Technologies and Formulation Design of Polysaccharide-Based Hydrogels for Drug Delivery
Abstract
1. Introduction
- gives the possibility to realize controlled release according to specific therapeutic needs
- ensures the protection against the action of environmental and physiological agents
- can modify pharmacokinetic and bio-distribution profiles
- can reduce clearance and side effects
- improve drug targeting.
- (1)
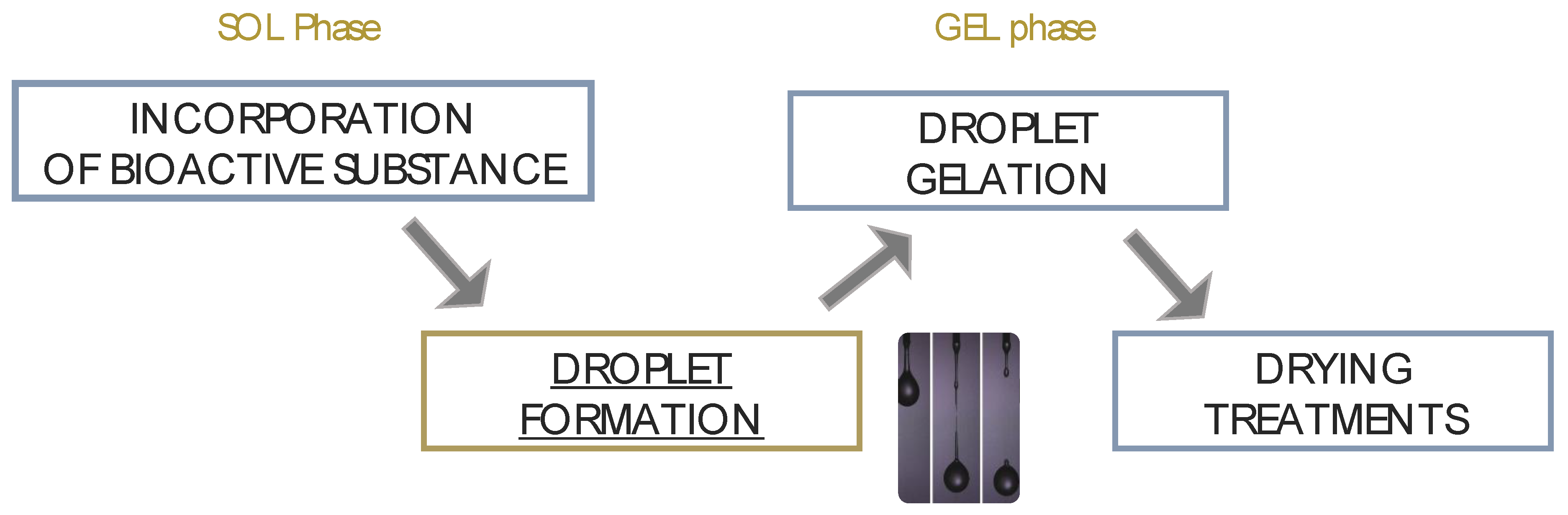
- Formation of droplets in a gaseous phase with following fall in a gelling medium.
- (2)
- Formation of droplets in a liquid phase that is immiscible with the polymeric solution; in this case, the mixing leads to an emulsion.
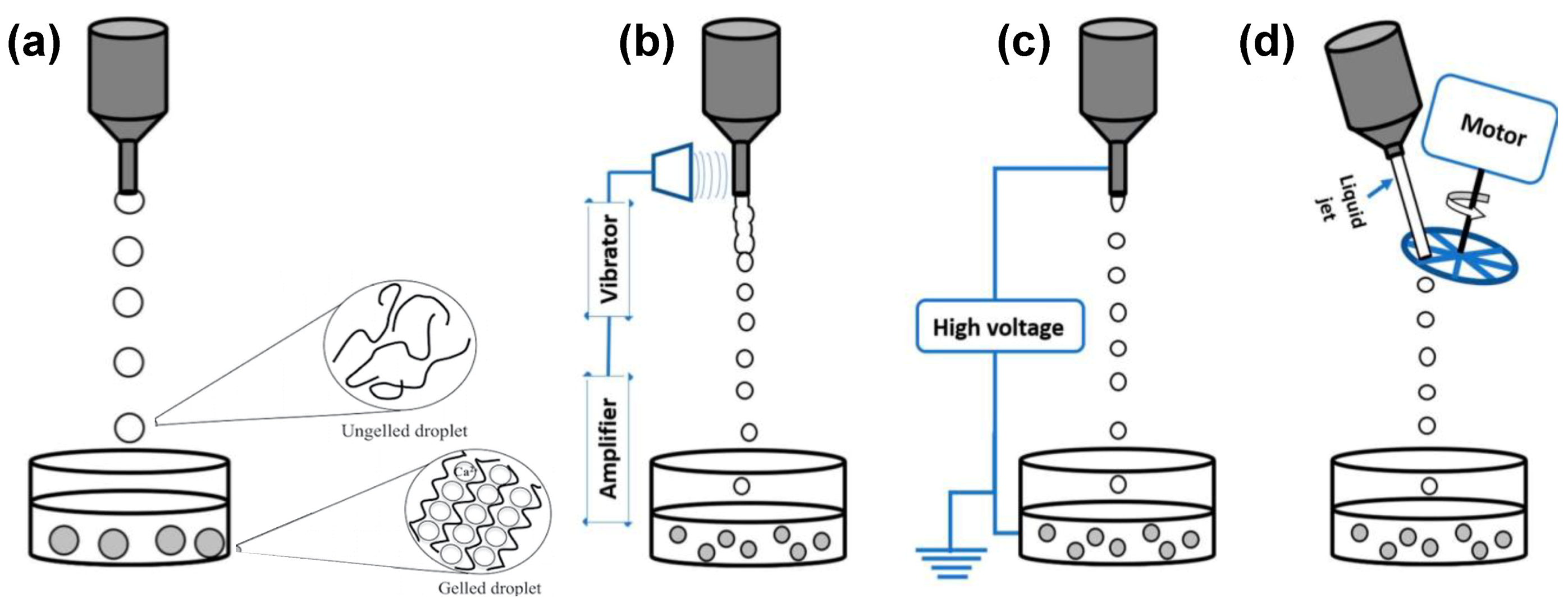
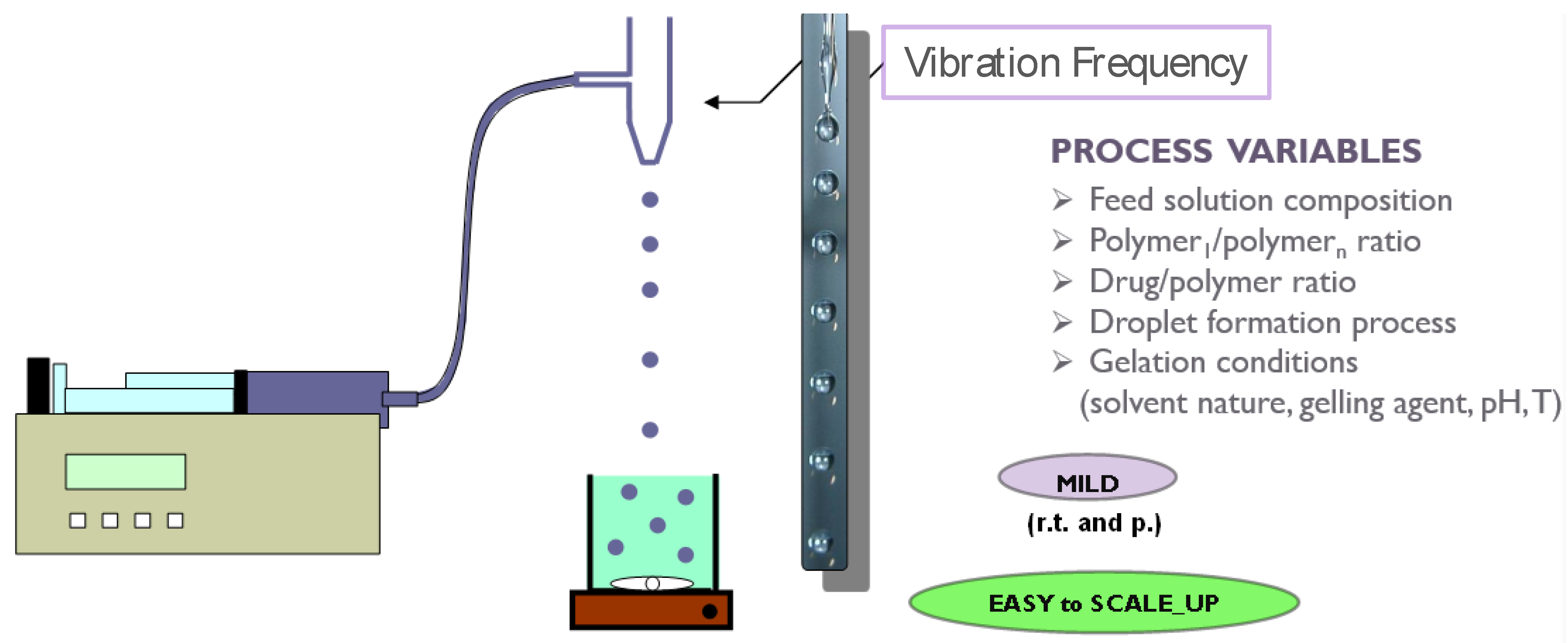
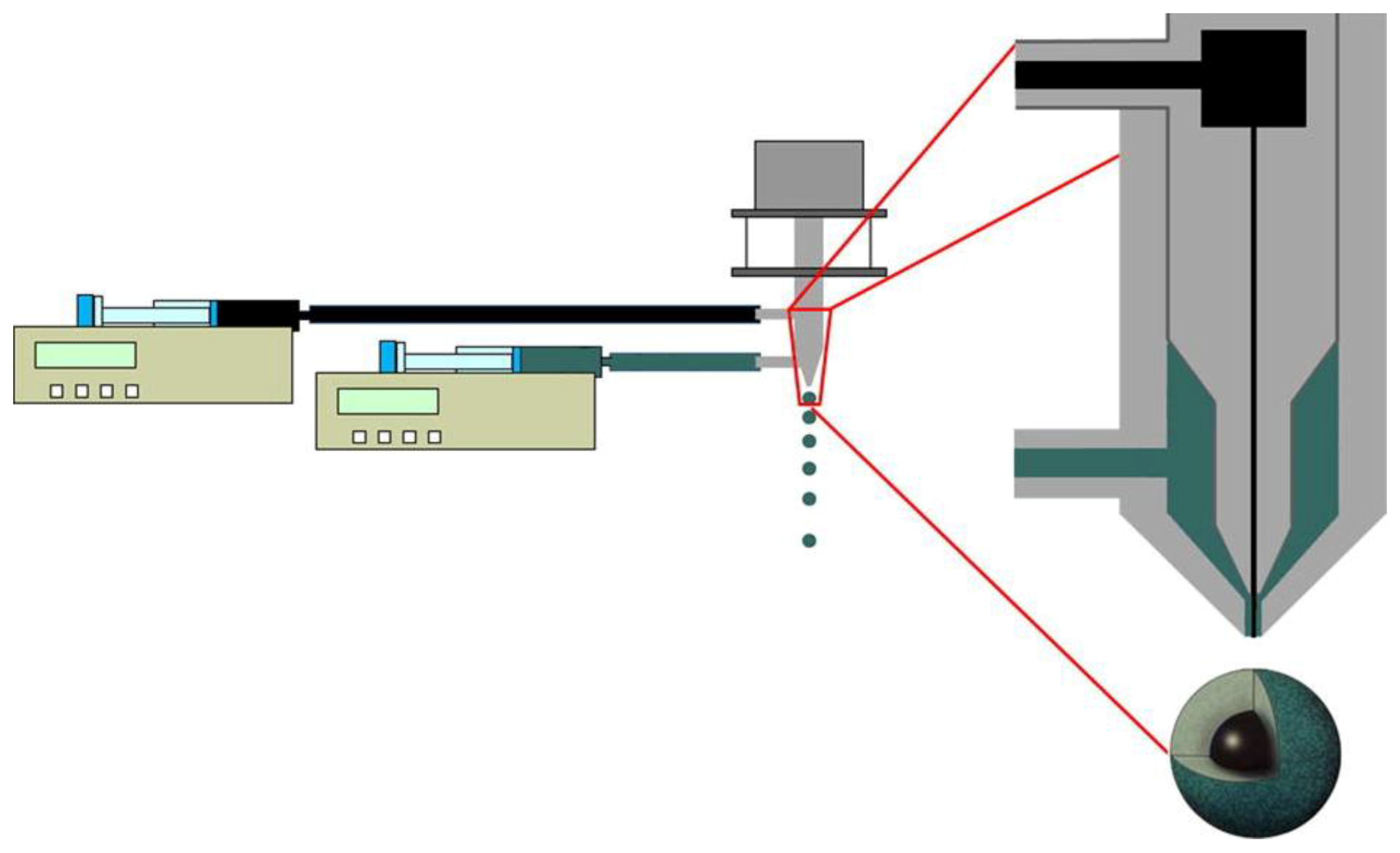
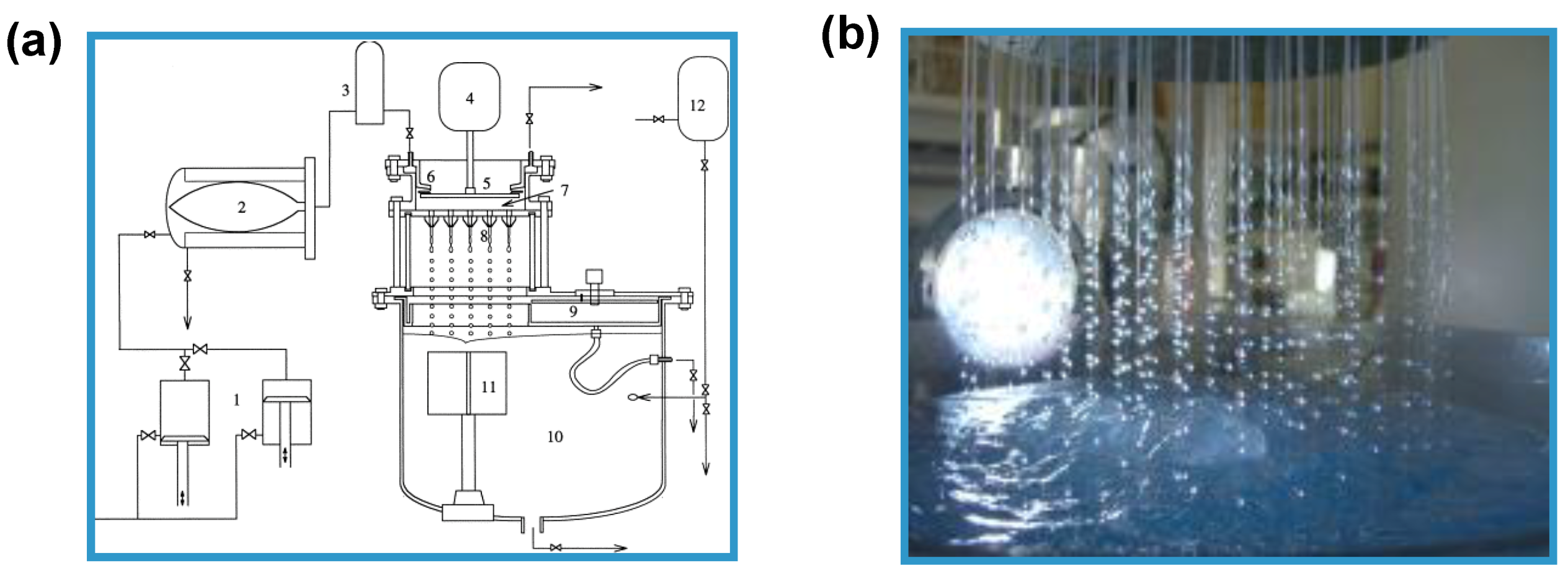
2. Prilling Technique to Produce Polymeric Droplets
Prilling or Laminar Jet Break-Up
