area and Its Application to the Prediction of Transport Phenomena. 2. Prediction of Blood–Brain Barrier Penetration. J. Pharm. Sci. 1999, 88, 815–821. [Google Scholar] [CrossRef]
- Bhatt, N.S.; Shah, A.K.; Raval, R.V.; Thakor, V. Novel Synthesis of Benzofurans. Curr. Sci. 1984, 53, 1289–1290. [Google Scholar]
- Kapewangolo, P.; Omolo, J.J.; Bruwer, R.; Fonteh, P.; Meyer, D. Antioxidant and Anti-Inflammatory Activity of Ocimum Labiatum Extract and Isolated Labdane Diterpenoid. J. Inflamm. Lond. Engl. 2015, 12, 1–13. [Google Scholar] [CrossRef]
- Moharram, F.A.-E.; Al-Gendy, A.A.; El-Shenawy, S.M.; Ibrahim, B.M.; Zarka, M.A. Phenolic profile, anti-inflammatory, antinociceptive, anti-ulcerogenic and hepatoprotective activities of Pimenta racemosa leaves. BMC Complement Altern Med. 2018, 18, 208. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are available from the authors. |
| Compd | IC50 | Compd | IC50 | Compd | IC50 |
|---|---|---|---|---|---|
| 2a | — | 5f | — | 8d | 4.30 ± 0.531 |
| 2b | — | 7a | 93.12 ± 0.727 | 8e | 32.85 ± 1.322 |
| 3a | — | 7b | 61.78 ± 2.719 | 8f | 35.36 ± 3.265 |
| 3b | 48.38 ± 4.616 | 7c | 120.12 ± 4.629 | 9a | — |
| 4a | — | 7d | 112.19 ± 4.391 | 9b | — |
| 4b | — | 7e | 71.67 ± 3.231 | 9c | — |
| 5a | — | 7f | 83.52 ± 1.849 | 9d | — |
| 5c | — | 8a | 14.51 ± 1.827 | 9e | — |
| 5d | 82.92 ± 3.300 | 8b | 19.25 ± 4.171 | 9f | — |
| 5e | — | 8c | 3.87 ± 0.409 | Ascorbic acid * | 2.83 ± 0.166 |
| Compd | Consensus Log P a | Gram +ve Bacteria | Gram −ve Bacteria | Fungi | ||||
|---|---|---|---|---|---|---|---|---|
| SA | BS | BM | EC | PA | SC | CA | ||
| 2a | 2.5 | 24 | 22 | 25 | 18 | 20 | 25 | 22 |
| 2b | 2.39 | 25 | 23 | 25 | 18 | 21 | 27 | 23 |
| 3a | 1.76 | - | 18 | 16 | 16 | 15 | - | - |
| 3b | 1.75 | 16 | - | - | 16 | 16 | 17 | 18 |
| 4a | 3.03 | 16 | - | - | - | - | - | - |
| 4b | 2.94 | 16 | - | - | 15 | 15 | - | - |
| 5a | 4.29 | 25 | 27 | 22 | 26 | 24 | 26 | 25 |
| 5c | 4.21 | 23 | 23 | 24 | 24 | 25 | 27 | 24 |
| 5d | 4.21 | 18 | - | - | - | - | 19 | 19 |
| 5e | 5.33 | 16 | 16 | 16 | 15 | 15 | 22 | 20 |
| 5f | 5.28 | 25 | 23 | 20 | 20 | 21 | 24 | 22 |
| 7a | 1.23 | 28 | 29 | 25 | 28 | 27 | 26 | 26 |
| 7b | 1.05 | - | - | - | - | - | - | - |
| 7c | 1.88 | 30 | 31 | 30 | 30 | 24 | 31 | 27 |
