Examining the Effects of (α4)3(β2)2 Nicotinic Acetylcholine Receptor-Selective Positive Allosteric Modulator on Acute Thermal Nociception in Rats
Abstract
1. Introduction
2. Results
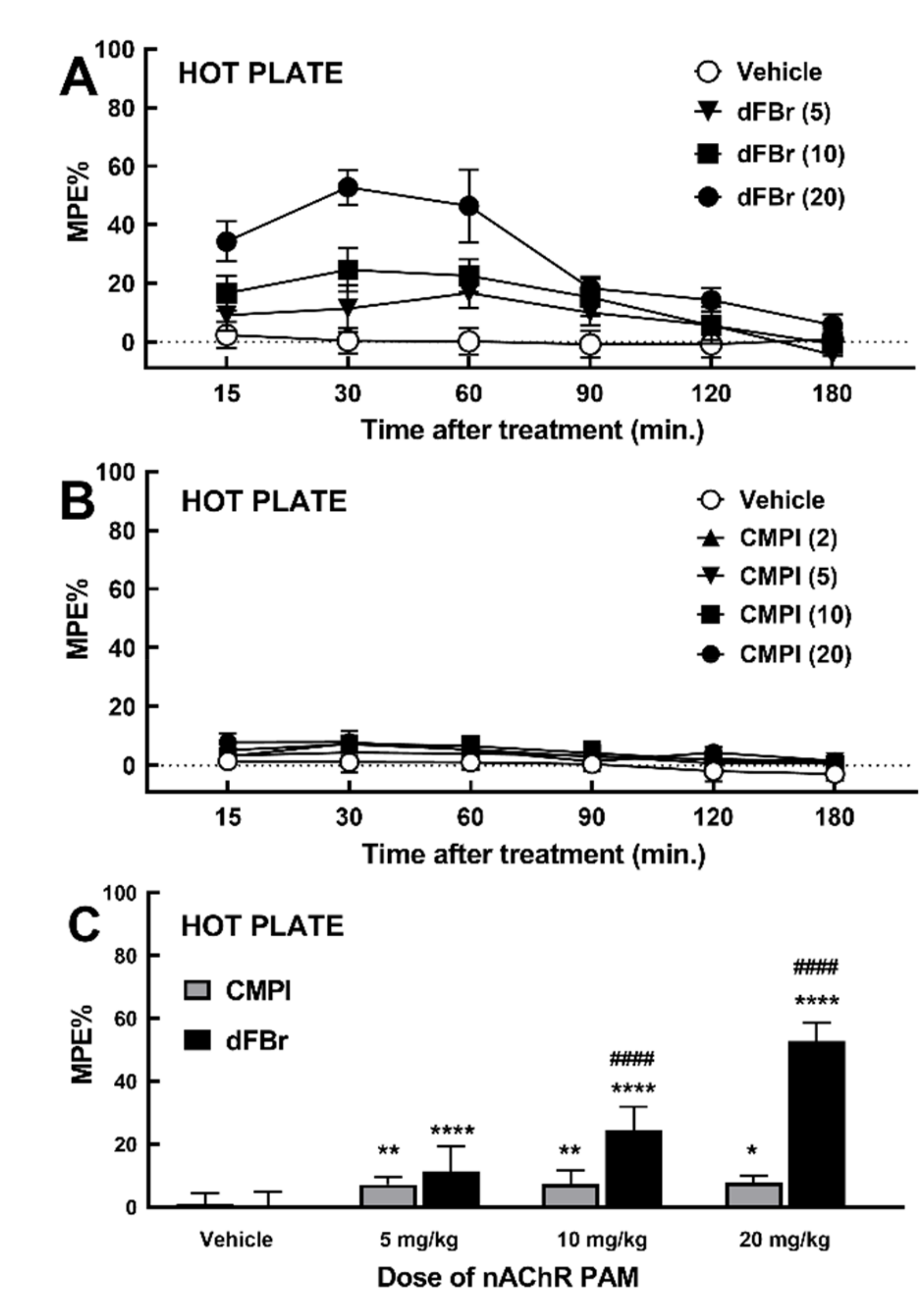
2.1. Effect of dFBr and CMPI on Acute Thermal Nociception Behavior in Hot Plate Test
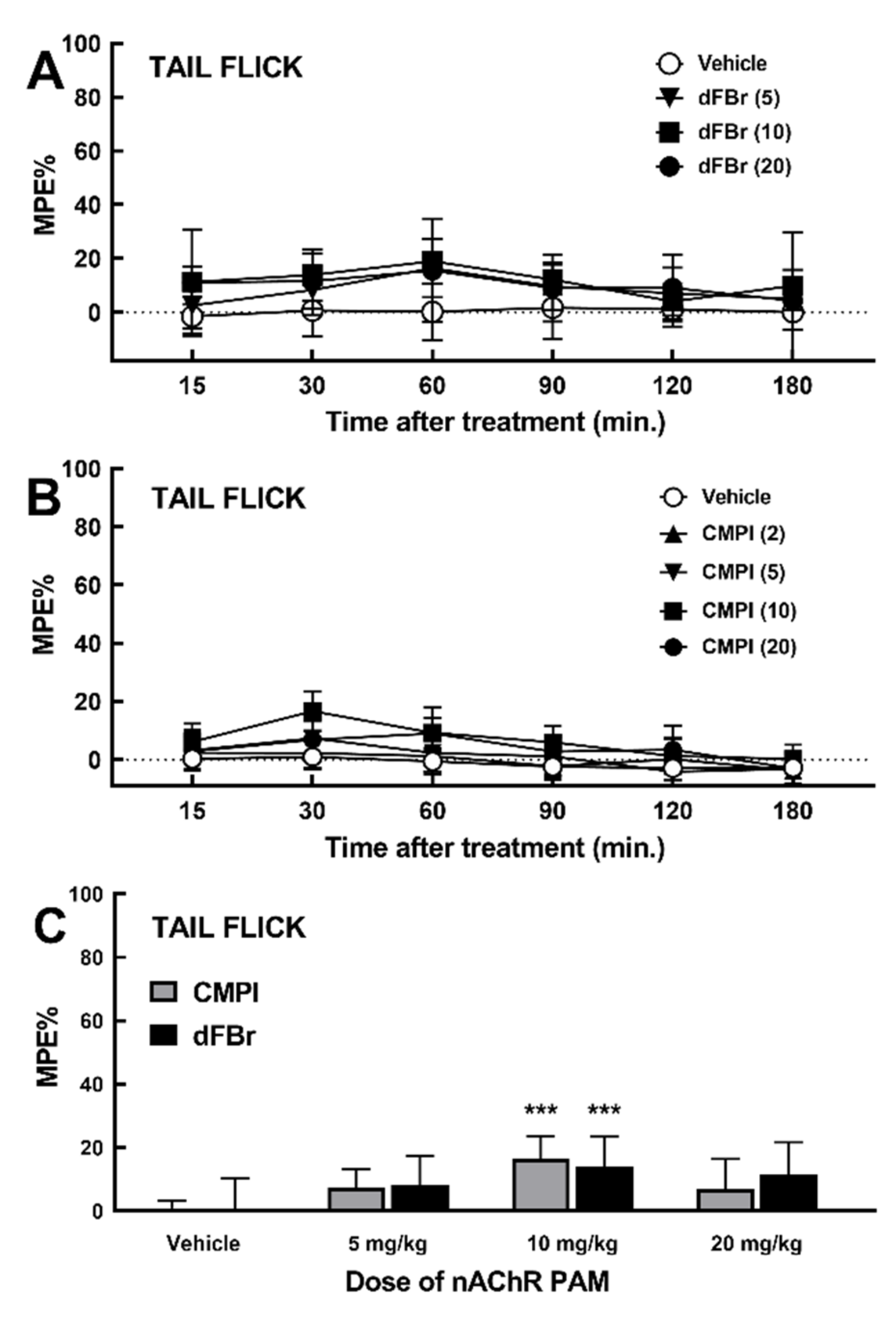
2.2. Effect of dFBr and CMPI on Acute Thermal Nociception Behavior in Tail Flick Test
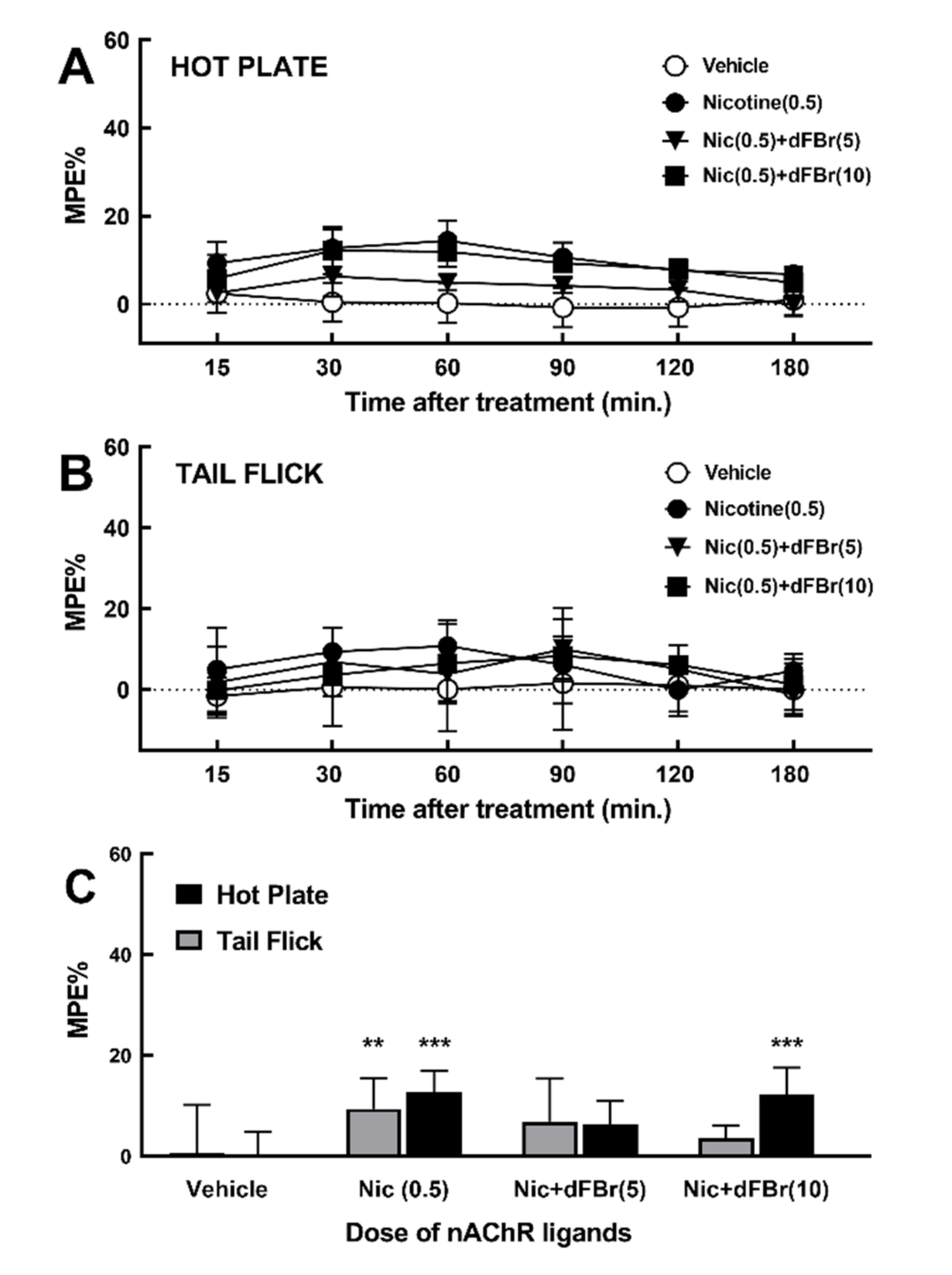
2.3. Effect of dFBr on Nicotine-Induced Acute Thermal Antinociception
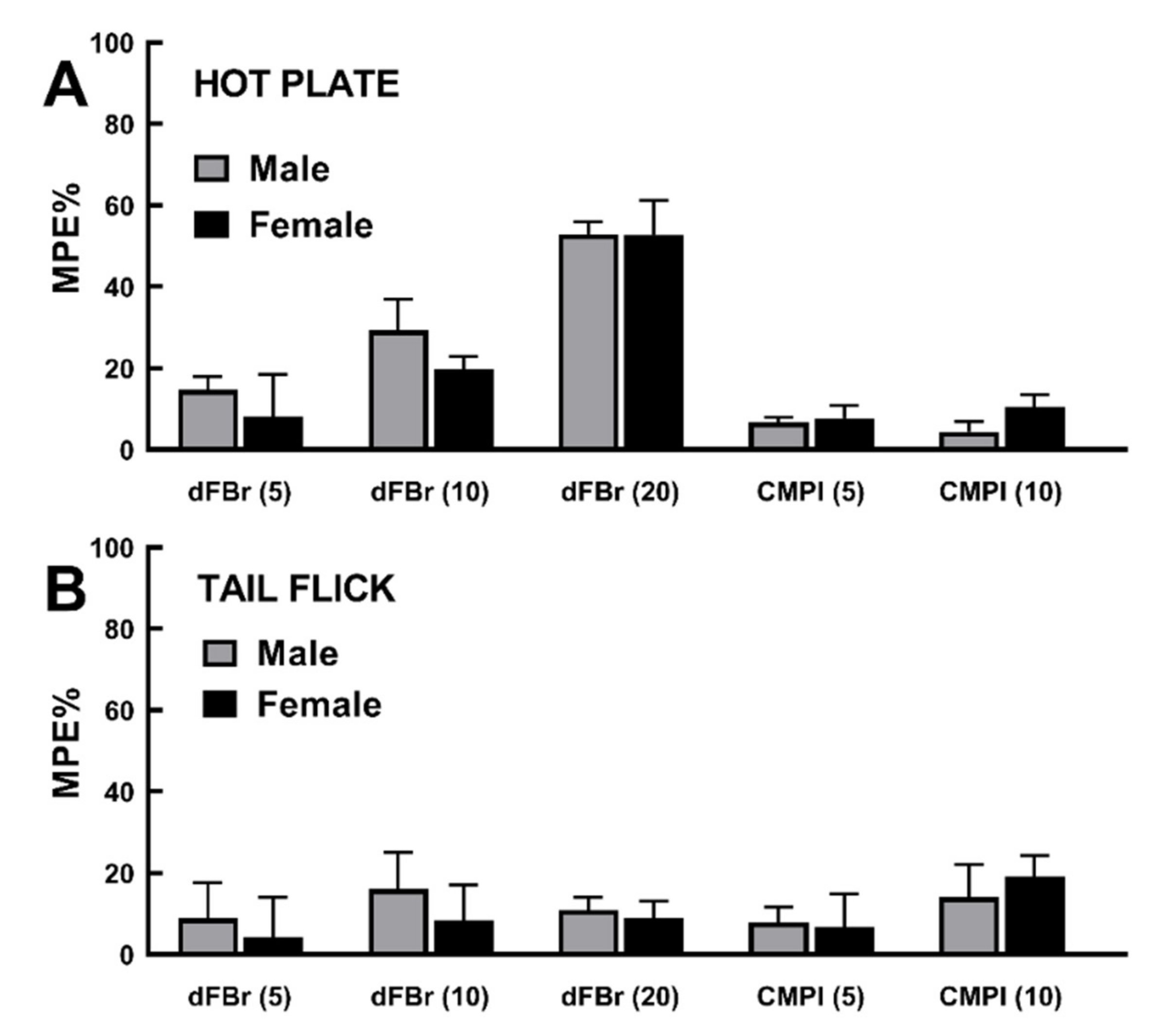
2.4. Rat Sex Does Not Influence dFBr and CMPI Modulation of Acute Thermal Nociception
3. Discussion
4. Materials and Methods
4.1. Animals
4.2. Drugs
4.3. Acute Thermal Nociceptive Tests
4.3.1. Hot Plate Test
4.3.2. Tail Flick Test
4.4. Data Analyses
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Dineley, K.T.; Pandya, A.A.; Yakel, J.L. Nicotinic ACh receptors as therapeutic targets in CNS disorders. Trends Pharmacol. Sci. 2015, 36, 96–108. [Google Scholar] [CrossRef] [PubMed]
- Zoli, M.; Pistillo, F.; Gotti, C. Diversity of native nicotinic receptor subtypes in mammalian brain. Neuropharmacology 2015, 96, 302–311. [Google Scholar] [CrossRef] [PubMed]
- DeDominicis, K.E.; Sahibzada, N.; Olson, T.T.; Xiao, Y.; Wolfe, B.B.; Kellar, K.J.; Yasuda, R.P. The (alpha4)3(beta2)2 Stoichiometry of the Nicotinic Acetylcholine Receptor Predominates in the Rat Motor Cortex. Mol. Pharmacol. 2017, 92, 327–337. [Google Scholar] [CrossRef] [PubMed]
- Nirogi, R.; Goura, V.; Abraham, R.; Jayarajan, P. alpha4beta2* neuronal nicotinic receptor ligands (agonist, partial agonist and positive allosteric modulators) as therapeutic prospects for pain. Eur. J. Pharmacol. 2013, 712, 22–29. [Google Scholar] [CrossRef]