3. Methods for the Gelation of Polymeric Droplets to Produce Gel-Particles
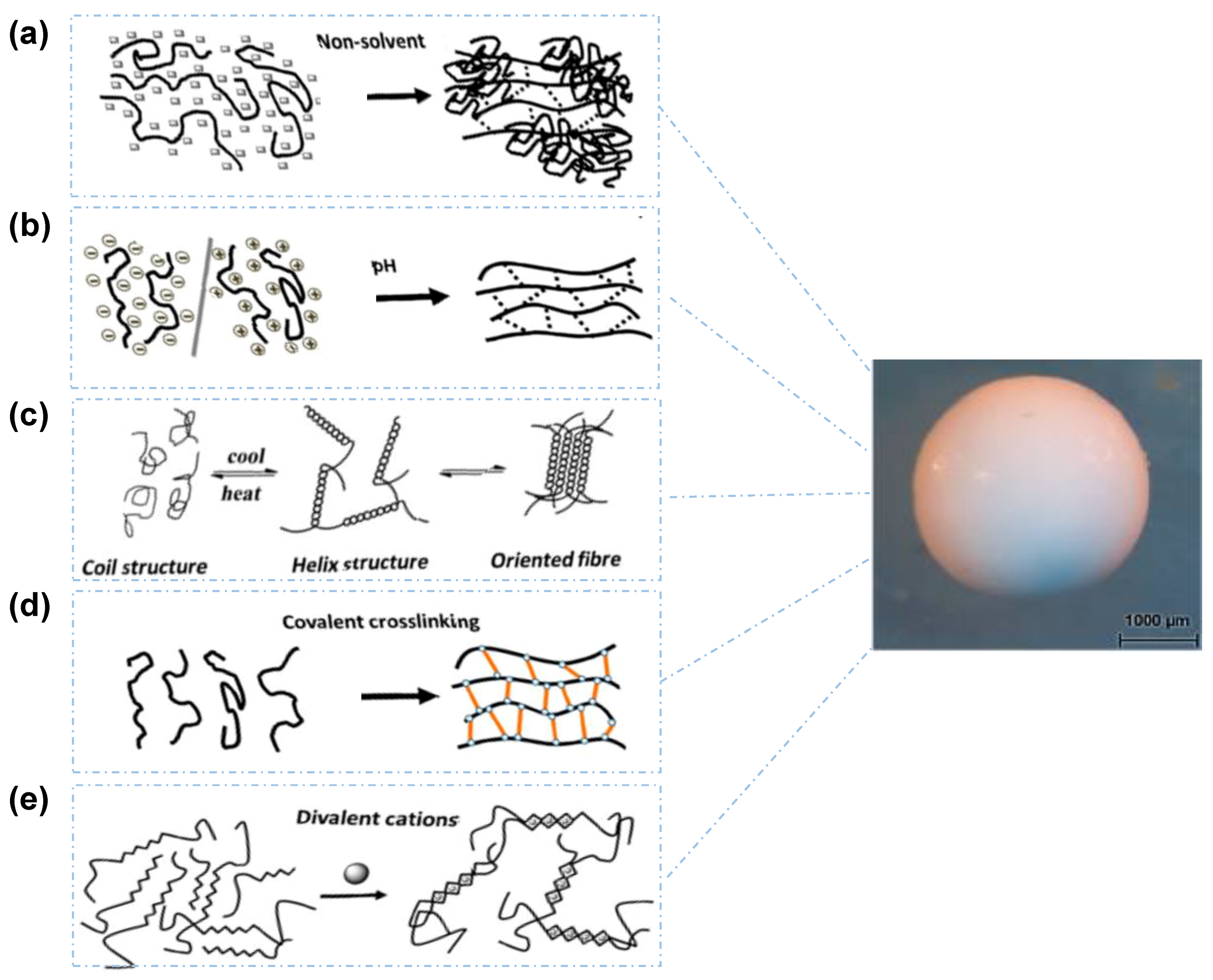
3.1. Non-Solvent Induced Phase Separation
3.2. pH-Induced Gelation
3.3. Temperature-Induced Gelation
3.4. Chemical Gelation
3.5. Ionotropic Cross-Linking
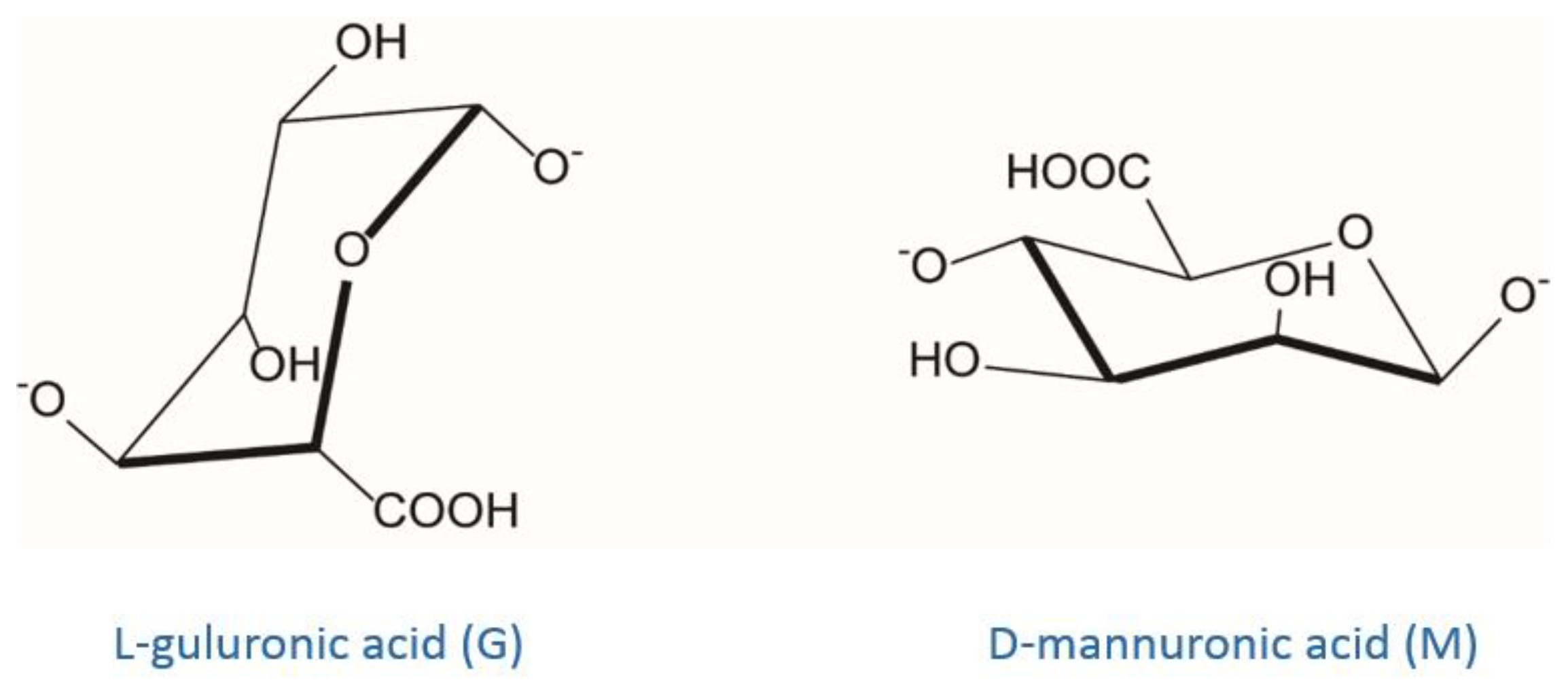
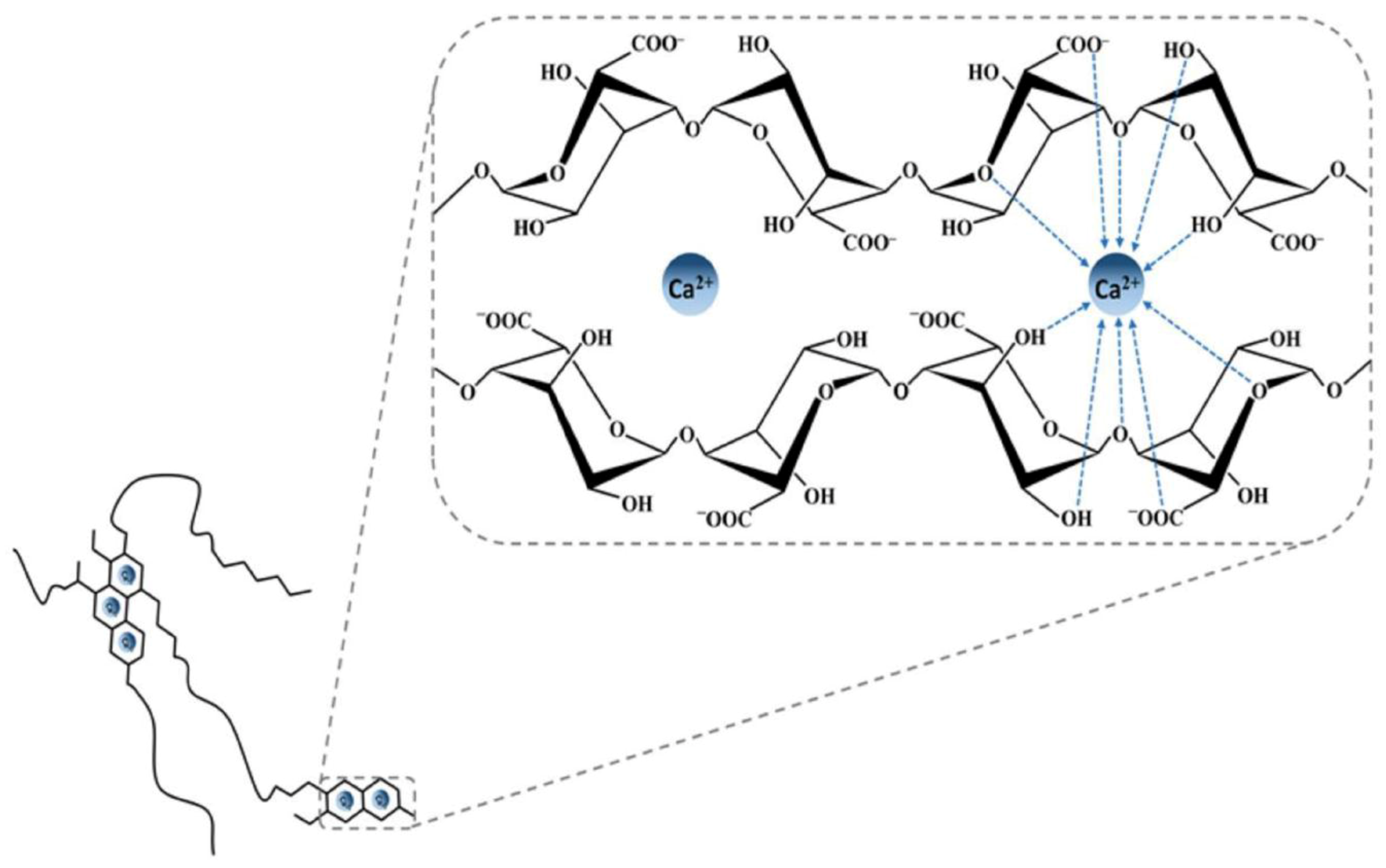
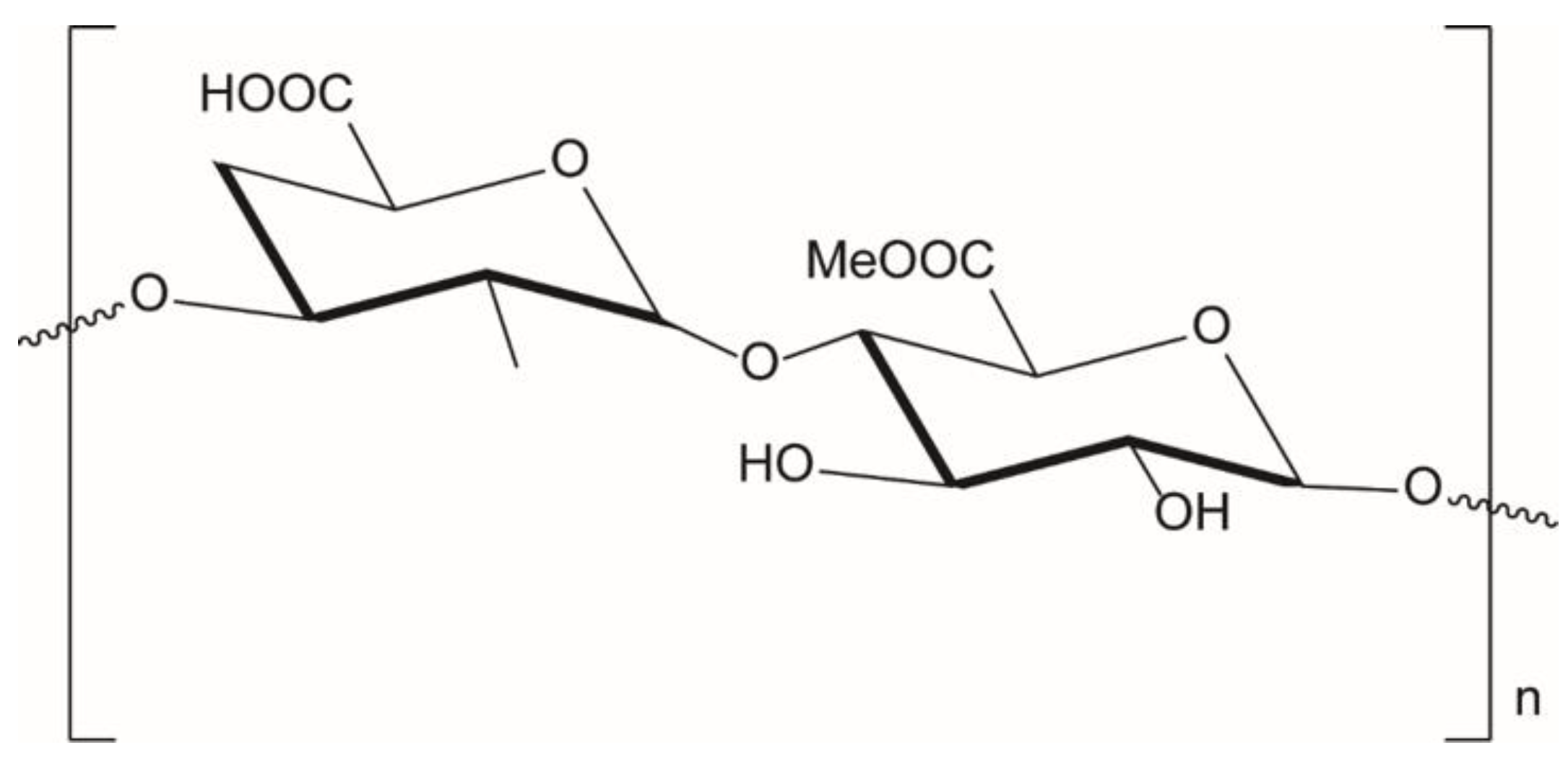
3.5.1. Alginate Ionic Cross-Linking
3.5.2. Pectin Ionic Cross-Linking
3.5.3. Chitosan Ionic Cross-Linking
3.6. Different Approaches to Hydrogel Formation by Ionotropic Cross-Linking
4. Influence of Drying Process on Gel Particle Characteristics
4.1. Conventional and Dielectric Drying to Produce Xerogels
4.2. Freeze-Drying to Produce Cryogels
4.3. Supercritical Assisted Drying to Produce Aerogels
5. Case-Studies: Polysaccharide-Based Hydrogel Particles Produced by Prilling/Ionotropic Gelation and Their Application as Drug Delivery Systems (DDS)
