Encapsidation of Different Plasmonic Gold Nanoparticles by the CCMV CP
Abstract
1. Introduction
2. Results
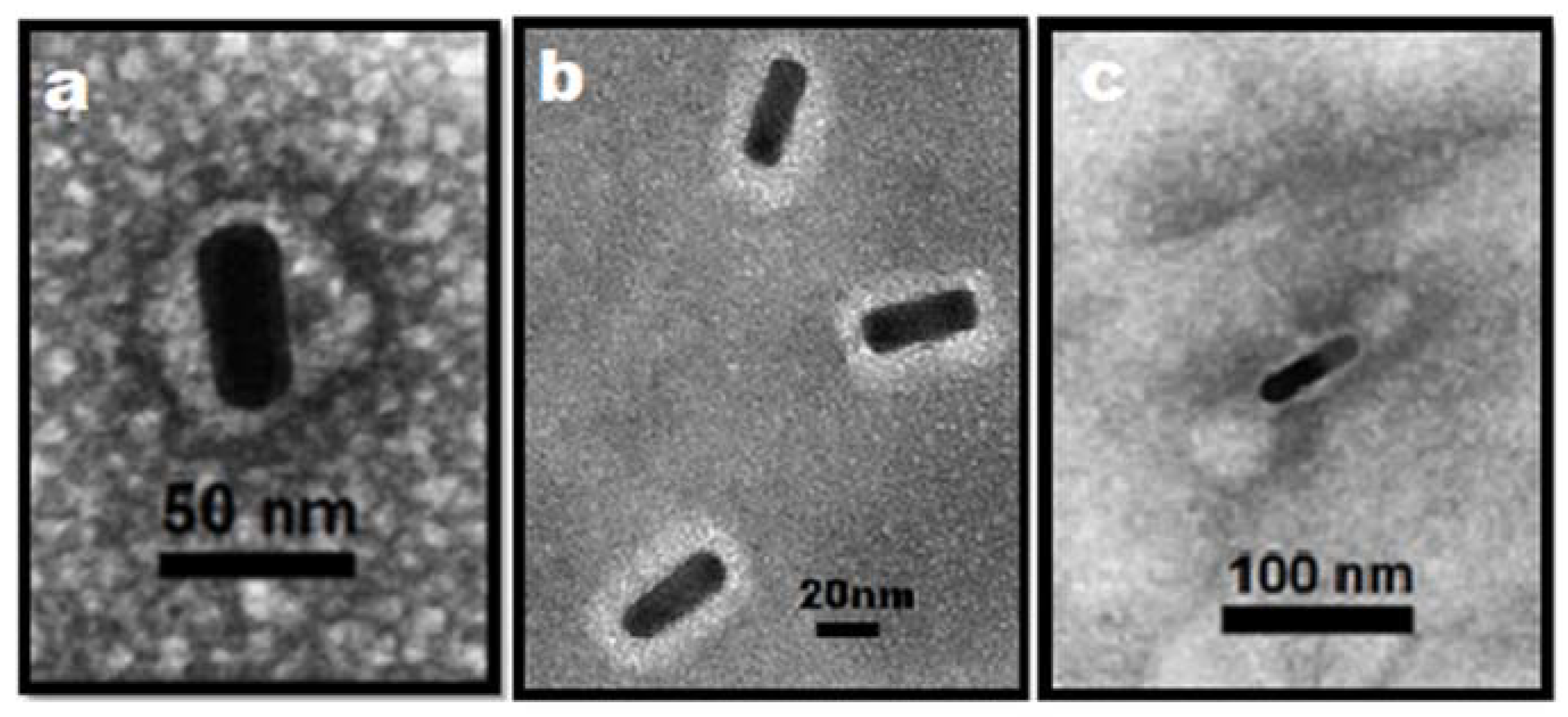
2.1. Synthesis of Gold Nanoparticles
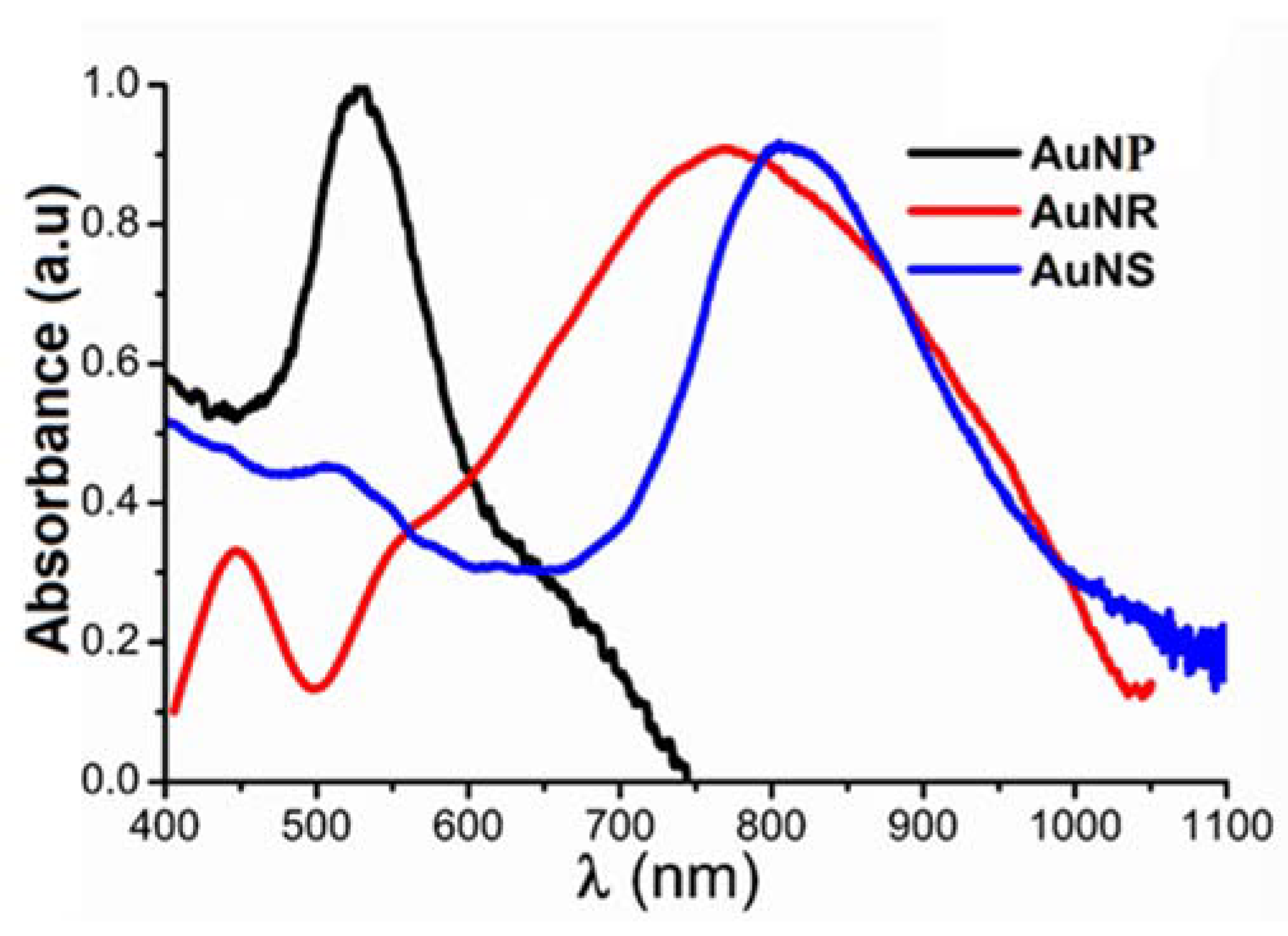
2.2. Absorption Spectra of Gold Nanoparticles
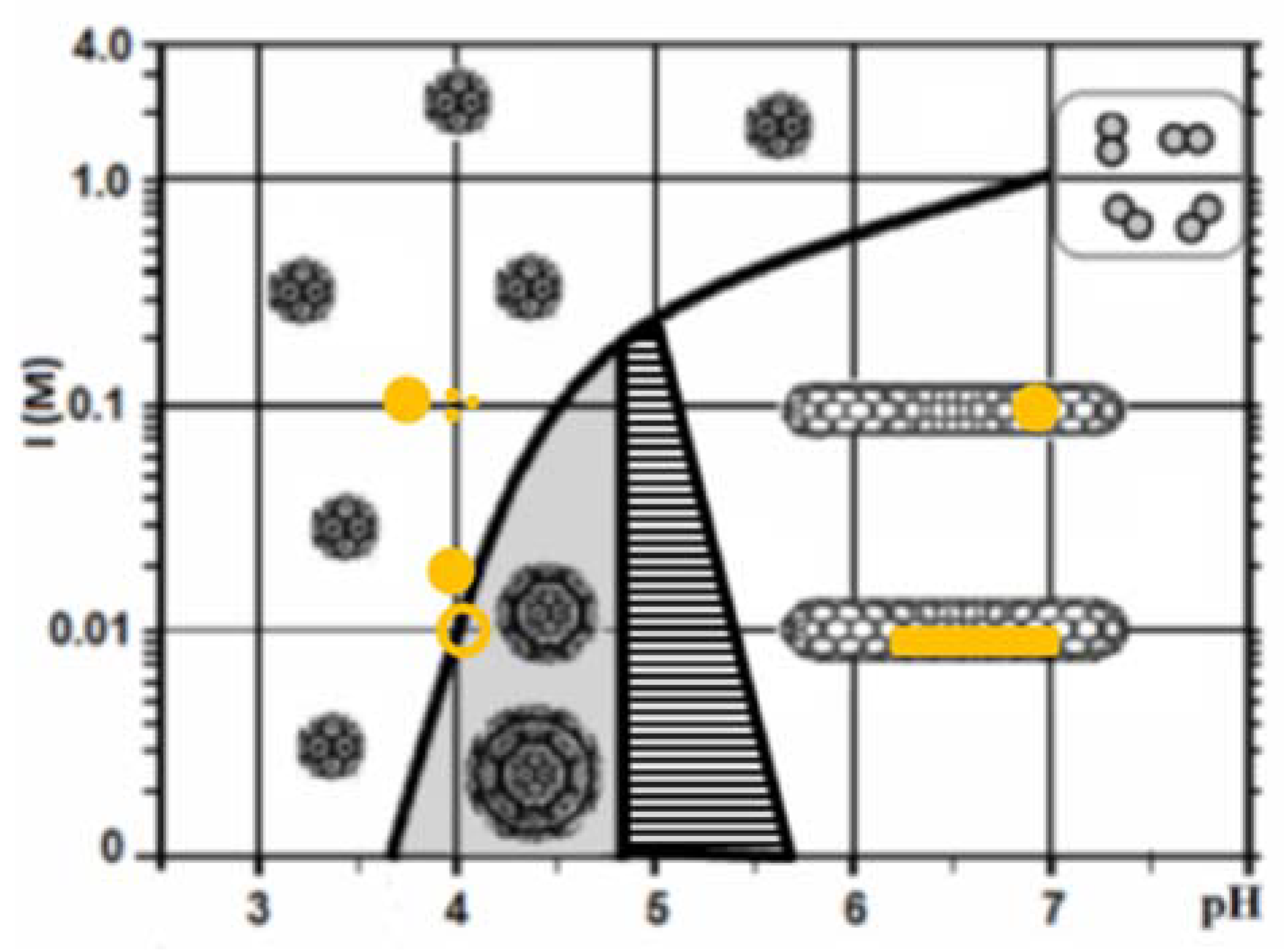
2.3. Encapsidation of AuNP
2.4. Encapsidartion of AuNSs
2.5. Encapsidation of AuNRs
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Synthesis of AuNP
4.3. Synthesis of AuNSs
4.4. Synthesis of AuNRs
4.5. CCMV CP Purification
4.6. AuNPs Encapsidation
4.7. Characterization Methods
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Chen, C.; Daniel, M.C.; Quinkert, Z.T.; De, M.; Stein, B.; Bowman, V.D.; Chipman, P.R.; Rotello, V.M.; Kao, C.C.; Dragnea, B. Nanoparticle-templated assembly of viral protein cages. Nano Lett. 2006, 6, 611–615. [Google Scholar] [CrossRef] [PubMed]
- Bancroft, J.B.; Bracker, C.E.; Wagner, G.W. Structures derived from cowpea chlorotic mottle and brome mosaic virus protein. Virology 1969, 38, 324–335. [Google Scholar] [CrossRef]
- De la Escosura, A.; Nolte, R.J.M.; Cornelissen, J.J.L.M. Viruses and protein cages as nanocontainers and nanoreactors. J. Mater. Chem. 2009, 19, 2274–2278. [Google Scholar] [CrossRef]
- Wyatt, S.D.; Kuhn, C.W. Replication and properties of cowpea chlorotic mottle virus in resistant cowpeas. Phytopathology 1979, 69, 125–129. [Google Scholar] [CrossRef]
- Bancroft, J.B.; Hiebert, E.; Rees, M.W.; Markham, R. Properties of cowpea chlorotic mottle virus, its protein and nucleic acid. Virology 1968, 34, 224–239. [Google Scholar] [CrossRef]
- Bancroft, J.B.; Hills, G.J.; Markham, R. A study of the self-assembly process in a small spherical virus. Formation of organized structures from protein subunits in vitro. Virology 1967, 31, 354–379. [Google Scholar] [CrossRef]