- Ueda, M.; Iida, Y.; Tominaga, A.; Yoneyama, T.; Ogawa, M.; Magata, Y.; Nishimura, H.; Kuge, Y.; Saji, H. Nicotinic acetylcholine receptors expressed in the ventralposterolateral thalamic nucleus play an important role in anti-allodynic effects. Br. J. Pharmacol. 2010, 159, 1201–1210. [Google Scholar] [CrossRef]
- Umana, I.C.; Daniele, C.A.; McGehee, D.S. Neuronal nicotinic receptors as analgesic targets: it’s a winding road. Biochem. Pharmacol. 2013, 86, 1208–1214. [Google Scholar] [CrossRef]
- Decker, M.W.; Rueter, L.E.; Bitner, R.S. Nicotinic acetylcholine receptor agonists: a potential new class of analgesics. Curr. Top. Med. Chem. 2004, 4, 369–384. [Google Scholar] [CrossRef]
- Hurst, R.; Rollema, H.; Bertrand, D. Nicotinic acetylcholine receptors: from basic science to therapeutics. Pharmacol. Ther. 2013, 137, 22–54. [Google Scholar] [CrossRef]
- Damaj, M.I.; Freitas, K.; Bagdas, D.; Flood, P. Nicotinic Receptors as Targets for Novel Analgesics and Anti-inflammatory Drugs. Recept. Ser. 2014, 26, 239–254. [Google Scholar] [CrossRef]
- Bannon, A.W.; Decker, M.W.; Holladay, M.W.; Curzon, P.; Donnelly-Roberts, D.; Puttfarcken, P.S.; Bitner, R.S.; Diaz, A.; Dickenson, A.H.; Porsolt, R.D.; et al. Broad-spectrum, non-opioid analgesic activity by selective modulation of neuronal nicotinic acetylcholine receptors. Science 1998, 279, 77–81. [Google Scholar] [CrossRef]
- Marubio, L.M.; del Mar Arroyo-Jimenez, M.; Cordero-Erausquin, M.; Lena, C.; Le Novere, N.; de Kerchove d’Exaerde, A.; Huchet, M.; Damaj, M.I.; Changeux, J.P. Reduced antinociception in mice lacking neuronal nicotinic receptor subunits. Nature 1999, 398, 805–810. [Google Scholar] [CrossRef] [PubMed]