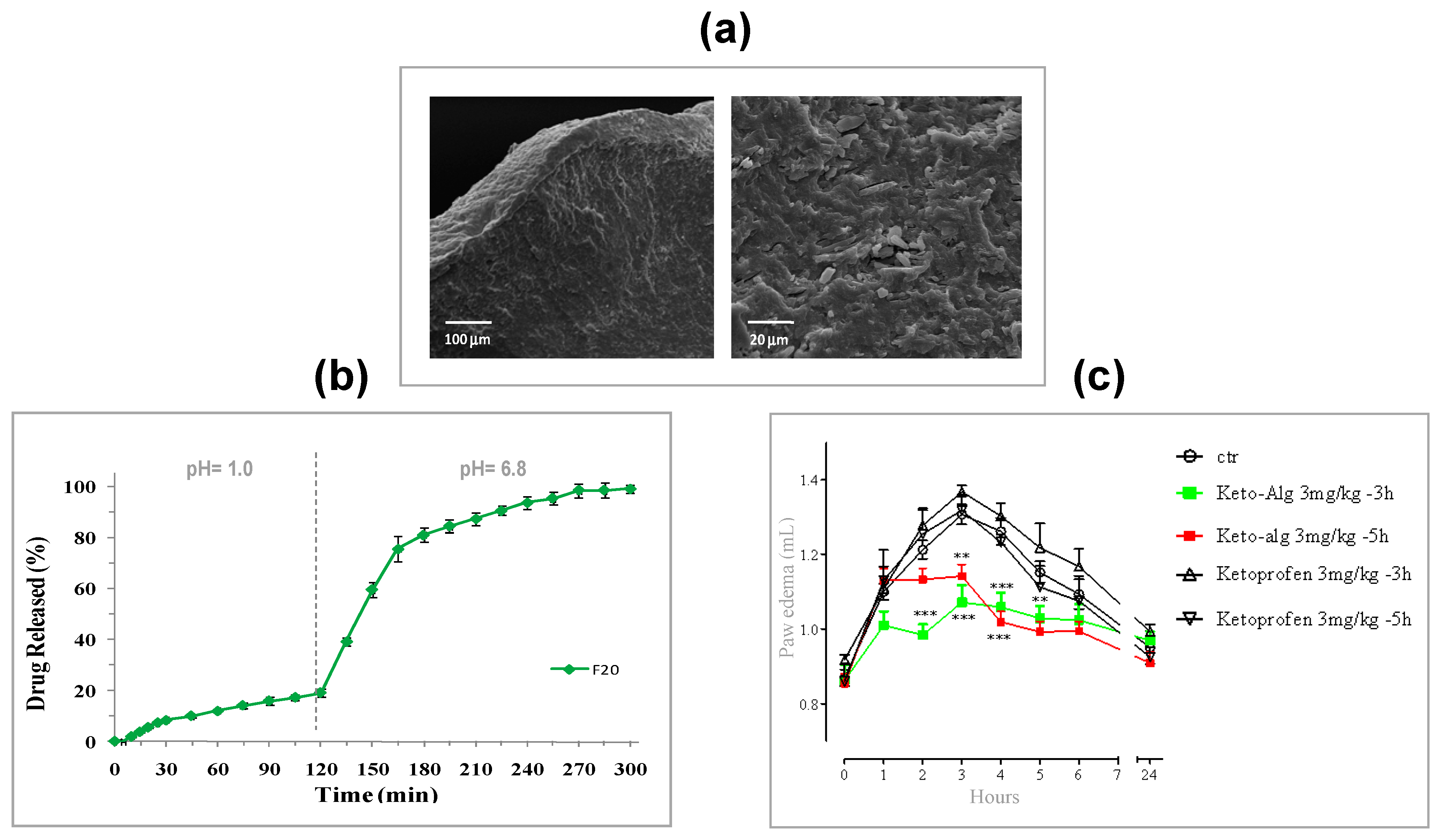
5.1. Design of PbHPs in Form of Xerogels
5.2. Investigation on the Effect of Different Cations on Gelation Process
5.3. Prilling to Obtain Floating PbHPs
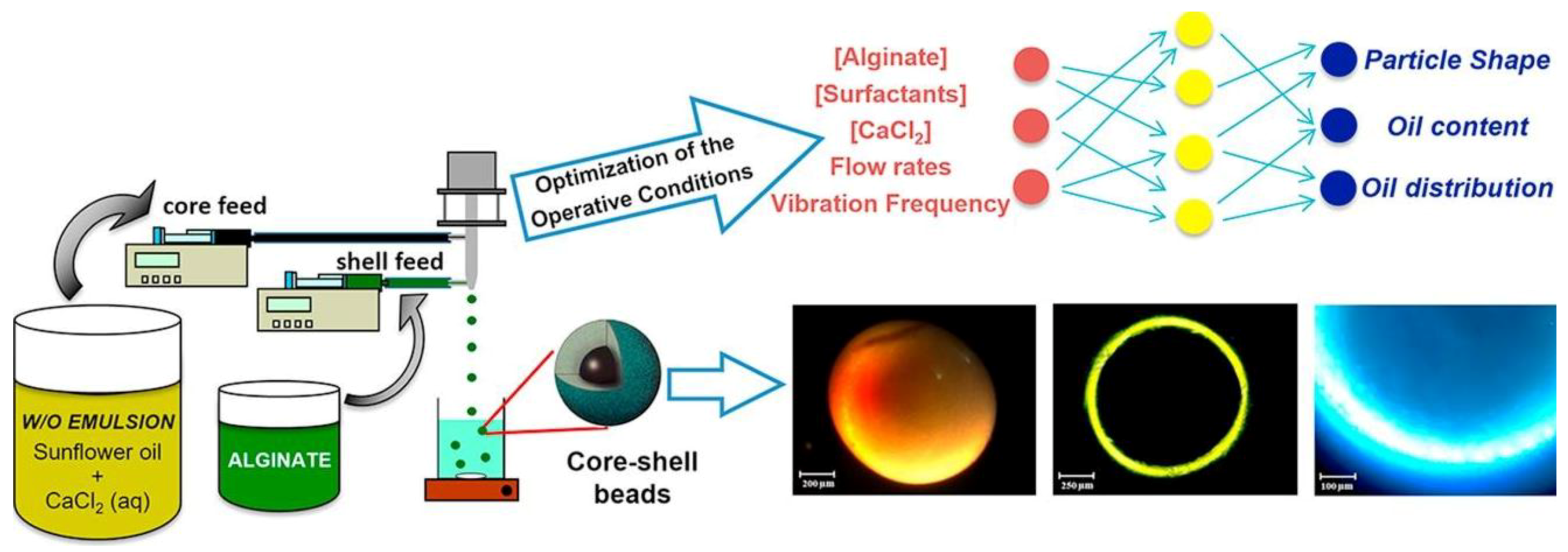
5.4. Core-Shell PbHPs
5.5. Design of PbHPs in Form of Aerogels
5.6. Design of Experiments (DoE) and Artificial Intelligence (AI) for the Productionof PbHP by Inverse Gelation Technique
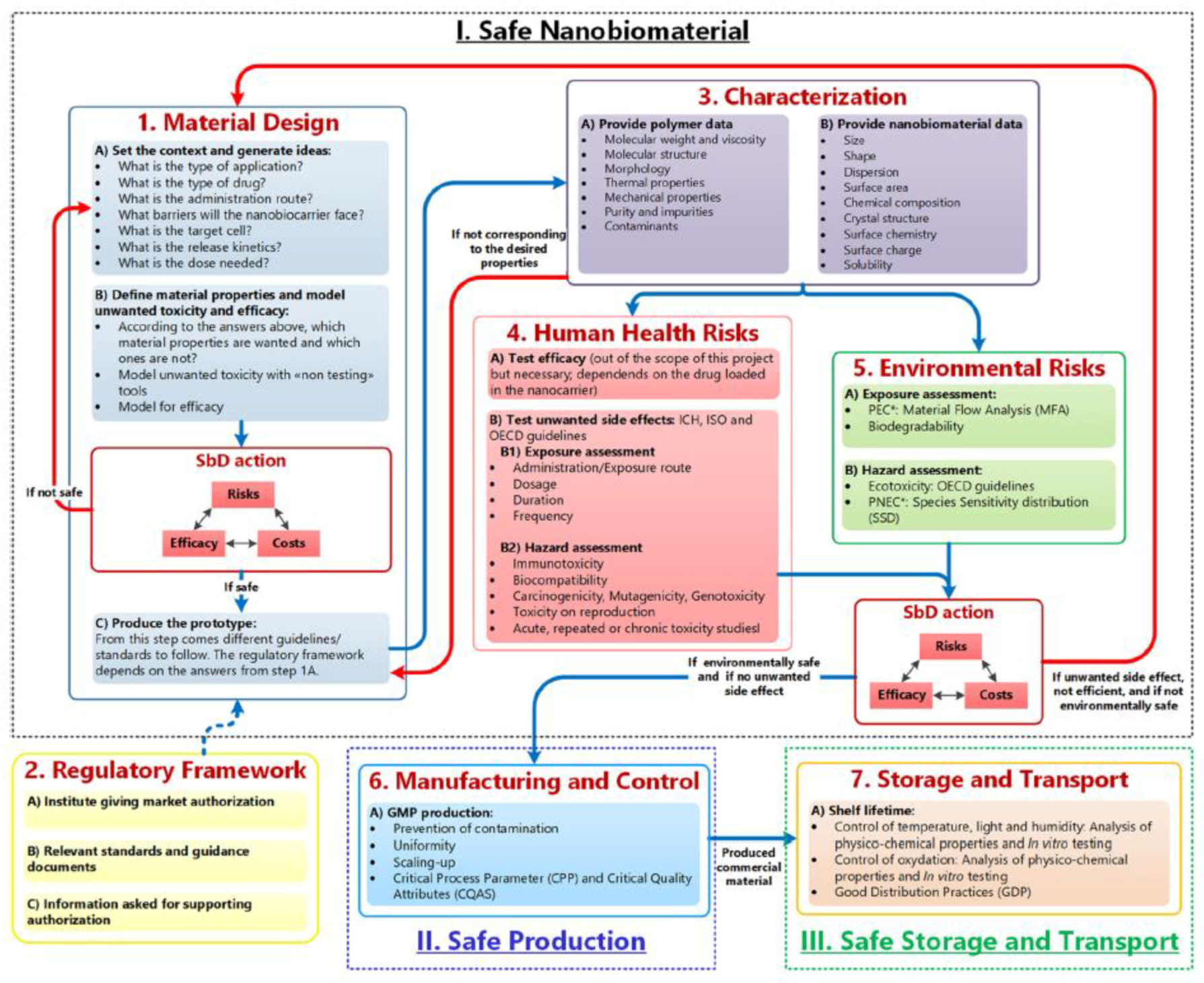
6. PbHPs: Safety by Design and Clinical Translations
- (1)
- understanding how properties of such materials influence the adsorption of bioactive molecules and their effect on cellular reactions,
- (2)
- standardized methods assessing material characteristics as well as biological reactions,
- (1)
- Material Design, Characterization, Human Health and Environmental Risks
- (2)
- Manufacturing and control
- (3)
- Storage and Transport.
7. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kim, K.K.; Pack, D.W. Microspheres for drug delivery. In Biomems and Biomedical Nanotechnology; Springer: Boston, MA, USA, 2006; pp. 19–50. [Google Scholar]
- Miao, T.; Wang, J.; Zeng, Y.; Liu, G.; Chen, X. Polysaccharide-Based Controlled Release Systems for Therapeutics Delivery and Tissue Engineering: From Bench to Bedside. Adv. Sci. (Weinh) 2018, 5, 1700513. [Google Scholar] [CrossRef] [PubMed]
- Dormer, N.H.; Berkland, C.J.; Singh, M. Monodispersed microencapsulation technology. In Microencapsulation in the Food Industry; Elsevier Science: Amsterdam, The Netherlands, 2014; pp. 111–123. [Google Scholar]
- Ganesan, K.; Budtova, T.; Ratke, L.; Gurikov, P.; Baudron, V.; Preibisch, I.; Niemeyer, P.; Smirnova, I.; Milow, B. Review on the production of polysaccharide aerogel particles. Mater. (Basel) 2018, 11, 2144. [Google Scholar] [CrossRef] [PubMed]
- Chan, E.-S. Preparation of Ca-alginate beads containing high oil content: Influence of process variables on encapsulation efficiency and bead properties. Carbohydr. Polym. 2011, 84, 1267–1275. [Google Scholar] [CrossRef]
- Smidsrød, O.; Skja, G. Alginate as immobilization matrix for cells. Trends Biotechnol. 1990, 8, 71–78. [Google Scholar] [CrossRef]
- Fundueanu, G.; Nastruzzi, C.; Carpov, A.; Desbrieres, J.; Rinaudo, M. Physico-chemical characterization of Ca-alginate microparticles produced with different methods. Biomaterials 1999, 20, 1427–1435. [Google Scholar] [CrossRef]
- Zohar-Perez, C.; Chet, I.; Nussinovitch, A. Irregular textural features of dried alginate-filler beads. Food Hydrocoll. 2004, 18, 249–258. [Google Scholar] [CrossRef]
- Chan, E.-S.; Lim, T.-K.; Voo, W.-P.; Pogaku, R.; Tey, B.T.; Zhang, Z. Effect of formulation of alginate beads on their mechanical behavior and stiffness. Particuology 2011, 9, 228–234. [Google Scholar] [CrossRef]
- Burey, P.; Bhandari, B.R.; Howes, T.; Gidley, M.J. Hydrocolloid Gel Particles: Formation, Characterization, and Application. Crit. Rev. Food Sci. Nutr 2008, 48, 361–377. [Google Scholar] [CrossRef]
- Krasaekoopt, W.; Bhandari, B.; Deeth, H. The influence of coating materials on some properties of alginate beads and survivability of microencapsulated probiotic bacteria. Int. Dairy J. 2004, 14, 737–743. [Google Scholar] [CrossRef]
- Krasaekoopt, W.; Bhandari, B.; Deeth, H.C. Survival of probiotics encapsulated in chitosan-coated alginate beads in yoghurt from UHT- and conventionally treated milk during storage. Lwt-Food Sci. Technol. 2006, 39, 177–183. [Google Scholar] [CrossRef]
- Nedovic, V.; Willaert, R. Alginate as a carrier for cell immobilisation. In Fundamentals of Cell Immobilisation Biotechnology; Springer Science & Business Media: Dordrecht, The Netherlands, 2013; Volume 8. [Google Scholar]
- Chan, E.-S.; Yim, Z.-H.; Phan, S.-H.; Mansa, R.F.; Ravindra, P. Encapsulation of herbal aqueous extract through absorption with ca-alginate hydrogel beads. Food Bioprod. Process. 2010, 88, 195–201. [Google Scholar] [CrossRef]
- Ching, S.H.; Bansal, N.; Bhandari, B. Alginate gel particles–A review of production techniques and physical properties. Crit. Rev. Food Sci. Nutr 2017, 57, 1133–1152. [Google Scholar] [CrossRef] [PubMed]
- Blandino, A.; Macías, M.; Cantero, D. Formation of calcium alginate gel capsules: Influence of sodium alginate and CaCl2 concentration on gelation kinetics. J. Biosci. Bioeng 1999, 88, 686–689. [Google Scholar] [CrossRef]
- Chan, E.-S.; Lee, B.-B.; Ravindra, P.; Poncelet, D. Prediction models for shape and size of ca-alginate macrobeads produced through extrusion–dripping method. J. Colloid Interface Sci. 2009, 338, 63–72. [Google Scholar] [CrossRef]
- Leong, J.-Y.; Lam, W.-H.; Ho, K.-W.; Voo, W.-P.; Lee, M.F.-X.; Lim, H.-P.; Lim, S.-L.; Tey, B.-T.; Poncelet, D.; Chan, E.-S. Advances in fabricating spherical alginate hydrogels with controlled particle designs by ionotropic gelation as encapsulation systems. Particuology 2016, 24, 44–60. [Google Scholar] [CrossRef]
- Prüsse, U.; Bilancetti, L.; Bučko, M.; Bugarski, B.; Bukowski, J.; Gemeiner, P.; Lewińska, D.; Manojlovic, V.; Massart, B.; Nastruzzi, C.; et al. Comparison of different technologies for alginate beads production. Chem. Pap. 2008, 62, 364. [Google Scholar] [CrossRef]
- Zvonar, A.; Kristl, J.; Kerč, J.; Grabnar, P.A. High celecoxib-loaded nanoparticles prepared by a vibrating nozzle device. J. Microencapsul. 2009, 26, 748–759. [Google Scholar] [CrossRef]
- Whelehan, M.; Marison, I.W. Microencapsulation using vibrating technology. J. Microencapsul. 2011, 28, 669–688. [Google Scholar] [CrossRef]
- Del Gaudio, P.; De Cicco, F.; Sansone, F.; Aquino, R.P.; Adami, R.; Ricci, M.; Giovagnoli, S. Alginate beads as a carrier for omeprazole/SBA-15 inclusion compound: A step towards the development of personalized paediatric dosage forms. Carbohydr. Polym. 2015, 133, 464–472. [Google Scholar] [CrossRef]
- Del Gaudio, P.; Colombo, P.; Colombo, G.; Russo, P.; Sonvico, F. Mechanisms of formation and disintegration of alginate beads obtained by prilling. Int. J. Pharm. 2005, 302, 1–9. [Google Scholar] [CrossRef]
- Tran, V.-T.; Benoît, J.-P.; Venier-Julienne, M.-C. Why and how to prepare biodegradable, monodispersed, polymeric microparticles in the field of pharmacy? Int. J. Pharm. 2011, 407, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Seifert, D.B.; Phillips, J.A. Production of small, monodispersed alginate beads for cell immobilization. Biotechnol. Prog. 1997, 13, 562–568. [Google Scholar] [CrossRef]
- Brandenberger, H.; Widmer, F. A new multinozzle encapsulation/immobilisation system to produce uniform beads of alginate. J. Biotechnol. 1998, 63, 73–80. [Google Scholar] [CrossRef]
- Heinzen, C.; Marison, I.; Berger, A.; von Stockar, U. Use of vibration technology for jet break-up for encapsulation of cells, microbes and liquids in monodisperse microcapsules. Landbauforsch. VölkenrodeSh241 2002, 19–25. [Google Scholar]
- Eckert, C.; Agnol, W.D.; Dallé, D.; Serpa, V.G.; Maciel, M.J.; Lehn, D.N.; de Souza, C.F.V. Development of alginate-pectin microparticles with dairy whey using vibration technology: Effects of matrix composition on the protection of Lactobacillus spp. from adverse conditions. Food Res. Int. 2018, 113, 65–73. [Google Scholar] [CrossRef]
- Del Gaudio, P.; Russo, P.; Rosaria Lauro, M.; Colombo, P.; Aquino, R.P. Encapsulation of ketoprofen and ketoprofen lysinate by prilling for controlled drug release. Aaps Pharmscitech 2009, 10, 1178–1185. [Google Scholar] [CrossRef]
- Lascol, M.; Bourgeois, S.; Barratier, C.; Marote, P.; Lantéri, P.; Bordes, C. Development of pectin microparticles by using ionotropic gelation with chlorhexidine as cross-linking agent. Int. J. Pharm. 2018, 542, 205–212. [Google Scholar] [CrossRef]
- Auriemma, G.; Cerciello, A.; Aquino, R.P.; Gaudio, P.D.; Fusco, B.M.; Russo, P. Pectin and zinc alginate: The right inner/outer polymer combination for core-shell drug delivery systems. Pharmaceutics 2020, 12, 87. [Google Scholar] [CrossRef]
- Auriemma, G.; Mencherini, T.; Russo, P.; Stigliani, M.; Aquino, R.P.; Del Gaudio, P. Prilling for the development of multi-particulate colon drug delivery systems: Pectin vs. pectin–alginate beads. Carbohydr. Polym. 2013, 92, 367–373. [Google Scholar] [CrossRef]
- Ghosal, S.K.; Talukdar, P.; Pal, T.K. Standardization of a newly designed vibrating capillary apparatus for the preparation of microcapsules. Chem. Eng. Technol. 1993, 16, 395–398. [Google Scholar] [CrossRef]
- Alisch, G.; Brauneis, E.; Pirstadt, B.; Iffland, N.; Brandau, E. Process and Plant for the Production of Spherical Alginate Pellets. U.S. Patent 5472648A, 5 December 1995. [Google Scholar]
- Dumas, H.; Tardy, M.; Rochat, M.; Tayot, J. Prilling process applied to collagen solutions. Drug Dev. Ind. Pharm. 1992, 18, 1395–1409. [Google Scholar] [CrossRef]
- Kim, K.; Pack, D.W.; Berkland, C. Microparticles. U.S. Patent 6669961B2, 30 December 2003. [Google Scholar]
- Ouwerx, C.; Velings, N.; Mestdagh, M.M.; Axelos, M.A.V. Physico-chemical properties and rheology of alginate gel beads formed with various divalent cations. Polym. Gels Netw. 1998, 6, 393–408. [Google Scholar] [CrossRef]
- Mazzitelli, S.; Tosi, A.; Balestra, C.; Nastruzzi, C.; Luca, G.; Mancuso, F.; Calafiore, R.; Calvitti, M. Production and characterization of alginate microcapsules produced by a vibrational encapsulation device. J. Biomater. Appl. 2008, 23, 123–145. [Google Scholar] [CrossRef] [PubMed]
- Berkland, C.; Kim, K.K.; Pack, D.W. Fabrication of PLG microspheres with precisely controlled and monodisperse size distributions. J. Control. Release 2001, 73, 59–74. [Google Scholar] [CrossRef]
- Poncelet, D. Microencapsulation: Fundamentals, methods and applications. In Surface Chemistry in Biomedical and Environmental Science; Springer Dordrecht: Berlin, Germany, 2006; pp. 23–34. [Google Scholar]
- Davarcı, F.; Turan, D.; Ozcelik, B.; Poncelet, D. The influence of solution viscosities and surface tension on calcium-alginate microbead formation using dripping technique. Food Hydrocoll. 2017, 62, 119–127. [Google Scholar] [CrossRef]
- Thu, B.; Smidsrod, O.; Skjåk-Bræk, G. Alginate gels-Some structure-function correlations relevant. Immobil. Cells Basics Appl. 1996, 11, 19–30. [Google Scholar]
- Del Gaudio, P.; Auriemma, G.; Russo, P.; Mencherini, T.; Campiglia, P.; Stigliani, M.; Aquino, R.P. Novel co-axial prilling technique for the development of core–shell particles as delayed drug delivery systems. Eur. J. Pharm. Biopharm. 2014, 87, 541–547. [Google Scholar] [CrossRef]
- De Cicco, F.; Russo, P.; Reverchon, E.; García-González, C.A.; Aquino, R.P.; Del Gaudio, P. Prilling and supercritical drying: A successful duo to produce core-shell polysaccharide aerogel beads for wound healing. Carbohydr. Polym. 2016, 147, 482–489. [Google Scholar] [CrossRef]
- Hiorth, M.; Versland, T.; Heikkilä, J.; Tho, I.; Sande, S.A. Immersion coating of pellets with calcium pectinate and chitosan. Int. J. Pharm. 2006, 308, 25–32. [Google Scholar] [CrossRef]
- Wong, T.W.; Nurjaya, S. Drug release property of chitosan–pectinate beads and its changes under the influence of microwave. Eur. J. Pharm. Biopharm. 2008, 69, 176–188. [Google Scholar] [CrossRef]
- Zhang, Y.; Wei, W.; Lv, P.; Wang, L.; Ma, G. Preparation and evaluation of alginate–chitosan microspheres for oral delivery of insulin. Eur. J. Pharm. Biopharm. 2011, 77, 11–19. [Google Scholar] [CrossRef] [PubMed]
- Luo, Y.; Wang, Q. Recent development of chitosan-based polyelectrolyte complexes with natural polysaccharides for drug delivery. Int. J. Biol. Macromol. 2014, 64, 353–367. [Google Scholar] [CrossRef]