- Zlotnick, A.; Mukhopadhyay, S. Virus assembly, allostery and antivirals. Trends Microbiol. 2011, 19, 14–23. [Google Scholar] [CrossRef]
- Vega-Acosta, J.R.; Cadena-Nava, R.D.; Gelbart, W.M.; Knobler, C.M.; Ruiz-Garcia, J. Electrophoretic Mobilities of a Viral Capsid, Its CP, and Their Relation to Viral Assembly. J. Phys. Chem. B 2014, 118, 1984–1989. [Google Scholar] [CrossRef]
- Kaiser, C.R.; Flenniken, M.L.; Gillitzer, E.; Harmsen, A.L.; Harmsen, A.G.; Jutila, M.A.; Young, M.J. Biodistribution studies of protein cage nanoparticles demonstrate broad tissue distribution and rapid clearance in vivo. Int. J. Nanome. 2007, 2, 715–733. [Google Scholar]
- Adolph, K.W.; Butler, P.J.G. Studies on the assembly of a spherical plant virus: I. States of aggregation of the isolated protein. J. Mol. Biol. 1974, 88, 327–341. [Google Scholar] [CrossRef]
- Lavelle, L.; Gingery, M.; Phillips, M.; Gelbart, W.M.; Knobler, C.M.; Cadena-Nava, R.D.; Vega-Acosta, J.R.; Pinedo-Torres, L.A.; Ruiz-Garcia, J. Phase diagram of self-assembled viral CP polymorphs. J. Phys. Chem. B 2009, 113, 3813–3819. [Google Scholar] [CrossRef] [PubMed]
- Vriend, G.; Verduin, B.J.M.; Hemminga, M.A. Role of the N-terminal part of the coat protein in the assembly of cowpea chlorotic mottle virus: A 500 MHz proton nuclear magnetic resonance study and structural calculations. J. Mol. Biol. 1986, 191, 453–460. [Google Scholar] [CrossRef]
- Jolley, C.; Klem, M.; Harrington, R.; Parise, J.; Douglas, T. Structure and photoelectrochemistry of a virus capsid-TiO2 nanocomposite. Nanoscale 2011, 3, 1004–1007. [Google Scholar] [CrossRef]
- Hu, Y.; Zandi, R.; Anavitarte, A.; Knobler, C.M.; Gelbart, W.M. Packaging of a Polymer by a Viral Capsid: The Interplay between Polymer Length and Capsid Size. Biophys. J. 2008, 94, 1428–1436. [Google Scholar] [CrossRef] [PubMed]