| 7d | 1.71 | 30 | 31 | 30 | 28 | 25 | 32 | 30 |
| 7e | 2.51 | 23 | 25 | 25 | 20 | 22 | 20 | 23 |
| 7f | 2.36 | 24 | 27 | 27 | 23 | 24 | 26 | 24 |
| 8a | 2.52 | 26 | 28 | 29 | 25 | 26 | 28 | 29 |
| 8b | 2.41 | 20 | 19 | 24 | 18 | 17 | 19 | 20 |
| 8c | 2.03 | 30 | 29 | 28 | 27 | 29 | 32 | 30 |
| 8d | 1.90 | 30 | 29 | 29 | 31 | 31 | 32 | 30 |
| 8e | 2.45 | - | 15 | 16 | - | - | - | - |
| 8f | 2.32 | 16 | 15 | 15 | - | - | - | - |
| 9a | 3.5 | 16 | 18 | 18 | - | - | - | - |
| 9b | 3.15 | 22 | 24 | 25 | 26 | 26 | 25 | 23 |
| 9c | 4.48 | 28 | 29 | 30 | 30 | 30 | 31 | 30 |
| 9d | 4.32 | 28 | 29 | 29 | 38 | 29 | 32 | 30 |
| 9e | 3.88 | 23 | 21 | 20 | 21 | 21 | 29 | 23 |
| 9f | 3.73 | 25 | 25 | 26 | 26 | 28 | 26 | 24 |
| Ciprofloxacin | 28 | 30 | 30 | 30 | 30 | - | - | |
| Ketoconazole | - | - | - | - | - | 30 | 28 | |
| Compd | Gram +ve Bacteria | Gram –ve Bacteria | Fungi | ||||
|---|---|---|---|---|---|---|---|
| SA | BS | BM | EC | PA | SC | CA | |
| 2a | 500 | 500 | 500 | 1000 | 500 | 500 | 500 |
| 2b | 500 | 500 | 500 | 1000 | 1000 | 500 | 500 |
| 3a | N.D. | 1000 | N.D. | N.D. | N.D. | N.D. | N.D. |
| 3b | N.D. | N.D. | N.D. | N.D. | N.D. | 1000 | 1000 |
| 5a | 250 | 250 | 500 | 250 | 500 | 500 | 500 |
| 5c | 500 | 500 | 500 | 500 | 500 | 500 | 500 |
| 5d | 1000 | N.D. | N.D. | N.D. | N.D. | 1000 | 1000 |
| 5e | N.D. | N.D. | N.D. | N.D. | N.D. | 500 | 500 |
| 5f | 500 | 500 | 1000 | 1000 | 1000 | 500 | 500 |
| 7a | 250 | 250 | 500 | 250 | 250 | 250 | 250 |
| 7c | 125 | 125 | 125 | 125 | 250 | 125 | 125 |
| 7d | 125 | 125 | 125 | 125 | 125 | 125 | 125 |
| 7e | 500 | 500 | 500 | 500 | 1000 | 1000 | 500 |
| 7f | 250 | 250 | 250 | 500 | 500 | 250 | 500 |
| 8a | 500 | 250 | 250 | 500 | 500 | 250 | 250 |
| 8b | 1000 | 1000 | 1000 | 1000 | 1000 | 1000 | 1000 |
| 8c | 125 | 125 | 250 | 250 | 250 | 125 | 125 |
| 8d | 125 | 125 | 250 | 125 | 125 | 125 | 125 |
| 9a | N.D. | 1000 | 1000 | N.D. | N.D. | N.D. | N.D. |
| 9b | 500 | 500 | 500 | 250 | 250 | 500 | 500 |
| 9c | 250 | 250 | 250 | 250 | 250 | 125 | 125 |
| 9d | 250 | 250 | 250 | 250 | 250 | 125 | 125 |
| 9e | 500 | 500 | 500 | 500 | 500 | 250 | 500 |
| 9f | 500 | 500 | 250 | 250 | 250 | 500 | 500 |
| Ciprofloxacin | 125 | 125 | 125 | 125 | 125 | N.D. | N.D. |