- Kesingland, A.C.; Gentry, C.T.; Panesar, M.S.; Bowes, M.A.; Vernier, J.M.; Cube, R.; Walker, K.; Urban, L. Analgesic profile of the nicotinic acetylcholine receptor agonists, (+)-epibatidine and ABT-594 in models of persistent inflammatory and neuropathic pain. Pain 2000, 86, 113–118. [Google Scholar] [CrossRef]
- Lynch, J.J., 3rd; Wade, C.L.; Mikusa, J.P.; Decker, M.W.; Honore, P. ABT-594 (a nicotinic acetylcholine agonist): anti-allodynia in a rat chemotherapy-induced pain model. Eur. J. Pharmacol. 2005, 509, 43–48. [Google Scholar] [CrossRef] [PubMed]
- Jain, K.K. Modulators of nicotinic acetylcholine receptors as analgesics. Curr. Opin. Investig. Drugs 2004, 5, 76–81. [Google Scholar]
- Lee, C.H.; Zhu, C.; Malysz, J.; Campbell, T.; Shaughnessy, T.; Honore, P.; Polakowski, J.; Gopalakrishnan, M. alpha4beta2 neuronal nicotinic receptor positive allosteric modulation: an approach for improving the therapeutic index of alpha4beta2 nAChR agonists in pain. Biochem. Pharmacol. 2011, 82, 959–966. [Google Scholar] [CrossRef]
- Williams, D.K.; Wang, J.; Papke, R.L. Positive allosteric modulators as an approach to nicotinic acetylcholine receptor-targeted therapeutics: advantages and limitations. Biochem. Pharmacol. 2011, 82, 915–930. [Google Scholar] [CrossRef]
- Rode, F.; Munro, G.; Holst, D.; Nielsen, E.O.; Troelsen, K.B.; Timmermann, D.B.; Ronn, L.C.; Grunnet, M. Positive allosteric modulation of alpha4beta2 nAChR agonist induced behaviour. Brain Res. 2012, 1458, 67–75. [Google Scholar] [CrossRef]