- De la Escosura, A.; Verwegen, M.; Sikkema, F.D.; Comellas-Aragones, M.; Kirilyuk, A.; Rasing, T.; Nolte, R.J.M.; Cornelissen, J.J.L.M. Viral capsids as templates for the production of monodisperse Prussian blue nanoparticles. Chem. Commun. 2008, 13, 1542–1544. [Google Scholar] [CrossRef] [PubMed]
- Cadena-Nava, R.D.; Comas-Garcia, M.; Garmann, R.F.; Rao, A.L.N.; Knobler, C.M.; Gelbart, W.M. Self-Assembly of Viral CP and RNA Molecules of Different Sizes: Requirement for a Specific High Protein/RNA Mass Ratio. J. Virol. 2012, 86, 3318–3326. [Google Scholar] [CrossRef]
- Sánchez-Sánchez, L.; Cadena-Nava, R.D.; Palomares, L.A.; Ruiz-Garcia, J.; Koay, M.S.; Cornelissen, J.J.; Vazquez-Duhalt, R. Chemotherapy pro-drug activation by biocatalytic virus-like nanoparticles containing cytochrome P450. Enzyme Microb. Technol. 2014, 60, 24–31. [Google Scholar] [CrossRef]
- Sardar, R.; Funston, A.M.; Mulvaney, P.; Murray, R.W. Gold Nanoparticles: Past, Present, and Future. Langmuir 2009, 25, 13840–13851. [Google Scholar] [CrossRef]
- Daniel, M.C.; Astruc, D. Gold Nanoparticles: Assembly, Supramolecular Chemistry, Quantum-Size-Related Properties, and Applications Toward Biology, Catalysis, and Nanotechnology. Chem. Rev. 2004, 104, 293–346. [Google Scholar] [CrossRef]
- Kimling, J.; Maier, M.; Okenve, B.; Kotaidis, V.; Ballot, H.; Plech, A. Turkevich Method for Gold Nanoparticle Synthesis Revisited. J. Phys. Chem. B 2006, 110, 15700–15707. [Google Scholar] [CrossRef]
- Jana, N.R.; Gearheart, L.; Murphy, C.J. Wet Chemical Synthesis of High Aspect Ratio Cylindrical Gold Nanorods. J. Phys. Chem. B 2001, 105, 4065–4067. [Google Scholar] [CrossRef]
- Khoury, C.; Vo-Dinh, T. Gold nanostars for surface-enhanced Raman scattering: Synthesis, characterization and optimization. J. Phys. Chem. C 2008, 112, 18849–18859. [Google Scholar] [CrossRef] [PubMed]