| Ketoconazole | N.D. | N.D. | N.D. | N.D. | N.D. | 125 | 125 |
| Compd | % Inhibition | Compd | % Inhibition | Compd | % Inhibition |
|---|---|---|---|---|---|
| 2a | 17.38 ± 1.63 | 5f | 26.83 ± 5.33 | 8d | 20.15 ± 7.31 |
| 2b | 74.68 ± 3.01 | 7a | 18.78 ± 2.05 | 8e | 18.5 ± 3.47 |
| 3a | 79.72 ± 4.51 | 7b | 5.93 ± 2.11 | 8f | 2.0 ± 1.4 |
| 3b | 49.28 ± 10.93 | 7c | 8.9 ± 2.04 | 9a | 62.14 ± 4.87 |
| 4a | 31.99 ± 2.45 | 7d | 36.84 ± 4.87 | 9b | — |
| 4b | 33.17 ± 2.79 | 7e | 19.8 ± 2.54 | 9c | 17.6 ± 6.86 |
| 5a | N.D. | 7f | 43.88 ± 5.6 | 9d | 31.88 ± 3.82 |
| 5c | 68.5 ± 6.23 | 8a | 44.7 ± 8.22 | 9e | 22.83 ± 4.88 |
| 5d | 43.4 ± 1.52 | 8b | 14.3 ± 2.21 | 9f | 21.7 ± 7.6 |
| 5e | 45.78 ± 1.62 | 8c | 41.69 ± 2.83 | Aspirin * | 45.83 ± 4.21 |
| Compd | IC50 (µM) a,b | SI c | |
|---|---|---|---|
| COX-1 | COX-2 | ||
| 2a | 116.66 | 2.74 | 42.58 |
| 2b | 201.16 | 4.25 | 47.33 |
| 3a | 183.27 | 9.81 | 18.68 |
| 3b | 7.73 | 2.45 | 3.16 |
| 4a | 12.49 | 7.99 | 1.56 |
| 4b | 57.46 | 25.23 | 2.28 |
| 5a | 1126.47 | 7.41 | 152.02 |
| 5b | 215.39 | 19.63 | 10.97 |
| 5c | 18.50 | 13.30 | 1.39 |
| 5d | 27.19 | 16.04 | 1.7 |
| 5e | 20.80 | 8.27 | 2.52 |
| 5f | 90.20 | 3.10 | 29.1 |
| 7a | 239.47 | 17.42 | 13.75 |
| 7b | 47.60 | 8.24 | 5.78 |
| 7c | 62.27 | 19.94 | 3.12 |
| 7d | 214.28 | 3.35 | 63.96 |
| 7e | 29.62 | 16.11 | 1.84 |
| 7f | 40.89 | 10.07 | 4.06 |
| 8a | 25.02 | 8.50 | 2.94 |
| 8b | 25.99 | 13.37 | 1.94 |
| 8c | 15.40 | 11.90 | 1.29 |
| 8d | 19.44 | 1.86 | 10.45 |
| 8e | 19.90 | 9.22 | 2.16 |
| 8f | 7.92 | 25.66 | 0.31 |
| Indomethacin | 1.88 | 15.14 | 0.12 |
| Celecoxib | 187.82 | 0.77 | 243.92 |
| Compd | Mol. Wt | miLogP | n-ON | n-OHNH | RO5 Violations | TPSA (Å2) |
|---|---|---|---|---|---|---|
| Rule | ≤500 | ≤5 | ≤10 | ≤5 | ≤1 | ≤140 |
| 2a | 375.34 | 3.10 | 8 | 2 | 0 | 135.08 |
| 2b | 320.30 | 2.40 | 6 | 2 | 0 | 102.40 |
| 3a | 393.36 | 2.05 | 9 | 4 | 0 | 154.39 |
| 3b | 338.32 | 1.35 | 7 | 4 | 0 | 121.70 |
| 4a | 430.42 | 3.58 | 9 | 0 | 0 | 124.66 |
| 4b | 375.38 | 2.88 | 7 | 0 | 0 | 91.98 |
| 5a | 463.45 | 5.16 | 8 | 0 | 1 | 121.42 |
| 5b | 408.41 | 4.45 | 6 | 0 | 0 | 88.74 |
| 5c | 553.53 | 4.79 | 11 | 0 | 2 | 149.13 |
| 5d | 498.49 | 4.08 | 9 | 0 | 0 | 116.44 |
| 5e | 532.34 | 6.44 | 8 | 0 | 2 | 121.42 |