- Uteshev, V.V. Allosteric Modulation of Nicotinic Acetylcholine Receptors: The Concept and Therapeutic Trends. Curr. Pharm. Des. 2016, 22, 1986–1997. [Google Scholar] [CrossRef]
- Mohamed, T.S.; Jayakar, S.S.; Hamouda, A.K. Orthosteric and Allosteric Ligands of Nicotinic Acetylcholine Receptors for Smoking Cessation. Front Mol. Neurosci. 2015, 8, 71. [Google Scholar] [CrossRef]
- Wilkerson, J.L.; Deba, F.; Crowley, M.L.; Hamouda, A.K.; McMahon, L.R. Advances in the In vitro and In vivo pharmacology of Alpha4beta2 nicotinic receptor positive allosteric modulators. Neuropharmacology 2020, 168, 108008. [Google Scholar] [CrossRef]
- Manetti, D.; Bellucci, C.; Chiaramonte, N.; Dei, S.; Teodori, E.; Romanelli, M.N. Designing selective modulators for the nicotinic receptor subtypes: challenges and opportunities. Future Med. Chem. 2018, 10, 433–459. [Google Scholar] [CrossRef]
- Pandya, A.; Yakel, J.L. Allosteric modulators of the alpha4beta2 subtype of neuronal nicotinic acetylcholine receptors. Biochem. Pharmacol. 2011, 82, 952–958. [Google Scholar] [CrossRef]
- Sala, F.; Mulet, J.; Reddy, K.P.; Bernal, J.A.; Wikman, P.; Valor, L.M.; Peters, L.; Konig, G.M.; Criado, M.; Sala, S. Potentiation of human alpha4beta2 neuronal nicotinic receptors by a Flustra foliacea metabolite. Neurosci. Lett. 2005, 373, 144–149. [Google Scholar] [CrossRef] [PubMed]
- Albrecht, B.K.; Berry, V.; Boezio, A.A.; Cao, L.; Clarkin, K.; Guo, W.; Harmange, J.C.; Hierl, M.; Huang, L.; Janosky, B.; et al. Discovery and optimization of substituted piperidines as potent, selective, CNS-penetrant alpha4beta2 nicotinic acetylcholine receptor potentiators. Bioorg. Med. Chem. Lett. 2008, 18, 5209–5212. [Google Scholar] [CrossRef] [PubMed]