- Senger, R.T.; Dag, S.; Ciraci, S. Chiral single-wall gold nanotubes. Phys. Rev. Lett. 2004, 93, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.-J.; Chiu, P.-H.; Wang, Y.-H.; Chen, W.R.; Meen, T.H. Synthesis of the Gold Nanocubes by Electrochemical Technique. J. Electrochem. Soc. 2006, 153, D129–D133. [Google Scholar] [CrossRef]
- Wang, B.; Yin, S.; Wang, G.; Buldum, A.; Zhao, J. Novel structures and properties of gold nanowires. Phys. Rev. Lett. 2001, 86, 2046–2049. [Google Scholar] [CrossRef]
- Sun, Y.; Xia, Y. Mechanistic Study on the Replacement Reaction between Silver Nanostructures and Chloroauric Acid in Aqueous Medium Mechanistic Study on the Replacement Reaction between Silver Nanostructures and Chloroauric Acid in Aqueous. J. Am. Chem. Soc. 2004, 126, 3892–3901. [Google Scholar] [CrossRef]
- Simpson, C.R.; Kohl, M.; Essenpreis, M.; Cope, M. Near-infrared optical properties of ex vivo human skin and subcutaneous tissues measured using the Monte Carlo inversion technique. Phys. Med. Biol. 1998, 43, 2465–2478. [Google Scholar] [CrossRef]
- Bashkatov, A.N.; Genina, E.A.; Kochubey, V.I.; Tuchin, V.V. Optical properties of human skin, subcutaneous and mucous tissues in the wavelength range from 400 to 2000 nm. J. Phys. D. Appl. Phys. 2005, 38, 2543–2555. [Google Scholar] [CrossRef]
- Jain, S.; Hirst, D.G.; O’Sullivan, J.M. Gold nanoparticles as novel agents for cancer therapy. Br. J. Radiol. 2012, 85, 101–113. [Google Scholar] [CrossRef]
- Liang, Z.; Li, X.; Xie, Y.; Liu, S. ‘Smart’ gold nanoshells for combined cancer chemotherapy and hyperthermia. Biomed. Mater. 2014, 9, 025012. [Google Scholar] [CrossRef]
- Huschka, R.; Barhoumi, A.; Liu, Q.; Roth, J.A.; Ji, L.; Halas, N.J. Gene silencing by gold nanoshell-mediated delivery and laser-triggered release of antisense oligonucleotide and siRNA. ACS Nano 2012, 6, 7681–7691. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; El-Sayed, I.H.; Qian, W.; El-Sayed, M.A. Cancer Cell Imaging and Photothermal Therapy in the Near-Infrared Region by Using Gold Nanorods. J. Am. Chem. Soc. 2006, 128, 2115–2120. [Google Scholar] [CrossRef] [PubMed]