- Brandau, T. Preparation of monodisperse controlled release microcapsules. Int. J. Pharm. 2002, 242, 179–184. [Google Scholar] [CrossRef]
- Lin, C.-x.; Zhan, H.-y.; Liu, M.-h.; Fu, S.-y.; Lucia, L.A. Novel preparation and characterization of cellulose microparticles functionalized in ionic liquids. Langmuir 2009, 25, 10116–10120. [Google Scholar] [CrossRef]
- Braun, M.; Guentherberg, N.; Lutz, M.; Magin, A.; Siemer, M.; Swaminathan, V.N.; Linner, B.; Ruslim, F.; Ramierz, G.A.F. Process for Producing Cellulose Beads from Solutions of Cellulose in Ionic Liquid. U.S. Patent 2708690A, 30 December 2010. [Google Scholar]
- Pérez-Madrigal, M.M.; Torras, J.; Casanovas, J.; Häring, M.; Alemán, C.; Díaz, D.D. Paradigm shift for preparing versatile m2+-free gels from unmodified sodium alginate. Biomacromolecules 2017, 18, 2967–2979. [Google Scholar] [CrossRef] [PubMed]
- Gurikov, P.; Smirnova, I. Non-conventional methods for gelation of alginate. Gels 2018, 4, 14. [Google Scholar] [CrossRef]
- Groult, S.; Budtova, T. Tuning structure and properties of pectin aerogels. Eur. Polym. J. 2018, 108, 250–261. [Google Scholar] [CrossRef]
- Ratanajiajaroen, P.; Ohshima, M. Preparation of highly porous β-chitin structure through nonsolvent–solvent exchange-induced phase separation and supercritical CO2 drying. J. Supercrit. Fluids 2012, 68, 31–38. [Google Scholar] [CrossRef]
- Tkalec, G.; Kranvogl, R.; Uzunalić, A.P.; Knez, Ž.; Novak, Z. Optimisation of critical parameters during alginate aerogels’ production. J. Non-Cryst. Solids 2016, 443, 112–117. [Google Scholar] [CrossRef]
- Tkalec, G.; Knez, Ž.; Novak, Z. Formation of polysaccharide aerogels in ethanol. Rsc Adv. 2015, 5, 77362–77371. [Google Scholar] [CrossRef]
- Silva, S.S.; Duarte, A.R.C.; Mano, J.F.; Reis, R.L. Design and functionalization of chitin-based microsphere scaffolds. Green Chem. 2013, 15, 3252–3258. [Google Scholar] [CrossRef]
- Oylum, H.; Yilmaz, E.; Yilmaz, O. Preparation of Chitin-g-poly (4-vinylpyridine) Beads. J. Macromol. Sci. Part. A 2013, 50, 221–229. [Google Scholar] [CrossRef]
- Tkalec, G.; Knez, Ž.; Novak, Z. Fast production of high-methoxyl pectin aerogels for enhancing the bioavailability of low-soluble drugs. J. Supercrit. Fluids 2015, 106, 16–22. [Google Scholar] [CrossRef]
- Wan, L.; Heng, P.; Chan, L. Drug encapsulation in alginate microspheres by emulsification. J. Microencapsul. 1992, 9, 309–316. [Google Scholar] [CrossRef] [PubMed]
- Paharia, A.; Yadav, A.K.; Rai, G.; Jain, S.K.; Pancholi, S.S.; Agrawal, G.P. Eudragit-coated pectin microspheres of 5-fluorouracil for colon targeting. Aaps Pharmscitech 2007, 8, E87–E93. [Google Scholar] [CrossRef]
- Vicini, S.; Mauri, M.; Wichert, J.; Castellano, M. Alginate gelling process: Use of bivalent ions rich microspheres. Polym. Eng. Sci. 2017, 57, 531–536. [Google Scholar] [CrossRef]
- Li, J.; He, J.; Huang, Y.; Li, D.; Chen, X. Improving surface and mechanical properties of alginate films by using ethanol as a co-solvent during external gelation. Carbohydr. Polym. 2015, 123, 208–216. [Google Scholar] [CrossRef]
- Donati, I.; Paoletti, S. Material properties of alginates. In Alginates: Biology and Applications; Springer: Berlin, Germany; Heidelberg, Germany, 2009; pp. 1–53. [Google Scholar]
- Draget, K.I.; Skjåk-Bræk, G.; Stokke, B.T. Similarities and differences between alginic acid gels and ionically crosslinked alginate gels. Food Hydrocoll. 2006, 20, 170–175. [Google Scholar] [CrossRef]
- Tsai, C.S. Biomacromolecules: Introduction to Structure, Function and Informatics; John Wiley & Sons: Hoboken, NJ, USA, 2007. [Google Scholar]
- Finch, P. Carbohydrates: Structures, Syntheses and Dynamics; Springer Science & Business Media: Dordrecht, The Netherlands, 2013. [Google Scholar]
- Gavillon, R. Preparation and Characterization of Ultra Porous Cellulosic Materials. Ph.D. Thesis, École Nationale Supérieure des Mines de Paris, Paris, France, 23 March 2007. [Google Scholar]
- Trygg, J.; Fardim, P.; Gericke, M.; Mäkilä, E.; Salonen, J. Physicochemical design of the morphology and ultrastructure of cellulose beads. Carbohydr. Polym. 2013, 93, 291–299. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, S.M.K.; Ganesan, K.; Milow, B.; Ratke, L. The effect of zinc oxide (ZnO) addition on the physical and morphological properties of cellulose aerogel beads. Rsc Adv. 2015, 5, 90193–90201. [Google Scholar] [CrossRef]
- Luo, X.; Zhang, L. Creation of regenerated cellulose microspheres with diameter ranging from micron to millimeter for chromatography applications. J. Chromatogr. A 2010, 1217, 5922–5929. [Google Scholar] [CrossRef]
- Blachechen, L.S.; Fardim, P.; Petri, D.F. Multifunctional cellulose beads and their interaction with gram positive bacteria. Biomacromolecules 2014, 15, 3440–3448. [Google Scholar] [CrossRef] [PubMed]
- Mao, B.; Divoux, T.; Snabre, P. Normal force controlled rheology applied to agar gelation. J. Rheol. 2016, 60, 473–489. [Google Scholar] [CrossRef]
- Holland, S.; Tuck, C.; Foster, T. Fluid Gels: A New Feedstock for High Viscosity Jetting. Food Biophys. 2018, 13, 175–185. [Google Scholar] [CrossRef]
- Alba, K.; Kontogiorgos, V. Seaweed polysaccharides (agar, alginate carrageenan). In Encyclopedia of Food Chemistry; Elsevier: Amsterdam, The Netherlands, 2018; pp. 240–250. [Google Scholar]
- Takemasa, M.; Chiba, A.; Date, M. Gelation mechanism of κ-and ι-carrageenan investigated by correlation between the strain−optical coefficient and the dynamic shear modulus. Macromolecules 2001, 34, 7427–7434. [Google Scholar] [CrossRef]
- Ratnayake, W.S.; Jackson, D.S. Starch gelatinization. Adv. Food Nutr. Res. 2009, 55, 221–268. [Google Scholar] [PubMed]
- Alvarez, M.D.; Fuentes, R.; Canet, W. Effects of pressure, temperature, treatment time, and storage on rheological, textural, and structural properties of heat-induced chickpea gels. Foods 2015, 4, 80–114. [Google Scholar] [CrossRef] [PubMed]
- Calabrese, V.; Muñoz-García, J.C.; Schmitt, J.; da Silva, M.A.; Scott, J.L.; Angulo, J.; Khimyak, Y.Z.; Edler, K.J. Understanding heat driven gelation of anionic cellulose nanofibrils: Combining saturation transfer difference (STD) NMR, small angle X-ray scattering (SAXS) and rheology. J. Colloid Interface Sci. 2019, 535, 205–213. [Google Scholar] [CrossRef] [PubMed]
- Appaw, C.; Gilbert, R.D.; Khan, S.A.; Kadla, J.F. Phase separation and heat-induced gelation characteristics of cellulose acetate in a mixed solvent system. Cellulose 2010, 17, 533–538. [Google Scholar] [CrossRef]
- Fatimi, A.; Tassin, J.-F.; Turczyn, R.; Axelos, M.A.V.; Weiss, P. Gelation studies of a cellulose-based biohydrogel: The influence of pH, temperature and sterilization. Acta Biomater. 2009, 5, 3423–3432. [Google Scholar] [CrossRef]
- Qin, W.; Li, J.; Tu, J.; Yang, H.; Chen, Q.; Liu, H. Fabrication of porous chitosan membranes composed of nanofibers by low temperature thermally induced phase separation, and their adsorption behavior for Cu2. Carbohydr. Polym. 2017, 178, 338–346. [Google Scholar] [CrossRef] [PubMed]
- Mi, F.L.; Tan, Y.C.; Liang, H.C.; Huang, R.N.; Sung, H.W. In vitro evaluation of a chitosan membrane cross-linked with genipin. J. Biomater. Sci. Polym. Ed. 2001, 12, 835–850. [Google Scholar] [CrossRef] [PubMed]
- Mirzaei, B.E.; Ramazani, S.A.A.; Shafiee, M.; Danaei, M. Studies on glutaraldehyde crosslinked chitosan hydrogel properties for drug delivery systems. Int. J. Polym. Mater. Polym. Biomater. 2013, 62, 605–611. [Google Scholar] [CrossRef]
- Dini, E.; Alexandridou, S.; Kiparissides, C. Synthesis and characterization of cross-linked chitosan microspheres for drug delivery applications. J. Microencapsul 2003, 20, 375–385. [Google Scholar] [CrossRef] [PubMed]
- Kulkarni, A.R.; Soppimath, K.S.; Aralaguppi, M.I.; Aminabhavi, T.M.; Rudzinski, W.E. Preparation of cross-linked sodium alginate microparticles using glutaraldehyde in methanol. Drug Dev. Ind. Pharm. 2000, 26, 1121–1124. [Google Scholar] [CrossRef]
- Almeida, P.F.; Almeida, A. Cross-linked alginate-gelatine beads: A new matrix for controlled release of pindolol. J. Control. Release 2004, 97, 431–439. [Google Scholar] [CrossRef]
- Kumar, S.; Haq, I.; Prakash, J.; Raj, A. Improved enzyme properties upon glutaraldehyde cross-linking of alginate entrapped xylanase from Bacillus licheniformis. Int. J. Biol. Macromol. 2017, 98, 24–33. [Google Scholar] [CrossRef]
- Jeon, J.G.; Kim, H.C.; Palem, R.R.; Kim, J.; Kang, T.J. Cross-linking of cellulose nanofiber films with glutaraldehyde for improved mechanical properties. Mater. Lett. 2019, 250, 99–102. [Google Scholar] [CrossRef]
- Buhus, G.; Popa, M.; Desbrieres, J. Hydrogels based on carboxymethylcellulose and gelatin for inclusion and release of chloramphenicol. J. Bioact. Compat. Polym. 2009, 24, 525–545. [Google Scholar] [CrossRef]
- Hongbo, T.; Yanping, L.; Min, S.; Xiguang, W. Preparation and property of crosslinking guar gum. Polym. J. 2012, 44, 211–216. [Google Scholar] [CrossRef]
- Sandolo, C.; Matricardi, P.; Alhaique, F.; Coviello, T. Effect of temperature and cross-linking density on rheology of chemical cross-linked guar gum at the gel point. Food Hydrocoll. 2009, 23, 210–220. [Google Scholar] [CrossRef]
- Usha, R.; Ramasami, T. Structure and conformation of intramolecularly cross-linked collagen. Colloids Surf. B Biointerfaces 2005, 41, 21–24. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Black, L.; Santacana-Laffitte, G.; Patrick Jr, C.W. Preparation and assessment of glutaraldehyde-crosslinked collagen-chitosan hydrogels for adipose tissue engineering. J. Biomed. Mater. Res. Part. A 2007, 81, 59–65. [Google Scholar] [CrossRef] [PubMed]
- George, M.; Abraham, T. pH sensitive alginate-guar gum hydrogel for the controlled delivery of protein drugs. Int. J. Pharm. 2007, 335, 123–129. [Google Scholar] [CrossRef] [PubMed]
- Distantina, S.; Rochmadi, S.; Fahrurrozi, M.; Wiratni, M. Preparation of hydrogel based on glutaraldehyde-crosslinked carrageenan. In Proceedings of the 3rd International Conference on Chemistry and Chemical Engineering IPCBEE, Singapore, 26–28 February 2012. [Google Scholar]
- Distantina, S.; Rochmadi, R.; Fahrurrozi, M.; Wiratni, W. Preparation and characterization of glutaraldehyde-crosslinked kappa carrageenan hydrogel. Eng. J. 2013, 17, 57–66. [Google Scholar] [CrossRef]
- Baki, E.; Denkbas, M.; Odabasi, E.; Kiliçay, N.O. Human serum albumin (HSA) adsorption with chitosan microspheres. J. Appl. Polym. Sci. 2002, 86, 3035–3039. [Google Scholar]