- Olsen, J.A.; Ahring, P.K.; Kastrup, J.S.; Gajhede, M.; Balle, T. Structural and functional studies of the modulator NS9283 reveal agonist-like mechanism of action at alpha4beta2 nicotinic acetylcholine receptors. J. Biol. Chem. 2014, 289, 24911–24921. [Google Scholar] [CrossRef] [PubMed]
- Zhu, C.Z.; Chin, C.L.; Rustay, N.R.; Zhong, C.; Mikusa, J.; Chandran, P.; Salyers, A.; Gomez, E.; Simler, G.; Lewis, L.G.; et al. Potentiation of analgesic efficacy but not side effects: co-administration of an alpha4beta2 neuronal nicotinic acetylcholine receptor agonist and its positive allosteric modulator in experimental models of pain in rats. Biochem. Pharmacol. 2011, 82, 967–976. [Google Scholar] [CrossRef] [PubMed]
- Bagdas, D.; Ergun, D.; Jackson, A.; Toma, W.; Schulte, M.K.; Damaj, M.I. Allosteric modulation of alpha4beta2* nicotinic acetylcholine receptors: Desformylflustrabromine potentiates antiallodynic response of nicotine in a mouse model of neuropathic pain. Eur. J. Pain 2018, 22, 84–93. [Google Scholar] [CrossRef]
- Weggel, L.A.; Pandya, A.A. Acute Administration of Desformylflustrabromine Relieves Chemically Induced Pain in CD-1 Mice. Molecules 2019, 24. [Google Scholar] [CrossRef]
- Gotti, C.; Clementi, F.; Fornari, A.; Gaimarri, A.; Guiducci, S.; Manfredi, I.; Moretti, M.; Pedrazzi, P.; Pucci, L.; Zoli, M. Structural and functional diversity of native brain neuronal nicotinic receptors. Biochem. Pharmacol. 2009, 78, 703–711. [Google Scholar] [CrossRef]
- Bertrand, D.; Terry, A.V., Jr. The wonderland of neuronal nicotinic acetylcholine receptors. Biochem. Pharmacol. 2018, 151, 214–225. [Google Scholar] [CrossRef]