- Loo, L.; Guenther, R.H.; Basnayake, V.R.; Lommel, S.A.; Franzen, S. Controlled encapsidation of gold nanoparticles by a viral protein shell. J. Am. Chem. Soc. 2006, 128, 4502–4503. [Google Scholar] [CrossRef] [PubMed]
- Dragnea, B.; Chen, C.; Kwak, E.-S.; Stein, B.; Kao, C.C. Gold nanoparticles as spectroscopic enhancers for in vitro studies on single viruses. J. Am. Chem. Soc. 2003, 125, 6374–6375. [Google Scholar] [CrossRef]
- Sun, J.; DuFort, C.; Daniel, M.-C.; Murali, A.; Chen, C.; Gopinath, K.; Stein, B.; De, M.; Rotello, V.M.; Holzenburg, A.; et al. Core-controlled polymorphism in virus-like particles. Proc. Natl. Acad. Sci. 2007, 104, 1354–1359. [Google Scholar] [CrossRef]
- Aniagyei, S.E.; Kennedy, C.J.; Stein, B.; Willits, D.A.; Douglas, T.; Young, M.J.; De, M.; Rotello, V.M.; Srisathiyanarayanan, D.; Kao, C.C.; et al. Synergistic effects of mutations and nanoparticle templating in the self-assembly of cowpea chlorotic mottle virus capsids. Nano Lett. 2009, 9, 393–398. [Google Scholar] [CrossRef]
- Liu, A.; Verwegen, M.; de Ruiter, M.V.; Maassen, S.J.; Traulsen, C.H.H.; Cornelissen, J.J. Protein cages as containers for gold nanoparticles. J. Phys. Chem. B 2016, 120, 6352–6357. [Google Scholar] [CrossRef]
- Zhang, G.D.; Jasinski, J.B.; Howell, J.L.; Patel, D.; Stephens, D.P.; Gobin, A.M. Tunability and stability of gold nanoparticles obtained from chloroauric acid and sodium thiosulfate reaction. Nanoscale Res. Lett. 2012, 7, 337. [Google Scholar] [CrossRef]
- Fröhlich, E. The role of surface charge in cellular uptake and cytotoxicity of medical nanoparticles. Int. J. Nanomed. 2012, 7, 5577–5591. [Google Scholar] [CrossRef]