- Nayak, U.Y.; Gopal, S.; Mutalik, S.; Ranjith, A.K.; Reddy, M.S.; Gupta, P.; Udupa, N. Glutaraldehyde cross-linked chitosan microspheres for controlled delivery of zidovudine. J. Microencapsul. 2009, 26, 214–222. [Google Scholar] [CrossRef] [PubMed]
- Rinki, K.; Dutta, P.K.; Hunt, A.J.; Macquarrie, D.J.; Clark, J.H. Chitosan aerogels exhibiting high surface area for biomedical application: Preparation, characterization, and antibacterial study. Int. J. Polym. Mater. 2011, 60, 988–999. [Google Scholar] [CrossRef]
- Thakkar, H.P.; Murthy, R.R. Effect of cross-linking agent on the characteristics of celecoxib loaded chitosan microspheres. Asian, J. Pharm. (Ajp): Free Full Text. Artic. Asian J. Pharm. 2014, 2. [Google Scholar] [CrossRef]
- Gangurde, H.H.; Chavan, N.V.; Mundada, A.S.; Derle, D.V.; Tamizharasi, S. Biodegradable Chitosan-Based Ambroxol Hydrochloride Microspheres: Effect of Cross-Linking Agents. J. Young Pharm. 2011, 3, 9–14. [Google Scholar] [CrossRef]
- Lee, K.Y.; Rowley, J.A.; Eiselt, P.; Moy, E.M.; Bouhadir, K.H.; Mooney, D.J. Controlling mechanical and swelling properties of alginate hydrogels independently by cross-linker type and cross-linking density. Macromolecules 2000, 33, 4291–4294. [Google Scholar] [CrossRef]
- Pedroso-Santana, S.; Fleitas-Salazar, N. Ionotropic gelation method in the synthesis of nanoparticles/microparticles for biomedical purposes. Polym. Int. 2020, 69, 443–447. [Google Scholar] [CrossRef]
- Racoviţă, S.; Vasiliu, S.; Popa, M.; Luca, C. Polysaccharides based on micro-and nanoparticles obtained by ionic gelation and their applications as drug delivery systems. Rev. Roum. De Chim. 2009, 54, 709–718. [Google Scholar]
- Giri, T.K.; Verma, S.; Alexander, A.; Ajazuddin, B.H.; Tripathy, M.; Tripathi, D. Crosslinked biodegradable alginate hydrogel floating beads for stomach site specific controlled delivery of metronidazole. Farmacia 2013, 61, 533–550. [Google Scholar]
- Patil, P.; Chavanke, D.; Wagh, M. A review on ionotropic gelation method: Novel approach for controlled gastroretentive gelispheres. Int J. Pharm Pharm Sci 2012, 4, 27–32. [Google Scholar]
- Laurienzo, P. Marine polysaccharides in pharmaceutical applications: An overview. Mar. Drugs 2010, 8, 2435–2465. [Google Scholar] [CrossRef]
- Patil, J.; Kamalapur, M.; Marapur, S.; Kadam, D. Ionotropic gelation and polyelectrolyte complexation: The novel techniques to design hydrogel particulate sustained, modulated drug delivery system: A review. Dig. J. Nanomater. Biostruct. 2010, 5, 241–248. [Google Scholar]
- Garner, J.; Park, K. Chemically modified natural polysaccharides to form gels. Polysacch. Cham Springer Int. Publ. 2015, 1555–1582. [Google Scholar]
- DeRamos, C.M.; Irwin, A.E.; Nauss, J.L.; Stout, B.E. 13C NMR and molecular modeling studies of alginic acid binding with alkaline earth and lanthanide metal ions. Inorg. Chim. Acta 1997, 256, 69–75. [Google Scholar] [CrossRef]
- Martău, G.A.; Mihai, M.; Vodnar, D.C. The use of chitosan, alginate, and pectin in the biomedical and food sector—biocompatibility, bioadhesiveness, and biodegradability. Polymers 2019, 11, 1837. [Google Scholar] [CrossRef]
- Braccini, I.; Pérez, S. Molecular basis of C(2+)-induced gelation in alginates and pectins: The egg-box model revisited. Biomacromolecules 2001, 2, 1089–1096. [Google Scholar] [CrossRef] [PubMed]
- Shu, X.; Zhu, K. Chitosan/gelatin microspheres prepared by modified emulsification and ionotropic gelation. J. Microencapsul. 2001, 18, 237–245. [Google Scholar] [PubMed]
- Shu, X.; Zhu, K. A novel approach to prepare tripolyphosphate/chitosan complex beads for controlled release drug delivery. Int. J. Pharm. 2000, 201, 51–58. [Google Scholar] [CrossRef]
- Panos, I.; Acosta, N.; Heras, A. New drug delivery systems based on chitosan. Curr. Drug Discov. Technol. 2008, 5, 333–341. [Google Scholar] [CrossRef] [PubMed]
- Smrdel, P.; Bogataj, M.; Mrhar, A. The influence of selected parameters on the size and shape of alginate beads prepared by ionotropic gelation. Sci. Pharm. 2008, 76, 77–90. [Google Scholar] [CrossRef]
- Lee, K.Y.; Mooney, D.J. Alginate: Properties and biomedical applications. Prog. Polym. Sci. 2012, 37, 106–126. [Google Scholar] [CrossRef]
- Draget, K.I. Alginates. In Handbook of Hydrocolloids; Elsevier: Amsterdam, The Netherlands, 2009; pp. 807–828. [Google Scholar]
- Sabra, W.; Zeng, A.-P.; Deckwer, W.-D. Bacterial alginate: Physiology, product quality and process aspects. Appl. Microbiol. Biotechnol. 2001, 56, 315–325. [Google Scholar] [CrossRef]
- d’Ayala, G.G.; Malinconico, M.; Laurienzo, P. Marine derived polysaccharides for biomedical applications: Chemical modification approaches. Molecules 2008, 13, 2069–2106. [Google Scholar] [CrossRef]
- Mørch, Ý.A.; Donati, I.; Strand, B.L.; Skjåk-Bræk, G. Effect of Ca2+, Ba2+, and Sr2+ on Alginate Microbeads. Biomacromolecules 2006, 7, 1471–1480. [Google Scholar] [CrossRef]
- Montanucci, P.; Terenzi, S.; Santi, C.; Pennoni, I.; Bini, V.; Pescara, T.; Basta, G.; Calafiore, R. Insights in behavior of variably formulated alginate-based microcapsules for cell transplantation. Biomed. Res. Int. 2015, 2015, 965804. [Google Scholar] [CrossRef]
- Brus, J.; Urbanova, M.; Czernek, J.; Pavelkova, M.; Kubova, K.; Vyslouzil, J.; Abbrent, S.; Konefal, R.; Horský, J.; Vetchy, D. Structure and dynamics of alginate gels cross-linked by polyvalent ions probed via solid state NMR spectroscopy. Biomacromolecules 2017, 18, 2478–2488. [Google Scholar] [CrossRef]
- BeMiller, J.N.; Whistler, R.L. Industrial Gums: Polysaccharides and Their Derivatives; Academic Press: Cambridge, MA, USA, 2012. [Google Scholar]
- Clark, A.H.; Ross-Murphy, S.B. Structural and mechanical properties of biopolymer gels. Biopolym. Adv. Polym. Sci. 1987, 83, 57–192. [Google Scholar]
- Chambin, O.; Dupuis, G.; Champion, D.; Voilley, A.; Pourcelot, Y. Colon-specific drug delivery: Influence of solution reticulation properties upon pectin beads performance. Int. J. Pharm. 2006, 321, 86–93. [Google Scholar] [CrossRef] [PubMed]
- El-Gibaly, I. Oral delayed-release system based on Zn-pectinate gel (ZPG) microparticles as an alternative carrier to calcium pectinate beads for colonic drug delivery. Int. J. Pharm. 2002, 232, 199–211. [Google Scholar] [CrossRef]
- Braccini, I.; Grasso, R.P.; Pérez, S. Conformational and configurational features of acidic polysaccharides and their interactions with calcium ions: A molecular modeling investigation. Carbohydr. Res. 1999, 317, 119–130. [Google Scholar] [CrossRef]
- Willaert, R.; Nedovic, V. Fundamentals of Cell Immobilisation Biotechnology; Kluwer: Alfen am Rhein, The Netherlands, 2004. [Google Scholar]
- Ahmed, S. Alginates: Applications in the Biomedical and Food Industries; John Wiley & Sons: Hoboken, NJ, USA, 2019. [Google Scholar]
- Brejnholt, S.M. Pectin. Food StabilisersThick. Gelling Agents 2009, 237–265. [Google Scholar]
- Chan, S.Y.; Choo, W.S.; Young, D.J.; Loh, X.J. Pectin as a rheology modifier: Origin, structure, commercial production and rheology. Carbohydr. Polym. 2017, 161, 118–139. [Google Scholar] [CrossRef]
- Rinaudo, M. Gelation of polysaccharides. J. Intell. Mater. Syst. Struct. 1993, 4, 210–215. [Google Scholar] [CrossRef]
- Toft, K.; Grasdalen, H.; Smidsrød, O. Synergistic Gelation of Alginates and Pectins; ACS Publications: Washington, DC, USA, 1986. [Google Scholar]
- Hasnain, M.S.; Nayak, A.K. Natural Polysaccharides in Drug Delivery and Biomedical Applications; Academic Press: Cambridge, MA, USA, 2019. [Google Scholar]
- Sacco, P.; Paoletti, S.; Cok, M.; Asaro, F.; Abrami, M.; Grassi, M.; Donati, I. Insight into the ionotropic gelation of chitosan using tripolyphosphate and pyrophosphate as cross-linkers. Int. J. Biol. Macromol. 2016, 92, 476–483. [Google Scholar] [CrossRef]
- Tharanathan, R.N.; Kittur, F.S. Chitin—The undisputed biomolecule of great potential. Crit. Rev. Food Sci. Nutr. 2003, 43, 61–87. [Google Scholar] [CrossRef]
- Santander-Ortega, M.; Peula-García, J.; Goycoolea, F.; Ortega-Vinuesa, J. Chitosan nanocapsules: Effect of chitosan molecular weight and acetylation degree on electrokinetic behaviour and colloidal stability. Colloids Surf. B Biointerfaces 2011, 82, 571–580. [Google Scholar] [CrossRef] [PubMed]
- Bodmeier, R.; Oh, K.-H.; Pramar, Y. Preparation and evaluation of drug-containing chitosan beads. Drug Dev. Ind. Pharm. 1989, 15, 1475–1494. [Google Scholar] [CrossRef]
- Kim, S.-K. Chitin and Chitosan Derivatives: Advances in Drug Discovery and Developments; CRC Press: Boca Raton, FL, USA, 2013. [Google Scholar]
- Sreekumar, S.; Goycoolea, F.M.; Moerschbacher, B.M.; Rivera-Rodriguez, G.R. Parameters influencing the size of chitosan-TPP nano- and microparticles. Sci. Rep. 2018, 8, 4695. [Google Scholar] [CrossRef] [PubMed]
- Ko, J.; Park, H.J.; Hwang, S.J.; Park, J.; Lee, J. Preparation and characterization of chitosan microparticles intended for controlled drug delivery. Int. J. Pharm. 2002, 249, 165–174. [Google Scholar] [CrossRef]
- Barakat, N.S.; Almurshedi, A.S. Preparation and characterization of chitosan microparticles for oral sustained delivery of gliclazide: In vitro/in vivo evaluation. Drug Dev. Res. 2011, 72, 235–246. [Google Scholar] [CrossRef]
- Hassani, S.; Laouini, A.; Fessi, H.; Charcosset, C. Preparation of chitosan-TPP nanoparticles using microengineered membranes-Effect of parameters and encapsulation of tacrine. Colloids Surf. A Physicochem. Eng. Asp. 2015, 482, 34–43. [Google Scholar] [CrossRef]
- Sacco, P.; Furlani, F.; De Marzo, G.; Marsich, E.; Paoletti, S.; Donati, I. Concepts for developing physical gels of chitosan and of chitosan derivatives. Gels 2018, 4, 67. [Google Scholar] [CrossRef]
- Huang, Y.; Lapitsky, Y. Monovalent salt enhances colloidal stability during the formation of chitosan/tripolyphosphate microgels. Langmuir 2011, 27, 10392–10399. [Google Scholar] [CrossRef]
- Periayah, M.H.; Halim, A.S.; Saad, A.Z.M. Chitosan: A promising marine polysaccharide for biomedical research. Pharm. Rev. 2016, 10, 39–42. [Google Scholar] [CrossRef]
- Chan, L.W.; Lee, H.Y.; Heng, P.W. Mechanisms of external and internal gelation and their impact on the functions of alginate as a coat and delivery system. Carbohydr. Polym. 2006, 63, 176–187. [Google Scholar] [CrossRef]
- Pavelková, M.; Kubová, K.; Vysloužil, J.; Kejdušová, M.; Vetchý, D.; Celer, V.; Molinková, D.; Lobová, D.; Pechová, A.; Vysloužil, J.; et al. Biological effects of drug-free alginate beads cross-linked by copper ions prepared using external ionotropic gelation. Aaps Pharmscitech 2017, 18, 1343–1354. [Google Scholar]