- Hamouda, A.K.; Deba, F.; Wang, Z.J.; Cohen, J.B. Photolabeling a Nicotinic Acetylcholine Receptor (nAChR) with an (alpha4)3(beta2)2 nAChR-Selective Positive Allosteric Modulator. Mol. Pharmacol. 2016, 89, 575–584. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.J.; Deba, F.; Mohamed, T.S.; Chiara, D.C.; Ramos, K.; Hamouda, A.K. Unraveling amino acid residues critical for allosteric potentiation of (alpha4)3(beta2)2-type nicotinic acetylcholine receptor responses. J. Biol. Chem. 2017, 292, 9988–10001. [Google Scholar] [CrossRef] [PubMed]
- Weltzin, M.M.; Schulte, M.K. Desformylflustrabromine Modulates alpha4beta2 Neuronal Nicotinic Acetylcholine Receptor High- and Low-Sensitivity Isoforms at Allosteric Clefts Containing the beta2 Subunit. J. Pharmacol. Exp. Ther. 2015, 354, 184–194. [Google Scholar] [CrossRef] [PubMed]
- Deba, F.; Ali, H.I.; Tairu, A.; Ramos, K.; Ali, J.; Hamouda, A.K. LY2087101 and dFBr share transmembrane binding sites in the (alpha4)3(beta2)2 Nicotinic Acetylcholine Receptor. Sci. Rep. 2018, 8, 1249. [Google Scholar] [CrossRef]
- AlSharari, S.; Bagdas, D.; Akbarali, H.; Cabral, G.A.; Carroll, F.I.; Damaj, I.; Lichtman, P. Sex Differences and Drug Dose Influence the Role of the alpha-7 Nicotinic Acetylcholine Receptor in the Mouse Dextran Sodium Sulfate-Induced Colitis Model. Am. J. Gastroenterol. 2015, 110, S776. [Google Scholar] [CrossRef]
- Liu, X. Positive allosteric modulation of alpha 4 beta 2 nicotinic acetylcholine receptors as a new approach to smoking reduction: evidence from a rat model of nicotine self-administration. Psychopharmacology 2013, 230, 203–213. [Google Scholar] [CrossRef]