- Yildiz, I.; Shukla, S.; Steinmetz, N.F. Applications of viral nanoparticles in medicine. Curr. Opin. Biotechnol. 2011, 22, 901–908. [Google Scholar] [CrossRef]
- Chang, C.B.; Knobler, C.M.; Gelbart, W.M.; Mason, T.G. Curvature dependence of viral protein structures on encapsidated nanoemulsion droplets. ACS Nano 2008, 2, 281–286. [Google Scholar] [CrossRef] [PubMed]
- Wilts, B.D.; Schaap, I.A.; Schmidt, C.F. Swelling and softening of the cowpea chlorotic mottle virus in response to pH shifts. Biophys. J. 2015, 108, 2541–2549. [Google Scholar] [CrossRef]
- Park, J.; Estrada, A.; Sharp, K.; Sang, K.; Schwartz, J.A.; Smith, D.K.; Coleman, C.; Payne, J.D.; Korgel, B.A.; Dunn, A.K.; et al. Two-photon-induced photoluminescence imaging of tumors using near-infrared excited gold nanoshells. Opt. Express 2008, 16, 1590–1599. [Google Scholar] [CrossRef] [PubMed]
- Loredo-Tovias, M.; Duran-Meza, A.L.; Villagrana-Escareño, M.V.; Vega-Acosta, R.; Reynaga-Hernández, E.; Flores-Tandy, L.M.; Valdes-Resendiz, O.E.; Cadena-Nava, R.D.; Alvizo-Paez, E.R.; Ruiz-Garcia, J. Encapsidated ultrasmall nanolipospheres as novel nanocarriers for highly hydrophobic anticancer drugs. Nanoscale 2017, 9, 11625–11631. [Google Scholar] [CrossRef] [PubMed]
- Park, H.; Lim, D.J.; Vines, J.B.; Yoon, J.H.; Ryu, N.E. Gold nanoparticles for photothermal cancer therapy. Frontiers in chemistry. Front. Chem. 2019, 7, 167. [Google Scholar]
- Sivaraman, S.K.; Kumar, S.; Santhanam, V. Monodisperse sub-10nm gold nanoparticles by reversing the order of addition in Turkevich method-The role of chloroauric acid. J. Colloid Interface Sci. 2011, 361, 543–547. [Google Scholar] [CrossRef]
- Duran-Meza, A.L.; Moreno-Gutierrez, D.S.; Ruiz-Robles, J.F.; Bañuelos-Frias, A.; Segovia-González, X.F.; Longoria-Hernandez, A.M.; Gomez, E.; Ruiz-Garcia, J. Synthesis and Characterization of Extremely Small Gold Nanoshells, and Comparison of Their Photothermal Conversion Capacity with Gold Nanorods. Nanoscale 2016, 8, 11091–11098. [Google Scholar] [CrossRef]