- Zhang, H.; Tumarkin, E.; Peerani, R.; Nie, Z.; Sullan, R.M.; Walker, G.C.; Kumacheva, E. Microfluidic production of biopolymer microcapsules with controlled morphology. J. Am. Chem. Soc. 2006, 128, 12205–12210. [Google Scholar] [CrossRef] [PubMed]
- Mikkelsen, A.; Elgsaeter, A. Density distribution of calcium-induced alginate gels. A numerical study. Biopolym.: Orig. Res. Biomol. 1995, 36, 17–41. [Google Scholar] [CrossRef]
- Pawar, S.N.; Edgar, K.J. Alginate derivatization: A review of chemistry, properties and applications. Biomaterials 2012, 33, 3279–3305. [Google Scholar] [CrossRef]
- Qun, L.; Ming, X.W.; Ting, Y.W.; Dong, L.X.; Yuan, Y.R.; Yun, L.J.; Jun, M.X. Studies on the membrane strength of alginate/chitosan microcapsule prepared by emulsification/internal gelation method. Chem. Res. Chin. Univ. 2002, 7. [Google Scholar]
- Ahmed, M.M.; El-Rasoul, S.A.; Auda, S.H.; Ibrahim, M.A. Emulsification/internal gelation as a method for preparation of diclofenac sodium–sodium alginate microparticles. Saudi Pharm. J. 2013, 21, 61–69. [Google Scholar] [CrossRef]
- Choi, B.; Park, H.J.; Hwang, S.; Park, J. Preparation of alginate beads for floating drug delivery system: Effects of CO2 gas-forming agents. Int. J. Pharm. 2002, 239, 81–91. [Google Scholar] [CrossRef]
- Vandenberg, G.W.; De La Noüe, J. Evaluation of protein release from chitosan-alginate microcapsules produced using external or internal gelation. J. Microencapsul. 2001, 18, 433–441. [Google Scholar] [CrossRef]
- Martins, E.; Renard, D.; Davy, J.; Marquis, M.; Poncelet, D. Oil core microcapsules by inverse gelation technique. J. Microencapsul 2015, 32, 86–95. [Google Scholar] [CrossRef]
- Abang, S.; Chan, E.-S.; Poncelet, D. Effects of process variables on the encapsulation of oil in ca-alginate capsules using an inverse gelation technique. J. Microencapsul. 2012, 29, 417–428. [Google Scholar] [CrossRef]
- Martins, E.; Poncelet, D.; Rodrigues, R.C.; Renard, D. Oil encapsulation in core-shell alginate capsules by inverse gelation II: Comparison between dripping techniques using W/O or O/W emulsions. J. Microencapsul. 2017, 34, 522–534. [Google Scholar] [CrossRef] [PubMed]
- Kabir, S.M.F.; Sikdar, P.P.; Haque, B.; Bhuiyan, M.A.R.; Ali, A.; Islam, M.N. Cellulose-based hydrogel materials: Chemistry, properties and their prospective applications. Prog. Biomater. 2018, 7, 153–174. [Google Scholar] [CrossRef] [PubMed]
- Chirani, N.; Gritsch, L.; Motta, F.L.; Fare, S. History and applications of hydrogels. J. Biomed. Sci. 2015, 4. [Google Scholar]
- Nicodemus, G.D.; Bryant, S.J. Cell encapsulation in biodegradable hydrogels for tissue engineering applications. Tissue Eng Part. B Rev. 2008, 14, 149–165. [Google Scholar] [CrossRef]
- Jen, A.C.; Wake, M.C.; Mikos, A.G. Hydrogels for cell immobilization. Biotechnol. Bioeng. 1996, 50, 357–364. [Google Scholar] [CrossRef]
- Bidarra, S.J.; Barrias, C.C.; Granja, P.L. Injectable alginate hydrogels for cell delivery in tissue engineering. Acta Biomater. 2014, 10, 1646–1662. [Google Scholar] [CrossRef]
- Racheva, M.; Julich-Gruner, K.K.; Nöchel, U.; Neffe, A.T.; Wischke, C.; Lendlein, A. Influence of drying procedures on network formation and properties of hydrogels from functionalized gelatin. Macromolecular Symposia 2018, 334, 24–32. [Google Scholar] [CrossRef]
- Hua, S.; Ma, H.; Li, X.; Yang, H.; Wang, A. pH-sensitive sodium alginate/poly(vinyl alcohol) hydrogel beads prepared by combined Ca2+ crosslinking and freeze-thawing cycles for controlled release of diclofenac sodium. Int. J. Biol. Macromol. 2010, 46, 517–523. [Google Scholar] [CrossRef]
- Rodríguez-Dorado, R.; López-Iglesias, C.; García-González, C.A.; Auriemma, G.; Aquino, R.P.; Del Gaudio, P. Design of aerogels, cryogels and xerogels of alginate: Effect of molecular weight, gelation conditions and drying method on particles’ micromeritics. Molecules 2019, 24, 1049. [Google Scholar] [CrossRef]
- Auriemma, G.; Cerciello, A.; Aquino, R.P. NSAIDS: Design and development of innovative oral delivery systems. Nonsteroidal Anti-Inflamm. Drugs 2017, 9, 51000. [Google Scholar]
- Al-Muhtaseb, S.A.; Ritter, J.A. Preparation and properties of resorcinol-formaldehyde organic and carbon gels. Adv. Mater. 2003, 15, 101–114. [Google Scholar] [CrossRef]
- Job, N.; Pirard, R.; Marien, J.; Pirard, J.-P. Porous carbon xerogels with texture tailored by pH control during sol–gel process. Carbon 2004, 42, 619–628. [Google Scholar] [CrossRef]
- Czakkel, O.; Marthi, K.; Geissler, E.; László, K. Influence of drying on the morphology of resorcinol–formaldehyde-based carbon gels. Microporous Mesoporous Mater. 2005, 86, 124–133. [Google Scholar] [CrossRef]
- Zubizarreta, L.; Arenillas, A.; Menéndez, J.; Pis, J.J.; Pirard, J.-P.; Job, N. Microwave drying as an effective method to obtain porous carbon xerogels. J. Non-Cryst. Solids 2008, 354, 4024–4026. [Google Scholar] [CrossRef]
- Zubizarreta, L.; Arenillas, A.; Domínguez, A.; Menéndez, J.; Pis, J. Development of microporous carbon xerogels by controlling synthesis conditions. J. Non-Cryst. Solids 2008, 354, 817–825. [Google Scholar] [CrossRef]
- Ganesan, K.; Dennstedt, A.; Barowski, A.; Ratke, L. Design of aerogels, cryogels and xerogels of cellulose with hierarchical porous structures. Mater. Des. 2016, 92, 345–355. [Google Scholar] [CrossRef]
- Léonard, A.; Job, N.; Blacher, S.; Pirard, J.-P.; Crine, M.; Jomaa, W. Suitability of convective air drying for the production of porous resorcinol-formaldehyde and carbon xerogels. Carbon 2005, 43, 1808–1811. [Google Scholar] [CrossRef]
- Job, N.; Panariello, F.; Marien, J.; Crine, M.; Pirard, J.-P.; Léonard, A. Synthesis optimization of organic xerogels produced from convective air-drying of resorcinol-formaldehyde gels. J. Non-Cryst. Solids 2006, 352, 24–34. [Google Scholar] [CrossRef]
- Menéndez, J.; Juárez-Pérez, E.; Ruisánchez, E.; Calvo, E.; Arenillas, A. A microwave-based method for the synthesis of carbon xerogel spheres. Carbon 2012, 50, 3555–3560. [Google Scholar] [CrossRef]
- Pour, G.; Beauger, C.; Rigacci, A.; Budtova, T. Xerocellulose: Lightweight, porous and hydrophobic cellulose prepared via ambient drying. J. Mater. Sci. 2015, 50, 4526–4535. [Google Scholar] [CrossRef]
- Auriemma, G.; Del Gaudio, P.; Barba, A.A.; d’Amore, M.; Aquino, R.P. A combined technique based on prilling and microwave assisted treatments for the production of ketoprofen controlled release dosage forms. Int. J. Pharm. 2011, 415, 196–205. [Google Scholar] [CrossRef]
- Aquino, R.P.; Auriemma, G.; d’Amore, M.; D’Ursi, A.M.; Mencherini, T.; Del Gaudio, P. Piroxicam loaded alginate beads obtained by prilling/microwave tandem technique: Morphology and drug release. Carbohydr. Polym. 2012, 89, 740–748. [Google Scholar] [CrossRef] [PubMed]
- Nussinovitch, A. Polymer Macro- and Micro-Gel Beads: Fundamentals and Applications; Springer Science & Business Media: Berlin, Germany, 2010. [Google Scholar]
- Stefanescu, D.M. Science and Engineering of Casting Solidification; Springer: Midtown Manhattan, NY, USA, 2015. [Google Scholar]
- Betz, M.; García-González, C.; Subrahmanyam, R.; Smirnova, I.; Kulozik, U. Preparation of novel whey protein-based aerogels as drug carriers for life science applications. J. Supercrit. Fluids 2012, 72, 111–119. [Google Scholar] [CrossRef]
- Jiménez-Saelices, C.; Seantier, B.; Cathala, B.; Grohens, Y. Spray freeze-dried nanofibrillated cellulose aerogels with thermal superinsulating properties. Carbohydr. Polym. 2017, 157, 105–113. [Google Scholar] [CrossRef] [PubMed]
- Tamon, H.; Ishizaka, H.; Yamamoto, T.; Suzuki, T. Influence of freeze-drying conditions on the mesoporosity of organic gels as carbon precursors. Carbon 2000, 38, 1099–1105. [Google Scholar] [CrossRef]
- Babić, B.; Kaluđerović, B.; Vračar, L.; Krstajić, N. Characterization of carbon cryogel synthesized by sol-gel polycondensation and freeze-drying. Carbon 2004, 42, 2617–2624. [Google Scholar] [CrossRef]
- Tonanon, N.; Siyasukh, A.; Tanthapanichakoon, W.; Nishihara, H.; Mukai, S.; Tamon, H. Improvement of mesoporosity of carbon cryogels by ultrasonic irradiation. Carbon 2005, 43, 525–531. [Google Scholar] [CrossRef]
- Pääkkö, M.; Vapaavuori, J.; Silvennoinen, R.; Kosonen, H.; Ankerfors, M.; Lindström, T.; Berglund, L.A.; Ikkala, O. Long and entangled native cellulose I nanofibers allow flexible aerogels and hierarchically porous templates for functionalities. Soft Matter 2008, 4, 2492–2499. [Google Scholar] [CrossRef]
- García-González, C.; Alnaief, M.; Smirnova, I. Polysaccharide-based aerogels—Promising biodegradable carriers for drug delivery systems. Carbohydr. Polym. 2011, 86, 1425–1438. [Google Scholar] [CrossRef]
- Maleki, H.; Durães, L.; García-González, C.A.; del Gaudio, P.; Portugal, A.; Mahmoudi, M. Synthesis and biomedical applications of aerogels: Possibilities and challenges. Adv. Colloid Interface Sci. 2016, 236, 1–27. [Google Scholar] [CrossRef]
- Smirnova, I.; Gurikov, P. Aerogels in chemical engineering: Strategies toward tailor-made aerogels. Annu. Rev. Chem. Biomol. Eng. 2017, 8, 307–334. [Google Scholar] [CrossRef] [PubMed]
- Nita, L.E.; Ghilan, A.; Rusu, A.G.; Neamtu, I.; Chiriac, A.P. New trends in bio-based aerogels. Pharmaceutics 2020, 12, 449. [Google Scholar] [CrossRef]
- Sanli, D.; Bozbag, S.; Erkey, C. Synthesis of nanostructured materials using supercritical CO2: Part, I. Physical transformations. J. Mater. Sci. 2012, 47, 2995–3025. [Google Scholar] [CrossRef]
- Diamond, L.W.; Akinfiev, N.N. Solubility of CO2 in water from 1.5 to 100 C and from 0.1 to 100 MPa: Evaluation of literature data and thermodynamic modelling. Fluid Phase Equilibria 2003, 208, 265–290. [Google Scholar] [CrossRef]
- Pasquali, I.; Bettini, R. Are pharmaceutics really going supercritical? Int. J. Pharm. 2008, 364, 176–187. [Google Scholar] [CrossRef] [PubMed]
- Liu, N.; Zhang, S.; Fu, R.; Dresselhaus, M.S.; Dresselhaus, G. Carbon aerogel spheres prepared via alcohol supercritical drying. Carbon 2006, 44, 2430–2436. [Google Scholar] [CrossRef]
- Russo, P.; Zacco, R.; Rekkas, D.M.; Politis, S.; Garofalo, E.; Del Gaudio, P.; Aquino, R.P. Application of experimental design for the development of soft-capsules through a prilling, inverse gelation process. J. Drug Deliv. Sci. Technol. 2019, 49, 577–585. [Google Scholar] [CrossRef]
- Rodríguez-Dorado, R.; Landín, M.; Altai, A.; Russo, P.; Aquino, R.P.; Del Gaudio, P. A novel method for the production of core-shell microparticles by inverse gelation optimized with artificial intelligent tools. Int. J. Pharm. 2018, 538, 97–104. [Google Scholar] [CrossRef]