- Mitra, S.; Mucha, M.; Khatri, S.N.; Glenon, R.; Schulte, M.K.; Bult-Ito, A. Attenuation of Compulsive-Like Behavior Through Positive Allosteric Modulation of alpha4beta2 Nicotinic Acetylcholine Receptors in Non-Induced Compulsive-Like Mice. Front Behav. Neurosci. 2016, 10, 244. [Google Scholar] [CrossRef]
- Moerke, M.J.; de Moura, F.B.; Koek, W.; McMahon, L.R. Effects of nicotine in combination with drugs described as positive allosteric nicotinic acetylcholine receptor modulators in vitro: discriminative stimulus and hypothermic effects in mice. Eur. J. Pharmacol. 2016, 786, 169–178. [Google Scholar] [CrossRef]
- Hamouda, A.K.; Jackson, A.; Bagdas, D.; Imad Damaj, M. Reversal of Nicotine Withdrawal Signs Through Positive Allosteric Modulation of alpha4beta2 Nicotinic Acetylcholine Receptors in Male Mice. Nicotine Tob. Res. 2018, 20, 903–907. [Google Scholar] [CrossRef]
- Dennis, S.G.; Melzack, R.; Gutman, S.; Boucher, F. Pain modulation by adrenergic agents and morphine as measured by three pain tests. Life Sci. 1980, 26, 1247–1259. [Google Scholar] [CrossRef]
- Langerman, L.; Zakowski, M.I.; Piskoun, B.; Grant, G.J. Hot plate versus tail flick: evaluation of acute tolerance to continuous morphine infusion in the rat model. J. Pharmacol. Toxicol. Methods 1995, 34, 23–27. [Google Scholar] [CrossRef]
- Hamouda, A.K.; Wang, Z.J.; Stewart, D.S.; Jain, A.D.; Glennon, R.A.; Cohen, J.B. Desformylflustrabromine (dFBr) and [3H]dFBr-Labeled Binding Sites in a Nicotinic Acetylcholine Receptor. Mol. Pharmacol. 2015, 88, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Alcaino, C.; Musgaard, M.; Minguez, T.; Mazzaferro, S.; Faundez, M.; Iturriaga-Vasquez, P.; Biggin, P.C.; Bermudez, I. Role of the Cys Loop and Transmembrane Domain in the Allosteric Modulation of alpha4beta2 Nicotinic Acetylcholine Receptors. J. Biol. Chem. 2017, 292, 551–562. [Google Scholar] [CrossRef]