- Vongsavat, V.; Vittur, B.M.; Bryan, W.W.; Kim, J.H.; Lee, T.R. Ultrasmall hollow gold–silver nanoshells with extinctions strongly red-shifted to the near-infrared. ACS Appl. Mater. Interfaces 2011, 3, 3616–3624. [Google Scholar] [CrossRef]
- Xu, D.; Mao, J.; He, Y.; Yeung, E.S. Size-tunable synthesis of high-quality gold nanorods under basic conditions by using H2O2 as the reducing agent. J. Mater. Chem. C 2014, 2, 4989–4996. [Google Scholar] [CrossRef]
- Michel, J.P.; Gingery, M.; Lavelle, L. Efficient purification of bromoviruses by ultrafiltration. J. Virol. Methods 2004, 122, 195–198. [Google Scholar] [CrossRef]
- Lavelle, L.; Michel, J.P.; Gingery, M. The disassembly, reassembly and stability of CCMV protein capsids. J. Virol. Methods 2007, 146, 311–316. [Google Scholar] [CrossRef] [PubMed]
- Burger, W.; Burge, M.J. Principles of Digital Image Processing; Springer: London, England, 2009; pp. 1–24. ISBN 978-1-84800-190-9. [Google Scholar]
- Elahi, N.; Kamali, M.; Baghersad, M.H. Recent biomedical applications of gold nanoparticles: A review. Talanta 2018, 184, 537–556. [Google Scholar] [CrossRef] [PubMed]
- Del Solar, V.; Contel, M. Metal-based antibody drug conjugates. Potential and challenges in their application as targeted therapies in cancer. J. Inorg. Biochem. 2019, 199, 110780. [Google Scholar] [CrossRef] [PubMed]
- Cruz, E.; Kayser, V. Synthesis and Enhanced Cellular Uptake In Vitro of Anti-HER2 Multifunctional Gold Nanoparticles. Cancers 2019, 11, 870. [Google Scholar] [CrossRef] [PubMed]
- Schoonen, L.; van Hest, J.C. Functionalization of protein-based nanocages for drug delivery applications. Nanoscale 2014, 6, 7124–7141. [Google Scholar] [CrossRef] [PubMed]