- Cerciello, A.; Del Gaudio, P.; Granata, V.; Sala, M.; Aquino, R.P.; Russo, P. Synergistic effect of divalent cations in improving technological properties of cross-linked alginate beads. Int. J. Biol. Macromol. 2017, 101, 100–106. [Google Scholar] [CrossRef]
- Cerciello, A.; Auriemma, G.; Morello, S.; Pinto, A.; Del Gaudio, P.; Russo, P.; Aquino, R.P. Design and in vivo anti-inflammatory effect of ketoprofen delayed delivery systems. J. Pharm. Sci. 2015, 104, 3451–3458. [Google Scholar] [CrossRef]
- Cerciello, A.; Auriemma, G.; Morello, S.; Aquino, R.P.; Del Gaudio, P.; Russo, P. Prednisolone delivery platforms: Capsules and beads combination for a right timing therapy. PLoS ONE 2016, 11, e0160266. [Google Scholar] [CrossRef]
- Cerciello, A.; Auriemma, G.; Del Gaudio, P.; Cantarini, M.; Aquino, R.P. Natural polysaccharides platforms for oral controlled release of ketoprofen lysine salt. Drug Dev. Ind. Pharm. 2016, 42, 2063–2069. [Google Scholar] [CrossRef] [PubMed]
- Cerciello, A.; Auriemma, G.; Del Gaudio, P.; Sansone, F.; Aquino, R.P.; Russo, P. A novel core-shell chronotherapeutic system for the oral administration of ketoprofen. J. Drug Deliv. Sci. Technol. 2016, 32, 126–131. [Google Scholar] [CrossRef]
- Auriemma, G.; Cerciello, A.; Sansone, F.; Pinto, A.; Morello, S.; Aquino, R.P. Polysaccharides based gastroretentive system to sustain piroxicam release: Development and in vivo prolonged anti-inflammatory effect. Int. J. Biol. Macromol. 2018, 120, 2303–2312. [Google Scholar] [CrossRef]
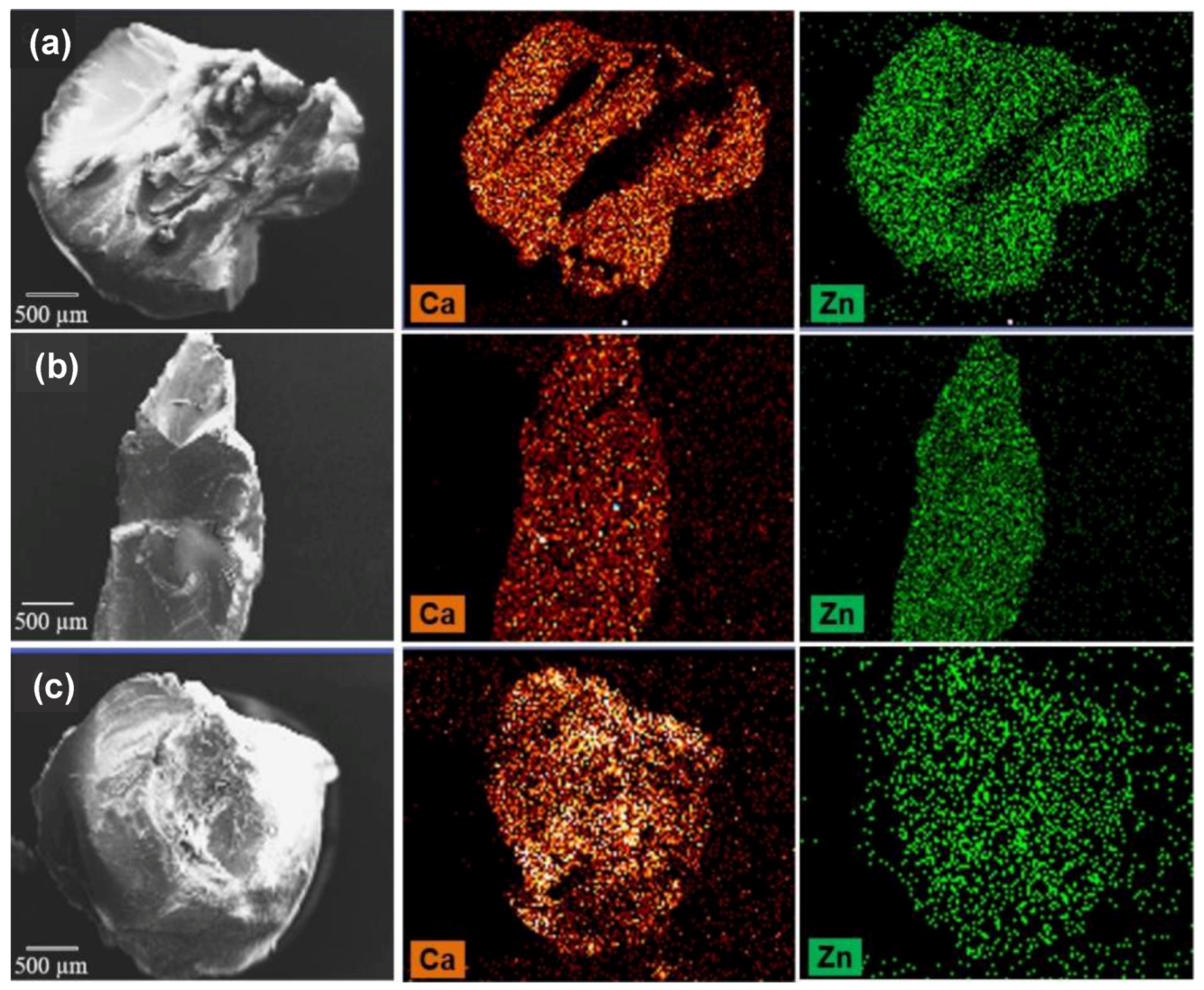
- Russo, P.; Morello, S.; Pinto, A.; Del Gaudio, P.; Auriemma, G.; Aquino, R.P. Zinc and calcium cations combination in the production of floating alginate beads as prednisolone delivery systems. Molecules 2020, 25, 1140. [Google Scholar] [CrossRef] [PubMed]
- Del Gaudio, P.; Auriemma, G.; Mencherini, T.; Della Porta, G.; Reverchon, E.; Aquino, R.P. Design of alginate-based aerogel for nonsteroidal anti-inflammatory drugs controlled delivery systems using prilling and supercritical-assisted drying. J. Pharm. Sci. 2013, 102, 185–194. [Google Scholar] [CrossRef] [PubMed]
- Della Porta, G.; Del Gaudio, P.; De Cicco, F.; Aquino, R.P.; Reverchon, E. Supercritical drying of alginate beads for the development of aerogel biomaterials: Optimization of process parameters and exchange solvents. Ind. Eng. Chem. Res. 2013, 52, 12003–12009. [Google Scholar] [CrossRef]
- Chan, L.W.; Jin, Y.; Heng, P.W.S. Cross-linking mechanisms of calcium and zinc in production of alginate microspheres. Int. J. Pharm. 2002, 242, 255–258. [Google Scholar] [CrossRef]
- Rasel, M.A.T.; Hasan, M. Formulation and evaluation of floating alginate beads of diclofenac sodium. Dhaka Univ. J. Pharm. Sci. 2012, 11, 29–35. [Google Scholar] [CrossRef]
- Celli, G.B.; Ghanem, A.; Brooks, M.S. Development and evaluation of floating alginate microspheres for oral delivery of anthocyanins—A preliminary investigation. Food Sci. Nutr. 2016, 5, 713–721. [Google Scholar] [CrossRef]
- Timilsena, Y.P.; Akanbi, T.O.; Khalid, N.; Adhikari, B.; Barrow, C.J. Complex coacervation: Principles, mechanisms and applications in microencapsulation. Int. J. Biol. Macromol. 2019, 121, 1276–1286. [Google Scholar] [CrossRef]
- Yadav, S.K.; Khan, G.; Bonde, G.V.; Bansal, M.; Mishra, B. Design, optimization and characterizations of chitosan fortified calcium alginate microspheres for the controlled delivery of dual drugs. Artif. CellsNanomed. Biotechnol. 2018, 46, 1180–1193. [Google Scholar] [CrossRef] [PubMed]
- Butstraen, C.; Salaün, F. Preparation of microcapsules by complex coacervation of gum Arabic and chitosan. Carbohydr. Polym. 2014, 99, 608–616. [Google Scholar] [CrossRef] [PubMed]
- Tiyaboonchai, W.; Ritthidej, G.C. Development of indomethacin sustained release microcapsules using chitosan-carboxymethyl-cellulose complex coacervation. Development 2003, 25, 246. [Google Scholar]
- Lisuzzo, L.; Cavallaro, G.; Parisi, F.; Milioto, S.; Fakhrullin, R.; Lazzara, G. Core/shell gel beads with embedded halloysite nanotubes for controlled drug release. Coatings 2019, 9, 70. [Google Scholar] [CrossRef]
- Lucinda-Silva, R.M.; Salgado, H.R.N.; Evangelista, R.C. Alginate-chitosan systems: In vitro controlled release of triamcinolone and in vivo gastrointestinal transit. Carbohydr. Polym. 2010, 81, 260–268. [Google Scholar] [CrossRef]
- Ren, Z.; Zhang, X.; Guo, Y.; Han, K.; Huo, N. Preparation and in vitro delivery performance of chitosan–alginate microcapsule for IgG. Food Agric. Immunol. 2017, 28, 1–13. [Google Scholar] [CrossRef]
- Assifaoui, A.; Loupiac, C.; Chambin, O.; Cayot, P. Structure of calcium and zinc pectinate films investigated by FTIR spectroscopy. Carbohydr. Res. 2010, 345, 929–933. [Google Scholar] [CrossRef]
- Das, S.; Ng, K.-Y.; Ho, P.C. Formulation and optimization of zinc-pectinate beads for the controlled delivery of resveratrol. Aaps Pharmscitech 2010, 11, 729–742. [Google Scholar] [CrossRef]
- Mallepally, R.R.; Bernard, I.; Marin, M.A.; Ward, K.R.; McHugh, M.A. Superabsorbent alginate aerogels. J. Supercrit. Fluids 2013, 79, 202–208. [Google Scholar] [CrossRef]
- Gurikov, P.; Smirnova, I. Amorphization of drugs by adsorptive precipitation from supercritical solutions: A review. J. Supercrit. Fluids 2018, 132, 105–125. [Google Scholar] [CrossRef]
- López-Iglesias, C.; Barros, J.; Ardao, I.; Monteiro, F.J.; Alvarez-Lorenzo, C.; Gómez-Amoza, J.L.; García-González, C.A. Vancomycin-loaded chitosan aerogel particles for chronic wound applications. Carbohydr. Polym. 2019, 204, 223–231. [Google Scholar] [CrossRef] [PubMed]
- Veronovski, A.; Tkalec, G.; Knez, Ž.; Novak, Z. Characterisation of biodegradable pectin aerogels and their potential use as drug carriers. Carbohydr. Polym. 2014, 113, 272–278. [Google Scholar] [CrossRef] [PubMed]
- Halamoda-Kenzaoui, B.; Baconnier, S.; Bastogne, T.; Bazile, D.; Boisseau, P.; Borchard, G.; Borgos, S.E.; Calzolai, L.; Cederbrant, K.; Di Felice, G.; et al. Bridging communities in the field of nanomedicine. Regul. Toxicol. Pharmacol. 2019, 106, 187–196. [Google Scholar] [CrossRef]
- Dalwadi, C.; Patel, G. Implementation of “Quality by Design (QbD)” Approach for the Development of 5-Fluorouracil Loaded Thermosensitive Hydrogel. Curr. Drug Deliv. 2016, 13, 512–527. [Google Scholar] [CrossRef]
- Schmutz, M.; Borges, O.; Jesus, S.; Borchard, G.; Perale, G.; Zinn, M.; Sips, Ä.A.; Soeteman-Hernandez, L.G.; Wick, P.; Som, C. A Methodological Safe-by-Design Approach for the Development of Nanomedicines. Front. Bioeng. Biotechnol. 2020, 8, 258. [Google Scholar] [CrossRef]
- van de Poel, I.; Robaey, Z. Safe-by-Design: From Safety to Responsibility. NanoEthics 2017, 11, 297–306. [Google Scholar] [CrossRef]
- Reason, J. Safety paradoxes and safety culture. Inj. Control. Saf. Promot. 2000, 7, 3–14. [Google Scholar] [CrossRef]
- Maresova, P.; Hajek, L.; Krejcar, O.; Storek, M.; Kuca, K. New Regulations on Medical Devices in Europe: Are They an Opportunity for Growth? Adm. Sci. 2020, 10, 16. [Google Scholar] [CrossRef]
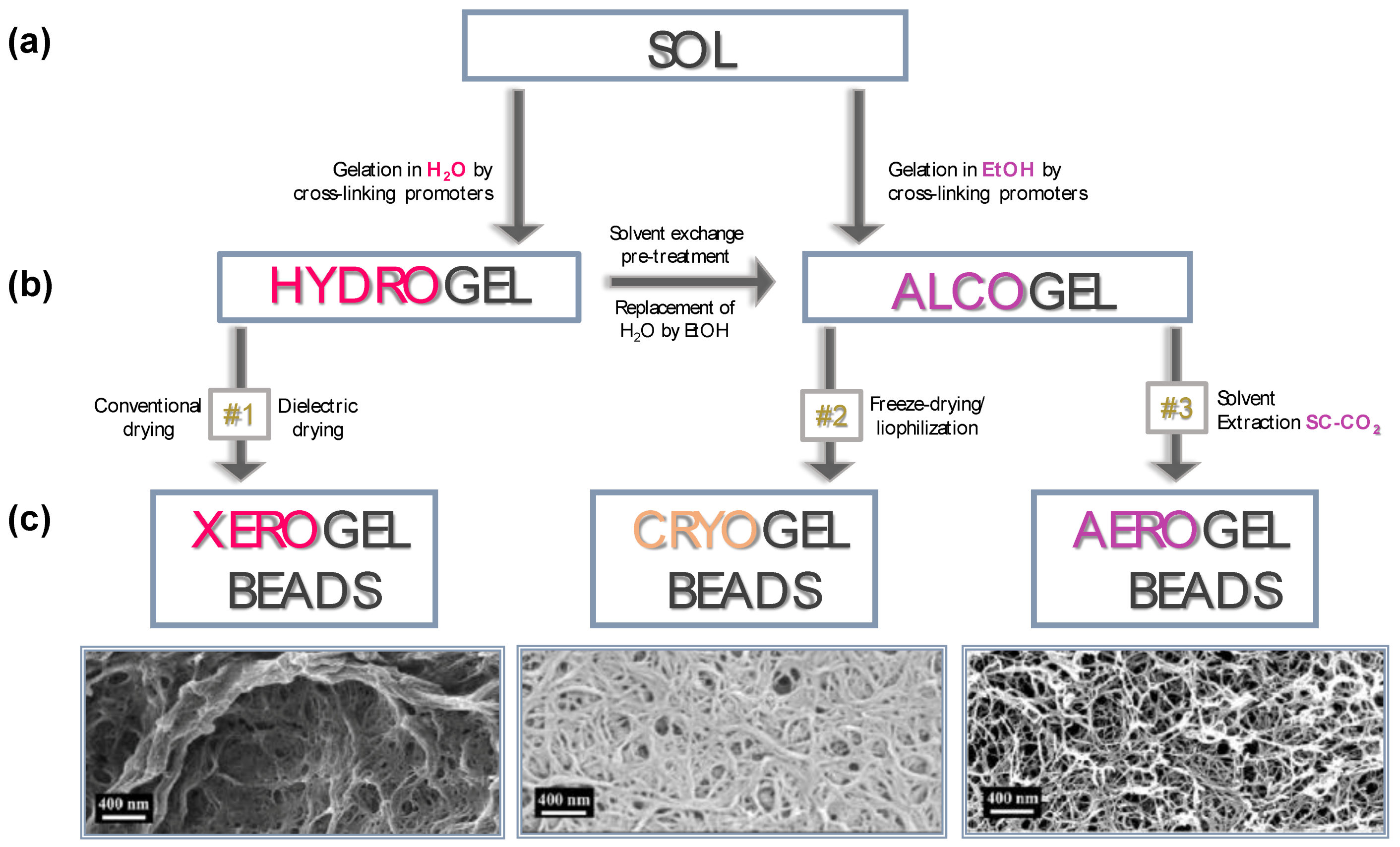

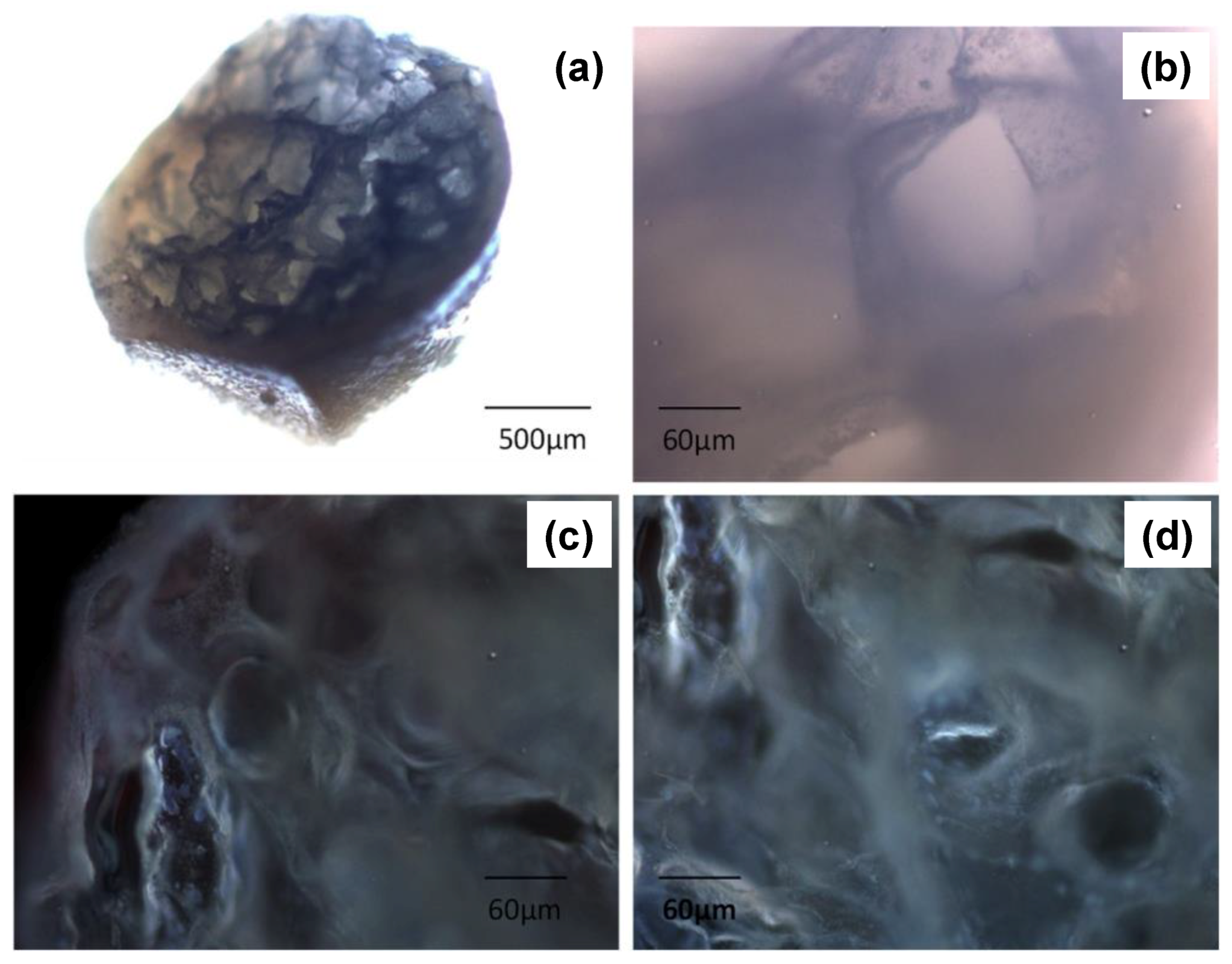
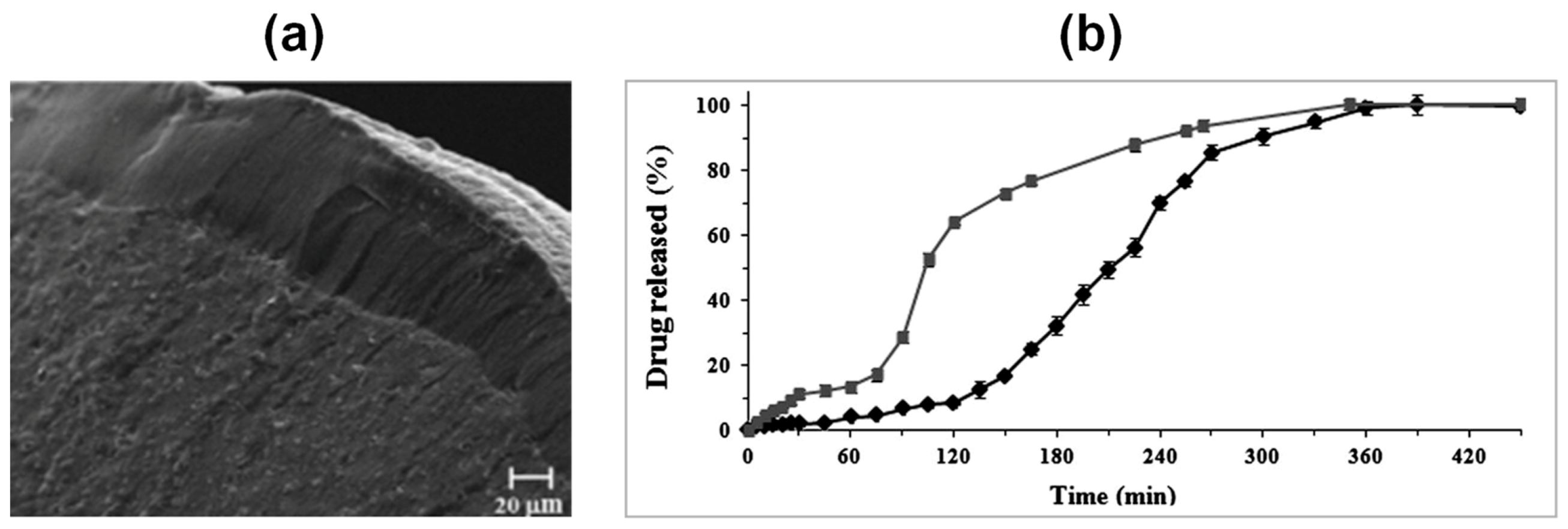
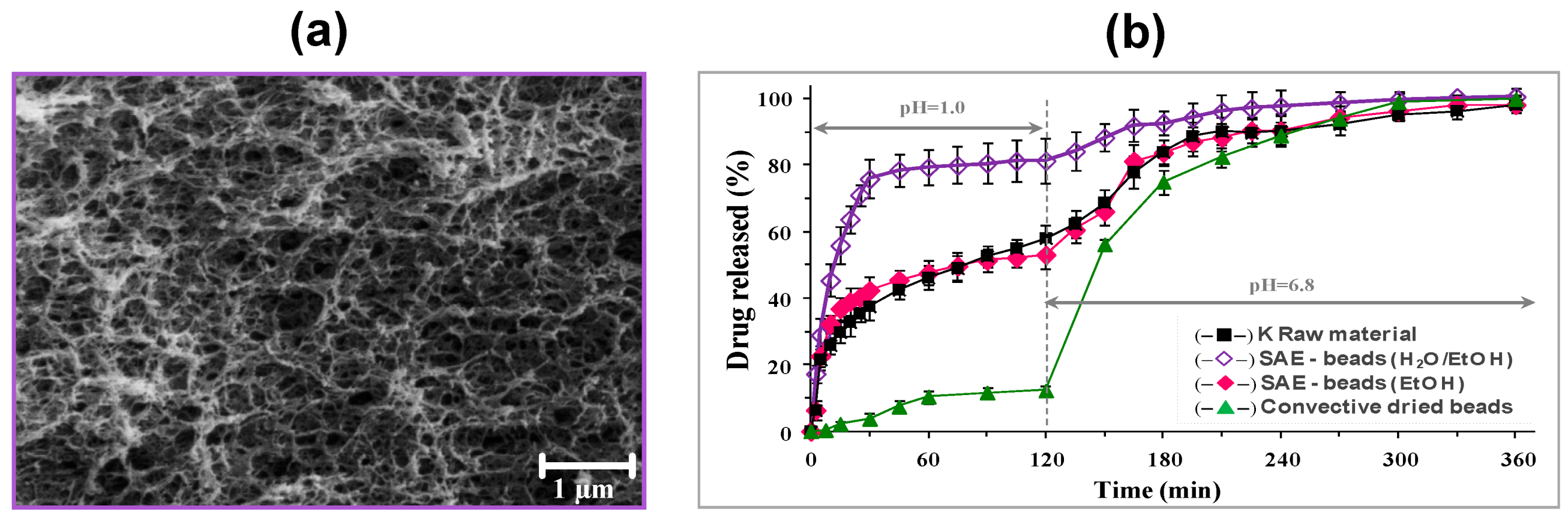
| Drying Method | Particle Inner Structure | Advantages | Disadvantages |
|---|---|---|---|
| Conventional Dielectric Drying | XEROGEL |
|
|
| Supercritical-Assisted Drying | AEROGEL |
|
|
| Freeze-Drying | CRYOGEL |
|
|
| Polysaccharide | Prilling Configuration | Ionic Cross-Linking Conditions | Drying Method | Type of Particle/ Inner Structure | Pharmaceutical Application | References |
|---|---|---|---|---|---|---|
| Alginate | Basic Apparatus | Inverse gelation Ca2+ | None | Soft alginate capsules | Topical administration | [199] |
| Alginate | Coaxial system | Inverse gelation Ca2+ | None | Hydrated core-shell beads loaded with hydrophobic substances | Microencapsulation of hydrophobic compounds into a hydrophilic matrix | [200] |
| Alginate | Basic Apparatus | External gelation Ca2+, Zn2+, Ca2+ plus Zn2+ | Conventional | Only core Xerogels | Delayed DDS for oral administration | [23,29,201,202,203] |
| Alginate | Basic Apparatus | External gelation Ca2+ | Dielectric | Only core Xerogels | Controlled DDS for oral administration | [181,182] |
| Pectin | Basic Apparatus | External gelation Zn2+ | Conventional | Only core Xerogels | Delayed DDS for oral administration | [204] |
| Pectin | Basic Apparatus plus enteric coating ES100 | External gelation Zn2+ | Conventional | Core/shell beads Xerogels | Colon targeted DDS for oral administration | [32,205] |
| Pectin and Alginate | Coaxial system | External gelation Zn2+ | Conventional | Core/shell beads Xerogels | Colon targeted DDS for oral administration | [31,43] |
| Alginate, Pectin and HPMC | Basic Apparatus | External gelation: Zn2+ | Conventional | Floating Hollow Beads | Floating and sustained release DDS for oral administration | [206] |
| Alginate | Basic Apparatus | (a) External gelation Zn2+ (b) Internal gelation Ca2+ | Conventional | Floating Hollow Beads | Floating and sustained release DDS for oral administration | [207] |
| Alginate | Basic Apparatus | External gelation Ca2+ | Supercritical-CO2 | Only core Aerogels | Immediate release DDS for oral administration | [208,209] |
| Alginate and Pectin | Coaxial system | External gelation Ca2+ | Supercritical-CO2 | Core/shell Aerogels | Topical application (Wound Healing) | [44] |
| Alginate | Basic Apparatus | External gelation Ca2+ |
|
| Controlled DDS for oral administration or topical application | [169] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Auriemma, G.; Russo, P.; Del Gaudio, P.; García-González, C.A.; Landín, M.; Aquino, R.P. Technologies and Formulation Design of Polysaccharide-Based Hydrogels for Drug Delivery. Molecules 2020, 25, 3156. https://doi.org/10.3390/molecules25143156
Auriemma G, Russo P, Del Gaudio P, García-González CA, Landín M, Aquino RP. Technologies and Formulation Design of Polysaccharide-Based Hydrogels for Drug Delivery. Molecules. 2020; 25(14):3156. https://doi.org/10.3390/molecules25143156
Chicago/Turabian StyleAuriemma, Giulia, Paola Russo, Pasquale Del Gaudio, Carlos A. García-González, Mariana Landín, and Rita Patrizia Aquino. 2020. "Technologies and Formulation Design of Polysaccharide-Based Hydrogels for Drug Delivery" Molecules 25, no. 14: 3156. https://doi.org/10.3390/molecules25143156
APA StyleAuriemma, G., Russo, P., Del Gaudio, P., García-González, C. A., Landín, M., & Aquino, R. P. (2020). Technologies and Formulation Design of Polysaccharide-Based Hydrogels for Drug Delivery. Molecules, 25(14), 3156. https://doi.org/10.3390/molecules25143156