- Freitas, K.; Negus, S.S.; Carroll, F.I.; Damaj, M.I. In vivo pharmacological interactions between a type II positive allosteric modulator of alpha7 nicotinic ACh receptors and nicotinic agonists in a murine tonic pain model. Br. J. Pharmacol. 2013, 169, 567–579. [Google Scholar] [CrossRef] [PubMed]

© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Deba, F.; Ramos, K.; Vannoy, M.; Munoz, K.; Akinola, L.S.; Damaj, M.I.; Hamouda, A.K. Examining the Effects of (α4)3(β2)2 Nicotinic Acetylcholine Receptor-Selective Positive Allosteric Modulator on Acute Thermal Nociception in Rats. Molecules 2020, 25, 2923. https://doi.org/10.3390/molecules25122923
Deba F, Ramos K, Vannoy M, Munoz K, Akinola LS, Damaj MI, Hamouda AK. Examining the Effects of (α4)3(β2)2 Nicotinic Acetylcholine Receptor-Selective Positive Allosteric Modulator on Acute Thermal Nociception in Rats. Molecules. 2020; 25(12):2923. https://doi.org/10.3390/molecules25122923
Chicago/Turabian StyleDeba, Farah, Kara Ramos, Matthew Vannoy, Kemburli Munoz, Lois S. Akinola, M. Imad Damaj, and Ayman K. Hamouda. 2020. "Examining the Effects of (α4)3(β2)2 Nicotinic Acetylcholine Receptor-Selective Positive Allosteric Modulator on Acute Thermal Nociception in Rats" Molecules 25, no. 12: 2923. https://doi.org/10.3390/molecules25122923
APA StyleDeba, F., Ramos, K., Vannoy, M., Munoz, K., Akinola, L. S., Damaj, M. I., & Hamouda, A. K. (2020). Examining the Effects of (α4)3(β2)2 Nicotinic Acetylcholine Receptor-Selective Positive Allosteric Modulator on Acute Thermal Nociception in Rats. Molecules, 25(12), 2923. https://doi.org/10.3390/molecules25122923