- Melancon, M.P.; Lu, W.; Yang, Z.; Zhang, R.; Cheng, Z.; Elliot, A.M.; Stafford, J.; Olson, T.; Zhang, J.Z.; Li, C. In vitro and in vivo targeting of hollow gold nanoshells directed at epidermal growth factor receptor for photothermal ablation therapy. Mol. Cancer Ther. 2008, 7, 1730–1739. [Google Scholar] [CrossRef]
- Villagrana-Escareño, M.V.; Reynaga-Hernández, E.; Galicia-Cruz, O.G.; Durán-Meza, A.L.; la Cruz-González, D.; Hernández-Carballo, C.Y.; Ruiz-Garcia, J. VLPs derived from the CCMV plant virus can directly transfect and deliver heterologous genes for translation into mammalian cells. BioMed. Res. Inter. 2019, 2019, 4630891. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are not available from the authors. |





© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Durán-Meza, A.L.; Escamilla-Ruiz, M.I.; Segovia-González, X.F.; Villagrana-Escareño, M.V.; Vega-Acosta, J.R.; Ruiz-Garcia, J. Encapsidation of Different Plasmonic Gold Nanoparticles by the CCMV CP. Molecules 2020, 25, 2628. https://doi.org/10.3390/molecules25112628
Durán-Meza AL, Escamilla-Ruiz MI, Segovia-González XF, Villagrana-Escareño MV, Vega-Acosta JR, Ruiz-Garcia J. Encapsidation of Different Plasmonic Gold Nanoparticles by the CCMV CP. Molecules. 2020; 25(11):2628. https://doi.org/10.3390/molecules25112628
Chicago/Turabian StyleDurán-Meza, Ana L., Martha I. Escamilla-Ruiz, Xochitl F. Segovia-González, Maria V. Villagrana-Escareño, J. Roger Vega-Acosta, and Jaime Ruiz-Garcia. 2020. "Encapsidation of Different Plasmonic Gold Nanoparticles by the CCMV CP" Molecules 25, no. 11: 2628. https://doi.org/10.3390/molecules25112628
APA StyleDurán-Meza, A. L., Escamilla-Ruiz, M. I., Segovia-González, X. F., Villagrana-Escareño, M. V., Vega-Acosta, J. R., & Ruiz-Garcia, J. (2020). Encapsidation of Different Plasmonic Gold Nanoparticles by the CCMV CP. Molecules, 25(11), 2628. https://doi.org/10.3390/molecules25112628