| 5f | 477.30 | 5.74 | 6 | 0 | 1 | 88.74 |
| 7a | 430.85 | 1.76 | 9 | 4 | 0 | 156.77 |
| 7b | 410.43 | 1.53 | 9 | 4 | 0 | 156.77 |
| 7c | 508.92 | 2.01 | 11 | 2 | 2 | 168.56 |
| 7d | 488.50 | 1.78 | 11 | 3 | 1 | 168.56 |
| 7e | 513.96 | 2.89 | 10 | 3 | 1 | 155.67 |
| 7f | 493.54 | 2.66 | 10 | 3 | 0 | 155.67 |
| 8a | 351.77 | 3.07 | 6 | 2 | 0 | 96.61 |
| 8b | 331.35 | 2.84 | 6 | 2 | 0 | 96.61 |
| 8c | 367.77 | 2.59 | 7 | 3 | 0 | 116.84 |
| 8d | 347.35 | 2.36 | 7 | 3 | 0 | 116.84 |
| 8e | 393.80 | 2.97 | 7 | 2 | 0 | 113.68 |
| 8f | 373.39 | 2.74 | 7 | 2 | 0 | 113.68 |
| 9a | 526.91 | 4.90 | 10 | 2 | 1 | 159.50 |
| 9b | 506.49 | 4.67 | 10 | 2 | 1 | 159.50 |
| 9c | 516.36 | 5.62 | 7 | 2 | 2 | 113.68 |
| 9d | 495.94 | 5.39 | 7 | 2 | 1 | 113.68 |
| 9e | 511.94 | 5.00 | 8 | 2 | 2 | 122.91 |
| 9f | 491.52 | 4.77 | 8 | 2 | 0 | 122.91 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alshibl, H.M.; Al-Abdullah, E.S.; Haiba, M.E.; Alkahtani, H.M.; Awad, G.E.A.; Mahmoud, A.H.; Ibrahim, B.M.M.; Bari, A.; Villinger, A. Synthesis and Evaluation of New Coumarin Derivatives as Antioxidant, Antimicrobial, and Anti-Inflammatory Agents. Molecules 2020, 25, 3251. https://doi.org/10.3390/molecules25143251
Alshibl HM, Al-Abdullah ES, Haiba ME, Alkahtani HM, Awad GEA, Mahmoud AH, Ibrahim BMM, Bari A, Villinger A. Synthesis and Evaluation of New Coumarin Derivatives as Antioxidant, Antimicrobial, and Anti-Inflammatory Agents. Molecules. 2020; 25(14):3251. https://doi.org/10.3390/molecules25143251
Chicago/Turabian StyleAlshibl, Hanan M., Ebtehal S. Al-Abdullah, Mogedda E. Haiba, Hamad M. Alkahtani, Ghada E.A. Awad, Ahlam H. Mahmoud, Bassant M.M. Ibrahim, Ahmed Bari, and Alexander Villinger. 2020. "Synthesis and Evaluation of New Coumarin Derivatives as Antioxidant, Antimicrobial, and Anti-Inflammatory Agents" Molecules 25, no. 14: 3251. https://doi.org/10.3390/molecules25143251
APA StyleAlshibl, H. M., Al-Abdullah, E. S., Haiba, M. E., Alkahtani, H. M., Awad, G. E. A., Mahmoud, A. H., Ibrahim, B. M. M., Bari, A., & Villinger, A. (2020). Synthesis and Evaluation of New Coumarin Derivatives as Antioxidant, Antimicrobial, and Anti-Inflammatory Agents. Molecules, 25(14), 3251. https://doi.org/10.3390/molecules25143251








